Patent application title: KITS FOR DETECTING AND MONITORING ENDOCRINE DISRUPTING CHEMICALS (EDCs)
Inventors:
IPC8 Class: AG01N3368FI
USPC Class:
Class name:
Publication date: 2015-06-18
Patent application number: 20150168416
Abstract:
Described herein are compositions, methods, a system, and kits for
detection of endocrine disruptor chemicals (EDCs) in environmental and
other samples, such as samples of water including but not limited to
waste water treatment plant effluent, using a live-cell
fluorescence-based nuclear translocation reporter system. Upon binding of
a ligand to a fluorescent-labeled reporter protein, the protein (and
therefore the fluorescence) is translocated in a ligand level-dependent
manner from the cytoplasm to the nucleus of live mammalian cells; this
translocation is detectable as diffuse (cytoplasmic) fluorescence
converting to localized, brightly fluorescent nuclei. The described
methods, kits and system can be used to reliably detect very low levels
of EDC contamination, including in high throughput analysis systems as
described.Claims:
1. A system for detecting or quantifying a ligand of a superfamily
receptor protein in a sample, the system comprising: a first mammalian
cell expressing a first traceable fusion protein; a second mammalian cell
expressing a second traceable fusion protein; and a detection system for
the detection of the cytoplasm-to-nuclear translocation of the marker
proteins, wherein the first and second traceable fusion proteins
independently comprise either: (1) a superfamily receptor protein, and a
marker protein domain; or (2) the cytoplasmic/nuclear translocation
domain of glucocorticoid receptor, the ligand binding domain of a
superfamily receptor protein, and a marker protein domain wherein at
least one of the traceable fusion proteins is GFP-mAhR, GFP-MR, or
GFP-PRB.
2. The system of claim 1, wherein the ligand is an agonist or an antagonist.
3. The system of claim 1, wherein the first traceable fusion protein binds a different ligand than the second traceable fusion protein.
4. The system of claim 3, wherein the marker protein domain is different in the first and second traceable fusion proteins.
5. The system of claim 1, wherein the marker protein domain is a fluorescent protein domain.
6. The system of claim of claim 1, wherein the ligand for the first or second traceable fusion protein is a natural ligand for the superfamily receptor protein of the traceable fusion protein, or a natural or synthetic compound that binds competitively therewith.
7. The system of claim 1, additionally comprising: one or more compounds and/or compositions that stably associate with a traceable fusion protein of the system in the absence of a ligand for the ligand binding domain of the traceable fusion protein, and which dissociates from the fusion protein in the presence of a ligand for the ligand binding domain of the traceable fusion protein.
8. The system of claim 1, wherein the mammalian cells are human cells.
9. The system of claim 1, comprising at least four mammalian cell lines each expressing a different traceable fusion protein, wherein at least one traceable fusion protein binds a glucocorticoid, at least one traceable fusion protein binds an androgen, at least one traceable fusion protein binds a progestin, and at least one traceable fusion protein binds an aryl hydrocarbon.
10. The system of claim 1, comprising at least four mammalian cell lines each expressing a different traceable fusion protein, wherein the traceable fusion proteins each bind one of a glucocorticoid, an androgen, a progestin, an aryl hydrocarbon, a thyroid hormone, or a mineralocorticoid.
11. The system of claim 1, provided as a kit.
12. A method for detecting or quantifying an endocrine disruptor chemical (EDC) ligand in a sample, the method comprising: contacting with the environmental sample a human cell expressing in its cytoplasm a fusion protein comprising: a receptor for the ligand, which receptor translocates from the cytoplasm to the nucleus upon ligand binding; and a marker protein domain; and detecting cytoplasmic to nuclear translocation of the fusion protein in response to binding a ligand of the ligand binding domain in the environmental sample, wherein the fusion protein is GFP-mAhR, GFP-MR, or GFP-PRB.
13. A method of determining the concentration of an endocrine disruptor chemical (EDC) ligand in a sample, the method comprising: contacting with the environmental sample a population of eukaryotic cells expressing in their cytoplasm a fusion protein comprising: a receptor for the ligand, which receptor translocates from the cytoplasm to the nucleus upon ligand binding; and a marker protein domain; and scanning one or more test cell(s) to obtain signal data from the marker of the fusion protein; converting the signal data to obtain the cellular location of the labeled protein in the test cell(s); and analyzing the signal data using an analysis system having an algorithm to calculate changes in distribution of the labeled fusion protein between the cytoplasm and the nucleus of the test cell(s), the analysis system having the capability of providing an accurate reading of the concentration of the ligand, wherein the fusion protein is GFP-mAhR, GFP-MR, or GFP-PRB.
14. The system of claim 19, wherein the environmental sample comprises a water sample, soil sample, or air sample.
15. The system of claim 14, wherein the sample comprises a water sample comprising surface water, sub-surface (ground) water, rain, run-off, well water, spring water, drinking water (processed or not), river water, estuary water, ocean water, effluent, treated sewage or untreated sewage.
16-18. (canceled)
19. The system of claim 1, wherein the sample is an environmental sample.
20. The method of claim 12, wherein the sample is an environmental sample.
21. The method of claim 13, wherein the sample is an environmental sample.
22. A traceable fusion protein capable of binding an endocrine disruptor chemical (EDC) ligand in a sample, comprising: a receptor for the ligand, which receptor translocates from the cytoplasm to the nucleus upon ligand binding; and a marker protein domain; wherein the ligand is an aryl hydrocarbon, a mineralocorticoid, or a progestin.
23. The traceable fusion protein of claim 22, comprising the polypeptide sequence shown in one of: SEQ ID NO: 10 (pRevTRE-GFP-mAhR); SEQ ID NO: 12 (eGFP-ratMR); or SEQ ID NO: 14 (eGFP-hPRB); or, encoded by the nucleic acid sequence shown in one of: SEQ ID NO: 9 (pRevTRE-GFP-mAhR); SEQ ID NO: 11 (eGFP-ratMR); or SEQ ID NO: 13 (eGFP-hPRB).
Description:
REFERENCE TO RELATED APPLICATION(S)
[0001] This application claims the benefit of the earlier filing date of U.S. Provisional Application No. 61/656,473, filed Jun. 6, 2012, which is incorporated herein in its entirety.
FIELD
[0002] This disclosure relates to methods, systems and kits for monitoring, detecting and/or measuring chemicals, such as endocrine disruptor chemicals, in environmental and other samples. This disclosure further relates to genetically engineered constructs that encode traceable fusion proteins that translocate from the cytoplasm to the nucleus in the presence of an endocrine disrupting chemical, and mammalian cells expressing one or more of such constructs.
BACKGROUND
[0003] An endocrine disruptor chemical (EDC) is an exogenous substance that impacts at least one function of an animal's endocrine system and consequently causes adverse health effects in an intact organism, its progeny, or (sub)populations. EDCs can cause adverse biological effects in animals and humans (Diamanti-Kandarakis et al., Horm. Metab Res 42:543-552, 2010).
[0004] Contamination of the environment, particularly water sources, with EDCs is a major concern for human health and threatens the integrity of aquatic ecosystems (Diamanti-Kandarakis et al., Endocr. Rev. 30:293-342, 2009; Deblonde et al., Int. J Hyg. Environ. Health 214:442-448, 2011). Harmful effects of synthetic progestogens (Zeilinger et al., Environ. Toxicol. Chem. 28:2663-2670, 2009; Paulos et al., Aquat. Toxicol. 99:256-262, 2010) and especially of estrogenic water contaminants (Iwanowicz et al., Environ. Toxicol. Chem. 28:1072-1083, 2009; Alvarez et al., Environ. Toxicol. Chem. 28:1084-1095, 2009; Caldwell et al., Environ. Sci. Technol. 42:7046-7054, 2008; Lange et al., Environ. Toxicol. Chem. 20:1216-1227, 2001; Blazer et al., Environ. Monit. Assess. DOI 10.1007/s10661-011-2266-5, 2011) on fish reproduction are well documented. In addition, there is growing concern that environmental contamination with EDCs has deleterious effects on human reproduction, breast development and cancer, prostate cancer, neuroendocrinology, thyroid metabolism and obesity, and cardiovascular endocrinology (Diamanti-Kandarakis et al., Endocr. Rev. 30:293-342, 2009).
[0005] Glucocorticoids act through the glucocorticoid and mineralocorticoids receptors (GR and MR, respectively). Glucocorticoid deficiency is associated with a number of complex symptoms and is a life-threatening condition (Arlt & Allolio, Lancet 361:1881-1893, 2003). Naturally occurring glucocorticoids are released in mammalian organisms during the circadian cycle. However, excess exposure to glucocorticoids is associated with immune suppression and variety of other deleterious side effects (Schacke et al., Pharmacol. Ther. 96:23-43, 2002). Unoccupied glucocorticoid receptor resides in the cytoplasm and is bound to various heat-shock proteins and immunophilins in a large multi-protein complex (Pratt & Toft, Endocr. Rev. 18:306-360, 1997; Pratt et al., Handb. Exp. Pharmacol. 172:111-138, 2006). Upon hormone binding, GR dissociates from the chaperones and translocates to the cell nucleus, where it interacts with GR regulatory elements (GREs) and elicits GR-specific transcription regulation (John et al., Mol. Cell 29:611-624, 2008).
[0006] At present, nothing is known about the prevalence of GCs activity in US water sources. However, using chemical methods, a few reports on water contamination in the Netherlands and China have demonstrated detectable levels of glucocorticoids (Schriks et al., Environ. Sci. Technol. 44:4766-4774, 2010; Change et al., Environ. Sci. Technol. 41:3462-3468, 2007). Another recent study has demonstrated that environmentally relevant concentrations of synthetic GCs have deleterious effects on fish (Kugathas & Sumpter, Environ. Sci. Technol. 45:2377-2383, 2011). The anti-inflammatory properties of the glucocorticoids make them highly prescribed pharmaceuticals. They could readily enter water sources and there are few sparse reports on water contamination with glucocorticoids (Schriks et al., Environ. Sci. Technol. 44:4766-4774, 2010; Chang et al., Environ. Sci. Technol. 41:3462-3468, 2007). Moreover, waste water treatment plants (WWTP) are not capable of efficiently removing glucocorticoids; it is well documented that anti-inflammatory chemicals are among the most resistant to treatment (30-40% of removal rate).
[0007] In spite of their importance, the levels of EDCs, such as steroidal EDCs, in the environment currently are not efficiently monitored and/or regulated. One of the reasons is that no high-throughput, reliable, low-cost detection methods exist for monitoring of biologically active EDCs. Current EDC detection relies on chemical analysis techniques (e.g., mass spectrometry, HPLC, GC, and other purely chemical analytical procedures), in vitro biologically-based but cell-free analysis techniques (e.g., purified receptor binding assays and immunoaffinity chromatography), in vitro cell-based analyses (e.g., cell proliferation assays and receptor-dependent gene expression assays, in human cells, or engineered yeast or bacterial cells), and in vivo analyses (e.g., uterotrophic and other growth/development assays in live rats or other animals). It is crucial to develop and implement novel high-throughput and low-cost methods for detection of EDCs in the environment. The need of such methods is well recognized in the field (Roy et al., J. Exp. Biol. 43:975-992, 2005). Existing methods for EDC detection may be sensitive, and in some instances are specific for individual ligands, but in general they are expensive, time-consuming, and largely incompatible with a large-scale sample testing.
SUMMARY
[0008] Described herein are methods, systems and kits for monitoring, detecting and/or measuring EDCs in environmental and other types of samples. These methods, systems and kits employ live mammalian (for instance, human) cells engineered to express at least one traceable fusion protein that changes its sub-cellular localization, for example translocates from the cytoplasm to the nucleus of the cell in the presence of an EDC. Specifically provided are cells and kits that detect more than one EDC, more than one class of EDC, and in some examples an array of EDCs simultaneously. Also provided are methods, systems and kits tailored for high throughput detection (and optionally quantification) of EDCs in environmental samples, such as water samples.
[0009] Provided herein in a first set of embodiments are systems for detecting or quantifying a ligand (e.g., an agonist or an antagonist) of a superfamily receptor protein in an environmental sample. In examples thereof, the system comprises a first mammalian cell expressing a first traceable fusion protein; a second mammalian cell expressing a second traceable fusion protein; and a detection system for the detection of the cytoplasm-to-nuclear translocation of the marker proteins, wherein the first and second traceable fusion proteins independently comprise either: (1) a superfamily receptor protein, and a marker protein domain; or (2) the cytoplasmic/nuclear translocation domain of glucocorticoid receptor, the ligand binding domain of a superfamily receptor protein, and a marker protein domain. Optionally, the mammalian cells are human cells.
[0010] Also described are systems for detecting or quantifying a ligand of a superfamily receptor protein in an environmental sample, wherein the ligand for the first or second traceable fusion protein is a natural ligand for the superfamily receptor protein of the traceable fusion protein, or a natural or synthetic compound that binds competitively therewith.
[0011] Also provided are systems for detecting or quantifying a ligand of a superfamily receptor protein in an environmental sample that additionally comprise one or more compounds and/or compositions that stably associate with a traceable fusion protein in the absence of a ligand for the ligand binding domain of the traceable fusion protein, and which dissociates from the fusion protein in the presence of a ligand for the ligand binding domain of the traceable fusion protein.
[0012] The systems described herein are optionally provided as kits.
[0013] Also described are methods for detecting or quantifying an endocrine disruptor chemical (EDC) ligand in an environmental sample, which methods involve contacting with the environmental sample a human cell expressing in its cytoplasm a fusion protein comprising: a receptor for the ligand, which receptor translocates from the cytoplasm to the nucleus upon ligand binding; and a marker protein domain; and then detecting cytoplasmic to nuclear translocation of the fusion protein in response to a ligand of the ligand binding domain in the water sample.
[0014] Additional described methods of determining the concentration of an endocrine disruptor chemical (EDC) ligand in an environmental sample involve contacting with the environmental sample a population of eukaryotic cells expressing in their cytoplasm a fusion protein comprising: a receptor for the ligand, which receptor translocates from the cytoplasm to the nucleus upon ligand binding; and a marker protein domain; and scanning one or more test cell(s) to obtain signal data from the marker of the fusion protein; converting the signal data to obtain the cellular location of the labeled protein in the test cell(s); and analyzing the signal data using an analysis system having an algorithm to calculate changes in distribution of the labeled fusion protein between the cytoplasm and the nucleus of the test cell(s), the analysis system having the capability of providing an accurate reading of the concentration of the ligand.
[0015] In any of the systems, kits, and methods described herein, the environmental sample can include a water sample, soil sample, or air sample. By way of example, where the environmental sample comprises a water sample, the sample includes in various embodiments one or more of surface water, sub-surface (ground) water, rain, run-off, well water, spring water, drinking water (processed or not), river water, estuary water, ocean water, effluent, treated sewage or untreated sewage.
[0016] Without limitation, the systems, methods and kits provided herein may use or include any cellular receptors including the following (generically termed) traceable fusion proteins: GFP-GR, GFP-AR, GFP-AhR, GFP-PR-B, GFP-GR-ER, GFP-GR-RAR, GFP-MR, and GFP-GR-TR, as well as fusion(s) in which the GFP is replaced with a different fluorescent domain that emits light at a different wavelength. By way of specific example, contemplated herein for use (singly or in combinations of two or more) in the described systems, methods and kits are the following traceable fusion proteins: eGFP-GR-ER310 (SEQ ID NOs: 1 & 2); eGFP-GR-TR216 (SEQ ID NOs: 3 & 4); pCI-nGFP-C656G (SEQ ID NOs: 5 & 6); eGFP-hAR (SEQ ID NOs: 7 & 8); pRefTRE-GFP-mAhR (SEQ ID NOs: 9 & 10); eGFP-ratMP (SEQ ID NO: 11 & 12); and/or eGFP-hPRB (SEQ ID NOs: 13 & 14).
[0017] The foregoing and other objects, features, and advantages of the invention will become more apparent from the following detailed description, which proceeds with reference to the accompanying figures.
BRIEF DESCRIPTION OF THE DRAWINGS
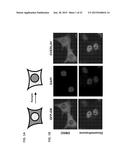
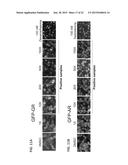
[0018] FIGS. 1A-E illustrates analysis of water samples for glucocorticoid and androgen contamination. FIG. 1A is a schematic representation of the GFP-tagged GR and AR receptor translocation in response to corresponding hormonal treatment. FIG. 1B is a series of micrographs showing GFP-GR translocation in a mammalian cell line (Walker et al., Methods (Comp. to Meth. Enzym.) 19:386-393, 1999) upon stimulation with dexamethasone for 30 minutes. Nuclei are stained with DAPI. Scale bar, 5 μm. FIG. 1C illustrates transcriptional activation of the GR-regulated Per1 gene by 10 water samples collected using a polar organic chemical integrative sampler (POCIS) are compared to transactivation induced by corticosterone. Data is normalized to DMSO alone. Blank and SS83 are POCIS negative controls. Error bars represent the mean±s.e.m., n=3. One of the water samples, SS97, induces complete GFP-GR translocation (image) and transcriptional activation of Per1 gene at a level higher than the activation induced by 100 nM corticosterone (graph). Scale bar, 5 μm. FIG. 1D is a GC/MS total ion chromatogram of HPLC fractionated sample SS97 (fraction 74-A), which revealed the presence of a complex mixture of volatile hydrocarbons, as indicated by the peaks. Database searching of the extracted MS spectra corresponding to peaks 1-3 showed structural similarity to known androstane-type steroids. GC/MS analysis of these peaks is presented in FIG. 3 and Table 3. FIG. 1E is a series of representative images of GFP-AR nuclear translocation in response to 100 nM of testosterone, androst-4-ene-3,6-dione, and sample SS97 (100×). Scale bar, 5 μm.
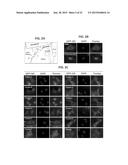
[0019] FIGS. 2A-C illustrates collection sites of the first sample set and testing of HPLC fractions from sample SS97 for GR translocation. FIG. 2A illustrates the geographic locations of the collection sites of the first sample set collected by POCIS. One of the samples (marked with red, SS97) tested positive for GFP-GR translocation, suggesting a presence of glucocorticoid activity. Negative samples are marked with green. FIG. 2B shows GFP-GR translocation in a mammalian cell line (Walker et al., Methods (Comp. to Meth. Enzym.) 19:386-393, 1999) upon stimulation with 100 nM corticosterone for 30 min. Nuclei are stained with DAPI. Scale bar, 5 μm. FIG. 2C is a series of micrographs illustrating testing of HPLC fractions of sample SS97 (Fractions A-K) for GFP-GR translocation to determine the presence of glucocorticoid activity. Four of 11 fractions tested positive for GFP-GR translocation (FIG. 2C). Because GC/MS analyses detected atrazine in sample SS97, we included it in our analysis (bottom right). However, atrazine did not induce GFP-GR translocation.
[0020] FIG. 3 illustrates that database searching of the extracted MS chromatographic peaks 1-3 (FIG. 1D) suggests that the compounds were similar in structure to known androstane-type steroids. Visual comparison of the mass spectra of chromatographic peaks 1-3 (FIG. 1D) with standard spectra from the AES 2010 database suggests similarities to known androstane-class compounds.
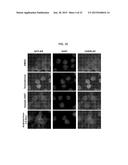
[0021] FIGS. 4A-E illustrates water sample screening by high throughput automated image analysis. FIG. 4A contains examples of images scored for cytoplasmic and nuclear segmentation from control and corticosterone treated 3617 cells. FIG. 4B is an illustration of a workflow for image-based screening of environmental contaminants with glucocorticoid activity using the Perkin Elmer Opera Image Screening System. FIG. 4C illustrates the automated image analysis output for a representative experiment. In this series, eight water samples from a total of 69 (well positions 2B, 2G, 3F, 4F, 9H, 10A, 10C, and 10E corresponding to samples R4E, R4A, R16W, R27, LF1a, 2a, 3a, and 4a (FIG. 4D) tested positive for GFP-GR translocation as indicated by color changes. Wells 1A-D represent four negative (DMSO) controls. Wells 1E-H are positive controls for cells treated with 100 nM corticosterone, and wells 10D, 10F and 10H are positive controls for cells treated with 100 nM dexamethasone. FIG. 4D and FIG. 4E are bar graphs illustrating quantitative analysis for GFP-GR and GFP-AR nuclear translocation, respectively. Translocation was calculated as a ratio of the nuclear versus cytoplasmic intensity, and each value was normalized to the control. Samples positive for glucocorticoid activity are marked with asterisks (P<0.01, single asterisks and P<0.05, double asterisks). Error bars represent the mean value±s.e.m, n=4.
[0022] FIGS. 5A-C illustrates concentration-dependent translocation of GFP-GR and GFP-AR in response to their respective hormones as detected by the Opera (Perkin Elmer) automated imaging analysis system. FIG. 5A is a bar graph illustrating that GFP-GR translocates to the nucleus in a concentration-dependent manner upon treatment with known concentrations of hydrocortisone, dexamethasone, or corticosterone. An algorithm for cytoplasm and nuclear segmentation of the cells was used to determine the mean GFP-GR intensity in both compartments and translocation was quantified as a ratio of these intensities. Each value was normalized to the control sample. Error bars represent the mean value±s.e.m, n=6 (P<0.05, asterisks). FIG. 5B is a bar graph illustrating that GFP-AR translocation in response to known concentrations of testosterone. Insert shows that testosterone concentrations as low as 0.1 nM induced a statistically significant increase in the GFP-AR translocation. Samples with P<0.05 are indicated by an asterisk. Error bars represent the mean value±s.e.m, n=6. FIG. 5C is a bar graph illustrating that androst-4-en-3,6-dione induces concentration-dependent translocation of the GFP-AR construct (P<0.05, asterisks). Error bars represent the mean value±s.e.m, n=6.
[0023] FIGS. 6A-B illustrates additional samples screened for GFP-GR nuclear translocation. FIG. 6A is an image analysis plate map showing a portion of GFP-GR expressing cells plated on a 384 well plate. Twenty two samples out of 38 (58%) tested positive for GFP-GR nuclear translocation. Wells 10A-H are negative controls (DMSO). Wells 1A-H and 9D, 9F, 9H are positive controls treated with 100 nM corticosterone and 100 nM dexamethasone, respectively. FIG. 6B is a bar graph GFP-GR nuclear translocation results summary. All samples in the gray box are POCIS negative controls. Positive samples are marked with asterisks (P<0.01, single asterisks and P<0.05, double asterisks). Error bars represent the mean value±s.e.m, n=4.
[0024] FIGS. 7A-B illustrates additional samples screened for GFP-AR nuclear translocation. FIG. 7A is an image analysis plate map showing a portion of GFP-AR expressing cells plated on a 384 well plate. Twenty one samples out of 40 (55%) tested positive for GFP-AR nuclear translocation. Wells 10A-H are negative controls (DMSO). Wells 1 A-H are positive controls for cells treated with 100 nM testosterone. FIG. 7B is a bar graph GFP-AR nuclear translocation results summary. All samples in the gray box are POCIS negative controls. Positive samples are marked with asterisks (P<0.01, single asterisks and P<0.05, double asterisks). Error bars represent the mean value±s.e.m, n=4.
[0025] FIG. 8 illustrates geographic locations of the collection sites and their contamination with glucocorticoid and androgenic activity. Negative samples are marked with green color. Samples positive for glucocorticoid activity are marked with black, androgen activity-positive samples are marked with pink, and samples positive for both activities are marked with red. Triangles indicate grab samples, while the circles indicate the use of POCIS membranes. For complete sample description (collection method as well as the time of collection and translocation activity) see Table 5A & 5B.
[0026] FIGS. 9A-F illustrates concentration-dependent translocation and transcriptional activation induced by newly collected grab samples at location SS97. FIG. 9A and FIG. 9B are bar graphs illustrating concentration-dependent GFP-GR and GFP-AR translocation for sample SS97 four years after the initial collection. Translocation is calculated from the automatic image analysis and expressed as a ratio of nuclear versus cytoplasmic intensity normalized to DMSO treated control. Samples positive for glucocorticoid and androgen activities are marked with asterisks (P<0.05). Error bars represent the mean value±s.e.m, n=4. Significant increase in the GFP-GR translocation is detectable in 1×dilution for this sample (inset). While the lowest concentration inducing significant GFP-AR translocation was 10× (B, insert). FIG. 9C shows a representative heat-map for concentration-dependent GFP-GR translocation indicated as nuclear versus cytoplasmic intensity. Dex (dexamethasone, 100 nM), Cst (corticosterone, 100 nM), and HC (hydrocortisone, 100 nM) are included as positive controls as indicated on the bar graph above (FIG. 9A). FIG. 9D is a representative heat-map for the concentration-dependent GFP-AR translocation. Testosterone (Testo, 100 nM) was included as a positive control, as indicated on the bar graph above (FIG. 9B). FIG. 9E is a pair of bar graphs illustrating concentration-dependent transcriptional activation of the GR-regulated genes, Tgm2 and Lcn2. All tested concentrations (including 1×) induced transcriptional responses of both genes, presented as fold change from the vehicle (DMSO) treated control. Dex (dexamethasone, 100 nM), Cst (corticosterone, 100 nM), and HC (hydrocortisone, 100 nM) are included as positive controls. Error bars represent the mean±s.e.m, n=4. FIG. 4F is a pair of bar graphs illustrating concentration-dependent transcriptional activation of the AR-regulated genes, NKX2.1 and RHOU by sample SS7 in LNCaP cells. Gene transcription was induced by the concentrations 10×, 20×, and 50× (P<0.05, asterisks), whereas higher concentrations seemed to have an inhibitory activity. Data is presented as fold change in comparison to the vehicle (DMSO) treated control. Androst-4-en-3,6-dione (A-4) (100 nM) and testosterone (Testo, 100 nM) were included as positive controls. Error bars represent the mean±s.e.m, n=4.
[0027] FIGS. 10A-B illustrates concentration-dependent GFP-GR and GFP-AR translocation induced by the newly collected sample from the same location as SS97. FIG. 10A is a series of representative images for the concentration-dependent GFP-GR translocation in response to known concentrations of the water sample at site SS97. Scale bar, 10 μm. FIG. 10B is a series of representative images for concentration-dependent GFP-AR translocation in response to known concentrations of the water sample at site SS97. Scale bar, 10 μm.
[0028] FIGS. 11A-B illustrates concentration-dependent GFP-GR and GFP-AR translocation induced by the newly collected sample from the same location as GL2W. FIG. 11A is a series of representative images for concentration-dependent GFP-GR translocation in response to known concentrations of the water sample at site GL2W. Scale bar, 10 μm. FIG. 11B is a series of representative images for the concentration-dependent GFP-AR translocation in response to known concentrations of the water sample at site GL2W. Scale bar, 10 μm.
[0029] FIGS. 12A-C illustrates concentration-dependent GFP-GR translocation and transcriptional activation of GR-regulated genes in response to the newly collected sample from the same location as GL2W. FIG. 12A is a bar graph illustrating translocation of GFP-GR in response to newly collected sample from the same location as GL2W (see also FIG. 11A). Translocation was calculated as a ratio of the nuclear versus cytoplasmic intensity and each value was further normalized to the value for the control sample. Samples positive for GFP-GR translocation are marked with asterisks (P<0.05). The lowest concentration inducing GFP-GR translocation is 10×. DMSO negative control, Dex (dexamethasone, 100 nM), Cst (corticosterone, 100 nM), and HC (hydrocortisone, 100 nM) were included as positive controls. Error bars represent the mean value±s.e.m, n=4. FIG. 12B is a representative raw data heat-map for GFP-GR translocation as in panel A. FIG. 12C is a series of bar graphs illustrating concentration-dependent transcriptional activation of three GR-regulated genes induced by newly collected sample from GL2W site. All concentrations induced transcriptional activation of at least one of the GR-regulated genes (P<0.05, asterisks). Transcription responses are presented as fold change in comparison to the vehicle control sample (DMSO). Dex (dexamethasone, 100 nM), Cst (corticosterone, 100 nM), and HC (hydrocortisone, 100 nM) are included as positive controls. Error bars represent the mean±s.e.m, n=4.
[0030] FIGS. 13A-C illustrates concentration-dependent GFP-AR translocation and transcriptional activation of AR-regulated genes in response to the newly collected sample from GL2W site. FIG. 13A is a bar graph illustrating quantification of the GFP-AR concentration-dependent translocation response for the newly collected water sample at the GL2W site (see also FIG. 11B). Translocation was calculated as a ratio of the nuclear versus cytoplasmic intensity and each value was further normalized to the value for the control sample. Samples positive for GFP-AR translocation are marked with asterisks (P<0.05). The lowest sample concentration inducing GFP-AR translocation is 10×. Testosterone (Testo, 100 nM) is included as a positive control. Error bars represent the mean value±s.e.m, n=4. FIG. 13B is a representative raw data heat-map for GFP-AR translocation as in panel A. FIG. 13C is a pair of graphs illustrating concentration-dependent transcriptional activation of AR-regulated genes induced by newly collected sample from GL2W site in LNCaP cells which express endogenous AR. Concentrations 10× to 50× induced transcriptional activation in the AR-regulated genes NKX2.1 and RHOU (P<0.05, asterisks). At 100× concentration, we observed a reduced NKX2.1 expression, and did not detect change in RHOU transcription, suggesting the presence of an inhibitory activity (possibly anti-androgenic) which is apparent at that concentration. Transcriptional responses are presented as fold change in comparison to the vehicle treated (DMSO) control sample. Androst-4-en-3,6-dione (100 nM) and testosterone (Testo, 100 nM) are included as positive controls. Error bars represent the mean±s.e.m, n=4.
[0031] FIGS. 14A-B illustrates testing of single-cell clones of the GFP-PR-B cell line for translocation response to known concentrations of progesterone. FIG. 14A shows the plate layout (four wells each, except for eight wells for clone 8122); FIG. 14B shows quantification of the translocation efficiency of each of the clones (bars in numerical order from left to right, 8119, 8122, 8124, 8127, and 8130, for each test condition). Four of the five tested clones (clones 8122, 8124, 8127, and 8130) are positive for translocation in response to progesterone.
Sequence Listing
[0032] The nucleic and amino acid sequences listed in the accompanying sequence listing are shown using standard letter abbreviations for nucleotide bases, and three letter code for amino acids, as defined in 37 C.F.R. 1.822. Only one strand of each nucleic acid sequence is shown, but the complementary strand is understood as included by any reference to the displayed strand. The Sequence Listing is submitted as an ASCII text file named Sequences.txt, created on Jun. 5, 2013, ˜100 KB, which is incorporated by reference herein.
[0033] SEQ ID NO: 1 is the nucleic acid sequence which encodes traceable fusion protein eGFP-GR-ER310. The sequence includes the following features at the indicated nucleotide positions: EGFP (1-717), GA Linker (718-747), hGR (748-2400), and hER (2401-3261).
TABLE-US-00001 ATGGTGAGCAAGGGCGAGGAGCTGTTCACCGGGGTGGTGCCCATCCTGGT CGAGCTGGACGGCGACGTAAACGGCCACAAGTTCAGCGTGTCCGGCGAGG GCGAGGGCGATGCCACCTACGGCAAGCTGACCCTGAAGTTCATCTGCACC ACCGGCAAGCTGCCCGTGCCCTGGCCCACCCTCGTGACCACCCTGACCTA CGGCGTGCAGTGCTTCAGCCGCTACCCCGACCACATGAAGCAGCACGACT TCTTCAAGTCCGCCATGCCCGAAGGCTACGTCCAGGAGCGCACCATCTTC TTCAAGGACGACGGCAACTACAAGACCCGCGCCGAGGTGAAGTTCGAGGG CGACACCCTGGTGAACCGCATCGAGCTGAAGGGCATCGACTTCAAGGAGG ACGGCAACATCCTGGGGCACAAGCTGGAGTACAACTACAACAGCCACAAC GTCTATATCATGGCCGACAAGCAGAAGAACGGCATCAAGGTGAACTTCAA GATCCGCCACAACATCGAGGACGGCAGCGTGCAGCTCGCCGACCACTACC AGCAGAACACCCCCATCGGCGACGGCCCCGTGCTGCTGCCCGACAACCAC TACCTGAGCACCCAGTCCGCCCTGAGCAAAGACCCCAACGAGAAGCGCGA TCACATGGTCCTGCTGGAGTTCGTGACCGCCGCCGGGATCACTCTCGGCA TGGACGAGCTGTACAAGGGCGCTGGAGCAGGGGCTGGAGCCGGAGCTGAC TCCAAAGAATCATTAACTCCTGGTAGAGAAGAAAACCCCAGCAGTGTGCT TGCTCAGGAGAGGGGAGATGTGATGGACTTCTATAAAACCCTAAGAGGAG GAGCTACTGTGAAGGTTTCTGCGTCTTCACCCTCACTGGCTGTCGCTTCT CAATCAGACTCCAAGCAGCGAAGACTTTTGGTTGATTTTCCAAAAGGCTC AGTAAGCAATGCGCAGCAGCCAGATCTGTCCAAAGCAGTTTCACTCTCAA TGGGACTGTATATGGGAGAGACAGAAACAAAAGTGATGGGAAATGACCTG GGATTCCCACAGCAGGGCCAAATCAGCCTTTCCTCGGGGGAAACAGACTT AAAGCTTTTGGAAGAAAGCATTGCAAACCTCAATAGGTCGACCAGTGTTC CAGAGAACCCCAAGAGTTCAGCATCCACTGCTGTGTCTGCTGCCCCCACA GAGAAGGAGTTTCCAAAAACTCACTCTGATGTATCTTCAGAACAGCAACA TTTGAAGGGCCAGACTGGCACCAACGGTGGCAATGTGAAATTGTATACCA CAGACCAAAGCACCTTTGACATTTTGCAGGATTTGGAGTTTTCTTCTGGG TCCCCAGGTAAAGAGACGAATGAGAGTCCTTGGAGATCAGACCTGTTGAT AGATGAAAACTGTTTGCTTTCTCCTCTGGCGGGAGAAGACGATTCATTCC TTTTGGAAGGAAACTCGAATGAGGACTGCAAGCCTCTCATTTTACCGGAC ACTAAACCCAAAATTAAGGATAATGGAGATCTGGTTTTGTCAAGCCCCAG TAATGTAACACTGCCCCAAGTGAAAACAGAAAAAGAAGATTTCATCGAAC TCTGCACCCCTGGGGTAATTAAGCAAGAGAAACTGGGCACAGTTTACTGT CAGGCAAGCTTTCCTGGAGCAAATATAATTGGTAATAAAATGTCTGCCAT TTCTGTTCATGGTGTGAGTACCTCTGGAGGACAGATGTACCACTATGACA TGAATACAGCATCCCTTTCTCAACAGCAGGATCAGAAGCCTATTTTTAAT GTCATTCCACCAATTCCCGTTGGTTCCGAAAATTGGAATAGGTGCCAAGG ATCTGGAGATGACAACTTGACTTCTCTGGGGACTCTGAACTTCCCTGGTC GAACAGTTTTTTCTAATGGCTATTCAAGCCCCAGCATGAGACCAGATGTA AGCTCTCCTCCATCCAGCTCCTCAACAGCAACAACAGGACCACCTCCCAA ACTCTGCCTGGTGTGCTCTGATGAAGCTTCAGGATGTCATTATGGAGTCT TAACTTGTGGAAGCTGTAAAGTTTTCTTCAAAAGAGCAGTGGAAGGACAG CACAATTACCTATGTGCTGGAAGGAATGATTGCATCATCGATAAAATTCG AAGAAAAAACTGCCCAGCATGCCGCTATCGAAAATGTCTTCAGGCTGGAA TGAACCTGGAAGCTCGAAAAACAAAGAAAAAAATAAAAGGAATTCAGCAG GCCACTACAGGAGTCTCACAAGAAACCTCTGAAAATCCTGGTAACAAAAC AATAGTTCCTGCAACGTTACCACAACTCACCCCTACCCTGGTGTCACTGT TGGAGGTTATTGAACCTGAAGTGTTATATGCAGGATATGATAGCTCTGTT CTGACGGCCGACCAGATGGTCAGTGCCTTGTTGGATGCTGAGCCCCCCAT ACTCTATTCCGAGTATGATCCTACCAGACCCTTCAGTGAAGCTTCGATGA TGGGCTTACTGACCAACCTGGCAGACAGGGAGCTGGTTCACATGATCAAC TGGGCGAAGAGGGTGCCAGGCTTTGTGGATTTGACCCTCCATGATCAGGT CCACCTTCTAGAATGTGCCTGGCTAGAGATCCTGATGATTGGTCTCGTCT GGCGCTCCATGGAGCACCCAGGGAAGCTACTGTTTGCTCCTAACTTGCTC TTGGACAGGAACCAGGGAAAATGTGTAGAGGGCATGGTGGAGATCTTCGA CATGCTGCTGGCTACATCATCTCGGTTCCGCATGATGAATCTGCAGGGAG AGGAGTTTGTGTGCCTCAAATCTATTATTTTGCTTAATTCTGGAGTGTAC ACATTTCTGTCCAGCACCCTGAAGTCTCTGGAAGAGAAGGACCATATCCA CCGAGTCCTGGACAAGATCACAGACACTTTGATCCACCTGATGGCCAAGG CAGGCCTGACCCTGCAGCAGCAGCACCAGCGGCTGGCCCAGCTCCTCCTC ATCCTCTCCCACATCAGGCACATGAGTAACAAAGGCATGGAGCATCTGTA CAGCATGAAGTGCAAGAACGTGGTGCCCCTCTATGACCTGCTGCTGGAGA TGCTGGACGCCCACCGCCTACATGCGCCCACTAGCCGTGGAGGGGCATCC GTGGAGGAGACGGACCAAAGCCACTTGGCCACTGCGGGCTCTACTTCATC GCATTCCTTGCAAAAGTATTACATCACGGGGGAGGCAGAGGGTTTCCCTG CCACGGTCTAA
[0034] SEQ ID NO: 2 is the amino acid sequence of traceable fusion protein eGFP-GR-ER310. The sequence includes the following features at the indicated amino acid positions: EGFP (1-239), GA Linker (240-249), hGR (250-800), and hER (801-1086).
