Patent application title: Systemic Carnitine Deficiency Gene and Uses Thereof
Inventors:
Jun-Ichi Nezu (Shizuoka, JP)
Akira Tsuji (Ishikawa, JP)
Asuka Ose (Chiba, JP)
Assignees:
Chugai Seiyaku Kabushiki Kaisha
IPC8 Class: AC12Q168FI
USPC Class:
435 6
Class name: Chemistry: molecular biology and microbiology measuring or testing process involving enzymes or micro-organisms; composition or test strip therefore; processes of forming such composition or test strip involving nucleic acid
Publication date: 2009-09-03
Patent application number: 20090220967
Inventors list |
Agents list |
Assignees list |
List by place |
Classification tree browser |
Top 100 Inventors |
Top 100 Agents |
Top 100 Assignees |
Usenet FAQ Index |
Documents |
Other FAQs |
Patent application title: Systemic Carnitine Deficiency Gene and Uses Thereof
Inventors:
Jun-Ichi Nezu
Akira Tsuji
Asuka Ose
Agents:
FISH & RICHARDSON PC
Assignees:
CHUGAI SEIYAKU KABUSHIKI KAISHA
Origin: MINNEAPOLIS, MN US
IPC8 Class: AC12Q168FI
USPC Class:
435 6
Abstract:
The gene responsible for systemic carnitine deficiency was found to be the
OCTN2 gene involved in the transportation of organic cations. This
invention enables tests for this disease by detecting whether or not the
OCTN2 gene has a mutation. Furthermore, systemic carnitine deficiency can
be treated using the normal OCTN2 gene and its protein.Claims:
1. A method of screening for a compound that regulates OCTN2 gene
expression, the method comprising:(a) contacting a test compound with (i)
OCTN2 DNA encoding OCTN2; or (ii) a cell comprising said DNA; and(b)
measuring OCTN2 gene expression, wherein an increase or decrease in gene
expression in the presence of the test compound, compared to in the
absence of the test compound, is indicative of a compound that regulates
OCTN2 gene expression.
2. The method of claim 1, wherein the OCTN2 DNA comprises a promoter region or an enhancer region of the OCTN2 gene.
3. The method of claim 1, wherein the OCTN2 DNA comprises the nucleotide sequence of SEQ ID NO:5.
4. The method of claim 1, wherein the test compound is selected from among proteins, peptides, synthetic compounds, natural compounds, genes and gene products.
5. The method of claim 4, wherein the test compound is a protein.
6. The method of claim 5, wherein the protein is a transcription factor.
7. A method of screening for a compound that regulates OCTN2 gene expression, the method comprising:(a) providing a vector comprising a reporter gene downstream of a promoter sequence from SEQ ID NO:5;(b) contacting a test compound with the vector; and(c) detecting expression of the reporter gene, wherein an increase or decrease in expression of the reporter gene in the presence of the test compound, compared to in the absence of the test compound, is indicative of a compound that regulates OCTN2 gene expression.
8. The method of claim 7, wherein the test compound binds to the promoter.
9. A method of screening for a compound that regulates OCTN2 gene expression, the method comprising:(a) providing an oligonucleotide DNA, wherein the oligonucleotide DNA comprises a regulatory sequence from SEQ ID NO:5 that is upstream of the OCTN2 coding sequence;(b) contacting a test compound with the oligonucleotide DNA; and(c) determining whether the test compound binds to the oligo DNA, wherein binding to the oligonucleotide DNA is an indication that the test compound is potentially able to regulate OCTN2 gene expression.
10. The method of claim 9, wherein the regulatory sequence is a promoter region of the OCTN2 gene.
11. A method of screening for a compound that regulates OCTN2 gene expression, the method comprising:(a) contacting a test compound with DNA that comprises a promoter region of the OCTN2 gene; and(b) measuring binding of the test compound to the promoter, wherein a compound that binds to the promoter is indicative of a compound that regulates OCTN2 gene expression.
12. The method of claim 11, wherein the DNA further comprises (i) OCTN2 genomic DNA (SEQ ID NO:5); (ii) a sequence that regulates transcription of SEQ ID NO:5; or (iii) DNA that encodes an OCTN2 protein comprising the amino acid sequence of SEQ ID NO:1.
13. The method of claim 11, wherein the DNA is bound to a solid support.
14. The method of claim 13, wherein the compound is purified by affinity chromatography.
15. The method of claim 11, wherein the test compound is a protein.
16. The method of claim 15, wherein the protein is a transcription factor.
17. A method of screening for a compound that regulates OCTN2 gene expression, the method comprising:(a) providing a cell comprising a reporter gene downstream of DNA that comprises a promoter region of the OCTN2 gene; and(b) detecting reporter gene activity in the cell, wherein an increase or decrease in reporter gene expression in the presence of the test compound, compared to in the absence of the test compound, is indicative of a compound that regulates OCTN2 gene expression.
18. The method of claim 17, wherein the test compound is a protein.
19. The method of claim 18, wherein the protein is a transcription factor.
20. A method of screening for a compound that enhances the activity of the OCTN2 protein, the method comprising:(a) contacting a test compound with an OCTN2 protein comprising the amino acid sequence of SEQ ID NO: 1; and(b) evaluating the activity of the protein, wherein an increase in activity in the presence of the test compound, compared to in the absence of the test compound, is indicative of a compound that enhances the activity of the OCTN2 protein.
21. The method of claim 20, wherein the test compound is introduced into a cell comprising a vector that expresses the OCTN2 protein.
22. The method of claim 21, wherein the activity of the OCTN2 protein is evaluated by measuring carnitine transport into the cell.
23. A pharmaceutical composition comprising an isolated protein comprising the amino acid sequence of SEQ ID NO: 1 as an active ingredient, wherein the protein is in an amount effective to increase cellular uptake of carnitine.
24. A method of testing whether an individual's genome carries a mutant OCTN2 allele that, in the homozygous state, may result in systemic carnitine deficiency, the method comprising(a) identifying an individual suspected of carrying the mutant allele; and(b) analyzing a nucleic acid sample from the individual to determine the presence or absence of a mutation in (i) DNA encoding OCTN2 (SEQ ID NO: 1) or (ii) OCTN2 genomic DNA (SEQ ID NO:5) or (iii) a sequence that regulates expression of SEQ ID NO:5, wherein the presence of the mutation in (i) or (ii) or (iii) indicates that the individual carries a mutant OCTN2 allele that, in the homozygous state, may result in systemic carnitine deficiency.
Description:
RELATED APPLICATIONS
[0001]This application is a divisional of U.S. application Ser. No. 10/940,500, filed on Sep. 13, 2004, to issue as U.S. Pat. No. 7,413,860 on Aug. 19, 2008, which is a divisional of U.S. application Ser. No. 09/798,743, filed on Mar. 2, 2001, now U.S. Pat. No. 6,790,831, which is a continuation of PCT/JP99/04853, filed on Sep. 7, 1999, and claims priority from Japanese Patent Application No. 10/252,683, filed on Sep. 7, 1998. The contents of each of these applications are incorporated in their entirety by reference herein.
INCORPORATION BY REFERENCE OF SEQUENCE LISTING FILED ELECTRONICALLY
[0002]An electronic version of the Sequence Listing is filed herewith, the contents of which are incorporated by reference in their entirety. The computer-readable file, created on Aug. 15, 2008, is 64.0 kilobytes in size and titled 14875073003SeqList.txt.
[0003]1. Technical Field
[0004]This invention relates to molecules used in the testing and treatment of systemic carnitine deficiency, as well as methods for testing the disease.
[0005]2. Background of the Invention
[0006]Systemic Carnitine Deficiency (SCD) is a human genetic disease inherited through autosomal recessive inheritance, the main symptoms being skeletal or cardiac muscle disorders (NIM 212140) (Roe, C. R. and Coates, P. M., Mitochondrial fatty acid oxidation disorder, The metabolic and molecular bases of inherited diseases 7th ed., edited by Scriver, C. R., Beaudet, A. L., Sly, W. S. and Valle, D., McGraw-Hill, New York, 1995, 1508-1509; Karpati, G. et al., The syndrome of systemic carnitine deficiency: clinical, morphologic, biochemical, and pathophysiologic features, Neurology 1975, 25:16-24). Serum carnitine levels and intra-tissue carnitine levels are known to be extremely low in these patients compared to healthy individuals. Carnitine is an indispensable co-factor in the long-chain fatty acid metabolism. A carnitine-mediated mechanism enables intracellular fatty acids to permeate mitochondrial outer and inner membranes, and energy is produced when these fatty acids undergo β-oxidation within the mitochondria (Walter, J. H., L-Carnitine, Arch Dis Child, 1996, 74:475-478; Bremer, J., Carnitine metabolism and functions, Physiol Rev, 1983, 1420-1480). The abnormal decrease of carnitine concentration in systemic carnitine deficiency patients is thought to be the direct cause of diseases in tissues such as muscles that require a large amount of energy. Membrane physiological studies done using fibroblasts from systemic carnitine deficiency patients have shown that these cells lack the mechanism to transport carnitine from the outside of the cell to the inside. A gene that encodes a protein involved in this mechanism is presumed to be the gene responsible for this disease (Tein, I. et al., Impaired skin fibroblast carnitine uptake in primary systemic carnitine deficiency manifested by childhood carnitine-responsive cardiomyopathy, Pediatr Res, 1990, 28:247-255). However, the gene responsible for systemic carnitine deficiency is yet to be isolated.
SUMMARY OF THE INVENTION
[0007]An objective of the present invention is to provide the gene responsible for systemic carnitine deficiency. Moreover, this invention aims to provide a molecule used in the testing and treatment of systemic carnitine deficiency, as well as a method for testing the disease.
[0008]The Inventors isolated several genes encoding proteins involved in the transport of organic cations. Among these, the Inventors discovered the human gene (human OCTN2 gene) having an activity to transport carnitine in a sodium ion dependent manner, and the corresponding mouse gene (mouse OCTN2 gene) (Japanese Patent Application Hei 9-260972, Japanese Patent Application Hei 10-156660). The Inventors thought that the isolated OCTN2 gene might be the gene responsible for systemic carnitine deficiency, and evaluated this possibility.
[0009]Specifically, the nucleotide sequence of the OCTN2 gene of the systemic carnitine deficiency mouse model and systemic carnitine deficiency patients were analyzed. As a result, the Inventors discovered the presence of various mutations in the OCTN2 gene of both the mouse model and systemic carnitine deficiency patients. In other words, for the first time in the world, the Inventors succeeded in revealing that systemic carnitine deficiency is caused by mutations in the OCTN2 gene.
[0010]Moreover, due to the close relationship of OCTN2 gene mutation and systemic carnitine deficiency, the Inventors found that this disease can be tested by examining whether or not there is a mutation in the OCTN2 gene of a patient.
[0011]It was also found that systemic carnitine deficiency could be treated by using the normal OCTN2 gene and its protein, to complete the invention.
[0012]Therefore, this invention relates to molecules used in the testing and treatment of systemic carnitine deficiency, as well as methods for testing the disease. More specifically, the present invention relates to:
[0013](1) a DNA for testing systemic carnitine deficiency, wherein the DNA hybridizes to a DNA comprising the nucleotide sequence of SEQ ID NO:5, or the transcription regulatory region thereof, and comprises at least 15 nucleotides;
[0014](2) a molecule as in any one of (a) to (c) below, which is used for the treatment of systemic carnitine deficiency,
[0015](a) a protein comprising the amino acid sequence of SEQ ID NO: 1,
[0016](b) a compound that promotes the activity of the protein comprising the amino acid sequence of SEQ ID NO: 1, or,
[0017](c) a DNA encoding the protein comprising the amino acid sequence of SEQ ID NO:1;
[0018](3) a pharmaceutical composition for treating systemic carnitine deficiency, comprising a molecule of (2) as the active ingredient;
[0019](4) a pharmaceutical composition for treating systemic carnitine deficiency, comprising an antibody binding to the protein comprising the amino acid sequence of SEQ ID NO: 1 as the active ingredient;
[0020](5) a test method for systemic carnitine deficiency comprising the detection of a mutation in the DNA encoding the protein comprising the amino acid sequence of SEQ ID NO: 1, or the transcription regulatory region of said DNA;
[0021](6) the test method for systemic carnitine deficiency of (5) comprising the steps of,
[0022](a) preparing a DNA sample from a patient,
[0023](b) amplifying patient-derived DNA using the DNA of (1) as a primer,
[0024](c) cleaving the amplified DNA,
[0025](d) separating the DNA fragments by their size,
[0026](e) hybridizing the DNA of (1) labeled by a detectable label as a probe to the DNA fragments separated, and,
[0027](f) comparing the size of the DNA fragment detected with a control from a healthy individual,
[0028](7) the test method for systemic carnitine deficiency of (5) comprising the steps of,
[0029](a) preparing an RNA sample from a patient,
[0030](b) separating the prepared RNA by size,
[0031](c) hybridizing the DNA of (1) labeled by a detectable label as a probe to the RNA fragments separated, and,
[0032](d) comparing the size of the RNA fragment detected with a control from a healthy individual,
[0033](8) the test method for systemic carnitine deficiency of (5) comprising the steps of,
[0034](a) preparing a DNA sample from a patient,
[0035](b) amplifying patient-derived DNA using the DNA of (1) as a primer,
[0036](c) dissociating the amplified DNA to single-stranded DNA,
[0037](d) separating the dissociated single-stranded DNA on a non-denaturing gel, and,
[0038](e) comparing the mobility of separated single stranded DNA on the gel with a control from a healthy individual,
[0039](9) the test method for systemic carnitine deficiency of (5) comprising the steps of,
[0040](a) preparing a DNA sample from a patient,
[0041](b) amplifying patient-derived DNA using the DNA of (1) as a primer,
[0042](c) separating the amplified DNA on a gel in which the concentration of the DNA denaturant gradually increases, and,
[0043](d) comparing the mobility of separated DNA on the gel with a control from a healthy individual.
[0044]The present invention is based on the finding by the present inventors that systemic carnitine deficiency is caused by a mutation in the gene named "OCTN2". First and foremost, this invention relates to a molecule used in the testing and treatment of systemic carnitine deficiency, as well as a method for testing the disease.
[0045]In the present invention, the genomic DNA region (for example, SEQ ID NO:5) containing OCTN2, or an oligonucleotide (probe and primer) that hybridizes to the nucleotide sequence of the regulatory region (comprising the intron, promoter, and enhancer sequences as well) of OCTN2 is used.
[0046]This oligonucleotide preferably hybridizes specifically to the genomic DNA region containing OCTN2, or the regulatory region of OCTN2. Herein, "hybridizes specifically" indicates that cross-hybridization does not significantly occur with DNA encoding other proteins, under normal hybridizing conditions, preferably under stringent conditions (for example, the conditions in Sambrook et al., Molecular Cloning second edition, Cold Spring Harbor Laboratory Press, New York, USA, 1989).
[0047]When using as a primer, the oligonucleotide is usually, 15 to 100 bp, preferably, 17 to 30 bp. The primer may be any, as long as it can amplify at least a part of the OCTN2 gene or the region regulating its expression. Such regions comprise, for example, the exon region of OCTN2, the intron region, the promoter region, and enhancer region.
[0048]On the other hand, the oligonucleotide used as a probe usually comprises at least 15 bp or more if it is a synthetic oligonucleotide. It is also possible to use a double stranded DNA obtained from a clone incorporated into a vector such as plasmid DNA. The probe may be any, as long as it specifically hybridizes to at least a part of the OCTN2 gene or the region regulating the expression of the gene. Regions to which the probe hybridizes include, for example, the exon region, intron region, promoter region, and enhancer region of the OCTN2 gene. When using as the probe, oligonucleotide or double stranded DNA is suitably labeled. Examples of labeling methods are, phosphorylating the 5' end of the oligonucleotide by 32p using T4 polynucleotide kinase, and incorporating a substrate nucleotide labeled by an isotope such as 32p, a florescent dye, or biotin, using the random hexamer oligonucleotide as a probe and using DNA polymerase such as the Klenow enzyme (random priming technique).
[0049]In the present invention, "a test method for systemic carnitine deficiency" includes not only a test for patients showing symptoms of systemic carnitine deficiency caused by a mutation of the OCTN2 gene, but also a test for detecting a mutation of the OCTN2 gene for determining whether or not the person tested is likely to develop systemic carnitine deficiency arising from a OCTN2 gene mutation. In other words, the risk of developing systemic carnitine deficiency may greatly increase in cases where one of the OCTN2 alleles develops a mutation, even when no symptoms are visible on the outside. Therefore, tests for specifying patients (carriers) having a mutation in an OCTN2 allele are also included in the invention.
[0050]In the present invention, a test method for systemic carnitine deficiency using the above oligonucleotides comprises the detection of a mutation in the OCTN2 gene or its transcription regulatory region. One embodiment of this method of testing is the direct determination of the nucleotide sequence of the patient's OCTN2 gene. For example, using the above oligonucleotide as the primer, the whole OCTN2 gene or a part of it is amplified by the Polymerase Chain Reaction (PCR) using as the template a DNA isolated from a patient suspected of having a disease caused by an OCTN2 mutation. By comparing this sequence with that of a healthy individual, it is possible to conduct a test for a disease arising from an OCTN2 gene mutation.
[0051]As the testing method of the invention, other than determining the nucleotide sequence of DNA derived directly from the patient, several other methods are also used. One such embodiment comprises the following steps of: (a) preparing a DNA sample from a patient; (b) amplifying the patient-derived DNA using the primer of this invention; (c) dissociating amplified DNA into single-stranded DNA; (d) separating the dissociated single-stranded DNA on a non-denaturing gel; and, (e) comparing the mobility of separated single stranded DNA on the gel with a control from a healthy individual.
[0052]An example of such a method is the PCR-single-strand conformation polymorphism (PCR-SSCP) method (Cloning and polymerase chain reaction-single-strand conformation polymorphism analysis of anonymous Alu repeats on chromosome 11, Genomics, 1992 Jan 1, 12(1):139-146; Detection of p53 gene mutations in human brain tumors by single-strand conformation polymorphism analysis of polymerase chain reaction products, Oncogene, 1991 August 1, 6(8):1313-1318; Multiple fluorescence-based PCR-SSCP analysis with postlabeling, PCR Methods Appl. 1995 April 1, 4(5):275-282). This method is comparatively easy to handle, and has various advantages such as requiring only a small amount of a sample, and therefore, is suitable for screening a large number of DNA samples. The principle of this method is as follows. When a double stranded DNA fragment is disassociated into single strands, each strand forms an original high-order structure depending on its nucleotide sequence. When these dissociated DNA strands are electrophoresed within a polyacrylamide gel free of denaturants, the single stranded DNAs that are complementary and have the same length, migrate to different positions according to the difference in their high-order structure. This high order structure of the single strands change even by a single nucleotide substitution showing different mobilities in polyacrylamide gel electrophoresis. Therefore, the presence of a mutation in a DNA fragment due to point mutation, deletion, or insertion can be detected by the change in mobility.
[0053]Specifically, first, the whole OCTN2 gene or a part of it is amplified by PCR, and such. A length of 200 to 400 bp is usually preferred amplified range. Regions amplified include all the exons and all the introns of the OCTN2 gene, as well as the promoter and enhancer of the OCTN2 gene. PCR can be done, for example, according to conditions described in Example 1. When amplifying the gene fragment by PCR, a primer labeled by an isotope such as 32p, a fluorescent dye, or biotin is used, or the DNA fragment synthesized by PCR after adding a substrate nucleotide labeled by an isotope such as 32p, a fluorescent dye, or biotin, is labeled. Labeling can also be done by adding to the synthesized DNA fragment a substrate nucleotide labeled by an isotope such as 32p, a fluorescent dye, or biotin, using the Klenow enzyme and such after the PCR reaction. The labeled DNA fragment thus obtained is denatured by heating and such, and electrophoresed in a polyacrylamide gel free of denaturants such as urea. Conditions for separating the DNA fragment can be improved by adding a suitable amount (about 5 to 10%) of glycerol to the polyacrylamide gel. Conditions of electrophoresis vary depending on the properties of the DNA fragment, but room temperature (from 20 to 25° C.) is usually used. When a preferable separation cannot be accomplished, the temperature that gives the optimum mobility at 4 to 30° C. is evaluated. Following electrophoresis, the mobility of the DNA fragment is detected by an autoradiography using X-ray films, a scanner that detects fluorescence, and so on, and analyzed. When a band having a difference in mobility is detected, this band is directly excised from the gel, re-amplified by PCR, and is directly sequenced to verify the presence of a mutation. Even when labeled DNA is not used, the band can be detected by staining the gel after electrophoresis with ethidium bromide, silver, and such.
[0054]Another embodiment of the test method of the present invention comprises the following steps of: (a) preparing a DNA sample from a patient; (b) amplifying patient-derived DNA using the primer of this invention; (c) cleaving the amplified DNA;
(d) separating the DNA fragments according to their size; (e) hybridizing the probe DNA of the invention labeled with a detectable label to the DNA fragments separated; and(f) comparing the size of the detected DNA fragment with a control from a healthy individual.
[0055]Such methods include those using Restriction Fragment Length Polymorphism (RFLP), PCR-RFLP method, and so on. Restriction enzymes are usually used to cleave DNA. Specifically, compared to a DNA fragment of a healthy individual, the size of one obtained following restriction enzyme treatment changes when a mutation exists at the recognition site of the restriction enzyme, or when nucleotides have been inserted or deleted in the DNA fragment resulting from restriction enzyme treatment. The portion containing the mutation is amplified by PCR, the amplified products are treated with each restriction enzyme and electrophoresed to detect the mutation as the difference of mobility. Alternatively, chromosomal DNA is cleaved with these restriction enzymes, and after electrophoresis, the presence or absence of a mutation can be detected by southern-blotting using the probe DNA of the invention. The restriction enzymes used can be suitably selected according to each mutation. This method can use not only genomic DNA, but also cDNA made by treating RNA prepared from patients with reverse transcriptase, cleaving this cDNA as-it-is with restriction enzymes, and then conducting southern blotting. It is also possible to examine the changes in mobility after amplifying the whole OCTN2 gene, or a part of it, by PCR using the above cDNA as the template, and cleaving the amplified products by restriction enzymes.
[0056]A similar detection is also possible using RNA prepared from patients instead of DNA. This method includes the steps of: (a) preparing an RNA sample from a patient; (b) separating the prepared RNA according to their size; (c) hybridizing the probe DNA of the invention labeled by a detectable label to the separated RNA; and (d) comparing the size of the detected RNA with a control from a healthy individual. In a specific example of this method, RNA prepared from a patient is electrophoresed, northern blotting is done using the probe of the invention to detect the mobility change.
[0057]Another embodiment of the method of the invention comprises the steps of:
(a) preparing a DNA sample from a patient; (b) amplifying patient-derived DNA using the primer of this invention; (c) separating the amplified DNA on a gel in which the concentration of the DNA denaturant gradually increases; and, (d) comparing mobility of the DNA separated upon the gel with a control from a healthy individual.
[0058]An example of such a method is denaturant gradient gel electrophoresis (DGGE). The whole OCTN2 gene or a part of it is amplified by a method such as PCR using the primer of the invention, and the amplified product is electrophoresed in a gel in which the concentration of the DNA denaturant gradually increases, and compared with a control from a healthy individual. In the case of a DNA having a mutation, the DNA fragment will become single stranded at a low denaturant concentration and the moving speed will become extremely slow. The presence or absence of a mutation can be detected by detecting the change in mobility.
[0059]Allele Specific Oligonucleotide (ASO) hybridization can be used alternatively when the aim is to detect a mutation at a specific site. When an oligonucleotide comprising a nucleotide sequence thought to have a mutation is prepared and this is hybridized with sample DNA, the hybrid formation efficiency will decrease when there is a mutation. This can be detected by southern blotting and by a method using the property of special fluorescent reagents that quench when intercalated into a hybrid gap. The detection by ribonuclease A mismatch cleavage method can also be used. Specifically, the whole OCTN2 gene, or a part of it, is amplified by a method such as PCR, and the amplified product is hybridized to labeled RNA prepared from OCTN2 cDNA and such incorporated into a plasmid vector, etc. The hybrids will be single stranded in the portion where a mutation exists. This portion is cleaved by ribonuclease A and the existence of a mutation can be detected by autoradiography, and such.
[0060]The present invention also relates to a test drug for systemic carnitine deficiency that comprises an antibody binding to the OCTN2 protein as the active ingredient. An antibody binding to the OCTN2 protein can be prepared using methods well known to those skilled in the art. Polyclonal antibodies can be made by, obtaining the serum of small animals such as rabbits immunized with the OCTN2 protein (apart from the natural protein, recombinant OCTN2 proteins expressed in suitable host cells (E. coli, yeasts, mammals, and such), such as recombinant OCTN2 protein expressed in E. coli as a fusion protein with GST) of the present invention, or a partial peptide. The serum is then purified by, for example, ammonium sulfate precipitation, protein A or protein G column chromatography, DEAE ion exchange chromatography, or an affinity chromatography using a column to which the protein of the present invention or synthetic peptide is coupled. Monoclonal antibodies can be made by immunizing small animals such as mice with the OCTN2 protein or a partial peptide thereof, excising the spleen from the mouse, homogenizing it and separating cells, fusing the cells with mouse myeloma cells using a reagent such as polyethylene glycol, and selecting clones that produce an antibody binding to the OCTN2 protein from the fused cells (hybridomas) produced. Next, the obtained hybridomas are transplanted into the abdominal cavity of a mouse, and ascites are extracted from the mouse. The obtained monoclonal antibodies can be purified by, for example, ammonium sulfate precipitation, protein A or protein G column chromatography, DEAE ion exchange chromatography, or an affinity chromatography using a column to which the OCTN2 protein or synthesized peptide is coupled. When using the antibody as a test drug, it is mixed with sterile water, physiological saline, plant oils, surfactants, lipids, solubilizers, stabilizers (BSA, gelatin, etc.), preservatives, and such, according to needs. An example of a test for systemic carnitine deficiency features the staining of tissues collected or cells isolated from a patient by the enzyme-labeled antibody method, fluorescence-labeled antibody method, and test for a deficiency, abnormal accumulation, or abnormal intracellular distribution of the OCTN2 protein. Testing can also be done by preparing a cell-extract of tissues collected or cells isolated from a systemic carnitine deficiency patient, separating the cell-extract by methods such as SDS-PAGE, transferring onto a nitrocellulose membrane, PVDF membrane, and such, and then staining this by a method (western blotting, immunoblotting, etc) using the above-described enzyme-labeled antibody method, etc.
[0061]The present invention also relates to a therapeutic drug for systemic carnitine deficiency. One such embodiment has the OCTN2 gene as the active ingredient. When using the OCTN2 gene as a therapeutic drug, it is given to the patient by oral, intravenous, topical administration and such, as the full length OCTN2 chromosomal DNA, a part of it, or by incorporating the OCTN2 DNA into a suitable vector, for example, adenovirus vector, adeno associated virus vector, retro virus vector, or plasmid DNA. The ex vivo method can also be used for administration apart from the in vivo method. The transition and absorption into tissues can be enhanced by enclosing the gene in a liposome prepared by micellization of phospholipids, or by adding a cationic lipid and forming a complex with genomic DNA. Therefore, the method of the invention can replace a patient's mutated OCTN2 gene by a normal gene, and also additionally administer the normal gene, thereby enabling the treatment of systemic carnitine deficiency.
[0062]Another embodiment of the invention relating to a therapeutic drug of systemic carnitine deficiency comprises the OCTN2 protein as the active ingredient. The amino acid sequences of human and mouse OCTN2 proteins are shown in SEQ ID NOs: 1 and 3, respectively. The OCTN2 protein can be prepared as a natural protein and also as a recombinant protein. The natural protein can be prepared by a method well known to one skilled in the art, for example, by isolating the OCTN2 protein from tissues or cells that show a high level expression of the protein (e.g. fetal kidney) by affinity chromatography using an antibody against a partial peptide of the OCTN2 protein. On the other hand, a recombinant protein can be prepared by culturing cells transformed by DNA (for example, SEQ ID NO:2) encoding the OCTN2 protein. Cells used for the production of recombinant proteins include mammalian cells such as, COS cells, CHO cells, and NIH3T3 cells, insect cells such as sf9 cells, yeast cells, and E. coli cells. Vectors for expressing the recombinant proteins within cells vary according to the host used, and normally, pcDNA3 (Invitrogen), pEF-BOS (Nucleic Acids Res. 1990, 18(17), 5322) and such are used as vectors for mammalian cells, the "BAC-to-BAC baculovirus expression system" (GIBCO BRL) and such are used for insect cells, "Pichia Expression Kit" (Invitrogen) and such are used for yeast cells, pGEX-5X-1 (Pharmacia), "QIAexpress system" (Qiagen) and such are used for E. coli cells. Vectors are introduced to hosts using, for example, the calcium phosphate method, DEAE dextran method, method using cationic liposome DOTAP (Boehringer Mannheim), and Superfect (Qiagen), electroporation method, calcium chloride method, and such. The recombinant protein can be purified from the transformant obtained usually using methods described in "The Qiaexpressionist handbook, Qiagen, Hilden, Germany".
[0063]When using the obtained OCTN2 protein as a therapeutic drug for treating systemic carnitine deficiency, the OCTN2 protein can be directly administered, or can be given after being formulated into a pharmaceutical composition by a well-known pharmaceutical manufacturing method. For example, the drug can be given after suitably combining with a generally used carrier or medium such as, sterilized water, physiological saline, plant oils, surfactants, lipids, solubilizers, stabilizers, preservatives, and such.
[0064]The dosage varies depending on factors such as the patient's body weight, age, healthiness, and method of administration, but a skilled artisan can suitably select the dosage. Usually, it is within the range from 0.01 to 1000 mg/kg. The administration can be done orally, intravenously, intramuscularly, or percutaneously. A skilled artisan can easily replace, add, or delete amino acid(s) in the amino acid sequence of the OCTN2 protein using a well-known method such as the site-specific mutation induction system using PCR (GIBCO-BRL, Gaithersburg, Maryland), site-specific mutagenesis using oligonucleotides (Kramer, W. and Fritz, H J, 1987, Methods in Enzymol, 154:350-367), the Kunkel method (Methods Enzymol., 1988, 85:2763-2766), and such.
[0065]Another embodiment of the therapeutic drug for systemic carnitine deficiency uses a compound that enhances the activity of the OCTN2 protein as the active ingredient. Such a compound can be screened as follows. For example, a plasmid expressing the OCTN2 protein is constructed, and this is introduced into HEK293 cells by the calcium phosphate method. Radiolabeled carnitine and a test compound are added to this transformant and the carnitine transporting activity into the cells is determined. A compound that can enhance the carnitine transporting activity is selected by comparing with the activity of the OCTN2 protein in the absence of the test compound. See Japanese Patent Application Hei 9-260972 and Hei 10-156660 for the detailed method.
[0066]Similar to the above-mentioned use of the OCTN2 protein as a therapeutic drug, the isolated compound can also be formulated into a pharmaceutical composition using well-known pharmaceutical manufacturing methods. The dose range is usually within 0.01 to 1000 mg/kg.
[0067]It is also conceivable to utilize the region regulating OCTN2 gene expression or a factor that binds to this region for the treatment of systemic carnitine deficiency.
[0068]The OCTN2 gene comprising the region that regulates OCTN2 gene expression is useful in the above-mentioned gene therapy as it can express the OCTN2 gene under normal expression regulation in vivo by introducing it into patients who lack the OCTN2 gene, or who have a defect in OCTN2 gene expression.
[0069]Moreover, if the promoter site is determined from the upstream region of the OCTN2 gene, a compound that regulates OCTN2 gene expression amount can be simply screened by using a reporter gene expression vector having the above promoter site through examining the influence of various compounds on the production of reporter gene products. Such a screening method comprises the following steps of, (a) constructing a vector in which a reporter gene is ligated to the downstream of the promoter site, (b) introducing the vector into a suitable cell, and, (c) detecting the reporter gene activity by contacting or introducing a test compound to the above cell. Examples of the test compound include, proteins, peptides, synthetic compounds, natural compounds, genes, gene products, and such.
[0070]A compound regulating OCTN2 gene expression can also be screened by contacting a test sample with the promoter site, and selecting a compound (such as a protein) that binds to the promoter site. For example, a synthetic oligo DNA and such having the nucleotide sequence of the promoter site is prepared, this is bound to a suitable support such as Sepharose, and contacted with a cell-extract, and such. Then, a transcription factor and such that binds to this promoter site and regulates OCTN2 gene expression can be purified by, for example, affinity chromatography.
DESCRIPTION OF DRAWINGS
[0071]FIG. 1 shows the direct sequencing of the mouse OCTN2 gene amplified by RT-PCR. wt/wt shows wild-type homologous mouse (SEQ ID NO:27), and jvs/jvs shows the jvs homologous mouse (SEQ ID NO:28). OCTN2 gene of the jvs mouse has a mutation at the nucleotide shown by the arrow.
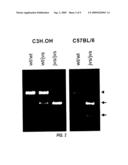
[0072]FIG. 2 is electrophoretic images showing the mutation in the OCTN2 gene of the jvs mouse, which was detected using the PCR-RFLP method (Cfr 131 cleavage). The fragment shown by the arrow head derives from the normal gene, and the fragments shown by the arrows were due to the mutated gene.
[0073]FIG. 3 shows results of the carnitine transporting activity assay of wild-type mouse OCTN2 and the mutant mouse OCTN2. A sodium-dependent carnitine transporting activity is seen for the wild type, whereas the mutant (Jvs) shows absolutely no activity. "Mock" is when a cDNA-non-containing vector was used as the control.
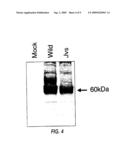
[0074]FIG. 4 is an electrophoretic image showing the results of western blot analysis using anti myc antibody. It can be seen that the wild-type OCTN2 protein (wild) and the mutant OCTN2 protein (Jvs) is produced in similar amounts. "Mock" is when a cDNA-non-containing vector was used as the control.
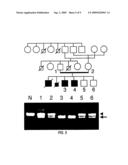
[0075]FIG. 5 shows the results of OCTN2 gene analysis in the KR family. The pedigree chart of this family is shown on top. Squares indicate males, circles females, filled ones individuals having systemic carnitine deficiency, and crossed squares indicate deceased individuals. An electrophoretic image showing the PCR results is given below. "N" shows the results of the normal gene used as the control. The fragments shown by the arrowhead are PCR products derived from the normal gene, and the fragments shown by the arrow derived from the gene where the defect exits.
[0076]FIG. 6 shows the results of sequencing exon 1 of the OCTN2 gene. Compared to the normal OCTN2 gene (upper panel; wild-type; nucleotides 214 to 234 of SEQ ID NO:5), the OCTN2 of systemic carnitine deficiency patients (lower panel; SEQ ID NO:29) belonging to the AK family, show an insertion of a cytosine residue at the position indicated by the arrow.
[0077]FIG. 7 shows the results of sequencing exon 2 of the OCTN2 gene. Compared to the normal OCTN2 gene (upper panel; wild-type; nucleotides 8,630 to 8,648 of SEQ ID NO:5), the OCTN2 of systemic carnitine deficiency patients (lower panel; SEQ ID NO:30) belonging to the AK family, show a single nucleotide substitution (A has substituted G) as indicated by the arrow.
[0078]FIG. 8 is electrophoretic images showing the results of the analysis of two-types of mutations seen in the OCTN2 gene of a systemic carnitine deficiency patient belonging to the AK family using a PCR-RFLP method utilizing BcnI and NlaIV, respectively. The pedigree chart of this family is shown on top. Square indicates a male, circles females, and the filled circle indicates a systemic carnitine deficiency patient. "N" shows the results of the normal gene used as the control. The fragments shown by the arrows derived from the mutant gene.
[0079]FIG. 9 shows the results of the sequencing analysis of the intron 8/exon 9 of the OCTN2 gene. Compared to the normal gene (normal; nucleotides 23,925 to 23,943 of SEQ ID NO:5), the gene deriving from the patient belonging to the TH family (patient; SEQ ID NO:31) has a splicing site mutation (AG to AA) in the 3' end of intron 8. The pedigree chart of this family is shown on top. Squares indicate males, the circle a female, and filled square indicates a systemic carnitine deficiency patient.
DETAILED DESCRIPTION OF THE INVENTION
[0080]The invention shall be described in detail below, but it is not to be construed as being limited thereto.
Example 1
[0081]Proof in mouse and human showing that the gene responsible for systemic carnitine deficiency (SCD) is OCTN2
[0082]The Inventors have previously isolated human cDNA encoding a protein having an activity to transport carnitine in a sodium-ion dependent manner, and also the corresponding mouse cDNA (Japanese Patent Application No. Hei 9-260972, Japanese Patent Application No. Hei 10-156660). The nucleotide sequences of the human and mouse OCTN2 cDNA isolated by the Inventors are shown in SEQ ID NO:2 and 4, respectively, and the amino acid sequences of the proteins encoded by these cDNAs are shown in SEQ ID NO: 1 and 3, respectively.
[0083]The Inventors drew up a working hypothesis that OCTN2 might be the gene responsible for systemic carnitine deficiency, and conducted experiments to prove this.
(1) OCTN2 Gene Analysis in Juvenile Visceral Steatosis (jvs) Mouse
[0084]The juvenile visceral steatosis (jvs) mouse was generated due to a mutation in the C3H.OH mouse. This jvs mouse shows symptoms similar to systemic carnitine deficiency patients, and shows an extremely low carnitine concentration within its blood and tissues. This phenotype is inherited by autosomal inheritance. From the above facts, the jvs mouse is considered to be a mouse model for systemic carnitine deficiency (Hashimoto, N. et al., Gene-dose effect on carnitine transport activity in embryonic fibroblasts of JVS mice as a model of human carnitine transporter deficiency, Biochem Pharmacol, 1998, 55:1729-1732). The Inventors examined the OCTN2 gene arrangement of the jvs mouse. Specifically, whole RNA was extracted from the kidney of a jvs homologous mouse, cDNA was synthesized, jvs mouse OCTN2 cDNA was amplified using this synthesized cDNA as the template by RT-PCR, and the sequence was examined by direct sequencing.
[0085]The amplification reaction by PCR was conducted as follows. For the 5' side fragment, the primers MONB 31 (5'-gataagcttacggtgtccccttattcccatacg-3'/SEQ ID NO:22) and MONB 20 (5'-cccatgccaacaaggacaaaaagc-3'/SEQ ID NO:23) were prepared. Then, amplification was done within a reaction solution (50 μl) containing, cDNA, 5 μl of 10× KOD buffer (Toyobo), 5 μl of 2 mM dNTPs, 2 μl of 25 mM MgCl2, 0.5 μl of KOD DNA polymerase (Toyobo), 1 μl of 20 μM MONB 31 primer, and 1 μl of 20 μM MONB 20 primer at 94° C. for 3 min, 30 cycles of "94° C. for 30 sec, 50° C. for 30 sec, and 74° C. for 1 min", and 72° C. for 10 min. As for the 3' side fragment, the primers MONB 6 (5'-tgtttttcgtgggtgtgctgatgg-3'/SEQ ID NO:24) and MONB 26 (5'-acagaacagaaaagccctcagtca-3'/SEQ ID NO:25) were prepared, and amplification was done within a reaction solution (50 μl) containing cDNA, 5 μl of 10× ExTaq buffer (TaKaRa), 4 μl of 2.5 mM dNTPs, 1 μl of a mixture of ExTaq DNA polymerase (TaKaRa) and anti Taq antibody (TaqStart antibody®, CLONTECH), 1 μl of 20 μM MONB 6 primer, and 1 μl of 20 μM MONB 26 primer, at 94° C. for 2 min, 30 cycles of "94° C. for 30 sec, 60° C. for 30 sec, and 74° C. for 2 min", and 72° C. for 10 min.
[0086]Sequencing revealed that the codon encoding the 352nd leucine (CTG) was mutated to a codon encoding arginine (CGG) (FIG. 1). This mutation can be detected by Restriction Fragment Length Polymorphism (PCR-RFLP) due to the presence of the Cfr13I restriction enzyme site. This method revealed that the jvs homologous mouse (jvs/jvs) had this mutation in both alleles, and that the heterologous mouse (wt/jvs) has both the mutated and wild type alleles (FIG. 2 left). This mutation was also found in the C57BL jvs mouse in which the genetic background has been replaced with that of the C57BL/6 mouse by backcrossing 12 times or more (FIG. 2 right). Since the C57BL jvs mouse was constructed after a series of selections using the jvs phenotype as an index, the jvs phenotype and OCTN2 mutations are considered to be very closely associated.
[0087]Next, the effect this mutation has on the carnitine transporting activity was examined. Plasmid DNA expressing wild-type mouse OCTN2, and those expressing mutated OCTN2 were separately introduced into BEK293 cells, and then, carnitine transporting ability was measured similar to the assay of human OCTN2 described in Japanese Patent Application Hei 10-156660 (FIG. 3). This revealed that although wild-type mouse OCTN2 shows a carnitine transporting activity similar to human OCTN2, the mutated OCTN2 has absolutely no activity. However, both proteins were confirmed to be expressed at a similar amount by a western blotting using an antibody against the c-myc epitope sequence (NH2-EQKLISEEDL-COOH; SEQ ID NO:26) added to the C terminus (FIG. 4).
[0088]Thus, the jvs mouse is thought to have developed the disease due to a functional deletion mutation of the OCTN2 gene.
(2) OCTN2 gene analysis in human systemic carnitine deficiency patients
[0089]A database search using human OCTN2 cDNA sequence revealed that the human OCTN2 genomic DNA sequence has been decoded by Lawrence Berkeley National Laboratory (LBNL) of the United States as a part of the human genome project. However, it was only recorded as several cosmid clone sequences, therefore, the inventors determined a complete human OCTN2 genomic DNA sequence (SEQ ID NO:5) by comparing with human OCTN2 cDNA sequence and suitably combining the clone sequences. The human OCTN2 gene is an about 26 kb gene comprising ten exons and nine introns. The eight pairs of primers shown below, which can amplify all the exons as eight fragments, were prepared from this gene arrangement.
[0090]Specifically, OCN2 43 (5'-GCAGGACCAAGGCGGCGGTGTCAG-3', SEQ ID NO:6) and OCN2 44 (5'-AGACTAGAGGAAAAACGGGATAGC-3', SEQ ID NO:7) for exon one; OCN2 25 (5'-AGATTTTTAGGAGCAAGCGTTAGA-3' SEQ ID NO:8) and OCN2 26 (5'-GAGGCAGACACCGTGGCACTACTA-3', SEQ ID NO:9) for exon two; OCN2 27 (5'-TTCACACCCACTTACTGGATGGAT-3' SEQ ID NO: 10) and OCN2 50 (5'-ATTCTGTTTTGTTTTGGCTCTTTT-3', SEQ ID NO: 11) for exons three and four; OCN2 31 (5'-AGCAGGGCCTGGGCTGACATAGAC-3', SEQ ID NO: 12) and OCN2 32 (5'-AAAGGACCTGACTCCAAGATGATA-3', SEQ ID NO: 13) for exon five; OCN2 33 (5'-TCTGACCACCTCTTCTTCCCATAC-3', SEQ ID NO: 14) and OCN2 34 (5'-GCCTCCTCAGCCACTGTCGGTAAC-3', SEQ ID NO: 15) for exon six; OCN2 35 (5'-ATGTTGTTCCTTTTGTTATCTTAT-3', SEQ ID NO: 16) and OCN2 36 (5'-CTTGTTTTCTTGTGTATCGTTATC-3', SEQ ID NO:17) for exon seven; OCN2 37 (5'-TATGTTTGTTTTGCTCTCAATAGC-3', SEQ ID NO:18) and OCN2 40 (5'-TCTGTGAGAGGGAGTTTGCGAGTA-3', SEQ ID NO: 19) for exon eight and nine; and, OCN2 41 (5'-TACGACCGCTTCCTGCCCTACATT-3', SEQ ID NO:20) and OCN2 42 (5'-TCATTCTGCTCCATCTTCATTACC-3', SEQ ID NO:21) for exon 10.
[0091]Next, human OCTN2 gene mutations in three families that have systemic carnitine deficiency patients, but no blood relationships were examined. The analysis is done by amplifying all the exons using the above primers and genomic DNA prepared from blood cells as the template, and subjecting the amplified products into direct sequencing.
[0092]The amplification reaction by PCR was done within a reaction solution (50 μl) containing 100 ng of genomic DNA, 5 μl of 10× ExTaq buffer (TaKaRa), 4 μl of 2.5 mM dNTPs, 1 μl of a mixture of ExTaq DNA polymerase (TaKaRa) and anti Taq antibody (TaqStart antibody®, CLONTECH), and 1 μl of each of the 20 μM primers. The reaction conditions were, 94° C. for 2 min, 36 cycles of "94° C. for 30 sec, 60° C. for 30 sec, and 74° C. for 2 min", and 72° C. for 10 min. However, in the case of exon one and exon five amplification, a reaction solution (50 μl) containing 100 ng genomic DNA, 25 μl of 2× GC buffer 1 (TaKaRa), 8 μl of 2.5 mM dNTPs, 0.5 μl of LA Taq DNA polymerase (TaKaRa), and 1 μl of each of the 20 μM primers, was used.
[0093]In the first family (KR family), a 113 bp deletion was found in first exon of the OCTN2 gene of a systemic carnitine deficiency patient (FIG. 5). This deletion affects the initiation codon and thus, a complete protein will not be produced. The next usable ATG codon present in the correct frame is at nucleotide no. 177, and in this case, it is thought that at least two transmembrane regions will be deleted. The two systemic carnitine deficiency patients in this family were found to contain this mutated OCTN2 gene in both alleles. On the other hand, the parents and the two brothers of the patient, who have not developed the disease, carry the mutation on just one allele.
[0094]In the second family (AK family), the systemic carnitine patients were found to contain two types of mutated OCTN2 genes. One mutation was a cytosine insertion just after the initiation codon, which is thought to cause a frame shift and prevent the proper protein from being produced (FIG. 6). The other mutation is a single base substitution (G to A) in the codon encoding the 132nd tryptophan (TGG). This mutation had altered the codon into a stop codon (TGA) (FIG. 7). These mutations are thought to prevent the production of active OCTN2 proteins in patients. These mutations can be detected by PCR-RFLP analysis using BcnI, NlaIV restriction enzymes, respectively, which revealed that the patient's parents who have not developed the disease, had one of each of the mutations, and the patient's sisters who have not developed the disease, do not have any mutated genes (FIG. 8).
[0095]In the third family (TH family), a mutation (AG to AA) was found in the splicing site in the 3' end of the intron eight of the OCTN2 gene (FIG. 9). This mutation prevents the gene from undergoing normal splicing, and thus, it is expected that the normal protein would not be produced. Sequencing analysis showed that the systemic carnitine deficiency patient belonging to this family had this mutation in both alleles. On the other hand, the patient's parents and one of the brothers who have not developed the disease had one mutated allele.
[0096]The above results revealed that systemic carnitine deficiency is a genetic disease caused by mutations in the OCTN2 gene. Thus, the present invention enables definitive diagnosis, prenatal diagnosis and such, of systemic carnitine deficiency by examining mutations in the OCTN2 gene using analyses described herein, as well as other methods. The present invention also enables treatment of systemic carnitine deficiency by treatments such as gene therapy using the OCTN2 gene.
INDUSTRIAL APPLICABILITY
[0097]The present invention revealed that the OCTN2 gene is the gene responsible for systemic carnitine deficiency, thus enabling tests for the disease by detecting mutations in the OCTN2 gene and its protein. Moreover, the present invention facilitates treatment of systemic carnitine deficiency by utilizing the OCTN2 gene and its protein.
Sequence CWU
1
311557PRTHomo sapiens 1Met Arg Asp Tyr Asp Glu Val Thr Ala Phe Leu Gly Glu
Trp Gly Pro1 5 10 15Phe
Gln Arg Leu Ile Phe Phe Leu Leu Ser Ala Ser Ile Ile Pro Asn 20
25 30Gly Phe Thr Gly Leu Ser Ser Val
Phe Leu Ile Ala Thr Pro Glu His 35 40
45Arg Cys Arg Val Pro Asp Ala Ala Asn Leu Ser Ser Ala Trp Arg Asn
50 55 60His Thr Val Pro Leu Arg Leu Arg
Asp Gly Arg Glu Val Pro His Ser65 70 75
80Cys Arg Arg Tyr Arg Leu Ala Thr Ile Ala Asn Phe Ser
Ala Leu Gly 85 90 95Leu
Glu Pro Gly Arg Asp Val Asp Leu Gly Gln Leu Glu Gln Glu Ser
100 105 110Cys Leu Asp Gly Trp Glu Phe
Ser Gln Asp Val Tyr Leu Ser Thr Ile 115 120
125Val Thr Glu Trp Asn Leu Val Cys Glu Asp Asp Trp Lys Ala Pro
Leu 130 135 140Thr Ile Ser Leu Phe Phe
Val Gly Val Leu Leu Gly Ser Phe Ile Ser145 150
155 160Gly Gln Leu Ser Asp Arg Phe Gly Arg Lys Asn
Val Leu Phe Val Thr 165 170
175Met Gly Met Gln Thr Gly Phe Ser Phe Leu Gln Ile Phe Ser Lys Asn
180 185 190Phe Glu Met Phe Val Val
Leu Phe Val Leu Val Gly Met Gly Gln Ile 195 200
205Ser Asn Tyr Val Ala Ala Phe Val Leu Gly Thr Glu Ile Leu
Gly Lys 210 215 220Ser Val Arg Ile Ile
Phe Ser Thr Leu Gly Val Cys Ile Phe Tyr Ala225 230
235 240Phe Gly Tyr Met Val Leu Pro Leu Phe Ala
Tyr Phe Ile Arg Asp Trp 245 250
255Arg Met Leu Leu Val Ala Leu Thr Met Pro Gly Val Leu Cys Val Ala
260 265 270Leu Trp Trp Phe Ile
Pro Glu Ser Pro Arg Trp Leu Ile Ser Gln Gly 275
280 285Arg Phe Glu Glu Ala Glu Val Ile Ile Arg Lys Ala
Ala Lys Ala Asn 290 295 300Gly Ile Val
Val Pro Ser Thr Ile Phe Asp Pro Ser Glu Leu Gln Asp305
310 315 320Leu Ser Ser Lys Lys Gln Gln
Ser His Asn Ile Leu Asp Leu Leu Arg 325
330 335Thr Trp Asn Ile Arg Met Val Thr Ile Met Ser Ile
Met Leu Trp Met 340 345 350Thr
Ile Ser Val Gly Tyr Phe Gly Leu Ser Leu Asp Thr Pro Asn Leu 355
360 365His Gly Asp Ile Phe Val Asn Cys Phe
Leu Ser Ala Met Val Glu Val 370 375
380Pro Ala Tyr Val Leu Ala Trp Leu Leu Leu Gln Tyr Leu Pro Arg Arg385
390 395 400Tyr Ser Met Ala
Thr Ala Leu Phe Leu Gly Gly Ser Val Leu Leu Phe 405
410 415Met Gln Leu Val Pro Pro Asp Leu Tyr Tyr
Leu Ala Thr Val Leu Val 420 425
430Met Val Gly Lys Phe Gly Val Thr Ala Ala Phe Ser Met Val Tyr Val
435 440 445Tyr Thr Ala Glu Leu Tyr Pro
Thr Val Val Arg Asn Met Gly Val Gly 450 455
460Val Ser Ser Thr Ala Ser Arg Leu Gly Ser Ile Leu Ser Pro Tyr
Phe465 470 475 480Val Tyr
Leu Gly Ala Tyr Asp Arg Phe Leu Pro Tyr Ile Leu Met Gly
485 490 495Ser Leu Thr Ile Leu Thr Ala
Ile Leu Thr Leu Phe Leu Pro Glu Ser 500 505
510Phe Gly Thr Pro Leu Pro Asp Thr Ile Asp Gln Met Leu Arg
Val Lys 515 520 525Gly Met Lys His
Arg Lys Thr Pro Ser His Thr Arg Met Leu Lys Asp 530
535 540Gly Gln Glu Arg Pro Thr Ile Leu Lys Ser Thr Ala
Phe545 550 55521831DNAHomo
sapiensCDS(124)..(1794) 2cggacggtct tgggtcgcct gctgcctggc ttgcctggtc
ggcggcgggt gccccgcgcg 60cacgcgcaaa gcccgccgcg ttcccagacc ccaggccgcg
ctctgtgggc ctctgagggc 120ggc atg cgg gac tac gac gag gtg acc gcc ttc
ctg ggc gag tgg ggg 168Met Arg Asp Tyr Asp Glu Val Thr Ala Phe Leu
Gly Glu Trp Gly1 5 10
15ccc ttc cag cgc ctc atc ttc ttc ctg ctc agc gcc agc atc atc ccc
216Pro Phe Gln Arg Leu Ile Phe Phe Leu Leu Ser Ala Ser Ile Ile Pro
20 25 30aat ggc ttc acc ggc ctg
tcc tcc gtg ttc ctg ata gcg acc ccg gag 264Asn Gly Phe Thr Gly Leu
Ser Ser Val Phe Leu Ile Ala Thr Pro Glu 35 40
45cac cgc tgc cgg gtg ccg gac gcc gcg aac ctg agc agc
gcc tgg cgc 312His Arg Cys Arg Val Pro Asp Ala Ala Asn Leu Ser Ser
Ala Trp Arg 50 55 60aac cac act
gtc cca ctg cgg ctg cgg gac ggc cgc gag gtg ccc cac 360Asn His Thr
Val Pro Leu Arg Leu Arg Asp Gly Arg Glu Val Pro His 65
70 75agc tgc cgc cgc tac cgg ctc gcc acc atc gcc aac
ttc tcg gcg ctc 408Ser Cys Arg Arg Tyr Arg Leu Ala Thr Ile Ala Asn
Phe Ser Ala Leu80 85 90
95ggg ctg gag ccg ggg cgc gac gtg gac ctg ggg cag ctg gag cag gag
456Gly Leu Glu Pro Gly Arg Asp Val Asp Leu Gly Gln Leu Glu Gln Glu
100 105 110agc tgt ctg gat ggc
tgg gag ttc agt cag gac gtc tac ctg tcc acc 504Ser Cys Leu Asp Gly
Trp Glu Phe Ser Gln Asp Val Tyr Leu Ser Thr 115
120 125att gtg acc gag tgg aac ctg gtg tgt gag gac gac
tgg aag gcc cca 552Ile Val Thr Glu Trp Asn Leu Val Cys Glu Asp Asp
Trp Lys Ala Pro 130 135 140ctc aca
atc tcc ttg ttc ttc gtg ggt gtg ctg ttg ggc tcc ttc att 600Leu Thr
Ile Ser Leu Phe Phe Val Gly Val Leu Leu Gly Ser Phe Ile 145
150 155tca ggg cag ctg tca gac agg ttt ggc cgg aag
aat gtg ctg ttc gtg 648Ser Gly Gln Leu Ser Asp Arg Phe Gly Arg Lys
Asn Val Leu Phe Val160 165 170
175acc atg ggc atg cag aca ggc ttc agc ttc ctg cag atc ttc tcg aag
696Thr Met Gly Met Gln Thr Gly Phe Ser Phe Leu Gln Ile Phe Ser Lys
180 185 190aat ttt gag atg ttt
gtc gtg ctg ttt gtc ctt gta ggc atg ggc cag 744Asn Phe Glu Met Phe
Val Val Leu Phe Val Leu Val Gly Met Gly Gln 195
200 205atc tcc aac tat gtg gca gca ttt gtc ctg ggg aca
gaa att ctt ggc 792Ile Ser Asn Tyr Val Ala Ala Phe Val Leu Gly Thr
Glu Ile Leu Gly 210 215 220aag tca
gtt cgt ata ata ttc tct acg tta gga gtg tgc ata ttt tat 840Lys Ser
Val Arg Ile Ile Phe Ser Thr Leu Gly Val Cys Ile Phe Tyr 225
230 235gca ttt ggc tac atg gtg ctg cca ctg ttt gct
tac ttc atc cga gac 888Ala Phe Gly Tyr Met Val Leu Pro Leu Phe Ala
Tyr Phe Ile Arg Asp240 245 250
255tgg cgg atg ctg ctg gtg gcg ctg acg atg ccg ggg gtg ctg tgc gtg
936Trp Arg Met Leu Leu Val Ala Leu Thr Met Pro Gly Val Leu Cys Val
260 265 270gca ctc tgg tgg ttc
atc cct gag tcc ccc cga tgg ctc atc tct cag 984Ala Leu Trp Trp Phe
Ile Pro Glu Ser Pro Arg Trp Leu Ile Ser Gln 275
280 285gga cga ttt gaa gag gca gag gtg atc atc cgc aag
gct gcc aaa gcc 1032Gly Arg Phe Glu Glu Ala Glu Val Ile Ile Arg Lys
Ala Ala Lys Ala 290 295 300aat ggg
att gtt gtg cct tcc act atc ttt gac ccg agt gag tta caa 1080Asn Gly
Ile Val Val Pro Ser Thr Ile Phe Asp Pro Ser Glu Leu Gln 305
310 315gac cta agt tcc aag aag cag cag tcc cac aac
att ctg gat ctg ctt 1128Asp Leu Ser Ser Lys Lys Gln Gln Ser His Asn
Ile Leu Asp Leu Leu320 325 330
335cga acc tgg aat atc cgg atg gtc acc atc atg tcc ata atg ctg tgg
1176Arg Thr Trp Asn Ile Arg Met Val Thr Ile Met Ser Ile Met Leu Trp
340 345 350atg acc ata tca gtg
ggc tat ttt ggg ctt tcg ctt gat act cct aac 1224Met Thr Ile Ser Val
Gly Tyr Phe Gly Leu Ser Leu Asp Thr Pro Asn 355
360 365ttg cat ggg gac atc ttt gtg aac tgc ttc ctt tca
gcg atg gtt gaa 1272Leu His Gly Asp Ile Phe Val Asn Cys Phe Leu Ser
Ala Met Val Glu 370 375 380gtc cca
gca tat gtg ttg gcc tgg ctg ctg ctg caa tat ttg ccc cgg 1320Val Pro
Ala Tyr Val Leu Ala Trp Leu Leu Leu Gln Tyr Leu Pro Arg 385
390 395cgc tat tcc atg gcc act gcc ctc ttc ctg ggt
ggc agt gtc ctt ctc 1368Arg Tyr Ser Met Ala Thr Ala Leu Phe Leu Gly
Gly Ser Val Leu Leu400 405 410
415ttc atg cag ctg gta ccc cca gac ttg tat tat ttg gct aca gtc ctg
1416Phe Met Gln Leu Val Pro Pro Asp Leu Tyr Tyr Leu Ala Thr Val Leu
420 425 430gtg atg gtg ggc aag
ttt gga gtc acg gct gcc ttt tcc atg gtc tac 1464Val Met Val Gly Lys
Phe Gly Val Thr Ala Ala Phe Ser Met Val Tyr 435
440 445gtg tac aca gcc gag ctg tat ccc aca gtg gtg aga
aac atg ggt gtg 1512Val Tyr Thr Ala Glu Leu Tyr Pro Thr Val Val Arg
Asn Met Gly Val 450 455 460gga gtc
agc tcc aca gca tcc cgc ctg ggc agc atc ctg tct ccc tac 1560Gly Val
Ser Ser Thr Ala Ser Arg Leu Gly Ser Ile Leu Ser Pro Tyr 465
470 475ttc gtt tac ctt ggt gcc tac gac cgc ttc ctg
ccc tac att ctc atg 1608Phe Val Tyr Leu Gly Ala Tyr Asp Arg Phe Leu
Pro Tyr Ile Leu Met480 485 490
495gga agt ctg acc atc ctg aca gcc atc ctc acc ttg ttt ctc cca gag
1656Gly Ser Leu Thr Ile Leu Thr Ala Ile Leu Thr Leu Phe Leu Pro Glu
500 505 510agc ttc ggt acc cca
ctc cca gac acc att gac cag atg cta aga gtc 1704Ser Phe Gly Thr Pro
Leu Pro Asp Thr Ile Asp Gln Met Leu Arg Val 515
520 525aaa gga atg aaa cac aga aaa act cca agt cac aca
agg atg tta aaa 1752Lys Gly Met Lys His Arg Lys Thr Pro Ser His Thr
Arg Met Leu Lys 530 535 540gat
ggt caa gaa agg ccc aca atc ctt aaa agc aca gcc ttc 1794Asp
Gly Gln Glu Arg Pro Thr Ile Leu Lys Ser Thr Ala Phe 545
550 555taacatcgct tccagtaagg gagaaactga agaggaa
18313557PRTMus musculus 3Met Arg Asp Tyr Asp Glu Val
Thr Ala Phe Leu Gly Glu Trp Gly Pro1 5 10
15Phe Gln Arg Leu Ile Phe Phe Leu Leu Ser Ala Ser Ile
Ile Pro Asn 20 25 30Gly Phe
Asn Gly Met Ser Ile Val Phe Leu Ala Gly Thr Pro Glu His 35
40 45Arg Cys Leu Val Pro His Thr Val Asn Leu
Ser Ser Ala Trp Arg Asn 50 55 60His
Ser Ile Pro Leu Glu Thr Lys Asp Gly Arg Gln Val Pro Gln Lys65
70 75 80Cys Arg Arg Tyr Arg Leu
Ala Thr Ile Ala Asn Phe Ser Glu Leu Gly 85
90 95Leu Glu Pro Gly Arg Asp Val Asp Leu Glu Gln Leu
Glu Gln Glu Ser 100 105 110Cys
Leu Asp Gly Trp Glu Tyr Asp Lys Asp Val Phe Leu Ser Thr Ile 115
120 125Val Thr Glu Trp Asp Leu Val Cys Lys
Asp Asp Trp Lys Ala Pro Leu 130 135
140Thr Thr Ser Leu Phe Phe Val Gly Val Leu Met Gly Ser Phe Ile Ser145
150 155 160Gly Gln Leu Ser
Asp Arg Phe Gly Arg Lys Asn Val Leu Phe Leu Thr 165
170 175Met Gly Met Gln Thr Gly Phe Ser Phe Leu
Gln Val Phe Ser Val Asn 180 185
190Phe Glu Met Phe Thr Val Leu Phe Val Leu Val Gly Met Gly Gln Ile
195 200 205Ser Asn Tyr Val Ala Ala Phe
Val Leu Gly Thr Glu Ile Leu Ser Lys 210 215
220Ser Ile Arg Ile Ile Phe Ala Thr Leu Gly Val Cys Ile Phe Tyr
Ala225 230 235 240Phe Gly
Phe Met Val Leu Pro Leu Phe Ala Tyr Phe Ile Arg Asp Trp
245 250 255Arg Met Leu Leu Leu Ala Leu
Thr Val Pro Gly Val Leu Cys Gly Ala 260 265
270Leu Trp Trp Phe Ile Pro Glu Ser Pro Arg Trp Leu Ile Ser
Gln Gly 275 280 285Arg Ile Lys Glu
Ala Glu Val Ile Ile Arg Lys Ala Ala Lys Ile Asn 290
295 300Gly Ile Val Ala Pro Ser Thr Ile Phe Asp Pro Ser
Glu Leu Gln Asp305 310 315
320Leu Asn Ser Thr Lys Pro Gln Leu His His Ile Tyr Asp Leu Ile Arg
325 330 335Thr Arg Asn Ile Arg
Val Ile Thr Ile Met Ser Ile Ile Leu Trp Leu 340
345 350Thr Ile Ser Val Gly Tyr Phe Gly Leu Ser Leu Asp
Thr Pro Asn Leu 355 360 365His Gly
Asp Ile Tyr Val Asn Cys Phe Leu Leu Ala Ala Val Glu Val 370
375 380Pro Ala Tyr Val Leu Ala Trp Leu Leu Leu Gln
Tyr Leu Pro Arg Arg385 390 395
400Tyr Ser Ile Ser Ala Ala Leu Phe Leu Gly Gly Ser Val Leu Leu Phe
405 410 415Met Gln Leu Val
Pro Ser Glu Leu Phe Tyr Leu Ser Thr Ala Leu Val 420
425 430Met Val Gly Lys Phe Gly Ile Thr Ser Ala Tyr
Ser Met Val Tyr Val 435 440 445Tyr
Thr Ala Glu Leu Tyr Pro Thr Val Val Arg Asn Met Gly Val Gly 450
455 460Val Ser Ser Thr Ala Ser Arg Leu Gly Ser
Ile Leu Ser Pro Tyr Phe465 470 475
480Val Tyr Leu Gly Ala Tyr Asp Arg Phe Leu Pro Tyr Ile Leu Met
Gly 485 490 495Ser Leu Thr
Ile Leu Thr Ala Ile Leu Thr Leu Phe Phe Pro Glu Ser 500
505 510Phe Gly Val Pro Leu Pro Asp Thr Ile Asp
Gln Met Leu Arg Val Lys 515 520
525Gly Ile Lys Gln Trp Gln Ile Gln Ser Gln Thr Arg Met Gln Lys Asp 530
535 540Gly Glu Glu Ser Pro Thr Val Leu
Lys Ser Thr Ala Phe545 550 55541888DNAMus
musculusCDS(60)..(1730) 4ctcccgcgcc acggtgtccc cttattccca tacgggcgct
gtgggaggct gaggacggc 59atg cgg gac tac gac gag gtg acc gcc ttc cta
ggc gag tgg ggg ccc 107Met Arg Asp Tyr Asp Glu Val Thr Ala Phe Leu
Gly Glu Trp Gly Pro1 5 10
15ttc cag cgc ctc atc ttc ttc ctg ctc agc gcc agc atc atc ccc aat
155Phe Gln Arg Leu Ile Phe Phe Leu Leu Ser Ala Ser Ile Ile Pro Asn
20 25 30ggc ttc aat ggt atg tcc atc
gtg ttc ctg gcg ggg acc ccg gag cac 203Gly Phe Asn Gly Met Ser Ile
Val Phe Leu Ala Gly Thr Pro Glu His 35 40
45cgt tgc ctt gtg cct cac acc gtg aac ctg agc agc gcg tgg cgc
aac 251Arg Cys Leu Val Pro His Thr Val Asn Leu Ser Ser Ala Trp Arg
Asn 50 55 60cac agt atc ccg ttg gag
acg aag gac gga cga cag gtg cct cag aaa 299His Ser Ile Pro Leu Glu
Thr Lys Asp Gly Arg Gln Val Pro Gln Lys65 70
75 80tgc cgc cgc tac cga ctg gcc acc atc gcc aac
ttc tct gag cta ggg 347Cys Arg Arg Tyr Arg Leu Ala Thr Ile Ala Asn
Phe Ser Glu Leu Gly 85 90
95ctg gag ccg ggg cgg gac gtg gac ctg gag cag ctg gag cag gag agc
395Leu Glu Pro Gly Arg Asp Val Asp Leu Glu Gln Leu Glu Gln Glu Ser
100 105 110tgc ctg gat ggc tgg gag
tac gac aag gac gtc ttc ctg tcc acc atc 443Cys Leu Asp Gly Trp Glu
Tyr Asp Lys Asp Val Phe Leu Ser Thr Ile 115 120
125gtg aca gag tgg gac ctg gtg tgt aag gat gac tgg aaa gcc
cca ctc 491Val Thr Glu Trp Asp Leu Val Cys Lys Asp Asp Trp Lys Ala
Pro Leu 130 135 140acc acc tcc ttg ttt
ttc gtg ggt gtg ctg atg ggc tcc ttc att tca 539Thr Thr Ser Leu Phe
Phe Val Gly Val Leu Met Gly Ser Phe Ile Ser145 150
155 160gga cag ctc tca gac agg ttt ggt cgc aag
aat gtg ctg ttt ttg acc 587Gly Gln Leu Ser Asp Arg Phe Gly Arg Lys
Asn Val Leu Phe Leu Thr 165 170
175atg ggc atg cag act ggc ttc agc ttc ctg cag gtc ttc tct gtg aac
635Met Gly Met Gln Thr Gly Phe Ser Phe Leu Gln Val Phe Ser Val Asn
180 185 190ttc gag atg ttt aca gtg
ctt ttt gtc ctt gtt ggc atg ggt cag atc 683Phe Glu Met Phe Thr Val
Leu Phe Val Leu Val Gly Met Gly Gln Ile 195 200
205tcc aac tac gtg gca gca ttt gtc ctg gga aca gaa att ctt
tcc aag 731Ser Asn Tyr Val Ala Ala Phe Val Leu Gly Thr Glu Ile Leu
Ser Lys 210 215 220tca att cga att ata
ttc gcc acc tta gga gtt tgc ata ttt tat gcg 779Ser Ile Arg Ile Ile
Phe Ala Thr Leu Gly Val Cys Ile Phe Tyr Ala225 230
235 240ttt ggc ttc atg gtg ctg cca ctg ttt gca
tac ttc atc aga gac tgg 827Phe Gly Phe Met Val Leu Pro Leu Phe Ala
Tyr Phe Ile Arg Asp Trp 245 250
255agg atg ctg ctg ctg gcg ctc act gtg cca ggg gtg cta tgt ggg gct
875Arg Met Leu Leu Leu Ala Leu Thr Val Pro Gly Val Leu Cys Gly Ala
260 265 270ctc tgg tgg ttc atc cct
gag tcc cca cga tgg ctc atc tct caa ggc 923Leu Trp Trp Phe Ile Pro
Glu Ser Pro Arg Trp Leu Ile Ser Gln Gly 275 280
285cga att aaa gag gca gag gtg atc atc cgc aaa gct gcc aaa
atc aat 971Arg Ile Lys Glu Ala Glu Val Ile Ile Arg Lys Ala Ala Lys
Ile Asn 290 295 300ggg att gtt gca cct
tcc act atc ttc gat cca agt gag tta caa gac 1019Gly Ile Val Ala Pro
Ser Thr Ile Phe Asp Pro Ser Glu Leu Gln Asp305 310
315 320tta aat tct acg aag cct cag ttg cac cac
att tat gat ctg atc cga 1067Leu Asn Ser Thr Lys Pro Gln Leu His His
Ile Tyr Asp Leu Ile Arg 325 330
335aca cgg aat atc agg gtc atc acc atc atg tct ata atc ctg tgg ctg
1115Thr Arg Asn Ile Arg Val Ile Thr Ile Met Ser Ile Ile Leu Trp Leu
340 345 350acc ata tca gtg ggc tat
ttt gga cta tct ctt gac act cct aac ttg 1163Thr Ile Ser Val Gly Tyr
Phe Gly Leu Ser Leu Asp Thr Pro Asn Leu 355 360
365cat ggg gac atc tat gtg aac tgc ttc cta ctg gcg gct gtt
gaa gtc 1211His Gly Asp Ile Tyr Val Asn Cys Phe Leu Leu Ala Ala Val
Glu Val 370 375 380cca gcc tat gtg ctg
gcc tgg ctg ttg ttg cag tac ttg ccc cgg cga 1259Pro Ala Tyr Val Leu
Ala Trp Leu Leu Leu Gln Tyr Leu Pro Arg Arg385 390
395 400tat tct atc tcg gct gcc ctt ttc ctg ggt
ggc agt gtc ctt ctc ttc 1307Tyr Ser Ile Ser Ala Ala Leu Phe Leu Gly
Gly Ser Val Leu Leu Phe 405 410
415atg cag ctg gtg cct tca gaa ttg ttt tac ttg tcc act gcc ctg gtg
1355Met Gln Leu Val Pro Ser Glu Leu Phe Tyr Leu Ser Thr Ala Leu Val
420 425 430atg gtg ggg aag ttt gga
atc acc tct gcc tac tcc atg gtc tat gtg 1403Met Val Gly Lys Phe Gly
Ile Thr Ser Ala Tyr Ser Met Val Tyr Val 435 440
445tac aca gct gag ctg tac ccc act gtg gtc aga aac atg ggt
gtg ggg 1451Tyr Thr Ala Glu Leu Tyr Pro Thr Val Val Arg Asn Met Gly
Val Gly 450 455 460gtc agc tcc aca gca
tcc cgc ctt ggc agc atc ctg tct ccc tac ttt 1499Val Ser Ser Thr Ala
Ser Arg Leu Gly Ser Ile Leu Ser Pro Tyr Phe465 470
475 480gtt tac cta ggt gcc tat gat cgc ttc ctg
cct tat att ctc atg gga 1547Val Tyr Leu Gly Ala Tyr Asp Arg Phe Leu
Pro Tyr Ile Leu Met Gly 485 490
495agt ctg acc atc ctg aca gct atc ctc acc ttg ttc ttc cct gag agc
1595Ser Leu Thr Ile Leu Thr Ala Ile Leu Thr Leu Phe Phe Pro Glu Ser
500 505 510ttt ggt gtc cct ctc cca
gat acc att gac cag atg cta agg gtc aaa 1643Phe Gly Val Pro Leu Pro
Asp Thr Ile Asp Gln Met Leu Arg Val Lys 515 520
525gga ata aaa cag tgg caa atc caa agc cag aca aga atg caa
aaa gat 1691Gly Ile Lys Gln Trp Gln Ile Gln Ser Gln Thr Arg Met Gln
Lys Asp 530 535 540ggt gaa gaa agc cca
aca gtc cta aag agc aca gcc ttc taacaccctg 1740Gly Glu Glu Ser Pro
Thr Val Leu Lys Ser Thr Ala Phe545 550
555tccagaaggc aaaaaactga ttggaaacct tcatgttgtc agaaatgctc tccatgactg
1800agggcttttc tgttctgtta accttgtgtc taacatgctc atggattggg gcatctgtcc
1860tggagagtca ccttcctcta gggacacc
1888525871DNAHomo
sapiensexon(1)..(614)intron(615)..(8636)exon(8637)..(8740)intron(8741)..(-
14409)exon(14410)..(14564)intron(14565)..(15590)exon(15591)..(15762)intron-
(15763)..(17282)exon(17283)..(17409)intron(17410)..(19178)exon(19179)..(19-
279)intron(19280)..(20947)exon(20948)..(21162)intron(21163)..(22690)exon(2-
2691)..(22873)intron(22874)..(23934)exon(23935)..(24070)intron(24071)..(24-
443)exon(24444)..(25871) 5gcggcccagg cccggaacct tccctggtcg tgcgccatat
gtaaggccag ccgcggcagg 60accaaggcgg cggtgtcagc tcgcgagcct accctccgcg
gacggtcttg ggtcgcctgc 120tgcctggctt gcctggtcgg cggcgggtgc cccgcgcgca
cgcgcaaagc ccgccgcgtt 180cccagacccc aggccgcgct ctgtgggcct ctgagggcgg
catgcgggac tacgacgagg 240tgaccgcctt cctgggcgag tgggggccct tccagcgcct
catcttcttc ctgctcagcg 300ccagcatcat ccccaatggc ttcaccggcc tgtcctccgt
gttcctgata gcgaccccgg 360agcaccgctg ccgggtgccg gacgccgcga acctgagcag
cgcctggcgc aaccacactg 420tcccactgcg gctgcgggac ggccgcgagg tgccccacag
ctgccgccgc taccggctcg 480ccaccatcgc caacttctcg gcgctcgggc tggagccggg
gcgcgacgtg gacctggggc 540agctggagca ggagagctgt ctggatggct gggagttcag
tcaggacgtc tacctgtcca 600ccattgtgac cgaggtgggt gccggcccct gctggggctg
agaccagggc tcggaggacc 660tgtcgcggtc cttgaacccg agctcctctc tcccagatgc
gcactggacg ctgtcactcc 720ccctccccca acggtcaaca ccctagcgat ggagaccctc
cagccaggtg gcttgggaac 780gcttcacgag gtgacctcca gccacagtgt gctcctccct
gcacaggtgg tcagtctggc 840ctcccgtcct gatggccact ttgaagaggg taccaggaag
gtcctggcgg tccctgggcg 900atgctctatg gccctgtgtg tccaggactt actctagttg
gggttggggg tggtaagtag 960cagagccagg acttgggcca ggggctatcc cgtttttcct
ctagtctctt gatttctttt 1020tagaagagaa gaaatacttc tctttcctga acttttaaaa
gttaaataaa gcatgtgtat 1080acaactgcct cttccctttt tcctctagtt actccttccc
ctaccgtcca caacccaaaa 1140acgacaatct ggtcatgccc tgtaagtaat tgtttgcctt
ttcccatggt cagttgtcag 1200tctttttttt tttttttttt ttttgagaca gagtctccct
ctgtcaccag gctggagcgc 1260agtcgtgtgt tcttggctca ctgcaacctt cgcagtcgtg
cgttcttggc tcactgcaat 1320cttcgccttc cgggctcaag tgattctcct gcctcagcct
ccgagtagct gtgagccacg 1380acgcccagct aatttttgtg tttttagtag agacggggtt
tcaccgtgtt ggccaggatg 1440gtctcgatct cttgacctcg tgatctgcct gcttctgcct
gcaaagctgg gattacaacc 1500gtgagccacc acgccaggcc gtcagttgtc actctttaag
atccattcat ctgaagatgg 1560gttcagggtg acttgttgac ctggaatatt ttctcaggta
ttatgaggca aggctgtcgg 1620ccagatttag ttaaagcata cagccttagg tcatagggtg
tgggggagcc tttctcattt 1680ctcatcccct tggattttcc ctctgggtgg ttttgtctgt
cccctccgaa cctgttggag 1740cagttgttgg agctggatgt aggaacatga tgttaatgat
gtatgtgttt tgtgtctttt 1800ttagacactg gcactctagc tccctgaagt ttcagcagca
ttgagtaagt agccagtgag 1860tagccctcat tgatagatag gctcactaaa tgtgcagatg
accaattcgc aggttagaga 1920aggcttccca gaagaggagg cctctcagtg agcctagggt
gttcagctca gcataatagt 1980gagctgaagg ccccaatgca gcagcaagaa accacccagc
aggacggcgg agttcacaga 2040gaggggagag ttcatagaga gggagagtgc cacaggccct
ggcacagctt caagccctgc 2100tggatgttgg tgctgagcct cccctcctgg agcctcagag
gggcttacag gggctctgga 2160gatcccaact gtgttgcttc ttggcgtcat cacccttcaa
tggagtctga gagctaccct 2220gggagatcca agtgtgtgtg catgtgtgtg ttttttcttt
ttggaaattt gatgtcccca 2280gcatttggac ctgctttctc cacatatatg tagtgggagt
gtgaccggag ccccactggg 2340atttcttagc tagtgacata gcttcagtgt ccagaggcat
catggcttga caaaagaggc 2400actccttgga ggtagccaac tgtgctttct ggcccatgaa
tccacaggaa attggaactt 2460tcagttgcca caaatcctgg gtcctagcct cacaggtggg
tgtgttacct tcccggaagc 2520cagtgagtac tatgaaagca gaggctgtcc ctgaggttgc
aggcagaggc cacagagggg 2580aacatgacac aggaatccct acaaattcta cttggggctg
cctaaagaag agggaagtag 2640tgaagcaaga agaagcacat ggcatctctt ggagttttac
attgacccct gagggttccc 2700cggcttactc tagtcacttg ttcctgcttt gctgcctcca
tcccacattg ggctgagtga 2760tggtggcatt gatgagctcc caaaggccag ctgtgccagg
gggtctgacc ttatcttgct 2820gccaatgtca gccttttgtt ttttaatatt tagactattt
atttagctgt cttagcagtt 2880tcaaaggagt tatgtgccct ttcacctact tatatgttgt
cagtctttgc agggaggcca 2940gattaatgct tagatctttg ttttgggcta ctggaatgct
tgacttgaag ttcagagctg 3000cttgttccca ggtgaacagc tactgctgga agttgctgca
tcaacattct aatggctttt 3060tctatggcct gttgtctttc aacccaaacc tggcctgctg
aacactgcat ctagtcccat 3120gcctgctaaa tgtctctaag cctgccctct gccccaaatc
atacataaag gtgtttgtaa 3180gtacactggt attgaattac tagtcatatt ttttccactg
aagactggaa cctcaggtgt 3240cctgtttgga tttttttaat ttgttcaagt taaagtacat
acatgtagta ccaaactttg 3300tggtggattt agatctttgc gtcttcctca atttctgaca
caattctgga tgcagaggga 3360ggttctcagg aaaattttta ttgaatgagt taatgaataa
tttaagaaat catctctaaa 3420gtttgagaac taaagaaaaa tagttcagtt cttagaaggg
aaacttgagg gtggctgaaa 3480aggattgact ggaatttttt aaaggaaatg tgactccccc
tgcccactga ctggggcttt 3540gatgccacat ggatgtggaa tgaggtgttg ggattggcag
agggaatctg ctagcaatta 3600ataaataaat aaatattggc agggcgtggt ggctcacgcc
tgtaatccca gcactttggg 3660aagccaaggc agcaggtcac ttgaggtcgg gagttcaaga
ccagtctggc caacatggtg 3720aaactccatc tctactaaaa atacaaaaat tagctgggtg
tggtggcaca tgcctgtatt 3780cccagctact tgggagactg aagcaggaga atcgcttgaa
cccaggaggc gggctttgca 3840gtgaaccgag atcgagcctc tgcactctag cctgggcgac
aaagcgagac tccatctcaa 3900aagaataata ataatattaa taaataaaaa tgatttatga
ggtaaaagag ttttatgccc 3960ccatgttcca ggaatagttt ggtggtccac atggttctcg
gctggcctct cctctggccc 4020ctcagtcatc cctggggtac tggggaatta gccaacccat
catgcagtgc ttcttggcca 4080tggactgccc catctgctgg aaacctgggt tgtttctgag
gttgtctggg ctgtccgctc 4140tttggtttca ccatagctct gtccagccta tggacagaca
ggttccttga gaaacttccg 4200gctgggtgtg gtggctcatg cctgtaaacc cagcactttg
ggaggctgag gcaggtggat 4260cacaaggcag gagttcgaga ccagcctggc caacatggtg
aaacctcgtc tccgctaaaa 4320atacaaaaat tagccctgcg tggtggcggg tgcctgtaat
cccagctact cgggagggag 4380gctgaggcag gagaatttct tgaacctggg aagtgggggt
tgcagtagcc aagatcgcac 4440cattgcatgc cagcctgggt gacaagagca agactccgtc
tcaaaaaaaa aaaaagaaaa 4500agaaaagaaa cttccaagct gctctgcatc gccttgctct
ccacctgtct gcttctaaga 4560agccctcggc ccagtcctgg gtgggactcc cactccctcc
ccattgtcct ggactagctt 4620ttctatcagc cttatcttgt gtagagacag atagtcttag
aagatgagag agccctcact 4680gttatcccca aagctgcctg gaggaaaagc cagagcaacc
tgggagctgg gaccggggct 4740gactctgggc agcagagacc cgagagacct ggaacttgaa
cctcactgtt acgcctttgt 4800tgatttctct cactcagggg acacacagac cctcatccag
ccttttgcag ctatatggca 4860aggcagagaa gccacttgcg gggtcccgtg gcccactatg
cacgtacata gtagacacat 4920ctggccatga gtggtcagat tgagccactc tctagctagc
tgacacctgt catcctgggt 4980caaatttctg acagttgaca caaagcaggg ggtcagggag
ccaaaaaaaa aaatggccag 5040gtgtggtggc tcttgcctgt aatcccaggg ctttgggagg
ccaaggtggg cagataacct 5100gaggtcagag ttcaagacca gcctggtcaa catggcaaaa
ccccgtctct actaaaaata 5160caaaaattag ccaggctggt ggtgcatccc tgtagtccca
gccactcagg aggctgaggc 5220atgagaatca cttgaacccg ggaggtggag gttgcagtga
gccgagattg tgccactgca 5280ctccagcctg ggagacagag caagactctg tttcaaaaga
aaaaaaagag tattctggag 5340attgaagttc aggagttcag ggttcatctc gactttgggc
agccaagcaa gaactaaagt 5400ataccaagat gttgaaggtt gatacctttt tatttatcga
ttcattcact cacctatgta 5460ccaaagagct cctgagcctc tcttcgatac agggggcact
gccaggagtt gtagaggatg 5520tgatagcaaa gataggaaat accttttctc tttgctctga
caacggtggg gcaaggattc 5580atcattgatt tcagcaggag gcaggataaa atgtgtgtag
gaatatagga atacatggca 5640atcagtaaca tgtggtacct agcagcatgt ctgactgttg
atacggtcag gctaggtaca 5700tcccctcagg gaagaacttc tgtcttaggg gcacacaccc
tatctttttt cccttcctgc 5760caattcacag gtaagaacat ttagtcccag ggaactatgt
catctctcta cttctcataa 5820ctgaaaaagc agtgccaatt atgtatgagg tataggagac
acaattctcc ctctttttaa 5880aaatgtttaa tagctttatt aaggtgtaaa tgacataaaa
actgcatgta gctaaagcat 5940gcaatttggc acacatgtat ctctacaccc ttgaaaccat
caccactaga aaggtgcatt 6000tctccccagg aagaggggca agtctaggcc ctttgccaga
gttgctccca gattgttttc 6060aggttgggcc tgcattcaca gctcagccag ctgaagggtg
acagcattag actcgtgacc 6120caaatcttaa acccacacat tccattttaa cattgatacc
tgtgatcatc agccagtata 6180gcccatccca tgtgccaggc ggaggcatca ataagctggt
ctgtagcagc tttaatcaca 6240gctggggagc caggagctaa ggaatgctag actcctttgt
aaacaattta agtagggggt 6300acttagcctg gactctatgt gcttctcatc cacctcttct
cagggagatt ctggccaagt 6360cctggaacct actgcagtcc atctggtggt tgacagatat
gtggacggat tggcaggctg 6420ggacccaatc tatgtttgcc cttgtgttca gttttgagac
ctagcacctt tcctgatcct 6480gctcacagac cccctgcggc caataggaaa gaagtgttaa
tgcatatttg cttttggagg 6540gcccaaagcc aggcccagag agttgtcaag ggcggtcagt
ggtgggtgga tggcagagtt 6600aaccaaggag ttacacacct gcctagacta aggacaggct
ggaccaagta gagagggtgg 6660ggctaaggga gcctgagaga tgctctgggg cctgtctcaa
aatgagcact atagtcaccc 6720tgtcccctgc agagattgtc tgacctggtt ttaggtcaca
cccaaccttg ccagccaagg 6780agtctttaga agcctgatat tgggagacct gtcctggggt
ctacaacccc agaactcact 6840gcagaagccc acgtggattg ctagtctagc tcagccatat
gggtccccaa ccctcacctc 6900atgatagtcc tgtgagaaac cgctgctgac cctttgttca
tgttttcatc ttttccacta 6960taaaagacat gctagctggg aaatagagcc catcatactc
aagagtggca ggagccaggt 7020cctggcccct gaagcttggc ctcacacaca gaggccggca
ccctgtcatc aattccctca 7080gctttttctc cgcctccact cccagtccta gatttagcag
ccatgtgtgg gtgggggcca 7140ctgcagggat acttacccac ctaccagaga gatggcctgt
gggtgctggc ccttctgagg 7200ctgtggaggt tggaggctgt ggcagcctgg gcagtcaggc
tgtggtcctc ccatgttctt 7260gactcctgct agtctgggct gcctcctgat taggggttgg
atgctccagt tcttccctgg 7320gttggggatt gccaccctac tcccagccca tccaggttca
cgcttattcc aaagcggagc 7380accagcagtg tgcctgctgc gggagttctc cgtgtccagc
ctgagggttg cctgccaacc 7440cctctgagag gtgcccggag gctgtgcgcc cacactgccc
agcagtgcgg agaagcaggc 7500ttgtttttcc ctgtcactgg cttggaagag atgctttgtt
ctagggagcc gcatgtcccc 7560ttgcctgcgt tgttggtgag gagccagcag gctccgtgga
gggcaggcta gcagcctggc 7620accagggagg caagggtctg agttcctagg agggtggttg
ctcatgtgag aagtctgcaa 7680aggttactac tgagcaccat ctctctgtct gaaaaacatt
tttcattttt ctgtgaacca 7740ctaagtttcc cgtttgggct ttcttcctgc ttttggctct
tgtttaggca ggcgtagcca 7800gatccaagcg tctggctgct tccccatgtc ttcagacctc
tttttctgtt catggtaact 7860atagatggaa ccacacattg gaagctggaa actcaagcgg
tgcagcctat tccttacccc 7920aatccctgtt ttacaaatgg ggaaatcaag gcacagcatg
gggtgatgct tatctgaggt 7980tggaagagtt aatagtagag tgggagctaa aacccagttt
cttacctcca agctcagggc 8040tttcagctgt aattgagcct agtatagttg gtgtgcagca
tcagggattc cagctctaaa 8100ggtcacaaaa aggacccggg ggtcattggc ccagggtggg
aacctgagca gagcaggtcc 8160agatggtgca ctctgtgccc tggccttagt ttcttgctgg
atgctttggc ccgtagagcc 8220ccagagccct gcttccagaa ccactccagt gacgttcatg
ccaatggcct gaaccccact 8280gagcgagggt gccctgcctc ttccacagcc ctgggctccg
ctcagatttt taggagcaag 8340cgttagaggc cttgctttct ccagggtcag catgtggaca
gaacacttac tctctgcctg 8400tctctcctcc tcaaaatgga agcaagacag tggggcctac
aatgctatga aaaacaggat 8460gggaaagaag cctgctctct gccttcctgc ccaggtgagc
catcacctga ctaagtgagt 8520tcacactcag agcgtgtggg gatggcagga tgttctgact
tcattttcca ggatgccttt 8580gctttaaaac cttttaaaaa gaagtgaatg atacaccccc
tttgctcatc ttgcagtgga 8640acctggtgtg tgaggacgac tggaaggccc cactcacaat
ctccttgttc ttcgtgggtg 8700tgctgttggg ctccttcatt tcagggcagc tgtcagacag
gtaaggtgtc tgtcttctgg 8760agcaccaggg gacctcagca ctgaggaaga agcgtgtgcc
tggcccttga tttcagttgg 8820tagtattctt tcagcgcagg gccctgtatt ttaaagaaga
ggaagctatg tctgtgatat 8880agactccatg cctagtaaga agagccaaca aatcatctga
ctccgtaatt cttgctaagt 8940aaagaaacct gagctgtcta agctgaatgt atctgtgatc
cggttgacta ggtaatatgc 9000catgattcac ttctgcagta gcctggcttg cctcccctgg
gtcactgtga ctctgtcatg 9060cccctgagca tgggagaggt tgacatcatg cacacatgca
catgtgctag attgtagatc 9120tgtagtagtg ccacggtgtc tgcctctgta gtcccaagaa
gaccagcatt ctctctgcaa 9180agtgaaagga gctctcacca gccactagtg gtatgaaaag
cagaactctt ttgtccacaa 9240ggctgatgcc ccttagctaa gtggcctgtg gttttggcat
ttactttatg acaggaggga 9300gaatagtgtt ttgatccatt tcttataagc aggttatttg
tataattcta aagcttttaa 9360ctcaaggaaa cattaacggc ttagagaatc ccaaacccct
cgaaattata tgcacaatgt 9420tagagactac atgtgagcat ttttttggag agaggtccgt
agctttcatg aagttcttag 9480aggggtccat tatgttgtgt cttctttccc tggagcttca
gggtctgtca gagaagactg 9540tgaagagagt agcagccttc agcaagtcct tggccacatg
gcacatgtga gaacacccca 9600caaatcggtg ggttagccgg tggaaaggag tcccagcatc
ttccctggtt tttaattcct 9660ggcctcaagc aatcctccct ccataacctc ccaaagtgct
gggattacag gcgtgagaca 9720ccatgcgcag ccagatattt tttattgttg ttgttttttt
ttggaaaagg agtttcactc 9780ttgttaccca ggctggagtg caatggcacg atctcggctc
actgcaacct ctgcctccca 9840ggttcaagag attctcctgt ctcagcctcc tgagttgctg
ggattacagg catgcaccac 9900catgcccagc taattttgta tttttaatag agacaaggtt
tctccatgtt ggtcaggctg 9960gtctcaaact cccgacctca ggtgatccac ccacctcggc
ctcccaaagt gctgggatta 10020caggcgcgag ccactgcacc tgcccagcca gatttttttt
taaaaagcag gttaacctgt 10080ttattattcc tactttacag atggagaaag tgagacagag
ggattaaata acttccccaa 10140ggtttcacag ctggcaagtg gcagagttag gatttggacc
caggtagtct tgctcctcta 10200ttgtgtatgg actactgttc taggtccctg ctgtcctaaa
acttgctttc tagcaaggtg 10260gaatgtatta aacaaccaag tgaggaagtc gttgttgtcc
ttggccatgg taagagatac 10320agagaagtgc agggtgccac aggagtgtct aacagaggag
gtcaggggca gcatccctga 10380gaaggtgaag catgagcgag agtgggaaga tgagtcgaaa
gtagccagct gaggggtaga 10440gaggagaaag aacatccagg cagggagaat agcaagtgct
aaagccgggg ctcatgaaaa 10500ggcatgggag caggacaaag tccgtgtggt agaggtgcgg
agagtggtgt taagatgaag 10560gggagaggca ggcagagccc tgggcagatg agcaaccagg
gcttagtgga tcacagttag 10620gactttgggc ttcagccaca gagcagcggt gggctactga
gattttttaa agcaatagtg 10680tgacaatcag atttgtcctt tttttttttt taaagatgct
ttgacaacct tgtgaagaaa 10740gaattgaagg gaagcaaaag gtgttgtaga gagaccaatt
aaaaggttgt cacagtagtt 10800catgccagag atgatggtgg catggcctag catgacggtg
gtagaaatgg agggaagtgg 10860taagaggtaa aatcaacaag acttgccgat gggctggatg
tcggaattgg gggaagaaag 10920ctttctggcc tgagtaactg ggtgaatgaa ggtacacttc
tctaagacag agaatgctgg 10980aaaagaacca cgttcatgga tattgagttc aatttgtgtg
tactaaattt ggggtgacta 11040tgagacccct aagtggagaa gtagagtgtg aagctggctg
tatggatatg gtactgcatg 11100agaggttttg gctagagaaa cacatgtagg atttgtcagc
atatagaacc ctcagcagag 11160ccccagatag ggatgaggtt gcctggggag aggggagtgg
ggaggggagg ggggaactgg 11220gggaggattg tgctatgctt agaggccacc tgagtggaca
tgggagattc cttctcacgg 11280agctaagtga cagctgcggc tcagaagaat gcccttgatg
tgaaaaggag ataccccaag 11340ttcagagtag aaatgcagtg tatttttctg ggtcagctgt
gtcatgggtc agcttaagaa 11400ccttcttgcc atgtgacaca atgattacct gaggaaagca
tttaagttcc aaaaggtcta 11460ttcccaggga aagtggaaac aaaatttgtg agtgtgttta
ggaccacttt agtctacaga 11520agggctaata tagtgttttt caaacctttc taaacatttt
ggccacagaa ctttcattaa 11580agtaggataa tttaagtcta gtaaatgaaa tacaccctaa
gtggctaaaa gtatggctgt 11640tctgcctgca gcccctgcct tcaattccca atgccctgcc
tcaagcctgt ctgtgccccc 11700ttggaaggcc cagggccctg tgggatggac agcttctgag
tgcattactt ctgagtaaac 11760cagttctaaa acctaagcta agtagattac ctgaaagcaa
tcaattcatg ccctaacttg 11820tctaggacat caatgtaact ttttatatag tataaaggtt
tcaccttctt tctggcctat 11880gaatatgtct gaaattcaag agcaattaaa gtactcctgg
gttgtcaaag ccctttatgt 11940aacactattg agttatcttt atgcgtctga cttgtaagag
atgcacaact ctaggaagaa 12000gtagactgct gtgtcctgtt tccaggtatg tgtgtgtttg
ccattttgtt gacagacttt 12060aaaagcaaac atttctggcc ccaaccctga actgccaagg
actggtgcta tgtaaagggt 12120tctctggatc tgtctcttcc ctaccatccc agggagctct
taggaaggga aagggcatag 12180agattatacc agcctgcctt gtggttagga accacccctt
ggttggcata tagaacatgc 12240ttgttaaaaa aaccatgcag gggaaagtag agtctactac
caggcgagag tttctcaacc 12300tcgaccctat taacattttg agccaaataa ttttgttcta
gggcattttc ctgggcattt 12360tagaatattt aacaacatat ctggcctcta ctcattagat
gccagtgaac cccaagtgat 12420ggaaaaaaaa aacaacaaca gaaaaaaacc tcttttattg
aggaaaaaca ccaaactctt 12480ccacatagtt gcaagacctt gtgcaatttg cctcctagcc
accactgtac tcttgaattg 12540cacgcctgat gccaaccaca ctggttcctc atgttcacca
tgccccctcc agccatgggg 12600gtgtgtggtc ttctcagagt ctgaagcatt ccccacccac
cccaacccac cccctgtggc 12660cttctttaac catgctggct aattcaggat ccctagttcc
ttatgacttt cctttaaaac 12720gtctaccaga aattggggga aaaaaagtgt tattatagga
ttaatgttgg tcttccccac 12780tatactgtga atatcattga gagcttggtc cctacacctt
aaatccccca tcgtcaacta 12840ttttttccca tctcagtgtc ccatgatcaa ggagaccctc
cctgaatgtc cagttcccca 12900acccttaccc ccagtccagg gtagcttcct tccttgtgcc
tctcattaac ctgcatgccg 12960atccttcagt gcacttgact cagtgtgtaa ttgtatattc
agtagcgtgt tgttagatta 13020aaatgtggtt aatatgtgtt tcaccagtta tactatgaca
ctccttaagg gcagaaacag 13080catctttttt aatttattga tatccaagtg ccctctataa
tagatgctca ataaacattg 13140aatgaaagtg ggtgtcagcc agtactggcc agactcaaac
tgaacccact gcttcccact 13200agcttgactt tttcctcctg tttgtggcac tctctttaaa
acaaaccaaa ataaacccaa 13260ttttaaaaac tttttaaaat gagcacggat acagaaaacc
acacagaaca aatgtgtagc 13320ttaatgaatt tttttcagag aaataacctt atgaccacca
ccaagtcgag cagtagaact 13380ttgctgtcca ctaagaagcc ctgtccatgt gccccatccc
aattacagca tcctctctct 13440ctccccatta agtaaccgct agcctgactc ctgtaataat
cacttccttg tgagtttttt 13500tagttttatt atcgaaatat gcatccttga cacaaattta
gtgttgccca cttaatatat 13560ttgatgtctt ttagtctact taatctatgg attctccttc
tatcgccttc tatgccttac 13620tgattatcta tgaagaacct gagctattcc acctatagaa
tttcccagtc tggatttgtt 13680gattgcacac tgatgatgca gttcagcaca ttcctctatg
ctctgcattt cctcaaaatt 13740ggcagttgga tccagagact tgagattcag gttctgattc
aggttcagtc cttttggcaa 13800gaccatagga agcatgcaat tcctgactgt ctctttatga
tgttaacagt aattagtata 13860taatgcatag atctattaat ccattggggg ctataaatgg
tattattcta attttattac 13920cttttcattt aaaagttaga atacttttgt acatgatact
acctcttatc tattattggt 13980tgctgttcac atagtttaca aaggaaaatc aggacaaatg
cttctttctc tttatttgcc 14040agttttcatt tataatgaat tgtttctctg ttattctcca
aatttggcag attctttttt 14100aaaaaaaata tcattatgaa tgtatggatt aaataattga
tgtatttcag tctcttgcaa 14160tcattatcat aattgtagca ttgcttttta ggcaaccctg
gtacccaggc tgtacatttg 14220tcatggggag tggggagggg gagaaatagc atgggcactg
tgagaccgag actgtccctg 14280gcagccagta ttctggcaac actgttcaca cccacttact
ggatggatct tgagaaagcc 14340ccacttggtg gagcccattc ctgctgccct tttccagctg
gttatctgtc actctccttt 14400tcttcccagg tttggccgga agaatgtgct gttcgtgacc
atgggcatgc agacaggctt 14460cagcttcctg cagatcttct cgaagaattt tgagatgttt
gtcgtgctgt ttgtccttgt 14520aggcatgggc cagatctcca actatgtggc agcatttgtc
ctgggtatgg ccatcaggtt 14580ggagttgagt acttgatcct gtatttcacc atcatcccat
cacctacctt tctggagaca 14640gctgtgatgt ccctcaaggg ggacagggtt tctaacaaaa
ctagccagag cttcctggtg 14700aaccttactt acaggcaggg aaactgagcc agacatgaga
ccagcctggg gtccccagca 14760gcacaatggc ctgacttctg atttccagtt cttttctggc
ctctgggctg tggctccttg 14820gtcttagtac ttggtagtca atttactagg actcaccaga
gatcctccat ttacaaaaag 14880ggcctgccac tgcacagggc tgagccagcc ccagaaagag
ggcggcatgg ttggaggagg 14940aggggctgtg actggcaagc ttgctaaggt agagaacccc
ttgtctgcag agcactgtgg 15000ctggtgatat ctacggacaa gaataaattg ataggaaggg
gctttcgtca ccttcagggt 15060tttaattcag agtgcacact gcagggcttt gtctcaaatg
tgccagcctg ttgtcactga 15120gaagctgcca ggccggcctg tgtttggagg aacctgactc
tagctgataa ggcctttgag 15180ttccttgggt tgtattgttg aaagggttgt tttttctttt
tatatttaat attctttcct 15240tgaggcttaa gtcagcatgt gctgacttag taatgacttc
acttttaata aattcttcct 15300catgtgagga ttaaaggggg cctaccatgg catctttagc
acatggcttc agaacatggc 15360gaaattttca agagagaact gttgcttggg ggcctgagag
gccacaggga tgtaccccca 15420ggagacagtc agacaggagg ggttcagaac gccatccgct
ccctagcgcc atgaacttag 15480agagagttct cgctgttttc ttgtctgtgt attcacaaag
ataccataaa aaattaataa 15540ggaaggaacc caaattaaac tgctaactcg acctcccttg
ttttgaacag ggacagaaat 15600tcttggcaag tcagttcgta taatattctc tacgttagga
gtgtgcatat tttatgcatt 15660tggctacatg gtgctgccac tgtttgctta cttcatccga
gactggcgga tgctgctggt 15720ggcgctgacg atgccggggg tgctgtgcgt ggcactctgg
tggtgagtgt gaccctgtgc 15780cccatgtgcc cactggcagg atgatttctg tctggccttc
actagagggc agcaacaacc 15840catgaatccc tattttgtct cccagagaca ggaagcatag
attataaatt atttcagaat 15900gttttctcca cactcaaaag agccaaaaca aaacagaatc
ccatgacagc aacagacttg 15960ctctcagccc tgtgctgggt tgccccaagt gtggggaaaa
atagcagtag ctgtgagaag 16020atggggtcca gcatgccctg taggaagttc ccaagcctca
gggcaggaca gtgtaggccc 16080tagttctggc tgtgtgctgc tgaagcctca tgccacaggc
actggcacca aaagcaagag 16140tcctcagggt agccacatgg aggaagccag gctccttctg
caccaccaag gtagaggagt 16200tgaacaggca gagaagaggc cattccagac caagagggga
acactgcaga ggtgctaagg 16260tgggaatcac cccttgcagg tggagaaggt gagatcacca
gcccaagtgg agcagagagc 16320atttcagggc atagtgggag agtaagccgc acatcatggg
gcccagtcat gaccgagggt 16380ggggggcggc tacctggtcc cagcaaggtg gaaaataata
tccatagagc actcaagtgc 16440cttgataaac atgctaattt ttttcctttt tttcttttct
ttttcttttt ttttcttttt 16500tttttctcag acaaagtctc tgttgcccag gctgtagtgc
agtggcgtga tttccactta 16560ctgcaacctc cgcctcccca gttcaagcga ttctcatgtc
tcagcctccc gagtagctgg 16620gactacaggt gcctgccacc gcacttggct aatttttttt
gtatttcttt ggagagacgg 16680gtttcactgc gttggccagg ctggtctgga actcctagcc
tcaaatgatc caccggcctt 16740ggcctcctaa agtgctggga ttataggagt gaaccactgc
acctggccaa acatgctatt 16800ttaggtagag tatctgacta atctgttgga taaatcaggg
gtagggtgag gagagaagag 16860aagctaaaag gccagtgcag aagcttctgt tggtgccggg
gacagggagg agagtgtagc 16920agggcctggg ctgacataga catgcacaga agccaggctt
ccggagccca tcttgcaccc 16980atctcctcag cccagcagat ggcaacactg ctcttcagaa
atggaggtgg ccagccagca 17040tggggatgcc gtcagggggt gcagggctct cccatttttg
tgcggtgtgg ggtacacata 17100agctcatcca ccccaggtta ttgctgcgtg tggatcagct
ctttgcttct ggcttgtgat 17160caccaaacat tccacaagct ctggttctgc aaccttattc
ccacctatgg ctgtgctcta 17220cctggtctgt gggtctgctg ttggcaggga ggcctcactg
agattggacc ttgtactgcc 17280aggttcatcc ctgagtcccc ccgatggctc atctctcagg
gacgatttga agaggcagag 17340gtgatcatcc gcaaggctgc caaagccaat gggattgttg
tgccttccac tatctttgac 17400ccgagtgagg taagcaccat gtgggtgtgg gtgagaggga
cagactgacc gtgatttgag 17460agcagcagca cccagccctg aagtcctccc tgctcacagc
agcccagccc tctctctgcc 17520caagccccaa ctgcccattc cccccatccc cccactcccc
acccccacac gggccctgtt 17580aacactcaga agttgaggaa taggttacag ctgcctcact
cttttcacca cgggtttcag 17640attttcattt tttacttcct ttctaggcaa tcatatattt
taaccattac ttctaacaat 17700aaatactctt tttgagtaat aggcctttca taaagtcagc
atttgggaaa atcattgttt 17760cttataccta aggtggcttg tcaccttaca aagctaaccc
caaacgtaaa atgtaaagca 17820caaatagatt tggagttaga agtatttcat ctcttgagta
ttagcaatta ttcattaaaa 17880agaaaaaaaa agtgtttagt ctctttctgc cctccaatgg
ttaattattg catatcatct 17940tggagtcagg tcctttttga tgtccacctc ttccccctac
cccacccctc ccgtcagccc 18000tgttctcaca caccatgact catttcttgg ctctacctag
ttcctggttc ttgcttttcc 18060ttcccgttcc ctctcctacc atctctgtag caggcagttt
tccttggtct cgtgactatg 18120agaggttaga agctgtaaat gctgcctggt ggggttctgg
aatgtgtctg tggtctgact 18180ggaagatgag gggttgggtg tgggaacagc cacaagcagc
cctgctgaag tgtgagaggc 18240aggcatggtt gggcttggaa aagagggaac agttattgta
gacagcggag gccaatggcc 18300actgccagcc ctgcagactt cccagtgagt ggtggcccag
cagccactgt cagcatgcac 18360cagaaagggg tcctgtgcgc aaaggtcagg caggagtgtg
gcagagggct tttaagttag 18420gtggttttgg gggcttttaa gtgaggggtc aatctgggtg
aatgcataag ccccactggc 18480atctttgagg aaatgaggct atttcagggg atactttcag
tccaaagttg accttttgtt 18540gaacttctaa ctctggaaaa acaagctcca aacctgggtt
tgcttaagaa agcaacatca 18600gtgtgtttag acgtgtggtt tattaatggc cttggctgtg
ctgaatttca taggaagtca 18660ctctgggtga agctcaggtc aattttcctg tttttctatt
tgaattcttt ttccctggaa 18720gcacaccagt aactacatag tataaggact caaaacatta
acttttaaaa aatatcagac 18780caataaacca cacagccagg tactctctct gacccagagg
gcagggagcc aggcttcggg 18840aggaatactt agaggcctcc ttggaatgtg gccaccgaca
ggaatatgtg ggggtgcagt 18900gaggaagctg tcagcctggg cctctgtctt cctgtaccct
tgagggactg gtcacttact 18960tttcctcatt ttcattcact ctgatttgtt actgacaagg
cctagggaag ttttcacagc 19020ctaaaacaca gtcagtatac ttactgttct tagaaacgta
acactccccg acgctgagat 19080gcagacagct aagatgccag ggattcaagt atgttattgt
gtgctctgag tctctgacca 19140cctcttcttc ccatacactt atgatgttgt tcctgcagtt
acaagaccta agttccaaga 19200agcagcagtc ccacaacatt ctggatctgc ttcgaacctg
gaatatccgg atggtcacca 19260tcatgtccat aatgctgtgg tatgtaaaag agacctgcct
gaggcttcca gacaaagctt 19320cttgaagtgg ccattgggcc tcttgtttac agacatgcct
cagacaaaat tcaaagccta 19380tgtcatcaga gagtgaaaag gatatgtctt gtgttagatg
gaaaaaatgg gcatgtcaca 19440attcttaatg ggatggaacc tcagaaaagg agaatgaaaa
caattgtgga ggctgttgtg 19500ggaaatatgg actctcgtgg ggaatctctc cagatcttaa
gatgaatcct tgcccaattt 19560gggtcattta gttcccgtct cctacccagt taccgacagt
ggctgaggag gccaggtagg 19620gcttttaaga aggatctgag tgaagacacc atgtcctgta
ggctgcagag gctgccagtt 19680actttctgga aatgtggaag tgggatgtgc tcctcctggg
atgtccataa acggtcctgg 19740agtcagggct atagcctaga tgtccttacc aggttcccac
taatgaggca aagtatgtca 19800gaaagggatt tgtgaattac cagggagagg aaacatgtcc
aagtgcacat cgctagcttt 19860tgctcagcgg ccgaaccctg ggattctagg cgacttctgg
agcctggtgg gttagcggtg 19920agaagatggg cgaggagggc ggacttcatc tcagagtcct
tattactagt ctcatccagc 19980tttgaggcag tcagccactg tgcctactga gggagtgcta
tgagtcaccc gcttccaagg 20040aatggcccag gatccctcca ggcagttcac cattccctga
gttggcctca agacaggagc 20100agcatgtagc ctgcaccaca gacatgcaag cctgtgatga
gtcacccact tttgtgttca 20160cccaggctct cctccctgct ctggatttcc tggggactca
tgcacatact ctttttattg 20220taccagctgt gtgttccacc tgcagatgag tcaaaacagt
ctaatccata aaggtctggt 20280ttgtcaaaga gtgtgggtca tcaacagaga gaatgcctac
tggggatgcc caggtcaggg 20340gtactgcagg gcatcctgat gagaggcagt gtggcccctc
cattgggagc cacctctctg 20400ctccacaagt accgcggggc tggtgtcagc tgtctctgac
cagcctcttc ctgactggtc 20460accacaggta gtgtgtgagg gtctctctcc aagtgtttga
cctaatgttg ttccttttgt 20520tatcttatcc cccaaatcct atcacacctc acttgatgtc
tgcctcctga ctcattctct 20580agctccttct gcagttgctg gatttgagga ggttcagctt
aggattttta aagctgaaag 20640gcaggttgga atttttcttt tcaatgaagt aaatctatct
gaattataca agcttttttg 20700ctgggacact gtctatatgg aaggctctga gagcgcactg
gcgcagggtt tacactgtac 20760cacttgggct ggggaaaatt atcttttgat ctatgaagta
agacgcaggg ttacagttac 20820tgctgcctta ctagtctctg cttaaagatg gtttggaatt
tactgaaata attgcattgt 20880aaaagttgta caggttggga aagatgtgga tactgctttt
ccagctttct tctgcactct 20940gtttcaggat gaccatatca gtgggctatt ttgggctttc
gcttgatact cctaacttgc 21000atggggacat ctttgtgaac tgcttccttt cagcgatggt
tgaagtccca gcatatgtgt 21060tggcctggct gctgctgcaa tatttgcccc ggcgctattc
catggccact gccctcttcc 21120tgggtggcag tgtccttctc ttcatgcagc tggtaccccc
aggtagggac catgtgcatc 21180tatggtttgg ggtcttcact gagtctctta ctgtctacca
ggctgtctca attaataaag 21240agaataaaat caagcccatc acagctccct tgcttatata
cattcttggc ctaaaaatca 21300atagaaagtg tcttctgaga ctagaacact tatggcctgg
gctttgaggg agtgggaaaa 21360agcagccatt ggggctgttg gttaatttta ctctgtaccc
aagttaatgt gctcatactg 21420ttttccactg cagaagaaga gggaagaaat agctatccca
ttcctttttt tcctggccct 21480gtcttcttta tttattcaac aaatagctat ggcatgccta
ccatgggctg ggcactgtgc 21540tcggtgataa cgatacacaa gaaaacaagc caggcagaac
cccaggccct catggaacgt 21600acaccctaga tgagaagaca gacaacaaac aagtaaataa
aatgcttaat atagttcaga 21660ctgtgttacc ttctaggaat acaaatgaag gacaatgccg
agttagttta catagtcaca 21720gatagtgtcc ctgaacaggg ggcagttcag tagaaatgta
cataaagtga cagaaagccc 21780tgaaaaagtc taggagaaca ttttaggaag aagaaatggc
aaaggcagcg accctgagca 21840ggggatgagc ctggcatgtt tgaggaggag ggagaagggg
aggggccaga ccactgagag 21900ggcctcacag acccttagca ggattttatt tctgaaacta
tcttagtatc ccacagatgg 21960gtgggaggta gccatttcca ataatttata gaacagttca
tgggccctca tctctccctc 22020tccatcactg tgcccagaga cttcagtgta cctgtagatt
tgggagcctc tgatggtcac 22080ttttgggccc atcaggctga gaacactgca cgggaacagc
tccccatggg atgtggcagg 22140aggagcccag aactgatgta gaggctcaca gctgagctca
gagtgacctt caggtcacac 22200atagctctcc catcagcaca gcacagagag attagaagat
caactcgaga ttctgatggc 22260ctatgatttt tttgaggtct gagtgggagg aaagcatgaa
atgagttaga actgaattct 22320ccattcatct aaacatcatg agttaattcc atagtgcctg
cagtgtgagg ttctggggtg 22380acagttaatc cctgacagac atgtctttaa tgacttatag
actgggaagc aggttgattg 22440gactattaag gagcttactc tggtggtctc caggttgagg
aaagtgcatg tccttatagc 22500tgcaggtccc agcctccttt cagcaatcaa tttggaggga
aatcttggct atagcccctt 22560cccccacaat aggaagtgat agaaactgac tccccaaaaa
atttgggaag aaagtatgtt 22620tgttttgctc tcaatagctg catgccatgg gttggtacct
actcctaccc tctttccttt 22680gcttctccag acttgtatta tttggctaca gtcctggtga
tggtgggcaa gtttggagtc 22740acggctgcct tttccatggt ctacgtgtac acagccgagc
tgtatcccac agtggtgaga 22800aacatgggtg tgggagtcag ctccacagca tcccgcctgg
gcagcatcct gtctccctac 22860ttcgtttacc ttggtaagtc ccatgagcca agggcacact
agagcaacgg gatggaagta 22920ctaactggct tgaatgtgag ctggaggttg cgtgttaaca
ggaaaacaag ttcatacagt 22980acatgggctc catccagtac tggatctttg gccgggaagg
gttcttgtcc cagtgcactg 23040gccctcactt tcaaatggaa aacaacctat agattaccta
gaaattgatg agaatattag 23100agggtttgtt tctgttttag ccatcccagg ccttccatca
gagactacaa ttcctttatc 23160ctaagaacct acagagtggt ttagggagcc agtgtgctta
gttggagaaa tttcttggaa 23220tcagagttta aaaggaacat gaggggaaag atgtccatgc
aagaggtctg atgaacgtaa 23280aattattata acctagagca ctatagagtg attttatctt
gtgtgaagat ccaccccatg 23340ccattttatg tagcaggtct ccagttttct cttctcagaa
ttatgtcttc atagcacctg 23400tggtttccct gcacatccct agccagtacc tctttaggga
gggtggcacc cacctgagag 23460tactcagagt gctttgtgaa catgctatgt agatctcaaa
gcaagcaaaa gcaccctgcc 23520taatctgaag gcagatcaca tgggctggga cacatctgca
gaggtggaag agttatttcc 23580atccctggac aagtacctca ggttccttgg aaacccaacc
ttggtaaata agaataatca 23640gcatggccca gaaataggaa taatcagcat ggcccagctc
ttctcctgca accgcccctt 23700tgtactcctc ccctgcatgg tggaacactg ctgggctctg
ggcatgcctg tgccagctct 23760gggttctgaa acctgtctag atgccagatt ctaatctgac
tgctcagact gtgagagatg 23820tgagaccaag aaggaaagtg atccccttcc agagtcctgg
gagcataaag gggtagatga 23880gagaccaagt ctaactgcag ccctgggcct gaggctccgt
ctgctttgcc ataggtgcct 23940acgaccgctt cctgccctac attctcatgg gaagtctgac
catcctgaca gccatcctca 24000ccttgtttct cccagagagc ttcggtaccc cactcccaga
caccattgac cagatgctaa 24060gagtcaaagg gtaagaagac ctcctctgtc agtgttgatg
cactgggtct gggtctggcc 24120aggtctcagg agcccctcac aatagagcta ctcgcaaact
ccctctcaca gacaccatgg 24180actagtttag ccattaaagg gttgtaaatg gcaaggtgct
tacttatagc ccatcctctc 24240tggtctgttc ctgtgtggac atgtcactat acacatctcc
atggcagtag ccgcactgga 24300taactcagag gctagaagaa acctttcaga atctgctgca
ggattctctt cccagggaag 24360atatcctcag ttcttgtttg tttggagact gggaggcatc
tttttaaaat gtgttactga 24420catatttttg cttgttttta tagaatgaaa cacagaaaaa
ctccaagtca cacaaggatg 24480ttaaaagatg gtcaagaaag gcccacaatc cttaaaagca
cagccttcta acatcgcttc 24540cagtaaggga gaaactgaag aggaaagact gtcttgccag
aaatggccag cttgtgcaga 24600ctccgagtcc ttcagtgaca aaggcctttg ctgtttgtcc
tcttgacctg tgtctgactt 24660gctcctggat gggcacccac actcagaggc tacatatggc
cctagagcac caccttcctc 24720tagggacact ggggctacct acagacaact tcatctaagt
cctaactatt acaatgatgg 24780actcagcacc tccaaagcag ttaatttttc actagaacca
gtgagatctg gaggaatgtg 24840agaagcatat gctaaatgta cattttaatt ttagactact
tgaaaaggcc cctaataagg 24900ctagaggtct aagtccccca cccctttccc cactcccctc
tagtggtgaa ctttagagga 24960aaaggaagta attgcacaag gagtttgatt cttacctttt
ctcagttaca gaggacatta 25020actggatcat tgcttcccca gggcaggaga gcgcagagct
agggaaagtg aaaggtaatg 25080aagatggagc agaatgagca gatgcagatc accagcaaag
tgcactgatg tgtgagctct 25140taagaccact cagcatgacg actgagtaga cttgtttaca
tctgatcaaa gcactgggct 25200tgtccaggct cataataaat gctccattga atctactatt
cttgttttcc actgctgtgg 25260aaacctcctt gctactatag cgtcttatgt atggtttaaa
ggaaatttat caggtgagag 25320agatgagcaa cgttgtcttt tctctcaaag ctgtaatgtg
ggttttgttt tactgtttat 25380ttgtttgttg ttgtatcctt ttctccttgt tatttgccct
tcagaatgca cttgggaaag 25440gctggttcct tagcctcctg gtttgtgtct tttttttttt
ttttttaaac acagaatcac 25500tctggcaatt gtctgcagct gccactggtg caaggcctta
ccagccctag cctctagcac 25560ttctctaagt gccaaaaaca gtgtcattgt gtgtgttcct
ttcttgatac ttagtcatgg 25620gaggatatta caaaaaagaa atttaaattg tgttcatagt
ctttcagagt agctcacttt 25680agtcctgtaa ctttattggg tgatattttg tgttcagtgt
aattgtcttc tctttgctga 25740ttatgttacc atggtactcc taaagcatat gcctcacctg
gttaaaaaag aacaaacatg 25800tttttgtgaa agctactgaa gtgccttggg aaatgagaaa
gttttaataa gtaaaatgat 25860tttttaaata t
25871624DNAArtificial SequenceDescription of
Artificial SequenceArtificially Synthesized Primer Sequence
6gcaggaccaa ggcggcggtg tcag
24724DNAArtificial SequenceDescription of Artificial SequenceArtificially
Synthesized Primer Sequence 7agactagagg aaaaacggga tagc
24824DNAArtificial SequenceDescription of
Artificial SequenceArtificially Synthesized Primer Sequence
8agatttttag gagcaagcgt taga
24924DNAArtificial SequenceDescription of Artificial SequenceArtificially
Synthesized Primer Sequence 9gaggcagaca ccgtggcact acta
241024DNAArtificial SequenceDescription of
Artificial SequenceArtificially Synthesized Primer Sequence
10ttcacaccca cttactggat ggat
241124DNAArtificial SequenceDescription of Artificial
SequenceArtificially Synthesized Primer Sequence 11attctgtttt
gttttggctc tttt
241224DNAArtificial SequenceDescription of Artificial
SequenceArtificially Synthesized Primer Sequence 12agcagggcct
gggctgacat agac
241324DNAArtificial SequenceDescription of Artificial
SequenceArtificially Synthesized Primer Sequence 13aaaggacctg
actccaagat gata
241424DNAArtificial SequenceDescription of Artificial
SequenceArtificially Synthesized Primer Sequence 14tctgaccacc
tcttcttccc atac
241524DNAArtificial SequenceDescription of Artificial
SequenceArtificially Synthesized Primer Sequence 15gcctcctcag
ccactgtcgg taac
241624DNAArtificial SequenceDescription of Artificial
SequenceArtificially Synthesized Primer Sequence 16atgttgttcc
ttttgttatc ttat
241724DNAArtificial SequenceDescription of Artificial
SequenceArtificially Synthesized Primer Sequence 17cttgttttct
tgtgtatcgt tatc
241824DNAArtificial SequenceDescription of Artificial
SequenceArtificially Synthesized Primer Sequence 18tatgtttgtt
ttgctctcaa tagc
241924DNAArtificial SequenceDescription of Artificial
SequenceArtificially Synthesized Primer Sequence 19tctgtgagag
ggagtttgcg agta
242024DNAArtificial SequenceDescription of Artificial
SequenceArtificially Synthesized Primer Sequence 20tacgaccgct
tcctgcccta catt
242124DNAArtificial SequenceDescription of Artificial
SequenceArtificially Synthesized Primer Sequence 21tcattctgct
ccatcttcat tacc
242233DNAArtificial SequenceDescription of Artificial
SequenceArtificially Synthesized Primer Sequence 22gataagctta
cggtgtcccc ttattcccat acg
332324DNAArtificial SequenceDescription of Artificial
SequenceArtificially Synthesized Primer Sequence 23cccatgccaa
caaggacaaa aagc
242424DNAArtificial SequenceDescription of Artificial
SequenceArtificially Synthesized Primer Sequence 24tgtttttcgt
gggtgtgctg atgg
242524DNAArtificial SequenceDescription of Artificial
SequenceArtificially Synthesized Primer Sequence 25acagaacaga
aaagccctca gtca 242610PRTHomo
sapiens 26Glu Gln Lys Leu Ile Ser Glu Glu Asp Leu1 5
102717DNAMus musculus 27tcctgtggct gaccata
172817DNAMus musculus 28tcctgtggcg gaccata
172922DNAHomo sapiens
29agggcggcat gccgggacta cg
223019DNAHomo sapiens 30cttgcagtga aacctggtg
193119DNAHomo sapiens 31tttgccataa gtgcctacg
19
User Contributions:
comments("1"); ?> comment_form("1"); ?>Inventors list |
Agents list |
Assignees list |
List by place |
Classification tree browser |
Top 100 Inventors |
Top 100 Agents |
Top 100 Assignees |
Usenet FAQ Index |
Documents |
Other FAQs |
User Contributions:
Comment about this patent or add new information about this topic: