Patent application title: Sodium-independent transporter carrying acidic amino acid and its gene
Inventors:
Hitoshi Endou (Kanagawa, JP)
Yoshikatsu Kanai (Tokyo, JP)
Assignees:
J-Pharma Co., Ltd.
IPC8 Class: AC07K1600FI
USPC Class:
5303879
Class name: Globulins immunoglobulin, antibody, or fragment thereof, other than immunoglobulin antibody, or fragment thereof that is conjugated or adsorbed binds specifically-identified amino acid sequence
Publication date: 2010-08-26
Patent application number: 20100216976
Claims:
1-17. (canceled)
18. An antibody directed to a protein selected from the group consisting of the following (A) and (B):(A) a protein consisting of the amino acid sequence represented by SEQ ID NO: 1; and(B) a protein having the ability to transport acidic amino acids and their analogues in a sodium-independent manner which consists of an amino acid sequence where one or several amino acids are deleted, substituted or added in the amino acid sequence represented by SEQ ID NO: 1.
19-28. (canceled)
29. The antibody of claim 18, wherein the protein is derived from mouse.
30. The antibody of claim 18, wherein the protein is derived from organ, tissue or cultured cell.
31. An antibody directed to a protein selected from the group consisting of the following (C) and (D):(C) a fusion protein of a protein consisting of the amino acid sequence represented by SEQ ID NO: 1 and a protein consisting of the amino acid sequence represented by SEQ ID NO: 3; and,(D) a protein having the ability to transport acidic amino acids and their analogues in a sodium-independent manner which consists of an amino acid sequence where one or several amino acids are deleted, substituted or added in the amino acid sequence of a fusion protein of a protein consisting of the amino acid sequence represented by SEQ ID NO: 1 and a protein consisting of the amino acid sequence represented by SEQ ID NO: 3.
32. The antibody of claim 31, wherein the protein is derived from mouse.
33. The antibody of claim 31, wherein the protein is derived from organ, tissue or cultured cell.
34. An antibody directed to a protein selected from the group consisting of the following (E) and (F):(E) a fusion protein of a protein consisting of the amino acid sequence represented by SEQ ID NO: 1 and a protein consisting of the amino acid sequence represented by SEQ ID NO: 5; and,(F) a protein having the ability to transport acidic amino acids and their analogues in a sodium-independent manner which consists of an amino acid sequence where one or several amino acids are deleted, substituted or added and in the amino acid sequence of a fusion protein of a protein consisting of the amino acid sequence represented by SEQ ID NO: 1 and a protein consisting of the amino acid sequence represented by SEQ ID NO: 5.
35. The antibody of claim 34, wherein the protein is derived from mouse.
36. The antibody of claim 34, wherein the protein is derived from organ, tissue or cultured cell.
Description:
TECHNICAL FIELD
[0001]The present invention relates to a protein associated with the sodium-independent transport of acidic amino acids and its analogue, fusion protein thereof, as well as a gene encoding said protein. The present invention also relates to a method for controlling the cell proliferation or for altering the in vivo pharmacokinetics of a pharmaceutical, toxic substance or xenobiotics by modulating an ability to transport acidic amino acids and its analogue possessed by a protein associated with the sodium-independent transport of acidic amino acids and its analogue, by means of employing said protein, its fusion protein, its specific antibody, or its function-promoting substance or function-suppressing substance, as well as an agent for controlling an ability to transport acidic amino acids and its analogue comprising said substances.
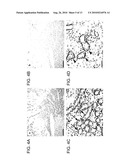
BACKGROUND ART
[0002]A cell always requires the uptake of an amino acid as a nutrition, and such a function is exerted by an amino acid transporter which is a membrane protein existing in a cell membrane. The amino acid transporter is distributed in a specific site in each tissue in a multicellular organism and plays an important role in expressing the specific function of each tissue. For example, in kidney cells and small intestine, it plays a role for epithelial absorption of amino acid in lumen and, in nerve tissues, it is in charge of recovery of amino acid as a neurotransmitter released as a result of neurotransmission and also of supply of amino acid as a precursor for neurotransmitter to nerve cells. Further, it exists in blood-brain barrier and placental barrier and makes permeation of the amino acid possible.
[0003]With regard to an amino acid transport mechanism, its identification and classification have been conducted using cultured cells and membrane specimens since 1960's and, reflecting the multiplicity of amino acid molecules, many transport systems have been described. However, there has been no independent transport system for each amino acid but most of the amino acid transports have been conducted by a few kinds of transport systems which transport several amino acids having similar side chains (Christensen, Physiol. Rev., volume 70, page 43, 1990).
[0004]Transport of acidic amino acids such as glutamic acid and aspartic acid having carboxyl group on a side chain has been believed to be carried out by both of a sodium-dependent transporter which requires sodium ion for its function and a sodium-independent transporter which does not require sodium ion for its function.
[0005]However, in a conventional method, it is difficult to analyze the details of the transport of an amino acid or its analogue via the acidic amino acid transport system and the in vivo functional roles, and it has been desired to enable a detailed functional analysis by isolating a gene of acidic amino acid transporter responsible for the function of the acidic amino acid transport system.
[0006]With regard to sodium-dependent acidic amino acid transporters, five kinds of glutamate transporters--EAAC1, GLT-1, GLAST, EAAT4 and EAAT5--have been cloned (Kanai, Curr. Opin. Cell Biol., volume 9, page 565, 1997; Kanai and Endou, Curr. Drug Metab., volume 2, page 339, 2001).
[0007]With regard to sodium-independent transporters, LAT1 (Kanai, et al., J. Biol. Chem., volume 273, pages 23629-23632, 1998) and LAT 2 (Segawa, et al., J. Biol. Chem., volume 274, pages 19745-19751, 1999) have been cloned as neutral amino acid transporters corresponding to a transport system L. It was also shown that LAT1 and LAT2 function only when they coexist with a cofactor 4F2hc which is a single membrane-spanning type protein. LAT1 shows an exchange transport activity which transports large-sized neutral amino acids such as leucine, isoleucine, valine, phenylalanine, tyrosine, tryptophan, methionine and histidine while LAT2 shows a broad substrate selectivity transporting small-sized neutral amino acids such as glycine, alanine, serine, cysteine and threonine in addition to large-sized neutral amino acids and they are not acidic amino transporters.
[0008]With regard to proteins analogous to LAT1 and LAT2, the above-mentioned y+LAT1 and y+LAT2 having the functions of a transport system y+L which transports neutral amino acids and basic amino acids have been cloned (Torrents, et al., J. Biol. Chem., volume 273, pages 32437-32445, 1998). It was also revealed that both of y+LAT1 and y+LAT2 function only when being coexisting with a cofactor 4F2hc. y+LAT1 and y+LAT2 mainly transport glutamine, leucine and isoleucine as neutral amino acids and do not transport acidic amino acids.
[0009]With regard to a transporter which requires a cofactor 4F2hc for expressing its function, Asc-1 which is a protein analogous to LAT1 to LAT2 was cloned (Fukasawa, et al., J. Biol. Chem., 275: 9690-9698, 2000). Asc-1 selectively transports alanine, serine, cysteine, threonine, glycine, etc., shows a substrate selectivity of amino acid transport system asc and does not transport acidic amino acids.
[0010]With regard to a transporter which requires another cofactor rBAT having an analogous structure to 4F2hc for expressing its function, BAT1 which is a protein analogous to LAT1 and LAT2 was cloned (Chairoungdua, et al., J. Biol. Chem., 274: 28845-28848, 1999). BAT1 transports cystine, neutral amino acids and basic amino acids and does not transport acidic amino acids.
[0011]As described above, molecular entity of a transporter which functions by binding to 4F2hc and rBAT was characterized and, the presence of a group of transporters which achieves a transport ability by forming heterodimer with a single membrane-spanning type protein and a heterodimeric amino acid transporter family was established.
[0012]Further, with regard to a transporter requiring a cofactor 4F2hc for expressing its function, xCT which is a protein analogous to LAT1 and LAT2 was cloned (Sato, et al., J. Biol. Chem., 274; 11455-11458, 1999). xCT transports cystine, glutamic acid and sodium aminoadipate in a sodium-independent manner and corresponds to an amino acid transport system Xc. xCT needs a negative charge of side chain of amino acid for recognition of substrate and is classified under sodium-independent acidic amino acid transporters (Kanai and Endou, Curr. Drug Metab., volume 2, page 339, 2001).
[0013]xCT transports glutamic acid but does not transport aspartic acid and its transport is suppressed by cystine. In addition, xCT is a transporter where expression is induced by oxidative stress and, except a few cases, its expression in common normal tissues is not detected. However, it has been reported that there is a sodium-independent glutamic acid and aspartic acid transporter which is not suppressed by cystine (Christensen, Physiol. Rev., volume 70, page 43, 1990) and it has been suggested that there is a sodium-independent acidic amino acid transporter other than xCT which has not been identified.
[0014]Further, Asc-2 which is a protein having an analogous structure to LAT1 and LAT2 and binds to unidentified protein other than rBAT or 4F2hc was cloned (Chairoungdua, et al., J. Biol. Chem., 276: 49390-49399, 2001). Asc-2 is not expressed in a cell membrane by itself, however, by preparing a fusion protein with 4F2hc or rBAT, it transfers to a cell membrane as a fusion protein and a transport activity can be detected. When Asc-2 is expressed in a cell membrane as a fusion protein with 4F2hc or rBAT, it shows a characteristic of a sodium-independent neutral amino acid transport system asc.
DISCLOSURE OF THE INVENTION
[0015]An object of the present invention is to provide a gene of a transporter which transports acidic amino acids such as glutamic acid and aspartic acid in a sodium-independent manner and also to provide a sodium-independent acidic amino acid transporter which is a polypeptide encoded by the gene.
[0016]Other objects will be apparent from the following description.
[0017]The present inventors have searched the EST (expressed sequence tag) database using a base sequence of translation region of cDNA of BAT1 and identified a base sequence analogous to BAT1. A base sequence of cDNA clone corresponding to the sequence has been decided and clarified that it encodes a novel protein. Further, a fusion protein of the translated product of the gene with 4F2hc or rBAT has been prepared and expressed in a cell membrane of oocyte of Xenopus. As a result, it has been clarified that the function of the translated product of the gene is a sodium-independent transporter which transports acidic amino acids such as glutamic acid and aspartic acid whereby the present invention has been achieved.
[0018]Thus, the present invention relates to a protein selected from the following (A) or (B).
[0019](A) protein comprising an amino acid sequence represented by SEQ ID NO: 1.
[0020](B) protein comprising an amino acid sequence where one or several amino acid(s) is/are deleted, substituted or added in the amino acid sequence represented by SEQ ID NO: 1 and having an ability of transport of acidic amino acids or its analogue in a sodium-independent manner.
[0021]The present invention also relates to a gene comprising DNA selected from the following (a) and (b).
[0022](a) DNA comprising a base sequence represented by SEQ ID NO: 2.
[0023](b) DNA hybridizing with DNA comprising the base sequence represented by SEQ ID NO: 2 under a stringent condition and encodes a protein having an ability of transport of acidic amino acids or its analogue in a sodium-independent manner.
[0024]The novel protein of the present invention having an ability of transport of acidic amino acids and its analogue in a sodium-independent manner or, in other words, an amino acid transporter AGT1 (aspartate/glutamate transporter 1) is expressed in a cell membrane and has an ability of transport (uptake) of acidic amino acids such as glutamic acid and aspartic acid in a highly affinitive manner by preparing a fusion protein with 4F2hc or rBAT.
[0025]Incidentally, the sodium-independent transporter AGT1 of the present invention which transports acidic amino acids is mainly expressed in the kidney in vivo.
BRIEF DESCRIPTION OF THE DRAWINGS
[0026]FIG. 1 shows the amino acid sequences of mouse AGT1, mouse Asc-2, rat LAT1, rat y+LAT1, mouse xCT and rat BAT1 for comparison with each other. The assumed membrane-spanning sites are shown by lines. A conserved cystine residue is shown by *, assumed cAMP-dependent phosphorylation site is shown by #, an assumed C-kinase-dependent phosphorylation site is shown by + and an assumed tyrosine phosphorylation site is shown by &.
[0027]FIG. 2 is a photographic picture as a substitute for drawing which shows the result of analysis of expression of AGT1 gene mRNA in various organ tissues of mouse by means of a northern blotting.
[0028]FIG. 3 is a photograph as a substitute for drawing which shows the result of the western blotting analysis by an anti-AGT1 antibody. It was carried out under a non-reducing condition (-) and a reducing condition (+) in a mouse kidney membrane specimen.
[0029]FIG. 4 is a photograph as a substitute for drawing which shows the result of immunohistological analysis of AGT1 by an anti-AGT1 antibody in a mouse kidney. a: slightly magnified image. Stainings are observed in proximal tubule of outer layer of medulla and in distal tubule of cortex. b: absorption experiment by antigen peptide. Stainings observed in "a" disappeared and specificity of staining was shown. c and d: highly magnified images of proximal tubule (c) and distal tubule (d). Stainings are observed in the side of basolateral membrane.
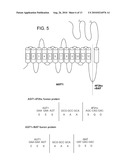
[0030]FIG. 5 is a schematic drawing of a fusion protein prepared by connecting AGT1 with 4F2hc or rBAT. Amino acid sequences and gene base sequences of the connection parts of AGT1-4F2hc fusion protein and AGT1-rBAT fusion protein are shown below in FIG. 5.
[0031]FIG. 6 shows the result of experiment of the uptake of aspartic acid by an oocyte into which mouse 4F2hc gene cRNA, AGT1 gene cRNA, AGT1 gene cRNA/mouse 4F2hc gene cRNA, AGT1-4F2hc fusion protein gene cRNA or AGT1-rBAT fusion protein gene cRNA is injected.
[0032]FIG. 7 shows the result of experiment of the uptake of aspartic acid by COS-7 cells into which mouse 4F2hc gene, mouse rBAT gene, AGT1 gene, AGT1 gene/mouse 4F2hc gene or AGT1 gene/mouse rBAT gene is injected.
[0033]FIG. 8 is a drawing which shows the result of investigating the expression of a fusion protein of AGT1 with 4F2hc (AGT1-4F2hc) in an oocyte cell membrane by a immunofluorescence analysis. As controls, investigations by a immunofluorescence analysis were carried out using an anti-4F2hc antibody (a, c and e) or an anti-AGT1 antibody in the oocyte into which water was injected (a and b), the oocyte into which AGT1 gene cRNA was injected and expressed (c and d) and the oocyte into which a fusion protein (AGT1-4F2hc) gene cRNA of AGT1 with 4F2hc was injected and expressed (e and f).
[0034]FIG. 9 shows the result of investigating the influence of added salt in an experiment of the uptake of aspartic acid by an oocyte into which a fusion protein (AGT1-4F2hc) gene cRNA of AGT1 with 4F2hc was injected.
[0035]FIG. 10 shows the result of investigating the influence of the concentration of substrate aspartic acid in an experiment of the uptake of aspartic acid by an oocyte into which a fusion protein (AGT1-4F2hc) gene cRNA of AGT1 with 4F2hc was injected.
[0036]FIG. 11 shows the result of investigating the influence of addition of various amino acids and analogous compounds on a system in an experiment of the uptake of aspartic acid by an oocyte into which a fusion protein (AGT1-4F2hc) gene cRNA of AGT1 with 4F2hc was injected.
[0037]FIG. 12 shows the result of investigating the influence of addition of various acidic amino acids and analogous compounds on a system in an experiment of the uptake of aspartic acid by an oocyte into which a fusion protein (AGT1-4F2hc) gene cRNA of AGT1 with 4F2hc was injected.
[0038]PDC: L-trans-pyrrolidine-2,4-dicarboxylate; DHK: dihydrokainate; **: the case where p<0.01 in Student's t-test to the data which does not constitute a pair.
[0039]FIG. 13 shows the result of investigating the uptake of radio-labeled amino acid by an oocyte into which a fusion protein (AGT1-4F2hc) gene cRNA of AGT1 with 4F2hc was injected.
BEST MODE FOR CARRYING OUT THE INVENTION
[0040]SEQ ID NO: 1 in the Sequence Listing which will be shown later represents an amino acid sequence (478 amino acids) of a sodium-independent transporter (mouse AGT1) derived from mouse transporting acidic amino acids and SEQ ID NO: 2 represents an amino acid sequence (478 amino acids) of protein encoded in a full-length cDNA base sequence (about 2.1 kbp) of the gene and a translation region thereof.
[0041]When the amino acid sequence represented by SEQ ID NO: 1 or the base sequence represented by SEQ ID NO: 2 which will be mentioned later was subjected to a homology search for all sequences included in the known protein databases (NBRF and SWISS-PROT) and DNA databases (GenBank and EMBL), no sequence was identical, whereby the sequence is believed to be novel.
[0042]With regard to the protein of the present invention, there may be exemplified, in addition to the one having an amino acid sequence represented by SEQ ID NO: 1, a protein having the amino acid sequence where one or several amino acid(s) is/are deleted, substituted or added in the amino acid sequence represented by SEQ ID NO: 1. Deletion, substitution or addition of amino acid(s) may be within such an extent that transport activity of a neutral amino acid is not lost and, usually, it is from 1 to about 96 or, preferably, from 1 to about 48. Such a protein usually has a homology to the amino acid sequence represented by SEQ ID NO: 1 to an extent of 1 to 80%, preferably, 1 to 90%.
[0043]With regard to the gene of the present invention, in addition to the one having a base sequence represented by SEQ ID NO: 2, it includes a gene containing DNA which can be hybridized with DNA having a base sequence represented by SEQ ID NO: 2 under a stringent condition. With regard to the DNA which can be hybridized, any substance will do so far as the protein encoded by the DNA has an ability of transporting a neutral amino acid. Such a DNA usually has a homology of base sequence of more than 70%, preferably more than 80% with the base sequence represented by SEQ ID NO: 2. Such a DNA includes a variant gene found in nature, an artificially modified variant gene, a homologous gene derived from other organism, and the like.
[0044]In the present invention, a hybridization under a stringent condition is usually carried out in such a manner that a hybridization is carried out for about 12 hours under the temperature of 37 to 42° C. in a hybridization solution of 5×SSC or having the identical salt concentration therewith, a preliminary washing is carried out upon necessity using a solution of 5×SSC or having the identical salt concentration therewith and then washing is carried out in a solution of 1×SSC or having the identical salt concentration therewith.
[0045]The sodium-independent transporter gene which transports acidic amino acids according to the present invention can be isolated and obtained by carrying out a screening using a tissue or a cell of appropriate mammals as a gene source. Examples of the mammals are non-human animals such as dog, cattle, horse, goat, sheep, monkey, pig, rabbit, rat and mouse, and in addition to those, human beings.
[0046]Screening and isolation of gene is able to be advantageously carried out by, for example, a homology cloning method.
[0047]For example, mouse or human kidney is used as a gene source and mRNA (poly(A)+RNA) is prepared therefrom. Then a cDNA library is constructed therefrom and cDNA is screened using a probe corresponding to a sequence analogous to BAT1 (such as GenBank®/EBI/DDBJ accession No AI314100) obtained by searching the EST (expressed sequence tag) database whereby a clone containing cDNA of Asc-2 gene can be obtained.
[0048]With regard to the obtained cDNA, the base sequence is determined by a conventional method and translation region is analyzed, whereby an amino acid sequence of the protein encoded thereby, i.e. AGT1, can be determined.
[0049]The fact that the resulting cDNA is a sodium-independent transporter which transports acidic amino acids, in other words, the gene product encoded with cDNA is a sodium-independent transporter which transports acidic amino acids can be tested, for example, by the following method. Thus, cDNA encoding a fusion protein of AGT1 with 4F2hc or rBAT is prepared using the resulting cDNA of AGT1 gene, then RNA (cRNA) which is prepared from the cDNA and complementary thereto is introduced into the oocyte to be expressed and an ability of transport (uptake) of acidic amino acids into the cells can be confirmed by measuring the uptake of the substrate into cells by a common uptake test using an appropriate acidic amino acid as a substrate (Kanai and Hediger, Nature, volume 360, pages 467-471, 1992).
[0050]AGT1 protein is synthesized by an in vitro translation method (Hediger, et al., Biochim. Biophys. Acta, volume 1064, page 360, 1991) using RNA (cRNA) prepared from the resulting cDNA of AGT1 gene and being complementary thereto and the size of the protein or the presence of sugar, etc. can be investigated by means of electrophoresis.
[0051]Since cDNA of 4F2hc gene has been reported already (Fukasawa, et al., J. Biol. Chem., 275: 9690-9698, 2000), it is possible to easily prepare a gene of 4F2hc from the sequence information by a PCR or the like.
[0052]Since cDNA of rBAT gene has been also reported already (Segawa, H., et al., Biochem. J, 328: 657-664, 2000), it is possible to easily prepare a gene of rBAT from the sequence information by a PCR or the like.
[0053]cDNA encoding a fusion protein of AGT1 with 4F2hc or rBAT is easily prepared by a PCR or the like from cDNA of AGT1 gene, cDNA of 4F2hc gene or cDNA of rBAT gene.
[0054]The characteristic of AGT1 such as substrate selectivity of AGT1 can be investigated by applying the similar uptake experiment to the expressed cells.
[0055]Homologous gene, chromosome gene, etc. derived from different tissues and different organisms can be isolated by screening an appropriate cDNA library or genomic DNA library prepared from different gene sources using the resulting cDNA of AGT1 gene.
[0056]It is also possible to isolate a gene from a cDNA library or a genomic DNA library by a conventional PCR (polymerase chain reaction) method using a synthetic primer designed on the basis of information of the disclosed base sequence of the gene of the present invention (the base sequence represented by SEQ ID NO: 2 or a part thereof).
[0057]DNA library such as a cDNA library or a genomic DNA library may be prepared by a method mentioned, for example, in "Molecular Cloning" by Sambrook, J., Fritsh, E. F. and Manitis, T. (Cold Spring Harbor Press, 1989). When there is a commercially available library, it can be used as well.
[0058]The sodium-independent transporter which transports acidic amino acids according to the present invention and the gene (AGT1) thereof may be produced by, for example, a gene recombination technique using cDNA encoding therefor. For example, DNA (such as cDNA) encoding AGT1 is incorporated into an appropriate expression vector and the resulting recombinant DNA can be introduced into an appropriate host cell. With regard to an expression system (host-vector system) for the production of polypeptide, there may be exemplified expression systems of bacteria, yeasts, insect cells and mammalian cells. Among those, it is preferred to use insect cells and mammalian cells for the preparation of functional proteins.
[0059]A fusion protein of the sodium-independent transporter which transports acidic amino acids according to the present invention with 4F2hc or rBAT or a gene thereof (AGT1-4F2hc or AGT1-rBAT) may be produced, for example, by a gene recombination technique using cDNA encoding it. For example, DNA (such as cDNA) encoding AGT1-4F2hc or AGT1-rBAT is incorporated into an appropriate expression vector and the resulting recombinant DNA can be introduced into an appropriate host cell. With regard to an expression system (host-vector system) for the production of polypeptide, there may be exemplified expression systems of bacteria, yeasts, insect cells and mammalian cells. Among those, it is preferred to use insect cells and mammalian cells for the preparation of functional proteins.
[0060]For example, when polypeptide is expressed in mammalian cells, DNA encoding the sodium-independent transporter AGT1 which transports acidic amino acids according to the present invention or DNA encoding a fusion protein of AGT1 with 4F2hc or rBAT is inserted into a downstream side of an appropriate promoter (such as cytomegalovirus promoter, SV40 promoter, LTR promoter, elongation 1 a promoter, etc.) in an appropriate expression vector (such as adenovirus vector, retrovirus vector, papilloma virus vector, vaccinia virus vector, SV40 vector, etc.) so that expression vector is constructed. Then, an appropriate animal cell is transformed using the resulting expression vector and the transformant is incubated in an appropriate medium whereby a desired polypeptide is produced. Examples of the mammalian cell used as a host are cell strains such as simian COS-7 cell, Chinese hamster CHO cell and human HeLa cell.
[0061]Accordingly, the present invention provides a vector, preferably an expression vector, which contains a gene encoding the above-mentioned gene of the present invention or for a protein in the gene and also provides a host cell (transformant) which is transformed using the vector.
[0062]With regard to the DNA encoding the sodium-independent transporter AGT1 which transports acidic amino acids, cDNA having the base sequence represented by SEQ ID NO: 2 may be used, for example, in addition to that, DNA corresponding to the amino acid sequence is designed and may be used as DNA encoding polypeptide without limiting to the above-mentioned cDNA sequence. In that case, with regard to a codon encoding one amino acid, 1 to 6 kinds are known for each, and although the used codon may be optionally selected, frequency of use of a codon of a host utilized for the expression may be taken into consideration to design a sequence having higher expression efficiency. DNA having the designed base sequence can be prepared by chemical synthesis of DNA, binding to fragmentation of the above-mentioned cDNA, partial modification of the base sequence, and the like. The artificial partial modification of and introduction of variation into base sequence may be carried out utilizing a primer comprising synthetic oligonucleotide encoding the desired modification by a site-specific mutagenesis (Mark, D. F., et al., Proceedings of National Academy of Sciences, volume 81, page 5662 (1984), etc.).
[0063]DNA encoding a fusion protein (AGT1-4F2hc or AGT1-rBAT) of the sodium-independent transporter AGT1 which transports acidic amino acids with 4F2hc or rBAT may be prepared, for example, using a base sequence represented by SEQ ID NO: 2 or SEQ ID NO: 4 or using cDNA having a base sequence represented by SEQ ID NO: 6 and, moreover, DNA corresponding to the amino acid sequence is designed and can be used as DNA encoding polypeptide without limiting to the above-mentioned cDNA sequences. In that case, with regard to a codon encoding one amino acid, 1 to 6 kinds are known for each, and although the used codon may be optionally selected, frequency of use of a codon of a host utilized for the expression may be taken into consideration to design a sequence having higher expression efficiency may be designed. DNA having the designed base sequence can be prepared by chemical synthesis of DNA, binding to fragmentation of the above-mentioned cDNA, partial modification of the base sequence, and the like. The artificial partial modification and introduction of variation into base sequence may be carried out utilizing a primer comprising synthetic oligonucleotide encoding the desired modification by a site-specific mutagenesis (Mark, D. F., et al., Proceedings of National Academy of Sciences, volume 81, page 5662 (1984), etc.).
[0064]The present invention also provides a nucleotide containing a partial sequence of continuous 14 or more bases, preferably 20 or more bases, in the base sequence represented by SEQ ID NO: 2 or the complementary sequence thereof.
[0065]The nucleotide of the present invention can be used as a probe for detection of a gene encoding a protein having an ability of transport of acidic amino acids or its analogue in a sodium-independent manner. It can be also used as a primer for obtaining a gene encoding the protein and the gene encoding a protein having high homology thereto. Further, it can be used for modulation of expression of a gene encoding a protein having an ability to transport acidic amino acids and its analogue in a sodium-independent manner by its anti-sense chain, etc.
[0066]It is possible to prepare the corresponding antibody using the sodium-independent transporter which transports acidic amino acids of the present invention or using a polypeptide having an immunological homology thereto. The antibody can be utilized for detection, purification, and the like. of the sodium-independent transporter which transports acidic amino acids. The antibody can be manufactured using the sodium-independent transporter which transports acidic amino acids according to the present invention, a fragment thereof, synthetic peptide having a partial sequence thereof, etc. as an antigen. Polyclonal antibody can be manufactured by a conventional method where antigen is inoculated to a host animal (such as rat and rabbit) and immune serum is recovered therefrom while monoclonal antibody can be manufactured by a conventional technique such as a hybridoma method.
[0067]The protein of the present invention has an ability of transporting acidic amino acids and its analogue in a sodium-independently manner and the ability is strongly affected in the presence of various substances. By screening a substance which inhibits or accelerates the ability, the ability of the present protein for transporting the substance can be controlled.
[0068]Accordingly, the present invention provides a method for detecting an effect of a test substance as a substrate on an ability of the protein of the present invention for transporting acidic amino acids or its analogue in a sodium-independent manner using the above-mentioned protein of the present invention.
[0069]The amino acid which is transported by the protein of the present invention is the substance essential for proliferation and growth of cells and for maintenance of life, and by controlling the uptake of such a substance into cells, proliferation, growth, etc. of cells can be controlled. Accordingly, the present invention provides a method for controlling the cell proliferation by modulating an ability of the protein for transporting the acidic amino acids and analogous substance thereto using the above-mentioned protein of the present invention, a specific antibody thereof or a function-promoting or function-suppressing substance thereof.
[0070]Gene of a fusion protein of the sodium-independent transporter AGT1 transporting the acidic amino acids with 4F2hc or rBAT according to the present invention and the expressed cell thereof can be used for an in vitro test for the efficiency of permeation of a substance at a cell membrane where AGT1 is present or at the site where the presence of AGT1 is assumed. In addition, a gene of a fusion protein of the sodium-independent transporter AGT1 transporting the acidic amino acids with 4F2hc or rBAT and the expressed cell thereof can be used for the development of a compound which efficiently permeates through a cell membrane where AGT1 is present or at the site where the presence of AGT1 is assumed. Further, a gene of a fusion protein of the sodium-independent transporter AGT1 transporting the acidic amino acids with 4F2hc or rBAT and the expressed cell thereof can be used for an in vitro test of pharmaceutical interaction at a cell membrane where AGT1 is present or at the site where the presence of AGT1 is assumed.
[0071]Accordingly, the present invention provides a method for changing the pharmacokinetics of pharmaceuticals or xenobiotics transported by the above-mentioned protein of the present invention by using the protein, a specific antibody thereof or a function-promoting or function-suppressing substance thereof, by modulating an ability of the protein for transport acidic amino acids or its analogue.
[0072]As described above, since the protein of the present invention has an ability to transport acidic amino acids or its analogue in a sodium-independent manner and this ability can be suppressed or promoted not only by the number of a protein existing in a cell, but also by the presence of various substances (in the presence of a function-suppressing substance, etc. or in the presence of a function-promoting substance, etc., respectively), the present invention provides a controlling agent for transport ability of a protein for acidic amino acids or its analogue possessed by the above-mentioned protein of the present invention which comprises the protein, a specific antibody thereof or a function-promoting substance or function-suppressing substance thereof.
[0073]Since the controlling agent for transport ability of the present invention can control the proliferation, growth, and the like. of cells, it can be used as a controlling agent for cell proliferation, and since the agent can modulate and control the pharmacokinetics of pharmaceutical, toxic substance or xenobiotics, it can be used as a controlling agent for pharmacokinetics of pharmaceutical, toxin or xenobiotics.
[0074]By suppressing the sodium-independent transporter AGT1 of the present invention which transports acidic amino acids, the permeation of a specific compound through the cell membrane where AGT1 is expressed or through the site where AGT1 is assumed to be present can be limited. In addition, a gene of a fusion protein of the sodium-independent transporter AGT1 of the present invention transporting the acidic amino acids with 4F2hc or rBAT and its expression cell can be used for the development of a pharmaceutical (such as a specific inhibitor for AGT1) which limits the permeation of a compound transported by AGT1 through a cell membrane or the site where AGT1 is assumed to be present.
[0075]Further, in accordance with the present invention, it has been found that the protein having an amino acid sequence represented by SEQ ID NO: 3 or NO: 5 comprises an ability to promote the transfer AGT1 into a cell membrane. Accordingly, the present invention provides a promoting agent for the transfer of AGT1 into a cell membrane containing a protein having an amino acid sequence represented by SEQ ID NO: 3 or NO: 5 or a protein having an amino acid sequence where one or several amino acid(s) of the above protein is/are deleted, substituted or added.
[0076]All of the contents mentioned in the specification of the Japanese patent application No. 2002-040,608 shall be incorporated into the present specification.
EXAMPLES
[0077]The present invention will now be illustrated in more detail by way of the following Examples although those Examples do not limit the present invention.
[0078]In the following Examples, each operation was carried out, unless otherwise clearly mentioned, according to a method mentioned in "Molecular Cloning" by Sambrook, J., Fritsh, E. F. and Manitis, T. (Cold Spring Harbor Press, 1989) or according to the Directions for Use of the commercially available products when the commercially available reagents or kits are used.
Example 1
[0079]Cloning and expression analysis of a sodium-independent transporter which transports acidic amino acids
[0080](1) Identification of Mouse cDNA of a Sodium-Independent Transporter which Transports Acidic Amino Acids
[0081]cDNA clone corresponding to the base sequence GenBank®/EBI/DDBJ accession No. AI314100 derived from mouse analogous to rat BAT1 obtained by searching the EST (expressed sequence tag) database using a base sequence of a translation region of rat BAT1 (Chairoungdua, et al., J. Biol. Chem., 274: 28845-28848, 1999) was purchased from IMAGE (Integrated and Molecular Analysis of Genomes and their Expression) (IMAGE clone I. D.: 1907807) and its fragment (1.8-kb) cleaved by a restriction enzyme XhoI was labeled with 32P-dCTP and used as a probe whereby a mouse kidney cDNA library was screened.
[0082]The cDNA library was prepared from poly(A)+RNA derived from mouse kidney using a kit for the synthesis of cDNA (trade name: Superscript Choice System, manufactured by Gibco) and incorporated into a site of phage vector AZipLox (manufactured by Gibco) cleaved by a restriction enzyme EcoRI. Hybridization by a probe labeled with 32P-dCTP was carried out for one night in a solution for hybridization of 37° C. and the filter membrane was washed with 0.1×SSC/0.1% SDS at 37° C. With regard to the solution for hybridization, a buffer of pH 6.5 containing 5×SSC, 3×Denhard's solution, 0.2% SDS, 10% dextran sulfate, 50% formamide, 0.01% Abtiform B (trade name; Sigma) (antifoaming agent), 0.2 mg/ml salmon sperm-modified DNA, 2.5 mM sodium pyrophosphate and 25 mM MES was used. The cDNA portion of λZipLox phage into which cDNA was incorporated was incorporated into a plasmid pZL1. The resulting cDNA-inserted fragment of clone was further incorporated into an NotI-cleaved site of a plasmid pcDNA 3.1 (Invitrogen).
[0083]A base sequence for the full-length cDNA was determined by a dye terminator cycle sequencing method (Applied Biosystems) using a synthetic primer for the determination of base sequence. Further, a base sequence of cDNA was analyzed by a conventional method and translation region of cDNA and amino acid sequence of the protein encoded thereby were determined.
[0084]Those sequences are represented in SEQ ID NO: 1 of the Sequence Listing which will be shown later.
[0085]AGT1 comprised a 48% homology to a mouse transporter Asc-2 corresponding to a neutral amino acid transport system asc. Further, AGT1 comprised a 35% homology to a rat transporter LAT1 and 37% to LAT2 corresponding to a neutral amino acid transport system L, 37% homology to a rat transporter y+LAT1 and 36% to a human transporter y+LAT2 corresponding to neutral and basic amino acid transport system y+L. Furthermore, AGT1 comprised a 37% homology to a mouse transporter Asc-1 corresponding to a neutral amino acid transport system asc, 37% to a mouse transporter xCT corresponding to a cystine and acidic amino acid transport system x, and 36% to a rat transporter BAT1 corresponding to a cystine, neutral and basic amino acid transport system b0,+. Still further, Asc-2 comprised a 30% homology to a mouse and human transporter CAT1 to 4 corresponding to a basic amino acid transport system y+.
[0086]Comparison of AGT1 with mouse Asc-2, rat LAT1, rat y+LAT1, mouse xCT and rat BAT1 in terms of amino acid sequences is shown in FIG. 1.
[0087]When an amino acid sequence of AGT1 was analyzed by an SOSUI algorithm (Hirokawa, T., et al., Bioinformatics, volume 14, page 378 (1998)), 12 membrane-spanning domains were assumed as shown in FIG. 1. In the third hydrophilic loop, conserved cysteine residues were present among Asc-2, LAT1, Asc-2, y+LAT1, xCT and BAT1. It is assumed that, via the cysteine residue, Asc-2 is binded to unknown cofactor via a disulfide bond. In addition, there were sites believed to be a cAMP-dependent phosphorylation site in the eighth hydrophilic loop, C-kinase-dependent phosphorylation sites in an N-terminal intracellular region and the sixth hydrophilic loop and a tyrosine phosphorylation site in an N-terminal intracellular region respectively.
[0088](2) Expression of AGT1 Gene in Various Tissues of Mouse (Analysis by Northern Blotting)
[0089]cDNA fragments corresponding to 43rd to 1836th base pair of AGT1 gene were amplified by a PCR, labeled with 32P-dCTP and using as a probe, a northern blotting was carried out in the following manner to RNA extracted from various tissues of mouse. 3 μg of poly(A)+RNA was subjected to an electrophoresis using 1% agarose/formaldehyde gel and then transferred to a nitrocellulose filter. The filter was subjected to a hybridization for one night using a hybridization solution containing Asc-2 cDNA fragments labeled with 32P-dCTP. The filter was washed at 65° C. with 0.1×SSC containing 0.1% SDS.
[0090]As a result of the northern blotting (FIG. 2), a band was detected at about 2.2 kb in the kidney.
[0091](3) Expression of AGT1 Protein in the Mouse Kidney
[0092]A specific antibody to synthetic oligopeptide (CIPDVSDDHIHEES) (mentioned in SEQ ID NO: 7 of Sequence Listing) corresponding to 465-478 of mouse AGT1 was prepared according to a method of Altman, et al. (Altman, et al., Proc. Natl. Acad. Sci. USA, volume 81, pages 2176-2180, 1984).
[0093]Membrane fraction of mouse kidney was prepared according to a method of Thorens, et al. (Thorens, et al., Cell, volume 55, pages 281-290, 1988). The protein sample was treated at 100° C. for 5 minutes in the presence (under a reducing condition) or absence (under a non-reducing condition) of 5% 2-mercaptoethanol, subjected to electrophoresis by an SDS-polyacrylamide gel, blotted to Hybond-P PVDV transfer membrane and treated with an anti-AGT1 antiserum (1:10,000).
[0094]As a result, in the mouse kidney, a band was detected near 250 kDa under a non-reducing condition by an anti-AGT1 antibody as shown in FIG. 3. Under a reducing condition, a band was detected near 40 kDa. From those results, it is suggested that AGT1 is binded to some protein by a disulfide bond.
[0095](4) Immunohistological Analysis of AGT1 Protein in the Mouse Kidney
[0096]According to a conventional method, a mouse kidney paraffin slice was treated with an anti-AGT1 antiserum (1:1,000) and colored with diaminobenzidine. Further, with an object of investigating the specificity of color development, an experiment of treating with an anti-AGT1 antiserum (1:1,000) in the presence of 50 mg/ml of an antigen peptide was also carried out.
[0097]As a result, in the mouse kidney, stainings were noted in proximal tubule of outer layer of medulla and in distal tubule of cortex as shown in FIG. 4a. As the stainings were not detected when an anti-AGT1 antiserum was made to act in the presence of an antigen peptide, the specificity in staining was shown (FIG. 4b). Further, when an observation was conducted with highly magnified, it was clarified that AGT1 protein was present in basolateral membrane of proximal tubule (FIG. 4c) and distal tubule (FIG. 4d).
Example 2
Preparation of a Fusion Protein of Sodium-Independent Transporter AGT1 Transporting Acidic Amino Acids with 4F2hc or with rBAT and Analysis of its Function
[0098](1) Preparation of a Fusion Protein of Sodium-Independent Transporter AGT1 Transporting Acidic Amino Acids with 4F2hc or rBAT
[0099]In order to prepare a fusion protein of AGT1 with rBAT(AGT1-rBAT), a PCR was carried out using synthetic oligo-DNA primers 5'-GCGCGAAGCTTACCTATAGGCAGAAACATTC-3' (in which, to a sequence corresponding to 4th to 23rd base pair of AGT1 cDNA were added a sequence corresponding to cleaved site with HindIII and GCGC at 5'-side; mentioned in SEQ ID NO: 8 of the Sequence Listing) and 5'-ATATGCGGCCGCACTTTCTTCATGTATGTGGT-3' (in which, to a sequence corresponding to 1473rd to 1492nd base pair of AGT1 cDNA were added a sequence corresponding to the cleaved site with NotI and ATAT at 5'-side; mentioned in SEQ ID NO: 9 of the Sequence Listing) where AGT1 cDNA was used as a template. The resulting PCR product was cleaved with HindIII and NotI and ligated to HindIII and NotI sites of mammalian cell expression vector pcDNA3.1(+) (Invitrogen). Further, a PCR was carried out using a synthetic oligo-DNA primers 5'-ATATGCGGCCGCAGATGAGGACAAAGGCAAGAG-3' (in which, to a sequence corresponding to the base pair immediately after translation initiation codon ATG of mouse rBAT to 21st as shown in SEQ ID NO: 6 were added a sequence corresponding to a site cleaved by NotI and ATAT at 5'-side; mentioned in SEQ ID NO: 10 in the Sequence Listing) and 5'-GCGCGCTCTAGAAATGCTTTAGTATTTGGCATAATC-3' (in which, to a sequence of 2228th to 2251st base pair of mouse rBAT as shown by SEQ ID NO: 6 were added a sequence corresponding to a site cleaved with XbaI and GCGC at 5'-side; mentioned in SEQ ID NO: 11 in the Sequence Listing) where rBAT cDNA was used as a template. The resulting PCR product was cleaved with NotI and XbaI and ligated to NotI and XbaI sites of the mammalian cell expression vector pcDNA3.1(+) into which the above-mentioned AGT1 PCR product was incorporated to prepare cDNA encoding a fusion protein of AGT1 with rBAT (FIG. 5).
[0100]In order to prepare a fusion protein of AGT1 with 4F2hc(AGT1-4F2hc), a PCR was carried out using synthetic oligo-DNA primers 5'-GCGCGAAGCTTACCTATAGGCAGAAACATTC-3' (in which, to a sequence corresponding to 4th to 23rd base pair of AGT1 cDNA were added a sequence corresponding to a site cleaved by HindIII and GCGC at 5'-side; mentioned in SEQ ID NO: 8 of the Sequence Listing) and 5'-ATATGCGGCCGCACTTTCTTCATGTATGTGGT-3' (in which, to a sequence corresponding to 1473rd to 1492nd base pair of AGT1 cDNA were added a sequence corresponding to a site cleaved by NotI and ATAT at 5'-side; mentioned in SEQ ID NO: 9 of the Sequence Listing) where AGT1 cDNA was used as a template. The resulting PCR product was cleaved with HindIII and NotI and ligated to HindIII and NotI sites of mammalian cell expression vector pcDNA3.1(+) (Invitrogen). Further, a PCR was carried out using synthetic oligo-DNA primers 5'-ATATGCGGCCGCAAGCCAGGACACCGAAGTGGA-3' (in which, to a sequence corresponding to the base pair immediately after translation initiation codon ATG of mouse 4F2hc to 21st as shown in SEQ ID NO: 4 were added a sequence corresponding to a site cleaved by NotI and ATAT at 5'-side; mentioned in SEQ ID NO: 12 in the Sequence Listing) and 55'-GCGCTCTAGACATGAGGCAGGGGTGATGTTIT-3' (in which, to a sequence corresponding to 1820th to 1838th base pair of mouse 4F2hc shown in SEQ ID NO: 4 were added a sequence corresponding to a site cleaved by XbaI and GCGC at 5'-side; mentioned in SEQ ID NO: 13 of the Sequence Listing) where 4F2hc cDNA was used as a template. The resulting PCR product was cleaved with NotI and XbaI and ligated to NotI and XbaI sites of mammalian cell expression vector pcDNA3.1(+) into which the above-mentioned AGT1 PCR product was incorporated to give cDNA encoding a fusion protein of AGT1 with 4F2hc (FIG. 5).
[0101](2) Expression of a Function of a Fusion Protein of a Sodium-Independent Transporter AGT1 Transporting Acidic Amino Acids with 4F2hc or rBAT
[0102]Comparisons were conducted for the uptake of aspartic acid when mouse AGT1 gene cRNA was expressed in the oocyte, when mouse AGT1 gene cRNA and mouse 4F2hc gene cRNA were expressed in the oocyte and when a fusion protein of ACT1 with 4F2hc or rBAT was expressed in the oocyte.
[0103]25 ng of mouse 4F2hc gene cRNA, 25 ng of AGT1 gene cRNA, 12.5 ng of AGT1 gene cRNA/12.5 ng of mouse 4F2hc gene cRNA, 25 ng of AGT1-4F2hc fusion protein gene cRNA or 25 ng of AGT1-rBAT fusion protein gene cRNA was injected into the oocyte to express and incubation was conducted for 3 days.
[0104]With regard to the oocyte into which mouse 4F2hc gene cRNA, AGT1 gene cRNA, AGT1 gene cRNA/mouse 4F2hc gene cRNA, AGT1-4F2hc fusion protein gene cRNA or AGT1-rBAT fusion protein gene cRNA was injected, experiments for the uptake of a substrate was carried out using aspartic acid as a substrate according to a method of Kanai, et al. (Kanai and Hediger, Nature, volume 360, pages 467-471, 1992) as follows. The oocyte were allowed to stand for 30 minutes in a sodium-free uptake solution (100 mM choline chloride, 2 mM potassium chloride, 1.8 mM calcium chloride, 1 mM magnesium chloride and 5 mM HEPES; pH 7.4) containing 14C-aspartic acid (20 mM) as a substrate and an uptake rate of the substrate was measured by the count of radioactivity incorporated into the cells.
[0105]As a result (FIG. 6), the levels of the uptake of aspartic acid in the oocyte where only 4F2hc was expressed, the oocyte where only AGT1 was expressed and the oocyte where both AGT1 and 4F2hc were co-expressed were similar to that in the control oocyte into which water was injected, while a higher uptake of aspartic acid was noted in the oocyte where AGT1-rBAT or AGT1-4F2hc was expressed.
[0106]It was investigated that rBAT or 4F2hc cannot be a direct cofactor of AGT1 using COS-7 cells. According to a method mentioned in Mizoguchi, et al., Kidney Int, 59: 1821-1833, 2001, plasmid DNA (each 1 mg) containing AGT1 cDNA, rBAT cDNA or 4F2hc cDNA was introduced into COS-7 cells using LIPOFECTAMINE 2000 Reagent (Life Technologies). After the introduction, the cells were incubated for two days in a 24-well plate and the uptake of 14C-aspartic acid (20 mM) was measured. Measurement of the uptake was conducted according to a method of Mizoguchi, et al., Kidney Int, 59: 1821-1833, 2001, in which it was started by removing the culture liquid and adding Dulbecco's PBS (manufactured by Gibco) containing 14C-aspartic acid, and completed by removing it and washing with ice-cooled Dulbecco's PBS. After the washing, it was dissolved with 0.1N NaOH and radioactivity was measured by a liquid scintillation counter.
[0107]As a result (FIG. 7), the levels of the uptake of aspartic acid in the oocyte where only 4F2hc was expressed, the oocyte where only rBAT was expressed, the oocyte where only AGT1 was expressed, the oocyte where both AGT1 and 4F2hc were co-expressed and the oocyte where both AGT1 and rBAT were co-expressed were similar to that the control oocyte into which a pcDNA 3.1 plasmid containing no inserted cDNA whereby it was confirmed that rBAT or 4F2hc was not a direct cofactor for AGT1.
[0108](3) Identification of Expression of a Fusion Protein of Sodium-Independent Transporter AGT1 Transporting Acidic Amino Acids with 4F2hc (AGT1-4F2hc) in Oocyte Cell Membrane by a Immunofluorescence Analysis
[0109]Whether the fact that when AGT1 was expressed in the oocyte, no function was observed while a fusion protein of AGT1 with 4F2hc (AGT1-4F2hc) showed a functional activity is due to the fact that AGT1 is not transported to a cell membrane while AGT1-4F2hc is transported to a cell membrane or not was investigated by a immunofluorescence analysis.
[0110]25 ng of AGT1 gene cRNA or 25 ng of the cRNA of a gene of a fusion protein of AGT1 with 4F2hc(AGT1-4F2hc) was injected into the oocyte to express, incubated for 3 days and, the oocyte was fixed in a 4% paraformaldehyde-phosphate buffer and prepared a paraffin section (3 mm) according to a conventional method. After removing the paraffin, the section was subjected to a blocking with 5% goat serum in 0.05M Tris buffer in a physiological saline containing 0.1% Tween 20 and treated with an affinity-purified anti-Asc-2 antibody or an affinity-purified anti-4F2hc antibody (Fukasawa, et al., J. Biol. Chem., 275: 9690-9698, 2000). Then, the section was treated with Alexa Fluor 488-labeled goat anti-rabbit IgG (Molecular Probe, Inc.), washed with 0.05M Tris buffer in physiological saline containing 0.1% Tween 20 and observed with Olympus Fluoview (FV500) confocal laser microscope (Olympus). Excitation was effected with argon laser. at 488 nm and fluorescence from Alexa Fluor 488 was detected using a BA505IF filter.
[0111]As a result (FIG. 8), in the oocyte in which AGT1 was expressed, an AGT1 protein detected in an anti-AGT1 antibody was not present in a cell membrane but remained inside the cell membrane (FIG. 8d), while in an oocyte in which a fusion protein of AGT1 with 4F2hc(AGT1-4F2hc) was expressed, an AGT1-4F2hc fusion protein expressed in a cell membrane was detected in both anti-4F2hc antibody (FIG. 8 e) and anti-AGT1 antibody (FIG. 80. In the control oocyte into which water was injected, no specific color development by anti-4F2hc antibody (FIG. 8a) or anti-AGT1 antibody (FIG. 8b) was observed. Accordingly, it was proven that the fact that no function was observed when AGT1 was expressed in the oocyte while a fusion protein of AGT1 with 4F2hc(AGT1-4F2hc) showed a functional activity is due to the fact that AGT1 is not transported to a cell membrane by itself, while its fusion protein with 4F2hc (AGT1-4F2hc) is transported to a cell membrane.
[0112](4) Salt-Dependency of Transport Activity of AGT1
[0113]In an uptake experiment of aspartic acid by the oocyte into which a cRNA of a gene of a fusion protein of AGT1 with 4F2hc or with rBAT (AGT1-4F2hc or AGT1-rBAT) was injected, influence of salt added to the medium was investigated.
[0114]An uptake experiment of aspartic acid was carried out according to the method mentioned in the above Example 2(2) using an oocyte into which a cRNA of a gene of a fusion protein of AGT1 with 4F2hc or with rBAT (AGT1-4F2hc or AGT1-rBAT) was injected. With regard to the uptake solution, a standard uptake solution (100 mM of choline chloride was exchanged with 100 mM sodium chloride) was used instead of a sodium-free uptake solution when the effect of sodium ion was investigated. A gluconic acid uptake solution (100 mM sodium chloride was exchanged with 100 mM sodium gluconate) was used instead of a standard uptake solution when the effect of chlorine ion was investigated.
[0115]As a result (FIG. 9), even when extracellular choline was exchanged with sodium or even when extracellular chlorine was exchanged with gluconate ion, it did not affect the uptake of aspartic acid at all. From the above, it was noted that Asc-2 is a transporter which acts independently on sodium ion and chlorine ion.
[0116](5) Michaelis-Menten Kinetic Analysis of AGT1
[0117]A Michaelis-Menten kinetic analysis of sodium-independent transporter AGT1 which transports acidic amino acids was carried out. The Michaelis-Menten kinetic analysis was conducted by investigating the change in the ratio of uptake of aspartic acid by the difference in the substrate aspartic acid concentration.
[0118]The aspartic acid uptake experiment was carried according to the method mentioned in the above Example 2(2) using the oocyte into which a cRNA of a gene of a fusion protein of AGT1 with 4F2hc or with rBAT (AGT1-4F2hc or AGT1-rBAT) was injected. As a result (FIG. 10), the Km value of aspartic acid transport by AGT1-4F2hc was 25.5+5.9 mM (mean value+standard error). The Km value of aspartic acid transport by AGT1-rBAT was 20.1+6.1.
[0119]The Michaelis-Menten kinetic analysis was similarly carried out in a fusion protein of AGT1 with 4F2hc (AGT1-4F2hc) in glutamic acid and Km value and Vmax value were calculated. Result of the above is shown in the following Table 1.
Table 1
[0120]Km values and Vmax values of substrate amino acid
TABLE-US-00001 Amino Acid Kma (μM) Vmaxb L-Aspartic acid 25.5 ± 5.9 (1.00) L-Glutamic acid 21.8 ± 6.5 0.63 ± 0.10 a, bV max value of L-glutamic acid is shown by the ratio to Vmax value of L-aspartic acid. Both Km and Vmax values are represented by mean value ± standard error.
[0121](6) Substrate Selectivity of AGT1 (Inhibition Experiment Using Added Amino Acid and Its Analogue)
[0122]In an uptake experiment of aspartic acid by an oocyte into which a cRNA of a gene of a fusion protein of AGT1 with 4F2hc (AGT1-4F2hc) was injected, the effect of addition of various amino acids and their analogues on the system was investigated.
[0123]An aspartic acid uptake experiment was carried out according to the method mentioned in the above Example 2(2) using an oocyte into which a cRNA of a gene of a fusion protein of AGT1 with 4F2hc (AGT1-4F2hc) was injected. However, a sodium-free uptake solution was used and the uptake of 14C-aspartic acid (20 mM) was measured in the presence and absence of 2 mM of various compounds (non-labeled).
[0124]As a result (FIG. 11), in aspartic acid, glutamic acid and cysteine, a significant cis-inhibiting effect was observed.
[0125]Basic amino acids, neutral amino acids except cysteine, cystine, 2-amino-2-norbornane-carboxylic acid (BCH) which is a transport system L-specific inhibitor, γ-aminoisobutyric acid, α-aminomethylisobutyric acid, D-aspartic acid and D-glutamic acid did no affect the uptake of 14C-aspartic acid mediated by AGT1-4F2hc (FIG. 11).
[0126]In an oocyte into which cRNA of a gene of a fusion protein of AGT1 with rBAT (AGT1-rBAT) were injected together, the effect of adding various amino acids and their analogues on the system was also investigated in an aspartic acid uptake experiment by an oocyte in a similar manner as in the case of AGT1-4F2hc.
[0127]As a result, in the case of AGT1-rBAT, the same result as in the case of AGT1-4F2hc was obtained, and in the case of a fusion protein of AGT1 with 4F2hc or with rBAT (AGT1-4F2hc or AGT1-rBAT), the 4F2hc or rBAT moiety did not affect the characteristic of substrate-binding site and, with regard to the information concerning the substrate selectivity obtained in a fusion protein, the AGT1 itself also reflects the transport characteristic.
[0128]In the substances analogous to acidic amino acids, threo-β-hydroxyaspartate (THA), L-serine-O-sulfate (SOS), L-cysteine sulfate and L-cysteate strongly inhibited the uptake of 14C-aspartic acid mediated by AGT1.
[0129]On the contrary, in the case of L-α-aminoadipate, L-homocysteate, L-trans-pyrrolidine-2,4-dicarboxylate (PDC) and dihydrokainate (DHK), no inhibition effect on the uptake of 14C-aspartic acid mediated by AGT1 was observed (FIG. 12).
[0130](7) Substrate Selectivity of AGT1 (Uptake Experiment Using Various Amino Acids and their Analogues as Substrates)
[0131]Various kinds of amino acids and their analogues were used as substrates and uptake by an oocyte into which a cRNA of a gene of a fusion protein of AGT1 with 4F2hc (AGT1-4F2hc) was injected was investigated.
[0132]The uptake experiment of various amino acids and their analogues was carried out according to the method mentioned in the above-mentioned Example 2(2) using an oocyte into which a cRNA of a gene of a fusion protein of AGT1 with 4F2hc (AGT1-4F2hc) was injected. With regard to a substrate, various compounds which were labeled with radioactivity were used instead of 14C-aspartic acid.
[0133]As a result, when L-glutamic acid (14C compound) was used as a substrate in addition to L-aspartic acid (14C compound) (FIG. 13), a substantial uptake into the oocyte was observed.
INDUSTRIAL APPLICABILITY
[0134]The sodium-independent transporter of the present invention which transports acidic amino acids and a gene thereof enables an in vitro investigation of transport of acidic amino acids and amino acid analogues including xenobiotics at the site where the transporter is expressed, and based on which, also enables an in vitro assumption of pharmacokinetic of these compounds. Further, the present invention is useful for the development of pharmaceutical which permeates efficiently through a site where the transporter is expressed. Furthermore, by modulating an ability to transport acidic amino acids and its analogue possessed by the transporter, the invention can be utilized for the development of a method for controlling a cell proliferation.
Sequence CWU
1
221478PRTMus sp.AGT1 1Met Ala Met Asp Ser Lys Lys Glu Ile Arg Leu Lys Arg
Glu Leu Gly1 5 10 15Tyr
Phe Trp Gly Thr Asn Phe Leu Ile Ile Asn Ile Ile Gly Ala Gly 20
25 30Ile Phe Val Ser Pro Lys Gly Val
Leu Gln His Ser Ser Met Asn Val 35 40
45Gly Val Ser Leu Cys Val Trp Ala Val Cys Ala Val Leu Thr Leu Thr
50 55 60Ser Ala Leu Cys Ser Ala Glu Ile
Gly Ile Thr Phe Pro Tyr Ser Gly65 70 75
80Ala His Tyr Tyr Phe Leu Lys Arg Cys Phe Gly Pro Leu
Val Ala Phe 85 90 95Leu
Arg Leu Trp Thr Ser Leu Phe Leu Gly Pro Gly Leu Ile Ala Ser
100 105 110Gln Ala Leu Leu Leu Ala Glu
Tyr Gly Val Gln Pro Phe Tyr Pro Ser 115 120
125Cys Ser Ala Pro Ile Leu Pro Arg Lys Cys Leu Ala Leu Ala Met
Leu 130 135 140Trp Ile Val Gly Ile Leu
Asn Ser Arg Gly Val Lys Glu Leu Ser Trp145 150
155 160Leu Gln Thr Val Ser Ser Val Leu Lys Val Gly
Ile Leu Gly Val Ile 165 170
175Ser Leu Ser Gly Leu Phe Leu Leu Val Arg Gly Lys Lys Glu Asn Val
180 185 190Gln Arg Leu Gln Asn Ala
Phe Asp Ala Glu Phe Pro Glu Val Ser Gln 195 200
205Leu Ile Glu Ala Ile Phe Gln Gly Tyr Phe Ala Phe Ser Gly
Gly Gly 210 215 220Cys Phe Thr Cys Ile
Ala Gly Glu Leu Lys Lys Pro Ser Lys Thr Ile225 230
235 240Pro Arg Cys Ile Phe Thr Gly Leu Pro Leu
Val Thr Val Val Tyr Leu 245 250
255Leu Ala Asn Ile Ser Tyr Leu Thr Val Leu Thr Pro Gln Glu Met Leu
260 265 270Ser Ser Asp Ala Val
Ala Leu Thr Trp Thr Asp Arg Val Ile Pro Gln 275
280 285Phe Thr Trp Thr Val Pro Phe Ala Ile Ser Ala Ser
Leu Phe Ile Asn 290 295 300Leu Val Ile
Asn Val Leu Glu Thr Ser Arg Val Leu Tyr Ile Ala Ser305
310 315 320Glu Asn Gly Gln Leu Pro Leu
Leu Phe Cys Ala Leu Asn Val His Ser 325
330 335Ser Pro Phe Ile Ala Val Leu Leu Ile Ile Ser Met
Ala Ser Ile Leu 340 345 350Ile
Val Leu Thr Asn Leu Ile Asp Leu Ile Asn Tyr Leu Tyr Phe Val 355
360 365Val Ser Ile Trp Thr Ala Leu Ser Ile
Ile Gly Ile Leu Lys Leu Arg 370 375
380Tyr Gln Glu Pro Asn Leu His Arg Pro Tyr Lys Val Phe Leu Pro Phe385
390 395 400Thr Phe Ile Ala
Leu Gly Ile Thr Leu Ser Leu Val Leu Ile Pro Leu 405
410 415Val Lys Ser Pro Lys Leu His Tyr Ile Tyr
Val Phe Leu Phe Leu Leu 420 425
430Ser Gly Leu Val Phe Tyr Val Pro Leu Ile His Phe Lys Val Lys Phe
435 440 445Val Trp Phe Gln Lys Leu Thr
Cys Tyr Leu Gln Leu Leu Phe Asn Ile 450 455
460Cys Ile Pro Asp Val Ser Asp Asp His Ile His Glu Glu Ser465
470 47522141DNAMus sp.CDS(59)..(1492)AGT1
2caaacctata ggcagaaaca ttcacagagt acaattttgt gaatgtaaac ttctctca
58atg gca atg gat tca aag aag gaa atc cgt ctt aag aga gaa ctt gga
106Met Ala Met Asp Ser Lys Lys Glu Ile Arg Leu Lys Arg Glu Leu Gly1
5 10 15tat ttc tgg ggg aca aac
ttt tta att att aat ata att ggt gca gga 154Tyr Phe Trp Gly Thr Asn
Phe Leu Ile Ile Asn Ile Ile Gly Ala Gly 20 25
30ata ttt gtg tcc ccc aag gga gtg ctc cag cac tct tcc
atg aat gtt 202Ile Phe Val Ser Pro Lys Gly Val Leu Gln His Ser Ser
Met Asn Val 35 40 45gga gtc tcc
ttg tgt gtt tgg gct gtc tgt gca gtg ctg acc ttg acc 250Gly Val Ser
Leu Cys Val Trp Ala Val Cys Ala Val Leu Thr Leu Thr 50
55 60agt gct ctc tgc tct gca gag atc ggg ata acc ttc
cca tac agt ggg 298Ser Ala Leu Cys Ser Ala Glu Ile Gly Ile Thr Phe
Pro Tyr Ser Gly65 70 75
80gct cac tat tat ttt tta aag cga tgc ttc ggc cct ctc gtg gca ttc
346Ala His Tyr Tyr Phe Leu Lys Arg Cys Phe Gly Pro Leu Val Ala Phe
85 90 95ctg agg ctt tgg act agc
ttg ttt ctc ggc cca ggc tta att gct agc 394Leu Arg Leu Trp Thr Ser
Leu Phe Leu Gly Pro Gly Leu Ile Ala Ser 100
105 110caa gct ctg cta ctg gct gag tat ggc gtt cag cct
ttt tat ccc agc 442Gln Ala Leu Leu Leu Ala Glu Tyr Gly Val Gln Pro
Phe Tyr Pro Ser 115 120 125tgc tct
gcc ccg att cta cca aga aaa tgt ctg gcc ctg gcg atg ctg 490Cys Ser
Ala Pro Ile Leu Pro Arg Lys Cys Leu Ala Leu Ala Met Leu 130
135 140tgg att gtg gga att ctg aat tct cgt ggt gta
aaa gag ctg tca tgg 538Trp Ile Val Gly Ile Leu Asn Ser Arg Gly Val
Lys Glu Leu Ser Trp145 150 155
160ctt cag aca gtg agc tca gtg ctg aag gtg ggc ata ctc ggt gtc att
586Leu Gln Thr Val Ser Ser Val Leu Lys Val Gly Ile Leu Gly Val Ile
165 170 175tcc ctc agc ggc ctg
ttc ttg ctg gtg aga ggg aag aag gag aat gta 634Ser Leu Ser Gly Leu
Phe Leu Leu Val Arg Gly Lys Lys Glu Asn Val 180
185 190caa agg ctt cag aat gcg ttt gat gcc gag ttc cca
gag gtc tct cag 682Gln Arg Leu Gln Asn Ala Phe Asp Ala Glu Phe Pro
Glu Val Ser Gln 195 200 205tta ata
gaa gct att ttc caa gga tac ttt gcg ttt tct ggc ggg gga 730Leu Ile
Glu Ala Ile Phe Gln Gly Tyr Phe Ala Phe Ser Gly Gly Gly 210
215 220tgc ttt aca tgt ata gca ggg gag ctg aag aaa
ccc agt aaa aca att 778Cys Phe Thr Cys Ile Ala Gly Glu Leu Lys Lys
Pro Ser Lys Thr Ile225 230 235
240cct aga tgc atc ttt aca gga ctg cct ctg gta act gtc gtg tac tta
826Pro Arg Cys Ile Phe Thr Gly Leu Pro Leu Val Thr Val Val Tyr Leu
245 250 255ctg gct aat att tcc
tac ctg aca gtt ctg aca ccc cag gaa atg ctc 874Leu Ala Asn Ile Ser
Tyr Leu Thr Val Leu Thr Pro Gln Glu Met Leu 260
265 270tct tca gat gct gtt gct ctt aca tgg aca gac agg
gtc att ccc caa 922Ser Ser Asp Ala Val Ala Leu Thr Trp Thr Asp Arg
Val Ile Pro Gln 275 280 285ttc aca
tgg act gtt cct ttt gct att tct gct tca ctg ttt atc aac 970Phe Thr
Trp Thr Val Pro Phe Ala Ile Ser Ala Ser Leu Phe Ile Asn 290
295 300ctt gtg att aat gta ctt gag aca tca aga gtg
tta tat att gca agt 1018Leu Val Ile Asn Val Leu Glu Thr Ser Arg Val
Leu Tyr Ile Ala Ser305 310 315
320gag aat ggc cag ctg cct ttg ttg ttt tgt gcc ctg aat gtc cat tcc
1066Glu Asn Gly Gln Leu Pro Leu Leu Phe Cys Ala Leu Asn Val His Ser
325 330 335tct ccc ttt ata gct
gtg cta cta att atc agt atg gca tcc att tta 1114Ser Pro Phe Ile Ala
Val Leu Leu Ile Ile Ser Met Ala Ser Ile Leu 340
345 350att gtc tta aca aac cta att gat ctg ata aac tat
ctc tat ttt gtt 1162Ile Val Leu Thr Asn Leu Ile Asp Leu Ile Asn Tyr
Leu Tyr Phe Val 355 360 365gtt tcc
att tgg act gcc tta tca ata ata gga atc ttg aaa ctg agg 1210Val Ser
Ile Trp Thr Ala Leu Ser Ile Ile Gly Ile Leu Lys Leu Arg 370
375 380tac caa gag ccc aat cta cac aga cca tat aag
gtg ttt tta ccg ttc 1258Tyr Gln Glu Pro Asn Leu His Arg Pro Tyr Lys
Val Phe Leu Pro Phe385 390 395
400aca ttc ata gcg ttg ggc atc acc ctg agc ttg gtt ttg atc ccg ctt
1306Thr Phe Ile Ala Leu Gly Ile Thr Leu Ser Leu Val Leu Ile Pro Leu
405 410 415gtc aag tct cca aag
ttg cat tat atc tat gtg ttc ctc ttc ctt ctc 1354Val Lys Ser Pro Lys
Leu His Tyr Ile Tyr Val Phe Leu Phe Leu Leu 420
425 430agt ggg ctg gtg ttt tac gtg cca ttg ata cac ttt
aaa gtg aag ttc 1402Ser Gly Leu Val Phe Tyr Val Pro Leu Ile His Phe
Lys Val Lys Phe 435 440 445gtt tgg
ttt cag aag ttg act tgc tat ctg cag tta ctg ttt aat att 1450Val Trp
Phe Gln Lys Leu Thr Cys Tyr Leu Gln Leu Leu Phe Asn Ile 450
455 460tgc atc cct gat gtg tct gat gac cac ata cat
gaa gaa agt 1492Cys Ile Pro Asp Val Ser Asp Asp His Ile His
Glu Glu Ser465 470 475tgaggaagaa
tagcctttgt agccatactg tgttccagat aaaggttaag tgtaagctaa 1552aatagtaatg
atggcaatgc tataaactga aatggtatat agaaaagtac ccaggaaatt 1612cctagttttt
aaaatatcaa aggaaatggc tgggtagatg actgtgctgt taagggcacc 1672agatgtcctt
ccaggggact gaagttcaat tcacaggccc cacttagaag ttcataacta 1732tctgtaattc
tagtcacaga ggatccaata tcctctactg gattctacca gcactgcatg 1792catctggagt
agagacatac atgtaggcac ccatgcacat ttacaagaag aaagaaagag 1852agaaagaaag
aaagaaagaa agaaagaaag aaagaaagaa agaaagaaag aaagggagag 1912ggagagagga
aggaaggaag gaaataagga aagaaggaag gaggaaagaa agacagtaaa 1972gaaagtattt
cacataacta aactgttttt attaaaaata aaatttctag cttgtatagc 2032ttcatgtagt
aagaatatct ttctcattct tctgtttatc tcatcgattt tctactgaat 2092gtattcttat
aataaaagtt actgatggaa attaaaaaaa aaaaaaaaa 21413526PRTMus
sp.4F2hc 3Met Ser Gln Asp Thr Glu Val Asp Met Lys Asp Val Glu Leu Asn
Glu1 5 10 15Leu Glu Pro
Glu Lys Gln Pro Met Asn Ala Ala Asp Gly Ala Ala Ala 20
25 30Gly Glu Lys Asn Gly Leu Val Lys Ile Lys
Val Ala Glu Asp Glu Thr 35 40
45Glu Ala Gly Val Lys Phe Thr Gly Leu Ser Lys Glu Glu Leu Leu Lys 50
55 60Val Ala Gly Ser Pro Gly Trp Val Arg
Thr Arg Trp Ala Leu Leu Leu65 70 75
80Leu Phe Trp Leu Gly Trp Leu Gly Met Leu Ala Gly Ala Val
Val Ile 85 90 95Ile Val
Arg Ala Pro Arg Cys Arg Glu Leu Pro Val Gln Arg Trp Trp 100
105 110His Lys Gly Ala Leu Tyr Arg Ile Gly
Asp Leu Gln Ala Phe Val Gly 115 120
125Arg Asp Ala Gly Gly Ile Ala Gly Leu Lys Ser His Leu Glu Tyr Leu
130 135 140Ser Thr Leu Lys Val Lys Gly
Leu Val Leu Gly Pro Ile His Lys Asn145 150
155 160Gln Lys Asp Glu Ile Asn Glu Thr Asp Leu Lys Gln
Ile Asn Pro Thr 165 170
175Leu Gly Ser Gln Glu Asp Phe Lys Asp Leu Leu Gln Ser Ala Lys Lys
180 185 190Lys Ser Ile His Ile Ile
Leu Asp Leu Thr Pro Asn Tyr Gln Gly Gln 195 200
205Asn Ala Trp Phe Leu Pro Ala Gln Ala Asp Ile Val Ala Thr
Lys Met 210 215 220Lys Glu Ala Leu Ser
Ser Trp Leu Gln Asp Gly Val Asp Gly Phe Gln225 230
235 240Phe Arg Asp Val Gly Lys Leu Met Asn Ala
Pro Leu Tyr Leu Ala Glu 245 250
255Trp Gln Asn Ile Thr Lys Asn Leu Ser Glu Asp Arg Leu Leu Ile Ala
260 265 270Gly Thr Glu Ser Ser
Asp Leu Gln Gln Ile Val Asn Ile Leu Glu Ser 275
280 285Thr Ser Asp Leu Leu Leu Thr Ser Ser Tyr Leu Ser
Asn Ser Thr Phe 290 295 300Thr Gly Glu
Arg Thr Glu Ser Leu Val Thr Arg Phe Leu Asn Ala Thr305
310 315 320Gly Ser Gln Trp Cys Ser Trp
Ser Val Ser Gln Ala Gly Leu Leu Ala 325
330 335Asp Phe Ile Pro Asp His Leu Leu Arg Leu Tyr Gln
Leu Leu Leu Phe 340 345 350Thr
Leu Pro Gly Thr Pro Val Phe Ser Tyr Gly Asp Glu Leu Gly Leu 355
360 365Gln Gly Ala Leu Pro Gly Gln Pro Ala
Lys Ala Pro Leu Met Pro Trp 370 375
380Asn Glu Ser Ser Ile Phe His Ile Pro Arg Pro Val Ser Leu Asn Met385
390 395 400Thr Val Lys Gly
Gln Asn Glu Asp Pro Gly Ser Leu Leu Thr Gln Phe 405
410 415Arg Arg Leu Ser Asp Leu Arg Gly Lys Glu
Arg Ser Leu Leu His Gly 420 425
430Asp Phe His Ala Leu Ser Ser Ser Pro Asp Leu Phe Ser Tyr Ile Arg
435 440 445His Trp Asp Gln Asn Glu Arg
Tyr Leu Val Val Leu Asn Phe Arg Asp 450 455
460Ser Gly Arg Ser Ala Arg Leu Gly Ala Ser Asn Leu Pro Ala Gly
Ile465 470 475 480Ser Leu
Pro Ala Ser Ala Lys Leu Leu Leu Ser Thr Asp Ser Ala Arg
485 490 495Gln Ser Arg Glu Glu Asp Thr
Ser Leu Lys Leu Glu Asn Leu Ser Leu 500 505
510Asn Pro Tyr Glu Gly Leu Leu Leu Gln Phe Pro Phe Val Ala
515 520 52541852DNAMus
sp.CDS(106)..(1683)4F2hc 4gctagcctca cggccacggg acgcctctct gaacggggat
ccaggcagga ttagagctgc 60ctcactgact acaggccgtg tcgtgtcacc gtttctgcag
gcacc atg agc cag gac 117
Met Ser Gln Asp 1acc
gaa gtg gac atg aaa gat gtg gag ctg aac gag cta gaa ccg gag 165Thr
Glu Val Asp Met Lys Asp Val Glu Leu Asn Glu Leu Glu Pro Glu5
10 15 20aag cag ccc atg aat gca
gcg gac ggg gcg gcg gcc ggg gag aag aac 213Lys Gln Pro Met Asn Ala
Ala Asp Gly Ala Ala Ala Gly Glu Lys Asn 25
30 35ggt ctg gtg aag atc aag gtg gcg gag gac gag acg
gag gcc ggg gtc 261Gly Leu Val Lys Ile Lys Val Ala Glu Asp Glu Thr
Glu Ala Gly Val 40 45 50aag
ttc acc ggc tta tcc aag gag gag cta ctg aag gta gcg ggc agc 309Lys
Phe Thr Gly Leu Ser Lys Glu Glu Leu Leu Lys Val Ala Gly Ser 55
60 65cct ggc tgg gtg cgc acc cgc tgg gcg
ctg ctg ctg ctc ttc tgg ctc 357Pro Gly Trp Val Arg Thr Arg Trp Ala
Leu Leu Leu Leu Phe Trp Leu 70 75
80ggt tgg ctg ggc atg ctg gcg ggc gcc gtg gtt atc atc gtt cgg gcg
405Gly Trp Leu Gly Met Leu Ala Gly Ala Val Val Ile Ile Val Arg Ala85
90 95 100ccg cgc tgc cgt
gag ctg cct gta cag agg tgg tgg cac aag ggc gcc 453Pro Arg Cys Arg
Glu Leu Pro Val Gln Arg Trp Trp His Lys Gly Ala 105
110 115ctc tac cgc atc ggc gac ctt cag gcc ttt
gta ggc cgg gat gcg gga 501Leu Tyr Arg Ile Gly Asp Leu Gln Ala Phe
Val Gly Arg Asp Ala Gly 120 125
130ggc ata gct ggt ctg aag agc cat ctg gag tac ttg agc acc ctg aag
549Gly Ile Ala Gly Leu Lys Ser His Leu Glu Tyr Leu Ser Thr Leu Lys
135 140 145gtg aag ggc ctg gtg tta ggc
cca att cac aag aac cag aag gat gaa 597Val Lys Gly Leu Val Leu Gly
Pro Ile His Lys Asn Gln Lys Asp Glu 150 155
160atc aat gaa acc gac ctg aaa cag att aat ccc act ttg ggc tcc cag
645Ile Asn Glu Thr Asp Leu Lys Gln Ile Asn Pro Thr Leu Gly Ser Gln165
170 175 180gaa gat ttt aaa
gac ctt cta caa agt gcc aag aaa aag agc att cac 693Glu Asp Phe Lys
Asp Leu Leu Gln Ser Ala Lys Lys Lys Ser Ile His 185
190 195atc att ttg gac ctc act ccc aac tac cag
ggc cag aat gcg tgg ttc 741Ile Ile Leu Asp Leu Thr Pro Asn Tyr Gln
Gly Gln Asn Ala Trp Phe 200 205
210ctc cct gct cag gct gac att gta gcc acc aaa atg aag gaa gct ctg
789Leu Pro Ala Gln Ala Asp Ile Val Ala Thr Lys Met Lys Glu Ala Leu
215 220 225agt tct tgg ttg cag gac ggt
gtg gat ggt ttc caa ttc cgg gat gtg 837Ser Ser Trp Leu Gln Asp Gly
Val Asp Gly Phe Gln Phe Arg Asp Val 230 235
240gga aag ctg atg aat gca ccc ttg tac ttg gct gag tgg cag aat atc
885Gly Lys Leu Met Asn Ala Pro Leu Tyr Leu Ala Glu Trp Gln Asn Ile245
250 255 260acc aag aac tta
agt gag gac agg ctt ttg att gca ggg act gag tcc 933Thr Lys Asn Leu
Ser Glu Asp Arg Leu Leu Ile Ala Gly Thr Glu Ser 265
270 275tct gac ctg cag caa att gtc aac ata ctt
gaa tcc acc agc gac ctg 981Ser Asp Leu Gln Gln Ile Val Asn Ile Leu
Glu Ser Thr Ser Asp Leu 280 285
290ctg ttg acc agc tcc tac ctg tca aat tcc act ttc act ggg gag cgt
1029Leu Leu Thr Ser Ser Tyr Leu Ser Asn Ser Thr Phe Thr Gly Glu Arg
295 300 305act gaa tcc cta gtc act agg
ttt ttg aat gcc act ggc agc caa tgg 1077Thr Glu Ser Leu Val Thr Arg
Phe Leu Asn Ala Thr Gly Ser Gln Trp 310 315
320tgc agc tgg agt gtg tcg caa gca gga ctc ctc gca gac ttt ata ccg
1125Cys Ser Trp Ser Val Ser Gln Ala Gly Leu Leu Ala Asp Phe Ile Pro325
330 335 340gac cat ctt ctc
cga ctc tac cag ctg ctg ctc ttc act ctg cca ggg 1173Asp His Leu Leu
Arg Leu Tyr Gln Leu Leu Leu Phe Thr Leu Pro Gly 345
350 355act cct gtt ttt agc tac ggg gat gag ctt
ggc ctt cag ggt gcc ctt 1221Thr Pro Val Phe Ser Tyr Gly Asp Glu Leu
Gly Leu Gln Gly Ala Leu 360 365
370cct gga cag cct gcg aag gcc cca ctc atg ccg tgg aat gag tcc agc
1269Pro Gly Gln Pro Ala Lys Ala Pro Leu Met Pro Trp Asn Glu Ser Ser
375 380 385atc ttt cac atc cca aga cct
gta agc ctc aac atg aca gtg aag ggc 1317Ile Phe His Ile Pro Arg Pro
Val Ser Leu Asn Met Thr Val Lys Gly 390 395
400cag aat gaa gac cct ggc tcc ctt ctt acc cag ttc cgg cgg ctg agt
1365Gln Asn Glu Asp Pro Gly Ser Leu Leu Thr Gln Phe Arg Arg Leu Ser405
410 415 420gac ctt cgg ggt
aag gag cgc tct ctg ttg cac ggt gac ttc cat gca 1413Asp Leu Arg Gly
Lys Glu Arg Ser Leu Leu His Gly Asp Phe His Ala 425
430 435ctg tct tcc tca cct gac ctc ttc tcc tac
ata cga cac tgg gac cag 1461Leu Ser Ser Ser Pro Asp Leu Phe Ser Tyr
Ile Arg His Trp Asp Gln 440 445
450aat gag cgt tac ctg gtg gtg ctc aac ttc cga gat tcg ggc cgg tca
1509Asn Glu Arg Tyr Leu Val Val Leu Asn Phe Arg Asp Ser Gly Arg Ser
455 460 465gcc agg cta ggg gcc tcc aac
ctc cct gct ggc ata agc ctg cca gcc 1557Ala Arg Leu Gly Ala Ser Asn
Leu Pro Ala Gly Ile Ser Leu Pro Ala 470 475
480agc gct aaa ctt ttg ctt agt acc gac agt gcc cgg caa agc cgt gag
1605Ser Ala Lys Leu Leu Leu Ser Thr Asp Ser Ala Arg Gln Ser Arg Glu485
490 495 500gag gac acc tcc
ctg aag ctg gaa aac ctg agc ctg aat cct tat gag 1653Glu Asp Thr Ser
Leu Lys Leu Glu Asn Leu Ser Leu Asn Pro Tyr Glu 505
510 515ggc ttg ctg tta cag ttc ccc ttt gtg gcc
tgatccttcc tatgcagaac 1703Gly Leu Leu Leu Gln Phe Pro Phe Val Ala
520 525ctaccaccct cctttgttct ccccaggcct
tttggattct agtcttcctc tccttgtttt 1763taaacttttg cagattacat acgaattctt
atactgggtg tttttgtctt caaataaaaa 1823catcacccct gcctcaaaaa aaaaaaaaa
18525685PRTMus sp.rBAT 5Met Asp Glu Asp
Lys Gly Lys Arg Asp Pro Ile Gln Met Ser Met Lys1 5
10 15Gly Cys Arg Thr Asn Asn Gly Phe Val Gln
Asn Glu Asp Ile Pro Glu 20 25
30Gln Asp Pro Asp Pro Gly Ser Arg Asp Thr Pro Gln Pro Asn Ala Val
35 40 45Ser Ile Pro Ala Pro Glu Glu Pro
His Leu Lys Ala Val Arg Pro Tyr 50 55
60Ala Gly Met Pro Lys Glu Val Leu Phe Gln Phe Ser Gly Gln Ala Arg65
70 75 80Tyr Arg Val Pro Arg
Glu Ile Leu Phe Trp Leu Thr Val Val Ser Val 85
90 95Phe Leu Leu Ile Gly Ala Thr Ile Ala Ile Ile
Val Ile Ser Pro Lys 100 105
110Cys Leu Asp Trp Trp Gln Ala Gly Pro Ile Tyr Gln Ile Tyr Pro Arg
115 120 125Ser Phe Lys Asp Ser Asp Lys
Asp Gly Asn Gly Asp Leu Lys Gly Ile 130 135
140Gln Glu Lys Leu Asp Tyr Ile Thr Ala Leu Asn Ile Lys Thr Leu
Trp145 150 155 160Ile Thr
Ser Phe Tyr Lys Ser Ile Phe Glu Asp Phe Arg Tyr Ala Val
165 170 175Glu Asp Ile Lys Glu Ile Asp
Pro Ile Phe Gly Thr Met Lys Asp Phe 180 185
190Glu Asn Leu Val Ala Ala Ile His Asp Lys Gly Leu Lys Leu
Ile Ile 195 200 205Asp Phe Ile Pro
Asn His Thr Ser Asp Lys His Pro Trp Phe Gln Ser 210
215 220Ser Arg Thr Arg Ser Gly Lys Tyr Thr Asp Tyr Tyr
Ile Trp His Asn225 230 235
240Cys Thr His Cys Gln Arg Val Pro Thr Pro Pro Asn Asn Trp Leu Ser
245 250 255Val Tyr Gly His Ser
Ser Trp His Phe Asp Glu Val Arg Glu Gln Cys 260
265 270Tyr Phe His Gln Phe Leu Arg Glu Gln Pro Asp Leu
Tyr Phe Arg Asn 275 280 285Pro Ala
Val Gln Glu Glu Ile Lys Glu Ile Ile Thr Phe Trp Leu Ser 290
295 300Lys Gly Val Asp Gly Phe Ser Phe Asp Ala Val
Lys Phe Leu Leu Glu305 310 315
320Ala Lys Asp Leu Arg Asn Glu Ile Gln Val Asn Thr Ser Gln Ile Pro
325 330 335Asp Thr Val Thr
His Tyr Ser Glu Leu Tyr His Asp Phe Thr Thr Thr 340
345 350Gln Val Gly Met His Asp Ile Val Arg Asp Phe
Arg Gln Thr Met Asn 355 360 365Gln
Tyr Ser Arg Glu Pro Gly Arg Tyr Arg Phe Met Gly Ala Glu Ala 370
375 380Ser Ala Glu Ser Ile Glu Arg Thr Met Met
Tyr Tyr Gly Leu Pro Phe385 390 395
400Ile Gln Glu Ala Asp Phe Pro Phe Asn Lys Tyr Phe Thr Thr Ile
Gly 405 410 415Thr Leu Ser
Gly His Thr Val Tyr Glu Val Ile Thr Ser Trp Met Glu 420
425 430Asn Met Pro Glu Gly Lys Trp Pro Asn Trp
Met Thr Gly Gly Pro Glu 435 440
445Thr Pro Arg Leu Thr Ser Arg Val Gly Ser Glu Tyr Val Asn Ala Met 450
455 460His Met Leu Leu Phe Thr Leu Pro
Gly Thr Pro Ile Thr Tyr Tyr Gly465 470
475 480Glu Glu Ile Gly Met Gly Asp Ile Ser Val Thr Asn
Phe Asn Glu Ser 485 490
495Tyr Asp Ser Thr Thr Leu Val Ser Lys Ser Pro Met Gln Trp Asp Asn
500 505 510Ser Ser Asn Ala Gly Phe
Thr Glu Ala Asn His Thr Trp Leu Pro Pro 515 520
525Asn Ser Asp Tyr His Thr Val Asn Val Asp Val Gln Lys Thr
Gln Pro 530 535 540Ser Ser Ala Leu Arg
Leu Tyr Gln Asp Leu Ser Leu Leu His Ala Thr545 550
555 560Glu Leu Val Leu Ser Arg Gly Trp Phe Cys
Leu Leu Arg Asp Asp Ser 565 570
575His Ser Val Val Tyr Thr Arg Glu Leu Asp Gly Ile Asp Asn Val Phe
580 585 590Leu Val Val Leu Asn
Phe Gly Glu Ser Ser Thr Val Leu Asn Leu Gln 595
600 605Gly Ile Ile Ser Asp Leu Pro Pro Glu Leu Arg Ile
Arg Leu Ser Thr 610 615 620Asn Ser Ala
Ser Lys Gly Ser Ala Val Asp Thr Arg Ala Ile Ser Leu625
630 635 640Glu Lys Gly Glu Gly Leu Val
Leu Glu His Ser Thr Lys Ala Pro Leu 645
650 655His Gln Gln Ala Ala Phe Arg Asp Arg Cys Phe Val
Ser Ser Arg Ala 660 665 670Cys
Tyr Ser Ser Ala Leu Asp Ile Leu Tyr Ser Ser Cys 675
680 68562287DNAMus sp.CDS(46)..(2100)rBAT 6gatccccctg
ctggaaagca ccaggaagag ctacacaggg tagac atg gat gag gac 57
Met Asp Glu Asp
1aaa ggc aag aga gac ccc atc caa atg agt atg
aag gga tgc cga acc 105Lys Gly Lys Arg Asp Pro Ile Gln Met Ser Met
Lys Gly Cys Arg Thr5 10 15
20aat aac ggg ttt gtc caa aat gaa gac att ccg gag cag gac cca gac
153Asn Asn Gly Phe Val Gln Asn Glu Asp Ile Pro Glu Gln Asp Pro Asp
25 30 35cca ggc tcc agg gac
acc cca cag ccc aac gcc gtg agt atc cct gct 201Pro Gly Ser Arg Asp
Thr Pro Gln Pro Asn Ala Val Ser Ile Pro Ala 40
45 50cca gag gag cct cac cta aag gcg gtg cgg ccc tat
gca ggg atg ccc 249Pro Glu Glu Pro His Leu Lys Ala Val Arg Pro Tyr
Ala Gly Met Pro 55 60 65aag gaa
gta ctc ttc cag ttc tcc ggc cag gct cgc tac cgg gtg ccc 297Lys Glu
Val Leu Phe Gln Phe Ser Gly Gln Ala Arg Tyr Arg Val Pro 70
75 80cga gag atc ctc ttc tgg ctc acc gtg gtt tcc
gtg ttc ctg ctc att 345Arg Glu Ile Leu Phe Trp Leu Thr Val Val Ser
Val Phe Leu Leu Ile85 90 95
100gga gcc acc ata gcc atc atc gtc atc tct cca aaa tgc ctt gac tgg
393Gly Ala Thr Ile Ala Ile Ile Val Ile Ser Pro Lys Cys Leu Asp Trp
105 110 115tgg caa gca ggt ccc
ata tac cag atc tac ccg agg tct ttt aag gac 441Trp Gln Ala Gly Pro
Ile Tyr Gln Ile Tyr Pro Arg Ser Phe Lys Asp 120
125 130agt gac aag gat ggg aat gga gac ctg aaa ggt atc
cag gag aag ctg 489Ser Asp Lys Asp Gly Asn Gly Asp Leu Lys Gly Ile
Gln Glu Lys Leu 135 140 145gac tat
atc act gct tta aac ata aag act ctt tgg atc act tcc ttt 537Asp Tyr
Ile Thr Ala Leu Asn Ile Lys Thr Leu Trp Ile Thr Ser Phe 150
155 160tat aaa tcg atc ttt gaa gac ttc aga tac gct
gtt gag gat atc aaa 585Tyr Lys Ser Ile Phe Glu Asp Phe Arg Tyr Ala
Val Glu Asp Ile Lys165 170 175
180gaa att gac cct att ttt gga aca atg aaa gat ttt gag aat ttg gtt
633Glu Ile Asp Pro Ile Phe Gly Thr Met Lys Asp Phe Glu Asn Leu Val
185 190 195gct gcc atc cat gac
aaa ggt tta aaa tta ata att gat ttc ata cca 681Ala Ala Ile His Asp
Lys Gly Leu Lys Leu Ile Ile Asp Phe Ile Pro 200
205 210aac cac act agt gac aaa cat cct tgg ttc caa tcg
agt agg aca cgg 729Asn His Thr Ser Asp Lys His Pro Trp Phe Gln Ser
Ser Arg Thr Arg 215 220 225agc gga
aaa tac acc gat tac tac atc tgg cac aac tgt acc cat tgt 777Ser Gly
Lys Tyr Thr Asp Tyr Tyr Ile Trp His Asn Cys Thr His Cys 230
235 240caa cgt gta ccc acc cct ccc aac aac tgg ctg
agt gtg tat gga cac 825Gln Arg Val Pro Thr Pro Pro Asn Asn Trp Leu
Ser Val Tyr Gly His245 250 255
260tcc agc tgg cac ttt gat gaa gta cga gag caa tgt tat ttt cac cag
873Ser Ser Trp His Phe Asp Glu Val Arg Glu Gln Cys Tyr Phe His Gln
265 270 275ttt ttg aga gag caa
cca gat tta tat ttc cga aat cct gct gtt caa 921Phe Leu Arg Glu Gln
Pro Asp Leu Tyr Phe Arg Asn Pro Ala Val Gln 280
285 290gag gaa ata aag gaa ata ata acg ttc tgg ctc tcg
aag ggt gtt gat 969Glu Glu Ile Lys Glu Ile Ile Thr Phe Trp Leu Ser
Lys Gly Val Asp 295 300 305ggg ttt
agt ttt gat gca gtt aaa ttt ctt ctg gaa gcg aag gat ctg 1017Gly Phe
Ser Phe Asp Ala Val Lys Phe Leu Leu Glu Ala Lys Asp Leu 310
315 320aga aat gaa atc caa gtg aat aca tcc caa att
ccg gac acg gtc acc 1065Arg Asn Glu Ile Gln Val Asn Thr Ser Gln Ile
Pro Asp Thr Val Thr325 330 335
340cac tac tca gag ctg tac cat gac ttc acc aca act cag gtg gga atg
1113His Tyr Ser Glu Leu Tyr His Asp Phe Thr Thr Thr Gln Val Gly Met
345 350 355cat gac atc gtc cga
gac ttc cgg cag acc atg aac cag tac agc agg 1161His Asp Ile Val Arg
Asp Phe Arg Gln Thr Met Asn Gln Tyr Ser Arg 360
365 370gag cct ggc aga tac cgg ttc atg ggg gcc gaa gcc
tca gct gag agc 1209Glu Pro Gly Arg Tyr Arg Phe Met Gly Ala Glu Ala
Ser Ala Glu Ser 375 380 385atc gag
agg acc atg atg tac tat ggc ttg cca ttt atc cag gaa gcc 1257Ile Glu
Arg Thr Met Met Tyr Tyr Gly Leu Pro Phe Ile Gln Glu Ala 390
395 400gac ttt cct ttc aac aag tac ttc acc aca ata
ggc act ctc tct ggg 1305Asp Phe Pro Phe Asn Lys Tyr Phe Thr Thr Ile
Gly Thr Leu Ser Gly405 410 415
420cat act gtc tat gaa gtt atc aca tcc tgg atg gaa aac atg cct gaa
1353His Thr Val Tyr Glu Val Ile Thr Ser Trp Met Glu Asn Met Pro Glu
425 430 435gga aaa tgg ccc aat
tgg atg act ggc gga ccg gag act cct cgg ctg 1401Gly Lys Trp Pro Asn
Trp Met Thr Gly Gly Pro Glu Thr Pro Arg Leu 440
445 450act tct cga gta ggg agt gag tat gtc aac gcc atg
cac atg ctc ctg 1449Thr Ser Arg Val Gly Ser Glu Tyr Val Asn Ala Met
His Met Leu Leu 455 460 465ttc aca
ctc ccg gga acg ccc atc act tac tat gga gag gaa atc ggg 1497Phe Thr
Leu Pro Gly Thr Pro Ile Thr Tyr Tyr Gly Glu Glu Ile Gly 470
475 480atg gga gac att tcc gtt aca aat ttc aac gag
agc tat gat agt act 1545Met Gly Asp Ile Ser Val Thr Asn Phe Asn Glu
Ser Tyr Asp Ser Thr485 490 495
500acc ctt gtc tcc aag tca ccg atg cag tgg gac aat agt tcc aat gct
1593Thr Leu Val Ser Lys Ser Pro Met Gln Trp Asp Asn Ser Ser Asn Ala
505 510 515ggg ttt act gag gcc
aac cac acc tgg cta cca cca aac tct gac tac 1641Gly Phe Thr Glu Ala
Asn His Thr Trp Leu Pro Pro Asn Ser Asp Tyr 520
525 530cac acc gtc aat gtg gat gtc caa aag acc cag ccg
agc tcc gca ctg 1689His Thr Val Asn Val Asp Val Gln Lys Thr Gln Pro
Ser Ser Ala Leu 535 540 545agg ctg
tat cag gat ctg agt cta ctc cat gcc aca gag ctg gtc ctc 1737Arg Leu
Tyr Gln Asp Leu Ser Leu Leu His Ala Thr Glu Leu Val Leu 550
555 560agc cgg ggc tgg ttt tgc ctc ttg aga gac gac
agt cac tct gtg gtg 1785Ser Arg Gly Trp Phe Cys Leu Leu Arg Asp Asp
Ser His Ser Val Val565 570 575
580tac aca aga gag ctg gac ggc ata gat aac gtc ttc ctc gtg gtt ctg
1833Tyr Thr Arg Glu Leu Asp Gly Ile Asp Asn Val Phe Leu Val Val Leu
585 590 595aat ttt gga gaa tca
tca act gtg cta aat cta cag ggg atc att tca 1881Asn Phe Gly Glu Ser
Ser Thr Val Leu Asn Leu Gln Gly Ile Ile Ser 600
605 610gat ctt cct cca gag ctg aga ata agg tta agt acc
aac tca gcc tcc 1929Asp Leu Pro Pro Glu Leu Arg Ile Arg Leu Ser Thr
Asn Ser Ala Ser 615 620 625aaa ggc
agt gct gtt gac acc cgt gcc att tct ctg gag aag gga gag 1977Lys Gly
Ser Ala Val Asp Thr Arg Ala Ile Ser Leu Glu Lys Gly Glu 630
635 640ggc ctg gtc ttg gag cac agc acg aag gct ccc
ctc cat cag cag gcc 2025Gly Leu Val Leu Glu His Ser Thr Lys Ala Pro
Leu His Gln Gln Ala645 650 655
660gct ttc aga gac aga tgc ttt gtt tcc agt cgg gcg tgc tac tcc agt
2073Ala Phe Arg Asp Arg Cys Phe Val Ser Ser Arg Ala Cys Tyr Ser Ser
665 670 675gca ctg gac atc ctc
tat agc tcg tgt tagggaggaa gctccctaag 2120Ala Leu Asp Ile Leu
Tyr Ser Ser Cys 680 685agatggccac ccagaacatc
acgtacgcac aggctgagca gactcatgaa tggcatcaat 2180tcttagatat ttctgtagca
cgatgcacgt tttttaaagt gtttaaagat tatgccaaat 2240actaaagcat ttaaatatga
aaaaaaaaaa aaaaaagcgg cgcgccg 2287714PRTArtificial
SequenceDescription of Artificial Sequence Synthetic partial peptide
7Cys Ile Pro Asp Val Ser Asp Asp His Ile His Glu Glu Ser1 5
10831DNAArtificial SequenceDescription of Artificial
Sequence Synthetic primer 8gcgcgaagct tacctatagg cagaaacatt c
31932DNAArtificial SequenceDescription of
Artificial Sequence Synthetic primer 9atatgcggcc gcactttctt
catgtatgtg gt 321033DNAArtificial
SequenceDescription of Artificial Sequence Synthetic primer
10atatgcggcc gcagatgagg acaaaggcaa gag
331136DNAArtificial SequenceDescription of Artificial Sequence Synthetic
primer 11gcgcgctcta gaaatgcttt agtatttggc ataatc
361233DNAArtificial SequenceDescription of Artificial Sequence
Synthetic primer 12atatgcggcc gcaagccagg acaccgaagt gga
331332DNAArtificial SequenceDescription of Artificial
Sequence Synthetic primer 13gcgctctaga catgaggcag gggtgatgtt tt
3214465PRTMus sp. 14Met Gln Leu Leu Arg Ala
Leu Gly Val Phe His Val Ser Met Ile Leu1 5
10 15Phe Ser Ala Thr Leu Gly Thr Gly Ile Phe Val Thr
Pro Lys Ala Val 20 25 30Leu
Lys Tyr Ser Ser Leu Asn Ile Pro Val Ser Leu Ser Ile Trp Ala 35
40 45Gly Cys Gly Leu Leu Ser Ile Met Ser
Ala Leu Cys Asn Ala Glu Ile 50 55
60Ala Thr Thr Tyr Pro Leu Ser Gly Ala Ser Tyr Tyr Phe Leu Lys Arg65
70 75 80Thr Leu Gly Ser Ser
Val Ala Phe Leu Ser Leu Trp Ile Lys Leu Phe 85
90 95Ala His Phe Leu Gly Ile Gly Ala Gln Cys Leu
Leu Ile Ala Thr Ser 100 105
110Val Ile Gln Cys Phe Tyr Ser Gly Cys Pro Ala Pro Glu Leu Pro Thr
115 120 125Lys Cys Leu Ala Leu Ala Ile
Leu Trp Ser Phe Gly Ile Val Ser Ala 130 135
140Arg Gly Ile Lys Thr Val Ala Trp Phe Asn Thr Val Ser Ser Phe
Ile145 150 155 160Lys Leu
Ser Val Leu Cys Leu Ile Ser Leu Thr Val Leu Leu Val Asn
165 170 175Gly Lys Lys Glu Asn Val Ser
Arg Phe Glu Asn Ala Leu Asp Ala Glu 180 185
190Leu Pro Asn Ala Ser Gln Ile Ala Asp Ala Ile Leu Gln Val
Ser Tyr 195 200 205Ser Tyr Leu Gly
Ser Ser Val Leu Ile Val Ile Ala Gly Glu Ile Lys 210
215 220Arg Pro Thr Glu Thr Ile Pro Lys Thr Leu Ile Tyr
Gly Ile Ser Ile225 230 235
240Val Thr Val Leu Tyr Leu Leu Thr Asn Ile Ser Tyr Leu Ala Val Leu
245 250 255Thr Ser Gln Glu Ile
Ile Phe Ser Asp Ser Val Gly Val Thr Trp Met 260
265 270Asn Arg Val Phe Pro Ser Ile Gln Trp Ile Ser Ser
Phe Leu Ile Ser 275 280 285Ala Phe
Leu Leu Gly Ser Val Ser Cys Gly Ile Val Ser Ala Ser Arg 290
295 300Val Phe Tyr Ser Ala Ser Gly Glu Gly Glu Phe
Pro Ser Ile Tyr Ser305 310 315
320Met Leu Asn Asp His His Ser Pro Ala Val Ala Asp Ile Gln Ile Val
325 330 335Ile Leu Ser Ser
Val Ala Ile Ile Ser Ser Ser Ile Ile Tyr Leu Val 340
345 350Lys Tyr Val Ser Leu Gly Ser Phe Cys Ile Asn
Leu Leu Gln Met Ile 355 360 365Gly
Leu Leu Lys Ile Arg Tyr Gln Asn Pro Asp Ile Pro Arg Pro Tyr 370
375 380Lys Val Trp Leu Pro Phe Ile Phe Gly Ser
Ile Ala Leu Ser Leu Phe385 390 395
400Leu Ile Phe Thr Pro Val Ile Gln Ser Pro Ser Ile Glu His Val
Tyr 405 410 415Gln Val Val
Phe Leu Phe Cys Gly Phe Leu Cys Tyr Trp Leu Gln Ala 420
425 430Asn Leu Asn Gly His Ala Thr Cys Phe Asp
Thr Ile Thr Cys Tyr Cys 435 440
445Gln Leu Leu Phe Asn Ile Ser Pro Ser Glu Asp Pro Glu Glu Gln Lys 450
455 460Asn46515512PRTMus sp. 15Met Ala
Val Ala Gly Ala Lys Arg Arg Ala Val Ala Ala Pro Ala Thr1 5
10 15Thr Ala Ala Glu Glu Glu Arg Gln
Ala Arg Glu Lys Met Leu Glu Ala 20 25
30Arg Arg Gly Asp Gly Ala Asp Pro Glu Gly Glu Gly Val Thr Leu
Gln 35 40 45Arg Asn Ile Thr Leu
Ile Asn Gly Val Ala Ile Ile Val Gly Thr Ile 50 55
60Ile Gly Ser Gly Ile Phe Val Thr Pro Thr Gly Val Leu Lys
Glu Ala65 70 75 80Gly
Ser Pro Gly Leu Ser Leu Val Val Trp Ala Val Cys Gly Val Phe
85 90 95Ser Ile Val Gly Ala Leu Cys
Tyr Ala Glu Leu Gly Thr Thr Ile Ser 100 105
110Lys Ser Gly Gly Asp Tyr Ala Tyr Met Leu Glu Val Tyr Gly
Ser Leu 115 120 125Pro Ala Phe Leu
Lys Leu Trp Ile Glu Leu Leu Ile Ile Arg Pro Ser 130
135 140Ser Gln Tyr Ile Val Ala Leu Val Phe Ala Thr Tyr
Leu Leu Lys Pro145 150 155
160Val Phe Pro Thr Cys Pro Val Pro Glu Glu Ala Ala Lys Leu Val Ala
165 170 175Cys Leu Cys Val Leu
Leu Leu Thr Ala Val Asn Cys Tyr Ser Val Lys 180
185 190Ala Ala Thr Arg Val Gln Asp Ala Phe Ala Ala Ala
Lys Leu Leu Ala 195 200 205Leu Ala
Leu Ile Ile Leu Leu Gly Phe Ile Gln Met Gly Lys Asp Ile 210
215 220Gly Gln Gly Asp Ala Ser Asn Leu His Gln Lys
Leu Ser Phe Glu Gly225 230 235
240Thr Asn Leu Asp Val Gly Asn Ile Val Leu Ala Leu Tyr Ser Gly Leu
245 250 255Phe Ala Tyr Gly
Gly Trp Asn Tyr Leu Asn Phe Val Thr Glu Glu Met 260
265 270Ile Asn Pro Tyr Arg Asn Leu Pro Leu Ala Ile
Ile Ile Ser Leu Pro 275 280 285Ile
Val Thr Leu Val Tyr Val Leu Thr Asn Leu Ala Tyr Phe Thr Thr 290
295 300Leu Ser Thr Asn Gln Met Leu Thr Ser Glu
Ala Val Ala Val Asp Phe305 310 315
320Gly Asn Tyr His Leu Gly Val Met Ser Trp Ile Ile Pro Val Phe
Val 325 330 335Gly Leu Ser
Cys Phe Gly Ser Val Asn Gly Ser Leu Phe Thr Ser Ser 340
345 350Arg Leu Phe Phe Val Gly Ser Arg Glu Gly
His Leu Pro Ser Ile Leu 355 360
365Ser Met Ile His Pro Gln Leu Leu Thr Pro Val Pro Ser Leu Val Phe 370
375 380Thr Cys Val Met Thr Leu Met Tyr
Ala Phe Ser Arg Asp Ile Phe Ser385 390
395 400Ile Ile Asn Phe Phe Ser Phe Phe Asn Trp Leu Cys
Val Ala Leu Ala 405 410
415Ile Ile Gly Met Met Trp Leu Arg Phe Lys Lys Pro Glu Leu Glu Arg
420 425 430Pro Ile Lys Val Asn Leu
Ala Leu Pro Val Phe Phe Ile Leu Ala Cys 435 440
445Leu Phe Leu Ile Ala Val Ser Phe Trp Lys Thr Pro Leu Glu
Cys Gly 450 455 460Ile Gly Phe Ala Ile
Ile Leu Ser Gly Leu Pro Val Tyr Phe Phe Gly465 470
475 480Val Trp Trp Lys Asn Lys Pro Lys Trp Ile
Leu Gln Val Ile Phe Ser 485 490
495Val Thr Val Leu Cys Gln Lys Leu Met Gln Val Val Pro Gln Glu Thr
500 505 51016512PRTRattus sp.
16Met Val Ala Ser Thr Lys Tyr Glu Val Ala Ala Gln Asn Glu Ala Asp1
5 10 15Glu Ala Asp Gly Ser Ala
Gln Gly Asp Gly Ala Gly Pro Ala Ala Glu 20 25
30Gln Val Lys Leu Lys Lys Glu Ile Ser Leu Leu Asn Gly
Val Cys Leu 35 40 45Ile Val Gly
Asn Met Ile Gly Ser Gly Ile Phe Val Ser Pro Lys Gly 50
55 60Val Leu Met Tyr Ser Ala Ser Phe Gly Leu Ser Leu
Val Ile Trp Ala65 70 75
80Val Gly Gly Ile Phe Ser Val Phe Gly Ala Leu Cys Tyr Ala Glu Leu
85 90 95Gly Thr Thr Ile Lys Lys
Ser Gly Ala Ser Tyr Ala Tyr Ile Leu Glu 100
105 110Ala Phe Gly Gly Phe Leu Ala Phe Ile Arg Leu Trp
Thr Ser Leu Leu 115 120 125Ile Ile
Glu Pro Thr Ser Gln Ala Val Ile Ala Ile Thr Phe Ala Asn 130
135 140Tyr Met Val Gln Pro Leu Phe Pro Ser Cys Gly
Ala Pro Tyr Ala Ala145 150 155
160Gly Arg Leu Leu Ala Ala Ala Cys Ile Cys Leu Leu Thr Phe Ile Asn
165 170 175Cys Ala Tyr Val
Lys Trp Gly Thr Leu Val Gln Asp Ile Phe Thr Tyr 180
185 190Ala Lys Val Leu Ala Leu Ile Ala Val Ile Ile
Ala Gly Ile Val Arg 195 200 205Leu
Gly Gln Gly Ala Thr Thr Asn Phe Glu Asp Ser Phe Glu Gly Ser 210
215 220Ser Phe Ala Met Gly Asp Ile Ala Leu Ala
Leu Tyr Ser Ala Leu Phe225 230 235
240Ser Tyr Ser Gly Trp Asp Thr Leu Asn Tyr Val Thr Glu Glu Ile
Arg 245 250 255Asn Pro Glu
Arg Asn Leu Pro Leu Ser Ile Gly Ile Ser Met Pro Ile 260
265 270Val Thr Ile Ile Tyr Leu Leu Thr Asn Val
Ala Tyr Tyr Ser Val Leu 275 280
285Asp Ile Lys Asp Ile Leu Ala Ser Asp Ala Val Ala Val Thr Phe Ala 290
295 300Asp Gln Ile Phe Gly Ile Phe Asn
Trp Thr Ile Pro Leu Ala Val Ala305 310
315 320Leu Ser Cys Phe Gly Gly Leu Asn Ala Ser Ile Val
Ala Ala Ser Arg 325 330
335Leu Leu Phe Val Gly Ser Arg Glu Gly His Leu Pro Asp Ala Ile Cys
340 345 350Met Ile His Val Glu Arg
Phe Thr Pro Val Pro Ser Leu Leu Phe Asn 355 360
365Gly Ile Leu Ala Leu Val Tyr Leu Cys Val Glu Asp Ile Phe
Gln Leu 370 375 380Ile Asn Tyr Tyr Ser
Phe Ser Tyr Trp Phe Phe Val Gly Leu Ser Ile385 390
395 400Val Gly Gln Leu Tyr Leu Arg Trp Lys Glu
Pro Asp Arg Pro Arg Pro 405 410
415Leu Lys Leu Ser Leu Phe Phe Pro Ile Val Phe Cys Leu Cys Thr Ile
420 425 430Phe Leu Val Ala Val
Pro Leu Tyr Ser Asp Thr Ile Asn Ser Leu Ile 435
440 445Gly Ile Gly Ile Ala Leu Ser Gly Leu Pro Phe Tyr
Phe Leu Ile Ile 450 455 460Arg Val Pro
Glu His Lys Arg Pro Leu Cys Leu Arg Arg Ile Val Ala465
470 475 480Ser Thr Thr Arg Tyr Leu Gln
Ile Ile Cys Met Ser Val Ala Ala Glu 485
490 495Met Asp Leu Glu Asp Gly Glu Leu Pro Lys Gln Gly
Pro Lys Ser Lys 500 505
51017502PRTRattus sp. 17Met Val Arg Lys Pro Val Val Ala Thr Ile Ser Lys
Gly Gly Tyr Leu1 5 10
15Gln Gly Asn Met Ser Gly Arg Leu Pro Ser Met Gly Asp Gln Glu Pro
20 25 30Pro Gly Gln Glu Lys Val Val
Leu Lys Lys Lys Ile Thr Leu Leu Arg 35 40
45Gly Val Ser Ile Ile Ile Gly Thr Val Ile Gly Ser Gly Ile Phe
Ile 50 55 60Ser Pro Lys Gly Ile Leu
Gln Asn Thr Gly Ser Val Gly Met Ser Leu65 70
75 80Val Phe Trp Ser Ala Cys Gly Val Leu Ser Leu
Phe Gly Ala Leu Ser 85 90
95Tyr Ala Glu Leu Gly Thr Ser Ile Lys Lys Ser Gly Gly His Tyr Thr
100 105 110Tyr Ile Leu Glu Val Phe
Gly Pro Leu Leu Ala Phe Val Arg Val Trp 115 120
125Val Glu Leu Leu Val Ile Arg Pro Gly Ala Thr Ala Val Ile
Ser Leu 130 135 140Ala Phe Gly Arg Tyr
Ile Leu Glu Pro Phe Phe Ile Gln Cys Glu Ile145 150
155 160Pro Glu Leu Ala Ile Lys Leu Val Thr Ala
Val Gly Ile Thr Val Val 165 170
175Met Val Leu Asn Ser Thr Ser Val Ser Trp Ser Ala Arg Ile Gln Ile
180 185 190Phe Leu Ile Phe Cys
Lys Leu Thr Ala Ile Leu Ile Ile Ile Val Pro 195
200 205Gly Val Ile Gln Leu Ile Lys Gly Gln Thr His His
Phe Lys Asp Ala 210 215 220Phe Ser Gly
Arg Asp Thr Ser Leu Met Gly Leu Pro Leu Ala Phe Tyr225
230 235 240Tyr Gly Met Tyr Ala Tyr Ala
Gly Trp Phe Tyr Leu Asn Phe Ile Thr 245
250 255Glu Glu Val Asp Asn Pro Glu Lys Thr Ile Pro Leu
Ala Ile Cys Ile 260 265 270Ser
Met Ala Ile Ile Thr Val Gly Tyr Val Leu Thr Asn Val Ala Tyr 275
280 285Phe Ile Thr Ile Ser Ala Glu Glu Leu
Leu Gln Ser Ser Ala Val Ala 290 295
300Val Ile Phe Ser Glu Arg Leu Leu Gly Lys Phe Ser Leu Ala Val Pro305
310 315 320Ile Phe Val Ala
Leu Ser Cys Phe Gly Ser Met Asn Gly Gly Val Phe 325
330 335Ala Val Ser Arg Leu Phe Tyr Val Ala Ser
Arg Glu Gly His Leu Pro 340 345
350Glu Ile Leu Ser Met Ile His Val His Lys His Thr Pro Leu Pro Ala
355 360 365Val Ile Val Leu His Pro Leu
Thr Met Val Met Leu Phe Ser Gly Asp 370 375
380Leu Tyr Ser Leu Leu Asn Phe Leu Ser Phe Ala Arg Trp Leu Phe
Met385 390 395 400Gly Leu
Ala Val Ala Gly Leu Ile Tyr Leu Arg Tyr Lys Arg Pro Asp
405 410 415Met His Arg Pro Phe Lys Val
Pro Leu Phe Ile Pro Ala Leu Phe Ser 420 425
430Phe Thr Cys Leu Phe Met Val Val Leu Ser Leu Tyr Ser Asp
Pro Phe 435 440 445Ser Thr Gly Val
Gly Phe Leu Ile Thr Leu Thr Gly Val Pro Ala Tyr 450
455 460Tyr Leu Phe Ile Val Trp Asp Lys Lys Pro Lys Trp
Phe Arg Arg Leu465 470 475
480Ser Asp Arg Ile Thr Arg Thr Leu Gln Ile Ile Leu Glu Val Val Pro
485 490 495Glu Asp Ser Lys Glu
Leu 50018487PRTRattus sp. 18Met Glu Glu Thr Ser Pro Arg Arg
Arg Arg Glu Asp Glu Lys Ser Val1 5 10
15His Ser Thr Glu Pro Lys Thr Thr Ser Leu Gln Lys Glu Val
Gly Leu 20 25 30Leu Ser Gly
Ile Cys Ile Ile Val Gly Thr Ile Ile Gly Ser Gly Ile 35
40 45Phe Ile Ser Pro Lys Ser Val Leu Ala Asn Thr
Glu Ser Val Gly Pro 50 55 60Cys Leu
Ile Ile Trp Ala Ala Cys Gly Val Leu Ala Thr Leu Gly Ala65
70 75 80Leu Cys Phe Ala Glu Leu Gly
Thr Met Ile Thr Lys Ser Gly Gly Glu 85 90
95Tyr Pro Tyr Leu Met Glu Ala Phe Gly Pro Ile Pro Ala
Tyr Leu Phe 100 105 110Ser Trp
Thr Ser Leu Ile Val Met Lys Pro Ser Ser Phe Ala Ile Ile 115
120 125Cys Leu Ser Phe Ser Glu Tyr Tyr Cys Ala
Ala Phe Tyr Leu Gly Cys 130 135 140Arg
Pro Pro Ala Val Val Val Lys Leu Leu Ala Ala Ala Ala Ile Leu145
150 155 160Leu Ile Thr Thr Val Asn
Ala Leu Ser Val Arg Leu Gly Ser Tyr Val 165
170 175Gln Asn Val Phe Thr Ala Ala Lys Leu Val Ile Val
Ala Ile Ile Ile 180 185 190Ile
Ser Gly Leu Val Leu Leu Ala Gln Gly Asn Val Lys Asn Phe Gln 195
200 205Asn Ser Phe Glu Gly Ser Gln Thr Ser
Val Gly Ser Ile Ser Leu Ala 210 215
220Phe Tyr Asn Gly Leu Trp Ala Tyr Asp Gly Trp Asn Gln Leu Asn Tyr225
230 235 240Ile Thr Glu Glu
Leu Arg Asn Pro Tyr Arg Asn Leu Pro Met Ala Ile 245
250 255Val Ile Gly Ile Pro Leu Val Thr Val Cys
Tyr Ile Leu Met Asn Ile 260 265
270Ala Tyr Phe Thr Val Met Thr Pro Thr Glu Leu Leu Gln Ser Gln Ala
275 280 285Val Ala Val Thr Phe Gly Asp
Arg Val Leu Tyr Pro Ala Ser Trp Val 290 295
300Val Pro Leu Phe Val Ala Phe Ser Thr Ile Gly Ala Ala Asn Gly
Thr305 310 315 320Cys Phe
Thr Ala Gly Arg Leu Ile Tyr Val Ala Gly Arg Glu Gly His
325 330 335Met Leu Lys Val Leu Ser Tyr
Ile Ser Val Lys Arg Leu Thr Pro Ala 340 345
350Pro Ala Leu Val Phe Tyr Gly Ile Ile Ala Ile Ile Tyr Ile
Ile Pro 355 360 365Gly Asp Ile Asn
Ser Leu Val Asn Tyr Phe Ser Phe Ala Ala Trp Leu 370
375 380Phe Tyr Gly Met Thr Ile Leu Gly Leu Val Val Met
Arg Phe Thr Arg385 390 395
400Lys Asp Leu Glu Arg Pro Ile Lys Val Pro Ile Phe Ile Pro Ile Ile
405 410 415Val Ile Leu Val Ser
Val Phe Leu Ile Leu Ala Pro Ile Ile Ser Ser 420
425 430Pro Ala Trp Glu Tyr Leu Tyr Cys Val Leu Phe Ile
Leu Ser Gly Leu 435 440 445Ile Phe
Tyr Phe Leu Phe Val His Tyr Lys Phe Arg Trp Ala Gln Lys 450
455 460Ile Ser Arg Pro Ile Thr Lys His Leu Gln Met
Leu Met Glu Val Val465 470 475
480Pro Pro Glu Lys Asp Pro Glu 4851927DNAArtificial
SequenceDescription of Artificial Sequence Synthetic oligonucleotide
19gaa gaa agt gcg gcc gca agc cag gac
27Glu Glu Ser Ala Ala Ala Ser Gln Asp1 5209PRTArtificial
SequenceDescription of Artificial Sequence Synthetic peptide 20Glu
Glu Ser Ala Ala Ala Ser Gln Asp1 52127DNAArtificial
SequenceDescription of Artificial Sequence Synthetic oligonucleotide
21gaa gaa agt gcg gcc gca gat gag gac
27Glu Glu Ser Ala Ala Ala Asp Glu Asp1 5229PRTArtificial
SequenceDescription of Artificial Sequence Synthetic peptide 22Glu
Glu Ser Ala Ala Ala Asp Glu Asp1 5
User Contributions:
Comment about this patent or add new information about this topic:
| People who visited this patent also read: | |
| Patent application number | Title |
|---|---|
| 20100217349 | Systems and Methods of Powered Muscle Stimulation Using an Energy Guidance Field |
| 20100217348 | Systems for Monitoring a Patient's Neurological Disease State |
| 20100217347 | NEUROSTIMULATION FOR THE TREATMENT OF PULMONARY DISORDERS |
| 20100217346 | METHOD AND APPARATUS FOR GASTROINTESTINAL STIMULATION VIA THE LYMPHATIC SYSTEM |
| 20100217345 | MICROPHONE FOR REMOTE HEALTH SENSING |