TABLE-US-00002 MVSKGEELFTGVVPILVELDGDVNGHKFSVSGEGEGDATYGKLTLKFICT TGKLPVPWPTLVTTLTYGVQCFSRYPDHMKQHDFFKSAMPEGYVQERTIF FKDDGNYKTRAEVKFEGDTLVNRIELKGIDFKEDGNILGHKLEYNYNSHN VYIMADKQKNGIKVNFKIRHNIEDGSVQLADHYQQNTPIGDGPVLLPDNH YLSTQSALSKDPNEKRDHMVLLEFVTAAGITLGMDELYKGAGAGAGAGAD SKESLTPGREENPSSVLAQERGDVMDFYKTLRGGATVKVSASSPSLAVAS QSDSKQRRLLVDFPKGSVSNAQQPDLSKAVSLSMGLYMGETETKVMGNDL GFPQQGQISLSSGETDLKLLEESIANLNRSTSVPENPKSSASTAVSAAPT EKEFPKTHSDVSSEQQHLKGQTGTNGGNVKLYTTDQSTFDILQDLEFSSG SPGKETNESPWRSDLLIDENCLLSPLAGEDDSFLLEGNSNEDCKPLILPD TKPKIKDNGDLVLSSPSNVTLPQVKTEKEDFIELCTPGVIKQEKLGTVYC QASFPGANIIGNKMSAISVHGVSTSGGQMYHYDMNTASLSQQQDQKPIFN VIPPIPVGSENWNRCQGSGDDNLTSLGTLNFPGRTVFSNGYSSPSMRPDV SSPPSSSSTATTGPPPKLCLVCSDEASGCHYGVLTCGSCKVFFKRAVEGQ HNYLCAGRNDCIIDKIRRKNCPACRYRKCLQAGMNLEARKTKKKIKGIQQ ATTGVSQETSENPGNKTIVPATLPQLTPTLVSLLEVIEPEVLYAGYDSSV LTADQMVSALLDAEPPILYSEYDPTRPFSEASMMGLLTNLADRELVHMIN WAKRVPGFVDLTLHDQVHLLECAWLEILMIGLVWRSMEHPGKLLFAPNLL LDRNQGKCVEGMVEIFDMLLATSSRFRMMNLQGEEFVCLKSIILLNSGVY TFLSSTLKSLEEKDHIHRVLDKITDTLIHLMAKAGLTLQQQHQRLAQLLL ILSHIRHMSNKGMEHLYSMKCKNVVPLYDLLLEMLDAHRLHAPTSRGGAS VEETDQSHLATAGSTSSHSLQKYYITGEAEGFPATV*
[0035] SEQ ID NO: 3 is the nucleic acid sequence which encodes traceable fusion protein eGFP-GR-TR216. The sequence includes the following features at the indicated nucleotide positions: EGFP (1-717), GA Linker (718-747), hGR (748-2400), and hTR (2401-3141).
TABLE-US-00003 ATGGTGAGCAAGGGCGAGGAGCTGTTCACCGGGGTGGTGCCCATCCTGGT CGAGCTGGACGGCGACGTAAACGGCCACAAGTTCAGCGTGTCCGGCGAGG GCGAGGGCGATGCCACCTACGGCAAGCTGACCCTGAAGTTCATCTGCACC ACCGGCAAGCTGCCCGTGCCCTGGCCCACCCTCGTGACCACCCTGACCTA CGGCGTGCAGTGCTTCAGCCGCTACCCCGACCACATGAAGCAGCACGACT TCTTCAAGTCCGCCATGCCCGAAGGCTACGTCCAGGAGCGCACCATCTTC TTCAAGGACGACGGCAACTACAAGACCCGCGCCGAGGTGAAGTTCGAGGG CGACACCCTGGTGAACCGCATCGAGCTGAAGGGCATCGACTTCAAGGAGG ACGGCAACATCCTGGGGCACAAGCTGGAGTACAACTACAACAGCCACAAC GTCTATATCATGGCCGACAAGCAGAAGAACGGCATCAAGGTGAACTTCAA GATCCGCCACAACATCGAGGACGGCAGCGTGCAGCTCGCCGACCACTACC AGCAGAACACCCCCATCGGCGACGGCCCCGTGCTGCTGCCCGACAACCAC TACCTGAGCACCCAGTCCGCCCTGAGCAAAGACCCCAACGAGAAGCGCGA TCACATGGTCCTGCTGGAGTTCGTGACCGCCGCCGGGATCACTCTCGGCA TGGACGAGCTGTACAAGGGCGCTGGAGCAGGGGCTGGAGCCGGAGCTGAC TCCAAAGAATCATTAACTCCTGGTAGAGAAGAAAACCCCAGCAGTGTGCT TGCTCAGGAGAGGGGAGATGTGATGGACTTCTATAAAACCCTAAGAGGAG GAGCTACTGTGAAGGTTTCTGCGTCTTCACCCTCACTGGCTGTCGCTTCT CAATCAGACTCCAAGCAGCGAAGACTTTTGGTTGATTTTCCAAAAGGCTC AGTAAGCAATGCGCAGCAGCCAGATCTGTCCAAAGCAGTTTCACTCTCAA TGGGACTGTATATGGGAGAGACAGAAACAAAAGTGATGGGAAATGACCTG GGATTCCCACAGCAGGGCCAAATCAGCCTTTCCTCGGGGGAAACAGACTT AAAGCTTTTGGAAGAAAGCATTGCAAACCTCAATAGGTCGACCAGTGTTC CAGAGAACCCCAAGAGTTCAGCATCCACTGCTGTGTCTGCTGCCCCCACA GAGAAGGAGTTTCCAAAAACTCACTCTGATGTATCTTCAGAACAGCAACA TTTGAAGGGCCAGACTGGCACCAACGGTGGCAATGTGAAATTGTATACCA CAGACCAAAGCACCTTTGACATTTTGCAGGATTTGGAGTTTTCTTCTGGG TCCCCAGGTAAAGAGACGAATGAGAGTCCTTGGAGATCAGACCTGTTGAT AGATGAAAACTGTTTGCTTTCTCCTCTGGCGGGAGAAGACGATTCATTCC TTTTGGAAGGAAACTCGAATGAGGACTGCAAGCCTCTCATTTTACCGGAC ACTAAACCCAAAATTAAGGATAATGGAGATCTGGTTTTGTCAAGCCCCAG TAATGTAACACTGCCCCAAGTGAAAACAGAAAAAGAAGATTTCATCGAAC TCTGCACCCCTGGGGTAATTAAGCAAGAGAAACTGGGCACAGTTTACTGT CAGGCAAGCTTTCCTGGAGCAAATATAATTGGTAATAAAATGTCTGCCAT TTCTGTTCATGGTGTGAGTACCTCTGGAGGACAGATGTACCACTATGACA TGAATACAGCATCCCTTTCTCAACAGCAGGATCAGAAGCCTATTTTTAAT GTCATTCCACCAATTCCCGTTGGTTCCGAAAATTGGAATAGGTGCCAAGG ATCTGGAGATGACAACTTGACTTCTCTGGGGACTCTGAACTTCCCTGGTC GAACAGTTTTTTCTAATGGCTATTCAAGCCCCAGCATGAGACCAGATGTA AGCTCTCCTCCATCCAGCTCCTCAACAGCAACAACAGGACCACCTCCCAA ACTCTGCCTGGTGTGCTCTGATGAAGCTTCAGGATGTCATTATGGAGTCT TAACTTGTGGAAGCTGTAAAGTTTTCTTCAAAAGAGCAGTGGAAGGACAG CACAATTACCTATGTGCTGGAAGGAATGATTGCATCATCGATAAAATTCG AAGAAAAAACTGCCCAGCATGCCGCTATCGAAAATGTCTTCAGGCTGGAA TGAACCTGGAAGCTCGAAAAACAAAGAAAAAAATAAAAGGAATTCAGCAG GCCACTACAGGAGTCTCACAAGAAACCTCTGAAAATCCTGGTAACAAAAC AATAGTTCCTGCAACGTTACCACAACTCACCCCTACCCTGGTGTCACTGT TGGAGGTTATTGAACCTGAAGTGTTATATGCAGGATATGATAGCTCTGTT GACGAGGAATGGGAGCTCATCAAAACTGTCACCGAAGCCCATGTGGCGAC CAACGCCCAAGGCAGCCACTGGAAGCAAAAACGGAAATTCCTGCCAGAAG ACATTGGACAAGCACCAATAGTCAATGCCCCAGAAGGTGGAAAGGTTGAC TTGGAAGCCTTCAGCCATTTTACAAAAATCATCACACCAGCAATTACCAG AGTGGTGGATTTTGCCAAAAAGTTGCCTATGTTTTGTGAGCTGCCATGTG AAGACCAGATCATCCTCCTCAAAGGCTGCTGCATGGAGATCATGTCCCTT CGCGCTGCTGTGCGCTATGACCCAGAAAGTGAGACTTTAACCTTGAATGG GGAAATGGCAGTGACACGGGGCCAGCTGAAAAATGGGGGTCTTGGGGTGG TGTCAGACGCCATCTTTGACCTGGGCATGTCTCTGTCTTCTTTCAACCTG GATGACACTGAAGTAGCCCTCCTTCAGGCCGTCCTGCTGATGTCTTCAGA TCGCCCGGGGCTTGCCTGTGTTGAGAGAATAGAAAAGTACCAAGATAGTT TCCTGCTGGCCTTTGAACACTATATCAATTACCGAAAACACCACGTGACA CACTTTTGGCCAAAACTCCTGATGAAGGTGACAGATCTGCGGATGATAGG AGCCTGCCATGCCAGCCGCTTCCTGCACATGAAGGTGGAATGCCCCACAG AACTCTTCCCCCCTTTGTTCTTGGAAGTGTTCGAGGATTAA
[0036] SEQ ID NO: 4 is the amino acid sequence of traceable fusion protein eGFP-GR-TR216. The sequence includes the following features at the indicated amino acid positions: EGFP (1-290), GA Linker (240-249), hGR (250-800), and hTR (801-1046).
TABLE-US-00004 MVSKGEELFTGVVPILVELDGDVNGHKFSVSGEGEGDATYGKLTLKFICT TGKLPVPWPTLVTTLTYGVQCFSRYPDHMKQHDFFKSAMPEGYVQERTIF FKDDGNYKTRAEVKFEGDTLVNRIELKGIDFKEDGNILGHKLEYNYNSHN VYIMADKQKNGIKVNFKIRHNIEDGSVQLADHYQQNTPIGDGPVLLPDNH YLSTQSALSKDPNEKRDHMVLLEFVTAAGITLGMDELYKGAGAGAGAGAD SKESLTPGREENPSSVLAQERGDVMDFYKTLRGGATVKVSASSPSLAVAS QSDSKQRRLLVDFPKGSVSNAQQPDLSKAVSLSMGLYMGETETKVMGNDL GFPQQGQISLSSGETDLKLLEESIANLNRSTSVPENPKSSASTAVSAAPT EKEFPKTHSDVSSEQQHLKGQTGTNGGNVKLYTTDQSTFDILQDLEFSSG SPGKETNESPWRSDLLIDENCLLSPLAGEDDSFLLEGNSNEDCKPLILPD TKPKIKDNGDLVLSSPSNVTLPQVKTEKEDFIELCTPGVIKQEKLGTVYC QASFPGANIIGNKMSAISVHGVSTSGGQMYHYDMNTASLSQQQDQKPIFN VIPPIPVGSENWNRCQGSGDDNLTSLGTLNFPGRTVFSNGYSSPSMRPDV SSPPSSSSTATTGPPPKLCLVCSDEASGCHYGVLTCGSCKVFFKRAVEGQ HNYLCAGRNDCIIDKIRRKNCPACRYRKCLQAGMNLEARKTKKKIKGIQQ ATTGVSQETSENPGNKTIVPATLPQLTPTLVSLLEVIEPEVLYAGYDSSV DEEWELIKTVTEAHVATNAQGSHWKQKRKFLPEDIGQAPIVNAPEGGKVD LEAFSHFTKIITPAITRVVDFAKKLPMFCELPCEDQIILLKGCCMEIMSL RAAVRYDPESETLTLNGEMAVTRGQLKNGGLGVVSDAIFDLGMSLSSFNL DDTEVALLQAVLLMSSDRPGLACVERIEKYQDSFLLAFEHYINYRKHHVT HFWPKLLMKVTDLRMIGACHASRFLHMKVECPTELFPPLFLEVFED*
[0037] SEQ ID NO: 5 is the nucleic acid sequence which encodes traceable fusion protein pCI-nGFP-C656G. The sequence includes the following features at the indicated nucleotide positions: 6X His tag (8-24), HA Tag (28-59), GFP (60-780), GA Linker (781-810), and Rat
[0038] GR (829-3213), containing within it the C656G mutation (single point mutation T->G, codon changes from TGC->GGC) (2791; underlined & bold)
TABLE-US-00005 ATGGCCCACCATCACCACCATCACGGATATCCATACGACGTGCCAGATTA CGCTCAGTCGAGTGCCATGAGTAAAGGAGAAGAACTTTTCACTGGAGTTG TCCCAATTCTTGTTGAATTAGATGGTGATGTTAATGGGCACAAATTTTCT GTCAGTGGAGAGGGTGAAGGTGATGCAACATACGGAAAACTTACCCTTAA ATTTATTTGCACTACTGGAAAACTACCTGTTCCTTGGCCAACACTTGTCA CTACTTTCACTTATGGTGTTCAATGCTTTTCAAGATACCCAGATCATATG AAACAGCATGACTTTTTCAAGAGTGCCATGCCCGAAGGTTATGTACAGGA AAGAACTATATTTTTCAAAGATGACGGGAACTACAAGACACGTGCTGAAG TCAAGTTTGAAGGTGATACCCTTGTTAATAGAATCGAGTTAAAAGGTATT GATTTTAAAGAAGATGGAAACATTCTTGGACACAAATTGGAATACAACTA TAACTCACACAATGTATACATCATGGCAGACAAACAAAAGAATGGAATCA AAGTTAACTTCAAAATTAGACACAACATTGAAGATGGAAGCGTTCAACTA GCAGACCATTATCAACAAAATACTCCAATTGGCGATGGCCCTGTCCTTTT ACCAGACAACCATTACCTGTCCACACAATCTGCCCTTTCGAAAGATCCCA ACGAAAAGAGAGACCACATGGTCCTTCTTGAGTTTGTAACAGCTGCTGGG ATTACACATGGCATGGATGAACTATACAAAGGCGCCGGCGCTGGTGCTGG TGCTGGCGCCATCAGCGCGCTGATCCTGGACTCCAAAGAATCCTTAGCTC CCCCTGGTAGAGACGAAGTCCCTGGCAGTTTGCTTGGCCAGGGGAGGGGG AGCGTAATGGACTTTTATAAAAGCCTGAGGGGAGGAGCTACAGTCAAGGT TTCTGCATCTTCGCCCTCAGTGGCTGCTGCTTCTCAGGCAGATTCCAAGC AGCAGAGGATTCTCCTTGATTTCTCGAAAGGCTCCACAAGCAATGTGCAG CAGCGACAGCAGCAGCAGCAGCAGCAGCAGCAGCAGCAGCAGCAGCAGCA GCAGCAGCAGCAGCCAGGCTTATCCAAAGCCGTTTCACTGTCCATGGGGC TGTATATGGGAGAGACAGAAACAAAAGTGATGGGGAATGACTTGGGCTAC CCACAGCAGGGCCAACTTGGCCTTTCCTCTGGGGAAACAGACTTTCGGCT TCTGGAAGAAAGCATTGCAAACCTCAATAGGTCGACCAGCGTTCCAGAGA ACCCCAAGAGTTCAACGTCTGCAACTGGGTGTGCTACCCCGACAGAGAAG GAGTTTCCCAAAACTCACTCGGATGCATCTTCAGAACAGCAAAATCGAAA AAGCCAGACCGGCACCAACGGAGGCAGTGTGAAATTGTATCCCACAGACC AAAGCACCTTTGACCTCTTGAAGGATTTGGAGTTTTCCGCTGGGTCCCCA AGTAAAGACACAAACGAGAGTCCCTGGAGATCAGATCTGTTGATAGATGA AAACTTGCTTTCTCCTTTGGCGGGAGAAGATGATCCATTCCTTCTCGAAG GGAACACGAATGAGGATTGTAAGCCTCTTATTTTACCGGACACTAAACCT AAAATTAAGGATACTGGAGATACAATCTTATCAAGTCCCAGCAGTGTGGC ACTACCCCAAGTGAAAACAGAAAAAGATGATTTCATTGAACTTTGCACCC CCGGGGTAATTAAGCAAGAGAAACTGGGCCCAGTTTATTGTCAGGCAAGC TTTTCTGGGACAAATATAATTGGTAATAAAATGTCTGCCATTTCTGTTCA TGGTGTGAGTACCTCTGGAGGACAGATGTACCACTATGACATGAATACAG CATCCCTTTCTCAGCAGCAGGATCAGAAGCCTGTTTTTAATGTCATTCCA CCAATTCCTGTTGGTTCTGAAAACTGGAATAGGTGCCAAGGCTCCGGAGA GGACAGCCTGACTTCCTTGGGGGCTCTGAACTTCCCAGGCCGGTCAGTGT TTTCTAATGGGTACTCAAGCCCTGGAATGAGACCAGATGTAAGCTCTCCT CCATCCAGCTCGTCAGCAGCCACGGGACCACCTCCCAAGCTCTGCCTGGT GTGCTCCGATGAAGCTTCAGGATGTCATTACGGGGTGCTGACATGTGGAA GCTGCAAAGTATTCTTTAAAAGAGCAGTGGAAGGACAGCACAATTACCTT TGTGCTGGAAGAAACGATTGCATCATTGATAAAATTCGAAGGAAAAACTG CCCAGCATGCCGCTATCGGAAATGTCTTCAGGCTGGAATGAACCTTGAAG CTCGAAAAACAAAGAAAAAAATCAAAGGGATTCAGCAAGCCACTGCAGGA GTCTCACAAGACACTTCGGAAAATCCTAACAAAACAATAGTTCCTGCAGC ATTACCACAGCTCACCCCTACCTTGGTGTCACTGCTGGAGGTGATTGAAC CCGAGGTGTTGTATGCAGGATATGATAGCTCTGTTCCAGATTCAGCATGG AGAATTATGACCACACTCAACATGTTAGGTGGGCGTCAAGTGATTGCAGC AGTGAAATGGGCAAAGGCGATACTAGGCTTGAGAAACTTACACCTCGATG ACCAAATGACCCTGCTACAGTACTCATGGATGTTTCTCATGGCATTTGCC TTGGGTTGGAGATCATACAGACAATCAAGCGGAAACCTGCTCTGCTTTGC TCCTGATCTGATTATTAATGAGCAGAGAATGTCTCTACCCGGCATGTATG ACCAATGTAAACACATGCTGTTTGTCTCCTCTGAATTACAAAGATTGCAG GTATCCTATGAAGAGTATCTCTGTATGAAAACCTTACTGCTTCTCTCCTC AGTTCCTAAGGAAGGTCTGAAGAGCCAAGAGTTATTTGATGAGATTCGAA TGACTTATATCAAAGAGCTAGGAAAAGCCATCGTCAAAAGGGAAGGGAAC TCCAGTCAGAACTGGCAACGGTTTTACCAACTGACAAAGCTTCTGGACTC CATGCATGAGGTGGTTGAGAATCTCCTTACCTACTGCTTCCAGACATTTT TGGATAAGACCATGAGTATTGAATTCCCAGAGATGTTAGCTGAAATCATC ACTAATCAGATACCAAAATATTCAAATGGAAATATCAAAAAGCTTCTGTT TCATCAAAAATGA
[0039] SEQ ID NO: 6 is the amino acid sequence of traceable fusion protein pCI-nGFP-C656G. The sequence includes the following features at the indicated amino acid positions: 6X His tag (3-8), HA Tag (10-18), GFP (23-260), GA Linker (261-270), and Rat GR (277-1070), containing within it the C656G mutation (Cysteine to Glycine) (931; underlined & bold).
TABLE-US-00006 MAHHHHHHGYPYDVPDYAQSSAMSKGEELFTGVVPILVELDGDVNGHKFS VSGEGEGDATYGKLTLKFICTTGKLPVPWPTLVTTFTYGVQCFSRYPDHM KQHDFFKSAMPEGYVQERTIFFKDDGNYKTRAEVKFEGDTLVNRIELKGI DFKEDGNILGHKLEYNYNSHNVYIMADKQKNGIKVNFKIRHNIEDGSVQL ADHYQQNTPIGDGPVLLPDNHYLSTQSALSKDPNEKRDHMVLLEFVTAAG ITHGMDELYKGAGAGAGAGAISALILDSKESLAPPGRDEVPGSLLGQGRG SVMDFYKSLRGGATVKVSASSPSVAAASQADSKQQRILLDFSKGSTSNVQ QRQQQQQQQQQQQQQQQQQQQPGLSKAVSLSMGLYMGETETKVMGNDLGY PQQGQLGLSSGETDFRLLEESIANLNRSTSVPENPKSSTSATGCATPTEK EFPKTHSDASSEQQNRKSQTGTNGGSVKLYPTDQSTFDLLKDLEFSAGSP SKDTNESPWRSDLLIDENLLSPLAGEDDPFLLEGNTNEDCKPLILPDTKP KIKDTGDTILSSPSSVALPQVKTEKDDFIELCTPGVIKQEKLGPVYCQAS FSGTNIIGNKMSAISVHGVSTSGGQMYHYDMNTASLSQQQDQKPVFNVIP PIPVGSENWNRCQGSGEDSLTSLGALNFPGRSVFSNGYSSPGMRPDVSSP PSSSSAATGPPPKLCLVCSDEASGCHYGVLTCGSCKVFFKRAVEGQHNYL CAGRNDCIIDKIRRKNCPACRYRKCLQAGMNLEARKTKKKIKGIQQATAG VSQDTSENPNKTIVPAALPQLTPTLVSLLEVIEPEVLYAGYDSSVPDSAW RIMTTLNMLGGRQVIAAVKWAKAILGLRNLHLDDQMTLLQYSWMFLMAFA LGWRSYRQSSGNLLCFAPDLIINEQRMSLPGMYDQCKHMLFVSSELQRLQ VSYEEYLCMKTLLLLSSVPKEGLKSQELFDEIRMTYIKELGKAIVKREGN SSQNWQRFYQLTKLLDSMHEVVENLLTYCFQTFLDKTMSIEFPEMLAEII TNQIPKYSNGNIKKLLFHQK*
[0040] SEQ ID NO: 7 is the nucleic acid sequence which encodes traceable fusion protein eGFP-hAR. The sequence includes the following features at the indicated nucleotide positions: EGFP (1-717), Vector polylinker (718-752), and hAR (753-3516).
TABLE-US-00007 ATGGTGAGCAAGGGCGAGGAGCTGTTCACCGGGGTGGTGCCCATCCTGGT CGAGCTGGACGGCGACGTAAACGGCCACAAGTTCAGCGTGTCCGGCGAGG GCGAGGGCGATGCCACCTACGGCAAGCTGACCCTGAAGTTCATCTGCACC ACCGGCAAGCTGCCCGTGCCCTGGCCCACCCTCGTGACCACCCTGACCTA CGGCGTGCAGTGCTTCAGCCGCTACCCCGACCACATGAAGCAGCACGACT TCTTCAAGTCCGCCATGCCCGAAGGCTACGTCCAGGAGCGCACCATCTTC TTCAAGGACGACGGCAACTACAAGACCCGCGCCGAGGTGAAGTTCGAGGG CGACACCCTGGTGAACCGCATCGAGCTGAAGGGCATCGACTTCAAGGAGG ACGGCAACATCCTGGGGCACAAGCTGGAGTACAACTACAACAGCCACAAC GTCTATATCATGGCCGACAAGCAGAAGAACGGCATCAAGGTGAACTTCAA GATCCGCCACAACATCGAGGACGGCAGCGTGCAGCTCGCCGACCACTACC AGCAGAACACCCCCATCGGCGACGGCCCCGTGCTGCTGCCCGACAACCAC TACCTGAGCACCCAGTCCGCCCTGAGCAAAGACCCCAACGAGAAGCGCGA TCACATGGTCCTGCTGGAGTTCGTGACCGCCGCCGGGATCACTCTCGGCA TGGACGAGCTGTACAAGTCCGGACTCAGATCTCGAGCTCAAGCTTCGAAT TCGATGGAAGTGCAGTTAGGGCTGGGAAGGGTCTACCCTCGGCCGCCGTC CAAGACCTACCGAGGAGCTTTCCAGAATCTGTTCCAGAGCGTGCGCGAAG TGATCCAGAACCCGGGCCCCAGGCACCCAGAGGCCGCGAGCGCAGCACCT CCCGGCGCCAGTTTGCTGCTGCTGCAGCAGCAGCAGCAGCAGCAGCAGCA GCAGCAGCAGCAGCAGCAGCAGCAGCAGCAGCAGCAGCAGCAAGAGACTA GCCCCAGGCAGCAGCAGCAGCAGCAGGGTGAGGATGGTTCTCCCCAAGCC CATCGTAGAGGCCCCACAGGCTACCTGGTCCTGGATGAGGAACAGCAACC TTCACAGCCGCAGTCGGCCCTGGAGTGCCACCCCGAGAGAGGTTGCGTCC CAGAGCCTGGAGCCGCCGTGGCCGCCAGCAAGGGGCTGCCGCAGCAGCTG CCAGCACCTCCGGACGAGGATGACTCAGCTGCCCCATCCACGTTGTCCCT GCTGGGCCCCACTTTCCCCGGCTTAAGCAGCTGCTCCGCTGACCTTAAAG ACATCCTGAGCGAGGCCAGCACCATGCAACTCCTTCAGCAACAGCAGCAG GAAGCAGTATCCGAAGGCAGCAGCAGCGGGAGAGCGAGGGAGGCCTCGGG GGCTCCCACTTCCTCCAAGGACAATTACTTAGGGGGCACTTCGACCATTT CTGACAACGCCAAGGAGTTGTGTAAGGCAGTGTCGGTGTCCATGGGCCTG GGTGTGGAGGCGTTGGAGCATCTGAGTCCAGGGGAACAGCTTCGGGGGGA TTGCATGTACGCCCCACTTTTGGGAGTTCCACCCGCTGTGCGTCCCACTC CTTGTGCCCCATTGGCCGAATGCAAAGGTTCTCTGCTAGACGACAGCGCA GGCAAGAGCACTGAAGATACTGCTGAGTATTCCCCTTTCAAGGGAGGTTA CACCAAAGGGCTAGAAGGCGAGAGCCTAGGCTGCTCTGGCAGCGCTGCAG CAGGGAGCTCCGGGACACTTGAACTGCCGTCTACCCTGTCTCTCTACAAG TCCGGAGCACTGGACGAGGCAGCTGCGTACCAGAGTCGCGACTACTACAA CTTTCCACTGGCTCTGGCCGGACCGCCGCCCCCTCCGCCGCCTCCCCATC CCCACGCTCGCATCAAGCTGGAGAACCCGCTGGACTACGGCAGCGCCTGG GCGGCTGCGGCGGCGCAGTGCCGCTATGGGGACCTGGCGAGCCTGCATGG CGCGGGTGCAGCGGGACCCGGTTCTGGGTCACCCTCAGCCGCCGCTTCCT CATCCTGGCACACTCTCTTCACAGCCGAAGAAGGCCAGTTGTATGGACCG TGTGGTGGTGGTGGGGGTGGTGGCGGCGGCGGCGGCGGCGGCGGCGGCGG CGGCGGCGGCGGCGGCGGCGGCGAGGCGGGAGCTGTAGCCCCCTACGGCT ACACTCGGCCCCCTCAGGGGCTGGCGGGCCAGGAAAGCGACTTCACCGCA CCTGATGTGTGGTACCCTGGCGGCATGGTGAGCAGAGTGCCCTATCCCAG TCCCACTTGTGTCAAAAGCGAAATGGGCCCCTGGATGGATAGCTACTCCG GACCTTACGGGGACATGCGTTTGGAGACTGCCAGGGACCATGTTTTGCCC ATTGACTATTACTTTCCACCCCAGAAGACCTGCCTGATCTGTGGAGATGA AGCTTCTGGGTGTCACTATGGAGCTCTCACATGTGGAAGCTGCAAGGTCT TCTTCAAAAGAGCCGCTGAAGGGAAACAGAAGTACCTGTGCGCCAGCAGA AATGATTGCACTATTGATAAATTCCGAAGGAAAAATTGTCCATCTTGTCG TCTTCGGAAATGTTATGAAGCAGGGATGACTCTGGGAGCCCGGAAGCTGA AGAAACTTGGTAATCTGAAACTACAGGAGGAAGGAGAGGCTTCCAGCACC ACCAGCCCCACTGAGGAGACAACCCAGAAGCTGACAGTGTCACACATTGA AGGCTATGAATGTCAGCCCATCTTTCTGAATGTCCTGGAAGCCATTGAGC CAGGTGTAGTGTGTGCTGGACACGACAACAACCAGCCCGACTCCTTTGCA GCCTTGCTCTCTAGCCTCAATGAACTGGGAGAGAGACAGCTTGTACACGT GGTCAAGTGGGCCAAGGCCTTGCCTGGCTTCCGCAACTTACACGTGGACG ACCAGATGGCTGTCATTCAGTACTCCTGGATGGGGCTCATGGTGTTTGCC ATGGGCTGGCGATCCTTCACCAATGTCAACTCCAGGATGCTCTACTTCGC CCCTGATCTGGTTTTCAATGAGTACCGCATGCACAAGTCCCGGATGTACA GCCAGTGTGTCCGAATGAGGCACCTCTCTCAAGAGTTTGGATGGCTCCAA ATCACCCCCCAGGAATTCCTGTGCATGAAAGCACTGCTACTCTTCAGCAT TATTCCAGTGGATGGGCTGAAAAATCAAAAATTCTTTGATGAACTTCGAA TGAACTACATCAAGGAACTCGATCGTATCATTGCATGCAAAAGAAAAAAT CCCACATCCTGCTCAAGACGCTTCTACCAGCTCACCAAGCTCCTGGACTC CGTGCAGCCTATTGCGAGAGAGCTGCATCAGTTCACTTTTGACCTGCTAA TCAAGTCACACATGGTGAGCGTGGACTTTCCGGAAATGATGGCAGAGATC ATCTCTGTGCAAGTGCCCAAGATCCTTTCTGGGAAAGTCAAGCCCATCTA TTTCCACACCCAGTGA
[0041] SEQ ID NO: 8 is the amino acid sequence of traceable fusion protein eGFP-hAR. The sequence includes the following features at the indicated amino acid positions: EGFP (1-239), Vector polylinker (240-251), and hAR (252-1171).
TABLE-US-00008 MVSKGEELFTGVVPILVELDGDVNGHKFSVSGEGEGDATYGKLTLKFICT TGKLPVPWPTLVTTLTYGVQCFSRYPDHMKQHDFFKSAMPEGYVQERTIF FKDDGNYKTRAEVKFEGDTLVNRIELKGIDFKEDGNILGHKLEYNYNSHN VYIMADKQKNGIKVNFKIRHNIEDGSVQLADHYQQNTPIGDGPVLLPDNH YLSTQSALSKDPNEKRDHMVLLEFVTAAGITLGMDELYKSGLRSRAQASN SMEVQLGLGRVYPRPPSKTYRGAFQNLFQSVREVIQNPGPRHPEAASAAP PGASLLLLQQQQQQQQQQQQQQQQQQQQQQQETSPRQQQQQQGEDGSPQA HRRGPTGYLVLDEEQQPSQPQSALECHPERGCVPEPGAAVAASKGLPQQL PAPPDEDDSAAPSTLSLLGPTFPGLSSCSADLKDILSEASTMQLLQQQQQ EAVSEGSSSGRAREASGAPTSSKDNYLGGTSTISDNAKELCKAVSVSMGL GVEALEHLSPGEQLRGDCMYAPLLGVPPAVRPTPCAPLAECKGSLLDDSA GKSTEDTAEYSPFKGGYTKGLEGESLGCSGSAAAGSSGTLELPSTLSLYK SGALDEAAAYQSRDYYNFPLALAGPPPPPPPPHPHARIKLENPLDYGSAW AAAAAQCRYGDLASLHGAGAAGPGSGSPSAAASSSWHTLFTAEEGQLYGP CGGGGGGGGGGGGGGGGGGGGGGGEAGAVAPYGYTRPPQGLAGQESDFTA PDVWYPGGMVSRVPYPSPTCVKSEMGPWMDSYSGPYGDMRLETARDHVLP IDYYFPPQKTCLICGDEASGCHYGALTCGSCKVFFKRAAEGKQKYLCASR NDCTIDKFRRKNCPSCRLRKCYEAGMTLGARKLKKLGNLKLQEEGEASST TSPTEETTQKLTVSHIEGYECQPIFLNVLEAIEPGVVCAGHDNNQPDSFA ALLSSLNELGERQLVHVVKWAKALPGFRNLHVDDQMAVIQYSWMGLMVFA MGWRSFTNVNSRMLYFAPDLVFNEYRMHKSRMYSQCVRMRHLSQEFGWLQ ITPQEFLCMKALLLFSIIPVDGLKNQKFFDELRMNYIKELDRIIACKRKN PTSCSRRFYQLTKLLDSVQPIARELHQFTFDLLIKSHMVSVDFPEMMAEI ISVQVPKILSGKVKPIYFHTQ*
[0042] SEQ ID NO: 9 is the nucleic acid sequence which encodes traceable fusion protein pRevTRE-GFP-mAhR. The sequence includes the following features at the indicated nucleotide positions: 6X His tag (8-27), HA Tag (28-58), GFP (59- 780), GA Linker (781- 810), and mouse AhR (829-3243).
TABLE-US-00009 ATGGCCCACCATCACCACCATCACGGATATCCATACGACGTGCCAGATTA CGCTCAGTCGAGTGCCATGAGTAAAGGAGAAGAACTTTTCACTGGAGTTG TCCCAATTCTTGTTGAATTAGATGGTGATGTTAATGGGCACAAATTTTCT GTCAGTGGAGAGGGTGAAGGTGATGCAACATACGGAAAACTTACCCTTAA ATTTATTTGCACTACTGGAAAACTACCTGTTCCTTGGCCAACACTTGTCA CTACTTTCACTTATGGTGTTCAATGCTTTTCAAGATACCCAGATCATATG AAACAGCATGACTTTTTCAAGAGTGCCATGCCCGAAGGTTATGTACAGGA AAGAACTATATTTTTCAAAGATGACGGGAACTACAAGACACGTGCTGAAG TCAAGTTTGAAGGTGATACCCTTGTTAATAGAATCGAGTTAAAAGGTATT GATTTTAAAGAAGATGGAAACATTCTTGGACACAAATTGGAATACAACTA TAACTCACACAATGTATACATCATGGCAGACAAACAAAAGAATGGAATCA AAGTTAACTTCAAAATTAGACACAACATTGAAGATGGAAGCGTTCAACTA GCAGACCATTATCAACAAAATACTCCAATTGGCGATGGCCCTGTCCTTTT ACCAGACAACCATTACCTGTCCACACAATCTGCCCTTTCGAAAGATCCCA ACGAAAAGAGAGACCACATGGTCCTTCTTGAGTTTGTAACAGCTGCTGGG ATTACACATGGCATGGATGAACTATACAAAGGCGCCGGCGCTGGAGCTGG TGCTGGCGCCATCAGCGCGCTGATCCTGAGCAGCGGCGCCAACATCACCT ATGCCAGCCGCAAGCGGCGCAAGCCGGTGCAGAAAACAGTAAAGCCCATC CCCGCTGAAGGAATTAAGTCAAATCCTTCTAAGCGACACAGAGACCGGCT GAACACAGAGTTAGACCGCCTGGCCAGCCTGCTGCCCTTCCCGCAAGATG TTATTAATAAGCTGGACAAACTCTCTGTTCTTAGGCTCAGCGTCAGCTAC CTGAGGGCCAAGAGCTTCTTTGATGTTGCATTAAAGTCCACCCCTGCTGA CAGAAATGGAGGCCAGGACCAGTGTAGAGCACAAATCAGAGACTGGCAGG ATTTGCAAGAAGGAGAGTTCTTGTTACAGGCGCTGAATGGCTTTGTGCTG GTTGTCACAGCAGATGCCTTGGTCTTCTATGCTTCCTCCACTATCCAAGA TTACCTGGGCTTTCAGCAGTCTGATGTCATCCATCAGAGCGTATATGAGC TCATCCATACAGAAGACCGGGCGGAATTCCAGCGCCAGCTTCACTGGGCT CTAAACCCAGACTCTGCACAAGGAGTGGACGAAGCCCATGGCCCTCCACA GGCAGCAGTCTATTATACCCCAGACCAGCTTCCTCCAGAGAACGCTTCTT TCATGGAGAGGTGCTTCAGGTGCCGGCTGAGGTGCCTGCTGGATAATTCA TCTGGTTTTCTGGCAATGAATTTCCAAGGGAGGTTAAAGTATCTTCATGG ACAGAACAAGAAAGGGAAGGACGGAGCGCTGCTTCCTCCACAACTGGCTT TGTTTGCAATAGCTACTCCACTTCAGCCACCCTCCATCCTGGAAATTCGA ACCAAAAACTTCATCTTCAGGACCAAACACAAGCTAGACTTCACACCTAT TGGTTGTGATGCCAAAGGGCAGCTTATTCTGGGCTATACAGAAGTAGAGC TGTGCACAAGAGGATCGGGGTACCAGTTCATCCATGCTGCAGACATACTT CACTGTGCAGAATCCCACATCCGCATGATTAAGACTGGAGAAAGTGGCAT GACAGTTTTCCGGCTTCTTGCAAAACACAGTCGCTGGAGGTGGGTCCAGT CCAATGCACGCTTGATTTACAGAAATGGAAGACCAGATTACATCATCGCC ACTCAGAGACCACTGACGGATGAAGAAGGACGAGAGCATTTACAGAAGCG AAGTACGTCGCTGCCCTTCATGTTTGCTACCGGAGAGGCTGTGTTGTACG AGATCTCCAGCCCTTTCTCTCCCATAATGGATCCCCTACCAATACGCACC AAAAGCAACACTAGCAGGAAAGACTGGGCTCCCCAGTCAACCCCAAGTAA GGATTCTTTCCACCCCAGTTCTCTTATGAGTGCCCTCATCCAGCAGGATG AGTCCATCTATCTGTGTCCTCCTTCAAGCCCTGCGCTGTTAGACAGCCAT TTTCTCATGGGCTCCGTGAGCAAGTGCGGGAGTTGGCAAGACAGCTTTGC GGCCGCAGGAAGTGAGGCTGCGCTGAAACATGAGCAAATTGGCCATGCTC AGGACGTGAACCTTGCACTCTCTGGCGGCCCCTCAGAGCTCTTTCCGGAT AATAAAAATAATGACTTGTACAGCATCATGAGGAACCTTGGGATTGATTT TGAAGATATCAGAAGCATGCAGAACGAGGAGTTCTTCAGAACTGACTCCA CCGCTGCTGGTGAGGTTGACTTCAAAGACATCGACATAACGGACGAAATC CTGACCTACGTGCAGGATTCCCTGAACAATTCAACTTTGCTGAACTCGGC TTGCCAGCAGCAGCCTGTGACTCAGCACCTAAGCTGTATGCTGCAGGAGC GCCTGCAACTAGAGCAACAGCAACAGCTTCAGCAGCCCCCGCCGCAGGCT CTGGAGCCCCAGCAGCAGCTGTGTCAGATGGTGTGCCCCCAGCAAGATCT GGGTCCGAAGCACACGCAAATCAACGGCACGTTTGCAAGTTGGAACCCCA CCCCTCCCGTGTCTTTCAACTGTCCCCAGCAGGAACTAAAGCACTATCAG CTCTTTTCCAGCTTACAGGGGACTGCTCAGGAATTTCCCTACAAACCAGA GGTGGACAGTGTGCCTTACACACAGAACTTTGCTCCCTGTAATCAGCCTC TGCTTCCAGAACATTCCAAGAGTGTGCAGTTGGACTTCCCTGGAAGGGAT TTTGAACCGTCCCTGCATCCCACTACTTCTAATTTAGATTTTGTCAGTTG TTTACAAGTTCCTGAAAACCAAAGTCATGGGATAAACTCACAGTCCGCCA TGGTCAGTCCTCAGGCATACTATGCTGGGGCCATGTCCATGTATCAGTGC CAGCCAGGGCCACAGCGCACCCCTGTGGACCAGACGCAGTACAGCTCTGA AATTCCAGGTTCTCAGGCATTCCTAAGCAAGGTGCAGAGTTGA
[0043] SEQ ID NO: 10 is the amino acid sequence of traceable fusion protein pRevTRE-GFP-mAhR. The sequence includes the following features at the indicated nucleotide positions: 6X His tag (3-8), HA Tag (9-18), GFP (23-260), GA Linker (261-270), and Mouse AhR (277-1080).
TABLE-US-00010 MAHHHHHHGYPYDVPDYAQSSAMSKGEELFTGVVPILVELDGDVNGHKFS VSGEGEGDATYGKLTLKFICTTGKLPVPWPTLVTTFTYGVQCFSRYPDHM KQHDFFKSAMPEGYVQERTIFFKDDGNYKTRAEVKFEGDTLVNRIELKGI DFKEDGNILGHKLEYNYNSHNVYIMADKQKNGIKVNFKIRHNIEDGSVQL ADHYQQNTPIGDGPVLLPDNHYLSTQSALSKDPNEKRDHMVLLEFVTAAG ITHGMDELYKGAGAGAGAGAISALILSSGANITYASRKRRKPVQKTVKPI PAEGIKSNPSKRHRDRLNTELDRLASLLPFPQDVINKLDKLSVLRLSVSY LRAKSFFDVALKSTPADRNGGQDQCRAQIRDWQDLQEGEFLLQALNGFVL VVTADALVFYASSTIQDYLGFQQSDVIHQSVYELIHTEDRAEFQRQLHWA LNPDSAQGVDEAHGPPQAAVYYTPDQLPPENASFMERCFRCRLRCLLDNS SGFLAMNFQGRLKYLHGQNKKGKDGALLPPQLALFAIATPLQPPSILEIR TKNFIFRTKHKLDFTPIGCDAKGQLILGYTEVELCTRGSGYQFIHAADIL HCAESHIRMIKTGESGMTVFRLLAKHSRWRWVQSNARLIYRNGRPDYIIA TQRPLTDEEGREHLQKRSTSLPFMFATGEAVLYEISSPFSPIMDPLPIRT KSNTSRKDWAPQSTPSKDSFHPSSLMSALIQQDESIYLCPPSSPALLDSH FLMGSVSKCGSWQDSFAAAGSEAALKHEQIGHAQDVNLALSGGPSELFPD NKNNDLYSIMRNLGIDFEDIRSMQNEEFFRTDSTAAGEVDFKDIDITDEI LTYVQDSLNNSTLLNSACQQQPVTQHLSCMLQERLQLEQQQQLQQPPPQA LEPQQQLCQMVCPQQDLGPKHTQINGTFASWNPTPPVSFNCPQQELKHYQ LFSSLQGTAQEFPYKPEVDSVPYTQNFAPCNQPLLPEHSKSVQLDFPGRD FEPSLHPTTSNLDFVSCLQVPENQSHGINSQSAMVSPQAYYAGAMSMYQC QPGPQRTPVDQTQYSSEIPGSQAFLSKVQS*
[0044] SEQ ID NO: 11 is the nucleic acid sequence which encodes traceable fusion protein eGFP-ratMR. The sequence includes the following features at the indicated nucleotide positions: EGFP (1-717), Vector polylinker (718-732), and rat MR (733-368).
TABLE-US-00011 ATGGTGAGCAAGGGCGAGGAGCTGTTCACCGGGGTGGTGCCCATCCTGGT CGAGCTGGACGGCGACGTAAACGGCCACAAGTTCAGCGTGTCCGGCGAGG GCGAGGGCGATGCCACCTACGGCAAGCTGACCCTGAAGTTCATCTGCACC ACCGGCAAGCTGCCCGTGCCCTGGCCCACCCTCGTGACCACCCTGACCTA CGGCGTGCAGTGCTTCAGCCGCTACCCCGACCACATGAAGCAGCACGACT TCTTCAAGTCCGCCATGCCCGAAGGCTACGTCCAGGAGCGCACCATCTTC TTCAAGGACGACGGCAACTACAAGACCCGCGCCGAGGTGAAGTTCGAGGG CGACACCCTGGTGAACCGCATCGAGCTGAAGGGCATCGACTTCAAGGAGG ACGGCAACATCCTGGGGCACAAGCTGGAGTACAACTACAACAGCCACAAC GTCTATATCATGGCCGACAAGCAGAAGAACGGCATCAAGGTGAACTTCAA GATCCGCCACAACATCGAGGACGGCAGCGTGCAGCTCGCCGACCACTACC AGCAGAACACCCCCATCGGCGACGGCCCCGTGCTGCTGCCCGACAACCAC TACCTGAGCACCCAGTCCGCCCTGAGCAAAGACCCCAACGAGAAGCGCGA TCACATGGTCCTGCTGGAGTTCGTGACCGCCGCCGGGATCACTCTCGGCA TGGACGAGCTGTACAAGTCCGGACTCAGATCTATGGAAACCAAAGGCTAC CACAGTCTCCCTGAAGGCCTAGACATGGAAAGGCGCTGGAGTCAAGTGTC TCAGACCTTGGAGCGTTCTTCACTTGGACCTGCAGAGAGGACCACTGAGA ACAACTACATGGAGATTGTCAACGTCAGCTGCGTTTCCGGTGCTATTCCG AACAACAGTACTCAAGGAAGCAGCAAAGAAAAGCACGAATTACTCCCTTA CATTCAGCAAGACAATAGTCGGTCTGGGATTTTGCCATCAGATATTAAAA CTGAGCTGGAATCCAAGGAACTTTCAGCCACGGTAGCTGAGTCCATGGGT TTATATATGGATTCTGTGAGAGATGCCGAGTACACTTACGATCAGCAGAA CCAACAAGGAAGCCTGAGCCCAACAAAGATTTATCAAAACATGGAGCAGC TGGTGAAGTTTTACAAAGAGAATGGTCACAGGTCCTCCACACTGAGTGCT ATGAGCAGACCTTTGAGGTCATTCATGCCTGACTCTGCAGCCTCCATGAA TGGTGGGGCCTTGCGTGCCATCGTGAAGAGCCCTATCATCTGTCATGAGA AGAGCTCTTCTGTTAGCAGCCCGCTGAACATGGCATCTTCAGTATGCAGC CCTGTGGGCATCAACTCCATGTCCTCCTCCACGACTAGCTTTGGTAGTTT CCCAGTGCACAGTCCCATCACTCAAGGAACCTCACTGACATGCTCCCCCA GTGTCGAAAACAGAGGCTCAAGGTCACACAGCCCCACACATGCGAGCAAC GTGGGCTCCCCTCTTTCAAGTCCATTGAGCAGCATGAAATCTCCAATCTC CAGCCCTCCAAGTCATTGCAGTGTAAAATCTCCAGTCTCCAGTCCAAATA ACGTCCCTCTGCGCTCCTCTGTGTCCAGCCCGGCAAATCTCAACAACTCA AGGTGCTCCGTTTCCAGCCCTTCCAACAACACCAACAATAGATCCACACT CTCCAGCCCGACAGCCAGCACAGTGGGGTCCATCGGCAGCCCCATCAGCA ATGCCTTCAGCTATGCCACTTCAGGCGCTTCTGCTGGAGCCGGTGCCATC CAGGATGTGGTTCCTAGTCCAGACACCCACGAGAAAGGTGCTCACGACGT TCCTTTCCCTAAGACAGAGGAAGTCGAGAAGGCCATCTCCAATGGTGTGA CTGGCCCGCTCAACATTGTCCAGTACATAAAATCAGAACCAGATGGGGCT TTCAGCAGTTCCTGTCTAGGAGGAAACAGCAAAATCAGCCCCAGTTCTCC ATTCTCTGTACCAATAAAGCAAGAGTCAAGCAAGCACTCATGTTCAGGCG CCTCTTTTAAAGGGAACCCCACGGTCAACCCATTTCCATTCATGGATGGC TCCTACTTTTCCTTTATGGATGATAAGGACTATTATTCCCTATCAGGAAT CTTAGGACCACCTGTGCCCGGCTTTGATGGTAGCTGCGAGGGCAGCGCAT TCCCAGTGGGGATTAAGCAAGAACCAGATGATGGGAGCTATTACCCTGAA GCCAGCATCCCATCATCTGCTATCGTTGGTGTGAATTCGGGTGGACAGTC CTTTCACTACCGGATTGGTGCTCAAGGTACAATATCTTTATCACGGTCAC CTAGAGACCAATCTTTCCAACACTTGAGTTCCTTCCCACCTGTCAATACA TTAGTGGAATCATGGAAACCACACGGTGACCTGTCATCTAGGAGAAGTGA TGGGTATCCCGTCCTAGAGTACATTCCAGAAAACGTGTCAAGCTCTACTT TACGAAGTGTTTCTACTGGATCCTCAAGACCTTCCAAGATCTGTTTGGTG TGTGGAGATGAGGCTTCTGGGTGTCACTATGGGGTAGTGACCTGTGGCAG CTGCAAAGTCTTCTTCAAAAGAGCCGTGGAAGGGCAACACAACTATCTGT GTGCTGGAAGAAATGACTGCATTATTGATAAGATTCGGCGAAAGAACTGT CCTGCCTGCAGGCTCCAGAAATGCCTTCAAGCTGGCATGAACTTAGGAGC TCGAAAGTCAAAGAAGCTGGGGAAGTTAAAAGGCCTCCACGAGGAGCAAC CACAGCAGCCCCCACCACCGCCACCCCAGAGTCCAGAAGAGGGGACCACA TACATCGCTCCGACCAAGGAGCCATCGGTGAACTCTGCGCTGGTCCCGCA GCTCACCTCCATTACGCATGCACTCACACCATCCCCGGCCATGATCCTGG AGAACATCGAGCCTGAGACCGTGTATGCAGGCTACGACAATTCCAAGCCC GACACCGCCGAGAGCCTGCTCTCCACGCTCAACCGCCTGGCAGGCAAACA GATGATCCAGGTCGTGAAGTGGGCCAAGGTACTTCCAGGATTTAAAAACT TGCCTCTCGAGGACCAAATCACCCTCATCCAGTATTCTTGGATGTGTCTA TCATCGTTCGCTTTGAGTTGGAGATCGTACAAACACACGAACAGCCAACT CCTCTATTTTGCTCCAGACCTAGTTTTTAATGAAGAGAAGATGCATCAGT CTGCCATGTATGAACTGTGCCAGGGGATGCGCCAGATCAGCCTTCAATTC GTGCGGCTGCAGCTGACCTTTGAGGAGTACTCCATAATGAAGGTTTTGCT GCTACTAAGCACAGTTCCAAAAGATGGCCTCAAGAGCCAGGCTGCATTTG AGGAGATGAGGACAAATTACATCAAAGAACTGAGGAAAATGGTCACCAAG TGTCCCAACAGTTCTGGACAGAGTTGGCAGAGGTTCTACCAACTGACCAA GCTTCTAGACTCCATGCATGATCTCGTGAGTGACCTGCTAGAATTCTGCT TCTACACCTTCCGAGAATCCCAGGCTCTGAAGGTGGAGTTTCCCGCCATG CTGGTGGAGATCATCACCGACCAGCTGCCAAAGGTGGAGTCTGGGAATGC CAAACCCCTTTACTTTCACAGAAAGTGA
[0045] SEQ ID NO: 12 is the amino acid sequence of traceable fusion protein EGFP-rMR. The sequence includes the following features at the indicated nucleotide positions: EGFP (1-239), Vector polylinker (240-244), and rat MR (245-1225).
TABLE-US-00012 MVSKGEELFTGVVPILVELDGDVNGHKFSVSGEGEGDATYGKLTLKFICT TGKLPVPWPTLVTTLTYGVQCFSRYPDHMKQHDFFKSAMPEGYVQERTIF FKDDGNYKTRAEVKFEGDTLVNRIELKGIDFKEDGNILGHKLEYNYNSHN VYIMADKQKNGIKVNFKIRHNIEDGSVQLADHYQQNTPIGDGPVLLPDNH YLSTQSALSKDPNEKRDHMVLLEFVTAAGITLGMDELYKSGLRSMETKGY HSLPEGLDMERRWSQVSQTLERSSLGPAERTTENNYMEIVNVSCVSGAIP NNSTQGSSKEKHELLPYIQQDNSRSGILPSDIKTELESKELSATVAESMG LYMDSVRDAEYTYDQQNQQGSLSPTKIYQNMEQLVKFYKENGHRSSTLSA MSRPLRSFMPDSAASMNGGALRAIVKSPIICHEKSSSVSSPLNMASSVCS PVGINSMSSSTTSFGSFPVHSPITQGTSLTCSPSVENRGSRSHSPTHASN VGSPLSSPLSSMKSPISSPPSHCSVKSPVSSPNNVPLRSSVSSPANLNNS RCSVSSPSNNTNNRSTLSSPTASTVGSIGSPISNAFSYATSGASAGAGAI QDVVPSPDTHEKGAHDVPFPKTEEVEKAISNGVTGPLNIVQYIKSEPDGA FSSSCLGGNSKISPSSPFSVPIKQESSKHSCSGASFKGNPTVNPFPFMDG SYFSFMDDKDYYSLSGILGPPVPGFDGSCEGSAFPVGIKQEPDDGSYYPE ASIPSSAIVGVNSGGQSFHYRIGAQGTISLSRSPRDQSFQHLSSFPPVNT LVESWKPHGDLSSRRSDGYPVLEYIPENVSSSTLRSVSTGSSRPSKICLV CGDEASGCHYGVVTCGSCKVFFKRAVEGQHNYLCAGRNDCIIDKIRRKNC PACRLQKCLQAGMNLGARKSKKLGKLKGLHEEQPQQPPPPPPQSPEEGTT YIAPTKEPSVNSALVPQLTSITHALTPSPAMILENIEPETVYAGYDNSKP DTAESLLSTLNRLAGKQMIQVVKWAKVLPGFKNLPLEDQITLIQYSWMCL SSFALSWRSYKHTNSQLLYFAPDLVFNEEKMHQSAMYELCQGMRQISLQF VRLQLTFEEYSIMKVLLLLSTVPKDGLKSQAAFEEMRTNYIKELRKMVTK CPNSSGQSWQRFYQLTKLLDSMHDLVSDLLEFCFYTFRESQALKVEFPAM LVEIITDQLPKVESGNAKPLYFHRK*
[0046] SEQ ID NO: 13 is the nucleic acid sequence which encodes traceable fusion protein eGFP-hPRB. The sequence includes the following features at the indicated nucleotide positions: eGFP (1-714), GA Linker (715-744), and hPRB (757-3555).
TABLE-US-00013 ATGAGTAAAGGAGAAGAACTTTTCACTGGAGTTGTCCCAATTCTTGTTGA ATTAGATGGTGATGTTAATGGGCACAAATTTTCTGTCAGTGGAGAGGGTG AAGGTGATGCAACATACGGAAAACTTACCCTTAAATTTATTTGCACTACT GGAAAACTACCTGTTCCTTGGCCAACACTTGTCACTACTTTCACTTATGG TGTTCAATGCTTTTCAAGATACCCAGATCATATGAAACAGCATGACTTTT TCAAGAGTGCCATGCCCGAAGGTTATGTACAGGAAAGAACTATATTTTTC AAAGATGACGGGAACTACAAGACACGTGCTGAAGTCAAGTTTGAAGGTGA TACCCTTGTTAATAGAATCGAGTTAAAAGGTATTGATTTTAAAGAAGATG GAAACATTCTTGGACACAAATTGGAATACAACTATAACTCACACAATGTA TACATCATGGCAGACAAACAAAAGAATGGAATCAAAGTTAACTTCAAAAT TAGACACAACATTGAAGATGGAAGCGTTCAACTAGCAGACCATTATCAAC AAAATACTCCAATTGGCGATGGCCCTGTCCTTTTACCAGACAACCATTAC CTGTCCACACAATCTGCCCTTTCGAAAGATCCCAACGAAAAGAGAGACCA CATGGTCCTTCTTGAGTTTGTAACAGCTGCTGGGATTACACATGGCATGG ATGAACTATACAAAGGCGCCGGCGCTGGTGCTGGTGCTGGCGCCATCAGC GCGTTGACTGAGCTGAAGGCAAAGGGTCCCCGGGCTCCCCACGTGGCGGG CGGCCCGCCCTCCCCCGAGGTCGGATCCCCACTGCTGTGTCGCCCAGCCG CAGGTCCGTTCCCGGGGAGCCAGACCTCGGACACCTTGCCTGAAGTTTCG GCCATACCTATCTCCCTGGACGGGCTACTCTTCCCTCGGCCCTGCCAGGG ACAGGACCCCTCCGACGAAAAGACGCAGGACCAGCAGTCGCTGTCGGACG TGGAGGGCGCATATTCCAGAGCTGAAGCTACAAGGGGTGCTGGAGGCAGC AGTTCTAGTCCCCCAGAAAAGGACAGCGGACTGCTGGACAGTGTCTTGGA CACTCTGTTGGCGCCCTCAGGTCCCGGGCAGAGCCAACCCAGCCCTCCCG CCTGCGAGGTCACCAGCTCTTGGTGCCTGTTTGGCCCCGAACTTCCCGAA GATCCACCGGCTGCCCCCGCCACCCAGCGGGTGTTGTCCCCGCTCATGAG CCGGTCCGGGTGCAAGGTTGGAGACAGCTCCGGGACGGCAGCTGCCCATA AAGTGCTGCCCCGGGGCCTGTCACCAGCCCGGCAGCTGCTGCTCCCGGCC TCTGAGAGCCCTCACTGGTCCGGGGCCCCAGTGAAGCCGTCTCCGCAGGC CGCTGCGGTGGAGGTTGAGGAGGAGGATGGCTCTGAGTCCGAGGAGTCTG CGGGTCCGCTTCTGAAGGGCAAACCTCGGGCTCTGGGTGGCGCGGCGGCT GGAGGAGGAGCCGCGGCTGTCCCGCCGGGGGCGGCAGCAGGAGGCGTCGC CCTGGTCCCCAAGGAAGATTCCCGCTTCTCAGCGCCCAGGGTCGCCCTGG TGGAGCAGGACGCGCCGATGGCGCCCGGGCGCTCCCCGCTGGCCACCACG GTGATGGATTTCATCCACGTGCCTATCCTGCCTCTCAATCACGCCTTATT GGCAGCCCGCACTCGGCAGCTGCTGGAAGACGAAAGTTACGACGGCGGGG CCGGGGCTGCCAGCGCCTTTGCCCCGCCGCGGAGTTCACCCTGTGCCTCG TCCACCCCGGTCGCTGTAGGCGACTTCCCCGACTGCGCGTACCCGCCCGA CGCCGAGCCCAAGGACGACGCGTACCCTCTCTATAGCGACTTCCAGCCGC CCGCTCTAAAGATAAAGGAGGAGGAGGAAGGCGCGGAGGCCTCCGCGCGC TCCCCGCGTTCCTACCTTGTGGCCGGTGCCAACCCCGCAGCCTTCCCGGA TTTCCCGTTGGGGCCACCGCCCCCGCTGCCGCCGCGAGCGACCCCATCCA GACCCGGGGAAGCGGCGGTGACGGCCGCACCCGCCAGTGCCTCAGTCTCG TCTGCGTCCTCCTCGGGGTCGACCCTGGAGTGCATCCTGTACAAAGCGGA GGGCGCGCCGCCCCAGCAGGGCCCGTTCGCGCCGCCGCCCTGCAAGGCGC CGGGCGCGAGCGGCTGCCTGCTCCCGCGGGACGGCCTGCCCTCCACCTCC GCCTCTGCCGCCGCCGCCGGGGCGGCCCCCGCGCTCTACCCTGCACTCGG CCTCAACGGGCTCCCGCAGCTCGGCTACCAGGCCGCCGTGCTCAAGGAGG GCCTGCCGCAGGTCTACCCGCCCTATCTCAACTACCTGAGGCCGGATTCA GAAGCCAGCCAGAGCCCACAATACAGCTTCGAGTCATTACCTCAGAAGAT TTGTTTAATCTGTGGGGATGAAGCATCAGGCTGTCATTATGGTGTCCTTA CCTGTGGGAGCTGTAAGGTCTTCTTTAAGAGGGCAATGGAAGGGCAGCAC AACTACTTATGTGCTGGAAGAAATGACTGCATCGTTGATAAAATCCGCAG AAAAAACTGCCCAGCATGTCGCCTTAGAAAGTGCTGTCAGGCTGGCATGG TCCTTGGAGGTCGAAAATTTAAAAAGTTCAATAAAGTCAGAGTTGTGAGA GCACTGGATGCTGTTGCTCTCCCACAGCCAGTGGGCGTTCCAAATGAAAG CCAAGCCCTAAGCCAGAGATTCACTTTTTCACCAGGTCAAGACATACAGT TGATTCCACCACTGATCAACCTGTTAATGAGCATTGAACCAGATGTGATC TATGCAGGACATGACAACACAAAACCTGACACCTCCAGTTCTTTGCTGAC AAGTCTTAATCAACTAGGCGAGAGGCAACTTCTTTCAGTAGTCAAGTGGT CTAAATCATTGCCAGGTTTTCGAAACTTACATATTGATGACCAGATAACT CTCATTCAGTATTCTTGGATGAGCTTAATGGTGTTTGGTCTAGGATGGAG ATCCTACAAACACGTCAGTGGGCAGATGCTGTATTTTGCACCTGATCTAA TACTAAATGAACAGCGGATGAAAGAATCATCATTCTATTCATTATGCCTT ACCATGTGGCAGATCCCACAGGAGTTTGTCAAGCTTCAAGTTAGCCAAGA AGAGTTCCTCTGTATGAAAGTATTGTTACTTCTTAATACAATTCCTTTGG AAGGGCTACGAAGTCAAACCCAGTTTGAGGAGATGAGGTCAAGCTACATT AGAGAGCTCATCAAGGCAATTGGTTTGAGGCAAAAAGGAGTTGTGTCGAG CTCACAGCGTTTCTATCAACTTACAAAACTTCTTGATAACTTGCATGATC TTGTCAAACAACTTCATCTGTACTGCTTGAATACATTTATCCAGTCCCGG GCACTGAGTGTTGAATTTCCAGAAATGATGTCTGAAGTTATTGCTGCACA ATTACCCAAGATATTGGCAGGGATGGTGAAACCCCTTCTCTTTCATAAAA AGTGA
[0047] SEQ ID NO: 14 is the amino acid sequence of traceable fusion protein eGFP-hPRB. The sequence includes the following features at the indicated and nucleotide positions: EGFP (1-238), GA Linker (239-248), and hPRB (253-1184).
TABLE-US-00014 MSKGEELFTGVVPILVELDGDVNGHKFSVSGEGEGDATYGKLTLKFICTT GKLPVPWPTLVTTFTYGVQCFSRYPDHMKQHDFFKSAMPEGYVQERTIFF KDDGNYKTRAEVKFEGDTLVNRIELKGIDFKEDGNILGHKLEYNYNSHNV YIMADKQKNGIKVNFKIRHNIEDGSVQLADHYQQNTPIGDGPVLLPDNHY LSTQSALSKDPNEKRDHMVLLEFVTAAGITHGMDELYKGAGAGAGAGAIS ALTELKAKGPRAPHVAGGPPSPEVGSPLLCRPAAGPFPGSQTSDTLPEVS AIPISLDGLLFPRPCQGQDPSDEKTQDQQSLSDVEGAYSRAEATRGAGGS SSSPPEKDSGLLDSVLDTLLAPSGPGQSQPSPPACEVTSSWCLFGPELPE DPPAAPATQRVLSPLMSRSGCKVGDSSGTAAAHKVLPRGLSPARQLLLPA SESPHWSGAPVKPSPQAAAVEVEEEDGSESEESAGPLLKGKPRALGGAAA GGGAAAVPPGAAAGGVALVPKEDSRFSAPRVALVEQDAPMAPGRSPLATT VMDFIHVPILPLNHALLAARTRQLLEDESYDGGAGAASAFAPPRSSPCAS STPVAVGDFPDCAYPPDAEPKDDAYPLYSDFQPPALKIKEEEEGAEASAR SPRSYLVAGANPAAFPDFPLGPPPPLPPRATPSRPGEAAVTAAPASASVS SASSSGSTLECILYKAEGAPPQQGPFAPPPCKAPGASGCLLPRDGLPSTS ASAAAAGAAPALYPALGLNGLPQLGYQAAVLKEGLPQVYPPYLNYLRPDS EASQSPQYSFESLPQKICLICGDEASGCHYGVLTCGSCKVFFKRAMEGQH NYLCAGRNDCIVDKIRRKNCPACRLRKCCQAGMVLGGRKFKKFNKVRVVR ALDAVALPQPVGVPNESQALSQRFTFSPGQDIQLIPPLINLLMSIEPDVI YAGHDNTKPDTSSSLLTSLNQLGERQLLSVVKWSKSLPGFRNLHIDDQIT LIQYSWMSLMVFGLGWRSYKHVSGQMLYFAPDLILNEQRMKESSFYSLCL TMWQIPQEFVKLQVSQEEFLCMKVLLLLNTIPLEGLRSQTQFEEMRSSYI RELIKAIGLRQKGVVSSSQRFYQLTKLLDNLHDLVKQLHLYCLNTFIQSR ALSVEFPEMMSEVIAAQLPKILAGMVKPLLFHKK*
[0048] SEQ ID NOs: 15-28 are primer sequences used for Q-PCR analysis, as follows:
TABLE-US-00015 Mouse cells SEQ ID (3134) Sequence NO: Per1 For CTTCTGGCAATGGCAAGGACTC 15 Per1 Rev CAGCATCATGCCATCATACACACA 16 Tgm2 For TGTCACCAGGGATGAGAGACGG 17 Tgm2 Rev TCCAAATCACACCTCTCCAGGAG 18 Lcn2 For ACCTCTCATTTCTTGCAGTTCCG 19 Lcn2 Rev CAGGATGGAGGTGACATTGTAGCT 20 β-Actin For AGTGTGACGTTGACATCCGTA 21 β-Actin Rev GCCAGAGCAGTAATCTCCTTCT 22 Human cells SEQ ID (LNCaP) Sequence NO: hNKX3.1 For TGACAGTGGGCTGTTTGTTC 23 hNKX3.1 Rev AAGACCCCAAGTGCCTTTCT 24 hRHOU For TTTCAAGGATGCTGGCTCTT 25 hRHOU Rev GGCCTCAGCTTGTCAAATTC 26 GAPDH For AAGGTGAAGGTCGGAGTCAAC 27 GAPDH Rev GGGGTCATTGATGGCAACAATA 28
DETAILED DESCRIPTION
I. Abbreviations
[0049] AhR aryl hydrocarbon receptor AR androgen receptor ATRA all-trans retinoic acid CAR constitutive androstane receptor cst corticosterone dex dexamethasone ER estrogen receptor protein (α, β) ERR estrogen-related receptor (α, β, γ) FP fluorescent protein FXR farnesoid X receptor GCNF germ cell nuclear factor GFP green fluorescent protein GFP-AhR chimera w/aryl hydrocarbon receptor and GFP GFP-AR chimera w/androgen receptor and GFP GFP-GR chimera w/GR (or GR*) and GFP (also, GR-GFP) GFP-GR-ER chimera w/ GR translocation domain, estrogen receptor, and GFP GFP-GR-RAR chimera w/GR translocation domain, retinoic acid receptor, and GFP GFP-GR-TR chimera w/GR translocation domain, thyroid hormone receptor, and GFP GFP-PR-B chimera w/GR translocation domain, progesterone B receptor, and GFP GR glucocorticoid receptor GR* modified GR with increased ligand binding affinity HNF4 hepatocyte nuclear factor-4 (α, γ) Hsp heat shock protein LBD ligand binding domain (of a receptor protein) LCA lithocholic acid LRH-1 liver receptor homolog-1 LTR long terminal repeat LXR liver X receptor (α, β) MR mineralocorticoid receptor MMTV mouse mammary tumor virus NF1 nuclear factor 1 NHRs nuclear hormone receptors
NRNC Nuclear Receptors Nomenclature Committee
[0050] OTF1 octomer transcription factor 1 POCIS polar organic chemical integrative samplers PPAR peroxisome proliferator-activated receptor (α, β/δ, γ) PR progesterone receptor PXR pregnane X receptor RAR retinoic acid receptor ROR RAR-related orphan receptor (α, β, γ)
RRE Rev Responsive Element
[0051] RXR retinoid-X receptor (α, β, γ) SF1 steroidogenic factor 1 SHP small heterodimer partner SRC1 steroid receptor coactivator 1 TR thyroid hormone receptor (α, β) T3 triiodothyronine T4 thyroxine UDCA ursodeoxycholic acid VDR vitamin D receptor WWTP waste water treatment plant
II. Terms
[0052] Unless otherwise noted, technical terms are used according to conventional usage. In order to facilitate review of the various embodiments of the invention, the following explanations of specific terms are provided:
[0053] Cell-based System: A system based employing live cells derived, isolated, or otherwise acquired from a living organism. This term includes, for example, a cell culture.
[0054] Chimera/Chimeric: A recombinant nucleic acid molecule generated by cloning portion(s) of one or more nucleic acid sequence(s) in-frame to one or more other nucleic acid sequence(s) to produce a single nucleic acid sequence capable of being transcribed into a polypeptide. A polypeptide expressed from such a nucleic acid sequence chimera is referred to as a "chimeric protein" or "protein chimera".
[0055] Contacting: To place in direct physical association, including in solid or in liquid form. Contacting can occur in vivo, for example by administering an agent to a subject, or in vitro for example with isolated cells or cell-cultures.
[0056] Control: Samples believed to be normal (e.g., representative of an activity or function in the absence of the variable being tested), as well as laboratory values, even though possibly arbitrarily set, keeping in mind that such values can vary from laboratory to laboratory. A control sample or group is practically identical to the test sample or group, except for the variable(s) of interest the effect of which is being tested, which is only applied to or found in the test group. A difference between a test sample and a control can be an increase or a decrease. The difference can be a qualitative difference or a quantitative difference, for example a statistically significant difference. In some examples, a difference is an increase or decrease, relative to a control, of at least about 10%, such as at least about 20%, at least about 30%, at least about 40%, at least about 50%, at least about 60%, at least about 70%, at least about 80%, at least about 90%, at least about 100%, at least about 150%, at least about 200%, at least about 250%, at least about 300%, at least about 350%, at least about 400%, at least about 500%, or greater than 500%.
[0057] Detect: To determine if an agent (such as a bacterium) is present or absent. In some examples this can further include quantification. For example, use of the disclosed methods permits detection of one or more target bacterium, such as by flow cytometry or fluorescence microscopy. Detection can be in bulk, so that a macroscopic number of molecules can be observed contemporaneously or simultaneously. Detection can also include detection of single events, such as a single bacterium.
[0058] Emission or emission signal: The light of a particular wavelength generated from a source. In particular examples, an emission signal is emitted from a fluorophore, such as a fluorescent protein, after the fluorophore absorbs light at its excitation wavelength(s).
[0059] Environmental sample: A sample obtained from the environment, for instance a water sample (e.g., of surface, sub-surface (ground), rain, run-off, well, spring, drinking, river, estuary, ocean, effluent, treated or untreated sewage, etc. water), soil sample (including soil samples that contain water), air sample, or a sample of another substance.
[0060] Excitation or excitation signal: The light of a particular wavelength necessary and/or sufficient to excite an electron transition to a higher energy level. In particular examples, an excitation is the light of a particular wavelength necessary and/or sufficient to excite a fluorophore, such as a fluorescent protein, to a state such that the fluorophore will emit a different (such as a longer) wavelength of light then the wavelength of light from the excitation signal.
[0061] Fluorescent property: A characteristic of a fluorescent molecule, such as a fluorescent protein, for example green fluorescent protein, red fluorescent protein, yellow fluorescent protein, cyan fluorescent protein and the like. Examples of fluorescent properties include the molar extinction coefficient at an appropriate excitation wavelength, the fluorescence quantum efficiency, the shape of the excitation spectrum or emission spectrum (the "fluorescence spectrum," the excitation wavelength maximum and emission wavelength maximum, the ratio of excitation amplitudes at two different wavelengths, the ratio of emission amplitudes at two different wavelengths, the excited state lifetime, or the fluorescence anisotropy. Quantifying fluorescence refers to the determination of the amount of fluorescence generated by a fluorophore, for example a fluorescent protein, which can be the quantity of photons emitted by a fluorophore. In some examples, fluorescence is quantified by measuring the intensity of a fluorescence signal at a particular wavelength, for example the wavelength of the emission maxima of a particular fluorophore, such as a fluorescent protein. Fluorescence intensity can also be quantified at a wavelength that is not the emission maxima of a particular fluorophore, for example to avoid emission spectra that overlap and thereby interfere with the emission maxima of a particular fluorophore, such as a particular fluorescent protein. In some examples, a fluorescence signal is emitted by a population of fluorescent proteins, for example fluorescent proteins present in a population of cells containing such fluorescent proteins. Such a signal can be quantified, for example to determine the number, or relative number of cells that emit such a fluorescent signal. Detecting a pattern of fluorescence refers to the correlation of a fluorescent signal to a specific location to determine the location where a fluorescence signal, such as a fluorescent signal of a particular wavelength, originates. In some examples, a pattern of fluorescence determines the location and or shape of the cells that emit a fluorescence signal, such as cells containing a fluorescent protein, including for example to sub-cellular localization of the fluorescent protein (e.g., cytoplasmic or nuclear) as well as the absolute or relative quantitative distribution between sub-cellular compartments and organelles.
[0062] Fluorescent protein: A protein capable of emission of a detectable (and therefore traceable) fluorescent signal. Fluorescent proteins can be characterized by the wavelength of their emission spectrum. For example, wildtype green fluorescent protein (GFP) has a fluorescent emission spectrum in the green part of the visible spectrum. In addition to green-fluorescent proteins, fluorescent proteins are well known that fluoresce in other regions of the visible spectrum, for example blue-fluorescent proteins, cyan-fluorescent proteins. yellow-fluorescent proteins, orange-fluorescent proteins. red-fluorescent proteins, and far-red fluorescent proteins. Non-limiting examples of fluorescent proteins can be found in the following patent documents: U.S. Pat. Nos. 5,804,387; 6,090,919; 6,096,865; 6,054,321; 5,625,048; 5,874,304; 5,777,079; 5,968,750; 6,020,192; 6,146,826; 6,969,597; 7,150,979; 7,157,565; and 7,166,444; and published international patent applications WO 07/085923; WO 07/052102, WO 04/058973, WO 04/044203, WO 03/062270; and WO 99/64592. Additional examples of fluorescent proteins are available from Clontech, Laboratories, Inc. (Mountain View, Calif.) under the trade name Living Colors®. Nucleic acids encoding such fluorescent proteins can be incorporated into mammalian expression vectors for use in producing the disclosed fluorescent traceable proteins and fluorescent cell-lines.
[0063] Fusion protein: Proteins that have at least two domains fused together, at least one domain comprising traceable (marker) feature (such as a fluorescent protein domain) and another domain or set of domains that provides translocation of the fusion protein from one cellular compartment or organelle to another in a cell in which it is expressed upon the specific binding of a ligand (EDC) to the fusion protein. Optionally the translocation and ligand binding features may be provided from the same source protein (e.g., in a GFP-GR traceable translocation fusion protein). In other embodiments, the translocation and ligand binding features are provided from two different source proteins (thus making a "chimeric receptor" portion of the fusion protein), such that the resultant traceable translocating fusion protein contains domains or subdomains from at least three different source proteins. An example of such a three-component fusion protein is the GFP-GR-ER protein which contains GFP, translocating features of or derived from GR, and the ligand binding domain of an estrogen receptor.
[0064] In general, the domains of the disclosed fusions are genetically fused together, in that nucleic acid molecules that encode each protein domain (or subdomain) are functionally linked together, for instance directly or through the use of a linker oligonucleotide, thereby producing a single fusion-encoding (chimeric) nucleic acid molecule. The translated product of such a fusion-encoding (chimeric) nucleic acid molecule is the traceable translocating fusion protein.
[0065] High throughput technique: A fast, automated or semi-automated analysis process, for instance to analyze many samples at once in order to detect or measure the presence (or absence) of a substance. In certain examples, combining modern robotics, data processing and control software, liquid handling devices, and sensitive detectors, high throughput techniques allows the rapid screening of many (e.g., hundreds or thousands) samples in a short period of time and often in a highly parallel manner.
[0066] Traceable marker protein domain: A protein domain that is detectable based on its inherent structural or functional characteristics, such as fluorescence.
[0067] Nuclear hormone receptor family: Nuclear hormone receptors (NHRs) function as ligand-activated transcription factors and have roles in diverse cellular processes ranging from mammalian development and differentiation to metabolic homeostasis (Mangelsdorf et al., Cell, 83(6):835-839, 1995; Adams et al., Science, 287:2185-2195, 2000). NHRs bind to sequence-specific DNA response elements on target gene promoters as homodimers, heterodimers, or monomers. Structural and functional analyses of the NHR family have demonstrated that the receptors are comprised of functional modular domains. The DNA binding domain (DBD) consists of a well characterized zinc finger motif which recognizes a degenerate six to eight nucleotide sequence on the target DNA. The ligand binding domain (LBD) resides in the C-terminal portion of the protein and shares a common, predominantly alpha helical fold (Mangelsdorf et al., Cell, 83(6):835-839, 1995). As implied, this domain of the receptor is where cognate ligands of the receptors interact and induce conformational changes associated with transcriptional activation. Many of the known ligands for these receptors are essential metabolic products including retinoids, thyroid hormone, vitamin D3, bile acids, oxysterols, and prostenoids that act through their cognate receptors to control metabolic homeostasis in the body (Gudas, J. Biol. Chem., 269(22):15399-15402, 1994). In addition, NHRs are also instrumental in the ability of the body to respond to and adapt to complex environmental cues.
[0068] Nucleic acid: A polymer composed of nucleotide units (ribonucleotides, deoxyribonucleotides, related naturally occurring structural variants, and synthetic non-naturally occurring analogs thereof) linked via phosphodiester bonds, related naturally occurring structural variants, and synthetic non-naturally occurring analogs thereof. Non-naturally occurring synthetic analogs include, for example and without limitation, phosphorothioates, phosphoramidates, methyl phosphonates, chiral-methyl phosphonates, 2-O-methyl ribonucleotides, peptide-nucleic acids (PNAs), and the like. Such polynucleotides can be synthesized, for example, using an automated DNA synthesizer. The term "oligonucleotide" typically refers to short polynucleotides, generally no greater than about 50 nucleotides. It will be understood that when a nucleotide sequence is represented by a DNA sequence (i.e., A, T, G, C), this also includes an RNA sequence (i.e., A, U, G, C) in which "U" replaces "T."
[0069] Conventional notation is used herein to describe nucleotide sequences: the left-hand end of a single-stranded nucleotide sequence is the 5'-end; the left-hand direction of a double-stranded nucleotide sequence is referred to as the 5'-direction. The direction of 5' to 3' addition of nucleotides to nascent RNA transcripts is referred to as the transcription direction. The DNA strand having the same sequence as an mRNA is referred to as the "coding strand;" sequences on the DNA strand having the same sequence as an mRNA transcribed from that DNA and which are located 5' to the 5'-end of the RNA transcript are referred to as "upstream sequences;" sequences on the DNA strand having the same sequence as the RNA and which are 3' to the 3' end of the coding RNA transcript are referred to as "downstream sequences."
[0070] "cDNA" refers to a DNA that is complementary or identical to an mRNA, in either single stranded or double stranded form.
[0071] "Encoding" refers to the inherent property of specific sequences of nucleotides in a polynucleotide, such as a gene, a cDNA, or an mRNA, to serve as templates for synthesis of other polymers and macromolecules in biological processes having either a defined sequence of nucleotides (i.e., rRNA, tRNA and mRNA) or a defined sequence of amino acids and the biological properties resulting therefrom. Thus, a gene encodes a protein if transcription and translation of mRNA produced by that gene produces the protein in a cell or other biological system. Both the coding strand (the nucleotide sequence of which is identical to the mRNA sequence and is usually provided in sequence listings) and non-coding strand (used as the template for transcription) of a gene or cDNA can be referred to as encoding the protein or other product of that gene or cDNA. Unless otherwise specified, a "nucleotide sequence encoding an amino acid sequence" includes all nucleotide sequences that are degenerate versions of each other and that encode the same amino acid sequence. Nucleotide sequences that encode proteins and RNA may include introns.
[0072] A first sequence is an "antisense" with respect to a second sequence if a polynucleotide whose sequence is the first sequence specifically hybridizes with a polynucleotide whose sequence is the second sequence.
[0073] Terms used to describe sequence relationships between two or more nucleotide sequences or amino acid sequences include "reference sequence," "selected from," "comparison window," "identical," "percentage of sequence identity," "substantially identical," "complementary," and "substantially complementary."
[0074] Operably linked: A first nucleic acid sequence is operably linked with a second nucleic acid sequence when the first nucleic acid sequence is placed in a functional relationship with the second nucleic acid sequence. For instance, a promoter is operably linked to a coding sequence (for example a coding sequence of an antibody or fragment therefore herein disclosed) if the promoter affects the transcription or expression of the coding sequence. Generally, operably linked DNA sequences are contiguous and, where necessary to join two protein-coding regions, in the same reading frame.
[0075] ORF (open reading frame): A series of nucleotide triplets (codons) coding for amino acids without any termination codons. These sequences are usually translatable into a peptide. In some examples an open reading frame encodes an antibody or antibody fragment, such as those disclosed herein.
[0076] Polypeptide: A polymer in which the monomers are amino acid residues that are joined together through amide bonds, for example γ amide bonds (for example from the γ position of a glutamic acid side chain) or α amide bonds. When the amino acids are alpha-amino acids, either the L-optical isomer or the D-optical isomer can be used. The terms "polypeptide" or "protein" as used herein is intended to encompass any amino acid sequence and include modified sequences such as glycoproteins. The term "polypeptide" is specifically intended to cover naturally occurring proteins, as well as those that are recombinantly or synthetically produced.
[0077] The term "polypeptide fragment" refers to a portion of a polypeptide which exhibits at least one useful epitope. The term "functional fragments of a polypeptide" refers to all fragments of a polypeptide that retain an activity of the polypeptide. Biologically functional fragments, for example, can vary in size from a polypeptide fragment as small as an epitope capable of binding an antibody molecule to a large polypeptide capable of participating in the characteristic induction or programming of phenotypic changes within a cell.
[0078] Sample: A material to be analyzed, for example to determine if it contains one or more target molecule such as an EDC. The term includes but is not limited to biological samples (e.g., obtained from a human or veterinary subject); food samples (e.g., vegetable, dairy, fruit or meat sample); environmental samples (e.g., soil, air, water, surfaces); industrial and construction samples; and the like. Specifically contemplated are samples of material known to or that potentially may leach (e.g., into the environment or surroundings) one or more compounds (molecules) with at least one potentially endocrine disrupting property, for instance upon degradation or aging of the material upon exposure to actual or simulated environmental or climactic conditions.
[0079] Sewage: Water-based fluid containing organic matter and solutes. Sewage may include feces and urine from human and non-human animals. Sewage may include waste from human activities, for example, blackwater (e.g., toilet and dishwasher waste) and grey water (e.g. waste water generated from washing activities). Residential, institutional, commercial and industrial establishments may produce sewage, including waste from toilets, baths, showers, kitchens, sinks, etc. Typically, sewage is waste intended to be carried away from the source of the waste, for example, carried to a sewage treatment facility.
[0080] Receptor Superfamily: Family of steroid, nuclear, and orphan receptor proteins having an identifiable ligand binding domain. The term as used herein is intended to encompass the known classic nuclear receptors, hormone receptors, and orphan receptors, as well as proteins having an identifiable ligand binding domain which may be discovered in the future.
TABLE-US-00016 TABLE 1 Representative Nuclear Receptors Subfamily Accession and Group NR/Gene Literature Names Number 1A NR1A1 TRα, c-erbA-1, THRA M24748 NR1A2 TRβ, c-erbA-2, THRB X04707 1B NR1B1 RARα X06538 NR1B2 RARβ, HAP Y00291 NR1B3 RARγ, RARD M57707 1C NR1C1 PPARα L02932 NR1C2 PPARβ, NUC1, PPARδ, FAAR L07592 NR1C3 PPARγ L40904 1D NR1D1 REVERBα, EAR1, EAR1A M24898 NR1D2 REVERBβ, EAR1β, BD73, RVR, L31785 HZF2 NR1D3 E75 X51548 1E NR1E1 E78, DR-78 U01087 1F NR1F1 RORα, RZRα U04897 NR1F2 RORβ, RZRβ Y08639 NR1F3 RORγ, TOR U16997 NR1F4 HR3, DHR3, MHR3, GHR3, CNR3, M90806 CHR3 U13075 1G NR1G1 CNR14 U13074 1H NR1H1 ECR M74078 NR1H2 UR, OR-1, NER1, RIP15, LXRβ U07132 NR1H3 RLD1, LXR, LXRα U22662 NR1H4 FXR, RIP14, HRR1 U09416 1I NR1I1 VDR J03258 NR1I2 ONR1, PXR, SXR, BXR X75163 NR1I3 MB67, CAR1, CARα Z30425 NR1I4 CAR2, CARβ AF00932 1J NR1J1 DHR96 U36792 1K NR1K1 NHR1 U19360 2A NR2A1 HNF4 X76930 NR2A2 HNF4G Z49826 NR2A3 HNF4B Z49827 NR2A4 DHNF4, HNF4D U70874 2B NR2B1 RXRA X52773 NR2B2 RXRB, H-2RIIBP, RCoR-1 M84820 NR2B3 RXRG X66225 NR2B4 USP, Ultraspiracle, 2C1, CF1 X52591 2C NR2C1 TR2, TR2-11 M29960 NR2C2 TR4, TAK1 L27586 2D NR2D1 DHR78 U36791 2E NR2E1 TLL, TLX, XTLL S72373 NR2E2 TLL, Tailless M34639 2F NR2F1 COUP-TFI, COUPTFA, EAR3, X12795 SVP44 NR2F2 COUP-TFII, COUPTFB, ARP1, M64497 SVP40 NR2F3 SVP, COUP-TF M28863 NR2F4 COUP-TFIII, COUPTFG X63092 NR2F5 SVP46 X70300 NR2F6 EAR2 X12794 3A NR3A1 ERα X03635 NR3A2 ERβ U57439 3B NR3B1 ERR1, ERRα X51416 NR3B2 ERR2, ERRβ X51417 3C NR3C1 GR X03225 NR3C2 MR M16801 NR3C3 PR M15716 NR3C4 AR M20132 4A NR4A1 NGFIB, TR3, N10, NUR77, NAK1 L13740 NR4A2 NURR1, NOT, RNR1, HZF-3, TINOR X75918 NR4A3 NOR1, MINOR D38530 NR4A4 DHR38, NGFIB U36762 CNR8, C48D5 U13076 5A NR5A1 SF1, ELP, FTZ-F1, AD4BP D88155 NR5A2 LRH1, xFF1rA, xFF1rB, FFLR, U93553 PHR, FTF NR5A3 FTZ-F1 M63711 5B NR5B1 DHR39, FTZF1B L06423 6A NR6A1 GCNF1, RTR U14666 0A NR0A1 KNI, Knirps X13331 NR0A2 KNRL, Knirps related X14153 NR0A3 EGON, Embryonic gonad, EAGLE X16631 NR0A4 ODR7 U16708 NR0A5 Trithorax M31617 0B NR0B1 DAX1, AHCH S74720 NR0B2 SHP L76571 # Derived from A unified nomenclature system for the nuclear receptor superfamily (Nuclear Receptors Nomenclature Committee, Cell 97(2): 161-163, 1999).
[0081] Unless otherwise explained, all technical and scientific terms used herein have the same meaning as commonly understood by one of ordinary skill in the art to which this invention belongs. The word "or" is intended to include "and" unless the context clearly indicates otherwise. Hence "comprising A or B" means including A, or B, or A and B. It is further to be understood that all base sizes or amino acid sizes, and all molecular weight or molecular mass values, given for nucleic acids or polypeptides are approximate, and are provided for description. Although methods and materials similar or equivalent to those described herein can be used in the practice or testing of the present invention, suitable methods and materials are described below. All publications, patent applications, patents, sequences defined by accession number, and other references mentioned herein are incorporated by reference in their entirety. In case of conflict, the present specification, including explanations of terms, will control. In addition, the materials, methods, and examples are illustrative only and not intended to be limiting.
III. Introduction and Overview of Several Embodiments
[0082] There is a growing interest in the possible health risks posed by endocrine-disrupting chemicals (EDCs) in the environment, water, food, and consumer products. Steroidal EDCs interfere with the normal function of the endocrine system and have been associated with developmental defects, metabolic disorders and cancer. Previous systems for the detection and monitoring of water sources for steroidal contamination relied on laborious analysis of their chemical structures.
[0083] Steroid hormones, such as estrogens, have been detected in water sources, and their deleterious effects are well documented. Considering that many natural steroids are rapidly metabolized, their derivatives are frequently not present in the currently existing libraries and thus cannot be identified. In addition, it is unclear whether EDCs detected by chemical methods can elicit specific biological responses in mammalian systems.
[0084] Described herein is are high-throughput assays (including automated assays) for biological testing of EDCs using mammalian cells that express GFP-tagged nuclear steroid receptor constructs. See also Stavreva et al. (Sci Rep. 2012;2:937. doi: 10.1038/srep00937. Epub Dec. 6, 2012). These assays re based on translocation of fluorescent marker(s) from the cytoplasm to the nucleus in the presence of a ligand that interacts with a specific receptor. Using this assay and studies of transcriptional activation, as described herein, water samples collected from 14 states in the U.S. have been screened; androgen activity was found in 35% of samples, and a previously unrecognized glucocorticoid (GC) activity in 27% of the samples. Androst-4-en-3,6-dione was identified in one of the samples. Androgen receptor (AR)-dependent nuclear translocation and transcriptional activation was confirmed for two AR-responsive genes, NKX3.1 and RHOU. NKX3.1 is a homeobox gene frequently deleted in prostate cancers, and RHOU is implicated in epidermal growth factor receptor signaling and cell migration. Glucocorticoid receptor (GR)-dependent transcriptional activation was detected using several targets. Induction of a circadian rhythm gene, Perl, was confirmed at concentrations equal to those present in one water sample. This water site was positive in a sample obtained by extraction of a filter (POCIS membranes), as well as a grab water sample obtained several years later. Widespread contamination with EDCs (including these two classes of steroid EDCs) is a possible health hazard not only for the aquatic ecosystems, but also humans. Testing for biological activity of glucocorticoids and many other steroid EDCs has not been previously performed. The herein described automated, highly reproducible, and low cost assays detect biologically active steroidal EDCs and is suitable for wide application in testing water samples, for instance for the analysis and monitoring of water contamination with steroidal EDCs.
[0085] Provided herein in a first set of embodiments are systems for detecting or quantifying a ligand (e.g., an agonist or an antagonist) of a superfamily receptor protein in an environmental sample. In examples thereof, the system comprises a first mammalian cell expressing a first traceable fusion protein; a second mammalian cell expressing a second traceable fusion protein; and a detection system for the detection of the cytoplasm-to-nuclear translocation of the marker proteins, wherein the first and second traceable fusion proteins independently comprise either: (1) a superfamily receptor protein, and a marker protein domain; or (2) the cytoplasmic/nuclear translocation domain of glucocorticoid receptor, the ligand binding domain of a superfamily receptor protein, and a marker protein domain. Optionally, the mammalian cells are human cells.
[0086] In examples of the described system for detecting or quantifying a ligand of a superfamily receptor protein in an environmental sample, the first traceable fusion protein binds a different ligand than the second traceable fusion protein.
[0087] In examples of the described system for detecting or quantifying a ligand of a superfamily receptor protein in an environmental sample, the marker protein domain is different in the first and second traceable fusion proteins. This enables differential detection of the two fusion proteins, for instance in the same analysis system or sample.
[0088] In any of the described system for detecting or quantifying a ligand of a superfamily receptor protein in an environmental sample, the marker protein domain may optionally be a fluorescent protein domain.
[0089] Without limitation, the systems provided herein may employ any cellular receptor including the following (generically termed) traceable fusion proteins: GFP-GR, GFP-AR, GFP-AhR, GFP-PR-B, GFP-GR-ER, GFP-GR-RAR, GFP-MR, and GFP-GR-TR, as well as fusion(s) in which the GFP is replaced with a different fluorescent domain that emits light at a different wavelength. By way of specific example, contemplated herein for use (singly or in combinations of two or more) in the described systems are the following traceable fusion proteins: eGFP-GR-ER310 (SEQ ID NOs: 1 & 2); eGFP-GR-TR216 (SEQ ID NOs: 3 & 4); pCI-nGFP-C656G (SEQ ID NOs: 5 & 6); eGFP-hAR (SEQ ID NOs: 7 & 8); pRefTRE-GFP-mAhR (SEQ ID NOs: 9 & 10); eGFP-ratMP (SEQ ID NO: 11 & 12); and/or eGFP-hPRB (SEQ ID NOs: 13 & 14).
[0090] Also described are systems for detecting or quantifying a ligand of a superfamily receptor protein in an environmental sample, wherein the ligand for the first or second traceable fusion protein is a natural ligand for the superfamily receptor protein of the traceable fusion protein, or a natural or synthetic compound that binds competitively therewith.
[0091] Also provided are systems for detecting or quantifying a ligand of a superfamily receptor protein in an environmental sample that additionally comprise one or more compounds and/or compositions that stably associate with a traceable fusion protein in the absence of a ligand for the ligand binding domain of the traceable fusion protein, and which dissociates from the fusion protein in the presence of a ligand for the ligand binding domain of the traceable fusion protein.
[0092] Yet another embodiment provides a system for detecting or quantifying a ligand of a superfamily receptor protein in an environmental sample, which system includes at least four mammalian cell lines each expressing a different traceable fusion protein, wherein at least one traceable fusion protein binds a glucocorticoid, at least one traceable fusion protein binds an androgen, at least one traceable fusion protein binds a progestin, and at least one traceable fusion protein binds an aryl hydrocarbon.
[0093] The systems described herein are optionally provided as kits.
[0094] Also described are methods for detecting or quantifying an endocrine disruptor chemical (EDC) ligand in an environmental sample, which methods involve contacting with the environmental sample a mammalian, optionally human, cell expressing in its cytoplasm a fusion protein comprising: a receptor for the ligand, which receptor translocates from the cytoplasm to the nucleus upon ligand binding; and a marker protein domain; and then detecting cytoplasmic to nuclear translocation of the fusion protein in response to a ligand of the ligand binding domain in the water sample.
[0095] Also described are methods of determining the concentration of an endocrine disruptor chemical (EDC) ligand in an environmental sample, which methods involve contacting with the environmental sample a population of eukaryotic cells expressing in their cytoplasm a fusion protein comprising: a receptor for the ligand, which receptor translocates from the cytoplasm to the nucleus upon ligand binding; and a marker protein domain; and scanning one or more test cell(s) to obtain signal data from the marker of the fusion protein; converting the signal data to obtain the cellular location of the labeled protein in the test cell(s); and analyzing the signal data using an analysis system having an algorithm to calculate changes in distribution of the labeled fusion protein between the cytoplasm and the nucleus of the test cell(s), the analysis system having the capability of providing an accurate reading of the concentration of the ligand.
[0096] Without limitation, the methods provided herein may employ any cellular receptor including the following (generically termed) traceable fusion proteins: GFP-GR, GFP-AR, GFP-AhR, GFP-PR-B, GFP-GR-ER, GFP-GR-RAR, GFP-MR, and GFP-GR-TR, as well as fusion(s) in which the GFP is replaced with a different fluorescent domain that emits light at a different wavelength. By way of specific example, contemplated herein for use (singly or in combinations of two or more) in the described methods are the following traceable fusion proteins: eGFP-GR-ER310 (SEQ ID NOs: 1 & 2); eGFP-GR-TR216 (SEQ ID NOs: 3 & 4);
[0097] pCI-nGFP-C656G (SEQ ID NOs: 5 & 6); eGFP-hAR (SEQ ID NOs: 7 & 8); pRefTRE-GFP-mAhR (SEQ ID NOs: 9 & 10); eGFP-ratMP (SEQ ID NO: 11 & 12); and/or eGFP-hPRB (SEQ ID NOs: 13 & 14).
[0098] In any of the provided systems, kits, and methods, the environmental sample can include a water sample, soil sample, or air sample. By way of example, where the environmental sample comprises a water sample, the sample includes in various embodiments one or more of surface water, sub-surface (ground) water, rain, run-off, well water, spring water, drinking water (processed or not), river water, estuary water, ocean water, effluent, treated sewage or untreated sewage.
[0099] Yet further embodiments are described herein.
IV. Systems and Kits for Detection of EDCs
[0100] Disclosed herein is use of a highly sensitive cytoplasm-to-nucleus translocation assay to monitor one or more EDC level or activity, for instance in water samples. A high throughput screen of water samples is described, using in one example a cell line expressing GFP-tagged glucocorticoid receptor (GR) (GFP-GR); the detected output that signals presence of EDC/ligand (such as glucocorticoid) in the sample is accumulation of fluorescence in the cell nucleus (that is, migration of the fluorescent fusion protein from the cytoplasm into the nucleus). By way of example, an automated imaging system (such as the PerkinElmer Opera® High Content Imaging system) can be used to image the change in fluorescence localization in individual cells; also contemplated are non-automated, direct microscopic examination embodiments.
[0101] Though the high-throughput, low cost, high sensitivity EDC detection system is described in detail with regard to a few exemplary traceable cytoplasm-to-nuclear marker proteins, it will be apparent to one of ordinary skill based on the teachings herein that the provided principles can be applied to the detection of other EDC compounds though use of different receptor fusions. Thus, specifically contemplated herein are methods and kits that employ cell lines expressing one or more (such as at least one two, three, four, five, six, seven, eight, or all nine) of the following detector (traceable) fusions: GFP-GR, GFP-AR, GFP-AhR, GFP-PR-B, GFP-GR-ER, GFP-GR-RAR, GFP-MR, GFP-hAR, and GFP-GR-TR, as well as fusion(s) in which the GFP is replaced with a different fluorescent domain that emits light at a different wavelength. Specifically contemplated are kits that include cell lines expressing two or more traceable fusion, where each is responsive to a different EDC and contains a differentially fluorescing traceable domain. Kits containing multiple cell lines, each of which expresses a different cytoplasm-to-nuclear traceable fusion protein are also contemplated. Also envisioned are GFP fusions to mutant, variant, or modified forms of nuclear receptors that manifest detectable subcellular relocalization in response to ligand.
A. Traceable Translocating Fusion Proteins with Non-chimeric Receptors
[0102] Embodiments provided herein employ cells that express a traceable fusion protein, which changes its sub-cellular localization (for example, translocates from the cytoplasm into the nucleus) upon binding of an EDC ligand compound. Examples of such traceable translocating fusion proteins comprise a traceable marker domain (exemplified by a fluorescent protein domain, such as GFP) functionally fused to a nuclear superfamily receptor domain that is sufficient for both recognition of the EDC ligand and for consequent translocation of the fusion protein from the cytoplasm into the nucleus.
[0103] Cell lines expressing other "non-chimeric" receptors (that is, not needing to be fused to the translocation domain of GR) such as GFP-AR (Klokk, et al., Mol. Cell Biol. 27:1823-1843, 2007), GFP-AhR (Elbi et al., Proc Natl Acad Sci USA 101(9):2876-2881, 2004), GFP-PR-B (Rayasam et al., Mol Cell Biol 25(6):2406-2418, 2005) have been generated previously in our laboratory. In addition to the GFP-GR, which could detect biologically active glucocorticoids in water samples, GFP-AR was used for the detection of androgen activity and positive results were obtained for many of the tested water samples.
[0104] A fluorescent protein (or protein domain) is a protein (or portion of a protein) that fluoresces in cells without adding exogenous cofactors. That is, it is a protein that can be expressed in cells and detected in these cells simply by exciting the protein with light and visualizing the resultant fluorescence. An example of such a fluorescent protein is the green fluorescent protein (GFP) originally isolated from the jellyfish Aequorea victoria. Another example of a fluorescent protein as defined herein is the green fluorescent protein originally isolated from Renilla reniforms, which exhibits a single absorption peak at 498 nm and an emission peak at 509 nm (Cubitt et al. TIBS 20: 448-455, 1995). It is also contemplated that modifications may be made to a fluorescent protein, as long as the resulting protein fluoresces when expressed in cells. The art recognizes many modifications that can be (and have been) made to existing fluorescent proteins, for instance to provide different excitation and emission peaks, to increase stability, that speed up the rate of the oxidation step of chromophore formation, that increase brightness at longer wavelengths, and that reduce pohotoisomerization and/or photobleaching.
[0105] Optionally, the fusion protein can include a linking peptide sequence between the traceable (e.g., fluorescent protein) domain and the steroid or other receptor domain. For example, a sequence of the amino acids glycine and alanine, or a sequence of alanine alone can be used; however, any sequence of amino acids and any length can be used that does not interfere with the binding of the receptor domain to its response element and that does not prevent fluorescence of the fluorescent protein. Typically, a linker peptide will range from two to about ten amino acids but maybe shorter or longer. Of course, certain linker peptides maybe preferred over others, e.g., the presence of four basic amino acids in a string of six might suffice as a nuclear localization signal so as to mislocate the uninduced state of the factor. A linker peptide can be used to separate the fluorescent protein structurally from the response element and can function to allow the fluorescent protein independently of the remaining portion of the chimeric protein. Typically, the traceable protein domain (e.g., fluorescent protein) can be fused to either the C-terminus or the N-terminus of the ligand receptor domain; however, the preferable construction for any specific fusion protein can readily be determined. Linker peptides can readily be introduced between the two protein domains in the fusion protein by producing a nucleic acid that encodes the fusion protein having the linker sequence between the detectable protein component and ligand responsive/translocation component.
[0106] The ligand binding domain (e.g., transcription factor component) of the traceable fusion protein can be derived from any selected mammal. Additionally, fusion proteins utilizing a transcription factor from one mammal can often be used in a cell from another mammal. For example, the glucocorticoid receptor amino acid sequence is highly conserved, particularly in the binding region among rat, human and mouse, and, for example, the rat glucocorticoid receptor binds with high affinity to the human glucocorticoid response element. However, in various embodiments provided herein, the ligand-binding and/or translocation component(s) of the traceable fusion proteins are based on human proteins.
[0107] Additional information regarding the construction of traceable translocating fusion proteins useful in the described methods and kits is provided in WO 97/20931 and U.S. Pat. Nos. 6,455,300, 7,312,032 and 8,058,395, each of which is incorporated by reference herein.
B. Traceable Translocating Fusion Proteins with Chimeric Receptors
[0108] Also contemplated are cell lines, methods, systems and kits that employ traceable chimeric receptors, that is traceable receptors that include a portion of a nuclear receptor for example responsible for cytoplasmic/nuclear translocation activity functionally fused to the ligand-binding portion of another superfamily receptor. A representative example is the GR-ER chimera (Martinez et al., J. Ster. Biochem Mol. Biol. 97:307-321, 2005), which contains the rat GR (rGR) N-terminus, DNA binding domain (DBD) and hinge regions upstream of a hybrid ligand binding domain (LBD) composed of GR helix 1 and partial loop 1-3 sequences linked to human ER alpha (hERa) LBD sequences starting with the C-terminus of loop 1-3.
[0109] The construction of examples of such chimeric traceable receptors is described in U.S. Patent Publication No. 2003/0077645, Mackem et al. (J. Biol. Chem. 276(49):45501-45504, 2001), and Martinez et al. (J. Ster. Biochem Mol. Biol. 97:307-321, 2005), each of which is incorporated herein by reference. These references describe production of fluorescently tagged fusion proteins that contain a chimera between the translocation features of glucocorticoid receptor and another nuclear superfamily receptor, such as the estrogen receptor, a retinoic acid receptor, and so forth. In each case, the labeled chimera is found in the cytoplasm in the absence of its cognate ligand), and is rapidly translocated to the nucleus in a dose-dependent manner upon exposure of the cell to ligand.
C. Additional Chimeric Receptors
[0110] GFP-GR (pCI-nGFP-C656G) and GFP-AR (eGFP-hAR) fusion proteins have been published ((Walker et al., Methods (Comp. to Meth. Enzym.) 19:386-393, 1999; Klokk et al., Mol. Cell Biol. 27:1823-1843, 2007) and have proven useful in detecting biologically active glucocorticoid and androgens in water samples, as described below in Example 1. Additional chimeras (eGFP-GR-ER310 and eGFP-GR-TR216) have been constructed. Mammalian cell lines can be readily prepared which express each of these chimeras under tetracycline regulation. The nucleotide sequences of eGFP-GR-ER310 (SEQ ID NO: 1), eGFP-GR-TR216 (SEQ ID NO: 2), pCI-nGFP-C656G (SEQ ID NO: 5), eGFP-hAR (SEQ ID NO: 7), pRevTRE-GFP-mAhR (SEQ ID NO: 9), eGFP-ratMR (SEQ ID NO: 11), and eGFP-hPRB (SEQ ID NO: 13) are provided. The encoded proteins are also provided herein (SEQ ID NOs: 2, 4, 6, 8, 10, 12, and 14, respectively).
D. Detection Cell Lines
[0111] The methods and kits described herein employ cell lines that express at least one traceable translocating fusion protein as described herein. For detection of the translocation of the labeled fusion protein from one sub-cellular compartment or organelle to another, any eukaryotic cell can be utilized, though mammalian cells and particularly human cells are contemplated. Representative methods for the construction of cell lines useful in the described methods and kits, as well as examples of such cell lines, are provided in WO 97/20931, U.S. Pat. Nos. 6,455,300, 7,312,032 and 8,058,395; U.S. Patent Publication No. 2003/0077645, Mackem et al. (J. Biol. Chem. 276(49):45501-45504, 2001), and Martinez et al. (J. Ster. Biochem Mol. Biol. 97:307-321, 2005), each of which is incorporated by reference herein.
[0112] In addition to steroid receptors, there are other ligand-dependent receptors (such as thyroid hormone receptor, retinoic acid receptor, retinoid X receptor, TCCD (dioxin) receptor
[0113] (AhR), fatty acid activatable receptors, etc.) and stimulus-dependent receptors (such as peroxisome proliferator activated receptor, growth factor-dependent receptors such as epidermal growth factor, nerve growth factor, etc.), and factors (such as CREB, NFAT, NFkB/IkB, etc.), and other receptors whose ligand remains to be defined (such as mammalian homologs of the Drosophila tailless, knirps, sevenup, FTZF1 genes, etc.). Many of these receptors or factors can be found listed in Parker, Steroid Hormone Action (Oxford University Press, New York, pp. 210, 1993), in Tsai & O'Malley (Annu. Rev. Biochem. 63:451-486, 1994), and in GenBank and other publicly available sequence databases, which contain additional receptors as well as the complete nucleotide sequences of the genes and cDNAs.
E. Methods for Detecting and/or Quantifying EDC(s) in Environmental Samples and Other Types of Samples
[0114] The present disclosure provides methods of detecting in an environmental sample the presence of an agonist or antagonist of a nuclear superfamily receptor comprising contacting the sample with a cell expressing at least one traceable translocating fusion protein and directly detecting the sub-cellular location of fluorescence (or other appropriate label) within the cell, the location of fluorescence aggregated in the nucleus indicating the presence in the environmental sample of at least one agonist or antagonist of the ligand-binding EDC receptor domain of the traceable translocation fusion protein. In addition to the exemplary methods described herein, methods generally applicable for detecting or quantifying a ligand using a traceable translocation fusion protein can be found in WO 97/20931, U.S. Pat. No. 6,455,300, 7,312,032 and 8,058,395; U.S. Patent Publication No. 2003/0077645, Mackem et al. (J. Biol. Chem. 276(49):45501-45504, 2001), and Martinez et al. (J. Ster. Biochem Mol. Biol. 97:307-321, 2005), each of which is incorporated by reference herein.
[0115] Direct detection means detection of the fluorescence emitted from the site in the cells when excited by (ultraviolet or visible) light, without the need for any additional chemical reactions or treatment of the cells. The fluorescence is directly detected by any device capable of detecting fluorescence, such as a fluorescent microscope, as visualized by the eye of the operator of the microscope at the time or as recorded from the microscope such as by photography of the field of view or through the use of photosensitive detectors. A fluorescent microscope, such as a confocal laser scanning microscope or an epifluorescent microscope, can be used, as is known in the art.
[0116] There is no requirement that cells be, e.g., fixed or stained or contacted by any additional reagents, in order to detect the translocation. Thus living cells can be assayed, and results obtained, shortly (for instance, about 30 minutes) after being contacted with a test sample. Furthermore, screening nuclear localization of fluorescence (translocation) can readily be adapted to high throughput computerized image analysis. Thus, the analysis of large numbers of samples can be automated, for instance for the repetitive examination of multiple environmental samples (for instance, from multiple sources or locations) as well as for the large-scale screening of sets of samples for multiple EDC contaminants.
[0117] Also provided is a method of detecting or monitoring the level of agonists and antagonists of a steroid receptor in an environmental sample, comprising obtaining periodic samples from a source or location over time (e.g., on a daily, weekly, monthly, bi-annually, annually, or longer basis), contacting the samples with cell(s) expressing one or more of the herein-described traceable translocating fusion proteins, and detecting the location of fluorescence within the cells. Optionally, the cells each or collectively express a set of two or more different traceable translocating fusion proteins that are responsive to different ligands (e.g., different EDCs known to or suspected of being contaminants in the environment being sampled). The amount or magnitude of fluorescence migration from the cytoplasm to the nucleus can be compared between the periodic samples, in order to monitor trends (upwards or downwards) in the levels of EDC contamination at a single location or set of locations over time. Decrease in the nuclear translocation in a later-obtained sample relative to an earlier-obtained sample indicating a decrease in level of the EDC agonist or antagonist of the receptor in the sample, while an increase in translocation into the nucleus in a later-obtained sample relative to an earlier-obtained sample indicates an increase in level of the EDC agonist or antagonist of the steroid receptor in the sample.
F. Automated High Throughput Analysis Systems and Image Analysis Software
[0118] Described herein is one embodiment of automated high throughput analysis of the presence of EDCs in environmental samples, using the PerkinElmer Opera® High Content Image Screening System and accompanying software package. With this teaching, additional high throughput applications of the herein described EDC detection and measuring methods are now enabled.
[0119] Contemplated herein is the use of "arrays" of cells that express one or more traceable translocating fusion protein, which cells are placed into an array such as a microchannel array, microtiter plate or other array system that permits separate cell samples to be contacted separately with different test samples (e.g., environmental samples, such as water samples, to be analyzed for the presence or level of one or more EDCs).
[0120] Arrays, as the term is used herein, are arrangements of addressable locations on a substrate; each address may contain a sample of one or more cells or cell lines. A "microarray" is an array that is miniaturized so as to require microscopic examination for evaluation of hybridization or other detection signals. "Macroarrays" are somewhat larger, such that at least the spot at each address is recognizable by the naked human eye. Primary examination for the presence and location of cellular fluorescence (or other traceable signals) in cells of the array is usually carried out using a microscope or other magnifying device.
[0121] Within an array, each arrayed cell sample is addressable, in that its location can be reliably and consistently determined within the at least the two dimensions of the array surface. Thus, in ordered arrays the location of each cell sample is assigned to the sample at the time when it is applied to the array and usually a key is provided in order to correlate each location with the appropriate "target" cell sample. Often, ordered arrays are arranged in a symmetrical grid pattern, but samples could be arranged in other patterns (e.g., in radially distributed lines or ordered clusters).
[0122] The data generated using an array according to this invention (e.g., subcellular localization of one or more traceable translocating fusion proteins) can be analyzed using computerized systems. For instance, the array can be read by a computerized "reader" or scanner and the quantification of the location traceable signal to individual cells and/or subcellular compartments within cells on the array carried out using computer algorithms. Such analysis of the array can be referred to as "automated detection" in that the data is being gathered by an automated reader system.
[0123] In the case of labels that emit detectable electromagnetic waves or particles, the emitted light (e.g., fluorescence or luminescence) or radioactivity can be detected by sensitive cameras, confocal scanners, image analysis devices, radioactive film or a Phosphoimager, which capture the signals (such as a color image or artificial color image) from the array. A computer with image analysis software then detects this image, and analyzes the intensity and/or pattern of the signal for each cell sample location (address), and optionally individual cells within a location, in the array. Signals can be compared between locations on a single array, or between arrays (such as a single array that is sequentially analyzed at different wavelengths in order to detect the localization of different traceable fusion proteins).
[0124] Computer algorithms can also be used for comparison between spots on a single array or on multiple arrays. In addition, the data from an array can be stored in a computer readable form.
[0125] Certain examples of automated array readers (scanners) will be controlled by a computer and software programmed to direct the individual components of the reader (e.g., mechanical components such as motors, analysis components such as signal interpretation and background subtraction). Optionally software may also be provided reader to control a graphic user interface and one or more systems for sorting, categorizing, storing, analyzing, or otherwise processing the data output of the reader.
[0126] By way of example, to "read" an array that contains cells expressing a traceably labeled translocating protein (e.g., a fusion protein that contains a GFP or other fluorescent protein domain), the array can be placed into (or onto, or below, etc., depending on the location of the detector system) the reader and a traceable signal detected by the reader. These detectable signals could be associated with an address identifier signal. The reader gathers information from each of the addresses, associates it with the address identifier signal, and recognizes addresses with a detectable signal (including a particular type of signal, such as relatively intense fluorescence localized to the nucleus of the cell) as distinct from those not producing such a signal (or producing a different kind of signal, such as diffuse fluorescence in the cytoplasm of cells). Certain readers are also capable of detecting intermediate levels of signal, between no signal at all and a high signal, such that quantification of signals at individual addresses is enabled. In particularly beneficial embodiments, the reader is equipped with software that enables it to map individual cells and cell nuclei in order to determine and measure the subcellular localization of the traceable signal.
[0127] Certain readers that can be used to collect data from the arrays used with the described methods, especially those that use a fluorescently tagged fusion protein, will include a light source for optical radiation emission. The wavelength of the excitation light will usually be in the UV or visible range, but in some situations may be extended into the infra-red range. A beam splitter can direct the reader-emitted excitation beam into the object lens, which for instance may be mounted such that it can move in the x, y and z directions in relation to the surface of the array substrate. The objective lens focuses the excitation light onto the array, and more particularly onto the (microbial cell) targets on the array. Light at longer wavelengths than the excitation light is emitted from addresses on the array that contain fluorescently-labeled probe molecules (i.e., those addresses containing a cell to which the probe binds).
[0128] In certain embodiments of the invention, the array may be movably disposed within the reader as it is being read, such that the array itself moves (for instance, rotates) while the reader detects information from each address. Alternatively, the array may be stationary within the reader while the reader detection system moves across or above or around the array to detect information from the addresses of the array. Specific movable-format array readers are known and described, for instance in U.S. Pat. No. 5,922,617, hereby incorporated in its entirety by reference. Examples of methods for generating optical data storage focusing and tracking signals are also known (see, for example, U.S. Pat. No. 5,461,599).
G. Kits for Screening an Environmental Sample, and Methods of Use Thereof
[0129] It is contemplated that the fusion proteins and cells described herein can be used to screen environmental samples for the presence and/or level of one or more EDCs, which methods optionally may be carried out using kits.
[0130] In one specific contemplated example, the kits are very simple--comprising one or more water-tight container(s) in which water or other environmental samples are collected (one sample per container). In examples of this embodiment, the containers are substantially non-reactive and contain no chemicals that might be transferred into the collected sample (e.g., by leaching) and thereby detected during subsequent sample analysis. By way of example only, containers may be 500 ml (or smaller) pre-cleaned glass bottles. Optionally, such kits may include system(s) for holding the sample containers (e.g., a rack or other device, including for instance providing the containers as part of a single unit such that two or more containers are joined to each other), including for instance for holding the containers during shipping or other transportation. Sample collection containers may be joined to each other, for instance in a plate or sheet configuration. In some instances, the kits include an acidifying agent that is used to acidify the environmental samples (e.g., to pH ˜3); an example of such acidifying agent is 6N hydrochloric acid (though one of ordinary skill in the art will recognize that other acidifying agents can be used). The kits may also be formatted to permit chilling of samples once they are collected, for instance chilling to about 4° C.
[0131] Capture kits of this relatively simple variety may be used in a remote-analysis system, where the samples are obtained from the environment at one location and then transported (e.g., by shipping the collected samples) to a remote location for analysis. Optionally, such kits are formatted so that the samples are shipped cold (e.g., under ice) so as to maintain a temperature of ˜4° C. from soon after collection until the samples are processed for analysis at the remote location (e.g., a centralized analysis center, such as a mail-order analysis site). Kits intended for shipping to a remote analysis site optionally may be provided with mailers, such as pre-paid mailers, and/or instructions for how to ship the samples to the analysis site after collection. It is contemplated that regular sample analysis systems may be employed, for instance where a site is (or set of sites are) sampled on a regular basis and the samples returned to the same analysis site on a daily, weekly, bi-weekly, monthly, quarterly, yearly, or other regular basis. In such instances, for instance where a water processing plant or other utility or agency is taking samples, a shipping system or pick-up/delivery system can be used for transporting the samples from the collection site(s). This may enable highly reliable sample movement, while helping to keep costs down for repeat sample analysis.
[0132] Also contemplated are analysis kits, which contemplate and enable the analysis of the sample(s) without needing to engage a remote analysis site. Such kits can be used with the methods described herein to detect (and optionally quantify) one or more EDCs from samples, such as environmental samples. Such analysis kits include (for instance, in addition to one or more components of the capture kits described above--such as sample collection containers) components of a herein described system used to analyze the environmental sample, including for instance one or more (e.g., two, three, four or more) mammalian cell line(s), each of which expresses at least one traceable fusion protein. In one embodiment, the kit implements a system for detecting or quantifying a ligand of a superfamily receptor protein in a water sample, the system comprising a first mammalian cell expressing a first traceable fusion protein, a second mammalian cell expressing a second traceable fusion protein; and (optionally) a detection system for detection of the cytoplasm-to-nuclear translocation of the marker proteins (for instance, detection of the sub-cellular localization of each marker protein), wherein the first and second traceable fusion proteins independently comprise either: (1) a superfamily receptor protein, and a marker protein domain; or (2) the cytoplasmic/nuclear translocation domain of glucocorticoid receptor, the ligand binding domain of a superfamily receptor protein, and a marker protein domain. Optionally, the analysis kit comprises one or more compounds and/or compositions that stably associate with a traceable fusion protein of the kit in the absence of a ligand for the ligand binding domain of the traceable fusion protein, and which compound/composition dissociates from the fusion protein in the presence of a ligand for the ligand binding domain of the traceable fusion protein.
[0133] Specifically contemplated are kits that comprise at least four mammalian cell lines each expressing a different traceable fusion protein, wherein at least one traceable fusion protein of the kit binds a glucocorticoid, at least one traceable fusion protein of the kit binds an androgen, at least one traceable fusion protein of the kit binds a progestin, and at least one traceable fusion protein of the kit binds an aryl hydrocarbon.
[0134] Optionally, analysis kits provided herein include a portable fluorescence reader programmed to detect and monitor translocation of one or more traceable translocating fusion proteins described herein. Portable in this context may refer to a device that can readily be carried by a person into the field. However, in additional embodiments, the portable fluorescence reader is of a size that requires a transport vehicle, for instance a portable analysis van that can be moved from location to location to provide analysis of environmental samples.
[0135] Kits may be provided with instructions, for instance instructions for the collection of environmental samples, instructions for after-collection processing of the samples, for analysis of the samples to detect one or more EDCs using the systems and cells and methods described herein, and so forth. The instructions can provide calibration curves or charts to compare with the determined (e.g., experimentally measured) values.
[0136] Without limitation, the kits provided herein may include any cellular receptor or receptor subdomain tagged florescent protein, including but not limited to the following (generically termed) traceable fusion proteins: GFP-GR, GFP-AR, GFP-AhR, GFP-PR-B, GFP-GR-ER, GFP-GR-RAR, GFP-MR, and GFP-GR-TR, as well as fusion(s) in which the GFP is replaced with a different fluorescent domain that emits light at a different wavelength. By way of specific example, contemplated herein for inclusion (singly or in combinations of two or more) in the described kits are the following traceable fusion proteins: eGFP-GR-ER310 (SEQ ID NOs: 1 & 2); eGFP-GR-TR216 (SEQ ID NOs: 3 & 4); pCI-nGFP-C656G (SEQ ID NOs: 5 & 6); eGFP-hAR (SEQ ID NOs: 7 & 8); pRefTRE-GFP-mAhR (SEQ ID NOs: 9 & 10); eGFP-ratMP (SEQ ID NO: 11 & 12); and/or eGFP-hPRB (SEQ ID NOs: 13 & 14).
[0137] The following example is provided to illustrate certain particular features and/or embodiments. The example should not be construed to limit the invention to the particular features or embodiments described.
EXAMPLE 1
Prevalent Glucocorticoid and Androgen Activity in U.S. Water Sources
[0138] Contamination of the environment with various endocrine disrupting chemicals (EDCs) is a major health concern. The presence of estrogenic compounds in water and their deleterious effects are well documented. However, effective detection and monitoring of EDCs, including corticosteroids and androgens, is lacking.
[0139] Described in this example (and in Stavreva et al., Sci Rep. 2012;2:937. doi: 10.1038/srep00937. Epub Dec. 6, 2012, incorporated herein by reference in its entirety) is use of a highly sensitive live cell assay based on sub-cellular relocalization of green fluorescent protein (GFP)-tagged nuclear receptors, exemplified by the glucocorticoid receptor (GR) and androgen receptor (AR) to test samples from US water sources for hormonal activity. This assay is based on the fact that unoccupied GR resides in the cytoplasm bound to various heat-shock proteins and immunophilins in a large multi-protein complex (Pratt & Tort, Endocr. Rev. 18:306-360, 1997; Pratt et al., Exp. Pharmacol. 111-138, 2006). Upon hormone binding, GR dissociates from the chaperones and translocates to the cell nucleus (FIGS. 1A and 1B), where it interacts with GR regulatory elements (GREs) to elicit GR-specific transcription regulation. This cell based assay is used to monitor glucocorticoid and androgen activity in water samples from more than 100 locations in the Northeast United States. Using cell lines expressing GFP-tagged glucocorticoid and androgen receptors (GFP-GR and GFP-AR, respectively) in high a throughput screening, combined with studies on transcription activation, previously unrecognized glucocorticoid activity was discovered in 27%, and androgen activity in 35%, of all tested water sources from 14 states. In addition to nuclear translocation, water samples induced GR and AR-dependent gene expression. Steroids of both classes impact body development, metabolism, and interfere with reproductive, endocrine, and immune systems. This prevalent contamination could negatively affect wildlife and humans.
[0140] The levels of steroidal EDCs in the environment are not efficiently monitored and/or regulated at present. One of the reasons is that a high-throughput, reliable, and low-cost detection methods for monitoring of biologically active steroidal EDCs, including corticosteroids, have not been developed and are in demand (Roy & Pereira, Indian J Exp. Biol 43:975-992, 2005).
[0141] Chemical methods for detection of EDCs reveal the presence of several classes of steroid hormones in water sources in China (Chang et al., Environ. Sci. Technol. 43:7691-7698, 2009).
[0142] Mass spectrometry data from wastewaters in the Netherlands (Schriks et al., Environ. Sci. Technol. 44:4766-4774, 2010) and also China (Chang et al., Environ. Sci. Technol. 41:3462-3468, 2007) suggest possible contamination with glucocorticoids and a recent study demonstrated that environmentally relevant concentrations of synthetic glucocorticoids have deleterious effects on fish (Kugathas & Sumpter, Environ. Sci. Technol. 45:2377-2383, 2011). However, it is unclear whether in U.S. water sources are also contaminated by EDCs and to what extent. It is also unclear whether the EDCs detected in water sources could elicit steroid-specific biological response(s) in mammalian systems.
[0143] Chemical methods for EDCs detection, although sensitive, are expensive, time-consuming, and largely incompatible with a large-scale sample testing. Therefore, it is crucial to develop, test, and implement methods for high-speed, reliable, and low-cost detection of biologically active EDCs in contaminated water and other environmental sources. Here we utilize a highly sensitive cellular assay based on cytoplasm to nucleus translocation to detect glucocorticoid activity in U.S. water sources.
Materials and Methods
Samples Collection
[0144] Environmental water samples were collected as part of ongoing U.S. Geological Survey (USGS) projects that were implemented to monitor the presence and effects of endocrine-disruptors and other contaminants of emerging concern. They were collected between 2005 and 2010 from different geographic locations in the United States (FIG. 8), and included discrete grab water samples, or samples collected via polar organic chemical integrative samplers (POCIS) (see, Miege et al., J. Environ. Monit., 14:626-635, 2012; epub Dec. 22, 2011). Samples in the R series were collected on or around National Wildlife Refuges in the Northeast and the GL series on tributaries of the Great Lakes, both were collected as part of collaborative projects with the US Fish and Wildlife Services. All other samples were collected as part of the USGS Chesapeake Bay Priority Ecosystems Science projects. Grab water samples were processed at the USGS, Leetown Science Center as described below.
[0145] POCIS samples: The POCIS membranes were shipped to the USGS, Columbia Environmental Research Center for analyte recovery. The procedures used for preparing the POCIS samples for analysis were described earlier (Alvarez et al., Environ. Toxicol. Chem. 28:1084-1095, 2009). Briefly, chemicals of interest were recovered from the POCIS sorbent using 50 mL of 1:1:8 (V:V:V) methanol:toluene:dichloromethane followed by 20 mL of ethyl acetate. The extracts were reduced by rotary evaporation, filtered, and composited into 2-POCIS equivalent samples thereby concentrating the amount of chemical present in each sample to aid in the detection.
[0146] Grab Water samples: Grab water samples were collected in 500 ml pre-cleaned amber glass bottles (I-Chem, Rockwood, Tenn.). Water was acidified to pH 3 using 6N hydrochloric acid, held on ice, and stored at 4° C. Within one week of collection, the preserved water samples were filtered through a GF/F filter (0.7 μm) using a solvent rinsed all-glass apparatus. Filters were rinsed with 1 ml of methanol to liberate soluble compounds from the retained suspended solids. Filtered samples and blanks were subjected to solid phase extraction (SPE) using OASIS® HLB (200 mg) glass cartridges (Waters Corporation, Milford, Mass.), following an existing protocol (Ciparis et al., Sci. Total Environ. 414:268-276, 2012). In short, cartridges were sequentially pre-conditioned and 400 ml of filtered samples were loaded onto the cartridge at a flow rate of 5-6 ml/minute (continuous vacuum). Analytes were eluted from the cartridge with 100% methanol and concentrated by rotary evaporation.
[0147] For biological testing, samples prepared as above were reconstituted in DMSO and diluted in growth media to a final 1,000× concentration from the original water volume while maintaining DMSO at <0.2%. Samples were added to cells for 30 minutes at 100 x concentration or as indicated in the text.
Cell Lines and Translocation Assay
[0148] The 3617 and 3108 cell lines are derivatives of 3134 mouse mammary adenocarcinoma cell line that express green fluorescent protein (GFP)-tagged GR (GFP-GR) and AR (GFP-AR), respectively from a chromosomal locus under control of the tetracycline-repressible promoter (Walker et al., Methods (Comp. to Meth. Enzym.) 19:386-393, 1999; Klokk et al., Mol. Cell Biol. 27:1823-1843, 2007). Prior to imaging, cells were grown overnight on 22-mm2 coverslips in DMEM medium containing 10% charcoal stripped serum (Hyclone, Logan, Utah) without tetracycline (to allow the expression of the GFP-GR or GFP-AR, respectively) at a density of 2×105 per 6-well plate. For the automated experiments conducted in 96 or 384 well plates, cell density was 10, 000 or 2,500 cells per well, respectively. Cells were treated with vehicle control, hormones (100 nM) or water samples for 30 min at 37° C. at a final concentration of 100×for water samples (unless otherwise specified). Additional negative controls contained samples that tested the activity of the POCIS membranes themselves.
[0149] Upon treatment, cells were fixed with 4% paraformaldehyde in PBS for 15 minutes and washed three times with PBS. Cells on the 22-mm2 coverslips were mounted in VECTASHIELD® mounting media with DAPI (Vector Laboratories, Inc.) and examined on a Leica DMRA microscope with Leica 100× 1.3-N.A. oil immersion objective. Images were acquired in green (GFP-GR and GFP-AR) and UV (DAPI) channel with SenSys (Photometrics) camera with KAF1400 chip configured to collect 0.067-μm-diameter pixels. For the automated experiments conducted in 96 or 384 well plates, cells were stained with DRAQ5 (BioStatus Limited) at a dilution of 1:5000 for 15 minutes and after three final washes with PBS were imaged either immediately on the Perkin Elmer Opera Image Screening System or kept in PBS at 4° C. for later imaging.
Automated Imaging and Analysis by Perkin Elmer Opera Image Screening System
[0150] A Perkin Elmer Opera Image Screening System was used for fully automated collection of fluorescence images of cells. This system employed a 40×water immersion objective lens, laser illuminated Nipkow disk, and cooled CCD cameras to digitally capture high resolution confocal fluorescence micrographs (300 nm pixel size with 2×2 camera pixel binning). An algorithm was customized using the Acapella image analysis software development kit (Perkin Elmer) to automatically segment both the nucleus and cytoplasm of each cell in the digital micrographs. The algorithm also measured the mean GFP-GR or GFP-AR intensity in both compartments, and translocation was calculated as a ratio of these intensities. Each value was further normalized to the value for the control (DMSO) sample.
Gene Transcription Analysis
[0151] For gene transcription studies, 3134 cells or LNCaP cells (expressing endogenous GR and AR, respectively) were plated in 24-well dishes 24 hours before each experiment in DMEM (3134 cells) or RPMI (LNCaP cells) media supplemented with charcoal stripped fetal bovine serum (Hyclone, Logan, Utah). Cells were treated with water samples, vehicle control (DMSO), or GR and AR specific hormones for 30 minutes. To reduce cell stress, these experiments were performed under conditions of stable CO2 and temperature levels throughout the duration of an experiment (though this is not essential for the assay). Cells were lysed in 600 μl of RLT buffer (with β-mercaptoethanol added) followed by syringe/needle shearing. Total RNA was extracted using the RNeasy Mini Kit (Qiagen), including a DNaseI digestion step (RNase free DNase Set, Qiagen). One microgram (μg) of RNA was reverse transcribed (iScript cDNA Synthesis Kit, BioRad) in 20 μl reaction volume and 0.5 μl was used per Q-PCR reaction using SyBr green and Bio-Rad IQ system (BioRad, Hercules, Calif.). Primer sequences were designed to amplify nascent RNA (amplicons that cross an exon/intron boundary). The primer sequences are shown below. PCR was performed as recommended by a manufacturer. Standard curves were created by 10-fold serial dilution of template. The expression data from three or more independent experiments were normalized to the expression of a control gene β-Actin (3134 cells) and GAPDH (LNCaP cells), the mean values and SEM were calculated and displayed as a fold change in relation to the control (DMSO treated) sample.
TABLE-US-00017 Primer sequences for Q-PCR analysis Mouse cells SEQ ID (3134) Sequence NO: Per1 For CTTCTGGCAATGGCAAGGACTC 15 Per1 Rev CAGCATCATGCCATCATACACACA 16 Tgm2 For TGTCACCAGGGATGAGAGACGG 17 Tgm2 Rev TCCAAATCACACCTCTCCAGGAG 18 Lcn2 For ACCTCTCATTTCTTGCAGTTCCG 19 Lcn2 Rev CAGGATGGAGGTGACATTGTAGCT 20 β-Actin For AGTGTGACGTTGACATCCGTA 21 β-Actin Rev GCCAGAGCAGTAATCTCCTTCT 22 Human cells SEQ ID (LNCaP) Sequence NO: hNKX3.1 For TGACAGTGGGCTGTTTGTTC 23 hNKX3.1 Rev AAGACCCCAAGTGCCTTTCT 24 hRHOU For TTTCAAGGATGCTGGCTCTT 25 hRHOU Rev GGCCTCAGCTTGTCAAATTC 26 GAPDH For AAGGTGAAGGTCGGAGTCAAC 27 GAPDH Rev GGGGTCATTGATGGCAACAATA 28
Statistical Analyses
[0152] Data were analyzed using the statistical functions of IBM SPSS Statistics 19 and SigmaPlot 11 (SPSS Inc., Chicago, Ill.). From the repeated experiments, the mean value was calculated for each sample. The mean values were used in a one-way analysis of variance test. If a significant F-value of P<0.05 was obtained, a Dunnett's multiple comparison versus the control group analysis was conducted.
TABLE-US-00018 TABLE 2 Collection sites and collection time of the first sample set. Collection Samples Date/year Location method Blank October-November 2005 POCIS WV2 October-November 2005 South Branch Potomac, Franklin, WV POCIS WV3 October-November 2005 South Branch Potomac, Springfield, WV POCIS WV4 October-November 2005 South Branch Potomac, Moorefield, WV POCIS WV5 October-November 2005 Elk River, WV POCIS WV6 October-November 2005 South Branch Potomac, Petersburg Gap, WV POCIS WV7 October-November 2005 Greenbrier River, WV POCIS SS83 Jun. 1, 2007 POCIS SS92 May 3, 2007-June 7, 2007 Gauley River, WV POCIS SS93 Apr. 11, 2007-May 9, 2007 Lower Conococheague River, MD POCIS SS94 Apr. 11, 2007-May 9, 2007 Upper Conococheague River, MD POCIS SS95 Apr. 27, 2007-May 31, 2007 South Branch Potomac, Petersburg Gap, WV POCIS SS97 Apr. 11, 2007-May 9, 2007 Lower Monocacy River, MD POCIS SS98 Apr. 5, 2007-May 9, 2007 South Fork, Shenandoah River, VA POCIS
TABLE-US-00019 TABLE 3 Synthetic glucocorticoids surveyed by monitoring the mass spectrometric data for the presence of the corresponding molecular ion in sample SS97 1 amcinonide 2 betamethasone 3 budesonide 4 clobetasone 5 clobetasol 6 propionate 7 desonide 8 fluocinonide 9 fluocinolone acetonide 10 fluocortolone 11 fluprednidene acetate 12 halcinonide 13 hydrocortisone 14 hydrocortisone-17-butyrate 15 methylprednisolone 16 mometasone 17 mometasone furoate 18 prednicarbate 19 prednisolone 20 prednisone 21 triamcinolone acetonide
TABLE-US-00020 TABLE 4 Closest EIMS library matches of the mass spectra of chromatographic peaks 1-3 in FIG. 1D. ~Observed Peak # MW Closest EIMS Library Match Structure 1 272 7α-Hydroxy-5α-androstan-3-one MW 290 ##STR00001## 2 286 Androst-4-en-3,6-dione MW 286 ##STR00002## or 4,4-Dimethyl-androst-5-ene MW 286 ##STR00003## 3 272 ? eα-Hydroxy-17-ethylidene-5α- androstane MW 302 ##STR00004##
Table 5A-5B contains information about additional samples tested for GFP-GR and GFP-AR translocation efficiency.
TABLE-US-00021 TABLE 5A Geographic location, time and methods of collection of the water samples. Collection Samples Date/year Location method Plate 1 R1 Sep. 2, 2008 Ohio River, Wheeling WV Grab sample R2 Sep. 3, 2008 Ohio River, Parkersburg, WV Grab sample R3 Sep. 3, 2008 Upstream Brighton Dam, Tridelphia reservoir, MD Grab sample R4A Sep. 10, 2008 Patuxent/Western Branch, MD Grab sample R4E Sep. 10, 2008 Patuxent/Western Branch, MD (WWTP effluent) Grab sample R5 Sep. 15, 2008 Seneca River, NY Grab sample R6 Sep. 16, 2008 Seneca River, NY Grab sample R7 Sep. 22, 2008 St. Croix River, ME Grab sample R8 Sep. 23, 2008 St. Croix River, ME Grab sample R9 Oct. 2, 2008 Sudbury river, MA Grab sample R10 Oct. 3, 2008 Sudbury river, MA Grab sample R13 Oct. 15, 2008 Rappahanock River, VA Grab sample R14 Oct. 15, 2008 Rappahanock River, VA Grab sample R15 Oct. 6, 2008 Rappahanock River, VA Grab sample R16 Oct. 27, 2008 Darby Creek, PA Grab sample R16W Oct. 27, 2008 Darby Creek, PA (WWTP effluent) Grab sample R21 Sep. 1, 2009 Missisquoi River, VT Grab sample R22 Sep. 1, 2009 Missisquoi River, VT Grab sample R22W Sep. 1, 2009 Missisquoi River, VT (WWTP effluent) Grab sample R23 Sep. 14, 2009 Penobscot River, ME Grab sample R24 Sep. 15, 2009 Penobscot River, ME Grab sample R26 Sep. 22, 2009 Peconic Lake, NY Grab sample R27 Sep. 28, 2009 Potomac River, Pohick, VA Grab sample R27W Sep. 28, 2009 Potomac River, Pohick, VA (WWTP effluent) Grab sample R28 Sep. 29, 2009 Burke Lake, VA Grab sample R29 Oct. 5, 2009 Delaware River, Cherry Valley, DE Grab sample R30 Oct. 6, 2009 Delaware River, Easton, PA Grab sample R31 Oct. 13, 2009 Susquehanna River, Garret Island, PA Grab sample R32 Oct. 14, 2009 Susquehanna River, Conowingo Dam, PA Grab sample Broad R32 CON Oct. 14, 2009 Susquehanna River, Conowingo Dam, PA Grab sample PSS2a May 14, 2010 Gooney Run, VA Grab sample PSS2b Jun. 14, 2010 Gooney Run, VA Grab sample PSS3a May 14, 2010 Passage Creek, VA Grab sample PSS3b Jun. 15, 2010 Passage Creek, VA Grab sample PSS4a May 14, 2010 Stony Creek (upstream), VA Grab sample PSS4b Jun. 15, 2010 Stony Creek (upstream), VA Grab sample PSS5a May 14, 2010 Stony Creek (downstream), VA Grab sample PSS5b Jun. 15, 2010 Stony Creek (downstream), VA Grab sample PSS6a May 13, 2010 Mill Creek, VA Grab sample PSS6b Jun. 14, 2010 Mill Creek, VA Grab sample PSS7a May 14, 2010 Hawksbill Creek, VA Grab sample PSS7b Jun. 14, 2010 Hawksbill Creek, VA Grab sample PSS8a May 13, 2010 Smith Creek (downstream), VA Grab sample PSS8b Jun. 14, 2010 Smith Creek (downstream), VA Grab sample PSS9a May 12, 2010 Naked Creek, VA Grab sample PSS9b Jun. 15, 2010 Naked Creek, VA Grab sample PSS10a May 13, 2010 Briery Branch, VA Grab sample PSS10b Jun. 15, 2010 Briery Branch, VA Grab sample PSS11a May 13, 2010 Smith Creek (upstream), VA Grab sample PSS11b Jun. 14, 2010 Smith Creek (upstream), VA Grab sample PSS12a May 13, 2010 Long Glade Creek, VA Grab sample PSS12b Jun. 14, 2010 Long Glade Creek, VA Grab sample PSS13a May 13, 2010 Linville Creek, VA Grab sample PSS13b Jun. 14, 2010 Linville Creek, VA Grab sample PSS14a May 13, 2010 Long Meadow Run, VA Grab sample PSS14b Jun. 14, 2010 Long Meadow Run, VA Grab sample PSS15a May 13, 2010 Muddy Creek, VA Grab sample PSS15b Jun. 15, 2010 Muddy Creek, VA Grab sample PSS16a May 12, 2010 Cooks Creek, VA Grab sample PSS16b Jun. 14, 2010 Cooks Creek, VA Grab sample Dairy Sep. 28, 2010 North Fork Shenandoah at Woodstock, VA Grab sample LF1a Oct. 15, 2008 WWTP1 effluent, Rappahannock river, Little Falls, Grab sample VA LF2a Oct. 15, 2008 WWTP2 effluent, Rappahannock river, Little Falls, Grab sample VA LF3a Oct. 15, 2008 WWTP3 effluent, Rappahannock river, Little Falls, Grab sample VA LF4a Oct. 15, 2008 WWTP4 effluent, Rappahannock river, Little Falls, Grab sample VA SUS1 May 12, 2009 Susquehanna river at Mahantango Access, PA Grab sample SUS2 May 13, 2009 West Branch Susquehanna river, PA Grab sample Plate 2 SUS5 Apr. 21, 2010-Jun. 1, 2010 Juniata River, Newport, PA POCIS SUS6 Apr. 21, 2010-Jun. 1, 2010 Susquehanna River at Mahantango Creek, PA POCIS DE1 Apr. 19, 2010-Jun. 3, 2010 Delaware River, Yardley, PA POCIS AL1 Apr. 28, 2010-Jun. 7, 2010 Allegheny River, Kittanning, PA POCIS PSS16 May 12, 2010-Jun. 14, 2010 Cooks Creek, VA POCIS PSS09 May 12, 2010-Jun. 15, 2010 Naked Creek, VA POCIS PSS15 May 13, 2010-Jun. 15, 2010 Muddy Creek, VA POCIS PSS10 May 13, 2010-Jun. 15, 2010 Briery Branch, VA POCIS PSS12 May 13, 2010-Jun. 14, 2010 Long Glade Creek, VA POCIS PSS11 May 13, 2010-Jun. 14, 2010 Smith Creek (upstream), VA POCIS PSS13 May 13, 2010-Jun. 14, 2010 Linville Creek, VA POCIS PSS14 May 13, 2010-Jun. 14, 2010 Long Meadow Run, VA POCIS PSS08 May 13, 2010-Jun. 14, 2010 Smith Creek (downstream), VA POCIS PSS06 May 13, 2010-Jun. 14, 2010 Mill Creek, VA POCIS PSS02 May 14, 2010-Jun. 14, 2010 Gooney Run, VA POCIS PSS07 May 14, 2010-Jun. 14, 2010 Hawksbill Creek, VA POCIS PSS03 May 14, 2010-Jun. 14, 2010 Passage Creek, VA POCIS PSS05 May 14, 2010-Jun. 14, 2010 Stony Creek (downstream), VA POCIS PSS04 May 14, 2010-Jun. 14, 2010 Stony Creek (upstream), VA POCIS FAB Jun. 1, 2010 Blank POCIS 1FB Jun. 1, 2010 Blank POCIS 2FB Jun. 1, 2010 Blank POCIS 3FB Jun. 1, 2010 Blank POCIS 4FV Jun. 1, 2010 Blank POCIS 5FB Jun. 1, 2010 Blank POCIS 6FB Jun. 1, 2010 Blank POCIS 7FB Jun. 1, 2010 Blank POCIS 8FB Jun. 1, 2010 Blank POCIS 9FB Jun. 1, 2010 Blank POCIS 10FB Jun. 1, 2010 Blank POCIS 11FB Jun. 1, 2010 Blank POCIS 12FB Jun. 1, 2010 Blank POCIS 13FB Jun. 1, 2010 Blank POCIS 14FB Jun. 1, 2010 Blank POCIS 15FB Jun. 1, 2010 Blank POCIS 16FB Jun. 1, 2010 Blank POCIS 17FB Jun. 1, 2010 Blank POCIS 18FB Jun. 1, 2010 Blank POCIS 19FB Jun. 1, 2010 Blank POCIS BLANK Jun. 1, 2010 Blank POCIS R38 Sep. 20, 2010 PA Erie (Pond H) Grab sample R39 Sep. 28, 2010 NJ Great Swamp (Hidden Valley Nursery) Grab sample R40 Oct. 4, 2010 VA Back Bay Grab sample R41 Oct. 5, 2010 VA Wilna Pond Grab sample R42 Oct. 6, 2010 VA Chandler's Mill Pond Grab sample R43 Oct. 13, 2010 MD Patuxent (Snowden Pond) Grab sample R44 Oct. 13, 2010 MD Patuxent (Cash Lake) Grab sample GL1 Oct. 1, 2010 Genesee River, NY Grab sample GL2W Oct. 1, 2010 St. Louis River, Duluth, MN (WWTP effluent) Grab sample GL2P Oct. 1, 2010 St. Louis River, Duluth, MN (Power Plant effluent) Grab sample GL5 Oct. 1, 2010 Fox river, Green Bay, WI Grab sample GL4CSOa Oct. 1, 2010 Swan Creek, Ohio Grab sample GL4CSOb Oct. 1, 2010 Swan Creek, Ohio Grab sample GL4CSOc Oct. 1, 2010 Swan Creek, Ohio Grab sample GL3a Oct. 1, 2010 Detroit River, MI Grab sample GL3b Oct. 1, 2010 Detroit River, MI Grab sample GL3c Oct. 1, 2010 Detroit River, MI Grab sample R36 Sep. 14, 2010 Missisquoi River (Gander Bay & Goose Bay), VT Grab sample R37 Sep. 16, 2010 Lake Umbagog, MA Grab sample
TABLE-US-00022 TABLE 5B Activity of the samples in the GFP-GR and GFP-AR translocation assays (P < 0.01 and P < 0.05, asterisks). GR translocation AR translocation Samples P < 0.01 P < 0.05 P < 0.01 P < 0.05 Plate 1 R1 FALSE FALSE FALSE FALSE R2 FALSE FALSE FALSE FALSE R3 FALSE FALSE FALSE FALSE R4A * * * * R4E * * * * R5 FALSE FALSE FALSE FALSE R6 FALSE FALSE FALSE FALSE R7 FALSE FALSE FALSE FALSE R8 FALSE FALSE * * R9 FALSE FALSE * * R10 FALSE FALSE * * R13 FALSE FALSE FALSE FALSE R14 FALSE FALSE FALSE FALSE R15 FALSE FALSE * * R16 FALSE FALSE * * R16W * * FALSE * R21 FALSE FALSE FALSE FALSE R22 FALSE FALSE FALSE FALSE R22W FALSE FALSE FALSE FALSE R23 FALSE FALSE FALSE FALSE R24 FALSE FALSE FALSE FALSE R26 FALSE FALSE FALSE FALSE R27 * * FALSE FALSE R27W FALSE FALSE FALSE FALSE R28 FALSE FALSE FALSE FALSE R29 FALSE FALSE FALSE FALSE R30 FALSE FALSE FALSE FALSE R31 FALSE FALSE FALSE FALSE R32 FALSE FALSE FALSE FALSE Broad R32 CON FALSE FALSE FALSE FALSE PSS2a FALSE FALSE FALSE FALSE PSS2b FALSE FALSE FALSE FALSE PSS3a FALSE FALSE FALSE FALSE PSS3b FALSE FALSE FALSE FALSE PSS4a FALSE FALSE FALSE FALSE PSS4b FALSE FALSE FALSE FALSE PSS5a FALSE FALSE FALSE FALSE PSS5b FALSE FALSE FALSE FALSE PSS6a FALSE FALSE FALSE FALSE PSS6b FALSE FALSE * * PSS7a FALSE FALSE FALSE FALSE PSS7b FALSE FALSE FALSE FALSE PSS8a FALSE FALSE FALSE FALSE PSS8b FALSE FALSE FALSE FALSE PSS9a FALSE FALSE FALSE FALSE PSS9b FALSE FALSE FALSE FALSE PSS10a FALSE FALSE FALSE FALSE PSS10b FALSE FALSE FALSE FALSE PSS11a FALSE FALSE FALSE FALSE PSS11b FALSE FALSE FALSE * PSS12a FALSE FALSE FALSE FALSE PSS12b FALSE FALSE * * PSS13a FALSE FALSE FALSE FALSE PSS13b FALSE FALSE * * PSS14a FALSE FALSE FALSE FALSE PSS14b FALSE FALSE * * PSS15a FALSE FALSE * * PSS15b FALSE FALSE FALSE FALSE PSS16a FALSE FALSE FALSE FALSE PSS16b FALSE FALSE * * Dairy FALSE FALSE FALSE FALSE LF1a FALSE * * * LF2a * * * * LF3a * * FALSE FALSE LF4a * * FALSE * SUS1 FALSE FALSE FALSE FALSE SUS2 FALSE FALSE FALSE FALSE Plate 2 SUS5 * * * * SUS6 * * * * DE1 * * * * AL1 * * * * PSS16 * * * * PSS09 * * * * PSS15 * * * * PSS10 * * * * PSS12 * * * * PSS11 * * FALSE * PSS13 * * * * PSS14 * * * * PSS08 * * FALSE * PSS06 * * * * PSS02 * * * * PSS07 * * * * PSS03 FALSE FALSE FALSE FALSE PSS05 * * * * PSS04 * * FALSE FALSE FAB FALSE FALSE FALSE FALSE 1FB FALSE FALSE FALSE FALSE 2FB FALSE FALSE FALSE FALSE 3FB FALSE FALSE FALSE FALSE 4FV FALSE FALSE FALSE FALSE 5FB FALSE FALSE FALSE FALSE 6FB FALSE FALSE FALSE FALSE 7FB FALSE FALSE FALSE FALSE 8FB FALSE FALSE FALSE FALSE 9FB FALSE FALSE FALSE FALSE 10FB FALSE FALSE FALSE FALSE 11FB FALSE FALSE FALSE FALSE 12FB FALSE FALSE FALSE FALSE 13FB FALSE FALSE FALSE FALSE 14FB FALSE FALSE FALSE FALSE 15FB FALSE FALSE FALSE FALSE 16FB FALSE FALSE FALSE FALSE 17FB FALSE FALSE FALSE FALSE 18FB FALSE FALSE FALSE FALSE 19FB FALSE FALSE FALSE FALSE BLANK FALSE FALSE FALSE FALSE R38 FALSE FALSE FALSE FALSE R39 FALSE FALSE FALSE FALSE R40 FALSE FALSE FALSE FALSE R41 FALSE FALSE FALSE FALSE R42 FALSE FALSE FALSE FALSE R43 FALSE FALSE FALSE FALSE R44 FALSE FALSE FALSE FALSE GL1 FALSE FALSE FALSE FALSE GL2W * * FALSE * GL2P FALSE FALSE FALSE FALSE GL5 FALSE FALSE FALSE FALSE GL4CSOa FALSE * * * GL4CSOb * * * * GL4CSOc FALSE FALSE * * GL3a FALSE FALSE FALSE FALSE GL3b FALSE FALSE FALSE FALSE GL3c * * FALSE FALSE R36 FALSE FALSE FALSE FALSE R37 FALSE FALSE FALSE FALSE
Results & Discussion
[0153] Accumulated data hint at an unacknowledged and potentially wide spread contamination with glucocorticoids (GCs) at biologically-relevant concentrations. To directly examine this possibility, 10 water samples collected from different locations (FIG. 2A and Table 2) were tested for GC activity using GFP-GR translocation assay. The presence of GC activity in one of these samples (SS97) was revealed by the accumulation of GFP-GR in the nucleus within 30 minutes (FIG. 1C, images). Furthermore, this sample induced transcriptional activity as measured by an increase of the nascent transcript of the GR-regulated gene, Per1, to a significantly higher level than the positive control, corticosterone (at a physiologically relevant dose of 100 nM) (FIG. 1C, graph).
[0154] In an attempt to determine the active constituent(s) in sample SS97, known corticosteroids (dexamethasone and corticosterone) were tested by high performance liquid chromatography/mass spectrometry (HPLC/MS) analysis to establish chromatographic retention times on a C18 HPLC column using published techniques (Chang et al., Environ. Sci. Technol. 43, 7691-7698, 2009). In addition, 20 other synthetic GCs were surveyed by monitoring the mass spectrometric data for the presence of the corresponding molecular ions (Table 3). Under these assay conditions, sample SS97 showed no evidence of any known compounds tested. Next, sample SS97 was subjected to HPLC fractionation followed by biological testing. Four of the eleven HPLC fractions showed activity in the nuclear translocation assay (FIG. 2C). Again, when these fractions were tested by ultra-performance liquid chromatography/mass spectrometry (UPLC/MS), no known GCs compounds were detected. The active fractions were also analyzed by gas chromatography/MS (GC/MS) (Mansilha et al., J Chromatogr. A 1217(43):6681-6691, 2010), and appeared similar in composition to volatile components. The mass spectra extracted from the GC/MS analysis were searched in both the NIST/EPA/NIH Mass Spectral Library 1998 and in the Wiley Mass Spectra Database of Androgens, Estrogens, and other Steroids 2010 (AES 2010), yielding no hits of high certainty for any of the peaks.
[0155] Visual comparison of the mass spectra of chromatographic peaks 1-3 (FIG. 1D) with standard spectra from the AES 2010 database (FIG. 3) suggested similarities to known androstane-class compounds (FIG. 1D, Table 4). One of these compounds, androst-4-en-3,6-dione (peak 2), was synthesized (Hunter & Priest, Steroids 71, 30, 2006) and further tested for biological activity. Androst-4-en-3,6-dione did not induce GFP-GR translocation (data not shown) whereas it induced GFP-tagged androgen receptor (GFP-AR) translocation (FIG. 1E) using a GFP-AR expressing cell line, 3108 (Klokk et al., Mol. Cell Biol. 27:1823-1843, 2007). These data suggest that, in addition to GCs, sample SS97 also contains androgenic activity.
[0156] Androgens are the original anabolic steroids and the precursor of all estrogens, the female sex hormones. Through their binding to androgen receptor (AR), they control the development and maintenance of male characteristics in vertebrates (Gottlieb et al., Reprod. Biomed. Online. 10:42-48, 2005). Similarly to the GR, AR is largely cytoplasmic in the absence of its ligand, and rapidly translocates to the nucleus in response to testosterone (Klokk et al., Mol. Cell Biol. 27:1823-1843, 2007) (FIG. 1E).
[0157] We conclude that environmental degradation and metabolic processes alter the structure of the glucocorticoid(s) in water samples, producing bioactive chemical structures which are not contained in the existing databases. Rapid transformation of hormonal steroids by aquatic microorganisms has been reported previously (Yin et al., Environ. Int. 28:545-551, 2002). We also conclude that, in contrast to the traditional chemical analysis, the translocation assay described herein is faster, cheaper, and also detects biologically relevant hormonal activity which cannot readily be discerned by chemical methods. Translocation assay allows unbiased "non-candidate" approach for detection of EDCs and could be used in a powerful combination with fractionation methods and "forensic chemistry" in the discovery of novel bioactive ligands.
[0158] Next, the search was expanded to screen over 100 additional samples from water sources throughout 14 states in the US (Table 5A) for both, glucocorticoid and androgen activities. To accomplish this screening, the GFP-GR- and GFP-AR-expressing cell lines (Walker et al., Methods (Comp. to Meth. Enzym.) 19:386-393, 1999; Klokk et al., Mol. Cell Biol. 27:1823-1843, 2007) were implemented in an automated imaging analysis system (Perkin Elmer Opera Image Screening System) and an algorithm for cytoplasm and nuclear segmentation was used to calculate translocation efficiency (FIGS. 4A and 4B). To test the sensitivity and reproducibility of the automated assay, translocation efficiency in response to known concentrations of the respective hormones was measured. GFP-GR translocated to the nucleus in a concentration-dependent manner in response to the rodent, human, as well as synthetic hormones (corticosterone, hydrocortisone, and dexamethasone, respectively) (FIG. 5A). The GFP-tagged AR also translocated to the nucleus in concentration-dependent manner in response to testosterone as well as synthesized androst-4-en-3,6-dione (FIGS. 5B and 5C). Confident in the sensitivity of the translocation assay, the additional water samples were tested after being divided into two plates: plate one [P1, (FIGS. 4D, 4E)] and plate two [P2, (FIGS. 6 and 6)]. Glucocorticoid activity was evident in over 28% (FIG. 4D and FIG. 6) and androgen activity in 37% (FIG. 4E and FIG. 7) of the 105 samples subjected to the high throughput screening (Tables 5A and 5B). When combined with the results obtained from the first manual screen of 10 samples (Table 2), glucocorticoid and androgen activity remained in the same range (27% and 35%, respectively). These results unambiguously demonstrate a wide spread contamination of the US water sources from 14 different states with both, glucocorticoid and androgenic activities (FIG. 3, Tables 5A and 5B).
[0159] Considering that the tested samples were collected over a span of several years (Tables 2 and 5A), we sought to determine whether the observed contaminations persist over time. Two of the previously identified contaminated sites (SS97 and GL2W) were revisited and new grab water samples were collected. As shown in FIGS. 4A and B (as well as in FIGS. S6, S7, S8, S9) both newly collected samples induced GFP-GR and GFP-AR nuclear translocation in a concentration-dependent manner, suggesting high and persisting water contamination at these sites. Tenfold concentrated samples from both locations were active in GR and AR translocation assays, and induced transcriptional activity. Moreover, at 1×concentration, sample SS97 induced significant GFP-GR translocation (FIG. 9A-insert) and activation of gene transcription from GR-responsive genes (FIG. 9E). These results indicate that the water at the SS97 location has biologically relevant glucocorticoid activity that is persistent over time.
[0160] Interestingly, 100× concentration of samples SS97 (FIG. 9F) and GL2W (FIG. 13C) were less potent than the lower doses in inducing gene transcription from AR-regulated genes. This could be an example of the well-known phenomenon of non-monotonic dose-response where the effects of the low doses of EDCs cannot be predicted by the effects observed at high doses (Vandenberg et al., Endocr. Rev., e-published ahead of printing on Mar. 14, 2012 as doi:10.1210/er.2011-1050). These results underscore the importance of examining the effects of a range of concentrations when using gene transcription analyses as a readout for the biological effect of EDCs. However, presence of inhibitory components or anti-estrogens cannot be ruled out and may warrant further investigation. In contrast, the GFP-GR and GFP-AR translocation assays were applicable to a wider range of concentrations including 100× doses. Thus, the translocation assay described herein is largely devoid of the non-monotonic dose-response effects observed by other detection methods, which makes it suitable for high-throughput screening.
SUMMARY
[0161] In conclusion, mammalian cell lines expressing GFP-tagged nuclear receptor constructs were utilized in an automated, highly reproducible, and low cost assay for detection of biologically active glucocorticoids and androgens in water sources. Using this high-throughput screening, combined with studies on transcriptional activation, glucocorticoid and androgen activities were discovered in water sources from 8 of 14 states in the US. This level of wide-spread contamination with steroids of both classes is a possible health hazard not only for the aquatic ecosystems, but may also negatively impact the human population. Largely unrestricted human activity with respect to many potential endocrine disruptors is of concern, and represents one of the main reasons for these wide-spread contaminations. Considering the largely unrestricted human activity in respect to many potential endocrine disruptors and limited methods (Roy & Pereira, Indian J Exp. Biol 43:975-992, 2005) for their detection in the environment, there is an urgent need of better assays for EDC screening. The results discussed here not only highlight the prevalence of contamination of water sources with glucocorticoid and androgen activities, but also introduce a novel approach for monitoring the quality of water. This approach can be readily extended to other nuclear receptors and applied to detection of various classes of EDCs in the environment.
EXAMPLE 2
Additional Traceable Fusion Proteins
[0162] This example provides additional traceable fusion proteins that can be used in the methods and kits described herein.
[0163] Using molecular cloning methods essentially similar to those employed in Example 1, the following additional traceable fusion proteins have been constructed: an aryl hydrocarbon (e.g., dioxin) sensitive fusion protein, pRevTRE-GFP-mAhR (SEQ ID NOs: 9/10); a mineralocorticoid (e.g., aldosterone) sensitive fusion protein, eGFP-ratMR (SEQ ID NOs: 11/12); and a progestin-sensitive fusion protein, eGFP-hPRB (SEQ ID NOs: 13/14). All three proteins were expressed in mammalian cells and exhibited the expected translocation in response to the corresponding ligand (dioxin, aldosterone, and progesterone--see Example 3) such as those described in Example 1.
EXAMPLE 3
Translocation in GFP-PR-B Construct-Expressing Cell Line Clones
[0164] This example demonstrates translocation response of four clonal lines expressing the traceable fusion protein GFP-PR-B upon exposure to defined amounts of progesterone.
[0165] Five mammalian clonal lines expressing the same traceable fusion protein, GFP-PR-B (eGFP-hPRB; SEQ ID NO: 14) were contacted with the indicated levels of progesterone, then assayed essentially as in Example 1 for translocation of the fusion protein into the nucleus from the cytoplasm. Four of the five clones tested (clones 8122, 8124, 8127, 8130) were positive for translocation in response to progesterone.
[0166] In view of the many possible embodiments to which the principles of the disclosed invention may be applied, it should be recognized that the illustrated embodiments are only preferred examples of the invention and should not be taken as limiting the scope of the invention. Rather, the scope of the invention is defined by the following claims. We therefore claim as our invention all that comes within the scope and spirit of these claims.
Sequence CWU
1
1
2813261DNAartificial sequenceeGFP-GR-ER310 encoding sequence 1atggtgagca
agggcgagga gctgttcacc ggggtggtgc ccatcctggt cgagctggac 60ggcgacgtaa
acggccacaa gttcagcgtg tccggcgagg gcgagggcga tgccacctac 120ggcaagctga
ccctgaagtt catctgcacc accggcaagc tgcccgtgcc ctggcccacc 180ctcgtgacca
ccctgaccta cggcgtgcag tgcttcagcc gctaccccga ccacatgaag 240cagcacgact
tcttcaagtc cgccatgccc gaaggctacg tccaggagcg caccatcttc 300ttcaaggacg
acggcaacta caagacccgc gccgaggtga agttcgaggg cgacaccctg 360gtgaaccgca
tcgagctgaa gggcatcgac ttcaaggagg acggcaacat cctggggcac 420aagctggagt
acaactacaa cagccacaac gtctatatca tggccgacaa gcagaagaac 480ggcatcaagg
tgaacttcaa gatccgccac aacatcgagg acggcagcgt gcagctcgcc 540gaccactacc
agcagaacac ccccatcggc gacggccccg tgctgctgcc cgacaaccac 600tacctgagca
cccagtccgc cctgagcaaa gaccccaacg agaagcgcga tcacatggtc 660ctgctggagt
tcgtgaccgc cgccgggatc actctcggca tggacgagct gtacaagggc 720gctggagcag
gggctggagc cggagctgac tccaaagaat cattaactcc tggtagagaa 780gaaaacccca
gcagtgtgct tgctcaggag aggggagatg tgatggactt ctataaaacc 840ctaagaggag
gagctactgt gaaggtttct gcgtcttcac cctcactggc tgtcgcttct 900caatcagact
ccaagcagcg aagacttttg gttgattttc caaaaggctc agtaagcaat 960gcgcagcagc
cagatctgtc caaagcagtt tcactctcaa tgggactgta tatgggagag 1020acagaaacaa
aagtgatggg aaatgacctg ggattcccac agcagggcca aatcagcctt 1080tcctcggggg
aaacagactt aaagcttttg gaagaaagca ttgcaaacct caataggtcg 1140accagtgttc
cagagaaccc caagagttca gcatccactg ctgtgtctgc tgcccccaca 1200gagaaggagt
ttccaaaaac tcactctgat gtatcttcag aacagcaaca tttgaagggc 1260cagactggca
ccaacggtgg caatgtgaaa ttgtatacca cagaccaaag cacctttgac 1320attttgcagg
atttggagtt ttcttctggg tccccaggta aagagacgaa tgagagtcct 1380tggagatcag
acctgttgat agatgaaaac tgtttgcttt ctcctctggc gggagaagac 1440gattcattcc
ttttggaagg aaactcgaat gaggactgca agcctctcat tttaccggac 1500actaaaccca
aaattaagga taatggagat ctggttttgt caagccccag taatgtaaca 1560ctgccccaag
tgaaaacaga aaaagaagat ttcatcgaac tctgcacccc tggggtaatt 1620aagcaagaga
aactgggcac agtttactgt caggcaagct ttcctggagc aaatataatt 1680ggtaataaaa
tgtctgccat ttctgttcat ggtgtgagta cctctggagg acagatgtac 1740cactatgaca
tgaatacagc atccctttct caacagcagg atcagaagcc tatttttaat 1800gtcattccac
caattcccgt tggttccgaa aattggaata ggtgccaagg atctggagat 1860gacaacttga
cttctctggg gactctgaac ttccctggtc gaacagtttt ttctaatggc 1920tattcaagcc
ccagcatgag accagatgta agctctcctc catccagctc ctcaacagca 1980acaacaggac
cacctcccaa actctgcctg gtgtgctctg atgaagcttc aggatgtcat 2040tatggagtct
taacttgtgg aagctgtaaa gttttcttca aaagagcagt ggaaggacag 2100cacaattacc
tatgtgctgg aaggaatgat tgcatcatcg ataaaattcg aagaaaaaac 2160tgcccagcat
gccgctatcg aaaatgtctt caggctggaa tgaacctgga agctcgaaaa 2220acaaagaaaa
aaataaaagg aattcagcag gccactacag gagtctcaca agaaacctct 2280gaaaatcctg
gtaacaaaac aatagttcct gcaacgttac cacaactcac ccctaccctg 2340gtgtcactgt
tggaggttat tgaacctgaa gtgttatatg caggatatga tagctctgtt 2400ctgacggccg
accagatggt cagtgccttg ttggatgctg agccccccat actctattcc 2460gagtatgatc
ctaccagacc cttcagtgaa gcttcgatga tgggcttact gaccaacctg 2520gcagacaggg
agctggttca catgatcaac tgggcgaaga gggtgccagg ctttgtggat 2580ttgaccctcc
atgatcaggt ccaccttcta gaatgtgcct ggctagagat cctgatgatt 2640ggtctcgtct
ggcgctccat ggagcaccca gggaagctac tgtttgctcc taacttgctc 2700ttggacagga
accagggaaa atgtgtagag ggcatggtgg agatcttcga catgctgctg 2760gctacatcat
ctcggttccg catgatgaat ctgcagggag aggagtttgt gtgcctcaaa 2820tctattattt
tgcttaattc tggagtgtac acatttctgt ccagcaccct gaagtctctg 2880gaagagaagg
accatatcca ccgagtcctg gacaagatca cagacacttt gatccacctg 2940atggccaagg
caggcctgac cctgcagcag cagcaccagc ggctggccca gctcctcctc 3000atcctctccc
acatcaggca catgagtaac aaaggcatgg agcatctgta cagcatgaag 3060tgcaagaacg
tggtgcccct ctatgacctg ctgctggaga tgctggacgc ccaccgccta 3120catgcgccca
ctagccgtgg aggggcatcc gtggaggaga cggaccaaag ccacttggcc 3180actgcgggct
ctacttcatc gcattccttg caaaagtatt acatcacggg ggaggcagag 3240ggtttccctg
ccacggtcta a
326121086PRTartificial sequenceeGFP-GR-ER310 traceable fusion protein
2Met Val Ser Lys Gly Glu Glu Leu Phe Thr Gly Val Val Pro Ile Leu 1
5 10 15 Val Glu Leu Asp
Gly Asp Val Asn Gly His Lys Phe Ser Val Ser Gly 20
25 30 Glu Gly Glu Gly Asp Ala Thr Tyr Gly
Lys Leu Thr Leu Lys Phe Ile 35 40
45 Cys Thr Thr Gly Lys Leu Pro Val Pro Trp Pro Thr Leu Val
Thr Thr 50 55 60
Leu Thr Tyr Gly Val Gln Cys Phe Ser Arg Tyr Pro Asp His Met Lys 65
70 75 80 Gln His Asp Phe Phe
Lys Ser Ala Met Pro Glu Gly Tyr Val Gln Glu 85
90 95 Arg Thr Ile Phe Phe Lys Asp Asp Gly Asn
Tyr Lys Thr Arg Ala Glu 100 105
110 Val Lys Phe Glu Gly Asp Thr Leu Val Asn Arg Ile Glu Leu Lys
Gly 115 120 125 Ile
Asp Phe Lys Glu Asp Gly Asn Ile Leu Gly His Lys Leu Glu Tyr 130
135 140 Asn Tyr Asn Ser His Asn
Val Tyr Ile Met Ala Asp Lys Gln Lys Asn 145 150
155 160 Gly Ile Lys Val Asn Phe Lys Ile Arg His Asn
Ile Glu Asp Gly Ser 165 170
175 Val Gln Leu Ala Asp His Tyr Gln Gln Asn Thr Pro Ile Gly Asp Gly
180 185 190 Pro Val
Leu Leu Pro Asp Asn His Tyr Leu Ser Thr Gln Ser Ala Leu 195
200 205 Ser Lys Asp Pro Asn Glu Lys
Arg Asp His Met Val Leu Leu Glu Phe 210 215
220 Val Thr Ala Ala Gly Ile Thr Leu Gly Met Asp Glu
Leu Tyr Lys Gly 225 230 235
240 Ala Gly Ala Gly Ala Gly Ala Gly Ala Asp Ser Lys Glu Ser Leu Thr
245 250 255 Pro Gly Arg
Glu Glu Asn Pro Ser Ser Val Leu Ala Gln Glu Arg Gly 260
265 270 Asp Val Met Asp Phe Tyr Lys Thr
Leu Arg Gly Gly Ala Thr Val Lys 275 280
285 Val Ser Ala Ser Ser Pro Ser Leu Ala Val Ala Ser Gln
Ser Asp Ser 290 295 300
Lys Gln Arg Arg Leu Leu Val Asp Phe Pro Lys Gly Ser Val Ser Asn 305
310 315 320 Ala Gln Gln Pro
Asp Leu Ser Lys Ala Val Ser Leu Ser Met Gly Leu 325
330 335 Tyr Met Gly Glu Thr Glu Thr Lys Val
Met Gly Asn Asp Leu Gly Phe 340 345
350 Pro Gln Gln Gly Gln Ile Ser Leu Ser Ser Gly Glu Thr Asp
Leu Lys 355 360 365
Leu Leu Glu Glu Ser Ile Ala Asn Leu Asn Arg Ser Thr Ser Val Pro 370
375 380 Glu Asn Pro Lys Ser
Ser Ala Ser Thr Ala Val Ser Ala Ala Pro Thr 385 390
395 400 Glu Lys Glu Phe Pro Lys Thr His Ser Asp
Val Ser Ser Glu Gln Gln 405 410
415 His Leu Lys Gly Gln Thr Gly Thr Asn Gly Gly Asn Val Lys Leu
Tyr 420 425 430 Thr
Thr Asp Gln Ser Thr Phe Asp Ile Leu Gln Asp Leu Glu Phe Ser 435
440 445 Ser Gly Ser Pro Gly Lys
Glu Thr Asn Glu Ser Pro Trp Arg Ser Asp 450 455
460 Leu Leu Ile Asp Glu Asn Cys Leu Leu Ser Pro
Leu Ala Gly Glu Asp 465 470 475
480 Asp Ser Phe Leu Leu Glu Gly Asn Ser Asn Glu Asp Cys Lys Pro Leu
485 490 495 Ile Leu
Pro Asp Thr Lys Pro Lys Ile Lys Asp Asn Gly Asp Leu Val 500
505 510 Leu Ser Ser Pro Ser Asn Val
Thr Leu Pro Gln Val Lys Thr Glu Lys 515 520
525 Glu Asp Phe Ile Glu Leu Cys Thr Pro Gly Val Ile
Lys Gln Glu Lys 530 535 540
Leu Gly Thr Val Tyr Cys Gln Ala Ser Phe Pro Gly Ala Asn Ile Ile 545
550 555 560 Gly Asn Lys
Met Ser Ala Ile Ser Val His Gly Val Ser Thr Ser Gly 565
570 575 Gly Gln Met Tyr His Tyr Asp Met
Asn Thr Ala Ser Leu Ser Gln Gln 580 585
590 Gln Asp Gln Lys Pro Ile Phe Asn Val Ile Pro Pro Ile
Pro Val Gly 595 600 605
Ser Glu Asn Trp Asn Arg Cys Gln Gly Ser Gly Asp Asp Asn Leu Thr 610
615 620 Ser Leu Gly Thr
Leu Asn Phe Pro Gly Arg Thr Val Phe Ser Asn Gly 625 630
635 640 Tyr Ser Ser Pro Ser Met Arg Pro Asp
Val Ser Ser Pro Pro Ser Ser 645 650
655 Ser Ser Thr Ala Thr Thr Gly Pro Pro Pro Lys Leu Cys Leu
Val Cys 660 665 670
Ser Asp Glu Ala Ser Gly Cys His Tyr Gly Val Leu Thr Cys Gly Ser
675 680 685 Cys Lys Val Phe
Phe Lys Arg Ala Val Glu Gly Gln His Asn Tyr Leu 690
695 700 Cys Ala Gly Arg Asn Asp Cys Ile
Ile Asp Lys Ile Arg Arg Lys Asn 705 710
715 720 Cys Pro Ala Cys Arg Tyr Arg Lys Cys Leu Gln Ala
Gly Met Asn Leu 725 730
735 Glu Ala Arg Lys Thr Lys Lys Lys Ile Lys Gly Ile Gln Gln Ala Thr
740 745 750 Thr Gly Val
Ser Gln Glu Thr Ser Glu Asn Pro Gly Asn Lys Thr Ile 755
760 765 Val Pro Ala Thr Leu Pro Gln Leu
Thr Pro Thr Leu Val Ser Leu Leu 770 775
780 Glu Val Ile Glu Pro Glu Val Leu Tyr Ala Gly Tyr Asp
Ser Ser Val 785 790 795
800 Leu Thr Ala Asp Gln Met Val Ser Ala Leu Leu Asp Ala Glu Pro Pro
805 810 815 Ile Leu Tyr Ser
Glu Tyr Asp Pro Thr Arg Pro Phe Ser Glu Ala Ser 820
825 830 Met Met Gly Leu Leu Thr Asn Leu Ala
Asp Arg Glu Leu Val His Met 835 840
845 Ile Asn Trp Ala Lys Arg Val Pro Gly Phe Val Asp Leu Thr
Leu His 850 855 860
Asp Gln Val His Leu Leu Glu Cys Ala Trp Leu Glu Ile Leu Met Ile 865
870 875 880 Gly Leu Val Trp Arg
Ser Met Glu His Pro Gly Lys Leu Leu Phe Ala 885
890 895 Pro Asn Leu Leu Leu Asp Arg Asn Gln Gly
Lys Cys Val Glu Gly Met 900 905
910 Val Glu Ile Phe Asp Met Leu Leu Ala Thr Ser Ser Arg Phe Arg
Met 915 920 925 Met
Asn Leu Gln Gly Glu Glu Phe Val Cys Leu Lys Ser Ile Ile Leu 930
935 940 Leu Asn Ser Gly Val Tyr
Thr Phe Leu Ser Ser Thr Leu Lys Ser Leu 945 950
955 960 Glu Glu Lys Asp His Ile His Arg Val Leu Asp
Lys Ile Thr Asp Thr 965 970
975 Leu Ile His Leu Met Ala Lys Ala Gly Leu Thr Leu Gln Gln Gln His
980 985 990 Gln Arg
Leu Ala Gln Leu Leu Leu Ile Leu Ser His Ile Arg His Met 995
1000 1005 Ser Asn Lys Gly Met
Glu His Leu Tyr Ser Met Lys Cys Lys Asn 1010 1015
1020 Val Val Pro Leu Tyr Asp Leu Leu Leu Glu
Met Leu Asp Ala His 1025 1030 1035
Arg Leu His Ala Pro Thr Ser Arg Gly Gly Ala Ser Val Glu Glu
1040 1045 1050 Thr Asp
Gln Ser His Leu Ala Thr Ala Gly Ser Thr Ser Ser His 1055
1060 1065 Ser Leu Gln Lys Tyr Tyr Ile
Thr Gly Glu Ala Glu Gly Phe Pro 1070 1075
1080 Ala Thr Val 1085 33141DNAartificial
sequenceeGFP-GR-TR216 encoding sequence 3atggtgagca agggcgagga gctgttcacc
ggggtggtgc ccatcctggt cgagctggac 60ggcgacgtaa acggccacaa gttcagcgtg
tccggcgagg gcgagggcga tgccacctac 120ggcaagctga ccctgaagtt catctgcacc
accggcaagc tgcccgtgcc ctggcccacc 180ctcgtgacca ccctgaccta cggcgtgcag
tgcttcagcc gctaccccga ccacatgaag 240cagcacgact tcttcaagtc cgccatgccc
gaaggctacg tccaggagcg caccatcttc 300ttcaaggacg acggcaacta caagacccgc
gccgaggtga agttcgaggg cgacaccctg 360gtgaaccgca tcgagctgaa gggcatcgac
ttcaaggagg acggcaacat cctggggcac 420aagctggagt acaactacaa cagccacaac
gtctatatca tggccgacaa gcagaagaac 480ggcatcaagg tgaacttcaa gatccgccac
aacatcgagg acggcagcgt gcagctcgcc 540gaccactacc agcagaacac ccccatcggc
gacggccccg tgctgctgcc cgacaaccac 600tacctgagca cccagtccgc cctgagcaaa
gaccccaacg agaagcgcga tcacatggtc 660ctgctggagt tcgtgaccgc cgccgggatc
actctcggca tggacgagct gtacaagggc 720gctggagcag gggctggagc cggagctgac
tccaaagaat cattaactcc tggtagagaa 780gaaaacccca gcagtgtgct tgctcaggag
aggggagatg tgatggactt ctataaaacc 840ctaagaggag gagctactgt gaaggtttct
gcgtcttcac cctcactggc tgtcgcttct 900caatcagact ccaagcagcg aagacttttg
gttgattttc caaaaggctc agtaagcaat 960gcgcagcagc cagatctgtc caaagcagtt
tcactctcaa tgggactgta tatgggagag 1020acagaaacaa aagtgatggg aaatgacctg
ggattcccac agcagggcca aatcagcctt 1080tcctcggggg aaacagactt aaagcttttg
gaagaaagca ttgcaaacct caataggtcg 1140accagtgttc cagagaaccc caagagttca
gcatccactg ctgtgtctgc tgcccccaca 1200gagaaggagt ttccaaaaac tcactctgat
gtatcttcag aacagcaaca tttgaagggc 1260cagactggca ccaacggtgg caatgtgaaa
ttgtatacca cagaccaaag cacctttgac 1320attttgcagg atttggagtt ttcttctggg
tccccaggta aagagacgaa tgagagtcct 1380tggagatcag acctgttgat agatgaaaac
tgtttgcttt ctcctctggc gggagaagac 1440gattcattcc ttttggaagg aaactcgaat
gaggactgca agcctctcat tttaccggac 1500actaaaccca aaattaagga taatggagat
ctggttttgt caagccccag taatgtaaca 1560ctgccccaag tgaaaacaga aaaagaagat
ttcatcgaac tctgcacccc tggggtaatt 1620aagcaagaga aactgggcac agtttactgt
caggcaagct ttcctggagc aaatataatt 1680ggtaataaaa tgtctgccat ttctgttcat
ggtgtgagta cctctggagg acagatgtac 1740cactatgaca tgaatacagc atccctttct
caacagcagg atcagaagcc tatttttaat 1800gtcattccac caattcccgt tggttccgaa
aattggaata ggtgccaagg atctggagat 1860gacaacttga cttctctggg gactctgaac
ttccctggtc gaacagtttt ttctaatggc 1920tattcaagcc ccagcatgag accagatgta
agctctcctc catccagctc ctcaacagca 1980acaacaggac cacctcccaa actctgcctg
gtgtgctctg atgaagcttc aggatgtcat 2040tatggagtct taacttgtgg aagctgtaaa
gttttcttca aaagagcagt ggaaggacag 2100cacaattacc tatgtgctgg aaggaatgat
tgcatcatcg ataaaattcg aagaaaaaac 2160tgcccagcat gccgctatcg aaaatgtctt
caggctggaa tgaacctgga agctcgaaaa 2220acaaagaaaa aaataaaagg aattcagcag
gccactacag gagtctcaca agaaacctct 2280gaaaatcctg gtaacaaaac aatagttcct
gcaacgttac cacaactcac ccctaccctg 2340gtgtcactgt tggaggttat tgaacctgaa
gtgttatatg caggatatga tagctctgtt 2400gacgaggaat gggagctcat caaaactgtc
accgaagccc atgtggcgac caacgcccaa 2460ggcagccact ggaagcaaaa acggaaattc
ctgccagaag acattggaca agcaccaata 2520gtcaatgccc cagaaggtgg aaaggttgac
ttggaagcct tcagccattt tacaaaaatc 2580atcacaccag caattaccag agtggtggat
tttgccaaaa agttgcctat gttttgtgag 2640ctgccatgtg aagaccagat catcctcctc
aaaggctgct gcatggagat catgtccctt 2700cgcgctgctg tgcgctatga cccagaaagt
gagactttaa ccttgaatgg ggaaatggca 2760gtgacacggg gccagctgaa aaatgggggt
cttggggtgg tgtcagacgc catctttgac 2820ctgggcatgt ctctgtcttc tttcaacctg
gatgacactg aagtagccct ccttcaggcc 2880gtcctgctga tgtcttcaga tcgcccgggg
cttgcctgtg ttgagagaat agaaaagtac 2940caagatagtt tcctgctggc ctttgaacac
tatatcaatt accgaaaaca ccacgtgaca 3000cacttttggc caaaactcct gatgaaggtg
acagatctgc ggatgatagg agcctgccat 3060gccagccgct tcctgcacat gaaggtggaa
tgccccacag aactcttccc ccctttgttc 3120ttggaagtgt tcgaggatta a
314141046PRTartificial
sequenceeGFP-GR-TR216 traceable fusion protein 4Met Val Ser Lys Gly Glu
Glu Leu Phe Thr Gly Val Val Pro Ile Leu 1 5
10 15 Val Glu Leu Asp Gly Asp Val Asn Gly His Lys
Phe Ser Val Ser Gly 20 25
30 Glu Gly Glu Gly Asp Ala Thr Tyr Gly Lys Leu Thr Leu Lys Phe
Ile 35 40 45 Cys
Thr Thr Gly Lys Leu Pro Val Pro Trp Pro Thr Leu Val Thr Thr 50
55 60 Leu Thr Tyr Gly Val Gln
Cys Phe Ser Arg Tyr Pro Asp His Met Lys 65 70
75 80 Gln His Asp Phe Phe Lys Ser Ala Met Pro Glu
Gly Tyr Val Gln Glu 85 90
95 Arg Thr Ile Phe Phe Lys Asp Asp Gly Asn Tyr Lys Thr Arg Ala Glu
100 105 110 Val Lys
Phe Glu Gly Asp Thr Leu Val Asn Arg Ile Glu Leu Lys Gly 115
120 125 Ile Asp Phe Lys Glu Asp Gly
Asn Ile Leu Gly His Lys Leu Glu Tyr 130 135
140 Asn Tyr Asn Ser His Asn Val Tyr Ile Met Ala Asp
Lys Gln Lys Asn 145 150 155
160 Gly Ile Lys Val Asn Phe Lys Ile Arg His Asn Ile Glu Asp Gly Ser
165 170 175 Val Gln Leu
Ala Asp His Tyr Gln Gln Asn Thr Pro Ile Gly Asp Gly 180
185 190 Pro Val Leu Leu Pro Asp Asn His
Tyr Leu Ser Thr Gln Ser Ala Leu 195 200
205 Ser Lys Asp Pro Asn Glu Lys Arg Asp His Met Val Leu
Leu Glu Phe 210 215 220
Val Thr Ala Ala Gly Ile Thr Leu Gly Met Asp Glu Leu Tyr Lys Gly 225
230 235 240 Ala Gly Ala Gly
Ala Gly Ala Gly Ala Asp Ser Lys Glu Ser Leu Thr 245
250 255 Pro Gly Arg Glu Glu Asn Pro Ser Ser
Val Leu Ala Gln Glu Arg Gly 260 265
270 Asp Val Met Asp Phe Tyr Lys Thr Leu Arg Gly Gly Ala Thr
Val Lys 275 280 285
Val Ser Ala Ser Ser Pro Ser Leu Ala Val Ala Ser Gln Ser Asp Ser 290
295 300 Lys Gln Arg Arg Leu
Leu Val Asp Phe Pro Lys Gly Ser Val Ser Asn 305 310
315 320 Ala Gln Gln Pro Asp Leu Ser Lys Ala Val
Ser Leu Ser Met Gly Leu 325 330
335 Tyr Met Gly Glu Thr Glu Thr Lys Val Met Gly Asn Asp Leu Gly
Phe 340 345 350 Pro
Gln Gln Gly Gln Ile Ser Leu Ser Ser Gly Glu Thr Asp Leu Lys 355
360 365 Leu Leu Glu Glu Ser Ile
Ala Asn Leu Asn Arg Ser Thr Ser Val Pro 370 375
380 Glu Asn Pro Lys Ser Ser Ala Ser Thr Ala Val
Ser Ala Ala Pro Thr 385 390 395
400 Glu Lys Glu Phe Pro Lys Thr His Ser Asp Val Ser Ser Glu Gln Gln
405 410 415 His Leu
Lys Gly Gln Thr Gly Thr Asn Gly Gly Asn Val Lys Leu Tyr 420
425 430 Thr Thr Asp Gln Ser Thr Phe
Asp Ile Leu Gln Asp Leu Glu Phe Ser 435 440
445 Ser Gly Ser Pro Gly Lys Glu Thr Asn Glu Ser Pro
Trp Arg Ser Asp 450 455 460
Leu Leu Ile Asp Glu Asn Cys Leu Leu Ser Pro Leu Ala Gly Glu Asp 465
470 475 480 Asp Ser Phe
Leu Leu Glu Gly Asn Ser Asn Glu Asp Cys Lys Pro Leu 485
490 495 Ile Leu Pro Asp Thr Lys Pro Lys
Ile Lys Asp Asn Gly Asp Leu Val 500 505
510 Leu Ser Ser Pro Ser Asn Val Thr Leu Pro Gln Val Lys
Thr Glu Lys 515 520 525
Glu Asp Phe Ile Glu Leu Cys Thr Pro Gly Val Ile Lys Gln Glu Lys 530
535 540 Leu Gly Thr Val
Tyr Cys Gln Ala Ser Phe Pro Gly Ala Asn Ile Ile 545 550
555 560 Gly Asn Lys Met Ser Ala Ile Ser Val
His Gly Val Ser Thr Ser Gly 565 570
575 Gly Gln Met Tyr His Tyr Asp Met Asn Thr Ala Ser Leu Ser
Gln Gln 580 585 590
Gln Asp Gln Lys Pro Ile Phe Asn Val Ile Pro Pro Ile Pro Val Gly
595 600 605 Ser Glu Asn Trp
Asn Arg Cys Gln Gly Ser Gly Asp Asp Asn Leu Thr 610
615 620 Ser Leu Gly Thr Leu Asn Phe Pro
Gly Arg Thr Val Phe Ser Asn Gly 625 630
635 640 Tyr Ser Ser Pro Ser Met Arg Pro Asp Val Ser Ser
Pro Pro Ser Ser 645 650
655 Ser Ser Thr Ala Thr Thr Gly Pro Pro Pro Lys Leu Cys Leu Val Cys
660 665 670 Ser Asp Glu
Ala Ser Gly Cys His Tyr Gly Val Leu Thr Cys Gly Ser 675
680 685 Cys Lys Val Phe Phe Lys Arg Ala
Val Glu Gly Gln His Asn Tyr Leu 690 695
700 Cys Ala Gly Arg Asn Asp Cys Ile Ile Asp Lys Ile Arg
Arg Lys Asn 705 710 715
720 Cys Pro Ala Cys Arg Tyr Arg Lys Cys Leu Gln Ala Gly Met Asn Leu
725 730 735 Glu Ala Arg Lys
Thr Lys Lys Lys Ile Lys Gly Ile Gln Gln Ala Thr 740
745 750 Thr Gly Val Ser Gln Glu Thr Ser Glu
Asn Pro Gly Asn Lys Thr Ile 755 760
765 Val Pro Ala Thr Leu Pro Gln Leu Thr Pro Thr Leu Val Ser
Leu Leu 770 775 780
Glu Val Ile Glu Pro Glu Val Leu Tyr Ala Gly Tyr Asp Ser Ser Val 785
790 795 800 Asp Glu Glu Trp Glu
Leu Ile Lys Thr Val Thr Glu Ala His Val Ala 805
810 815 Thr Asn Ala Gln Gly Ser His Trp Lys Gln
Lys Arg Lys Phe Leu Pro 820 825
830 Glu Asp Ile Gly Gln Ala Pro Ile Val Asn Ala Pro Glu Gly Gly
Lys 835 840 845 Val
Asp Leu Glu Ala Phe Ser His Phe Thr Lys Ile Ile Thr Pro Ala 850
855 860 Ile Thr Arg Val Val Asp
Phe Ala Lys Lys Leu Pro Met Phe Cys Glu 865 870
875 880 Leu Pro Cys Glu Asp Gln Ile Ile Leu Leu Lys
Gly Cys Cys Met Glu 885 890
895 Ile Met Ser Leu Arg Ala Ala Val Arg Tyr Asp Pro Glu Ser Glu Thr
900 905 910 Leu Thr
Leu Asn Gly Glu Met Ala Val Thr Arg Gly Gln Leu Lys Asn 915
920 925 Gly Gly Leu Gly Val Val Ser
Asp Ala Ile Phe Asp Leu Gly Met Ser 930 935
940 Leu Ser Ser Phe Asn Leu Asp Asp Thr Glu Val Ala
Leu Leu Gln Ala 945 950 955
960 Val Leu Leu Met Ser Ser Asp Arg Pro Gly Leu Ala Cys Val Glu Arg
965 970 975 Ile Glu Lys
Tyr Gln Asp Ser Phe Leu Leu Ala Phe Glu His Tyr Ile 980
985 990 Asn Tyr Arg Lys His His Val Thr
His Phe Trp Pro Lys Leu Leu Met 995 1000
1005 Lys Val Thr Asp Leu Arg Met Ile Gly Ala Cys
His Ala Ser Arg 1010 1015 1020
Phe Leu His Met Lys Val Glu Cys Pro Thr Glu Leu Phe Pro Pro
1025 1030 1035 Leu Phe Leu
Glu Val Phe Glu Asp 1040 1045 53213DNAartificial
sequencepCI-nGFP-C656G encoding sequence 5atggcccacc atcaccacca
tcacggatat ccatacgacg tgccagatta cgctcagtcg 60agtgccatga gtaaaggaga
agaacttttc actggagttg tcccaattct tgttgaatta 120gatggtgatg ttaatgggca
caaattttct gtcagtggag agggtgaagg tgatgcaaca 180tacggaaaac ttacccttaa
atttatttgc actactggaa aactacctgt tccttggcca 240acacttgtca ctactttcac
ttatggtgtt caatgctttt caagataccc agatcatatg 300aaacagcatg actttttcaa
gagtgccatg cccgaaggtt atgtacagga aagaactata 360tttttcaaag atgacgggaa
ctacaagaca cgtgctgaag tcaagtttga aggtgatacc 420cttgttaata gaatcgagtt
aaaaggtatt gattttaaag aagatggaaa cattcttgga 480cacaaattgg aatacaacta
taactcacac aatgtataca tcatggcaga caaacaaaag 540aatggaatca aagttaactt
caaaattaga cacaacattg aagatggaag cgttcaacta 600gcagaccatt atcaacaaaa
tactccaatt ggcgatggcc ctgtcctttt accagacaac 660cattacctgt ccacacaatc
tgccctttcg aaagatccca acgaaaagag agaccacatg 720gtccttcttg agtttgtaac
agctgctggg attacacatg gcatggatga actatacaaa 780ggcgccggcg ctggtgctgg
tgctggcgcc atcagcgcgc tgatcctgga ctccaaagaa 840tccttagctc cccctggtag
agacgaagtc cctggcagtt tgcttggcca ggggaggggg 900agcgtaatgg acttttataa
aagcctgagg ggaggagcta cagtcaaggt ttctgcatct 960tcgccctcag tggctgctgc
ttctcaggca gattccaagc agcagaggat tctccttgat 1020ttctcgaaag gctccacaag
caatgtgcag cagcgacagc agcagcagca gcagcagcag 1080cagcagcagc agcagcagca
gcagcagcag cagccaggct tatccaaagc cgtttcactg 1140tccatggggc tgtatatggg
agagacagaa acaaaagtga tggggaatga cttgggctac 1200ccacagcagg gccaacttgg
cctttcctct ggggaaacag actttcggct tctggaagaa 1260agcattgcaa acctcaatag
gtcgaccagc gttccagaga accccaagag ttcaacgtct 1320gcaactgggt gtgctacccc
gacagagaag gagtttccca aaactcactc ggatgcatct 1380tcagaacagc aaaatcgaaa
aagccagacc ggcaccaacg gaggcagtgt gaaattgtat 1440cccacagacc aaagcacctt
tgacctcttg aaggatttgg agttttccgc tgggtcccca 1500agtaaagaca caaacgagag
tccctggaga tcagatctgt tgatagatga aaacttgctt 1560tctcctttgg cgggagaaga
tgatccattc cttctcgaag ggaacacgaa tgaggattgt 1620aagcctctta ttttaccgga
cactaaacct aaaattaagg atactggaga tacaatctta 1680tcaagtccca gcagtgtggc
actaccccaa gtgaaaacag aaaaagatga tttcattgaa 1740ctttgcaccc ccggggtaat
taagcaagag aaactgggcc cagtttattg tcaggcaagc 1800ttttctggga caaatataat
tggtaataaa atgtctgcca tttctgttca tggtgtgagt 1860acctctggag gacagatgta
ccactatgac atgaatacag catccctttc tcagcagcag 1920gatcagaagc ctgtttttaa
tgtcattcca ccaattcctg ttggttctga aaactggaat 1980aggtgccaag gctccggaga
ggacagcctg acttccttgg gggctctgaa cttcccaggc 2040cggtcagtgt tttctaatgg
gtactcaagc cctggaatga gaccagatgt aagctctcct 2100ccatccagct cgtcagcagc
cacgggacca cctcccaagc tctgcctggt gtgctccgat 2160gaagcttcag gatgtcatta
cggggtgctg acatgtggaa gctgcaaagt attctttaaa 2220agagcagtgg aaggacagca
caattacctt tgtgctggaa gaaacgattg catcattgat 2280aaaattcgaa ggaaaaactg
cccagcatgc cgctatcgga aatgtcttca ggctggaatg 2340aaccttgaag ctcgaaaaac
aaagaaaaaa atcaaaggga ttcagcaagc cactgcagga 2400gtctcacaag acacttcgga
aaatcctaac aaaacaatag ttcctgcagc attaccacag 2460ctcaccccta ccttggtgtc
actgctggag gtgattgaac ccgaggtgtt gtatgcagga 2520tatgatagct ctgttccaga
ttcagcatgg agaattatga ccacactcaa catgttaggt 2580gggcgtcaag tgattgcagc
agtgaaatgg gcaaaggcga tactaggctt gagaaactta 2640cacctcgatg accaaatgac
cctgctacag tactcatgga tgtttctcat ggcatttgcc 2700ttgggttgga gatcatacag
acaatcaagc ggaaacctgc tctgctttgc tcctgatctg 2760attattaatg agcagagaat
gtctctaccc ggcatgtatg accaatgtaa acacatgctg 2820tttgtctcct ctgaattaca
aagattgcag gtatcctatg aagagtatct ctgtatgaaa 2880accttactgc ttctctcctc
agttcctaag gaaggtctga agagccaaga gttatttgat 2940gagattcgaa tgacttatat
caaagagcta ggaaaagcca tcgtcaaaag ggaagggaac 3000tccagtcaga actggcaacg
gttttaccaa ctgacaaagc ttctggactc catgcatgag 3060gtggttgaga atctccttac
ctactgcttc cagacatttt tggataagac catgagtatt 3120gaattcccag agatgttagc
tgaaatcatc actaatcaga taccaaaata ttcaaatgga 3180aatatcaaaa agcttctgtt
tcatcaaaaa tga 321361070PRTartificial
sequencepCI-nGFP-C656G traceable fusion protein 6Met Ala His His His His
His His Gly Tyr Pro Tyr Asp Val Pro Asp 1 5
10 15 Tyr Ala Gln Ser Ser Ala Met Ser Lys Gly Glu
Glu Leu Phe Thr Gly 20 25
30 Val Val Pro Ile Leu Val Glu Leu Asp Gly Asp Val Asn Gly His
Lys 35 40 45 Phe
Ser Val Ser Gly Glu Gly Glu Gly Asp Ala Thr Tyr Gly Lys Leu 50
55 60 Thr Leu Lys Phe Ile Cys
Thr Thr Gly Lys Leu Pro Val Pro Trp Pro 65 70
75 80 Thr Leu Val Thr Thr Phe Thr Tyr Gly Val Gln
Cys Phe Ser Arg Tyr 85 90
95 Pro Asp His Met Lys Gln His Asp Phe Phe Lys Ser Ala Met Pro Glu
100 105 110 Gly Tyr
Val Gln Glu Arg Thr Ile Phe Phe Lys Asp Asp Gly Asn Tyr 115
120 125 Lys Thr Arg Ala Glu Val Lys
Phe Glu Gly Asp Thr Leu Val Asn Arg 130 135
140 Ile Glu Leu Lys Gly Ile Asp Phe Lys Glu Asp Gly
Asn Ile Leu Gly 145 150 155
160 His Lys Leu Glu Tyr Asn Tyr Asn Ser His Asn Val Tyr Ile Met Ala
165 170 175 Asp Lys Gln
Lys Asn Gly Ile Lys Val Asn Phe Lys Ile Arg His Asn 180
185 190 Ile Glu Asp Gly Ser Val Gln Leu
Ala Asp His Tyr Gln Gln Asn Thr 195 200
205 Pro Ile Gly Asp Gly Pro Val Leu Leu Pro Asp Asn His
Tyr Leu Ser 210 215 220
Thr Gln Ser Ala Leu Ser Lys Asp Pro Asn Glu Lys Arg Asp His Met 225
230 235 240 Val Leu Leu Glu
Phe Val Thr Ala Ala Gly Ile Thr His Gly Met Asp 245
250 255 Glu Leu Tyr Lys Gly Ala Gly Ala Gly
Ala Gly Ala Gly Ala Ile Ser 260 265
270 Ala Leu Ile Leu Asp Ser Lys Glu Ser Leu Ala Pro Pro Gly
Arg Asp 275 280 285
Glu Val Pro Gly Ser Leu Leu Gly Gln Gly Arg Gly Ser Val Met Asp 290
295 300 Phe Tyr Lys Ser Leu
Arg Gly Gly Ala Thr Val Lys Val Ser Ala Ser 305 310
315 320 Ser Pro Ser Val Ala Ala Ala Ser Gln Ala
Asp Ser Lys Gln Gln Arg 325 330
335 Ile Leu Leu Asp Phe Ser Lys Gly Ser Thr Ser Asn Val Gln Gln
Arg 340 345 350 Gln
Gln Gln Gln Gln Gln Gln Gln Gln Gln Gln Gln Gln Gln Gln Gln 355
360 365 Gln Gln Gln Pro Gly Leu
Ser Lys Ala Val Ser Leu Ser Met Gly Leu 370 375
380 Tyr Met Gly Glu Thr Glu Thr Lys Val Met Gly
Asn Asp Leu Gly Tyr 385 390 395
400 Pro Gln Gln Gly Gln Leu Gly Leu Ser Ser Gly Glu Thr Asp Phe Arg
405 410 415 Leu Leu
Glu Glu Ser Ile Ala Asn Leu Asn Arg Ser Thr Ser Val Pro 420
425 430 Glu Asn Pro Lys Ser Ser Thr
Ser Ala Thr Gly Cys Ala Thr Pro Thr 435 440
445 Glu Lys Glu Phe Pro Lys Thr His Ser Asp Ala Ser
Ser Glu Gln Gln 450 455 460
Asn Arg Lys Ser Gln Thr Gly Thr Asn Gly Gly Ser Val Lys Leu Tyr 465
470 475 480 Pro Thr Asp
Gln Ser Thr Phe Asp Leu Leu Lys Asp Leu Glu Phe Ser 485
490 495 Ala Gly Ser Pro Ser Lys Asp Thr
Asn Glu Ser Pro Trp Arg Ser Asp 500 505
510 Leu Leu Ile Asp Glu Asn Leu Leu Ser Pro Leu Ala Gly
Glu Asp Asp 515 520 525
Pro Phe Leu Leu Glu Gly Asn Thr Asn Glu Asp Cys Lys Pro Leu Ile 530
535 540 Leu Pro Asp Thr
Lys Pro Lys Ile Lys Asp Thr Gly Asp Thr Ile Leu 545 550
555 560 Ser Ser Pro Ser Ser Val Ala Leu Pro
Gln Val Lys Thr Glu Lys Asp 565 570
575 Asp Phe Ile Glu Leu Cys Thr Pro Gly Val Ile Lys Gln Glu
Lys Leu 580 585 590
Gly Pro Val Tyr Cys Gln Ala Ser Phe Ser Gly Thr Asn Ile Ile Gly
595 600 605 Asn Lys Met Ser
Ala Ile Ser Val His Gly Val Ser Thr Ser Gly Gly 610
615 620 Gln Met Tyr His Tyr Asp Met Asn
Thr Ala Ser Leu Ser Gln Gln Gln 625 630
635 640 Asp Gln Lys Pro Val Phe Asn Val Ile Pro Pro Ile
Pro Val Gly Ser 645 650
655 Glu Asn Trp Asn Arg Cys Gln Gly Ser Gly Glu Asp Ser Leu Thr Ser
660 665 670 Leu Gly Ala
Leu Asn Phe Pro Gly Arg Ser Val Phe Ser Asn Gly Tyr 675
680 685 Ser Ser Pro Gly Met Arg Pro Asp
Val Ser Ser Pro Pro Ser Ser Ser 690 695
700 Ser Ala Ala Thr Gly Pro Pro Pro Lys Leu Cys Leu Val
Cys Ser Asp 705 710 715
720 Glu Ala Ser Gly Cys His Tyr Gly Val Leu Thr Cys Gly Ser Cys Lys
725 730 735 Val Phe Phe Lys
Arg Ala Val Glu Gly Gln His Asn Tyr Leu Cys Ala 740
745 750 Gly Arg Asn Asp Cys Ile Ile Asp Lys
Ile Arg Arg Lys Asn Cys Pro 755 760
765 Ala Cys Arg Tyr Arg Lys Cys Leu Gln Ala Gly Met Asn Leu
Glu Ala 770 775 780
Arg Lys Thr Lys Lys Lys Ile Lys Gly Ile Gln Gln Ala Thr Ala Gly 785
790 795 800 Val Ser Gln Asp Thr
Ser Glu Asn Pro Asn Lys Thr Ile Val Pro Ala 805
810 815 Ala Leu Pro Gln Leu Thr Pro Thr Leu Val
Ser Leu Leu Glu Val Ile 820 825
830 Glu Pro Glu Val Leu Tyr Ala Gly Tyr Asp Ser Ser Val Pro Asp
Ser 835 840 845 Ala
Trp Arg Ile Met Thr Thr Leu Asn Met Leu Gly Gly Arg Gln Val 850
855 860 Ile Ala Ala Val Lys Trp
Ala Lys Ala Ile Leu Gly Leu Arg Asn Leu 865 870
875 880 His Leu Asp Asp Gln Met Thr Leu Leu Gln Tyr
Ser Trp Met Phe Leu 885 890
895 Met Ala Phe Ala Leu Gly Trp Arg Ser Tyr Arg Gln Ser Ser Gly Asn
900 905 910 Leu Leu
Cys Phe Ala Pro Asp Leu Ile Ile Asn Glu Gln Arg Met Ser 915
920 925 Leu Pro Gly Met Tyr Asp Gln
Cys Lys His Met Leu Phe Val Ser Ser 930 935
940 Glu Leu Gln Arg Leu Gln Val Ser Tyr Glu Glu Tyr
Leu Cys Met Lys 945 950 955
960 Thr Leu Leu Leu Leu Ser Ser Val Pro Lys Glu Gly Leu Lys Ser Gln
965 970 975 Glu Leu Phe
Asp Glu Ile Arg Met Thr Tyr Ile Lys Glu Leu Gly Lys 980
985 990 Ala Ile Val Lys Arg Glu Gly Asn
Ser Ser Gln Asn Trp Gln Arg Phe 995 1000
1005 Tyr Gln Leu Thr Lys Leu Leu Asp Ser Met His
Glu Val Val Glu 1010 1015 1020
Asn Leu Leu Thr Tyr Cys Phe Gln Thr Phe Leu Asp Lys Thr Met
1025 1030 1035 Ser Ile Glu
Phe Pro Glu Met Leu Ala Glu Ile Ile Thr Asn Gln 1040
1045 1050 Ile Pro Lys Tyr Ser Asn Gly Asn
Ile Lys Lys Leu Leu Phe His 1055 1060
1065 Gln Lys 1070 73516DNAartificial sequenceeGFP-hAR
encoding sequence 7atggtgagca agggcgagga gctgttcacc ggggtggtgc ccatcctggt
cgagctggac 60ggcgacgtaa acggccacaa gttcagcgtg tccggcgagg gcgagggcga
tgccacctac 120ggcaagctga ccctgaagtt catctgcacc accggcaagc tgcccgtgcc
ctggcccacc 180ctcgtgacca ccctgaccta cggcgtgcag tgcttcagcc gctaccccga
ccacatgaag 240cagcacgact tcttcaagtc cgccatgccc gaaggctacg tccaggagcg
caccatcttc 300ttcaaggacg acggcaacta caagacccgc gccgaggtga agttcgaggg
cgacaccctg 360gtgaaccgca tcgagctgaa gggcatcgac ttcaaggagg acggcaacat
cctggggcac 420aagctggagt acaactacaa cagccacaac gtctatatca tggccgacaa
gcagaagaac 480ggcatcaagg tgaacttcaa gatccgccac aacatcgagg acggcagcgt
gcagctcgcc 540gaccactacc agcagaacac ccccatcggc gacggccccg tgctgctgcc
cgacaaccac 600tacctgagca cccagtccgc cctgagcaaa gaccccaacg agaagcgcga
tcacatggtc 660ctgctggagt tcgtgaccgc cgccgggatc actctcggca tggacgagct
gtacaagtcc 720ggactcagat ctcgagctca agcttcgaat tcgatggaag tgcagttagg
gctgggaagg 780gtctaccctc ggccgccgtc caagacctac cgaggagctt tccagaatct
gttccagagc 840gtgcgcgaag tgatccagaa cccgggcccc aggcacccag aggccgcgag
cgcagcacct 900cccggcgcca gtttgctgct gctgcagcag cagcagcagc agcagcagca
gcagcagcag 960cagcagcagc agcagcagca gcagcagcag caagagacta gccccaggca
gcagcagcag 1020cagcagggtg aggatggttc tccccaagcc catcgtagag gccccacagg
ctacctggtc 1080ctggatgagg aacagcaacc ttcacagccg cagtcggccc tggagtgcca
ccccgagaga 1140ggttgcgtcc cagagcctgg agccgccgtg gccgccagca aggggctgcc
gcagcagctg 1200ccagcacctc cggacgagga tgactcagct gccccatcca cgttgtccct
gctgggcccc 1260actttccccg gcttaagcag ctgctccgct gaccttaaag acatcctgag
cgaggccagc 1320accatgcaac tccttcagca acagcagcag gaagcagtat ccgaaggcag
cagcagcggg 1380agagcgaggg aggcctcggg ggctcccact tcctccaagg acaattactt
agggggcact 1440tcgaccattt ctgacaacgc caaggagttg tgtaaggcag tgtcggtgtc
catgggcctg 1500ggtgtggagg cgttggagca tctgagtcca ggggaacagc ttcgggggga
ttgcatgtac 1560gccccacttt tgggagttcc acccgctgtg cgtcccactc cttgtgcccc
attggccgaa 1620tgcaaaggtt ctctgctaga cgacagcgca ggcaagagca ctgaagatac
tgctgagtat 1680tcccctttca agggaggtta caccaaaggg ctagaaggcg agagcctagg
ctgctctggc 1740agcgctgcag cagggagctc cgggacactt gaactgccgt ctaccctgtc
tctctacaag 1800tccggagcac tggacgaggc agctgcgtac cagagtcgcg actactacaa
ctttccactg 1860gctctggccg gaccgccgcc ccctccgccg cctccccatc cccacgctcg
catcaagctg 1920gagaacccgc tggactacgg cagcgcctgg gcggctgcgg cggcgcagtg
ccgctatggg 1980gacctggcga gcctgcatgg cgcgggtgca gcgggacccg gttctgggtc
accctcagcc 2040gccgcttcct catcctggca cactctcttc acagccgaag aaggccagtt
gtatggaccg 2100tgtggtggtg gtgggggtgg tggcggcggc ggcggcggcg gcggcggcgg
cggcggcggc 2160ggcggcggcg gcgaggcggg agctgtagcc ccctacggct acactcggcc
ccctcagggg 2220ctggcgggcc aggaaagcga cttcaccgca cctgatgtgt ggtaccctgg
cggcatggtg 2280agcagagtgc cctatcccag tcccacttgt gtcaaaagcg aaatgggccc
ctggatggat 2340agctactccg gaccttacgg ggacatgcgt ttggagactg ccagggacca
tgttttgccc 2400attgactatt actttccacc ccagaagacc tgcctgatct gtggagatga
agcttctggg 2460tgtcactatg gagctctcac atgtggaagc tgcaaggtct tcttcaaaag
agccgctgaa 2520gggaaacaga agtacctgtg cgccagcaga aatgattgca ctattgataa
attccgaagg 2580aaaaattgtc catcttgtcg tcttcggaaa tgttatgaag cagggatgac
tctgggagcc 2640cggaagctga agaaacttgg taatctgaaa ctacaggagg aaggagaggc
ttccagcacc 2700accagcccca ctgaggagac aacccagaag ctgacagtgt cacacattga
aggctatgaa 2760tgtcagccca tctttctgaa tgtcctggaa gccattgagc caggtgtagt
gtgtgctgga 2820cacgacaaca accagcccga ctcctttgca gccttgctct ctagcctcaa
tgaactggga 2880gagagacagc ttgtacacgt ggtcaagtgg gccaaggcct tgcctggctt
ccgcaactta 2940cacgtggacg accagatggc tgtcattcag tactcctgga tggggctcat
ggtgtttgcc 3000atgggctggc gatccttcac caatgtcaac tccaggatgc tctacttcgc
ccctgatctg 3060gttttcaatg agtaccgcat gcacaagtcc cggatgtaca gccagtgtgt
ccgaatgagg 3120cacctctctc aagagtttgg atggctccaa atcacccccc aggaattcct
gtgcatgaaa 3180gcactgctac tcttcagcat tattccagtg gatgggctga aaaatcaaaa
attctttgat 3240gaacttcgaa tgaactacat caaggaactc gatcgtatca ttgcatgcaa
aagaaaaaat 3300cccacatcct gctcaagacg cttctaccag ctcaccaagc tcctggactc
cgtgcagcct 3360attgcgagag agctgcatca gttcactttt gacctgctaa tcaagtcaca
catggtgagc 3420gtggactttc cggaaatgat ggcagagatc atctctgtgc aagtgcccaa
gatcctttct 3480gggaaagtca agcccatcta tttccacacc cagtga
351681171PRTartificial sequenceeGFP-hAR traceable fusion
protein 8Met Val Ser Lys Gly Glu Glu Leu Phe Thr Gly Val Val Pro Ile Leu
1 5 10 15 Val Glu
Leu Asp Gly Asp Val Asn Gly His Lys Phe Ser Val Ser Gly 20
25 30 Glu Gly Glu Gly Asp Ala Thr
Tyr Gly Lys Leu Thr Leu Lys Phe Ile 35 40
45 Cys Thr Thr Gly Lys Leu Pro Val Pro Trp Pro Thr
Leu Val Thr Thr 50 55 60
Leu Thr Tyr Gly Val Gln Cys Phe Ser Arg Tyr Pro Asp His Met Lys 65
70 75 80 Gln His Asp
Phe Phe Lys Ser Ala Met Pro Glu Gly Tyr Val Gln Glu 85
90 95 Arg Thr Ile Phe Phe Lys Asp Asp
Gly Asn Tyr Lys Thr Arg Ala Glu 100 105
110 Val Lys Phe Glu Gly Asp Thr Leu Val Asn Arg Ile Glu
Leu Lys Gly 115 120 125
Ile Asp Phe Lys Glu Asp Gly Asn Ile Leu Gly His Lys Leu Glu Tyr 130
135 140 Asn Tyr Asn Ser
His Asn Val Tyr Ile Met Ala Asp Lys Gln Lys Asn 145 150
155 160 Gly Ile Lys Val Asn Phe Lys Ile Arg
His Asn Ile Glu Asp Gly Ser 165 170
175 Val Gln Leu Ala Asp His Tyr Gln Gln Asn Thr Pro Ile Gly
Asp Gly 180 185 190
Pro Val Leu Leu Pro Asp Asn His Tyr Leu Ser Thr Gln Ser Ala Leu
195 200 205 Ser Lys Asp Pro
Asn Glu Lys Arg Asp His Met Val Leu Leu Glu Phe 210
215 220 Val Thr Ala Ala Gly Ile Thr Leu
Gly Met Asp Glu Leu Tyr Lys Ser 225 230
235 240 Gly Leu Arg Ser Arg Ala Gln Ala Ser Asn Ser Met
Glu Val Gln Leu 245 250
255 Gly Leu Gly Arg Val Tyr Pro Arg Pro Pro Ser Lys Thr Tyr Arg Gly
260 265 270 Ala Phe Gln
Asn Leu Phe Gln Ser Val Arg Glu Val Ile Gln Asn Pro 275
280 285 Gly Pro Arg His Pro Glu Ala Ala
Ser Ala Ala Pro Pro Gly Ala Ser 290 295
300 Leu Leu Leu Leu Gln Gln Gln Gln Gln Gln Gln Gln Gln
Gln Gln Gln 305 310 315
320 Gln Gln Gln Gln Gln Gln Gln Gln Gln Gln Gln Glu Thr Ser Pro Arg
325 330 335 Gln Gln Gln Gln
Gln Gln Gly Glu Asp Gly Ser Pro Gln Ala His Arg 340
345 350 Arg Gly Pro Thr Gly Tyr Leu Val Leu
Asp Glu Glu Gln Gln Pro Ser 355 360
365 Gln Pro Gln Ser Ala Leu Glu Cys His Pro Glu Arg Gly Cys
Val Pro 370 375 380
Glu Pro Gly Ala Ala Val Ala Ala Ser Lys Gly Leu Pro Gln Gln Leu 385
390 395 400 Pro Ala Pro Pro Asp
Glu Asp Asp Ser Ala Ala Pro Ser Thr Leu Ser 405
410 415 Leu Leu Gly Pro Thr Phe Pro Gly Leu Ser
Ser Cys Ser Ala Asp Leu 420 425
430 Lys Asp Ile Leu Ser Glu Ala Ser Thr Met Gln Leu Leu Gln Gln
Gln 435 440 445 Gln
Gln Glu Ala Val Ser Glu Gly Ser Ser Ser Gly Arg Ala Arg Glu 450
455 460 Ala Ser Gly Ala Pro Thr
Ser Ser Lys Asp Asn Tyr Leu Gly Gly Thr 465 470
475 480 Ser Thr Ile Ser Asp Asn Ala Lys Glu Leu Cys
Lys Ala Val Ser Val 485 490
495 Ser Met Gly Leu Gly Val Glu Ala Leu Glu His Leu Ser Pro Gly Glu
500 505 510 Gln Leu
Arg Gly Asp Cys Met Tyr Ala Pro Leu Leu Gly Val Pro Pro 515
520 525 Ala Val Arg Pro Thr Pro Cys
Ala Pro Leu Ala Glu Cys Lys Gly Ser 530 535
540 Leu Leu Asp Asp Ser Ala Gly Lys Ser Thr Glu Asp
Thr Ala Glu Tyr 545 550 555
560 Ser Pro Phe Lys Gly Gly Tyr Thr Lys Gly Leu Glu Gly Glu Ser Leu
565 570 575 Gly Cys Ser
Gly Ser Ala Ala Ala Gly Ser Ser Gly Thr Leu Glu Leu 580
585 590 Pro Ser Thr Leu Ser Leu Tyr Lys
Ser Gly Ala Leu Asp Glu Ala Ala 595 600
605 Ala Tyr Gln Ser Arg Asp Tyr Tyr Asn Phe Pro Leu Ala
Leu Ala Gly 610 615 620
Pro Pro Pro Pro Pro Pro Pro Pro His Pro His Ala Arg Ile Lys Leu 625
630 635 640 Glu Asn Pro Leu
Asp Tyr Gly Ser Ala Trp Ala Ala Ala Ala Ala Gln 645
650 655 Cys Arg Tyr Gly Asp Leu Ala Ser Leu
His Gly Ala Gly Ala Ala Gly 660 665
670 Pro Gly Ser Gly Ser Pro Ser Ala Ala Ala Ser Ser Ser Trp
His Thr 675 680 685
Leu Phe Thr Ala Glu Glu Gly Gln Leu Tyr Gly Pro Cys Gly Gly Gly 690
695 700 Gly Gly Gly Gly Gly
Gly Gly Gly Gly Gly Gly Gly Gly Gly Gly Gly 705 710
715 720 Gly Gly Gly Gly Glu Ala Gly Ala Val Ala
Pro Tyr Gly Tyr Thr Arg 725 730
735 Pro Pro Gln Gly Leu Ala Gly Gln Glu Ser Asp Phe Thr Ala Pro
Asp 740 745 750 Val
Trp Tyr Pro Gly Gly Met Val Ser Arg Val Pro Tyr Pro Ser Pro 755
760 765 Thr Cys Val Lys Ser Glu
Met Gly Pro Trp Met Asp Ser Tyr Ser Gly 770 775
780 Pro Tyr Gly Asp Met Arg Leu Glu Thr Ala Arg
Asp His Val Leu Pro 785 790 795
800 Ile Asp Tyr Tyr Phe Pro Pro Gln Lys Thr Cys Leu Ile Cys Gly Asp
805 810 815 Glu Ala
Ser Gly Cys His Tyr Gly Ala Leu Thr Cys Gly Ser Cys Lys 820
825 830 Val Phe Phe Lys Arg Ala Ala
Glu Gly Lys Gln Lys Tyr Leu Cys Ala 835 840
845 Ser Arg Asn Asp Cys Thr Ile Asp Lys Phe Arg Arg
Lys Asn Cys Pro 850 855 860
Ser Cys Arg Leu Arg Lys Cys Tyr Glu Ala Gly Met Thr Leu Gly Ala 865
870 875 880 Arg Lys Leu
Lys Lys Leu Gly Asn Leu Lys Leu Gln Glu Glu Gly Glu 885
890 895 Ala Ser Ser Thr Thr Ser Pro Thr
Glu Glu Thr Thr Gln Lys Leu Thr 900 905
910 Val Ser His Ile Glu Gly Tyr Glu Cys Gln Pro Ile Phe
Leu Asn Val 915 920 925
Leu Glu Ala Ile Glu Pro Gly Val Val Cys Ala Gly His Asp Asn Asn 930
935 940 Gln Pro Asp Ser
Phe Ala Ala Leu Leu Ser Ser Leu Asn Glu Leu Gly 945 950
955 960 Glu Arg Gln Leu Val His Val Val Lys
Trp Ala Lys Ala Leu Pro Gly 965 970
975 Phe Arg Asn Leu His Val Asp Asp Gln Met Ala Val Ile Gln
Tyr Ser 980 985 990
Trp Met Gly Leu Met Val Phe Ala Met Gly Trp Arg Ser Phe Thr Asn
995 1000 1005 Val Asn Ser
Arg Met Leu Tyr Phe Ala Pro Asp Leu Val Phe Asn 1010
1015 1020 Glu Tyr Arg Met His Lys Ser Arg
Met Tyr Ser Gln Cys Val Arg 1025 1030
1035 Met Arg His Leu Ser Gln Glu Phe Gly Trp Leu Gln Ile
Thr Pro 1040 1045 1050
Gln Glu Phe Leu Cys Met Lys Ala Leu Leu Leu Phe Ser Ile Ile 1055
1060 1065 Pro Val Asp Gly Leu
Lys Asn Gln Lys Phe Phe Asp Glu Leu Arg 1070 1075
1080 Met Asn Tyr Ile Lys Glu Leu Asp Arg Ile
Ile Ala Cys Lys Arg 1085 1090 1095
Lys Asn Pro Thr Ser Cys Ser Arg Arg Phe Tyr Gln Leu Thr Lys
1100 1105 1110 Leu Leu
Asp Ser Val Gln Pro Ile Ala Arg Glu Leu His Gln Phe 1115
1120 1125 Thr Phe Asp Leu Leu Ile Lys
Ser His Met Val Ser Val Asp Phe 1130 1135
1140 Pro Glu Met Met Ala Glu Ile Ile Ser Val Gln Val
Pro Lys Ile 1145 1150 1155
Leu Ser Gly Lys Val Lys Pro Ile Tyr Phe His Thr Gln 1160
1165 1170 93243DNAartificial
sequencepRevTRE-GFP-mAhR encoding sequence 9atggcccacc atcaccacca
tcacggatat ccatacgacg tgccagatta cgctcagtcg 60agtgccatga gtaaaggaga
agaacttttc actggagttg tcccaattct tgttgaatta 120gatggtgatg ttaatgggca
caaattttct gtcagtggag agggtgaagg tgatgcaaca 180tacggaaaac ttacccttaa
atttatttgc actactggaa aactacctgt tccttggcca 240acacttgtca ctactttcac
ttatggtgtt caatgctttt caagataccc agatcatatg 300aaacagcatg actttttcaa
gagtgccatg cccgaaggtt atgtacagga aagaactata 360tttttcaaag atgacgggaa
ctacaagaca cgtgctgaag tcaagtttga aggtgatacc 420cttgttaata gaatcgagtt
aaaaggtatt gattttaaag aagatggaaa cattcttgga 480cacaaattgg aatacaacta
taactcacac aatgtataca tcatggcaga caaacaaaag 540aatggaatca aagttaactt
caaaattaga cacaacattg aagatggaag cgttcaacta 600gcagaccatt atcaacaaaa
tactccaatt ggcgatggcc ctgtcctttt accagacaac 660cattacctgt ccacacaatc
tgccctttcg aaagatccca acgaaaagag agaccacatg 720gtccttcttg agtttgtaac
agctgctggg attacacatg gcatggatga actatacaaa 780ggcgccggcg ctggagctgg
tgctggcgcc atcagcgcgc tgatcctgag cagcggcgcc 840aacatcacct atgccagccg
caagcggcgc aagccggtgc agaaaacagt aaagcccatc 900cccgctgaag gaattaagtc
aaatccttct aagcgacaca gagaccggct gaacacagag 960ttagaccgcc tggccagcct
gctgcccttc ccgcaagatg ttattaataa gctggacaaa 1020ctctctgttc ttaggctcag
cgtcagctac ctgagggcca agagcttctt tgatgttgca 1080ttaaagtcca cccctgctga
cagaaatgga ggccaggacc agtgtagagc acaaatcaga 1140gactggcagg atttgcaaga
aggagagttc ttgttacagg cgctgaatgg ctttgtgctg 1200gttgtcacag cagatgcctt
ggtcttctat gcttcctcca ctatccaaga ttacctgggc 1260tttcagcagt ctgatgtcat
ccatcagagc gtatatgagc tcatccatac agaagaccgg 1320gcggaattcc agcgccagct
tcactgggct ctaaacccag actctgcaca aggagtggac 1380gaagcccatg gccctccaca
ggcagcagtc tattataccc cagaccagct tcctccagag 1440aacgcttctt tcatggagag
gtgcttcagg tgccggctga ggtgcctgct ggataattca 1500tctggttttc tggcaatgaa
tttccaaggg aggttaaagt atcttcatgg acagaacaag 1560aaagggaagg acggagcgct
gcttcctcca caactggctt tgtttgcaat agctactcca 1620cttcagccac cctccatcct
ggaaattcga accaaaaact tcatcttcag gaccaaacac 1680aagctagact tcacacctat
tggttgtgat gccaaagggc agcttattct gggctataca 1740gaagtagagc tgtgcacaag
aggatcgggg taccagttca tccatgctgc agacatactt 1800cactgtgcag aatcccacat
ccgcatgatt aagactggag aaagtggcat gacagttttc 1860cggcttcttg caaaacacag
tcgctggagg tgggtccagt ccaatgcacg cttgatttac 1920agaaatggaa gaccagatta
catcatcgcc actcagagac cactgacgga tgaagaagga 1980cgagagcatt tacagaagcg
aagtacgtcg ctgcccttca tgtttgctac cggagaggct 2040gtgttgtacg agatctccag
ccctttctct cccataatgg atcccctacc aatacgcacc 2100aaaagcaaca ctagcaggaa
agactgggct ccccagtcaa ccccaagtaa ggattctttc 2160caccccagtt ctcttatgag
tgccctcatc cagcaggatg agtccatcta tctgtgtcct 2220ccttcaagcc ctgcgctgtt
agacagccat tttctcatgg gctccgtgag caagtgcggg 2280agttggcaag acagctttgc
ggccgcagga agtgaggctg cgctgaaaca tgagcaaatt 2340ggccatgctc aggacgtgaa
ccttgcactc tctggcggcc cctcagagct ctttccggat 2400aataaaaata atgacttgta
cagcatcatg aggaaccttg ggattgattt tgaagatatc 2460agaagcatgc agaacgagga
gttcttcaga actgactcca ccgctgctgg tgaggttgac 2520ttcaaagaca tcgacataac
ggacgaaatc ctgacctacg tgcaggattc cctgaacaat 2580tcaactttgc tgaactcggc
ttgccagcag cagcctgtga ctcagcacct aagctgtatg 2640ctgcaggagc gcctgcaact
agagcaacag caacagcttc agcagccccc gccgcaggct 2700ctggagcccc agcagcagct
gtgtcagatg gtgtgccccc agcaagatct gggtccgaag 2760cacacgcaaa tcaacggcac
gtttgcaagt tggaacccca cccctcccgt gtctttcaac 2820tgtccccagc aggaactaaa
gcactatcag ctcttttcca gcttacaggg gactgctcag 2880gaatttccct acaaaccaga
ggtggacagt gtgccttaca cacagaactt tgctccctgt 2940aatcagcctc tgcttccaga
acattccaag agtgtgcagt tggacttccc tggaagggat 3000tttgaaccgt ccctgcatcc
cactacttct aatttagatt ttgtcagttg tttacaagtt 3060cctgaaaacc aaagtcatgg
gataaactca cagtccgcca tggtcagtcc tcaggcatac 3120tatgctgggg ccatgtccat
gtatcagtgc cagccagggc cacagcgcac ccctgtggac 3180cagacgcagt acagctctga
aattccaggt tctcaggcat tcctaagcaa ggtgcagagt 3240tga
3243101080PRTartificial
sequencepRevTRE-GFP-mAhR traceable fusion protein 10Met Ala His His His
His His His Gly Tyr Pro Tyr Asp Val Pro Asp 1 5
10 15 Tyr Ala Gln Ser Ser Ala Met Ser Lys Gly
Glu Glu Leu Phe Thr Gly 20 25
30 Val Val Pro Ile Leu Val Glu Leu Asp Gly Asp Val Asn Gly His
Lys 35 40 45 Phe
Ser Val Ser Gly Glu Gly Glu Gly Asp Ala Thr Tyr Gly Lys Leu 50
55 60 Thr Leu Lys Phe Ile Cys
Thr Thr Gly Lys Leu Pro Val Pro Trp Pro 65 70
75 80 Thr Leu Val Thr Thr Phe Thr Tyr Gly Val Gln
Cys Phe Ser Arg Tyr 85 90
95 Pro Asp His Met Lys Gln His Asp Phe Phe Lys Ser Ala Met Pro Glu
100 105 110 Gly Tyr
Val Gln Glu Arg Thr Ile Phe Phe Lys Asp Asp Gly Asn Tyr 115
120 125 Lys Thr Arg Ala Glu Val Lys
Phe Glu Gly Asp Thr Leu Val Asn Arg 130 135
140 Ile Glu Leu Lys Gly Ile Asp Phe Lys Glu Asp Gly
Asn Ile Leu Gly 145 150 155
160 His Lys Leu Glu Tyr Asn Tyr Asn Ser His Asn Val Tyr Ile Met Ala
165 170 175 Asp Lys Gln
Lys Asn Gly Ile Lys Val Asn Phe Lys Ile Arg His Asn 180
185 190 Ile Glu Asp Gly Ser Val Gln Leu
Ala Asp His Tyr Gln Gln Asn Thr 195 200
205 Pro Ile Gly Asp Gly Pro Val Leu Leu Pro Asp Asn His
Tyr Leu Ser 210 215 220
Thr Gln Ser Ala Leu Ser Lys Asp Pro Asn Glu Lys Arg Asp His Met 225
230 235 240 Val Leu Leu Glu
Phe Val Thr Ala Ala Gly Ile Thr His Gly Met Asp 245
250 255 Glu Leu Tyr Lys Gly Ala Gly Ala Gly
Ala Gly Ala Gly Ala Ile Ser 260 265
270 Ala Leu Ile Leu Ser Ser Gly Ala Asn Ile Thr Tyr Ala Ser
Arg Lys 275 280 285
Arg Arg Lys Pro Val Gln Lys Thr Val Lys Pro Ile Pro Ala Glu Gly 290
295 300 Ile Lys Ser Asn Pro
Ser Lys Arg His Arg Asp Arg Leu Asn Thr Glu 305 310
315 320 Leu Asp Arg Leu Ala Ser Leu Leu Pro Phe
Pro Gln Asp Val Ile Asn 325 330
335 Lys Leu Asp Lys Leu Ser Val Leu Arg Leu Ser Val Ser Tyr Leu
Arg 340 345 350 Ala
Lys Ser Phe Phe Asp Val Ala Leu Lys Ser Thr Pro Ala Asp Arg 355
360 365 Asn Gly Gly Gln Asp Gln
Cys Arg Ala Gln Ile Arg Asp Trp Gln Asp 370 375
380 Leu Gln Glu Gly Glu Phe Leu Leu Gln Ala Leu
Asn Gly Phe Val Leu 385 390 395
400 Val Val Thr Ala Asp Ala Leu Val Phe Tyr Ala Ser Ser Thr Ile Gln
405 410 415 Asp Tyr
Leu Gly Phe Gln Gln Ser Asp Val Ile His Gln Ser Val Tyr 420
425 430 Glu Leu Ile His Thr Glu Asp
Arg Ala Glu Phe Gln Arg Gln Leu His 435 440
445 Trp Ala Leu Asn Pro Asp Ser Ala Gln Gly Val Asp
Glu Ala His Gly 450 455 460
Pro Pro Gln Ala Ala Val Tyr Tyr Thr Pro Asp Gln Leu Pro Pro Glu 465
470 475 480 Asn Ala Ser
Phe Met Glu Arg Cys Phe Arg Cys Arg Leu Arg Cys Leu 485
490 495 Leu Asp Asn Ser Ser Gly Phe Leu
Ala Met Asn Phe Gln Gly Arg Leu 500 505
510 Lys Tyr Leu His Gly Gln Asn Lys Lys Gly Lys Asp Gly
Ala Leu Leu 515 520 525
Pro Pro Gln Leu Ala Leu Phe Ala Ile Ala Thr Pro Leu Gln Pro Pro 530
535 540 Ser Ile Leu Glu
Ile Arg Thr Lys Asn Phe Ile Phe Arg Thr Lys His 545 550
555 560 Lys Leu Asp Phe Thr Pro Ile Gly Cys
Asp Ala Lys Gly Gln Leu Ile 565 570
575 Leu Gly Tyr Thr Glu Val Glu Leu Cys Thr Arg Gly Ser Gly
Tyr Gln 580 585 590
Phe Ile His Ala Ala Asp Ile Leu His Cys Ala Glu Ser His Ile Arg
595 600 605 Met Ile Lys Thr
Gly Glu Ser Gly Met Thr Val Phe Arg Leu Leu Ala 610
615 620 Lys His Ser Arg Trp Arg Trp Val
Gln Ser Asn Ala Arg Leu Ile Tyr 625 630
635 640 Arg Asn Gly Arg Pro Asp Tyr Ile Ile Ala Thr Gln
Arg Pro Leu Thr 645 650
655 Asp Glu Glu Gly Arg Glu His Leu Gln Lys Arg Ser Thr Ser Leu Pro
660 665 670 Phe Met Phe
Ala Thr Gly Glu Ala Val Leu Tyr Glu Ile Ser Ser Pro 675
680 685 Phe Ser Pro Ile Met Asp Pro Leu
Pro Ile Arg Thr Lys Ser Asn Thr 690 695
700 Ser Arg Lys Asp Trp Ala Pro Gln Ser Thr Pro Ser Lys
Asp Ser Phe 705 710 715
720 His Pro Ser Ser Leu Met Ser Ala Leu Ile Gln Gln Asp Glu Ser Ile
725 730 735 Tyr Leu Cys Pro
Pro Ser Ser Pro Ala Leu Leu Asp Ser His Phe Leu 740
745 750 Met Gly Ser Val Ser Lys Cys Gly Ser
Trp Gln Asp Ser Phe Ala Ala 755 760
765 Ala Gly Ser Glu Ala Ala Leu Lys His Glu Gln Ile Gly His
Ala Gln 770 775 780
Asp Val Asn Leu Ala Leu Ser Gly Gly Pro Ser Glu Leu Phe Pro Asp 785
790 795 800 Asn Lys Asn Asn Asp
Leu Tyr Ser Ile Met Arg Asn Leu Gly Ile Asp 805
810 815 Phe Glu Asp Ile Arg Ser Met Gln Asn Glu
Glu Phe Phe Arg Thr Asp 820 825
830 Ser Thr Ala Ala Gly Glu Val Asp Phe Lys Asp Ile Asp Ile Thr
Asp 835 840 845 Glu
Ile Leu Thr Tyr Val Gln Asp Ser Leu Asn Asn Ser Thr Leu Leu 850
855 860 Asn Ser Ala Cys Gln Gln
Gln Pro Val Thr Gln His Leu Ser Cys Met 865 870
875 880 Leu Gln Glu Arg Leu Gln Leu Glu Gln Gln Gln
Gln Leu Gln Gln Pro 885 890
895 Pro Pro Gln Ala Leu Glu Pro Gln Gln Gln Leu Cys Gln Met Val Cys
900 905 910 Pro Gln
Gln Asp Leu Gly Pro Lys His Thr Gln Ile Asn Gly Thr Phe 915
920 925 Ala Ser Trp Asn Pro Thr Pro
Pro Val Ser Phe Asn Cys Pro Gln Gln 930 935
940 Glu Leu Lys His Tyr Gln Leu Phe Ser Ser Leu Gln
Gly Thr Ala Gln 945 950 955
960 Glu Phe Pro Tyr Lys Pro Glu Val Asp Ser Val Pro Tyr Thr Gln Asn
965 970 975 Phe Ala Pro
Cys Asn Gln Pro Leu Leu Pro Glu His Ser Lys Ser Val 980
985 990 Gln Leu Asp Phe Pro Gly Arg Asp
Phe Glu Pro Ser Leu His Pro Thr 995 1000
1005 Thr Ser Asn Leu Asp Phe Val Ser Cys Leu Gln
Val Pro Glu Asn 1010 1015 1020
Gln Ser His Gly Ile Asn Ser Gln Ser Ala Met Val Ser Pro Gln
1025 1030 1035 Ala Tyr Tyr
Ala Gly Ala Met Ser Met Tyr Gln Cys Gln Pro Gly 1040
1045 1050 Pro Gln Arg Thr Pro Val Asp Gln
Thr Gln Tyr Ser Ser Glu Ile 1055 1060
1065 Pro Gly Ser Gln Ala Phe Leu Ser Lys Val Gln Ser
1070 1075 1080 113678DNAartificial
sequenceeGFP-ratMR encoding sequence 11atggtgagca agggcgagga gctgttcacc
ggggtggtgc ccatcctggt cgagctggac 60ggcgacgtaa acggccacaa gttcagcgtg
tccggcgagg gcgagggcga tgccacctac 120ggcaagctga ccctgaagtt catctgcacc
accggcaagc tgcccgtgcc ctggcccacc 180ctcgtgacca ccctgaccta cggcgtgcag
tgcttcagcc gctaccccga ccacatgaag 240cagcacgact tcttcaagtc cgccatgccc
gaaggctacg tccaggagcg caccatcttc 300ttcaaggacg acggcaacta caagacccgc
gccgaggtga agttcgaggg cgacaccctg 360gtgaaccgca tcgagctgaa gggcatcgac
ttcaaggagg acggcaacat cctggggcac 420aagctggagt acaactacaa cagccacaac
gtctatatca tggccgacaa gcagaagaac 480ggcatcaagg tgaacttcaa gatccgccac
aacatcgagg acggcagcgt gcagctcgcc 540gaccactacc agcagaacac ccccatcggc
gacggccccg tgctgctgcc cgacaaccac 600tacctgagca cccagtccgc cctgagcaaa
gaccccaacg agaagcgcga tcacatggtc 660ctgctggagt tcgtgaccgc cgccgggatc
actctcggca tggacgagct gtacaagtcc 720ggactcagat ctatggaaac caaaggctac
cacagtctcc ctgaaggcct agacatggaa 780aggcgctgga gtcaagtgtc tcagaccttg
gagcgttctt cacttggacc tgcagagagg 840accactgaga acaactacat ggagattgtc
aacgtcagct gcgtttccgg tgctattccg 900aacaacagta ctcaaggaag cagcaaagaa
aagcacgaat tactccctta cattcagcaa 960gacaatagtc ggtctgggat tttgccatca
gatattaaaa ctgagctgga atccaaggaa 1020ctttcagcca cggtagctga gtccatgggt
ttatatatgg attctgtgag agatgccgag 1080tacacttacg atcagcagaa ccaacaagga
agcctgagcc caacaaagat ttatcaaaac 1140atggagcagc tggtgaagtt ttacaaagag
aatggtcaca ggtcctccac actgagtgct 1200atgagcagac ctttgaggtc attcatgcct
gactctgcag cctccatgaa tggtggggcc 1260ttgcgtgcca tcgtgaagag ccctatcatc
tgtcatgaga agagctcttc tgttagcagc 1320ccgctgaaca tggcatcttc agtatgcagc
cctgtgggca tcaactccat gtcctcctcc 1380acgactagct ttggtagttt cccagtgcac
agtcccatca ctcaaggaac ctcactgaca 1440tgctccccca gtgtcgaaaa cagaggctca
aggtcacaca gccccacaca tgcgagcaac 1500gtgggctccc ctctttcaag tccattgagc
agcatgaaat ctccaatctc cagccctcca 1560agtcattgca gtgtaaaatc tccagtctcc
agtccaaata acgtccctct gcgctcctct 1620gtgtccagcc cggcaaatct caacaactca
aggtgctccg tttccagccc ttccaacaac 1680accaacaata gatccacact ctccagcccg
acagccagca cagtggggtc catcggcagc 1740cccatcagca atgccttcag ctatgccact
tcaggcgctt ctgctggagc cggtgccatc 1800caggatgtgg ttcctagtcc agacacccac
gagaaaggtg ctcacgacgt tcctttccct 1860aagacagagg aagtcgagaa ggccatctcc
aatggtgtga ctggcccgct caacattgtc 1920cagtacataa aatcagaacc agatggggct
ttcagcagtt cctgtctagg aggaaacagc 1980aaaatcagcc ccagttctcc attctctgta
ccaataaagc aagagtcaag caagcactca 2040tgttcaggcg cctcttttaa agggaacccc
acggtcaacc catttccatt catggatggc 2100tcctactttt cctttatgga tgataaggac
tattattccc tatcaggaat cttaggacca 2160cctgtgcccg gctttgatgg tagctgcgag
ggcagcgcat tcccagtggg gattaagcaa 2220gaaccagatg atgggagcta ttaccctgaa
gccagcatcc catcatctgc tatcgttggt 2280gtgaattcgg gtggacagtc ctttcactac
cggattggtg ctcaaggtac aatatcttta 2340tcacggtcac ctagagacca atctttccaa
cacttgagtt ccttcccacc tgtcaataca 2400ttagtggaat catggaaacc acacggtgac
ctgtcatcta ggagaagtga tgggtatccc 2460gtcctagagt acattccaga aaacgtgtca
agctctactt tacgaagtgt ttctactgga 2520tcctcaagac cttccaagat ctgtttggtg
tgtggagatg aggcttctgg gtgtcactat 2580ggggtagtga cctgtggcag ctgcaaagtc
ttcttcaaaa gagccgtgga agggcaacac 2640aactatctgt gtgctggaag aaatgactgc
attattgata agattcggcg aaagaactgt 2700cctgcctgca ggctccagaa atgccttcaa
gctggcatga acttaggagc tcgaaagtca 2760aagaagctgg ggaagttaaa aggcctccac
gaggagcaac cacagcagcc cccaccaccg 2820ccaccccaga gtccagaaga ggggaccaca
tacatcgctc cgaccaagga gccatcggtg 2880aactctgcgc tggtcccgca gctcacctcc
attacgcatg cactcacacc atccccggcc 2940atgatcctgg agaacatcga gcctgagacc
gtgtatgcag gctacgacaa ttccaagccc 3000gacaccgccg agagcctgct ctccacgctc
aaccgcctgg caggcaaaca gatgatccag 3060gtcgtgaagt gggccaaggt acttccagga
tttaaaaact tgcctctcga ggaccaaatc 3120accctcatcc agtattcttg gatgtgtcta
tcatcgttcg ctttgagttg gagatcgtac 3180aaacacacga acagccaact cctctatttt
gctccagacc tagtttttaa tgaagagaag 3240atgcatcagt ctgccatgta tgaactgtgc
caggggatgc gccagatcag ccttcaattc 3300gtgcggctgc agctgacctt tgaggagtac
tccataatga aggttttgct gctactaagc 3360acagttccaa aagatggcct caagagccag
gctgcatttg aggagatgag gacaaattac 3420atcaaagaac tgaggaaaat ggtcaccaag
tgtcccaaca gttctggaca gagttggcag 3480aggttctacc aactgaccaa gcttctagac
tccatgcatg atctcgtgag tgacctgcta 3540gaattctgct tctacacctt ccgagaatcc
caggctctga aggtggagtt tcccgccatg 3600ctggtggaga tcatcaccga ccagctgcca
aaggtggagt ctgggaatgc caaacccctt 3660tactttcaca gaaagtga
3678121225PRTartificial
sequenceeGFP-ratMR traceable fusion protein 12Met Val Ser Lys Gly Glu Glu
Leu Phe Thr Gly Val Val Pro Ile Leu 1 5
10 15 Val Glu Leu Asp Gly Asp Val Asn Gly His Lys
Phe Ser Val Ser Gly 20 25
30 Glu Gly Glu Gly Asp Ala Thr Tyr Gly Lys Leu Thr Leu Lys Phe
Ile 35 40 45 Cys
Thr Thr Gly Lys Leu Pro Val Pro Trp Pro Thr Leu Val Thr Thr 50
55 60 Leu Thr Tyr Gly Val Gln
Cys Phe Ser Arg Tyr Pro Asp His Met Lys 65 70
75 80 Gln His Asp Phe Phe Lys Ser Ala Met Pro Glu
Gly Tyr Val Gln Glu 85 90
95 Arg Thr Ile Phe Phe Lys Asp Asp Gly Asn Tyr Lys Thr Arg Ala Glu
100 105 110 Val Lys
Phe Glu Gly Asp Thr Leu Val Asn Arg Ile Glu Leu Lys Gly 115
120 125 Ile Asp Phe Lys Glu Asp Gly
Asn Ile Leu Gly His Lys Leu Glu Tyr 130 135
140 Asn Tyr Asn Ser His Asn Val Tyr Ile Met Ala Asp
Lys Gln Lys Asn 145 150 155
160 Gly Ile Lys Val Asn Phe Lys Ile Arg His Asn Ile Glu Asp Gly Ser
165 170 175 Val Gln Leu
Ala Asp His Tyr Gln Gln Asn Thr Pro Ile Gly Asp Gly 180
185 190 Pro Val Leu Leu Pro Asp Asn His
Tyr Leu Ser Thr Gln Ser Ala Leu 195 200
205 Ser Lys Asp Pro Asn Glu Lys Arg Asp His Met Val Leu
Leu Glu Phe 210 215 220
Val Thr Ala Ala Gly Ile Thr Leu Gly Met Asp Glu Leu Tyr Lys Ser 225
230 235 240 Gly Leu Arg Ser
Met Glu Thr Lys Gly Tyr His Ser Leu Pro Glu Gly 245
250 255 Leu Asp Met Glu Arg Arg Trp Ser Gln
Val Ser Gln Thr Leu Glu Arg 260 265
270 Ser Ser Leu Gly Pro Ala Glu Arg Thr Thr Glu Asn Asn Tyr
Met Glu 275 280 285
Ile Val Asn Val Ser Cys Val Ser Gly Ala Ile Pro Asn Asn Ser Thr 290
295 300 Gln Gly Ser Ser Lys
Glu Lys His Glu Leu Leu Pro Tyr Ile Gln Gln 305 310
315 320 Asp Asn Ser Arg Ser Gly Ile Leu Pro Ser
Asp Ile Lys Thr Glu Leu 325 330
335 Glu Ser Lys Glu Leu Ser Ala Thr Val Ala Glu Ser Met Gly Leu
Tyr 340 345 350 Met
Asp Ser Val Arg Asp Ala Glu Tyr Thr Tyr Asp Gln Gln Asn Gln 355
360 365 Gln Gly Ser Leu Ser Pro
Thr Lys Ile Tyr Gln Asn Met Glu Gln Leu 370 375
380 Val Lys Phe Tyr Lys Glu Asn Gly His Arg Ser
Ser Thr Leu Ser Ala 385 390 395
400 Met Ser Arg Pro Leu Arg Ser Phe Met Pro Asp Ser Ala Ala Ser Met
405 410 415 Asn Gly
Gly Ala Leu Arg Ala Ile Val Lys Ser Pro Ile Ile Cys His 420
425 430 Glu Lys Ser Ser Ser Val Ser
Ser Pro Leu Asn Met Ala Ser Ser Val 435 440
445 Cys Ser Pro Val Gly Ile Asn Ser Met Ser Ser Ser
Thr Thr Ser Phe 450 455 460
Gly Ser Phe Pro Val His Ser Pro Ile Thr Gln Gly Thr Ser Leu Thr 465
470 475 480 Cys Ser Pro
Ser Val Glu Asn Arg Gly Ser Arg Ser His Ser Pro Thr 485
490 495 His Ala Ser Asn Val Gly Ser Pro
Leu Ser Ser Pro Leu Ser Ser Met 500 505
510 Lys Ser Pro Ile Ser Ser Pro Pro Ser His Cys Ser Val
Lys Ser Pro 515 520 525
Val Ser Ser Pro Asn Asn Val Pro Leu Arg Ser Ser Val Ser Ser Pro 530
535 540 Ala Asn Leu Asn
Asn Ser Arg Cys Ser Val Ser Ser Pro Ser Asn Asn 545 550
555 560 Thr Asn Asn Arg Ser Thr Leu Ser Ser
Pro Thr Ala Ser Thr Val Gly 565 570
575 Ser Ile Gly Ser Pro Ile Ser Asn Ala Phe Ser Tyr Ala Thr
Ser Gly 580 585 590
Ala Ser Ala Gly Ala Gly Ala Ile Gln Asp Val Val Pro Ser Pro Asp
595 600 605 Thr His Glu Lys
Gly Ala His Asp Val Pro Phe Pro Lys Thr Glu Glu 610
615 620 Val Glu Lys Ala Ile Ser Asn Gly
Val Thr Gly Pro Leu Asn Ile Val 625 630
635 640 Gln Tyr Ile Lys Ser Glu Pro Asp Gly Ala Phe Ser
Ser Ser Cys Leu 645 650
655 Gly Gly Asn Ser Lys Ile Ser Pro Ser Ser Pro Phe Ser Val Pro Ile
660 665 670 Lys Gln Glu
Ser Ser Lys His Ser Cys Ser Gly Ala Ser Phe Lys Gly 675
680 685 Asn Pro Thr Val Asn Pro Phe Pro
Phe Met Asp Gly Ser Tyr Phe Ser 690 695
700 Phe Met Asp Asp Lys Asp Tyr Tyr Ser Leu Ser Gly Ile
Leu Gly Pro 705 710 715
720 Pro Val Pro Gly Phe Asp Gly Ser Cys Glu Gly Ser Ala Phe Pro Val
725 730 735 Gly Ile Lys Gln
Glu Pro Asp Asp Gly Ser Tyr Tyr Pro Glu Ala Ser 740
745 750 Ile Pro Ser Ser Ala Ile Val Gly Val
Asn Ser Gly Gly Gln Ser Phe 755 760
765 His Tyr Arg Ile Gly Ala Gln Gly Thr Ile Ser Leu Ser Arg
Ser Pro 770 775 780
Arg Asp Gln Ser Phe Gln His Leu Ser Ser Phe Pro Pro Val Asn Thr 785
790 795 800 Leu Val Glu Ser Trp
Lys Pro His Gly Asp Leu Ser Ser Arg Arg Ser 805
810 815 Asp Gly Tyr Pro Val Leu Glu Tyr Ile Pro
Glu Asn Val Ser Ser Ser 820 825
830 Thr Leu Arg Ser Val Ser Thr Gly Ser Ser Arg Pro Ser Lys Ile
Cys 835 840 845 Leu
Val Cys Gly Asp Glu Ala Ser Gly Cys His Tyr Gly Val Val Thr 850
855 860 Cys Gly Ser Cys Lys Val
Phe Phe Lys Arg Ala Val Glu Gly Gln His 865 870
875 880 Asn Tyr Leu Cys Ala Gly Arg Asn Asp Cys Ile
Ile Asp Lys Ile Arg 885 890
895 Arg Lys Asn Cys Pro Ala Cys Arg Leu Gln Lys Cys Leu Gln Ala Gly
900 905 910 Met Asn
Leu Gly Ala Arg Lys Ser Lys Lys Leu Gly Lys Leu Lys Gly 915
920 925 Leu His Glu Glu Gln Pro Gln
Gln Pro Pro Pro Pro Pro Pro Gln Ser 930 935
940 Pro Glu Glu Gly Thr Thr Tyr Ile Ala Pro Thr Lys
Glu Pro Ser Val 945 950 955
960 Asn Ser Ala Leu Val Pro Gln Leu Thr Ser Ile Thr His Ala Leu Thr
965 970 975 Pro Ser Pro
Ala Met Ile Leu Glu Asn Ile Glu Pro Glu Thr Val Tyr 980
985 990 Ala Gly Tyr Asp Asn Ser Lys Pro
Asp Thr Ala Glu Ser Leu Leu Ser 995 1000
1005 Thr Leu Asn Arg Leu Ala Gly Lys Gln Met Ile
Gln Val Val Lys 1010 1015 1020
Trp Ala Lys Val Leu Pro Gly Phe Lys Asn Leu Pro Leu Glu Asp
1025 1030 1035 Gln Ile Thr
Leu Ile Gln Tyr Ser Trp Met Cys Leu Ser Ser Phe 1040
1045 1050 Ala Leu Ser Trp Arg Ser Tyr Lys
His Thr Asn Ser Gln Leu Leu 1055 1060
1065 Tyr Phe Ala Pro Asp Leu Val Phe Asn Glu Glu Lys Met
His Gln 1070 1075 1080
Ser Ala Met Tyr Glu Leu Cys Gln Gly Met Arg Gln Ile Ser Leu 1085
1090 1095 Gln Phe Val Arg Leu
Gln Leu Thr Phe Glu Glu Tyr Ser Ile Met 1100 1105
1110 Lys Val Leu Leu Leu Leu Ser Thr Val Pro
Lys Asp Gly Leu Lys 1115 1120 1125
Ser Gln Ala Ala Phe Glu Glu Met Arg Thr Asn Tyr Ile Lys Glu
1130 1135 1140 Leu Arg
Lys Met Val Thr Lys Cys Pro Asn Ser Ser Gly Gln Ser 1145
1150 1155 Trp Gln Arg Phe Tyr Gln Leu
Thr Lys Leu Leu Asp Ser Met His 1160 1165
1170 Asp Leu Val Ser Asp Leu Leu Glu Phe Cys Phe Tyr
Thr Phe Arg 1175 1180 1185
Glu Ser Gln Ala Leu Lys Val Glu Phe Pro Ala Met Leu Val Glu 1190
1195 1200 Ile Ile Thr Asp Gln
Leu Pro Lys Val Glu Ser Gly Asn Ala Lys 1205 1210
1215 Pro Leu Tyr Phe His Arg Lys 1220
1225 133555DNAartificial sequenceeGFP-hPRB encoding sequence
13atgagtaaag gagaagaact tttcactgga gttgtcccaa ttcttgttga attagatggt
60gatgttaatg ggcacaaatt ttctgtcagt ggagagggtg aaggtgatgc aacatacgga
120aaacttaccc ttaaatttat ttgcactact ggaaaactac ctgttccttg gccaacactt
180gtcactactt tcacttatgg tgttcaatgc ttttcaagat acccagatca tatgaaacag
240catgactttt tcaagagtgc catgcccgaa ggttatgtac aggaaagaac tatatttttc
300aaagatgacg ggaactacaa gacacgtgct gaagtcaagt ttgaaggtga tacccttgtt
360aatagaatcg agttaaaagg tattgatttt aaagaagatg gaaacattct tggacacaaa
420ttggaataca actataactc acacaatgta tacatcatgg cagacaaaca aaagaatgga
480atcaaagtta acttcaaaat tagacacaac attgaagatg gaagcgttca actagcagac
540cattatcaac aaaatactcc aattggcgat ggccctgtcc ttttaccaga caaccattac
600ctgtccacac aatctgccct ttcgaaagat cccaacgaaa agagagacca catggtcctt
660cttgagtttg taacagctgc tgggattaca catggcatgg atgaactata caaaggcgcc
720ggcgctggtg ctggtgctgg cgccatcagc gcgttgactg agctgaaggc aaagggtccc
780cgggctcccc acgtggcggg cggcccgccc tcccccgagg tcggatcccc actgctgtgt
840cgcccagccg caggtccgtt cccggggagc cagacctcgg acaccttgcc tgaagtttcg
900gccataccta tctccctgga cgggctactc ttccctcggc cctgccaggg acaggacccc
960tccgacgaaa agacgcagga ccagcagtcg ctgtcggacg tggagggcgc atattccaga
1020gctgaagcta caaggggtgc tggaggcagc agttctagtc ccccagaaaa ggacagcgga
1080ctgctggaca gtgtcttgga cactctgttg gcgccctcag gtcccgggca gagccaaccc
1140agccctcccg cctgcgaggt caccagctct tggtgcctgt ttggccccga acttcccgaa
1200gatccaccgg ctgcccccgc cacccagcgg gtgttgtccc cgctcatgag ccggtccggg
1260tgcaaggttg gagacagctc cgggacggca gctgcccata aagtgctgcc ccggggcctg
1320tcaccagccc ggcagctgct gctcccggcc tctgagagcc ctcactggtc cggggcccca
1380gtgaagccgt ctccgcaggc cgctgcggtg gaggttgagg aggaggatgg ctctgagtcc
1440gaggagtctg cgggtccgct tctgaagggc aaacctcggg ctctgggtgg cgcggcggct
1500ggaggaggag ccgcggctgt cccgccgggg gcggcagcag gaggcgtcgc cctggtcccc
1560aaggaagatt cccgcttctc agcgcccagg gtcgccctgg tggagcagga cgcgccgatg
1620gcgcccgggc gctccccgct ggccaccacg gtgatggatt tcatccacgt gcctatcctg
1680cctctcaatc acgccttatt ggcagcccgc actcggcagc tgctggaaga cgaaagttac
1740gacggcgggg ccggggctgc cagcgccttt gccccgccgc ggagttcacc ctgtgcctcg
1800tccaccccgg tcgctgtagg cgacttcccc gactgcgcgt acccgcccga cgccgagccc
1860aaggacgacg cgtaccctct ctatagcgac ttccagccgc ccgctctaaa gataaaggag
1920gaggaggaag gcgcggaggc ctccgcgcgc tccccgcgtt cctaccttgt ggccggtgcc
1980aaccccgcag ccttcccgga tttcccgttg gggccaccgc ccccgctgcc gccgcgagcg
2040accccatcca gacccgggga agcggcggtg acggccgcac ccgccagtgc ctcagtctcg
2100tctgcgtcct cctcggggtc gaccctggag tgcatcctgt acaaagcgga gggcgcgccg
2160ccccagcagg gcccgttcgc gccgccgccc tgcaaggcgc cgggcgcgag cggctgcctg
2220ctcccgcggg acggcctgcc ctccacctcc gcctctgccg ccgccgccgg ggcggccccc
2280gcgctctacc ctgcactcgg cctcaacggg ctcccgcagc tcggctacca ggccgccgtg
2340ctcaaggagg gcctgccgca ggtctacccg ccctatctca actacctgag gccggattca
2400gaagccagcc agagcccaca atacagcttc gagtcattac ctcagaagat ttgtttaatc
2460tgtggggatg aagcatcagg ctgtcattat ggtgtcctta cctgtgggag ctgtaaggtc
2520ttctttaaga gggcaatgga agggcagcac aactacttat gtgctggaag aaatgactgc
2580atcgttgata aaatccgcag aaaaaactgc ccagcatgtc gccttagaaa gtgctgtcag
2640gctggcatgg tccttggagg tcgaaaattt aaaaagttca ataaagtcag agttgtgaga
2700gcactggatg ctgttgctct cccacagcca gtgggcgttc caaatgaaag ccaagcccta
2760agccagagat tcactttttc accaggtcaa gacatacagt tgattccacc actgatcaac
2820ctgttaatga gcattgaacc agatgtgatc tatgcaggac atgacaacac aaaacctgac
2880acctccagtt ctttgctgac aagtcttaat caactaggcg agaggcaact tctttcagta
2940gtcaagtggt ctaaatcatt gccaggtttt cgaaacttac atattgatga ccagataact
3000ctcattcagt attcttggat gagcttaatg gtgtttggtc taggatggag atcctacaaa
3060cacgtcagtg ggcagatgct gtattttgca cctgatctaa tactaaatga acagcggatg
3120aaagaatcat cattctattc attatgcctt accatgtggc agatcccaca ggagtttgtc
3180aagcttcaag ttagccaaga agagttcctc tgtatgaaag tattgttact tcttaataca
3240attcctttgg aagggctacg aagtcaaacc cagtttgagg agatgaggtc aagctacatt
3300agagagctca tcaaggcaat tggtttgagg caaaaaggag ttgtgtcgag ctcacagcgt
3360ttctatcaac ttacaaaact tcttgataac ttgcatgatc ttgtcaaaca acttcatctg
3420tactgcttga atacatttat ccagtcccgg gcactgagtg ttgaatttcc agaaatgatg
3480tctgaagtta ttgctgcaca attacccaag atattggcag ggatggtgaa accccttctc
3540tttcataaaa agtga
3555141184PRTartificial sequenceeGFP-hPRB traceable fusion protein 14Met
Ser Lys Gly Glu Glu Leu Phe Thr Gly Val Val Pro Ile Leu Val 1
5 10 15 Glu Leu Asp Gly Asp Val
Asn Gly His Lys Phe Ser Val Ser Gly Glu 20
25 30 Gly Glu Gly Asp Ala Thr Tyr Gly Lys Leu
Thr Leu Lys Phe Ile Cys 35 40
45 Thr Thr Gly Lys Leu Pro Val Pro Trp Pro Thr Leu Val Thr
Thr Phe 50 55 60
Thr Tyr Gly Val Gln Cys Phe Ser Arg Tyr Pro Asp His Met Lys Gln 65
70 75 80 His Asp Phe Phe Lys
Ser Ala Met Pro Glu Gly Tyr Val Gln Glu Arg 85
90 95 Thr Ile Phe Phe Lys Asp Asp Gly Asn Tyr
Lys Thr Arg Ala Glu Val 100 105
110 Lys Phe Glu Gly Asp Thr Leu Val Asn Arg Ile Glu Leu Lys Gly
Ile 115 120 125 Asp
Phe Lys Glu Asp Gly Asn Ile Leu Gly His Lys Leu Glu Tyr Asn 130
135 140 Tyr Asn Ser His Asn Val
Tyr Ile Met Ala Asp Lys Gln Lys Asn Gly 145 150
155 160 Ile Lys Val Asn Phe Lys Ile Arg His Asn Ile
Glu Asp Gly Ser Val 165 170
175 Gln Leu Ala Asp His Tyr Gln Gln Asn Thr Pro Ile Gly Asp Gly Pro
180 185 190 Val Leu
Leu Pro Asp Asn His Tyr Leu Ser Thr Gln Ser Ala Leu Ser 195
200 205 Lys Asp Pro Asn Glu Lys Arg
Asp His Met Val Leu Leu Glu Phe Val 210 215
220 Thr Ala Ala Gly Ile Thr His Gly Met Asp Glu Leu
Tyr Lys Gly Ala 225 230 235
240 Gly Ala Gly Ala Gly Ala Gly Ala Ile Ser Ala Leu Thr Glu Leu Lys
245 250 255 Ala Lys Gly
Pro Arg Ala Pro His Val Ala Gly Gly Pro Pro Ser Pro 260
265 270 Glu Val Gly Ser Pro Leu Leu Cys
Arg Pro Ala Ala Gly Pro Phe Pro 275 280
285 Gly Ser Gln Thr Ser Asp Thr Leu Pro Glu Val Ser Ala
Ile Pro Ile 290 295 300
Ser Leu Asp Gly Leu Leu Phe Pro Arg Pro Cys Gln Gly Gln Asp Pro 305
310 315 320 Ser Asp Glu Lys
Thr Gln Asp Gln Gln Ser Leu Ser Asp Val Glu Gly 325
330 335 Ala Tyr Ser Arg Ala Glu Ala Thr Arg
Gly Ala Gly Gly Ser Ser Ser 340 345
350 Ser Pro Pro Glu Lys Asp Ser Gly Leu Leu Asp Ser Val Leu
Asp Thr 355 360 365
Leu Leu Ala Pro Ser Gly Pro Gly Gln Ser Gln Pro Ser Pro Pro Ala 370
375 380 Cys Glu Val Thr Ser
Ser Trp Cys Leu Phe Gly Pro Glu Leu Pro Glu 385 390
395 400 Asp Pro Pro Ala Ala Pro Ala Thr Gln Arg
Val Leu Ser Pro Leu Met 405 410
415 Ser Arg Ser Gly Cys Lys Val Gly Asp Ser Ser Gly Thr Ala Ala
Ala 420 425 430 His
Lys Val Leu Pro Arg Gly Leu Ser Pro Ala Arg Gln Leu Leu Leu 435
440 445 Pro Ala Ser Glu Ser Pro
His Trp Ser Gly Ala Pro Val Lys Pro Ser 450 455
460 Pro Gln Ala Ala Ala Val Glu Val Glu Glu Glu
Asp Gly Ser Glu Ser 465 470 475
480 Glu Glu Ser Ala Gly Pro Leu Leu Lys Gly Lys Pro Arg Ala Leu Gly
485 490 495 Gly Ala
Ala Ala Gly Gly Gly Ala Ala Ala Val Pro Pro Gly Ala Ala 500
505 510 Ala Gly Gly Val Ala Leu Val
Pro Lys Glu Asp Ser Arg Phe Ser Ala 515 520
525 Pro Arg Val Ala Leu Val Glu Gln Asp Ala Pro Met
Ala Pro Gly Arg 530 535 540
Ser Pro Leu Ala Thr Thr Val Met Asp Phe Ile His Val Pro Ile Leu 545
550 555 560 Pro Leu Asn
His Ala Leu Leu Ala Ala Arg Thr Arg Gln Leu Leu Glu 565
570 575 Asp Glu Ser Tyr Asp Gly Gly Ala
Gly Ala Ala Ser Ala Phe Ala Pro 580 585
590 Pro Arg Ser Ser Pro Cys Ala Ser Ser Thr Pro Val Ala
Val Gly Asp 595 600 605
Phe Pro Asp Cys Ala Tyr Pro Pro Asp Ala Glu Pro Lys Asp Asp Ala 610
615 620 Tyr Pro Leu Tyr
Ser Asp Phe Gln Pro Pro Ala Leu Lys Ile Lys Glu 625 630
635 640 Glu Glu Glu Gly Ala Glu Ala Ser Ala
Arg Ser Pro Arg Ser Tyr Leu 645 650
655 Val Ala Gly Ala Asn Pro Ala Ala Phe Pro Asp Phe Pro Leu
Gly Pro 660 665 670
Pro Pro Pro Leu Pro Pro Arg Ala Thr Pro Ser Arg Pro Gly Glu Ala
675 680 685 Ala Val Thr Ala
Ala Pro Ala Ser Ala Ser Val Ser Ser Ala Ser Ser 690
695 700 Ser Gly Ser Thr Leu Glu Cys Ile
Leu Tyr Lys Ala Glu Gly Ala Pro 705 710
715 720 Pro Gln Gln Gly Pro Phe Ala Pro Pro Pro Cys Lys
Ala Pro Gly Ala 725 730
735 Ser Gly Cys Leu Leu Pro Arg Asp Gly Leu Pro Ser Thr Ser Ala Ser
740 745 750 Ala Ala Ala
Ala Gly Ala Ala Pro Ala Leu Tyr Pro Ala Leu Gly Leu 755
760 765 Asn Gly Leu Pro Gln Leu Gly Tyr
Gln Ala Ala Val Leu Lys Glu Gly 770 775
780 Leu Pro Gln Val Tyr Pro Pro Tyr Leu Asn Tyr Leu Arg
Pro Asp Ser 785 790 795
800 Glu Ala Ser Gln Ser Pro Gln Tyr Ser Phe Glu Ser Leu Pro Gln Lys
805 810 815 Ile Cys Leu Ile
Cys Gly Asp Glu Ala Ser Gly Cys His Tyr Gly Val 820
825 830 Leu Thr Cys Gly Ser Cys Lys Val Phe
Phe Lys Arg Ala Met Glu Gly 835 840
845 Gln His Asn Tyr Leu Cys Ala Gly Arg Asn Asp Cys Ile Val
Asp Lys 850 855 860
Ile Arg Arg Lys Asn Cys Pro Ala Cys Arg Leu Arg Lys Cys Cys Gln 865
870 875 880 Ala Gly Met Val Leu
Gly Gly Arg Lys Phe Lys Lys Phe Asn Lys Val 885
890 895 Arg Val Val Arg Ala Leu Asp Ala Val Ala
Leu Pro Gln Pro Val Gly 900 905
910 Val Pro Asn Glu Ser Gln Ala Leu Ser Gln Arg Phe Thr Phe Ser
Pro 915 920 925 Gly
Gln Asp Ile Gln Leu Ile Pro Pro Leu Ile Asn Leu Leu Met Ser 930
935 940 Ile Glu Pro Asp Val Ile
Tyr Ala Gly His Asp Asn Thr Lys Pro Asp 945 950
955 960 Thr Ser Ser Ser Leu Leu Thr Ser Leu Asn Gln
Leu Gly Glu Arg Gln 965 970
975 Leu Leu Ser Val Val Lys Trp Ser Lys Ser Leu Pro Gly Phe Arg Asn
980 985 990 Leu His
Ile Asp Asp Gln Ile Thr Leu Ile Gln Tyr Ser Trp Met Ser 995
1000 1005 Leu Met Val Phe Gly
Leu Gly Trp Arg Ser Tyr Lys His Val Ser 1010 1015
1020 Gly Gln Met Leu Tyr Phe Ala Pro Asp Leu
Ile Leu Asn Glu Gln 1025 1030 1035
Arg Met Lys Glu Ser Ser Phe Tyr Ser Leu Cys Leu Thr Met Trp
1040 1045 1050 Gln Ile
Pro Gln Glu Phe Val Lys Leu Gln Val Ser Gln Glu Glu 1055
1060 1065 Phe Leu Cys Met Lys Val Leu
Leu Leu Leu Asn Thr Ile Pro Leu 1070 1075
1080 Glu Gly Leu Arg Ser Gln Thr Gln Phe Glu Glu Met
Arg Ser Ser 1085 1090 1095
Tyr Ile Arg Glu Leu Ile Lys Ala Ile Gly Leu Arg Gln Lys Gly 1100
1105 1110 Val Val Ser Ser Ser
Gln Arg Phe Tyr Gln Leu Thr Lys Leu Leu 1115 1120
1125 Asp Asn Leu His Asp Leu Val Lys Gln Leu
His Leu Tyr Cys Leu 1130 1135 1140
Asn Thr Phe Ile Gln Ser Arg Ala Leu Ser Val Glu Phe Pro Glu
1145 1150 1155 Met Met
Ser Glu Val Ile Ala Ala Gln Leu Pro Lys Ile Leu Ala 1160
1165 1170 Gly Met Val Lys Pro Leu Leu
Phe His Lys Lys 1175 1180
1522DNAArtificial SequenceSynthetic primer 15cttctggcaa tggcaaggac tc
221624DNAArtificial
SequenceSynthetic primer 16cagcatcatg ccatcataca caca
241722DNAArtificial SequenceSynthetic primer
17tgtcaccagg gatgagagac gg
221823DNAArtificial SequenceSynthetic primer 18tccaaatcac acctctccag gag
231923DNAArtificial
SequenceSynthetic primer 19acctctcatt tcttgcagtt ccg
232024DNAArtificial SequenceSynthetic primer
20caggatggag gtgacattgt agct
242121DNAArtificial SequenceSynthetic primer 21agtgtgacgt tgacatccgt a
212222DNAArtificial
SequenceSynthetic primer 22gccagagcag taatctcctt ct
222320DNAArtificial SequenceSynthetic primer
23tgacagtggg ctgtttgttc
202420DNAArtificial SequenceSynthetic primer 24aagaccccaa gtgcctttct
202520DNAArtificial
SequenceSynthetic primer 25tttcaaggat gctggctctt
202620DNAArtificial SequenceSynthetic primer
26ggcctcagct tgtcaaattc
202721DNAArtificial SequenceSynthetic primer 27aaggtgaagg tcggagtcaa c
212822DNAArtificial
SequenceSynthetic primer 28ggggtcattg atggcaacaa ta
22
User Contributions:
Comment about this patent or add new information about this topic: