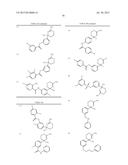
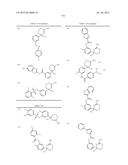
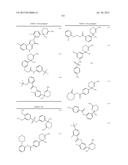
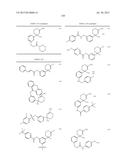
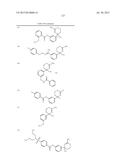
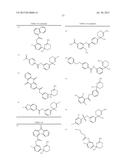
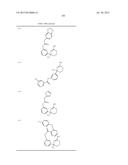
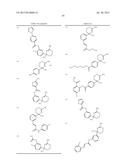
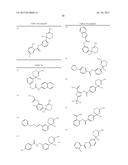
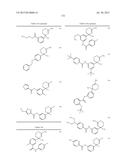
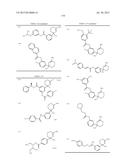
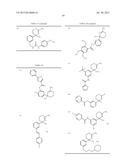
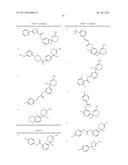
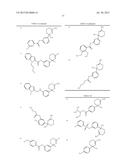
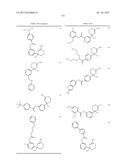
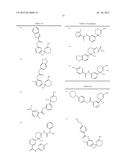
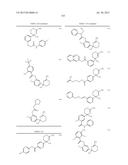
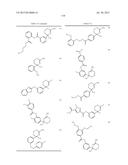
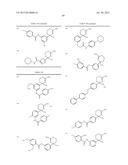
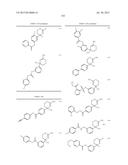
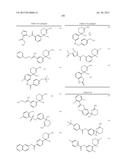
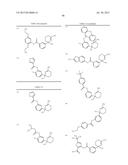
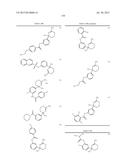
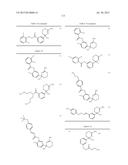
Patent application title: AMINODIHYDROTHIAZINE DERIVATIVES
Inventors:
Naotake Kobayashi (Osaka-Shi, JP)
Kazuo Ueda (Osaka-Shi, JP)
Naohiro Itoh (Koka-Shi, JP)
Shinji Suzuki (Osaka-Shi, JP)
Gaku Sakaguchi (Koka-Shi, JP)
Akira Kato (Koka-Shi, JP)
Akira Yukimasa (Osaka-Shi, JP)
Akihiro Hori (Osaka-Shi, JP)
Yuji Koriyama (Osaka-Shi, JP)
Hidekazu Haraguchi (Osaka-Shi, JP)
Ken Yasui (Osaka-Shi, JP)
Yasuhiko Kanda (Osaka-Shi, JP)
IPC8 Class: AC07D41714FI
USPC Class:
Class name:
Publication date: 2015-07-30
Patent application number: 20150210680
Abstract:
A composition having BACE 1 inhibitory activity containing a compound
represented by the general formula (I):
##STR00001## wherein ring A is an optionally substituted carbocyclic
group or an optionally substituted heterocyclic group; E is lower
alkylene; X is S, O, or NR1; R1 is a hydrogen atom or lower
alkyl; R2a, R2b, R3a, R3b, R4a and R4b is
each independently a hydrogen atom, halogen, or hydroxy etc.; n and m are
each independently an integer of 0 to 3; n+m is an integer of 0 to 3;
R5 is a hydrogen atom or substituted lower alkyl; its
pharmaceutically acceptable salt, or a solvate thereof.Claims:
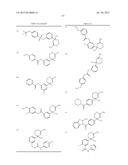
1. A compound represented by formula (I): ##STR01369## wherein ring A
is an optionally substituted carbocyclic group, the carbocyclic group
being substituted optionally with at least one selected from the group
consisting of (A) the substituent α, the substituent α being
at least one selected from the group consisting of halogen, hydroxy,
lower alkoxy, hydroxy lower alkoxy, lower alkoxy lower alkoxy, acyl,
acyloxy, carboxy, lower alkoxycarbonyl, amino, acylamino, lower
alkylamino, lower alkylthio, carbamoyl, lower alkylcarbamoyl, hydroxy
lower alkylcarbamoyl, sulfamoyl, lower alkylsulfamoyl, lower
alkylsulfinyl, cyano, nitro, aryl, and heterocyclic group; (B) lower
alkyl optionally substituted with one or more substituents selected from
the group of substituent α; (C) amino lower alkyl substituted with
one or more substituents selected from the group of substituent α;
(D) hydroxyimino lower alkyl; (E) lower alkoxyimino lower alkyl; (F)
lower alkenyl optionally substituted with one or more substituents
selected from the group of substituent α; (G) lower alkynyl
optionally substituted with one or more substituents selected from the
group of substituent α; (H) lower alkoxy optionally substituted
with one or more substituents selected from the group of substituent
α; (I) lower alkylthio optionally substituted with one or more
substituents selected from the group of substituent α; (J) lower
alkylamino substituted with one or more substituents selected from the
group of substituent α; (K) lower alkylsulfonyl optionally
substituted with one or more substituents selected from the group of
substituent α; (L) aryl lower alkoxycarbonyl optionally substituted
with one or more substituents selected from the group of substituent
α, azido, and lower alkyl; (M) acyl substituted with one or more
substituents selected from the group of substituent α; and the ring
part of the carbocyclic carbonyl and the heterocyclic carbonyl is
substituted optionally with at least one selected from the group of lower
alkyl; the substituent group α; and lower alkyl substituted with at
least one selected from the group of the substituent α; (N)
cycloalkyl optionally substituted with one or more substituents selected
from the group of substituent α, azido, and lower alkyl; (O) lower
alkylsulfinyl optionally substituted with one or more substituents
selected from the group of substituent α; (P) sulfamoyl; (Q) aryl
optionally substituted with one or more substituents selected from the
group of substituent α, azido, and lower alkyl; (R) heterocyclic
group optionally substituted with one or more substituents selected from
the group of substituent α, azido, and lower alkyl; (S) aryloxy
optionally substituted with one or more substituents selected from the
group of substituent α, azido, and lower alkyl; (T) heterocyclicoxy
optionally substituted with one or more substituents selected from the
group of substituent α, azido, and lower alkyl; (U) arylthio
optionally substituted with one or more substituents selected from the
group of substituent α, azido, and lower alkyl; (V)
heterocyclicthio optionally substituted with one or more substituents
selected from the group of substituent α, azido, and lower alkyl;
(W) arylamino optionally substituted with one or more substituents
selected from the group of substituent α, azido, and lower alkyl;
(X) heterocyclicamino optionally substituted with one or more
substituents selected from the group of substituent α, azido, and
lower alkyl; (Y) aryl lower alkylamino optionally substituted with one or
more substituents selected from the group of substituent α, azido,
and lower alkyl; (Z) heterocyclic lower alkylamino optionally substituted
with one or more substituents selected from the group of substituent
α, azido, and lower alkyl; (AA) lower alkyl sulfamoyl optionally
substituted with one or more substituents selected from the group of
substituent α; (AB) aryl sulfamoyl optionally substituted with one
or more substituents selected from the group of substituent α,
azido, and lower alkyl; (AC) heterocyclic sulfamoyl optionally
substituted with one or more substituents selected from the group of
substituent α, azido, and lower alkyl; (AD) arylsulfonyl
substituted optionally with one or more substituents selected from the
group of substituent α, azido, and lower alkyl; (AE) heterocyclic
sulfonyl optionally substituted with one or more substituents selected
from the group of substituent α, azido, and lower alkyl; (AF) aryl
carbamoyl optionally substituted with one or more substituents selected
from the group of substituent α, azido, and lower alkyl; (AG)
heterocyclic carbamoyl optionally substituted with one or more
substituents selected from the group of substituent α, azido, and
lower alkyl; (AH) aryl lower alkylcarbamoyl optionally substituted with
one or more substituents selected from the group of substituent α,
azido, and lower alkyl; (AI) heterocyclic lower alkylcarbamoyl optionally
substituted with one or more substituents selected from the group of
substituent α, azido, and lower alky; (AJ) aryloxycarbonyl
optionally substituted with one or more substituents selected from the
group of substituent α, azido, and lower alkyl; (AK)
heterocyclicoxycarbonyl optionally substituted with one or more
substituents selected from the group of substituent α, azido, and
lower alkyl; (AL) lower alkylenedioxy substituted optionally with
halogen; (AM) oxo; (AN) azido; and (AO) one of the following formulae:
##STR01370## ##STR01371## wherein Q1, Q2 and Q3 are each
independently a bond, optionally substituted lower alkylene or optionally
substituted lower alkenylene, lower alkylene and lower alkenylene being
substituted optionally with the substituent α; Q4 is
optionally substituted lower alkylene or optionally substituted lower
alkenylene, lower alkylene and lower alkenylene being substituted
optionally with the substituent α; W1 and W2 are each
independently O or S; W3 is O, S or NR12; R12 is a
hydrogen atom, lower alkyl, hydroxy lower alkyl, lower alkoxy lower
alkyl, lower alkoxycarbonyl lower alkyl, carbocyclic group lower alkyl or
acyl; R14 is a hydrogen atom or lower alkyl; ring B is an optionally
substituted carbocyclic group or an optionally substituted heterocyclic
group, the carbocyclic group and the heterocyclic group being substituted
optionally with at least one selected from the group consisting of (a)
the substituent α; (b) lower alkyl optionally substituted with the
substituent α; (c) amino lower alky substituted with the
substituent α; (d) hydroxyimino lower alkyl; (e) lower alkoxyimino
lower alkyl; (f) lower alkenyl optionally substituted with the
substituent α; (g) lower alkynyl optionally substituted with the
substituent α; (h) lower alkoxy optionally substituted with the
substituent α; (i) lower alkylthio optionally substituted with the
substituent α; (i) lower alkylamino substituted with the
substituent α; (k) lower alkylsulfonyl optionally substituted with
the substituent α; (l) aryl lower alkoxycarbonyl optionally
substituted with one or more substituents selected from the group of
substituent α and lower alkyl; (m) acyl substituted with the
substituent α; and the ring part of the carbocyclic carbonyl and
the heterocyclic carbonyl is substituted optionally with at least one
selected from the group of lower alkyl; the substituent group α;
and lower alkyl substituted with at least one selected from the group of
the substituent α, (n) lower alkylsulfonyl optionally substituted
with the substituent α; (o) sulfamoyl; (p) lower alkyl sulfamoyl
optionally substituted with one or more substituents selected from the
group of substituent α; (q) cycloalkyl optionally substituted with
one or more substituents selected from the group of substituent α,
azido and lower alkyl; (r) aryl optionally substituted with one or more
substituents selected from the group of substituent α, azido and
lower alkyl; (s) heterocyclic group optionally substituted with one or
more substituents selected from the group of substituent α, azido
and lower alkyl; (t) aryloxy optionally substituted with one or more
substituents selected from the group of substituent α, azido and
lower alkyl; (u) heterocyclicoxy optionally substituted with one or more
substituents selected from the group of substituent α, azido and
lower alkyl; (v) arylthio optionally substituted with one or more
substituents selected from the group of substituent α, azido and
lower alkyl; (w) heterocyclic thio optionally substituted with one or
more substituents selected from the group of substituent α, azido
and lower alkyl; (x) arylamino optionally substituted with one or more
substituents selected from the group of substituent α, azido and
lower alkyl; (y) heterocyclic amino optionally substituted with one or
more substituents selected from the group of substituent α, azido
and lower alkyl; (z) aryl lower alkylamino optionally substituted with
one or more substituents selected from the group of substituent α,
azido and lower alkyl; (aa) heterocyclic lower alkylamino optionally
substituted with one or more substituents selected from the group of
substituent α, azido and lower alkyl; (ab) arylsulfamoyl optionally
substituted with one or more substituents selected from the group of
substituent α, azido and lower alkyl; (ac) heterocyclic sulfamoyl
optionally substituted with one or more substituents selected from the
group of substituent α, azido and lower alkyl; (ad) arylsulfonyl
optionally substituted with one or more substituents selected from the
group of substituent α, azido and lower alkyl; (ae) heterocyclic
sulfonyl optionally substituted with one or more substituents selected
from the group of substituent α, azido and lower alkyl; (af)
arylcarbamoyl optionally substituted with one or more substituents
selected from the group of substituent α, azido and lower alkyl;
(ag) heterocyclic carbamoyl optionally substituted with one or more
substituents selected from the group of substituent α, azido and
lower alkyl; (ah) aryl lower alkylcarbamoyl optionally substituted with
one or more substituents selected from the group of substituent α,
azido and lower alkyl; (ai) heterocyclic lower alkylcarbamoyl optionally
substituted with one or more substituents selected from the group of
substituent α, azido and lower alkyl; (aj) aryloxy carbonyl
optionally substituted with one or more substituents selected from the
group of substituent α, azido and lower alkyl; (ak)
heterocyclicoxycarbonyl optionally substituted with one or more
substituents selected from the group of substituent α, azido and
lower alkyl; (al) lower alkylenedioxy substituted optionally with
halogen; and (am) oxo; Alk2 is optionally substituted lower alkyl,
lower alkyl being substituted optionally with at least one selected from
the group consisting of the substituent α; p is an integer of 1 or
2; ##STR01372## Alk1 is lower alkylene or lower alkenylene;
R0 is a hydrogen atom, lower alkyl, or acyl; X is O; R2a and
R2b are each independently a hydrogen atom, hydroxy, optionally
substituted lower alkyl, optionally substituted lower alkenyl, optionally
substituted amino, optionally substituted amidino, optionally substituted
acyl, optionally substituted carbamoyl, optionally substituted
carbamoylcarbonyl, optionally substituted lower alkylsulfonyl, optionally
substituted arylsulfonyl, an optionally substituted carbocyclic group, or
an optionally substituted heterocyclic group; lower alkyl being
substituted optionally with at least one selected from the group
consisting of the substituent α, lower alkenyl being substituted
optionally with the substituent α, amino being substituted
optionally with at least one selected from the group consisting of lower
alkyl, acyl, hydroxy, lower alkoxy, lower alkoxycarbonyl, a carbocyclic
group and a heterocyclic group, amidino being substituted optionally with
at least one selected from the group consisting of lower alkyl, acyl,
hydroxy, lower alkoxy, lower alkoxycarbonyl, a carbocyclic group and a
heterocyclic group, acyl includes aliphatic acyl of a carbon number of 1
to 10, carbocyclic carbonyl and heterocyclic carbonyl; acyl being
substituted optionally with the substituent α; and the ring part of
the carbocyclic carbonyl and the heterocyclic carbonyl is substituted
optionally with at least one selected from the group of lower alkyl; the
substituent group α; and lower alkyl substituted with at least one
selected from the group of the substituent α, carbamoyl being
substituted optionally with at least one selected from the group
consisting of lower alkyl, acyl, hydroxy, lower alkoxy, lower
alkoxycarbonyl, a carbocyclic group and a heterocyclic group,
carbamoylcarbonyl being substituted optionally with at least one selected
from the group consisting of lower alkyl, acyl, hydroxy, lower alkoxy,
lower alkoxycarbonyl, a carbocyclic group and a heterocyclic group, lower
alkylsulfonyl being substituted optionally with the substituent α,
arylsulfonyl being substituted optionally with at least one selected from
the group consisting of the substituent α and a lower alkyl, and
the carbocyclic group and the heterocyclic group being substituted
optionally with at least one selected from the group consisting of the
substituent α and lower alkyl; R3a, R3b, R4a, and
R4b are each independently a hydrogen atom, halogen, hydroxy,
optionally substituted lower alkyl, optionally substituted lower alkenyl,
optionally substituted acyl, carboxy, optionally substituted lower
alkoxycarbonyl, optionally substituted amino, optionally substituted
carbamoyl, an optionally substituted carbocyclic group, or an optionally
substituted heterocyclic group; lower alkyl being substituted optionally
with at least one selected from the group consisting of the substituent
α, lower alkenyl being substituted optionally with the substituent
α, acyl being substituted optionally with the substituent α;
and the ring part of the carbocyclic carbonyl and the heterocyclic
carbonyl is substituted optionally with at least one selected from the
group of lower alkyl; the substituent group α; and lower alkyl
substituted with at least one selected from the group of the substituent
α, lower alkoxycarbonyl being substituted optionally with the
substituent α, amino being substituted optionally with at least one
selected from the group consisting of lower alkyl, acyl, hydroxy, lower
alkoxy, lower alkoxycarbonyl, a carbocyclic group and a heterocyclic
group,
carbamoyl being substituted optionally with at least one selected from the group consisting of lower alkyl, acyl, hydroxy, lower alkoxy, lower alkoxycarbonyl, a carbocyclic group and a heterocyclic group, and the carbocyclic group and the heterocyclic group being substituted optionally with at least one selected from the group consisting of the substituent α and a lower alkyl; n=0, 1, 2 or 3, m=0, 1, 2 or 3, with the proviso that n+m=3, each R3a, each R3b, each R4a, and each R4b may be independently different; R5 is a hydrogen atom, optionally substituted lower alkyl, optionally substituted lower alkenyl, optionally substituted lower alkynyl, an optionally substituted carbocyclic group, an optionally substituted heterocyclic group; lower alkyl being substituted optionally with at least one selected from the group consisting of the substituent α, lower alkenyl being substituted optionally with the substituent α, lower alkynyl being substituted optionally with the substituent α, and the carbocyclic group and the heterocyclic group being substituted optionally with at least one selected from the group consisting of the substituent α and a lower alkyl; wherein acyl includes aliphatic acyl of a carbon number of 1 to 10, carbocyclic carbonyl and heterocyclic carbonyl; when ##STR01373## R5 and ring A can be taken together to form ##STR01374## wherein R5a and R5b are each independently a hydrogen atom or lower alkyl; s is an integer of 1 to 4; each R5a and each R5b may be different; its pharmaceutically acceptable salt, or a solvate thereof.
2. (canceled)
3. (canceled)
4. The compound according to claim 1, wherein E is a bond, its pharmaceutically acceptable salt, or a solvate thereof.
5. (canceled)
6. (canceled)
7. (canceled)
8. The compound according to claim 1, wherein R5 is optionally substituted lower alkyl, optionally substituted lower alkenyl, optionally substituted lower alkynyl, an optionally substituted carbocyclic group, or an optionally substituted heterocyclic group, lower alkyl being substituted optionally with at least one selected from the group consisting of the substituent α, the substituent α being at least one selected from the group consisting of halogen, hydroxy, lower alkoxy, hydroxy lower alkoxy, lower alkoxy lower alkoxy, acyl, acyloxy, carboxy, lower alkoxycarbonyl, amino, acylamino, lower alkylamino, lower alkylthio, carbamoyl, lower alkylcarbamoyl, hydroxy lower alkylcarbamoyl, sulfamoyl, lower alkylsulfamoyl, lower alkylsulfinyl, cyano, nitro, aryl, and heterocyclic group, lower alkenyl being substituted optionally with the substituent α, lower alkynyl being substituted optionally with the substituent α, and the carbocyclic group and the heterocyclic group being substituted optionally with at least one selected from the group consisting of the substituent α and lower alkyl, its pharmaceutically acceptable salt, or a solvate thereof.
9. The compound according to claim 1, wherein R2a is a hydrogen atom; R2b is a hydrogen atom, optionally substituted lower alkyl, optionally substituted acyl, optionally substituted lower alkylsulfonyl, or optionally substituted amidino, lower alkyl being substituted optionally with at least one selected from the group consisting of the substituent α, the substituent α being at least one selected from the group consisting of halogen, hydroxy, lower alkoxy, hydroxy lower alkoxy, lower alkoxy lower alkoxy, acyl, acyloxy, carboxy, lower alkoxycarbonyl, amino, acylamino, lower alkylamino, lower alkylthio, carbamoyl, lower alkylcarbamoyl, hydroxy lower alkylcarbamoyl, sulfamoyl, lower alkylsulfamoyl, lower alkylsulfinyl, cyano, nitro, aryl, and heterocyclic group, acyl being substituted optionally with the substituent α, and the ring part of the carbocyclic carbonyl and the heterocyclic carbonyl is substituted optionally with at least one selected from the group of lower alkyl; the substituent group α; and lower alkyl substituted with at least one selected from the group of the substituent α, lower alkylsulfonyl being substituted optionally with the substituent α, and amidino being substituted optionally with at least one selected from the group consisting of lower alkyl, acyl, hydroxy, lower alkoxy, lower alkoxycarbonyl, a carbocyclic group and a heterocyclic group, its pharmaceutically acceptable salt, or a solvate thereof.
10. The compound according to claim 1, wherein NR2aR2b is represented by the formula: ##STR01375## R6, R7, and R8 are each independently a hydrogen atom, lower alkyl, or acyl; Y is optionally substituted lower alkylene, optionally substituted lower alkenylene, or optionally substituted lower alkynylene; lower alkylene being substituted optionally with the substituent α, the substituent α being at least one selected from the group consisting of halogen, hydroxy, lower alkoxy, hydroxy lower alkoxy, lower alkoxy lower alkoxy, acyl, acyloxy, carboxy, lower alkoxycarbonyl, amino, acylamino, lower alkylamino, lower alkylthio, carbamoyl, lower alkylcarbamoyl, hydroxy lower alkylcarbamoyl, sulfamoyl, lower alkylsulfamoyl, lower alkylsulfinyl, cyano, nitro, aryl, and heterocyclic group, lower alkenylene being substituted optionally with the substituent α, and lower alkynylene being substituted optionally with the substituent α; Z is O or S; its pharmaceutically acceptable salt, or a solvate thereof.
11. The compound according to claim 1, wherein ring A is optionally substituted phenyl, the phenyl being substituted optionally with at least one selected from the group consisting of the above (A) to (AO), its pharmaceutically acceptable salt, or a solvate thereof.
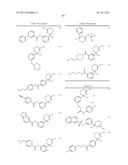
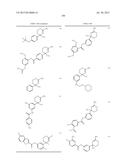
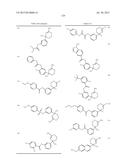
12. The compound according to claim 1, wherein ring A is represented by the formula: ##STR01376## wherein R9, R10, and R11 are a hydrogen atom or G; G is <A>, <B>, <C>, <D>, <E>, <F>, <G>, or <H>, where <A>, <B>, <C>, <D>, <E>, <F>, <G> and <H> are the following: <A> halogen, hydroxy, cyano, nitro, mercapto, optionally substituted lower alkyl, optionally substituted lower alkoxy, optionally substituted lower alkenyl, optionally substituted lower alkynyl, optionally substituted acyl, optionally substituted acyloxy, carboxy, optionally substituted lower alkoxycarbonyl, optionally substituted lower alkoxycarbonyloxy, optionally substituted aryloxycarbonyloxy, optionally substituted amino, optionally substituted carbamoyl, optionally substituted carbamoyloxy, optionally substituted lower alkylthio, optionally substituted arylthio, optionally substituted lower alkylsulfonyl, optionally substituted arylsulfonyl, optionally substituted lower alkylsulfinyl, optionally substituted arylsulfinyl, optionally substituted lower alkylsulfonyloxy, optionally substituted arylsulfonyloxy, optionally substituted sulfamoyl, an optionally substituted carbocyclic group, optionally substituted carbocyclicoxy, an optionally substituted heterocyclic group, or optionally substituted heterocyclicoxy, lower alkyl being substituted optionally with at least one selected from the group consisting of the substituent α, a hydroxyimino group, and a lower alkoxyimino group, the substituent α being at least one selected from the group consisting of halogen, hydroxy, lower alkoxy, hydroxy lower alkoxy, lower alkoxy lower alkoxy, acyl, acyloxy, carboxy, lower alkoxycarbonyl, amino, acylamino, lower alkylamino, lower alkylthio, carbamoyl, lower alkylcarbamoyl, hydroxy lower alkylcarbamoyl, sulfamoyl, lower alkylsulfamoyl, lower alkylsulfinyl, cyano, nitro, aryl, and heterocyclic group, lower alkoxy being substituted optionally with the substituent α, lower alkenyl being substituted optionally with the substituent α, lower alkynyl being substituted optionally with the substituent α, acyl includes aliphatic acyl of a carbon number of 1 to 10, carbocyclic carbonyl and heterocyclic carbonyl; and acyl being substituted optionally with the substituent α; and the ring part of the "carbocyclic carbonyl" and the "heterocyclic carbonyl" is substituted optionally with at least one selected from the group of "lower alkyl"; the "substituent group α"; and "lower alkyl substituted with at least one selected from the group of the substituent α", acyloxy includes aliphatic acyloxy of a carbon number of 1 to 10, carbocyclic carbonyloxy and heterocyclic carbonyloxy; and acyloxy being substituted optionally with the substituent α; and the ring part of the "carbocyclic carbonyloxy" and the "heterocyclic carbonyloxy" is substituted optionally with at least one selected from the group of "lower alkyl"; the "substituent group α"; and "lower alkyl substituted with at least one selected from the group of the substituent α", lower alkoxycarbonyl being substituted optionally with the substituent α, lower alkoxycarbonyloxy being substituted optionally with the substituent α, aryloxycarbonyloxy being substituted optionally with at least one selected from the group consisting of lower alkyl and the substituent α, amino being substituted optionally with at least one selected from the group consisting of lower alkyl, acyl, hydroxy, lower alkoxy, lower alkoxycarbonyl, a carbocyclic group and a heterocyclic group, carbamoyl being substituted optionally with at least one selected from the group consisting of lower alkyl, acyl, hydroxy, lower alkoxy, lower alkoxycarbonyl, a carbocyclic group and a heterocyclic group, carbamoyloxy being substituted optionally with at least one selected from the group consisting of lower alkyl, acyl, hydroxy, lower alkoxy, lower alkoxycarbonyl, a carbocyclic group and a heterocyclic group, lower alkylthio being substituted optionally with the substituent α, arylthio being substituted optionally with at least one selected from the group consisting of lower alkyl and the substituent α, lower alkylsulfonyl being substituted optionally with the substituent α, arylsulfonyl being substituted optionally with at least one selected from the group consisting of the substituent α, azido, and lower alkyl, lower alkylsulfinyl being substituted optionally with the substituent α, arylsulfinyl being substituted optionally with at least one selected from the group consisting of lower alkyl and the substituent α, lower alkylsulfonyloxy being substituted optionally with the substituent α, arylsulfonyloxy being substituted optionally with the substituent α, sulfamoyl being substituted optionally with the substituent α, and the carbocyclic group, carbocyclicoxy, the heterocyclic group, and heterocyclicoxy being substituted optionally with at least one selected from the group consisting of lower alkyl and the substituent α; <B> one of the following (i), (ii), (iv), (vi), (viii) and (x): ##STR01377## wherein each Q1 is optionally substituted lower alkylene, lower alkylene being substituted optionally with the substituent α; Q2 and Q3 are each independently a bond, optionally substituted lower alkylene or optionally substituted lower alkenylene, lower alkylene and lower alkenylene being substituted optionally with the substituent α; W1 and W2 are each independently O or S; W3 is O, S, or NR12; R12 is a hydrogen atom, lower alkyl, hydroxy lower alkyl, lower alkoxy lower alkyl, lower alkoxycarbonyl lower alkyl, carbocyclic lower alkyl, or acyl; and ring B is an optionally substituted carbocyclic group or an optionally substituted heterocyclic group, the carbocyclic group and the heterocyclic group being substituted optionally with at least one selected from the group consisting of (a) the substituent α; (b) lower alkyl optionally substituted with the substituent α; (c) amino lower alky substituted with the substituent α; (d) hydroxyimino lower alkyl; (e) lower alkoxyimino lower alkyl; (f) lower alkenyl optionally substituted with the substituent α; (g) lower alkynyl optionally substituted with the substituent α; (h) lower alkoxy optionally substituted with the substituent α; (i) lower alkylthio optionally substituted with the substituent α; (j) lower alkylamino substituted with the substituent α; (k) lower alkylsulfonyl optionally substituted with the substituent α; (l) aryl lower alkoxycarbonyl optionally substituted with one or more substituents selected from the group of substituent α and lower alkyl; (m) acyl substituted with the substituent α; (n) lower alkylsulfonyl optionally substituted with the substituent α; (o) sulfamoyl; (p) lower alkyl sulfamoyl optionally substituted with one or more substituents selected from the group of substituent α; (q) cycloalkyl optionally substituted with one or more substituents selected from the group of substituent α, azido and lower alkyl; (r) aryl optionally substituted with one or more substituents selected from the group of substituent α, azido and lower alkyl; (s) heterocyclic group optionally substituted with one or more substituents selected from the group of substituent α, azido and lower alkyl; (t) aryloxy optionally substituted with one or more substituents selected from the group of substituent α, azido and lower alkyl; (u) heterocyclicoxy optionally substituted with one or more substituents selected from the group of substituent α, azido and lower alkyl; (v) arylthio optionally substituted with one or more substituents selected from the group of substituent α, azido and lower alkyl; (w) heterocyclic thio optionally substituted with one or more substituents selected from the group of substituent α, azido and lower alkyl; (x) arylamino optionally substituted with one or more substituents selected from the group of substituent α, azido and lower alkyl; (y) heterocyclic amino optionally substituted with one or more substituents selected from the group of substituent α, azido and lower alkyl; (z) aryl lower alkylamino optionally substituted with one or more substituents selected from the group of substituent α, azido and lower alkyl; (aa) heterocyclic lower alkylamino optionally substituted with one or more substituents selected from the group of substituent α, azido and lower alkyl; (ab) arylsulfamoyl optionally substituted with one or more substituents selected from the group of substituent α, azido and lower alkyl; (ac) heterocyclic sulfamoyl optionally substituted with one or more substituents selected from the group of substituent α, azido and lower alkyl; (ad) arylsulfonyl optionally substituted with one or more substituents selected from the group of substituent α, azido and lower alkyl; (ae) heterocyclic sulfonyl optionally substituted with one or more substituents selected from the group of substituent α, azido and lower alkyl; (af) arylcarbamoyl optionally substituted with one or more substituents selected from the group of substituent α, azido and lower alkyl; (ag) heterocyclic carbamoyl optionally substituted with one or more substituents selected from the group of substituent α, azido and lower alkyl; (ah) aryl lower alkylcarbamoyl optionally substituted with one or more substituents selected from the group of substituent α, azido and lower alkyl; (ai) heterocyclic lower alkylcarbamoyl optionally substituted with one or more substituents selected from the group of substituent α, azido and lower alkyl; (aj) aryloxy carbonyl optionally substituted with one or more substituents selected from the group of substituent α, azido and lower alkyl; (ak) heterocyclicoxycarbonyl optionally substituted with one or more substituents selected from the group of substituent α, azido and lower alkyl; (al) lower alkylenedioxy substituted optionally with halogen; and (am) oxo; <C> one of the following (iii), (v), (vii), (ix) and (xii): ##STR01378## wherein each Q2 is optionally substituted lower alkylene, lower alkylene being substituted optionally with the substituent α; Q1 and Q3 are each independently a bond, optionally substituted lower alkylene or optionally substituted lower alkenylene, lower alkylene and lower alkenylene being substituted optionally with the substituent α; W1 and W2 are each independently O or S; W3 is O, S, or NR12; R12 is a hydrogen atom, lower alkyl, hydroxy lower alkyl, lower alkoxy lower alkyl, lower alkoxycarbonyl lower alkyl, carbocyclic lower alkyl, or acyl; p is an integer of 1 or 2; and ring B is an optionally substituted carbocyclic group or an optionally substituted heterocyclic group, the carbocyclic group and the heterocyclic group being substituted optionally with at least one selected from the group consisting of the above (a) to (am); and R14 is a hydrogen atom or lower alkyl; <D> one of the following (iii), (v), (vi) and (xi): ##STR01379## wherein each Q1, Q2 and Q4 are each independently optionally substituted lower alkylene, lower alkylene being substituted optionally with the substituent α; Q3 are each independently a bond, optionally substituted lower alkylene, or optionally substituted lower alkenylene, lower alkylene and lower alkenylene being substituted optionally with the substituent α; W1 is O or S; W3 is O, S, or NR12; R12 is a hydrogen atom, lower alkyl, hydroxy lower alkyl, lower alkoxy lower alkyl, lower alkoxycarbonyl lower alkyl, carbocyclic lower alkyl, or acyl; and ring B is an optionally substituted carbocyclic group or an optionally substituted heterocyclic group, the carbocyclic group and the heterocyclic group being substituted optionally with at least one selected from the group consisting of the above (a) to (am); <E> one of the following (i), (ii), (iv), (vi), (viii) and (x): ##STR01380## wherein each Q1 is optionally substituted lower alkenylene, lower alkenylene being substituted optionally with the substituent α; Q2 and Q3 are each independently a bond, optionally substituted lower alkylene or optionally substituted lower alkenylene, lower alkylene and lower alkenylene being substituted optionally with the substituent α; W1 and W2 are each independently O or S; W3 is O, S, or NR12; R12 is a hydrogen atom, lower alkyl, hydroxy lower alkyl, lower alkoxy lower alkyl, lower alkoxycarbonyl lower alkyl, carbocyclic lower alkyl, or acyl; ring B is an optionally substituted carbocyclic group or an optionally substituted heterocyclic group, the carbocyclic group and the heterocyclic group being substituted optionally with at least one selected from the group consisting of the above (a) to (am); <F> one of the following (iii), (v), (vii), and (ix): ##STR01381## wherein Q2 is optionally substituted lower alkenylene, lower alkenylene being substituted optionally with the substituent α; Q1 and Q3 are each independently a bond, optionally substituted lower alkylene or optionally substituted lower alkenylene, lower alkylene and lower alkenylene being substituted optionally with the substituent α; W1 and W2 are each independently O or S; W3 is O, S, or NR12; R12 is a hydrogen atom, lower alkyl, hydroxy lower alkyl, lower alkoxy lower alkyl, lower alkoxycarbonyl lower alkyl, carbocyclic lower alkyl, or acyl; p is an integer of 1 or 2; ring B is an optionally substituted carbocyclic group or an optionally substituted heterocyclic group, the carbocyclic group and the heterocyclic group being substituted optionally with at least one selected from the group consisting of the above (a) to (am); and R14 is a hydrogen atom or lower alkyl; <G> one of the following (ii), (iv),(v), (ix), (xiii) and (xiv): ##STR01382## wherein each Q1, Q2 and Q3 are each independently a bond, optionally substituted lower alkylene, or optionally substituted lower alkenylene; lower alkylene and lower alkenylene being substituted optionally with the substituent α; W1 is O or S; W3 is O, S, or NR12; R12 is a hydrogen atom, lower alkyl, hydroxy lower alkyl, lower alkoxy lower alkyl, lower alkoxycarbonyl lower alkyl, carbocyclic lower alkyl, or acyl; Alk2 is optionally substituted lower alkyl, lower alkyl being substituted optionally with at least one selected from the group consisting of the substituent
α; p is an integer of 1 or 2; and ring B is an optionally substituted carbocyclic group or an optionally substituted heterocyclic group, the carbocyclic group and the heterocyclic group being substituted optionally with at least one selected from the group consisting of the above (a) to (am); <H> one of the following (i), (viii) and (xv): ##STR01383## wherein Q2 is a bond, optionally substituted lower alkylene, or optionally substituted lower alkenylene, lower alkylene and lower alkenylene being substituted optionally with the substituent α; W3 is O, S, or NR12, R12 is a hydrogen atom, lower alkyl, hydroxy lower alkyl, lower alkoxy lower alkyl, lower alkoxycarbonyl lower alkyl, carbocyclic lower alkyl, or acyl; Alk2 is optionally substituted lower alkyl, lower alkyl being substituted optionally with at least one selected from the group consisting of the substituent α; ring B is an optionally substituted carbocyclic group or an optionally substituted heterocyclic group, the carbocyclic group and the heterocyclic group being substituted optionally with at least one selected from the group consisting of the above (a) to (am); each G may be independently different; its pharmaceutically acceptable salt, or a solvate thereof.
13. The compound according to claim 12, wherein R9, R10, and R11 are a hydrogen atom, halogen, hydroxy, cyano, nitro, mercapto, optionally substituted lower alkyl, optionally substituted lower alkoxy, optionally substituted lower alkenyl, optionally substituted lower alkynyl, optionally substituted acyl, optionally substituted acyloxy, carboxy, optionally substituted lower alkoxycarbonyl, optionally substituted lower alkoxycarbonyloxy, optionally substituted lower aryloxycarbonyloxy, optionally substituted amino, optionally substituted carbamoyl, optionally substituted carbamoyloxy, optionally substituted lower alkylthio, optionally substituted lower arylthio, optionally substituted lower alkylsulfonyl, optionally substituted arylsulfonyl, optionally substituted lower alkylsulfinyl, optionally substituted arylsulfinyl, optionally substituted lower alkylsulfonyloxy, optionally substituted arylsulfonyloxy, optionally substituted sulfamoyl, an optionally substituted carbocyclic group, optionally substituted carbocyclicoxy, an optionally substituted heterocyclic group, or optionally substituted heterocyclicoxy; lower alkyl being substituted optionally with at least one selected from the group consisting of the substituent α, a hydroxyimino group, and a lower alkoxyimino group, the substituent α being at least one selected from the group consisting of halogen, hydroxy, lower alkoxy, hydroxy lower alkoxy, lower alkoxy lower alkoxy, acyl, acyloxy, carboxy, lower alkoxycarbonyl, amino, acylamino, lower alkylamino, lower alkylthio, carbamoyl, lower alkylcarbamoyl, hydroxy lower alkylcarbamoyl, sulfamoyl, lower alkylsulfamoyl, lower alkylsulfinyl, cyano, nitro, aryl, and heterocyclic group, lower alkoxy being substituted optionally with the substituent α, lower alkenyl being substituted optionally with the substituent α, lower alkynyl being substituted optionally with the substituent α, acyl includes aliphatic acyl of a carbon number of 1 to 10, carbocyclic carbonyl and heterocyclic carbonyl; and acyl being substituted optionally with the substituent α; and the ring part of the "carbocyclic carbonyl" and the "heterocyclic carbonyl" is substituted optionally with at least one selected from the group of "lower alkyl"; the "substituent group α"; and "lower alkyl substituted with at least one selected from the group of the substituent α". acyloxy includes aliphatic acyloxy of a carbon number of 1 to 10, carbocyclic carbonyloxy and heterocyclic carbonyloxy; and acyloxy being substituted optionally with the substituent α; and the ring part of the "carbocyclic carbonyloxy" and the "heterocyclic carbonyloxy" is substituted optionally with at least one selected from the group of "lower alkyl"; the "substituent group α"; and "lower alkyl substituted with at least one selected from the group of the substituent α", lower alkoxycarbonyl being substituted optionally with the substituent α, lower alkoxycarbonyloxy being substituted optionally with the substituent α, aryloxycarbonyloxy being substituted optionally with at least one selected from the group consisting of lower alkyl and the substituent α, amino being substituted optionally with at least one selected from the group consisting of lower alkyl, acyl, hydroxy, lower alkoxy, lower alkoxycarbonyl, a carbocyclic group and a heterocyclic group, carbamoyl being substituted optionally with at least one selected from the group consisting of lower alkyl, acyl, hydroxy, lower alkoxy, lower alkoxycarbonyl, a carbocyclic group and a heterocyclic group, carbamoyloxy being substituted optionally with at least one selected from the group consisting of lower alkyl, acyl, hydroxy, lower alkoxy, lower alkoxycarbonyl, a carbocyclic group and a heterocyclic group, lower alkylthio being substituted optionally with the substituent α, arylthio being substituted optionally with at least one selected from the group consisting of lower alkyl and the substituent α, lower alkylsulfonyl being substituted optionally with the substituent α, arylsulfonyl being substituted optionally with at least one selected from the group consisting of the substituent α and lower alkyl, lower alkylsulfinyl being substituted optionally with the substituent α, arylsulfinyl being substituted optionally with at least one selected from the group consisting of lower alkylsulfonyloxy being substituted optionally with the substituent α, arylsulfonyloxy being substituted optionally with the substituent α, sulfamoyl being substituted optionally with the substituent α, and the carbocyclic group, carbocyclicoxy, the heterocyclic group, and heterocyclicoxy being substituted optionally with at least one selected from the group consisting of lower alkyl and the substituent α; and G is represented by the formula: ##STR01384## ##STR01385## Q1, Q2, and Q3 are each independently a bond, optionally substituted lower alkylene, or optionally substituted lower alkenylene; lower alkylene and lower alkenylene being substituted optionally with the substituent α; Q4 is optionally substituted lower alkylene or optionally substituted lower alkenylene; lower alkylene and lower alkenylene being substituted optionally with the substituent α; W1 and W2 are each independently O or S; W3 is O, S, or NR12; R12 is a hydrogen atom, lower alkyl, hydroxy lower alkyl, lower alkoxy lower alkyl, lower alkoxycarbonyl lower alkyl, carbocyclic lower alkyl, or acyl; R14 is a hydrogen atom or lower alkyl; ring B is an optionally substituted carbocyclic group or an optionally substituted heterocyclic group; the carbocyclic group and the heterocyclic group being substituted optionally with at least one selected from the group consisting of the above (a) to (am); Alk2 is optionally substituted lower alkyl; lower alkyl being substituted optionally with at least one selected from the group consisting of the substituent α; p is 1 or 2; if there are multiple W1, multiple W3 and multiple R12, each are independently different, its pharmaceutically acceptable salt, or a solvate thereof.
14. The compound according to claim 13, wherein ring B is aryl optionally substituted with one or more substituents selected from the group consisting of halogen, hydroxy, optionally substituted lower alkyl, optionally substituted lower alkoxy, optionally substituted acyl, optionally substituted amino, cyano, optionally substituted carbamoyl, an optionally substituted carbocyclic group, optionally substituted carbocyclicoxy, and an optionally substituted heterocyclic group or heteroaryl optionally substituted with one or more substituents selected from the group consisting of halogen, hydroxy, optionally substituted lower alkyl, optionally substituted lower alkoxy, optionally substituted acyl, optionally substituted amino, cyano, optionally substituted carbamoyl, an optionally substituted carbocyclic group, optionally substituted carbocyclicoxy, and an optionally substituted heterocyclic group, lower alkyl being substituted optionally with the substituent α, the substituent α being at least one selected from the group consisting of halogen, hydroxy, lower alkoxy, hydroxy lower alkoxy, lower alkoxy lower alkoxy, acyl, acyloxy, carboxy, lower alkoxycarbonyl, amino, acylamino, lower alkylamino, lower alkylthio, carbamoyl, lower alkylcarbamoyl, hydroxy lower alkylcarbamoyl, sulfamoyl, lower alkylsulfamoyl, lower alkylsulfinyl, cyano, nitro, aryl, and heterocyclic group, lower alkoxy being substituted optionally with the substituent α, acyl includes aliphatic acyl of a carbon number of 1 to 10, carbocyclic carbonyl and heterocyclic carbonyl; and acyl being substituted optionally with the substituent α; and the ring part of the "carbocyclic carbonyl" and the "heterocyclic carbonyl" is substituted optionally with at least one selected from the group of "lower alkyl"; the "substituent group α"; and "lower alkyl substituted with at least one selected from the group of the substituent α", amino being substituted optionally with at least one selected from the group consisting of lower alkyl, acyl, hydroxy, lower alkoxy, lower alkoxycarbonyl, a carbocyclic group and a heterocyclic group, carbamoyl being substituted optionally with at least one selected from the group consisting of lower alkyl, acyl, hydroxy, lower alkoxy, lower alkoxycarbonyl, a carbocyclic group and a heterocyclic group, and the carbocyclic group, carbocyclicoxy, and the heterocyclic group being substituted optionally with at least one selected from the group consisting of lower alkyl and the substituent α, its pharmaceutically acceptable salt, or a solvate thereof.
15. The compound according to claim 13, wherein G is represented by the formula: ##STR01386## its pharmaceutically acceptable salt, or a solvate thereof.
16. The compound according to claim 1, wherein R5 is C1 to C3 alkyl, its pharmaceutically acceptable salt, or a solvate thereof.
17. The compound according to claim 1, wherein R5 is methyl, its pharmaceutically acceptable salt, or a solvate thereof.
18. The compound according to claim 1, wherein R3a and R3b are each independently a hydrogen atom, halogen, hydroxy, optionally substituted lower alkyl, optionally substituted lower alkoxy, or optionally substituted aryl, lower alkyl being substituted optionally with at least one selected from the group consisting of the substituent α, the substituent α being at least one selected from the group consisting of halogen, hydroxy, lower alkoxy, hydroxy lower alkoxy, lower alkoxy lower alkoxy, acyl, acyloxy, carboxy, lower alkoxycarbonyl, amino, acylamino, lower alkylamino, lower alkylthio, carbamoyl, lower alkylcarbamoyl, hydroxy lower alkylcarbamoyl, sulfamoyl, lower alkylsulfamoyl, lower alkylsulfinyl, cyano, nitro, aryl, and heterocyclic group, lower alkoxy being substituted optionally with the substituent α, and aryl being substituted optionally with the substituent α or lower alkyl, its pharmaceutically acceptable salt, or a solvate thereof.
19. The compound according to claim 1, wherein all of R3a and all of R3b are hydrogen atoms, its pharmaceutically acceptable salt, or a solvate thereof.
20. (canceled)
21. (canceled)
22. The compound according to claim 1, wherein n=3 and m=0, its pharmaceutically acceptable salt, or a solvate thereof.
23. A pharmaceutical composition containing the compound as defined in claim 1, its pharmaceutically acceptable salt, or a solvate thereof as an active ingredient, and a pharmaceutically acceptable carrier.
Description:
CROSS-REFERENCE TO RELATED APPLICATIONS
[0001] This application is a Division of application Ser. No. 13/941,082 filed Jul. 12, 2013, which is a Division of application Ser. No. 13/768,557 filed Feb. 15, 2013, which is a Division of application Ser. No. 13/243,971, filed Sep. 23, 2011, which is a Division of application Ser. No. 12/089,409, filed Apr. 7, 2008, which is a U.S. National Stage of PCT/JP2006/321015, filed Oct. 23, 2006, which applications are incorporated herein by reference.
TECHNICAL FIELD
[0002] The present invention relates to a compound which has reducing effect to produce amyloid β protein and is useful as an agent for treating disease induced by production, secretion and/or deposition of amyloid β protein.
BACKGROUND ART
[0003] In the brain of Alzheimer's patient, the peptide composed of about 40 amino acids residue as is called amyloid β protein, that accumulates to form insoluble specks (senile specks) outside nerve cells is widely observed. It is concerned that this senile specks kill nerve cells to cause Alzheimer's disease. The therapeutic agents for Alzheimer's disease, such as decomposition agents of amyloid β protein and amyloid β vaccine, are under investigation.
[0004] Secretase is an enzyme which cleaves amyloid β precursor protein (APP) in cell and produce amyloid β protein. The enzyme which controls the production of N terminus of amyloid β protein is called as BACE 1 (beta-site APP-cleaving enzyme 1, β-secretase). It is thought that inhibition of this enzyme leads to reduction of producing amyloid β protein and that the therapeutic agent for Alzheimer's disease will be created by the inhibition.
[0005] Patent Literature 1 describes the compounds which are similar to those of the present invention, and the compounds have NO synthase enzyme inhibitory activity and are useful for dementia.
[0006] Patent Literatures 2 to 4 and Non-patent Literatures 1 and 2 describe the compounds which are similar to those of the present invention, and are useful for hypertensive agent, analgesic like morphine, or tranquilizers, intermediate for medicine, analgesic respectively.
[0007] Patent Literature 5 to 13 are known as BACE 1 inhibitor, however, all compounds in these literatures have different structures from the present invention.
[0008] [Patent Literature 1] International Patent Application Publication WO96/014842
[0009] [Patent Literature 2] U.S. Pat. No. 3,235,551
[0010] [Patent Literature 3] U.S. Pat. No. 3,227,713
[0011] [Patent Literature 4] JP Application Publication H09-067355
[0012] [Patent Literature 5] International Patent Application Publication WO01/187293
[0013] [Patent Literature 6] International Patent Application Publication WO04/014843
[0014] [Patent Literature 7] JP Application Publication 2004-149429
[0015] [Patent Literature 8] International Patent Application Publication WO02/96897
[0016] [Patent Literature 9] International Patent Application Publication WO04/043916
[0017] [Patent Literature 10] International Patent Application Publication WO2005/058311
[0018] [Patent Literature 11] International Patent Application Publication WO2005/097767
[0019] [Patent Literature 12] International Patent Application Publication WO2006/041404
[0020] [Patent Literature 13] International Patent Application Publication WO2006/041405
[0021] [(Non-Patent Literature 1] Journal of Heterocyclic Chemistry, 14, 717-723 (1977)
[0022] [Non-Patent Literature 2] Journal of Organic Chemistry, 33, 8, 3126-3132 (1968)
DISCLOSURE OF INVENTION
Problems to be Solved by the Invention
[0023] The present invention provides compounds which have reducing effects to produce amyloid β protein, especially BACE 1 inhibitory activity, and are useful as an agent for treating disease induced by production, secretion and/or deposition of amyloid β protein.
Means to Solve the Problems
[0024] The present invention provides:
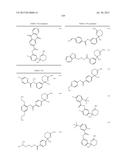
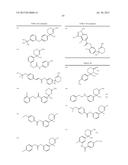
(a) a composition having BACE 1 inhibitory activity containing a compound represented by the general formula (I):
##STR00002##
wherein ring A is an optionally substituted carbocyclic group or an optionally substituted heterocyclic group;
##STR00003##
Alk1 is lower alkylene or lower alkenylene; R0 is a hydrogen atom, lower alkyl or acyl;
X is S, O, or NR1;
[0025] R1 is a hydrogen atom or lower alkyl; R2a and R2b are each independently a hydrogen atom, hydroxy, optionally substituted lower alkyl, optionally substituted lower alkenyl, optionally substituted amino, optionally substituted amidino, optionally substituted acyl, optionally substituted carbamoyl, optionally substituted carbamoylcarbonyl, optionally substituted lower alkylsulfonyl, optionally substituted arylsulfonyl, an optionally substituted carbocyclic group or an optionally substituted heterocyclic group; R3a, R3b, R4a and R4b are each independently a hydrogen atom, halogen, hydroxy, optionally substituted lower alkyl, optionally substituted lower alkenyl, optionally substituted acyl, carboxy, optionally substituted lower alkoxycarbonyl, optionally substituted amino, optionally substituted carbamoyl, an optionally substituted carbocyclic group or an optionally substituted heterocyclic group; n and m are each independently an integer of 0 to 3; n+m is an integer of 1 to 3; each R3a, each R3b, each R4a, and each R4b may be independently different; R5 is a hydrogen atom, optionally substituted lower alkyl, optionally substituted lower alkenyl, optionally substituted lower alkenyl, an optionally substituted carbocyclic group or an optionally substituted heterocyclic group;
##STR00004##
R5 and ring A can be taken together to form
##STR00005##
wherein R5a and R5b are each independently a hydrogen atom or lower alkyl; s is an integer of 1 to 4; each R5a and each R5b may be different; with the proviso that the compound wherein n+m is 2; R5 is a hydrogen atom; and ring A is non-substituted phenyl is excluded, its pharmaceutically acceptable salt, or a solvate thereof, (a1) a composition having BACE 1 inhibitory activity containing a compound represented by the general formula (I):
##STR00006##
wherein ring A is an optionally substituted carbocyclic group or an optionally substituted heterocyclic group;
##STR00007##
Alk1 is lower alkylene; R0 is a hydrogen atom, lower alkyl or acyl;
X is S, O, or NR1;
[0026] R1 is a hydrogen atom or lower alkyl; R2a and R2b are each independently a hydrogen atom, hydroxy, optionally substituted lower alkyl, optionally substituted lower alkenyl, optionally substituted amino, optionally substituted amidino, optionally substituted acyl, optionally substituted carbamoyl, optionally substituted lower alkylsulfonyl, optionally substituted arylsulfonyl, an optionally substituted carbocyclic group or an optionally substituted heterocyclic group; R3a, R3b, R4a, and R4b are each independently a hydrogen atom, halogen, hydroxy, optionally substituted lower alkyl, optionally substituted lower alkenyl, optionally substituted acyl, carboxy, optionally substituted lower alkoxycarbonyl, optionally substituted amino, optionally substituted carbamoyl, an optionally substituted carbocyclic group or an optionally substituted heterocyclic group; n and m are each independently an integer of 0 to 3; n+m is an integer of 1 to 3; each R3a, each R3b, each R4a, and each R4b may be independently different; R5 is a hydrogen atom, optionally substituted lower alkyl, optionally substituted lower alkenyl, optionally substituted lower alkynyl, an optionally substituted carbocyclic group or an optionally substituted heterocyclic group;
##STR00008##
R5 and ring A can be taken together to form
##STR00009##
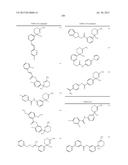
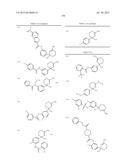
wherein R5a and R5b are each independently a hydrogen atom or lower alkyl; s is an integer of 1 to 4; each R5a and each R5b may be different; with the proviso that the compound wherein n+m is 2; R5 is a hydrogen atom; and ring A is non-substituted phenyl is excluded, its pharmaceutically acceptable salt, or a solvate thereof, (b) a composition having BACE 1 inhibitory activity according to (a), wherein X is S, (c) a composition having BACE 1 inhibitory activity according to (a), wherein n is 2, and m is 0, (d) a composition having BACE 1 inhibitory activity according to (a), wherein E is a bond, (e) a compound represented by the general formula (I):
##STR00010##
wherein each symbols are the same as described in (a), with the proviso that the compounds as shown below; i) wherein n+m is 2, R5 is a hydrogen atom, and ring A is non-substituted phenyl; ii) wherein n is 2, m is 0, R2a is a hydrogen atom, R2b is a hydrogen atom or acetyl, R5 is methyl, and ring A is phenyl or 4-methoxyphenyl; iii) wherein n is 2, m is 0, R2a is a hydrogen atom, R2b is a hydrogen atom or acetyl, R6 is ethyl, and ring A is 3,4-dimethoxyphenyl; iv) wherein n is 2, m is 0, R2a is a hydrogen atom, R2b is a hydrogen atom or acetyl, and R5 and ring A is phenyl; v) wherein n is 2, m is 0, R2a and R2b is a hydrogen atom, R5 and ring A are taken together to form
##STR00011##
wherein Me is methyl, and each symbols are the same as described above; and vi) wherein n+m is 2, R5 is a hydrogen atom, ring A is phenyl substituted with one or two substituent(s) selected from the group of hydroxy, halogen, lower alkyl, lower alkoxy, nitro, amino, lower alkylcarbonylamino, mercapto, lower alkylthio, and carbamoyl, non-substituted phenyl, or non-substituted naphthyl; are excluded, its pharmaceutically acceptable salt, or a solvate thereof, (f) the compound according to (e), wherein X is S, its pharmaceutically acceptable salt, or a solvate thereof, (g) the compound according to (e) or (i), wherein n is 2, and m is 0, its pharmaceutically acceptable salt, or a solvate thereof, (h) the compound according to any one of (e) to (g), wherein R5 is optionally substituted lower alkyl, optionally substituted lower alkenyl, optionally substituted lower alkynyl, an optionally substituted carbocyclic group or an optionally substituted heterocyclic group, its pharmaceutically acceptable salt, or a solvate thereof, (i) the compound according to any one of (e) to (h), wherein R2a is a hydrogen atom; R2b is a hydrogen atom, optionally substituted lower alkyl, optionally substituted acyl, optionally substituted lower alkylsulfonyl, or optionally substituted amidino, its pharmaceutically acceptable salt, or a solvate thereof, (j) the compound according to any one of (e) to (h), wherein NR2aR2b is represented by the formula:
##STR00012##
R6, R7, and R8 are each independently a hydrogen atom, lower alkyl or acyl, Y is optionally substituted lower alkylene, optionally substituted lower alkenylene or optionally substituted lower alkenylene;
Z is O or S;
[0027] its pharmaceutically acceptable salt, or a solvate thereof, (k) the compound according to any one of (e) to (j), wherein ring A is substituted phenyl, its pharmaceutically acceptable salt, or a solvate thereof, (l) the compound according to any one of (e) to (j), wherein ring A is represented by the formula:
##STR00013##
wherein R9, R10 and R11 are hydrogen atom or G; G is halogen, hydroxy, cyano, nitro, mercapto, optionally substituted lower alkyl, optionally substituted lower alkoxy, optionally substituted lower alkenyl, optionally substituted lower alkynyl, optionally substituted acyl, optionally substituted acyloxy, carboxy, optionally substituted lower alkoxycarbonyl, optionally substituted lower alkoxycarbonyloxy, optionally substituted aryloxycarbonyloxy, optionally substituted amino, optionally substituted carbamoyl, optionally substituted carbamoyloxy, optionally substituted lower alkylthio, optionally substituted arylthio, optionally substituted lower alkylsulfonyl, optionally substituted arylsulfonyl, optionally substituted lower alkylsulfinyl, optionally substituted arylsulfinyl, optionally substituted lower alkylsulfonyloxy, optionally substituted arylsulfonyloxy, an optionally substituted carbocyclic group, optionally substituted carbocyclicoxy, an optionally substituted heterocyclic group or optionally substituted heterocyclicoxy; each G may be independently different; its pharmaceutically acceptable salt, or a solvate thereof, (m) the compound according to (l), wherein G is represented by the formula:
##STR00014## ##STR00015## ##STR00016##
Q1, Q2, w and Q3 are each independently a bond, optionally substituted lower alkylene, or optionally substituted lower alkenylene; Q4 is optionally substituted lower alkylene or optionally substituted lower alkenylene; W1 and W2 are each independently O or S;
W3 is O, S or NR12;
[0028] R12 is a hydrogen atom, lower alkyl, hydroxy lower alkyl, lower alkoxy lower alkyl, lower alkoxycarbonyl lower alkyl, carbocyclic lower alkyl or acyl; R14 is a hydrogen atom or lower alkyl; ring B is an optionally substituted carbocyclic group or an optionally substituted heterocyclic group; Alk2 is optionally substituted lower alkyl; p is for 2; if there are multiple W1, multiple W3, and multiple R12, each may be independently different; in (xii), the position of an oxygen atom may be cis or trans to a substituent R14, its pharmaceutically acceptable salt, or a solvate thereof, (n) the compound according to (m), wherein ring B is aryl optionally substituted with one or more substituents selected from the group of halogen, hydroxy, optionally substituted lower alkyl, optionally substituted lower alkoxy, optionally substituted acyl, optionally substituted amino, cyano, optionally substituted carbamoyl, an optionally substituted carbocyclic group, optionally substituted carbocyclicoxy or an optionally substituted heterocyclic group, or heteroaryl optionally substituted with one or more substituents selected from the group of halogen, hydroxy, optionally substituted lower alkyl, optionally substituted lower alkoxy, optionally substituted acyl, optionally substituted amino, cyano, optionally substituted carbamoyl, an optionally substituted carbocyclic group, optionally substituted carbocyclicoxy or an optionally substituted heterocyclic group, its pharmaceutically acceptable salt, or a solvate thereof, (o) the compound according to (m), wherein G is represented by the formula:
##STR00017##
wherein, each symbols are the same as described above, its pharmaceutically acceptable salt, or a solvate thereof, (p) the compound according to any one of (e) to (o), wherein R5 is C1 to C3 alkyl, its pharmaceutically acceptable salt, or a solvate thereof, (q) the compound according to any one of (e) to (o), wherein R5 is methyl, its pharmaceutically acceptable salt, or a solvate thereof, (r) the compound according to any one of (e) to (q), wherein R3a and R3b are each independently a hydrogen atom, halogen, hydroxy, optionally substituted lower alkyl, optionally substituted lower alkoxy or optionally substituted aryl, its pharmaceutically acceptable salt, or a solvate thereof, (s) the compound according to any one of (e) to (q), wherein R3a and R3b are both hydrogen atoms, its pharmaceutically acceptable salt, or a solvate thereof, (t) a pharmaceutical composition containing the compound according to any one of (e) to (s), its pharmaceutically acceptable salt, or a solvate thereof as an active ingredient, (u) a composition having BACE 1 inhibitory activity containing the compound according to any one of (e) to (s), its pharmaceutically acceptable salt, or a solvate thereof, (v) a composition having BACE 1 inhibitory activity containing the compound according to any one of (a) to (d) or (u) as amyloid β reducing agent, (w) a composition having BACE 1 inhibitory activity according to any one of (a) to (d), (u) or (v) as therapeutic agent for disease induced by production, secretion and/or deposition of amyloid β protein, (x) a composition having BACE 1 inhibitory activity according to any one of (a) to (d), (u) or (v) as therapeutic agent for Alzheimer's disease. in addition, the present invention provides: (y) a method for treating disease induced by production, secretion and/or deposition of amyloid β protein comprising administering the compound as defined in any one of formula (I) in above (a), its pharmaceutically acceptable salt, or a solvate thereof, (z) use of compound as defined in any one of formula (I) in above (a), its pharmaceutically acceptable salt, or a solvate thereof, in the manufacture of a medicament for the treatment of disease induced by production, secretion and/or deposition of amyloid β protein, (aa) a method for treating Alzheimer's disease characterizing in administering the compound as defined in any one of formula (I) in above (a), its pharmaceutically acceptable salt, or a solvate thereof, (ab) use of compound as defined in any one of formula (I) in above (a), its pharmaceutically acceptable salt, or a solvate thereof, in the manufacture of a medicament for the treatment of Alzheimer's disease.
Effect of the Invention
[0029] The compounds in this invention are useful as an agent for treating disease such as Alzheimer's disease induced by production, secretion and/or deposition of amyloid β protein.
BEST MODE FOR CARRYING OUT THE INVENTION
[0030] As used herein, the "halogen" includes fluorine, chlorine, bromine, and iodine. A halogen part of the "halogeno lower alkyl", the "halogeno lower alkoxy", the "halogeno acyl", the "halogeno lower alkylthio" and the "halogeno lower alkoxycarbonyl" is the same.
[0031] The "lower alkyl" includes a straight or branched alkyl of a carbon number of 1 to 15, preferably a carbon number of 1 to 10, further preferably a carbon number of 1 to 6, and more further preferably a carbon number of 1 to 3, and examples include methyl, ethyl, n-propyl, isopropyl, n-butyl, isobutyl, sec-butyl, tert-butyl, n-pentyl, isopentyl, neopentyl, hexyl, isohexyl, n-heptyl, isoheptyl, n-octyl, isooctyl, n-nonyl, and n-decyl.
[0032] A lower alkyl part of the "carbocyclic lower alkyl", the "lower alkoxy", the "halogeno lower alkyl", the "halogen lower alkoxy", the "halogeno lower alkylthio", the "hydroxy lower alkyl", the "lower alkoxycarbonyl", the "halogen lower alkoxycarbonyl", the "lower alkoxycarbonyl lower alkyl", the "lower alkoxycarbonyloxy", the "lower alkylamino", the "lower alkylcarbonylamino", the "lower alkoxycarbonylamino", the "lower alkoxy lower alkyl", the "lower alkylcarbamoyl", the "hydroxy lower alkylcarbamoyl", the "amino lower alkyl", the "hydroxy imino lower alkyl", the "lower alkoxy imino lower alkyl", the "lower alkylthio", the "lower alkylsulfonyl", the "lower alkyl sulfamoyl", the "lower alkylsulfinyl", the "lower alkylsulfonyloxy", the "lower alkoxycarbonyl lower alkynyl", the "lower alkylthio lower alkyl", the "aryl lower alkyl", the "aryl lower alkylamino", the "aryl lower alkoxycarbonyl", the "aryl lower alkylcarbamoyl", the "heterocyclic group lower alkylamino" and the "heterocyclic group lower alkylcarbamoyl" is the same as that of the aforementioned "lower alkyl".
[0033] The example of the "optionally substituted lower alkyl" as a substituent of ring A is lower alkyl optionally substituted with one or more substituents selected from the "substituent group α", "hydroxyimino" and "lower alkoxyimino"; the group defined as above (i), (ii), (iv), (vi), (viii), (x) (wherein each Q1 is optionally substituted lower alkylene); the group defined as (v), (vii), (ix) (wherein Q2 is optionally substituted lower alkylene); and the group (xii).
[0034] In other "optionally substituted lower alkyl" is optionally substituted with one or more substituents selected from the "substituent group α".
[0035] The "substituent group α" is selected from the group of halogen, hydroxy, lower alkoxy, hydroxy lower alkoxy, lower alkoxy lower alkoxy, acyl, acyloxy, carboxy, lower alkoxycarbonyl, amino, acylamino, lower alkylamino, lower alkylthio, carbamoyl, lower alkylcarbamoyl, hydroxy lower alkylcarbamoyl, sulfamoyl, lower alkylsulfamoyl, lower alkylsulfinyl, cyano, nitro, aryl, and heterocyclic group.
[0036] Especially as a substituent of the "optionally substituted lower alkyl" in Alk2, halogen, hydroxy, lower alkoxy, lower alkoxy lower alkoxy, lower alkoxycarbonyl, amino, acylamino, lower alkylamino and/or lower alkylthio are preferable.
[0037] The example of the "optionally substituted lower alkoxy" as a substituent of ring A is lower alkoxy optionally substituted with one or more substituents selected from the above "substituent group α"; above (iii) wherein Q1 is optionally substituted lower alkylene, Q2 is a bond, W2 is O; above (v) wherein Q1 is optionally substituted lower alkylene, Q2 is a bond, W3 is O; above (vi) wherein Q1 is a bond, Q2 is optionally substituted lower alkylene, W2 is O; or above (xi) wherein Q4 is optionally substituted lower alkylene, W2 is O.
[0038] In other case, the substituents of the "optionally substituted lower alkoxy", the "optionally substituted lower alkoxycarbonyl", the "optionally substituted lower alkoxycarbonyloxy", the "optionally substituted lower alkylsulfonyl", the "optionally substituted lower alkylsulfinyl", the "optionally substituted lower alkylsulfonyloxy" and the "optionally substituted lower alkylthio" are one or more substituents selected from the "substituent group α".
[0039] The "lower alkenyl" includes a straight or branched alkenyl of a carbon number of 2 to 15, preferably a carbon number of 2 to 10, further preferably a carbon number of 2 to 6 and more further preferably a carbon number of 2 to 4 having one or more double bonds at an arbitrary position. Specifically examples include vinyl, allyl, propenyl, isopropenyl, butenyl, isobutenyl, prenyl, butadienyl, pentenyl, isopentenyl, hexenyl, isohexenyl, hexadienyl, heptenyl, octenyl, nonenyl, decenyl, undecenyl, dodenyl, tridecenyl, tetradecenyl, and pentadecenyl.
[0040] The "lower alkynyl" includes a straight or branched alkynyl of a carbon number of 2 to 10, preferably a carbon number of 2 to 8, further preferably a carbon number of 3 to 6, having one or more triple bonds at an arbitrary position. Specifically, examples include ethynyl, propenyl, butynyl, pentynyl, hexynyl, heptynyl, octynyl, nonynyl, and decynyl. These may further have a double bond at an arbitrary position.
[0041] A lower alkynyl part of the "lower alkoxycarbonyl lower alkynyl" is the same as that of above "lower alkynyl".
[0042] The example of the "optionally substituted lower alkenyl" as a substituent of ring A is lower alkenyl optionally substituted with one or more substituents selected from the above "substituent group α"; above (i), (ii), (iv), (vi), (viii) or (x), wherein Q1 is optionally substituted lower alkenylene; (v), (vii) or (ix), wherein Q2 is optionally substituted lower alkenylene.
[0043] In other case, the substituents of the "optionally substituted lower alkenyl" and the "optionally substituted lower alkynyl" are one or more substituents selected from the "substituent group α".
[0044] The example of the "optionally substituted lower amino" as a substituent of ring A is amino optionally substituted with one or more substituents selected from the group of lower alkyl, acyl, hydroxy, lower alkoxy, lower alkoxycarbonyl, a carbocyclic group and a heterocyclic group; (ii), wherein Q1 is a bond; (iv), wherein Q1 is a bond; (v), wherein Q2 is a bond, W3 is NR12; (ix), wherein Q2 is a bond; (xiii); or (xiv).
[0045] The example of the "optionally substituted carbamoyl" as a substituent of ring A is carbamoyl optionally substituted with one or more substituents selected from the group of lower alkyl, acyl, hydroxy, lower alkoxy, lower alkoxycarbonyl, a carbocyclic group and a heterocyclic group; (i), (viii), wherein each Q1 is bond; or (xv).
[0046] In other case, the substituents of the "optionally substituted amino", the "optionally substituted amidino", the "optionally substituted carbamoyl", the "optionally substituted carbamoylcarbonyl", and the "optionally substituted carbamoyloxy" are one or two substituents selected from the group of lower alkyl, acyl, hydroxy, lower alkoxy, lower alkoxycarbonyl, a carbocyclic group and a heterocyclic group, and the like.
[0047] The "acyl" includes acyl of a carbon number of 1 to 10, carbocyclic carbonyl and heterocyclic carbonyl. Specifically, formyl, acetyl, propyonyl, butylyl, isobutylyl, valeryl, pivaloyl, hexanoyl, acryloyl, propioloyl, methacryloyl, crotonoyl, benzoyl, cyclohexanecarbonyl, pyridinecarbonyl, furancarbonyl, thiophenecarbonyl, benzothiazolcarbonyl, pyradinecarbonyl, piperidinecarbonyl, thiomorpholinocarbonyl, and the like.
[0048] The part of the acyl of the "halogenoacyl", the "acylamino" and the "acyloxy" is the same as the aforementioned "acyl".
[0049] The substituent of the "optionally substituted acyl" and "optionally substituted acyloxy" is one or more substituents selected from the group of the "substituent group α". The ring part of the "carbocyclic carbonyl" and the "heterocyclic carbonyl" is optionally substituted with one or more substituents selected from the group of "lower alkyl"; the "substituent group α"; and "lower alkyl substituted with one or more substituents selected from the group of the substituent α".
[0050] The "carbocyclic group" includes cycloalkyl, cycloalkenyl, aryl and non-aromatic fused carbocyclic group.
[0051] The "cycloalkyl" includes a carbocyclic group of a carbon number of 3 to 10, preferably a carbon number of 3 to 8, further preferably a carbon number of 4 to 8, and examples include, cyclopropyl, cyclobutyl, cyclopentyl, cyclohexyl, cycloheptyl, cyclooctyl, cyclononyl, and cyclodecyl, and the like.
[0052] The "cycloalkenyl" includes cycloalkenyl having one or more double bonds at an arbitrary position in a ring of the aforementioned cycloalkyl, and examples include cyclopropenyl, cyclobutenyl, cyclopentenyl, cyclohexenyl, cycloheptynyl, cyclooctynyl, and cyclohexadienyl, and the like.
[0053] The "aryl" includes phenyl, naphthyl, anthryl, and phenanthryl, and the like, and phenyl is particularly preferable.
[0054] The "non-aromatic fused a carbocyclic group" includes group fused with two or more ring groups selected from the group of the above "cycloalkyl", the "cycloalkenyl" and the "aryl". Specifically, examples include indanyl, indenyl, tetrahydronaphthyl, and fluorenyl, and the like.
[0055] The carbocyclic part of the "carbocyclicoxy", and the "carbocyclic lower alkyl" is the same as the aforementioned "carbocyclic group".
[0056] The aryl part of the "aryl lower alkyl", the "aryloxy", the "aryloxycarbonyl", the "aryloxycarbonyloxy", the "aryl lower alkoxycarbonyl", the "arylthio", the "arylamino", the "aryl lower alkylamino", the "arylsulfonyl", the "arylsulfonyloxy", the "arylsulfinyl", the "arylsulfamoyl", the "arylcarbamoyl" and the "aryl lower alkylcarbamoyl" is the same as the aforementioned "aryl".
[0057] The "heterocyclic group" includes a heterocyclic group having one or more heteroatoms arbitrary selected from O, S, and N in a ring, specifically includes a 5- to 6-membered heteroaryl such as pyrrolyl, imidazolyl, pyrazolyl, pyridyl, pyridazinyl, pyrimidinyl, pyrazinyl, triazolyl, triazinyl, tetrazolyl, isoxazolyl, oxazolyl, oxadiazolyl, isothiazolyl, thiazolyl, thiadiazolyl, isothiazolyl, furyl and thienyl; a bicyclic fused heterocyclic group such as indolyl, isoindolyl, indazolyl, indolidinyl, indolinyl, isoindolinyl, quinolyl, isoquinolyl, cinnolinyl, phthalazinyl, quinazolinyl, naphthridinyl, quinoxalinyl, purinyl, pteridinyl, benzopyranyl, benzimidazolyl, benzioxazolyl, benzoxazolyl, benzoxadiazolyl, benzoisothiazolyl, benzothiazolyl, benzothiadiazolyl, benzofuryl, isobenzofuryl, benzothienyl, benzotriazolyl, imidazopyridyl, pyrazolopyridyl, triazolopyridyl, imidazothiazolyl, pyrazinopyridazinyl, quinazolinyl, quinolyl, isoquinolyl, naphthyridinyl, dihydrobenzofuryl, tetrahydroquinolyl, tetrahydroisoquinolyl, dihydrobenzoxazine, tetrahydrobenzothienyl; a tricyclic fused heterocyclic group such as carbazolyl, acridinyl, xanthenyl, phenothiazinyl, phenoxathiinyl, phenoxazinyl, dibenzofuryl, and imidazoquinolyl; a non-aromatic heterocyclic group such as dioxanyl, thiiranyl, oxyranyl, oxathioranyl, azethidinyl, thianyl, pyrrolidinyl, pyrrolinyl, imidazolidinyl, imidazolinyl, pyrazolidinyl, pyrazolinyl, piperidyl, piperazinyl, morpholinyl, morpholino, thiomorpholinyl, thiomorpholino, dihydropyridyl, dihydrobenzoimidazolyl, tetrahydropyridyl, tetrahydrofuryl, tetrahydropyranyl, tetrahydrothiazolyl, tetrahydroisothiazolyl, dihydroxadinyl, hexahydroazepinyl, tetrahydroazepyinyl. Preferable is a 5- to 6-membered heteroaryl, or a non-aromatic heterocyclic group.
[0058] The heterocyclic part of the "heterocyclicoxy", the "heterocyclic thio", the "heterocyclic carbonyl", the "heterocyclic amino", the "heterocyclic carbonylamino", the "heterocyclic sulfamoyl", the "heterocyclic sulfonyl", the "heterocyclic carbamoyl", the "heterocyclicoxycarbonyl", the "heterocyclic lower alkylamino" and the "heterocyclic lower alkyl carbamoyl" is the same as the aforementioned "heterocyclic group".
[0059] The example of the substituent of the "optionally substituted carbocyclic group" and the "optionally substituted heterocyclic group" in ring A is;
the substituent α, wherein preferable is for example, halogen, hydroxy, acyl, acyloxy, carboxy, lower alkoxycarbonyl, carbamoyl, amino, lower alkylamino, lower alkylthio; lower alkyl substituted with one or more substituents selected from the group of substituent α, wherein preferable is halogen, hydroxy, lower alkoxy, lower alkoxycarbonyl, and the like; amino lower alkyl substituted with one or more substituents selected from the group of substituent α, wherein preferable is acyl, lower alkyl and for lower alkoxy, and the like; hydroxyimino lower alkyl; lower alkoxyimino lower alkyl; lower alkenyl substituted with one or more substituents selected from the group of substituent α, wherein preferable is lower alkoxycarbonyl, halogen and for halogeno lower alkoxycarbonyl, and the like; lower alkynyl substituted with one or more substituents selected from the group of substituent α, wherein preferable is for example, lower alkoxycarbonyl, lower alkoxy substituted with one or more substituents selected from the group of substituent α, wherein preferable is for example, lower alkyl carbamoyl and for hydroxy lower alkyl carbamoyl, lower alkylthio substituted with one or more substituents selected from the group of substituent α, lower alkylamino substituted with one or more substituents selected from the group of substituent α, lower alkylsulfonyl substituted with one or more substituents selected from the group of substituent α, aryl lower alkoxycarbonyl substituted with one or more substituents selected from the group of substituent α, azido, and lower alkyl, acyl substituted with one or more substituents selected from the group of substituent α, cycloalkyl substituted with one or more substituents selected from the group of substituent α, azido, and lower alkyl, lower alkylsulfinyl substituted with one or more substituents selected from the group of substituent α, sulfamoyl, aryl substituted with one or more substituents selected from the group of substituent α, azido, and lower alkyl, heterocyclic group substituted with one or more substituents selected from the group of substituent α, azido, and lower alkyl, aryloxy substituted with one or more substituents selected from the group of substituent α, azido, and lower alkyl, heterocyclicoxy substituted with one or more substituents selected from the group of substituent α, azido, and lower alkyl, arylthio substituted with one or more substituents selected from the group of substituent α, azido, and lower alkyl, heteroarylthio substituted with one or more substituents selected from the group of substituent α, azido, and lower alkyl, arylamino substituted with one or more substituents selected from the group of substituent α, azido, and lower alkyl, heterocyclicamino substituted with one or more substituents selected from the group of substituent α, azido, and lower alkyl, aryl lower alkylamino substituted with one or more substituents selected from the group of substituent α, azido, and lower alkyl, heterocyclic lower alkylamino substituted with one or more substituents selected from the group of substituent α, azido, and lower alkyl, lower alkyl sulfamoyl substituted with one or more substituents selected from the group of substituent α, aryl sulfamoyl substituted with one or more substituents selected from the group of substituent α, azido, and lower alkyl, heterocyclic sulfamoyl substituted with one or more substituents selected from the group of substituent α, azido, and lower alkyl, arylsulfonyl substituted with one or more substituents selected from the group of substituent α, azido, and lower alkyl, heterocyclic sulfonyl substituted with one or more substituents selected from the group of substituent α, azido, and lower alkyl, aryl carbamoyl substituted with one or more substituents selected from the group of substituent α, azido, and lower alkyl, heterocyclic carbamoyl substituted with one or more substituents selected from the group of substituent α, azido, and lower alkyl, aryl lower alkylcarbamoyl substituted with one or more substituents selected from the group of substituent α, azido, and lower alkyl, heterocyclic lower alkylcarbamoyl substituted with one or more substituents selected from the group of substituent α, azido, and lower alkyl, aryloxycarbonyl substituted with one or more substituents selected from the group of substituent α, azido, and lower alkyl, heterocyclicoxycarbonyl substituted with one or more substituents selected from the group of substituent α, azido, and lower alkyl, lower alkylenedioxy optionally substituted with halogen; oxo; azido;
##STR00018## ##STR00019## ##STR00020##
wherein Q1, Q2 and Q3 are each independently a bond, optionally substituted lower alkylene or optionally substituted lower alkenylene; Q4 is optionally substituted lower alkylene or optionally substituted lower alkenylene; W1 and W2 are each independently O or S;
W3 is O, S or NR12;
[0060] R12 is a hydrogen atom, lower alkyl, hydroxy lower alkyl, lower alkoxy lower alkyl, lower alkoxycarbonyl lower alkyl, carbocyclic group lower alkyl or acyl; R14 is a hydrogen atom or lower alkyl; ring B is an optionally substituted carbocyclic group or an optionally substituted heterocyclic group; Alk2 is optionally substituted lower alkyl; and the ring A is optionally substituted with one or more substituents selected from these groups.
[0061] If there are multiple W1, multiple W3, and multiple R12, each may be independently different.
[0062] In addition, an oxygen atom in (xii) may be cis or trans position to the substituent R14.
[0063] The substituent of the "substituted phenyl" is, in the same way, phenyl substituted with one or two substituents selected preferably from the group of the substituent α or (i) to (xv).
[0064] The substituent of the "optionally substituted carbocyclic group" or the "optionally substituted heterocyclic group" in ring B is optionally substituted with one or more substituents selected from the following group of, for example;
the substituent α, wherein preferable is halogen, hydroxy, lower alkoxy, carboxy, lower alkoxycarbonyl, acyl, amino, lower alkylamino, acylamino, carbamoyl, lower alkylcarbamoyl, cyano, and nitro, and the like; lower alkyl substituted with one or more substituents selected from the group of the substituent α, wherein preferable is halogen, hydroxy, and lower alkoxy, and the like; amino lower alkyl, hydroxyimino lower alkyl, or lower alkoxyimino lower alkyl, substituted with one or more substituents selected from the group of substituent α; lower alkenyl substituted with one or more substituents selected from the group of substituent α; lower alkynyl substituted with one or more substituents selected from the group of substituent α; lower alkoxy substituted with one or more substituents selected from the group of substituent α, wherein preferable is halogen, hydroxy, and the like; lower alkylthio substituted with one or more substituents selected from the group of substituent α, wherein preferable is halogen; lower alkylamino substituted with one or more substituents selected from the group of substituent α, wherein preferable is amino; lower alkylsulfonyl substituted with one or more substituents selected from the group of substituent α; aryl lower alkoxycarbonyl substituted with one or more substituents selected from the group of substituent α and lower alkyl; acyl substituted with one or more substituents selected from the group of substituent α, wherein preferable is halogen; lower alkylsulfonyl substituted with one or more substituents selected from the group of substituent α; sulfamoyl; lower alkyl sulfamoyl substituted with one or more substituents selected from the group of substituent α; cycloalkyl substituted with one or more substituents selected from the group of substituent α, azido and lower alkyl; aryl substituted with one or more substituents selected from the group of substituent α, azido and lower alkyl; heterocyclic group substituted with one or more substituents selected from the group of substituent α, azido and lower alkyl, wherein preferable is halogen, lower alkyl, and the like; aryloxy substituted with one or more substituents selected from the group of substituent α, azido and lower alkyl; heterocyclicoxy substituted with one or more substituents selected from the group of substituent α, azido and lower alkyl; arylthio substituted with one pr more substituents selected from the group of substituent α, azido and lower alkyl, wherein preferable is halogen, hydroxy, lower alkoxy, acyl, and the like; heterocyclic thio substituted with one or more substituents selected from the group of substituent α, azido and lower alkyl; arylamino substituted with one or more substituents selected from the group of substituent α, azido and lower alkyl, wherein preferable is halogen, hydroxy, lower alkoxy, acyl; heterocyclic amino substituted with one or more substituents selected from the group of substituent α, azido and lower alkyl; aryl lower alkylamino substituted with one or more substituents selected from the group of substituent α, azido and lower alkyl, wherein preferable is halogen, hydroxy, lower alkoxy, acyl; heterocyclic lower alkylamino substituted with one or more substituents selected from the group of substituent α, azido and lower alkyl; arylsulfamoyl substituted with one or more substituents selected from the group of substituent α azido and lower alkyl; heterocyclic sulfamoyl substituted with one or more substituents selected from the group of substituent α, azido and lower alkyl; arylsulfonyl substituted with one or more substituents selected from the group of substituent α, azido and lower alkyl; heterocyclic sulfonyl substituted with one or more substituents selected from the group of substituent α, azido and lower alkyl; arylcarbamoyl substituted with one or more substituents selected from the group of substituent α, ado and lower alkyl; heterocyclic carbamoyl substituted with one or more substituents selected from the group of substituent α, azido and lower alkyl; aryl lower alkylcarbamoyl substituted with one or more substituents selected from the group of substituent α, azido and lower alkyl; heterocyclic lower alkylcarbamoyl substituted with one or more substituents selected from the group of substituent α, azido and lower alkyl; aryloxy carbonyl substituted with one or more substituents selected from the group of substituent α, azido and lower alkyl; heterocyclicoxycarbonyl substituted with one or more substituents selected from the group of substituent α, azido and lower alkyl; lower alkylenedioxy optionally substituted with halogen; oxo; and the like.
[0065] In other case, the substituent of the "optionally substituted carbocyclic group", the "optionally substituted heterocyclic group", the "optionally substituted carbocyclicoxy", the "optionally substituted arylsulfonyl", the "optionally substituted aryloxycarbonyloxy", the "optionally substituted heterocyclicoxy", the "optionally substituted arylsulfinyl", the "optionally substituted arylsulfonyloxy", the "optionally substituted arylthio" is one or more substituents selected from the group of "lower alkyl" and the "substituent α".
"heteroaryl" include aromatic ring group in the aforementioned "heterocyclic group".
[0066] The substituent of the "optionally substituted 5- to 6-membered heteroaryl" is the same as the substituent of the "optionally substituted heterocyclic group" in the aforementioned "ring B". Preferable is one or more substituent selected from lower alkyl and a substituent α.
[0067] The "lower alkylene" includes a straight or branched bivalent carbon chain of a carbon number of 1 to 10, preferably a carbon number of 1 to 6, further preferably a carbon number of 1 to 3. Specifically, examples include methylene, dimethylene, trimethylene, teteramethylene, and methyltrimethylene, and the like.
[0068] The part of lower alkylene of the "lower alkylenedioxy" is the same as the aforementioned "lower alkylene".
[0069] The "lower alkenylene" includes a straight or branched bivalent carbon chain of a carbon number of 2 to 10, preferably a carbon number of 2 to 6, further preferably a carbon number of 2 to 4 having double bond at an arbitrary position. Specifically, examples include vinylene, propenylene, butenylene, butadienylene, methylpropenylene, pentenylene, and hexenylene, and the like.
[0070] The "lower alkynylene" includes a straight or branched bivalent carbon chain of a carbon number of 2 to 10, preferably a carbon number of 2 to 6, further preferably a carbon number of 2 to 4 having triple bond at an arbitrary position. Specifically, examples include ethynylene, propynylene, butynylene, pentynylene, and hexynylene, and the like.
[0071] The substituent of the "optionally substituted lower alkylene", the "optionally substituted lower alkenylene", the "optionally substituted lower alkynylene" is the substituent α, preferable is halogen, hydroxy and the like.
[0072] The "each R3a, each R3b, each R4a, and each R4b may be independently different" means when n is 2 or 3, two or three R3b may be independently different, and two or three R3b may be independently different. In the same way, when m is 2 or 3, two or three R4a may be independently different, and two or three R4b may be independently different.
##STR00021##
R5 and ring A can be taken together to form
##STR00022##
means for example, include the following structures.
##STR00023##
wherein each symbols are the same as described above; preferably, R5a and R5b are all hydrogen atoms.
[0073] In this description, "solvate" includes, for example, a solvate with an organic solvent and a hydrate, and the like. When hydrate is formed, arbitrary number of water molecules may be coordinated.
[0074] The compound (I) includes a pharmaceutically acceptable salt. Examples include salts with alkali metals (lithium, sodium or potassium, and the like), alkaline earth metals (magnesium or calcium, and the like), ammonium, organic bases or amino acids, and salts with inorganic acids (hydrochloric acid, sulfuric acid, nitric acid, hydrobromic acid, phosphoric acid or hydroiodic acid, and the like), and organic acid (acetic acid, trifluoroacetic acid, citric acid, lactic acid, tartaric acid, oxalic acid, maleic acid, fumaric acid, manderic acid, glutaric acid, malic acid, benzoic acid, phthalic acid, benzenesulfonic acid, p-toluenesulfonic acid, methanesulfonic acid, or ethanesulfonic acid, and the like). Particularly, hydrochloric acid, phosphoric acid, tartaric acid, or methanesulfonic acid is preferable. These salts can be formed by a conventional method.
[0075] In addition, the compound (I) is not limited to a specific isomer, but includes all possible isomers (keto-enol isomer, imine-enamine isomer, diastereo isomer, optical isomer, and rotational isomer, and the like) and racemates. For example, the compound (I), wherein R2a is a hydrogen atom, includes following tautomer.
##STR00024##
[0076] The compound (I) in this invention can be prepared by the process described in, for example Non-patent Document 1 or following process.
[0077] The synthesis of aminodihydrothiazine ring; Method A
##STR00025##
[0078] In formula, at least either R2b or R2c is a hydrogen atom, either R3c or R3d is each independently a hydrogen atom, halogen, hydroxy, optionally substituted lower alkyl, optionally substituted lower alkenyl, optionally substituted acyl, carboxy, optionally substituted lower alkoxycarbonyl, optionally substituted amino, optionally substituted carbamoyl, an optionally substituted carbocyclic group or an optionally substituted heterocyclic group. Other symbols are the same as described above.
(Step 1)
[0079] To a solution of compound (a), which is commercially available or prepared by known method, in appropriate solvent or mixture of solvents, such as ether, tetrahydrofuran, and the like is added the Grignard reagent having substituent corresponds to the target compound; for example vinylmagnesium chloride, vinylmagnesium bromide, or propenylmagnesium bromide, and the like; at -100° C. to 50° C., preferably -80° C. to 0° C. The mixture is reacted for 0.2 to 24 hours, preferably 0.5 to 5 hours, to obtain compound (b).
(Step 2)
[0080] The compound (b) in solvent, such as toluene or absence of solvent is treated with thiourea derivatives having substituent corresponds to the target compound, such as thiourea, N-methylthiourea, N,N'-dimethylthiourea, and the like in the presence of an acid or mixture of acids, such as acetic acid, trifluoroacetic acid, hydrochloric acid, or sulfuric acid, and the like. The mixture is reacted at -20° C. to 100° C., preferably 0° C. to 50° C. for 0.5 to 120 hours, preferably 1 to 72 hours, to obtain the compound (c).
(Step 3)
[0081] The compound (c) in solvent, such as toluene or absence of solvent is treated with an acid or mixture of acids, such as trifluoroacetic acid, methanesulfonic acid, trifluoromethanesulfonic acid, and the like. The mixture is reacted at -20° C. to 100° C., preferably 0° C. to 60° C. for 0.6 to 120 hours, preferably 1 to 72 hours, to obtain the compound (I-2), wherein R2b is a hydrogen atom, or the compound (I-1), wherein R2c is a hydrogen atom.
[0082] The synthesis of aminodihydrothiazine ring; Method B
##STR00026##
[0083] In formula, L is leaving group such as halogen or sulfonyloxy, and the like. Other symbols are the same as described above.
(Step 1)
[0084] The compound (d) which is commercially available or prepared by known method is reacted with thiocyanic acid; for example, sodium thiocyanic acid, ammonium thiocyanic acid, and the like; in solvent; for example, toluene, chloroform, tetrahydrofuran, and the like; in the presence of acid; for example, water, hydrochloric acid, sulfuric acid, and the like; at 0° C. to 150° C., preferably 20° C. to 100° C. for 0.5 to 24 hours, preferably 1 to 12 hours, to obtain the compound (e).
(Step 2)
[0085] To the compound (e) in solvent or mixture of solvents; for example, tetrahydrofuran, methanol, ethanol, water, and the like; in the presence or the absence of buffer like sodium dihydorgen phosphate, and the like; reducing agent; for example sodium borohydride, and the like; is added and the mixture is reacted at -80° C. to 50° C., preferably -20° C. to 20° C. for 0.1 to 24 hours, preferably 0.5 to 12 hours, to obtain the compound (f).
(Step 3)
[0086] The compound (f) in the presence or the absence of solvent; for example, toluene, dichloromethane, and the like; is reacted with halogenating agent; for example thionyl chloride, phosphorus oxychloride, carbon tetrabromide-triphenylphosphine, and the like; at -80° C. to 50° C., preferably -20° C. to 20° C. for 0.1 to 24 hours, preferably 0.5 to 12 hours, to obtain the compound (g). Alternatively, the compound (f) in the presence or the absence of solvent; for example, toluene, dichloromethane, and the like; under base; for example triethylamine, and the like; is reacted with sulfonating agent; for example, methanesulfonyl chloride, p-toluenesulfonylchloride, and the like; at -80° C. to 50° C., preferably -20° C. to 20° C. for 0.1 to 24 hours, preferably 0.5 to 12 hours, to obtain the compound (g).
(Step 4)
[0087] To the compound (g) in solvent or mixture of solvents, for example methanol, ethanol, water, and the like; is reacted with primary amine; for example, ammonia or methylamine, and the like; at -20° C. to 80° C., preferably 0° C. to 40° C. for 0.5 to 48 hours, preferably 1 to 24 hours, to obtain the compound (I-3).
[0088] The synthesis of aminodihydrothiazine ring; Method C
##STR00027##
[0089] In formula, R is a hydrogen atom or protective groups of carboxyl group. Other symbols are the same as described above.
(Step 1)
[0090] The compound (h) which is commercially available or prepared by known method is reacted with reducing agent; for example, lithium aluminium hydride, diisobutyl aluminium hydride, and the like; in solvent; for example tetrahydrofuran, ether, and the like; at -80° C. to 150° C., preferably 25° C. to 100° C. for 0.1 to 24 hours, preferably 0.5 to 12 hours, to obtain the compound (i).
(Step 2)
[0091] The compound (i) in solvent; for example, toluene, chloroform, tetrahydrofuran, and the like; in the presence or the absence of base; for example, diisopropylethylamine, triethylamine, pyridine, sodium hydroxide, and the like; is reacted with corresponding isothiocyanate; for example, 4-methoxybenzylisothiocyanate, t-butylisothiocyanate, and the like; or corresponding thiocarbamoylhalide; for example, N,N-dimethylthiocarbamoylchloride, N,N-diethylthiocarbamoylchloride, and the like; at 0° C. to 150° C., preferably 20° C. to 100° C. for 0.5 to 120 hours, preferably 1 to 72 hours, to obtain the compound (j).
(Step 3)
[0092] The compound (j) in solvent; for example, acetonitrile, toluene, dichloromethane, and the like; is reacted with halogenating agent; for example thionyl chloride, phosphorus oxychloride, carbon tetrabromide-triphenylphosphine, and the like; at -80° C. to 50° C., preferably -20° C. to 20° C. for 0.1 to 24 hours, preferably 0.5 to 12 hours, or alternatively, the compound (j) in solvent; for example, toluene, dichloromethane, and the like; in the presence of base; for example triethylamine, and the like; is reacted with sulfonating agent; for example, methanesulfonyl chloride, p-toluenesulfonylchloride, and the like; at -80° C. to 50° C., preferably -20° C. to 20° C. for 0.1 to 24 hours, preferably 0.5 to 12 hours. The obtained halogenated compound or sulfonylated compound is reacted with base; for example, diisopropylamine, potassium carbonate, sodium hydrogencarbonate, sodium hydride, sodium hydroxide, and the like; at 0° C. to 150° C., preferably 20° C. to 100° C. for 0.5 to 120 hours, preferably 1 to 72 hours, to obtain the compound (I-4).
[0093] The synthesis of aminodihydrothiazine ring; Method D
[0094] The synthesis of aminothiazoline ring; Method A
[0095] The synthesis of tetrahydrothiazepine ring; Method A
##STR00028##
[0096] In formula, L is leaving group such as halogen or sulfonyloxy, and the like; m is an integer of 1 to 3; and the other symbols are the same as described above.
(Step 1)
[0097] The compound (k) which is commercially available or prepared by known method is reacted with azide reagent; for example, sodium azide, and the like; in solvent; for example N,N-dimethylformamide, tetrahydrofuran, and the like; at 0° C. to 200° C., preferably 40° C. to 150° C. for 0.5 to 24 hours, preferably 1 to 12 hours, to obtain the compound (l).
(Step 2)
[0098] The compound (l) is reacted with reducing agent; for example, lithium aluminium hydride, diisobutyl aluminium hydride, and the like; in solvent; for example tetrahydrofuran, ether, and the like; at -80° C. to 150° C., preferably 25° C. to 100° C. for 0.1 to 24 hours, preferably 0.5 to 12 hours, to obtain the compound (m).
(Step 3)
[0099] The compound (m) in solvent; for example, toluene, chloroform, tetrahydrofuran, and the like; is reacted with corresponding isothiocyanate; for example, methylisothiocyanate, ethylisothiocyanate, and the like; or corresponding thiocarbamoylhalide; for example, N,N-dimethylthiocarbamoylchloride, N,N-diethylthiocarbamoylchloride, and the like; at 0° C. to 150° C., preferably 20° C. to 100° C. for 0.5 to 120 hours, preferably 1 to 72 hours, to obtain the compound (n).
(Step 4)
[0100] The compound (n) in solvent; for example, acetonitrile, toluene, dichloromethane and the like; is reacted with halogenating agent; for example thionyl chloride, phosphorus oxychloride, carbon tetrabromide-triphenylphosphine, and the like; at -80° C. to 50° C., preferably -20° C. to 20° C. for 0.1 to 24 hours, preferably 0.5 to 12 hours, or alternatively, the compound (n) in solvent; for example, toluene, dichloromethane, and the like; in the presence of base; for example diisopropylethylamine, triethylamine, and the like; is reacted with sulfonating agent; for example, methanesulfonyl chloride, p-toluenesulfonylchloride, and the like; at -80° C. to 50° C., preferably -20° C. to 20° C. for 0.1 to 24 hours, preferably 0.5 to 12 hours. The obtained halogenated compound or sulfonylated compound is reacted with base; for example, diisopropylamine, potassium carbonate, sodium hydrogencarbonate, sodium hydride, sodium hydroxide, and the like; at 0° C. to 150° C., preferably 20° C. to 100° C. for 0.5 to 120 hours, preferably 1 to 72 hours, to obtain the compound (I-5).
[0101] The synthesis of aminodihydrothiazine ring; Method E
[0102] The synthesis of aminothiazoline ring; Method B
[0103] The synthesis of tetrahydrothiazepine ring; Method B
##STR00029##
[0104] In formula, at lease one of R2b and R2c is a hydrogen atom, n is an integer of 1 to 3, and the other symbols are the same as described above.
(Step 1)
[0105] The compound (o) which is commercially available or prepared by known method is reacted with substituted thiourea; for example, thiourea, N-methylthiourea, N,N,-dimethylthiourea, N,N'-dimethylthiourea, and the like; in solvent; for example, ethanol, methanol, tetrahydrofuran, toluene, and the like; at -20° C. to 200° C., preferably 0° C. to 150° C. for 0.5 to 200 hours, preferably 1 to 120 hours, to obtain the compound (p).
(Step 2)
[0106] To the compound (p) in solvent or mixture of solvents; for example, ether, tetrahydrofuran, and the like; the Grignard reagent having substituent corresponding to target compound; for example methylmagnesium chloride, ethylmagnesium bromide, or benzylmagnesium bromide, and the like; is added at -100° C. to 50° C., preferably -80° C. to 30° C., and the mixture is reacted for 0.2 to 24 hours, preferably 0.5 to 5 hours, to obtain the compound (q).
(Step 3)
[0107] To the compound (q) in the presence or the absence of solvent; for example, toluene, and the like; acid or mixture of acids, such as trifluoroacetic acid, methanesulfonic acid, trifluoromethanesulfonic acid, and the like; is added and the mixture is reacted at -20° C. to 100° C., preferably 0° C. to 50° C. for 0.5 to 200 hours, preferably 1 to 150 hours, to obtain the compound (I-6) (wherein R2c is H), or the compound (I-7) (wherein R2b is H).
[0108] The synthesis of aminodihydrothiazine ring; Method F
##STR00030##
[0109] In formula, each symbols are the same as described above.
(Step 1)
[0110] The compound (r) which is commercially available or prepared by known method is reacted with ammonium chloride in solvent; for example, acetic acid, and the like; at 0° C. to 200° C., preferably 10° C. to 100° C. for 0.1 to 100 hours, preferably 0.5 to 24 hours, to obtain the compound (s).
(Step 2)
[0111] The compound (s) is reacted with reducing agent; for example, lithium aluminium hydride, diisobutyl aluminium hydride, and the like; in solvent; for example trahydrofuran, ether, and the like; at -80° C. to 150° C., preferably 0° C. to 100° C. for 0.1 to 24 hours, preferably 0.5 to 12 hours, to obtain the compound (t).
(Step 3)
[0112] The compound (t) in solvent; for example, toluene, chloroform, tetrahydrofuran, and the like; in the presence or the absence of base; for example, diisopropylethylamine; triethylamine, pyridine, sodium hydroxide, and the like; is reacted with corresponding isothiocyanate; for example, 4-methoxybenzylisothiocyanate, t-butylisothiocyanate, and the like; or corresponding carbamoylhalide; for example, N,N-dimethylthiocarbamoylchloride, N,N-diethylthiocarbamoylchloride, and the like; at 0° C. to 150° C., preferably 20° C. to 100° C. for 0.5 to 120 hours, preferably 1 to 72 hours, to obtain the compound (u).
(Step 4)
[0113] The compound (u) in solvent; for example, acetonitrile, toluene, dichloromethane, and the like; is reacted with halogenating agent; for example thionyl chloride, phosphorus oxychloride, carbon tetrabromide-triphenylphosphine, and the like; at -80° C. to 50° C., preferably -20° C. to 20° C. for 0.1 to 24 hours, preferably 0.5 to 12 hours, or alternatively, the compound (u) in solvent; for example, toluene, dichloromethane, and the like; in the presence of base; for example triethylamine, and the like; is reacted with sulfonating agent; for example, methanesulfonyl chloride, p-toluenesulfonylchloride, and the like; at -80° C. to 50° C., preferably -20° C. to 20° C. for 0.1 to 24 hours, preferably 0.5 to 12 hours. The obtained halogenated compound or sulfonylated compound is reacted with base; for example, diisopropylamine, potassium carbonate, sodium hydrogencarbonate, sodium hydride, sodium hydroxide, and the like; at 0° C. to 150° C., preferably 20° C. to 100° C. for 0.5 to 120 hours, preferably 1 to 72 hours, to obtain the compound (I-8).
[0114] The synthesis of aminodihydrooxazine ring; Method A
[0115] The synthesis of aminotetrahydrooxazepine ring; Method A
##STR00031##
[0116] In formula, each symbols are the same as described above.
(Step 1)
[0117] The compound (n) which is obtained by Step 3 (the compound (m) to the compound (n)) of "The synthesis of aminodihydrothiazine ring; Method D", in solvent; for example, methanol, ethanol, N,N-dimethylformamide, tetrahydrofuran, and the like; in the presence or the absence of base; for example, diisopropylethylamine, triethylamine, pyridine, sodium hydroxide, and the like; is reacted with alkylating agent; for example, methyl iodide, dimethyl sulfate, benzyl bromide, and the like; at 0° C. to 200° C., preferably 40° C. to 150° C. for 0.1 to 48 hours, preferably 0.5 to 24 hours, to obtain the compound (v).
(Step 2)
[0118] The compound (v) in solvent; for example, N, N-dimethylformamide, tetrahydrofuran, dichloromethane, and the like; in the presence or the absence of base; for example, diisopropylethylamine, triethylamine, pyridine, sodium hydroxide, and the like; is reacted with metallic oxide; for example, silver oxide, mercury oxide, manganese dioxide, and the like; at 0° C. to 200° C., preferably 10° C. to 150° C. for 1 to 120 hours, preferably 0.5 to 100 hours, to obtain the compound (I-9).
[0119] The synthesis of aminodihydrooxazine ring; Method B
[0120] The synthesis of aminoxazoline ring
[0121] The synthesis of aminotetrahydrooxazepine ring; Method B
##STR00032##
[0122] In formula, R15 is optionally substituted lower alkyl; for example, t-butyl, benzyl, and the like; R16 is hydrogen atom or lower alkyl; n is an integer of 1 to 3, and the other symbols are the same as described above.
(Step 1)
[0123] The compound (w) which is commercially available or prepared by known method in solvent; for example, toluene, t-butylalcohol, tetrahydrofuran, and the like; in the presence of base; for example, diisopropylethylamine, triethylamine, pyridine, and the like; is reacted with azide reagent; for example, diphenyl phosphoryl azide, and the like; at 0° C. to 200° C., preferably 40° C. to 150° C. for 1 to 48 hours, preferably 0.5 to 24 hours, to obtain the compound (x).
(Step 2)
[0124] The compound (x) in solvent; for example, toluene, xylene, N,N-dimethylformamide, tetrahydrofuran, and the like; is reacted with alcohol; for example, t-butylalcohol, 3,4-dimethoxybenzylalcohol, 4-methoxybenzylalcohol, and the like; at 0° C. to 300° C., preferably 50° C. to 200° C. for 1 to 800 hours, preferably 5 to 500 hours, to obtain the compound (y).
(Step 3)
[0125] The compound (y) in the presence or the absence of solvent; for example, water, toluene, dichloromethane, methanol, 1,4-dioxane, acetic acid, ethyl acetate, and the like; in the presence of acid; for example, hydrochloric acid, sulfuric acid, hydrobromic acid, trifluoroacetic acid, and the like; at 0° C. to 200° C., preferably 25° C. to 150° C. for 0.1 to 48 hours, preferably 0.5 to 24 hours, to obtain the compound (z).
(Step 4)
[0126] The compound (z) in solvent; for example, toluene, chloroform, tetrahydrofuran, and the like; in the presence of base; for example, diisopropylethylamine, triethylamine, pyridine, and the like; is reacted with corresponding isothiocyanate, or thiocarbamoylhalide corresponding to target compound; for example, N,N-dimethylthiocarbamoylchloride, N,N-diethylthiocarbamoylchloride, and the like; at 0° C. to 150° C., preferably 20° C. to 100° C. for 0.5 to 120 hours, preferably 1 to 72 hours, to obtain the compound (aa).
(Step 5)
[0127] The compound (aa) in solvent; for example, methanol, ethanol, N,N-dimethylformamide, tetrahydrofuran, and the like; in the presence or the absence of base; for example, diisopropylethylamine, triethylamine, pyridine, sodium hydroxide, and the like; is reacted with alkylating agent; for example, methyl iodide, dimethyl sulfate, benzyl bromide, and the like; at 0° C. to 200° C., preferably 40° C. to 150° C. for 1 to 48 hours, preferably 0.5 to 24 hours, to obtain the compound (ab).
(Step 6)
[0128] The compound (ab) in solvent; for example, N,N-dimethylformamide, tetrahydrofuran, dichloromethane, and the like; in the presence of base; for example, diisopropylethylamine, triethylamine, pyridine, sodium hydroxide, and the like; is reacted with metallic oxide; for example, silver oxide, mercury oxide, manganese dioxide, and the like; at 0° C. to 200° C., preferably 10° C. to 150° C. for 1 to 120 hours, preferably 0.5 to 100 hours, to obtain the compound (I-10).
[0129] The synthesis of aminotetrahydropyrimidine ring
##STR00033##
[0130] In formula, each symbols are the same as described above.
(Step 1)
[0131] To the compound (ac) prepared by known method in solvent; for example, N,N-dimethylformamide, methanol, and the like; is reacted with azide reagent; for example, sodium azide, lithium azide, and the like; at 20° C. to 150° C., preferably 50° C. to 100° C. for 0.5 to 120 hours, preferably 1 to 72 hours, to obtain the compound (ad).
(Step 2)
[0132] To the suspension of lithium aluminium hydride in solvent; for example, tetrahydrofuran, or ether, and the like; the compound (ad) dissolved in solvent; for example, tetrahydrofuran, or diethyl ether, and the like; is added under nitrogen atmosphere, at -80° C. to 20° C., preferably -30° C. to 0° C., and the mixture is reacted for 1 minute to 10 hours, preferably 10 minutes to 1 hour, or alternatively to the compound (ad) in solvent; for example, ethanol, isopropanol, or n-butanol, and the like; Raney-Nickel is added at 10° C. to 110° C., preferably 50° C. to 80° C., and reacted for 1 minute to 10 hours, preferably 10 minutes to 1 hour, to obtain the compound (ae).
(Step 3)
[0133] The compound (ae) in solvent; for example, tetrahydrofuran, dichloromethane, and the like; in the presence of acid; for example, acetic acid, or propionic acid, and the like; is reacted with reducing agent; for example, sodium cyanoborohydride, sodium triacetoxyborohydride, and the like; at -50° C. to 100° C., preferably 0° C. to 50° C., for 0.1 to 48 hours, preferably 0.5 to 24 hours, or the compound (ae) in solvent; for example, tetrahydrofuran, N,N-dimethylformamide, and the like; in the presence of dehydrating agent; for example, 1-ehthyl-3-(3-dimethylaminopropyl)carbodiimide-N-hydroxybenzotriazole, carbonyldiimidazole, and the like; or in the presence of base; for example, triethylamine, potassium carbonate, and the like; is reacted with carboxylic acid; for example, formic acid, acetic acid, and the like; at -50° C. to 100° C., preferably 0° C. to 50° C. for 0.1 to 48 hours, preferably 0.5 to 16 hours, to obtain the compound (a0. And next, to the suspension of lithium aluminium hydride in solvent; for example, tetrahydrofuran, or diethyl ether, and the like; the aforementioned amide compound dissolved in solvent; for example, tetrahydrofuran, or ether, and the like; is added at -50° C. to 60° C., preferably 0° C. to 50° C., and the mixture is reacted for 1 minute to 48 hours, preferably 10 minutes to 10 hours, to obtain the compound (af).
(Step 4)
[0134] The compound (ae) or the compound (af) in solvent; for example, acetonitrile, tetrahydrofuran, N,N-dimethylformamide, and the like; is reacted with 3,5-dimethylpyrazole-1-carboxyamidine or S-methylthiourea at 0° C. to 150° C., preferably 20° C. to 100° C., and the mixture is reacted for 0.5 to 120 hours, preferably 1 to 24 hours, to obtain the compound (ag).
(Step 5)
[0135] To the compound (ag) (wherein at least either R2b or R2c is a hydrogen atom) in the presence or the absence of solvent; for example, toluene, and the like; acid; for example, trifluoroacetic acid, methanesulfonic acid, trifluoromethanesulfonic acid, and the like, or the mixture thereof; is added and the mixture is reacted at -20° C. to 100° C., preferably 0° C. to 50° C., and the mixture is reacted for 0.5 to 120 hours, preferably 1 to 72 hours, to obtain the compound (I-2) (wherein R2b is a hydrogen atom) or the compound (I-1) (wherein R2c is a hydrogen atom) respectively. Proviso, if R2a, R2b, and R2c have fragile structure under acidic condition; for example, t-butyloxycarbonyl, and the like; R2a, R2b, and R2c in the compound (I-1) or the compound (I-2) may be transformed into a hydrogen atom.
[0136] The synthesis of aminothiazoline ring; Method C
##STR00034##
[0137] In formula, Hal is halogen, and other symbols are the same as described above.
(Step 1)
[0138] The compound (ah) which is commercially available or prepared by known method in solvent; for example, toluene, chloroform, tetrahydrofuran, and the like; or in mixed-solvent; for example, chloroform-water, and the like; is reacted with halogen; for example, including iodine, bromine, chorine; phase transfer catalyst; for example, sodium thiocyanic acid, ammonium thiocyanic acid, and the like; at 0° C. to 150° C., preferably 20° C. to 100° C., for 0.5 to 48 hours, preferably 1 to 24 hours, to obtain the compound (ai).
(Step 2)
[0139] The compound (ai) in solvent; for example, toluene, chloroform, tetrahydrofuran, and the like; is reacted with amine having substituent corresponding to target compound; for example ammonia, methylamine, diethylamine, and the like; at 0° C. to 150° C., preferably 20° C. to 100° C., for 0.5 to 48 hours, preferably 1 to 24 hours, to obtain the compound (I-11).
[0140] The aminoacyl derivative-1
##STR00035##
[0141] In formula, R17 is optionally substituted lower alkyl, an optionally substituted carbocyclic group or an optionally substituted heterocyclic group, and the other symbols are the same as described above.
[0142] The compound (I-12) wherein R2b is a hydrogen atom in the presence or the absence of solvent; for example, tetrahydrofuran, dichloromethane, and the like; in the presence of base; for example, pyridine, triethylamine, and the like; is reacted with acylating agent having substituent corresponding to target compound; for example, benzoyl chloride, 2-furoyl chloride, acetic anhydride, and the like; at -80° C. to 100° C., preferably -20° C. to 40° C., for 0.1 to 24 hours, preferably 1 to 12 hours, or alternatively, the compound (I-12) in solvent; for example, N,N-dimethylformamide, tetrahydrofuran, dichloromethane, and the like; in the presence of dehydrating agent; for example, dicyclohexylcarbodiimide, carbonyldiimidazole, and the like; is reacted with carboxylic acid having substituent corresponding to target compound; for example, amino acid, glycolic acid, and the like; at -80° C. to 100° C., preferably -20° C. to 40° C., for 0.1 to 24 hours, preferably 1 to 12 hours, to obtain the compound (I-13) and/or the compound (I-14) (wherein R2a is a hydrogen atom).
[0143] The guanidino derivative
##STR00036##
[0144] In formula, each symbols are the same as described above.
[0145] The compound (I-12) wherein R2b is a hydrogen atom in solvent; for example, acetonitrile, tetrahydrofuran, N,N-dimethylformamide, and the like; in the presence or the absence of base; for example, triethylamine, sodium hydrogencarbonate, and the like; is reacted with 3,5-dimethylpyrazole-1-carboxyamidine, or S-methylisothiourea etc. at 0° C. to 150° C., preferably 20° C. to 100° C., for 0.5 to 120 hours, preferably 1 to 24 hours, to obtain the compound (I-15).
[0146] The carbamoyl derivatives
##STR00037##
[0147] In formula, CONR18R19 is optionally substituted carbamoyl, and the other symbols axe the same as described above.
[0148] The compound (I-16) having a carboxyl group as substituent of ring A in solvent; for example, N,N-dimethylformamide, tetrahydrofuran, dichloromethane, and the like; in the presence of dehydrating agent; for example, dicyclohexylcarbodiimide, carbonyldiimidazole, dicyclohexylcarbodiimide-N-hydroxybenzotriazole, and the like; is reacted with primary amine or secondary amine (aniline, 2-aminopyridine, dimethylamine etc.) at -80° C. to 100° C., preferably -20° C. to 40° C., for 0.1 to 24 hours, preferably 1 to 12 hours, to obtain the compound (I-17).
[0149] The acylamino derivative-2
##STR00038##
[0150] In formula, NHR20 is optionally substituted amino; NR20COR21 is optionally substituted acyl amino, optionally substituted ureido, carboxy amino having substituent on oxygen atom, and the other symbols are the same as described above.
[0151] The compound (I-18) having an optionally substituted amino group on ring A in the presence or the absence of solvent; for example, tetrahydrofuran, dichloromethane, and the like; in the presence or the absence of base; for example, pyridine, triethylamine, and the like; is reacted with reagent including acid chloride, acid anhydride, chloroformate ester derivatives, isocyanate derivatives (benzoyl chloride, 2-furoyl chloride, acetic anhydride, benzyl chloroformate, di-t-butyl dicarbonate, phenyl isocyanate etc.), at -80° C. to 100° C., preferably -20° C. to 40° C., for 0.1 to 24 hours, preferably 1 to 12 hours. Or alternatively, the compound (I-18) having an optionally substituted amino group on ring A in solvent; for example, N,N-dimethylformamide, tetrahydrofuran, dichloromethane, and the like; in the presence of dehydrating agent; for example, dicyclohexylcarbodiimide, carbonyldiimidazole, dicyclohexylcarbodiimide-N-hydroxybenzotriazole, and the like; is reacted with carboxylic acid having substituent corresponding to target compound; for example, benzoic acid, 2-pyridinecarboxylic acid, and the like; at -80° C. to 100° C., preferably -20° C. to 40° C., for 0.1 to 24 hours, preferably 1 to 12 hours, to obtain the compound (I-19).
[0152] The alkylamino derivatives
##STR00039##
[0153] In formula, NHR22 is optionally substituted amino, R22 is lower alkyl.
[0154] The compound (I-18) having an amino group on ring A in solvent; for example, dichloromethane, tetrahydrofuran, and the like; in the presence or the absence of acid; for example, acetic acid, and the like; is reacted with aldehyde having substituent corresponding to target compound; for example, benzaldehyde, pyridine-2-carboaldehyde, and the like; and reducing agent; for example, sodium borohydride, sodium triacetoxyborohydride, and the like; at -80° C. to 100° C., preferably 0° C. to 40° C., for 0.5 to 150 hours, preferably 1 to 24 hours, to obtain the compound (I-20).
[0155] The substituted alkoxy derivatives
##STR00040##
[0156] In formula, R23 is optionally substituted lower alkyl, an optionally substituted carbocyclic group or an optionally substituted heterocyclic group, etc., and the other symbols are the same as described above.
[0157] The compound (I-21) having a hydroxy group as substituent of A ring in solvent; for example, N,N-dimethylformamide, tetrahydrofuran, and the like; in the presence of base; for example potassium carbonate, sodium hydroxide, sodium hydride, and the like; is reacted with alkylating agent having substituent corresponding to target compound; for example, benzylchloride, methyl iodide, and the like; at -80° C. to 100° C., preferably 0° C. to 40° C., for 0.5 to 150 hours, preferably 1 to 24 hours, or alternatively, the compound (I-18) in solvent; for example, N,N-dimethylformamide, tetrahydrofuran, and the like; under Mitsunobu reagent; for example triphenylphosphine-azodicarboxylic acid ethyl ester, and the like; is reacted with alcohol; for example, 2-aminoethanol, and the like; at -80° C. to 100° C., preferably 0° C. to 40° C., for 0.5 to 72 hours, preferably 1 to 24 hours, to obtain the compound (I-22).
[0158] The introduction of substituent with palladium coupling reaction
##STR00041##
[0159] In formula, Hal is halogen, G is optionally substituted lower alkenyl, optionally substituted lower alkynyl, optionally substituted lower alkoxycarbonyl, an optionally substituted carbocyclic group or an optionally substituted heterocyclic group etc., and the other symbols are the same as described above.
[0160] The compound (I-23) having halogen as substituent of A ring in solvent; for example, tetrahydrofuran, N,N-dimethylformamide, 1,2-dimethoxyethane, methanol, and the like; in the presence of base; for example, triethylamine, sodium carbonate, and the like; palladium catalyst; for example, palladium acetate, palladium chloride, and the like; and ligand; for example triphenylphosphine, and the like; is reacted with compound having substituent corresponding to target compound (styrene, propargyl alcohol, aryl boronic acid, carbon monoxide), with or without microwave irradiation, at -80° C. to 150° C., preferably 0° C. to 100° C., for 0.5 to 72 hours, preferably 1 to 24 hours, to obtain the compound (I-24).
[0161] The oxime derivatives
##STR00042##
[0162] In formula, in R24 is a hydrogen atom or optionally substituted lower alkyl etc., R25 is a hydrogen atom, optionally substituted lower alkyl, optionally substituted lower alkenyl or an optionally substituted carbocyclic group or an optionally substituted heterocyclic group etc., and the other symbols are the same as described above.
[0163] The compound (I-25) having an acyl group as substituent of A ring in solvent; for example, methanol, ethanol, and the like; in the presence or the absence of additives; for example, potassium acetate, and the like; is reacted with hydroxylamine having substituent corresponding to target compound (hydroxylamine, methoxylamine, O-benzylhydroxylamine, etc.) or the salt thereof at 0° C. to 100° C., preferably 0° C. to 40° C., for 0.5 to 150 hours, preferably 1 to 72 hours, to obtain the compound (I-26).
[0164] In all of above mentioned steps, if a compound having substituent which interrupts the reaction; (for example, hydroxy, mercapto, amino, formyl, carbonyl, carboxyl, etc.), the substituent of the compound is protected by methods described in Protective Groups in Organic Synthesis, Theodora W Green (John Wiley & Sons) beforehand, and is deprotected at preferable step.
[0165] The compound (I) in this invention presented below; in particular, X is S, and E is a bond or methylene; is preferable.
1) A compound represented by the general formula (I'),
##STR00043##
in formula, t is 0 or 1, the other symbols are the same as above (a), with the proviso that the compounds represented below; i) wherein n+m is 2, R5 is a hydrogen atom, and ring A is unsubstituted phenyl; ii) wherein n is 2, m is 0, R2a is a hydrogen atom, R2b is a hydrogen atom or acetyl, R5 is methyl, and ring A is phenyl or 4-methoxyphenyl; iii) Wherein n is 2, m is 0, R2a is a hydrogen atom, R2b is a hydrogen atom or acetyl, R5 is ethyl, and ring A is 3,4-dimethoxyphenyl; iv) wherein n is 2, m is 0, R2a is a hydrogen atom, R2b is a hydrogen atom or acetyl, R5 and ring A are phenyl; v) wherein n is 2, m is 0, R2a and R2b is a hydrogen atom, R5 and ring A are taken together to form
##STR00044##
and vi) the compound, wherein n+m is 1 or 2; R5 is a hydrogen atom; ring A is phenyl substituted by one or two substituent selected from hydroxy, halogen, lower alkyl, lower alkoxy, nitro, amino, lower alkyl carbonylamino, mercapto, lower alkylthio, carbamoyl, lower alkylamino, lower alkyl carbamoyl and lower alkoxycarbonyl; non-substituted phenyl, or non-substituted naphthyl; are excluded.
[0166] In addition, in formula (I'), preferable is the compound represented below.
2) The compound, wherein n is 1 and m is 0 (this compound is represented by nm-1), 3) the compound, wherein n is 2 and m is 0 (this compound is represented by nm-2), 4) the compound, wherein n is 3 and m is 0 (this compound is represented by nm-3), 5) the compound, wherein R2a is a hydrogen atom; R2b is a hydrogen atom, optionally substituted lower alkyl, optionally substituted acyl, optionally substituted lower alkylsulfonyl, or optionally substituted amidino (this compound is represented by R2-1), 6) the compound, wherein R2a is a hydrogen atom; R2b is a hydrogen atom, optionally substituted lower alkyl or optionally substituted acyl (this compound is represented by R2-2), 7) the compound, wherein NR2aR2b is represented by the following formula:
##STR00045##
wherein each symbols are the same as described above. R6, R7 and R8 are each independently a hydrogen atom, lower alkyl or acyl, Y is optionally substituted lower alkylene, optionally substituted lower alkenylene or optionally substituted lower alkynylene, Z is O or S (this compound is represented by R2-3), 8) the compound, wherein NR2aR2b is NH2 (this compound is represented by R2-4), 9) the compound, wherein ring A is substituted phenyl or substituted pyridyl (this compound is represented by A-1), 10) the compound, wherein ring A is represented by the following formula:
##STR00046##
wherein R9, R10 and R11 is a hydrogen atom or G, G is halogen, hydroxy, cyano, nitro, mercapto, optionally substituted lower alkyl, optionally substituted lower alkoxy, optionally substituted lower alkenyl, optionally substituted lower alkynyl, optionally substituted acyl, optionally substituted acyloxy, carboxy, optionally substituted lower alkoxycarbonyl, optionally substituted lower alkoxycarbonyloxy, optionally substituted aryloxycarbonyloxy, optionally substituted amino, optionally substituted carbamoyl, optionally substituted carbamoyloxy, optionally substituted lower alkylthio, optionally substituted arylthio, optionally substituted lower alkylsulfonyl, optionally substituted arylsulfonyl, optionally substituted lower alkylsulfinyl, optionally substituted arylsulfinyl, optionally substituted lower alkylsulfonyloxy, optionally substituted arylsulfonyloxy, optionally substituted sulfamoyl, an optionally substituted carbocyclic group, optionally substituted carbocyclicoxy, an optionally substituted heterocyclic group or optionally substituted heterocyclicoxy, each G may be different (this compound is represented by A-2), 11) the compound, wherein ring A is represented by the following formula:
##STR00047##
wherein R9 and R10 are each independently a hydrogen atom, halogen, hydroxy, optionally substituted lower alkyl, cyano, nitro, optionally substituted lower alkoxy, optionally substituted acyl, optionally substituted amino, optionally substituted carbamoyl, optionally substituted carbamoyloxy, optionally substituted lower alkylsulfonyl, optionally substituted arylsulfonyl, optionally substituted lower alkylsulfonyloxy, optionally substituted arylsulfonyloxy, an optionally substituted carboncyclic group, optionally substituted carbocyclicoxy, an optionally substituted heterocyclic group or optionally substituted heterocyclicoxy, G is the same as described above 10) (this compound is represented by A-3), 12) the compound, wherein ring A is represented by the following formula:
##STR00048##
wherein R9 and R10 is the same as described in 11), G is the same as described in 10) (this compound is represented by A-4), 13) the compound, wherein ring A, R9, and R10 are defined in 11), G is optionally substituted amino (this compound is represented by A-5), 14) the compound, wherein ring A, R9 and R19 are defined in 11), G is optionally substituted arylcarbonylamino or optionally substituted heterocyclic carbonylamino, 15) the compound, wherein ring A, R9 and R10 are defined in 11), G is optionally substituted heterocyclic carbonylamino (this compound is represented by A-6), 16) the compound, wherein ring A is defined in 11), G is represented by the following formula:
##STR00049## ##STR00050## ##STR00051##
wherein Q1, Q2 and Q3 are each independently a bond, optionally substituted lower alkylene or optionally substituted lower alkenylene; Q4 is optionally substituted lower alkylene or optionally substituted lower alkenylene; W1 and W2 are each independently O or S;
W3 is O, S or NR12;
[0167] R12 is a hydrogen atom, lower alkyl, hydroxy lower alkyl, lower alkoxy lower alkyl, lower alkoxycarbonyl lower alkyl, carbocyclic lower alkyl or acyl; R14 is hydrogen atom or lower alkyl; ring B is an optionally substituted carbocyclic group or an optionally substituted heterocyclic group; Alk2 is optionally substituted lower alkyl; R9 and R19 are the same as described in 11) (this compound is represented by A-7), 17) the compound, wherein ring A, R9 and R10 are the group defined in 11); G is the group defined in 16); ring B is aryl optionally substituted with one or more substituents selected from halogen, hydroxy, optionally substituted lower alkyl, optionally substituted lower alkoxy, optionally substituted acyl, optionally substituted amino, cyano, optionally substituted carbamoyl, an optionally substituted carbocyclic group, optionally substituted carbocyclicoxy or an optionally substituted heterocyclic group or heteroaryl optionally substituted with one or more substituents selected from halogen, hydroxy, optionally substituted lower alkyl, optionally substituted lower alkoxy, optionally substituted acyl, optionally substituted amino, cyano, optionally substituted carbamoyl, an optionally substituted carbocyclic group, optionally substituted carbocyclicoxy or an optionally substituted heterocyclic group; and the other symbols are the same as described in 16) (this compound is represented by A-8), 18) the compound, wherein ring A, R9 and R10 are defined in 11), G is represented by the following formula:
##STR00052##
in formula, wherein each symbols are the same as described in 16) (this compound is represented by A-9), 19) the compound, wherein ring A is represented by the following formula:
##STR00053##
G is defined in 16), ring B is optionally substituted aryl or optionally substituted heteroaryl, either R9 or R10 is a hydrogen atom; and the other is a hydrogen atom, halogen, optionally substituted lower alkyl, cyano, nitro, optionally substituted lower alkoxy, optionally substituted amino, optionally substituted carbamoyl, optionally substituted lower alkylsulfonyl, an optionally substituted carbocyclic group or an optionally substituted heterocyclic group (this compound is represented by A-10), 20) the compound, wherein ring A is represented by the following formula:
##STR00054##
G is defined in 18), the other symbols are the same as described in 19) (this compound is represented by A-11), 21) the compound, wherein ring A is represented by the following formula:
##STR00055##
G is defined in 16), ring B is optionally substituted phenyl, 6- to 6-membered heteroaryl, benzothiazolyl or benzothienyl, R9 and R10 are the same as described in 19) (this compound is represented by A-12), 22) the compound, wherein ring A is represented by the following formula:
##STR00056##
G is defined in 18), ring B is defined in 21), R9 and R19 are the same as described in 19) (this compound is represented by A-13), 23) the compound, wherein ring A is represented by the following formula:
##STR00057##
wherein R9 is a hydrogen atom, halogen, optionally substituted lower alkyl, cyano, nitro, optionally substituted lower alkoxy, optionally substituted amino, optionally substituted carbamoyl, optionally substituted lower alkylsulfonyl, an optionally substituted carbocyclic group or an optionally substituted heterocyclic group, ring B is the same as described in 21); R12 is a hydrogen atom or lower alkyl (this compound is represented by A-14), 24) the compound, wherein R6 is a hydrogen atom or 01 to C3 alkyl (this compound is represented by R5-1), 25) the compound, wherein R6 is C1 to C3 alkyl (this compound is represented by R5-2), 26) the compound, wherein R6 is methyl (this compound is represented by R5-3), 27) the compound, wherein R3a and R3b are each independently a hydrogen atom, halogen, hydroxy, optionally substituted lower alkyl, optionally substituted lower alkoxy or optionally substituted aryl (this compound is represented by R3-1), 28) the compound wherein, R3a is a hydrogen atom, halogen, hydroxy, optionally substituted lower alkyl, optionally substituted lower alkoxy or optionally substituted aryl, R3b is a hydrogen atom, one R3 is a hydrogen atom when n is 2, one or two R2a is(are) a hydrogen atom when n is 3 (this compound is represented by R3-2), 29) the compound, wherein R3a and R3b are all hydrogen atoms (this compound is represented by R3-3), and in a compound represented by the general formula (I'), a compound, wherein the combination of n, m, R2a, R2b, ring A, R5, R3a, and R3b, (nm, R2, A, R5, R3) is the following compound. (nm, R2, A, R5, R3)=(nm-1,R2-1,A-1,R5-1,R3-1),(nm-1,R2-1,A-1,R5-1,R3-2),(nm- -1,R2-1,A-1,R5-2,R3-1),(nm-1,R2-1,A-1,R5-2,R3-2),(nm-1,R2-1,A-1,R5-3,R3-1)- ,(nm-1,R2-1,A-1,R5-3,R3-2),(nm-1,R2-1,A-2,R5-1,R3-1),(nm-1,R2-1,A-2,R5-1,R- 3-2),(nm-1,R2-1,A-2,R5-2,R3-1),(nm-1,R2-1,A-2,R5-2,R3-2),(nm-1,R2-1,A-2,R5- -3,R3-1),(nm-1,R2-1,A-2,R5-3,R3-2),(nm-1, R2-1,A-3,R5-1,R3-1),(nm-1,R2-1,A-3,R5-1,R3-2),(nm-1,R2-1,A-3,R5-2,R3-1),(- nm-1,R2-1,A-3,R5-2,R3-2),(nm-1,R2-1,A-3,R5-3,R3-1),(nm-1,R2-1,A-3,R5-3,R3-- 2),(nm-1,R2-1,A-4,R5-1,R3-1),(nm-1,R2-1,A-4,R5-1,R3-2),(nm-1,R2-1,A-4,R5-2- ,R3-1),(nm-1,R2-1, A-4,R5-2,R3-2),(nm-1,R2-1,A-4,R5-3,R3-1),(nm-1,R2-1,A-4,R5-3,R3-2),(nm-1,- R2-1,A-5,R5-1,R3-1),(nm-1,R2-1,A-5,R5-1,R3-2),(nm-1,R2-1,A-5,R5-2,R3-1),(n- m-1,R2-1,A-5,R5-2,R3-2),(nm-1,R2-1,A-5,R5-3,R3-1),(nm-1,R2-1,A-5,R5-3,R3-2- ),(nm-1,R2-1,A-6, R5-1,R3-1),(nm-1,R2-1,A-6,R5-1,R3-2),(nm-1,R2-1,A-6,R5-2,R3-1),(nm-1,R2-1- ,A-6,R5-2,R3-2),(nm-1,R2-1,A-6,R5-3,R3-1),(nm-1,R2-1,A-6,R5-3,R3-2),(nm-1,- R2-1,A-7,R5-1,R3-1),(nm-1,R2-1,A-7,R5-1,R3-2),(nm-1,R2-1,A-7,R5-2,R3-1),(n- m-1,R2-1,A-7,R5-2, R3-2),(nm-1,R2-1,A-7,R5-3,R3-1),(nm-1,R2-1,A-7,R5-3,R3-2),(nm-1,R2-1,A-8,- R5-1,R3-1),(nm-1,R2-1,A-8,R5-1,R3-2),(nm-1,R2-1,A-8,R5-2,R3-1),(nm-1,R2-1,- A-8,R5-2,R3-2),(nm-1,R2-1,A-8,R5-3,R3-1),(nm-1,R2-1,A-8,R5-3,R3-2),(nm-1,R- 2-1,A-9,R5-1,R3-1),(nm-1,R2-1,A-9,R5-1,R3-2),(nm-1,R2-1,A-9,R5-2,R3-1),(nm- -1,R2-1,A-9,R5-2,R3-2),(nm-1,R2-1,A-9,R5-3,R3-1),(nm-1,R2-1,A-9,R5-3,R3-2)- ,(nm-1,R2-1,A-10,R5-1,R3-1),(nm-1,R2-1,A-10,R5-1,R3-2),(nm-1,R2-1,A-10,R5-- 2,R3-1),(nm-1,R2-1,A-10,R5-2,R3-2),(nm-1,R2-1,A-10,R5-3,R3-1),(nm-1,R2-1,A- -10,R5-3,R3-2),(nm-1,R2-1,A-11,R5-1,R3-1),(nm-1,R2-1,A-11,R5-1,R3-2),(nm-1- ,R2-1,A-11,R5-2,R3-1),(nm-1,R2-1,A-11,R5-2, R3-2),(nm-1,R2-1,A-11,R5-3,R3-1),(nm-1,R2-1,A-11,R5-3,R3-2),(nm-1,R2-1,A-- 12,R5-1,R3-1),(nm-1,R2-1,A-12,R5-1,R3-2),(nm-1,R2-1,A-12,R5-2,R3-1),(nm-1,- R2-1,A-12,R5-2,R3-2),(nm-1,R2-1,A-12,R5-3,R3-1),(nm-1,R2-1,A-12,R5-3,R3-2)- ,(nm-1,R2-1,A-13, R5-1,R3-1),(nm-1,R2-1,A-13,R5-1,R3-2),(nm-1,R2-1,A-13,R5-2,R3-1),(nm-1,R2- -1,A-13,R5-2,R3-2),(nm-1,R2-1,A-13,R5-3,R3-1),(nm-1,R2-1,A-13,R5-3,R3-2),(- nm-1,R2-1,A-14,R5-1,R3-1),(nm-1,R2-1,A-14,R5-1,R3-2),(nm-1,R2-1,A-14,R5-2,- R3-1),(nm-1,R2-1, A-14,R5-2,R3-2),(nm-1,R2-1,A-14,R5-3,R3-1),(nm-1,R2-1,A-14,R5-3,R3-2),(nm- -1,R2-2,A-1,R5-1,R3-1),(nm-1,R2-2,A-1,R5-1,R3-2),(nm-1,R2-2, A-1,R5-2,R3-2),(nm-1,R2-2,A-1,R5-3,R3-1),(nm-1,R2-2,A-1,R5-3,R3-2),(nm-1,- R2-2,A-2,R5-1,R3-1),(nm-1,R2-2,A-2,R5-1,R3-2),(nm-1,R2-2,A-2,R5-2,R3-1),(n- m-1,R2-2,A-2,R5-2,R3-2),(nm-1,R2-2,A-2,R5-3,R3-1),(nm-1,R2-2,A-2,R5-3,R3-2- ),(nm-1,R2-2,A-3, R5-1,R3-1),(nm-1,R2-2,A-3,R5-1,R3-2),(nm-1,R2-2,A-3,R5-2,R3-1),(nm-1,R2-2- ,A-3,R5-2,R3-2),(nm-1,R2-2,A-3,R5-3,R3-1),(nm-1,R2-2,A-3,R5-3,R3-2),(nm-1,- R2-2,A-4,R5-1,R3-1),(nm-1,R2-2,A-4,R5-1,R3-2),(nm-1,R2-2,A-4,R5-2,R3-1),(n- m-1,R2-2,A-4,R5-2, R3-2),(nm-1,R2-2,A-4,R5-3,R3-1),(nm-1,R2-2,A-5,R5-1,R3-1),(nm-1,R2-2,A-5,- R5-1,R3-2),(nm-1,R2-2,A-5,R5-2,R3-1),(nm-1,R2-2,A-5,R5-2,R3-2),(nm-1,R2-2,- A-5,R5-3,R3-1),(nm-1,R2-2,A-5,R5-3,R3-2),(nm-1,R2-2,A-6,R5-1,R3-1),(nm-1,R- 2-2,A-6,R5-1,R3-2),(nm-1,R2-2,A-6,R5-2,R3-1),(nm-1,R2-2,A-6,R5-2,R3-2),(nm- -1,R2-2,A-6,R5-3,R3-1),(nm-1,R2-2,A-6,R5-3,R3-2),(nm-1,R2-2,A-7,R5-1,R3-1)- ,(nm-1,R2-2,A-7,R5-1,R3-2),(nm-1,R2-2,A-7,R5-2,R3-1),(nm-1,R2-2,A-7,R5-2,R- 3-2),(nm-1,R2-2,A-7,R5-3,R3-1),(nm-1,R2-2,A-7,R5-3,R3-2),(nm-1,R2-2,A-8,R5- -1,R3-1),(nm-1,R2-2,A-8,R5-1,R3-2),(nm-1,R2-2,A-8,R5-2,R3-1),(nm-1,R2-2,A-- 8,R5-2,R3-2),(nm-1, R2-2,A-8,R5-3,R3-1),(nm-1,R2-2,A-8,R5-3,R3-2),(nm-1,R2-2,A-9,R5-1,R3-1),(- nm-1,R2-2,A-9,R5-1,R3-2),(nm-1,R2-2,A-9,R5-2,R3-1),(nm-1,R2-2,A-9,R5-2,R3-- 2),(nm-1,R2-2,A-9,R5-3,R3-1),(nm-1,R2-2,A-10,R5-1,R3-1),(nm-1,R2-2,A-10,R5- -1,R3-2),(nm-1,R2-2,A-10,R5-2,R3-1),(nm-1,R2-2,A-10,R5-2,R3-2),(nm-1,R2-2,- A-10,R5-3,R3-1),(nm-1,R2-2,A-10,R5-3,R3-2),(nm-1,R2-2,A-11,R5-1,R3-1),(nm-- 1, R2-2,A-11,R5-1,R3-2),(nm-1,R2-2,A-11,R5-2,R3-1),(nm-1,R2-2,A-11,R5-2,R3- -2),(nm-1,R2-2,A-11,R5-3,R3-1),(nm-1,R2-2,A-11,R5-3,R3-2),(nm-1,R2-2,A-12,- R5-1,R3-1),(nm-1,R2-2,A-12,R5-1,R3-2),(nm-1,R2-2,A-12,R5-2,R3-1),(nm-1,R2-- 2,A-12,R5-2,R3-2), (nm-1,R2-2,A-12,R5-3,R3-1),(nm-1,R2-2,A-12,R5-3,R3-2),(nm-1,R2-2,A-13,R5-- 1,R3-1),(nm-1,R2-2,A-13,R5-1,R3-2),(nm-1,R2-2,A-13,R5-2,R3-1),(nm-1,R2-2,A- -13,R5-2,R3-2),(nm-1,R2-2,A-13,R5-3,R3-1),(nm-1,R2-2,A-13,R5-3,R3-2),(nm-1- ,R2-2,A-14,R5-1, R3-1),(nm-1,R2-2,A-14,R5-1,R3-2),(nm-1,R2-2,A-14,R5-2,R3-1),(nm-1,R2-2,A-- 14,R5-2,R3-2),(nm-1,R2-2,A-14,R5-3,R3-1),(nm-1,R2-2,A-14,R5-3,R3-2),(nm-1,- R2-3,A-1,R5-1,R3-1),(nm-1,R2-3,A-1,R5-1,R3-2),(nm-1,R2-3,A-1,R5-2,R3-1),(n- m-1,R2-3,A-1,R5-2,R3-2),(nm-1,R2-3,A-1,R5-3,R3-1),(nm-1,R2-3,A-1,R5-3,R3-2- ),(nm-1,R2-3,A-2,R5-1, R3-1),(nm-1,R2-3,A-2,R5-1,R3-2),(nm-1,R2-3,A-2,R5-2,R3-1),(nm-1,R2-3,A-2,- R5-2,R3-2),(nm-1,R2-3,A-2,R5-3,R3-1),(nm-1,R2-3,A-3,R5-1,R3-1),(nm-1,R2-3,- A-3,R5-1,R3-2),(nm-1,R2-3,A-3,R5-2,R3-1),(nm-1,R2-3,A-3,R5-2,R3-2),(nm-1,R- 2-3,A-3,R5-3,R3-1),(nm-1,R2-3,A-3,R5-3,R3-2),(nm-1,R2-3,A-4,R5-1,R3-1),(nm- -1,R2-3,A-4,R5-1,R3-2),(nm-1,R2-3,A-4,R5-2,R3-1),(nm-1,R2-3,A-4,R5-2,R3-2)- ,(nm-1,R2-3,A-4,R5-3,R3-1),(nm-1,R2-3,A-4,R5-3,R3-2),(nm-1,R2-3,A-5,R5-1,R- 3-1),(nm-1,R2-3,A-5,R5-1,R3-2),(nm-1,R2-3,A-5,R5-2,R3-1),(nm-1,R2-3,A-5,R5- -2,R3-2),(nm-1,R2-3,A-6,R5-1,R3-1),(nm-1, R2-3,A-6,R5-1,R3-2),(nm-1,R2-3,A-6,R5-2,R3-1),(nm-1,R2-3,A-6,R5-2,R3-2),(- nm-1,R2-3,A-6,R5-3,R3-1),(nm-1,R2-3,A-6,R5-3,R3-2),(nm-1,R2-3,A-7,R5-1,R3-- 1),(nm-1,R2-3,A-7,R5-1,R3-2),(nm-1,R2-3,A-7,R5-2,R3-1),(nm-1,R2-3,A-7,R5-2- ,R3-2),(nm-1,R2-3, A-7,R5-3,R3-1),(nm-1,R2-3,A-7,R5-3,R3-2),(nm-1,R2-3,A-8,R5-1,R3-1),(nm-1,- R2-3,A-8,R5-1,R3-2),(nm-1,R2-3,A-8,R5-2,R3-1),(nm-1,R2-3,A-8,R5-2,R3-2),(n- m-1,R2-3,A-8,R5-3,R3-1),(nm-1,R2-3,A-8,R5-3,R3-2),(nm-1,R2-3,A-9,R5-1,R3-1- ),(nm-1,R2-3,A-9, R5-1,R3-2),(nm-1,R2-3,A-9,R5-2,R3-1),(nm-1,R2-3,A-9,R5-2,R3-2),(nm-1,R2-3- ,A-9,R5-3,R3-1),(nm-1,R2-3,A-10,R5-1,R3-1),(nm-1,R2-3,A-10, R5-1,R3-2),(mar 1,R2-3,A-10,R5-2,R3-1),(nm-1,R2-3,A-10,R5-2,R3-2),(nm-1,R2-3,A-10,R5-3,R3- -1),(nm-1,R2-3,A-10,R5-3,R3-2),(nm-1,R2-3,A-11,R5-1,R3-1),(nm-1,R2-3,A-11,- R5-1,R3-2),(nm-1,R2-3,A-11,R5-2,R3-1),(nm-1,R2-3,A-11,R5-2,R3-2),(nm-1,R2-- 3, A-11,R5-3,R3-1),(nm-1,R2-3,A-11,R5-3,R3-2),(nm-1,R2-3,A-12,R5-1,R3-1),(- nm-1,R2-3,A-12,R5-1,R3-2),(nm-1,R2-3,A-12,R5-2,R3-1),(nm-1,R2-3,A-12,R5-2,- R3-2),(nm-1,R2-3,A-12,R5-3,R3-1),(nm-1,R2-3,A-12,R5-3,R3-2),(nm-1,R2-3,A-1- 3,R5-1,R3-1),(nm-1, R2-3,A-13,R5-1,R3-2),(nm-1,R2-3,A-13,R5-2,R3-1),(nm-1,R2-3,A-13,R5-2,R3-2- ),(nm-1,R2-3,A-13,R5-3,R3-1),(nm-1,R2-3,A-13,R5-3,R3-2),(nm-1,R2-3,A-14,R5- -1,R3-1),(nm-1,R2-3,A-14,R5-1,R3-2),(nm-1,R2-3,A-14,R5-2,R3-1),(nm-1,R2-3,- A-14,R5-2,R3-2), (nm-1,R2-3,A-14,R5-3,R3-1),(nm-1,R2-3,A-14,R5-3,R3-2),(nm-1,R2-4,A-1,R5-1- ,R3-1),(nm-1,R2-4,A-1,R5-1,R3-2),(nm-1,R2-4,A-1,R5-2,R3-1),(nm-1,R2-4,A-1,- R5-2,R3-2),(nm-1,R2-4,A-1,R5-3,R3-1),(nm-1,R2-4,A-1,R5-3,R3-2),(nm-1,R2-4,- A-2,R5-1,R3-1),(nm-1,R2-4,A-2,R5-1,R3-2),(nm-1,R2-4,A-2,R5-2,R3-1),(nm-1,R- 2-4,A-2,R5-2,R3-2),(nm-1,R2-4,A-2,R5-3,R3-1),(nm-1,R2-4,A-2,R5-3,R3-2),1,R- 3-1),(nm-1,R2-4,A-3,R5-1,R3-2),(nm-1,R2-4,A-3,R5-2,R3-1),(nm-1,R2-4,A-3,R5- -2,R3-2),(nm-1, R2-4,A-3,R5-3,R3-1),(nm-1,R2-4,A-3,R5-3,R3-2),(nm-1,R2-4,A-4,R5-1,R3-1),(- nm-1,R2-4,A-4,R5-1,R3-2),(nm-1,R2-4,A-4,R5-2,R3-1),(nm-1,R2-4,A-4,R5-2,R3-- 2),(nm-1,R2-4,A-4,R5-3,R3-1),(nm-1,R2-4,A-5,R5-1,R3-1),(nm-1,R2-4, A-5,R5-1,R3-2),(nm-1,R2-4,A-5,R5-2,R3-1),(nm-1,R2-4,A-5,R5-2,R3-2),(nm-1,- R2-4,A-5,R5-3,R3-1),(nm-1,R2-4,A-5,R5-3,R3-2),(nm-1,R2-4,A-6,R5-1,R3-1),(n- m-1,R2-4,A-6,R5-1,R3-2),(nm-1,R2-4,A-6,R5-2,R3-1),(nm-1,R2-4,A-6,R5-2,R3-2- ),(nm-1,R2-4,A-6, R5-3,R3-1),(nm-1,R2-4,A-6,R5-3,R3-2),(nm-1,R2-4,A-7,R5-1,R3-1),(nm-1,R2-4- ,A-7,R5-1,R3-2),(nm-1,R2-4,A-7,R5-2,R3-1),(nm-1,R2-4,A-7,R5-2,R3-2),(nm-1,- R2-4,A-7,R5-3,R3-1),(nm-1,R2-4,A-8,R5-1,R3-1),(nm-1,R2-4,A-8,R5-1, R3-2),(nm-1,R2-4,A-8,R5-2,R3-1),(nm-1,R2-4,A-8,R5-2,R3-2),(nm-1,R2-4,A-8,- R5-3,R3-1),(nm-1,R2-4,A-8,R5-3,R3-2),(nm-1,R2-4,A-9,R5-1,R3-1),(nm-1,R2-4,- A-9,R5-1,R3-2),(nm-1,R2-4,A-9,R5-2,R3-1),(nm-1,R2-4,A-9,R5-2,R3-2),(nm-1,R- 2-4,A-9,R5-3,R3-1),(nm-1,R2-4,A-9,R5-3,R3-2),(nm-1,R2-4,A-10,R5-1,R3-1),(n- m-1,R2-4,A-10,R5-1,R3-2),(nm-1,R2-4,A-10,R5-2,R3-1),(nm-1,R2-4,A-10,R5-2,R- 3-2),(nm-1,R2-4,A-10,R5-3,R3-1),(nm-1,R2-4,A-10,R5-3,R3-2),(nm-1,R2-4,A-11- ,R5-1,R3-1),(nm-1,R2-4,A-11,R5-1, R3-2),(nm-1,R2-4,A-11,R5-2,R3-1),(nm-1,R2-4,A-11,R5-2,R3-2),(nm-1,R2-4,A-- 11,R5-3,R3-1),(nm-1,R2-4,A-11,R5-3,R3-2),(nm-1,R2-4,A-12,R5-1,R3-1),(nm-1,- R2-4,A-12,R5-1,R3-2),(nm-1,R2-4,A-12,R5-2,R3-1),(nm-1,R2-4,A-12,R5-2,R3-2)- ,(nm-1,R2-4,A-12, R5-3,R3-1),(nm-1,R2-4,A-12,R5-3,R3-2),(nm-1,R2-4,A-13,R5-1,R3-1),(nm-1,R2- -4,A-13, R5-1,R3-2),(nm-1,R2-4,A-13,R5-2,R3-1),(nm-1,R2-4,A-13,R5-2,R3-2),- (nm-1,R2-4,A-13,R5-3,R3-1),(nm-1,R2-4,A-13,R5-3,R3-2),(nm-1,R2-4,A-14,R5-1- ,R3-1),(nm-1,R2-4, A-14,R5-1,R3-2),(nm-1,R2-4,A-14,R5-2,R3-1),(nm-1,R2-4,A-14,R5-2,R3-2),(nm- -1,R2-4,A-14,R5-3,R3-1),(nm-1,R2-4,A-14,R5-3,R3-2), (nm-2,R2-1,A-1,R5-1,R3-1),(nm-2,R2-1,A-1,R5-1,R3-2),(nm-2,R2-1,A-1,R5-2,R- 3-1),(nm-2,R2-1,A-1,R5-2,R3-2),(nm-2,R2-1,A-1,R5-3,R3-1),(nm-2,R2-1,A-1,R5- -3,R3-2),(nm-2,R2-1,A-2,R5-1,R3-1),(nm-2,R2-1,A-2,R5-1,R3-2),(nm-2,R2-1,A-- 2,R5-2,R3-1),(nm-2,R2-1,A-2,R5-2,R3-2),(nm-2,R2-1,A-2,R5-3,R3-1),(nm-2,R2-- 1,A-2,R5-3,R3-2),(nm-2, R2-1,A-3,R5-1,R3-1),(nm-2,R2-1,A-3,R5-1,R3-2),(nm-2,R2-1,A-3,R5-2,R3-1),(- nm-2,R2-1,A-3,R5-2,R3-2),(nm-2,R2-1,A-3,R5-3,R3-1),(nm-2,R2-1,A-3,R5-3,R3-- 2),(nm-2,R2-1,A-4,R5-1,R3-1),(nm-2,R2-1,A-4,R5-1,R3-2),(nm-2,R2-1,A-4,R5-2- ,R3-1),(nm-2,R2-1, A-4,R5-2,R3-2),(nm-2,R2-1,A-4,R5-3,R3-1),(nm-2,R2-1,A-4,R5-3,R3-2),(nm-2,- R2-1,A-5,R5-1,R3-1),(nm-2,R2-1,A-5,R5-1,R3-2),(nm-2,R2-1,A-5,R5-2,R3-1),(n- m-2,R2-1,A-5,R5-2,R3-2),(nm-2,R2-1,A-5,R5-3,R3-1),(nm-2,R2-1,A-6, R5-1,R3-1),(nm-2,R2-1,A-6,R5-1,R3-2),(nm-2,R2-1,A-6,R5-2,R3-1),(nm-2,R2-1- ,A-6,R5-2,R3-2),(nm-2,R2-1,A-6,R5-3,R3-1),(nm-2,R2-1,A-6,R5-3,R3-2),(nm-2,- R2-1,A-7,R5-1,R3-1),(nm-2,R2-1,A-7,R5-1,R3-2),(nm-2,R2-1,A-7,R5-2,R3-1),(n- m-2,R2-1,A-7,R5-2, R3-2),(nm-2,R2-1,A-7,R5-3,R3-1),(nm-2,R2-1,A-7,R5-3,R3-2),(nm-2,R2-1,A-8,- R5-1,R3-1),(nm-2,R2-1,A-8,R5-1,R3-2),(nm-2,R2-1,A-8,R5-2,R3-1),(nm-2,R2-1,- A-8,R5-2,R3-2),(nm-2,R2-1,A-8,R5-3,R3-1),(nm-2,R2-1,A-9,R5-1,R3-1),(nm-2,R- 2-1,A-9,R5-1,R3-2),(nm-2,R2-1,A-9,R5-2,R3-1),(nm-2,R2-1,A-9,R5-2,R3-2),(nm- -2,R2-1,A-9,R5-3,R3-1),(nm-2,R2-1,A-9,R5-3,R3-2),(nm-2,R2-1,A-10,R5-1,R3-1- ),(nm-2,R2-1,A-10,R5-1,R3-2),(nm-2,R2-1,A-10,R5-2,R3-1),(nm-2,R2-1,A-10,R5- -2,R3-2),(nm-2,R2-1,A-10,R5-3,R3-1),(nm-2,R2-1,A-10,R5-3,R3-2),(nm-2,R2-1,- A-11,R5-1,R3-1),(nm-2,R2-1,A-11,R5-1,R3-2),(nm-2,R2-1,A-11,R5-2,R3-1),(nm-- 2,R2-1,A-11,R5-2, R3-2),(nm-2,R2-1,A-11,R5-3,R3-1),(nm-2,R2-1,A-11,R5-3,R3-2),(nm-2,R2-1,A-- 12,R5-1,R3-1),(nm-2,R2-1,A-12,R5-1,R3-2),(nm-2,R2-1,A-12,R5-2,R3-1),(nm-2,- R2-1,A-12,R5-2,R3-2),(nm-2,R2-1,A-12,R5-3,R3-1),(nm-2,R2-1,A-12,R5-3,R3-2)- ,(nm-2,R2-1,A-13, R5-1,R3-1),(nm-2,R2-1,A-13,R5-1,R3-2),(nm-2,R2-1,A-13,R5-2,R3-1),(nm-2,R2- -1,A-13, R5-2,R3-2),(nm-2,R2-1,A-13,R5-3,R3-1),(nm-2,R2-1,A-13,R5-3,R3-2),- (nm-2,R2-1,A-14,R5-1,R3-1),(nm-2,R2-1,A-14,R5-1,R3-2),(nm-2,R2-1,A-14,R5-2- ,R3-1),(nm-2,R2-1, A-14,R5-2,R3-2),(nm-2,R2-1,A-14,R5-3,R3-1),(nm-2,R2-1,A-14,R5-3,R3-2),(nm- -2,R2-2,A-1,R5-1,R3-1),(nm-2,R2-2,A-1,R5-1,R3-2),(nm-2,R2-2,A-1,R5-2,R3-1)- ,(nm-2,R2-2, A-1,R5-2,R3-2),(nm-2,R2-2,A-1,R5-3,R3-1),(nm-2,R2-2,A-1,R5-3,R3-2),(nm-2,- R2-2,A-2,R5-1,R3-1),(nm-2,R2-2,A-2,R5-1,R3-2),(nm-2,R2-2,A-2,R5-2,R3-1),(n- m-2,R2-2,A-2,R5-2,R3-2),(nm-2,R2-2,A-2,R5-3,R3-1),(nm-2,R2-2,A-2,R5-3,R3-2- ),(nm-2,R2-2,A-3, R5-1,R3-1),(nm-2,R2-2,A-3,R5-1,R3-2),(nm-2,R2-2,A-3,R5-2,R3-1),(nm-2,R2-2- ,A-3,R5-2,R3-2),(nm-2,R2-2,A-3,R5-3,R3-1),(nm-2,R2-2,A-3,R5-3,R3-2),(nm-2,- R2-2,A-4,R5-1,R3-1),(nm-2,R2-2,A-4,R5-1,R3-2),(nm-2,R2-2,A-4,R5-2,R3-1),(n- m-2,R2-2,A-4,R5-2, R3-2),(nm-2,R2-2,A-4,R5-3,R3-1),(nm-2,R2-2,A-4,R5-3,R3-2),(nm-2,R2-2,A-5,- R5-1,R3-1),(nm-2,R2-2,A-5,R5-1,R3-2),(nm-2,R2-2,A-5,R5-2,R3-1),(nm-2,R2-2,- A-5,R5-2,R3-2),(nm-2,R2-2,A-5,R5-3,R3-1),(nm-2,R2-2,A-5,R5-3,R3-2),(nm-2,R- 2-2,A-6,R5-1,R3-1),(nm-2,R2-2,A-6,R5-1,R3-2),(nm-2,R2-2,A-6,R5-2,R3-1),(nm- -2,R2-2,A-6,R5-2,R3-2),(nm-2,R2-2,A-6,R5-3,R3-1),(nm-2,R2-2,A-6,R5-3,R3-2)- ,(nm-2,R2-2,A-7,R5-1,R3-1),(nm-2,R2-2,A-7,R5-2,R3-1),(nm-2,R2-2,A-7,R5-2,R- 3-2),(nm-2,R2-2,A-7,R5-3,R3-1),(nm-2,R2-2,A-7,R5-3,R3-2),(nm-2,R2-2,A-8,R5- -1,R3-1),(nm-2,R2-2,A-8,R5-2,R3-1),(nm-2,R2-2,A-8,R5-2,R3-2),(nm-2, R2-2,A-8,R5-3,R3-1),(nm-2,R2-2,A-8,R5-3,R3-2),(nm-2,R2-2,A-9,R5-1,R3-1),(- nm-2,R2-2,A-9,R5-1,R3-2),(nm-2,R2-2,A-9,R5-2,R3-1),(nm-2,R2-2,A-9,R5-2,R3-- 2),(nm-2,R2-2,A-9,R5-3,R3-1),(nm-2,R2-2,A-9,R5-3,R3-2),(nm-2,R2-2,A-10,R5-- 1,R3-1),(nm-2,R2-2,A-10,R5-1,R3-2),(nm-2,R2-2,A-10,R5-2,R3-1),(nm-2,R2-2,A- -10,R5-2,R3-2),(nm-2,R2-2,A-10,R5-3,R3-1),(nm-2,R2-2,A-10,R5-3,R3-2),(nm-2- ,R2-2,A-11,R5-1,R3-1),(nm-2, R2-2,A-11,R5-1,R3-2),(nm-2,R2-2,A-11,R5-2,R3-1),(nm-2,R2-2,A-11,R5-2,R3-2- ),(nm-2,R2-2,A-11,R5-3,R3-1),(nm-2,R2-2,A-11,R5-3,R3-2),(nm-2,R2-2,A-12,R5- -1,R3-1),(nm-2,R2-2,A-12,R5-1,R3-2),(nm-2,R2-2,A-12,R5-2,R3-1),(nm-2,R2-2,- A-12,R5-2,R3-2), (nm-2,R2-2,A-12,R5-3,R3-1),(nm-2,R2-2,A-12,R5-3,R3-2),(nm-2,R2-2,A-13,R5-- 1,R3-1),(nm-2,R2-2,A-13,R5-1,R3-2),(nm-2,R2-2,A-13,R5-2,R3-1),(nm-2,R2-2,A- -13,R5-2,R3-2),(nm-2,R2-2,A-13,R5-3,R3-1),(nm-2,R2-2,A-13,R5-3,R3-2),(nm-2- ,R2-2,A-14,R5-1, R3-1),(nm-2,R2-2,A-14,R5-1,R3-2),(nm-2,R2-2,A-14,R5-2,R3-1),(nm-2,R2-2,A-- 14,R5-2,R3-2),(nm-2,R2-2,A-14,R5-3,R3-1),(nm-2,R2-2,A-14,R5-3,R3-2),(nm-2,- R2-3,A-1,R5-1,R3-1),(nm-2,R2-3,A-1,R5-1,R3-2),(nm-2,R2-3,A-1,R5-2,R3-1),(n- m-2,R2-3,A-1,R5-2,R3-2),(nm-2,R2-3,A-1,R5-3,R3-1),(nm-2,R2-3,A-1,R5-3,R3-2- ),(nm-2,R2-3,A-2,R5-1, R3-1),(nm-2,R2-3,A-2,R5-1,R3-2),(nm-2,R2-3,A-2,R5-2,R3-1),(nm-2,R2-3,A-2,- R5-2,R3-2),(nm-2,R2-3,A-2,R5-3,R3-1),(nm-2,R2-3,A-2,R5-3,R3-2),(nm-2,R2-3,- A-3,R5-1,R3-1),(nm-2,R2-3,A-3,R5-1,R3-2),(nm-2,R2-3,A-3,R5-2,R3-1),(nm-2,R- 2-3,A-3,R5-2,R3-2),(nm-2,R2-3,A-3,R5-3,R3-1),(nm-2,R2-3,A-3,R5-3,R3-2),(nm- -2,R2-3,A-4,R5-1,R3-1),(nm-2,R2-3,A-4,R5-1,R3-2),(nm-2,R2-3,A-4,R5-2,R3-1)- ,(nm-2,R2-3,A-4,R5-2,R3-2),(nm-2,R2-3,A-4,R5-3,R3-1),(nm-2,R2-3,A-4,R5-3,R- 3-2),(nm-2,R2-3,A-5,R5-1,R3-1),(nm-2,R2-3,A-5,R5-1,R3-2),(nm-2,R2-3,A-5,R5- -2,R3-1),(nm-2,R2-3,A-5,R5-2,R3-2),(nm-2,R2-3,A-5,R5-3,R3-1),(nm-2,R2-3,A-- 5,R5-3,R3-2),(nm-2,R2-3,A-6,R5-1,R3-1),(nm-2, R2-3,A-6,R5-1,R3-2),(nm-2,R2-3,A-6,R5-2,R3-1),(nm-2,R2-3,A-6,R5-2,R3-2),(- nm-2,R2-3,A-6,R5-3,R3-1),(nm-2,R2-3,A-6,R5-3,R3-2),(nm-2,R2-3,A-7,R5-1,R3-- 1),(nm-2,R2-3,A-7,R5-1,R3-2),(nm-2,R2-3,A-7,R5-2,R3-1),(nm-2,R2-3,A-7,R5-2- ,R3-2),(nm-2,R2-3, A-7,R5-3,R3-1),(nm-2,R2-3,A-7,R5-3,R3-2),(nm-2,R2-3,A-8,R5-1,R3-1),(nm-2,- R2-3,A-8,R5-1,R3-2),(nm-2,R2-3,A-8,R5-2,R3-1),(nm-2,R2-3,A-8,R5-2,R3-2),(n- m-2,R2-3,A-8,R5-3,R3-1),(nm-2,R2-3,A-8,R5-3,R3-2),(nm-2,R2-3,A-9,R5-1,R3-1- ),(nm-2,R2-3,A-9, R5-1,R3-2),(nm-2,R2-3,A-9,R5-2,R3-1),(nm-2,R2-3,A-9,R5-2,R3-2),(nm-2,R2-3- ,A-9,R5-3,R3-1),(nm-2,R2-3,A-9,R5-3,R3-2),(nm-2,R2-3,A-10,R5-1,R3-1),(nm-2- ,R2-3,A-10, R5-1,R3-2),(nm-2,R2-3,A-10,R5-2,R3-1),(nm-2,R2-3,A-10,R5-2,R3-2),(nm-2,R2-
-3,A-10,R5-3,R3-1),(nm-2,R2-3,A-10,R5-3,R3-2),(nm-2,R2-3,A-11,R5-1,R3-1),(- nm-2,R2-3,A-11,R5-1,R3-2),(nm-2,R2-3,A-11,R5-2,R3-1),(nm-2,R2-3,A-11,R5-2,- R3-2),(nm-2,R2-3, A-11,R5-3,R3-1),(nm-2,R2-3,A-11,R5-3,R3-2),(nm-2,R2-3,A-12,R5-1,R3-1),(nm- -2,R2-3,A-12,R5-1,R3-2),(nm-2,R2-3,A-12,R5-2,R3-1),(nm-2,R2-3,A-12,R5-2,R3- -2),(nm-2,R2-3,A-12,R5-3,R3-1),(nm-2,R2-3,A-12,R5-3,R3-2),(nm-2,R2-3,A-13,- R5-1,R3-1),(nm-2, R2-3,A-13,R5-1,R3-2),(nm-2,R2-3,A-13,R5-2,R3-1),(nm-2,R2-3,A-13,R5-2,R3-2- ),(nm-2,R2-3,A-13,R5-3,R3-1),(nm-2,R2-3,A-13,R5-3,R3-2),(nm-2,R2-3,A-14,R5- -1,R3-1),(nm-2,R2-3,A-14,R5-1,R3-2),(nm-2,R2-3,A-14,R5-2,R3-1),(nm-2,R2-3,- A-14,R5-2,R3-2), (nm-2,R2-3,A-14,R5-3,R3-1),(nm-2,R2-3,A-14,R5-3,R3-2),(nm-2,R2-4,A-1,R5-1- ,R3-1),(nm-2,R2-4,A-1,R5-1,R3-2),(nm-2,R2-4,A-1,R5-2,R3-1),(nm-2,R2-4,A-1,- R5-2,R3-2),(nm-2,R2-4,A-1,R5-3,R3-1),(nm-2,R2-4,A-1,R5-3,R3-2),(nm-2,R2-4,- A-2,R5-1,R3-1),(nm-2,R2-4,A-2,R5-1,R3-2),(nm-2,R2-4,A-2,R5-2,R3-1),(nm-2,R- 2-4,A-2,R5-2,R3-2),(nm-2,R2-4,A-2,R5-3,R3-1),(nm-2,R2-4,A-2,R5-3,R3-2),(nm- -2,R2-4,A-3,R5-1,R3-1),(nm-2,R2-4,A-3,R5-1,R3-2),(nm-2,R2-4,A-3,R5-2,R3-1)- ,(nm-2,R2-4,A-3,R5-2,R3-2),(nm-2, R2-4,A-3,R5-3,R3-1),(nm-2,R2-4,A-3,R5-3,R3-2),(nm-2,R2-4,A-4,R5-1,R3-1),(- nm-2,R2-4,A-4,R5-2,R3-1),(nm-2,R2-4,A-4,R5-2,R3-2),(nm-2,R2-4,A-4,R5-3,R3-- 1),(nm-2,R2-4,A-4,R5-3,R3-2),(nm-2,R2-4,A-5,R5-1,R3-1),(nm-2,R2-4, A-5,R5-1,R3-2),(nm-2,R2-4,A-5,R5-2,R3-1),(nm-2,R2-4,A-5,R5-2,R3-2),(nm-2,- R2-4,A-5,R5-3,R3-1),(nm-2,R2-4,A-5,R5-3,R3-2),(nm-2,R2-4,A-6,R5-1,R3-1),(n- m-2,R2-4,A-6,R5-1,R3-2),(nm-2,R2-4,A-6,R5-2,R3-1),(nm-2,R2-4,A-6,R5-2,R3-2- ),(nm-2,R2-4,A-6, R5-3,R3-1),(nm-2,R2-4,A-6,R5-3,R3-2),(nm-2,R2-4,A-7,R5-1,R3-1),(nm-2,R2-4- ,A-7,R5-1,R3-2),(nm-2,R2-4,A-7,R5-2,R3-1),(nm-2,R2-4,A-7,R5-2,R3-2),(nm-2,- R2-4,A-7,R5-3,R3-1),(nm-2,R2-4,A-7,R5-3,R3-2),(nm-2,R2-4,A-8,R5-1,R3-1),(n- m-2,R2-4,A-8,R5-1, R3-2),(nm-2,R2-4,A-8,R5-2,R3-1),(nm-2,R2-4,A-8,R5-2,R3-2),(nm-2,R2-4,A-8,- R5-3,R3-1),(nm-2,R2-4,A-8,R5-3,R3-2),(nm-2,R2-4,A-9,R5-1,R3-1),(nm-2,R2-4,- A-9,R5-1,R3-2),(nm-2,R2-4,A-9,R5-2,R3-1),(nm-2,R2-4,A-9,R5-2,R3-2),(nm-2,R- 2-4,A-9,R5-3,R3-1),(nm-2,R2-4,A-9,R5-3,R3-2),(nm-2,R2-4,A-10,R5-1,R3-1),(n- m-2,R2-4,A-10,R5-1,R3-2),(nm-2,R2-4,A-10,R5-2,R3-1),(nm-2,R2-4,A-10,R5-2,R- 3-2),(nm-2,R2-4,A-10,R5-3,R3-1),(nm-2,R2-4,A-10,R5-3,R3-2),(nm-2,R2-4,A-11- ,R5-1,R3-1),(nm-2,R2-4,A-11,R5-1, R3-2),(nm-2,R2-4,A-11,R5-2,R3-1),(nm-2,R2-4,A-11,R5-2,R3-2),(nm-2,R2-4,A-- 11,R5-3,R3-1),(nm-2,R2-4,A-11,R5-3,R3-2),(nm-2,R2-4,A-12,R5-1,R3-1),(nm-2,- R2-4,A-12,R5-1,R3-2),(nm-2,R2-4,A-12,R5-2,R3-1),(nm-2,R2-4,A-12,R5-2,R3-2)- ,(nm-2,R2-4,A-12, R5-3,R3-1),(nm-2,R2-4,A-12,R5-3,R3-2),(nm-2,R2-4,A-13,R5-1,R3-1),(nm-2,R2- -4,A-13, R5-1,R3-2),(nm-2,R2-4,A-13,R5-2,R3-1),(nm-2,R2-4,A-13,R5-2,R3-2),- (nm-2,R2-4,A-13,R5-3,R3-1),(nm-2,R2-4,A-13,R5-3,R3-2),(nm-2,R2-4,A-14,R5-1- ,R3-1),(nm-2,R2-4, A-14,R5-1,R3-2),(nm-2,R2-4,A-14,R5-2,R3-1),(nm-2,R2-4,A-14,R5-2,R3-2),(nm- -2,R2-4,A-14,R5-3,R3-1),(nm-2,R2-4,A-14,R5-3,R3-2), (nm-3,R2-1,A-1,R5-1,R3-1),(nm-3,R2-1,A-1,R5-1,R3-2),(nm-3,R2-1,A-1,R5-2,R- 3-1),(nm-3,R2-1,A-1,R5-2,R3-2),(nm-3,R2-1,A-1,R5-3,R3-1),(nm-3,R2-1,A-1,R5- -3,R3-2),(nm-3,R2-1,A-2,R5-1,R3-1),(nm-3,R2-1,A-2,R5-1,R3-2),(nm-3,R2-1,A-- 2,R5-2,R3-1),(nm-3,R2-1,A-2,R5-3,R3-1),(nm-3,R2-1,A-2,R5-3,R3-2),(nm-3, R2-1,A-3,R5-1,R3-1),(nm-3,R2-1,A-3,R5-1,R3-2),(nm-3,R2-1,A-3,R5-2,R3-1),(- nm-3,R2-1,A-3,R5-2,R3-2),(nm-3,R2-1,A-3,R5-3,R3-1),(nm-3,R2-1,A-4,R5-1,R3-- 1),(nm-3,R2-1,A-4,R5-2,R3-1),(nm-3,R2-1, A-4,R5-2,R3-2),(nm-3,R2-1,A-4,R5-3,R3-1),(nm-3,R2-1,A-4,R5-3,R3-2),(nm-3,- R2-1,A-5,R5-1,R3-1),(nm-3,R2-1,A-5,R5-1,R3-2),(nm-3,R2-1,A-5,R5-2,R3-1),(n- m-3,R2-1,A-5,R5-2,R3-2),(nm-3,R2-1,A-5,R5-3,R3-1),(nm-3,R2-1,A-5,R5-3,R3-2- ),(nm-3,R2-1,A-6, R5-1,R3-1),(nm-3,R2-1,A-6,R5-1,R3-2),(nm-3,R2-1,A-6,R5-2,R3-1),(nm-3,R2-1- ,A-6,R5-2,R3-2),(nm-3,R2-1,A-6,R5-3,R3-1),(nm-3,R2-1,A-6,R5-3,R3-2),(nm-3,- R2-1,A-7,R5-1,R3-1),(nm-3,R2-1,A-7,R5-1,R3-2),(nm-3,R2-1,A-7,R5-2,R3-1),(n- m-3,R2-1,A-7,R5-2, R3-2),(nm-3,R2-1,A-7,R5-3,R3-1),(nm-3,R2-1,A-8,R5-1,R3-1),(nm-3,R2-1,A-8,- R5-1,R3-2),(nm-3,R2-1,A-8,R5-2,R3-1),(nm-3,R2-1,A-8,R5-2,R3-2),(nm-3,R2-1,- A-8,R5-3,R3-1),(nm-3,R2-1,A-8,R5-3,R3-2),(nm-3,R2-1,A-9,R5-1,R3-1),(nm-3,R- 2-1,A-9,R5-1,R3-2),(nm-3,R2-1,A-9,R5-2,R3-1),(nm-3,R2-1,A-9,R5-2,R3-2),(nm- -3,R2-1,A-9,R5-3,R3-1),(nm-3,R2-1,A-9,R5-3,R3-2),(nm-3,R2-1,A-10,R5-1,R3-1- ),(nm-3,R2-1,A-10,R5-1,R3-2),(nm-3,R2-1,A-10,R5-2,R3-1),(nm-3,R2-1,A-10,R5- -2,R3-2),(nm-3,R2-1,A-10,R5-3,R3-1),(nm-3,R2-1,A-10,R5-3,R3-2),(nm-3,R2-1,- A-11,R5-1,R3-1),(nm-3,R2-1,A-11,R5-1,R3-2),(nm-3,R2-1,A-11,R5-2,R3-1),(nm-- 3,R2-1,A-11,R5-2, R3-2),(nm-3,R2-1,A-11,R5-3,R3-1),(nm-3,R2-1,A-11,R5-3,R3-2),(nm-3,R2-1,A-- 12,R5-1,R3-1),(nm-3,R2-1,A-12,R5-1,R3-2),(nm-3,R2-1,A-12,R5-2,R3-1),(nm-3,- R2-1,A-12,R5-2,R3-2),(nm-3,R2-1,A-12,R5-3,R3-1),(nm-3,R2-1,A-12,R5-3,R3-2)- ,(nm-3,R2-1,A-13, R5-1,R3-1),(nm-3,R2-1,A-13,R5-1,R3-2),(nm-3,R2-1,A-13,R5-2,R3-1),(nm-3,R2- -1,A-13, R5-2,R3-2),(nm-3,R2-1,A-13,R5-3,R3-1),(nm-3,R2-1,A-13,R5-3,R3-2),- (nm-3,R2-1,A-14,R5-1,R3-1),(nm-3,R2-1,A-14,R5-1,R3-2),(nm-3,R2-1,A-14,R5-2- ,R3-1),(nm-3,R2-1, A-14,R5-2,R3-2),(nm-3,R2-1,A-14,R5-3,R3-1),(nm-3,R2-1,A-14,R5-3,R3-2),(nm- -3,R2-2,A-1,R5-1,R3-1),(nm-3,R2-2,A-1,R5-1,R3-2),(nm-3,R2-2,A-1,R5-2,R3-1)- ,(nm-3,R2-2, A-1,R5-2,R3-2),(nm-3,R2-2,A-1,R5-3,R3-1),(nm-3,R2-2,A-1,R5-3,R3-2),(nm-3,- R2-2,A-2,R5-1,R3-1),(nm-3,R2-2,A-2,R5-1,R3-2),(nm-3,R2-2,A-2,R5-2,R3-1),(n- m-3,R2-2,A-2,R5-2,R3-2),(nm-3,R2-2,A-2,R5-3,R3-1),(nm-3,R2-2,A-2,R5-3,R3-2- ),(nm-3,R2-2,A-3, R5-1,R3-1),(nm-3,R2-2,A-3,R5-1,R3-2),(nm-3,R2-2,A-3,R5-2,R3-1),(nm-3,R2-2- ,A-3,R5-2,R3-2),(nm-3,R2-2,A-3,R5-3,R3-1),(nm-3,R2-2,A-3,R5-3,R3-2),(nm-3,- R2-2,A-4,R5-1,R3-1),(nm-3,R2-2,A-4,R5-1,R3-2),(nm-3,R2-2,A-4,R5-2,R3-1),(n- m-3,R2-2,A-4,R5-2, R3-2),(nm-3,R2-2,A-4,R5-3,R3-1),(nm-3,R2-2,A-4,R5-3,R3-2),(nm-3,R2-2,A-5,- R5-1,R3-1),(nm-3,R2-2,A-5,R5-1,R3-2),(nm-3,R2-2,A-5,R5-2,R3-1),(nm-3,R2-2,- A-5,R5-2,R3-2),(nm-3,R2-2,A-5,R5-3,R3-1),(nm-3,R2-2,A-5,R5-3,R3-2),(nm-3,R- 2-2,A-6,R5-1,R3-1),(nm-3,R2-2,A-6,R5-1,R3-2),(nm-3,R2-2,A-6,R5-2,R3-1),(nm- -3,R2-2,A-6,R5-2,R3-2),(nm-3,R2-2,A-6,R5-3,R3-1),(nm-3,R2-2,A-6,R5-3,R3-2)- ,(nm-3,R2-2,A-7,R5-1,R3-1),(nm-3,R2-2,A-7,R5-1,R3-2),(nm-3,R2-2,A-7,R5-2,R- 3-1),(nm-3,R2-2,A-7,R5-2,R3-2),(nm-3,R2-2,A-7,R5-3,R3-1),(nm-3,R2-2,A-7,R5- -3,R3-2),(nm-3,R2-2,A-8,R5-1,R3-1),(nm-3,R2-2,A-8,R5-1,R3-2),(nm-3,R2-2,A-- 8,R5-2,R3-1),(nm-3,R2-2,A-8,R5-2,R3-2),(nm-3, R2-2,A-8,R5-3,R3-1),(nm-3,R2-2,A-8,R5-3,R3-2),(nm-3,R2-2,A-9,R5-1,R3-1),(- nm-3,R2-2,A-9,R5-1,R3-2),(nm-3,R2-2,A-9,R5-2,R3-1),(nm-3,R2-2,A-9,R5-2;R3-- 2),(nm-3,R2-2,A-9,R5-3,R3-1),(nm-3,R2-2,A-9,R5-3,R3-2),(nm-3,R2-2,A-10,R5-- 1,R3-1),(nm-3,R2-2,A-10,R5-1,R3-2),(nm-3,R2-2,A-10,R5-2,R3-1),(nm-3,R2-2,A- -10,R5-2,R3-2),(nm-3,R2-2,A-10,R5-3,R3-1),(nm-3,R2-2,A-10,R5-3,R3-2),(nm-3- ,R2-2,A-11,R5-1,R3-1),(nm-3, R2-2,A-11,R5-1,R3-2),(nm-3,R2-2,A-11,R5-2,R3-1),(nm-3,R2-2,A-11,R5-2,R3-2- ),(nm-3,R2-2,A-11,R5-3,R3-1),(nm-3,R2-2,A-11,R5-3,R3-2),(nm-3,R2-2,A-12,R5- -1,R3-1),(nm-3,R2-2,A-12,R5-1,R3-2),(nm-3,R2-2,A-12,R5-2,R3-1),(nm-3,R2-2,- A-12,R5-2,R3-2), (nm-3,R2-2,A-12,R5-3,R3-1),(nm-3,R2-2,A-12,R5-3,R3-2),(nm-3,R2-2,A-13,R5-- 1,R3-1),(nm-3,R2-2,A-13,R5-1,R3-2),(nm-3,R2-2,A-13,R5-2,R3-1),(nm-3,R2-2,A- -13,R5-2,R3-2),(nm-3,R2-2,A-13,R5-3,R3-1),(nm-3,R2-2,A-13,R5-3,R3-2),(nm-3- ,R2-2,A-14,R5-1, R3-1),(nm-3,R2-2,A-14,R5-1,R3-2),(nm-3,R2-2,A-14,R5-2,R3-1),(nm-3,R2-2,A-- 14,R5-2,R3-2),(nm-3,R2-2,A-14,R5-3,R3-1),(nm-3,R2-2,A-14,R5-3,R3-2),(nm-3,- R2-3,A-1,R5-1,R3-1),(nm-3,R2-3,A-1,R5-1,R3-2),(nm-3,R2-3,A-1,R5-2,R3-1),(n- m-3,R2-3,A-1,R5-2,R3-2),(nm-3,R2-3,A-1,R5-3,R3-1),(nm-3,R2-3,A-1,R5-3,R3-2- ),(nm-3,R2-3,A-2,R5-1, R3-1),(nm-3,R2-3,A-2,R5-1,R3-2),(nm-3,R2-3,A-2,R5-2,R3-1),(nm-3,R2-3,A-2,- R5-2,R3-2),(nm-3,R2-3,A-2,R5-3,R3-1),(nm-3,R2-3,A-2,R5-3,R3-2),(nm-3,R2-3,- A-3,R5-1,R3-1),(nm-3,R2-3,A-3,R5-1,R3-2),(nm-3,R2-3,A-3,R5-2,R3-1),(nm-3,R- 2-3,A-3,R5-2,R3-2),(nm-3,R2-3,A-3,R5-3,R3-1),(nm-3,R2-3,A-3,R5-3,R3-2),(nm- -3,R2-3,A-4,R5-1,R3-1),(nm-3,R2-3,A-4,R5-1,R3-2),(nm-3,R2-3,A-4,R5-2,R3-1)- ,(nm-3,R2-3,A-4,R5-2,R3-2),(nm-3,R2-3,A-4,R5-3,R3-1),(nm-3,R2-3,A-4,R5-3,R- 3-2),(nm-3,R2-3,A-5,R5-1,R3-1),(nm-3,R2-3,A-5,R5-1,R3-2),(nm-3,R2-3,A-5,R5- -2,R3-1),(nm-3,R2-3,A-5,R5-2,R3-2),(nm-3,R2-3,A-5,R5-3,R3-1),(nm-3,R2-3,A-- 5,R5-3,R3-2),(nm-3,R2-3,A-6,R5-1,R3-1),(nm-3, R2-3,A-6,R5-1,R3-2),(nm-3,R2-3,A-6,R5-2,R3-1),(nm-3,R2-3,A-6,R5-2,R3-2),(- nm-3,R2-3,A-6,R5-3,R3-1),(nm-3,R2-3,A-6,R5-3,R3-2),(nm-3,R2-3,A-7,R5-1,R3-- 1),(nm-3,R2-3,A-7,R5-1,R3-2),(nm-3,R2-3,A-7,R5-2,R3-1),(nm-3,R2-3,A-7,R5-2- ,R3-2),(nm-3,R2-3, A-7,R5-3,R3-1),(nm-3,R2-3,A-7,R5-3,R3-2),(nm-3,R2-3,A-8,R5-1,R3-1),(nm-3,- R2-3,A-8,R5-1,R3-2),(nm-3,R2-3,A-8,R5-2,R3-1),(nm-3,R2-3,A-8,R5-2,R3-2),(n- m-3,R2-3,A-8,R5-3,R3-1),(nm-3,R2-3,A-9,R5-1,R3-1),(nm-3,R2-3,A-9, R5-1,R3-2),(nm-3,R2-3,A-9,R5-2,R3-1),(nm-3,R2-3,A-9,R5-2,R3-2),(nm-3,R2-3- ,A-9,R5-3,R3-1),(nm-3,R2-3,A-9,R5-3,R3-2),(nm-3,R2-3,A-10,R5-1,R3-1),(nm-3- ,R2-3,A-10, R5-1,R3-2),(nm-3,R2-3,A-10,R5-2,R3-1),(nm-3,R2-3,A-10,R5-2,R3-2),(nm-3,R2- -3,A-10,R5-3,R3-1),(nm-3,R2-3,A-10,R5-3,R3-2),(nm-3,R2-3,A-11,R5-1,R3-1),(- nm-3,R2-3,A-11,R5-1,R3-2),(nm-3,R2-3,A-11,R5-2,R3-1),(nm-3,R2-3,A-11,R5-2,- R3-2),(nm-3,R2-3, A-11,R5-3,R3-1),(nm-3,R2-3,A-11,R5-3,R3-2),(nm-3,R2-3,A-12,R5-1,R3-1),(nm- -3,R2-3,A-12,R5-1,R3-2),(nm-3,R2-3,A-12,R5-2,R3-1),(nm-3,R2-3,A-12,R5-2,R3- -2),(nm-3,R2-3,A-12,R5-3,R3-1),(nm-3,R2-3,A-12,R5-3,R3-2),(nm-3,R2-3,A-13,- R5-1,R3-1),(nm-3, R2-3,A-13,R5-1,R3-2),(nm-3,R2-3,A-13,R5-2,R3-1),(nm-3,R2-3,A-13,R5-2,R3-2- ),(nm-3,R2-3,A-13,R5-3,R3-1),(nm-3,R2-3,A-13,R5-3,R3-2),(nm-3,R2-3,A-14,R5- -1,R3-1),(nm-3,R2-3,A-14,R5-1,R3-2),(nm-3,R2-3,A-14,R5-2,R3-1),(nm-3,R2-3,- A-14,R5-2,R3-2), (nm-3,R2-3,A-14,R5-3,R3-1),(nm-3,R2-3,A-14,R5-3,R3-2),(nm-3,R2-4,A-1,R5-1- ,R3-1),(nm-3,R2-4,A-1,R5-1,R3-2),(nm-3,R2-4,A-1,R5-2,R3-1),(nm-3,R2-4,A-1,- R5-2,R3-2),(nm-3,R2-4,A-1,R5-3,R3-1),(nm-3,R2-4,A-1,R5-3,R3-2),(nm-3,R2-4,- A-2,R5-1,R3-1),(nm-3,R2-4,A-2,R5-1,R3-2),(nm-3,R2-4,A-2,R5-2,R3-1),(nm-3,R- 2-4,A-2,R5-2,R3-2),(nm-3,R2-4,A-2,R5-3,R3-1),(nm-3,R2-4,A-2,R5-3,R3-2),(nm- -3,R2-4,A-3,R5-1,R3-1),(nm-3,R2-4,A-3,R5-1,R3-2),(nm-3,R2-4,A-3,R5-2,R3-1)- ,(nm-3,R2-4,A-3,R5-2,R3-2),(nm-3, R2-4,A-3,R5-3,R3-1),(nm-3,R2-4,A-3,R5-3,R3-2),(nm-3,R2-4,A-4,R5-1,R3-1),(- nm-3,R2-4,A-4,R5-1,R3-2),(nm-3,R2-4,A-4,R5-2,R3-1),(nm-3,R2-4,A-4,R5-2,R3-- 2),(nm-3,R2-4,A-4,R5-3,R3-1),(nm-3,R2-4,A-4,R5-3,R3-2),(nm-3,R2-4,A-5,R5-1- ,R3-1),(nm-3,R2-4, A-5,R5-1,R3-2),(nm-3,R2-4,A-5,R5-2,R3-1),(nm-3,R2-4,A-5,R5-2,R3-2),(nm-3,- R2-4,A-5,R5-3,R3-1),(nm-3,R2-4,A-5,R5-3,R3-2),(nm-3,R2-4,A-6,R5-1,R3-1),(n- m-3,R2-4,A-6,R5-1,R3-2),(nm-3,R2-4,A-6,R5-2,R3-1),(nm-3,R2-4,A-6,R5-2,R3-2- ),(nm-3,R2-4,A-6, R5-3,R3-1),(nm-3,R2-4,A-6,R5-3,R3-2),(nm-3,R2-4,A-7,R5-1,R3-1),(nm-3,R2-4- ,A-7,R5-1,R3-2),(nm-3,R2-4,A-7,R5-2,R3-1),(nm-3,R2-4,A-7,R5-2,R3-2),(nm-3,- R2-4,A-7,R5-3,R3-1),(nm-3,R2-4,A-7,R5-3,R3-2),(nm-3,R2-4,A-8,R5-1,R3-1),(n- m-3,R2-4,A-8,R5-1, R3-2),(nm-3,R2-4,A-8,R5-2,R3-1),(nm-3,R2-4,A-8,R5-2,R3-2),(nm-3,R2-4,A-8,- R5-3,R3-1),(nm-3,R2-4,A-8,R5-3,R3-2),(nm-3,R2-4,A-9,R5-1,R3-1),(nm-3,R2-4,- A-9,R5-1,R3-2),(nm-3,R2-4,A-9,R5-2,R3-1),(nm-3,R2-4,A-9,R5-2,R3-2),(nm-3,R- 2-4,A-9,R5-3,R3-1),(nm-3,R2-4,A-9,R5-3,R3-2),(nm-3,R2-4,A-10,R5-1,R3-1),(n- m-3,R2-4,A-10,R5-1,R3-2),(nm-3,R2-4,A-10,R5-2,R3-1),(nm-3,R2-4,A-10,R5-2,R- 3-2),(nm-3,R2-4,A-10,R5-3,R3-1),(nm-3,R2-4,A-10,R5-3,R3-2),(nm-3,R2-4,A-11- ,R5-1,R3-1),(nm-3,R2-4,A-11,R5-1, R3-2),(nm-3,R2-4,A-11,R5-2,R3-1),(nm-3,R2-4,A-11,R5-2,R3-2),(nm-3,R2-4,A-- 11,R5-3,R3-1),(nm-3,R2-4,A-11,R5-3,R3-2),(nm-3,R2-4,A-12,R5-1,R3-1),(nm-3,- R2-4,A-12,R5-1,R3-2),(nm-3,R2-4,A-12,R5-2,R3-1),(nm-3,R2-4,A-12,R5-2,R3-2)- ,(nm-3,R2-4,A-12, R5-3,R3-1),(nm-3,R2-4,A-12,R5-3,R3-2),(nm-3,R2-4,A-13,R5-1,R3-1),(nm-3,R2- -4,A-13, R5-1,R3-2),(nm-3,R2-4,A-13,R5-2,R3-1),(nm-3,R2-4,A-13,R5-2,R3-2),- (nm-3,R2-4,A-13,R5-3,R3-1),(nm-3,R2-4,A-13,R5-3,R3-2),(nm-3,R2-4,A-14,R5-1- ,R3-1),(nm-3,R2-4, A-14,R5-1,R3-2),(nm-3,R2-4,A-14,R5-2,R3-1),(nm-3,R2-4,A-14,R5-2,R3-2),(nm- -3,R2-4,A-14,R5-3,R3-1),(nm-3,R2-4,A-14,R5-3,R3-2),(nm-3,R2-4,A-14,R5-3,R3- -3).
[0168] In a compound represented by the general formula (I'), a compound, wherein the combination of n, m, R2a, R2b, ring A, R5, R3a, and R3b (nm, R2, A, R5, R3) is one of the above compound, and E is a bond.
[0169] The compounds of the invention can be employed in the treatment and/or prevention of disease associated with the generation, secretion or deposition of β-amyloid protein, such as dementia of the Alzheimer's type (Alzheimer's disease, senile dementia of Alzheimer type), Down's syndrome, memory impairment, prion disease (Creutzfeldt-Jakob disease), mild cognitive impairment (MCI), Dutch type of hereditary cerebral hemorrhage with amyloidosis, cerebral amyloid angiopathy, other type of degenerative dementia, mixed dementia with Alzheimer's and vascular type, dementia with Parkinson's Disease, dementia with progressive supranuclear palsy, dementia with Cortico-basal degeneration, Alzheimer's disease with diffuse Lewy body disease, age-related macular degeneration, Parkinson's Disease, amyloid angiopathy and so on.
[0170] The compounds of the invention can be administrated in combination with other pharmaceutical agents such as other therapeutic drugs for Alzheimer's disease, acetylcholinesterase inhibitors and so on. The compounds of the invention can be treated with concomitantly with the anti-dementia agents such as Donepezil Hydrochloride, Tacrine, Galantamine, Rivastigmine, Zanapezil, Memantine, Vinpocetine.
[0171] When the present compound is administered to a human, it can be administered orally as powders, granules, tablets, capsules, pills, solutions, or the like, or parenterally as injectables, suppositories, transdermal absorbable agents, absorbable agents, or the like. In addition, the present compound can be formulated into pharmaceutical preparations by adding pharmaceutical additives such as excipients, binders, wetting agents, disintegrating agents, lubricants and the like, which are suitable for formulations and an effective amount of the present compound.
[0172] A dose is different depending on state of disease, an administration route, and an age and a weight of a patient, and is usually 0.1 μg to 1 g/day, preferably 0.01 to 200 mg/day when orally administered to an adult, and is usually 0.1 μg to 10 g/day, preferably 0.1 to 2 g/day when parenterally administered.
[0173] Following examples and test examples illustrate the present invention in more detail, but the present invention is not limited by these examples.
[0174] In example, the meaning of each abbreviation is following.
Me methyl Et ethyl iPr or Pri isopropyl Ph phenyl Bn benzyl Boc t-butoxycarbonyl TBDPS t-butyldiphenylsilyl
Example 1
The Synthesis of Compound 588
##STR00058## ##STR00059##
[0175] Step 1
[0176] Under nitrogen atmosphere, the compound (1-1) (7.98 g) was dissolved into diethyl ether (330 ml)-tetrahydrofuran (36 ml), vinylmagnesium chloride in tetrahydrofuran solution (1.32 mol/L, 44.8 ml) was added under cooling with dryice-acetone bath, and stirred for 20 min. Then, the reaction solution was stirred for 30 min under cooling with ice-water bath and stirred for 36 min at room temperature. And then, saturated ammonium chloride solution was added to the mixture, the mixture was extracted with ethyl acetate, and organic layer was washed with saturated ammonium chloride solution, saturated sodium hydrogencarbonate solution, and brine, and dried over anhydrous magnesium sulfate, and the solvent was evaporated. Then, the residue was purified by silica gel column chromatography to afford the compound (1-2) (6.00 g).
[0177] 1H-NMR (CDCl3): 1.63 (3H, s), 2.08 (1H, br), 5.20 (1H, dd, J=10.6, 1.6 Hz), 6.31 (1H, dd, J=17.1, 1.6 Hz), 6.09 (1H, m), 7.46 (1H, m), 7.52 (1H, dd, J=3.4, 2.6 Hz), 7.80 (1H, dd, J=3.9, 2.6 Hz), 8.06 (1H, br)
Step 2
[0178] The compound (1-2) (6.36 g) was dissolved into acetic acid (30 ml), and added thiourea (1.50 g), 1 mol/L hydrochloride-acetic acid solution (20.7 ml). The reaction mixture was stirred at room temperature for 3 hours, then stirred at 40° C. for 3 hours, then stirred at room temperature for 66 hours, and at 40° C. for 19 hours. Thiourea (0.450 g), and 1 mol/L hydrochloric acid-acetic acid solution (7.53 ml) was added, and stirred at 40° C. for 23 hours. After the consumption of the compound (1-2), the solvent was evaporated under reduced pressure, then the obtained residue was crystallized from methanol-diethyl ether to afford the compound (1-3) (5.23 g) as crystal. On the other hand, mother liquid was evaporated under reduced pressure, and the compound (1-3) (3.00 g) was obtained as crude solid product.
[0179] 1H-NMR (DMSO-d6): 2.09 (3H, 5), 4.10 (2H, d, J=7.3 Hz), 5.94 (1H, t, J=7.7 Hz), 7.50 (1H, s), 7.75 (1H, s), 7.87 (1H, s), 9.17 (3H, br), 11.46 (1H, s)
Step 3
[0180] The compound (1-3) (5.23 g) dissolved in trifluoroacetic acid (25 ml) was added methanesulfonic acid (2.14 ml) dropwise under cooling with ice-water bath. After addition, the reaction mixture was stirred at room temperature for 3.5 hours. After the consumption of the compound (1-3), the solvent was evaporated under reduced pressure. To the residue obtained was added water and sodium carbonate, and then extracted with ethyl acetate. The organic layer was washed with saturated sodium hydrogencarbonate solution, and was dried over anhydrous magnesium sulfate, and the solvent was evaporated under reduced pressure to afford the compound (1-4) (4.90 g) as crude product.
[0181] 1H-NMR (CDCl3): 1.53 (3H, s), 1.90 (1H, m), 2.09 (1H, m), 2.74 (1H, m), 2.97 (1H, m), 4.32 (2H, br), 7.34 (1H, t, J=1.6 Hz), 7.37 (1H, t, J=1.8 Hz), 7.86 (1H, t, J=1.8 Hz)
Step 4
[0182] Under nitrogen atmosphere, the compound (1-4) (4.90 g) dissolved in tetrahydrofuran was added di-t-butyl-dicarbonate (2.97 g) and triethylamine (1.89 ml) under cooling with ice-water bath and then stirred for 2 hours. The reaction mixture was stirred at room temperature for 3 hours. The reaction mixture was added water, and then extracted with ethyl acetate. The organic layer was washed with water, and dried over anhydrous magnesium sulfate, then the solvent was evaporated under reduced pressure. Then the obtained residue was crystallized from ethyl acetate-diethyl ether to afford the compound (1-5) (4.62 g) as crystal.
[0183] 1H-NMR (CDCl3): 1.36 (9H, s), 1.72 (3H, s), 2.10 (1H, m), 2.41 (1H, m), 2.62 (1H, m), 2.75 (1H, m), 7.22 (1H, s), 7.48 (1H, s), 8.29 (1H, s)
Step 5
[0184] The compound (1-5) (1.00 g) was dissolved into tetrahydrofuran (8.7 ml), and 1 mol/L lithium hydroxide (4.43 ml) was added and stirred at 50° C. for 4 hours. Water was added to the reaction mixture, and the mixture was extracted with ethyl acetate, and the organic layer was washed with water, brine successively, and dried over anhydrous magnesium sulfate, and the solution was evaporated under reduced pressure. The obtained residue was purified by medium-pressured silica gel column chromatography to afford the compound (1-6) (0.668 g).
[0185] 1H-NMR (CDCl3): 1.51 (9H, 5), 1.63 (3H, 5), 2.06 (1H, m), 2.40 (1H, m), 2.68-2.74 (2H, m), 3.83 (2H, br), 6.51 (1H, t, J=1.8 Hz), 6.72-6.74 (2H, m)
Step 6
[0186] The compound (1-6) (20.0 mg) was dissolved into 4 mol/L hydrochloric acid in 1,4-dioxane, and the mixture was stirred for 16 hours. The reaction solvent was evaporated under reduced pressure and the obtained residue was crystallized from methanol-diethyl ether to afford the compound (588) (14.7 mg).
[0187] 1H-NMR (DMSO-d6): 1.59 (3H, s), 2.09-2.76 (4H, m), 6.44 (1H, t, J=1.6 Hz), 6.60 (1H, t J=1.9 Hz), 6.71 (1H, t, J=2.0 Hz), 10.4 (111, s)
Example 2
The Synthesis of Compound 835
##STR00060##
[0188] Step 1
[0189] The compound (2-1) (2020 mg) was dissolved into chloroform (20 ml), then water (4 ml) and sodium thiocyanic acid (1470 mg) were added at room temperature with stirring, and then sulfuric acid (1.94 ml) was added dropwise under cooling with ice-water bath. After an addition was complete, the reaction mixture was warmed to room temperature and then stirred for 345 minutes, then stirred at 60° C. overnight. Because the compound (2-1) was remained (checked by TLC), the reaction mixture was cooled to room temperature, then sodium thiocyanic acid (1470 mg), water (5 ml) and sulfuric acid (1.94 ml) were added successively. After the reaction mixture was warmed to 60° C., the mixture was stirred for 1 day. Saturated sodium carbohydrate solution was added to the reaction mixture to be basic condition under cooling with ice-water bath, and then the reaction mixture was extracted with ethyl acetate. The organic layer was washed with brine, then dried over anhydrous magnesium sulfate. The solvent was evaporated and the obtained residue was purified by silica gel column chromatography to afford the compound (2-2) (968 mg).
[0190] 1H-NMR (CDCl3, 270 MHz): 1.99 (3H, s), 3.55 (1H, d, J=16.1 Hz), 3.69 (1H, d, J=16.1 Hz), 7.12-7.64 (8H, m), 7.82-7.96 (2H, m)
Step 2
[0191] The compound (2-2) (842 mg) was dissolved into ethanol (8.4 ml), sodium dihydorgen phosphate, sodium borohydride (113.2 mg), and water (2.8 ml), were added successively under cooling with ice-water bath with stirring, and the mixture was stirred for 30 minutes. After the consumption of the compound (2-2) (checked by TLC), ethyl acetate and water were added to the reaction mixture under cooling with ice-water bath, and then stirred for a few minutes. The reaction mixture was extracted with ethyl acetate. The organic layer was washed with water, brine successively, and dried over anhydrous magnesium sulfate. The solvent was evaporated to afford the compound (2-3) (904.8 mg) as crude product.
Step 3
[0192] To a solution of compound (2-3) (900 mg) in toluene (10 ml) was added a solution of thionyl chloride (0.7 ml) in toluene (5 ml) under cooling with ice-water bath, and then stirred for 1 hour. After the consumption of the compound (2-3) (checked by TLC), the reaction solvent was evaporated under reduced pressure to afford the compound (2-4) (1076.8 mg) as crude product.
Step 4
[0193] The compound (2-4) (1070 mg) was dissolved into about 7 mol/L ammonia in methanol (20 ml) at room temperature, then the mixture was stirred for 1 day. After the consumption of the compound (2-4) (checked by TLC), the reaction solvent was evaporated under reduced pressure to afford the compound (835) (2633 mg) as crude product.
Example 3
The Synthesis of Compound 561
##STR00061##
[0194] Step 1
[0195] To tetrahydrofuran (30 ml) under cooling with ice-water bath with stirring, lithium aluminium hydride (0.63 g) was added portionwise, then a solution of compound (3-1) (1.94 g) in tetrahydrofuran (40 ml) was added dropwise. The reaction mixture was reacted for 20 minutes at room temperature, then reacted for 3 hours under reflux. Then ice was added in small portions under cooling, and then stirred for 1 day at room temperature. The reaction mixture was filtered and the filtrate was evaporated under reduced pressure, and the residue was purified by silica gel column chromatography to afford the compound (3-2) (0.90 g).
[0196] 1H-NMR (CDCl3): 1.22 (3H, s), 3.08 (1H, d, J=12.5 Hz), 3.34 (1H, d, J=12.5 Hz), 3.85 (1H, d, J=11.0 Hz), 4.11 (1H, d, J=11.0 Hz), 7.21-7.25 (1H, m), 7.34-7.40 (2H, m), 7.46-7.50 (2H, m).
Step 2
[0197] The compound (3-2) (0.90 g) was dissolved into tetrahydrofuran (15 ml), t-butylisothiocyanate (0.69 g) in tetrahydrofuran (5 ml) was added under cooling with ice-water bath with stirring. The reaction mixture was stirred for 3 days at room temperature, water was added and extracted with dichloromethane. The organic layer was dried over anhydrous magnesium sulfate, then the solvent was evaporated under reduced pressure. The residue was purified by silica gel column chromatography to afford the compound (3-3) (1.33 g).
[0198] 1H-NMR (CDCl3): 1.12 (9H, s), 1.34 (3H, s), 3.15 (1H, br), 3.76 (1H, d, J=11.2 Hz), 3.87 (1H, dd, J=14.2, 4.6 Hz), 4.13 (1H, d, J=11.2 Hz), 4.23 (1H, dd, J=14.2, 6.6 Hz), 5.18 (1H, br), 6.01 (1H, br), 7.23-7.28 (1H, m), 7.34-7.41 (4H, m).
Step 3
[0199] The compound (3-3) (315 mg) was dissolved into acetonitrile (3 ml), triphenylphosphine (440 mg), and carbon tetrachloride (520 mg) in acetonitrile (3 ml) were added under cooling with ice-water bath with stirring. The reaction mixture was stirred for 1 hour at room temperature, and then potassium carbonate (460 mg) was added and stirred for 2 days at room temperature. Then water was added to the reaction mixture and the mixture was extracted with dichloromethane. The organic layer was dried over anhydrous magnesium sulfate, and then the solvent was evaporated under reduced pressure. The residue was purified by silica gel column chromatography to afford the compound (3-4) (0.23 g).
[0200] 1H-NMR (CDCl3): 1.30 (9H, s), 1.36 (3H, s), 3.13 (1H, d, J=12.2 Hz), 3.24 (1H, dd, J=12.2, 2.3 Hz), 3.51 (1H, br), 3.53 (1H, d, J=15.2 Hz), 3.99 (1H, dd, J=15.2, 2.3 Hz), 7.20-7.25 (1H, m), 7.30-7.36 (2H, m), 7.39-7.43 (2H, m).
Step 4
[0201] To the compound (3-4) (0.22 g), conc. hydrochloric acid (4.5 ml) was added, then stirred for 2 hours under reflux, and then the reaction solvent was evaporated under reduced pressure. The obtained residue was crystallized from methanol-diethyl ether to afford the compound (561) (0.16 g).
[0202] 1H-NMR (DMSO-d6): 1.33 (3H, s), 3.33-3.49 (2H, m), 3.65-3.96 (2H, m), 7.29 (1H, t. J=7.6 Hz), 7.40 (2H, t. J=7.6 Hz), 7.48 (2H, t. J=7.6 Hz).
Example 4
The Synthesis of Compound 534
##STR00062##
[0203] Step 1
[0204] The compound (4-1) (0.72 g) was dissolved into N,N-dimethylformamide (15 ml), then sodium azide (0.31 g) was added. The reaction mixture was stirred at 100° C. for 13 hours, then water was added and the mixture was extracted with diethyl ether, the organic layer was dried over anhydrous magnesium sulfate to afford the compound (4-2) (0.71 g) as crude product.
Step 2
[0205] To a solution of the compound (4-2) (0.71 g) in tetrahydrofuran (10 ml), lithium aluminium hydride (0.14 g) was added portionwise under cooling with ice-water bath with stirring, then stirred for 2 hours at room temperature. After the consumption of the starting material, ice was added in small portions, then stirred for 18 hours at room temperature. The reaction mixture was filtered then filtrate was evaporated under reduced pressure to afford the compound (4-3) (0.89 g) as crude product.
Step 3
[0206] The compound (4-3) (0.89 g) was dissolved into tetrahydrofuran (10 ml), then t-butylisothiocyanate (0.56 g) in tetrahydrofuran (5 ml) was added under cooling with ice-water bath with stirring. The reaction mixture was stirred for 4 hours at room temperature, and water was added, and then extracted with dichloromethane, and the organic layer was dried over anhydrous magnesium sulfate. Then the residue was purified by silica gel column chromatography to afford the compound (4-4) (0.72 g).
[0207] 1H-NMR (CDCl3): 1.39 (9H, s), 2.08 (3H, s), 2.09-2.15 (2H, m), 3.37-3.44 (1H, m), 3.80-3.87 (1H, m), 5.97 (1H, br.), 6.86 (1H, br.), 7.28-7.43 (5H, m).
Step 4
[0208] The compound (4-4) (120 mg) was dissolved into acetonitrile (2 ml), triphenylphosphine (170 mg), and carbon tetrachloride (200 mg) in acetonitrile (1 ml) were added under cooling with ice-water bath with stirring. The reaction mixture was stirred for 5 hours at room temperature, and then potassium carbonate (177 mg) was added and stirred for 6 days at room temperature. Then water was added to the reaction mixture and the mixture was extracted with dichloromethane, the organic layer was dried over anhydrous magnesium sulfate, then the solvent was evaporated under reduced pressure. The residue was purified by silica gel column chromatography to afford the compound (4-6) (0.06 g).
[0209] 1H-NMR (CDCl3): 1.35 (9H, s), 1.59 (3H, s), 1.91 (1H, ddd, J=13.5, 8.8, 6.0 Hz), 2.06 (1H, dt, J=13.6, 6.0 Hz), 3.00 (1H, ddd, J=16.1, 8.8, 6.0 Hz), 3.30 (1H, dt, J=16.1, 6.0 Hz), 7.24-7.38 (6H, m).
Step 6
[0210] To the compound (4-5) (0.06 g), conc. hydrochloric acid (3 ml) was added, then the mixture was stirred for 1 hour under reflux, and the solvent was evaporated under reduced pressure. The obtained residue was crystallized from methanol-water to afford the compound (634) (0.02 g).
[0211] 1H-NMR (DMSO-d6): 1.43 (3H, s), 1.77 (1H, dt. J=8.4, 3.4 Hz), 2.11 (1H, d. J=9.2 Hz), 2.48-2.50 (1H, m), 2.83-2.99 (1H, m), 6.12 (1H, br), 6.65 (1H, br), 7.21-7.24 (1H, m), 7.31-7.37 (4H, m).
Example 5
The Synthesis of Compound 1008
##STR00063##
[0212] Step 1
[0213] The compound (5-1) (3.00 g) was dissolved into ethanol (30 ml), and thiourea (1.13 g) was added, and then the mixture was refluxed for 26 hours, and the solvent was evaporated under reduced pressure. The obtained residue was crystallized from ethyl acetate/hexane to afford the compound (5-2) (4.03 g).
[0214] 1H-NMR (DMSO-d6): 1.95 (2H, quint, J=6.8 Hz), 3.13 (2H, t, J=6.8 Hz), 3.21 (2H, t, J=6.8 Hz), 3.85 (3H, s), 7.06 (2H, d, J=8.8 Hz), 7.95 (2H, d, J=8.8 Hz), 9.18 (4H, br).
Step 2
[0215] The compound (5-2) (1.00 g) was dissolved into tetrahydrofuran (25 ml), then di-t-butyl-dicarbonate (1.74 g), and triethylamine (0.88 g) were added, and then the mixture was stirred for 3 hours at room temperature. Water was added to the reaction mixture, and the mixture was extracted with dichloromethane. The organic layer was dried over anhydrous magnesium sulfate, and then the solvent was evaporated under reduced pressure. The residue was purified by silica gel column chromatography to afford the compound (5-3) (1.24 g).
[0216] 1H-NMR (CDCl3): 1.50 (9H, 5), 2.07-2.17 (2H, m), 2.98 (2H, t, J=7.8 Hz), 3.09 (2H, t, J=6.3 Hz), 6.95 (2H, d, J=8.9 Hz), 7.95 (2H, d, J=8.9 Hz).
Step 3
[0217] The compound (5-3) (1.18 g) was dissolved into tetrahydrofuran (12 ml), then 0.9 mol/L methylmagnesium bromide in tetrahydrofuran solution (10.1 ml) was added under cooling with acetonitrile-dryice bath with stirring, and then reaction mixture was stirred for 1 hour, then stirred for 30 minutes at room temperature. After the reaction, saturated ammonium chloride solution was added under cooling with ice-water bath with stirring, then the mixture was extracted with diethyl ether, and the organic layer was dried over anhydrous magnesium sulfate, and then the solvent was evaporated under reduced pressure. The obtained residue was purified by silica gel column chromatography to afford the compound (5-4) (0.39 g).
[0218] 1H-NMR(CDCl3): 1.51 (9H, s), 1.63 (3H, s), 1.55-1.65 (2H, m), 1.87-1.91 (2H, m), 2.96-3.1.2 (2H, m), 6.86 (2H, d, J=8.9 Hz), 7.36 (2H, d, J=8.9 Hz).
Step 4
[0219] The compound (5-4) (0.24 g) was dissolved into trifluoroacetic acid (6 ml), and stirred for 20 hours at room temperature, then the reaction solvent was evaporated under reduced pressure. To the residue, water and saturated sodium hydrogencarbonate was added, and then extracted with dichloromethane. The organic layer was dried over anhydrous sodium sulfate, and then the solvent was evaporated under reduced pressure. The residue was purified by silica gel column chromatography to afford the compound (1008) (0.06 g).
[0220] 1H-NMR (CDCl3): 1.54 (3H, s), 1.77-1.87 (1H, m), 1.90-1.97 (1H, m), 2.20-2.36 (2H, m), 2.67-2.79 (2H, m), 3.81 (3H, s), 5.30 (2H, br), 6.87 (2H, d, J=9.0 Hz), 7.33 (2H, d, J=9.0 Hz).
Example 6
The Synthesis of Compound 783
##STR00064##
[0221] Step 1
[0222] The compound (6-1) (0.55 g) was dissolved into methanol (7 ml), and methyl iodide (0.36 g) was added at room temperature with stirring. The mixture was stirred at room temperature for 18 hours, then the reaction solvent was evaporated under reduced pressure to afford the compound (6-2) (0.92 g) as crude product.
Step 2
[0223] The compound (6-2) (0.92 g) was dissolved into tetrahydrofuran (7 ml), then triethylamine (0.24 g) and silver oxide (1.1 g) was added. The mixture was stirred at room temperature for 3 days, then the insolubles was removed by filtration, then the filtrate was evaporated under reduced pressure, and then the obtained residue was purified by silica gel column chromatography to afford the compound (6-3) (0.31 g).
[0224] 1H-NMR (CDCl3): 1.35 (9H, s), 1.60 (3H, s), 1.92 (1H, ddd, J=9.2, 5.8, 3.4 Hz), 2.07 (1H, dt, J=9.2, 3.4 Hz), 3.00 (1H, ddd, J=9.2, 5.8, 3.4 Hz), 3.30 (1H, dt, J=9.2, 3.4 Hz), 7.24-7.38 (5H, m).
Step 3
[0225] To the compound (6-3) (0.22 g), conc. hydrochloric acid (3 ml) was added, then the mixture was stirred for 1 hour under reflux, and then the reaction solvent was evaporated under reduced pressure. The obtained residue was crystallized from water to afford the compound (783) (0.13 g).
[0226] 1H-NMR (DMSO-d6): 1.44 (3H, s), 1.78 (1H, dt. J=12.4, 4.2 Hz), 2.12 (1H, d. J=8.9 Hz), 2.51-2.52 (1H, m), 2.96 (1H, d. J=4.2 Hz), 6.12 (1H, br), 6.66 (1H, br), 7.21-7.24 (1H, m), 7.32-7.37 (4H, m).
Example 7
The Synthesis of Compound 69
##STR00065##
[0227] Step 1
[0228] A solution of the compound (7-1) (1.93 g), diphenylphosphoryl azide (1.60 g), and triethylamine (0.59 g) in toluene (20 ml) was stirred at 80° C. for 3 hours, and water was added, and then the mixture was extracted with diethyl ether. The organic layer was dried over anhydrous sodium sulfate, and then the solvent was evaporated under reduced pressure. The obtained residue was purified by silica gel column chromatography to afford the compound (7-2) (1.69 g).
[0229] 1H-NMR (CDCl3): 1.00 (9H, s), 1.72 (3H, 5), 2.17-2.22 (2H, m), 3.49-3.58 (1H, m), 3.70-3.80 (1H, m), 7.20-7.42 (10H, m), 7.58-7.63 (5H, m).
Step 2
[0230] The compound (7-2) (1.68 g) was dissolved into toluene (9 ml), and 3,4-dimethoxybenzylalcohol (0.79 g) was added, the mixture was refluxed for 8 hours. To the reaction mixture, water was added, then the mixture was extracted with dichloromethane, and the organic layer was dried over anhydrous sodium sulfate, and the solvent was evaporated under reduced pressure. The obtained residue was purified by silica gel column chromatography to afford the compound (7-3) (2.09 g).
[0231] 1H-NMR (CDCl3): 1.03 (9H, s), 1.87 (3H, s), 2.04 (2H, m), 3.48 (1H, m), 3.51 (1H, m), 3.62 (3H, s), 3.65 (3H, s), 4.95 (1H, d, J=12.2 Hz), 5.03 (1H, d, J=12.2 Hz), 6.80-7.09 (3H, m), 7.22-7.42 (10H, m), 7.56-7.64 (5H, m).
Step 3
[0232] The compound (7-3) (2.09 g) was dissolved into 1,4-dioxane (15 ml), and 4 mol/L hydrochloric acid-1,4-dioxane (15 ml) solution was added, then stirred at room temperature for 24 hours. To the reaction mixture, water and 1 mol/L--sodium hydroxide solution were added and extracted with dichloromethane, then the organic layer was dried over anhydrous sodium sulfate, and then the solvent was evaporated under reduced pressure. The obtained residue was purified by silica gel column chromatography to afford the compound (7-4) (0.45 g).
[0233] 1H-NMR (CDCl3): 1.57 (3H, s), 1.07-1.98 (2H, m), 3.48-3.56 (1H, m), 3.72-3.86 (1H, m), 7.23-7.45 (15H, m).
Step 4
[0234] The compound (7-4) (0.44 g) was dissolved into tetrahydrofuran (15 ml), t-butylisothiocyanate (0.41 g) and diisopropylethylamine (0.46 g) were added. After the mixture was stirred at room temperature for 3 days, water was added, and extracted with dichloromethane, then the organic layer was dried over anhydrous sodium sulfate, and then the solvent was evaporated under reduced pressure. The obtained residue was purified by silica gel column chromatography to afford the compound (7-5) (0.17 g).
[0235] 1H-NMR (CDCl3): 1.79 (3H, s), 1.82-2.20 (2H, m), 3.71-3.81 (2H, m), 5.09 (1H, br), 7.30-7.52 (5H, m).
Step 5
[0236] The compound (7-5) (0.17 g) was dissolved into tetrahydrofuran (3.4 ml), then methyl iodide (0.11 g) was added at room temperature with stirring. The mixture was stirred for 23 hours, the reaction solvent was evaporated under reduced pressure to afford the compound (7-6) (0.28 g) as crude product.
Step 6
[0237] The compound (7-6) (0.28 g) was dissolved into tetrahydrofuran (5 ml), then triethylamine (74 mg) and silver oxide (0.34 g) were added. The mixture was stirred at room temperature for 20 hours, then insolubles were removed by filtration, and then the filtrate was evaporated under reduced pressure. The obtained residue was purified by silica gel column chromatography to afford the compound (7-7) (0.14 g).
[0238] 1H-NMR (CDCl3): 1.36 (9H, s), 1.49 (3H, s), 1.96-2.09 (2H, m), 2.77-3.83 (1H, m), 4.05-4.10 (1H, m), 7.19 (1H, t, J=7.3 Hz), 7.31 (2H, t, J=7.3 Hz), 7.44 (2H, d, J=7.3 Hz).
Step 7
[0239] To the compound (7-7) (0.12 g) conc. hydrochloric acid (9 ml) was added, then stirred for 1 hour under reflux, and then the reaction solvent was evaporated under reduced pressure. The obtained residue was crystallized from methanol-water to afford the compound (69) (0.10 g).
[0240] 1H-NMR (DMSO-d6): 1.65 (3H, s), 2.28-2.35 (1H, m), 2.39-2.44 (1H, m), 3.97 (1H, dt, J=7.8, 3.0 Hz), 4.53 (1H, dt, J=7.8, 3.0 Hz), 7.32-7.44 (5H, m), 8.44 (2H, br), 10.33 (1H, s).
Example 8
The Synthesis of Compound 256
##STR00066## ##STR00067##
[0241] Step 1
[0242] The compound (8-1) (4890 mg) was dissolved into N,N-dimethylformamide (100 ml), then sodium azide (5720 mg) was added at room temperature with stirring, and the solution was warmed to 80° C., and stirred for 12 hours. After the consumption of the compound (8-1) (checked by TLC), the reaction mixture was cooled to room temperature, then diethyl ether and water were added, and then the mixture was extracted with diethyl ether. The organic layer was washed with brine, and dried over magnesium sulfate. The solvent was evaporated under reduced pressure to afford the compound (8-2) (4940 mg) as crude product.
Step 2
[0243] To the suspension of lithium aluminium hydride (1080 mg) in tetrahydrofuran (90 ml) under nitrogen atmosphere under cooling with ice-water bath, the compound (8-2) (4940 mg) in tetrahydrofuran (15 ml) solution was added, the reaction mixture was stirred for 30 minutes. After the consumption of the compound (8-2) (checked by TLC), 1 mol/L sodium hydroxide solution was added under cooling with ice-water bath, then stirred for a while. The generated gel was removed with filtration, and the mother liquid was extracted with diethyl ether. The organic layer was washed with brine, and dried over magnesium sulfate. The solvent was evaporated under reduced pressure to afford the compound (8-3) (4219.1 mg) as crude product.
Step 3
[0244] The compound (8-3) (800 mg) was dissolved into acetonitrile (16 ml), the compound (8-4) (1840 mg) was added with stirring at room temperature, and stirred for 13 hours. After the consumption of the compound (8-3) (checked by no, the reaction solvent was evaporated under reduced pressure, the obtained residue was purified by silica gel column chromatography to afford the compound (8-5) (1550.7 mg).
[0245] 8-5-(Z): 1H-NMR (CDCl3, 270 MHz): 1.49 (18H, s), 2.06 (3H, d, J=1.4 Hz), 3.91-4.00 (2H, m), 5.54 (1H, td, J=7.1, 1.4 Hz), 7.12-7.41 (5H, m), 8.17-8.25 (1H, m), 11.47 (1H, s)
[0246] 8-5-(E): 1H-NMR (CDCl3, 270 MHz): 1.49 (9H, s), 1.52 (9H, s), 2.09 (3H, d, J=1.5 Hz), 4.24 (2H, dd, J=6.6, 5.3 Hz), 5.80 (1H, td, J=6.6, 1.5 Hz), 7.21-7.48 (5H, m), 8.28-8.38 (1H, m), 11.51 (1H, s)
Step 4
[0247] The compound (8-5) (474.1 mg) was dissolved into trifluoroacetic acid (4.5 ml) under cooling with ice-water bath, then warmed to room temperature, and stirred for 4 hours. After the consumption of the compound (8-5) (checked by NMR), the reaction mixture was poured into floating ice--1 mol/L sodium hydroxide solution to be neutralized, then the mixture was extracted with ethyl acetate. The organic layer was washed with brine, and dried over magnesium sulfate. The solvent was evaporated under reduced pressure to afford the compound (8-6) (326.4 mg) as crude product.
Step 5
[0248] The compound (8-6) (326.4 mg) was dissolved into 1,4-dioxane (2.4 ml), sodium hydroxide (195 mg) and water (1.2 ml) were added successively, then di-t-butyl dicarbonate (0.84 ml) was added under cooling with ice-water bath. The reaction mixture was warmed to room temperature, and stirred for 15 hours, then the consumption of the compound (8-6) was checked by LC-MS. After added water to the reaction mixture, the mixture was extracted with ethyl acetate. The organic layer was washed with brine, and dried over magnesium sulfate. The solvent was evaporated under reduced pressure, and the obtained residue was purified by silica gel column chromatography to afford the compound (8-7) (113.6 mg).
[0249] 1H-NMR (CDCl3, 400 MHz): 1.46 (9H, s), 1.51 (9H, s), 1.64 (3H, s), 2.06 (1H, ddd, J=13.4, 11.4, 5.0 Hz), 2.27 (1H, dt, J=13.4, 4.6 Hz), 3.15 (1H, ddd, J=12.9, 11.3, 4.6 Hz), 3.70 (1H, dt, J=12.9, 4.7 Hz), 7.23-7.29 (1H, m), 7.33-7.38 (4H, m)
Step 6
[0250] The compound (8-7) (110 mg) was dissolved into 4 mol/L hydrochloric acid-1,4-dioxane solution (1 ml) under cooling ice-water bath, the mixture was warmed to room temperature, and stirred for 2 days, then the consumption of the compound (8-7) was checked by LC-MS, and diethyl ether and water were added at room temperature. After separation of diethyl ether layer, water layer was evaporated under reduced pressure. To the obtained residue, methanol was added, then the generated crystal was filtered. The methanol in mother liquid was evaporated under reduced pressure to afford the compound (256) (69 mg).
[0251] 1H-NMR (DMSO-d6, 400 MHz): 1.57 (3H, s), 1.87-1.96 (1H, m), 2.30 (1H, dt, J=13.6, 3.8 Hz), 2.60 (1H, td, J=12.0, 3.7 Hz), 3.25 (1H, ddd, J=12.8, 8.2, 4.4 Hz), 6.93 (2H, s), 7.27-7.44 (5H, m), 7.94 (1H, s), 8.63 (1H, s)
Example 9
The Synthesis of Compound 24
##STR00068##
[0252] Step 1
[0253] The compound (9-1) (0.39 g) was dissolved into chloroform (20 ml), iodine (1.53 g), potassium thiocyanate (1.25 g), catalytic amount of tetrabutylammonium chloride, and water (1 ml) were added at room temperature, then stirred for 15 hours. To the reaction mixture, 10% thiosodium sulfate solution and water were added, and the mixture was extracted with dichloromethane. The organic layer was dried over anhydrous sodium sulfate, the solvent was evaporated under reduced pressure. The obtained residue was purified by silica gel column chromatography to afford the compound (9-2) (0.56 g).
[0254] 1H-NMR (CDCl3): 1.95 (3H, s), 3.62 (2H, s), 7.30-7.40 (4H, m).
Step 2
[0255] To a solution of the compound (9-2) (0.56 g) in tetrahydrofuran (10 ml), t-butylamine (0.24 g) was added and stirred at room temperature for 18 hours. The reaction solvent was evaporated under reduced pressure, then the obtained residue was purified by silica gel column chromatography to afford the compound (9-3) (190 mg).
[0256] 1H-NMR (CDCl3): 1.43 (9H, s), 1.56 (3H, s), 3.27 (1H, d, J=10.6 Hz), 3.36 (1H, d, J=10.6 Hz), 7.28 (2H, d, J=8.2 Hz), 7.43 (2H, d, J=8.2 Hz).
Step 3
[0257] To the compound (9-3) (190 mg), conc. hydrochloric acid (3 ml) was added, then stirred at 100° C. for 3 hours. To the reaction mixture, 6 mol/L sodium hydroxide was added to neutralize, the mixture was extracted with dichloromethane. The organic layer was dried with anhydrous sodium sulfate, and the solvent was evaporated under reduced pressure. The obtained residue was purified by silica gel column chromatography, then crystallized from dichloromethane/n-hexane to afford the compound (24) (110 mg).
[0258] 1H-NMR (CDCl3): 1.62 (3H, s), 3.47 (1H, d, J=10.6 Hz), 3.52 (1H, d, J=10.6 Hz), 4.59 (2H, br), 7.29 (2H, d, J=8.6 Hz), 7.39 (2H, d, J=8.6 Hz).
Example 10
The Synthesis of Compound 48
##STR00069##
[0259] Step 1
[0260] The compound (10-1) (79.6 mg) and (10-2) (120 mg) were dissolved into N,N-dimethylformamide (3 ml), then 1-hydroxybenzotriazole (54.6 mg) and N,N'-diisopropylcarbodiimide (0.063 ml) were added, then the reaction mixture was stirred overnight at room temperature. Then after the consumption of the compound (10-1), water was added, and the mixture was extracted with ethyl acetate. The organic layer was washed with brine, and dried over magnesium sulfate, and the solvent was evaporated under reduced pressure. The obtained residue was purified by silica gel column chromatography to afford the compound (10-3) (110.2 mg) as crude product of diastereomer.
[0261] 1H-NMR (CDCl3): 0.78-1.00 (6H, m), 1.14 (9/2H, e), 1.16 (9/2H, s) 1.52 (3/2H, s), 1.54 (3/2H, s) 1.86-2.28 (3H, m), 2.56-2.89 (2H, m), 3.80 (3/2H, s), 3.81 (3/2H, s) 4.04-4.14 (1H, m), 6.80-6.91 (2H, m), 7.08-7.22 (2H, m), 7.30-7.51 (6H, m), 7.61-7.76 (4H, m)
Step 2
[0262] The compound (10-3) (100 mg) was dissolved into tetrahydrofuran (3 ml) under nitrogen atmosphere, then 1 mol/L tetrabutylammonium fluoride in tetrahydrofuran (0.18 ml) was added at 0° C. with stirring, then the reaction mixture was stirred at 0° C. for 5 minutes. After the consumption of the compound (10-3), water was added, and the mixture was extracted with ethyl acetate. The organic layer was washed with brine, and dried over magnesium sulfate, then the solvent was evaporated under reduced pressure. The obtained residue was purified by silica gel column chromatography to afford the compound (48) (40.7 mg) as a mixture of diastereomers.
[0263] 1H-NMR (CDCl3): 0.80-0.90 (3H, m) 1.01-1.12 (3H, m) 1.70 (3H, m), 2.02-2.31 (2H, m) 2.39-2.55 (1H, m), 2.61-2.90 (2H, m) 3.53-3.70 (1H, m) 3.81 (3H, m), 3.96-4.08 (1H, m) 6.87-6.96 (2H, m), 7.13-7.22 (2H, m)
Example 11
The Synthesis of Compound 707
##STR00070##
[0264] Step 1
[0265] The compound (11-1) (150 mg) was dissolved into acetonitrile (5 ml), then the compound (11-2) (219.6 mg) was added at room temperature with stirring, and then the reaction mixture was warmed to 60° C., and stirred for 25 hours. The compound (11-1) was remained (checked by TLC). The reaction solvent was evaporated under reduced pressure, then the obtained residue was purified by silica gel column chromatography to afford the compound (11-1) (211.4 mg).
[0266] 1H-NMR (CDCl3, 400 MHz): 1.46 (9H, s), 1.50 (9H, s), 1.57 (3H, s), 1.90 (1H, ddd, J=13.7, 10.0, 3.8 Hz) 2.11 (1H, ddd, J=13.7, 6.5, 3.7 Hz) 2.68-2.76 (1H, m), 2.86-2.93 (1H, m), 3.88 (3H, s), 6.91 (1H, t, J=8.6 Hz) 6.99-7.03 (1H, m), 7.06 (1H, dd, J=13.0, 2.2 Hz), 10.14 (1H, s), 13.93 (1H, s)
Step 2
[0267] The compound (11-3) (210 mg) was dissolved into 4 mol/L hydrochloric acid in 1,4-dioxane (4 ml) under cooling with ice-water bath, then the mixture was warmed to room temperature and stirred for 67 hours. After the consumption of the compound (11-3) (checked by LC/MS), the reaction solvent was evaporated under reduced pressure. The obtained residue was crystallized from methanol-diethyl ether, and crystal was collected by filtration and washed with diethyl ether to afford compound (707) (140.2 mg).
[0268] 1H-NMR (DMSO-d6, 400 MHz): 1.56 (3H, s), 1.90-2.01 (1H, m), 2.43-2.62 (2H, m), 2.95-3.03 (1H, m), 3.84 (3H, s), 7.10-7.27 (3H, m), 7.76 (3H, br s), 8.26 (1H, br s), 9.42 (1H, s)
Example 12
The Synthesis of Compound 845
##STR00071##
[0269] Step 1
[0270] The compound (12-1) (50 mg) and piperidine (17.9 mg) were dissolved into N,N-dimethylformamide (2 ml), then O-(7-azabenzotriazo-1-yl)-1,1,3,3-tetramethyluroniumhexafluorophosphate (79.8 mg) was added, and then the mixture was stirred at room temperature for 40 hours. After the consumption of the compound (12-1), the solvent was evaporated under reduced pressure with heating. To the obtained residue, saturated sodium hydrogencarbonate solution was added, and extracted with ethyl acetate. The organic layer was washed with brine, and dried over magnesium sulfate, then the solvent was evaporated under reduced pressure. The obtained residue was purified by silica gel column chromatography to afford the compound (845) (30.7 mg).
[0271] 1H-NMR (CDCl3): 1.60 (3H, s), 1.51-1.82 (6H, m), 1.87-1.98 (1H, m), 2.09-2.19 (1H, m), 2.91-2.97 (2H, m), 3.64-3.68 (4H, m), 6.73 (1H, d, J=4.05 Hz), 7.14 (1H, d, J=4.05 Hz)
Example 13
The Synthesis of Compound 1262
##STR00072##
[0272] Step 1
[0273] The compound (13-1) (50.0 mg) was dissolved into tetrahydrofuran (1 ml) under nitrogen atmosphere, then triethylamine (19 μl), and 4-bromobenzoyl chloride (30.1 mg) were added under cooling with ice-water bath, and stirred for 40 minutes. The reaction solvent was evaporated under reduced pressure, and then the obtained residue was dissolved into ethyl acetate. The solution was washed with saturated sodium hydrogencarbonate solution, and dried over magnesium sulfate, and then the solvent was evaporated under reduced pressure. The generated crystal was collected by filtration to afford the compound (13-2) (57.2 mg).
[0274] 1H-NMR (CDCl3): 1.48 (9H, s), 1.68 (3H, s), 2.08 (1H, m), 2.44 (1H, m), 2.65 (1H, m), 2.78 (1H, m), 7.18 (1H, s), 7.32 (1H, s), 7.64 (2H, d, J=8.2 Hz), 7.78 (2H, d, J=8.2 Hz), 8.15 (1H, s), 8.25 (1H, br)
Step 2
[0275] The compound (13-2) (62.3 mg) was dissolved into 4 mol/L hydrochloric acid-1,4-dioxane and stirred for 24 hours. The reaction solvent was evaporated under reduced pressure. The obtained residue was crystallized from methanol/diethyl ether to afford the compound (1262) (44.7 mg).
[0276] 1H-NMR (DMSO-d6): 1.67 (3H, s), 2.10 (1H, m), 2.50-2.61 (3H, m), 7.33 (1H, s), 7.74 (1H, s), 7.77 (2H, d, J=8.6 Hz), 7.91 (2H, d, J=8.6 Hz), 8.08 (1H, s), 10.6 (1H, s)
Example 14
The Synthesis of Compound 753
##STR00073##
[0277] Step 1
[0278] The compound (14-1) (46 mg) was dissolved into dichloromethane (2 ml), then 4-chlorobenzaldehyde (20 mg) and acetic acid (17 mg) was added at room temperature, and then stirred for 20 minutes, and then sodium triacetoxyborohydride (45 mg) was added under cooling with ice-water bath. The mixture was stirred at room temperature for 14 hours, and then water was added and extracted with dichloromethane. The organic layer was dried over sodium sulfate, and then the solvent was evaporated under reduced pressure. The obtained residue was purified by silica gel column chromatography to afford the compound (14-2) (52 mg).
[0279] 1H-NMR(CDCl3): 1.50 (9H, s), 1.64 (3H, s), 2.02-2.10 (1H, m), 2.40 (1H, dt, J=14.0, 4.1 Hz), 2.62-2.74 (2H, m), 4.30 (2H, s), 6.49 (1H, ddd, J=, 7.8, 2.0, 0.8 Hz), 6.52 (1H, t, J=2.0 Hz), 6.60 (1H, ddd, J=, 7.8, 2.0, 0.8 Hz), 7.16 (1H, t, J=7.8 Hz), 7.18-7.33 (4H, m).
Step 2
[0280] To the compound (14-2) (52 mg), 4 mol/L hydrochloric acid in 1,4-dioxane solution (4 ml) was added, then the mixture was stirred at room temperature for 4 days, and then the reaction solvent was evaporated under reduced pressure. The obtained residue was crystallized from methanol/diethyl ether to afford the compound (753) (42 mg).
[0281] 1H-NMR (DMSO-d6): 1.58 (3H, s), 2.00 (1H, ddd, J=, 14.3, 11.3, 3.3 Hz),2.49-2.57 (2H, m), 3.07 (1H, dt, J=12.7, 3.3 Hz), 4.27 (2H, s), 6.47 (1H, d, J=8.2 Hz), 6.51-6.53 (2H, m), 7.08 (1H, t, J=8.2 Hz), 7.37 (4H, s), 8.80 (2.H, br).
Example 15
The Synthesis of Compound 1135
##STR00074##
[0282] Step 1
[0283] To a solution of the compound (15-1) (101 mg), 2-propanol (56 μl), and triphenylphosphine (189 mg) in tetrahydrofuran (2 ml), diethyl azodicarboxylate (2.2 mol/L) in toluene (328 μl) was added dropwise, then stirred for 1 hour at room temperature. After the consumption of the compound (15-1), the solvent was evaporated under reduced pressure. The obtained residue was purified by silica gel column chromatography to afford the compound (15-2) (280 mg) as a mixture of triphenylphosphine oxide and diethyl hydrazodicarboxylate.
Step 2
[0284] To the suspension of 5-chloropyridine-2-carboxylic acid (47 mg) in toluene (1 ml), N,N-dimethylformamide (1 drop) and tlaionylchloride (91 μl) were added and stirred at 100° C. for 1 hour. The solvent was evaporated under reduced pressure, then the obtained residue was dissolved into tetrahydrofuran (1 ml), and then the mixture of the compound (15-2) (280 mg), and pyridine (194 μl) in tetrahydrofuran (0.5 ml) were added dropwise at 0° C. and stirred for 10 minutes. After the consumption of the compound (15-2), water was added and the mixture was extracted with ethyl acetate. The organic layer was washed with water, and then the solvent was evaporated under reduced pressure. The obtained residue was purified by silica gel column chromatography to afford the compound (15-3) (68 mg) as a mixture of diethyl hydrazodicarboxylate.
Step 3
[0285] To the compound (15-3) (68 mg) as a mixture of diethyl hydrazodicarboxylate, 4 mol/L in hydrochloric acid in 1,4-dioxane solution (1 ml) was added, then the mixture was stirred at room temperature for 16 hours. After the consumption of the compound (44), the reaction solvent was evaporated under reduced pressure. The obtained residue was crystallized from 2-propanol/diethyl ether to afford the compound (1135) (36 mg).
[0286] 1H-NMR (DMSO-6): 1.30 (3H, d, J=6.4 Hz), 1.31 (3H, d, J=6.4 Hz), 1.65 (3H, s), 2.04-2.11 (1H, m), 2.50-2.64 (2H, m), 3.12-3.16 (1H, m), 4.61 (1H, sep, J=6.4 Hz), 6.66 (1H, t, J=2.0 Hz), 7.48 (1H, t, J=2.0 Hz), 7.60 (1H, t, J=2.0 Hz), 8.16 (1H, dd, J=8.4, 0.8 Hz), 8.22 (1H, dd, J=8.4, 2.4 Hz), 8.79 (1H, dd, J=2.4, 0.8 Hz), 10.33 (1H, a), 10.72 (1H, s).
Example 16
The Synthesis of Compound 161
##STR00075##
[0287] Step 1
[0288] The compound (16-1) (200 mg), palladium acetate (4.7 mg), and tri-(o-tolyl)phosphine (12.5 mg), were dissolved into N,N-dimethylformamide (2 ml) under nitrogen atmosphere, then n-butylamine (0.196 ml), and p-chlorostyrene (0.074 ml) were added at room temperature with stirring, then the solution was warmed to 80° C., and stirred for 3 hours. After the consumption of the compound (16-1) (checked by TLC), the reaction mixture was cooled to room temperature, and saturated ammonium chloride solution was added to the mixture. The mixture was extracted with ethyl acetate, the organic layer was washed with water and brine, and dried over magnesium sulfate, and then the solvent was evaporated under reduced pressure. The obtained residue was purified by silica gel column chromatography to afford the compound (16-2) (213.1 mg).
[0289] 1H-NMR (CDCl3, 400 MHz): 1.54 (18H, s), 1.64 (3H, s), 1.96 (1H, ddd, J=13.7, 9.1, 4.0 Hz) 2.10 (1H, ddd, J=13.7, 8.1, 3.4 Hz) 2.86 (1H, ddd, J=12.3, 9.1, 3.4 Hz), 3.03 (1H, ddd, J=12.3, 8.1, 4.0 Hz), 7.08 (1H, d, J=16.4 Hz) 7.15 (1H, d, J=16.4 Hz), 7.27-7.40 (5H, m) 7.44 (2H, d, J=8.8 Hz), 7.58 (1H, 5)
Step 2
[0290] The compound (16-2) (213 mg) was dissolved into 4 mol/L hydrochloric acid in 1,4-dioxane (5 ml) under cooling with ice-water bath, then the mixture was warmed to room temperature and stirred for 63 hours. After the consumption of the compound (16-2) (checked by LC/MS), the reaction mixture was diluted with diethyl ether. The generated crystal was collected by filtration, and washed with diethyl ether to afford the compound (161) (108.6 mg).
[0291] 1H-NMR (DMSO-d6, 400 MHz): 1.69 (3H, s), 2.08-2.18 (1H, m), 2.56-2.70 (2H, m), 3.13-3.20 (1H, m), 7.23 (1H, d, J=8.0 Hz), 7.31 (1H, d, J=17.0 Hz), 7.35 (1H, d, J=17.0 Hz), 7.45 (2H, d, J=8.6 Hz), 7.46 (1H, t, 7.6 Hz), 7.59 (1H, d, J=2.0 Hz), 7.61-7.64 (1H, m), 7.64 (2H, d, J=8.6 Hz), 8.53-9.50 (2H, br), 10.67 (1H, br
Example 17
The Synthesis of Compound 597
##STR00076##
[0292] Step 1
[0293] The solution of compound (17-1) (135 mg), O-methxylhydroxylamine hydrochloride (39 mg), and potassium acetate (27 mg) in methanol (3 ml) was stirred at room temperature for 16 hours, then water was added. The mixture was extracted with dichloromethane, the organic layer was dried over anhydrous sodium sulfate, then the solvent was evaporated under reduced pressure. The obtained residue was purified by silica gel column chromatography to afford the compound (17-2) (110 mg).
[0294] 1H-NMR (CDCl3): 1.51 (9H, s), 1.70 (3H, s), 2.14 (1H, ddd, J=14.4, 11.4, 3.4 Hz), 2.22 (3H, s), 2.48 (1H, m), 2.65 (1H, dt, J=12.6, 11.4 Hz), 2.78 (1H, ddd, J=12.6, 5.6, 3.4 Hz), 4.00 (3H, 5), 7.30 (1H, d, J=7.8 Hz), 7.38 (1H, d, J=7.8 Hz), 7.54-7.57 (2H, m).
Step 2
[0295] To the compound (17-2) (110 mg), 4 mol/L hydrochloric acid in 1,4-dioxane (4.5 ml) solution was added and stirred for 4 days at room temperature, then the reaction solvent was evaporated under reduced pressure. The obtained residue was crystallized from methanol/diethyl ether to afford compound (597) (65 mg).
[0296] 1H-NMR (DMSO-d6): 1.67 (3H, s), 2.08-2.15 (1H, m), 2.20 (3H, s), 2.56-2.64 (2H, m), 3.14-3.17 (1H, m), 3.92 (3H, s), 7.37 (1H, d, J=8.0 Hz), 7.48 (1H, d, J=8.0 Hz), 7.56 (1H, s), 7.62 (1H, d, J=8.0 Hz).
[0297] The other compounds were synthesized in the same way. The structural formulas and physical constants are shown below.
TABLE-US-00001 TABLE 1 Compound No. Chemical structure 1 ##STR00077## 2 ##STR00078## 3 ##STR00079## 4 ##STR00080## 5 ##STR00081## 6 ##STR00082## 7 ##STR00083## 8 ##STR00084## 9 ##STR00085##
TABLE-US-00002 TABLE 2 10 ##STR00086## 11 ##STR00087## 12 ##STR00088## 13 ##STR00089## 14 ##STR00090## 15 ##STR00091## 16 ##STR00092## 17 ##STR00093## 18 ##STR00094##
TABLE-US-00003 TABLE 3 19 ##STR00095## 20 ##STR00096## 21 ##STR00097## 22 ##STR00098## 23 ##STR00099## 24 ##STR00100## 25 ##STR00101## 26 ##STR00102## 27 ##STR00103## 28 ##STR00104##
TABLE-US-00004 TABLE 4 29 ##STR00105## 30 ##STR00106## 31 ##STR00107## 32 ##STR00108## 33 ##STR00109## 34 ##STR00110## 35 ##STR00111## 36 ##STR00112## 37 ##STR00113## 38 ##STR00114## 39 ##STR00115##
TABLE-US-00005 TABLE 5 40 ##STR00116## 41 ##STR00117## 42 ##STR00118## 43 ##STR00119## 44 ##STR00120## 45 ##STR00121## 46 ##STR00122## 47 ##STR00123##
TABLE-US-00006 TABLE 6 48 ##STR00124## 49 ##STR00125## 50 ##STR00126## 51 ##STR00127## 52 ##STR00128## 53 ##STR00129## 54 ##STR00130## 55 ##STR00131##
TABLE-US-00007 TABLE 7 56 ##STR00132## 57 ##STR00133## 58 ##STR00134## 59 ##STR00135## 60 ##STR00136## 61 ##STR00137## 62 ##STR00138## 63 ##STR00139## 64 ##STR00140## 65 ##STR00141##
TABLE-US-00008 TABLE 8 66 ##STR00142## 67 ##STR00143## 68 ##STR00144## 69 ##STR00145## 70 ##STR00146## 71 ##STR00147## 72 ##STR00148## 73 ##STR00149## 74 ##STR00150##
TABLE-US-00009 TABLE 9 75 ##STR00151## 76 ##STR00152## 77 ##STR00153## 78 ##STR00154## 79 ##STR00155## 80 ##STR00156## 81 ##STR00157## 82 ##STR00158##
TABLE-US-00010 TABLE 10 83 ##STR00159## 84 ##STR00160## 85 ##STR00161## 86 ##STR00162## 87 ##STR00163## 88 ##STR00164## 89 ##STR00165## 90 ##STR00166## 91 ##STR00167##
TABLE-US-00011 TABLE 11 92 ##STR00168## 93 ##STR00169## 94 ##STR00170## 95 ##STR00171## 96 ##STR00172## 97 ##STR00173## 98 ##STR00174## 99 ##STR00175## 100 ##STR00176## 101 ##STR00177##
TABLE-US-00012 TABLE 12 102 ##STR00178## 103 ##STR00179## 104 ##STR00180## 105 ##STR00181## 106 ##STR00182## 107 ##STR00183## 108 ##STR00184## 109 ##STR00185##
TABLE-US-00013 TABLE 13 110 ##STR00186## 111 ##STR00187## 112 ##STR00188## 113 ##STR00189## 114 ##STR00190## 115 ##STR00191## 116 ##STR00192## 117 ##STR00193## 118 ##STR00194## 119 ##STR00195##
TABLE-US-00014 TABLE 14 120 ##STR00196## 121 ##STR00197## 122 ##STR00198## 124 ##STR00199## 125 ##STR00200## ##STR00201## 126 ##STR00202## 127 ##STR00203## 128 ##STR00204## 129 ##STR00205## 130 ##STR00206##
TABLE-US-00015 TABLE 15 131 ##STR00207## 132 ##STR00208## 133 ##STR00209## 134 ##STR00210## 135 ##STR00211## 136 ##STR00212## 137 ##STR00213## 138 ##STR00214## 139 ##STR00215## 140 ##STR00216## 141 ##STR00217##
TABLE-US-00016 TABLE 16 142 ##STR00218## 143 ##STR00219## 144 ##STR00220## 145 ##STR00221## 146 ##STR00222## 147 ##STR00223## 148 ##STR00224## 149 ##STR00225## 150 ##STR00226##
TABLE-US-00017 TABLE 17 151 ##STR00227## 152 ##STR00228## 153 ##STR00229## 154 ##STR00230## 155 ##STR00231## 156 ##STR00232## 157 ##STR00233## 158 ##STR00234## 159 ##STR00235##
TABLE-US-00018 TABLE 18 160 ##STR00236## 161 ##STR00237## 162 ##STR00238## 163 ##STR00239## 164 ##STR00240## 165 ##STR00241## 166 ##STR00242##
TABLE-US-00019 TABLE 19 167 ##STR00243## 168 ##STR00244## 169 ##STR00245## 170 ##STR00246## 171 ##STR00247## 172 ##STR00248## 173 ##STR00249## 174 ##STR00250## 175 ##STR00251## 176 ##STR00252##
TABLE-US-00020 TABLE 20 177 ##STR00253## 178 ##STR00254## 179 ##STR00255## 180 ##STR00256## 181 ##STR00257## 182 ##STR00258## 183 ##STR00259## 184 ##STR00260## 185 ##STR00261## 186 ##STR00262## 187 ##STR00263## 188 ##STR00264##
TABLE-US-00021 TABLE 21 189 ##STR00265## 190 ##STR00266## 191 ##STR00267## 192 ##STR00268## 193 ##STR00269## 194 ##STR00270## 195 ##STR00271## 196 ##STR00272##
TABLE-US-00022 TABLE 22 197 ##STR00273## 198 ##STR00274## 199 ##STR00275## 200 ##STR00276## 201 ##STR00277## 202 ##STR00278## 203 ##STR00279## 204 ##STR00280## 205 ##STR00281## 206 ##STR00282##
TABLE-US-00023 TABLE 23 207 ##STR00283## 208 ##STR00284## 209 ##STR00285## 210 ##STR00286## 211 ##STR00287## 212 ##STR00288## 213 ##STR00289## 214 ##STR00290## 215 ##STR00291##
TABLE-US-00024 TABLE 24 216 ##STR00292## 217 ##STR00293## 218 ##STR00294## 219 ##STR00295## 220 ##STR00296## 221 ##STR00297## 222 ##STR00298## 223 ##STR00299## 224 ##STR00300##
TABLE-US-00025 TABLE 25 225 ##STR00301## 226 ##STR00302## 227 ##STR00303## 228 ##STR00304## 229 ##STR00305## 230 ##STR00306## 231 ##STR00307## 232 ##STR00308## 233 ##STR00309## 234 ##STR00310##
TABLE-US-00026 TABLE 26 235 ##STR00311## 236 ##STR00312## 237 ##STR00313## 238 ##STR00314## 239 ##STR00315## 240 ##STR00316## 241 ##STR00317## 242 ##STR00318## 243 ##STR00319## 244 ##STR00320## 245 ##STR00321## 246 ##STR00322##
TABLE-US-00027 TABLE 27 247 ##STR00323## 248 ##STR00324## 249 ##STR00325## 250 ##STR00326## 251 ##STR00327## 252 ##STR00328## 253 ##STR00329## 254 ##STR00330## 255 ##STR00331##
TABLE-US-00028 TABLE 28 256 ##STR00332## 257 ##STR00333## 258 ##STR00334## 259 ##STR00335## 260 ##STR00336## 261 ##STR00337## 262 ##STR00338## 263 ##STR00339## 264 ##STR00340## 265 ##STR00341## 266 ##STR00342##
TABLE-US-00029 TABLE 29 267 ##STR00343## 268 ##STR00344## 269 ##STR00345## 270 ##STR00346## 271 ##STR00347## 272 ##STR00348## 273 ##STR00349## 274 ##STR00350## 275 ##STR00351##
TABLE-US-00030 TABLE 30 276 ##STR00352## 277 ##STR00353## 278 ##STR00354## 279 ##STR00355## 280 ##STR00356## 281 ##STR00357## 282 ##STR00358## 283 ##STR00359## 284 ##STR00360##
TABLE-US-00031 TABLE 31 285 ##STR00361## 286 ##STR00362## 287 ##STR00363## 288 ##STR00364## 289 ##STR00365## 290 ##STR00366## 291 ##STR00367## 292 ##STR00368## 293 ##STR00369##
TABLE-US-00032 TABLE 32 294 ##STR00370## 295 ##STR00371## 296 ##STR00372## 297 ##STR00373## 298 ##STR00374## 299 ##STR00375## 300 ##STR00376## 301 ##STR00377## 302 ##STR00378##
TABLE-US-00033 TABLE 33 303 ##STR00379## 304 ##STR00380## 305 ##STR00381## 306 ##STR00382## 307 ##STR00383## 308 ##STR00384## 309 ##STR00385##
TABLE-US-00034 TABLE 34 310 ##STR00386## 311 ##STR00387## 312 ##STR00388## 313 ##STR00389## 314 ##STR00390## 315 ##STR00391## 316 ##STR00392## 317 ##STR00393##
TABLE-US-00035 TABLE 35 318 ##STR00394## 319 ##STR00395## 320 ##STR00396## 321 ##STR00397## 322 ##STR00398## 323 ##STR00399## 324 ##STR00400## 325 ##STR00401## 326 ##STR00402## 327 ##STR00403## 328 ##STR00404##
TABLE-US-00036 TABLE 36 329 ##STR00405## 330 ##STR00406## 331 ##STR00407## 332 ##STR00408## 333 ##STR00409## 334 ##STR00410## 335 ##STR00411## 336 ##STR00412## 337 ##STR00413## 338 ##STR00414## 339 ##STR00415##
TABLE-US-00037 TABLE 37 340 ##STR00416## 341 ##STR00417## 342 ##STR00418## 343 ##STR00419## 344 ##STR00420## 345 ##STR00421## 346 ##STR00422## 347 ##STR00423## 348 ##STR00424##
TABLE-US-00038 TABLE 38 349 ##STR00425## 350 ##STR00426## 351 ##STR00427## 352 ##STR00428## 353 ##STR00429## 354 ##STR00430## 355 ##STR00431## 356 ##STR00432## 357 ##STR00433##
TABLE-US-00039 TABLE 39 358 ##STR00434## 359 ##STR00435## 360 ##STR00436## 361 ##STR00437## 362 ##STR00438## 363 ##STR00439## 364 ##STR00440## 365 ##STR00441## 366 ##STR00442## 367 ##STR00443## 368 ##STR00444## 369 ##STR00445##
TABLE-US-00040 TABLE 40 370 ##STR00446## 371 ##STR00447## 372 ##STR00448## 373 ##STR00449## 374 ##STR00450## 375 ##STR00451## 376 ##STR00452## 377 ##STR00453## 378 ##STR00454##
TABLE-US-00041 TABLE 41 379 ##STR00455## 380 ##STR00456## 381 ##STR00457## 382 ##STR00458## 383 ##STR00459## 384 ##STR00460## 385 ##STR00461## 386 ##STR00462## 387 ##STR00463## 388 ##STR00464## 389 ##STR00465##
TABLE-US-00042 TABLE 42 390 ##STR00466## 391 ##STR00467## 392 ##STR00468## 393 ##STR00469## 394 ##STR00470## 395 ##STR00471## 396 ##STR00472## 397 ##STR00473##
TABLE-US-00043 TABLE 43 398 ##STR00474## 399 ##STR00475## 400 ##STR00476## 401 ##STR00477## 402 ##STR00478## 403 ##STR00479## 404 ##STR00480## 405 ##STR00481## 406 ##STR00482## 407 ##STR00483##
TABLE-US-00044 TABLE 44 408 ##STR00484## 409 ##STR00485## 410 ##STR00486## 411 ##STR00487## 412 ##STR00488## 413 ##STR00489## 414 ##STR00490## 415 ##STR00491## 416 ##STR00492##
TABLE-US-00045 TABLE 45 417 ##STR00493## 418 ##STR00494## 419 ##STR00495## 420 ##STR00496## 421 ##STR00497## 422 ##STR00498## 423 ##STR00499## 424 ##STR00500##
TABLE-US-00046 TABLE 46 425 ##STR00501## 426 ##STR00502## 427 ##STR00503## 428 ##STR00504## 429 ##STR00505## 430 ##STR00506## 431 ##STR00507## 432 ##STR00508## 433 ##STR00509##
TABLE-US-00047 TABLE 47 434 ##STR00510## 435 ##STR00511## 436 ##STR00512## 437 ##STR00513## 438 ##STR00514## 439 ##STR00515## 440 ##STR00516## 441 ##STR00517## 442 ##STR00518##
TABLE-US-00048 TABLE 48 443 ##STR00519## 444 ##STR00520## 445 ##STR00521## 446 ##STR00522## 447 ##STR00523## 448 ##STR00524## 449 ##STR00525## 450 ##STR00526## 451 ##STR00527## 452 ##STR00528## 453 ##STR00529##
TABLE-US-00049 TABLE 49 454 ##STR00530## 455 ##STR00531## 456 ##STR00532## 457 ##STR00533## 458 ##STR00534## 459 ##STR00535## 460 ##STR00536## 461 ##STR00537## 462 ##STR00538## 463 ##STR00539## 464 ##STR00540##
TABLE-US-00050 TABLE 50 465 ##STR00541## 466 ##STR00542## 467 ##STR00543## 468 ##STR00544## 469 ##STR00545## 470 ##STR00546## 471 ##STR00547## 472 ##STR00548## 473 ##STR00549## 474 ##STR00550##
TABLE-US-00051 TABLE 51 475 ##STR00551## 476 ##STR00552## 477 ##STR00553## 478 ##STR00554## 479 ##STR00555## 480 ##STR00556## 481 ##STR00557## 482 ##STR00558## 483 ##STR00559##
TABLE-US-00052 TABLE 52 484 ##STR00560## 485 ##STR00561## 486 ##STR00562## 487 ##STR00563## 488 ##STR00564## 489 ##STR00565## 490 ##STR00566## 491 ##STR00567## 492 ##STR00568##
TABLE-US-00053 TABLE 53 493 ##STR00569## 494 ##STR00570## 495 ##STR00571## 496 ##STR00572## 497 ##STR00573## 498 ##STR00574## 499 ##STR00575## 500 ##STR00576## 501 ##STR00577## 502 ##STR00578## 503 ##STR00579##
TABLE-US-00054 TABLE 54 504 ##STR00580## 505 ##STR00581## 506 ##STR00582## 507 ##STR00583## 508 ##STR00584## 509 ##STR00585## 510 ##STR00586## 511 ##STR00587## 512 ##STR00588## 513 ##STR00589## 514 ##STR00590##
TABLE-US-00055 TABLE 55 515 ##STR00591## 516 ##STR00592## 517 ##STR00593## 518 ##STR00594## 519 ##STR00595## 520 ##STR00596## 521 ##STR00597## 522 ##STR00598## 523 ##STR00599## 524 ##STR00600##
TABLE-US-00056 TABLE 56 525 ##STR00601## 526 ##STR00602## 527 ##STR00603## 528 ##STR00604## 529 ##STR00605## 530 ##STR00606## 531 ##STR00607## 532 ##STR00608## 533 ##STR00609## 534 ##STR00610## 535 ##STR00611##
TABLE-US-00057 TABLE 57 536 ##STR00612## 537 ##STR00613## 538 ##STR00614## 539 ##STR00615## 540 ##STR00616## 541 ##STR00617## 542 ##STR00618## 543 ##STR00619## 544 ##STR00620## 545 ##STR00621## 546 ##STR00622##
TABLE-US-00058 TABLE 58 547 ##STR00623## 548 ##STR00624## 549 ##STR00625## 550 ##STR00626## 551 ##STR00627## 552 ##STR00628## 553 ##STR00629## 554 ##STR00630## 555 ##STR00631##
TABLE-US-00059 TABLE 59 556 ##STR00632## 557 ##STR00633## 558 ##STR00634## 559 ##STR00635## 560 ##STR00636## 561 ##STR00637## 562 ##STR00638## 563 ##STR00639## 564 ##STR00640## 565 ##STR00641## 566 ##STR00642##
TABLE-US-00060 TABLE 60 567 ##STR00643## 568 ##STR00644## 569 ##STR00645## 570 ##STR00646## 571 ##STR00647## 572 ##STR00648## 573 ##STR00649## 574 ##STR00650##
TABLE-US-00061 TABLE 61 ##STR00651## 575 ##STR00652## 576 ##STR00653## 577 ##STR00654## 578 ##STR00655## 579 ##STR00656## 580 ##STR00657## 581 ##STR00658## 582 ##STR00659## 583 ##STR00660## 584
TABLE-US-00062 TABLE 62 ##STR00661## 585 ##STR00662## 586 ##STR00663## 587 ##STR00664## 588 ##STR00665## 589 ##STR00666## 590 ##STR00667## 591
TABLE-US-00063 TABLE 63 ##STR00668## 592 ##STR00669## 593 ##STR00670## 594 ##STR00671## 595 ##STR00672## 596 ##STR00673## 597 ##STR00674## 598 ##STR00675## 599 ##STR00676## 600 ##STR00677## 601 ##STR00678## 602
TABLE-US-00064 TABLE 64 ##STR00679## 603 ##STR00680## 604 ##STR00681## 605 ##STR00682## 606 ##STR00683## 607 ##STR00684## 608 ##STR00685## 609 ##STR00686## 610 ##STR00687## 611 ##STR00688## 612 ##STR00689## 613
TABLE-US-00065 TABLE 65 ##STR00690## 614 ##STR00691## 615 ##STR00692## 616 ##STR00693## 617 ##STR00694## 618 ##STR00695## 619 ##STR00696## 620 ##STR00697## 621 ##STR00698## 623 ##STR00699## 624
TABLE-US-00066 TABLE 66 ##STR00700## 625 ##STR00701## 626 ##STR00702## 627 ##STR00703## 628 ##STR00704## 629 ##STR00705## 630 ##STR00706## 631 ##STR00707## 632 ##STR00708## 633 ##STR00709## 634 ##STR00710## 635
TABLE-US-00067 TABLE 67 ##STR00711## 636 ##STR00712## 637 ##STR00713## 638 ##STR00714## 639 ##STR00715## 640 ##STR00716## 641 ##STR00717## 642 ##STR00718## 643
TABLE-US-00068 TABLE 68 ##STR00719## 644 ##STR00720## 645 ##STR00721## 646 ##STR00722## 647 ##STR00723## 648 ##STR00724## 649 ##STR00725## 650 ##STR00726## 651 ##STR00727## 652 ##STR00728## 653 ##STR00729## 654 ##STR00730## 655 ##STR00731## 656
TABLE-US-00069 TABLE 69 ##STR00732## 657 ##STR00733## 658 ##STR00734## 659 ##STR00735## 660 ##STR00736## 661 ##STR00737## 662 ##STR00738## 663 ##STR00739## 664 ##STR00740## 665
TABLE-US-00070 TABLE 70 ##STR00741## 666 ##STR00742## 667 ##STR00743## 668 ##STR00744## 669 ##STR00745## 670 ##STR00746## 671 ##STR00747## 672 ##STR00748## 673 ##STR00749## 674
TABLE-US-00071 TABLE 71 ##STR00750## 675 ##STR00751## 676 ##STR00752## 677 ##STR00753## 678 ##STR00754## 679 ##STR00755## 680 ##STR00756## 681 ##STR00757## 682 ##STR00758## 683
TABLE-US-00072 TABLE 72 ##STR00759## 684 ##STR00760## 685 ##STR00761## 686 ##STR00762## 687 ##STR00763## 688 ##STR00764## 689 ##STR00765## 690 ##STR00766## 691 ##STR00767## 692
TABLE-US-00073 TABLE 73 ##STR00768## 693 ##STR00769## 694 ##STR00770## 695 ##STR00771## 696 ##STR00772## 697 ##STR00773## 698 ##STR00774## 699 ##STR00775## 700 ##STR00776## 701 ##STR00777## 702
TABLE-US-00074 TABLE 74 ##STR00778## 703 ##STR00779## 704 ##STR00780## 705 ##STR00781## 706 ##STR00782## 707 ##STR00783## 708 ##STR00784## 709 ##STR00785## 710 ##STR00786## 711 ##STR00787## 712
TABLE-US-00075 TABLE 75 ##STR00788## 713 ##STR00789## 714 ##STR00790## 715 ##STR00791## 716 ##STR00792## 717 ##STR00793## 718 ##STR00794## 719
TABLE-US-00076 TABLE 76 720 ##STR00795## 721 ##STR00796## 722 ##STR00797## 723 ##STR00798## 724 ##STR00799## 725 ##STR00800## 728 ##STR00801## 727 ##STR00802## 728 ##STR00803## 729 ##STR00804##
TABLE-US-00077 TABLE 77 730 ##STR00805## 731 ##STR00806## 732 ##STR00807## 734 ##STR00808## 735 ##STR00809## 736 ##STR00810## 737 ##STR00811## 738 ##STR00812## 739 ##STR00813## 740 ##STR00814##
TABLE-US-00078 TABLE 78 741 ##STR00815## 742 ##STR00816## 743 ##STR00817## 744 ##STR00818## 745 ##STR00819## 746 ##STR00820## 747 ##STR00821## 748 ##STR00822## 749 ##STR00823##
TABLE-US-00079 TABLE 79 750 ##STR00824## 751 ##STR00825## 752 ##STR00826## 753 ##STR00827## 754 ##STR00828## 755 ##STR00829## 756 ##STR00830## 757 ##STR00831## 758 ##STR00832## 759 ##STR00833##
TABLE-US-00080 TABLE 80 760 ##STR00834## 761 ##STR00835## 762 ##STR00836## 763 ##STR00837## 764 ##STR00838## 765 ##STR00839## 766 ##STR00840## 767 ##STR00841## 768 ##STR00842## 769 ##STR00843##
TABLE-US-00081 TABLE 81 770 ##STR00844## 771 ##STR00845## 772 ##STR00846## 773 ##STR00847## 774 ##STR00848## 775 ##STR00849## 776 ##STR00850## 777 ##STR00851##
TABLE-US-00082 TABLE 82 778 ##STR00852## 779 ##STR00853## 780 ##STR00854## 781 ##STR00855## 782 ##STR00856## 783 ##STR00857## 784 ##STR00858## 785 ##STR00859## 786 ##STR00860## 787 ##STR00861## 788 ##STR00862## 789 ##STR00863##
TABLE-US-00083 TABLE 83 790 ##STR00864## 791 ##STR00865## 792 ##STR00866## 793 ##STR00867## 794 ##STR00868## 795 ##STR00869## 796 ##STR00870## 797 ##STR00871## 798 ##STR00872##
TABLE-US-00084 TABLE 84 799 ##STR00873## 800 ##STR00874## 801 ##STR00875## 802 ##STR00876## 803 ##STR00877## 804 ##STR00878## 805 ##STR00879## 806 ##STR00880## 807 ##STR00881## 808 ##STR00882## 809 ##STR00883##
TABLE-US-00085 TABLE 85 810 ##STR00884## 811 ##STR00885## 812 ##STR00886## 813 ##STR00887## 814 ##STR00888## 815 ##STR00889## 816 ##STR00890## 817 ##STR00891## 818 ##STR00892##
TABLE-US-00086 TABLE 86 819 ##STR00893## 820 ##STR00894## 821 ##STR00895## 822 ##STR00896## 823 ##STR00897## 824 ##STR00898## 825 ##STR00899## 826 ##STR00900## 827 ##STR00901##
TABLE-US-00087 TABLE 87 828 ##STR00902## 829 ##STR00903## 830 ##STR00904## 831 ##STR00905## 832 ##STR00906## 833 ##STR00907## 834 ##STR00908## 835 ##STR00909## 836 ##STR00910##
TABLE-US-00088 TABLE 88 837 ##STR00911## 838 ##STR00912## 839 ##STR00913## 840 ##STR00914## 841 ##STR00915## 842 ##STR00916## 843 ##STR00917## 844 ##STR00918## 845 ##STR00919##
TABLE-US-00089 TABLE 89 846 ##STR00920## 847 ##STR00921## 848 ##STR00922## 849 ##STR00923## 850 ##STR00924## 851 ##STR00925## 852 ##STR00926## 853 ##STR00927## 854 ##STR00928## 855 ##STR00929##
TABLE-US-00090 TABLE 90 856 ##STR00930## 857 ##STR00931## 858 ##STR00932## 859 ##STR00933## 860 ##STR00934## 861 ##STR00935## 862 ##STR00936## 863 ##STR00937## 864 ##STR00938## 865 ##STR00939##
TABLE-US-00091 TABLE 91 ##STR00940## 866 ##STR00941## 867 ##STR00942## 868 ##STR00943## 869 ##STR00944## 870 ##STR00945## 871 ##STR00946## 872 ##STR00947## 873 ##STR00948## 874 ##STR00949## 875
TABLE-US-00092 TABLE 92 ##STR00950## 876 ##STR00951## 877 ##STR00952## 878 ##STR00953## 879 ##STR00954## 880 ##STR00955## 881 ##STR00956## 882 ##STR00957## 883
TABLE-US-00093 TABLE 93 ##STR00958## 884 ##STR00959## 885 ##STR00960## 886 ##STR00961## 887 ##STR00962## 888 ##STR00963## 889 ##STR00964## 890 ##STR00965## 891 ##STR00966## 892 ##STR00967## 893 ##STR00968## 894
TABLE-US-00094 TABLE 94 ##STR00969## 895 ##STR00970## 896 ##STR00971## 897 ##STR00972## 898 ##STR00973## 899 ##STR00974## 900 ##STR00975## 901 ##STR00976## 902 ##STR00977## 903 ##STR00978## 904
TABLE-US-00095 TABLE 95 ##STR00979## 905 ##STR00980## 906 ##STR00981## 907 ##STR00982## 908 ##STR00983## 909 ##STR00984## 910 ##STR00985## 911 ##STR00986## 912 ##STR00987## 913 ##STR00988## 914
TABLE-US-00096 TABLE 96 ##STR00989## 915 ##STR00990## 916 ##STR00991## 917 ##STR00992## 918 ##STR00993## 919 ##STR00994## 920 ##STR00995## 921 ##STR00996## 922
TABLE-US-00097 TABLE 97 ##STR00997## 923 ##STR00998## 924 ##STR00999## 925 ##STR01000## 926 ##STR01001## 927 ##STR01002## 928 ##STR01003## 929 ##STR01004## 930 ##STR01005## 931 ##STR01006## 932
TABLE-US-00098 TABLE 98 ##STR01007## 933 ##STR01008## 934 ##STR01009## 935 ##STR01010## 936 ##STR01011## 937 ##STR01012## 938 ##STR01013## 939 ##STR01014## 940
TABLE-US-00099 TABLE 99 ##STR01015## 941 ##STR01016## 942 ##STR01017## 943 ##STR01018## 944 ##STR01019## 945 ##STR01020## 946 ##STR01021## 947 ##STR01022## 948 ##STR01023## 949
TABLE-US-00100 TABLE 100 ##STR01024## 950 ##STR01025## 951 ##STR01026## 952 ##STR01027## 953 ##STR01028## 954 ##STR01029## 955 ##STR01030## 956 ##STR01031## 957
TABLE-US-00101 TABLE 101 ##STR01032## 958 ##STR01033## 959 ##STR01034## 960 ##STR01035## 961 ##STR01036## 962 ##STR01037## 963 ##STR01038## 964
TABLE-US-00102 TABLE 102 ##STR01039## 965 ##STR01040## 966 ##STR01041## 967 ##STR01042## 968 ##STR01043## 969 ##STR01044## 970 ##STR01045## 971 ##STR01046## 972
TABLE-US-00103 TABLE 103 ##STR01047## 973 ##STR01048## 974 ##STR01049## 975 ##STR01050## 976 ##STR01051## 977 ##STR01052## 978 ##STR01053## 979 ##STR01054## 980
TABLE-US-00104 TABLE 104 ##STR01055## 981 ##STR01056## 982 ##STR01057## 983 ##STR01058## 984 ##STR01059## 985 ##STR01060## 986 ##STR01061## 987 ##STR01062## 988 ##STR01063## 989
TABLE-US-00105 TABLE 105 ##STR01064## 990 ##STR01065## 991 ##STR01066## 992 ##STR01067## 993 ##STR01068## 994 ##STR01069## 995 ##STR01070## 996 ##STR01071## 997 ##STR01072## 998
TABLE-US-00106 TABLE 106 990 ##STR01073## 1000 ##STR01074## 1001 ##STR01075## 1002 ##STR01076## 1003 ##STR01077## 1004 ##STR01078## 1005 ##STR01079## 1006 ##STR01080## 1007 ##STR01081##
TABLE-US-00107 TABLE 107 1008 ##STR01082## 1009 ##STR01083## 1010 ##STR01084## 1011 ##STR01085## 1012 ##STR01086## 1013 ##STR01087## 1014 ##STR01088## 1015 ##STR01089## 1016 ##STR01090## 1017 ##STR01091##
TABLE-US-00108 TABLE 108 1018 ##STR01092## 1019 ##STR01093## 1020 ##STR01094## 1021 ##STR01095## 1022 ##STR01096## 1023 ##STR01097## 1024 ##STR01098## 1025 ##STR01099##
TABLE-US-00109 TABLE 109 1026 ##STR01100## 1027 ##STR01101## 1028 ##STR01102## 1029 ##STR01103## 1030 ##STR01104## 1031 ##STR01105## 1032 ##STR01106## 1033 ##STR01107## 1034 ##STR01108## 1035 ##STR01109##
TABLE-US-00110 TABLE 110 1036 ##STR01110## 1037 ##STR01111## 1038 ##STR01112## 1039 ##STR01113## 1040 ##STR01114## 1041 ##STR01115## 1042 ##STR01116## 1043 ##STR01117## 1044 ##STR01118##
TABLE-US-00111 TABLE 111 1045 ##STR01119## 1046 ##STR01120## 1047 ##STR01121## 1048 ##STR01122## 1049 ##STR01123## 1050 ##STR01124## 1051 ##STR01125## 1052 ##STR01126##
TABLE-US-00112 TABLE 112 1053 ##STR01127## 1054 ##STR01128## 1055 ##STR01129## 1056 ##STR01130## 1057 ##STR01131## 1058 ##STR01132## 1059 ##STR01133## 1060 ##STR01134## 1061 ##STR01135## 1062 ##STR01136##
TABLE-US-00113 TABLE 113 1063 ##STR01137## 1064 ##STR01138## 1065 ##STR01139## 1066 ##STR01140## 1067 ##STR01141## 1068 ##STR01142## 1069 ##STR01143## 1070 ##STR01144## 1071 ##STR01145##
TABLE-US-00114 TABLE 114 1072 ##STR01146## 1073 ##STR01147## 1074 ##STR01148## 1075 ##STR01149## 1076 ##STR01150## 1077 ##STR01151## 1078 ##STR01152## 1079 ##STR01153## 1080 ##STR01154##
TABLE-US-00115 TABLE 115 1081 ##STR01155## 1082 ##STR01156## 1083 ##STR01157## 1084 ##STR01158## 1085 ##STR01159## 1086 ##STR01160## 1087 ##STR01161## 1088 ##STR01162## 1089 ##STR01163## 1090 ##STR01164##
TABLE-US-00116 TABLE 116 1091 ##STR01165## 1092 ##STR01166## 1093 ##STR01167## 1094 ##STR01168## 1095 ##STR01169## 1096 ##STR01170## 1097 ##STR01171## 1098 ##STR01172##
TABLE-US-00117 TABLE 117 1099 ##STR01173## 1100 ##STR01174## 1101 ##STR01175## 1102 ##STR01176## 1103 ##STR01177## 1105 ##STR01178## 1106 ##STR01179## 1107 ##STR01180##
TABLE-US-00118 TABLE 118 1108 ##STR01181## 1109 ##STR01182## 1110 ##STR01183## 1111 ##STR01184## 1112 ##STR01185## 1113 ##STR01186## 1114 ##STR01187## 1115 ##STR01188##
TABLE-US-00119 TABLE 119 1116 ##STR01189## 1117 ##STR01190## 1118 ##STR01191## 1119 ##STR01192## 1120 ##STR01193## 1121 ##STR01194## 1122 ##STR01195## 1123 ##STR01196## 1124 ##STR01197##
TABLE-US-00120 TABLE 120 1125 ##STR01198## 1126 ##STR01199## 1127 ##STR01200## 1128 ##STR01201## 1129 ##STR01202## 1130 ##STR01203## 1131 ##STR01204## 1132 ##STR01205##
TABLE-US-00121 TABLE 121 ##STR01206## 1133 ##STR01207## 1134 ##STR01208## 1135 ##STR01209## 1136 ##STR01210## 1137 ##STR01211## 1138 ##STR01212## 1139 ##STR01213## 1140
TABLE-US-00122 TABLE 122 ##STR01214## 1141 ##STR01215## 1142 ##STR01216## 1143 ##STR01217## 1144 ##STR01218## 1145 ##STR01219## 1146 ##STR01220## 1147 ##STR01221## 1148 ##STR01222## 1149 ##STR01223## 1150 ##STR01224## 1151 ##STR01225## 1152
TABLE-US-00123 TABLE 123 ##STR01226## 1153 ##STR01227## 1154 ##STR01228## 1155 ##STR01229## 1156 ##STR01230## 1157 ##STR01231## 1158 ##STR01232## 1159 ##STR01233## 1160
TABLE-US-00124 TABLE 124 ##STR01234## 1161 ##STR01235## 1162 ##STR01236## 1163 ##STR01237## 1164 ##STR01238## 1165 ##STR01239## 1166 ##STR01240## 1167 ##STR01241## 1168
TABLE-US-00125 TABLE 125 ##STR01242## 1169 ##STR01243## 1170 ##STR01244## 1171 ##STR01245## 1172 ##STR01246## 1173 ##STR01247## 1174 ##STR01248## 1175 ##STR01249## 1176 ##STR01250## 1177
TABLE-US-00126 TABLE 126 ##STR01251## 1178 ##STR01252## 1179 ##STR01253## 1180 ##STR01254## 1181 ##STR01255## 1182 ##STR01256## 1183 ##STR01257## 1184 ##STR01258## 1185 ##STR01259## 1186 ##STR01260## 1187 ##STR01261## 1188
TABLE-US-00127 TABLE 127 ##STR01262## 1189 ##STR01263## 1190 ##STR01264## 1191 ##STR01265## 1192 ##STR01266## 1193 ##STR01267## 1194 ##STR01268## 1195 ##STR01269## 1196 ##STR01270## 1197 ##STR01271## 1198
TABLE-US-00128 TABLE 128 ##STR01272## 1199 ##STR01273## 1200 ##STR01274## 1201 ##STR01275## 1202 ##STR01276## 1203 ##STR01277## 1204 ##STR01278## 1205 ##STR01279## 1206 ##STR01280## 1207
TABLE-US-00129 TABLE 129 ##STR01281## 1208 ##STR01282## 1209 ##STR01283## 1210 ##STR01284## 1211 ##STR01285## 1212 ##STR01286## 1213 ##STR01287## 1214 ##STR01288## 1215 ##STR01289## 1216 ##STR01290## 1217
TABLE-US-00130 TABLE 130 ##STR01291## 1218 ##STR01292## 1219 ##STR01293## 1220 ##STR01294## 1221 ##STR01295## 1222 ##STR01296## 1223 ##STR01297## 1224 ##STR01298## 1225 ##STR01299## 1226
TABLE-US-00131 TABLE 131 ##STR01300## 1227 ##STR01301## 1228 ##STR01302## 1229 ##STR01303## 1230 ##STR01304## 1231 ##STR01305## 1232 ##STR01306## 1233 ##STR01307## 1234 ##STR01308## 1235 ##STR01309## 1236
TABLE-US-00132 TABLE 132 ##STR01310## 1237 ##STR01311## 1238 ##STR01312## 1239 ##STR01313## 1240 ##STR01314## 1241 ##STR01315## 1242 ##STR01316## 1243 ##STR01317## 1244 ##STR01318## 1245 ##STR01319## 1246
TABLE-US-00133 TABLE 133 ##STR01320## 1247 ##STR01321## 1248 ##STR01322## 1249 ##STR01323## 1250 ##STR01324## 1251 ##STR01325## 1252 ##STR01326## 1253 ##STR01327## 1254 ##STR01328## 1255 ##STR01329## 1256 ##STR01330## 1257
TABLE-US-00134 TABLE 134 ##STR01331## 1258 ##STR01332## 1259 ##STR01333## 1260 ##STR01334## 1261 ##STR01335## 1262 ##STR01336## 1263 ##STR01337## 1264 ##STR01338## 1265 ##STR01339## 1266 ##STR01340## 1267 ##STR01341## 1268 ##STR01342## 1269
TABLE-US-00135 TABLE 135 ##STR01343## 1270 ##STR01344## 1271 ##STR01345## 1272 ##STR01346## 1273 ##STR01347## 1274 ##STR01348## 1275 ##STR01349## 1276 ##STR01350## 1277 ##STR01351## 1279 ##STR01352## 1280 ##STR01353## 1281 ##STR01354## 1282
##STR01355##
TABLE-US-00136 TABLE 136 Com- Melting UV pound Point MS (λmax: No. (° C.) 1H-NMR (d) (m/z) nm) 1 213.4 305.3 3 285 (dec.) 4 amorphous 219 5 215, 262 6 147-148 8 214-217 9 oil 220 18 181-183 23 213.4 272.2 305.3 24 116-117 26 182-184 30 267.4 33 253.3 305.3 37 amorphous 219, 275 38 240-244 (dec.) 39 285.2 42 187-188 43 218.1 275.7 48 230 275 57 197-198 58 234-240 62 198-201 69 194-195 71 216.9 268.6 73 266-269 77 d in d20-DMSO:1.67(3H, s), 422.543 2.13-2.06(1H, m), 2.63- 2.55(2H, m), 3.16-3.13 (4H, m), 3.65-3.63(2H, m), 4.76-4.73 (2H, m), 7.15-7.08 (2H, m), 7.30(1H, t, J = 8.0 Hz), 7.35(1H, s), 7.42(1H, t, J = 8.0Hz), 7.60(1H, d, J = 8.0 Hz), 7.69(1H, d, J = 8.0 Hz), 7.73(1H, brs), 7.86(1H, d, J = 8.0 Hz), 10.52(1H, s) 78 1H-NMR (CDCl3) 365 d: 1.76 (3H, s), [M + 1] 2.02 (1H, s), 2.58 (1H, d, J = 14.1 Hz), 2.78 (2H, d, J = 6.9 Hz), 3.80 (3H, d, J = 13.1 Hz), 4.54 (2H, s), 6.45 (1H, s), 6.55- 6.57 (2H, m), 6.66 (1H, d, J =8.7 Hz), 7:10 (1H, t, J = 7.0 Hz), 7.22 (2H, td, J = 71, 1.4 Hz), 7.34 (1H, d, J= 9.1 Hz), 7.56 (1H, d, J =7.7 Hz). 80 220.4 280.4
TABLE-US-00137 TABLE 137 Com- Melting UV pound Point MS (λmax: No. (° C.) 1H-NMR (d) (m/z) nm) 85 147-148 1.54(3H, s), 1.75-1.86(1H, m), 2.08-2.18(1H, m), 2.33(3H, s), 2.63-2.74(1H, m), 2.81-2.90(1H, m), 4.36(2H, br), 7.13(2H, d, J = 8.6 Hz), 7.20(2H, d, J = 8.6 Hz) (solvent: CDCl3) 86 141-142 91 372 201 [M + 1] 206 296 216 96 309 97 d in d13-DMS0:1.64(3H, s), 2.03-1.97(1H, m), 2.63 2.57(2H, m), 3.28-3.25(1H, m), 7.22(1H, q, J = 12.4, 9.0 Hz), 7.82-7.77(2H, m), 8.60(1H, s), 8.79(1H, s), 10.37(1H, s) 99 221-224 101 264-265 104 amorphous 229, 280 113 1.58 (s, 3H), 1.88 (ddd, J = 14.1, 10.9, 3.7 Hz, 1H), 2.24 (ddd, J = 14.1, 5.9, 3.5 Hz, 1H), 2.73 (ddd, J = 12.3,10.9, 3.5 Hz, 1H), 2.88 (ddd, J = 12.3, 5.9, 3.7 Hz, 1H), 3.83 (d, J = 15.4 Hz, 1H), 3.87 (d, J = 15.4 Hz, 1H), 7.02-7.04 (m,1H), 7.25-7.31 (m, 2H), 7.36 (d, J = 2.0 Hz, 1H), 7.45-7.50 (m, 2H), 8.52 (d, J = 5.2Hz, 1H), 9.43 (s, 1H) (solvent: CDCl3) 114 214.5 306.5 115 d in d6-DMSO:1.47(3H, s), 1.80-1.74(1H, m, 2.22- 2.18(1H, m), 2.60-2.55(1H, m), 2.96-2.93(1H, m), 6.14(1H, s), 6.93(1H, s), 7.09-7.04(2H, m), 7.63-7.61(1H, m), 7.68-7.66(1H, m), 9.85(1H, s), 11.63(1H, brs) 120 amorphous 213 121 166-167 125 >300 126 amorphous 229, 271 127 280-285 128 159-163 129 219-222 130 128-131 1.56 (3H, s), 1.83-1.93 (1H, m), 344 2.16 (1H, dq, J = [M + 1] 13.85, 3.41 Hz), 2.29 (3H, s), 2.72-2.77 (1H, m), 2.90-2.94 (1H, m), 4.13 (3H, s), 6.42 (1H, s), 7.10- 7.14 (1H, m), 7.32 (1H, d, J = 7.91 Hz), 7.37-7.38 (1H, m), 7.60-7.63 (1H, m). (solvent: CDCl3) 132 147-150 134 228.5
TABLE-US-00138 TABLE 138 Com- Melting UV pound Point MS (λmax: No. (° C.) 1H-NMR (d) (m/z) nm) 139 287-290 1.77 (s, 3H), g.10 (ddd, J = 14.0, 10.8, 3.6 Hz, 1H), 2.64-2.70 (411, m), 2.76 (td, J = 12.8, 3.6 Hz, 1H), 2.90 (dt, J = 12.8, 3.6 Hz, 1H), 7.05 (ddd, J = 8.0, 2.0, 0.8 Hz, 1H), 7.41 (t, J = 8.0 Hz, 1H), 7.69-7.72 (m, 2H), 8.32 (dd, J = 8.0, 0.8 Hz, 1H), 8.40 (dd, J = 8.0, 2.0 Hz, 1H), 9.14 (dd, J = 2.0, 0.8 Hz, 1H) (solvent: CDCl3 + CD3OD) 141 d in d17-DMSO:1.41(3H, s), 1.75-1.70(1H, m), 2.03- 1.99(1H, m), 2.62-2.56(1H, m), 2.94-2.89(1H, m), 3.89(3H, s), 6.88(1H, d, J = 8.8Hz), 7.05(1H, d, J = 7.6 Hz), 7.24(1H, t, J = 8.0 Hz), 7.66-7.63(3H, m), 8.45-8.44(1H, m), 9.90(1H, s) 148 362[M+1] 200 286 208 212 218 262 149 143-145 157 d in d6-DMSO: 1.20(6H, d, J = 6.6 Hz), 1.41(3H, s), 1.65-1.77(1H, m), 1.96-2.07 (1H, m), 2.55-2.63(1H, m), 2.85-2.95(1H, m), 4.04-4.16 (1H, m), 5.79(2H, bs), 7.07(1H, d, J = 8.1 Hz), 7.25(1H, t, J = 8.1 Hz), 7.72- 7.78(3H, m), 7.93(1H, s), 8.64 (1H, s), 9.96(1H, s). 159 amorphous 285 161 247-251 163 amorphous 164 91-96 1.68(s, 3H), 2.07-2.15(m, 1H), 3.13-3.20(m; 1H), 7.12(d, J = 7.6 Hz, 1H), 7.46 (t, J = 7.6 Hz, 1H), 7.90- 7.94(m, 2H), 8.83(br s, 1H), 8.96(br s, 1H), 9.31(br s, 1H), 10.36(s, 1H), 10.86(s, 1H) 165 246-248 166 amorphous 220, 275 176 amorphous 217, 278 178 224-225 181 261.5 189 259 193 266-268 196 212 202 117-118 0.85(3H, t, J = 7.3 Hz), 1.02-1.19(1H, m), 1.34- 1.54(1H, m), 1.72-1.89 (3H, m), 2.04-2.15(1H, m), 2.61-2.82(2H, m), 3.80(3H, s), 4.32(2H, br), 6.85(2H, d, J = 8.9 Hz), 7.18 (2H, d, J = 3.9Hk) (solvent: CDCl3)
TABLE-US-00139 TABLE 139 Com- Melting UV pound Point MS (λmax: No. (° C.) 1H-NMR (d) (m/z) nm) 204 205-208 1.64 (d, J = 1.2 Hz, 3H), 1.95 (ddd, J = 14.0, 10.8, 3.6 Hz, 1H), 2.45 (ddd, J = 14.0, 6.4, 3.6 Hz, 1H), 2.75 (ddd, J = 12.4, 10.8, 3.6 Hz, 1H), 2.99 (ddd, J = 12.4, 6.4, 3.6 Hz, 1H), 7.09 (dd, J = 11.6, 8.8 Hz, 1H), 7.47 (dd, J = 7.2, 2.8 Hz, 1H), 8.03 (ddd, J = 8.8, 4.4, 2.8 Hz, 1H), 8.89 (s, 2 H), 9.75 (s, 1H) (solvent: CDCl3) 213 oil 216, 272 214 212.2 292.3 356.5 216 242.7 220 191-193 363 [M + 3] 361 [M + 1] 287 285 224 oil 1.58(3H, s), 1.87(1H, ddd, 222 J = 13.9, 10.5, 3.7), 2.13(1H, ddd, J = 13.9, 6.3, 3.7), 2.25(3H, s), 2.68(1H, ddd, J = 12.1, 10.5, 6.2), 2.89(1H, ddd, J = 12.1, 6.3, 3.7), 5.23(2H, s), 7.28-7.48(4H, m), 7.60(1 H, s), 7.75(1H, d, J = 8.0), 8.56(1H, dd, J = 5.0, 1.4),.8.70(1H, d, J = 1.4) (solvent: CDCl3) 227 213 232 1 H-NMR (CDCl3) d: 1.59 378 (3H, s), 1.83-1.90 (1H, m), [M + 1] 2.35-2.47 (4 H, m), 2.60-2.67 (1H, m), 2.87-2.92 (1H, m), 4.70 (2H, br s), 6.87-6.98 (2H, m), 7.16 (1H, d, J = 6.6 Hz), 7.27 (2H, d, J = 7.8 Hz), 7.61 (2H, d, J = 8.1 Hz). 233 oil 224, 272 235 196-200 238 1H-NMR (CDCl3) d: 1.68 (3H, s), 362 1.97-2.00 (1H, m), [M + 1] 2.53 (1H, dt, J = 14.4, 3.7 Hz), 2.63-2.79 (2H, m), 4.52 (2H, s), 6.56-6.66 (3H, m), 7.17 (1H, t, J = 8.0 Hz), 7.43-7.52 (3H, m), 7.81 (4H, dd, J = 11.6, 5.7 Hz). 241 187-190 1H-NMR (DMSO-d6) d: 1.49 (3H, s), 1.78-1.86 (1H, m), 2.13-2.21 (1H, m), 2.59- 2.67 (1H, m), 2.96-3.02 (1H, m), 7.11 (1H, t, J = 10.7 Hz), 7.29 (1H, t, J = 7.8 Hz), 7.45 (1H, t, J =7.5 Hz), 7.66 (1H, d, J = 8.8 Hz), 7.74-7.78 (1H, m), 7.80-7.83 (1H, m), 8.21 (1H, d, J = 8.6 Hz), 10.25 (1H, s). 243 182-184 1.46(s, 3H), 1.75-1.83 (m, 1H), 2.08-2.16(m, 1H), 2.55-2.63(m, 1H), 2.92-2.98 (m, 1H), 4.02(s, 3H), 7.11(d, J = 8.0 Hz, 1H), 7.31(t, J = 8.0 Hz, 1H), 7.77(d, J = 8.0 Hz, 1H), 7.82(br s, 1H), 8.41(d, J = 1.2 Hz, 1H), 8.90(d, J = 1.2 Hz, 1H), 10.38(s, 1H) (solvent: CDCl3)
TABLE-US-00140 TABLE 140 Com- Melting UV pound Point MS (λmax: No. (° C.) 1H-NMR (d) (m/z) nm) 244 222-224 251 351 200 [M + 1] 311 204 275 215 285 255 238-239 256 oil 215, 257 259 amorphous 1.58(3H, s), 2.01(1H, ddd, 229 J = 15.2, 12.2, 3.4), 2.46- 298 2.56(2H, m), 3.07(1H, ddd, J = 13.3, 5.7, 3.5), 4.24(2H, s), 6.53(1H, d, J = 7.6), 6.59-6.61(2H, m), 7.09-7.12(1H, m), 7.11(2H, d, J = 7.6), 7.24(2H, d, J = 7.6), 8.82(2H, br) (solvent: DMSO-d6) 263 363 200 [M + 1] 287 284 267 114-115 268 214.5 298.2 271 oil 229, 276 275 (CDCl3) 1.66(3H, d, J = 1.2 Hz), 1.98(1H, ddd, J = 14.0, 10.4, 3.7 Hz), 2.47(1H, ddd, J = 14.0, 6.7, 3.5 Hz), 2.79(1H, ddd, J = 12.0, 10.4, 3.5 Hz), 3.02(1H, ddd, J = 12,0, 6.7, 3.7 Hz), 4.45 (2H, br), 6.16(2H, br), 7.04 7.11(2H, m), 7.38(1H, dd, J = 7.2, 2.9 Hz), 7.88(1H, d, J = 2.0 Hz), 7.96(1H, ddd, J = 8.9, 4.2, 2.9 Hz), 9.88(1H, s) 277 216 228 281 279 214.5 292.3 281 amorphous 1.55(3H, s), 233 1.83(1H, ddd, J = 13.9, 301 10.6, 3.9), 2.10(1H, ddd, J = 13.9, 6.5, 3.6), 2.67 (1H, ddd, J = 12.2, 10.6, 3.6), 2.87(1H, ddd, J = 12.2, 6.5, 3.9), 4.49(2H, d, J = 5.6), 4.85(1H, br), 6.38(1H, dt, J = 8.5, 0.9), 6.59(1H, ddd, J = 7.2, 5.2, 0.9), 7.21- 7.24(2H, m), 7.28-7.32 (2H, m), 7.40(1H, ddd, J = 8.5, 7.2, 1.8), 8.11(1H, ddd, J =5.2, 1.8, 0.8) (solvent: CDCl3) 282 146-147 284 181.5 293 1.57 (s, 3H), 1.78-1.89 (m, 1H), 2.10-2.19 (m, 1H), . 2.69 (ddd, J = 11.9, 10.8, 3.5 Hz, 1H), 2.83-2.91 (m, 1H), 7.15-7.35 (m, 5H) (solvent: CDCl3) 299 293.5
TABLE-US-00141 TABLE 141 Com- Melting UV pound Point MS (λmax: No. (° C.) 1H-NMR (d) (m/z) nm) 301 (CDCl3) 1.53(3H, s), 1.80(1H, ddd, J = 14.0, 10.4, 3.6 Hz), 2.12(1H, ddd, J = 14.0, 6.0, 3.6 Hz), 2.75(1H, ddd, J = 12.0, 10.4, 3.6 Hz), 2.85(1H, ddd, J = 12,0, 6.0, 3.6 Hz), 3.64(2H, s), 4.32(2H, br), 6.55(1H, ddd, J = 8.0, 2.0, 0.8 Hz), 6.66 (1H, t, J = 2.0 Hz), 6.70(1H, ddd, J = 8.0, 2.0, 0.8 Hz), 7.11 (1H, t, J = 8.0 Hz) 302 122-126 1.41(s, 3H), 1.67-1.76 (m, 1H), 1.98-2.06(m, 1H), 2.55-2.63(m, 1H), 2.86-2.94 (m, 1H), 3.19(s, 6H), 5.75(s, 2H), 7.08(d, J = 8.0 Hz, 1H), 7.26(t, J = 8.0 Hz, 1H), 7.73(d, J = 8:0 Hz, 1H), 7.76(br s, 1H), 8.16(s, 1H), 8.73(s, 1H), 10.00 (s, 1H)(solvent: CDCl3) 306 231, 258, 289 307 1.83 (ddd, J = 13.9, 10.3, 3.6 Hz, 1H), 2.13 (ddd, J = 13.6, 6.2, 3.5 Hz, 1H), 2.53 (s, 3H), 2.66-2.75 (m, 1H), 2.90 (ddd, J = 12.2, 6.3, 3.8 Hz, 1H), 7.09 (d, J = 7.8 Hz, 1H), 7.32 (t, J = 8.0 Hz, 1H), 7.37 (s, 1H), 7.63 (d, J = 7.8 Hz, 1H), 8.79 (s, 1H) (solvent: CDCl3) 308 167-168 309 241-244 319 308.9 329 238-239 330 213.4 263.9 332 212.2 333 154-158 339 217-218 341 amorphous 216 249 342 184-187 344 (DMSO) 1.49(3H, s), 1.73-1.85 (1H, m), 2.15- 2.28(1H, m), 2.54-2.66(1H, m), 2.92-3.04(1H, m), 5.86(2H, s), 7.03-7.25 (3H, m), 7.40-7.48(2H, m), 7.64-7.78(3H, m), 10.31 (1H, s), 11.74(1H, s) . 353 279.3 364.5 354 102-103 356 amorphous 1.73 (s, 3H), 2.09-2.17 267 (m, 1H), 2.40(s, 3H), 2.65- 2.73 (m, 2H), 3.15-3.23 (m, 1H), 3.81(s, 3H), 7.07 (d, J = 7.2 Hz, 2H), 7.29 (br s, 1H), 7.36 (d, J = 8.0 Hz, 2H), 7.61 (d, J = 8.0 Hz, 2H), 7.78 (br s, 1H), 7.90 (d, J = 7.2 Hz, 2H), 8.00 (br s, 1H), 10.32 (s, 11-1)(solvent: DMSO-d6) 357 amorphous 224, 298
TABLE-US-00142 TABLE 142 Com- Melting UV pound Point MS (λmax: No. (° C.) 1H-NMR (d) (m/z) nm) 358 1.57 (3 H, s), 1.80-1.91 360 (1H, m), 2.15-2.18 (1H, m) [M + 1] 2.70-2.94 (2H, m), 3.94 (3H, s), 4.67 (2H, s), 6.75 (1H, s), 7.05-7.08 (1H, m), 7.31 (1H, t, J = 7.91 Hz), 7.53 (1H, t, J = 1.98 Hz), 7.64-7.67 (1H, m), 8.64 (1H, s).(solvent: CDCl3) 359 212-214 1.46(s, 3H), 1.73-1.83(m, 1H), 2.13-2.20(m, 1H), 2.54-2.61(m, 1H), 2.62(s, 3H), 2.93-3.00(m, 1H), 5.84(br s, 2H), 7.12(dd, J = 12.0, 8.8 Hz, 1H), 7.73- 7.78(m, 1H), 7.81(dd, J = 72, 2.4 Hz, 1H), 8.68(s 1H), 9.13(s, 1H), 10.59 (s, 1H)(solvent: CDCl3) 360 amorphous 222 361 280.4 364 oil 344 227, 271 [M + 1] 367 (CDCl3) 1.78(3H, s), 2.07(1 H, ddd, J = 14.0, 12,4, 3.6 Hz), 2.61(1 H, br d, J = 14.0 Hz), 2.84(1 H, td, J = 12.4, 3.2 Hz), 2.94(1H, td, J = 12.4, 3.6 Hz), 4.08(3H, s), 7.07(1H, ddd, J = 8.0, 2.0, 0.8 Hz), 7.40(1 H, t, J = 8.0 Hz), 7.63(1H, ddd, J = 8.0, 2.0, 0.8 Hz), 7.74(1H, t, J = 2.0 Hz), 8.18(1H, d, J = 1.2 Hz), 9.02(1H, d, J = 1.2 Hz), 9.56(1H, s) 375 217 380 181-182 0.86 (t, J = 7.2 Hz, 3H), 1.82-1.98 (m, 3H), 2.24 (br, 1H), 2.74 (td, J = 12.0, 3.6 Hz, 1H), 2.84 (dt, J =12.0, 4.0 Hz, 1H), 7.08 (ddd, J = 8.0, 2.0, 0.8 Hz, 1H), 7.37 (t, J = 8.0 Hz, 1H), 7.58 (t, J = 2.0 Hz, 2H), 7.76 (ddd, J = 8.0, 2.0, 0.8 Hz, 1H), 7.88 (dd, J = 8.4, 2.4 Hz, 1H), 8.25 (dd, J = 8.4, 0.8 Hz, 1H), 8.57 (dd, J = 2.4,0.8 Hz, 1H), 9.84 (s, 1H) (solvent: CDCl3) 383 oil 225, 269, 288 389 amorphous 292 393 213.4 316.0 395 amorphous 217, 269 396 211-213 1.64 (s, 3H), 1.96 (ddd, J = 14.0, 10.4, 4.0 Hz, 1H), 2.44 (ddd, J = 14.0, 6.8, 3.6 Hz, 1H), 2.75 (ddd, J = 12.4, 10.4, 3.6 Hz, 1H), , 2.99 (ddd, J = 12.4, 6.8, 4.0 Hz, 1H), 4.50 (2H, br), 7.08 (dd, J = 11.6, 8.8 Hz, 1H), 7.45 (dd, J = 6.8, 2.8 Hz, 1H), 8.01 (ddd, J = 8.8, 4.4, 2.8 Hz, 1H), 8.16 (ddd, J = 8.0, 2.0, 0.8 Hz, 1H), 8.43 (d, J = 8.0 Hz, 1H), 8.89 (dd, J = 2.0.0.8 Hz, 1H), 9.91 (s, 1H) (solvent: CDCl3) 401 106-107
TABLE-US-00143 TABLE 143 Com- Melting UV pound Point MS (λmax: No. (° C.) 1H-NMR (d) (m/z) nm) 405 192-194 1.41(s, 3H), 1.68-1.77(m, 1H), 1.96-2.05(m, 1H), 2.55-2.63(m, 1H), 2.88-2.95 (m, 1H), 4.15(s, 3H), 5.74(s, 2H), 7.13(d, J = 8.0 Hz, 1H), 7.29(t, J = 8.0 Hz, 1H), 7.44(d, J = 8.8 Hz, 1H), 7.75(d, J = 8.0 Hz, 1H), 7.86(br s, 1H), 8.20(d, J = 8.8 Hz, 1H), 10.73(s, 1H) (solvent: CDCl3) 406 276.9 408 221-224 1.74 (3H, s) 2.28 (2H, m) 2.67 (2H, m) 2.91 (3H, s) 3.82 (3H, s), 6.90 (2H, d, J = 9.0), 7.19 (2H, d, J = 9.0) (solvent: CDCl3) 409 oil 215 410 178-182 1.37(d, J = 6.0 Hz, 6H), 1.42 (5, 3H), 1.70-1.78(m, 1H), 2.00-2.08(m, 1H), 2.53-2.61(m, 1H), 2.88- 2.95(m, 1H), 5.36(quintet, J = 6.0 Hz, 1H), 7.11(d, J = 8.0 Hz 1H), 7.29(t, J = 8.0 Hz, 1H), 7.75(d, J = 8.0 Hz, 1H), 7.80(br s, 1H), 8.32 (d, J = 1.2 Hz, 1H), 8.87(d, J = 1.2 Hz, 1H), 10.32(s, 1H) (solvent: CDCl3) 411 218, 264 413 251-254 415 amorphous 226, 290 417 137-139 422 (CDCl3) 1.45(3H, s), 1.70-1.84(1H, m), 1.96- 2.04(1H, m), 2.88-2.96 (1H, m), 3.04-3.14(1H, m), 6.86(1H, d, J = 15.9Hz), 6.42(1H, d, J = 15.9Hz), 7.22- 7.41(5H, m) 426 211.0 312.4 427 216 429 oil 211 259 430 (DMSO) 1.07(3H, s), 1.53-1.66(4H, m), 2.50- 2.70(2H, m), 2.92-3.10(2H, m), 5.48(1H, s), 7.11- 7.21(3H, m), 7.23-7.29(2H, m) 432 oil 216, 272 436 254-256 441 161-165 443 1H-NMR (CDCl3) d: 1.55 (4H, s), 362 1.74-1.80 (1H, m), [M + 1] 2.13-2.17 (1H, m), 2.68-2.73 (2H, m), 4.33 (1H, br s), 4.48 (2H, d, J = 4.0 Hz), 4.76 (2H, t, J = 20.1 Hz), 6.52 (1H, dd, J = 7.9, 1.8 Hz), 6.63-6.65 (2H, m), 7.13 (1H, t, J = 1.8 Hz), 7.45-7.51 (2H, m), 7.79-7.82 (4H, m).
TABLE-US-00144 TABLE 144 Compound Melting Point MS UV No. (° C.) 1H-NMR (d) (m/z) (λmax: nm) 444 214-215 1.41(s, 3H), 1.66-1.76(m, 1H), 1.97-2.05(m, 1H), 2.53-2.62(m, 1H), 2.62(s, 3H), 2.86-2.93(m, 1H), 5.79(br s, 2H), 7.12(d, J = 8.0 Hz, 1H), 7.28(t, J = 8.0 Hz, 1H), 7.74(d, J = 8.0 Hz, 1H), 7.81(br s, 1H), 8.68(s, 1H), 9.14(s, 1H), 10.52(s, 1H) (solvent: CDCl3) 445 92-93 446 oil 1.57(3H, s), 1.86(1H, ddd, J = 13.9, 10.4, 3.7), 219 2.13(1H, ddd, J = 13.9, 6.5, 3.6), 2.25(3H, s), 252 2.35(3H, s), 2.70(1H, ddd, J = 12.2, 10.4, 3.6), 2.89(1H, ddd, J = 12.2, 6.5, 3.7), 4.35(2H, br), 5.19(2H, s), 7.17(2H, d, J = 8.0), 7.31-7.34(4H, m), 7.50(1H, ddd, J = 5.8, 3.0, 1.8), 7.55-7.60(1H, m) (solvent: CDCl3) 448 d in d6-DMSO: 1.41(3H, s), 1.67-1.75(1H, m), 1.98- 2.05(1H, m), 2.52-2.61(1H, m), 2.86-2.94(1H, m), 5.79(2H, bs), 7.14(1H, d, J = 7.8 Hz), 7.30(1H, t, J = 7.8 Hz), 7.73(1H, bd, J = 7.8 Hz), 7.81(1H, t, J = 1.8 Hz), 8.94(1H, m), 9.11(1H, m), 10.63(1H, bs). 452 132-134 456 147-149 457 153-155 465 194.6 466 211 470 281 (dec.) 482 1.60 (s, 3H), 1.91 (ddd, J = 14.0, 10.8, 4.0 Hz, 1H), 2.23 (ddd, J = 14.0, 6.4, 3.6 Hz, 1H), 2.77 (ddd, J = 12.0, 10.8, 3.6 Hz, 1H), 2.93 (ddd, J = 12.0, 6.4, 4.0 Hz, 1H), 7.16 (ddd, J = 8.0, 2.0, 0.8 Hz, 1H), 7.37 (t, J = 8.0 Hz, 1H), 7.61 (t, J = 2.0 Hz, 1H), 7.75 (ddd, J = 8.0, 2.0, 0.8 Hz, 1H), 8.14 (d, J = 1.6 Hz, 1H), 8.80(d, J = 1.6 Hz, 1H), 9.79 (s, 1H) (solvent: CDCl3) 483 224-227 211, 289 490 1.64 (3H, s) 2.03-2.12 (1H, m) 2.49-2.62 (m) 3.12- 3.16 (1H m) 7.22 (1H, dd, J = 4.2 Hz) 7.27 (1H, bs) 7.75 (1H bs) 7.87 (1H, dd, J = 4.2 Hz) 8.04 (1H, s) 8.12 (1H, dd, J = 4.2 Hz) 10.64 (1H, s) 10.72 (1H, s) (solvent: DMSO-d6) 1.58 (s, 3H), 1.85-1.96 (m, 1H), 2.15-2.24 (m, 1H), 2.50 (s, 3H), 2.67 (s, 3H), 2.71-2.81 (m, 1H), 2.90- 491 2.98 (m, 1H), 7.13 (d, J = 6.2 Hz, 1H), 7.35 (t, J = 8.0 Hz, 1H), 7.40 (s, 1H), 7.55 (d, J = 7.6 Hz, 1H) (solvent: CDCl3) 493 216
TABLE-US-00145 TABLE 145 Compound Melting Point MS UV No. (° C.) 1H-NMR (d) (m/z) (λmax: nm) 494 d in d6-DMSO: 1.37(3H, s), 1.62-1.70(1H, m), 2.0- 366[M + 1] 2.12(1H, m), 2.40-2.50(1H, m), 2.79-2.83(1H, m), 3.82(3H, s), 4.52(2H, d, J = 5.4 Hz), 6.19(1H, m), 6.54(1H, d, J = 7.8 Hz), 6.62(1H, d, J = 8.1 Hz), 6.75 (1H, s), 7.01(1H, t, J = 8.1 Hz), 7.14-7.25(2H, m), 7.51(1H, d, J = 8.1 Hz), 7.60(1H, d, J = 7.5 Hz). 496 152-154 497 d in d6-DMSO:1.48(3H, s), 1.83-1.77(1H, m), 2.61- 2.56(1H, m), 2.99-2.95(1H, m), 3.86(3H, s), 6.07(1H, s), 6.95(1H, s), 7.03-7.02(1H, m), 7.09-7.06(1H, m), 7.58-7.57(1H, m), 7.64-7.62(1H, m), 9.83(1H, s) 498 122-125 500 181-184 501 155-156 502 137-138 504 209-219 511 211-214 1.58 (s, 3H), 1.90 (ddd, J = 14.0, 10.0, 3.6 Hz, 1H), 2.15 (ddd, J = 14.0, 6.8, 3.6 Hz, 1H), 2.77 (ddd, J = 12.4, 10.0, 3.6 Hz, 1H), 2.94 (ddd, J = 12.4, 6.8; 3.6 Hz, 1H), 4.34 (2H, br), 7.17 (ddd, J = 8.0, 2.0, 0.8 Hz, 1H), 7.38 (t, J = 8.0 Hz, 1H), 7.50 (d, J = 2.0 Hz, 1H), 7.56 (td, J = 2.0 Hz, 1H), 7.70 (ddd, J = 8.0, 2.0, 0.8 Hz, 1H), 8.08 (d, J = 1.6 Hz), 9.70 (s, 1H) (solvent: CDCl3) 515 204-206 1.61 (s, 3H), 1.90 (ddd, J = 14.0, 10.8, 3.6 Hz, 1H), 2.22 (ddd, J = 14.0, 6.0, 3.6 Hz, 1H), 2.77 (ddd, J = 12.4, 10.8, 3.6 Hz, 1H), 2.93 (ddd, J = 12.4, 6.0, 3.6 Hz, 1H), 7.15 (ddd, J = 8.0, 2.0, 0.8 Hz, 1H), 7.39 (t, J = 8.0 Hz, 1H), 7.65 (t, J = 2.0 Hz, 1H), 7.80 (ddd, J = 8.0, 2.0, 0.8 Hz, 1H), 8.89 (s, 2H), 9.77 (s, 1H) (solvent: CDCl3) 516 292.3 525 105-106 528 173-174 1.60 (s, 3H), 1.89 (ddd, J = 14.0, 10.8, 3.6 Hz, 1H), 2.22 (ddd, J = 14.0, 6.4, 3.2 Hz, 1H), 2.44 (s, 3H), 2.77 (ddd, J = 12.4, 10.8, 3.2 Hz, 1H), 2.91 (ddd, J = 12.4, 6.4, 3.6 Hz, 1H), 4.50 (br, 2H), 7.11 (ddd, J = 8.0, 2.0, 0.8 Hz, 1H), 7.35 (t, J = 8.0 Hz, 1H), 7.67- 7.71 (m, 2H), 7.74 (ddd, J = 8.0, 2.0, 0.8 Hz, 1H), 8.18 (d, J = 8.4 Hz, 1H), 8.44(d, J = 1.6 Hz, 1H), 9.98 (s, 1H) (solvent: CDCl3) 532 305.3 533 180-181 534 201-204 549 100-101 551 139-141 554 216
TABLE-US-00146 TABLE 146 Compound Melting Point MS UV No. (° C.) 1H-NMR (d) (m/z) (λmax: nm) 556 (CDCl3) 1.67(3H, d, J = 1.2 Hz), 1.98(1H, ddd, J = 14.0, 10.4, 3.7 Hz), 2.47(1H, ddd, J = 14.0, 6.7, 3.5 Hz), 2.79(1H, ddd, J = 12.0, 10.4, 3.5 Hz), 3.02(1H, ddd, J = 12,0, 6.7, 3.7 Hz), 4.11(3H, s), 4.45(2H, br), 7.10(1H, dd, J = 11.7, 8.8 Hz), 7.41(1H, dd, J = 6.9, 2.8 Hz), 8.04(1H, ddd, J = 8.8, 4.0, 2.8 Hz), 8.20(1H, d, J = 1.4 Hz); 9.06(1H, d, J = 1.4 Hz), 9.51(1H, s) 558 358[M + 1] 200 282 559 224 560 d in d10-DMSO:1.72(3H, s), 2.12-2.05(1H, m), 2.71- 2.61(2H, m), 3.22-3.19(1H, m), 6.52(1H, s), 7.26(1H, q, J = 11.6, 9.2 Hz), 7.55(1H, s), 7.66-7.62(2H, m), 7.79-7.77(1H, m), 7.90-7.88(1H, m), 8.07(1H, s), 10.42(1H, s), 11.55(1H, s) 561 235-240 567 oil 212 570 186-187 573 112-114 577 d in d19-DMSO:2.14-2.07(1H, m), 2.88-2.70(3H, m), 3.07, 3.26(2H, abq, J = 12.0 Hz), 3.73(3H, s), 5.40(2H, s), 6.51(1H, s), 6.85(1H, d, J = 12.0 Hz), 7.34(1H, d, J = 8.0 Hz) 584 152-153 586 d in d7-DMSO:1.71(3H, s), 2.10-2.04(1H, m), 2.69- 2.59(2H, m), 3.20-3.17(1H, m), 4.00(3H, s), 7.13(1H, d, J = 7.4 Hz), 7.33-7.23(3H, m), 7.55(1H, d, J = 8.4 Hz), 7.72-7.68(1H, m), 7.92-7.90(1H, m), 10.60(1H, s) 588 155-156 593 oil 226 595 oil 1.56(3H, s), 1.86(1H, ddd, J = 13.9, 10.1, 3.7), 220 2.11(1H, ddd, J = 13.9, 6.6, 3.6), 2.32(3H, s), 2.70(1H, ddd, J = 12.3, 10.1, 3.6), 2.90(1H, ddd, J = 12.3, 6.6, 3.7), 5.25(2H, s), 7.29-7.35(4H, m), 7.47(1H, dt, J = 6.8, 2.0), 7.56-7.58(1H, m), 8.59(2H, d, J = 6.0) (solvent: CDCl3) 596 215 597 192-194 600 178-180 601 181-192 1.59 (3H, s), 1.85-1.95 (1H, m), 2.15-2.22 (1H, m), 375[M + 1] 2.72-2.78 (1H, m), 2.88-2.96 (1H, m), 4.31 (3H, s), 7.13 (1H, d, J = 7.25 Hz), 7.33 (1H, t, J = 7.91 Hz), 7.59 (1H, s), 7.68 (1H, d, J = 7.91 Hz), 7.75 (1H, s). (solvent: CDCl3) 602 272-285 (dec.)
TABLE-US-00147 TABLE 147 Compound Melting Point MS UV No. (° C.) 1H-NMR (d) (m/z) (λmax: nm) 605 230-233 1.63 (s, 3H), 1.94 (ddd, J = 14.0, 10.4, 3.6 Hz, 1H), 2.44 (ddd, J = 14.0, 6.4, 3.6 Hz, 1H), 2.75 (ddd, J = 12.4, 10.4,3.6 Hz, 1H),, 2.98 (ddd, J = 12.4, 6.4, 3.6 Hz, 1H), 4.50 (2H, br), 7.06 (dd, J = 11.6, 8.8 Hz, 1H), 7.40 (dd, J = 7.2, 2.8 Hz, 1H), 7.59 (ddd, J = 8.8, 8.0, 2.8 Hz, 1H), 7.99 (ddd, J = 8.8, 4.4, 2.8 Hz, 1H), 8.33 (dd, J = 8.8, 4.4 Hz, 1H), 8.45 (d, J = 2.8 Hz, 1H), 9.78 (s, 1H) (solvent: CDCl3) 608 213.4 304.1 611 200-202 613 238 618 1.74(s, 3H), 1.97-2.07(m, 1H), 2.45-2.55(m, 1H), 2.77-2.85(m, 1H), 2.84(s, 3H), 2.90-2.96(m, 1H), 7.11(d, J = 8.0 Hz, 1H), 7.42(t, J = 8.0 Hz, 1H), 7.57(d, J = 8.8 Hz, 1H), 7.70(d, J = 8.0 Hz, 1H), 7.74(br s, 1H), 8.29(d, J = 8.8 Hz, 1H), 10.12(s, 1H) (solvent: CDCl3) 620 212, 253 625 107-109 629 d in d14-DMSO:1.66(3H, s), 2.11-2.05(1H, m), 2.37(3H, s), 2.63-2.53(2H, m), 3.14-3.11(1H, m), 7.08-7.04(2H, t, J = 7.0 Hz), 7.43-7.35(4H, m), 7.83- 7.80(2H, m), 10.39(1H, s), 11.69(1H, s) 630 1.28 (3H, t, J = 7.7 Hz), 1.96 (1H, ddd, J = 3.8, 9.9, 301[M + 1] 13.7 Hz), 2.19 (1H, ddd, J = 3.5, 7.0, 13.7 Hz), 2.74 (1H, ddd, J = 3.6, 9.9, 12.2 Hz), 2.93 (1H, ddd, J = 3.8, 7.0, 12.1 Hz), 4.05-4.49 (4H, m), 7.40-7.50 (3H, m), 7.77-7.86 (1H, m) (solvent: CDCl3) 634 (CDCl3) 1.67(3H, d, J = 1.2 Hz), 1.98(1H, ddd, J = 14.0, 10.4, 3.7 Hz), 2.47(1H, ddd, J = 14.0, 6.7, 3.5 Hz), 2.79(1H, ddd, J = 12.0, 10.4, 3.5 Hz), 3.02(1H, ddd, J = 12,0, 6.7, 3.7 Hz), 4.11(3H, s), 4.45(2H, br), 7.10(1H, dd, J = 11.7, 8.8 Hz), 7.41(1H, dd, J = 6.9, 2.8 Hz), 8.04(1H, ddd, J = 8.8, 4.0, 2.8 Hz), 8.20(1H, d, J = 1.4 Hz), 9.06(1H, d, J = 1.4 Hz), 9.51(1H, s) 636 118-119 637 229, 275 643 155-157 1.60 (s, 3H), 1.90 (ddd, J = 14.0, 10.4, 3.6 Hz, 1H), 2.20 (ddd, J = 14.0, 6.8, 3.6 Hz; 1H), 2.77 (ddd, J = 12.0, 10.4, 3.6 Hz, 1H),, 2.93 (ddd, J = 12.0, 6.8, 3.6 Hz, 1H), 4.59 (brs, 1H), 7.16 (ddd, J = 8.0, 2.0, 0.8 Hz, 1H), 7.37 (t, J = 8.0 Hz, 1H), 7.67 (t, J = 2.0 Hz, 1H), 7.71 (ddd, J = 8.0, 2.0, 0.8 Hz, 1H), 7.87 (dd, J = 10.0, 1.2 Hz, 1H), 8.73 (d, J = 1.2 Hz, 1H), 9.74 (s, 1H) (solvent: CDCl3) 644 201-203
TABLE-US-00148 TABLE 148 Compound Melting Point MS UV No. (° C.) 1 H-NMR (d) (m/z) (λmax: nm) 645 oil 1.58(3H, s), 1.87(1H, ddd, J = 14.0, 10.4, 3.6), 222 2.16(1H, ddd, J = 14.0, 6.3, 3.5), 2.34(3H, s), 2.70(1H, ddd, J = 12.3, 10.4, 3.5), 2.90(1H, ddd, J = 12.3, 6.3, 3.6), 5.38(2H, s), 7.18-7.33(3H, m), 7.42(1H, d, J = 8.0), 7.49-7.60(2H, m), 7.69(1H, dt, J = 7.7, 1.9), 8.59(1H, ddd, J = 4.9, 1.9, 1.1) (solvent: CDCl3) 649 161-162 651 193-196 1.59 (s, 3H), 1.90 (ddd, J = 14.0, 10.4, 3.6 Hz, 1H), 2.18 (ddd, J = 14.0, 6.4, 3.6 Hz, 1H), 2.76 (ddd, J = 12.4, 10.4, 3.6 Hz, 1H),, 2.93 (ddd, J = 12.4, 6..4, 3.6 Hz, 1H), 4.42 (br, 2 H), 7.17 (ddd, J = 8.0, 2.0, 0.8 Hz, 1H), 7.38.(t, J = 8.0 Hz, 1H), 7.64 (t, J = 2.0 Hz, 1H), 7.77 (ddd, J = 8.0, 2.0, 0.8 Hz, 1H), 8.20 (dd, J = 8.0, 2.0 Hz, 1H), 8.44 (dd, J = 8.0, 0.8 Hz, 1H), 8.91 (dd, J = 2.0, 0.8 Hz, 1H), 9.87 (s, 1H) (solvent: CDCl3) 652 d in d21-DMSO:1.67(3H, s), 2.14-2.07(1H, m), 2.62- 2.57(2H, m), 3.17-3.14(1H, m), 5.74(1H, s), 7.14(1H, d, J = 8.0 Hz), 7.44(1H, t, J = 8.0 Hz), 7.85- 7.81(2H, m), 8.01(1H, d, J = 12.0 Hz), 8.16(1H, d, J = 8.0 Hz), 8.77(1H, s), 10.95(1H, s) 653 193-194 654 oil 257 657 199-203 660 amorphous 223, 266 661 d in d9-DMSO:1.30(3H, t, J = 7.0 Hz), 1.69(3H, s), 2.10-2.04(1H, m), 2.20(3H, s), 2.67-2.62(2H, m), 3.20-3.17(1H, m), 4.40(2H, q, J = 14.0, 7.0 Hz), 6.83(1H, s), 7.25(1H, q, J = 12.0, 9.0 Hz), 7.62- 7.61(1H, m), 7.85-7.83(1H, m), 10.42(1H, s) 664 amorphous 225, 267 667 amorphous 226 673 oil 224 677 amorphous 216 680 159-160 1.63(3H, 5), 1.65-1.80(1H, m), 2.53-2.64(1H, m), 2.75-2.88(2H, m), 3.83(3H, s), 4.32(2H, br), 6.87- 6.96(2H, m), 7.19-7.33(2H, m) (solvent : CDCl3) 681 d in d6-DMSO: 1.43(3H, s), 1.66-1.74(1H, m), 2.02- 338[M + 1] 2.07(1H, m), 2.56-2.63(1H, m), 2.85-2.90(1H, m), 5.80(2H, bs), 6.91(1H, d, J = 7.8 Hz), 6.96-6.98(2H, m), 7.25(1H, t, J = 7.8 Hz), 7.2-7.36(2H, m), 7.40(1H, m), 7.89-7.92(1H, m), 9.42(1H, bs), 10.78(1H, bs). 683 166-168
TABLE-US-00149 TABLE 149 Compound Melting Point MS UV No. (° C.) 1H-NMR (d) (m/z) (λmax: nm) 687 164-167 1.60 (3H, s), 1.84-1.95 (1H, m), 2.21-2.26 (1H, m), 388[M + 1] 2.73-2.94 (2H, m), 3.92 (3H, s), 4.25 (3H, s), 7.10 (1H, d, J = 7.58 Hz), 7.34 (1H, t, J = 7.91 Hz), 7.40 (1H, s), 7.57 (1H, br s), 7.66 (1H, d, J = 7.91 Hz), 8.67 (1H, s). (solvent: CDCl3) 692 (CDCl3) 1.50(3H, s), 1.75-1.88(1H, m), 2.00- 2.10(1H, m), 2.91-2.99(1H, m), 3.08-3.18(1H, m), 6.21(1H, d, J = 15.9 Hz), 6.59(1H, d, J = 15.9 Hz), 7.42- 7.47(3H, m), 7.59(1H, dd, J = 8.6, 2,0 Hz), 7.74- 7.83(4H, m) 698 269 700 177-178 701 1.61(s, 3H), 1.90(m, 1H), 2.25(m, 1H), 2.81(m, 1H), 2.92(m, 1H), 3.86(s, 3H), 6.71(t-like, J = 1.8 Hz, 1H), 7.12(t-like, J = 1.8 Hz, 1H), 7.53(t-like, J = 1.8 Hz, 1H), 7.89(dd, J = 8.3 Hz, 2.4 Hz, 1H), 8.24(d, J = 8.3 Hz, 1H), 8.58(d, J = 2.4 Hz, 1H), 9.85(br, 1H) (solvent: CDCl3) 702 1H-NMR (CDCl3) d: 1.65 (3H, s), 1.91-1.98 (1H, m), 2.57-2.62 (1H, m), 2.68-2.75 (1H, m), 2.92-2.97 (1H, m), 4.18 (3H, s), 6.82 (1H, br s), 7.02-7.08 (1H, m), 7.28-7.32 (1H, m), 7.44 (1H, s), 7.92-7.96 (1H, m). 707 167-174 709 99-100 0.82(3H, t, J = 7.3 Hz), 1.72-1.90(3H, m), 2.06- 2.15(1H, m), 2.61-2.82(2H, m), 3.80(3H, s), 4.36(2H, br), 6.86(2H, d, J = 8.9 Hz), 7.17(2H, d, J = 8.9 Hz) (solvent: CDCl3) 717 157-162 1.58 (s, 3H), 1.90 (ddd, J = 14.0, 10.4, 3.6 Hz, 1H), 2.15 (ddd, J = 14.0, 6.8, 3.6 Hz, 1H), 2.76 (ddd, J = 12.4, 10.4, 3.6 Hz, 1H), 2.94 (ddd, J = 12.4, 6.8, 3.6 Hz, 1H), 3.49 (1H, S), 3.76 (2H, br), 7.17 (ddd, J = 8.0, 2.0, 0.8 Hz, 1H), 7.36 (t, J = 8.0 Hz, 1H), 7.38 (d, J = 1.6 Hz, 1H), 7.50 (t, J = 2.0 Hz, 1H), 7.73 (ddd, J = 8.0, 2.0, 0.8 Hz, 1H), 8.22 (d, J = 2.4 Hz), 9.26 (d, J = 2.4 Hz, 1H), 10.12 (s, 1H) (solvent: CDCl3) 719 oil 226 254 720 133-138 725 amorphous 1.62 (s, 3H), 1.96-2.03(m, 1H), 2.38-2.49 (m, 1H), 265 2.63-2.71 (m, 1H), 3.05-3.12 (m, 1H), 6.73 (dd, J = 3.2, 1.6 Hz, 2H), 7.35(d, J = 3.2 Hz, 1H), 7.37 (br s, 1H), 7.57 (d, J = 3.4 Hz, 2H), 7.67 (d, J = 8.4 Hz, 2H), 7.77 (br s, 1H), 7.96(br s, 1H), 8.01(br s, 1H), 10.35 (s, 1H) (solvent: DMSO-d6) 728 179-182 729 167-169
TABLE-US-00150 TABLE 150 Compound Melting Point MS UV No. (° C.) 1H-NMR (d) (m/z) (λmax: nm) 730 211.0 289.9 731 91-94 732 amorphous 211 735 166-168 737 1H-NMR (CDCl3) d: 1.59 (3H, s), 1.87-1.94 (1H, m), 2.47-2.53 (1H, m), 2.67-2.73 (1H, m), 2.93-2.99 (1H, m), 4.10 (3H, s), 6.62 (1H, s), 7.04 (1H; t, J = 10.2 Hz), 7.33 (1H, d, J = 4.3 Hz), 7.85 (1H, br s). 738 181-183 739 285 740 250 (dec.) 743 148-150 1.60 (s, 3H), 179-2.93 (m, 4H), 4.48 (2H, br); 7.09 (d, J = 2.0 Hz, 1H), 7.12 (ddd, J = 7.6, 2.0, 0.8 Hz, 1H), 7.18 (t, J = 2.0 Hz, 1H), 7.36 (d, J = 7.6, 2.0, 0.8 Hz, 1H), 7.43 (t, J = 7.6 Hz, 1H), 8.21(d, J = 2.0 Hz) (solvent: CDCl3) 744 d in d8-DMSO:1.47(3H, s), 1.82-1.78(1H, m), 2.22- 2.18(1H, m), 2.62-2.56(1H, m), 3.00-2.96(1H, m), 6.79(1H, s), 6.63(1H, s), 7.08-7.03(1H, m), 7.51(1H, s), 7.64-7.57(2H, m), 9.57(1H, s), 11.25(1H, s) 753 amorphous 225, 299 756 110-111 1.55(3H, s), 1.76-1.87(1H, m), 2.08-2.17(1H, m), 2.35(3H, s), 2.65-2.76(1H, m), 2.82-2.92(1H, m), 4.35(2H, br), 7.01-7.25(4H, m) (solvent: CDCl3) 758 156-157 766 336[M + 1] 203 260 212 767 98-100 768 1.60 (3H, d, J = 1.3 Hz), 1.89-1.99 (1H, m), 2.29 362[M + 1] 213 (3H, s), 2.37-2.42 (1H, m), 2.70-2.75 (1H, m), 2.96- 263 3.00(1H, m), 4.12 (3H, s), 6.39 (1H, s), 7.04 (1H, dd, J = 11.5, 8.9 Hz), 7.18 (1H, dd, J = 6.9, 2.6 Hz), 7.60(1H, s), 7.82-7.86 (1H, m). (solvent: CDCl3) 771 417[M + 1] 201 341 774 1H-NMR (CDCl3) d: 1.77 (3H, s), 2.11-2.21 (1H, m), 400[M + 1] 2.71-2.80 (1H, m), 2.87-2.99 (2H, m), 6.91 (1H, d, J = 6.9 Hz), 7.28 (2H, s), 7.47 (1H, t, J = 8.1 Hz), 7.75 (1H, t, J = 8.6 Hz), 8.04 (1H, dd, J = 8.6, 2.3 Hz), 8.29 (1H, d, J = 8.2 Hz), 8.46 (1H, d, J = 2.2 Hz).
TABLE-US-00151 TABLE 151 Compound Melting Point MS UV No. (° C.) 1H-NMR (d) (m/z) (λmax: nm) 781 1.63 (s, 3H), 1.92 (ddd, J = 14.0, 10.8, 4.0 Hz, 1H), 2.29 (m, 1H), 2.78 (ddd, J = 12.4, 10.8, 3.6 Hz, 1H),, 2.91 (ddd, J = 12.4, 6.4, 4.0 Hz, 1H), 3.94 (3H, s), 7.09 (ddd, J = 8.0, 2.0, 0.8 Hz, 1H), 7.34 (dd, J = 8.8, 2.8 Hz, 1H), 7.35 (t, J = 8.0 Hz, 1H), 7.68 (t, J = 2.0 Hz, 1H), 7.71 (ddd, J = 8.0, 2.0, 0.8 Hz, 1H), 8.24 (d, J = 8.8 Hz, 1H), 8.28 (d, J = 2.8 Hz, 1H), 9.86 (s, 1H) (solvent: CDCl3) 783 205-206 786 1.66(3H, s), 2.10(1H, m), 2.57-2.64(2H, m), 3.16(1H, m), 6.74(1H, s), 7.30(1H, s), 7.36(1H, s), 7.74(1H, s), 7.98(1H, s), 8.06(1H, s), 10.33(1H, s), 10.47(1H, s) (solvent: DMSO-d6) 790 amorphous 223, 290 791 d in d18-DMSO:1.41(3H, s), 1.76-1.69(1H, m), 2.02- 1.98(1H, m), 2.62-2.55(1H, m), 2.92-2.89(1H, m), 7.13(1H, d, J = 7.6 Hz), 7.29(1H, t, J = 7.6 Hz), 7.62- 7.59(2H, m), 8.71(1H, s), 9.28(1H, s), 10.46(1H, brs) 792 299.4 793 269 (dec.) 797 213.4 312.4 799 215, 240 800 225, 275 802 1.63 (s, 3H), 1.92 (ddd, J = 14.0, 11.2, 3.6 Hz, 1H), 2.28 (br, 1H), 2.78 (ddd, J = 12.4, 11.2, 3.6 Hz, 1H), 2.81 (s, 3H), 2.92 (ddd, J = 12.4, 6.4, 4.0 Hz, 1H), 7.10 (ddd, J = 8.0, 2.0, 0.8 Hz, 1H), 7.35 (t, J = 8.0 Hz, 1H), 7.56 (t, J = 2.0 Hz, 1H), 7.65 (d, J = 2.4 Hz, 1H), 7.74 (ddd, J = 8.0, 2.0, 0.8 Hz, 1H), 8:41 (d, J = 2.4 Hz, 1H), 10.03 (s, 1H) (solvent: CDCl3) 803 271 804 135-136 810 47-48 811 138-139 813 204-205 182 (s, 3H), 1.89-1.94 (m, 1H), 2.78 (ddd, J = 12.4, 6.4,3.6 Hz, 1H), 4.50 (2H, br), 7.06 (dd, J = 11.6, 8.8 Hz, 1H), 7.40 (dd, J = 7.2, 2.8 Hz, 1H), 7.59 (ddd, J = 8.8, 8.0, 2.8 Hz, 1H), 7.99 (ddd, J = 8.8, 4.4, 2.8 Hz, 1H), 8.33 (dd, J = 8.8, 4.4 Hz, 1H), 8.45 (d, J = 2.8 Hz, 1H), 9.78 (s, 1H)(solvent: CDCl3) 814 oil 218, 272 816 214.5
TABLE-US-00152 TABLE 152 Compound Melting Point MS UV No. (° C.) 1H-NMR (d) (m/z) (λmax: nm) 820 (CDCl3) 1.66(3H, d, J = 1.2 Hz), 1.98(1H, ddd, J = 14.0, 10.4, 3.7 Hz), 2.47(1H, ddd, J = 14.0, 6.7, 3.5 Hz), 2.79(1H, ddd, J = 12.0, 10.4, 3.5 Hz), 3.02(1H, ddd, J = 12,0, 6.7, 3.7 Hz), 4.45(2H, br), 6.16(2H, br), 7.04- 7.11(2H, m), 7.38(1H, dd, J = 7.2, 2.9 Hz), 7.88(1H, d, J = 2.0 Hz), 7:96(1 H, ddd, J = 8,9, 4.2, 2.9 Hz), 9.88(1H, s) 822 279 827 134-137 214.5 284.0 832 212, 299 833 oil 212, 273 834 217, 267 835 139-140 836 221.6 279.3 840 223-225 848 oil 223, 254 849 143-145 850 d in d16-DMSO:1.41(3H, s), 1.75-1.70(1H, m), 2.02- 1.99(1H, m), 2.61-2.56(1H, m), 2.93-2.88(1H, m), 7.13(1H, d, J = 8.0), 7.29(1H, t, J = 7.8 Hz), 7.35(1H, q, J = 8.4, 2.4 Hz), 7.66-7.63(2H, m), 8.52;8.47(1H, m), 8.81(1H, s), 10.44(1H, s) 851 82-83 1.55(3H, s), 1.76-1.88(1H, m), 2.10-2.18(1H, m), 2.66-2.77(1H, m), 2.82-2.91(1H, m), 3.81(3H, s), 6.73-6.78(1H, m), 6.88-6.92(2H, m), 7.21-7.29(1H, m) (solvent: CDCl3) 855 oil 219 859 350[M + 1] 200 274 208 254 863 192-194 1.39(t, J = 7.2 Hz, 3H), 1.42(s, 3H), 1.71-1.79(m, 1H), 2.02-2.10(m, 1H), 2.55-2.62(m, 1H), 2.88- 2.96(m, 1H), 4.47(q, J = 7.2 Hz, 2H), 5.70-6.20(br s, 2H), 7.11(d, J = 8.0 Hz, 1H), 7.29(t, J = 8.0 Hz, 1H), 7.75(d, J = 8.0 Hz, H), 7.80(br s, 1H), 8.38(d, J = 1.2 Hz, 1H), 8.87(d, J = 1.2 Hz, 1H), 10.34(s, 1H) (solvent: CDCl3) 866 293.5 869 1.65 (s, 3H), 1.90-2.01 (m, 3H), 2.32 (br, 1H), 2.80 (td, J = 12.0, 3.6 Hz, 1H), 2.85 (t, J = 8.0 Hz, 2H), 2.92 (ddd, J = 12.0, 5.6, 3.6, 1H), 3.75 (t, J = 8.0 Hz, 2H), 7.11 (ddd, J = 8.0, 2.0, 0.8 Hz, 1H), 7.37 (t, J = 8.0 Hz, 1H), 7.70 (t, J = 2.0 Hz, 1H), 7.73-7.76 (m, 2H), 8.22 (d, J = 7.6 Hz, 1H), 8.48 (d, J = 2.0 Hz, 1H), 10.00 (s, 1H) (solvent: CDCl3) 871 212-213
TABLE-US-00153 TABLE 153 Compound Melting Point MS UV No. (° C.) 1H-NMR (d) (m/z) (λmax: nm) 875 oil 222, 271 876 oil 222 878 oil 211 881 141-144 887 262.7 892 251 (dec.) 893 d in d12-DMSO:1.70(3H, s), 2.10-2.04(1H, m), 2.69- 2.59(2H, m), 3.20-3.17(1H, m), 6.80(1H, brs), 7.26- 7.20(1H, m), 7.88-7.81(3H, m), 10.35(1H, s) 13.53(1H, brs) 895 378[M + 1] 202 302 208 216 221 265 896 amorphous 219, 264 897 212-214 900 205-207 1.61 (s, 3H), 1.91 (ddd, J = 14.0, 10.8, 4.0 Hz, 1H), 2.23 (ddd, J = 14.0, 6.4, 3.6 Hz, 1H), 2.77 (ddd, J = 12.4, 10.8, 3.6 Hz, 1H), 2.92 (ddd, J = 12.4, 6.4, 4.0 Hz, 1H), 7.15 (ddd, J = 8.0, 2.0, 0.8 Hz, 1H), 7.38 (t, J = 8.0 Hz, 1H), 7.65 (t, J = 2.0 Hz, 1H), 7.79 (ddd, J = 8.0, 2.0, 0.8 Hz, 1H), 8.99 (s, 2H), 9.78 (s, 1H) (solvent: CDCl3) 906 212.2 273.4 350.5 908 d in d15-DMSO:1.66(3H, s), 2.11-2.05(1H, m), 2.37(3H, s), 2.63-2.54(2H, m), 3.16-3.11(1H, m), 3.16(3H, s), 7.08-6.96(3H, m), 7.49-7.41(3H, m), 7.85-7.81(2H, m), 10.52(1H, s)11.69(1H, s) 910 oil 211, 276 916 131-132 926 1.89(3H, s), 2.15(1H, m), 2.71-2.82(2H, m), 2.96(1H, m), 3.04(3H, d, J = 4.9), 7.35(1H, dd, J = 8.7, 1.8), 7.50-7.55(2H, m), 7.74(1H, s), 7.82-7.90(3H, s), 10.40(1H, br), 11.36(1H, Br) (solvent: CDCl3) 928 1.20(t, J = 7.6 Hz, 3H), 1.53(br s, 3H), 1.82-1.97(m, 1H), 2.39(s, 3H), 2.61(q, J = 7.6 Hz, 2H), 2.99- 3.07(m, 1H), 6.93(br s, 1H), 7.33(d, J = 8.4 Hz, 2H), 7.54-7.58(m, 2H), 7.87(d, J = 8.4 Hz, 2H), 10.13(s, 1H) (solvent: CDCl3) 930 132.1-134.4 328[M + 1] 931 299 933 amorphous 212, 259
TABLE-US-00154 TABLE 154 Compound Melting Point MS UV No. (° C.) 1H-NMR (d) (m/z) (λmax: nm) 935 161-165 1.62 (s, 3H), 1.91 (ddd, J = 14.0, 10.4, 4.0 Hz, 1H), 2.24 (ddd, J = 14.0, 6.4, 3.6 Hz, 1H), 2.80 (ddd, J = 12.0, 10.4, 3.6 Hz, 1H), 2.93 (ddd, J = 12.0, 6.4, 4.0 Hz, 1H), 7.15 (ddd, J = 8.0, 2.0, 1.2 Hz, 1H), 7.39 (t, J = 8.0 Hz, 1H), 7.66 (ddd, J = 8.4, 7.2, 1.2 Hz, 1H), 7.75 (t, J = 2.0 Hz, 1H), 7.80-7.84 (m, 2H), 7.93 (ddd, J = 8.0, 2.0, 1.2 Hz), 8.21 (d, J = 8.4 Hz, 1H), 8.38 (d, J = 8.0 Hz, 1H), 8.41 (d, J = 8.0 Hz, 1H), 10.25 (s, 1H) (solvent: CDCl3) 936 169-170 939 d in d6-DMSO:1.72(3H, s), 2.11-2.05(1H, m), 2.70- 2.60(2H, m), 3.21-3.18(1H, m), 7.20(1H, d, J = 9.2 Hz), 7.28(1H, q, J = 11.6, 9.2 Hz), 8.56-7.54(2H, m), 7.69(1H, s), 7.90-7.85(2H, m), 10.69(1H, s), 12.17(1H, brs) 941 220 944 amorphous 219, 256 946 1.61 (s, 3H), 1.91 (ddd, J = 14.0, 10.8, 3.6 Hz, 1H), 2.26 (ddd, J = 14.0, 6.4, 3.6 Hz, 1H), 2.77 (ddd, J = 12.4, 10.8, 3.6 Hz, 1H),, 2.92 (ddd, J = 12.4, 6.4, 3.6 Hz, 1H), 7.13 (ddd, J = 8.0, 2.0, 1.2 Hz, 1H), 7.36 (t, J = 8.0 Hz, 1H), 7.61 (t, J = 2.0 Hz, 1H), 7.72 (ddd, J = 8.0, 2.0, 1.2 Hz, 1H), 7.91 (d, J = 2.4 Hz, 1H), 8.49 (d, J = 2.4 Hz, 1H), 9.75 (s, 1H)(solvent: CDCl3) 947 215.7 276.9 960 261.5 964 185-187 966 oil 216 968 107-109 970 1.57 (s, 3H), 1.78-1.89 (m, 1H), 2.10-2.19 (m, 1H), 2.69 (ddd, J = 11.9, 10.8, 3.5 Hz, 1H), 2.83-2.91 (m, 1H), 7.15-7.35 (m, 5H) (solvent: CDCl3) 971 (DMSO) 1.49(3H, s), 1.73-1.86(1H, m), 2.16- 2.30(1H, m), 2.54-2.65(1H, m), 2.92-3.03(1H, m), 5.86(2H, s), 7.04-7.18(2H, m), 7.38-7.50(3H, m), 7.66-7.78(2H, m), 10.35(1H, s), 11.84(1H, s) 972 1.51 (3H, s) 1.91-1.95 (1H, m) 2.37 (3H,s) 3.00-3.05 (1H, m) 7.24 (1H s) 7.33 (2H, d J = 9.0 Hz) 7.66 (1H,s) 7.85 (2H, d J = 9.0 Hz) 8.03 (1H, s) 10.37 (1H, s) (solvent: DMSO-d6) 974 amorphous 219 978 oil 222 984 255.7 318.4 990 126-129 994 130-131
TABLE-US-00155 TABLE 155 Compound Melting Point MS UV No. (° C.) 1H-NMR (d) (m/z) (λmax: nm) 998 amorphous 229, 290 1005 191-193 1006 88-90 2.42-2.47(2H, m), 2.80-2.86(2H, m), 7.78(6H, s), 6.83(4H, d, J = 8.9 Hz), 7.22(4H, d, J = 8.9 Hz) (solvent: CDCl3) 1008 125-126 1010 90-91 1014 206-210 1020 216.9 245.1 1028 105-106 1034 212.2 286.4 1035 247-251 (dec.) 1037 amorphous 224, 272 1039 amorphous 217 249 1043 277-281 1044 (DMSO) 1.12(3H, s), 1.60(2H, d, J = 6.2 Hz), 1.73(2H, d, J = 8.6 Hz), 2.65-2.90(2H, m), 2.93-3.13(2H, m), 5.55(1H, s), 7.34-7.52(3H, m), 7.68(1H, s), 7.79- 7.90(3H, m) 1052 1.75(s, 3H), 2.12-2.21(m, 1H), 2.40(s, 3H), 2.65- 2.73(m, 2H), 3.17-3.23(m, 1H), 7.37(d, J = 8.4 Hz, 2H), 7.40-7.44(m, 1H), 7.77(br s, 1H), 7.92-7.99(m, 5H), 8.47(br s, 1H), 8.70(d, J = 4.8 Hz, 1H), 10.37(s, 1H), 10.41(s, 1H) (solvent: CDCl3) 1055 169-170 1.56(3H, s), 1.78-1.89(1H; m), 2.04-2.15(1H, m), 2.68-2.79(1H, m), 2.86-2.95(1H, m), 4.32(2H, br), 6.94-7.02(4H, m), 7.05-7.12(1H, m), 7.25-7.37(4H, m) (solvent: CDCl3) 1056 219 1059 262-267 1061 216 1062 136-137 1.53(3H, s), 1.76-1.88(1H, m), 2.03-2.13(1H, m), 2.63-2.73(1H, m), 2.85-2.94(1H, m), 4.35(2H, br), 7.23-7.32(4H, m) (solvent: CDCl3) 1064 84-85 1.52(3H, s), 1.73-1.89(1H, m), 1.97-2.07(1H, m), 2.64-2.81(1H, m), 2.82-2.91(1H, m),2.87(3H, s), 3.77(3H, s), 4.10(1H, brs), 6.84(2H, d, J = 8.9 Hz), 7.28(2H, d, J = 8.6 Hz) (solvent: CDCl3) 1067 162-165 1068 132-134 230 1069 194-196 1074 324[M + 1] 200 248 207 1076 amorphous 217
TABLE-US-00156 TABLE 156 Compound Melting Point MS UV No. (° C.) 1H-NMR (d) (m/z) (λmax: nm) 1084 146-149 1087 311.2 1088 amorphous 1.55(3H, s), 1.83(1H, ddd, J = 13.9, 10.5, 3.7), 229 2.09(1H, ddd, J = 13.9, 6,6, 3.6), 2.67(1H, ddd, J = 318 12.3, 10.5, 3.6), 2.88(1H, ddd, J = 12.3, 6.6, 3.7), 4.48(2H, d, J = 6.0), 4.91(1H, br), 6.33(1H, dd, J = 8..8, 0.8), 7.19(1H, d, J = 7.3, 7.23-7.30(2H, m), 7.35(1H, dd, J = 8.8, 2.8), 8.05(1H, dd, J = 2.8, 0.8) (solvent: CDCl3) 1094 216, 322 1100 278 (dec.) 1107 oil 1.58(3H, s), 1.90(1H, ddd, J = 13.9, 10.1, 3.7), 226 2.14(1H, ddd, J = 13.9, 6.8, 3.6), 2.69(1H, ddd, J = 284 12.2, 10.1, 3.6), 2.94(1H, ddd, J = 12.2, 6.8, 3.7), 3.81(3H, s), 4.62(2H, s), 6.90(2H, d, J = 8.8), 7.30(2H, d, J = 8.8), 7.43(1H, t, J = 7.4), 7.57(1H, ddd, J = 7.4, 1.6, 1.2), 7.81(1H, ddd, J = 7.6, 1.6, 1.2), 7.95(1H, t, J = 1.6) (solvent: CDCl3) 1109 134-140 1110 109-110 1111 118-119 1114 121-124 1115 167-170 1.63 (s, 3H), 1.93 (ddd, J = 14.0, 10.4, 4.0 Hz, 1H), 2.24 (ddd, J = 14.0, 6.4, 3.6 Hz, 1H), 2.81 (ddd, J = 12.4, 10.4, 3.6 Hz, 1H), 2.96 (ddd, J = 12.4, 6.4, 4.0 Hz, 1H), 4.49 (br, 2 H), 7.19 (ddd, J = 8.0, 2.0, 0.8 Hz, 1H), 7.42 (t, J = 8.0 Hz, 1H), 7.74 (t, J = 2.0 Hz, 1H), 7.84 (ddd, J = 8.0, 2.0, 0.8 Hz, 1H), 7.88-7.95 (m, 2H), 8.22-8.26 (m, 2H), 9.80 (s, 1H), 9.89 (s, 1H) (solvent: CDCl3) 1116 oil 220, 255, 307 1119 153-157 1120 213-214 1124 169-172 225 1125 195-198 222 256 289 1131 189-191 1132 175-180 (dec) 1133 amorphous 219, 292 1135 255-260 (dec.) 1139 140-141 1140 oil 218 1142 182-186 (dec.)
TABLE-US-00157 TABLE 157 Compound Melting Point MS UV No. (° C.) 1H-NMR (d) (m/z) (λmax: nm) 1147 214.5 275.7 1150 221.6 279.3 1153 156-159 1160 1.64 (3H, s) 2.02-2.12 (1H, m) 2.54-2.63 (1H, m) 3.11-3.16 (1H, m) 7.28 (1H, s) 7.70 (1H, dd J = 8.1 Hz) 7.85 (1H,s) 8.04-8.17 (2H, m) 8.28 (1H s) 8.74 (1H d J = 5.1 Hz) 10.81 (1H, s) 10.96 (1H, s) (solvent: DMSO-d6) 1161 192-193 1166 290-295 444[M + 3] 442[M + 1] 368 366 1172 1.55 (3H, s) 1.94-2.03 (1H,m) 2.18-2.27 (1H, m) 2.32 (3H, s) 3.03-3.07 (1H, m) 7.05 (1H. s) 7.09 (1H, s) 7.14 (1H, s) 7.37 (2H, d J = 9.0 Hz) 7.66 (2H, d J = 9.0 Hz) 10.65 (1H, s) 10.70 (1H,s) (solvent: DMSO-d6) 1181 194-195 1.60(3H, s), 1.81-1.93(1H, m), 2.13-2.22(1H, m), 2.70-2.81(1H, m), 2.86-2.96(1H, m), 4.36(2H, br), 7.29-7.46(5H, m), 7.53-7.61(4H, m) (solvent: CDCl3) 1184 149-150 1185 225.1 280.4 1193 182-183 1194 344[M + 1] 209 268 214 261 1197 250-255 (dec.) 1199 274-283 1205 oil E 213, 273 Z 219, 275 1207 106-108 1211 1.77 (s, 3H), 1.98- 2.54 (m, 2H), 2.81 (s, 3H), 2.81- 2.94 (m, 2H), 3.93 (s, 3H), 7.03 (ddd, J = 8.0, 2.0, 0.8 Hz, 1H), 7.08 (d, J = 2.4 Hz, 1H), 7.36 (t, J = 8.0 Hz, 1H), 7.63 (t, J = 2.0 Hz, 1H), 7.69 (ddd, J = 8.0, 2.0, 0.8 Hz, 1H), 8.14 (d, J = 2.4 Hz, 1H), 10.13 (s, 1H) (solvent: CDCl3) 1213 406[M + 1] 20 330 209 213
TABLE-US-00158 TABLE 158 Compound Melting Point MS UV No. (° C.) 1H-NMR (d) (m/z) (λmax: nm) 1215 amorphous 1.64 (s, 3H), 2.07 (ddd, J = 14.1, 11.5, 3.8 Hz, 1H), 2.17 (s, 3H), 2.39 (ddd, J = 14.1, 5.3, 3.5 Hz, 1H), 2.72 (ddd, J = 12.6, 11.5, 3.5 Hz, 1H), 2.80 (ddd, J = 12.6, 5.3, 3.8 Hz, 1H), 3.21 (t, J = 8.9 Hz, 2H), 4.58 (t, J = 8.9 Hz, 2H), 6.76 (d, J = 8.4 Hz, 1H), 6.97-7.02 (m, 1H), 7.08-7.11 (m, 1H) (solvent: CDCl3) 1216 305.3 1217 263-266 1221 amorphous 220, 253 1223 226.3 280.4 1224 d in d11-DMSO:1.46(3H, s), 1.83-1.77(1H, m), 2.18- 2.15(1H, m), 2.61-2.56(1H, m), 2.99-2.95(1H, m), 7.08(1H, q, J = 12.0, 8.4 Hz), 7.72-7.66(2H, m), 7.79(2H, d, J = 9.2)9.67(1H, s) 1228 oil 224 1230 232-234 1240 216.9 285.2 1241 194-195 1242 d in d21-DMSO:1.41(3H, m), 1.75-1.68(1H, m), 2.04- 1.99(1H, m), 2.61-2.56(1H, m), 2.89(4H, s), 5.75(2H, brs), 7.07(1H, d, J = 4.0 Hz), 7.25(1H, t, J = 8.0 Hz), 7.72(1H, d, J = 8.0 Hz), 7.75(1H, 5), 7.83(1H, brs), 7.96(1H, s), 8.67(1H, s), 9.96(1H, s) 1243 amorphous 1.58(3H, s), 2.00(1H, ddd, J = 14.3, 11.5; 3.1), 223 2.53(1H, m), 2.56(1H, m), 3.07(1H, dt, J = 12.5, 299 3.1), 4.26(2H, s), 6.47-6.56(3H, m), 7.07-7.15(1H, m), 7.12(2H, t, J = 8.8), 7.39(2H, dd, J = 8.8, 5.6), 8.76(2H, br) (solvent: DMSO-d6) 1244 268-288 1.68 (s, 3H), 2.11 (ddd, J = 15.2,12.0, 4.0 Hz, 1H), 219 2.57-2.64 (m, 2H), 3.16 (dt, J = 12.0, 4.0 Hz, 1H), 288 7.13 (ddd, J = 8.0, 2.0, 0.8 Hz, 1H), 7.46 (t, J = 8.0 Hz, 1H), 7.89 (t, J = 2.0 Hz, 1H), 7.97 (ddd, J = 8.0, 2.0, 0.8 Hz, 1H), 8.35 (d, J = 8.0 Hz, 1H), 8.52 (dd, J = 8.0, 2.4 Hz, 1H), 9.12 (d, J = 2.4 Hz, 1H), 10.68 (s, 1H), 10.92 (s, 1H) (solvent: DMSO-d6) 1245 oil 286 1247 211 1255 242.7 1257 amorphous 211 1258 352[M + 1] 228 276 301 1261 179-180 1262 278-281
TABLE-US-00159 TABLE 159 Compound Melting Point MS UV No. (° C.) 1H-NMR (d) (m/z) (λmax: nm) 1263 1H-NMR(d in d6-DMSO): 1.41(3H, s), 1.65-1.77 387[M + 1] (1H, m), 1.95-2.07(1H, m), 2.54-2.63(1H, m), 2.84- 2.94(1 H, m), 3.39-3.46(2H, m), 3.53-3.61(2H, m), 4.83(1H, t, J = 5:4 Hz), 5.79(2H, bs), 7.07(1H, d, J = 7.5 Hz), 7.25(1H, t, J = 7.8 Hz), 7.73(1H, d, J = 7.8 Hz), 7.76(1H, m), 7.87-7.93(1H, m), 8.62(1H, d, J = 1.2 Hz), 8.63(1H, d, J = 1.2 Hz), 9.97(1H, s). 1264 1H-NMR(d in d6-DMSO): 1.41(3H, s), 1.65-1.77 413[M + 1] (1H, m), 1.95-2.07(1H, m), 2.53-2.63(1H, m), 2.84- 2.95(1H, m), 3.73(8H, s), 5.79(2H, bs), 7.09(1H, d, J = 7.8 Hz), 7.26(1H, t, J = 7.8 Hz), 7.72(1H, d, J = 7.8 Hz), 7.75-7.78(1H, m), 8.34(1H, d, J = 1.2 Hz), 8.76(1H, d, J = 1.2 Hz), 10.08(1H, bs). 1265 1H-NMR (DMSO-d6) d: 1.42 (3H, s), 1.70-1.76 (1H, m), 2.02-2.05 (1H, m), 2.56-2.59 (1H, m), 2.87-2.93 (2H, m), 7.07 (1H, d, J = 7.6 Hz), 7.23-7.26 (3H, m), 7.72-7.74 (2H, m), 7.93 (1H, s), 8.60 (1H, s), 9.99 (1H, s). 1266 1H-NMR(d in d6-DMSO): 1.43(3H, s), 1.70-1.81 369[M + 1] (1H, m), 1.97-2.10(1H, m), 2.55-2.64(1H, m), 2.89- 2.95(1H, m), 5.84(2H, bs), 7.17(1H, d, J = 7.8 Hz), 7.33(1H, t, J = 7.8 Hz), 9.98(1H, d, J = 1.2 Hz), 10.01(1H, d, J = 1.2 Hz), 10.74(1H, bs). 1267 1H-NMR (CDCl3) d: 1.82-1.91 (1H, m), 2.04 (3H, s), 338[M + 1] 2.22 (1H, ddd, J = 13.8, 52, 3.6 Hz), 2.67 (1H, dt, J = 16.7, 5.8 Hz), 2.80 (1H, dt, J = 12.4, 4.7 Hz), 6.95 (2H, d, J = 8.1 Hz), 7.06 (2H, td, J = 7.8, 1.2 Hz), 7.18 (1H, td, J = 7.6, 1.1 Hz), 7.27 (1H, d, J = 1.7 Hz), 7.32 (1H, d, J = 7.9 Hz), 7.42-7.44 (2H, m), 7.80 (1H, dd, J = 8.0, 1.9 Hz). 1268 1H-NMR (CDCl3) d: 1.62 (3H, s), 1.89 (1H, t, J = 327[M + 1] 12.3 Hz), 2.27-2.30 (1H, m), 2.69-2.76 (1H, m), 2.85- 2.88 (1H, m), 7.11 (1H, dd, J = 11.4, 7.7 Hz), 7.30- 7.53 (2H, m), 7.63 (1H, s), 7.71 (1H, d, J = 6.9 Hz). 1269 1H-NMR (DMSO-d6) d: 1.40 (3H, s), 1.70-1.73 (1H, m), 1.99-2.02 (1H, m), 2.57-2.60 (1H, m), 2.88-2.90 (1H, m), 3.29 (3H, s), 3.52 (4H, s), 5.75 (2H, br s), 7.07 (1H, d, J = 7.6 Hz), 7.25 (1H, t, J = 7.7 Hz), 7.72 (1H, d, J = 8.3 Hz), 7.75 (1H, s), 7.92 (1H, br s), 8.03 (1H, s), 8.64 (1H, s), 9.96 (1H, s). 1271 1H-NMR(d in d6-DMSO): 1.41(3H, s), 1.65-1.75 397[M + 1] (1H, m), 1.99-2.06(5H, m), 2.52-2.61(1H, m), 2.85- 2.93(1H, m), 3.55(4H, t, J = 6.6 Hz), 5.79(2H, bs), 7.05(1H, d, J = 7.8 Hz), 7.25(1H, t, J = 7.8 Hz), 7.70- 7.75(1H, m), 7.73-7.77(1H, m), 7.97(1H, d, J = 1.2 Hz), 8.72(1H, d, J = 1.2 Hz), 10.00(1H, s).
TABLE-US-00160 TABLE 160 Compound Melting Point MS UV No. (° C.) 1H-NMR (d) (m/z) (λmax: nm) 1272 (CDCl3) 1.61(3H, s), 1.85-1.96(1H, m), 2.17- 2.27(1H, m), 2.69-2.79(1H, m), 2.87-2.97(1H, m), 7.17 (1H, d, J = 8.1 Hz), 7.38 (1H, t, J = 8.1 Hz), 7.48- 7.74(5H, m), 8.40(2H, d, J = 7.5 Hz) 1273 1H-NMR (CDCl3) d: 1.58 (3H, s), 1.89 (1H, t, J = 395[M + 1] 11.2 Hz), 2.27 (1H, s), 2.75-2.82 (2H, m), 6.61 (1H, dd, J = 20.3, 8.4 Hz), 7.10 (1H, d, J = 7.2 Hz), 7.37 (1H, dd, J = 15.0, 8.8 Hz), 7.90 (1H, d, J = 7.6 Hz), 8.10 (1H, d, J = 3.2 Hz), 9.37 (1H, d, J = 4.9 Hz), 9.69 (1H, s). 1274 1H-NMR (CDCl3) d: 1.61 (3H, s), 1.84-1.93 (1H, 327[M + 1] m), 2.30 (1H, t, J = 13.1 Hz), 2.77-2.86 (2H, m), 6.64 (1H, dd, J = 20.6, 8.6 Hz), 7.13 (1H, d, J = 7.9 Hz), 7.38-7.43 (1H, m), 7.93 (1H, d, J = 8.1 Hz), 8.13 (1H, s), 9.40 (1H, d, J = 4.9 Hz), 9.72 (1H, s). 1275 1H-NMR (DMSO-d6) d: 1.40 (3H, s), 1.70-1.72 (1H, m), 2.01-2.04 (1H, m), 2.18 (6H, s), 2.44 (2H, t, J = 6.3 Hz), 2.56-2.59 (1H, m), 2.86-2.92 (1H, m), 7.06 (1H, d, J = 7.6 Hz), 7.25 (1H, t, J = 7.7 Hz), 7.71-7.73 (3H, m), 8.0211H, s), 8.64 (1H, s), 9.95 (1H, s). 1276 1H-NMR (DMSO-d6) d: 1.70-1.73 (1H, m), 1.99- 2.02 (1H, m), 2.57-2.60 (1H, m), 2.88-2.91 (1H, m), 3.04 (3H, s), 3.4.3 (3H, t, J = 6.3 Hz), 3.79-3.81 (2H, m), 5.75 (3H, br s), 7.08 (1H, d, J = 7.3 Hz), 7.26 (1H, t, J = 7.8 Hz), 7.72 (1H, d, J = 7.8 Hz), 7.76 (1H, s), 8.04 (1H, s), 8.09 (1H, br s), 8.70 (1H, s), 10.01 (1H, s). 1279 1H-NMR (CDCl3) d: 1.73 (3H, s), 2.04 (1H, dt, J = 328[M + 1] 18.2, 6.5 Hz), 2.45 (1H, d, J = 13.6 Hz), 2.78 (2H, t, J = 11.8 Hz), 2.89 (2H, t, J = 11.5 Hz), 6.60 (1H, s), 6.99 (1H, d, J = 8.2 Hz), 7.34 (1H, t, J = 8.0 Hz), 7.48 (1H, s), 7.70 (1 H, d, J = 8.2 Hz). 1280 1H-NMR(d in d6-DMSO): 1.42(3H, s), 1.68-1.82 426[M + 1] (1H, m), 2.02-2.09(1H, m), 2.23(3H, s), 2.43(4H, t, J = 5.1 Hz), 2.53-2.61(1H, m), 2.87-2.95(1H, m), 3.73(4H, t, J = 5.1 Hz), 6.01(2H, bs), 7.07(1H, d, J = 7.8 Hz), 7.26(1H, t, J = 7.8 Hz), 7.73(1H, d, J = 7.8 Hz), 7.73-7.78(1H, m), 8.33(1H, d, J = 1.2 Hz), 8.72(1H, d, J = 1.2 Hz), 10.06(1H, s). 1281 1H-NMR(d in d6-DMSO): 1.40(3H, s), 1.30-1.50 427[M + 1] (2H, m), 1.69-1.76(1H, m), 1.82-1.88(2H, m), 2.01- 2.07(1H, m), 2.52-2.61(1H, m), 2.86-2.94(1H, m), 3.76-3.83(1H, m), 4.10-4.18(2H, m), 4.82(1H, d, J = 4.2 Hz), 5.91(2H, bs), 7.07(1H, d, J = 7.8 Hz), 7.26(1H, t, J = 7.8 Hz), 7.70-7.77(2H, m), 8.33(1H, d, J = 1.2 Hz), 8.70(1H, d, J = 1.2 Hz), 10.02(1H, s).
TABLE-US-00161 TABLE 161 compound No. MS(m/z) 2 336[M + 1] 76 363[M + 3] 7 394[M + 1] 361[M + 1] 10 431[M + 3] 79 310[M + 1] 429[M + 1] 81 386[M + 1] 11 356[M + 1] 82 308[M + 1] 12 354[M + 1] 83 336[M + 1] 13 363[M + 3] 84 380[M + 1] 361[M + 1] 87 415[M + 1] 14 394[M + 1] 88 426[M + 1] 15 409[M + 1] 89 370[M + 1] 16 425[M + 1] 90 354[M + 1] 17 374[M + 1] 92 417[M + 1] 19 362[M + 3] 93 407[M + 1] 360[M + 1] 94 350[M + 1] 20 438[M + 1] 95 406[M + 3] 21 380[M + 3] 404[M + 1] 378[M + 1] 98 398[M + 3] 22 380[M + 3] 396[M + 1] 378[M + 1] 100 332[M + 1] 25 354[M + 1] 102 424[M + 3] 27 338[M + 1] 422[M + 1] 28 356[M + 1] 103 444[M + 1] 29 372[M + 1] 105 424[M + 1] 31 378[M + 1] 348 32 417[M + 1] 106 490[M + 1] 34 358[M + 1] 414 35 398[M + 3] 107 414[M + 3] 396[M + 1] 412[M + 1] 36 370[M + 1] 108 332[M + 1] 40 416[M + 1] 109 412[M + 1] 340 110 404[M + 1] 41 414[M + 1] 111 469[M + 1] 44 362[M + 3] 393 360[M + 1] 112 377[M + 1] 45 365[M + 1] 116 408[M + 1] 46 362[M + 1] 117 413[M + 1] 47 416[M + 3] 118 372[M + 1] 414[M + 1] 119 424[M + 1] 49 394[M + 3] 122 338[M + 1] 392[M + 1] 124 471[M + 1] 50 292[M + 1] 131 412[M + 3] 51 388[M + 1] 410[M + 1] 52 360[M + 1] 133 404[M + 1] 284 135 416[M + 1] 53 380[M + 1] 136 380[M + 1] 54 332[M + 1] 137 327[M + 1] 55 412[M + 3] 138 394[M + 1] 410[M + 1] 140 456[M + 1] 56 397[M + 1] 142 446[M + 1] 395[M + 1] 143 399[M + 1] 59 412[M + 1] 144 432[M + 1] 60 422[M + 1] 145 394[M + 3] 420[M + 1] 392[M + 1] 61 394[M + 1] 146 433[M + 3] 63 366[M + 1] 431[M + 1] 64 441[M + 1] 147 324[M + 1] 385 150 410[M + 1] 65 384[M + 1] 151 458[M + 3] 66 398[M + 1] 456[M + 1] 67 386[M + 1] 152 371[M + 1] 310 153 398[M + 1] 68 376[M + 1] 154 401[M + 1] 70 372[M + 1] 155 322[M + 1] 72 330[M + 1] 156 332[M + 3] 74 322[M + 1] 330[M + 1] 75 412[M + 1] 158 394[M + 1]
TABLE-US-00162 TABLE 162 160 427[M + 1] 230 368[M + 1] 162 416[M + 3] 231 336[M + 1] 414[M + 1] 234 376[M + 1] 167 392[M + 3] 236 392[M + 1] 390[M + 1] 237 348[M + 1] 168 380[M + 3] 239 384[M + 1] 378[M + 1] 240 341[M + 1] 169 346[M + 1] 242 446[M + 1] 170 356[M + 1] 245 374[M + 1] 171 334[M + 1] 246 390[M + 1] 172 376[M + 3] 314 374[M + 1] 247 374[M + 1] 173 424[M + 3] 248 370[M + 1] 422[M + 3] 249 336[M + 1] 174 369[M + 1] 250 366[M + 1] 175 410[M + 1] 252 401[M + 1] 177 357[M + 1] 253 397[M + 1] 179 334[M + 1] 254 434[M + 1] 180 426[M + 1] 257 321[M + 1] 182 396[M + 3] 258 398[M + 1] 394[M + 1] 260 440[M + 1] 183 372[M + 1] 261 308[M + 1] 184 346[M + 1] 262 466[M + 3] 185 330[M + 1] 464[M + 1] 186 393[M + 3] 264 336[M + 1] 391[M + 1] 265 435[M + 1] 187 374[M + 1] 266 432[M + 3] 188 423[M + 1] 430[M + 1] 190 278[M + 1] 269 372[M + 1] 191 448[M + 1] 296 192 436[M + 3] 270 338[M + 1] 434[M + 1] 272 349[M + 1] 194 384[M + 1] 273 406[M + 3] 195 369[M + 1] 404[M + 1] 197 382[M + 1] 274 380[M + 1] 198 355[M + 1] 276 398[M + 3] 199 361[M + 1] 396[M + 1] 200 356[M + 1] 278 404[M + 1] 280 280 433[M + 3] 201 452[M + 1] 431[M + 1] 203 397[M + 1] 283 322[M + 1] 205 427[M + 1] 285 340[M + 1] 206 386[M + 3] 286 433[M + 1] 310 431[M + 1] 207 384[M + 1] 287 440[M + 1] 208 386[M + 3] 288 354[M + 1] 384[M + 1] 289 341[M + 1] 209 371[M + 1] 290 363[M + 3] 210 366 291 361[M + 1] 211 442[M + 1] 317[M + 1] 366 292 426[M + 1] 212 345[M + 1] 294 424[M + 3] 215 425[M + 3] 422[M + 1] 423[M + 1] 295 394[M + 3] 217 362[M + 1] 392[M + 1] 218 322 [M + 1] 296 389[M + 1] 219 347[M + 1] 297 448[M + 3] 221 444[M + 1] 446[M + 1] 222 329[M + 1] 299 363[M + 3] 223 413[M + 1] 361[M + 1] 225 402[M + 1] 300 356[M + 1] 226 390[M + 1] 303 366[M + 1] 228 383[M + 1] 304 402[M + 1] 229 366[M + 1] 305 407[M + 3] 405[M + 1] 310 411[M + 1]
TABLE-US-00163 TABLE 163 311 388[M + 1] 394 363[M + 3] 312 428[M + 1] 361[M + 1] 313 453[M + 1] 397 460[M + 3] 314 368[M + 1] 458[M + 1] 315 322[M + 1] 398 408[M + 1] 316 386[M + 1] 399 372[M + 1] 317 328[M + 1] 400 374[M + 1] 318 382[M + 1] 402 372[M + 1] 320 327[M + 1] 296 321 392[M + 1] 403 436[M + 1] 322 404[M + 1] 404 376[M + 3] 328 374[M + 1] 323 394[M + 1] 407 449[M + 3] 324 384[M + 1] 447[M + 1] 325 399[M + 1] 412 410[M + 1] 326 440[M + 1] 414 331[M + 1] 364 416 282[M + 1] 327 314[M + 1] 418 322[M + 1] 328 384[M + 1] 419 420[M + 3] 331 360[M + 1] 418[M + 1] 334 412[M + 1] 420 332[M + 1] 335 316[M + 1] 421 388[M + 3] 336 356[M + 1] 386[M + 1] 337 428[M + 1] 423 412[M + 3] 338 466[M + 3] 410[M + 1] 464[M + 1] 424 370[M + 1] 340 344[M + 1] 425 378[M + 1] 343 399[M + 1] 428 350[M + 1] 345 412[M + 1] 431 391[M + 1] 346 384[M + 1] 433 454[M + 3] 347 430[M + 1] 452[M + 1] 348 341[M + 1] 434 448[M + 3] 349 335[M + 1] 446[M + 1] 350 412[M + 1] 435 431[M + 3] 351 322[M + 1] 429[M + 1] 352 327[M + 1] 437 382[M + 1] 355 397[M + 1] 400[M + 1] 362 366[M + 1] 438 324 363 376[M + 3] 439 380[M + 1] 374[M + 1] 440 358[M + 1] 365 366[M + 1] 442 394[M + 1] 366 409[M + 1] 318 368 384[M + 1] 447 370[M + 1] 369 394[M + 3] 449 336[M + 1] 396[M + 1] 450 455[M + 1] 371 398[M + 3] 451 390[M + 3] 396[M + 1] 388[M + 1] 372 348[M + 1] 453 358[M + 1] 373 358[M + 1] 454 407[M + 1] 374 364[M + 1] 331 376 412[M + 1] 455 296[M + 1] 377 425[M + 1] 458 382[M + 1] 378 380[M + 3] 459 392[M + 1] 378[M + 1] 460 431[M + 1] 379 377[M + 1] 461 369[M + 1] 381 409[M + 1] 462 381[M + 3] 382 340[M + 1] 379[M + 1] 384 388[M + 1] 463 440[M + 3] 385 384[M + 1] 438[M + 1] 386 352[M + 1] 464 338[M + 1] 387 376[M + 1] 262 388 440[M + 1] 467 387[M + 1] 390 407[M + 1] 468 439[M + 1] 331 363 391 362[M + 1] 469 360[M + 1] 392 390[M + 1] 471 363[M + 3] 361[M + 1]
TABLE-US-00164 TABLE 164 472 378[M + 1] 555 383[M + 1] 473 414[M + 1] 557 304[M + 1] 474 334[M + 1] 562 374[M + 1] 475 317[M + 1] 563 366[M + 1] 476 324[M + 1] 564 395[M + 1] 477 437[M + 1] 565 336[M + 1] 478 379[M + 1] 566 427[M + 1] 479 394[M + 1] 351 480 370[M + 1] 568 362[M + 3] 481 431[M + 1] 360[M + 1] 484 314[M + 3] 569 356[M + 1] 312[M + 1] 571 356[M + 1] 485 448[M + 1] 572 473[M + 3] 486 350[M + 1] 471[M + 1] 487 338[M + 1] 574 381[M + 3] 488 306[M + 1] 379[M + 1] 489 335[M + 1] 575 360[M + 1] 492 380[M + 1] 576 384[M + 1] 495 334[M + 1] 578 344[M + 1] 499 370[M + 1] 579 370[M + 1] 503 412[M + 1] 580 347[M + 1] 505 363[M + 3] 581 409[M + 1] 361[M + 1] 582 334[M + 1] 506 386[M + 1] 583 392[M + 1] 507 400[M + 1] 585 358[M + 1] 508 372[M + 1] 587 348[M + 1] 509 414[M + 1] 589 407[M + 3] 338 405[M + 1] 510 374[M + 1] 590 410[M + 3] 512 320[M + 1] 408[M + 1] 513 420[M + 3] 591 460[M + 1] 418[M + 1] 384 514 372[M + 1] 592 380[M + 3] 517 369[M + 1] 378[M + 1] 518 376[M + 1] 594 390[M + 1] 519 411[M + 1] 598 394[M + 1] 520 395[M + 1] 599 377[M + 1] 521 372[M + 1] 603 398[M + 3] 522 390[M + 1] 396[M + 1] 523 414[M + 1] 604 395[M + 1] 524 341[M + 1] 606 358[M + 1] 526 426[M + 1] 607 362[M + 1] 527 381[M + 3] 609 413[M + 1] 379[M + 1] 610 409[M + 1] 529 320[M + 1] 612 385[M + 1] 530 390[M + 3] 614 322[M + 1] 388[M + 1] 615 441[M + 1] 531 410[M + 1] 616 346[M + 3] 535 356[M + 1] 344[M + 1] 536 372[M + 1] 270 537 377[M + 1] 268 538 406[M + 1] 617 406[M + 3] 539 411[M + 1] 404[M + 1] 540 354[M + 1] 619 404[M + 1] 541 342[M + 1] 621 366[M + 1] 542 361[M + 1] 623 422[M + 1] 543 344[M + 1] 346 544 412[M + 1] 624 370[M + 1] 545 366[M + 1] 626 402[M + 1] 546 383[M + 1] 627 398[M + 3] 547 430[M + 1] 396[M + 1] 428[M + 1] 628 413[M + 1] 548 427[M + 1] 631 370[M + 1] 550 340[M + 1] 632 414[M + 3] 552 400[M + 1] 412[M + 1] 553 304[M + 1]
TABLE-US-00165 TABLE 165 633 322[M + 1] 721 322[M + 1] 635 420[M + 1] 722 377[M + 1] 638 408[M + 1] 723 440[M + 1] 639 386[M + 1] 364 310 724 457[M + 3] 640 370[M + 1] 455[M + 1] 641 437[M + 1] 726 362[M + 1] 642 380[M + 1] 727 366[M + 1] 646 395[M + 1] 734 370[M + 1] 647 334[M + 1] 736 338[M + 1] 648 403[M + 1] 741 404[M + 1] 650 370[M + 1] 742 351[M + 1] 655 362[M + 1] 745 386[M + 1] 656 308[M + 1] 746 370[M + 1] 658 430[M + 1] 294 659 340[M + 3] 747 336[M + 1] 388[M + 1] 748 381[M + 3] 662 330[M + 1] 379[M + 1] 663 334[M + 1] 749 416[M + 1] 665 316[M + 1] 340 666 345[M + 1] 750 437[M + 1] 668 430[M + 1] 751 362[M + 1] 669 377[M + 1] 752 352[M + 3] 670 368[M + 3] 350[M + 1] 366[M + 1] 754 366[M + 1] 671 334[M + 1] 755 354[M + 1] 672 442[M + 1] 757 425[M + 1] 674 340[M + 1] 759 346[M + 1] 675 306[M + 1] 760 344[M + 1] 676 392[M + 1] 761 402[M + 1] 678 386[M + 1] 762 251[M + 1] 679 426[M + 1] 763 355[M + 1] 682 414[M + 3] 764 362[M + 3] 412[M + 1] 360[M + 1] 684 364[M + 1] 765 392[M + 3] 685 369[M + 1] 390[M + 1] 686 446[M + 1] 769 366[M + 1] 688 414[M + 1] 770 372[M + 1] 689 306[M + 1] 772 292[M + 1] 690 348[M + 1] 773 424[M + 1] 691 452[M + 1] 775 396[M + 3] 693 371[M + 1] 394[M + 1] 694 448[M + 1] 776 388[M + 1] 695 364[M + 1] 777 383[M + 1] 696 392[M + 3] 778 404[M + 1] 390[M + 1] 779 398[M + 1] 697 358[M + 1] 780 366[M + 1] 699 426[M + 1] 782 368[M + 1] 703 451[M + 3] 784 369[M + 1] 449[M + 1] 785 431[M + 3] 704 342[M + 1] 429[M + 1] 705 372[M + 1] 787 473[M + 1] 706 368[M + 1] 397 708 383[M + 1] 788 375[M + 1] 710 396[M + 3] 789 467[M + 1] 394[M + 1] 794 327[M + 1] 711 351[M + 1] 795 384[M + 1] 712 376[M + 1] 796 370[M + 1] 713 398[M + 3] 798 370[M + 1] 396[M + 1] 801 404[M + 3] 714 366[M + 1] 402[M + 1] 715 454[M + 1] 805 376[M + 1] 716 381[M + 3] 806 411[M + 1] 379[M + 1] 807 356[M + 1] 718 388[M + 1]
TABLE-US-00166 TABLE 166 808 354[M + 1] 889 451[M + 1] 809 400[M + 1] 449[M + 1] 324 890 400[M + 1] 812 425[M + 1] 881 292[M + 1] 815 386[M + 1] 894 347[M + 1] 817 377[M + 1] 412[M + 3] 818 398[M + 1] 898 410[M + 1] 819 352[M + 1] 898 397[M + 1] 821 336[M + 1] 901 411[M + 1] 823 362[M + 1] 902 377[M + 1] 824 363[M + 1] 903 370[M + 1] 287 904 422[M + 1] 825 420[M + 1] 905 392[M + 1] 826 430[M + 1] 907 308[M + 1] 828 377[M + 1] 909 393[M + 1] 829 437[M + 1] 911 415[M + 1] 830 370[M + 1] 912 383[M + 1] 831 327[M + 1] 913 413[M + 1] 837 324[M + 1] 914 400[M + 1] 248 915 389[M + 1] 838 377[M + 1] 313 839 376[M + 3] 917 358[M + 1] 374[M + 1] 918 433[M + 3] 841 363[M + 3] 431[M + 1] 361[M + 1] 919 354[M + 1] 842 386[M + 1] 920 381[M + 3] 843 466[M + 3] 379[M + 1] 464[M + 1] 921 389[M + 1] 844 381[M + 1] 922 413[M + 1] 845 324[M + 1] 337 248 923 437[M + 1] 846 358[M + 1] 924 376[M + 1] 847 373[M + 1] 925 390[M + 1] 852 489[M + 1] 927 355[M + 1] 853 376[M + 1] 929 370[M + 1] 854 448[M + 1] 932 380[M + 3] 856 420[M + 1] 378[M + 1] 344 934 507[M + 1] 857 341[M + 1] 937 388[M + 1] 858 383[M + 1] 938 366[M + 1] 860 370[M + 1] 940 388[M + 1] 861 334[M + 3] 942 378[M + 1] 332[M + 1] 943 413[M + 1] 862 358[M + 1] 945 372[M + 1] 864 392[M + 1] 948 462[M + 1] 865 398[M + 3] 949 363[M + 1] 396[M + 1] 950 368[M + 1] 867 399[M + 1] 951 412[M + 1] 868 430[M + 1] 952 378[M + 1] 870 362[M + 3] 953 318[M + 1] 360[M + 1] 954 363[M + 3] 872 428[M + 1] 361[M + 1] 873 351[M + 1] 955 406[M + 3] 874 341[M + 1] 404[M + 1] 877 399[M + 1] 956 292[M + 1] 323 957 398[M + 3] 879 332[M + 1] 396[M + 1] 880 363[M + 3] 958 310[M + 1] 361[M + 1] 959 406[M + 3] 882 426[M + 1] 404[M + 1] 883 360[M + 1] 961 382[M + 3] 884 320[M + 1] 360[M + 1] 885 361[M + 1] 962 327[M + 1] 886 380[M + 1] 963 392[M + 1] 888 292[M + 1]
TABLE-US-00167 TABLE 167 965 438[M + 3] 1036 350[M + 1] 436[M + 1] 1038 364[M + 1] 967 425[M + 3] 1040 317[M + 1] 423[M + 1] 1041 407[M + 1] 969 413[M + 1] 1042 382[M + 1] 973 386[M + 1] 1045 425[M + 3] 407[M + 3] 423[M + 1] 975 405[M + 1] 1046 366[M + 1] 976 358[M + 1] 1047 390[M + 1] 977 369[M + 1] 1048 440[M + 1] 979 395[M + 1] 1049 396[M + 1] 980 402[M + 1] 1050 400[M + 1] 981 392[M + 3] 1051 315[M + 1] 390[M + 1] 1053 363[M + 3] 982 366[M + 1] 381[M + 1] 983 379[M + 1] 1054 360[M + 1] 985 408[M + 1] 1057 427[M + 1] 986 440[M + 3] 1058 360[M + 1] 438[M + 1] 1060 381[M + 3] 987 356[M + 1] 379[M + 1] 988 294[M + 1] 1063 395[M + 1] 989 332[M + 1] 1065 451[M + 1] 991 358[M + 1] 449[M + 1] 992 477(M + 1] 1068 485[M + 1] 993 416[M + 3] 1070 380[M + 3] 414[M + 1] 378[M + 1] 995 425[M + 3] 1071 345[M + 1] 423[M + 1] 1072 381[M + 3] 996 416[M + 3] 379[M + 1] 414[M + 1] 1073 397[M + 1] 997 363[M + 3] 1075 342[M + 1] 361[M + 1] 1077 344[M + 1] 999 336[M + 1] 1078 370[M + 1] 1000 388[M + 1] 1079 387[M + 1] 312 1080 370[M + 1] 1001 374[M + 1] 294 1002 400[M + 1] 1081 355[M + 1] 1003 394[M + 1] 1082 398[M + 3] 1004 397[M + 1] 396[M + 1] 1007 448[M + 1] 1083 318[M + 1] 372 1085 439[M + 3] 1009 366[M + 1] 437[M + 1] 1011 419[M + 1] 1086 428[M + 1] 1012 316[M + 1] 1089 399[M + 1] 1013 431[M + 1] 1090 398[M + 1] 1015 372[M + 1] 1091 434[M + 3] 1016 470[M + 1] 432[M + 1] 1017 413[M + 1] 1092 398[M + 3] 1018 386[M + 1] 396[M + 1] 1019 433[M + 3] 1093 401[M + 1] 431[M + 1] 1095 400[M + 1] 1021 464[M + 1] 1096 409[M + 1] 1022 384[M + 1] 1097 384[M + 1] 1023 407[M + 3] 1098 395[M + 1] 405[M + 1] 1099 511[M + 4] 1024 346[M + 1] 510[M + 3] 1025 455[M + 3] 509[M + 2] 453[M + 1] 508[M + 1] 1026 425[M + 1] 1101 350[M + 1] 1027 444[M + 1] 1102 442[M + 1] 1029 410[M + 1] 1103 397[M + 1] 1030 413[M + 1] 1105 372[M + 1] 1031 404[M + 1] 1106 346[M + 1] 1032 472[M + 1] 1108 383[M + 1] 396 1112 445[M + 1] 1033 377[M + 1]
TABLE-US-00168 TABLE 168 1113 358[M + 1] 1187 427[M + 1] 1117 394[M + 1] 1188 350[M + 1] 1118 336[M + 1] 1189 408[M + 3] 260 406[M + 1] 1121 392[M + 3] 1190 386[M + 1] 390[M + 1] 1191 377[M + 1] 1122 322[M + 1] 1192 335[M + 1] 1123 316[M + 1] 412[M + 3] 1126 386[M + 1] 1195 410[M + 1] 1127 368[M + 1] 1196 380[M + 1] 1128 416[M + 3] 1198 398[M + 1] 414[M + 1] 322 1129 341[M + 1] 1200 352[M + 1] 1130 432[M + 1] 1201 424[M + 3] 1134 396[M + 1] 422[M + 1] 1136 396[M + 3] 1202 369[M + 1] 394[M + 1] 1203 420[M + 1] 1137 292[M + 1] 1204 398[M + 3] 1138 413[M + 1] 396[M + 1] 1141 344[M + 1] 1206 416[M + 1] 1143 384[M + 1] 1208 344[M + 1] 1144 446[M + 1] 1209 422[M + 1] 1145 390[M + 1] 1210 408[M + 1] 314 1212 391[M + 1] 1146 405[M + 1] 1214 360[M + 1] 1148 380[M + 1] 1218 372[M + 1] 304 1219 470[M + 1] 1149 364[M + 1] 1220 264[M + 1] 1151 442[M + 1] 1222 362[M + 3] 1152 365[M + 1] 360[M + 1] 1154 318[M + 1] 1225 413[M + 1] 1155 427[M + 1] 1226 374[M + 1] 1156 368[M + 1] 1227 425[M + 1] 1157 366[M + 1] 1229 455[M + 3] 1158 415[M + 3] 453[M + 1] 413[M + 1] 1231 413[M + 1] 1159 414[M + 3] 1232 340[M + 1] 412[M + 1] 1233 394[M + 1] 1162 370[M + 1] 1234 416[M + 3] 294 414[M + 1] 1163 416[M + 3] 1235 427[M + 1] 414[M + 1] 1236 348[M + 1] 1164 396[M + 1] 272 320 1237 353[M + 1] 1165 361[M + 1] 1238 419[M + 1] 1167 424[M + 1] 1239 416[M + 3] 348 414[M + 1] 1188 428[M + 1] 1246 474[M + 1] 1169 422[M + 1] 1248 414[M + 1] 1170 411[M + 1] 1249 338[M + 1] 1171 390[M + 3] 1250 352[M + 1] 388[M + 1] 1251 393[M + 1] 1173 361[M + 1] 1252 357[M + 1] 1174 342[M + 1] 1253 430[M + 1] 1175 431[M + 1] 1254 412[M + 1] 1176 345[M + 1] 1256 333[M + 1] 1177 376[M + 3] 1259 358[M + 1] 374[M + 1] 1280 348[M + 1] 1178 351[M + 1] 1270 374[M + 1] 1179 344[M + 1] 1282 362[M + 1] 1180 398[M + 3] 396[M + 1] 1182 426[M + 1] 376[M + 3] 1183 374[M + 1] 1186 374[M + 1] 298
##STR01356## ##STR01357##
[0298] In above structural formula (Ia) to (Ih), the combination of NR2aR2b, R3c, R3d, R5 and G (NR2aR2b, R3c, R3d, R5, G) are the following compounds. (NHMe,H,H,Me,CONHPh),(NHMe,H,H,Me,CONH-3-pyridyl),(NHMe,H,H,Me,NHCOPh),(N- HMe,H,H,Me,NHCO-2-furyl),(NHMe,H,H,Me,NHCONHPh),(NHMe,H,H, Me,NHCOCONHPh),(NHMe,H,H,Et,CONHPh),(NHMe,H,H,Et,CONH-3-pyridyl),(NHMe,H,- H,Et,NHCOPh),(NHMe,H,H,Et,NHCO-2-furyl,(NHMe,H,H,Et,NHCONHPh),(NHMe,H,H,Et- ,NHCOCONHPh),(NHMe,H,H,CH2OH,CONHPh),(NHMe,H,H,CH2OH,CONH-3-pyridyl),(NHMe- ,H,H,CH2OH,NHCOPh),(NHMe,H,H,CH2OH, NHCO-2-furyl),CH2OH,NHCONHPh),(NHMe,H,H,CH2OH,NHCOCONHPh),(NHMe,H,Me,Me,C- ONHPh),(NHMe,H,Me,Me,CONH-3-pyridyl),(NHMe,H, Me,Me,NHCOPh),(NHMe,H,Me,Me,NHCO-2-furyl),(NHMe,H,Me,Me,NHCONHPh),(NHMe,H- ,Me,Me,NHCOCONHPh),(NHMe,H,Me,Et,CONHPh),(NHMe,H,Me,Et,CONH-3-pyridyl),(NH- Me,H,Me,Et,NHCOPh),(NHMe,H,Me,Et,NHCO-2-furyl),(NHMe,H,Me,Et,NHCONHPh),(NH- Me,H,Me,Et,NHCOCONHPh),(NHMe,H,Me,CH2OH, CONHPh),(NHMe,H,Me,CH2OH,CONH-3-pyridyl),(NHMe,H,Me,CH2OH,NHCOPh),(NHMe,H- ,Me,CH2OH,NHCO-2-furyl),(NHMe,H,Me,CH2OH,NHCONHPh),(NHMe,H,Me,CH2OH,NHCOCO- NHPh),(NHMe,H,Ph,Me,CONHPh),(NHMe,H,Ph,Me, CONH-3-pyridyl),(NHMe,H,Ph,Me,NHCOPh),(NHMe,H,Ph,Me,NHCO-2-furyl),(NHMe,H- ,Ph,Me,NHCONHPh),(NHMe,H,Ph,Me,NHCOCONHPh),(NEHMe,H,Ph,Et,CONHPh),(NHMe,H,- Ph,Et,CONH-3-pyridyl),(NHMe,H,Ph,Et,NHCOPh),(NHMe,H,Ph,Et,NHCO-2-furyl),(N- HMe,H,Ph,Et,NHCONHPh),(NHMe,H,Ph,Et,NHCOCONHPh),(NHMe,H,Ph,CH2OH,CONHPh),(- NHMe,H,Ph,CH2OH,CONH-3-pyridyl),(NHMe,H,Ph,CH2OH,NHCOPh),(NHMe,H,Ph,CH2OH,- NHCO-2-furyl),(NHMe,H,Ph,CH2OH,NHCONHPh),(NHMe,H,Ph,CH2OH,NHCOCONHPh),(NHM- e,H,OH,Me,CONHPh),(NHMe,H,OH,Me,CONH-3-pyridyl),(NHMe,H,OH,Me,NHCOPh),(NHM- e,H, OH,Me,NHCO-2-furyl),(NHMe,H,OH,Me,NHCONHPh),(NHMe,H,OH,Me,NHCOCONHPh)- ,(NHMe,H,OH,Et,CONHPh),(NHMe,H,OH,Et,CONH-3-pyridyl),(NHMe,H, OH,Et,NHCOPh),(NHMe,H,OH,Et,NHCO-2-furyl),(NHMe,H,OH,Et,NHCONHPh), (NHMe,H,OH,Et,NHCOCONHPh),(NHMe,H,OH,CH2OH,CONHPh),(NHMe,H,OH,CH2OH,CONH-- 3-pyridyl),(NHMe,H,OH,CH2OH,NHCOPh),(NHMe,H,OH,CH2OH,NHCO-2-furyl),(NHMe,H- ,OH,CH2OH,NHCONHPh),(NHMe,H,OH,CH2OH,NHCOCONHPh),(NHMe,Me,H,Me,CONHPh),(NH- Me,Me,H,Me,CONH-3-pyridyl),(NHMe,Me,H,Me,NHCOPh),(NHMe,Me,H,Me,NHCO-2-fury- l),(NHMe,Me,H,Me,NHCONHPh),(NHMe,Me,H,Me,NHCOCONHPh),(NHMe,Me,H,Et,CONPh),- (NHMe,Me,H,Et,CONH-3-pyridyl),(NHMe,Me,H,Et,NHCOPh),(NHMe,Me,H,Et,NHCO-2-f- uryl),(NHMe,Me,H,Et,NHCONHPh),(NHMe,Me,H,Et,NHCOCONHPh),(NHMe,Me,H,CH2OH,C- ONHPh),(NHMe,Me,H,CH2OH,CONH-3-pyridyl),(NHMe,Me,H,CH2OH,NHCOPh),(NHMe,Me,- H,CH2OH,NHCO-2-furyl),(NHMe,Me,H,CH2OH,NHCONHPh),(NHMe,Me,H,CH2OH,NHCOCONH- Ph),(NHMe,Me,Me,Me,CONHPh),(NHMe,Me,Me,Me,CONH-3-pyridyl),(NHMe,Me,Me,Me,N- HCOPh),(NHMe,Me,Me,Me,NHCO-2-furyl),(NHMe,Me,Me,Me,NHCONHPh),(NHMe,Me,Me,M- e,NHCOCONHPh),(NHMe,Me,Me,Et,CONHPh),(NHMe,Me,Me,Et,CONH-3-pyridyl),(NHfMe- ,Me,Me,Et,NHCOPh),(NHMe,Me,Me,Et,NHCO-2-furyl),(NHMe,Me,Me,Et,NHCONHPh),(N- HMe, Me,Me,Et,NHCOCONHPh),(NHMe,Me,Me,CH2OH,CONHPh),(NHMe,Me;Me,CH2OH,CONH- -3-pyridyl),(NHMe,Me,Me,CH2OH,NHCOPh),(NHMe,Me,Me,CH2OH,NHCO-2-furyl),(NHM- e,Me,Me,CH2OH,NHCONHPh),(NHMe,Me,Me,CH2OH,NHCOCONHPh),(NHMe,Me,Ph,Me,CONHP- h),(NHMe,Me,Ph,Me,CONH-3-pyridyl),(NHMe,Me,Ph,Me,NHCOPh),(NHMe,Me,Ph,Me,NH- CO-2-furyl),(NHMe,Me,Ph,Me,NHCONHPh),(NHMe,Me,Ph,Me,NHCOCONHPh),(NHMe,Me,P- h,Et,CONHPh),(NHMe,Me,Ph,Et,CONH-3-pyridyl),(NHMe,Me,Ph,Et,NHCOPh),(NHMe,M- e,Ph,Et,NHCO-2-furyl),(NHMe,Me,Ph,Et,NHCONHPh),(NHMe,Me,Ph,Et,NHCOCONHPh),- (NHMe,Me,Ph,CH2OH,CONHPh),(NHMe,Me,Ph,CH2OH,CONH-3-pyridyl),(NHMe,Me,Ph,CH- 2OH,NHCOPh),(NHMe,Me,Ph,CH2OH,NHCO-2-furyl),(NHMe,Me,Ph,CH2OH,NHCONHPh),(N- HMe,Me,Ph,CH2OH,NHCOCONHPh),(NHMe,Me,OH,Me,CONHPh),(NHMe,Me,OH,Me,CONH-3-p- yridyl),(NHMe,Me,OH,Me,NHCOPh),(NHMe, Me,OH,Me,NHCO-2-furyl),(NHMe,Me,OH,Me,NHCONHPh),(NHMe,Me,OH,Me,NHCOCONHPh- ),(NHMe,Me,OH,Et,CONHPh),(NHMe,Me,OH,Et,CONH-3-pyridyl),(NHMe,Me,OH,Et,NHC- OPh),(NHMe,Me,OH,Et,NHCO-2-furyl),(NHMe,Me,OH,Et, NHCONHPh),(NHMe,Me,OH,Et,NHCOCONHPh),(NHMe,Me,OH,CH2OH,CONHPh),(NHMe,Me,O- H,CH2OH,CONH-3-pyridyl),(NHMe,Me,OH,CH2OH,NHCOPh),(NHMe,Me,OH,CH2OH,NHCO-2- -furyl),(NHMe,Me,OH,CH2OH,NHCONHPh),(NHMe,Me,OH,CH2OH,NHCOCONHPh),(NHMe,Ph- ,H,Me,CONHPh),(NHMe,Ph,H,Me,CONH-3-pyridyl),(NHMe,Ph,H,Me,NHCOPh),(NHMe,Ph- ,H,Me,NHCO-2-furyl,(NHMe,Ph,H,Me,NHCONHPh),(NHMe,Ph,H,Me,NHCOCONHPh),(NHMe- ,Ph,H,Et,CONHPh),(NHMe,Ph,H,Et,CONH-3-pyridyl),(NHMe,Ph,H,Et,NHCOPh),(NHMe- ,Ph,H,Et,NHCO-2-furyl),(NHMe,Ph,H,Et,NHCONHPh),(NHMe,Ph,H,Et,NHCOCONHPh),(- NHMe,Ph,H,CH2OH,CONHPh),(NHMe,Ph,H,CH2OH,CONH-3-pyridyl),(NHMe,Ph,H,CH2OH,- NHCOPh),(NHMe,Ph,H,CH2OH,NHCO-2-furyl),(NHMe,Ph,H,CH2OH,NHCONHPh),(NHMe,Ph- ,H,CH2OH,NHCOCONHPh),(NHRMe,Ph,Me,Me,CONHPh),(NHMe,Ph,Me,Me,CONH-3-pyridyl- ),(NHMe,Ph,Me,Me,NHCOPh),(NHMe,Ph,Me,Me,NHCO-2-furyl),(NHMe,Ph,Me,Me,NHCON- HPh),(NHMe,Ph,Me,Me,NHCOCONHPh),(NHMe,Ph,Me,Et,CONHPh),(NHMe,Ph,Me,Et,CONH- -3-pyridyl),(NHMe,Ph,Me,Et,NHCOPh),(NHMe,Ph,Me,Et,NHCO-2-furyl),(NHMe,Ph,M- e,Et,NHCONHPh),(NHMe,Ph,Me,Et,NHCOCONHPh),(NHMe,Ph,Me,CH2OH,CONHPh),(NHMe,- Ph,Me,CH2OH,CONH-3-pyridyl),(NHMe,Ph,Me,CH2OH,NHCOPh),(NHMe,Ph,Me,CH2OH,NH- CO-2-furyl),(NHMe,Ph,Me,CH2OH,NHCONHPh),(NHMe,Ph,Me,CH2OH,NHCOCONHPh),(NHM- e,Ph,Ph,Me,CONHPh),(NHMe,Ph,Ph,Me,CONH-3-pyridyl),(NHMe,Ph,Ph,Me,NHCOPh),(- NHMe,Ph,Ph,Me,NHCO2-furyl),(NHMe,Ph,Ph,Me,NHCONHPh),(NHMe,Ph,Ph,Me,NHCOCON- HPh),(NHMe,Ph,Ph,Et,CONHPh),(NHMe,Ph,Ph,Et,CONH-3-pyridyl),(NHMe,Ph,Ph,Et,- NHCOPh),(NHMe,Ph,Ph,Et, NHCO-2-furyl),(NHMe,Ph,Ph,Et,NHCONHPh),(NHMe,Ph,Ph,Et,NHCOCONHPh), (NHMe,Ph,Ph,CH2OH,CONHPh),(NHMe,Ph,Ph,CH2OH,CONH-3-pyridyl),(NH Me, Ph,Ph,CH2OH,NHCOPh),(NHMe,Ph,Ph,CH2OH,NHCO-2-furyl),(NHMe,Ph,Ph,CH2H,NHCO- NHPh),(NHMe,Ph,Ph,CH2OH,NHCOCONHPh),(NHMe,Ph,OH,Me,CONHPh),(NHMe,Ph,OH,Me,- CONH-3-pyridyl),(NHMe,Ph,OH,Me,NHCOPh),(NHMe, Ph,OH,Me,NHCO-2-furyl),(NHMe,Ph,OH,Me,NHCONHPh),(NHMe,Ph,OH,Me,NHCOCONHPh- ),(NHMe,Ph,OH,Et,CONHPh),(NHMe,Ph,OH,Et,CONH-3-pyridyl),(NHMe,Ph,OH,Et,NHC- OPh),(NHMe,Ph,OH,Et,NHCO-2-furyl),(NHMe,Ph,OH,Et,NHCONHPh),(NHMe,Ph,OH,Et,- NHCOCONHPh),(NHMe,Ph,OH,CH2OH,CONHPh),(NHMe,Ph,OH,CH2OH,CONH-3-pyridyl),(N- HMe,Ph,OH,CH2OH,NHCOPh),(NHMe,Ph,OH,CH2OH,NHCO-2-furyl),(NHMe,Ph,OH,CH2OH,- NHCONHPh),(NHMe,Ph,OH,CH2OH,NHCOCONHPh),
(NHCH2CH2OH,H,H,Me,CONHPh),(NHCH2CH2OH,H,H,Me,CONH-3-pyridyl),(NHCH2CH2OH- ,H,H,Me,NHCOPh),(NHCH2CH2OH,H,H,Me,NHCO-2-furyl),(NHCH2CH2OH,H,H,Me,NHCONP- h),(NHCH2CH2OH,H,H,Me,NHCOCONHPh),(NHCH2CH2OH,H,H,Et,CONHPh),(NHCH2CH2OH,H- ,H,Et,CONH-3-pyridyl),(NHCH2CH2OH,H,H,Et,NHCOPh),(NHCH2CH2OH,H,H,Et,NHCO-2- -furyl),(NHCH2CH2OH,H,H,Et,NHCONHPh),(NHCH2CH2OH,H,H,Et,NHCOCONHPh),(NHCH2- CH2OH,H,H,CH2OH,CONHPh),(NHCH2CH2OH,H,H,CH2OH,CONH-3-pyridyl),(NHCH2CH2OH,- H,H,CH2OH,NHCOPh),(NHCH2CH2OH,H,H,CH2OH,NHCO-2-furyl),(NHCH2CH2OH,H,H,CH2O- H,NHCONPh),(NHCH2CH2OH,H,H,CH2OH,NCOCONHPh),(NHCH2CH2OH,H,Me,Me,CONHPh),(N- HCH2CH2OH,H,Me,Me,CONH-3-pyridyl),(NHCH2CH2OH,H,Me,Me,NHCOPh),(NHCH2CH2OH,- H,Me,Me,NHCO-2-furyl),(NHCH2CH2OH,H,Me,Me,NHCONHPh),(NHCH2CH2OH,H,Me,Me,NH- COCONHPh),(NHCH2CH2OH,H,Me,Et,CONHPh),(NHCH2CH2OH,H,Me,Et,CONH-3-pyridyl),- (NHCH2CH2OH,H,Me,Et,NHCOPh),(NHCH2CH2OH,H,Me,Et,NHCO-2-furyl),(NHCH2CH2OH,- H,Me,Et,NHCONHPh),(NHCH2CH2OH,H,Me,Et,NHCOCONHPh),(NHCH2CH2OH,H,Me,CH2OH,C- ONHPh),(NHCH2CH2OH,H,Me,CH2OH,CONH-3-pyridyl),(NHCH2CH2OH,H,Me,CH2OH,NHCOP- h),(NHCH2CH2OH, H,Me,CH2OH,NHCO-2-furyl),(NHCH2CH2OH,H,Me,CH2OH,NHCONPh),(NHCH2CH2OH,H,Me- ,CH2OH,NHCOCONHPh),(NHCH2CH2OH,H,Ph,Me,CONHPh),(NHCH2CH2OH,H,Ph,Me,CONH-3-- pyridyl),(NHCH2CH2OH,H,Ph,Me,NHCOPh),(NHCH2CH2OH,H,Ph,Me,NHCO-2-furyl),(NH- CH2CH2OH,H,Ph,Me,NHCONHPh), (NHCH2CH2OH,H,Ph,Me,NHCOCONHPh),(NHCH2CH2OH,H,Ph,Et,CONHPh),(NHCH2CH2OH,H- ,Ph,Et,CONH-3-pyridyl),(NHCH2CH2OH,H,Ph,Et,NHCOPh),(NHCH2CH2OH,H,Ph,Et,NHC- O-2-furyl),(NHCH2CH2OH,H,Ph,Et,NHCONHPh),(NHCH2CH2OH,H,Ph,Et,NHCOCONHPh),(- NHCH2CH2OH,H,Ph,CH2OH,CONHPh),(NHCH2CH2OH,H,Ph,CH2OH,CONH-3-pyridyl),(NHCH- 2CH2OH,H,Ph,CH2OH, NHCOPh),(NHCH2CH2OH,H,Ph,CH2OH,NHCO-2-furyl),(NHCH2CH2OH,H,Ph,CH2OH,NHCON- HPh),(NHCH2CH2OH,H,Ph,CH2OH,NHCOCONHPh),(NHCH2CH2OH,H,OH,Me,CONHPh),(NHCH2- CH2OH,H,OH,Me,CONH-3-pyridyl),(NHCH2CH2OH,H,OH,Me,NHCOPh),(NHCH2CH2OH,H,OH- ,Me,NHCO-2-furyl),(NHCH2CH2OH,H,OH,Me,NHCONHPh),(NHCH2CH2OH,H,OH,Me,NHCOCO- NHPh),(NHCH2CH2OH,H,OH,Et,CONHPh),(NHCH2CH2OH,H,OH,Et,CONH-3-pyridyl),(NHC- H2CH2OH,H,OH,Et,NHCOPh),(NHCH2CH2OH,H,OH,Et,NHCO-2-furyl),(NHCH2CH2OH,H,OH- ,Et,NHCONHPh),(NHCH2CH2OH,H,OH,Et,NHCOCONHPh),(NHCH2CH2OH,H,OH,CH2OH,CONHP- h),(NHCH2CH2OH,H,OH,CH2OH,CONH-3-pyridyl),(NHCH2CH2OH,H,OH,CH2OH,NHCOPh),(- NHCH2CH2OH,H,OH,CH2OH,NHCO-2-fu yl),(NHCH2CH2OH,H,OH,CH2OH,NHCONHPh),(NHCH2CH2OH,H,OH,CH2OH,NHCOCONHPh),(- NHCH2CH2OH,Me,H,Me,CONHPh),(NHCH2CH2OH,Me,H,Me,CONH-3-pyridyl),(NHCH2CH2OH- ,Me,H,Me,NHCOPh),(NHCH2CH2OH,Me,H,Me,NHCO-2-furyl),(NHCH2CH2OH,Me,H,Me,NHC- ONHPh),(NHCH2CH2OH,Me,H,Me,NHCOCONHPh),(NHCH2CH2OH,Me,H,Et,CONHPh),(NHCH2C- H2OH,Me,H,Et,CONH-3-pyridyl),(NHCH2CH2OH,Me,H,Et,NHCOPh),(NHCH2CH2OH,Me,H,- Et,NHCO-2-furyl),(NHCH2CH2OH,Me,H,Et,NHCONHPh),(NHCH2CH2OH,Me,H,Et,NHCOCON- HPh),(NHCH2CH2OH,Me,H,CH2OH,CONHPh),(NHCH2CH2OH,Me,H,CH2OH,CONH-3-pyridyl)- ,(NHCH2CH2OH,Me,H,CH2OH,NHCOPh),(NHCH2CH2OH,Me,H,CH2OH,NHCO-2-furyl),(NHCH- 2CH2OH,Me,H,CH2OH,NHCONHPh),(NHCH2CH2OH,Me,H,CH2OH,NHCOCONHPh),(NHCH2CH2OH- ,Me,Me,Me,CONHPh),(NHCH2CH2OH,Me,Me,Me,CONH-3-pyridyl),(NHCH2CH2OH, Me,Me,Me,NHCOPh),(NHCH2CH2OH,Me,Me,Me,NHCO-2-furyl),(NHCH2CH2OH,Me,Me,Me,- NHCONHPh),(NHCH2CH2OH,Me,Me,Me,NHCOCONHPh),(NHCH2CH2OH,Me,Me,Et,CONHPh),(N- HCH2CH2OH,Me,Me,Et,CONH3-pyridyl),(NHCH2CH2OH,Me,Me,Et,NHCOPh),(NHCH2CH2OH- ,Me,Me,Et,NHCO-2-furyl),(NHCH2CH2OH,Me,Me,Et,NHCONHPh),(NHCH2CH2OH,Me,Me,E- t,NHCOCONHPh),(NHCH2CH2OH,Me,Me,CH2OH,CONHPh),(NHCH2CH2OH,Me,Me,CH2OH,CONH- -3-pyridyl),(NHCH2CH2OH,Me,Me,CH2OH,NHCOPh),(NHCH2CH2OH,Me,Me,CH2OH,NHCO-2- -furyl),(NHCH2CH2OH,Me,Me,CH2OH,NHCONHPh),(NHCH2CH2OH,Me,Me,CH2OH,NHCOCONH- Ph),(NHCH2CH2OH,Me,Ph,Me,CONHPh),(NHCH2CH2OH,Me,Ph,Me,CONH-3-pyridyl),(NHC- H2CH2OH,Me,Ph,Me,NHCOPh),(NHCH2CH2OH,Me,Ph,Me,NHCO-2-furyl),(NHCH2CH2OH,Me- ,Ph,Me,NHCONHPh),(NHCH2CH2OH,Me,Ph,Me,NHCOCONHPh),(NHCH2CH2OH,Me,Ph,Et,CON- HPh),(NHCH2CH2OH,Me,Ph,Et,CONH-3-pyridyl),(NHCH2CH2OH,Me,Ph,Et,NHCOPh),(NH- CH2CH2OH,Me,Ph,Et,NHCO-2-furyl),(NHCH2CH2OH,Me,Ph,Et,NHCONHPh),(NHCH2CH2OH- ,Me,Ph,Et,NHCOCONHPh),(NHCH2CH2OH,Me,Ph,CH2OH,CONHPh),(NHCH2CH2OH,Me,Ph,CH- 2OH,CONH-3-pyridyl,(NHCH2CH2OH,Me, Ph,CH2OH,NHCOPh),(NHCH2CH2OH,Me,Ph,CH2OH,NHCO-2-furyl),(NHCH2CH2OH,Me,Ph,- CH2OH,NHCONHPh),(NHCH2CH2OH,Me,Ph,CH2OH,NHCOCONHPh),(NHCH2CH2OH,Me,OH,Me,C- ONHPh),(NHCH2CH2OH,Me,OH,Me,CONH-3-pyridyl),(NHCH2CH2OH,Me,OH,Me,NHCOPh),(- NHCH2CH2OH,Me,OH,Me,NHCO-2-furyl,(NHCH2CH2OH,Me,OH,Me,NHCONHPh),(NHCH2CH2O- H,Me,OH,Me, NHCOCONHPh),(NHCH2CH2OH,Me,OH,Et,CONHPh),(NHCH2CH2OH,Me,OH, Et,CONH-3-pyridyl),(NHCH2CH2OH,Me,OH,Et,NHCOPh),(NHCH2CH2OH,Me,OH,Et,NHCO- -2-furyl),(NHCH2CH2OH,Me,OH,Et,NHCONHPh),(NHCH2CH2OH,Me,OH,Et,NHCOCONHPh),- (NHCH2CH2OH,Me,OH,CH2OH,CONHPh),(NHCH2CH2OH,Me,OH,CH2OH,CONH-3-pyridyl),(N- HCH2CH2OH,Me,OH,CH2OH,NHCOPh),(NHCH2CH2OH,Me,OH,CH2OH,NHCO-2-furyl,(NHCH2C- H2OH,Me,OH,CH2OH,NHCONHPh),(NHCH2CH2OH,Me,OH,CH2OH,NHCOCONHPh),(NHCH2CH2OH- ,Ph,H,Me,CONHPh),(NHCH2CH2OH,Ph,H,Me,CONH-3-pyridyl,(NHCH2CH2OH,Ph,H,Me,NH- COPh),(NHCH2CH2OH,Ph,H,Me,NHCO-2-furyl),(NHCH2CH2OH,Ph,H,Me,NHCONHPh),(NHC- H2CH2OH,Ph,H,Me,NHCOCONHPh),(NHCH2CH2OH,Ph,H,Et,CONHPh),(NHCH2CH2OH,Ph,H,E- t,CONH-3-pyridyl,(NHCH2CH2OH,Ph,H,Et,NHCOPh),(NHCH2CH2OH,Ph,H,Et,NHCO-2-fu- ryl,(NHCH2CH2OH, Ph,H,Et,NHCONHPh),(NHCH2CH2OH,Ph,H,Et,NHCOCONHPh),(NHCH2CH2OH,Ph,H,CH2OH,- CONHPh),(NHCH2CH2OH,Ph,H,CH2OH,CONH-3-pyridyl),(NHCH2CH2OH,Ph,H,CH2OH,NHCO- Ph),(NHCH2CH2OH,Ph,H,CH2OH,NHCO-2-furyl),(NH CH2CH2OH,Ph,H,CH2OH,NHCONHPh),(NHCH2CH2OH,Ph,H,CH2OH,NHCOCONHPh),(NHCH2CH- 2OH,Ph,Me,Me,CONHPh),(NHCH2CH2OH,Ph,Me,Me,CONH-3-pyridyl),(NHCH2CH2OH,Ph,M- e,Me,NHCOPh),(NHCH2CH2OH,Ph,Me,Me,NHCO-2-furyl),(NHCH2CH2OH,Ph,Me,Me,NHCON- HPh),(NHCH2CH2OH,Ph,Me, Me,NHCOCONHPh),(NHCH2CH2OH,Ph,Me,Et,CONHPh),(NHCH2CH2OH,Ph,Me,Et,CONH-3-p- yridyl),(NHCH2CH2OH,Ph,Me,Et,NHCOPh),(NHCH2CH2OH,Ph, Me,Et,NHCO-2-furyl),(NHCH2CH2OH,Ph,Me,Et,NHCONHPh),(NHCH2CH2OH,Ph,Me,Et,N- HCOCONHPh),(NHCH2CH2OH,Ph,Me,CH2OH,CONHPh),(NHCH2CH2OH,Ph,Me,CH2OH,CONH-3-- pyridyl,(NHCH2CH2OH,Ph,Me,CH2OH,NHCOPh), (NHCH2CH2OH,Ph,Me,CH2OH,NHCO-2-furyl),(NHCH2CH2OH,Ph,Me,CH2OH, NHCONHPh),(NHCH2CH2OH,Ph,Me,CH2OH,NHCOCONHPh),(NHCH2CH2OH, Ph,Ph,Me,CONHPh),(NHCH2CH2OH,Ph,Ph,Me,CONH-3-pyridyl),(NHCH2CH2OH,Ph,Ph,M- e,NHCOPh),(NHCH2CH2OH,Ph,Ph,Me,NHCO-2-furyl,(NHCH2CH2OH,Ph,Ph,Me,NHCONHPh)- ,(NHCH2CH2OH,Ph,Ph,Me,NHCOCONHPh),(NHCH2CH2OH,Ph,Ph,Et,CONHPh),(NHCH2CH2OH- ,Ph,Ph,Et,CONH-3-pyridyl),(NHCH2CH2OH,Ph,Ph,Et,NHCOPh),(NHCH2CH2OH,Ph,Ph,E- t,NHCO-2-furyl),(NHCH2CH2OH,Ph,Ph,Et,NHCONHPh),(NHCH2CH2OH,Ph,Ph,EtNHCOCON- HPh),(NHCH2CH2OH,Ph,Ph,CH2OH,CONHPh),(NHCH2CH2OH,Ph,Ph,CH2OH,CONH-3-pyridy- l),(NHCH2CH2OH,Ph,Ph,CH2OH,NHCOPh),(NHCH2CH2OH,Ph,Ph,CH2OH,NHCO-2-furyl),(- NHCH2CH2OH,Ph,Ph,CH2OH,NHCONHPh),(NHCH2CH2OH,Ph,Ph,CH2OH,NHCOCONHPh),(NHCH- 2CH2OH,Ph,OH,Me,CONHPh),(NHCH2CH2OH,Ph,OH,Me,CONH-3-pyridyl),(NHOH2CH2OH,P- h,OH,Me,NHCOPh),(NHCH2CH2OH,Ph,OH,Me,NHCO-2-furyl),(NHCH2CH2OH,Ph,OH,Me,NH- CONHPh),(NHCH2CH2OH,Ph,OH,Me,NHCOCONHPh),(NHCH2CH2OH,Ph,OH,Et,CONHPh),(NHC- H2CH2OH,Ph,OH,Et,CONH-3-pyridyl),(NHCH2CH2OH,Ph,OH,Et,NHCOPh),(NHCH2CH2OH,- Ph,OH,Et,NHCO-2-furyl),(NHCH2CH2OH,Ph,OH,Et,NHCONHPh),(NHCH2CH2OH,Ph,OH,Et- ,NHCOCONHPh),(NHCH2CH2OH,Ph,OH,CH2OH,CONHPh),(NHCH2CH2OH,Ph,OH,CH2OH,CONH-- 3-pyridyl),(NHCH2CH2OH,Ph,OH, CH2OH,NHCOPh),(NHCH2CH2OH,Ph,OH,CH2OH,NHCO-2-furyl),(NHCH2CH2OH,Ph,OH,CH2- OH,NHCONHPh),(NHCH2CH2OH,Ph,OH,CH2OH,NHCOCONHPh), (NHCH2CONH2,H,H,Me,CONHPh),(NHCH2CONH2,H,H,Me,CONH-3-pyridyl),(NHCH2CONH2- ,H,H,Me,NHCOPh),(NHCH2CONH2,H,H,Me,NHCO-2-furyl),(NHCH2CONH2,H,H,Me,NHCONH- Ph),(NHCH2CONH2,H,H,Me,NHCOCONHPh),(NHCH2CONH2,H,H,Et,CONHPh),(NHCH2CONH2,- H,H,Et,CONH-3-pyridyl),(NHCH2CONH2,H,H,Et,NCOPh),(NHCH2CONH2,H,H,Et,NHCO-2- -furyl),(NHCH2CONH2,H,H,Et,NHCONHPh),(NHCH2CONH2,H,H,Et,NHCOCONHPh),(NHCH2- CONH2,H,H,CH2OH,CONHPh),(NHCH2CONH2,H,H,CH2OH,CONH-3-pyridyl),(NHCH2CONH2,- H,H,CH2OH,NHCOPh),(NHCH2CONH2,H,H,CH2OH,NHCO-2-furyl),(NHCH2CONH2,H,H,CH2O- H,NHCONHPh),(NHCH2CONH2,H,H,CH2OH,NHCOCONHPh),(NHCH2CONH2,H,Me,Me,CONHPh),- (NHCH2CONH2,H,Me,Me,CONH-3-pyridyl),(NHCH2CONH2,H,Me,Me,NHCOPh),(NHCH2CONH- 2,H,Me,Me,NHCO-2-furyl),(NHCH2CONH2,H,Me,Me,NHCONHPh),(NHCH2CONH2,H,Me,Me,- NHCOCONHPh),(NHCH2CONH2,H,Me,Et,CONHPh),(NHCH2CONH2,H,Me,Et,CONH-3-pyridyl- ),(NHCH2CONH2,H,Me,Et,NHCOPh),(NHCH2CONH2,H,Me,Et,NHCO-2-furyl),(NHCH2CONH- 2,H,Me,Et,NHCONHPh),(NHCH2CONH2,H,Me,Et,NHCOCONHPh),(NHCH2CONH2,H,Me,CH2OH- ,CONHPh),(NHCH2CONH2,H,Me,CH2OH,CONH-3-pyridyl),(NHCH2CONH2,H,Me,CH2OH,NHC- OPh),(NHCH2CONH2, H,Me,CH2OH,NHCO-2-furyl),(NHCH2CONH2,H,Me,CH2OH,NHCONHPh),(NHCH2CONH2,H,M- e,CH2OH,NHCOCONHPh),(NHCH2CONH2,H,Ph,Me,CONHPh),(NHCH2CONH2,H,Ph,Me,CONH-3- -pyridyl),(NHCH2CONH2,H,Ph,Me,NHCOPh),(NHCH2CONH2,H,Ph,Me,NHCO-2-furyl),(N- HCH2CONH2,H,Ph,Me,NHCONHPh), (NHCH2CONH2,H,Ph,Me,NHCOCONHPh),(NHCH2CONH2,H,Ph,Et,CONHPh),(NHCH2CONH2,H- ,Ph,Et,CONH-3-pyridyl),(NHCH2CONH2,H,Ph,Et,NHCOPh),(NHCH2CONH2,H,Ph,Et,NHC- O-2-furyl),(NHCH2 CONH2,H,Ph,Et,NCONHPh),(NHCH2CONH2,H,Ph,Et,NHCOCONPh),(NHCH2CONH2,H,Ph,CH- 2OH,CONHPh),(NHCH2CONH2,H,Ph,CH2OH,CONH-3-pyridyl),(NHCH2CONH2,H,Ph,CH2OH, NHCOPh),(NHCH2CONH2,H,Ph,CH2OH,NHCO-2-furyl),(NHCH2CONH2,H,Ph,CH2OH,NHCON- HPh),(NHCH2CONH2,H,Ph,CH2OH,NHCOCONHPh),(NHCH2CONH2,H,OH,Me,CONHPh),(NHCH2- CONH2,H,OH,Me,CONH-3-pyridyl),(NHCH2CONH2,H,OH,Me,NHCOPh),(NHCH2CONH2,H,OH- ,Me,NHCO-2-furyl),(NHCH2CONH2,H,OH,Me,NHCONHPh),(NHCH2CONH2,H,OH,Me,NHCOCO- NHPh),(NHCH2CONH2,H,OH,Et,CONHPh),(NHCH2CONH2,H,OH,Et,CONH-3-pyridyl),(NHC- H2CONH2,H,OH,Et,NHCOPh),(NHCH2CONH2,H,OH,Et,NHCO-2-furyl),(NHCH2CONH2,H,OH- ,Et,NHCONHPh),(NHCH2CONH2,H,OH,Et,NHCOCONHPh),(NHCH2CONH2,H,OH,CH2OH,CONHP- h),(NHCH2CONH2,H,OH,CH2OH,CONH-3-pyridyl),(NHCH2CONH2,H,OH,CH2OH,NHCOPh),(- NHCH2CONH2,H,OH,CH2OH,NHCO-2-furyl),(NHCH2CONH2,H,OH,CH2OH,NHCONHPh),(NHCH- 2CONH2,H,OH, CH2OH,NHCO CONHPh),(NHCH2CONH2,Me,H,Me,CONHPh),(NHCH2 CONH2, Me,H,Me,CONH-3-pyridyl),(NHCH2CONH2,Me,H,Me,NHCOPh),(NHCH2CONH2, Me,H,Me,NHCO-2-furyl),(NHCH2CONH2,Me,H,Me,NHCONHPh),(NHCH2CONH2,Me,H,Me,N- HCOCONHPh),(NHCH2CONH2,Me,H,Et,CONHPh),(NHCH2CONH2,Me,H,Et,CONH-3-pyridyl)- ,(NHCH2CONH2,Me,H,Et,NHCOPh),(NHCH2CONH2,Me,H,Et,NHCO-2-furyl),(NHCH2CONH2- ,Me,H,Et,NHCONHPh),(NHCH2CONH2,Me,H,Et,NHCOCONHPh),(NHCH2CONH2,Me,H,CH2OH,- CONHPh),(NHCH2CONH2,Me,H,CH2OH,CONH-3-pyridyl),(NHCH2CONH2,Me,H,CH2OH,NHCO- Ph),(NHCH2CONH2,Me,H,CH2OH,NHCO-2-furyl),(NHCH2CONH2,Me,H,CH2OH,NHCONHPh),- (NHCH2CONH2,Me,H,CH2OH,NHCOCONHPh),(NHCH2CONH2,Me, Me,Me,CONHPh),(NHCH2CONH2,Me,Me,Me,CONH-3-pyridyl),(NHCH2CONH2, Me,Me,Me,NHCOPh),(NHCH2CONH2,Me,Me,Me,NHCO-2-furyl),(NHCH2CONH2,Me,Me,Me,- NHCONHPh),(NHCH2CONH2,Me,Me,Me,NHCOCONHPh),(NHCH2CONH2,Me,Me,Et,CONHPh),(N- HCH2CONH2,Me,Me,Et,CONH-3-pyridyl),(NHCH2CONH2,Me,Me,Et,NHCOPh),(NHCH2CONH- 2,Me,Me,Et,NHCO-2-furyl),(NHCH2CONH2,Me,Me,Et,NHCONHPh),(NHCH2CONH2,Me,Me,- Et,NHCOCONHPh),(NHCH2CONH2,Me,Me,CH2OH,CONHPh),(NHCH2 CONH2,Me,Me,CH2OH,CONH-3-pyridyl),(NHCH2CONH2,Me,Me,CH2OH,NHCOPh),(NHCH2C- ONH2,Me,Me,CH2OH,NHCO-2-furyl),(NHCH2CONH2,Me,Me,CH2OH,NHCONHPh),(NHCH2CON- H2,Me,Me,CH2OH,NHCOCONHPh),(NHCH2CONH2,Me,Ph,Me,CONHPh),(NHCH2CONH2,Me,Ph,- Me,CONH-3-pyridyl),(NHCH2CONH2,Me,Ph,Me,NHCOPh),(NHCH2CONH2,Me,Ph,Me,NHCO-- 2-furyl),(NHCH2CONH2,Me,Ph,Me,NHCONHPh),(NHCH2CONH2,Me,Ph,Me,NHCOCONHPh),(- NHCH2CONH2,Me,Ph,Et,CONHPh),(NHCH2CONH2,Me,Ph,Et,CONH-3-pyridyl),(NHCH2CON- H2,Me,Ph,Et,NRCOPh), (NHCH2CONH2,Me,Ph,Et,NHCO-2-furyl),(NHCH2CONH2,Me,Ph,Et,NHCONHPh),(NHCH2C- ONH2,Me,Ph,Et,NHCOCONHPh),(NHCH2CONH2,Me,Ph,CH2OH,CONHPh),(NHCH2CONH2,Me,P- h,CH2OH,CONH-3-pyridyl),(NHCH2CONH2,Me,Ph,CH2OH,NHCOPh),(NHCH2CONH2,Me,Ph,- CH2OH,NHCO-2-furyl),(NHCH2CONH2,Me,Ph,CH2OH,NHCONHPh),(NHCH2CONH2,Me,Ph,CH- 2OH,NHCOCONHPh),(NHCH2CONH2,Me,OH,Me,CONHPh),(NHCH2CONH2,Me,OH,Me,CONH-3-p- yridyl),(NHCH2CONH2,Me,OH,Me,NHCOPh),(NHCH2CONH2,Me,OH,Me,NHCO-2-furyl),(N- HCH2CONH2,Me,OH,Me,NHCONHPh),(NHCH2CONH2,Me,OH,Me, NHCO CONHPh),(NHCH2CONH2,Me,OH,Et,CONHPh),(NHCH2CONH2,Me,OH, Et,CONH-3-pyridyl),(NHCH2CONH2,Me,OH,Et,NCOPh),(NHCH2CONH2,Me,OH,Et,NHCO-- 2-furyl),(NHCH2CONH2,Me,OH,Et,NHCONPh),(NHCH2CONH2,Me,OH,Et,NHCOCONHPh),(N- HCH2 CONH2,Me,OH,CH2OH,CONHPh),(NHCH2CONH2,Me,OH,CH2OH,CONH-3-pyridyl),(NH- CH2CONH2,Me,OH,CH2OH,NHCOPh),(NHCH2 CONH2,Me,OH,CH2OH,NHCO-2-furyl),(NHCH2CONH2,Me,OH,CH2OH,NHCONHPh),(NHCH2C- ONH2,Me,OH,CH2OH,NHCO CONHPh),(NHCH2 CONH2,Ph,H,Me,CONHPh),(NHCH2CONH2,Ph,H,Me,CONH-3-pyridyl),(NHCH2CONH2,Ph,- H,Me,NHCOPh),(NHCH2CONH2,Ph,H,Me,NHCO-2-furyl),(NHCH2CONH2,Ph,H,Me,NHCONHP- h),(NHCH2CONH2,Ph,H,Me,NHCOCONHPh),(NHCH2CONH2,Ph,H,Et,CONHPh),(NHCH2CONH2- ,Ph,H,Et,CONH-3-pyridyl),(NHCH2CONH2,Ph,H,Et,NHCOPh),(NHCH2CONH2,Ph,H,Et,N- HCO-2-furyl),(NHCH2CONH2,Ph,H,Et,NHCONHPh),(NHCH2CONH2,Ph,H,Et,NHCOCONHPh)- ,(NHCH2CONH2,Ph,H,CH2OH,CONHPh),(NHCH2OCONH2,Ph,H,CH2OH,CONH-3-pyridyl),(N- HCH2CONH2,Ph,H,CH2OH,NHCOPh),(NHCH2CONH2,Ph,H,CH2OH,NHCO-2-furyl),(NHCH2CO- NH2,Ph,H,CH2OH,NHCONHPh),(NHCH2CONH2,Ph,H,CH2OH,NHCOCONHPh),(NHCH2CONH2,Ph- ,Me,Me,CONHPh),(NHCH2CONH2,Ph,Me,Me,CONH-3-pyridyl),(NHCH2CONH2,Ph,Me,Me,N- HCOPh),(NHCH2CONH2,Ph,Me,Me, NHCO-2-furyl),(NHCH2CONH2,Ph,Me,Me,NHCONHPh),(NHCH2CONH2,Ph,Me, Me,NHCOCONHPh),(NHCH2CONH2,Ph,Me,Et,CONHPh),(NHCH2CONH2,Ph,Me,Et,CONH-3-p- yridyl),(NHCH2CONH2,Ph,Me,Et,NHCOPh),(NHCH2CONH2,Ph, Me,Et,NHCO-2-fu yl),(NHCH2CONH2,Ph,Me,Et,NHCONHPh),(NHCH2CONH2,Ph,Me,Et,NHCOCONHPh),(NHCH- 2 CONH2,Ph,Me,CH2OH,CONHPh),(NHCH2 CONH2,Ph,Me,CH2OH,CONH-3-pyridyl),(NHCH2OCONH2,Ph,Me,CH2OH,NHCOPh), (NHCH2CONH2,Ph,Me,CH2OH,NHCO-2-furyl),(NHCH2CONH2,Ph,Me,CH2OH, NHCONHPh),(NHCH2CONH2,Ph,Me,CH2OH,NHCOCONHPh),(NHCH2CONH2, Ph,Ph,Me,CONHPh),(NHCH2CONH2,Ph,Ph,Me,CONH-3-pyridyl),(NHCH2CONH 2,Ph,Ph,Me,NHCOPh),(NHCH2CONH2,Ph,Ph,Me,NHCO-2-furyl),(NHCH2CONH2,Ph,Ph,M- e,NHCONHPh),(NHCH2CONH2,Ph,Ph,Me,NHCOCONHPh),(NHCH2CONH2,Ph,Ph,Et,CONHPh),- (NHCH2CONH2,Ph,Ph,Et,CONH-3-pyridyl,(NHCH2CONH2,Ph,Ph,Et,NHCOPh),(NHCH2CON- H2,Ph,Ph,Et,NHCO-2-furyl),(NHCH2CONH2,Ph,Ph,Et,NHCONHPh),(NHCH2CONH2,Ph,Ph- ,Et,NHCOCONHPh),(NHCH2CONH2,Ph,Ph,CH2OH,CONHPh),(NHCH2CONH2,Ph,Ph,CH2OH,CO- NH-3-pyridyl)(NHCH2CONH2,Ph,Ph,CH2OH,NHCOPh),(NHCH2CONH2,Ph,Ph,CH2OH,NHCO-- 2-furyl),(NH CH2CONH2,Ph,Ph,CH2OH,NHCONHPh),(NHCH2OCONH2,Ph,Ph,CH2OH,NHCOCONHPh),(NHCH- 2CONH2,Ph,OH,Me,CONHPh),(NHCH2CONH2,Ph,OH,Me,CONH3-pyridyl),(NHCH2 CONH2,Ph,OH,Me,NHCOPh),(NHCH2CONH2,Ph,OH,Me,NHCO2-furyl),(NHCH2CONH2,Ph,O- H,Me,NHCONHPh),(NHCH2CONH2,Ph,OH,Me,NHCOCONHPh),(NHCH2CONH2,Ph,OH,Et,CONHP- h),(NHCH2CONH2,Ph,OH,Et,CONH-3-pyridyl),(NHCH2CONH2,Ph,OH,Et,NHCOPh),(NHCH- 2CONH2,Ph,OH,Et,HCO-2-furyl),(NHCH2CONH2,Ph,OH,Et,NHCONHPh),(NHCH2CONH2,Ph- ,OH,Et,NHCOCONHPh),(NHCH2CONH2,Ph,OH,CH2OH,CONHPh),(NHCH2CONH2,Ph,OH,CH2OH- ,CONH-3-pyridyl),(NHCH2CONH2,Ph,OH,CH2OH,NHCOPh),(NHCH2CONH2,Ph,OH,CH2OH,N- HCO-2-furyl),(NHCH2CONH2,Ph,OH,CH2OH,NHCONHPh),(NHCH2 CONH2,Ph,OH,CH2OH,NHCO CONHPh), (NHCH(Bn)CONH2,H,H,Me,CONHPh),(NHCH(Bn)CONH2,H,H,Me,CONH-3-pyridyl),(NHCH- (Bn)CONH2,H,H,Me,NBCOPh),(NHCH(Bn)CONH2,H,H,Me,NHCO-2-furyl),(NHCH(Bn)CONH- 2,H,H,Me,NHCONHPh),(NHCH(Bn)CONH2,H,H,Me,NHCOCONHPh),(NHCH(Bn)CONH2,H,H,Et- ,CONHPh),(NHCH(Bn)CONH2,H,H,Et,CONH-3-pyridyl),(NHCH(Bn)CONH2,H,H,Et,NHCOP- h),(NHCH(Bn)CONH2,H,H,Et,NHCO-2-furyl),(NHCH(Bn)CONH2,H,H,Et,NHCONHPh),(NH- CH(Bn)CONH2,H, H,Et,NHCOCONHPh),(NHCH(Bn)CONH2,H,H,CH2OH,CONHPh),(NHCH(Bn)CONH2,H,H,CH2O- H,CONH-3-pyridyl),(NHCH(Bn)CONH2,H,H,CH2OH,NHCOPh),(NHCH(Bn)CONH2,H,H,CH2O- H,NHCO-2-furyl),(NHCH(Bn)CONH2,H,H,CH2OH, NHCONHPh),(NHCH(Bn)CONH2,H,H,CH2OH,NHCOCONHPh),(NHCH(Bn)CONH2,H,Me,Me,CON- HPh),(NHCH(Bn)CONH2,H,Me,Me,CONH-3-pyridyl),(NHCH(Bn)CONH2,H,Me,Me,NHCOPh)- ,(NHCH(Bn)CONH2,H,Me,Me,NHCO-2-furyl),(NHCH(Bn)CONH2,H,Me,Me,NHCONHPh),(NH- CH(Bn)CONH2,H,Me,Me,NHCOCONHPh),(NHCH(Bn)CONH2,H,Me,Et,CONHPh),(NHCH(Bn)CO- NH2,H,Me,Et,CONH-3-pyridyl),(NHCH(Bn)CONH2,H,Me,Et,NHCOPh),(NHCH(Bn)CONH2,- H,Me,Et,NHCO-2-furyl),(NHCH(Bn)CONH2,H,Me,Et,NHCONHPh),(NHCH(Bn)CONH2,H,Me- , Et,NHCOCONHPh),(NHCH(Bn)CONH2,H,Me,CH2OH,CONHPh),(NHCH(Bn)CONH2,H,Me,CH2- OH,CONH-3-pyridyl),(NHCH(Bn)CONH2,H,Me,CH2OH,NHCOPh),(NHCH(Bn)CONH2,H,Me,C- H2OH,NHCO-2-furyl),(NHCH(Bn)CONH2,H,Me,CH2OH,NHCONHPh),(NHCH(Bn)CONH2,H,Me- ,CH2OH,NHCOCONHPh),(NHCH(Bn)CONH2,H,Ph,Me,CONHPh),(NHCH(Bn)CONH2,H,Ph,Me,C- ONH-3-pyridyl),(NHCH(Bn)CONH2,H,Ph,Me,NHCOPh),(NHCH(Bn)CONH2,H,Ph,Me,NHCO-- 2-furyl),(NHCH(Bn)CONH2,H,Ph,Me,NHCONHPh),(NHCH(Bn)CONH2,H,Ph,Me,NHCONHPh)- ,(NHCH(Bn)CONH2,H,Ph,Et,CONHPh),(NHCH(Bn)CONH2,H,Ph,Et,CONH-3-pyridyl),(NC- H(Bn)CONH2,H,Ph,Et,NBCOPh),(NHCH(Bn)CONH2,H,Ph,Et, NHCO-2-furyl),(NHCH(Bn)CONH2,H,Ph,Et,NBCONHPh),(NHCH(Bn)CONH2,H, Ph,Et,NBCOCONHPh),(NHCH(Bn)CONH2,H,Ph,CH2OH,CONHPh),(NHCH(Bn)CONH2,H,Ph,C- H120,CONH-3-pyridyl),(NHCH(Bn)CONH2,H,Ph,CH2OH,NHCONHPh),(NHCH(Bn)CONH2,H,- Ph,CH2OH,NHCO-2-furyl),(NHCH(Bn)CONH2,H,Ph,CH2OH,NBCONHPh),(NHCH(Bn)CONH2,- H,Ph,CH2OH,NHCOCONHPh),(NHCH(Bn)CONH2,H,OH,Me,CONHPh),(NHCH(Bn)CONH2,H,OH,- Me,CONH-3-pyridyl),(NHCH(Bn)CONH2,H,OH,Me,NHCOPh),(NHCH(Bn)CONH2,H,OH,Me,N- HCO2-furyl),(NHCH(Bn)CONH2,H,OH,Me,NHCONHPh),(NHCH(Bn)CONH2,H,OH,Me,NHCOCO- NHPh),(NHCH(Bn)CONH2,H,OH,Et,CONHPh),(NHCH(Bn)CONH2,H,OH, Et,CONH-3-pyridyl),(NHCH(Bn)CONH2,H,OH,Et,NHCOPh),(NHCH(Bn)CONH2, H,OH,Et,NHCO-2-furyl),(NHCH(Bn)CONH2,H,OH,Et,NHCONHPh),(NHCH(Bn)CONH2,H,O- H,Et,NHCOCONHPh),(NHCH(Bn)CONH2,H,OH,CH2OH,CONHPh),(NHCH(Bn)CONH2,H,OH,CH2- OH,CONH-3-pyridyl),(NHCH(Bn)CONH2,H,OH,CH2OH,NHCOPh),(NHCH(Bn)CONH2,H,OH,C- H2OH,NHCO-2-furyl),(NHCH(Bn)CONH2,H,OH,CH2OH,NHCONHPh),(NHCH(Bn)CONH2,H,OH- ,CH2OH,NHCOCONHPh),(NHCH(Bn)CONH2,Me,H,Me,CONHPh),(NHCH(Bn)CONH2,Me,H,Me,C- ONH-3-pyridyl),(NHCH(Bn)CONH2,Me,H,Me,NHCOPh),(NHCH(Bn)CONH2,Me,H,Me, NHCO-2-furyl),(NHCH(Bn)CONH2,Me,H,Me,NHCONHPb),(NHCH(Bn)CONH2,Me,H,Me,NHC- OCONHPh),(NHCH(Bn)CONH2,Me,H,Et,CONHPh),(NHCH(Bn)CONH2,Me,H,Et,CONH-3-pyri- dyl),(NHCH(Bn)CONH2,Me,H,Et,NHCOPh),(NHCH(Bn)CONH2,Me,H,Et,NHCO-2-furyl),(- NHCH(Bn)CONH2,Me,H,Et,NHCONHPh),(NHCH(Bn)CONH2,Me,H,Et,NHCOCONHPh),(NHCH(B- n)CONH2,Me,H,CH2OH,CONHPh),(NHCH(Bn)CONH2,Me,H,CH2OH,CONH-3-pyridyl),(NHCH-
(Bn)CONH2,Me, H,CH2OH,NHCOPh),(NHCH(Bn)CONH2,Me,H,CH2OH,NHCO-2-furyl),(NHCH(Bn)CONH2,Me- ,H,CH2OH,NHCONHPh),(NHCH(Bn)CONH2,Me,H,CH2OH,NHCO CONHPh),(NHCH(Bn)CONH2,Me,Me,Me,CONHPh),(NHCH(Bn)CONH2,Me,Me, Me,CONH-3-pyridyl),(NHCH(Bn)CONH2,Me,Me,Me,NHCOPh),(NHCH(Bn)CONH2,Me,Me,M- e,NHCO-2-furyl),(NHCH(Bn)CONH2,Me,Me,Me,NHCONHPh),(NHCH(Bn)CONH2,Me,Me,Me,- NHCO CONHPh),(NHCH(Bn)CONH2,Me,Me,Et,CONHPh), (NHCH(Bn)CONH2,Me,Me,Et,CONH-3-pyridyl),(NHCH(Bn)CONH2,Me,Me,Et,NHCOPh),(- NHCH(Bn)CONH2,Me,Me,Et,NHCO-2-furyl),(NHCH(Bn)CONH2,Me,Me,Et,NHCONHPh),(NH- CH(Bn)CONH2,Me,Me,Et,NHCOCONHPh),(NHCH(Bn)CONH2,Me,Me,CH2OH,CONHPh),(NHCH(- Bn)CONH2,Me,Me,CH2OH,CONH-3-pyridyl), (NHCH(Bn)CONH2,Me,Me,CH2OH,NHCOPh),(NHCH(Bn)CONH2,Me,Me,CH2OH,NHCO-2-fury- l),(NHCH(Bn)CONH2,Me,Me,CH2OH,NHCONHPh),(NHCH(Bn)CONH2,Me,Me,CH2OH,NHCOCON- HPh),(NHCH(Bn)CONH2,Me,Ph,Me,CONHPh), (NHCH(Bn)CONH2,Me,Ph,Me,CONH-3-pyridyl),(NHCH(Bn)CONH2,Me,Ph,Me,NHCOPh),(- NHCH(Bn)CONH2,Me,Ph,Me,NHCO2-furyl),(NHCH(Bn)CONH2,Me,Ph,Me,NHCONHPh),(NHC- H(Bn)CONH2,Me,Ph,Me,NHCOCONHPh),(NHCH(Bn)CONH12,Me,Ph,Et,CONHPh),(NHCH(Bn)- CONH2,Me,Ph,Et,CONH-3-pyridyl),(NHCH(Bn)CONH2,Me,Ph,Et,NHCOPh),(NHCH(Bn)CO- NH2,Me,Ph,Et,NHCO-2-furyl),(NHCH(Bn)CONH2,Me,Ph,Et,NHCONHPh),(NHCH(Bn)CONH- 2,Me,Ph,Et,NHCOCONHPh),(NHCH(Bn)CONH2,Me,Ph,CH2OH,CONHPh),(NHCH(Bn)CONH2,M- e,Ph, CH2OH,CONH-3-pyridyl),(NHCH(Bn)CONH2,Me,Ph,CH2OH,NHCOPh),(NHCH(Bn)CO- NH2,Me,Ph,CH2OH,NHCO-furyl),(NHCH(Bn)CONH2,Me,Ph,CH2OH,NHCONHPh),(NHCH(Bn)- CONH2,Me,Ph,CH2OH,NHCOCONHPh),(NHCH(Bn)CONH2,Me,OH,Me,CONHPh),(NHCH(Bn)CON- H2,Me,OH,Me,CONH-3-pyridyl),(NHCH(Bn)CONH2,Me,OH,Me,NHCOPh),(NHCH(Bn)CONH2- ,Me,OH,Me,NHCO2-furyl),(NHCH(Bn)CONH2,Me,OH,Me,NHCONHPh),(NHCH(Bn)CONH2,Me- ,OH,Me,NHCOCONHPh),(NHCH(Bn)CONH2,Me,OH,Et,CONHPh),(NHCH(Bn)CONH2,Me,OH,Et- ,CONH-3-pyridyl),(NHCH(Bn)CONH2,Me,OH,Et,NHCOPh),(NHCH(Bn)CONH2,Me,OH,Et,N- HCO-2-furyl),(NHCH(Bn)CONH2,Me,OH,Et,NHCONHPh),(NHCH(Bn)CONH2,Me,OH,Et,NHC- OCONHPh),(NHCH(Bn)CONH2,Me,OH,CH2OH,CONHPh),(NHCH(Bn)CONH2,Me,OH,CH2OH,CON- H-3-pyridyl),(NHCH(Bn)CONH2,Me,OH,CH2OH,NHCOPh),(NHCH(Bn)CONH2,Me,OH,CH2OH- ,NHCO-2-furyl),(NHCH(Bn)CONH2,Me,OH,CH2OH,NHCONHPh),(NHCH(Bn)CONH2,Me,OH,C- H2OH,NHCO CONHPh),(NHCH(Bn)CONH2,Ph,H,Me,CONHPh),(NHCH(Bn)CONH2,Ph,H,Me,CO- NH-3-pyridyl),(NHCH(Bn)CONH2,Ph,H,Me,NHCOPh),(NHCH(Bn)CONH2,Ph,H,Me,NHCO-2- -furyl),(NHCH(Bn)CONH2,Ph,H,Me,NHCONHPh),(NHCH(Bn)CONH2,Ph,H,Me,NHCOCONHPh- ),(NHCH(Bn)CONH2,Ph,H,Et,CONHPh),(NHCH(Bn)CONH2,Ph,H,Et,CONH-3-pyridyl),(N- HCH(Bn)CONH2,Ph,H,Et,NHCOPh),(NHCH(Bn)CONH2,Ph,H,Et,NHCO-2-furyl),(NHCH(Bn- )CONH2,Ph,H,Et,NHCONHPh),(NHCH(Bn)CONH2,Ph,H,Et,NHCOCONHPh),(NHCH(Bn)CONH2- ,Ph,H, CH2OH,CONHPh),(NHCH(Bn)CONH2,Ph,H,CH2OH,CONH-3-pyridyl),(NHCH(Bn)CO- NH2,Ph,H,CH2OH,NHCOPh),(NHCH(Bn)CONH2,Ph,H,CH2OH,NHCO-2-furyl),(NHCH(Bn)CO- NH2,Ph,H,CH2OH,NHCONHPh),(NHCH(Bn)CONH2,Ph,H,CH2OH,NHCOCONHPh),(NHCH(Bn)CO- NH2,Ph,Me,Me,CONHPh),(NHCH(Bn)CONH2,Ph,Me,Me,CONH-3-pyridyl),(NHCH(Bn)CONH- 2,Ph,Me,Me,NHCOPh),(NHCH(Bn)CONH2,Ph,Me,Me,NHCO-2-furyl),(NHCH(Bn)CONH2,Ph- ,Me,Me,NHCONHPh), (NHCH(Bn)CONH2,Ph,Me,Me,NHCOCONHPh),(NHCH(Bn)CONH2,Ph,Me,Et,CONHPh),(NHCH- (Bn)CONH2,Ph,Me,Et,CONH-3-pyridyl),(NHCH(Bn)CONH2,Ph,Me,Et,NHCOPh),(NHCH(B- n)CONH2,Ph,Me,Et,NHCO-2-furyl)(NHCH(Bn)CONH2,Ph,Me,Et,NHCONHPh),(NHCH(Bn)C- ONH2,Ph,Me,Et,NHCO CONHPh),(NHCH(Bn)CONH2,Ph,Me,CH2OH,CONHPh),(NHCH(Bn)CONH2,Ph,Me,CH2OH,CON- H-3-pyridyl),(NHCH(Bn)CONH2,Ph,Me,CH2OH,NHCOPh),(NHCH(Bn)CONH2,Ph,Me,CH2OH- ,NHCO-2-furyl),(NHCH(Bn)CONH2,Ph,Me,CH2OH,NHCONHPh),(NHCH(Bn)CONH2,Ph,Me,C- H2OH,NHCOCONHPh),(NHCH(Bn)CONH2,Ph,Ph,Me,CONHPh),(NHCH(Bn)CONH2,Ph,Ph,Me,C- ONH-3-pyridyl),(NHCH(Bn)CONH2,Ph,Ph,Me, NHCOPh),(NHCH(Bn)CONH2,Ph,Ph,Me,NHCO-2-furyl),(NHCH(Bn)CONH2,Ph,Ph,Me,NHC- ONHPh),(NHCH(Bn)CONH2,Ph,Ph,Me,NHCOCONHPh),(NHCH(Bn)CONH2,Ph,Ph,Et,CONHPh)- ,(NHCH(Bn)CONH2,Ph,Ph,Et,CONH-3-pyridyl),(NHCH(Bn)CONH2,Ph,Ph,Et,NHCOPh),(- NHCH(Bn)CONH2,Ph,Ph,Et,NHCO-2-furyl),(NHCH(Bn)CONH2,Ph,Ph,Et,NHCONHPh),(NH- CH(Bn)CONH2,Ph,Ph,Et,NHCOCONHPh),(NHCH(Bn)CONH2,Ph,Ph,CH2OH,CONHPh),(NHCH(- Bn)CONH2,Ph,Ph,CH2OH,CONH-3-pyridyl),(NHCH(Bn)CONH2,Ph,Ph,CH2OH,NHCOPh),(N- HCH(Bn)CONH2,Ph,Ph,CH2OH,NHCO-2-furyl),(NHCH(Bn)CONH2,Ph,Ph,CH2OH,NHCONHPh- ),(NHCH(Bn)CONH2,Ph,Ph,CH2OH,NHCOCONHPh),(NHCH(Bn)CONH2,Ph,OH,Me,CONHPh),(- NHCH(Bn)CONH2,Ph,OH,Me,CONH-3-pyridyl),(NHCH(Bn)CONH2,Ph,OH,Me,NHCOPh),(NH- CH(Bn)CONH2,Ph,OH,Me,NHCO-2-furyl),(NHCH(Bn)CONH2,Ph,OH,Me,NHCONHPh),(NHCH- (Bn)CONH2,Ph,OH,Me,NHCOCONHPh),(NHCH(Bn)CONH2,Ph,OH,Et,CONHPh),(NHCH(Bn)CO- NH2,Ph,OH,Et, CONH-3-pyridyl),(NHCH(Bn)CONH2,Ph,OH,Et,NHCOPh),(NHCH(Bn)CONH2,Ph,OH,Et,N- HCO-2-furyl),(NHCH(Bn)CONH2,Ph,OH,Et,NHCONHPh),(NHCH(Bn)CONH2,Ph,OH,Et,NHC- OCONHPh),(NHCH(Bn)CONH2,Ph,OH,CH2OH,CONHPh),(NHCH(Bn)CONH2,Ph,OH,CH2OH,CON- H-3-pyridyl),(NHCH(Bn)CONH2,Ph,OH,CH2OH,NHCOPh),(NHCH(Bn)CONH2,Ph,OH,CH2OH- ,NHCO-2-furyl),(NHCH(Bn)CONH2,Ph,OH,CH2OH,NHCONHPh),(NHCH(Bn)CONH2,Ph,OH,C- H2OH,NHCOCONHPh),
(NHCH(Me)CH2OH,H,H,Me,CONHPh),(NHCH(Me)CH2OH,H,H,Me,CONH-3-pyridyl),(NHCH- (Me)CH2OH,H,H,Me,NHCOPh),(NHCH(Me)CH2OH,H,H,Me,NHCO-2-furyl),(NHCH(Me)CH2O- H,H,H,Me,NHCONHPh),(NHCH(Me)CH2OH,H,H,Me,NHCOCONHPh),(NHCH(Me)CH2OH,H,H,Et- ,CONHPh),(NHCH(Me)CH2OH,H,H,Et, CONH-3-pyridyl),(NHCH(Me)CH2OH,H,H,Et,NHCOPh),(NHCH(Me)CH2OH,H,H, Et,NHCO-2-furyl),(NHCH(Me)CH2OH,H,H,Et,NHCONHPh),(NHCH(Me)CH2OH, H,H,Et,NHCOCONHPh),(NHCH(Me)CH2OH,H,H,CH2OH,CONHPh),(NHCH(Me)CH2OH,H,H,CH- 2OH,CONH-3-pyridyl),(NHCH(Me)CH2OH,H,H,CH2OH,NHCOPh),(NHCH(Me)CH2OH,H,H,CH- 2OH,NHCO-2-furyl),(NHCH(Me)CH2OH,H,H,CH2OH,NHCONHPh),(NHCH(Me)CH2OH,H,H,CH- 2OH,NHCOCONHPh),(NHCH(Me)CH2OH,H,Me,Me,CONHPh),(NHCH(Me)CH2OH,H,Me,Me,CONH- -3-pyridyl),(NHCH(Me)CH2OH,H,Me,Me,NHCOPh),(NHCH(Me)CH2OH,H,Me,Me,NHCO-2-f- uryl),(NHCH(Me)CH2OH,H,Me,Me,NHCONHPh),(NHCH(Me)CH2OH,H,Me,Me,NHCOCONHPh),- (NHCH(Me)CH2OH,H,Me,Et,CONHPh),(NHCH(Me)CH2OH,H,Me,Et,CONH-3-pyridyl),(NHC- H(Me)CH2OH,H,Me,Et,NHCOPh),(NHCH(Me)CH2OH,H,Me,Et,NHCO-2-furyl),(NHCH(Me)C- H2OH,H,Me,Et,NHCONHPh),(NHCH(Me)CH2OH,H,Me,Et,NHCOCONHPh),(NHCH(Me)CH2OH,H- ,Me,CH2OH,CONHPh),(NHCH(Me)CH2OH,H,Me,CH2OH,CONH-3-pyridyl),(NHCH(Me)CH2OH- ,H,Me,CH2OH,NHCOPh),(NHCH(Me)CH2OH,H,Me,CH2OH,NHCO-2-furyl),(NHCH(Me)CH2OH- ,H,Me,CH2OH,NHCONHPh),(NHCH(Me)CH2OH,H,Me,CH2OH,NHCOCONHPh),(NHCH(Me)CH2OH- ,H,Ph,Me,CONHPh),(NHCH(Me)CH2OH,H,Ph,Me,CONH-3-pyridyl),(NHCH(Me)CH2OH,H,P- h,Me,NHCOPh),(NHCH(Me)CH2OH,H,Ph,Me,NHCO-2-furyl),(NHCH(Me)CH2OH,H,Ph,Me,N- HCONHPh),(NHCH(Me)CH2OH,H,Ph,Me, NHCOCONHPh),(NHCH(Me)CH2OH,H,Ph,Et,CONHPh),(NHCH(Me)CH2OH,H,Ph,Et,CONH-3-- pyridyl),(NHCH(Me)CH2OH,H,Ph,Et,NHCOPh),(NHCH(Me)CH2OH,H,Ph,Et,NHCO-2-fury- l),(NHCH(Me)CH2OH,H,Ph,Et,NHCONHPh),(NHCH(Me)CH2OH,H,Ph,Et,NHCOCONHPh),(NH- CH(Me)CH2OH,H,Ph,CH2OH,CONHPh),(NHCH(Me)CH2OH,H,Ph,CH2OH,CONH-3-pyridyl),(- NHCH(Me)CH2OH,H,Ph,CH2OH,NHCOPh),(NHCH(Me)CH2OH,H,Ph,CH2OH,NHCO-2-furyl),(- NHCH(Me)CH2OH,H,Ph,CH2OH,NHCONHPh),(NHCH(Me)CH2OH,H,Ph,CH2OH,NHCOCONHPh),(- NHCH(Me)CH2OH,H,OH,Me,CONHPh),(NHCH(Me)CH2OH,H,OH,Me,CONH-3-pyridyl),(NHCH- (Me)CH2OH,H,OH,Me,NHCOPh),(NHCH(Me)CH2OH,H,OH, Me,NHCO-2-furyl),(NHCH(Me)CH2OH,H,OH,Me,NHCONHPh),(NHCH(Me)CH2OH,H,OH,Me,- NHCOCONHPh),(NHCH(Me)CH2OH,H,OH,Et,CONHPh),(NHCH(Me)CH2OH,H,OH,Et,CONH-3-p- yridyl),(NHCH(Me)CH2OH,H,OH,Et,NHCOPh),(NHCH(Me)CH2OH,H,OH,Et,NHCO-2-furyl- ),(NHCH(Me)CH2OH,H,OH,Et,NHCONHPh),(NHCH(Me)CH2OH,H,OH,Et,NHCOCONHPh),(NHC- H(Me)CH2OH,H,OH,CH2OH,CONHPh),(NHCH(Me)CH2OH,H,OH,CH2OH,CONH-3-pyridy- l),(NHCH(Me)CH2OH,H,OH,CH2OH,NHCOPh),(NHCH(Me)CH2OH,H,OH,CH2OH,NHCO-2-fury- l),(NHCH(Me)CH2OH,H,OH,CH2OH,NHCONHPh),(NHCH(Me)CH2OH,H,OH,CH2OH,NHCOCONHP- h),(NHCH(Me)CH2OH,Me,H,Me,CONHPh),(NHCH(Me)CH2OH,Me,H,Me,CONH-3-pyridyl),(- NHCH(Me)CH2OH,Me,H,Me,NHCOPh),(NHCH(Me)CH2OH,Me,H,Me,NHCO-2-furyl),(NHCH(M- e)CH2OH,Me,H,Me,NHCONHPh), (NHCH(Me)CH2OH,Me,H,Me,NHCOCONHPh),(NHCH(Me)CH2OH,Me,H,Et,CONHPh),(NHCH(M- e)CH2OH,Me,H,Et,CONH-3-pyridyl),(NHCH(Me)CH2OH,Me,H,Et,NHCOPh),(NHCH(Me)CH- 2OH,Me,H,Et,NHCO-2-furyl),(NHCH(Me)CH2OH,Me,H,Et,NHCONHPh),(NHCH(Me)CH2OH,- Me,H,Et,NHCOCONHPh),(NHCH(Me)CH2OH,Me,H,CH2OH,CONHPh),(NHCH(Me)CH2OH,Me,H,- CH2OH,CONH-3-pyridyl), (NHCH(Me)CH2OH,Me,H,CH2OH,NHCOPh),(NHCH(Me)CH2OH,Me,H,CH2OH, NHCO-2-furyl),(NHCH(Me)CH2OH,Me,H,CH2OH,NHCONHPh),(NHCH(Me)CH2OH,Me,H,CH2- OH,NHCO CONHPh),(NHCH(Me)CH2OH,Me,Me,Me,CONHPh),(NHCH(Me)CH2OH,Me,Me,Me,CO- NH-3-pyridyl),(NHCH(Me)CH2OH,Me,Me,Me,NHCOPh),(NHCH(Me)CH2OH,Me,Me,Me,NHCO- -2-furyl),(NHCH(Me)CH2OH,Me,Me, Me,NHCONHPh),(NHCH(Me)CH2OH,Me,Me,Me,NHCOCONHPh),(NHCH(Me)CH2OH,Me,Me,Et,- CONHPh),(NHCH(Me)CH2OH,Me,Me,Et,CONH-3-pyridyl),(NHCH(Me)CH2OH,Me,Me,Et,NH- COPh),(NHCH(Me)CH2OH,Me,Me,Et,NHCO-2-furyl),(NHCH(Me)CH2OH,Me,Me,Et,NHCONH- Ph),(NHCH(Me)CH2OH,Me,Me,Et,NHCOCONHPh),(NHCH(Me)CH2OH,Me,Me,CH2OH,CONHPh)- ,(NHCH(Me)CH2OH,Me, Me,CH2OH,CONH-3-pyridyl),(NHCH(Me)CH2OH,Me,Me,CH2OH,NHCOPh),(NHCH(Me)CH2O- H,Me,Me,CH2OH,NHCO-2-furyl),(NHCH(Me)CH2OH,Me,Me,CH2OH,NHCONHPh),(NHCH(Me)- CH2OH,Me,Me,CH2OH,NHCO CONHPh),(NHCH(Me)CH2OH,Me,Ph,Me,CONHPh),(NHCH(Me)CH2OH,Me,Ph,Me,CONH-3-py- ridyl),(NHCH(Me)CH2OH,Me,Ph,Me,NHCOPh),(NHCH(Me)CH2OH,Me,Ph,Me,NHCO-2-fury- l),(NHCH(Me)CH2OH,Me,Ph,Me,NHCONHPh),(NHCH(Me)CH2OH,Me,Ph,Me, NHCOCONHPh),(NHCH(Me)CH2OH,Me,Ph,Et,CONHPh),(NHCH(Me)CH2OH,Me,Ph,Et,CONH-- 3-pyridyl),(NHCH(Me)CH2OH,Me,Ph,Et,NHCOPh),(NHCH(Me)CH2OH,Me,Ph,Et,NHCO-2-- furyl),(NHCH(Me)CH2OH,Me,Ph,Et,NHCONPh),(NHCH(Me)CH2OH,Me,Ph,Et,NHCOCONHPh- ),(NHCH(Me)CH2OH,Me,Ph,CH2OH,CONHPh),(NHCH(Me)CH2OH,Me,Ph,CH2OH,CONH-3-pyr- idyl),(NHCH(Me)CH2OH,Me,Ph,CH2OH,NHCOPh),(NHCH(Me)CH2OH,Me,Ph,CH2OH,NHCO-2- -furyl),(NHCH(Me)CH2OH,Me,Ph,CH2OH,NHCONHPh),(NHCH(Me)CH2OH,Me,Ph,CH2OH,NH- COCONHPh),(NHCH(Me)CH2OH,Me,OH,Me,CONHPh),(NHCH(Me)CH2OH,Me,OH,Me,CONH-3-p- yridyl),(NHCH(Me)CH2OH,Me,OH,Me,NHCOPh),(NHCH(Me)CH2OH,Me,OH,Me,NHCO-2-fur- yl),(NHCH(Me)CH2OH,Me,OH,Me,NHCONHPh),(NHCH(Me)CH2OH,Me,OH,Me,NHCOCONHPh),- (NHCH(Me)CH2OH,Me,OH,Et,CONHPh),(NHCH(Me)CH2OH,Me,OH,Et,CONH-3-pyridyl),(N- HCH(Me)CH2OH,Me,OH,Et,NHCOPh),(NHCH(Me)CH2OH,Me,OH,Et,NHCO-2-furyl),(NHCH(- Me)CH2OH,Me,OH,Et,NHCONHPh),(NHCH(Me)CH2OH,Me,OH,Et,NHCO CONHPh),(NHCH(Me)CH2OH,Me,OH,CH2OH,CONHPh),(NHCH(Me)CH2OH,Me,OH,CH2OH,CON- H-3-pyridyl),(NHCH(Me)CH2OH,Me,OH,CH2OH,NHCOPh),(NHCH(Me)CH2OH,Me,OH,CH2OH- ,NHCO-2-furyl),(NHCH(Me)CH2OH,Me,OH,CH2OH,NHCONHPh),(NHCH(Me)CH2OH,Me,OH,C- H2OH,NHCOCONHPh),(NHCH(Me)CH2OH,Ph,H, Me,CONHPh),(NHCH(Me)CH2OH,Ph,H,Me,CONH-3-pyridyl),(NHCH(Me)CH2OH,Ph,H,Me,- NHCOPh),(NHCH(Me)CH2OH,Ph,H,Me,NHCO-2-furyl),(NHCH(Me)CH2OH,Ph,H,Me,NHCONH- Ph),(NHCH(Me)CH2OH,Ph,H,Me,NHCOCONHPh),(NHCH(Me)CH2OH,Ph,H,Et,CONHPh),(NHC- H(Me)CH2OH,Ph,H,Et,CONH-3-pyridyl),(NHCH(Me)CH2OH,Ph,H,Et,NHCOPh),(NHCH(Me- )CH2OH,Ph,H,Et,NHCO-2-furyl),(NHCH(Me)CH2OH,Ph,H,Et,NHCONHPh),(NHCH(Me)CH2- OH,Ph,H,Et,NHCOCONHPh),(NHCH(Me)CH2OH,Ph,H,CH2OH,CONHPh),(NHCH(Me)CH2OH,Ph- ,H,CH2OH,CONH-3-pyridyl),(NHCH(Me)CH2OH,Ph,H,CH2OH,NHCOPh),(NHCH(Me)CH2OH,- Ph,H,CH2OH,NHCO-2-furyl),(NHCH(Me)CH2OH,Ph,H,CH2OH,NHCONHPh),(NHCH(Me)CH2O- H,Ph,H,CH2OH,NHCOCONHPh),(NHCH(Me)CH2OH, Ph,Me,Me,CONHPh),(NHCH(Me)CH2OH,Ph,Me,Me,CONH-3-pyridyl),(NHCH(Me)CH2OH,P- h,Me,Me,NHCOPh),(NHCH(Me)CH2OH,Ph,Me,Me,NHCO-2-furyl),(NHCH(Me)CH2OH,Ph,Me- ,Me,NHCONHPh),(NHCH(Me)CH2OH,Ph,Me,Me,NHCOCONHPh),(NHCH(Me)CH2OH,Ph,Me,Et,- CONHPh),(NHCH(Me)CH2OH,Ph,Me,Et,CONH-3-pyridyl),(NHCH(Me)CH2OH,Ph,Me,Et,NH- COPh),(NHCH(Me)CH2OH,Ph, Me,Et,NHCO-2-furyl),(NHCH(Me)CH2OH,Ph,Me,Et,NHCONHPh),(NHCH(Me)CH2OH,Ph,M- e,Et,NHCOCONHPh),(NHCH(Me)CH2OH,Ph,Me,CH2OH,CONHPh),(NHCH(Me)CH2OH,Ph,Me,C- H2OH,CONH-3-pyridyl),(NHCH(Me)CH2OH,Ph,Me,CH2OH,NHCOPh),(NHCH(Me)CH2OH,Ph,- Me,CH2OH,NHCO-2-furyl),(NHCH(Me)CH2OH,Ph,Me,CH2OH,NHCONHPh),(NHCH(Me)CH2OH- ,Ph,Me,CH2OH,NHCOCONHPh),(NHCH(Me)CH2OH,Ph,Ph,Me,CONHPh),(NHCH(Me)CH2OH,Ph- ,Ph,Me, CONH-3-pyridyl),(NHCH(Me)CH2OH,Ph,Ph,Me,NHCOPh),(NHCH(Me)CH2OH,Ph,- Ph,Me,NHCO-2-furyl),(NHCH(Me)CH2OH,Ph,Ph,Me,NHCONHPh),(NHCH(Me)CH2OH,Ph,Ph- ,Me,NHCOCONHPh),(NHCH(Me)CH2OH,Ph,Ph,Et,CONHPh),(NHCH(Me)CH2OH,Ph,Ph,Et,CO- NH-3-pyridyl),(NHCH(Me)CH2OH,Ph,Ph,Et,NHCOPh),(NHCH(Me)CH2OH,Ph,Ph,Et,NHCO- -2-furyl),(NHCH(Me)CH2OH,Ph,Ph,Et,NCONHPh),(NHCH(Me)CH2OH,Ph,Ph,Et,NHCOCON- HPh),(NHCH(Me)CH2OH,Ph, Ph,CH2OH,CONHPh),(NHCH(Me)CH2OH,Ph,Ph,CH2OH,CONH-3-pyridyl),(NHCH(Me)CH2O- H,Ph,Ph,CH2OH,NHCOPh),(NHCH(Me)CH2OH,Ph,Ph,CH2OH,NHCO-2-furyl),(NHCH(Me)CH- 2OH,Ph,Ph,CH2OH,NHCONHPh),(NHCH(Me)CH2OH,Ph,Ph,CH2OH,NHCOCONHPh),(NHCH(Me)- CH2OH,Ph,OH,Me,CONHPh),(NHCH(Me)CH2OH,Ph,OH,Me,CONH-3-pyridyl),(NHCH(Me)CH- 2OH,Ph,OH,Me,NHCOPh),(NHCH(Me)CH2OH,Ph,OH,Me,NHCO-2-furyl),(NHCH(Me)CH2OH,- Ph,OH,Me,NHCONHPh),(NHCH(Me)CH2OH,Ph,OH,Me,NHCOCONHPh),(NHCH(Me)CH2OH, Ph,OH,Et,CONHPh),(NHCH(Me)CH2OH,Ph,OH,Et,CONH-3-pyridyl),(NHCH(Me)CH2OH,P- h,OH,Et,NHCOPh),(NHCH(Me)CH2OH,Ph,OH,Et,NHCO-2-furyl),(NHCH(Me)CH2OH,Ph,OH- ,Et,NHCONHPh),(NHCH(Me)CH2OH,Ph,OH,Et,NHCOCONHPh),(NHCH(Me)CH2OH,Ph,OH,CH2- OH,CONHPh),(NHCH(Me)CH2OH,Ph,OH, CH2OH,CONH-3-pyridyl),(NHCH(Me)CH2OH,Ph,OH,CH2OH,NHCOPh),(NHCH(Me)CH2OH,P- h,OH,CH2OH,NHCO-2-furyl),(NHCH(Me)CH2OH,Ph,OH,CH2OH,NHCONHPh),(NHCH(Me)CH2- OH,Ph,OH,CH2OH,NHCOOONHPh), (NHCH(Me)CONHMe,H,H,Me,CONHPh),(NHCH(Me)CONHMe,H,H,Me,CONH-3-pyridyl),(NH- CH(Me)CONHMe,H,H,Me,NHCOPh),(NHCH(Me)CONHMe,H,H,Me,NHCO-2-furyl),(NHCH(Me)- CONHMe,H,H,Me,NHCONHPh),(NHCH(Me)CONHMe,H,H,Me,NHCOCONHPh),(NHCH(Me)CONHMe- ,H,H,Et,CONHPh),(NHCH(Me)CONHMe,H,H,Et,CONH-3-pyridyl),(NHCH(Me)CONHMe,H,H- ,Et,NHCOPh),(NHCH(Me)CONHMe,H,H,Et,NHCO-2-furyl),(NHCH(Me)CONHMe,H,H,Et,NH- CONHPh),(NHCH(Me)CONHMe,H,H,Et,NHCOCONHPh),(NHCH(Me)CONHMe,H,H,CH2OH,CONHP- h),(NHCH(Me)CONHMe,H,H,CH2OH,CONH-3-pyridyl),(NHCH(Me)CONHMe,H,H,CH2OH,NHC- OPh),(NHCH(Me)CONHMe,H,H,CH2OH,NHCO-2-furyl),(NHCH(Me)CONHMe,H,H,CH2OH,NHC- ONHPh),(NHCH(Me)CONHMe,H,H,CH2OH,NHCOCONHPh),(NHCH(Me)CONHMe,H,Me,Me,CONHP- h),(NHCH(Me)CONHMe,H,Me,Me,CONH-3-pyridyl),(NHCH(Me)CONHMe,H,Me,Me,NHCOPh)- ,(NHCH(Me)CONHMe,H,Me,Me,NHCO-2-furyl),(NHCH(Me)CONHMe,H,Me,Me,NHCONHPh),(- NHCH(Me)CONHMe,H,Me,Me,NHCOCONHPh),(NHCH(Me)CONHMe,H, Me,Et,CONHPh),(NHCH(Me)CONMe,H,Me,Et,CONH-3-pyridyl),(NHCH(Me)CONHMe,H,Me- ,Et,NHCOPh),(NHCH(Me)CONHMe,H,Me,Et,NHCO-2-furyl),(NHCH(Me)CONHMe,H,Me,Et,- NHCONHPh),(NHCH(Me)CONHMe,H,Me,Et,NHCOCONHPh),(NHCH(Me)CONHMe,H,Me,CH2OH,C- ONHPh),(NHCH(Me)CONHMe,H,Me,CH2OH,CONH-3-pyridyl),(NHCH(Me)CONHMe,H,Me,CH2- OH,NHCOPh),(NHCH(Me)CONHMe,H,Me,CH2OH,NHCO-2-furyl),(NHCH(Me)CONHMe,H,Me,C- H2OH, NHCONHPh),(NHCH(Me)CONHMe,H,Me,CH2OH,NHCOCONHPh),(NHCH(Me)CONHMe,H,P- h,Me,CONHPh),(NHCH(Me)CONHMe,H,Ph,Me,CONH-3-pyridyl),(NHCH(Me)CONHMe,H,Ph,- Me,NHCOPh),(NHCH(Me)CONHMe,H,Ph,Me,NHCO-2-furyl),(NHCH(Me)CONHMe,H,Ph,Me,N- HCONHPh),(NHCH(Me)CONHMe,H,Ph,Me,NHCOCONHPh),(NHCH(Me)CONHMe,H,Ph,Et,CONHP- h),(NHCH(Me)CONHMe,H,Ph,Et,CONH-3-pyridyl),(NHCH(Me)CONHMe,H,Ph,Et,NHCOPh)- ,(NHCH(Me)CONHMe,H,Ph,Et,NHCO-2-furyl),(NHCH(Me)CONHMe,H,Ph,Et,NHCONHPh),(- NHCH(Me)CONHMe,H,Ph,Et,NHCOCONHPh),(NHCH(Me)CONHMe,H,Ph,CH2OH,CONHPh),(NHC- H(Me)CONHMe,H,Ph,CH2OH,CONH-3-pyridyl),(NHCH(Me)CONHMe,H,Ph,CH2OH,NHCOPh),- (NHCH(Me)CONHMe,H,Ph,CH2OH,NHCO-2-furyl),(NHCH(Me)CONHMe,H,Ph,CH2OH,NHCONH- Ph),(NHCH(Me)CONHMe,H,Ph, CH2OH,NHCOCONHPh),(NHCH(Me)CONHMe,H,OH,Me,CONHPh),(NHCH(Me)CONHMe,H,OH,Me- ,CONH-3-pyridyl),(NCH(Me)CONHMe,H,OH,Me,NHCOPh),(NHCH(Me)CONHMe,H,OH,Me,NH- CO-2-furyl),(NHCH(Me)CONHMe,H,OH,Me,NHCONHPh),(NHCH(Me)CONHMe,H,OH,Me,NHCO- CONHPh),(NHCH(Me)CONHMe,H,OH,Et,CONHPh),(NHCH(Me)CONHMe,H,OH,Et,CONH-3-pyr- idyl),(NHCH(Me)CONHMe,H,OH,Et,NHCOPh),(NHCH(Me)CONHMe,H,OH,Et,NHCO-2-furyl- ),(NHCH(Me)CONHMe,H,OH,Et,NHCONHPh),(NHCH(Me)CONHMe,H,OH,Et,NHCOCONHPh),(N- HCH(Me)CONHMe,H,OH,CH2OH,CONHPh),(NHCH(Me)CONHMe,H,OH,CH2OH,CONH-3-pyridyl- ),(NHCH(Me)CONHMe,H,OH,CH2OH,NHCOPh),(NHCH(Me)CONHMe,H,OH,CH2OH,NHCO-2-fur- yl),(NHCH(Me)CONH2Me,H,OH,CH2OH,NHCONHPh),(NHCH(Me)CONHMe,H,OH,CH2OH,NHCOC- ONHPh),(NHCH(Me)CONHMe,Me,H,Me,CONHPh),(NHCH(Me)CONHMe,Me,H,Me,CONH-3-pyri- dyl),(NHCH(Me)CONHMe,Me,H,Me,NHCOPh),(NHCH(Me)CONHMe,Me,H,Me,NHCO-2-furyl)- ,(NHCH(Me)CONHMe,Me,H,Me,NHCONHPh),(NHCH(Me)CONHMe,Me,H,Me,NHCOCONHPh),(NH- CH(Me)CONHMe,Me,H,Et,CONHPh),(NHCH(Me)CONHMe,Me,H,Et,CONH-3-pyridyl),(NHCH- (Me)CONHMe,Me,H,Et,NHCOPh),(NHCH(Me)CONHMe,Me,H,Et,NHCO-2-furyl),(NHCH(Me)- CONHMe,Me,H,Et,NHCONHPh),(NHCH(Me)CONHMe,Me,H,Et,NHCOCONHPh),(NHCH(Me)CONH- Me,Me,H,CH2OH,CONHPh),(NHCH(Me)CONHMe,Me,H,CH2OH,CONH-3-pyridyl),(NHCH(Me)- CONHMe,Me,H,CH2OH,NHCOPh),(NHCH(Me)CONHMe,Me,H,CH2OH, NHCO-2-furyl),(NHCH(Me)CONHMe,Me,H,CH2OH,NHCONHPh),(NHCH(Me)CONHMe,Me,H,C- H2OH,NHCOCONHPh),(NHCH(Me)CONHMe,Me,Me,Me,CONHPh),(NHCH(Me)CONHMe,Me,Me,Me- ,CONH-3-pyridyl),(NHCH(Me)CONHMe,Me,Me, Me,NHCOPh),(NHCH(Me)CONHMe,Me,Me,Me,NHCO-2-furyl),(NHCH(Me)CONHMe,Me,Me,M- e,NHCONHPh),(NHCH(Me)CONHMe,Me,Me,Me,NHCOCONHPh),(NHCH(Me)CONHMe,Me,Me,Et,- CONHPh),(NHCH(Me)CONHMe,Me,Me,Et,CONH-3-pyridyl),(NHCH(Me)CONHMe,Me,Me,Et,- NHCOPh),(NHCH(Me)CONHMe,Me,Me, Et,NHCO-2-furyl),(NHCH(Me)CONHMe,Me,Me,Et,NHCONHPh),(NHCH(Me)CONHMe,Me,Me- ,Et,NHCOCONHPh),(NHCH(Me)CONHMe,Me,Me,CH2OH,CONHPh), (NHCH(Me)CONHMe,Me,Me,CH2OH,CONH-3-pyridyl),(NHCH(Me)CONHMe,Me, Me,CH2OH,NHCOPh),(NHCH(Me)CONHMe,Me,Me,CH2OH,NHCO-2-furyl),(NHCH(Me)CONHM- e,Me,Me,CH2OH,NHCONHPh),(NHCH(Me)CONHMe,Me,Me,CH2OH,NHCOCONHPh),(NHCH(Me)C- ONHMe,Me,Ph,Me,CONHPh),(NHCH(Me)CONHMe,Me,Ph,Me,CONH-3-pyridyl),(NHCH(Me)C- ONHMe,Me,Ph,Me,NHCOPh),(NHCH(Me)CONHMe,Me,Ph,Me,NHCO-2-furyl),(NHCH(Me)CON- HMe,Me,Ph,Me,NHCONHPh),(NHCH(Me)CONHMe,Me,Ph,Me,NHCOCONHPh),(NHCH(Me)CONHM- e,Me,Ph,Et,CONHPh),(NHCH(Me)CONHMe,Me,Ph,Et,CONH-3-pyridyl),(NHCH(Me)CONHM- e,Me,Ph,Et,NHCOPh),(NHCH(Me)CONHMe,Me,Ph,Et,NHCO-2-furyl),(NHCH(Me)CONHMe,- Me,Ph,Et,NHCONHPh),(NHCH(Me)CONHMe,Me,Ph,Et,NHCOCONHPh),(NHCH(Me)CONHMe,Me- ,Ph,CH2OH,CONHPh),(NHCH(Me)CONHMe,Me,Ph,CH2OH,CONH-3-pyridyl),(NHCH(Me)CON- HMe,Me,Ph,CH2OH,NHCOPh),(NHCH(Me)CONHMe,Me,Ph,CH2OH,NHCO-2-furyl),(NHCH(Me- )CONHMe,Me,Ph,CH2OH,NHCONHPh),(NHCH(Me)CONHMe,Me,Ph,CH2OH,NHCOCONHPh),(NHC- H(Me)CONHMe,Me,OH,Me,CONHPh),(NHCH(Me)CONHMe,Me,OH,Me,CONH-3-pyridyl),(NHC- H(Me)CONHMe,Me,OH,Me,NHCOPh),(NHCH(Me)CONHMe, Me,OH,Me,NHCO2-furyl),(NHCH(Me)CONHMe,Me,OH,Me,NHCONHPh),(NHCH(Me)CONHMe,- Me,OH,Me,NHCOCONHPh),(NHCH(Me)CONHMe,Me,OH,Et,CONHPh),(NHCH(Me)CONHMe,Me,O- H,Et,CONH-3-pyridyl),(NHCH(Me)CONHMe,Me,OH,Et,NHCOPh),(NHCH(Me)CONHMe,Me,O- H,Et,NHRCO-2-furyl),(NCH(Me)CONHMe,Me,OH,Et,NHCONHPh),(NHCH(Me)CONHMe,Me,O- H,Et,NHCOCONHPh)(NHCH(Me)CONHMe,Me,OH,CH2OH,CONHPh),(NHCH(Me)CONHMe,Me,OH,- CH2OH,CONH-3-pyridyl),(NHCH(Me)CONHMe,Me,OH,CH2OH,NHCOPh),(NHCH(Me)CONHMe,- Me,OH,CH2OH,NHCO-2-furyl),(NHCH(Me)CONHMe,Me,OH,CH2OH,NHCONHPh),(NHCH(Me)C- ONHMe,Me,OH,CH2OH,NHCOCONHPh),(NHCH(Me)CONHMe,Ph,H,Me,CONHPh),(NHCH(Me)CON- HMe,Ph,H,Me,CONH-3-pyridyl),(NHCH(Me)CONHMe,Ph,H,Me,NHCOPh),(NHCH(Me)CONHM- e,Ph,H,Me,NHCO-2-furyl),(NHCH(Me)CONHMe,Ph,H,Me,NHCONHPh),(NHCH(Me)CONHMe,- Ph,H,Me,NHCOCONHPh),(NHCH(Me)CONHMe,Ph,H,Et,CONHPh),(NHCH(Me)CONHMe,Ph,H,E- t,CONH-3-pyridyl),(NHCH(Me)CONHMe,Ph,H,Et,NHCOPh),(NHCH(Me)CONHMe,Ph,H,Et,- NHCO-2-furyl),(NHCH(Me)CONHMe,Ph,H,Et,NHCONHPh),(NHCH(Me)CONHMe,Ph,H,Et,NH- COCONHPh),(NHCH(Me)CONHMe,Ph,H,CH2OH,CONHPh),(NHCH(Me)CONHMe,Ph,H,CH2OH,CO- NH-3-pyridyl),(NHCH(Me)CONHMe,Ph,H,CH2OH,NHCOPh),(NHCH(Me)CONHMe,Ph,H,CH2O- H,NHCO-2-furyl),(NHCH(Me)CONHMe,Ph,H,CH2OH,NHCONHPh),(NHCH(Me)CONHMe,Ph, H,CH2OH,NHCOCONHPh),(NHCH(Me)CONHMe,Ph,Me,Me,CONHPh),(NHCH(Me)CONHMe,Ph,M- e,Me,CONH-3-pyridyl),(NHCH(Me)CONHMe,Ph,Me,Me,NHCOPh),(NHCH(Me)CONHMe,Ph,M- e,Me,NHCO-2-furyl),(NHCH(Me)CONHMe,Ph,Me, Me,NHCONHPh),(NHCH(Me)CONHMe,Ph,Me,Me,NHCONHPh),(NHCH(Me)CONHMe,Ph,Me,Et,- CONHPh),(NHCH(Me)CONHMe,Ph,Me,Et,CONH-3-pyridyl),(NHCH(Me)CONHMe,Ph,Me,Et,- NHCOPh),(NHCH(Me)CONHMe,Ph,Me,Et,NHCO-2-furyl),(NHCH(Me)CONHMe,Ph,Me,Et,NH- CONHPh),(NHCH(Me)CONHMe,Ph,Me, Et,NHCO CONHPh),(NHCH(Me)CONHMe,Ph,Me,CH2OH,CONHPh),(NHCH(Me)CONHMe,Ph,Me,CH2OH,C- ONH-3-pyridyl),(NHCH(Me)CONHMe,Ph,Me,CH2OH, NHCOPh),(NHCH(Me)CONHMe,Ph,Me,CH2OH,NHCO-2-furyl),(NHCH(Me)CONHMe,Ph,Me,C- H2OH,NHCONHPh),(NHCH(Me)CONHMe,Ph,Me,CH2OH,NHCOCONHPh),(NHCH(Me)CONHMe,Ph,- Ph,Me,CONHPh),(NHCH(Me)CONHMe,Ph,Ph,Me,CONH-3-pyridyl),(NHCH(Me)CONHMe,Ph,- Ph,Me,NHCOPh),(NHCH(Me)CONHMe,Ph,Ph,Me,NHCO-2-furyl),(NHCH(Me)CONHMe,Ph,Ph- ,Me,NHECONHPh),(NHCH(Me)CONHMe,Ph,Ph,Me,NHCOCONHPh),(NHCH(Me)CONHMe,Ph,Ph,- Et,CONHPh),(NHCH(Me)CONHMe,Ph,Ph,Et,CONH-3-pyridyl),(NHCH(Me)CONHMe,Ph,Ph,- Et,NHCOPh),(NHCH(Me)CONHMe,Ph,Ph,Et,NHCO-2-furyl),(NHCH(Me)CONHMe,Ph,Ph,Et- ,NHCONHPh),(NHCH(Me)CONHMe,Ph,Ph,Et,NHCOCONHPh),(NHCH(Me)CONHMe,Ph,Ph,CH2O- H,CONHPh),(NHCH(Me)CONHMe,Ph,Ph,CH2OH,CONH-3-pyridy),(NHCH(Me)CONHMe,Ph,Ph- ,CH2OH,NHCOPh),(NHCH(Me)CONHMe,Ph,Ph,CH2OH,NHCO-2-furyl),(NHCH(Me)CONHMe,P- h,Ph,CH2OH,NHCONHPh),(NHCH(Me)CONHMe,Ph,Ph,CH2OH,NHCOCONHPh),(NHCH(Me)CONH- Me,Ph,OH,Me,CONHPh),(NHCH(Me)CONHMe,Ph,OH,Me,CONH3-pyridy),(NHCH(Me)CONHMe- ,Ph,OH,Me,NHCOPh),(NHCH(Me)CONHMe,Ph,OH,Me,NHCO-2-furyl),(NHCH(Me)CONHMe,P- h,OH,Me,NHCONHPh),(NHCH(Me)CONHMe,Ph, OH,Me,NHCOCONHTPh),(NHCH(Me)CONHMe,Ph,OH,Et,CONHPh),(NHCH(Me)CONHMe,Ph,OH- ,Et,CONH-3-pyridyl),(NHCH(Me)CONHMe,Ph,OH,Et,NHCOPh),(NHCH(Me)CONHMe,Ph,OH- ,Et,NHCO-2-furyl),(NHCH(Me)CONHMe,Ph,OH,Et,NHCONHPh),(NHCH(Me)CONHMe,Ph,OH- ,Et,NHCO CONHPh),(NHCH(Me)CONHMe,Ph,OH,CH2OH,CONHPh),(NHCH(Me)CONHMe,Ph,OH- ,CH2OH,CONH-3-pyridyl),(NHCH(Me)CONHMe,Ph,OH,CH2OH,NHCOPh),(NHCH(Me)CONHMe- ,Ph,OH,CH2OH,NHCO-2-furyl),(NHCH(Me)CONBMe,Ph,OH,CH2OH,NHCONHPh),(NHCH(Me)- CONHMe,Ph,OH,CH2OH,NHCOCONHPh), (NHCOCH(iPr)OH,H,H,Me,CONHPh),(NHCOCH(iPr)OH,H,H,Me,CONH-3-pyridyl),(NHCO- CH(iPr)OH,H,H,Me,NHCOPh),(NHCOCH(iPr)OH,H,H,Me,NHCO-2-furyl) (NHCOCH(iPr)OH,H,H,Me,NHCONHPh),(NHCOCH(iPr)OH,H,H,Me,NHCOCONHPh),(NHCOCH- (iPr) OH,H,H,Et,CONHPh),(NHCOCH(iPr)OH,H,H,Et,CONH-3-pyridyl),(NHCOCH(iPr)- OH,H,H,Et,NHCOPh),(NHCOCH(iPr)OH,H,H,Et,NHCO-2-furyl),(NHCOCH(iPr) OH,H,H,Et,NHCONHPh),(NHCOCH(iPr)OH,H,H,Et,NHCONHPh),(NHCOCH(iPr) OH,H,H,CH2OH,CONHPh),(NHCOCH(iPr) OH,H,H,CH2OH,CONH3-pyridyl),(NHCOCH(iPr)OH,H,H,CH2OH,NHCOPh),(NHCOCH(iPr)- OH,H,H,CH2OH,NHCO-2-furyl),(NHCOCH(iPr) OH,H,H,CH2OH,NHCONHPh),(NHCOCH(iPr)OH,H,H,CH2OH,NHCOCONHPh),(NHCOCH(iPr)O- H,H,Me,Me,CONHPh),(NHCOCH(iPr)OH,H,Me,Me,CONH-3-pyridyl),(NHCOCH(iPr)OH,H,- Me,Me,NHCOPh),(NHCOCH(iPr)OH,H,Me,Me,NHCO2-furyl),(NHCOCH(iPr)OH,H,Me,Me,N- HCONHPh),(NHCOCH(iPr)OH,H,Me,Me,NHCOCONHPh),(NHCOCH(iPr)OH,H,Me,Et,CONHPh)- ,(NHCOCH(iPr)OH,H,Me,Et,CONH-3-pyridyl),(NHCOCH(iPr)OH,H,Me,Et,NHCOPh),(NH- COCH(iPr)OH,H,Me,Et,NHCO-2-furyl),(NHCOCH(iPr)OH,H,Me,Et,NHCONHPh),(NHCOCH- (iPr)OH,H,Me,Et,NHCOCONHPh),(NHCOCH(iPr) OH,H,Me,CH2OH,CONHPh),(NHCOCH(iPr) OH,H,Me,CH2OH,CONH-3-pyridyl),(NHCOCH(iPr)OH,H,Me,CH2OH,NHCOPh),(NHCOCH(i- Pr) OH,H,Me,CH2OH, NHCO-2-furyl),(NHCOCH(iPr)OH,H,Me,CH2OH,NHCONHPh),(NHCOCH(iPr)OH,H,Me,CH2- OH,NHCOCONHPh),(NHCOCH(iPr)OH,H,Ph,Me,CONHPh),(NHCOCH(iPr)OH,H,Ph,Me,CONH-- 3-pyridyl),(NHCOCH(iPr)OH,H,Ph,Me,NHCOPh),(NHCOCH(iPr)OH,H,Ph,Me,NHCO-2-fu- ryl),(NHCOCH(iPr)OH,H,Ph,Me,NHCONHPh),(NHCOCH(iPr)OH,H,Ph,Me,NHCOCONHPh),(- NHCOCH(iPr)OH,H,Ph,Et,CONHPh),(NHCOCH(iPr)OH,H,Ph,Et,CONH-3-pyridyl),(NHCO-
CH(iPr)OH,H,Ph,Et,NHCOPh),(NHCOCH(iPr)OH,H,Ph,Et,NHCO-2-fu yl),(NHCOCH(iPr)OH,H,Ph,Et,NHCONHPh),(NHCOCH(iPr)OH,H,Ph,Et,NHCOCONHPh),(- NHCOCH(iPr)OH,H,Ph,CH2OH,CONHPh),(NHCOCH(iPr)OH,H,Ph,CH2OH,CONH-3-pyridyl)- ,(NHCOCH(iPr)OH,H,Ph,CH2OH,NHCOPh),(NHCOCH(iPr)OH,H,Ph,CH2OH,NHCO-2-furyl)- ,(NHCOCH(iPr) OH,H,Ph,CH2OH,NHCONHPh),(NHCOCH(iPr)OH,H,Ph,CH2OH,NHCOCONHPh),(NHCOCH(iPr- )OH,H,OH,Me,CONHPh),(NHCOCH(iPr)OH,H,OH,Me,CONH-3-pyridyl),(NHCOCH(iPr)OH,- H,OH,Me,NHCOPh),(NHCOCH(iPr)OH,H, OH,Me,NHCO-2-furyl),(NHCOCH(iPr)OH,H,OH,Me,NHCONHPh),(NHCOCH(iPr)OH,H,OH,- Me,NHCOCONHPh),(NHCOCH(iPr)OH,H,OH,Et,CONHPh),(NHCOCH(iPr) OH,H,OH,Et,CONH-3-pyridyl),(NHCOCH(iPr)OH,H,OH,Et,NHCOPh),(NHCOCH(iPr) OH,H,OH,Et,NHCO-2-furyl),(NHCOCH(iPr)OH,H,OH,Et,NHCONHPh),(NHCOCH(iPr)OH,- H,OH,Et,NHCOCONHPh),(NHCOCH(iPr)OH,H,OH,CH2OH,CONHPh),(NHCOCH(iPr)OH,H,OH,- CH2OH,CONH-3-pyridyl),(NHCOCH(iPr)OH,H, OH,CH2OH,NHCOPh),(NHCOCH(iPr) OH,H,OH,CH2OH,NHCO-2-furyl),(NHCOCH(iPr)OH,H,OH,CH2OH,NHCONHPh),(NHCOCH(i- Pr)OH,H,OH,CH2OH,NHCOCONHPh),(NHCOCH(iPr)OH,Me,H,Me,CONHPh),(NHCOCH(iPr)OH- ,Me,H,Me,CONH-3-pyridyl),(NHCOCH(iPr)OH,Me,H,Me,NHCOPh),(NHCOCH(iPr)OH,Me,- H,Me,NHCO2-furyl),(NHCOCH(iPr)OH,Me,H,Me,NHCONHPh),(NHCOCH(iPr)OH,Me,H,Me,- NHCOCONHPh),(NHCOCH(iPr)OH,Me,H,Et,CONHPh),(NHCOCH(iPr)OH,Me,H,Et,CONH-3-p- yridyl),(NHCOCH(iPr)OH,Me,H,Et,NHCOPh),(NHCOCH(iPr)OH,Me,H,Et,NHCO-2-furyl- ),(NHCOCH(iPr)OH,Me,H,Et,NHCONHPh),(NHCOCH(iPr)OH,Me,H,Et,NHCOCONHPh),(NHC- OCH(iPr)OH,Me,H,CH2OH,CONHPh),(NHCOCH(iPr)OH,Me,H,CH2OH,CONH-3-pyridyl),(N- HCOCH(iPr)OH,Me,H,CH2OH,NHCOPh),(NHCOCH(iPr)OH,Me,H,CH2OH,NHCO-2-furyl),(N- HCOCH(iPr)OH, Me,H,CH2OH,NHCONHPh),(NHCOCH(iPr)OH,Me,H,CH2OH,NHCOCONHPh),(NHCOCH(iPr)OH- ,Me,Me,Me,CONHPh),(NHCOCH(iPr)OH,Me,Me,Me,CONH-3-pyridyl),(NHCOCH(iPr)OH,M- e,Me,Me,NHCOPh),(NHCOCH(iPr)OH,Me,Me,Me,NHCO-2-furyl),(NHCOCH(iPr)OH,Me,Me- ,Me,NHCONHPh),(NHCOCH(iPr)OH,Me,Me, Me,NHCOCONHPh),(NHCOCH(iPr)OH,Me,Me,Et,CONHPh),(NHCOCH(iPr)OH, Me,Me,Et,CONH-3-pyridyl),(NHCOCH(iPr)OH,Me,Me,Et,NHCOPh),(NHCOCH(iPr)OH,M- e,Me,Et,NHCO-2-furyl),(NHCOCH(iPr)OH,Me,Me,Et,NHCONHPh),(NHCOCH(iPr)OH,Me,- Me,Et,NHCOCONHPh),(NHCOCH(iPr)OH,Me,Me,CH2OH,CONH7Ph),(NHCOCH(iPr)OH,Me,Me- ,CH2OH,CONH-3-pyridyl),(NHCOCH(iPr)OH,Me, Me,CH2OH,NHCOPh),(NHCOCH(iPr)OH,Me,Me,CH2OH,NHCO-2-furyl),(NHCOCH(iPr)OH,- Me,Me,CH2OH,NHCONHPh),(NHCOCH(iPr)OH,Me,Me,CH2OH,NHCOCONHPh),(NHCOCH(iPr) OH,Me,Ph,Me,CONHPh),(NHCOCH(iPr)OH,Me,Ph,Me,CONH-3-pyridyl),(NHCOCH(iPr)O- H,Me,Ph,Me,NHCOPh),(NHCOCH(iPr)OH,Me, Ph,Me,NHCO-2-furyl),(NHCOCH(iPr)OH,Me,Ph,Me,NHCONHPh),(NHCOCH(iPr)OH,Me,P- h,Me,NHCOCONHPh),(NHCOCH(iPr)OH,Me,Ph,Et,CONHPh),(NHCOCH(iPr)OH,Me,Ph,Et,C- ONH-3-pyridyl),(NHCOCH(iPr)OH,Me,Ph,Et,NHCOPh),(NHCOCH(iPr)OH,Me,Ph,Et,NHC- O-2-furyl),(NCOCH(iPr)OH,Me,Ph,Et,NCONHPh),(NHCOCH(iPr)OH,Me,Ph,Et,NHCOCON- HPh),(NHCOCH(iPr)OH,Me,Ph,CH2OH,CONHPh),(NH C OCH(iPr) OH,Me,Ph,CH2 OH,CONH-3-pyridyl,(NHCOCH(iPr) OH,Me,Ph,CH2OH,NHCOPh),(NHCOCH(iPr)OH,Me,Ph,CH2OH,NHCO-2-furyl),(NHCOCH(i- Pr)OH,Me,Ph,CH2OH,NHCONHPh),(NHCOCH(iPr)OH,Me,Ph,CH2OH, NHCOCONHPh),(NHCOCH(iPr)OH,Me,OH,Me,CONHPh),(NHCOCH(iPr)OH,Me, OH,Me,CONH-3-pyridyl),(NHCOCH(iPr)OH,Me,OH,Me,NHCOPh),(NHCOCH(iPr)OH,Me,O- H,Me,NHCO-2-furyl),(NHCOCH(iPr)OH,Me,OH,Me,NHCONHPh),(NHCOCH(iPr)OH,Me,OH,- Me,NHCOCONHPh),(NHCOCH(iPr)OH,Me,OH,Et,CONHPh),(NHCOCH(iPr) OH,Me,OH,Et,CONH-3-pyridyl),(NHCOCH(iPr) OH,Me,OH,Et,NHCOPh),(NHCOCH(iPr)OH,Me,OH,Et,NHCO-2-furyl),(NHCOCH(iPr)OH,- Me,OH,Et, NHCONHPh),(NHCOCH(iPr)OH,Me,OH,Et,NHCOCONHPh),(NHCOCH(iPr)OH, Me,OH,CH2OH,CONHPh),(NHCOCH(iPr)OH,Me,OH,CH2OH,CONH-3-pyridy),(NHCOCH(iPr- )OH,Me,OH,CH2OH,NHCOPh),(NHCOCH(iPr)OH,Me,OH,CH2OH, NHCO-2-furyl),(NHCOCH(iPr) OH,Me,OH,CH2OH,NHCONHPh),(NHCOCH(iPr) OH,Me,OH,CH2OH,NHCOCONHPh),(NHCOCH(iPr)OH,Ph,H,Me,CONHPh),(NHCOCH(iPr)OH,- Ph,H,Me,CONH-3-pyridyl),(NHCOCH(iPr)OH,Ph,H,Me,NHCOPh),(NHCOCH(iPr) OH,Ph,H,Me,NHCO-2-furyl),(NHCOCH(iPr)OH,Ph,H,Me,NHCONPh),(NHCOCH(iPr)OH,P- h,H,Me,NHCOCONHPh),(NHCOCH(iPr)OH,Ph,H,Et,CONHPh),(NHCOCH(iPr)OH,Ph,H,Et,C- ONH-3-pyridyl),(NHCOCH(iPr)OH,Ph,H,Et,NHCOPh),(NHCOCH(iPr) OH,Ph,H,Et,NHCO-2-furyl),(NHCOCH(iPr)OH,Ph,H,Et,NHCONHPh),(NHCOCH(iPr)OH,- Ph,H,Et,NHCOCONHPh),(NHCOCH(iPr)OH,Ph,H,CH2OH,CONHPh),(NHCOCH(iPr)OH,Ph,H,- CH2OH,CONH-3-pyridyl),(NHCOCH(iPr)OH,Ph,H,CH2OH,NHCOPh),(NHCOCH(iPr)OH,Ph,- H,CH2OH,NHCO-2-furyl)(NHCOCH(iPr)OH,Ph,H,CH2OH,NHCONHPh),(NHCOCH(iPr)OH,Ph- ,H,CH2OH,NHCOCONHPh),(NHCOCH(iPr)OH,Ph,Me,Me,CONHPh),(NHCOCH(iPr)OH,Ph,Me,- Me,CONH-3-pyridyl),(NHCOCH(iPr)OH,Ph,Me,Me,NHCOPh),(NHCOCH(iPr)OH, Ph,Me,Me,NHCO-2-furyl),(NHCOCH(iPr)OH,Ph,Me,Me,NHCONHPh),(NHCOCH(iPr)OH,P- h,Me,Me,NHCOCONHPh),(NHCOCH(iPr)OH,Ph,Me,Et,CONHPh),(NHCOCH(iPr)OH,Ph,Me,E- t,CONH-3-pyridyl),(NHCOCH(iPr)OH,Ph,Me,Et,NHCOPh),(NHCOCH(iPr)OH,Ph,Me,Et,- NHCO-2-furyl),(NHCOCH(iPr)OH,Ph,Me,Et,NHCONHPh),(NHCOCH(iPr)OH,Ph,Me,Et,NH- COCONHPh),(NHCOCH(iPr)OH,Ph,Me,CH2OH,CONHPh),(NHCOCH(iPr) OH,Ph,Me,CH2OH,CONH3-pyridyl),(NHCOCH(iPr) OH,Ph,Me,CH2OH,NHCOPh),(NHCOCH(iPr) OH,Ph,Me,CH2OH,NHCO-2-furyl), (NHCOCH(iPr)OH,Ph,Me,CH2OH,NHCONHPh),(NHCOCH(iPr)OH,Ph,Me,CH2OH,NHCOCONHP- h),(NHCOCH(iPr)OH,Ph,Ph,Me,CONHPh),(NHCOCH(iPr)OH,Ph,Ph,Me,CONH-3-pyridyl)- ,(NHCOCH(iPr)OH,Ph,Ph,Me,NHCOPh),(NHCOCH(iPr)OH,Ph,Ph,Me,NHCO-2-furyl),(NH- COCH(iPr)OH,Ph,Ph,Me,NHCONHPh),(NHCOCH(iPr)OH,Ph,Ph,Me,NHCOCONHPh),(NHCOCH- (iPr)OH,Ph,Ph,Et,CONHPh),(NHCOCH(iPr)OH,Ph,Ph,Et,CONH-3-pyridyl),(NHCOCH(i- Pr)OH,Ph,Ph,Et,NHCOPh), (NHCOCH(iPr)OH,Ph,Ph,Et,NHCO-2-furyl),(NHCOCH(iPr)OH,Ph,Ph,Et,NHCONHPh),(- NCOCH(iPr)OH,Ph,Ph,Et,NHCOCONHPh),(NHCOCH (iPr)OH,Ph,Ph,CH2OH,CONHPh),(NHCOCH(iPr)OH,Ph,Ph,CH2OH,CONH-3-pyridyl),(N- HCOCH(iPr)OH,Ph,Ph,CH2OH,NHCOPh),(NHCOCH(iPr) OH,Ph,Ph,CH2OH,NHCO-2-furyl),(NHCOCH(iPr)OH,Ph,Ph,CH2OH,NHCONHPh),(NHCOCH- (iPr) OH,Ph,Ph,CH2OH, NHCOCONHPh),(NHCOCH(iPr)OH,Ph,OH,Me,CONHPh),(NHCOCH(iPr)OH,Ph, OH,Me,CONH-3-pyridy),(NHCOCH(iPr)OH,Ph,OH,Me,NHCOPh),(NHCOCH(iPr)OH,Ph,OH- ,Me,NHCO-2-furyl),(NHCOCH(iPr)OH,Ph,OH,Me,NHCONHPh),(NHCOCH(iPr)OH,Ph,OH,M- e,NHCOCONHPh),(NHCOCH(iPr)OH,Ph,OH,Et,CONHPh),(NH C OCH (iPr) OH,Ph,OH,Et,CONH-3-pyridyl),(NHCOCH(iPr)OH,Ph,OH,Et,NHCOPh),(NHCOCH(iPr)O- H,Ph,OH,Et,NHCO-2-furyl),(NHCOCH(iPr)OH,Ph,OH,Et,NHCONHPh),(NHCOCH(iPr)OH,- Ph,OH,Et,NHCOCONHPh),(NHCOCH(iPr)OH,Ph,OH,CH2OH,CONHPh),(NHCOCH(iPr)OH,Ph,- OH,CH2OH,CONH-3-pyridyl),(NHCOCH(iPr)OH,Ph,OH,CH2OH,NHCOPh),(NHCOCH(iPr) OH,Ph,OH,CH2OH,NHCO-2-furyl),(NHCOCH(iPr)OH,Ph,OH,CH2OH,NHCONHPh),(NHCOCH- (iPr)OH,Ph,OH,CH2OH,NHCOCONHPh), (NHSO2Me,H,H,Me,CONHPh),(NHSO2Me,H,H,Me,CONH-3-pyridyl),(NHSO2Me, H,H,Me,NHCOPh),(NHSO2Me,H,H,Me,NHCO-2-furyl),(NHSO2Me,H,H,Me,NHCONHPh),(N- HSO2Me,H,H,Me,NHCOCONHPh),(NHSO2Me,H,H,Et,CONHPh),(NHSO2Me,H,H,Et,CONH-3-p- yridyl),(NHSO2Me,H,H,Et,NHCOPh),(NHSO2Me,H,H,Et, NHCO-2-furyl),(NHSO2Me,H,H,Et,NHCONHPh),(NHSO2Me,H,H,Et,NHCOCONHPh),(NHSO- 2Me,H,H,CH2OH,CONHPh),(NHSO2Me,H,H,CH2OH,CONH-3-pyridyl),(NHSO2Me,H,H,CH2O- H,NHCOPh),(NHSO2Me,H,H,CH2OH,NHCO-2-furyl),(NHSO2Me,H,H,CH2OH,NHCONHPh),(N- HSO2Me,H,H,CH2OH,NHCOCONHPh),(NHSO2Me,H,Me,Me,CONHPh),(NHSO2Me,H,Me,Me,CON- H-3-pyridyl),(NHSO2Me, H,Me,Me,NHCOPh),(NHSO2Me,H,Me,Me,NHCO-2-furyl),(NHSO2Me,H,Me,Me,NHCONHPh)- ,(NHSO2Me,H,Me,Me,NHCOCONHPh),(NHSO2Me,H,Me,Et,CONHPh),(NHSO2Me,H,Me,Et,CO- NH-3-pyridyl),(NHSO2Me,H,Me,Et,NHCOPh),(NHSO2Me,H,Me,Et,NHCO-2-furyl),(NHS- O2Me,H,Me,Et,NHCONHPh),(NHSO2Me,H,Me,Et,NHCOCONHPh),(NHSO2Me,H,Me,CH2OH,CO- NHPh),(NHSO2Me,H,Me,CH2OH, CONH-3-pyridyl),(NHSO2Me,H,Me,CH2OH,NHCOPh),(NHSO2Me,H,Me,CH2OH, NHCO-2-furyl),(NHSO2Me,H,Me,CH2OH,NHCONHPh),(NHSO2Me,H,Me,CH2OH,NHCOCONHP- h),(NHSO2Me,H,Ph,Me,CONHPh),(NHSO2Me,H,Ph,Me,CONH-3-pyridyl),(NHSO2Me,H,Ph- ,Me,NHCOPh),(NHSO2Me,H,Ph,Me,NHCO-2-furyl),(NHSO2Me,H,Ph,Me,NHCONHPh),(NHS- O2Me,H,Ph,Me,NHCOCONHPh),(NHSO2Me, H,Ph,Et,CONHPh),(NHSO2Me,H,Ph,Et,CONH-3-pyridyl),(NHSO2Me,H,Ph,Et,NHCOPh)- ,(NHSO2Me,H,Ph,Et,NHCO-2-furyl),(NHSO2Me,H,Ph,Et,NHCONHPh),(NHSO2Me,H,Ph,E- t,NHCOCONHPh),(NHSO2Me,H,Ph,CH2OH,CONHPh),(NHSO2Me,H,Ph,CH2OH,CONH-3-pyrid- yl),(NHSO2Me,H,Ph,CH2OH,NHCOPh),(NHSO2Me,H,Ph,CH2OH,NHCO-2-furyl),(NHSO2Me- ,H,Ph,CH2OH,NHCONHPh),(NHSO2Me,H,Ph,CH2OH,NHCOCONHPh),(NHSO2Me,H,OH,Me,CON- HPh),(NHSO2Me,H,OH,Me,CONH-3-pyridyl),(NHSO2Me,H,OH,Me,NHCOPh),(NHSO2Me,H,- OH,Me, NHCO-2-furyl),(NHSO2Me,H,OH,Me,NHCONHPh),(NHSO2Me,H,OH,Me,NHCOCONHP- h),(NHSO2Me,H,OH,Et,CONHPh),(NHSO2Me,H,OH,Et,CONH-3-pyridyl),(NHSO2Me,H,OH- ,Et,NHCOPh),(NHSO2Me,H,OH,Et,NHCO-2-furyl),(NHSO2Me,H,OH,Et,NHCONHPh),(NHS- O2Me,H,OH,Et,NHCOCONHPh),(NHSO2Me,H,OH,CH2OH,CONHPh),(NHSO2Me,H,OH,CH2OH,C- ONH-3-pyridyl),(NHSO2Me,H,OH,CH2OH,NHCOPh),(NHSO2Me,H,OH,CH2OH,NHCO-2-fury- l),(NHSO2Me,H,OH,CH2OH,NHCONHPh),(NHSO2Me,H,OH,CH2OH,NHCOCONHPh),(NHSO2Me,- Me,H, Me,CONHPh),(NHSO2Me,Me,H,Me,CONH-3-pyridyl),(NHSO2Me,Me,H,Me,NHCOPh)- ,(NHSO2Me,Me,H,Me,NHCO-2-furyl),(NHSO2Me,Me,H,Me,NHCONHPh),(NHSO2Me,Me,H,M- e,NHCOCONHPh),(NHSO2Me,Me,H,Et,CONHPh),(NHSO2Me,Me,H,Et,CONH-3-pyridyl),(N- HSO2Me,Me,H,Et,NHCOPh),(NHSO2Me,Me,H,Et,NHCO-2-furyl),(NHSO2Me,Me,H,Et,NHC- ONHPh),(NHSO2Me,Me,H,Et,NHCOCONHPh),(NHSO2Me,Me,H,CH2OH,CONHPh),(NHSO2Me,M- e,H,CH2OH,CONH-3-pyridyl),(NHSO2Me,Me,H,CH2OH,NHCOPh),(NHSO2Me,Me,H,CH2OH,- NHCO-2-furyl),(NHSO2Me,Me,H,CH2OH,NHCONHPh),(NHSO2Me,Me,H,CH2OH,NHCOCONHPh- ),(NHSO2Me,Me,Me,Me,CONHPh),(NHSO2Me,Me,Me,Me,CONH-3-pyridyl),(NHSO2Me,Me,- Me,Me,NHCOPh),(NHSO2Me,Me,Me,Me,NHCO-2-furyl),(NHSO2Me,Me,Me,Me,NHCONHPh),- (NHSO2Me,Me,Me,Me,NHCOCONHPh),(NHSO2Me,Me,Me,Et,CONHPh),(NHSO2Me,Me,Me,Et,- CONH-3-pyridyl),(NHSO2Me,Me,Me,Et,NHCOPh),(NHSO2Me,Me,Me,Et,NHCO-2-furyl),- (NHSO2Me,Me,Me,Et,NHCONHPh),(NHSO2Me,Me,Me,Et,NHCOCONHPh),(NHSO2Me,Me,Me,C- H2OH,CONHPh),(NHSO2Me,Me,Me,CH2OH,CONH-3-pyridyl),(NHSO2Me,Me,Me,CH2OH,NHC- OPh),(NHSO2Me,Me,Me,CH2OH,NHCO-2-furyl),(NHSO2Me,Me,Me,CH2OH,NHCONHPh),(NH- SO2Me,Me,Me,CH2OH,NHCOCONHPh),(NHSO2Me,Me,Ph,Me,CONHPh),(NHSO2Me,Me,Ph,Me,- CONH-3-pyridyl),(NHSO2Me,Me,Ph,Me,NHCOPh),(NHSO2Me,Me,Ph,Me,NHCO-2-furyl),- (NHSO2Me,Me,Ph,Me,NHCONHPh),(NHSO2Me,Me,Ph,Me,NHCOCONHPh),(NHSO2Me,Me,Ph,E- t,CONHPh),(NHSO2Me,Me,Ph,Et,CONH-3-pyridyl),(NHSO2Me,Me,Ph,Et,NHCOPh),(NHS- O2Me,Me,Ph,Et,NHCO-2-furyl),(NHSO2Me,Me,Ph,Et,NHCONHPh),(NHSO2Me,Me,Ph,Et,- NHCOCONHPh),(NHSO2Me,Me,Ph,CH2OH,CONHPh),(NHSO2Me,Me,Ph,CH2OH,CONH-3-pyrid- yl),(NHSO2Me,Me,Ph,CH2OH,NHCOPh),(NHSO2Me,Me,Ph,CH2OH,NHCO-2-furyl),(NHSO2- Me,Me,Ph,CH2OH,NHCONHPh),(NHSO2Me,Me,Ph,CH2OH,NHCOCONHPh),(NHSO2Me,Me,OH,M- e,CONHPh),(NHSO2Me,Me,OH,Me,CONH-3-pyridyl),(NHSO2Me,Me,OH,Me,NHCOPh),(NHS- O2Me,Me,OH,Me,NHCO-2-furyl),(NHSO2Me,Me,OH,Me,NHCONHPh),(NHSO2Me,Me,OH,Me,- NHCOCONHPh),(NHSO2Me,Me,OH,Et,CONHPh),(NHSO2Me,Me,OH,Et,CONH-3-pyridyl),(N- HSO2Me,Me,OH,Et, NHCOPh),(NHSO2Me,Me,OH,Et,NHCO-2-furyl),(NHSO2Me,Me,OH,Et,NHCONHPh),(NHSO- 2Me,Me,OH,Et,NHCOCONHPh),(NHSO2Me,Me,OH,CH2OH,CONHPh),(NHSO2Me,Me,OH,CH2OH- ,CONH-3-pyridyl),(NHSO2Me,Me,OH,CH2OH,NHCO Ph),(NHSO2Me,Me,OH,CH2OH,NHCO-2-furyl),(NHSO2Me,Me,OH,CH2OH,NHCONHPh),(NH- SO2Me,Me,OH,CH2OH,NHCOCONHPh),(NHSO2Me,Ph,H,Me,CONHPh),(NHSO2Me,Ph,H,Me,CO- NH-3-pyridyl),(NHSO2Me,Ph,H,Me,NHCOPh),(NHSO2Me,Ph,H,Me,NHCO-2-furyl),(NHS- O2Me,Ph,H,Me,NHCONHPh),(NHSO2Me,Ph,H,Me,NHCOCONHPh),(NHSO2Me,Ph,H,Et,CONHP- h),(NHSO2Me,Ph,H,Et,CONH-3-pyridyl),(NHSO2Me,Ph,H,Et,NHCOPh),(NHSO2Me,Ph,H- ,Et,NHCO2-furyl), (NHSO2Me,Ph,H,Et,NHCONHPh),(NHSO2Me,Ph,H,Et,NHCOCONHPh),(NHSO2Me,Ph,H,CH2- OH,CONHPh),(NHSO2Me,Ph,H,CH2OH,CONH-3-pyridyl),(NHSO2Me,Ph,H,CH2OH,NHCOPh)- ,(NHSO2Me,Ph,H,CH2OH,NHCO-2-furyl),(NHSO2Me, Ph,H,CH2OH,NHCONHPh),(NHSO2Me,Ph,H,CH2OH,NHCO CONHPh),(NHSO2Me,Ph,Me,Me,CONHPh),(NHSO2Me,Ph,Me,Me,CONH-3-pyridyl),(NHSO- 2Me,Ph, Me,Me,NHCOPh),(NHSO2Me,Ph,Me,Me,NHCO-2-furyl),(NHSO2Me,Ph,Me,Me,NH- CONPh),(NHSO2Me,Ph,Me,Me,NHCOCONHPh),(NHSO2Me,Ph,Me,Et,CONHPh),(NHSO2Me,Ph- ,Me,Et,CONH-3-pyridyl),(NHSO2Me,Ph,Me,Et,NHCOPh),(NHSO2Me,Ph,Me,Et,NHCO2-f- uryl),(NHSO2Me,Ph,Me,Et,NHCONHPh),(NHSO2Me,Ph,Me,Et,NHCOCONHPh),(NHSO2Me,P- h,Me,CH2OH,CONHPh),(NHSO2Me,Ph,Me,CH2OH,CONH-3-pyridyl),(NHSO2Me,Ph,Me,CH2- OH,NHCOPh),(NHSO2Me,Ph,Me,CH2OH,NHCO-2-furyl),(NHSO2Me,Ph,Me,CH2OH,NHCONHP- h),(NHSO2Me, Ph,Me,CH2OH,NHCOCONHPh),(NHSO2Me,Ph,Ph,Me,CONHPh),(NHSO2Me,Ph, Ph,Me,CONH-3-pyridyl),(NHSO2Me,Ph,Ph,Me,NHCOPh),(NHSO2Me,Ph,Ph,Me,NHCO-2-- furyl),(NHSO2Me,Ph,Ph,Me,NHCONHPh),(NHSO2Me,Ph,Ph,Me,NHCOCONHPh),(NHSO2Me,- Ph,Ph,Et,CONHPh),(NHSO2Me,Ph,Ph,Et,CONH-3-pyridyl),(NHSO2Me,Ph,Ph,Et,NHCOP- h),(NHSO2Me,Ph,Ph,Et,NHCO-2-furyl),(NHSO2Me,Ph,Ph,Et,NHCONHPh),(NHSO2Me,Ph- ,Ph,Et,NHCOCONHPh),(NHSO2Me,Ph,Ph,CH2OH,CONHPh),(NHSO2Me,Ph,Ph,CH2OH,CONH-- 3-pyridyl),(NHSO2Me,Ph,Ph, CH2OH,NHCOPh),(NHSO2Me,Ph,Ph,CH2OH,NHCO-2-furyl),(NHSO2Me,Ph,Ph,CH2OH,NHC- ONHPh),(NHSO2Me,Ph,Ph,CH2OH,NHCOCONHPh),(NHSO2Me,Ph, OH,Me,CONHPh),(NHSO2Me,Ph,OH,Me,CONH-3-pyridyl),(NHSO2Me,Ph,OH,Me, NHCOPh),(NHSO2Me,Ph,OH,Me,NHCO-2-furyl),(NHSO2Me,Ph,OH,Me,NHCONHPh),(NHSO- 2Me,Ph,OH,Me,NHCOCONHPh),(NHSO2Me,Ph,OH,Et,CONHPh),(NHSO2Me,Ph,OH,Et,CONH-- 3-pyridyl),(NHSO2Me,Ph,OH,Et,NHCOPh),(NHSO2Me,Ph,OH,Et,NHCO-2-furyl),(NHSO- 2Me,Ph,OH,Et,NHCONHPh),(NHSO2Me,Ph,OH, Et,NHCOCONHPh),(NHSO2Me,Ph,OH,CH2OH,CONHPh),(NHSO2Me,Ph,OH,CH2OH,CONH-3-p- yridyl),(NHSO2Me,Ph,OH,CH2OH,NHCOPh),(NHSO2Me,Ph,OH, CH2OH,NHCO-2-furyl),(NHSO2Me,Ph,OH,CH2OH,NHCONHPh),(NHSO2Me,Ph, OH,CH2OH,NHCO CONHPh), (NH2,H,H,Me,CONHPh),(NH2,H,H,Me,CONH-3-pyridyl),(NH2,H,H,Me,NHCOPh), (NH2,H,H,Me,NHCO-2-furyl),(NH2,H,H,Me,NHCONHPh),(NH2,H,H,Me,NHCOCONPh),(N- H2,H,H,Et,CONHPh),(NH2,H,H,Et,CONH-3-pyridyl),(NH12,H,H,Et,NHCOPh),(NH2,H,- H,Et,NHCO-2-furyl),(NH12,H,H,Et,NHCONHPh),(NH2,H,H,Et,NHCOCONHPh),(NH2,H,H- ,CH2OH,CONHPh),(NH2,H,H,CH2OH,CONH-3-pyridyl),(NH2,H,H,CH2OH,NHCONHPh),(NH- 2,H,H,CH2OH,NHCOCONHPh),(NH2,H,Me, Me,CONHPh),(NH2,H,Me,Me,CONH-3-pyridyl),(NH2,H,Me,Me,NHCONHPh),(NH2,H,Me,- Me,NHCOCONHPh),(NH2,H,Me,Et,CONHPh),(NH2,H,Me,Et,CONH-3-pyridyl),(NH2,H,Me- ,Et,NHCOPh),(NH2,H,Me,Et,NHCO-2-fury),(NH2,H,Me,Et,NHCONHPh),(NH2,H,Me,Et,- NHCOCONHPh),(NH2,H,Me,CH2OH,CONHPh),(NH12,H, Me,CH2OH,CONH-3-pyridyl),(NH2,H,Me,CH2OH,NHCONHPh),(NH2,H,Me,CH2OH,NHCO CONHPh),(NH2,H,Ph,Me,CONHPh),(NH2,H,Ph,Me,CONH-3-pyridyl),(NH2,H,Ph,Me,NH- CONHPh),(NH2,H,Ph,Me,NHCOCONHPh),(NH2,H,Ph,Et,CONHPh),(NH2,H,Ph,Et,CONH-3-- pyridyl),(NH2,H,Ph,Et,NHCOPh),(NH2,H,Ph,Et,NHCO-2-furyl,(NH2,H,Ph,Et,NHCON- HPh),(NH2,H,Ph,Et,NHCOCONHPh),(NH2,H, Ph,CH2OH,CONHPh),(NH2,H,Ph,CH2OH,CONH-3-pyridyl),(NH2,H,Ph,CH2OH,NHCONHPh- ),(NH2,H,Ph,CH2OH,NHCOCONHPh),(NH2,H,OH,Me,CONHPh),(NH2,H,OH,Me,CONH-3-pyr- idyl),(NH2,H,OH,Me,NHCONHPh),(NIH2,H,OH,Me,NHCOCONHPh),(NH2,H,OH,Et,CONHPh- ),(NH2,H,OH,Et,CONH-3-pyridyl),(NH2,H,OH, Et,NHCOPh),(NH2,H,OH,Et,NHCO-2-furyl),(NH2,H,OH,Et,NHCONHPh),(NH2,H,OH,Et- ,NHCOCONHPh),(NH2,H,OH,CH2OH,CONHPh),(NH2,H,OH,CH2OH,CONH-3-pyridyl),(NH2,- H,OH,CH2OH,NHCONHPh),(NH2,H,OH,CH2OH,NHCOCONHPh),(NH2,Me,H,Me,CONHPh),(NH1- 2,Me,H,Me,CONH-3-pyridyl),(NH2,Me,H,Me, NHCONHPh),(NH12,Me,H,Me,NHCOCONHPh),(NH2,Me,H,Et,CONHPh),(NH2,Me,H,Et,CON- H-3-pyridyl),(NH12,Me,H,Et,NHCOPh),(NH2,Me,H,Et,NHCO-2-fury),(NH2,Me,H,Et,- NHCONHPh),(NH2,Me,H,Et,NHCOCONHPh),(NH2,Me,H,CH2OH,CONHPh),(NH2,Me,H,CH2OH- ,CONH-3-pyridy),(NH12,Me,H,CH2OH,NHCONHPh), (NH2,Me,H,CH2OH,NHCOCONHPh),(NH2,Me,Me,Me,CONHPh),(NH2,Me,Me,Me,CONH-3-py- ridyl),(NH2,Me,Me,Me,NHCONHPh),(NH2,Me,Me,Me,NHCOCONHPh),(NH2,Me,Me,Et,CON- HPh),(NH2,Me,Me,Et,CONH-3-pyridyl),(NH2,Me,Me,Et,NHCOPh),(NH2,Me,Me,Et,NHC- O-2-furyl),(NH2,Me,Me,Et,NHCONHPh),(NH2,Me, Me,Et,NHCOCONHPh),(NH2,Me,Me,CH2OH,CONHPh),(NH2,Me,Me,CH2OH,CONH-3-pyridy- l),(NH2,Me,Me,CH2OH,NHCONHPh),(NH2,Me,Me,CH2OH,NHCOCONHPh),(NH2,Me,Ph,Me,C- ONHPh),(NH2,Me,Ph,Me,CONH-3-pyridyl),(NH2,Me,Ph, Me,NHCOPh),(NH2,Me,Ph,Me,NHCO-2-furyl),(NH2,Me,Ph,Me,NHCONHPh),(NH2,Me,Ph- ,Me,NHCOCONHPh),(NH2,Me,Ph,Et,CONHPh),(NH2,Me,Ph,Et,CONH-3-pyridyl),(NH2,M- e,Ph,Et,NHCOPh),(NH2,Me,Ph,Et,NHCO-2-furyl),(NH2,Me,Ph,Et, NHCONHPh),(NH2,Me,Ph,Et,NHCOCONHPh),(NH2,Me,Ph,CH2OH,CONHPh),(NH2,Me,Ph,C- H2OH,CONH-3-pyridyl),(NH2,Me,Ph,CH2OH,NHCONHPh),(NH2,Me, Ph,CH2OH,NHCOCONHPh),(NH2,Me,OH,Me,CONHPh),(NH2,Me,OH,Me,CONH-3-pyridyl),- (NH2,Me,OH,Me,NHCONHPh),(NH2,Me,OH,Me,NHCOCONHPh),(NH2,Me,OH,Et,CONHPh),(N- H2,Me,OH,Et,CONH-3-pyridyl),(NH2,Me,OH,Et,NHCOPh),(NH2,Me,OH,Et,NHCO-2-fur- yl),(NH2,Me,OH,Et,NHCONHPh),(NH2,Me,OH,Et, NHCOCONHPh),(NH2,Me,OH,CH2OH,CONHPh),(NH2,Me,OH,CH2OH,CONH-3-pyridyl),(NH- 2,Me,OH,CH2OH,NHCONHPh),(NH2,Me,OH,CH2OH,NHCOCONHPh),(NH2,Ph,H,Me,CONHPh),- (NH12,Ph,H,Me,CONH-3-pyridyl),(NH2,Ph,H,Me,NHCONHPh),(NH2,Ph,H,Me,NHCOCONH- Ph),(NH2,Ph,H,Et,CONHPh),(NH2,Ph,H,Et,CONH-3-pyridyl),(NH2,Ph,H,Et,NHCOPh)- ,(NH12,Ph,H,Et,NHCO-2-fury),(NH2,Ph,H,Et,NHCONHPh),(NH2,Ph,H,Et,NHCOCONHPh- ),(NH2,Ph,H,CH2OH,CONHPh),(NH2,Ph,H,CH2OH,CONH-3-pyridyl),(NH2,Ph,H,CH2OH,- NHCONHPh),(NH2,Ph,H,CH2OH,NHCOCONHPh),(NH2,Ph,Me,Me,CONHPh),(NH2,Ph,Me,Me,- CONH-3-pyridyl),(NH2,Ph,Me,Me,NHCONHPh),(NH2,Ph,Me,Me,NHCOCONHPh),(NH2, Ph,Me,Et,CONHPh),(NH2,Ph,Me,Et,CONH-3-pyridyl),(NH2,Ph,Me,Et,NHCOPh),(NH2- ,Ph,Me,Et,NHCO-2-furyl),(NH2,Ph,Me,Et,NHCONHPh),(NH2,Ph,Me,Et,NHCOCONHPh),- (NH12,Ph,Me,CH2OH,CONHPh),(NH2,Ph,Me,CH2OH,CONH-3-pyridyl),(NH2,Ph,Me,CH2O- H,NHCONHPh),(NH2,Ph,Me,CH2OH,NHCOCONHPh),(NH2,Ph,Ph,Me,CONHPh),(NH2,Ph,Ph,- Me,CONH-3-pyridyl),(NH2,Ph,Ph,Me,NHCOPh),(NH2,Ph,Ph,Me,NHCO-2-furyl),(NH2,- Ph,Ph,Me,NHCONHPh),(NH2,Ph,Ph,Me,NHCOCONHPh),(NH2,Ph,Ph,Et,CONHPh),(NIH2,P- h,Ph,Et,CONH-3-pyridy),(NH2, Ph,Ph,Et,NHCOPh),(NH2,Ph,Ph,Et,NHCO-2-furyl),(NH2,Ph,Ph,Et,NHCONHPh),(NH2- ,Ph,Ph,Et,NHCOCONHPh),(NH2,Ph,Ph,CH2OH,CONHPh),(NH2,Ph,Ph,CH2OH,CONH-3-pyr-
idyl),(NH2,Ph,Ph,CH2OH,NHCOPh),(NH2,Ph,Ph,CH2OH,NHCO-2-furyl),(NH2,Ph,Ph,C- H2OH,NHCONHPh),(NH2,Ph,Ph,CH2OH,NHCOCONPh), (NIH2,Ph,OH,Me,CONHPh),(NH2,Ph,OH,Me,CONH-3-pyridyl),(NH2,Ph,OH,Me,NHCONH- Ph),(NH2,Ph,OH,Me,NHCOCONHPh),(NH2,Ph,OH,Et,CONHPh),(NH2,Ph,OH,Et,CONH-3-p- yridyl),(NH2,Ph,OH,Et,NHCOPh),(NH2,Ph,OH,Et,NHCO-2-furyl),(NH2,Ph,OH,Et,NH- CONPh),(NH2,Ph,OH,Et,NHOCONHPh),(NH2,Ph,OH,CH2OH,CONHPh),(NH2,Ph,OH,CH2OH,- CONH-3-pyridyl),(NH2,Ph,OH,CH2OH,NHCOPh),(NH2,Ph,OH,CH2OH,NHCO-2-furyl),(N- H2,Ph,OH,CH2OH,NCONHPh), (NH2,Ph,OH,CH2OH,NHCOCONHPh), (NHCH2CH(OH)CH2OH,H,H,Me,CONHPh),(NHCH2CH(OH)CH2OH,H,H,Me,CONH-3-pyridyl)- ,(NHCH2CH(OH)CH2OH,H,H,Me,NHCOPh),(NHCH2CH(OH)CH2OH,H,H,Me,NHCO-2-furyl),(- NHCH2CH(OH)CH2OH,H,H,Me,NHCONHPh),(NHCH2CH(OH)CH2OH,H,H,Me,NHCOCONHPh),(NH- CH2CH(OH)CH2OMe,H,H,Me,CONHPh),(NHCH2CH(OH)CH2OMe,H,H,Me,CONH-3-pyridy),(N- HCH2CH(OH)CH2OMe,H,H,Me,NHCOPh),(NHCH2CH(OH)CH2OMe,H,H,Me,NHCO-2-furyl),(N- HCH2CH(OH)CH2OMe,H,H,Me,NHCONHPh),(NHCH2CH(OH)CH2OMe,H,H,Me,NHCOCONHPh),(N- HCH2CH(OH)CH2NH2,H,H,Me,CONHPh),(NHCH2CH(OH)CH2NH2,H,H,Me,CONH-3-pyridyl),- (NHCH2CH(OH)CH2NH2,H,H,Me,NHCOPh),(NHCH2CH(OH)CH2NH2,H,H,Me,NHCO-2-furyl),- (NHCH2CH(OH)CH2NH2,H,H,Me, NHCONHPh),(NHCH2CH(OH)CH2NH2,H,H,Me,NHCOCONHPh),(NHCH2CH(OH)CH2NHMe,H,H,M- e,CONHPh),(NHCH2CH(OH)CH2NHMe,H,H,Me,CONH-3-pyridyl),(NHCH2CH(OH)CH2NHMe,H- ,H,Me,NHCOPh),(NHCH2CH(OH)CH2NHMe, H,H,Me,NHCO-2-furyl),(NHCH2CH(OH)CH2NHMe,H,H,Me,NHCONHPh),(NHCH2CH(OH)CH2- NHMe,H,H,Me,NHCOCONHPh),(NHCH2CH(OH)CH2NHCOMe,H,H,Me,CONHPh),(NHCH2CH(OH)C- H2NHCOMe,H,H,Me,CONH-3-pyridyl),(NHCH2CH(OH)CH2NHCOMe,H,H,Me,NHCOPh),(NHCH- 2CH(OH)CH2NHCOMe,H,H, Me,NHCO-2-furyl),(NHCH2CH(OH)CH2NHCOMe,H,H,Me,NHCONHPh),(NHCH2CH(OH)CH2NH- COMe,H,H,Me,NHCOCONHPh),(NHCH2CH(OH)CH2N(Me)Me,H,H,Me,CONHPh),(NHCH2CH(OH)- CH2N(Me)Me,H,H,Me,CONH-3-pyridyl),(NHCH2CH(OH)OCH2N(Me)Me,H,H,Me,NHCOPh),(- NHCH2CH(OH)CH2N(Me)Me,H,H,Me,NHCO-2-furyl),(NHCH2CH(OH)CH2N(Me)Me,H,H,Me,N- HCONHPh),(NHCH2CH(OH)CH2N(Me)Me,H,H,Me,NHCOCONHPh),(NHC(O)C(O)NH2,H,H,Me,C- ONHPh),(NHC(O)C(O)NH2,H,H,Me,CONH-3-pyridy),(NHC(O) C(O)NH2,H,H,Me,NHCOPh),(NHC(O)C(O)NH2,H,H,Me,NHCO-2-furyl),(NHC(O)C(O)NH2- ,H,H,Me,NHCONHPh),(NHC(O)C(O)NH2,H,H,Me,NHCOCONHPh),(NC(O)C(O)NHMe,H,H,Me,- CONHPh),(NHC(O)C(O)NHMe,H,H,Me,CONH-3-pyridyl),(NHC(O)C(O)NHMe,H,H, Me,NHCOPh),(NHC(O)C(O)NHMe,H,H,Me,NHCO-2-furyl),(NHC(O)C(O)NHMe,H, H,Me,NHCONHPh),(NHC(O) C(O)NHMe,H,H,Me,NHCOCONHPh),(NHC(O)C(O)N(Me)Me,H,H,Me,CONHPh),(NHC(O)C(O)- N(Me)Me,H,H,Me,CONH-3-pyridyl),(NHC(O) C(O)N(Me)Me,H,H,Me,NHCOPh),(NHC(O)C(O)N(Me)Me,H,H,Me,NHCO-2-furyl),(NHC(O- )C(O)N(Me)Me,H,H,Me,NHCONHP),(NHC(O)C(O)N(Me)Me,H,H,Me,NHCOCONHPh).
##STR01358##
[0299] In above structural formula (Ii) or (Ij), the combination of B, Linker, A, R5 (B, Linker, A, R5) are the following compounds.
TABLE-US-00169 TABLE 169 B B1 Ph-- B2 2-piridyl- B3 4-Me--Ph-- B4 cHex-- B5 cHex--CH2-- Linker L1 ##STR01359## L2 ##STR01360## L3 ##STR01361## L4 ##STR01362## L5 ##STR01363## A A1 ##STR01364## A2 ##STR01365## A3 ##STR01366## A4 ##STR01367## A5 ##STR01368## R5 R51 --CN R52 --C≡CH R53 --C≡CMe R54 --CF3 R55 --CH2Cl R56 CHCl2
(B, Linker, A, R5)=(B1,L1,A1,R51),(B1,L1,A1,R52),(B1,L1,A1,R53),(B1,L1,A1,R54),(B1,- L1,A1,R55),(B1,L1,A1,R56),(B1,L1,A2,R51),(B1,L1,A2,R52),(B1,L1,A2,R53),(B1- ,L1,A2,R54),(B1,L1,A2,R55),(B1,L1,A2,R56),(B1,L1,A3,R51),(B1,L1,A3,R52),(B- 1,L1,A3,R53),(B1,L1,A3,R54),(B1,L1,A3,R55),(B1,L1,A3,R56),(B1,L1,A4,R51),(- B1,L1,A4,R52),(B1,L1,A4,R53),(B1,L1,A4,R54),(B1,L1,A4,R55),(B1,L1,A4,R56),- (B1,L1,A5,R51),(B1,L1,A5,R52),(B1,L1,A5,R53),(B1,L1,A5,R54),(B1,L1,A5,R55)- ,(B1,L1,A5,R56),(B1,L2,A1,R51),(B1, L2,A1,R52),(B1,L2,A1,R53),(B1,L2,A1,R54),(B1,L2,A1,R55),(B1,L2,A1,R56),(B- 1,L2, A2,R51),(B1,L2,A2,R52),(B1,L2,A2,R53),(B1,L2,A2,R54),(B1,L2,A2,R55),- (B1,L2,A2, R56),(B1,L2,A3,R51),(B1,L2,A3,R52),(B1,L2,A3,R53),(B1,L2,A3,R54- ),(B1,L2,A3,R55),(B1,L2,A3,R56),(B1,L2,A4,R51),(B1,L2,A4,R52),(B1,L2,A4,R5- 3),(B1,L2,A4,R54),(B1,L2,A4,R55),(B1,L2,A4,R56),(B1,L2,A5,R51),(B1,L2,A5,R- 52),(B1,L2,A5,R53),(B1, L2,A5,R54),(B1,L2,A5,R55),(B1,L2,A5,R56),(B1,L3,A1,R51),(B1,L3,A1,R52),(B- 1,L3, A1,R53),(B1,L3,A1,R54),(B1,L3,A1,R55),(B1,L3,A1,R56),(B1,L3,A2,R51),- (B1,L3,A2, R52),(B1,L3,A2,R53),(B1,L3,A2,R54),(B1,L3,A2,R55),(B1,L3,A2,R56- ),(B1,L3,A3,R51),(B1,L3,A3,R52,(B1,L3,A3,R53),(B1,L3,A3,R54),(B1,L3,A3,R55- ),(B1,L3,A3,R56),(B1,L3,A4,R51),(B1,L3,A4,R52),(B1,L3,A4,R53),(B1,L3,A4,R5- 4),(B1,L3,A4,R55),(B1, L3,A4,R56),(B1,L3,A5,R51),(B1,L3,A5,R52),(B1,L3,A5,R53),(B1,L3,A5,R54),(B- 1,L3, A5,R55),(B1,L3,A5,R56),(B1,L4,A1,R51),(B1,L4,A1,R52),(B1,L4,A1,R53),- (B1,L4,A1, R54),(B1,L4,A1,R55),(B1,L4,A1,R56),(B1,L4,A2,R51),(B1,L4,A2,R52- ),(B1,L4,A2,R53),(B1,L4,A2,R54),(B1,L4,A2,R55),(B1,L4,A2,R56),(B1,L4,A3,R5- 1),(B1,L4,A3,R52),(B1,L4,A3,R53),(B1,L4,A3,R54),(B1,L4,A3,R55),(B1,L4,A3,R- 56),(B1,L4,A4,R51),(B1, L4,A4,R52),(B1,L4,A4,R53),(B1,L4,A4,R54),(B1,L4,A4,R55),(B1,L4,A4,R56),(B- 1,L4, A5,R51),(B1,L4,A5,R52),(B1,L4,A5,R53),(B1,L4,A5,R54),(B1,L4,A5,R55),- (B1,L4,A5, R56),(B1,L5,A1,R51),(B1,L5,A1,R52),(B1,L5,A1,R53),(B1,L5,A1,R54- ),(B1,L5,A1,R55),(B1,L5,A1,R56),(B1,L5,A2,R51),(B1,L5,A2,R52),(B1,L5,A2,R5- 3),(B1,L5,A2,R54),(B1,L5,A2,R55),(B1,L5,A2,R56),(B1,L5,A3,R51),(B1,L5,A3,R- 52),(B1,L5,A3,R53),(B1, L5,A3,R54),(B1,L5,A3,R55),(B1,L5,A3,R56),(B1,L5,A4,R51),(B1,L5,A4,R52),(B- 1,L5, A4,R53),(B1,L5,A4,R54),(B1,L5,A4,R55),(B1,L5,A4,R56),(B1,L5,A5,R51),- (B1,L5,A5, R52),(B1,L5,A5,R53),(B1,L5,A5,R54),(B1,L5,A5,R55),(B1,L5,A5,R56- ),(B2,L1,A1,R51),(B2,L1,A1,R52),(B2,L1,A1,R53),(B2,L1,A1,R54),(B2,L1,A1,R5- 5),(B2,L1,A1,R56),(B2,L1,A2,R51),(B2,L1,A2,R52),(B2,L1,A2,R53),(B2,L1,A2,R- 54),(B2,L1,A2,R55),(B2, L1,A2,R56),(B2,L1,A3,R51),(B2,L1,A3,R52),(B2,L1,A3,R53),(B2,L1,A3,R54),(B- 2,L1, A3,R55),(B2,L1,A3,R56),(B2,L1,A4,R51),(B2,L1,A4,R52),(B2,L1,A4,R53),- (B2,L1,A4, R54),(B2,L1,A4,R55),(B2,L1,A4,R56),(B2,L1,A5,R51),(B2,L1,A5,R52- ),(B2,L1,A5,R53),(B2,L1,A5,R54),(B2,L1,A5,R55),(B2,L1,A5,R56),(B2,L2,A1,R5- 1),(B2,L2,A1,R52),(B2,L2,A1,R53),(B2,L2,A1,R54),(B2,L2,A1,R55),(B2,L2,A1,R- 56),(B2,L2,A2,R51),(B2, L2,A2,R52),(B2,L2,A2,R53),(B2,L2,A2,R54),(B2,L2,A2,R55),(B2,L2,A2,R56),(B- 2,L2, A3,R51),(B2,L2,A3,R52),(B2,L2,A3,R3),(B2,L2,A3,R54),(B2,L2,A3,R55),(- B2,L2,A3, R56),(B2,L2,A4,R51),(B2,L2,A4,R52),(B2,L2,A4,R53),(B2,L2,A4,R54)- ,(B2,L2,A4,R55),(B2,L2,A4,R56),(B2,L2,A5,R51),(B2,L2,A5,R52),(B2,L2,A5,R53- ),(B2,L2,A5,R54),(B2,L2,A5,R55),(B2,L2,A5,R56),(B2,L3,A1,R51),(B2,L3,A1,R5- 2),(B2,L3,A1,R53),(B2, L3,A1,R54),(B2,L3,A1,R55),(B2,L3,A1,R56),(B2,L3,A2,R51),(B2,L3,A2,R52),(B- 2,L3, A2,R53),(B2,L3,A2,R54),(B2,L3,A2,R55),(B2,L3,A2,R56),(B2,L3,A3,R51),- (B2,L3,A3, R52),(B2,L3,A3,R53),(B2,L3,A3,R54),(B2,L3,A3,R55),(B2,L3,A3,R56- ),(B2,L3,A4,R51),(B2,L3,A4,R52),(B2,L3,A4,R53),(B2,L3,A4,R54),(B2,L3,A4,R5- 5),(B2,L3,A4,R56),(B2,L3,A5,R51),(B2,L3,A5,R52),(B2,L3,A5,R53),(B2,L3,A5,R- 54),(B2,L3,A5,R55),(B2, L3,A5,R56),(B2,L4,A1,R51),(B2,L4,A1,R52),(B2,L4,A1,R53),(B2,L4,A1,R54),(B- 2,L4, A1,R55),(B2,L4,A1,R56),(B2,L4,A2,R51),(B2,L4,A2,R52),(B2,L4,A2,R53),- (B2,L4,A2, R54),(B2,L4,A2,R55),(B2,L4,A2,R56),(B2,L4,A3,R51),(B2,L4,A3,R52- ),(B2,L4,A3,R53),(B2,L4,A3,R54),(B2,L4,A3,R55),(B2,L4,A3,R56),(B2,L4,A4,R5- 1),(B2,L4,A4,R52),(B2,L4,A4,R53),(B2,L4,A4,R54),(B2,L4,A4,R55),(B2,L4,A4,R- 56),(B2,L4,A5,R51),(B2, L4,A5,R52),(B2,L4,A5,R53),(B2,L4,A5,R54),(B2,L4,A5,R55),(B2,L4,A5,R56),(B- 2,L5, A1,R51),(B2,L5,A1,R52),(B2,L5,A1,R53),(B2,L5,A1,R54),(B2,L5,A1,R55),- (B2,L5,A1, R56),(B2,L5,A2,R51),(B2,L5,A2,R52),(B2,L5,A2,R53),(B2,L5,A2,R54- ),(B2,L5,A2,R55),(B2,L5,A2,R56),(B2,L5,A3,R51),(B2,L5,A3,R52),(B2,L5,A3,R5- 3),(B2,L5,A3,R54),(B2,L5,A3,R55),(B2,L5,A3,R56),(B2,L5,A4,R51),(B2,L5,A4,R- 52),(B2,L5,A4,R53),(B2, L5,A4,R54),(B2,L5,A4,R55),(B2,L5,A4,R56),(B2,L5,A5,R51),(B2,L5,A5,R52),(B- 2,L5, A5,R53),(B2,L5,A5,R54),(B2,L5,A5,R55),(B2,L5,A5,R56),(B3,L1,A1,R51),- (B3,L1,A1, R52),(B3,L1,A1,R53),(B3,L1,A1,R54),(B3,L1,A1,R55),(B3,L1,A1,R56- ),(B3,L1,A2,R51),(B3,L1,A2,R52),(B3,L1,A2,R53),(B3,L1,A2,R54),(B3,L1,A2,R5- 5),(B3,L1,A2,R56),(B3,L1,A3,R51),(B3,L1,A3,R52),(B3,L1,A3,R53),(B3,L1,A3,R- 54),(B3,L1,A3,R55),(B3, L1,A3,R56),(B3,L1,A4,R51),(B3,L1,A4,R52),(B3,L1,A4,R53),(B3,L1,A4,R54),(B- 3,L1, A4,R55),(B3,L1,A4,R56),(B3,L1,A5,R51),(B3,L1,A5,R52),(B3,L1,A5,R53),- (B3,L1,A5, R54),(B3,L1,A5,R55),(B3,L1,A5,R56),(B3,L2,A1,R51),(B3,L2,A1,R52- ),(B3,L2,A1,R53),(B3,L2,A1,R54),(B3,L2,A1,R55),(B3,L2,A1,R56),(B3,L2,A2,R5- 1),(B3,L2,A2,R52),(B3,L2,A2,R53),(B3,L2,A2,R54),(B3,L2,A2,R55),(B3,L2,A2,R- 56),(B3,L2,A3,R51),(B3, L2,A3,R52),(B3,L2,A3,R53),(B3,L2,A3,R54),(B3,L2,A3,R55),(B3,L2,A3,R56),(B- 3,L2, A4,R51),(B3,L2,A4,R52),(B3,L2,A4,R53),(B3,L2,A4,R54),(B3,L2,A4,R55),- (B3,L2,A4, R56),(B3,L2,A5,R51),(B3,L2,A5,R52),(B3,L2,A5,R53),(B3,L2,A5,R54- ),(B3,L2,A5,R55),(B3,L2,A5,R56),(B3,L3,A1,R51),(B3,L3,A1,R52),(B3,L3,A1,R5- 3),(B3,L3,A1,R54),(B3,L3,A1,R55),(B3,L3,A1,R56),(B3,L3,A2,R51),(B3,L3,A2,R- 52),(B3,L3,A2,R53),(B3, L3,A2,R54),(B3,L3,A2,R55),(B3,L3,A2,R56),(B3,L3,A3,R51),(B3,L3,A3,R52),(B- 3,L3, A3,R53),(B3,L3,A3,R54),(B3,L3,A3,R55),(B3,L3,A3,R56),(B3,L3,A4,R51),- (B3,L3,A4, R52),(B3,L3,A4,R53),(B3,L3,A4,R54),(B3,L3,A4,R55),(B3,L3,A4,R56- ),(B3,L3,A5,R51),(B3,L3,A5,R52),(B3,L3,A5,R53),(B3,L3,A5,R54),(B3,L3,A5,R5- 5),(B3,L3,A5,R56),(B3,L4,A1,R51),(B3,L4,A1,R52),(B3,L4,A1,R53),(B3,L4,A1,R- 54),(B3,L4,A1,R55),(B3, L4,A1,R56),(B3,L4,A2,R51),(B3,L4,A2,R52),(B3,L4,A2,R53),(B3,L4,A2,R54),(B- 3,L4, A2,R55),(B3,L4,A2,R56),(B3,L4,A3,R51),(B3,L4,A3,R52),(B3,L4,A3,R53),- (B3,L4,A3, R54),(B3,L4,A3,R55),(B3,L4,A3,R56),(B3,L4,A4,R51),(B3,L4,A4,R52- ),(B3,L4,A4,R53),(B3,L4,A4,R54),(B3,L4,A4,R55),(B3,L4,A4,R56),(B3,L4,A5,R5- 1),(B3,L4,A5,R52),(B3,L4,A5,R53),(B3,L4,A5,R54),(B3,L4,A5,R55),(B3,L4,A5,R- 56),(B3,L5,A1,R51),(B3, L5,A1,R52),(B3,L5,A1,R53),(B3,L5,A1,R54),(B3,L5,A1,R55),(B3,L5,A1,R56),(B- 3,L5, A2,R51),(B3,L5,A2,R52),(B3,L5,A2,R53),(B3,L5,A2,R54),(B3,L5,A2,R55),- (B3,L5,A2, R56),(B3,L5,A3,R51),(B3,L5,A3,R52),(B3,L5,A3,R53),(B3,L5,A3,R54- ),(B3,L5,A3,R55),(B3,L5,A3,R56),(B3,L5,A4,R51),(B3,L5,A4,R52),(B3,L5,A4,R5- 3),(B3,L5,A4,R54),(B3,L5,A4,R55),(B3,L5,A4,R56),(B3,L5,A5,R51),(B3,L5,A5,R- 52),(B3,L5,A5,R53),(B3, L5,A5,R54),(B3,L5,A5,R55),(B3,L5,A5,R56),(B4,L1,A1,R51),(B4,L1,A1,R52),(B- 4,L1, A1,R53),(B4,L1,A1,R54),(B4,L1,A1,R55),(B4,L1,A1,R56),(B4,L1,A2,R51),- (B4,L1,A2, R52),(B4,L1,A2,R53),(B4,L1,A2,R54),(B4,L1,A2,R55),(B4,L1,A2,R56- ),(B4,L1,A3,R51),(B4,L1,A3,R52),(B4,L1,A3,R53),(B4,L1,A3,R54),(B4,L1,A3,R5- 5),(B4,L1,A3,R56),(B4,L1,A4,R51),(B4,L1,A4,R52),(B4,L1,A4,R53),(B4,L1,A4,R- 54),(B4,L1,A4,R55),(B4, L1,A4,R56),(B4,L1,A5,R51),(B4,L1,A5,R52),(B4,L1,A5,R53),(B4,L1,A5,R54),(B- 4,L1, A5,R55),(B4,L1,A5,R56),(B4,L2,A1,R51),(B4,L2,A1,R52),(B4,L2,A1,R53),- (B4,L2,A1, R54),(B4,L2,A1,R55),(B4,L2,A1,R56),(B4,L2,A2,R51),(B4,L2,A2,R52- ),(B4,L2,A2,R53),(B4,L2,A2,R54),(B4,L2,A2,R55),(B4,L2,A2,R56),(B4,L2,A3,R5- 1),(B4,L2,A3,R52),(B4,L2,A3,R53),(B4,L2,A3,R54),(B4,L2,A3,R55),(B4,L2,A3,R- 56),(B4,L2,A4,R51),(B4, L2,A4,R52),(B4,L2,A4,R53),(B4,L2,A4,R54),(B4,L2,A4,R55),(B4,L2,A4,R56),(B- 4,L2, A5,R51),(B4,L2,A5,R52),(B4,L2,A5,R53),(B4,L2,A5,R54),(B4,L2,A5,R55),- (B4,L2,A5, R56),(B4,L3,A1,R51),(B4,L3,A1,R52),(B4,L3,A1,R53),(B4,L8,A1,R54- ),(B4,L3,A1,R55),(B4,L3,A1,R56),(B4,L3,A2,R51),(B4,L3,A2,R52),(B4,L3,A2,R5- 3),(B4,L3,A2,R54),(B4,L3,A2,R55),(B4,L3,A2,R56),(B4,L3,A3,R51),(B4,L3,A3,R- 52),(B4,L3,A3,R53),(B4, L3,A3,R54),(B4,L3,A3,R55),(B4,L3,A3,R56),(B4,L3,A4,R51),(B4,L3,A4,R52),(B- 4,L3, A4,R53),(B4,L3,A4,R54),(B4,L3,A4,R55),(B4,L3,A4,R56),(B4,L3,A5,R51),- (B4,L3,A5, R52),(B4,L3,A5,R53),(B4,L3,A5,R54),(B4,L3,A5,R55),(B4,L3,A5,R56- ),(B4,L4,A1,R51),(B4,L4,A1,R52),(B4,L4,A1,R53),(B4,L4,A1,R54),(B4,L4,A1,R5- 5),(B4,L4,A1,R56),(B4,L4,A2,R51),(B4,L4,A2,R52),(B4,L4,A2,R53),(B4,L4,A2,R- 54),(B4,L4,A2,R55),(B4, L4,A2,R56),(B4,L4,A3,R51),(B4,L4,A3,R52),(B4,L4,A3,R53),(B4,L4,A3,R54),(B- 4,L4, A3,R55),(B4,L4,A3,R56),(B4,L4,A4,R51),(B4,L4,A4,R52),(B4,L4,A4,R53),- (B4,L4,A4, R54),(B4,L4,A4,R55),(B4,L4,A4,R56),(B4,L4,A5,R51),(B4,L4,A5,R52- ),(B4,L4,A5,R53),(B4,L4,A5,R54),(B4,L4,A5,R55),(B4,L4,A5,R56),(B4,L5,A1,R5- 1),(B4,L5,A1,R52),(B4,L5,A1,R53),(B4,L5,A1,R54),(B4,L5,A1,R55),(B4,L5,A1,R- 56),(B4,L5,A2,R51),(B4, L5,A2,R52),(B4,L5,A2,R53),(B4,L5,A2,R54),(B4,L5,A2,R55),(B4,L5,A2,R56),(B- 4,L5, A3,R51),(B4,L5,A3,R52),(B4,L5,A3,R53),(B4,L5,A3,R54),(B4,L5,A3,R55),- (B4,L5,A3, R56),(B4,L5,A4,R51),(B4,L5,A4,R52),(B4,L5,A4,R53),(B4,L5,A4,R54- ),(B4,L5,A4,R55),(B4,L5,A4,R56),(B4,L5,A5,R51),(B4,L5,A5,R52),(B4,L5,A5,R5- 3),(B4,L5,A5,R54),(B4,L5,A5,R55),(B4,L5,A5,R56),(B5,L1,A1,R51),(B5,L1,A1,R- 52),(B5,L1,A1,R53),(B5, L1,A1,R54),(B5,L1,A1,R55),(B5,L1,A1,R56),(B5,L1,A2,R51),(B5,L1,A2,R52),(B- 5,L1, A2,R53),(B5,L1,A2,R54),(B5,L1,A2,R55),(B5,L1,A2,R56),(B5,L1,A3,R51),- (B5,L1,A3, R52),(B5,L1,A3,R53),(B5,L1,A3,R54),(B5,L1,A3,R55),(B5,L1,A3,R56- ),(B5,L1,A4,R51),(B5,L1,A4,R52),(B5,L1,A4,R53),(B5,L1,A4,R54),(B5,L1,A4,R5- 5),(B5,L1,A4,R56),(B5,L1,A5,R51),(B5,L1,A5,R52),(B5,L1,A5,R53),(B5,L1,A5,R- 54),(B5,L1,A5,R55),(B5, L1,A5,R56),(B5,L2,A1,R51),(B5,L2,A1,R52),(B5,L2,A1,R53),(B5,L2,A1,R54),(B- 5,L2, A1,R55),(B5,L2,A1,R56),(B5,L2,A2,R51),(B5,L2,A2,R52),(B5,L2,A2,R53),- (B5,L2,A2, R54),(B5,L2,A2,R55),(B5,L2,A2,R56),(B5,L2,A3,R51),(B5,L2,A3,R52- ),(B5,L2,A3,R53),(B5,L2,A3,R54),(B5,L2,A3,R55),(B5,L2,A3,R56),(B5,L2,A4,R5- 1),(B5,L2,A4,R52),(B5,L2,A4,R53),(B5,L2,A4,R54),(B5,L2,A4,R55),(B5,L2,A4,R- 56),(B5,L2,A5,R51),(B5, L2,A5,R52),(B5,L2,A5,R53),(B5,L2,A5,R54),(B5,L2,A5,R55),(B5,L2,A5,R56),(B- 5,L3, A1,R51),(B5,L3,A1,R52),(B5,L3,A1,R53),(B5,L3,A1,R54),(B5,L3,A1,R55),- (B5,L3,A1, R56),(B5,L3,A2,R51),(B5,L3,A2,R52),(B5,L3,A2,R53),(B5,L3,A2,R54- ),(B5,L3,A2,R55),(B5,L3,A2,R56),(B5,L3,A3,R51),(B5,L3,A3,R52),(B5,L3,A3,R5- 3),(B5,L3,A3,R54),(B5,L3,A3,R55),(B5,L3,A3,R56),(B5,L3,A4,R51),(B5,L3,A4,R- 52),(B5,L3,A4,R53),(B5, L3,A4,R54),(B5,L3,A4,R55),(B5,L3,A4,R56),(B5,L3,A5,R51),(B5,L3,A5,R52),(B- 5,L3, A5,R53),(B5,L3,A5,R54),(B5,L3,A5,R55),(B5,L3,A5,R56),(B5,L4,A1,R51),- (B5,L4,A1, R52),(B5,L4,A1,R53),(B5,L4,A1,R54),(B5,L4,A1,R55),(B5,L4,A1,R56- ),(B5,L4,A2,R51),(B5,L4,A2,R52),(B5,L4,A2,R53),(B5,L4,A2,R54),(B5,L4,A2,R5- 5),(B5,L4,A2,R56),(B5,L4,A3,R51),(B5,L4,A3,R52),(B5,L4,A3,R53),(B5,L4,A3,R- 54),(B5,L4,A3,R55),(B5, L4,A3,R56),(B5,L4,A4,R51),(B5,L4,A4,R52),(B5,L4,A4,R53),(B5,L4,A4,R54),(B- 5,L4, A4,R55),(B5,L4,A4,R56),(B5,L4,A5,R51),(B5,L4,A5,R52),(B5,L4,A5,R53),- (B5,L4,A5, R54),(B5,L4,A5,R55),(B5,L4,A5,R56),(B5,L5,A1,R51),(B5,L5,A1,R52- ),(B5,L5,A1,R53),(B5,L5,A1,R54),(B5,L5,A1,R55),(B5,L5,A1,R56),(B5,L5,A2,R5- 1),(B5,L5,A2,R52),(B5,L5,A2,R53),(B5,L5,A2,R54),(B5,L5,A2,R55),(B5,L5,A2,R- 56),(B5,L5,A3,R51),(B5, L5,A3,R52),(B5,L5,A3,R53),(B5,L5,A3,R54),(B5,L5,A3,R55),(B5,L5,A3,R56),(B- 5,L5, A4,R51),(B5,L5,A4,R52),(B5,L5,A4,R53),(B5,L5,A4,R54),(B5,L5,A4,R55),- (B5,L5,A4, R56),(B5,L5,A5,R51),(B5,L5,A5,R52),(B5,L5,A5,R53),(B5,L5,A5,R54- ),(B5,L5,A5,R55),(B5,L5,A5,R56).
Test: Measurement of Inhibition of β-Secretase Activity
[0300] Zero point five μL of the test compounds (dissolved in N,N'-dimethylsulfoxide) were incubated with 48.5 μL of the fluorescence-quenched peptide substrate solution (Biotin-XSEVNLDAEFRHDSGC-Eu:X=ε-amino-n-capronic acid, Eu=Europium cryptate) and 1 μL of recombinant human BACE-1 protein (R&D systems) for 3 h at 30° C. in the 96 well half-area plate (black color plate, Costar). The substrate peptide was synthesized by reacting with Biotin-XSEVNLDAEFRHDSGC (Peptide Institute) and Cryptate TBPCOOH mono SMP (CIS bio international). The final-concentration of the substrate peptide and recombinant human BACE-1 protein were 18 nM and 7.4 nM, respectively. The enzymatic reaction was performed in sodium acetate buffer (50 mM sodium acetate (pH5.0), 0.008% Triton X-100). After the reaction, a 50 μL of 8.0 g/mL Streptavidin-XL665 (CIS bio international) dissolved in phosphate buffer (150 mM K2HPO4--KH2PO4 (pH 7.0), 0.008% Triton X-100, 0.8 M KF) was add to each well and incubated for 1 h at 30° C. Then, the fluorescence intensity (excitation wavelength 320 nm, emission wavelength 620 nM and 665 nM) in each well was measured using Wallac 1420 multilabel counter (Perkin Elmer life sciences). The enzymatic activity was calculated by the each fluorescence intensity ratio ([ratio of fluorescence at 665 nm to that at 620 nm]×10,000). IC50 values of test compounds were indicated in table 170.
TABLE-US-00170 TABLE 170 Compound IC50 value No. (μM) 1186 5.7 639 7.9 1000 8.0 246 2.9 269 7.0 1010 2.8 417 5.0 161 7.7 220 1.5 1207 4.0 998 4.5 1205 8.1 616 5.9 504 2.5 799 6.7 490 5.1 972 0.45 1160 0.72 753 5.6 786 0.156 165 0.0394 1132 2.563 570 0.149 1014 0.165 731 0.278 1262 0.140 964 0.264 793 0.061 625 1.288 498 0.930 26 1.977 465 3.239 1197 0.912 395 1.500 896 8.497 660 4.586 664 3.642 176 1.479 284 0.229 912 0.175 212 0.220 163 2.278 1244 0.130 52 10.0 698 0.165 96 0.163 822 0.243 739 0.049 832 0.222 897 0.816 1100 0.037 740 0.505 436 0.160 1043 0.027 1199 0.032 73 0.435 127 0.054 309 0.833 1135 2.296 1035 0.174
[0301] The IC50 value of the following compounds were less than 100 μM by the same test.
[0302] 3, 4, 6, 8, 12, 17, 18, 30, 31, 35, 36, 38, 39, 42, 43, 57, 61, 67, 67, 71, 77, 78, 80, 85, 97, 99, 105, 106, 113, 114, 115, 117, 120, 121, 125, 128, 129, 130, 134, 139, 144, 154, 157, 159, 164, 172, 175, 178, 181, 182, 186, 189, 200, 200, 201, 204, 207, 209, 211, 214, 215, 216, 228, 232, 240, 241, 243, 243, 243, 251, 255, 259, 267, 273, 275, 278, 279, 281, 282, 293, 298, 299, 300, 302, 303, 307, 314, 319, 321, 322, 326, 328, 330, 333, 335, 339, 341, 344, 345, 346, 348, 352, 353, 357, 358, 359, 359, 359, 360, 361, 363, 369, 370, 373, 378, 380, 383, 389, 390, 393, 396, 397, 402, 405, 406, 409, 410, 413, 415, 426, 442, 443, 444, 451, 452, 454, 456, 463, 467, 469, 472, 472, 479, 480, 482, 482, 483, 491, 493, 497, 500, 501, 502, 509, 511, 515, 516, 517, 527, 528, 532, 542, 544, 549, 550, 551, 558, 560, 568, 569, 575, 578, 584, 586, 588, 591, 600, 607, 608, 611, 613, 618, 620, 629, 634, 634, 637, 643, 646, 652, 657, 661, 671, 677, 681, 687, 691, 708, 711, 719, 720, 723, 725, 728, 729, 730, 732, 735, 743, 746, 756, 758, 761, 770, 775, 781, 787, 788, 790, 791, 792, 796, 797, 802, 803, 804, 808, 809, 813, 816, 819, 820, 824, 833, 835, 836, 847, 850, 861, 865, 866, 871, 876, 887, 893, 894, 900, 905, 906, 908, 910, 919, 922, 928, 932, 933, 935, 936, 939, 941, 943, 944, 946, 947, 949, 959, 966, 971, 984, 986, 988, 990, 1004, 1005, 1007, 1009, 1013, 1020, 1028, 1034, 1039, 1046, 1055, 1062, 1063, 1069, 1074, 1077, 1084, 1089, 1096, 1099, 1108, 1109, 1114, 1124, 1125, 1131, 1140, 1142, 1145, 1147, 1148, 1150, 1164, 1165, 1172, 1174, 1184, 1185, 1193, 1211, 1217, 1221, 1237, 1241, 1243, 1255, 1256, 1257, 1258, 1261, 1263, 1264, 1265, 1266, 1268, 1269, 1270, 1271, 1272, 1274, and so on.
Formulation Example 1
[0303] A granule containing the following ingredients is prepared.
TABLE-US-00171 Ingredient Compound represented by formula (I) 10 mg Lactose 700 mg Corn starch 274 mg HPC-L 16 mg 1000 mg
[0304] The compound represented by the formula (I) and lactose are passed through a 60 mesh sieve. Corn starch is passed through a 120 mesh sieve. These are mixed with a V-type mixer. To a mixed powder is added a HPC-L (lower viscosity hydroxypropylcellulose) aqueous solution, the materials are kneaded, granulated (extrusion granulation, pore diameter 0.5 to 1 mm), and dried. The resulting dry granule is passed through a sieve using a vibration sieve (12/60 mesh) to obtain a granule.
Formulation Example 2
[0305] A granule for filling into a capsule containing the following ingredients is prepared.
TABLE-US-00172 Ingredient Compound represented by formula (I) 15 mg Lactose 90 mg Corn starch 42 mg HPC-L 3 mg 150 mg
[0306] The compound represented by the formula (I) and lactose are passed through a 60 mesh sieve. Corn starch is passed through a 120 mesh sieve. These are mixed, to a mixed powder is added a HPC-L solution, the materials are kneaded, granulated, and dried. The resulting dry granule is size-adjusted, 150 mg of which is filled into a No. 4 hard gelatin capsule.
Formulation Example 3
[0307] A tablet containing the following ingredients is prepared.
TABLE-US-00173 Ingredient Compound represented by the formula (I) 10 mg Lactose 90 mg Microcrystalline cellulose 30 mg CMC-Na 15 mg Magnesium stearate 5 mg 150 mg
[0308] The compound represented by the formula (I), lactose, microcrystalline cellulose, CMC-Na (carboxymethylcellulose sodium salt) are passed through a 60 mesh sieve, and mixed. Into a mixed powder is mixed magnesium stearate to obtain a mixed powder for tabletting. The present mixed powder is compressed to obtain 150 mg of a tablet.
Formulation Example 4
[0309] The following ingredients are warmed, mixed, and sterilized to obtain an injectable.
TABLE-US-00174 Ingredient Compound represented by the formula (I) 3 mg Nonionic surfactant 15 mg Purified water for injection 1 ml
INDUSTRIAL APPLICABILITY
[0310] The present invention is useful as an agent for treating disease induced by production, secretion and/or deposition of amyloid β.
User Contributions:
Comment about this patent or add new information about this topic: