Patent application title: COMPOSITIONS AND METHODS FOR USE IN RECOMBINATIONAL CLONING OF NUCLEIC ACIDS
Inventors:
James Hartley (Frederick, MD, US)
Michael Brasch (Gaithersburg, MD, US)
Gary Temple (Washington, MD, US)
David Cheo (Kensington, MD, US)
David Cheo (Kensington, MD, US)
IPC8 Class: AC12N1511FI
USPC Class:
435 912
Class name: Nucleotide polynucleotide (e.g., nucleic acid, oligonucleotide, etc.) acellular exponential or geometric amplification (e.g., pcr, etc.)
Publication date: 2015-04-02
Patent application number: 20150093787
Abstract:
The present invention relates generally to compositions and methods for
use in recombinational cloning of nucleic acid molecules. In particular,
the invention relates to nucleic acid molecules encoding one or more
recombination sites or portions thereof, to nucleic acid molecules
comprising one or more of these recombination site nucleotide sequences
and optionally comprising one or more additional physical or functional
nucleotide sequences. The invention also relates to vectors comprising
the nucleic acid molecules of the invention, to host cells comprising the
vectors or nucleic acid molecules of the invention, to methods of
producing polypeptides using the nucleic acid molecules of the invention,
and to polypeptides encoded by these nucleic acid molecules or produced
by the methods of the invention. The invention also relates to antibodies
that bind to one or more polypeptides of the invention or epitopes
thereof. The invention also relates to the use of these compositions in
methods for recombinational cloning of nucleic acids, in vitro and in
vivo, to provide chimeric DNA molecules that have particular
characteristics and/or DNA segments.Claims:
1. An isolated nucleic acid molecule comprising a nucleotide sequence
selected from the group of nucleotide sequences consisting of an attB1
nucleotide sequence as set forth in FIG. 9, an attB2 nucleotide sequence
as set forth in FIG. 9, an attP1 nucleotide sequence as set forth in FIG.
9, an attP2 nucleotide sequence as set forth in FIG. 9, an attL1
nucleotide sequence as set forth in FIG. 9, an attL2 nucleotide sequence
as set forth in FIG. 9, an attR1 nucleotide sequence as set forth in FIG.
9, an attR2 nucleotide sequence as set forth in FIG. 9, a polynucleotide
complementary thereto, and a mutant, fragment, or derivative thereof.
2-9. (canceled)
10. The isolated nucleic acid molecule of claim 1, further comprising one or more functional or structural nucleotide sequences selected from the group consisting of one or more multiple cloning sites, one or more localization signals, one or more transcription termination sites, one or more transcriptional regulatory sequences, one or more translational signals, one or more origins of replication, one or more fusion partner peptide-encoding nucleic acid molecules, one or more protease cleavage sites, and one or more 5' polynucleotide extensions.
11. The nucleic acid molecule of claim 10, wherein said transcriptional regulatory sequence is a promoter, an enhancer, or a repressor.
12. The nucleic acid molecule of claim 10, wherein said fusion partner peptide-encoding nucleic acid molecule encodes glutathione S-transferase (GST), hexahistidine (HiS6) or thioredoxin (Trx).
13. The nucleic acid molecule of claim 10, wherein said 5' polynucleotide extension consists of from one to five nucleotide bases.
14. The nucleic acid molecule of claim 13, wherein said 5' polynucleotide extension consists of four or five guanine nucleotide bases.
15. A primer nucleic acid molecule suitable for amplifying a target nucleotide sequence, comprising the isolated nucleic acid molecule of claim for a portion thereof linked to a target-specific nucleotide sequence useful in amplifying said target nucleotide sequence.
16. The primer nucleic acid molecule of claim 15, wherein said primer comprises an attB1 nucleotide sequence having the sequence shown in FIG. 9 or a portion thereof, or a polynucleotide complementary to the sequence shown in FIG. 9 or a portion thereof.
17. The primer nucleic acid molecule of claim 15, wherein said primer comprises an attB2 nucleotide sequence having the sequence shown in FIG. 9 or a portion thereof, or a polynucleotide complementary to the sequence shown in FIG. 9 or a portion thereof.
18. The primer nucleic acid molecule of claim 15, further comprising a 5' terminal extension of four or five guanine bases.
19. A vector comprising the isolated nucleic acid molecule of claim 1.
20. The vector of claim 19, wherein said vector is an Expression Vector.
21. (canceled)
22. A method of synthesizing or amplifying one or more nucleic acid molecules comprising: (a) mixing one or more nucleic acid templates with at least one polypeptide having polymerase or reverse transcriptase activity and at least a first primer comprising a template-specific sequence that is complementary to or capable of hybridizing to said templates and at least a second primer comprising all or a portion of a recombination site wherein said at least a portion of said second primer is homologous to or complementary to at least a portion of said first primer; and (b) incubating said mixture under conditions sufficient to synthesize or amplify one or more nucleic acid molecules complementary to all or a portion of said templates and comprising one or more recombination sites or portions thereof at one or both termini of said molecules.
23-25. (canceled)
26. An isolated nucleic acid molecule comprising one or more att recombination sites comprising at least one mutation in its core region that increases the specificity of interaction between said recombination site and a second att recombination site.
27. The isolated nucleic acid molecule of claim 26, wherein said mutation is at least one substitution mutation of at least one nucleotide in the seven basepair overlap region of said core region of said recombination site.
28. The isolated nucleic acid molecule of claim 26, wherein said nucleic acid molecule comprises the sequence NNNATAC, wherein "N" refers to any nucleotide with the proviso that if one of the first three nucleotides in the consensus sequence is a TIU, then at least one of the other two of the first three nucleotides is not a TIU.
29. An isolated nucleic acid molecule comprising one or more mutated att recombination sites comprising at least one mutation in its core region that enhances the efficiency of recombination between a first nucleic acid molecule comprising said mutated att recombination site and a second nucleic acid molecule comprising a second recombination site that interacts with said mutated att recombination site.
30. The isolated nucleic acid molecule of claim 29, wherein said mutated att recombination site is a mutated attL site comprising a core region having the nucleotide sequence caacttnntnnnannaagttg (SEQID NO:92), wherein "n" represents any nucleotide.
31. The isolated nucleic acid molecule of claim 30, wherein said mutated attL recombination site comprises a core region having a nucleotide sequence selected from agcctgctttattatactaagttggcatta (attL5; SEQ ID NO:87) and agcctgcttttttatattaagttggcatta (attL6; SEQ ID NO:88).
32. The isolated nucleic acid molecule of claim 29, wherein said mutated att recombination site comprises a core region having a nucleotide sequence selected from the group consisting of ggggacaactttgtacaaaaaagttggct (attB1.6; SEQ ID NO:105), ggggacaactttgtacaagaaagctgggt (attB2.2; SEQ ID NO:97), and ggggacaactttgtacaagaaagttgggt (attB2.10; SEQ ID NO: 107).
33-38. (canceled)
Description:
CROSS REFERENCE TO RELATED APPLICATIONS
[0001] The present application claims the benefit of the filing dates of U.S. Provisional Application Nos. 60/122,389, filed Mar. 2, 1999, 60/126,049, filed Mar. 23, 1999, and 60/136,7844, filed May 28, 1999. The present application is also related to U.S. application Ser. No. 08/486,139, filed Jun. 7, 1995 (now abandoned), Ser. No. 08/663,002, filed Jun. 7, 1996 (now U.S. Pat. No. 5,888,732), Ser. No. 09/005,476, filed Jan. 12, 1998, Ser. No. 09/177,387, filed Oct. 23, 1998, Ser. No. 09/233,492, filed Jan. 20, 1999, Ser. No. 09/233,493, filed Jan. 20, 1999, Ser. No. 09/296,280, filed Apr. 22, 1999, Ser. No. 09/296,281, filed Apr. 22, 1999, Ser. No. 09/432,085, filed Nov. 2, 1999, and Ser. No. 09/438,358, filed Nov. 12, 1999. The disclosures of all of the applications cross-referenced above are incorporated by reference herein in their entireties.
BACKGROUND OF THE INVENTION
[0002] 1. Field of the Invention
[0003] The present invention relates generally to recombinant DNA technology. More particularly, the present invention relates to compositions and methods for use in recombinational cloning of nucleic acid molecules. The invention relates specifically to nucleic acid molecules encoding one or more recombination sites or one or more partial recombination sites, particularly attB, attP, attL, and attR, and fragments, mutants, variants and derivatives thereof. The invention also relates to such nucleic acid molecules wherein the one or more recombination site nucleotide sequences is operably linked to the one or more additional physical or functional nucleotide sequences. The invention also relates to vectors comprising the nucleic acid molecules of the invention, to host cells comprising the vectors or nucleic acid molecules of the invention, to methods of producing polypeptides and RNAs encoded by the nucleic acid molecules of the invention, and to polypeptides encoded by these nucleic acid molecules or produced by the methods of the invention, which may be fusion proteins. The invention also relates to antibodies that bind to one or more polypeptides of the invention or epitopes thereof, which may be monoclonal or polyclonal antibodies. The invention also relates to the use of these nucleic acid molecules, vectors, polypeptides and antibodies in methods for recombinational cloning of nucleic acids, in vitro and in vivo, to provide chimeric DNA molecules that have particular characteristics and/or DNA segments. More particularly, the antibodies of the invention may be used to identify and/or purify proteins or fusion proteins encoded by the nucleic acid molecules or vectors of the invention, or to identify and/or purify the nucleic acid molecules of the invention.
[0004] 2. Related Art
[0005] Site-Specific Recombinases.
[0006] Site-specific recombinases are proteins that are present in many organisms (e.g. viruses and bacteria) and have been characterized to have both endonuclease and ligase properties. These recombinases (along with associated proteins in some cases) recognize specific sequences of bases in DNA and exchange the DNA segments flanking those segments. The recombinases and associated proteins are collectively referred to as "recombination proteins" (see, e.g., Landy, A., Current Opinion in Biotechnology 3:699-707 (1993)).
[0007] Numerous recombination systems from various organisms have been described. See, e.g., Hoess et al., Nucleic Acids Research 14(6):2287 (1986); Abremski et al., J. Biol. Chem. 261(1): 391 (1986); Campbell, J. Bacteriol. 174(23):7495 (1992); Qian et al., J. Biol. Chem. 267(11):7794 (1992); Araki et al., J. Mol. Biol. 225(1):25 (1992); Maeser and Kahnmann Mol. Gen. Genet. 230:170-176) (1991); Esposito et al., Nucl. Acids Res. 25(18):3605 (1997).
[0008] Many of these belong to the integrase family of recombinases (Argos et al. EMBO J. 5:433-440 (1986); Voziyanov et al., Nucl. Acids Res. 27:930 (1999)). Perhaps the best studied of these are the Integrase/att system from bacteriophage λ (Landy, A. Current Opinions in Genetics and Devel. 3:699-707 (1993)), the Cre/loxP system from bacteriophage P1 (Hoess and Abremski (1990) In Nucleic Acids and Molecular Biology, vol. 4. Eds.: Eckstein and Lilley, Berlin-Heidelberg: Springer-Verlag; pp. 90-109), and the FLP/FRT system from the Saccharomyces cerevisiae 2μ circle plasmid (Broach et al. Cell 29:227-234 (1982)).
[0009] Backman (U.S. Pat. No. 4,673,640) discloses the in vivo use of λ recombinase to recombine a protein producing DNA segment by enzymatic site-specific recombination using wild-type recombination sites attB and attP.
[0010] Hasan and Szybalski (Gene 56:145-151 (1987)) discloses the use of λ Int recombinase in vivo for intramolecular recombination between wild type attP and attB sites which flank a promoter. Because the orientations of these sites are inverted relative to each other, this causes an irreversible flipping of the promoter region relative to the gene of interest.
[0011] Palazzolo et al. Gene 88:25-36 (1990), discloses phage lambda vectors having bacteriophage λ arms that contain restriction sites positioned outside a cloned DNA sequence and between wild-type loxP sites. Infection of E. coli cells that express the Cre recombinase with these phage vectors results in recombination between the loxP sites and the in vivo excision of the plasmid replicon, including the cloned cDNA.
[0012] Posfai et al. (Nucl. Acids Res. 22:2392-2398 (1994)) discloses a method for inserting into genomic DNA partial expression vectors having a selectable marker, flanked by two wild-type FRT recognition sequences. FLP site-specific recombinase as present in the cells is used to integrate the vectors into the genome at predetermined sites. Under conditions where the replicon is functional, this cloned genomic DNA can be amplified.
[0013] Bebee et al. (U.S. Pat. No. 5,434,066) discloses the use of site-specific recombinases such as Cre for DNA containing two loxP sites for in vivo recombination between the sites.
[0014] Boyd (Nucl. Acids Res. 21:817-821 (1993)) discloses a method to facilitate the cloning of blunt-ended DNA using conditions that encourage intermolecular ligation to a dephosphorylated vector that contains a wild-type loxP site acted upon by a Cre site-specific recombinase present in E. coli host cells.
[0015] Waterhouse et al. (WO 93/19172 and Nucleic Acids Res. 21 (9):2265 (1993)) disclose an in vivo method where light and heavy chains of a particular antibody were cloned in different phage vectors between loxP and loxP 511 sites and used to transfect new E. coli cells. Cre, acting in the host cells on the two parental molecules (one plasmid, one phage), produced four products in equilibrium: two different cointegrates (produced by recombination at either loxP or loxP 511 sites), and two daughter molecules, one of which was the desired product.
[0016] Schlake & Bode (Biochemistry 33:12746-12751 (1994)) discloses an in vivo method to exchange expression cassettes at defined chromosomal locations, each flanked by a wild type and a spacer-mutated FRT recombination site. A double-reciprocal crossover was mediated in cultured mammalian cells by using this FLP/FRT system for site-specific recombination.
[0017] Hartley et al. (U.S. Pat. No. 5,888,732) disclose compositions and methods for recombinational exchange of nucleic acid segments and molecules, including for use in recombinational cloning of a variety of nucleic acid molecules in vitro and in vivo, using a combination of wildtype and mutated recombination sites and recombination proteins.
[0018] Transposases.
[0019] The family of enzymes, the transposases, has also been used to transfer genetic information between replicons. Transposons are structurally variable, being described as simple or compound, but typically encode the recombinase gene flanked by DNA sequences organized in inverted orientations. Integration of transposons can be random or highly specific. Representatives such as Tn7, which are highly site-specific, have been applied to the in vivo movement of DNA segments between replicons (Lucklow et al., J. Virol. 67:4566-4579 (1993)).
[0020] Devine and Boeke Nucl. Acids Res. 22:3765-3772 (1994), discloses the construction of artificial transposons for the insertion of DNA segments, in vitro, into recipient DNA molecules. The system makes use of the integrase of yeast TY1 virus-like particles. The DNA segment of interest is cloned, using standard methods, between the ends of the transposon-like element TY1. In the presence of the TY1 integrase, the resulting element integrates randomly into a second target DNA molecule.
[0021] Recombination Sites.
[0022] Also key to the integration/recombination reactions mediated by the above-noted recombination proteins and/or transposases are recognition sequences, often termed "recombination sites," on the DNA molecules participating in the integration/recombination reactions. These recombination sites are discrete sections or segments of DNA on the participating nucleic acid molecules that are recognized and bound by the recombination proteins during the initial stages of integration or recombination. For example, the recombination site for Cre recombinase is loxP which is a 34 base pair sequence comprised of two 13 base pair inverted repeats (serving as the recombinase binding sites) flanking an 8 base pair core sequence. See FIG. 1 of Sauer, B., Curr. Opin. Biotech. 5:521-527 (1994). Other examples of recognition sequences include the attB, attP, attL, and attR sequences which are recognized by the recombination protein λ Int. attB is an approximately 25 base pair sequence containing two 9 base pair core-type Int binding sites and a 7 base pair overlap region, while attP is an approximately 240 base pair sequence containing core-type Int binding sites and arm-type Int binding sites as well as sites for auxiliary proteins integration host factor (IHF), FIS and excisionase (Xis). See Landy, Curr. Opin. Biotech. 3:699-707 (1993); see also U.S. Pat. No. 5,888,732, which is incorporated by reference herein.
[0023] DNA Cloning.
[0024] The cloning of DNA segments currently occurs as a daily routine in many research labs and as a prerequisite step in many genetic analyses. The purpose of these clonings is various, however, two general purposes can be considered: (1) the initial cloning of DNA from large DNA or RNA segments (chromosomes, YACs, PCR fragments, mRNA, etc.), done in a relative handful of known vectors such as pUC, pGem, pBlueScript, and (2) the subcloning of these DNA segments into specialized vectors for functional analysis. A great deal of time and effort is expended both in the transfer of DNA segments from the initial cloning vectors to the more specialized vectors. This transfer is called subcloning.
[0025] The basic methods for cloning have been known for many years and have changed little during that time. A typical cloning protocol is as follows:
[0026] (1) digest the DNA of interest with one or two restriction enzymes;
[0027] (2) gel purify the DNA segment of interest when known;
[0028] (3) prepare the vector by cutting with appropriate restriction enzymes, treating with alkaline phosphatase, gel purify etc., as appropriate;
[0029] (4) ligate the DNA segment to the vector, with appropriate controls to eliminate background of uncut and self-ligated vector;
[0030] (5) introduce the resulting vector into an E. coli host cell;
[0031] (6) pick selected colonies and grow small cultures overnight;
[0032] (7) make DNA minipreps; and
[0033] (8) analyze the isolated plasmid on agarose gels (often after diagnostic restriction enzyme digestions) or by PCR.
[0034] The specialized vectors used for subcloning DNA segments are functionally diverse. These include but are not limited to: vectors for expressing nucleic acid molecules in various organisms; for regulating nucleic acid molecule expression; for providing tags to aid in protein purification or to allow tracking of proteins in cells; for modifying the cloned DNA segment (e.g., generating deletions); for the synthesis of probes (e.g., riboprobes); for the preparation of templates for DNA sequencing; for the identification of protein coding regions; for the fusion of various protein-coding regions; to provide large amounts of the DNA of interest, etc. It is common that a particular investigation will involve subcloning the DNA segment of interest into several different specialized vectors.
[0035] As known in the art, simple subclonings can be done in one day (e.g., the DNA segment is not large and the restriction sites are compatible with those of the subcloning vector). However, many other subclonings can take several weeks, especially those involving unknown sequences, long fragments, toxic genes, unsuitable placement of restriction sites, high backgrounds, impure enzymes, etc. Subcloning DNA fragments is thus often viewed as a chore to be done as few times as possible.
[0036] Several methods for facilitating the cloning of DNA segments have been described, e.g., as in the following references.
[0037] Ferguson, J., et al. Gene 16:191 (1981), discloses a family of vectors for subcloning fragments of yeast DNA. The vectors encode kanamycin resistance. Clones of longer yeast DNA segments can be partially digested and ligated into the subcloning vectors. If the original cloning vector conveys resistance to ampicillin, no purification is necessary prior to transformation, since the selection will be for kanamycin.
[0038] Hashimoto-Gotoh, T., et al. Gene 41:125 (1986), discloses a subcloning vector with unique cloning sites within a streptomycin sensitivity gene; in a streptomycin-resistant host, only plasmids with inserts or deletions in the dominant sensitivity gene will survive streptomycin selection.
[0039] Accordingly, traditional subcloning methods, using restriction enzymes and ligase, are time consuming and relatively unreliable. Considerable labor is expended, and if two or more days later the desired subclone can not be found among the candidate plasmids, the entire process must then be repeated with alternative conditions attempted. Although site specific recombinases have been used to recombine DNA in vivo, the successful use of such enzymes in vitro was expected to suffer from several problems. For example, the site specificities and efficiencies were expected to differ in vitro; topologically linked products were expected; and the topology of the DNA substrates and recombination proteins was expected to differ significantly in vitro (see, e.g., Adams et al, J. Mol. Biol. 226:661-73 (1992)). Reactions that could go on for many hours in vivo were expected to occur in significantly less time in vitro before the enzymes became inactive. In addition, the stabilities of the recombination enzymes after incubation for extended periods of time in in vitro reactions was unknown, as were the effects of the topologies (i.e., linear, coiled, supercoiled, etc.) of the nucleic acid molecules involved in the reaction. Multiple DNA recombination products were expected in the biological host used, resulting in unsatisfactory reliability, specificity or efficiency of subcloning. Thus, in vitro recombination reactions were not expected to be sufficiently efficient to yield the desired levels of product.
[0040] Accordingly, there is a long felt need to provide an alternative subcloning system that provides advantages over the known use of restriction enzymes and ligases.
SUMMARY OF THE INVENTION
[0041] The present invention relates to nucleic acid molecules encoding one or more recombination sites or one or more partial recombination sites, particularly attB, attP, attL, and attR, and fragments, mutants, variants and derivatives thereof. The invention also relates to such nucleic acid molecules comprising one or more of the recombination site nucleotide sequences or portions thereof and one or more additional physical or functional nucleotide sequences, such as those encoding one or more multiple cloning sites, one or more transcription termination sites, one or more transcriptional regulatory sequences (e.g., one or more promoters, enhancers, or repressors), one or more translational signal sequences, one or more nucleotide sequences encoding a fusion partner protein or peptide (e.g., GST, His6 or thioredoxin), one or more selection markers or modules, one or more nucleotide sequences encoding localization signals such as nuclear localization signals or secretion signals, one or more origins of replication, one or more protease cleavage sites, one or more desired proteins or peptides encoded by a gene or a portion of a gene, and one or more 5' or 3' polynucleotide tails (particularly a poly-G tail). The invention also relates to such nucleic acid molecules wherein the one or more recombination site nucleotide sequences is operably linked to the one or more additional physical or functional nucleotide sequences.
[0042] The invention also relates to primer nucleic acid molecules comprising the recombination site nucleotide sequences of the invention (or portions thereof), and to such primer nucleic acid molecules linked to one or more target-specific (e.g., one or more gene-specific) primer nucleic acid sequences. Such primers may also comprise sequences complementary or homologous to DNA or RNA sequences to be amplified, e.g., by PCR, RT-PCR, etc. Such primers may also comprise sequences or portions of sequences useful in the expression of protein genes (ribosome binding sites, localization signals, protease cleavage sites, repressor binding sites, promoters, transcription stops, stop codons, etc.). Said primers may also comprise sequences or portions of sequences useful in the manipulation of DNA molecules (restriction sites, transposition sites, sequencing primers, etc.). The primers of the invention may be used in nucleic acid synthesis and preferably are used for amplification (e.g., PCR) of nucleic acid molecules. When the primers of the invention include target- or gene-specific sequences (any sequence contained within the target to be synthesized or amplified including translation signals, gene sequences, stop codons, transcriptional signals (e.g., promoters) and the like), amplification or synthesis of target sequences or genes may be accomplished. Thus, the invention relates to synthesis of a nucleic acid molecules comprising mixing one or more primers of the invention with a nucleic acid template, and incubating said mixture under conditions sufficient to make a first nucleic acid molecule complementary to all or a portion of said template. Thus, the invention relates specifically to a method of synthesizing a nucleic acid molecule comprising:
[0043] (a) mixing a nucleic acid template with a polypeptide having polymerase activity and one or more primers comprising one or more recombination sites or portions thereof; and
[0044] (b) incubating said mixture under conditions sufficient to synthesize a first nucleic acid molecule complementary to all or a portion of said template and which preferably comprises one or more recombination sites or portions thereof. Such method of the invention may further comprise incubating said first synthesized nucleic acid molecule under conditions sufficient to synthesize a second nucleic acid molecule complementary to all or a portion of said first nucleic acid molecule. Such synthesis may provide for a first nucleic acid molecule having a recombination site or portion thereof at one or both of its termini.
[0045] In a preferred aspect, for the synthesis of the nucleic acid molecules, at least two primers are used wherein each primer comprises a homologous sequence at its terminus and/or within internal sequences of each primer (which may have a homology length of about 2 to about 500 bases, preferably about 3 to about 100 bases, about 4 to about 50 bases, about 5 to about 25 bases and most preferably about 6 to about 18 base overlap). In a preferred aspect, the first such primer comprises at least one target-specific sequence and at least one recombination site or portion thereof while the second primer comprises at least one recombination site or portion thereof. Preferably, the homologous regions between the first and second primers comprise at least a portion of the recombination site. In another aspect, the homologous regions between the first and second primers may comprise one or more additional sequences, e.g., expression signals, translational start motifs, or other sequences adding functionality to the desired nucleic acid sequence upon amplification. In practice, two pairs of primers prime synthesis or amplification of a nucleic acid molecule. In a preferred aspect, all or at least a portion of the synthesized or amplified nucleic acid molecule will be homologous to all or a portion of the template and further comprises a recombination site or a portion thereof at least one terminus and preferably both termini of the synthesized or amplified molecule. Such synthesized or amplified nucleic acid molecule may be double stranded or single stranded and may be used in the recombinational cloning methods of the invention. The homologous primers of the invention provide a substantial advantage in that one set of the primers may be standardized for any synthesis or amplification reaction. That is, the primers providing the recombination site sequences (without the target specific sequences) can be pre-made and readily available for use. This in practice allows the use of shorter custom made primers that contain the target specific sequence needed to synthesize or amplify the desired nucleic acid molecule. Thus, this provides reduced time and cost in preparing target specific primers (e.g., shorter primers containing the target specific sequences can be prepared and used in synthesis reactions). The standardized primers, on the other hand, may be produced in mass to reduce cost and can be readily provided (e.g., in kits or as a product) to facilitate synthesis of the desired nucleic acid molecules.
[0046] Thus, in one preferred aspect, the invention relates to a method of synthesizing or amplifying one or more nucleic acid molecules comprising:
[0047] (a) mixing one or more nucleic acid templates with at least one polypeptide having polymerase or reverse transcriptase activity and at least a first primer comprising a template specific sequence (complementary to or capable of hybridizing to said templates) and at least a second primer comprising all or a portion of a recombination site wherein said at least a portion of said second primer is homologous to or complementary to at least a portion of said first primer; and
[0048] (b) incubating said mixture under conditions sufficient to synthesize or amplify one or more nucleic acid molecules complementary to all or a portion of said templates and comprising one or more recombination sites or portions thereof at one and preferably both termini of said molecules.
[0049] More specifically, the invention relates to a method of synthesizing or amplifying one or more nucleic acid molecules comprising:
[0050] (a) mixing one or more nucleic acid templates with at least one polypeptide having polymerase or reverse transcriptase activity and at least a first primer comprising a template specific sequence (complementary to or capable of hybridizing to said templates) and at least a portion of, a recombination site, and at least a second primer comprising all or a portion of a recombination site wherein said at least a portion of said recombination site on said second primer is complementary to or homologous to at least a portion of said recombination site on said first primer; and
[0051] (b) incubating said mixture under conditions sufficient to synthesize or amplify one or more nucleic acid molecules complementary to all or a portion of said templates and comprising one or more recombination sites or portions thereof at one and preferably both termini of said molecules.
[0052] In a more preferred aspect, the invention relates to a method of amplifying or synthesizing one or more nucleic acid molecules comprising:
[0053] (a) mixing one or more nucleic acid templates with at least one polypeptide having polymerase or reverse transcriptase activity and one or more first primers comprising at least a portion of a recombination site and a template specific sequence (complementary to or capable of hybridizing to said template);
[0054] (b) incubating said mixture under conditions sufficient to synthesize or amplify one or more first nucleic acid molecules complementary to all or a portion of said templates wherein said molecules comprise at least a portion of a recombination site at one and preferably both termini of said molecules;
[0055] (c) mixing said molecules with one or more second primers comprising one or more recombination sites, wherein said recombination sites of said second primers are homologous to or complementary to at least a portion of said recombination sites on said first nucleic acid molecules; and
[0056] (d) incubating said mixture under conditions sufficient to synthesize or amplify one or more second nucleic acid molecules complementary to all or a portion of said first nucleic acid molecules and which comprise one or more recombination sites at one and preferably both termini of said molecules.
[0057] The invention also relates to vectors comprising the nucleic acid molecules of the invention, to host cells comprising the vectors or nucleic acid molecules of the invention, to methods of producing polypeptides encoded by the nucleic acid molecules of the invention, and to polypeptides encoded by these nucleic acid molecules or produced by the methods of the invention, which may be fusion proteins. The invention also relates to antibodies that bind to one or more polypeptides of the invention or epitopes thereof, which may be monoclonal or polyclonal antibodies. The invention also relates to the use of these nucleic acid molecules, primers, vectors, polypeptides and antibodies in methods for recombinational cloning of nucleic acids, in vitro and in vivo, to provide chimeric DNA molecules that have particular characteristics and/or DNA segments.
[0058] The antibodies of the invention may have particular use to identify and/or purify peptides or proteins (including fusion proteins produced by the invention), and to identify and/or purify the nucleic acid molecules of the invention or portions thereof.
[0059] The methods for in vitro or in vivo recombinational cloning of nucleic acid molecule generally relate to recombination between at least a first nucleic acid molecule having at least one recombination site and a second nucleic acid molecule having at least one recombination site to provide a chimeric nucleic acid molecule. In one aspect, the methods relate to recombination between and first vector having at least one recombination site and a second vector having at least one recombination site to provide a chimeric vector. In another aspect, a nucleic acid molecule having at least one recombination site is combined with a vector having at least one recombination site to provide a chimeric vector. In a most preferred aspect, the nucleic acid molecules or vectors used in recombination comprise two or more recombination sites. In a more specific embodiment of the invention, the recombination methods relate to a Destination Reaction (also referred to herein as an "LR reaction") in which recombination occurs between an Entry clone and a Destination Vector. Such a reaction transfers the nucleic acid molecule of interest from the Entry Clone into the Destination Vector to create an Expression Clone. The methods of the invention also specifically relate to an Entry or Gateward reaction (also referred to herein as a "BP reaction") in which an Expression Clone is recombined with a Donor vector to produce an Entry clone. In other aspects, the invention relates to methods to prepare Entry clones by combining an Entry vector with at least one nucleic acid molecule (e.g., gene or portion of a gene). The invention also relates to conversion of a desired vector into a Destination Vector by including one or more (preferably at least two) recombination sites in the vector of interest. In a more preferred aspect, a nucleic acid molecule (e.g., a cassette) having at least two recombination sites flanking a selectable marker (e.g., a toxic gene or a genetic element preventing the survival of a host cell containing that gene or element, and/or preventing replication, partition or heritability of a nucleic acid molecule (e.g., a vector or plasmid) comprising that gene or element) is added to the vector to make a Destination Vector of the invention.
[0060] Preferred vectors for use in the invention include prokaryotic vectors, eukaryotic vectors, or vectors which may shuttle between various prokaryotic and/or eukaryotic systems (e.g. shuttle vectors). Preferred prokaryotic vectors for use in the invention include but are not limited to vectors which may propagate and/or replicate in gram negative and/or gram positive bacteria, including bacteria of the genera Escherichia, Salmonella, Proteus, Clostridium, Klebsiella, Bacillus, Streptomyces, and Pseudomonas and preferably in the species E. coli. Eukaryotic vectors for use in the invention include vectors which propagate and/or replicate and yeast cells, plant cells, mammalian cells, (particularly human and mouse), fungal cells, insect cells, nematode cells, fish cells and the like. Particular vectors of interest include but are not limited to cloning vectors, sequencing vectors, expression vectors, fusion vectors, two-hybrid vectors, gene therapy vectors, phage display vectors, gene-targeting vectors, PACs, BACs, YACs, MACs, and reverse two-hybrid vectors. Such vectors may be used in prokaryotic and/or eukaryotic systems depending on the particular vector.
[0061] In another aspect, the invention relates to kits which may be used in carrying out the methods of the invention, and more specifically relates to cloning or subcloning kits and kits for carrying out the LR Reaction (e.g., making an Expression Clone), for carrying out the BP Reaction (e.g., making an Entry Clone), and for making Entry Clone and Destination Vector molecules of the invention. Such kits may comprise a carrier or receptacle being compartmentalized to receive and hold therein any number of containers. Such containers may contain any number of components for carrying out the methods of the invention or combinations of such components. In particular, a kit of the invention may comprise one or more components (or combinations thereof) selected from the group consisting of one or more recombination proteins or auxiliary factors or combinations thereof, one or more compositions comprising one or more recombination proteins or auxiliary factors or combinations thereof (for example, GATEWAY® LR Clonase® Enzyme Mix or GATEWAY® BP Clonase® Enzyme Mix), one or more reaction buffers, one or more nucleotides, one or more primers of the invention, one or more restriction enzymes, one or more ligases, one or more polypeptides having polymerase activity (e.g., one or more reverse transcriptases or DNA polymerases), one or more proteinases (e.g., proteinase K or other proteinases), one or more Destination Vector molecules, one or more Entry Clone molecules, one or more host cells (e.g. competent cells, such as E. coli cells, yeast cells, animal cells (including mammalian cells, insect cells, nematode cells, avian cells, fish cells, etc.), plant cells, and most particularly E. coli DB3.1 host cells, such as E. coli LIBRARY EFFICIENCY® DB3.1® Competent Cells), instructions for using the kits of the invention (e.g., to carry out the methods of the invention), and the like. In related aspects, the kits of the invention may comprise one or more nucleic acid molecules encoding one or more recombination sites or portions thereof, particularly one or more nucleic acid molecules comprising a nucleotide sequence encoding the one or more recombination sites or portions thereof of the invention. Preferably, such nucleic acid molecules comprise at least two recombination sites which flank a selectable marker (e.g., a toxic gene and/or antibiotic resistance gene). In a preferred aspect, such nucleic acid molecules are in the form of a cassette (e.g., a linear nucleic acid molecule comprising one or more and preferably two or more recombination sites or portions thereof).
[0062] Kits for inserting or adding recombination sites to nucleic acid molecules of interest may comprise one or more nucleases (preferably restriction endonucleases), one or more ligases, one or more topoisomerases, one or more polymerases, and one or more nucleic acid molecules or adapters comprising one or more recombination sites. Kits for integrating recombination sites into one or more nucleic acid molecules of interest may comprise one or more components (or combinations thereof) selected from the group consisting of one or more integration sequences comprising one or more recombination sites. Such integration sequences may comprise one or more transposons, integrating viruses, homologous recombination sequences, RNA molecules, one or more host cells and the like.
[0063] Kits for making the Entry Clone molecules of the invention may comprise any or a number of components and the composition of such kits may vary depending on the specific method involved. Such methods may involve inserting the nucleic acid molecules of interest into an Entry or Donor Vector by the recombinational cloning methods of the invention, or using conventional molecular biology techniques (e.g., restriction enzyme digestion and ligation). In a preferred aspect, the Entry Clone is made using nucleic acid amplification or synthesis products. Kits for synthesizing Entry Clone molecules from amplification or synthesis products may comprise one or more components (or combinations thereof) selected from the group consisting of one or more Donor Vectors (e.g., one or more attP vectors including, but not limited to, pDONR201 (FIG. 49), pDONR202 (FIG. 50), pDONR203 (FIG. 51), pDONR204 (FIG. 52), pDONR205 (FIG. 53), pDONR206 (FIG. 53), and the like), one or more polypeptides having polymerase activity (preferably DNA polymerases and most preferably thermostable DNA polymerases), one or more proteinases, one or more reaction buffers, one or more nucleotides, one or more primers comprising one or more recombination sites or portions thereof, and instructions for making one or more Entry Clones.
[0064] Kits for making the Destination vectors of the invention may comprise any number of components and the compositions of such kits may vary depending on the specific method involved. Such methods may include the recombination methods of the invention or conventional molecular biology techniques (e.g., restriction endonuclease digestion and ligation). In a preferred aspect, the Destination vector is made by inserting a nucleic acid molecule comprising at least one recombination site (or portion thereof) of the invention (preferably a nucleic acid molecule comprising at least two recombination sites or portions thereof flanking a selectable marker) into a desired vector to convert the desired vector into a Destination vector of the invention. Such kits may comprise at least one component (or combinations thereof) selected from the group consisting of one or more restriction endonucleases, one or more ligases, one or more polymerases, one or more nucleotides, reaction buffers, one or more nucleic acid molecules comprising at least one recombination site or portion thereof (preferably at least one nucleic acid molecule comprising at least two recombination sites flanking at least one selectable marker, such as a cassette comprising at least one selectable marker such as antibiotic resistance genes and/or toxic genes), and instructions for making such Destination vectors.
[0065] The invention also relates to kits for using the antibodies of the invention in identification and/or isolation of peptides and proteins (which may be fusion proteins) produced by the nucleic acid molecules of the invention, and for identification and/or isolation of the nucleic acid molecules of the invention or portions thereof. Such kits may comprise one or more components (or combination thereof) selected from the group consisting of one or more antibodies of the invention, one or more detectable labels, one or more solid supports and the like.
[0066] Other preferred embodiments of the present invention will be apparent to one of ordinary skill in light of what is known in the art, in light of the following drawings and description of the invention, and in light of the claims.
BRIEF DESCRIPTION OF THE DRAWINGS
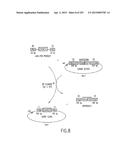
[0067] FIG. 1 depicts one general method of the present invention, wherein the starting (parent) DNA molecules can be circular or linear. The goal is to exchange the new subcloning vector D for the original cloning vector B. It is desirable in one embodiment to select for AD and against all the other molecules, including the Cointegrate. The square and circle are sites of recombination: e.g., lox (such as loxP) sites, att sites, etc. For example, segment D can contain expression signals, protein fusion domains, new drug markers, new origins of replication, or specialized functions for mapping or sequencing DNA. It should be noted that the cointegrate molecule contains Segment D (Destination vector) adjacent to segment A (Insert), thereby juxtaposing functional elements in D with the insert in A. Such molecules can be used directly in vitro (e.g., if a promoter is positioned adjacent to a gene-for in vitro transcription/translation) or in vivo (following isolation in a cell capable of propagating ccdB-containing vectors) by selecting for the selection markers in Segments B+D. As one skilled in the art will recognize, this single step method has utility in certain envisioned applications of the invention.
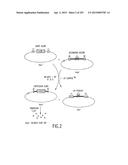
[0068] FIG. 2 is a more detailed depiction of the recombinational cloning system of the invention, referred to herein as the "GATEWAY® Cloning System." This figure depicts the production of Expression Clones via a "Destination Reaction," which may also be referred to herein as an "LR Reaction." A kanr vector (referred to herein as an "Entry clone") containing a DNA molecule of interest (e.g., a gene) localized between an attL1 site and an attL2 site is reacted with an ampr vector (referred to herein as a "Destination Vector") containing a toxic or "death" gene localized between an attR1 site and an attR2 site, in the presence of GATEWAY® LR Clonase® Enzyme Mix (a mixture of Int, IHF and Xis). After incubation at 25° C. for about 60 minutes, the reaction yields an ampr Expression Clone containing the DNA molecule of interest localized between an attB1 site and an attB2 site, and a kanr byproduct molecule, as well as intermediates. The reaction mixture may then be transformed into host cells (e.g., E. coli) and clones containing the nucleic acid molecule of interest may be selected by plating the cells onto ampicillin-containing media and picking ampr colonies.
[0069] FIG. 3 is a schematic depiction of the cloning of a nucleic acid molecule from an Entry clone into multiple types of Destination vectors, to produce a variety of Expression Clones. Recombination between a given Entry clone and different types of Destination vectors (not shown), via the LR Reaction depicted in FIG. 2, produces multiple different Expression Clones for use in a variety of applications and host cell types.
[0070] FIG. 4 is a detailed depiction of the production of Entry Clones via a "BP reaction," also referred to herein as an "Entry Reaction" or a "Gateward Reaction." In the example shown in this figure, an ampr expression vector containing a DNA molecule of interest (e.g., a gene) localized between an attB1 site and an attB2 site is reacted with a kanr Donor vector (e.g., an attP vector; here, GATEWAY® pDONR201 (see FIG. 49A-C)) containing a toxic or "death" gene localized between an attP1 site and an attP2 site, in the presence of GATEWAY® BP Clonase® Enzyme Mix (a mixture of Int and IHF). After incubation at 25° C. for about 60 minutes, the reaction yields a kanr Entry clone containing the DNA molecule of interest localized between an attL1 site and an attL2 site, and an ampr by-product molecule. The Entry clone may then be transformed into host cells (e.g., E. coli) and clones containing the Entry clone (and therefore the nucleic acid molecule of interest) may be selected by plating the cells onto kanamycin-containing media and picking kanr colonies. Although this figure shows an example of use of a kanr Donor vector, it is also possible to use Donor vectors containing other selection markers, such as the gentamycin resistance or tetracycline resistance markers, as discussed herein.
[0071] FIG. 5 is a more detailed schematic depiction of the LR ("Destination") reaction (FIG. 5A) and the BP ("Entry" or "Gateward") reaction (FIG. 5B) of the GATEWAY® Cloning System, showing the reactants, products and byproducts of each reaction.
[0072] FIG. 6 shows the sequences of the attB1 (SEQ ID NO:1) and attB2 (SEQ ID NO:2) sites flanking a gene of interest after subcloning into a Destination Vector to create an Expression Clone.
[0073] FIG. 7 is a schematic depiction of four ways to make Entry Clones using the compositions and methods of the invention: 1. using restriction enzymes and ligase; 2. starting with a cDNA library prepared in an attL Entry Vector; 3. using an Expression Clone from a library prepared in an attB Expression Vector via the BxP reaction; and 4. recombinational cloning of PCR fragments with terminal attB sites, via the BxP reaction. Approaches 3 and 4 rely on recombination with a Donor vector (here, an attP vector such as pDONR201 (see FIG. 49A-C), pDONR202 (see FIG. 50A-C), pDONR203 (see FIG. 51A-C), pDONR204 (see FIG. 52A-C), pDONR205 (see FIG. 53A-C), or pDONR206 (see FIG. 54A-C), for example) that provides an Entry Clone carrying a selection marker such as kanr, genr, tetr, or the like.
[0074] FIG. 8 is a schematic depiction of cloning of a PCR product by a BxP (Entry or Gateward) reaction. A PCR product with 25 bp terminal attB sites (plus four Gs) is shown as a substrate for the BxP reaction. Recombination between the attB-PCR product of a gene and a Donor vector (which donates an Entry Vector that carries kanr) results in an Entry Clone of the PCR product.
[0075] FIG. 9 is a listing of the nucleotide sequences of the recombination sites designated herein as attB1, attB2, attP1, attP2, attL1, attL2, attR1 and attR2 (SEQ ID NOs:1-8, respectively). Sequences are written conventionally, from 5' to 3'.
[0076] FIGS. 10-20: The plasmid backbone for all the Entry Vectors depicted herein is the same, and is shown in FIG. 10A for the Entry Vector pENTR1A. For other Entry Vectors shown in FIGS. 11-20, only the sequences shown in Figure "A" for each figure set (i.e., FIG. 11A, FIG. 12A, etc.) are different (within the attL1-attL2 cassettes) from those shown in FIG. 10--the plasmid backbone is identical.
[0077] FIG. 10 is a schematic depiction of the physical map and cloning sites (FIG. 10A), and the nucleotide sequence (FIG. 10B) (SEQ ID NO:118), of the Entry Vector pENTR1A.
[0078] FIG. 11 is a schematic depiction of the cloning sites (FIG. 11A) and the nucleotide sequence (FIG. 11B) (SEQ ID NO:119) of the Entry Vector pENTR2B.
[0079] FIG. 12 is a schematic depiction of the cloning sites (FIG. 12A) and the nucleotide sequence (FIG. 12B) (SEQ ID NO:120) of the Entry Vector pENTR3C.
[0080] FIG. 13 is a schematic depiction of the cloning sites (FIG. 13A) and the nucleotide sequence (FIG. 13B) (SEQ ID NO:121) of the Entry Vector pENTR4.
[0081] FIG. 14 is a schematic depiction of the cloning sites (FIG. 14A) and the nucleotide sequence (FIG. 14B) (SEQ ID NO:122) of the Entry Vector pENTR5.
[0082] FIG. 15 is a schematic depiction of the cloning sites (FIG. 15A) and the nucleotide sequence (FIG. 15B) (SEQ ID NO:123) of the Entry Vector pENTR6.
[0083] FIG. 16 is a schematic depiction of the cloning sites (FIG. 16A) and the nucleotide sequence (FIG. 16B) (SEQ ID NO:124) of the Entry Vector pENTR7.
[0084] FIG. 17 is a schematic depiction of the cloning sites (FIG. 17A) and the nucleotide sequence (FIG. 17B) (SEQ ID NO:125) of the Entry Vector pENTR8.
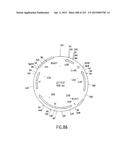
[0085] FIG. 18 is a schematic depiction of the cloning sites (FIG. 18A) and the nucleotide sequence (FIG. 18B) (SEQ ID NO:126) of the Entry Vector pENTR9.
[0086] FIG. 19 is a schematic depiction of the cloning sites (FIG. 19A) and the nucleotide sequence (FIG. 19B) (SEQ ID NO:127) of the Entry Vector pENTR10.
[0087] FIG. 20 is a schematic depiction of the cloning sites (FIG. 20A) and the nucleotide sequence (FIG. 20B) (SEQ ID NO:128) of the Entry Vector pENTR11.
[0088] FIG. 21 is a schematic depiction of the physical map and the Trc expression cassette (FIG. 21A) showing the promoter sequences at -35 and at -10 from the initiation codon, and the nucleotide sequence (FIG. 21B-D) (SEQ ID NO:129), of Destination Vector pDEST1. This vector may also be referred to as pTrc-DEST1.
[0089] FIG. 22 is a schematic depiction of the physical map and the His6 expression cassette (FIG. 22A) showing the promoter sequences at -35 and at -10 from the initiation codon, and the nucleotide sequence (FIG. 22B-D) (SEQ ID NO:130), of Destination Vector pDEST2. This vector may also be referred to as pHis6-DEST2.
[0090] FIG. 23 is a schematic depiction of the physical map and the GST expression cassette (FIG. 23A) showing the promoter sequences at -35 and at -10 from the initiation codon, and the nucleotide sequence (FIG. 23B-D) (SEQ ID NO:131), of Destination Vector pDEST3. This vector may also be referred to as pGST-DEST3.
[0091] FIG. 24 is a schematic depiction of the physical map and the His6-Trx expression cassette (FIG. 24A) showing the promoter sequences at -35 and at -10 from the initiation codon and a TEV protease cleavage site, and the nucleotide sequence (FIG. 24B-D) (SEQ ID NO:132), of Destination Vector pDEST4. This vector may also be referred to as pTrx-DEST4.
[0092] FIG. 25 is a schematic depiction of the attR1 and attR2 sites (FIG. 25A), the physical map (FIG. 25B), and the nucleotide sequence (FIGS. 25C-E) (SEQ ID NO:133), of Destination Vector pDEST5. This vector may also be referred to as pSPORT(+)-DEST5.
[0093] FIG. 26 is a schematic depiction of the attR1 and attR2 sites (FIG. 26A), the physical map (FIG. 26B), and the nucleotide sequence (FIGS. 26C-E) (SEQ ID NO:134), of Destination Vector pDEST6. This vector may also be referred to as pSPORT(-)-DEST6.
[0094] FIG. 27 is a schematic depiction of the attR1 site, CMV promoter, and the physical map (FIG. 27A), and the nucleotide sequence (FIGS. 27B-D) (SEQ ID NO:135), of Destination Vector pDEST7. This vector may also be referred to as pCMV-DEST7.
[0095] FIG. 28 is a schematic depiction of the attR1 site, baculovirus polyhedrin promoter, and the physical map (FIG. 28A), and the nucleotide sequence (FIG. 28B-D) (SEQ ID NO:136), of Destination Vector pDEST8. This vector may also be referred to as pFastBac-DEST8.
[0096] FIG. 29 is a schematic depiction of the attR1 site, Semliki Forest Virus promoter, and the physical map (FIG. 29A), and the nucleotide sequence (FIGS. 29B-G) (SEQ ID NO:137), of Destination Vector pDEST9. This vector may also be referred to as pSFV-DEST9.
[0097] FIG. 30 is a schematic depiction of the attR1 site, baculovirus polyhedrin promoter, His6 fusion domain, and the physical map (FIG. 30A), and the nucleotide sequence (FIG. 30B-D) (SEQ ID NO:138), of Destination Vector pDEST10. This vector may also be referred to as pFastBacHT-DEST10.
[0098] FIG. 31 is a schematic depiction of the attR1 cassette containing a tetracycline-regulated CMV promoter and the physical map (FIG. 31A), and the nucleotide sequence (FIGS. 31B-E) (SEQ ID NO:139), of Destination Vector pDEST11. This vector may also be referred to as pTet-DEST11.
[0099] FIG. 32 is a schematic depiction of the attR1 site, the start of the mRNA of the CMV promoter, and the physical map (FIG. 32A), and the nucleotide sequence (FIGS. 32B-E) (SEQ ID NO:140), of Destination Vector pDEST12.2. This vector may also be referred to as pCMVneo-DEST12, as pCMV-DEST12, or as pDEST12.
[0100] FIG. 33 is a schematic depiction of the attR1 site, the λPL promoter, and the physical map (FIG. 33A), and the nucleotide sequence (FIGS. 33B-D) (SEQ ID NO:141), of Destination Vector pDEST13. This vector may also be referred to as pλPL-DEST13.
[0101] FIG. 34 is a schematic depiction of the attR1 site, the T7 promoter, and the physical map (FIG. 34A), and the nucleotide sequence (FIG. 34B-D) (SEQ ID NO:142), of Destination Vector pDEST14. This vector may also be referred to as pPT7-DEST14.
[0102] FIG. 35 is a schematic depiction of the attR1 site, the T7 promoter, and the N-terminal GST fusion sequence, and the physical map (FIG. 35A), and the nucleotide sequence (FIG. 35B-D) (SEQ ID NO:143), of Destination Vector pDEST15. This vector may also be referred to as p17 GST-DEST15.
[0103] FIG. 36 is a schematic depiction of the attR1 site, the 17 promoter, and the N-terminal thioredoxin fusion sequence, and the physical map (FIG. 36A), and the nucleotide sequence (FIG. 36B-D) (SEQ ID NO:144), of Destination Vector pDEST16. This vector may also be referred to as pT7 Trx-DEST16.
[0104] FIG. 37 is a schematic depiction of the attR1 site, the 17 promoter, and the N-terminal His6 fusion sequence, and the physical map (FIG. 37A), and the nucleotide sequence (FIG. 37B-D) (SEQ ID NO:145), of Destination Vector pDEST17. This vector may also be referred to as pT7 His-DEST17.
[0105] FIG. 38 is a schematic depiction of the attR1 site and the p10 baculovirus promoter, and the physical map (FIG. 38A), and the nucleotide sequence (FIG. 38B-D) (SEQ ID NO:146), of Destination Vector pDEST18. This vector may also be referred to as pFBp10-DEST18.
[0106] FIG. 39 is a schematic depiction of the attR1 site, and the 39k baculovirus promoter, and the physical map (FIG. 39A), and the nucleotide sequence (FIG. 39B-D) (SEQ ID NO:147), of Destination Vector pDEST19. This vector may also be referred to as pFB39k-DEST19.
[0107] FIG. 40 is a schematic depiction of the attR1 site, the polh baculovirus promoter, and the N-terminal GST fusion sequence, and the physical map (FIG. 40A), and the nucleotide sequence (FIG. 40B-D) (SEQ ID NO:148), of Destination Vector pDEST20. This vector may also be referred to as pFB GST-DEST20.
[0108] FIG. 41 is a schematic depiction of a 2-hybrid vector with a DNA-binding domain, the attR1 site, and the ADH promoter, and the physical map (FIG. 41A), and the nucleotide sequence (FIGS. 41B-F) (SEQ ID NO:149), of Destination Vector pDEST21. This vector may also be referred to as pDB Leu-DEST21.
[0109] FIG. 42 is a schematic depiction of a 2-hybrid vector with an activation domain, the attR1 site, and the ADH promoter, and the physical map (FIG. 42A), and the nucleotide sequence (FIGS. 42B-E) (SEQ ID NO:150), of Destination Vector pDEST22. This vector may also be referred to as pPC86-DEST22.
[0110] FIG. 43 is a schematic depiction of the attR1 and attR2 sites, the T7 promoter, and the C-terminal His6 fusion sequence, and the physical map (FIG. 43A), and the nucleotide sequence (FIG. 43B-D) (SEQ ID NO:151), of Destination Vector pDEST23. This vector may also be referred to as pC-term-His6-DEST23.
[0111] FIG. 44 is a schematic depiction of the attR1 and attR2 sites, the T7 promoter, and the C-terminal GST fusion sequence, and the physical map (FIG. 44A), and the nucleotide sequence (FIG. 44B-D) (SEQ ID NO:152), of Destination Vector pDEST24. This vector may also be referred to as pC-term-GST-DEST24.
[0112] FIG. 45 is a schematic depiction of the attR1 and attR2 sites, the T7 promoter, and the C-terminal thioredoxin fusion sequence, and the physical map (FIG. 45A), and the nucleotide sequence (FIG. 45B-D) (SEQ ID NO:153), of Destination Vector pDEST25. This vector may also be referred to as pC-term-Trx-DEST25.
[0113] FIG. 46 is a schematic depiction of the attR1 site, the CMV promoter, and an N-terminal His6 fusion sequence, and the physical map (FIG. 46A), and the nucleotide sequence (FIG. 46B-D) (SEQ ID NO:154), of Destination Vector pDEST26. This vector may also be referred to as pCMV-SPneo-His-DEST26.
[0114] FIG. 47 is a schematic depiction of the attR1 site, the CMV promoter, and an N-terminal GST fusion sequence, and the physical map (FIG. 47A), and the nucleotide sequence (FIGS. 47B-E) (SEQ ID NO:155), of Destination Vector pDEST27. This vector may also be referred to as pCMV-Spneo-GST-DEST27.
[0115] FIG. 48 is a depiction of the physical map (FIG. 48A), the cloning sites (FIG. 48B), and the nucleotide sequence (FIG. 48C-D) (SEQ ID NO:156), for the attB cloning vector plasmid pEXP501. This vector may also be referred to equivalently herein as pCMV.SPORT6, pCMVSPORT6, and pCMVSport6.
[0116] FIG. 49 is a depiction of the physical map (FIG. 49A), and the nucleotide sequence (FIG. 49B-C) (SEQ ID NO:157), for the Donor plasmid pDONR201 which donates a kanamycin-resistant vector in the BP Reaction. This vector may also be referred to as pAttPkanr Donor Plasmid, or as pAttPkan Donor Plasmid
[0117] FIG. 50 is a depiction of the physical map (FIG. 50A), and the nucleotide sequence (FIG. 50B-C) (SEQ ID NO:158), for the Donor plasmid pDONR202 which donates a kanamycin-resistant vector in the BP Reaction.
[0118] FIG. 51 is a depiction of the physical map (FIG. 51A), and the nucleotide sequence (FIG. 51B-C) (SEQ ID NO:159), for the Donor plasmid pDONR203 which donates a kanamycin-resistant vector in the BP Reaction.
[0119] FIG. 52 is a depiction of the physical map (FIG. 52A), and the nucleotide sequence (FIG. 52B-C) (SEQ ID NO:160), for the Donor plasmid pDONR204 which donates a kanamycin-resistant vector in the BP Reaction.
[0120] FIG. 53 is a depiction of the physical map (FIG. 53A), and the nucleotide sequence (FIG. 53B-C) (SEQ ID NO:161), for the Donor plasmid pDONR205 which donates a tetracycline-resistant vector in the BP Reaction.
[0121] FIG. 54 is a depiction of the physical map (FIG. 54A), and the nucleotide sequence (FIG. 54B-C) (SEQ ID NO:162), for the Donor plasmid pDONR206 which donates a gentamycin-resistant vector in the BP Reaction. This vector may also be referred to as pENTR22 attP Donor Plasmid, pAttPGenr Donor Plasmid, or pAttPgent Donor Plasmid.
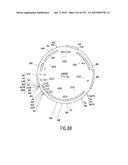
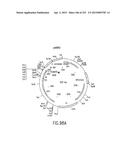
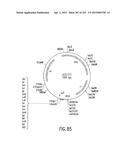
[0122] FIG. 55 depicts the attB1 site, and the physical map, of an Entry Clone (pENTR7) of CAT subcloned into the Destination Vector pDEST2 (FIGS. 22A-D)
[0123] FIG. 56 depicts the DNA components of Reaction B of the one-tube BxP reaction described in Example 16, pEZC7102 and attB-tet-PCR.
[0124] FIG. 57 is a physical map of the desired product of Reaction B of the one-tube BxP reaction described in Example 16, tetx7102.
[0125] FIG. 58 is a physical map of the Destination Vector pEZC8402.
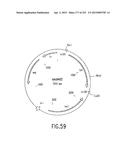
[0126] FIG. 59 is a physical map of the expected tetr subclone product, tetx8402, resulting from the LxR Reaction with tetx7102 (FIG. 57) plus pEZC8402 (FIG. 58).
[0127] FIG. 60 is a schematic depiction of the bacteriophage lambda recombination pathways in E. coli.
[0128] FIG. 61 is a schematic depiction of the DNA molecules participating in the LR Reaction. Two different co-integrates form during the LR Reaction (only one of which is shown here), depending on whether attL1 and attR1 or attL2 and attR2 are first to recombine. In one aspect, the invention provides directional cloning of a nucleic acid molecule of interest, since the recombination sites react with specificity (attL1 reacts with attR1; attL2 with attR2; attB1 with attP1; and attB2 with attP2). Thus, positioning of the sites allows construction of desired vectors having recombined fragments in the desired orientation.
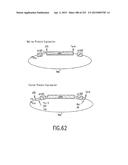
[0129] FIG. 62 is a depiction of native and fusion protein expression using the recombinational cloning methods and compositions of the invention. In the upper figure depicting native protein expression, all of the translational start signals are included between the attB1 and attB2 sites; therefore, these signals must be present in the starting Entry Clone. The lower figure depicts fusion protein expression (here showing expression with both N-terminal and C-terminal fusion tags so that ribosomes read through attB1 and attB2 to create the fusion protein). Unlike native protein expression vectors, N-terminal fusion vectors have their translational start signals upstream of the attB1 site.
[0130] FIG. 63 is a schematic depiction of three GATEWAY® Cloning System cassettes. Three blunt-ended cassettes are depicted which convert standard expression vectors to Destination Vectors. Each of the depicted cassettes provides amino-terminal fusions in one of three possible reading frames, and each has a distinctive restriction cleavage site as shown.
[0131] FIG. 64 shows the physical maps of plasmids containing three attR reading frame cassettes, pEZC15101 (reading frame A; FIG. 64A), pEZC15102 (reading frame B; Figure MB), and pEZC15103 (reading frame C; FIG. 64C).
[0132] FIG. 65 depicts the attB primers used for amplifying the tee and ampr genes from pBR322 bp the cloning methods of the invention.
[0133] FIG. 66 is a table listing the results of recombinational cloning of the tee and ampr PCR products made using the primers shown in FIG. 65.
[0134] FIG. 67 is a graph showing the effect of the number of guanines (G's) contained on the 5' end of the PCR primers on the cloning efficiency of PCR products. It is noted, however, that other nucleotides besides guanine (including A, T, C, U or combinations thereof) may be used as 5' extensions on the PCR primers to enhance cloning efficiency of PCR products.
[0135] FIG. 68 is a graph showing a titration of various amounts of attP and attB reactants in the BxP reaction, and the effects on cloning efficiency of PCR products.
[0136] FIG. 69 is a series of graphs showing the effects of various weights (FIG. 69A) or moles (FIG. 69B) of a 256 bp PCR product on formation of colonies, and on efficiency of cloning of the 256 bp PCR product into a Donor Vector (FIG. 69C).
[0137] FIG. 70 is a series of graphs showing the effects of various weights (FIG. 70A) or moles (FIG. 70B) of a 1 kb PCR product on formation of colonies, and on efficiency of cloning of the 1 kb PCR product into a Donor Vector (FIG. 70C).
[0138] FIG. 71 is a series of graphs showing the effects of various weights (FIG. 71A) or moles (FIG. 71B) of a 1.4 kb PCR product on formation of colonies, and on efficiency of cloning of the 1.4 kb PCR product into a Donor Vector (FIG. 71C).
[0139] FIG. 72 is a series of graphs showing the effects of various weights (FIG. 72A) or moles (FIG. 72B) of a 3.4 kb PCR product on formation of colonies, and on efficiency of cloning of the 3.4 kb PCR product into a Donor Vector (FIG. 72C).
[0140] FIG. 73 is a series of graphs showing the effects of various weights (FIG. 73A) or moles (FIG. 73B) of a 4.6 kb PCR product on formation of colonies, and on efficiency of cloning of the 4.6 kb PCR product into a Donor Vector (FIG. 73C).
[0141] FIG. 74 is photograph of an ethidium bromide-stained gel of a titration of a 6.9 kb PCR product in a BxP reaction.
[0142] FIG. 75 is a graph showing the effects of various amounts of a 10.1 kb PCR product on formation of colonies upon cloning of the 10.1 kb PCR product into a Donor Vector.
[0143] FIG. 76 is photograph of an ethidium bromide-stained gel of a titration of a 10.1 kb PCR product in a BxP reaction.
[0144] FIG. 77 is a table summarizing the results of the PCR product cloning efficiency experiments depicted in FIGS. 69-74, for PCR fragments ranging in size from 0.256 kb to 6.9 kb.
[0145] FIG. 78 is a depiction of the sequences at the ends of attR Cassettes (SEQ ID NOs:163-170). Sequences contributed by the Cmr-ccdB cassette are shown, including the outer ends of the flanking attR sites (boxed). The staggered cleavage sites for Int are indicated in the boxed regions. Following recombination with an Entry Clone, only the outer sequences in attR sites contribute to the resulting attB sites in the Expression Clone. The underlined sequences at both ends dictate the different reading frames (reading frames A, B, or C, with two alternative reading frame C cassettes depicted) for fusion proteins.
[0146] FIG. 79 is a depiction of several different attR cassettes (SEQ ID NOs:171-173) (in reading frames A, B, or C) which may provide fusion codons at the amino-terminus of the encoded protein.
[0147] FIG. 80 illustrates the single-cutting restriction sites in an attR reading frame A cassette of the invention.
[0148] FIG. 81 illustrates the single-cutting restriction sites in an attR reading frame B cassette of the invention.
[0149] FIG. 82 illustrates the single-cutting restriction sites in two alternative attR reading frame C cassettes of the invention (FIGS. 82A and 82B) depicted in FIG. 78.
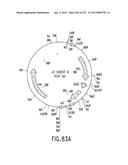
[0150] FIG. 83 shows the physical map (FIG. 83A), and the nucleotide sequence (FIG. 83B-C) (SEQ ID NO:174), for an attR reading frame C parent plasmid prfC Parent III, which contains an attR reading frame C cassette of the invention (alternative A in FIGS. 78 and 82).
[0151] FIG. 84 is a physical map of plasmid pEZC1301.
[0152] FIG. 85 is a physical map of plasmid pEZC1313.
[0153] FIG. 86 is a physical map of plasmid pEZ14032.
[0154] FIG. 87 is a physical map of plasmid pMAB58.
[0155] FIG. 88 is a physical map of plasmid pMAB62.
[0156] FIG. 89 is a depiction of a synthesis reaction using two pairs of homologous primers of the invention.
[0157] FIG. 90 is a schematic depiction of the physical map (FIG. 90A), and the nucleotide sequence (FIG. 90B-D) (SEQ ID NO:175), of Destination Vector pDEST28.
[0158] FIG. 91 is a schematic depiction of the physical map (FIG. 91A), and the nucleotide sequence (FIG. 91B-D) (SEQ ID NO:176), of Destination Vector pDEST29.
[0159] FIG. 92 is a schematic depiction of the physical map (FIG. 92A), and the nucleotide sequence (FIG. 92B-D) (SEQ ID NO:177), of Destination Vector pDEST30.
[0160] FIG. 93 is a schematic depiction of the physical map (FIG. 93A), and the nucleotide sequence (FIG. 93B-D) (SEQ ID NO:178), of Destination Vector pDEST31.
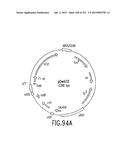
[0161] FIG. 94 is a schematic depiction of the physical map (FIG. 94A), and the nucleotide sequence (FIGS. 94B-F) (SEQ ID NO:179), of Destination Vector pDEST32.
[0162] FIG. 95 is a schematic depiction of the physical map (FIG. 95A), and the nucleotide sequence (FIGS. 95B-E) (SEQ ID NO:180), of Destination Vector pDEST33.
[0163] FIG. 96 is a schematic depiction of the physical map (FIG. 96A), and the nucleotide sequence (FIG. 96B-D) (SEQ ID NO:181), of Destination Vector pDEST34.
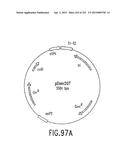
[0164] FIG. 97 is a depiction of the physical map (FIG. 97A), and the nucleotide sequence (FIG. 97B-C) (SEQ ID NO:182), for the Donor plasmid pDONR207 which donates a gentamycin-resistant vector in the BP Reaction.
[0165] FIG. 98 is a schematic depiction of the physical map (FIG. 98A), and the nucleotide sequence (FIG. 98B-D) (SEQ ID NO:183), of the 2-hybrid vector pMAB85.
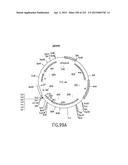
[0166] FIG. 99 is a schematic depiction of the physical map (FIG. 99A), and the nucleotide sequence (FIG. 99B-D) (SEQ ID NO:184), of the 2-hybrid vector pMAB86.
DETAILED DESCRIPTION OF THE INVENTION
Definitions
[0167] In the description that follows, a number of terms used in recombinant DNA technology are utilized extensively. In order to provide a clear and consistent understanding of the specification and claims, including the scope to be given such terms, the following definitions are provided.
[0168] Byproduct: is a daughter molecule (a new clone produced after the second recombination event during the recombinational cloning process) lacking the segment which is desired to be cloned or subcloned.
[0169] Cointegrate: is at least one recombination intermediate nucleic acid molecule of the present invention that contains both parental (starting) molecules. It will usually be linear. In some embodiments it can be circular. RNA and polypeptides may be expressed from cointegrates using an appropriate host cell strain, for example E. coli DB3.1 (particularly E. coli LIBRARY EFFICIENCY® DB3.1® Competent Cells), and selecting for both selection markers found on the cointegrate molecule.
[0170] Host: is any prokaryotic or eukaryotic organism that can be a recipient of the recombinational cloning Product, vector, or nucleic acid molecule of the invention. A "host," as the term is used herein, includes prokaryotic or eukaryotic organisms that can be genetically engineered. For examples of such hosts, see Maniatis et al., Molecular Cloning: A Laboratory Manual, Cold Spring Harbor Laboratory, Cold Spring Harbor, N.Y. (1982).
[0171] Insert or Inserts: include the desired nucleic acid segment or a population of nucleic acid segments (segment A of FIG. 1) which may be manipulated by the methods of the present invention. Thus, the terms Insert(s) are meant to include a particular nucleic acid (preferably DNA) segment or a population of segments. Such Insert(s) can comprise one or more nucleic acid molecules.
[0172] Insert Donor: is one of the two parental nucleic acid molecules (e.g. RNA or DNA) of the present invention which carries the Insert. The Insert Donor molecule comprises the Insert flanked on both sides with recombination sites. The Insert Donor can be linear or circular. In one embodiment of the invention, the Insert Donor is a circular DNA molecule and further comprises a cloning vector sequence outside of the recombination signals (see FIG. 1). When a population of Inserts or population of nucleic acid segments are used to make the Insert Donor, a population of Insert Donors results and may be used in accordance with the invention. Examples of such Insert Donor molecules are GATEWAY® Entry Vectors, which include but are not limited to those Entry Vectors depicted in FIGS. 10-20, as well as other vectors comprising a gene of interest flanked by one or more attL sites (e.g., attL1, attL2, etc.), or by one or more attB sites (e.g., attB1, attB2, etc.) for the production of library clones.
[0173] Product: is one of the desired daughter molecules comprising the A and D sequences which is produced after the second recombination event during the recombinational cloning process (see FIG. 1). The Product contains the nucleic acid which was to be cloned or subcloned. In accordance with the invention, when a population of Insert Donors are used, the resulting population of Product molecules will contain all or a portion of the population of Inserts of the Insert Donors and preferably will contain a representative population of the original molecules of the Insert Donors.
[0174] Promoter: is a DNA sequence generally described as the 5'-region of a gene, located proximal to the start codon. The transcription of an adjacent DNA segment is initiated at the promoter region. A repressible promoter's rate of transcription decreases in response to a repressing agent. An inducible promoter's rate of transcription increases in response to an inducing agent. A constitutive promoter's rate of transcription is not specifically regulated, though it can vary under the influence of general metabolic conditions.
[0175] Recognition sequence: Recognition sequences are particular sequences which a protein, chemical compound, DNA, or RNA molecule (e.g., restriction endonuclease, a modification methylase, or a recombinase) recognizes and binds. In the present invention, a recognition sequence will usually refer to a recombination site. For example, the recognition sequence for Cre recombinase is loxP which is a 34 base pair sequence comprised of two 13 base pair inverted repeats (serving as the recombinase binding sites) flanking an 8 base pair core sequence. See FIG. 1 of Sauer, B., Current Opinion in Biotechnology 5:521-527 (1994). Other examples of recognition sequences are the attB, attP, attL, and attR sequences which are recognized by the recombinase enzyme λ Integrase. attB is an approximately 25 base pair sequence containing two 9 base pair core-type Int binding sites and a 7 base pair overlap region. attP is an approximately 240 base pair sequence containing core-type Int binding sites and arm-type hit binding sites as well as sites for auxiliary proteins integration host factor (IHF), FIS and excisionase (Xis). See Landy, Current Opinion in Biotechnology 3:699-707 (1993). Such sites may also be engineered according to the present invention to enhance production of products in the methods of the invention. When such engineered sites lack the P1 or H1 domains to make the recombination reactions irreversible (e.g., attR or attP), such sites may be designated attR' or attP' to show that the domains of these sites have been modified in some way.
[0176] Recombination proteins: include excisive or integrative proteins, enzymes, co-factors or associated proteins that are involved in recombination reactions involving one or more recombination sites, which may be wild-type proteins (See Landy, Current Opinion in Biotechnology 3:699-707 (1993)), or mutants, derivatives (e.g., fusion proteins containing the recombination protein sequences or fragments thereof), fragments, and variants thereof.
[0177] Recombination site: is a recognition sequence on a DNA molecule participating in an integration/recombination reaction by the recombinational cloning methods of the invention. Recombination sites are discrete sections or segments of DNA on the participating nucleic acid molecules that are recognized and bound by a site-specific recombination protein during the initial stages of integration or recombination. For example, the recombination site for Cre recombinase is loxP which is a 34 base pair sequence comprised of two 13 base pair inverted repeats (serving as the recombinase binding sites) flanking an 8 base pair core sequence. See FIG. 1 of Sauer, B., Curr. Opin. Biotech. 5:521-527 (1994). Other examples of recognition sequences include the attB, attP, attL, and attR sequences described herein, and mutants, fragments, variants and derivatives thereof, which are recognized by the recombination protein λ Int and by the auxiliary proteins integration host factor (IHF), FIS and excisionase (Xis). See Landy, Curr. Opin. Biotech. 3:699-707 (1993).
[0178] Recombinational Cloning: is a method described herein, whereby segments of nucleic acid molecules or populations of such molecules are exchanged, inserted, replaced, substituted or modified, in vitro or in vivo. By "in vitro" and "in vivo" herein is meant recombinational cloning that is carried out outside of host cells (e.g., in cell-free systems) or inside of host cells (e.g., using recombination proteins expressed by host cells), respectively.
[0179] Repression cassette: is a nucleic acid segment that contains a repressor or a Selectable marker present in the subcloning vector.
[0180] Selectable marker: is a DNA segment that allows one to select for or against a molecule (e.g., a replicon) or a cell that contains it, often under particular conditions. These markers can encode an activity, such as, but not limited to, production of RNA, peptide, or protein, or can provide a binding site for RNA, peptides, proteins, inorganic and organic compounds or compositions and the like. Examples of Selectable markers include but are not limited to: (1) DNA segments that encode products which provide resistance against otherwise toxic compounds (e.g., antibiotics); (2) DNA segments that encode products which are otherwise lacking in the recipient cell (e.g., tRNA genes, auxotrophic markers); (3) DNA segments that encode products which suppress the activity of a gene product; (4) DNA segments that encode products which can be readily identified (e.g., phenotypic markers such as β-galactosidase, green fluorescent protein (GFP), and cell surface proteins); (5) DNA segments that bind products which are otherwise detrimental to cell survival and/or function; (6) DNA segments that otherwise inhibit the activity of any of the DNA segments described in Nos. 1-5 above (e.g., antisense oligonucleotides); (7) DNA segments that bind products that modify a substrate (e.g. restriction endonucleases); (8) DNA segments that can be used to isolate or identify a desired molecule (e.g. specific protein binding sites); (9) DNA segments that encode a specific nucleotide sequence which can be otherwise non-functional (e.g., for PCR amplification of subpopulations of molecules); (10) DNA segments, which when absent, directly or indirectly confer resistance or sensitivity to particular compounds; (11) DNA segments that encode products which are toxic in recipient cells; (12) DNA segments that inhibit replication, partition or heritability of nucleic acid molecules that contain them; and/or (13) DNA segments that encode conditional replication functions, e.g., replication in certain hosts or host cell strains or under certain environmental conditions (e.g., temperature, nutritional conditions, etc.).
[0181] Selection scheme: is any method which allows selection, enrichment, or identification of a desired Product or Product(s) from a mixture containing an Entry Clone or Vector, a Destination Vector, a Donor Vector, an Expression Clone or Vector, any intermediates (e.g. a Cointegrate or a replicon), and/or Byproducts. The selection schemes of one preferred embodiment have at least two components that are either linked or unlinked during recombinational cloning. One component is a Selectable marker. The other component controls the expression in vitro or in vivo of the Selectable marker, or survival of the cell (or the nucleic acid molecule, e.g., a replicon) harboring the plasmid carrying the Selectable marker. Generally, this controlling element will be a repressor or inducer of the Selectable marker, but other means for controlling expression or activity of the Selectable marker can be used. Whether a repressor or activator is used will depend on whether the marker is for a positive or negative selection, and the exact arrangement of the various DNA segments, as will be readily apparent to those skilled in the art. A preferred requirement is that the selection scheme results in selection of or enrichment for only one or more desired Products. As defined herein, selecting for a DNA molecule includes (a) selecting or enriching for the presence of the desired DNA molecule, and (b) selecting or enriching against the presence of DNA molecules that are not the desired DNA molecule.
[0182] In one embodiment, the selection schemes (which can be carried out in reverse) will take one of three forms, which will be discussed in terms of FIG. 1. The first, exemplified herein with a Selectable marker and a repressor therefore, selects for molecules having segment D and lacking segment C. The second selects against molecules having segment C and for molecules having segment D. Possible embodiments of the second form would have a DNA segment carrying a gene toxic to cells into which the in vitro reaction products are to be introduced. A toxic gene can be a DNA that is expressed as a toxic gene product (a toxic protein or RNA), or can be toxic in and of itself. (In the latter case, the toxic gene is understood to carry its classical definition of "heritable trait".)
[0183] Examples of such toxic gene products are well known in the art, and include, but are not limited to, restriction endonucleases (e.g., DpnI), apoptosis-related genes (e.g. ASK1 or members of the bcl-2/ced-9 family), retroviral genes including those of the human immunodeficiency virus (HIV), defensins such as NP-1, inverted repeats or paired palindromic DNA sequences, bacteriophage lytic genes such as those from ΦX174 or bacteriophage T4; antibiotic sensitivity genes such as rpsL, antimicrobial sensitivity genes such as pheS, plasmid killer genes, eukaryotic transcriptional vector genes that produce a gene product toxic to bacteria, such as GATA-1, and genes that kill hosts in the absence of a suppressing function, e.g., kicB, ccdB, ΦX174 E (Liu, Q. et al., Curr. Biol. 8:1300-1309 (1998)), and other genes that negatively affect replicon stability and/or replication. A toxic gene can alternatively be selectable in vitro, e.g., a restriction site.
[0184] Many genes coding for restriction endonucleases operably linked to inducible promoters are known, and may be used in the present invention. See, e.g. U.S. Pat. No. 4,960,707 (DpnI and DpnII); U.S. Pat. Nos. 5,000,333, 5,082,784 and 5,192,675 (KpnI); U.S. Pat. No. 5,147,800 (NgoAIII and NgoAI); U.S. Pat. No. 5,179,015 (FspI and HaeIII): U.S. Pat. No. 5,200,333 (HaeII and TaqI); U.S. Pat. No. 5,248,605 (HpaII); U.S. Pat. No. 5,312,746 (ClaI); U.S. Pat. No. 5,231,021 and U.S. Pat. No. 5,304,480 (XhoI and XhoII); U.S. Pat. No. 5,334,526 (AluI); U.S. Pat. No. 5,470,740 (NsiI); U.S. Pat. No. 5,534,428 (SstI/SacI); U.S. Pat. No. 5,202,248 (NcoI); U.S. Pat. No. 5,139,942 (NdeI); and U.S. Pat. No. 5,098,839 (PacI). See also Wilson, G. G., Nucl. Acids Res. 19:2539-2566 (1991); and Lunnen, K. D., et al., Gene 74:25-32 (1988).
[0185] In the second form, segment D carries a Selectable marker. The toxic gene would eliminate transformants harboring the Vector Donor, Cointegrate, and Byproduct molecules, while the Selectable marker can be used to select for cells containing the Product and against cells harboring only the Insert Donor.
[0186] The third form selects for cells that have both segments A and D in cis on the same molecule, but not for cells that have both segments in trans on different molecules. This could be embodied by a Selectable marker that is split into two inactive fragments, one each on segments A and D.
[0187] The fragments are so arranged relative to the recombination sites that when the segments are brought together by the recombination event, they reconstitute a functional Selectable marker. For example, the recombinational event can link a promoter with a structural nucleic acid molecule (e.g., a gene), can link two fragments of a structural nucleic acid molecule, or can link nucleic acid molecules that encode a heterodimeric gene product needed for survival, or can link portions of a replicon.
[0188] Site-specific recombinase: is a type of recombinase which typically has at least the following four activities (or combinations thereof): (1) recognition of one or two specific nucleic acid sequences; (2) cleavage of said sequence or sequences; (3) topoisomerase activity involved in strand exchange; and (4) ligase activity to reseal the cleaved strands of nucleic acid. See Sauer, B., Current Opinions in Biotechnology 5:521-527 (1994). Conservative site-specific recombination is distinguished from homologous recombination and transposition by a high degree of sequence specificity for both partners. The strand exchange mechanism involves the cleavage and rejoining of specific DNA sequences in the absence of DNA synthesis (Landy, A. (1989) Ann. Rev. Biochem. 58:913-949).
[0189] Subcloning vector: is a cloning vector comprising a circular or linear nucleic acid molecule which includes preferably an appropriate replicon. In the present invention, the subcloning vector (segment D in FIG. 1) can also contain functional and/or regulatory elements that are desired to be incorporated into the final product to act upon or with the cloned DNA Insert (segment A in FIG. 1). The subcloning vector can also contain a Selectable marker (preferably DNA).
[0190] Vector: is a nucleic acid molecule (preferably DNA) that provides a useful biological or biochemical property to an Insert. Examples include plasmids, phages, autonomously replicating sequences (ARS), centromeres, and other sequences which are able to replicate or be replicated in vitro or in a host cell, or to convey a desired nucleic acid segment to a desired location within a host cell. A Vector can have one or more restriction endonuclease recognition sites at which the sequences can be cut in a determinable fashion without loss of an essential biological function of the vector, and into which a nucleic acid fragment can be spliced in order to bring about its replication and cloning. Vectors can further provide primer sites, e.g., for PCR, transcriptional and/or translational initiation and/or regulation sites, recombinational signals, replicons, Selectable markers, etc. Clearly, methods of inserting a desired nucleic acid fragment which do not require the use of homologous recombination, transpositions or restriction enzymes (such as, but not limited to, UDG cloning of PCR fragments (U.S. Pat. No. 5,334,575, entirely incorporated herein by reference), T:A cloning, and the like) can also be applied to clone a fragment into a cloning vector to be used according to the present invention. The cloning vector can further contain one or more selectable markers suitable for use in the identification of cells transformed with the cloning vector.
[0191] Vector Donor: is one of the two parental nucleic acid molecules (e.g. RNA or DNA) of the present invention which carries the DNA segments comprising the DNA vector which is to become part of the desired Product. The Vector Donor comprises a subcloning vector D (or it can be called the cloning vector if the Insert Donor does not already contain a cloning vector (e.g., for PCR fragments containing attB sites; see below)) and a segment C flanked by recombination sites (see FIG. 1). Segments C and/or D can contain elements that contribute to selection for the desired Product daughter molecule, as described above for selection schemes. The recombination signals can be the same or different, and can be acted upon by the same or different recombinases. In addition, the Vector Donor can be linear or circular. Examples of such Vector Donor molecules include GATEWAY® Destination Vectors, which include but are not limited to those Destination Vectors depicted in FIGS. 21-47 and 90-96.
[0192] Primer: refers to a single stranded or double stranded oligonucleotide that is extended by covalent bonding of nucleotide monomers during amplification or polymerization of a nucleic acid molecule (e.g. a DNA molecule). In a preferred aspect, a primer comprises one or more recombination sites or portions of such recombination sites. Portions of recombination sites comprise at least 2 bases (or basepairs, abbreviated herein as "bp"), at least 5-200 bases, at least 10-100 bases, at least 15-75 bases, at least 15-50 bases, at least 15-25 bases, or at least 16-25 bases, of the recombination sites of interest, as described in further detail below and in the Examples. When using portions of recombination sites, the missing portion of the recombination site may be provided as a template by the newly synthesized nucleic acid molecule. Such recombination sites may be located within and/or at one or both termini of the primer. Preferably, additional sequences are added to the primer adjacent to the recombination site(s) to enhance or improve recombination and/or to stabilize the recombination site during recombination. Such stabilization sequences may be any sequences (preferably G/C rich sequences) of any length. Preferably, such sequences range in size from 1 to about 1000 bases, 1 to about 500 bases, and 1 to about 100 bases, 1 to about 60 bases, 1 to about 25, 1 to about 10, 2 to about 10 and preferably about 4 bases. Preferably, such sequences are greater than 1 base in length and preferably greater than 2 bases in length.
[0193] Template: refers to double stranded or single stranded nucleic acid molecules which are to be amplified, synthesized or sequenced. In the case of double stranded molecules, denaturation of its strands to form a first and a second strand is preferably performed before these molecules will be amplified, synthesized or sequenced, or the double stranded molecule may be used directly as a template. For single stranded templates, a primer complementary to a portion of the template is hybridized under appropriate conditions and one or more polypeptides having polymerase activity (e.g. DNA polymerases and/or reverse transcriptases) may then synthesize a nucleic acid molecule complementary to all or a portion of said template. Alternatively, for double stranded templates, one or more promoters may be used in combination with one or more polymerases to make nucleic acid molecules complementary to all or a portion of the template. The newly synthesized molecules, according to the invention, may be equal or shorter in length than the original template. Additionally, a population of nucleic acid templates may be used during synthesis or amplification to produce a population of nucleic acid molecules typically representative of the original template population.
[0194] Adapter: is an oligonucleotide or nucleic acid fragment or segment (preferably DNA) which comprises one or more recombination sites (or portions of such recombination sites) which in accordance with the invention can be added to a circular or linear Insert Donor molecule as well as other nucleic acid molecules described herein. When using portions of recombination sites, the missing portion may be provided by the Insert Donor molecule. Such adapters may be added at any location within a circular or linear molecule, although the adapters are preferably added at or near one or both termini of a linear molecule. Preferably, adapters are positioned to be located on both sides (flanking) a particular nucleic acid molecule of interest. In accordance with the invention, adapters may be added to nucleic acid molecules of interest by standard recombinant techniques (e.g. restriction digest and ligation). For example, adapters may be added to a circular molecule by first digesting the molecule with an appropriate restriction enzyme, adding the adapter at the cleavage site and reforming the circular molecule which contains the adapter(s) at the site of cleavage. In other aspects, adapters may be added by homologous recombination, by integration of RNA molecules, and the like. Alternatively, adapters may be ligated directly to one or more and preferably both termini of a linear molecule thereby resulting in linear molecule(s) having adapters at one or both termini. In one aspect of the invention, adapters may be added to a population of linear molecules, (e.g. a cDNA library or genomic DNA which has been cleaved or digested) to form a population of linear molecules containing adapters at one and preferably both termini of all or substantial portion of said population.
[0195] Adapter-Primer: is primer molecule which comprises one or more recombination sites (or portions of such recombination sites) which in accordance with the invention can be added to a circular or linear nucleic acid molecule described herein. When using portions of recombination sites, the missing portion may be provided by a nucleic acid molecule (e.g., an adapter) of the invention. Such adapter-primers may be added at any location within a circular or linear molecule, although the adapter-primers are preferably added at or near one or both termini of a linear molecule. Examples of such adapter-primers and the use thereof in accordance with the methods of the invention are shown in Example 25 herein. Such adapter-primers may be used to add one or more recombination sites or portions thereof to circular or linear nucleic acid molecules in a variety of contexts and by a variety of techniques, including but not limited to amplification (e.g., PCR), ligation (e.g., enzymatic or chemical/synthetic ligation), recombination (e.g., homologous or non-homologous (illegitimate) recombination) and the like.
[0196] Library: refers to a collection of nucleic acid molecules (circular or linear). In one embodiment, a library may comprise a plurality (i.e., two or more) of DNA molecules, which may or may not be from a common source organism, organ, tissue, or cell. In another embodiment, a library is representative of all or a portion or a significant portion of the DNA content of an organism (a "genomic" library), or a set of nucleic acid molecules representative of all or a portion or a significant portion of the expressed nucleic acid molecules (a cDNA library) in a cell, tissue, organ or organism. A library may also comprise random sequences made by de novo synthesis, mutagenesis of one or more sequences and the like. Such libraries may or may not be contained in one or more vectors.
[0197] Amplification: refers to any in vitro method for increasing a number of copies of a nucleotide sequence with the use of a polymerase. Nucleic acid amplification results in the incorporation of nucleotides into a DNA and/or RNA molecule or primer thereby forming a new molecule complementary to a template. The formed nucleic acid molecule and its template can be used as templates to synthesize additional nucleic acid molecules. As used herein, one amplification reaction may consist of many rounds of replication. DNA amplification reactions include, for example, polymerase chain reaction (PCR). One PCR reaction may consist of 5-100 "cycles" of denaturation and synthesis of a DNA molecule.
[0198] Oligonucleotide: refers to a synthetic or natural molecule comprising a covalently linked sequence of nucleotides which are joined by a phosphodiester bond between the 3' position of the deoxyribose or ribose of one nucleotide and the 5' position of the deoxyribose or ribose of the adjacent nucleotide. This term may be used interchangeably herein with the terms "nucleic acid molecule" and "polynucleotide," without any of these terms necessarily indicating any particular length of the nucleic acid molecule to which the term specifically refers.
[0199] Nucleotide: refers to a base-sugar-phosphate combination. Nucleotides are monomeric units of a nucleic acid molecule (DNA and RNA). The term nucleotide includes ribonucleoside triphosphates ATP, UTP, CTG, GTP and deoxyribonucleoside triphosphates such as dATP, dCTP, dITP, dUTP, dGTP, dTTP, or derivatives thereof. Such derivatives include, for example, [αS]dATP, 7-deaza-dGTP and 7-deaza-dATP. The term nucleotide as used herein also refers to dideoxyribonucleoside triphosphates (ddNTPs) and their derivatives. Illustrated examples of dideoxyribonucleoside triphosphates include, but are not limited to, ddATP, ddCTP, ddGTP, ddITP, and ddTTP. According to the present invention, a "nucleotide" may be unlabeled or detectably labeled by well known techniques. Detectable labels include, for example, radioactive isotopes, fluorescent labels, chemiluminescent labels, bioluminescent labels and enzyme labels.
[0200] Hybridization: The terms "hybridization" and "hybridizing" refers to base pairing of two complementary single-stranded nucleic acid molecules (RNA and/or DNA) to give a double stranded molecule. As used herein, two nucleic acid molecules may be hybridized, although the base pairing is not completely complementary. Accordingly, mismatched bases do not prevent hybridization of two nucleic acid molecules provided that appropriate conditions, well known in the art, are used. In some aspects, hybridization is said to be under "stringent conditions." By "stringent conditions" as used herein is meant overnight incubation at 42° C. in a solution comprising: 50% formamide, 5×SSC (150 mM NaCl, 15 mM trisodium citrate), 50 mM sodium phosphate (pH 7.6), 5×Denhardt's solution, 10% dextran sulfate, and 20 g/ml denatured, sheared salmon sperm DNA, followed by washing the filters in 0.1×SSC at about 65° C.
[0201] Other terms used in the fields of recombinant DNA technology and molecular and cell biology as used herein will be generally understood by one of ordinary skill in the applicable arts.
Overview
[0202] Two reactions constitute the recombinational cloning system of the present invention, referred to herein as the "GATEWAY® Cloning System," as depicted generally in FIG. 1. The first of these reactions, the LR Reaction (FIG. 2), which may also be referred to interchangeably herein as the Destination Reaction, is the main pathway of this system. The LR Reaction is a recombination reaction between an Entry vector or clone and a Destination Vector, mediated by a cocktail of recombination proteins such as the GATEWAY® LR Clonase® Enzyme Mix described herein. This reaction transfers nucleic acid molecules of interest (which may be genes, cDNAs, cDNA libraries, or fragments thereof) from the Entry Clone to an Expression Vector, to create an Expression Clone.
[0203] The sites labeled L, R, B, and P are respectively the attL, attR, attB, and attP recombination sites for the bacteriophage λ recombination proteins that constitute the Clonase cocktail (referred to herein variously as "Clonase" or "GATEWAY® LR Clonase® Enzyme Mix" (for recombination protein mixtures mediating attL×attR recombination reactions, as described herein) or "GATEWAY® BP Clonase® Enzyme Mix" (for recombination protein mixtures mediating attB×attP recombination reactions, as described herein)). The Recombinational Cloning reactions are equivalent to concerted, highly specific, cutting and ligation reactions. Viewed in this way, the recombination proteins cut to the left and right of the nucleic acid molecule of interest in the Entry Clone and ligate it into the Destination vector, creating a new Expression Clone.
[0204] The nucleic acid molecule of interest in an Expression Clone is flanked by the small attB1 and attB2 sites. The orientation and reading frame of the nucleic acid molecule of interest are maintained throughout the subcloning, because attL1 reacts only with attR1, and attL2 reacts only with attR2. Likewise, attB1 reacts only with attP1, and attB2 reacts only with attP2. Thus, the invention also relates to methods of controlled or directional cloning using the recombination sites of the invention (or portions thereof), including variants, fragments, mutants and derivatives thereof which may have altered or enhanced specificity. The invention also relates more generally to any number of recombination site partners or pairs (where each recombination site is specific for and interacts with its corresponding recombination site). Such recombination sites are preferably made by mutating or modifying the recombination site to provide any number of necessary specificities (e.g., attB1-10, attP1-10, attL1-10, attR1-10, etc.), non-limiting examples of which are described in detail in the Examples herein.
[0205] When an aliquot from the recombination reaction is transformed into host cells (e.g., E. coli) and spread on plates containing an appropriate selection agent, e.g., an antibiotic such as ampicillin with or without methicillin, cells that take up the desired clone form colonies. The unreacted Destination Vector does not give ampicillin-resistant colonies, even though it carries the ampicillin-resistance gene, because it contains a toxic gene, e.g., ccdB. Thus selection for ampicillin resistance selects for E. coli cells that carry the desired product, which usually comprise >90% of the colonies on the ampicillin plate.
[0206] To participate in the Recombinational (or "GATEWAY®") Cloning Reaction, a nucleic acid molecule of interest first may be cloned into an Entry Vector, creating an Entry Clone. Multiple options are available for creating Entry Clones, including: cloning of PCR sequences with terminal attB recombination sites into Entry Vectors; using the GATEWAY® Cloning System recombination reaction; transfer of genes from libraries prepared in GATEWAY® Cloning System vectors by recombination into Entry Vectors; and cloning of restriction enzyme-generated fragments and PCR fragments into Entry Vectors by standard recombinant DNA methods. These approaches are discussed in further detail herein.
[0207] A key advantage of the GATEWAY® Cloning System is that a nucleic acid molecule of interest (or even a population of nucleic acid molecules of interest) present as an Entry Clone can be subcloned in parallel into one or more Destination Vectors in a simple reactions for anywhere from about 30 seconds to about 60 minutes (preferably about 1-60 minutes, about 1-45 minutes, about 1-30 minutes, about 2-60 minutes, about 2-45 minutes, about 2-30 minutes, about 1-2 minutes, about 30-60 minutes, about 45-60 minutes, or about 30-45 minutes). Longer reaction times (e.g., 2-24 hours, or overnight) may increase recombination efficiency, particularly where larger nucleic acid molecules are used, as described in the Examples herein. Moreover, a high percentage of the colonies obtained carry the desired Expression Clone. This process is illustrated schematically in FIG. 3, which shows an advantage of the invention in which the molecule of interest can be moved simultaneously or separately into multiple Destination Vectors. In the LR Reaction, one or both of the nucleic acid molecules to be recombined may have any topology (e.g., linear, relaxed circular, nicked circular, supercoiled, etc.), although one or both are preferably linear.
[0208] The second major pathway of the GATEWAY® Cloning System is the BP Reaction (FIG. 4), which may also be referred to interchangeably herein as the Entry Reaction or the Gateward Reaction. The BP Reaction may recombine an Expression Clone with a Donor Plasmid (the counterpart of the byproduct in FIG. 2). This reaction transfers the nucleic acid molecule of interest (which may have any of a variety of topologies, including linear, coiled, supercoiled, etc.) in the Expression Clone into an Entry Vector, to produce a new Entry Clone. Once this nucleic acid molecule of interest is cloned into an Entry Vector, it can be transferred into new Expression Vectors, through the LR Reaction as described above. In the BP Reaction, one or both of the nucleic acid molecules to be recombined may have any topology (e.g., linear, relaxed circular, nicked circular, supercoiled, etc.), although one or both are preferably linear.
[0209] A useful variation of the BP Reaction permits rapid cloning and expression of products of amplification (e.g., PCR) or nucleic acid synthesis. Amplification (e.g., PCR) products synthesized with primers containing terminal 25 bp attB sites serve as efficient substrates for the Gateward Cloning reaction. Such amplification products may be recombined with a Donor Vector to produce an Entry Clone (see FIG. 7). The result is an Entry Clone containing the amplification fragment. Such Entry Clones can then be recombined with Destination Vectors--through the LR Reaction--to yield Expression Clones of the PCR product.
[0210] Additional details of the LR Reaction are shown in FIG. 5A. The GATEWAY® LR Clonase® Enzyme Mix that mediates this reaction contains lambda recombination proteins Int (Integrase), Xis (Excisionase), and IHF (Integration Host Factor). In contrast, the GATEWAY® BP Clonase® Enzyme Mix, which mediates the BP Reaction (FIG. 5B), comprises Int and IHF alone.
[0211] The recombination (att) sites of each vector comprise two distinct segments, donated by the parental vectors. The staggered lines dividing the two portions of each att site, depicted in FIGS. 5A and 5B, represent the seven-base staggered cut produced by Int during the recombination reactions. This structure is seen in greater detail in FIG. 6, which displays the attB recombination sequences of an Expression Clone, generated by recombination between the attL1 and attL2 sites of an Entry Clone and the attR1 and attR2 sites of a Destination Vector.
[0212] The nucleic acid molecule of interest in the Expression Clone is flanked by attB sites: attB1 to the left (amino terminus) and attB2 to the right (carboxy terminus). The bases in attB1 to the left of the seven-base staggered cut produced by Int are derived from the Destination vector, and the bases to the right of the staggered cut are derived from the Entry Vector (see FIG. 6). Note that the sequence is displayed in triplets corresponding to an open reading frame. If the reading frame of the nucleic acid molecule of interest cloned in the Entry Vector is in phase with the reading frame shown for attB1, amino-terminal protein fusions can be made between the nucleic acid molecule of interest and any GATEWAY® Cloning System Destination Vector encoding an amino-terminal fusion domain. Entry Vectors and Destination Vectors that enable cloning in all three reading frames are described in more detail herein, particularly in the Examples.
[0213] The LR Reaction allows the transfer of a desired nucleic acid molecule of interest into new Expression Vectors by recombining a Entry Clone with various Destination Vectors. To participate in the LR or Destination Reaction, however, a nucleic acid molecule of interest preferably is first converted to a Entry Clone. Entry Clones can be made in a number of ways, as shown in FIG. 7.
[0214] One approach is to clone the nucleic acid molecule of interest into one or more of the Entry Vectors, using standard recombinant DNA methods, with restriction enzymes and ligase. The starting DNA fragment can be generated by restriction enzyme digestion or as a PCR product. The fragment is cloned between the attL1 and attL2 recombination sites in the Entry Vector. Note that a toxic or "death" gene (e.g., ccdB), provided to minimize background colonies from incompletely digested Entry Vector, must be excised and replaced by the nucleic acid molecule of interest.
[0215] A second approach to making an Entry Clone (FIG. 7) is to make a library (genomic or cDNA) in an Entry Vector, as described in detail herein. Such libraries may then be transferred into Destination Vectors for expression screening, for example in appropriate host cells such as yeast cells or mammalian cells.
[0216] A third approach to making Entry Clones (FIG. 7) is to use Expression Clones obtained from cDNA molecules or libraries prepared in Expression Vectors. Such cDNAs or libraries, flanked by attB sites, can be introduced into a Entry. Vector by recombination with a Donor Vector via the BP Reaction. If desired, an entire Expression Clone library can be transferred into the Entry Vector through the BP Reaction. Expression Clone cDNA libraries may also be constructed in a variety of prokaryotic and eukaryotic GATEWAY®-modified vectors (e.g., the pEXP501 Expression Vector (see FIG. 48), and 2-hybrid and attB library vectors), as described in detail herein, particularly in the Examples below.
[0217] A fourth, and potentially most versatile, approach to making an Entry Clone (FIG. 7) is to introduce a sequence for a nucleic acid molecule of interest into an Entry Vector by amplification (e.g., PCR) fragment cloning. This method is diagramed in FIG. 8. The DNA sequence first is amplified (for example, with PCR) as outlined in detail below and in the Examples herein, using primers containing one or more bp, two or more bp, three or more bp, four or more bp, five or more bp, preferably six or more bp, more preferably 6-25 bp (particularly 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24 or 25) by of the attB nucleotide sequences (such as, but not limited to, those depicted in FIG. 9), and optionally one or more, two or more, three or more, four or more, and most preferably four or five or more additional terminal nucleotide bases which preferably are guanines. The PCR product then may be converted to a Entry Clone by performing a BP Reaction, in which the attB-PCR product recombines with a Donor Vector containing one or more attP sites. Details of this approach and protocols for PCR fragment subcloning are provided in Examples 8 and 21-25.
[0218] A variety of Entry Clones may be produced by these methods, providing a wide array of cloning options; a number of specific Entry Vectors are also available commercially from Life Technologies, Inc. (Rockville, Md.). The Examples herein provide a more in-depth description of selected Entry Vectors and details of their cloning sites. Choosing the optimal Entry Vector for a particular application is discussed in Example 4.
[0219] Entry Vectors and Destination Vectors should be constructed so that the amino-terminal region of a nucleic acid molecule of interest (e.g., a gene, cDNA library or insert, or fragment thereof) will be positioned next to the attL1 site. Entry Vectors preferably contain the rrnB transcriptional terminator upstream of the attL1 site. This sequence ensures that expression of cloned nucleic acid molecules of interest is reliably "off" in E. coli, so that even toxic genes can be successfully cloned. Thus, Entry Clones may be designed to be transcriptionally silent. Note also that Entry Vectors, and hence Entry Clones, may contain the kanamycin antibiotic resistance (kanr) gene to facilitate selection of host cells containing Entry Clones after transformation. In certain applications, however, Entry Clones may contain other selection markers, including but not limited to a gentamycin resistance (genr) or tetracycline resistance (tetr) gene, to facilitate selection of host cells containing Entry Clones after transformation.
[0220] Once a nucleic acid molecule of interest has been cloned into an Entry Vector, it may be moved into a Destination Vector. The upper right portion of FIG. 5A shows a schematic of a Destination Vector. The thick arrow represents some function (often transcription or translation) that will act on the nucleic acid molecule of interest in the clone. During the recombination reaction, the region between the attR1 and attR2 sites, including a toxic or "death" gene (e.g., ccdB), is replaced by the DNA segment from the Entry Clone. Selection for recombinants that have acquired the ampicillin resistance (ampr) gene (carried on the Destination Vector) and that have also lost the death gene ensures that a high percentage (usually >90%) of the resulting colonies will contain the correct insert.
[0221] To move a nucleic acid molecule of interest into a Destination Vector, the Destination Vector is mixed with the Entry Clone comprising the desired nucleic acid molecule of interest, a cocktail of recombination proteins (e.g., GATEWAY® LR Clonase® Enzyme Mix) is added, the mixture is incubated (preferably at about 25° C. for about 60 minutes, or longer under certain circumstances, e.g. for transfer of large nucleic acid molecules, as described below) and any standard host cell (including bacterial cells such as E. coli; animal cells such as insect cells, mammalian cells, nematode cells and the like; plant cells; and yeast cells) strain is transformed with the reaction mixture. The host cell used will be determined by the desired selection (e.g., E. coli DB3.1, available commercially from Life Technologies, Inc., allows survival of clones containing the ccdB death gene, and thus can be used to select for cointegrate molecules--i.e., molecules that are hybrids between the Entry Clone and Destination Vector). The Examples below provide further details and protocols for use of Entry and Destination Vectors in transferring nucleic acid molecules of interest and expressing RNAs or polypeptides encoded by these nucleic acid molecules in a variety of host cells.
[0222] The cloning system of the invention therefore offers multiple advantages:
[0223] Once a nucleic acid molecule of interest is cloned into the GATEWAY® Cloning System, it can be moved into and out of other vectors with complete fidelity of reading frame and orientation. That is, since the reactions proceed whereby attL1 on the Entry Clone recombines with attR1 on the Destination Vector, the directionality of the nucleic acid molecule of interest is maintained or may be controlled upon transfer from the Entry Clone into the Destination Vector. Hence, the GATEWAY® Cloning System provides a powerful and easy method of directional cloning of nucleic acid molecule of interest.
[0224] One-step cloning or subcloning: Mix the Entry Clone and the Destination Vector with Clonase, incubate, and transform.
[0225] Clone PCR products readily by in vitro recombination, by adding attB sites to PCR primers. Then directly transfer these Entry Clones into Destination Vectors. This process may also be carried out in one step (see Examples below).
[0226] Powerful selections give high reliability: >90% (and often >99%) of the colonies contain the desired DNA in its new vector.
[0227] One-step conversion of existing standard vectors into GATEWAY® Cloning System vectors.
[0228] Ideal for large vectors or those with few cloning sites.
[0229] Recombination sites are short (25 bp), and may be engineered to contain no stop codons or secondary structures.
[0230] Reactions may be automated, for high-throughput applications (e.g., for diagnostic purposes or for therapeutic candidate screening).
[0231] The reactions are economical: 0.3 μg of each DNA; no restriction enzymes, phosphatase, ligase, or gel purification. Reactions work well with miniprep DNA.
[0232] Transfer multiple clones, and even libraries, into one or more Destination Vectors, in a single experiment.
[0233] A variety of Destination Vectors may be produced, for applications including, but not limited to:
[0234] Protein expression in E. coli: native proteins; fusion proteins with GST, His6, thioredoxin, etc., for purification, or one or more epitope tags; any promoter useful in expressing proteins in E. coli may be used, such as ptrc, λPL, and T7 promoters.
[0235] Protein expression in eukaryotic cells: CMV promoter, baculovirus (with or without His6 tag), Semliki Forest virus, Tet regulation.
[0236] DNA sequencing (all lac primers), RNA probes, phagemids (both strands)
[0237] A variety of Entry Vectors (for recombinational cloning entry by standard recombinant DNA methods) may be produced:
[0238] Strong transcription stop just upstream, for genes toxic to E. coli.
[0239] Three reading frames.
[0240] With or without TEV protease cleavage site.
[0241] Motifs for prokaryotic and/or eukaryotic translation.
[0242] Compatible with commercial cDNA libraries.
[0243] Expression Clone cDNA (attB) libraries, for expression screening, including 2-hybrid libraries and phage display libraries, may also be constructed.
Recombination Site Sequences
[0244] In one aspect, the invention relates to nucleic acid molecules, which may or may not be isolated nucleic acid molecules, comprising one or more nucleotide sequences encoding one or more recombination sites or portions thereof. In particular, this aspect of the invention relates to such nucleic acid molecules comprising one or more nucleotide sequences encoding attB, attP, attL, or attR, or portions of these recombination site sequences. The invention also relates to mutants, derivatives, and fragments of such nucleic acid molecules. Unless otherwise indicated, all nucleotide sequences that may have been determined by sequencing a DNA molecule herein were determined using manual or automated DNA sequencing, such as dideoxy sequencing, according to methods that are routine to one of ordinary skill in the art (Sanger, F., and Coulson, A. R., J. Mol. Biol. 94:444-448 (1975); Sanger, F., et al., Proc. Natl. Acad. Sci. USA 74:5463-5467 (1977)). All amino acid sequences of polypeptides encoded by DNA molecules determined herein were predicted by conceptual translation of a DNA sequence determined as above. Therefore, as is known in the art for any DNA sequence determined by these approaches, any nucleotide sequence determined herein may contain some errors. Nucleotide sequences determined by such methods are typically at least about 90% identical, more typically at least about 95% to at least about 99.9% identical to the actual nucleotide sequence of the sequenced DNA molecule. As is also known in the art, a single insertion or deletion in a determined nucleotide sequence compared to the actual sequence will cause a frame shift in translation of the nucleotide sequence such that the predicted amino acid sequence encoded by a determined nucleotide sequence will be completely different from the amino acid sequence actually encoded by the sequenced DNA molecule, beginning at the point of such an insertion or deletion.
[0245] Unless otherwise indicated, each "nucleotide sequence" set forth herein is presented as a sequence of deoxyribonucleotides (abbreviated A, G, C and T). However, by "nucleotide sequence" of a nucleic acid molecule or polynucleotide is intended, for a DNA molecule or polynucleotide, a sequence of deoxyribonucleotides, and for an RNA molecule or polynucleotide, the corresponding sequence of ribonucleotides (A, G, C and U), where each thymidine deoxyribonucleotide (T) in the specified deoxyribonucleotide sequence is replaced by the ribonucleotide uridine (U). Thus, the invention relates to sequences of the invention in the form of DNA or RNA molecules, or hybrid DNA/RNA molecules, and their corresponding complementary DNA, RNA, or DNA/RNA strands.
[0246] In a first such aspect, the invention provides nucleic acid molecules comprising one or more nucleotide sequences encoding attB1, or mutants, fragments, variants or derivatives thereof. Such nucleic acid molecules may comprise an attB1 nucleotide sequence having the sequence set forth in FIG. 9, such as: ACAAGTTTGTACAAAAAAGCAGGCT (SEQ ID NO:1), or a nucleotide sequence complementary to the nucleotide sequence set forth in FIG. 9 for attB1, or mutants, fragments, variants or derivatives thereof. As one of ordinary skill will appreciate, however, certain mutations, insertions, or deletions of one or more bases in the attB1 sequence contained in the nucleic acid molecules of the invention may be made without compromising the structural and functional integrity of these molecules; hence, nucleic acid molecules comprising such mutations, insertions, or deletions in the attB1 sequence are encompassed within the scope of the invention.
[0247] In a related aspect, the invention provides nucleic acid molecules comprising one or more nucleotide sequences encoding attB2, or mutants, fragments, variants or derivatives thereof. Such nucleic acid molecules may comprise an attB2 nucleotide sequence having the sequence set forth in FIG. 9, such as: ACCCAGCTTTCTTGTACAAAGTGGT (SEQ ID NO:2), or a nucleotide sequence complementary to the nucleotide sequence set forth in FIG. 9 for attB2, or mutants, fragments, variants or derivatives thereof. As noted above for attB1, certain mutations, insertions, or deletions of one or more bases in the attB2 sequence contained in the nucleic acid molecules of the invention may be made without compromising the structural and functional integrity of these molecules; hence, nucleic acid molecules comprising such mutations, insertions, or deletions in the attB2 sequence are encompassed within the scope of the invention.
[0248] A recombinant host cell comprising a nucleic acid molecule containing attB1 and attB2 sites (the vector pEXP501, also known as pCMVSport6; see FIG. 48), E. coli DB3.1(pCMVSport6), was deposited on Feb. 27, 1999, with the Collection, Agricultural Research Culture Collection (NRRL), 1815 North University Street, Peoria, Ill. 61604 USA, as Deposit No. NRRL B-30108. The attB1 and attB2 sites within the deposited nucleic acid molecule are contained in nucleic acid cassettes in association with one or more additional functional sequences as described in more detail below.
[0249] In another related aspect, the invention provides nucleic acid molecules comprising one or more nucleotide sequences encoding attP1, or mutants, fragments, variants or derivatives thereof. Such nucleic acid molecules may comprise an attP1 nucleotide sequence having the sequence set forth in FIG. 9, such as: TACAGGTCACTAATACCATCTAAGTAGTTGATTCATAGTGA-CTGGATATGTTGTGTTTTACAGTATTATGTA- GTCTGTTTTTTAT-GCAAAATCTAATTTAATATATTGATATTTATATCATTTTACGTT-TCTCGTTCAGCTTTT- TTGTACAAAGTTGGCATTATAAAAAAGCATTG-CTCATCAATTTGTTGCAACGAACAGGTCACTATCAGTCAAA- ATAA-AATCATTATTTG (SEQ ID NO:3), or a nucleotide sequence complementary to the nucleotide sequence set forth in FIG. 9 for attP1, or mutants, fragments, variants or derivatives thereof. As noted above for attB1, certain mutations, insertions, or deletions of one or more bases in the attP1 sequence contained in the nucleic acid molecules of the invention may be made without compromising the structural and functional integrity of these molecules; hence, nucleic acid molecules comprising such mutations, insertions, or deletions in the attP1 sequence are encompassed within the scope of the invention.
[0250] In another related aspect, the invention provides nucleic acid molecules comprising one or more nucleotide sequences encoding attP2, or mutants, fragments, variants or derivatives thereof. Such nucleic acid molecules may comprise an attP2 nucleotide sequence having the sequence set forth in FIG. 9, such as: CAAATAATGATTTTATTTTGACTGATAGTGACCTGTTCGTTG-CAACAAATTGATAAGCAATGCTTTCTTATA- ATGCCAACTTT-GTACAAGAAAGCTGAACGAGAAACGTAAAATGATA-TAAATATCAATATATTAAATTAGATT- TTGCATAAAAAACAG-ACTACATAATACTGTAAAACACAACATATCCAGTCACTATGAATCAA-CTACTTAGAT- GGTATTAGTGACCTGTA (SEQ ID NO:4), or a nucleotide sequence complementary to the nucleotide sequence set forth in FIG. 9 for attP2, or mutants, fragments, variants or derivatives thereof. As noted above for attB1, certain mutations, insertions, or deletions of one or more bases in the attP2 sequence contained in the nucleic acid molecules of the invention may be made without compromising the structural and functional integrity of these molecules; hence, nucleic acid molecules comprising such mutations, insertions, or deletions in the attP2 sequence are encompassed within the scope of the invention.
[0251] A recombinant host cell comprising a nucleic acid molecule (the attP vector pDONR201, also known as pENTR21-attPkan or pAttPkan; see FIG. 49) containing attP1 and attP2 sites, E. coli DB3.1(pAttPkan) (also called E. coli DB3.1(pAHKan)), was deposited on Feb. 27, 1999, with the Collection, Agricultural Research Culture Collection (NRRL), 1815 North University Street, Peoria, Ill. 61604 USA, as Deposit No. NRRL B-30099. The attP1 and attP2 sites within the deposited nucleic acid molecule are contained in nucleic acid cassettes in association with one or more additional functional sequences as described in more detail below.
[0252] In another related aspect, the invention provides nucleic acid molecules comprising one or more nucleotide sequences encoding attR1, or mutants, fragments, variants or derivatives thereof. Such nucleic acid molecules may comprise an attR1 nucleotide sequence having the sequence set forth in FIG. 9, such as: ACAAGTTTGTACAAAAAAGCTGAACGAG-AAACGTAAAATGATATAAATATCAATATATTAAATTAGATTTTG- CAT-AAAAAACAGACTACATAATACTGTAAAACACAACATATCCAGTCA-CTATG (SEQ ID NO:5), or a nucleotide sequence complementary to the nucleotide sequence set forth in FIG. 9 for attR1, or mutants, fragments, variants or derivatives thereof. As noted above for attB1, certain mutations, insertions, or deletions of one or more bases in the attR1 sequence contained in the nucleic acid molecules of the invention may be made without compromising the structural and functional integrity of these molecules; hence, nucleic acid molecules comprising such mutations, insertions, or deletions in the attR1 sequence are encompassed within the scope of the invention.
[0253] In another related aspect, the invention provides nucleic acid molecules comprising one or more nucleotide sequences encoding attR2, or mutants, fragments, variants or derivatives thereof. Such nucleic acid molecules may comprise an attR2 nucleotide sequence having the sequence set forth in FIG. 9, such as: GCAGGTCGACCATAGTGACTGGATAT-GTTGTGTTTTACAGTATTATGTAGTCTGTTTTTTATGCAAAATCTA- -ATTTAATATATTGATATTTATATCATTTTACGTTTCTCGTTCAGCTT-TCTTGTACAAAGTGGT (SEQ ID NO:6), or a nucleotide sequence complementary to the nucleotide sequence set forth in FIG. 9 for attR2, or mutants, fragments, variants or derivatives thereof. As noted above for attB1, certain mutations, insertions, or deletions of one or more bases in the attR2 sequence contained in the nucleic acid molecules of the invention may be made without compromising the structural and functional integrity of these molecules; hence, nucleic acid molecules comprising such mutations, insertions, or deletions in the attR2 sequence are encompassed within the scope of the invention.
[0254] Recombinant host cell strains containing attR1 sites apposed to cloning sites in reading frame A, reading frame B, and reading frame C, E. coli DB3.1(pEZC15101) (reading frame A; see FIG. 64A), E. coli DB3.1(pEZC15102) (reading frame B; see FIG. 64B), and E. coli DB3.1(pEZC15103) (reading frame C; see FIG. 64C), and containing corresponding attR2 sites, were deposited on Feb. 27, 1999, with the Collection, Agricultural Research Culture Collection (NRRL), 1815 North University Street, Peoria, Ill. 61604 USA, as Deposit Nos. NRRL B-30103, NRRL B-30104, and NRRL B-30105, respectively. The attR1 and attR2 sites within the deposited nucleic acid molecules are contained in nucleic acid cassettes in association with one or more additional functional sequences as described in more detail below.
[0255] In another related aspect, the invention provides nucleic acid molecules comprising one or more nucleotide sequences encoding attL1, or mutants, fragments, variants and derivatives thereof. Such nucleic acid molecules may comprise an attL1 nucleotide sequence having the sequence set forth in FIG. 9, such as: CAA ATA ATG ATT TTA TTT TGA CTG ATA GTG ACC TGT TCG TTG CAA CAA ATT GAT AAG CAA TGC TTT TTT ATA ATG CCA ACT TTG TAC AAA AAA GCA GGC T (SEQ ID NO:7), or a nucleotide sequence complementary to the nucleotide sequence set forth in FIG. 9 for attL1, or mutants, fragments, variants or derivatives thereof. As noted above for attB1, certain mutations, insertions, or deletions of one or more bases in the attL1 sequence contained in the nucleic acid molecules of the invention may be made without compromising the structural and functional integrity of these molecules; hence, nucleic acid molecules comprising such mutations, insertions, or deletions in the attL1 sequence are encompassed within the scope of the invention.
[0256] In another related aspect, the invention provides nucleic acid molecules comprising one or more nucleotide sequences encoding attL2, or mutants, fragments, variants and derivatives thereof. Such nucleic acid molecules may comprise an attL2 nucleotide sequence having the sequence set forth in FIG. 9, such as: C AAA TAA TGA TTT TAT TTT GAC TGA TAG TGA CCT GTT CGT TGC AAC AAA TTG ATA AGC AAT GCT TTC TTA TAA TGC CAA CTT TGT ACA AGA AAG CTG GGT (SEQ ID NO:8), or a nucleotide sequence complementary to the nucleotide sequence set forth in FIG. 9 for attL2, or mutants, fragments, variants or derivatives thereof. As noted above for attB1, certain mutations, insertions, or deletions of one or more bases in the attL2 sequence contained in the nucleic acid molecules of the invention may be made without compromising the structural and functional integrity of these molecules; hence, nucleic acid molecules comprising such mutations, insertions, or deletions in the attL2 sequence are encompassed within the scope of the invention.
[0257] Recombinant host cell strains containing attL1 sites apposed to cloning sites in reading frame A, reading frame B, and reading frame C, E. coli DB3.1(pENTR1A) (reading frame A; see FIG. 10), E. coli DB3.1(pENTR2B) (reading frame B; see FIG. 11), and E. coli DB3.1 (pENTR3C) (reading frame C; see FIG. 12), and containing corresponding attL2 sites, were deposited on Feb. 27, 1999, with the Collection, Agricultural Research Culture Collection (NRRL), 1815 North University Street, Peoria, Ill. 61604 USA, as Deposit Nos. NRRL B-30100, NRRL B-30101, and NRRL B-30102, respectively. The attL1 and attL2 sites within the deposited nucleic acid molecules are contained in nucleic acid cassettes in association with one or more additional functional sequences as described in more detail below.
[0258] Each of the recombination site sequences described herein or portions thereof, or the nucleotide sequence cassettes contained in the deposited clones, may be cloned or inserted into a vector of interest (for example, using the recombinational cloning methods described herein and/or standard restriction cloning techniques that are routine in the art) to generate, for example, Entry Vectors or Destination Vectors which may be used to transfer a desired segment of a nucleic acid molecule of interest (e.g., a gene, cDNA molecule, or cDNA library) into a desired vector or into a host cell.
[0259] Using the information provided herein, such as the nucleotide sequences for the recombination site sequences described herein, an isolated nucleic acid molecule of the present invention encoding one or more recombination sites or portions thereof may be obtained using standard cloning and screening procedures, such as those for cloning cDNAs using mRNA as starting material. Preferred such methods include PCR-based cloning methods, such as reverse transcriptase-PCR (RT-PCR) using primers such as those described herein and in the Examples below. Alternatively, vectors comprising the cassettes containing the recombination site sequences described herein are available commercially from Life Technologies, Inc. (Rockville, Md.).
[0260] The invention is also directed to nucleic acid molecules comprising one or more of the recombination site sequences or portions thereof and one or more additional nucleotide sequences, which may encode functional or structural sites such as one or more multiple cloning sites, one or more transcription termination sites, one or more transcriptional regulatory sequences (which may be promoters, enhancers, repressors, and the like), one or more translational signals (e.g., secretion signal sequences), one or more origins of replication, one or more fusion partner peptides (particularly glutathione S-transferase (GST), hexahistidine (His6), and thioredoxin (Trx)), one or more selection markers or modules, one or more nucleotide sequences encoding localization signals such as nuclear localization signals or secretion signals, one or more origins of replication, one or more protease cleavage sites, one or more genes or portions of genes encoding a protein or polypeptide of interest, and one or more 5' polynucleotide extensions (particularly an extension of guanine residues ranging in length from about 1 to about 20, from about 2 to about 15, from about 3 to about 10, from about 4 to about 10, and most preferably an extension of 4 or 5 guanine residues at the 5' end of the recombination site nucleotide sequence. The one or more additional functional or structural sequences may or may not flank one or more of the recombination site sequences contained on the nucleic acid molecules of the invention.
[0261] In some nucleic acid molecules of the invention, the one or more nucleotide sequences encoding one or more additional functional or structural sites may be operably linked to the nucleotide sequence encoding the recombination site. For example, certain nucleic acid molecules of the invention may have a promoter sequence operably linked to a nucleotide sequence encoding a recombination site or portion thereof of the invention, such as a T7 promoter, a phage lambda PL promoter, an E. coli lac, tip or tac promoter, and other suitable promoters which will be familiar to the skilled artisan.
[0262] Nucleic acid molecules of the present invention, which may be isolated nucleic acid molecules, may be in the form of RNA, such as mRNA, or in the form of DNA, including, for instance, cDNA and genomic DNA obtained by cloning or produced synthetically, or in the form of DNA-RNA hybrids. The nucleic acid molecules of the invention may be double-stranded or single-stranded. Single-stranded DNA or RNA may be the coding strand, also known as the sense strand, or it may be the non-coding strand, also referred to as the anti-sense strand. The nucleic acid molecules of the invention may also have a number of topologies, including linear, circular, coiled, or supercoiled.
[0263] By "isolated" nucleic acid molecule(s) is intended a nucleic acid molecule, DNA or RNA, which has been removed from its native environment. For example, recombinant DNA molecules contained in a vector are considered isolated for the purposes of the present invention. Further examples of isolated DNA molecules include recombinant DNA molecules maintained in heterologous host cells, and those DNA molecules purified (partially or substantially) from a solution whether produced by recombinant DNA or synthetic chemistry techniques. Isolated RNA molecules include in vivo or in vitro RNA transcripts of the DNA molecules of the present invention.
[0264] The present invention further relates to mutants, fragments, variants and derivatives of the nucleic acid molecules of the present invention, which encode portions, analogs or derivatives of one or more recombination sites. Variants may occur naturally, such as a natural allelic variant. By an "allelic variant" is intended one of several alternate forms of a gene occupying a given locus on a chromosome of an organism (see Lewin, B., ed., Genes II, John Wiley & Sons, New York (1985)). Non-naturally occurring variants may be produced using art-known mutagenesis techniques, such as those described hereinbelow.
[0265] Such variants include those produced by nucleotide substitutions, deletions or additions or portions thereof, or combinations thereof. The substitutions, deletions or additions may involve one or more nucleotides. The variants may be altered in coding regions, non-coding regions, or both. Alterations in the coding regions may produce conservative or non-conservative amino acid substitutions, deletions or additions. Especially preferred among these are silent substitutions, additions and deletions, which do not alter the properties and activities of the encoded polypeptide(s) or portions thereof, and which also do not substantially alter the reactivities of the recombination site nucleic acid sequences in recombination reactions. Also especially preferred in this regard are conservative substitutions.
[0266] Particularly preferred mutants, fragments, variants, and derivatives of the nucleic acid molecules of the invention include, but are not limited to, insertions, deletions or substitutions of one or more nucleotide bases within the 15 bp core region (GCTTTTTTATACTAA) (SEQ ID NO:9) which is identical in all four wildtype lambda att sites, attB, attP, attL and attR (see U.S. application Ser. No. 08/663,002, filed Jun. 7, 1996 (now U.S. Pat. No. 5,888,732), Ser. No. 09/005,476, filed Jan. 12, 1998, and Ser. No. 09/177,387, filed Oct. 23, 1998, which describes the core region in further detail, and the disclosures of which are incorporated herein by reference in their entireties). Analogously, the core regions in attB1, attP1, attL1 and attR1 are identical to one another, as are the core regions in attB2, attP2, attL2 and attR2. Particularly preferred in this regard are nucleic acid molecules comprising insertions, deletions or substitutions of one or more nucleotides within the seven by overlap region (TTTATAC, which is defined by the cut sites for the integrase protein and is the region where strand exchange takes place) that occurs within this 15 bp core region (GCTTTTTTATACTAA) (SEQ ID NO:9). Examples of such preferred mutants, fragments, variants and derivatives according to this aspect of the invention include, but are not limited to, nucleic acid molecules in which the thymine at position 1 of the seven by overlap region has been deleted or substituted with a guanine, cytosine, or adenine; in which the thymine at position 2 of the seven by overlap region has been deleted or substituted with a guanine, cytosine, or adenine; in which the thymine at position 3 of the seven by overlap region has been deleted or substituted with a guanine, cytosine, or adenine; in which the adenine at position 4 of the seven by overlap region has been deleted or substituted with a guanine, cytosine, or thymine; in which the thymine at position 5 of the seven by overlap region has been deleted or substituted with a guanine, cytosine, or adenine; in which the adenine at position 6 of the seven by overlap region has been deleted or substituted with a guanine, cytosine, or thymine; and in which the cytosine at position 7 of the seven by overlap region has been deleted or substituted with a guanine, thymine, or adenine; or any combination of one or more such deletions and/or substitutions within this seven by overlap region. As described in detail in Example 21 herein, mutants of the nucleic acid molecules of the invention in which substitutions have been made within the first three positions of the seven by overlap (TTTATAC) have been found in the present invention to strongly affect the specificity of recombination, mutant nucleic acid molecules in which substitutions have been made in the last four positions (TTTATAC) only partially alter recombination specificity, and mutant nucleic acid molecules comprising nucleotide substitutions outside of the seven by overlap, but elsewhere within the 15 bp core region, do not affect specificity of recombination but do influence the efficiency of recombination. Hence, in an additional aspect, the present invention is also directed to nucleic acid molecules comprising one or more recombination site nucleotide sequences that affect recombination specificity, particularly one or more nucleotide sequences that may correspond substantially to the seven base pair overlap within the 15 bp core region, having one or more mutations that affect recombination specificity. Particularly preferred such molecules may comprise a consensus sequence (described in detail in Example 21 herein) such as NNNATAC, wherein "N" refers to any nucleotide (i.e., may be A, G, T/U or C), with the proviso that if one of the first three nucleotides in the consensus sequence is a T/U, then at least one of the other two of the first three nucleotides is not a T/U.
[0267] In a related aspect, the present invention is also directed to nucleic acid molecules comprising one or more recombination site nucleotide sequences that enhance recombination efficiency, particularly one or more nucleotide sequences that may correspond substantially to the core region and having one or more mutations that enhance recombination efficiency. By sequences or mutations that "enhance recombination efficiency" is meant a sequence or mutation in a recombination site, preferably in the core region (e.g., the 15 bp core region of att recombination sites), that results in an increase in cloning efficiency (typically measured by determining successful cloning of a test sequence, e.g., by determining CFU/ml for a given cloning mixture) when recombining molecules comprising the mutated sequence or core region as compared to molecules that do not comprise the mutated sequence or core region (e.g., those comprising a wildtype recombination site core region sequence). More specifically, whether or not a given sequence or mutation enhances recombination efficiency may be determined using the sequence or mutation in recombinational cloning as described herein, and determining whether the sequence or mutation provides enhanced recombinational cloning efficiency when compared to a non-mutated (e.g., wildtype) sequence. Methods of determining preferred cloning efficiency-enhancing mutations for a number of recombination sites, particularly for att recombination sites, are described herein, for example in Examples 22-25. Examples of preferred such mutant recombination sites include but are not limited to the attL consensus core sequence of caacttnntnnnannaagttg (SEQ ID NO:92) (wherein "n" represents any nucleotide), for example the attL5 sequence agcctgctttattatactaagttggcatta (SEQ ID NO:10) and the attL6 sequence agcctgcttttttatattaagttggcatta (SEQ ID NO:11); the attB1.6 sequence ggggacaactttgtacaaaaaagttggct (SEQ ID NO:12); the attB2.2 sequence ggggacaactttgtacaagaaagctgggt (SEQ ID NO:13); and the attB2.10 sequence ggggacaactttgtacaagaaagttgggt (SEQ ID NO:14). Those of skill in the art will appreciate that, in addition to the core region, other portions of the att site may affect the efficiency of recombination. There are five so-called arm binding sites for the integrase protein in the bacteriophage lambda attP site, two in attR (P1 and P2), and three in attL (P'1, P'2 and P'3). Compared to the core binding sites, the integrase protein binds to arm sites with high affinity and interacts with core and arm sites through two different domains of the protein. As with the core binding site a consensus sequence for the arm binding site consisting of C/AAGTCACTAT has been inferred from sequence comparison of the five arm binding sites and seven non-att sites (Ross and Landy, Proc. Natl. Acad. Sci. USA 79:7724-7728 (1982)). Each arm site has been mutated and tested for its effect in the excision and integration reactions (Numrych et al., Nucl. Acids Res. 18:3953 (1990)). Hence, specific sites are utilized in each reaction in different ways, namely, the P1 and P'3 sites are essential for the integration reaction whereas the other three sites are dispensable to the integration reaction to varying degrees. Similarly, the P2, P'1 and P'2 sites are most important for the excision reaction, whereas P1 and P'3 are completely dispensable. Interestingly, when P2 is mutated the integration reaction occurs more efficiently than with the wild type attP site. Similarly, when P1 and P'3 are mutated the excision reaction occurs more efficiently. The stimulatory effect of mutating integrase arm binding sites can be explained by removing sites that compete or inhibit a specific recombination pathway or that function in a reaction that converts products back to starting substrates. In fact there is evidence for an XIS-independent LR reaction (Abremski and Gottesman, J. Mol. Biol. 153:67-78 (1981)). Thus, in addition to modifications in the core region of the att site, the present invention contemplates the use of att sites containing one or more modifications in the integrase arm-type binding sites. In some preferred embodiments, one or more mutations may be introduced into one or more of the P1, P'1, P2, P'2 and P'3 sites. In some preferred embodiments, multiple mutations may be introduced into one or more of these sites. Preferred such mutations include those which increase the recombination in vitro. For example, in some embodiments mutations may be introduced into the arm-type binding sites such that integrative recombination, corresponding to the BP reaction, is enhanced. In other embodiments, mutations may be introduced into the arm-type binding sites such that excisive recombination, corresponding to the LR reaction, is enhanced. Of course, based on the guidance contained herein, particularly in the construction and evaluation of effects of mutated recombination sites upon recombinational specificity and efficiency, analogous mutated or engineered sequences may be produced for other recombination sites described herein (including but not limited to lox, FRT, and the like) and used in accordance with the invention. For example, much like the mutagenesis strategy used to select core binding sites that enhance recombination efficiency, similar strategies can be employed to select changes in the arms of attP, attL and attR, and in analogous sequences in other recombination sites such as lox, FRT and the like, that enhance recombination efficiency. Hence, the construction and evaluation of such mutants is well within the abilities of those of ordinary skill in the art without undue experimentation. One suitable methodology for preparing and evaluating such mutations is found in Numrych, et al., (1990) Nucleic Acids Research 18(13): 3953-3959.
[0268] Other mutant sequences and nucleic acid molecules that may be suitable to enhance recombination efficiency will be apparent from the description herein, or may be easily determined by one of ordinary skill using only routine experimentation in molecular biology in view of the description herein and information that is readily available in the art
[0269] Since the genetic code is well known in the art, it is also routine for one of ordinary skill in the art to produce degenerate variants of the nucleic acid molecules described herein without undue experimentation. Hence, nucleic acid molecules comprising degenerate variants of nucleic acid sequences encoding the recombination sites described herein are also encompassed within the scope of the invention.
[0270] Further embodiments of the invention include isolated nucleic acid molecules comprising a polynucleotide having a nucleotide sequence at least 50% identical, at least 60% identical, at least 70% identical, at least 75% identical, at least 80% identical, at least 85% identical, at least 90% identical, and more preferably at least 95%, 96%, 97%, 98% or 99% identical to the nucleotide sequences of the seven by overlap region within the 15 bp core region of the recombination sites described herein, or the nucleotide sequences of attB1, attB2, attP1, attP2, attL1, attL2, attR1 or attR2 as set forth in FIG. 9 (or portions thereof), or a nucleotide sequence complementary to any of these nucleotide sequences, or fragments, variants, mutants, and derivatives thereof.
[0271] By a polynucleotide having a nucleotide sequence at least, for example, 95% "identical" to a reference nucleotide sequence encoding a particular recombination site or portion thereof is intended that the nucleotide sequence of the polynucleotide is identical to the reference sequence except that the polynucleotide sequence may include up to five point mutations (e.g., insertions, substitutions, or deletions) per each 100 nucleotides of the reference nucleotide sequence encoding the recombination site. For example, to obtain a polynucleotide having a nucleotide sequence at least 95% identical to a reference attB1 nucleotide sequence, up to 5% of the nucleotides in the attB1 reference sequence may be deleted or substituted with another nucleotide, or a number of nucleotides up to 5% of the total nucleotides in the attB1 reference sequence may be inserted into the attB1 reference sequence. These mutations of the reference sequence may occur at the 5' or 3' terminal positions of the reference nucleotide sequence or anywhere between those terminal positions, interspersed either individually among nucleotides in the reference sequence or in one or more contiguous groups within the reference sequence.
[0272] As a practical matter, whether any particular nucleic acid molecule is at least 50%, 60%, 70%, 75%, 80%, 85%, 90%, 95%, 96%, 97%, 98% or 99% identical to, for instance, a given recombination site nucleotide sequence or portion thereof can be determined conventionally using known computer programs such as DNAsis software (Hitachi Software, San Bruno, Calif.) for initial sequence alignment followed by ESEE version 3.0 DNA/protein sequence software (cabot@trog.mbb.sfu.ca) for multiple sequence alignments. Alternatively, such determinations may be accomplished using the BESTFIT program (Wisconsin Sequence Analysis Package, Genetics Computer Group, University Research Park, 575 Science Drive, Madison, Wis. 53711), which employs a local homology algorithm (Smith and Waterman, Advances in Applied Mathematics 2: 482-489 (1981)) to find the best segment of homology between two sequences. When using DNAsis, ESEE, BESTFIT or any other sequence alignment program to determine whether a particular sequence is, for instance, 95% identical to a reference sequence according to the present invention, the parameters are set such that the percentage of identity is calculated over the full length of the reference nucleotide sequence and that gaps in homology of up to 5% of the total number of nucleotides in the reference sequence are allowed.
[0273] The present invention is directed to nucleic acid molecules at least 50%, 60%, 70%, 75%, 80%, 85%, 90%, 95%, 96%, 97%, 98% or 99% identical to the attB1, attB2, attP1, attP2, attL1, attL2, attR1 or attR2 nucleotide sequences as set forth in FIG. 9, or to the nucleotide sequence of the deposited clones, irrespective of whether they encode particular functional polypeptides. This is because even where a particular nucleic acid molecule does not encode a particular functional polypeptide, one of skill in the art would still know how to use the nucleic acid molecule, for instance, as a hybridization probe or a polymerase chain reaction (PCR) primer.
[0274] Mutations can also be introduced into the recombination site nucleotide sequences for enhancing site specific recombination or altering the specificities of the reactants, etc. Such mutations include, but are not limited to: recombination sites without translation stop codons that allow fusion proteins to be encoded; recombination sites recognized by the same proteins but differing in base sequence such that they react largely or exclusively with their homologous partners allowing multiple reactions to be contemplated; and mutations that prevent hairpin formation of recombination sites. Which particular reactions take place can be specified by which particular partners are present in the reaction mixture.
[0275] There are well known procedures for introducing specific mutations into nucleic acid sequences. A number of these are described in Ausubel, F. M. et al., Current Protocols in Molecular Biology, Wiley Interscience, New York (1989-1996). Mutations can be designed into oligonucleotides, which can be used to modify existing cloned sequences, or in amplification reactions. Random mutagenesis can also be employed if appropriate selection methods are available to isolate the desired mutant DNA or RNA. The presence of the desired mutations can be confirmed by sequencing the nucleic acid by well known methods.
[0276] The following non-limiting methods can be used to modify or mutate a given nucleic acid molecule encoding a particular recombination site to provide mutated sites that can be used in the present invention:
[0277] 1. By recombination of two parental DNA sequences by site-specific (e.g. attL and attR to give attP) or other (e.g. homologous) recombination mechanisms where the parental DNA segments contain one or more base alterations resulting in the final mutated nucleic acid molecule;
[0278] 2. By mutation or mutagenesis (site-specific, PCR, random, spontaneous, etc) directly of the desired nucleic acid molecule;
[0279] 3. By mutagenesis (site-specific, PCR, random, spontaneous, etc) of parental DNA sequences, which are recombined to generate a desired nucleic acid molecule;
[0280] 4. By reverse transcription of an RNA encoding the desired core sequence; and
[0281] 5. By de novo synthesis (chemical synthesis) of a sequence having the desired base changes, or random base changes followed by sequencing or functional analysis according to methods that are routine in the art.
[0282] The functionality of the mutant recombination sites can be demonstrated in ways that depend on the particular characteristic that is desired. For example, the lack of translation stop codons in a recombination site can be demonstrated by expressing the appropriate fusion proteins. Specificity of recombination between homologous partners can be demonstrated by introducing the appropriate molecules into in vitro reactions, and assaying for recombination products as described herein or known in the art. Other desired mutations in recombination sites might include the presence or absence of restriction sites, translation or transcription start signals, protein binding sites, particular coding sequences, and other known functionalities of nucleic acid base sequences. Genetic selection schemes for particular functional attributes in the recombination sites can be used according to known method steps. For example, the modification of sites to provide (from a pair of sites that do not interact) partners that do interact could be achieved by requiring deletion, via recombination between the sites, of a DNA sequence encoding a toxic substance. Similarly, selection for sites that remove translation stop sequences, the presence or absence of protein binding sites, etc., can be easily devised by those skilled in the art.
[0283] Accordingly, the present invention also provides a nucleic acid molecule, comprising at least one DNA segment having at least one, and preferably at least two, engineered recombination site nucleotide sequences of the invention flanking a selectable marker and/or a desired DNA segment, wherein at least one of said recombination site nucleotide sequences has at least one engineered mutation that enhances recombination in vitro in the formation of a Cointegrate DNA or a Product DNA. Such engineered mutations may be in the core sequence of the recombination site nucleotide sequence of the invention; see U.S. application Ser. No. 08/486,139, filed Jun. 7, 1995, Ser. No. 08/663,002, filed Jun. 7, 1996 (now U.S. Pat. No. 5,888,732), Ser. No. 09/005,476, filed Jan. 12, 1998, and Ser. No. 09/177,387, filed Oct. 23, 1998, the disclosures of which are all incorporated herein by reference in their entireties.
[0284] While in the preferred embodiment the recombination sites differ in sequence and do not interact with each other, it is recognized that sites comprising the same sequence, which may interact with each other, can be manipulated or engineered to inhibit recombination with each other. Such conceptions are considered and incorporated herein. For example, a protein binding site (e.g., an antibody-binding site, a histone-binding site, an enzyme-binding site, or a binding site for any nucleic acid molecule-binding protein) can be engineered adjacent to one of the sites. In the presence of the protein that recognizes the engineered site, the recombinase fails to access the site and another recombination site in the nucleic acid molecule is therefore used preferentially. In the cointegrate this site can no longer react since it has been changed, e.g., from attB to attL. During or upon resolution of the cointegrate, the protein can be inactivated (e.g., by antibody, heat or a change of buffer) and the second site can undergo recombination.
[0285] The nucleic acid molecules of the invention can have at least one mutation that confers at least one enhancement of said recombination, said enhancement selected from the group consisting of substantially (i) favoring integration; (ii) favoring recombination; (ii) relieving the requirement for host factors; (iii) increasing the efficiency of said Cointegrate DNA or Product DNA formation; (iv) increasing the specificity of said Cointegrate DNA or Product DNA formation; and (v) adding or deleting protein binding sites.
[0286] In other embodiments, the nucleic acid molecules of the invention may be PCR primer molecules, which comprise one or more of the recombination site sequences described herein or portions thereof, particularly those shown in FIG. 9 (or sequences complementary to those shown in FIG. 9), or mutants, fragments, variants or derivatives thereof, attached at the 3' end to a target-specific template sequence which specifically interacts with a target nucleic acid molecule which is to be amplified. Primer molecules according to this aspect of the invention may further comprise one or more, (e.g., 1, 2, 3, 4, 5, 10, 20, 25, 50, 100, 500, 1000, or more) additional bases at their 5' ends, and preferably comprise one or more (particularly four or five) additional bases, which are preferably guanines, at their 5' ends, to increase the efficiency of the amplification products incorporating the primer molecules in the recombinational cloning system of the invention. Such nucleic acid molecules and primers are described in detail in the examples herein, particularly in Examples 22-25.
[0287] Certain primers of the invention may comprise one or more nucleotide deletions in the attB1, attB2, attP1, attP2, attL1, attL2, attR1 or attR2 sequences as set forth in FIG. 9. In one such aspect, for example, attB2 primers may be constructed in which one or more of the first four nucleotides at the 5' end of the attB2 sequence shown in FIG. 9 have been deleted. Primers according to this aspect of the invention may therefore have the sequence:
TABLE-US-00001 (attB2(-1)): (SEQ ID NO: 15) CCCAGCTTTCTTGTACAAAGTGGTnnnnnnnnnnnnn...n (attB2(-2)): (SEQ ID NO: 16) CCAGCTTTCTTGTACAAAGTGGTnnnnnnnnnnnnnn...n (attB2(-3)): (SEQ ID NO: 17) CAGCTTTCTTGTACAAAGTGGTnnnnnnnnnnnnnnn...n (attB2(-4)): (SEQ ID NO: 18) AGCTTTCTTGTACAAAGTGGTnnnnnnnnnnnnnnnn...n,
wherein "nnnnnnnnnnnnn . . . n" at the 3' end of the primer represents a target-specific sequence of any length, for example from one base up to all of the bases of a target nucleic acid molecule (e.g., a gene) or a portion thereof, the sequence and length which will depend upon the identity of the target nucleic acid molecule which is to be amplified.
[0288] The primer nucleic acid molecules according to this aspect of the invention may be produced synthetically by attaching the recombination site sequences depicted in FIG. 9, or portions thereof, to the 5' end of a standard PCR target-specific primer according to methods that are well-known in the art. Alternatively, additional primer nucleic acid molecules of the invention may be produced synthetically by adding one or more nucleotide bases, which preferably correspond to one or more, preferably five or more, and more preferably six or more, contiguous nucleotides of the au nucleotide sequences described herein (see, e.g., Example 20 herein; see also U.S. application Ser. No. 08/663,002, filed Jun. 7, 1996 (now U.S. Pat. No. 5,888,732), Ser. No. 09/005,476, filed Jan. 12, 1998, and Ser. No. 09/177,387, filed Oct. 23, 1998, the disclosures of which are all incorporated herein by reference in their entireties), to the 5' end of a standard PCR target-specific primer according to methods that are well-known in the art, to provide primers having the specific nucleotide sequences described herein. As noted above, primer nucleic acid molecules according to this aspect of the invention may also optionally comprise one, two, three, four, five, or more additional nucleotide bases at their 5' ends, and preferably will comprise four or five guanines at their 5' ends. In one particularly preferred such aspect, the primer nucleic acid molecules of the invention may comprise one or more, preferably five or more, more preferably six or more, still more preferably 6-18 or 6-25, and most preferably 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24 or 25, contiguous nucleotides or by of the attB1 or attB2 nucleotide sequences depicted in FIG. 9 (or nucleotides complementary thereto), linked to the 5' end of a target-specific (e.g., a gene-specific) primer molecule. Primer nucleic acid molecules according to this aspect of the invention include, but are not limited to, attB1- and attB2-derived primer nucleic acid molecules having the following nucleotide sequences:
TABLE-US-00002 (SEQ ID NO: 19) ACAAGTTTGTACAAAAAAGCAGGCT-nnnnnnnnnnnnn...n (SEQ ID NO: 20) ACCACTTTGTACAAGAAAGCTGGGT-nnnnnnnnnnnnn...n (SEQ ID NO: 21) TGTACAAAAAAGCAGGCT-nnnnnnnnnnnnn...n (SEQ ID NO: 22) TGTACAAGAAAGCTGGGT-nnnnnnnnnnnnn...n (SEQ ID NO: 23) ACAAAAAAGCAGGCT-nnnnnnnnnnnnn...n (SEQ ID NO: 24) ACAAGAAAGCTGGGT-nnnnnnnnnnnnn...n (SEQ ID NO: 25) AAAAAGCAGGCT-nnnnnnnnnnnnn...n (SEQ ID NO: 26) AGAAAGCTGGGT-nnnnnnnnnnnnn...n (SEQ ID NO: 27) AAAAGCAGGCT-nnnnnnnnnnnnn...n (SEQ ID NO: 28) GAAAGCTGGGT-nnnnnnnnnnnnn...n (SEQ ID NO: 29) AAAGCAGGCT-nnnnnnnnnnnnn...n (SEQ ID NO: 30) AAAGCTGGGT-nnnnnnnnnnnnn...n AAGCAGGCT-nnnnnnnnnnnnn...n AAGCTGGGT-nnnnnnnnnnnnn...n AGCAGGCT-nnnnnnnnnnnnn...n AGCTGGGT-nnnnnnnnnnnnn...n GCAGGCT-nnnnnnnnnnnnn...n GCTGGGT-nnnnnnnnnnnnn...n CAGGCT-nnnnnnnnnnnnn...n CTGGGT-nnnnnnnnnnnnn...n,
wherein "nnnnnnnnnnnnn . . . n" at the 3' end of the primer represents a target-specific sequence of any length, for example from one base up to all of the bases of a target nucleic acid molecule (e.g., a gene) or a portion thereof, the sequence and length which will depend upon the identity of the target nucleic acid molecule which is to be amplified.
[0289] Of course, it will be apparent to one of ordinary skill from the teachings contained herein that additional primer nucleic acid molecules analogous to those specifically described herein may be produced using one or more, preferably five or more, more preferably six or more, still more preferably ten or more, 15 or more, 20 or more, 25 or more, 30 or more, etc. (through to and including all) of the contiguous nucleotides or by of the attP1, attP2, attL1, attL2, attR1 or attR2 nucleotide sequences depicted in FIG. 9 (or nucleotides complementary thereto), linked to the 5' end of a target-specific (e.g., a gene-specific) primer molecule. As noted above, such primer nucleic acid molecules may optionally further comprise one, two, three, four, five, or more additional nucleotide bases at their 5' ends, and preferably will comprise four guanines at their 5' ends. Other primer molecules comprising the attB1, attB2, attP1, attP2, attL1, attL2, attR1 and attR2 sequences depicted in FIG. 9, or portions thereof, may be made by one of ordinary skill without resorting to undue experimentation in accordance with the guidance provided herein.
[0290] The primers of the invention described herein are useful in producing PCR fragments having a nucleic acid molecule of interest flanked at each end by a recombination site sequence (as described in detail below in Example 9), for use in cloning of PCR-amplified DNA fragments using the recombination system of the invention (as described in detail below in Examples 8, 19 and 21-25).
Vectors
[0291] The invention also relates to vectors comprising one or more of the nucleic acid molecules of the invention, as described herein. In accordance with the invention, any vector may be used to construct the vectors of the invention. In particular, vectors known in the art and those commercially available (and variants or derivatives thereof) may in accordance with the invention be engineered to include one or more nucleic acid molecules encoding one or more recombination sites (or portions thereof), or mutants, fragments, or derivatives thereof, for use in the methods of the invention. Such vectors may be obtained from, for example, Vector Laboratories Inc., InVitrogen, Promega, Novagen, New England Biolabs, Clontech, Roche, Pharmacia, EpiCenter, OriGenes Technologies Inc., Stratagene, Perkin Elmer, Pharmingen, Life Technologies, Inc., and Research Genetics. Such vectors may then for example be used for cloning or subcloning nucleic acid molecules of interest. General classes of vectors of particular interest include prokaryotic and/or eukaryotic cloning vectors, Expression Vectors, fusion vectors, two-hybrid or reverse two-hybrid vectors, shuttle vectors for use in different hosts, mutagenesis vectors, transcription vectors, vectors for receiving large inserts and the like.
[0292] Other vectors of interest include viral origin vectors (M13 vectors, bacterial phage λ vectors, bacteriophage P1 vectors, adenovirus vectors, herpesvirus vectors, retrovirus vectors, phage display vectors, combinatorial library vectors), high, low, and adjustable copy number vectors, vectors which have compatible replicons for use in combination in a single host (pACYC184 and pBR322) and eukaryotic episomal replication vectors (pCDM8).
[0293] Particular vectors of interest include prokaryotic Expression Vectors such as pcDNA II, pSL301, pSE280, pSE380, pSE420, pTrcHisA, B, and C, pRSET A, B, and C (Invitrogen, Inc.), pGEMEX-1, and pGEMEX-2 (Promega, Inc.), the pET vectors (Novagen, Inc.), pTrc99A, pKK223-3, the pGEX vectors, pEZZ18, pRIT2T, and pMC1871 (Pharmacia, Inc.), pKK233-2 and pKK388-1 (Clontech, Inc.), and pProEx-HT (Life Technologies, Inc.) and variants and derivatives thereof. Destination Vectors can also be made from eukaryotic Expression Vectors such as pFastBac, pFastBac HT, pFastBac DUAL, pSFV, and pTet-Splice (Life Technologies, Inc.), pEUK-C1, pPUR, pMAM, pMAMneo, pBI101, pBI121, pDR2, pCMVEBNA, and pYACneo (Clontech), pSVK3, pSVL, pMSG, pCH110, and pKK232-8 (Pharmacia, Inc.), p3'SS, pXT1, pSG5, pPbac, pMbac, pMC1neo, and pOG44 (Stratagene, Inc.), and pYES2, pAC360, pBlueBacHis A, B, and C, pVL1392, pBsueBacIII, pCDM8, pcDNA1, pZeoSV, pcDNA3 pREP4, pCEP4, and pEBVHis (Invitrogen, Inc.) and variants or derivatives thereof.
[0294] Other vectors of particular interest include pUC18, pUC19, pBlueScript, pSPORT, cosmids, phagemids, YACs (yeast artificial chromosomes), BACs (bacterial artificial chromosomes), MACs (mammalian artificial chromosomes), pQE70, pQE60, pQE9 (Quiagen), pBS vectors, PhageScript vectors, BlueScript vectors, pNH8A, pNH16A, pNH18A, pNH46A (Stratagene), pcDNA3 (InVitrogen), pGEX, pTrsfus, pTrc99A, pET-5, pET-9, pKK223-3, pKK233-3, pDR540, pRIT5 (Pharmacia), pSPORT1, pSPORT2, pCMVSPORT2.0 and pSV-SPORT1 (Life Technologies, Inc.) and variants or derivatives thereof.
[0295] Additional vectors of interest include pTrxFus, pThioHis, pLEX, pTrcHis, pTrcHis2, pRSET, pBlueBacHis2, pcDNA3.1/His, pcDNA3.1(-)/Myc-His, pSecTag, pEBVHis, pPIC9K, pPIC3.5K, pAO815, pPICZ, pPICZα, pGAPZ, pGAPZα, pBlueBac4.5, pBlueBacHis2, pMelBac, pSinRep5, pSinHis, pIND, pIND(SP1), pVgRXR, pcDNA2.1. pYES2, pZErO1.1, pZErO-2.1, pCR-Blunt, pSE280, pSE380, pSE420, pVL1392, pVL1393, pCDM8, pcDNA1.1, pcDNA1.1/Amp, pcDNA3.1, pcDNA3.1/Zeo, pSe, SV2, pRc/CMV2, pRc/RSV, pREP4, pREP7, pREP8, pREP9, pREP10, pCEP4, pEBVHis, pCR3.1, pCR2.1, pCR3.1-Uni, and pCRBac from Invitrogen; λExCell, λgt11, pTrc99A, pKK223-3, pGEX-1λT, pGEX-2T, pGEX-2TK, pGEX-4T-1, pGEX-4T-2, pGEX-4T-3, pGEX-3X, pGEX-5X-1, pGEX-5X-2, pGEX-5X-3, pEZZ18, pRIT2T, pMC1871, pSVK3, pSVL, pMSG, pCH110, pKK232-8, pSL1180, pNEO, and pUC4K from Pharmacia; pSCREEN-1b(+), pT7Blue(R), pT7Blue-2, pCITE-4abc(+), pOCUS-2, pTAg, pET-32 LIC, pET-30 LIC, pBAC-2cp LIC, pBACgus-2cp LIC, pT7Blue-2 LIC, pT7Blue-2, λSCREEN-1, λBlueSTAR, pET-3abcd, pET-7abc, pET9abcd, pET11abcd, pET12abc, pET-14b, pET-15b, pET-16b, pET-17b-pET-17xb, pET-19b, pET-20b(+), pET-21abcd(+), pET-22b(+), pET-23abcd(+), pET-24abcd(+), pET-25b(+), pET-26b(+), pET-27b(+), pET-28abc(+), pET-29abc(+), pET-30abc(+), pET-31b(+), pET-32abc(+), pET-33b(+), pBAC-1, pBACgus-1, pBAC4x-1, pBACgus4x-1, pBAC-3cp, pBACgus-2cp, pBACsurf-1, plg, Signal plg, pYX, Selecta Vecta-Neo, Selecta Vecta-Hyg, and Selecta Vecta-Gpt from Novagen; pLexA, pB42AD, pGBT9, pAS2-1, pGAD424, pACT2, pGAD GL, pGAD GH, pGAD10, pGilda, pEZM3, pEGFP, pEGFP-1, pEGFP-N, pEGFP-C, pEBFP, pGFPuv, pGFP, p6xHis-GFP, pSEAP2-Basic, pSEAP2-Contral, pSEAP2-Promoter, pSEAP2-Enhancer, pβgal-Basic, pβgal-Control, pβgal-Promoter, pβgal-Enhancer, pCMVβ, pTet-Off, pTet-On, pTK-Hyg, pRetro-Off, pRetro-On, pIRES1neo, pIRES1hyg, pLXSN, pLNCX, pLAPSN, pMAMneo, pMAMneo-CAT, pMAMneo-LUC, pPUR, pSV2neo, pYEX 4T-1/2/3, pYEX-S 1, pBacPAK-His, pBacPAK8/9, pAcUW31, BacPAK6, pTrip1Ex, λgt10, λgt11, pWE15, and λTripIEx from Clontech; Lambda ZAP II, pBK-CMV, pBK-RSV, pBluescript II KS+/-, pBluescript II SK+/-, pAD-GALA-, pBD-GAL4 Cam, pSurfscript, Lambda FIX II, Lambda DASH, Lambda EMBL3, Lambda EMBL4, SuperCos, pCR-Scrigt Amp, pCR-Script Cam, pCR-Script Direct, pBS+/-, pBC KS+/-, pBC SK+/-, Phagescript, pCAL-n-EK, pCAL-n, pCAL-c, pCAL-kc, pET-3abcd, pET-11abcd, pSPUTK, pESP-1, pCMVLacI, pOPRSVI/MCS, pOPI3 CAT, pXT1, pSG5, pPbac, pMbac, pMC1neo, pMC1neo Poly A, pOG44, pOG45, pFRTβGAL, pNEOβGAL, pRS403, pRS404, pRS405, pRS406, pRS413, pRS414, pRS415, and pRS416 from Stratagene.
[0296] Two-hybrid and reverse two-hybrid vectors of particular interest include pPC86, pDBLeu, pDBTrp, pPC97, p2.5, pGAD1-3, pGAD10, pACt, pACT2, pGADGL, pGADGH, pAS2-1, pGAD424, pGBT8, pGBT9, pGAD-GAL4, pLexA, pBD-GAL4, pHISi, pHISi-1, placZi, pB42AD, pDG202, pJK202, pJG4-5, pNLexA, pYESTrp and variants or derivatives thereof.
[0297] Yeast Expression Vectors of particular interest include pESP-1, pESP-2, pESC-His, pESC-Trp, pESC-URA, pESC-Leu (Stratagene), pRS401, pRS402, pRS411, pRS412, pRS421, pRS422, and variants or derivatives thereof.
[0298] According to the invention, the vectors comprising one or more nucleic acid molecules encoding one or more recombination sites, or mutants, variants, fragments, or derivatives thereof, may be produced by one of ordinary skill in the art without resorting to undue experimentation using standard molecular biology methods. For example, the vectors of the invention may be produced by introducing one or more of the nucleic acid molecules encoding one or more recombination sites (or mutants, fragments, variants or derivatives thereof) into one or more of the vectors described herein, according to the methods described, for example, in Maniatis et al., Molecular Cloning: A Laboratory Manual, Cold Spring Harbor Laboratory, Cold Spring Harbor, N.Y. (1982). In a related aspect of the invention, the vectors may be engineered to contain, in addition to one or more nucleic acid molecules encoding one or more recombination sites (or portions thereof), one or more additional physical or functional nucleotide sequences, such as those encoding one or more multiple cloning sites, one or more transcription termination sites, one or more transcriptional regulatory sequences (e.g., one or more promoters, enhancers, or repressors), one or more selection markers or modules, one or more genes or portions of genes encoding a protein or polypeptide of interest, one or more translational signal sequences, one or more nucleotide sequences encoding a fusion partner protein or peptide (e.g., GST, His6 or thioredoxin), one or more origins of replication, and one or more 5' or 3' polynucleotide tails (particularly a poly-G tail). According to this aspect of the invention, the one or more recombination site nucleotide sequences (or portions thereof) may optionally be operably linked to the one or more additional physical or functional nucleotide sequences described herein.
[0299] Preferred vectors according to this aspect of the invention include, but are not limited to: pENTR1A (FIGS. 10A and 10B), pENTR2B (FIGS. 11A and 11B), pENTR3C (FIGS. 12A and 12B), pENTR4 (FIGS. 13A and 13B), pENTR5 (FIGS. 14A and 14B), pENTR6 (FIGS. 15A and 15B), pENTR7 (FIGS. 16A and 16B), pENTR8 (FIGS. 17A and 17B), pENTR9 (FIGS. 18A and 18B), pENTR10 (FIGS. 19A and 19B), pENTR11 (FIGS. 20A and 20B), pDEST1 (FIGS. 21A-D), pDEST2 (FIG. 22A-D), pDEST3 (FIG. 23A-D), pDEST4 (FIG. 24A-D), pDEST5 (FIG. 25A-D), pDEST6 (FIG. 26A-D), pDEST7 (FIG. 27A-C), pDEST8 (FIG. 28A-D), pDEST9 (FIG. 29A-E), pDEST10 (FIG. 30A-D), pDEST11 (FIG. 31A-D), pDEST12.2 (also known as pDEST12) (FIG. 32A-D), pDEST13 (FIG. 33A-C), pDEST14 (FIG. 34A-D), pDEST15 (FIG. 35A-D), pDEST16 (FIG. 36A-D), pDEST17 (FIG. 37A-D), pDEST18 (FIG. 38A-D), pDEST19 (FIG. 39A-D), pDEST20 (FIG. 40A-D), pDEST21 (FIG. 41A-E), pDEST22 (FIG. 42A-D), pDEST23 (FIG. 43A-D), pDEST24 (FIG. 44A-D), pDEST25 (FIG. 45A-D), pDEST26 (FIG. 46A-D), pDEST27 (FIG. 47A-D), pEXP501 (also known as pCMVSPORT6) (FIG. 48A-B), pDONR201 (also known as pENTR21 attP vector or pAttPkan Donor Vector) (FIG. 49), pDONR202 (FIG. 50), pDONR203 (also known as pEZ15812) (FIG. 51), pDONR204 (FIG. 52), pDONR205 (FIG. 53), pDONR206 (also known as pENTR22 attP vector or pAttPgen Donor Vector) (FIG. 54), pMAB58 (FIG. 87), pMAB62 (FIG. 88), pDEST28 (FIG. 90), pDEST29 (FIG. 91), pDEST30 (FIG. 92), pDEST31 (FIG. 93), pDEST32 (FIG. 94), pDEST33 (FIG. 95), pDEST34 (FIG. 96), pDONR207 (FIG. 97), pMAB85 (FIG. 98), pMAB86 (FIG. 99), and fragments, mutants, variants, and derivatives thereof. However, it will be understood by one of ordinary skill that the present invention also encompasses other vectors not specifically designated herein, which comprise one or more of the isolated nucleic acid molecules of the invention encoding one or more recombination sites or portions thereof (or mutants, fragments, variants or derivatives thereof), and which may further comprise one or more additional physical or functional nucleotide sequences described herein which may optionally be operably linked to the one or more nucleic acid molecules encoding one or more recombination sites or portions thereof. Such additional vectors may be produced by one of ordinary skill according to the guidance provided in the present specification.
Polymerases
[0300] Preferred polypeptides having reverse transcriptase activity (i.e., those polypeptides able to catalyze the synthesis of a DNA molecule from an RNA template) for use in accordance with the present invention include, but are not limited to Moloney Murine Leukemia Virus (M-MLV) reverse transcriptase, Rous Sarcoma Virus (RSV) reverse transcriptase, Avian Myeloblastosis Virus (AMV) reverse transcriptase, Rous Associated Virus (RAV) reverse transcriptase, Myeloblastosis Associated Virus (MAV) reverse transcriptase, Human Immunodeficiency Virus (HIV) reverse transcriptase, retroviral reverse transcriptase, retrotransposon reverse transcriptase, hepatitis B reverse transcriptase, cauliflower mosaic virus reverse transcriptase and bacterial reverse transcriptase. Particularly preferred are those polypeptides having reverse transcriptase activity that are also substantially reduced in RNAse H activity (i.e., "RNAse H.sup.-" polypeptides). By a polypeptide that is "substantially reduced in RNase H activity" is meant that the polypeptide has less than about 20%, more preferably less than about 15%, 10% or 5%, and most preferably less than about 2%, of the RNase H activity of a wildtype or RNase H.sup.+ enzyme such as wildtype M-MLV reverse transcriptase. The RNase H activity may be determined by a variety of assays, such as those described, for example, in U.S. Pat. No. 5,244,797, in Kotewicz, M. L. et al., Nucl. Acids Res. 16:265 (1988) and in Gerard, G. F., et al., FOCUS 14(5):91 (1992), the disclosures of all of which are fully incorporated herein by reference. Suitable RNAse Hpolypeptides for use in the present invention include, but are not limited to, M-MLV H.sup.- reverse transcriptase, RSV H.sup.- reverse transcriptase, AMV H.sup.- reverse transcriptase, RAV H.sup.- reverse transcriptase, MAV H.sup.- reverse transcriptase, HIV H.sup.- reverse transcriptase, THERMOSCRIPT® reverse transcriptase and THERMOSCRIPT ® II reverse transcriptase, and SUPERSCRIPT® I reverse transcriptase and SUPERSCRIPT® IIreverse transcriptase, which are obtainable, for example, from Life Technologies, Inc. (Rockville, Md.). See generally published PCT application WO 98/47912.
[0301] Other polypeptides having nucleic acid polymerase activity suitable for use in the present methods include thermophilic DNA polymerases such as DNA polymerase I, DNA polymerase III, Klenow fragment, T7 polymerase, and T5 polymerase, and thermostable DNA polymerases including, but not limited to, Thermus thermophilus (Tth) DNA polymerase, Thermus aquaticus (Taq) DNA polymerase, Thermotoga neopolitana (Tne) DNA polymerase, Thermotoga maritima (Tma) DNA polymerase, Thermococcus litoralis (Tli or VENT®) DNA polymerase, Pyrococcus furiosus (Pfu) DNA polymerase, Pyrococcus species GB-D (or DEEPVENT®) DNA polymerase, Pyrococcus woosii (Pwo) DNA polymerase, Bacillus sterothermophilus (Bst) DNA polymerase, Sulfolobus acidocaldarius (Sac) DNA polymerase, Thermoplasma acidophilum (Tac) DNA polymerase, Thermus flavus (Tfl/Tub) DNA polymerase, Thermus ruber (Tru) DNA polymerase, Thermus brockianus (DYNAZYME®) DNA polymerase, Methanobacterium thermoautotrophicum (Mth) DNA polymerase, and mutants, variants and derivatives thereof. Such polypeptides are available commercially, for example from Life Technologies, Inc. (Rockville, Md.), New Englan BioLabs (Beverly, Mass.), and Sigma/Aldrich (St. Louis, Mo.).
Host Cells
[0302] The invention also relates to host cells comprising one or more of the nucleic acid molecules or vectors of the invention, particularly those nucleic acid molecules and vectors described in detail herein. Representative host cells that may be used according to this aspect of the invention include, but are not limited to, bacterial cells, yeast cells, plant cells and animal cells. Preferred bacterial host cells include Escherichia spp. cells (particularly E. coli cells and most particularly E. coli strains DH10B, Stb12, DH5α, DB3, DB3.1 (preferably E. coli LIBRARY EFFICIENCY® DB3.1® Competent Cells; Life Technologies, Inc., Rockville, Md.), DB4 and DB5; see U.S. Provisional Application No. 60/122,392, filed on Mar. 2, 1999, the disclosure of which is incorporated by reference herein in its entirety), Bacillus spp. cells (particularly B. subtilis and B. megaterium cells), Streptomyces spp. cells, Erwinia spp. cells, Klebsiella spp. cells, Serratia spp. cells (particularly S. marcessans cells), Pseudomonas spp. cells (particularly P. aeruginosa cells), and Salmonella spp. cells (particularly S. typhimurium and S. typhi cells). Preferred animal host cells include insect cells (most particularly Drosophila melanogaster cells, Spodoptera frugiperda Sf9 and Sf21 cells and Trichoplusa High-Five cells), nematode cells (particularly C. elegans cells), avian cells, amphibian cells (particularly Xenopus laevis cells), reptilian cells, and mammalian cells (most particularly CHO, COS, VERO, BILK and human cells). Preferred yeast host cells include Saccharomyces cerevisiae cells and Pichia pastoris cells. These and other suitable host cells are available commercially, for example from Life Technologies, Inc. (Rockville, Md.), American Type Culture Collection (Manassas, Va.), and Agricultural Research Culture Collection (NRRL; Peoria, Ill.).
[0303] Methods for introducing the nucleic acid molecules and/or vectors of the invention into the host cells described herein, to produce host cells comprising one or more of the nucleic acid molecules and/or vectors of the invention, will be familiar to those of ordinary skill in the art. For instance, the nucleic acid molecules and/or vectors of the invention may be introduced into host cells using well known techniques of infection, transduction, transfection, and transformation. The nucleic acid molecules and/or vectors of the invention may be introduced alone or in conjunction with other the nucleic acid molecules and/or vectors. Alternatively, the nucleic acid molecules and/or vectors of the invention may be introduced into host cells as a precipitate, such as a calcium phosphate precipitate, or in a complex with a lipid. Electroporation also may be used to introduce the nucleic acid molecules and/or vectors of the invention into a host. Likewise, such molecules may be introduced into chemically competent cells such as E. coli. If the vector is a virus, it may be packaged in vitro or introduced into a packaging cell and the packaged virus may be transduced into cells. Hence, a wide variety of techniques suitable for introducing the nucleic acid molecules and/or vectors of the invention into cells in accordance with this aspect of the invention are well known and routine to those of skill in the art. Such techniques are reviewed at length, for example, in Sambrook, J., et al., Molecular Cloning, a Laboratory Manual, 2nd Ed., Cold Spring Harbor, N.Y.: Cold Spring Harbor Laboratory Press, pp. 16.30-16.55 (1989), Watson, J. D., et al., Recombinant DNA, 2nd Ed., New York: W.H. Freeman and Co., pp. 213-234 (1992), and Winnacker, E.-L., From Genes to Clones, New York: VCH Publishers (1987), which are illustrative of the many laboratory manuals that detail these techniques and which are incorporated by reference herein in their entireties for their relevant disclosures.
Polypeptides
[0304] In another aspect, the invention relates to polypeptides encoded by the nucleic acid molecules of the invention (including polypeptides and amino acid sequences encoded by all possible reading frames of the nucleic acid molecules of the invention), and to methods of producing such polypeptides. Polypeptides of the present invention include purified or isolated natural products, products of chemical synthetic procedures, and products produced by recombinant techniques from a prokaryotic or eukaryotic host, including, for example, bacterial, yeast, insect, mammalian, avian and higher plant cells.
[0305] The polypeptides of the invention may be produced by synthetic organic chemistry, and are preferably produced by standard recombinant methods, employing one or more of the host cells of the invention comprising the vectors or isolated nucleic acid molecules of the invention. According to the invention, polypeptides are produced by cultivating the host cells of the invention (which comprise one or more of the nucleic acid molecules of the invention, preferably contained within an Expression Vector) under conditions favoring the expression of the nucleotide sequence contained on the nucleic acid molecule of the invention, such that the polypeptide encoded by the nucleic acid molecule of the invention is produced by the host cell. As used herein, "conditions favoring the expression of the nucleotide sequence" or "conditions favoring the production of a polypeptide" include optimal physical (e.g., temperature, humidity, etc.) and nutritional (e.g., culture medium, ionic) conditions required for production of a recombinant polypeptide by a given host cell. Such optimal conditions for a variety of host cells, including prokaryotic (bacterial), mammalian, insect, yeast, and plant cells will be familiar to one of ordinary skill in the art, and may be found, for example, in Sambrook, J., et al., Molecular Cloning, A Laboratory Manual, 2nd Ed., Cold Spring Harbor, N.Y.: Cold Spring Harbor Laboratory Press, (1989), Watson, J. D., et al., Recombinant DNA, 2nd Ed., New York: W.H. Freeman and Co., and Winnacker, E.-L., From Genes to Clones, New York: VCH Publishers (1987).
[0306] In some aspects, it may be desirable to isolate or purify the polypeptides of the invention (e.g., for production of antibodies as described below), resulting in the production of the polypeptides of the invention in isolated form. The polypeptides of the invention can be recovered and purified from recombinant cell cultures by well-known methods of protein purification that are routine in the art, including ammonium sulfate or ethanol precipitation, acid extraction, anion or cation exchange chromatography, phosphocellulose chromatography, hydrophobic interaction chromatography, affinity chromatography, hydroxylapatite chromatography and lectin chromatography. For example, His6 or GST fusion tags on polypeptides made by the methods of the invention may be isolated using appropriate affinity chromatography matrices which bind polypeptides bearing His6 or GST tags, as will be familiar to one of ordinary skill in the art. Polypeptides of the present invention include naturally purified products, products of chemical synthetic procedures, and products produced by recombinant techniques from a prokaryotic or eukaryotic host, including, for example, bacterial, yeast, higher plant, insect and mammalian cells. Depending upon the host employed in a recombinant production procedure, the polypeptides of the present invention may be glycosylated or may be non-glycosylated. In addition, polypeptides of the invention may also include an initial modified methionine residue, in some cases as a result of host-mediated processes.
[0307] Isolated polypeptides of the invention include those comprising the amino acid sequences encoded by one or more of the reading frames of the polynucleotides comprising one or more of the recombination site-encoding nucleic acid molecules of the invention, including those encoding attB1, attB2, attP1, attP2, attL1, attL2, attR1 and attR2 having the nucleotide sequences set forth in FIG. 9 (or nucleotide sequences complementary thereto), or fragments, variants, mutants and derivatives thereof; the complete amino acid sequences encoded by the polynucleotides contained in the deposited clones described herein; the amino acid sequences encoded by polynucleotides which hybridize under stringent hybridization conditions to polynucleotides having the nucleotide sequences encoding the recombination site sequences of the invention as set forth in FIG. 9 (or a nucleotide sequence complementary thereto); or a peptide or polypeptide comprising a portion or a fragment of the above polypeptides. The invention also relates to additional polypeptides having one or more additional amino acids linked (typically by peptidyl bonds to form a nascent polypeptide) to the polypeptides encoded by the recombination site nucleotide sequences or the deposited clones. Such additional amino acid residues may comprise one or more functional peptide sequences, for example one or more fusion partner peptides (e.g., GST, His6, Trx, etc.) and the like.
[0308] As used herein, the terms "protein," "peptide," "oligopeptide" and "polypeptide" are considered synonymous (as is commonly recognized) and each term can be used interchangeably as the context requires to indicate a chain of two or more amino acids, preferably five or more amino acids, or more preferably ten or more amino acids, coupled by (a) peptidyl linkage(s), unless otherwise defined in the specific contexts below. As is commonly recognized in the art, all polypeptide formulas or sequences herein are written from left to right and in the direction from amino terminus to carboxy terminus.
[0309] It will be recognized by those of ordinary skill in the art that some amino acid sequences of the polypeptides of the invention can be varied without significant effect on the structure or function of the polypeptides. If such differences in sequence are contemplated, it should be remembered that there will be critical areas on the protein which determine structure and activity. In general, it is possible to replace residues which form the tertiary structure, provided that residues performing a similar function are used. In other instances, the type of residue may be completely unimportant if the alteration occurs at a non-critical region of the polypeptide.
[0310] Thus, the invention further includes variants of the polypeptides of the invention, including allelic variants, which show substantial structural homology to the polypeptides described herein, or which include specific regions of these polypeptides such as the portions discussed below. Such mutants may include deletions, insertions, inversions, repeats, and type substitutions (for example, substituting one hydrophilic residue for another, but not strongly hydrophilic for strongly hydrophobic as a rule). Small changes or such "neutral" or "conservative" amino acid substitutions will generally have little effect on activity.
[0311] Typical conservative substitutions are the replacements, one for another, among the aliphatic amino acids Ala, Val, Leu and Ile; interchange of the hydroxylated residues Ser and Thr; exchange of the acidic residues Asp and Glu; substitution between the amidated residues Asn and Gln; exchange of the basic residues Lys and Arg; and replacements among the aromatic residues Phe and Tyr.
[0312] Thus, the fragment, derivative or analog of the polypeptides of the invention, such as those comprising peptides encoded by the recombination site nucleotide sequences described herein, may be (i) one in which one or more of the amino acid residues are substituted with a conservative or non-conservative amino acid residue (preferably a conservative amino acid residue), and such substituted amino acid residue may be encoded by the genetic code or may be an amino acid (e.g., desmosine, citrulline, ornithine, etc.) that is not encoded by the genetic code; (ii) one in which one or more of the amino acid residues includes a substituent group (e.g., a phosphate, hydroxyl, sulfate or other group) in addition to the normal "R" group of the amino acid; (iii) one in which the mature polypeptide is fused with another compound, such as a compound to increase the half-life of the polypeptide (for example, polyethylene glycol), or (iv) one in which additional amino acids are fused to the mature polypeptide, such as an immunoglobulin Fc region peptide, a leader or secretory sequence, a sequence which is employed for purification of the mature polypeptide (such as GST) or a proprotein sequence. Such fragments, derivatives and analogs are intended to be encompassed by the present invention, and are within the scope of those skilled in the art from the teachings herein and the state of the art at the time of invention.
[0313] The polypeptides of the present invention are preferably provided in an isolated form, and preferably are substantially purified. Recombinantly produced versions of the polypeptides of the invention can be substantially purified by the one-step method described in Smith and Johnson, Gene 67:31-40 (1988). As used herein, the term "substantially purified" means a preparation of an individual polypeptide of the invention wherein at least 50%, preferably at least 60%, 70%, or 75% and more preferably at least 80%, 85%, 90%, 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98% or 99% (by mass) of contaminating proteins (i.e., those that are not the individual polypeptides described herein or fragments, variants, mutants or derivatives thereof) have been removed from the preparation.
[0314] The polypeptides of the present invention include those which are at least about 50% identical, at least 60% identical, at least 65% identical, more preferably at least about 70%, at least about 75%, at least about 80%, at least about 85%, at least about 90%, at least about 95%, at least about 96%, at least about 97%, at least about 98% or at least about 99% identical, to the polypeptides described herein. For example, preferred attB1-containing polypeptides of the invention include those that are at least about 50% identical, at least 60% identical, at least 65% identical, more preferably at least about 70%, at least about 75%, at least about 80%, at least about 85%, at least about 90%, at least about 95%, at least about 96%, at least about 97%, at least about 98% or at least about 99% identical, to the polypeptide(s) encoded by the three reading frames of a polynucleotide comprising a nucleotide sequence of attB1 having a nucleic acid sequence as set forth in FIG. 9 (or a nucleic acid sequence complementary thereto), to a polypeptide encoded by a polynucleotide contained in the deposited cDNA clones described herein, or to a polypeptide encoded by a polynucleotide hybridizing under stringent conditions to a polynucleotide comprising a nucleotide sequence of attB1 having a nucleic acid sequence as set forth in FIG. 9 (or a nucleic acid sequence complementary thereto). Analogous polypeptides may be prepared that are at least about 65% identical, more preferably at least about 70%, at least about 75%, at least about 80%, at least about 85%, at least about 90%, at least about 95%, at least about 96%, at least about 97%, at least about 98% or at least about 99% identical, to the attB2, attP1, attP2, attL1, attL2, attR1 and attR2 polypeptides of the invention as depicted in FIG. 9. The present polypeptides also include portions or fragments of the above-described polypeptides with at least 5, 10, 15, 20, or 25 amino acids.
[0315] By a polypeptide having an amino acid sequence at least, for example, 65% "identical" to a reference amino acid sequence of a given polypeptide of the invention is intended that the amino acid sequence of the polypeptide is identical to the reference sequence except that the polypeptide sequence may include up to 35 amino acid alterations per each 100 amino acids of the reference amino acid sequence of a given polypeptide of the invention. In other words, to obtain a polypeptide having an amino acid sequence at least 65% identical to a reference amino acid sequence, up to 35% of the amino acid residues in the reference sequence may be deleted or substituted with another amino acid, or a number of amino acids up to 35% of the total amino acid residues in the reference sequence may be inserted into the reference sequence. These alterations of the reference sequence may occur at the amino (N-) or carboxy (C-) terminal positions of the reference amino acid sequence or anywhere between those terminal positions, interspersed either individually among residues in the reference sequence or in one or more contiguous groups within the reference sequence. As a practical matter, whether a given amino acid sequence is, for example, at least 65% identical to the amino acid sequence of a given polypeptide of the invention can be determined conventionally using known computer programs such as those described above for nucleic acid sequence identity determinations, or more preferably using the CLUSTAL W program (Thompson, J. D., et al., Nucleic Acids Res. 22:4673-4680 (1994)).
[0316] The polypeptides of the present invention can be used as molecular weight markers on SDS-PAGE gels or on molecular sieve gel filtration columns using methods well known to those of skill in the art. In addition, as described in detail below, the polypeptides of the present invention can be used to raise polyclonal and monoclonal antibodies which are useful in a variety of assays for detecting protein expression, localization, detection of interactions with other molecules, or for the isolation of a polypeptide (including a fusion polypeptide) of the invention.
[0317] In another aspect, the present invention provides a peptide or polypeptide comprising an epitope-bearing portion of a polypeptide of the invention, which may be used to raise antibodies, particularly monoclonal antibodies, that bind specifically to a one or more of the polypeptides of the invention. The epitope of this polypeptide portion is an immunogenic or antigenic epitope of a polypeptide of the invention. An "immunogenic epitope" is defined as a part of a protein that elicits an antibody response when the whole protein is the immunogen. These immunogenic epitopes are believed to be confined to a few loci on the molecule. On the other hand, a region of a protein molecule to which an antibody can bind is defined as an "antigenic epitope." The number of immunogenic epitopes of a protein generally is less than the number of antigenic epitopes (see, e.g., Geysen et al., Proc. Natl. Acad. Sci. USA 81:3998-4002 (1983)).
[0318] As to the selection of peptides or polypeptides bearing an antigenic epitope (i.e., that contain a region of a protein molecule to which an antibody can bind), it is well-known in the art that relatively short synthetic peptides that mimic part of a protein sequence are routinely capable of eliciting an antiserum that reacts with the partially mimicked protein (see, e.g., Sutcliffe, J. G., et al., Science 219:660-666 (1983)). Peptides capable of eliciting protein-reactive sera are frequently represented in the primary sequence of a protein, can be characterized by a set of simple chemical rules, and are not confined to the immunodominant regions of intact proteins (i.e., immunogenic epitopes) or to the amino or carboxy termini. Peptides that are extremely hydrophobic and those of six or fewer residues generally are ineffective at inducing antibodies that bind to the mimicked protein; longer peptides, especially those containing proline residues, usually are effective (Sutcliffe, J. G., et al., Science 219:660-666 (1983)).
[0319] Epitope-bearing peptides and polypeptides of the invention designed according to the above guidelines preferably contain a sequence of at least five, more preferably at least seven or more amino acids contained within the amino acid sequence of a polypeptide of the invention. However, peptides or polypeptides comprising a larger portion of an amino acid sequence of a polypeptide of the invention, containing about 30 to about 50 amino acids, or any length up to and including the entire amino acid sequence of a given polypeptide of the invention, also are considered epitope-bearing peptides or polypeptides of the invention and also are useful for inducing antibodies that react with the mimicked protein. Preferably, the amino acid sequence of the epitope-bearing peptide is selected to provide substantial solubility in aqueous solvents (i.e., the sequence includes relatively hydrophilic residues and highly hydrophobic sequences are preferably avoided); sequences containing proline residues are particularly preferred.
[0320] Non-limiting examples of epitope-bearing polypeptides or peptides that can be used to generate antibodies specific for the polypeptides of the invention include certain epitope-bearing regions of the polypeptides comprising amino acid sequences encoded by polynucleotides comprising one or more of the recombination site-encoding nucleic acid molecules of the invention, including those encoding attB1, attB2, attP1, attP2, attL1, attL2, attR1 and attR2 having the nucleotide sequences set forth in FIG. 9 (or a nucleotide sequence complementary thereto); the complete amino acid sequences encoded by the three reading frames of the polynucleotides contained in the deposited clones described herein; and the amino acid sequences encoded by all reading frames of polynucleotides which hybridize under stringent hybridization conditions to polynucleotides having the nucleotide sequences encoding the recombination site sequences (or portions thereof) of the invention as set forth in FIG. 9 (or a nucleic acid sequence complementary thereto). Other epitope-bearing polypeptides or peptides that may be used to generate antibodies specific for the polypeptides of the invention will be apparent to one of ordinary skill in the art based on the primary amino acid sequences of the polypeptides of the invention described herein, via the construction of Kyte-Doolittle hydrophilicity and Jameson-Wolf antigenic index plots of the polypeptides of the invention using, for example, PROTEAN computer software (DNASTAR, Inc.; Madison, Wis.).
[0321] The epitope-bearing peptides and polypeptides of the invention may be produced by any conventional means for making peptides or polypeptides including recombinant means using nucleic acid molecules of the invention. For instance, a short epitope-bearing amino acid sequence may be fused to a larger polypeptide which acts as a carrier during recombinant production and purification, as well as during immunization to produce anti-peptide antibodies. Epitope-bearing peptides also may be synthesized using known methods of chemical synthesis (see, e.g., U.S. Pat. No. 4,631,211 and Houghten, R. A., Proc. Natl. Acad. Sci. USA 82:5131-5135 (1985), both of which are incorporated by reference herein in their entireties).
[0322] As one of skill in the art will appreciate, the polypeptides of the present invention and epitope-bearing fragments thereof may be immobilized onto a solid support, by techniques that are well-known and routine in the art. By "solid support" is intended any solid support to which a peptide can be immobilized. Such solid supports include, but are not limited to nitrocellulose, diazocellulose, glass, polystyrene, polyvinylchloride, polypropylene, polyethylene, dextran, Sepharose, agar, starch, nylon, beads and microtitre plates. Linkage of the peptide of the invention to a solid support can be accomplished by attaching one or both ends of the peptide to the support. Attachment may also be made at one or more internal sites in the peptide. Multiple attachments (both internal and at the ends of the peptide) may also be used according to the invention. Attachment can be via an amino acid linkage group such as a primary amino group, a carboxyl group, or a sulfhydryl (SH) group or by chemical linkage groups such as with cyanogen bromide (CNBr) linkage through a spacer. For non-covalent attachments to the support, addition of an affinity tag sequence to the peptide can be used such as GST (Smith, D. B., and Johnson, K. S., Gene 67:31 (1988)), polyhistidines (Hochuli, E., et al., J. Chromatog. 411:77 (1987)), or biotin. Such affinity tags may be used for the reversible attachment of the peptide to the support. Such immobilized polypeptides or fragments may be useful, for example, in isolating antibodies directed against one or more of the polypeptides of the invention, or other proteins or peptides that recognize other proteins or peptides that bind to one or more of the polypeptides of the invention, as described below.
[0323] As one of skill in the art will also appreciate, the polypeptides of the present invention and the epitope-bearing fragments thereof described herein can be combined with one or more fusion partner proteins or peptides, or portions thereof, including but not limited to GST, His6, Trx, and portions of the constant domain of immunoglobulins (Ig), resulting in chimeric or fusion polypeptides. These fusion polypeptides facilitate purification of the polypeptides of the invention (EP 0 394 827; Traunecker et al., Nature 331:84-86 (1988)) for use in analytical or diagnostic (including high-throughput) format.
Antibodies
[0324] In another aspect, the invention relates to antibodies that recognize and bind to the polypeptides (or epitope-bearing fragments thereof) or nucleic acid molecules (or portions thereof) of the invention. In a related aspect, the invention relates to antibodies that recognize and bind to one or more polypeptides encoded by all reading frames of one or more recombination site nucleic acid sequences or portions thereof, or to one or more nucleic acid molecules comprising one or more recombination site nucleic acid sequences or portions thereof, including but not limited to att sites (including attB1, attB2, attP1, attP2, attL1, attL2, attR1, attR2 and the like), lox sites (e.g., loxP, loxP511, and the like), FRT, and the like, or mutants, fragments, variants and derivatives thereof. See generally U.S. Pat. No. 5,888,732, which is incorporated herein by reference in its entirety. The antibodies of the present invention may be polyclonal or monoclonal, and may be prepared by any of a variety of methods and in a variety of species according to methods that are well-known in the art. See, for instance, U.S. Pat. No. 5,587,287; Sutcliffe, J. G., et al., Science 2/9:660-666 (1983); Wilson et al., Cell 37: 767 (1984); and Bittle, F. J., et al., J. Gen. Viral. 66:2347-2354 (1985). Antibodies specific for any of the polypeptides or nucleic acid molecules described herein, such as antibodies specifically binding to one or more of the polypeptides encoded by the recombination site nucleotide sequences, or one or more nucleic acid molecules, described herein or contained in the deposited clones, antibodies against fusion polypeptides (e.g., binding to fusion polypeptides between one or more of the fusion partner proteins and one or more of the recombination site polypeptides of the invention, as described herein), and the like, can be raised against the intact polypeptides or polynucleotides of the invention or one or more antigenic polypeptide fragments thereof.
[0325] As used herein, the term "antibody" (Ab) may be used interchangeably with the terms "polyclonal antibody" or "monoclonal antibody" (mAb), except in specific contexts as described below. These terms, as used herein, are meant to include intact molecules as well as antibody fragments (such as, for example, Fab and F(ab')2 fragments) which are capable of specifically binding to a polypeptide or nucleic acid molecule of the invention or a portion thereof. It will therefore be appreciated that, in addition to the intact antibodies of the invention, Fab, F(ab')2 and other fragments of the antibodies described herein, and other peptides and peptide fragments that bind one or more polypeptides or polynucleotides of the invention, are also encompassed within the scope of the invention. Such antibody fragments are typically produced by proteolytic cleavage of intact antibodies, using enzymes such as papain (to produce Fab fragments) or pepsin (to produce F(ab')2 fragments). Antibody fragments, and peptides or peptide fragments, may also be produced through the application of recombinant DNA technology or through synthetic chemistry.
[0326] Epitope-bearing peptides and polypeptides, and nucleic acid molecules or portions thereof, of the invention may be used to induce antibodies according to methods well known in the art, as generally described herein (see, e.g., Sutcliffe, et al., supra; Wilson, et al., supra; and Bittle, F. J., et al., J. Gen. Virol. 66:2347-2354 (1985)).
[0327] Polyclonal antibodies according to this aspect of the invention may be made by immunizing an animal with one or more of the polypeptides or nucleic acid molecules of the invention described herein or portions thereof according to standard techniques (see, e.g., Harlow, E., and Lane, D., Antibodies: A Laboratory Manual, Cold Spring Harbor, N.Y.: Cold Spring Harbor Laboratory Press (1988); Kaufman, P. B., et al., In: Handbook of Molecular and Cellular Methods in Biology and Medicine, Boca Raton, Fla.: CRC Press, pp. 468-469 (1995)). For producing antibodies that recognize and bind to the polypeptides or nucleic acid molecules of the invention or portions thereof, animals may be immunized with free peptide or free nucleic acid molecules; however, antibody titer may be boosted by coupling of the peptide to a macromolecular carrier, such as albumin, KLH, or tetanus toxoid (particularly for producing antibodies against the nucleic acid molecules of the invention or portions thereof; see Harlow and Lane, supra, at page 154), or to a solid phase carrier such as a latex or glass microbead. For instance, peptides containing cysteine may be coupled to carrier using a linker such as m-maleimidobenzoyl-N-hydroxysuccinimide ester (MBS), while other peptides may be coupled to carrier using a more general linking agent such as glutaraldehyde. Animals such as rabbits, rats and mice may be immunized with either free (if the polypeptide immunogen is larger than about 25 amino acids in length) or carrier-coupled peptides or nucleic acid molecules, for instance, by intraperitoneal and/or intradermal injection of emulsions containing about 100 μg peptide, polynucleotide, or carrier protein, and Freund's adjuvant. Several booster injections may be needed, for instance, at intervals of about two weeks, to provide a useful titer of antibody which can be detected, for example, by ELISA assay using free peptide or nucleic acid molecule adsorbed to a solid surface. In another approach, cells expressing one or more of the polypeptides or polynucleotides of the invention or an antigenic fragment thereof can be administered to an animal in order to induce the production of sera containing polyclonal antibodies, according to routine immunological methods. In yet another method, a preparation of one or more of the polypeptides or polynucleotides of the invention is prepared and purified as described herein, to render it substantially free of natural contaminants. Such a preparation may then be introduced into an animal in order to produce polyclonal antisera of greater specific activity. The titer of antibodies in serum from an immunized animal, regardless of the method of immunization used, may be increased by selection of anti-peptide or anti-polynucleotide antibodies, for instance, by adsorption to the peptide or polynucleotide on a solid support and elution of the selected antibodies according to methods well known in the art.
[0328] In an alternative method, the antibodies of the present invention are monoclonal antibodies (or fragments thereof which bind to one or more of the polypeptides of the invention). Such monoclonal antibodies can be prepared using hybridoma technology (Kohler et al., Nature 256:495 (1975); Kohler et al., Eur. J. Immunol. 6:511 (1976); Kohler et al., Eur. J. Immunol. 6:292 (1976); Hammerling et al., In: Monoclonal Antibodies and T-Cell Hybridomas, Elsevier, N.Y., pp. 563-681 (1981)). In general, such procedures involve immunizing an animal (preferably a mouse) with a polypeptide or polynucleotide of the invention (or a fragment thereof), or with a cell expressing a polypeptide or polynucleotide of the invention (or a fragment thereof). The splenocytes of such mice are extracted and fused with a suitable myeloma cell line. Any suitable myeloma cell line may be employed in accordance with the present invention; however, it is preferable to employ the parent myeloma cell line (SP2O), available from the American Type Culture Collection, Rockville, Md. After fusion, the resulting hybridoma cells are selectively maintained in HAT medium, and then cloned by limiting dilution as described by Wands et al. (Gastroenterol. 80:225-232 (1981)). The hybridoma cells obtained through such a selection are then assayed to identify clones which secrete antibodies capable of binding one or more of the polypeptides or nucleic acid molecules of the invention, or fragments thereof. Hence, the present invention also provides hybridoma cells and cell lines producing monoclonal antibodies of the invention, particularly that recognize and bind to one or more of the polypeptides or nucleic acid molecules of the invention.
[0329] Alternatively, additional antibodies capable of binding to one or more of the polypeptides of the invention, or fragments thereof, may be produced in a two-step procedure through the use of anti-idiotypic antibodies. Such a method makes use of the fact that antibodies are themselves antigens, and that, therefore, it is possible to obtain an antibody which binds to a second antibody. In accordance with this method, antibodies specific for one or more of the polypeptides or polynucleotides of the invention, prepared as described above, are used to immunize an animal, preferably a mouse. The splenocytes of such an animal are then used to produce hybridoma cells, and the hybridoma cells are screened to identify clones which produce an antibody whose ability to bind to an antibody specific for one or more of the polypeptides or polynucleotides of the invention can be blocked by polypeptides of the invention themselves. Such antibodies comprise anti-idiotypic antibodies to the antibodies recognizing one or more of the polypeptides or polynucleotides of the invention, and can be used to immunize an animal to induce formation of further antibodies specific for one or more of the polypeptides or polynucleotides of the invention.
[0330] For use, the antibodies of the invention may optionally be detectably labeled by covalent or non-covalent attachment of one or more labels, including but not limited to chromogenic, enzymatic, radioisotopic, isotopic, fluorescent, toxic, chemiluminescent, or nuclear magnetic resonance contrast agents or other labels.
[0331] Examples of suitable enzyme labels include malate dehydrogenase, staphylococcal nuclease, delta-5-steroid isomerase, yeast-alcohol dehydrogenase, alpha-glycerol phosphate dehydrogenase, triose phosphate isomerase, peroxidase, alkaline phosphatase, asparaginase, glucose oxidase, beta-galactosidase, ribonuclease, urease, catalase, glucose-6-phosphate dehydrogenase, glucoamylase, and acetylcholine esterase.
[0332] Examples of suitable radioisotopic labels include 3H, 111In, 125I, 131I, 32P, 35S, 14C, 51Cr, 57To, 58Co, 59Fe, 75Se, 152Eu, 90Y, 67Cu, 217Ci, 211At, 212Pb, 47Sc, 109Pd, etc. 111In is a preferred isotope where in vivo imaging is used since its avoids the problem of dehalogenation of the 125I or 131I-labeled monoclonal antibody by the liver. In addition, this radionucleotide has a more favorable gamma emission energy for imaging (Perkins et al., Eur. J. Nucl. Med. 10:296-301 (1985); Carasquillo et al., J. Nucl. Med. 28:281-287 (1987)). For example, 111In coupled to monoclonal antibodies with 1-(P-isothiocyanatobenzyl)-DPTA has shown little uptake in non-tumorous tissues, particularly the liver, and therefore enhances specificity of tumor localization (Esteban et al., J. Nucl. Med. 28:861-870 (1987)).
[0333] Examples of suitable non-radioactive isotopic labels include 157Gd, 55Mn, 162Dy, 52Tr, and 56Fe.
[0334] Examples of suitable fluorescent labels include an 152Eu label, a fluorescein label, an isothiocyanate label, a rhodamine label, a phycoerythrin label, a phycocyanin label, an allophycocyanin label, an o-phthaldehyde label, a green fluorescent protein (GFP) label, and a fluorescamine label.
[0335] Examples of suitable toxin labels include diphtheria toxin, ricin, and cholera toxin.
[0336] Examples of chemiluminescent labels include a luminal label, an isoluminal label, an aromatic acridinium ester label, an imidazole label, an acridinium salt label, an oxalate ester label, a luciferin label, a luciferase label, and an aequorin label.
[0337] Examples of nuclear magnetic resonance contrasting agents include heavy metal nuclei such as Gd, Mn, and iron.
[0338] Typical techniques for binding the above-described labels to the antibodies of the invention are provided by Kennedy et al., Clin. Chim. Acta 70:1-31 (1976), and Schurs et al., Clin. Chim. Acta 81:1-40 (1977). Coupling techniques mentioned in the latter are the glutaraldehyde method, the periodate method, the dimaleimide method, the m-maleimidobenzyl-N-hydroxy-succinimide ester method, all of which methods are incorporated by reference herein.
[0339] It will be appreciated by one of ordinary skill that the antibodies of the present invention may alternatively be coupled to a solid support, to facilitate, for example, chromatographic and other immunological procedures using such solid phase-immobilized antibodies. Included among such procedures are the use of the antibodies of the invention to isolate or purify polypeptides comprising one or more epitopes encoded by the nucleic acid molecules of the invention (which may be fusion polypeptides or other polypeptides of the invention described herein), or to isolate or purify polynucleotides comprising one or more recombination site sequences of the invention or portions thereof. Methods for isolation and purification of polypeptides (and, by analogy, polynucleotides) by affinity chromatography, for example using the antibodies of the invention coupled to a solid phase support, are well-known in the art and will be familiar to one of ordinary skill. The antibodies of the invention may also be used in other applications, for example to cross-link or couple two or more proteins, polypeptides, polynucleotides, or portions thereof into a structural and/or functional complex. In one such use, an antibody of the invention may have two or more distinct epitope-binding regions that may bind, for example, a first polypeptide (which may be a polypeptide of the invention) at one epitope-binding region on the antibody and a second polypeptide (which may be a polypeptide of the invention) at a second epitope-binding region on the antibody, thereby bringing the first and second polypeptides into close proximity to each other such that the first and second polypeptides are able to interact structurally and/or functionally (as, for example, linking an enzyme and its substrate to carry out enzymatic catalysis, or linking an effector molecule and its receptor to carry out or induce a specific binding of the effector molecule to the receptor or a response to the effector molecule mediated by the receptor). Additional applications for the antibodies of the invention include, for example, the preparation of large-scale arrays of the antibodies, polypeptides, or nucleic acid molecules of the invention, or portions thereof, on a solid support, for example to facilitate high-throughput screening of protein or RNA expression by host cells containing nucleic acid molecules of the invention (known in the art as "chip array" protocols; see, e.g., U.S. Pat. Nos. 5,856,101, 5,837,832, 5,770,456, 5,744,305, 5,631,734, and 5,593,839, which are directed to production and use of chip arrays of polypeptides (including antibodies) and polynucleotides, and the disclosures of which are incorporated herein by reference in their entireties). By "solid support" is intended any solid support to which an antibody can be immobilized. Such solid supports include, but are not limited to nitrocellulose, diazocellulose, glass, polystyrene, polyvinylchloride, polycarbonate, polypropylene, polyethylene, dextran, Sepharose, agar, starch, nylon, beads and microtitre plates. Preferred are beads made of glass, latex or a magnetic material. Linkage of an antibody of the invention to a solid support can be accomplished by attaching one or both ends of the antibody to the support. Attachment may also be made at one or more internal sites in the antibody. Multiple attachments (both internal and at the ends of the antibody) may also be used according to the invention. Attachment can be via an amino acid linkage group such as a primary amino group, a carboxyl group, or a sulfhydryl (SH) group or by chemical linkage groups such as with cyanogen bromide (CNBr) linkage through a spacer. For non-covalent attachments, addition of an affinity tag sequence to the peptide can be used such as GST (Smith, D. B., and Johnson, K. S., Gene 67:31 (1988)), polyhistidines (Hochuli, E., et al., J. Chromatog. 411:77 (1987)), or biotin. Alternatively, attachment can be accomplished using a ligand which binds the Fc region of the antibodies of the invention, e.g., protein A or protein G. Such affinity tags may be used for the reversible attachment of the antibodies to the support. Peptides may also be recognized via specific ligand-receptor interactions or using phage display methodologies that will be familiar to the skilled artisan, for their ability to bind polypeptides of the invention or fragments thereof.
Kits
[0340] In another aspect, the invention provides kits which may be used in producing the nucleic acid molecules, polypeptides, vectors, host cells, and antibodies, and in the recombinational cloning methods, of the invention. Kits according to this aspect of the invention may comprise one or more containers, which may contain one or more of the nucleic acid molecules, primers, polypeptides, vectors, host cells, or antibodies of the invention. In particular, a kit of the invention may comprise one or more components (or combinations thereof) selected from the group consisting of one or more recombination proteins (e.g., Int) or auxiliary factors (e.g. IHF and/or Xis) or combinations thereof, one or more compositions comprising one or more recombination proteins or auxiliary factors or combinations thereof (for example, GATEWAY® LR Clonase® Enzyme Mix or GATEWAY® BP Clonase® Enzyme Mix) one or more Destination Vector molecules (including those described herein), one or more Entry Clone or Entry Vector molecules (including those described herein), one or more primer nucleic acid molecules (particularly those described herein), one or more host cells (e.g. competent cells, such as E. coli cells, yeast cells, animal cells (including mammalian cells, insect cells, nematode cells, avian cells, fish cells, etc.), plant cells, and most particularly E. coli DB3, DB3.1 (preferably E. coli LIBRARY EFFICIENCY® DB3.1® Competent Cells; Life Technologies, Inc., Rockville, Md.), DB4 and DB5; see U.S. Provisional Application No. 60/122,392, filed on Mar. 2, 1999, and the corresponding U.S. Utility application Ser. No. 09/518,188 of Hartley et al., entitled "Cells Resistant to Toxic Genes and Uses Thereof," filed on even day herewith, the disclosures of which are incorporated by reference herein in its entirety), and the like. In related aspects, the kits of the invention may comprise one or more nucleic acid molecules encoding one or more recombination sites or portions thereof, such as one or more nucleic acid molecules comprising a nucleotide sequence encoding the one or more recombination sites (or portions thereof) of the invention, and particularly one or more of the nucleic acid molecules contained in the deposited clones described herein. Kits according to this aspect of the invention may also comprise one or more isolated nucleic acid molecules of the invention, one or more vectors of the invention, one or more primer nucleic acid molecules of the invention, and/or one or more antibodies of the invention. The kits of the invention may further comprise one or more additional containers containing one or more additional components useful in combination with the nucleic acid molecules, polypeptides, vectors, host cells, or antibodies of the invention, such as one or more buffers, one or more detergents, one or more polypeptides having nucleic acid polymerase activity, one or more polypeptides having reverse transcriptase activity, one or more transfection reagents, one or more nucleotides, and the like. Such kits may be used in any process advantageously using the nucleic acid molecules, primers, vectors, host cells, polypeptides, antibodies and other compositions of the invention, for example in methods of synthesizing nucleic acid molecules (e.g., via amplification such as via PCR), in methods of cloning nucleic acid molecules (preferably via recombinational cloning as described herein), and the like.
Optimization of Recombinational Cloning System
[0341] The usefulness of a particular nucleic acid molecule, or vector comprising a nucleic acid molecule, of the invention in methods of recombinational cloning may be determined by any one of a number of assay methods. For example, Entry and Destination vectors of the present invention may be assessed for their ability to function (i.e., to mediate the transfer of a nucleic acid molecule, DNA segment, gene, cDNA molecule or library from a cloning vector to an Expression Vector) by carrying out a recombinational cloning reaction as described in more detail in the Examples below and as described in U.S. application Ser. No. 08/663,002, filed Jun. 7, 1996 (now U.S. Pat. No. 5,888,732), Ser. No. 09/005,476, filed Jan. 12, 1998, Ser. No. 09/177,387, filed Oct. 23, 1998, and 60/108,324, filed Nov. 13, 1998, the disclosures of which are incorporated by reference herein in their entireties. Alternatively, the functionality of Entry and Destination Vectors prepared according to the invention may be assessed by examining the ability of these vectors to recombine and create cointegrate molecules, or to transfer a nucleic acid molecule of interest, using an assay such as that described in detail below in Example 19. Analogously, the formulation of compositions comprising one or more recombination proteins or combinations thereof, for example GATEWAY® LR Clonase® Enzyme Mix and GATEWAY® BP Clonase® Enzyme Mix, may be optimized using assays such as those described below in Example 18.
Uses
[0342] There are a number of applications for the compositions, methods and kits of the present invention. These uses include, but are not limited to, changing vectors, targeting gene products to intracellular locations, cleaving fusion tags from desired proteins, operably linking nucleic acid molecules of interest to regulatory genetic sequences (e.g., promoters, enhancers, and the like), constructing genes for fusion proteins, changing copy number, changing replicons, cloning into phages, and cloning, e.g., PCR products, genomic DNAs, and cDNAs. In addition, the nucleic acid molecules, vectors, and host cells of the invention may be used in the production of polypeptides encoded by the nucleic acid molecules, in the production of antibodies directed against such polypeptides, in recombinational cloning of desired nucleic acid sequences, and in other applications that may be enhanced or facilitated by the use of the nucleic acid molecules, vectors, and host cells of the invention.
[0343] In particular, the nucleic acid molecules, vectors, host cells, polypeptides, antibodies, and kits of the invention may be used in methods of transferring one or more desired nucleic acid molecules or DNA segments, for example one or more genes, cDNA molecules or cDNA libraries, into a cloning or Expression Vector for use in transforming additional host cells for use in cloning or amplification of, or expression of the polypeptide encoded by, the desired nucleic acid molecule or DNA segment. Such recombinational cloning methods which may advantageously use the nucleic acid molecules, vectors, and host cells of the invention, are described in detail in the Examples below, and in commonly owned U.S. application Ser. No. 08/486,139, filed Jun. 7, 1995, Ser. No. 08/663,002, filed Jun. 7, 1996 (now U.S. Pat. No. 5,888,732), Ser. No. 09/005,476, filed Jan. 12, 1998, Ser. No. 09/177,387, filed Oct. 23, 1998, and 60/108,324, filed Nov. 13, 1998, the disclosures of all of which are incorporated by reference herein in their entireties.
[0344] It will be understood by one of ordinary skill in the relevant arts that other suitable modifications and adaptations to the methods and applications described herein are readily apparent from the description of the invention contained herein in view of information known to the ordinarily skilled artisan, and may be made without departing from the scope of the invention or any embodiment thereof. Having now described the present invention in detail, the same will be more clearly understood by reference to the following examples, which are included herewith for purposes of illustration only and are not intended to be limiting of the invention.
EXAMPLES
Example 1
Recombination Reactions of Bacteriophage λ
[0345] The E. coli bacteriophage λ can grow as a lytic phage, in which case the host cell is lysed, with the release of progeny virus. Alternatively, lambda can integrate into the genome of its host by a process called lysogenization (see FIG. 60). In this lysogenic state, the phage genome can be transmitted to daughter cells for many generations, until conditions arise that trigger its excision from the genome. At this point, the virus enters the lytic part of its life cycle. The control of the switch between the lytic and lysogenic pathways is one of the best understood processes in molecular biology (M. Ptashne, A Genetic Switch, Cell Press, 1992).
[0346] The integrative and excisive recombination reactions of A, performed in vitro, are the basis of Recombinational Cloning System of the present invention. They can be represented schematically as follows:
attB×attPattL×attR (where "×" signifies recombination)
[0347] The four att sites contain binding sites for the proteins that mediate the reactions. The wild type attP, attB, attL, and attR sites contain about 243, 25, 100, and 168 base pairs, respectively. The attB×attP reaction (hereinafter referred to as a "BP Reaction," or alternatively and equivalently as an "Entry Reaction" or a "Gateward Reaction") is mediated by the proteins hit and IHF. The attL×attR reaction (hereinafter referred to as an "LR Reaction," or alternatively and equivalently as a "Destination Reaction") is mediated by the proteins hit, IHF, and Xis. Int (integrate) and Xis (excisionase) are encoded by the A genome, while IHF (integration host factor) is an E. coli protein. For a general review of lambda recombination, see: A. Landy, Ann. Rev. Biochem. 58: 913-949 (1989).
Example 2
Recombination Reactions of the Recombinational Cloning System
[0348] The LR Reaction--the exchange of a DNA segment from an Entry Clone to a Destination Vector--is the in vitro version of the A excision reaction:
attL×attRattB+attP.
[0349] There is a practical imperative for this configuration: after an LR Reaction in one configuration of the present method, an att site usually separates a functional motif (such as a promoter or a fusion tag) from a nucleic acid molecule of interest in an Expression Clone, and the 25 bp attB site is much smaller than the attP, attL, and attR sites.
[0350] Note that the recombination reaction is conservative, i.e., there is no net synthesis or loss of base pairs. The DNA segments that flank the recombination sites are merely switched. The wild type A recombination sites are modified for purposes of the GATEWAY® Cloning System, as follows:
[0351] To create certain preferred Destination Vectors, a part (43 bp) of attR was removed, to make the excisive reaction irreversible and more efficient (W. Bushman et al., Science 230: 906, 1985). The attR sites in preferred Destination Vectors of the invention are 125 bp in length. Mutations were made to the core regions of the att sites, for two reasons: (1) to eliminate stop codons, and (2) to ensure specificity of the recombination reactions (i.e., attR1 reacts only with attL1, attR2 reacts only with attL2, etc.).
[0352] Other mutations were introduced into the short (5 bp) regions flanking the 15 bp core regions of the attB sites to minimize secondary structure formation in single-stranded forms of attB plasmids, e.g., in phagemid ssDNA or in mRNA. Sequences of attB1 and attB2 to the left and right of a nucleic acid molecule of interest after it has been cloned into a Destination Vector are given in FIG. 6.
[0353] FIG. 61 illustrates how an Entry Clone and a Destination Vector recombine in the LR Reaction to form a co-integrate, which resolves through a second reaction into two daughter molecules. The two daughter molecules have the same general structure regardless of which pair of sites, attL1 and attR1 or attL2 and attR2, react first to form the co-integrate. The segments change partners by these reactions, regardless of whether the parental molecules are both circular, one is circular and one is linear, or both are linear. In this example, selection for ampicillin resistance carried on the Destination Vector, which also carries the death gene ccdB, provides the means for selecting only for the desired attB product plasmid.
Example 3
Protein Expression in the Recombinational Cloning System
[0354] Proteins are expressed in vivo as a result of two processes, transcription (DNA into RNA), and translation (RNA into protein). For a review of protein expression in prokaryotes and eukaryotes, see Example 13 below. Many vectors (pUC, BlueScript, pGem) use interruption of a transcribed lacZ gene for blue-white screening. These plasmids, and many Expression Vectors, use the lac promoter to control expression of cloned genes. Transcription from the lac promoter is turned on by adding the inducer IPTG. However, a low level of RNA is made in the absence of inducer, i.e., the lac promoter is never completely off. The result of this "leakiness" is that genes whose expression is harmful to E. coli may prove difficult or impossible to clone in vectors that contain the lac promoter, or they may be cloned only as inactive mutants.
[0355] In contrast to other gene expression systems, nucleic acid molecules cloned into an Entry Vector may be designed not to be expressed. The presence of the strong transcriptional terminator rrnB (Orosz, et al., Eur. J. Biochem. 201: 653, 1991) just upstream of the attL1 site keeps transcription from the vector promoters (drug resistance and replication origin) from reaching the cloned gene. However, if a toxic gene is cloned into a Destination Vector, the host may be sick, just as in other expression systems. But the reliability of subcloning by in vitro recombination makes it easier to recognize that this has happened--and easier to try another expression option in accordance with the methods of the invention, if necessary.
Example 4
Choosing the Right Entry Vector
[0356] There are two kinds of choices that must be made in choosing the best Entry Vector, dictated by (1) the particular DNA segment that is to be cloned, and (2) what is to be accomplished with the cloned DNA segment. These factors are critical in the choice of Entry Vector used, because when the desired nucleic acid molecule of interest is moved from the Entry Vector to a Destination Vector, all the base pairs between the nucleic acid molecule of interest and the Int cutting sites in attL1 and attL2 (such as in FIG. 6) move into the Destination Vector as well. For genomic DNAs that are not expressed as a result of moving into a Destination Vector, these decisions are not as critical.
[0357] For example, if an Entry Vector with certain translation start signals is used, those sequences will be translated into amino acids if an amino-terminal fusion to the desired nucleic acid molecule of interest is made. Whether the desired nucleic acid molecule of interest is to be expressed as fusion protein, native protein, or both, dictates whether translational start sequences must be included between the attB sites of the clone (native protein) or, alternatively, supplied by the Destination Vector (fusion protein). In particular, Entry Clones that include translational start sequences may prove less suitable for making fusion proteins, as internal initiation of translation at these sites can decrease the yield of N-terminal fusion protein. These two types of expression afforded by the compositions and methods of the invention are illustrated in FIG. 62.
[0358] No Entry Vector is likely to be optimal for all applications. The nucleic acid molecule of interest may be cloned into any of several optimal Entry Vectors.
[0359] As an example, consider pENTR7 (FIG. 16) and pENTR11 (FIG. 20), which are useful in a variety of applications, including (but not limited to):
[0360] Cloning cDNAs from most of the commercially available libraries. The sites to the left and right of the ccdB death gene have been chosen so that directional cloning is possible if the DNA to be cloned does not have two or
[0361] Cloning of genes directionally: SalI, BamHI, XmnI (blunt), or KpnI on the left of ccdB; NotI, XhoI, XbaI, or EcoRV (blunt), on the right.
[0362] Cloning of genes or gene fragments with a blunt amino end at the XmnI site. The XmnI site has four of the six most favored bases for eukaryotic expression (see Example 13, below), so that if the first three bases of the DNA to be cloned are ATG, the open reading frame (ORF) will be expressed in eukaryotic cells (e.g., mammalian cells, insect cells, yeast cells) when it is transcribed in the appropriate Destination Vector. In addition, in pENTR11, a Shine-Dalgarno sequence is situated 8 bp upstream, for initiating protein synthesis in a prokaryotic host cell (particularly a bacterial cell, such as E. coli) at an ATG.
[0363] Cleaving off amino terminal fusions (e.g., His6, GST, or thioredoxin) using the highly specific TEV (Tobacco Etch Virus) protease (available from Life Technologies, Inc.). If the nucleic acid molecule of interest is cloned at the blunt XmnI site, TEV cleavage will leave two amino acids on the amino end of the expressed protein.
[0364] Selecting against uncut or singly cut Entry Vector molecules during cloning with restriction enzymes and ligase. If the ccdB gene is not removed with a double digest, it will kill any recipient E. coli cell that does not contain a mutation that makes the cell resistant to ccdB (see U.S. Provisional Application No. 60/122,392, filed on Mar. 2, 1999, the disclosure of which is incorporated by reference herein in its entirety).
[0365] Allowing production of amino fusions with ORFs in all cloning sites. There are no stop codons (in the attL1 reading frame) upstream of the ccdB gene.
[0366] In addition, pENTR11 is also useful in the following applications:
[0367] Cloning cDNAs that have an NcoI site at the initiating ATG into the NcoI site. Similar to the XmnI site, this site has four of the six most favored bases for eukaryotic expression. Also, a Shine-Dalgarno sequence is situated 8 bp upstream, for initiating protein synthesis in a prokaryotic host cell (particularly a bacterial cell, such as E. coli) at an ATG.
[0368] Producing carboxy fusion proteins with ORFs positioned in phase with the reading frame convention for carboxy-terminal fusions (see FIG. 20A).
[0369] Table 1 lists some non-limiting examples of Entry Vectors and their characteristics, and FIGS. 10-20 show their cloning sites. All of the Entry Vectors listed in Table 1 are available commercially from Life Technologies, Inc., Rockville, Md. Other Entry Vectors not specifically listed here, which comprise alternative or additional features may be made by one of ordinary skill using routine methods of molecular and cellular biology, in view of the disclosure contained herein.
TABLE-US-00003 TABLE 1 Examples of Entry Vectors Native Class of Protein in Protein Mnemonic Entry Distinctive Amino Native Protein Eukaryotic Synthesis Designation Name Vector Cloning Sites Fusions in E. coli Cells Features pENTR- Minimal Alternative Reading frame Good Poor Good Minimal amino 1A, 2B, 3C blunt RF Reading A, B, or C; blunt acids between A, B, C Frame cut closest to tag and protein; Vectors attL1 no SD pENTR4 Minimal Restr. Enz. Nco I site Good Poor Good Good Kozac; no Nco Cleavage (common in euk. SD Vectors cDNAs) closest to attL1 pENTR5 Minimal Restr. Enz. Nde I site closest Good Poor Poor at Nde I, No SD; poor Nde Cleavage to attL1 Good at Xmn Kozac at Nde, Vectors I good at Xmn pENTR6 Minimal Restr. Enz. Sph I site closest Good Poor Poor at Sph I, No SD; poor Sph Cleavage to attL1 Good at Xmn Kozac at Sph, Vectors I good at Xmn pENTR7 TEV Blunt TEV Xmn I (blunt) is Good Poor Good at Xmn TEV protease Cleavage first cloning site I site leaves Gly-Thr Site Present after TEV site on amino end of protein; no SD pENTR8 TEV Nco TEV Nco I is first Good Poor Good TEV protease Cleavage cloning site after leaves Gly-Thr Site Present TEV site on amino end of protein; no SD pENTR9 TEV Nde TEV Nde I is first Good Poor Poor TEV protease Cleavage cloning site after leaves Gly-Thr Site Present TEV site on amino end of protein; no SD, poor Kozac pENTR10 Nde with Good SD for Strong SD; Nde I Poor Good Poor Strong SD, SD E. coli site, no TEV internal starts in Expression amino fusions. Poor Kz. No TEV pENTR11 2 X Good SD for Xmn I (blunt) Good Good Good Strong SD/Koz SD + Kozac E. coli and Nco I sites Internal starts in Expression each preceded by amino fusions. SD and Kozac No TEV
[0370] Entry vectors pENTR1A (FIGS. 10A and 10B), pENTR2B (FIGS. 11A and 11B), and pENTR3C (FIGS. 12A and 12B) are almost identical, except that the restriction sites are in different reading frames. Entry vectors pENTR4 (FIGS. 13A and 13B), pENTR5 (FIGS. 14A and 14B), and pENTR6 (FIGS. 15A and 15B) are essentially identical to pENTR1A, except that the blunt DraI site has been replaced with sites containing the ATG methionine codon: NcoI in pENTR4, NdeI in pENTR5, and SphI in pENTR6. Nucleic acid molecules that contain one of these sites at the initiating ATG can be conveniently cloned in these Entry vectors. The NcoI site in pENTR4 is especially useful for expression of nucleic acid molecules in eukaryotic cells, since it contains many of the bases that give efficient translation (see Example 13, below). (Nucleic acid molecules of interest cloned into the NdeI site of pENTR5 are not expected to be highly expressed in eukaryotic cells, because the cytosine at position -3 from the initiating ATG is rare in eukaryotic genes.)
[0371] Entry vectors pENTR7 (FIGS. 16A and 16B), pENTR8 (FIGS. 17A and 17B), and pENTR9 (FIGS. 18A and 18B) contain the recognition site for the TEV protease between the attL1 site and the cloning sites. Cleavage sites for XmnI (blunt), NcoI, and NdeI, respectively, are the most 5' sites in these Entry vectors. Amino fusions can be removed efficiently if nucleic acid molecules are cloned into these Entry vectors. TEV protease is highly active and highly specific.
Example 5
Controlling Reading Frame
[0372] One of the trickiest tasks in expression of cloned nucleic acid molecules is making sure the reading frame is correct. (Reading frame is important if fusions are being made between two ORFs, for example between a nucleic acid molecule of interest and a His6 or GST domain.) For purposes of the present invention, the following convention has been adopted: The reading frame of the DNA cloned into any Entry Vector must be in phase with that of the attB1 site shown in FIG. 16A, pENTR7. Notice that the six As of the attL1 site are split into two lysine codons (aaa aaa). The Destination Vectors that make amino fusions were constructed such that they enter the attR1 site in this reading frame. Destination Vectors for carboxy terminal fusions were also constructed, including those containing His6 (pDEST23; FIG. 43), GST (pDEST24; FIG. 44), or thioredoxin (pDEST25; FIG. 45) C-terminal fusion sequences.
[0373] Therefore, if a nucleic acid molecule of interest is cloned into an Entry Vector so that the aaa aaa reading frame within the attL1 site is in phase with the nucleic acid molecule's ORF, amino terminal fusions will automatically be correctly phased, for all the fusion tags. This is a significant improvement over the usual case, where each different vector can have different restriction sites and different reading frames.
[0374] See Example 15 for a practical example of how to choose the most appropriate combinations of Entry Vector and Destination Vector.
Materials
[0375] Unless otherwise indicated, the following materials were used in the remaining Examples included herein:
5×LR Reaction Buffer:
[0376] 200-250 mM (preferably 250 mM) Tris-HCl, pH 7.5
[0377] 250-350 mM (preferably 320 mM) NaCl
[0378] 1.25-5 mM (preferably 4.75 mM) EDTA
[0379] 12.5-35 mM (preferably 22-35 mM, and most preferably 35 mM)
[0380] Spermidine-HCl
[0381] 1 mg/ml bovine serum albumin
GATEWAY® LR Clonase® Enzyme Mix:
per 4 μl of 1×LR Reaction Buffer:
[0381]
[0382] 150 ng carboxy-His6-tagged Int (see U.S. Appl. No. 60/108,324, filed Nov. 13, 1998, and Ser. No. 09/438,358, filed Nov. 12, 1999, both entirely incorporated by reference herein)
[0383] 25 ng carboxy-His6-tagged Xis (see U.S. Appl. No. 60/108,324, filed Nov. 13, 1998, and Ser. No. 09/438,358, filed Nov. 12, 1999, both entirely incorporated by reference herein)
[0384] 30 ng IHF
[0385] 50% glycerol
5×BP Reaction Buffer:
[0385]
[0386] 125 mM Tris-HCl, pH 7.5
[0387] 110 mM NaCl
[0388] 25 mM EDTA
[0389] 25 mM Spermidine-HCl
[0390] 5 mg/ml bovine serum albumin
GATEWAY® BP Clonase® Enzyme Mix:
[0391] Per 4 μl of 1×BP Reaction Buffer:
[0392] 200 ng carboxy-His6-tagged Int (see U.S. Appl. No. 60/108,324, filed Nov. 13, 1998, and Ser. No. 09/438,358, filed Nov. 12, 1999, both entirely incorporated by reference herein)
[0393] 80 ng IHF
[0394] 50% glycerol
10× Clonase Stop Solution:
[0394]
[0395] 50 mM Tris-HCl, pH 8.0
[0396] 1 mM EDTA
[0397] 2 mg/ml Proteinase K
Example 6
LR ("Destination") Reaction
[0398] To create a new Expression Clone containing the nucleic acid molecule of interest (and which may be introduced into a host cell, ultimately for production of the polypeptide encoded by the nucleic acid molecule), an Entry Clone or Vector containing the nucleic acid molecule of interest, prepared as described herein, is reacted with a Destination Vector. In the present example, a β-Gal gene flanked by attL sites is transferred from an Entry Clone to a Destination Vector.
Materials needed:
[0399] 5×LR Reaction buffer
[0400] Destination Vector (preferably linearized), 75-150 ng/μl
[0401] Entry Clone containing nucleic acid molecule of interest, 100-300 ng in 8 μl TE buffer
[0402] Positive control Entry Clone (pENTR-β-Gal) DNA (See note, below)
[0403] Positive control Destination Vector, pDEST1 (pTrc), 75 ng/μl
[0404] GATEWAY® LR Clonase® Enzyme Mix (stored at -80° C.)
[0405] 10× Clonase Stop solution
[0406] pUC19 DNA, 10 pg/μl
[0407] Chemically competent E. coli cells (competence: ≧1×107 CFU/μg), 400 μl.
[0408] LB Plates containing ampicillin (100 μg/ml) and methicillin (200 μg/ml)±X-gal and IPTG (See below)
Notes:
[0409] Preparation of the Entry Clone DNA: Miniprep DNA that has been treated with RNase works well. A reasonably accurate quantitation (±50%) of the DNA to be cloned is advised, as the GATEWAY® reaction appears to have an optimum of about 100-300 ng of Entry Clone per 20 μl of reaction mix.
[0410] The positive control Entry Clone, pENTR-β-Gal, permits functional analysis of clones based on the numbers of expected blue vs. white colonies on LB plates containing IPTG+Bluo-gal (or X-gal), in addition to ampicillin (100 μg/ml) and methicillin (200 μg/ml). Because β-Galactosidase is a large protein, it often yields a less prominent band than many smaller proteins do on SDS protein gels.
[0411] In the Positive Control Entry Vector pENTR-β-Gal, the coding sequence of β-Gal has been cloned into pENTR11 (FIGS. 20A and 20B), with translational start signals permitting expression in E. coli, as well as in eukaryotic cells. The positive control Destination Vector, for example pDEST1 (FIG. 21), is preferably linearized.
[0412] To prepare X-gal+IPTG plates, either of the following protocols may be used:
[0413] A. With a glass rod, spread over the surface of an LB agar plate: 40 μl of 20 mg/ml X-gal (or Bluo-gal) in DMF plus 4 μl 200 mg/ml IPTG. Allow liquid to adsorb into agar for 3-4 hours at 37° C. before plating cells.
[0414] B. To liquid LB agar at ˜45° C., add: X-gal (or Bluo-Gal) (20 mg/ml in DMF) to make 50 μg/ml and IPTG (200 mM in water) to make 0.5-1 mM, just prior to pouring plates. Store X-gal and Bluo-Gal in a light-shielded container.
[0415] Colony color may be enhanced by placing the plates at 5° C. for a few hours after the overnight incubation at 37° C. Protocol B can give more consistent colony color than A, but A is more convenient when selection plates are needed on short notice.
[0416] Recombination in Clonase reactions continues for many hours. While incubations of 45-60 minutes are usually sufficient, reactions with large DNAs, or in which both parental DNAs are supercoiled, or which will be transformed into cells of low competence, can be improved with longer incubation times, such as 2-24 hours at 25° C.
Procedure:
[0417] 1. Assemble reactions as follows (combine all components at room temperature, except GATEWAY® LR Clonase® Enzyme Mix ("Clonase LR"), before removing Clonase LR from frozen storage):
TABLE-US-00004 Tube 1 Tube 2 Tube 3 Tube 4 Component Neg. Pos. Neg. Test p-Gate-βGal, (Posi- 4 μl 4 μl tive control Entry Clone) 75 ng/μl pDEST1 (Positive con- 4 μl 4 μl trol Destination Vector), 75 ng/μl Your Entry Clone 1-8 μl.sup. 1-8 μl.sup. (100-300 ng) Destination Vector for 4 μl 4 μl your nucleic acid molecule, 75 ng/μl 5 X LR Reaction Buffer 4 μl 4 μl 4 μl 4 μl TE 8 μl 4 μl To 20 μl .sup. To 16 μl .sup. GATEWAY ® LR -- 4 μl -- 4 μl Clonase ® Enzyme Mix (store at -80° C., add last) Total Volume 20 μl 20 μl 20 μl 20 μl
2. Remove the GATEWAY® LR Clonase® Enzyme Mix from the -80° C. freezer, place immediately on ice. The Clonase takes only a few minutes to thaw. 3. Add 4 μl of GATEWAY® LR Clonase® Enzyme Mix to reactions #2 and #4; 4. Return GATEWAY® LR Clonase® Enzyme Mix to -80° C. freezer. 5. Incubate tubes at 25° for at least 60 minutes. 6. Add 2 μl Clonase Stop solution to all reactions. Incubate for 20 min at 37° C. (This step usually increases the total number of colonies obtained by 10-20 fold.) 7. Transform 2 μl into 100 μl competent E. coli. Select on plates containing ampicillin at 100 μg/ml.
Example 7
Transformation of E. coli
[0418] To introduce cloning or Expression Vectors prepared using the recombinational cloning system of the invention, any standard E. coli transformation protocol should be satisfactory. The following steps are recommended for best results:
[0419] 1. Let the mixture of competent cells and Recombinational Cloning System reaction product stand on ice at least 15 minutes prior to the heat-shock step. This gives time for the recombination proteins to dissociate from the DNA, and improves the transformation efficiency.
[0420] 2. Expect the reaction to be about 1%-5% efficient, i.e., 2 μl of the reaction should contain at least 100 pg of the Expression Clone plasmid (taking into account the amounts of each parental plasmid in the reaction, and the subsequent dilution). If the E. coli cells have a competence of 107 CFU/μg, 100 pg of the desired clone plasmid will give about 1000 colonies, or more, if the entire transformation is spread on one ampicillin plate.
[0421] 3. Always do a control pUC DNA transformation. If the number of colonies is not what you expect, the pUC DNA transformation gives you an indication of where the problem was.
Example 8
Preparation of attB-PCR Product
[0422] For preparation of attB-PCR products in the PCR cloning methods described in Example 9 below, PCR primers containing attB1 and attB2 sequences are used. The attB1 and attB2 primer sequences are as follows:
TABLE-US-00005 attB1: (SEQ ID NO: 31) 5'-GGGGACAAGTTTGTACAAAAAAGCAGGCT-(template- specific sequence)-3' attB2: (SEQ ID NO: 32) 5'-GGGGACCACTTTGTACAAGAAAGCTGGGT-(template- specific sequence)-3'
The attB1 sequence should be added to the amino primer, and the attB2 sequence to the carboxy primer. The 4 guanines at the 5' ends of each of these primers enhance the efficiency of the minimal 25 bp attB sequences as substrates for use in the cloning methods of the invention.
[0423] Standard PCR conditions may be used to prepare the PCR product. The following suggested protocol employs PLATINUM Taq DNA Polymerase High Fidelity®, available commercially from Life Technologies, Inc. (Rockville, Md.). This enzyme mix eliminates the need for hot starts, has improved fidelity over Taq, and permits synthesis of a wide range of amplicon sizes, from 200 bp to 10 kb, or more, even on genomic templates.
Materials needed:
[0424] PLATINUM Taq DNA Polymerase High Fidelity® (Life Technologies, Inc.)
[0425] attB1- and attB2-containing primer pair (see above) specific for your template
[0426] DNA template (linearized plasmid or genomic DNA)
[0427] 10× High Fidelity PCR Buffer
[0428] 10 mM dNTP mix
[0429] PEG/MgCl2 Mix (30% PEG 8000, 30 mM MgCl2)
Procedure:
[0430] 1.) Assemble the reaction as follows:
TABLE-US-00006 Reaction with Reaction with Component Plasmid Target Genomic Target 10X High Fidelity PCR Buffer 5 μl 5 μl dNTP Mix 10 mM 1 μl 1 μl MgSO4, 50 mM 2 μl 2 μl attB1 Primer, 10 μM 2 μl 1 μl attB2 Primer, 10 μM 2 μl 1 μl Template DNA 1-5 ng* ≧100 ng PLATINUM Taq High Fidelity 2 μl 1 μl Water to 50 μl to 50 μl *Use of higher amounts of plasmid template may permit fewer cycles (10-15) of PCR
2.) Add 2 drops mineral oil, as appropriate.
3.) Denature for 30 sec. at 94° C.
[0431] 4.) Perform 25 cycles:
[0432] 94° C. for 15 sec-30 sec
[0433] 55° C. for 15 sec-30 sec
[0434] 68° C. for 1 min per kb of template.
5.) Following the PCR reaction, apply 1-2 μl of the reaction mixture to an agarose gel, together with size standards (e.g., 1 Kb Plus Ladder, Life Technologies, Inc.) and quantitation standards (e.g., Low Mass Ladder, Life Technologies, Inc.), to assess the yield and uniformity of the product.
[0435] Purification of the PCR product is recommended, to remove attB primer dimers which can clone efficiently into the Entry Vector. The following protocol is fast and will remove DNA <300 bp in size:
6.) Dilute the 50 μl PCR reaction to 200 μl with TE. 7.) Add 100 μl PEG/MgCl2 Solution. Mix and centrifuge immediately at 13,000 RPM for 10 min at room temperature. Remove the supernatant (pellet is clear and hard to see). 8.) Dissolve the pellet in 50 μl TE and check recovery on a gel.
[0436] If the starting PCR template is a plasmid that contains the gene for Kanr, it is advisable to treat the completed PCR reaction with the restriction enzyme DpnI, to degrade the plasmid since unreacted residual starting plasmid is a potential source of false-positive colonies from the transformation of the GATEWAY® Cloning System reaction. Adding ˜5 units of DpnI to the completed PCR reaction and incubating for 15 min at 37° C. will eliminate this potential problem. Heat inactivate the DpnI at 65° C. for 15 min, prior to using the PCR product in the GATEWAY® Cloning System reaction.
Example 9
Cloning attB-PCR Products into Entry Vectors Via the BP ("Gateward") Reaction
[0437] The addition of 5'-terminal attB sequences to PCR primers allows synthesis of a PCR product that is an efficient substrate for recombination with a Donor (attP) Plasmid in the presence of GATEWAY® BP Clonase® Enzyme Mix. This reaction produces an Entry Clone of the PCR product (See FIG. 8).
[0438] The conditions of the Gateward Cloning reaction with an attB PCR substrate are similar to those of the BP Reaction (see Example 10 below), except that the attB-PCR product (see Example 8) substitutes for the Expression Clone, and the attB-PCR positive control (attB-tetr) substitutes for the Expression Clone Positive Control (GFP).
Materials needed:
[0439] 5×BP Reaction Buffer
[0440] Desired attB-PCR product DNA, 50-100 ng in ≦8 μl TE.
[0441] Donor (attP) Plasmid (FIGS. 49-54), 75 ng/μl supercoiled DNA
[0442] attB-tetr PCR product positive control, 25 ng/μl
[0443] GATEWAY® BP Clonase® Enzyme Mix (stored at -80° C.)
[0444] 10× Clonase Stop Solution
[0445] pUC19 DNA, 10 pg/μl.
[0446] Chemically competent E. coli cells (competence: ≧1×107 CFU/μg), 400 μl
Notes:
[0446]
[0447] Preparation of attB-PCR DNA: see Example 8.
[0448] The Positive Control attB-tetrPCR product contains a functional copy of the tee gene of pBR322, with its own promoter. By plating the transformation of the control BP Reaction on kanamycin (50 μg/ml) plates (if kanr Donor Plasmids are used; see FIGS. 49-52) or an alternative selection agent (e.g., gentamycin, if genr Donor Plasmids are used; see FIG. 54), and then picking about 50 of these colonies onto plates with tetracycline (20 μg/ml), the percentage of Entry Clones containing functional tetr among the colonies from the positive control reaction can be determined (% Expression Clones=(number of tetr+kanr (or genr) colonies/kanr (or genr) colonies).
Procedure:
[0449] 1. Assemble reactions as follows. Combine all components except GATEWAY® BP Clonase® Enzyme Mix, before removing GATEWAY® BP Clonase® Enzyme Mix from frozen storage.
TABLE-US-00007 Neg. Pos. Test Component Tube 1 Tube 2 Tube 3 attB-PCR product, 50-100 ng 1-8 μl.sup. Donor (attP) Plasmid 75 ng/μl 2 μl 2 μl 2 μl attB-PCR tetr control DNA (75 4 μl ng/μl) 5 X BP Reaction Buffer 4 μl 4 μl 4 μl TE 10 μl 6 μl To 16 μl .sup. GATEWAY ® BP Clonase ® 4 μl 4 μl 4 μl Enzyme Mix (store at -80° C., add last) Total Volume 20 μl 20 μl 20 μl
2. Remove the GATEWAY® BP Clonase® Enzyme Mix from the -80° C. freezer, place immediately on ice. The Clonase takes only a few minutes to thaw. 3. Add 4 μl of GATEWAY® BP Clonase® Enzyme Mix to the subcloning reaction, mix. 4. Return GATEWAY® BP Clonase® Enzyme Mix to -80° C. freezer. 5. Incubate tubes at 25° for at least 60 minutes. 6. Add 2 μl Proteinase K (2 μg/μl) to all reactions. Incubate for 20 min at 37° C. 7. Transform 2 μl into 100 μl competent E. coli, as per 3.2, above. Select on LB plates containing kanamycin, 50 μg/ml.
Results:
[0450] In initial experiments, primers for amplifying tetR and ampR from pBR322 were constructed containing only the tetR- or ampR-specific targeting sequences, the targeting sequences plus attB1 (for forward primers) or attB2 (for reverse primers) sequences shown in FIG. 9, or the attB1 or attB2 sequences with a 5' tail of four guanines. The construction of these primers is depicted in FIG. 65. After PCR amplification of tetR and ampR from pBR322 using these primers and cloning the PCR products into host cells using the recombinational cloning system of the invention, the results shown in FIG. 66 were obtained. These results demonstrated that primers containing attB sequences provided for a somewhat higher number of colonies on the tetracycline and ampicillin plates. However, inclusion of the 5' extensions of four or five guanines on the primers in addition to the attB sequences provided significantly better cloning results, as shown in FIGS. 66 and 67. These results indicate that the optimal primers for cloning of PCR products using recombinational cloning will contain the recombination site sequences with a 5' extension of four or five guanine bases.
[0451] To determine the optimal stoichiometry between attB-containing PCR products and attP-containing Donor plasmid, experiments were conducted where the amount of PCR product and Donor plasmid were varied during the BP Reaction. Reaction mixtures were then transformed into host cells and plated on tetracycline plates as above. Results are shown in FIG. 68. These results indicate that, for optimal recombinational cloning results with a PCR product in the size range of the tet gene, the amounts of attP-containing Donor plasmids are between about 100-500 ng (most preferably about 200-300 ng), while the optimal concentrations of attB-containing PCR products is about 25-100 ng (most preferably about 100 ng), per 20 μl reaction.
[0452] Experiments were then conducted to examine the effect of PCR product size on efficiency of cloning via the recombinational cloning approach of the invention. PCR products containing attB1 and attB2 sites, at sizes 256 bp, 1 kb, 1.4 kb, 3.4 kb, 4.6 kb, 6.9 kb and 10.1 kb were prepared and cloned into Entry vectors as described above, and host cells were transformed with the Entry vectors containing the cloned PCR products. For each PCR product, cloning efficiency was calculated relative to cloning of pUC 19 positive control plasmids as follows:
Cloning Efficiency = CFU / ng attB PCR product CFU / ng pUC19 control × Size ( kb ) PCR product Size ( kb ) pUC19 control ##EQU00001##
[0453] The results of these experiments are depicted in FIGS. 69A-69C (for 256 bp PCR fragments), 70A-70C (for 1 kb PCR fragments), 71A-71C (for 1.4 kb PCR fragments), 72A-72C (for 3.4 kb PCR fragments), 73A-73C (for 4.6 kb PCR fragments), 74 (for 6.9 kb PCR fragments), and 75-76 (for 10.1 kb PCR fragments). The results shown in these figures are summarized in FIG. 77, for different weights and moles of input PCR DNA.
[0454] Together, these results demonstrate that attB-containing PCR products ranging in size from about 0.25 kb to about 5 kb clone relatively efficiently in the recombinational cloning system of the invention. While PCR products larger than about 5 kb clone less efficiently (apparently due to slow resolution of cointegrates), longer incubation times during the recombination reaction appears to improve the efficiency of cloning of these larger PCR fragments. Alternatively, it may also be possible to improve efficiency of cloning of large (>about 5 kb) PCR fragments by using lower levels of input attP Donor plasmid and perhaps attB-containing PCR product, and/or by adjusting reaction conditions (e.g., buffer conditions) to favor more rapid resolution of the cointegrates.
Example 0.10
The BP Reaction
[0455] One purpose of the Gateward ("Entry") reaction is to convert an Expression Clone into an Entry Clone. This is useful when you have isolated an individual Expression Clone from an Expression Clone cDNA library, and you wish to transfer the nucleic acid molecule of interest into another Expression Vector, or to move a population of molecules from an attB or attL library. Alternatively, you may have mutated an Expression Clone and now wish to transfer the mutated nucleic acid molecule of interest into one or more new Expression Vectors. In both cases, it is necessary first to convert the nucleic acid molecule of interest to an Entry Clone.
Materials needed:
[0456] 5×BP Reaction Buffer
[0457] Expression Clone DNA, 100-300 ng in ≦8 μl TE.
[0458] Donor (attP) Vector, 75 ng/μl, supercoiled DNA
[0459] Positive control attB-tet-PCR DNA, 25 ng/μl
[0460] GATEWAY® BP Clonase® Enzyme Mix (stored at -80° C.)
[0461] Clonase Stop Solution (Proteinase K, 2 μg/μl).
Notes:
[0462] Preparation of the Expression. Clone DNA: Miniprep DNA treated with RNase works well.
[0463] 1. As with the LR Reaction (see Example 14), the BP Reaction is strongly influenced by the topology of the reacting DNAs. In general, the reaction is most efficient when one of the DNAs is linear and the other is supercoiled, compared to reactions where the DNAs are both linear or both supercoiled. Further, linearizing the attB Expression Clone (anywhere within the vector) will usually give more colonies than linearizing the Donor (attP) Plasmid. If finding a suitable cleavage site within your Expression Clone vector proves difficult, you may linearize the Donor (attP) Plasmid between the attP1 and attP2 sites (for example, at the NcoI site), avoiding the ccdB gene. Maps of Donor (attP) Plasmids are given in FIGS. 49-54.
Procedure:
[0464] 1. Assemble reactions as follows. Combine all components at room temperature, except GATEWAY® BP Clonase® Enzyme Mix, before removing GATEWAY® BP Clonase® Enzyme Mix from freezer.
TABLE-US-00008 Neg. Pos. Test Component Tube 1 Tube 2 Tube 3 Positive Control, attB-tet-PCR DNA, 4 μl 4 μl 25 ng/μl Desired attB Expression Clone DNA 1-8 μl.sup. (100 ng) linearized Donor (attP) Plasmid, 75 ng/μl 2 μl 2 μl 2 μl 5 X BP Reaction Buffer 4 μl 4 μl 4 μl TE 10 μl 6 μl To 16 μl .sup. GATEWAY ® BP Clonase ® Enzyme -- 4 μl 4 μl Mix (store at -80° C., add last) Total Volume 20 μl 20 μl 20 μl
2. Remove the GATEWAY® BP Clonase® Enzyme Mix from the -80° C. freezer, place immediately on ice. The mixture takes only a few minutes to thaw. 3. Add 4 μl of GATEWAY® BP Clonase® Enzyme Mix to the subcloning reaction, mix. 4. Return GATEWAY® BP Clonase® Enzyme Mix to -80° C. freezer. 5. Incubate tubes at 25° for at least 60 minutes. If both the attB and attP DNAs are supercoiled, incubation for 2-24 hours at 25° C. is recommended. 6. Add 2 μl Clonase Stop Solution. Incubate for 10 min at 37° C. 7. Transform 2 μl into 100 μl competent E. coli, as above. Select on LB plates containing 50 μg/ml kanamycin.
Example 11
Cloning PCR Products into Entry Vectors Using Standard Cloning Methods
Preparation of Entry Vectors for Cloning of PCR Products
[0465] All of the Entry Vectors of the invention contain the death gene ccdB as a stuffer between the "left" and "right" restriction sites. The advantage of this arrangement is that there is virtually no background from vector that has not been cut with both restriction enzymes, because the presence of the ccdB gene will kill all standard E. coli strains. Thus it is necessary to cut each Entry Vector twice, to remove the ccdB fragment.
[0466] We strongly recommend that, after digestion of the Entry Vector with the second restriction enzyme, you treat the reaction with phosphatase (calf intestine alkaline phosphatase, CIAP or thermosensitive alkaline phosphatase, TSAP). The phosphatase can be added directly to the reaction mixture, incubated for an additional time, and inactivated. This step dephosphorylates both the vector and ccdB fragments, so that during subsequent ligation there is less competition between the ccdB fragment and the DNA of interest for the termini of the Entry Vector.
Blunt Cloning of PCR products
[0467] Generally PCR products do not have 5' phosphates (because the primers are usually 5' OH), and they are not necessarily blunt. (On this latter point, see Brownstein, et al., BioTechniques 20: 1006, 1996 for a discussion of how the sequence of the primers affects the addition of single 3' bases.) The following protocol repairs these two defects.
[0468] In a 0.5 ml tube, ethanol precipitate about 40 ng of PCR product (as judged from an agarose gel).
[0469] 1. Dissolve the precipitated DNA in 10 μl comprising 1 μl 10 mM rATP, 1 μl mixed 2 mM dNTPs (i.e., 2 mM each dATP, dCTP, dTTP, and dGTP), 2 μl 5×T4 polynucleotide kinase buffer (350 mM Tris HCl (pH7.6), 50 mM MgCl2, 500 mM KCl, 5 mM 2-mercaptoethanol) 10 units T4 polynucleotide kinase, 1 μl T4 DNA polymerase, and water to 10 μl.
[0470] 2. Incubate the tube at 37° for 10 minutes, then at 65° for 15 minutes, cool, centrifuge briefly to bring any condensate to the tip of the tube.
[0471] 3. Add 5 μl of the PEG/MgCl2 solution, mix and centrifuge at room temperature for 10 minutes. Discard supernatant.
[0472] 4. Dissolve the invisible precipitate in 10 μl containing 2 μl 5×T4 DNA ligase buffer (Life Technologies, Inc.), 0.5 units T4 DNA ligase, and about 50 ng of blunt, phosphatase-treated Entry Vector. 5. Incubate at 25° for 1 hour, then 65° for 10 minutes. Add 90 μl TE, transform 10 μl into 50-100 μl competent E. coli cells. 6. Plate on kanamycin.
Note:
[0473] In the above protocol, steps b-c simultaneously polish the ends of the PCR product (through the exonuclease and polymerase activities of T4 DNA polymerase) and phosphorylate the 5' ends (using T4 polynucleotide kinase). It is necessary to inactivate the kinase, so that the blunt, dephosphorylated vector in step e cannot self ligate. Step d (the PEG precipitation) removes all small molecules (primers, nucleotides), and has also been found to improve the yield of cloned PCR product by 50 fold.
Cloning PCR Products after Digestion with Restriction Enzymes
[0474] Efficient cloning of PCR products that have been digested with restriction enzymes includes three steps: inactivation of Taq DNA polymerase, efficient restriction enzyme cutting, and removal of small DNA fragments.
[0475] Inactivation of Taq DNA Polymerase:
[0476] Carryover of Taq DNA polymerase and dNTPs into a RE digestion significantly reduces the success in cloning a PCR product (D. Fox et al., FOCUS 20(1):15, 1998), because Taq DNA polymerase can fill in sticky ends and add bases to blunt ends. Either TAQQUENCH® (obtainable from Life Technologies, Inc.; Rockville, Md.) or extraction with phenol can be used to inactivate the Taq.
[0477] Efficient Restriction Enzyme Cutting:
[0478] Extra bases on the 5' end of each PCR primer help the RE cut near ends of PCR products. With the availability of cheap primers, adding 6 to 9 bases on the 5' sides of the restriction sites is a good investment to ensure that most of the ends are digested. Incubation of the DNA with a 5-fold excess of restriction enzyme for an hour or more helps ensure success.
[0479] Removal of Small Molecules Before Ligation:
[0480] Primers, nucleotides, primer dimers, and small fragments produced by the restriction enzyme digestion, can all inhibit or compete with the desired ligation of the PCR product to the cloning vector. This protocol uses PEG precipitation to remove small molecules.
Protocol for Cutting the Ends of PCR Products with Restriction Enzyme(s):
1. Inactivation of Taq DNA Polymerase in the PCR Product:
[0481] Option A: Extraction with Phenol
[0482] A1. Dilute the PCR reaction to 200 μl with TB. Add an equal volume of phenol:chloroform:isoamyl alcohol, vortex vigorously for 20 seconds, and centrifuge for 1 minute at room temperature. Discard the lower phase.
[0483] A2. Extract the phenol from the DNA and concentrate as follows. Add an equal volume of 2-butanol (colored red with "Oil Red O" from Aldrich, if desired), vortex briefly, centrifuge briefly at room temperature. Discard the upper butanol phase. Repeat the extraction with 2-butanol. This time the volume of the lower aqueous phase should decrease significantly. Discard the upper 2-butanol phase.
[0484] A3. Ethanol precipitate the DNA from the aqueous phase of the above extractions. Dissolve in a 200 μl of a suitable restriction enzyme (RE) buffer. Option B: Inactivation with TaqQuench
[0485] B1. Ethanol precipitate an appropriate amount of PCR product (100 ng to 1 μg), dissolve in 200 μl of a suitable RE buffer.
[0486] B2. Add 2 μl TaqQuench. 2. Add 10 to 50 units of restriction enzyme and incubate for at least 1 hour. Ethanol precipitate if necessary to change buffers for digestion at the other end of the PCR product. 3. Add 1/2 volume of the PEG/MgCl2 mix to the RE digestion. Mix well and immediately centrifuge at room temperature for 10 minutes. Discard the supernatant (pellet is usually invisible), centrifuge again for a few seconds, discard any remaining supernatant. 4. Dissolve the DNA in a suitable volume of TE (depending on the amount of PCR product in the original amplification reaction) and apply an aliquot to an agarose gel to confirm recovery. Apply to the same gel 20-100 ng of the appropriate Entry Vector that will be used for the cloning.
Example 12
Determining the Expected Size of the GATEWAY® Cloning Reaction Products
[0487] If you have access to a software program that will electronically cut and splice sequences, you can create electronic clones to aid you in predicting the sizes and restriction patterns of GATEWAY® Cloning System recombination products.
[0488] The cleavage and ligation steps performed by the enzyme Int in the GATEWAY® Cloning System recombination reactions mimic a restriction enzyme cleavage that creates a 7-bp 5'-end overhang followed by a ligation step that reseals the ends of the daughter molecules. The recombination proteins present in the Clonase cocktails (see Example 19 below) recognize the 15 bp core sequence present within all four types of att sites (in addition to other flanking sequences characteristic of each of the different types of att sites).
[0489] By treating these sites in your software program as if they were restriction sites, you can cut and splice your Entry Clones with various Destination Vectors and obtain accurate maps and sequences of the expected results from your GATEWAY® Cloning System reactions.
Example 13
Protein Expression
Brief Review of Protein Expression
[0490] Transcription:
[0491] The most commonly used promoters in E. coli Expression Vectors are variants of the lac promoter, and these can be turned on by adding IPTG to the growth medium. It is usually good to keep promoters off until expression is desired, so that the host cells are not made sick by the overabundance of some heterologous protein. This is reasonably easy in the case of the lac promoters used in E. coli. One needs to supply the lac I gene (or its more productive relative, the lac Iq gene) to make lac repressor protein, which binds near the promoter and keeps transcription levels low. Some Destination Vectors for E. coli expression carry their own lac Iq gene for this purpose. (However, lac promoters are always a little "on," even in the absence of IPTG.)
[0492] Controlling transcription in eukaryotic cells is not nearly so straightforward or efficient. The tetracycline system of Bujard and colleagues is the most successful approach, and one of the Destination Vectors (pDEST11; FIG. 31) has been constructed to supply this function.
[0493] Translation:
[0494] Ribosomes convert the information present in mRNA into protein. Ribosomes scan RNA molecules looking for methionine (AUG) codons, which begin nearly all nascent proteins. Ribosomes must, however, be able to distinguish between AUG codons that code for methionine in the middle of proteins from those at the start. Most often ribosomes choose AUGs that are 1) first in the RNA (toward the 5' end), and 2) have the proper sequence context. In E. coli the favored context (first recognized by Shine and Dalgarno, Eur. J. Biochem. 57: 221 (1975)) is a run of purines (As and Gs) from five to 12 bases upstream of the initiating AUG, especially AGGAGG or some variant.
[0495] In eukaryotes, a survey of translated mRNAs by Kozak (J. Biol. Chem. 266: 19867 (1991)) has revealed a preferred sequence context, gcc Acc ATGG, around the initiating methionine, with the A at -3 being most important, and a purine at +4 (where the A of the ATG is +1), preferably a G, being next most influential. Having an A at -3 is enough to make most ribosomes choose the first AUG of an mRNA, in plants, insects, yeast, and mammals. (For a review of initiation of protein synthesis in eukaryotic cells, see: Pain, V. M. Eur. J. Biochem. 236:747-771, 1996.)
[0496] Consequences of Translation Signals for GATEWAY® Cloning System:
[0497] First, translation signals (Shine-Dalgarno in E. coli, Kozak in eukaryotes) have to be close to the initiating ATG. The attB site is 25 base pairs long. Thus if translation signals are desired near the natural ATG of the nucleic acid molecule of interest, they must be present in the Entry Clone of that nucleic acid molecule of interest. Also, when a nucleic acid molecule of interest is moved from an Entry Clone to a Destination vector, any translation signals will move along. The result is that the presence or absence of Shine-Dalgarno and/or Kozak sequences in the Entry Clone must be considered, with the eventual Destination Vectors to be used in mind.
[0498] Second, although ribosomes choose the 5' ATG most often, internal ATGs are also used to begin protein synthesis. The better the translation context around this internal ATG, the more internal translation initiation will be seen. This is important in the GATEWAY® Cloning System, because you can make an Entry Clone of your nucleic acid molecule of interest, and arrange to have Shine-Dalgarno and/or Kozak sequences near the ATG. When this cassette is recombined into a Destination Vector that transcribes your nucleic acid molecule of interest, you get native protein. If you want, you can make a fusion protein in a different Destination Vector, since the Shine-Dalgarno and/or Kozak sequences do not contain any stop signals in the same reading frame. However, the presence of these internal translation signals may result in a significant amount of native protein being made, contaminating, and lowering the yield of, your fusion protein. This is especially likely with short fusion tags, like His6.
[0499] A good compromise can be recommended. If an Entry Vector like pENTR7 (FIG. 16) or pENTR8 (FIG. 17) is chosen, the Kozak bases are present for native eukaryotic expression. The context for E. coli translation is poor, so the yield of an amino-terminal fusion should be good, and the fusion protein can be digested with the TEV protease to make near-native protein following purification.
[0500] Recommended Conditions for Synthesis of Proteins in E. coli:
[0501] When making proteins in E. coli it is advisable, at least initially, to incubate your cultures at 30° C., instead of at 37° C. Our experience indicates that proteins are less likely to form aggregates at 30° C. In addition, the yields of proteins from cells grown at 30° C. frequently are improved.
[0502] The yields of proteins that are difficult to express may also be improved by inducing the cultures in mid-log phase of growth, using cultures begun in the morning from overnight growths, as opposed to harvesting directly from an overnight culture. In the latter case, the cells are preferably in late log or stationary growth, which can favor the formation of insoluble aggregates.
Example 14
Constructing Destination Vectors from Existing Vectors
[0503] Destination Vectors function because they have two recombination sites, attR1 and attR2, flanking a chloramphenicol resistance (CmR) gene and a death gene, ccdB. The GATEWAY® Cloning System recombination reactions exchange the entire Cassette (except for a few bases comprising part of the attB sites) for the DNA segment of interest from the Entry Vector. Because attR1, CmR, ccdB gene, and attR2 are contiguous, they can be moved on a single DNA segment. If this Cassette is cloned into a plasmid, the plasmid becomes a Destination Vector. FIG. 63 shows a schematic of the GATEWAY® Cloning System Cassette; attR cassettes in all three reading frames contained in vectors pEZC15101, pEZC15102 and pEZC15103 are shown in FIGS. 64A, 64B, and 64C, respectively.
[0504] The protocol for constructing a Destination Vector is presented below. Keep in mind the following points:
[0505] Destination Vectors must be constructed and propagated in one of the DB strains of E. coli (e.g., DB3.1, and particularly E. coli LIBRARY EFFICIENCY® DB3.1® Competent Cells) available from Life Technologies, Inc. (and described in detail in U.S. Provisional Application No. 60/122,392, filed on Mar. 2, 1999, which is incorporated herein by reference), because the ccdB death gene will kill any E. coli strain that has not been mutated such that it will survive the presence of the ccdB gene.
[0506] If your Destination Vector will be used to make a fusion protein, a GATEWAY® Cloning System cassette with the correct reading frame must be used. The nucleotide sequences of the ends of the cassettes are shown in FIG. 78. The reading frame of the fusion protein domain must be in frame with the core region of the attR1 site (for an amino terminal fusion) so that the six As are translated into two lysine codons. For a C-terminal fusion protein, translation through the core region of the attR2 site should be in frame with -TAC-AAA-, to yield -Tyr-Lys-.
[0507] Note that each reading frame Cassette has a different unique restriction site between the chloramphenicol resistance and ccdB genes (MluI for reading frame A, BglII for reading frame B, and XbaI for reading frame C; see FIG. 63).
[0508] Most standard vectors can be converted to Destination Vectors, by inserting the Entry Cassette into the MCS of that vector.
Protocol for Making a Destination Vector
[0509] 1. If the vector will make an amino fusion protein, it is necessary to keep the "aaa aaa" triplets in attR1 in phase with the triplets of the fusion protein. Determine which Entry cassette to use as follows:
[0510] a.) Write out the nucleotide sequence of the existing vector near the restriction site into which the Entry cassette will be cloned. These must be written in triplets corresponding to the amino acid sequence of the fusion domain.
[0511] b.) Draw a vertical line through the sequence that corresponds to the restriction site end, after it has been cut and made blunt, i.e., after filling in a protruding 5' end or polishing a protruding 3' end.
[0512] c.) Choose the appropriate reading frame cassette:
[0513] If the coding sequence of the blunt end ends after a complete codon triplet, use the reading frame A cassette. See FIGS. 78, 79 and 80.
[0514] If the coding sequence of the blunt end ends in a single base, use the reading frame B cassette. See FIGS. 78, 79 and 81.
[0515] If the coding sequence of the blunt end ends in two bases, use the reading frame C cassette. See FIGS. 78, 79, 82A-B, and 83A-C. 2. Cut one to five micrograms of the existing plasmid at the position where you wish your nucleic acid molecule of interest (flanked by att sites) to be after the recombination reactions. Note: it is better to remove as many of the MCS restriction sites as possible at this step. This makes it more likely that restriction enzyme sites within the GATEWAY® Cloning System Cassette will be unique in the new plasmid, which is important for linearizing the Destination Vector (Example 14, below). 3. Remove the 5' phosphates with alkaline phosphatase. While this is not mandatory, it increases the probability of success. 4. Make the end(s) blunt with fill-in or polishing reactions. For example, to 1 μg of restriction enzyme-cut, ethanol-precipitated vector DNA, add:
[0516] i. 20 μl 5×T4 DNA Polymerase Buffer (165 mM Tris-acetate (pH 7.9), 330 mM Na acetate, 50 mM Mg acetate, 500 μg/ml BSA, 2.5 mM DTT)
[0517] ii. 5 μl 10 mM dNTP mix
[0518] iii. 1 Unit of T4 DNA Polymerase
[0519] iv. Water to a final volume of 100
[0520] v. Incubate for 15 min at 37° C. 5. Remove dNTPs and small DNA fragments: Ethanol precipitate (add three volumes of room temperature ethanol containing 0.1 M sodium acetate, mix well, immediately centrifuge at room temperature 5-10 minutes), dissolve wet precipitate in 200 μl TE, add 100 μl 30% PEG 8000, 30 mM MgCl2, mix well, immediately centrifuge for 10 minutes at room temperature, discard supernatant, centrifuge again a few seconds, discard any residual liquid. 6. Dissolve the DNA to a final concentration of 10-50 ng per microliter. Apply 20-100 ng to a gel next to supercoiled plasmid and linear size standards to confirm cutting and recovery. The cutting does not have to be 100% complete, since you will be selecting for the chloramphenicol marker on the Entry cassette. 7. In a 10 μl ligation reaction combine 10-50 ng vector, 10-20 ng of Entry Cassette (FIG. 79), and 0.5 units T4 DNA ligase in ligase buffer. After one hour (or overnight, whichever is most convenient), transform 1 μl into one of the DB strains of competent E. coli cells with a gyrA462 mutation (See U.S. Provisional Application No. 60/122,392, filed on Mar. 2, 1999, which is incorporated herein by reference), preferably DB3.1, and most preferably E. coli LIBRARY EFFICIENCY® DB3.1® Competent Cells. The ccdB gene on the Entry Cassette will kill other strains of E. coli that have not been mutated so as to survive the presence of the ccdB gene. 8. After expression in SOC medium, plate 10 μl and 100 μl on chloramphenicol-containing (30 μg/ml) plates, incubate at 37° C. 9. Pick colonies, make miniprep DNA. Treat the miniprep with RNase A and store in TE. Cut with the appropriate restriction enzyme to determine the orientation of the Cassette. Choose clones with the attR1 site next to the amino end of the protein expression function of the plasmid.
Notes on Using Destination Vectors
[0520]
[0521] We have found that about ten-fold more colonies result from a GATEWAY® Cloning System reaction if the Destination Vector is linear or relaxed. If the competent cells you use are highly competent (>108 per microgram), linearizing the Destination Vector is less essential.
[0522] The site or sites used for the linearization must be within the Entry Cassette. Sites that cut once or twice within each cassette are shown in FIGS. 80-82.
[0523] Minipreps of Destination Vectors will work fine, so long as they have been treated with RNase. Since most DB strains are endA-(See U.S. Provisional Application No. 60/122,392, filed on Mar. 2, 1999, which is incorporated herein by reference), minipreps can be digested with restriction enzymes without a prior phenol extraction.
[0524] Reading the OD260 of miniprep DNA is inaccurate unless the RNA and ribonucleotides have been removed, for example, by a PEG precipitation.
Example 15
Some Options in Choosing Appropriate Entry Vectors and Destination Vectors: An Example
[0525] In some applications, it may be desirable to express a nucleic acid molecule of interest in two forms: as an amino-terminal fusion in E. coli, and as a native protein in eukaryotic cells. This may be accomplished in any of several ways:
[0526] Option 1:
[0527] Your choices depend on your nucleic acid molecule of interest and the fragment that contains it, as well as the available Entry Vectors. For eukaryotic translation, you need consensus bases according to Kozak (J. Biol. Chem. 266:19867, 1991) near the initiating methionine (ATG) codon. All of the Entry Vectors offer this motif upstream of the XmnI site (blunt cutter). One option is to amplify your nucleic acid molecule of interest, with its ATG, by PCR, making the amino end blunt and the carboxy end containing the natural stop codon followed by one of the "right side" restriction sites (EcoRI, NotI, XhoI, EcoRV, or XbaI of the pENTR vectors).
[0528] If you know your nucleic acid molecule of interest does not have, for example, an XhoI site, you can make a PCR product that has this structure (SEQ ID NO:33):
TABLE-US-00009 Xho I 5' ATG nnn nnn --- nnn TAA ctc gag nnn nnn 3' 3' tac nnn nnn --- nnn att gag ctc nnn nnn 5'
After cutting with XhoI, the fragment is ready to clone:
TABLE-US-00010 5' ATG nnn nnn --- nnn TAA c 3' 3' tac nnn nnn --- nnn att gag ct 5'
(If you follow this example, don't forget to put a phosphate on the amino oligo.)
[0529] Option 2:
[0530] This PCR product could be cloned into two Entry Vectors to give the desired products, between the XmnI and XhoI sites: pENTR1A (FIGS. 10A, 10B) or pENTR7 (FIGS. 16A, 16B). If you clone into pENTR1A, amino fusions will have the minimal number of amino acids between the fusion domain and your nucleic acid molecule of interest, but the fusion cannot be removed with TEV protease. The converse is true of clones in pENTR7, i.e., an amino fusion can be cleaved with TEV protease, at the cost of more amino acids between the fusion and your nucleic acid molecule of interest.
[0531] In this example, let us choose to clone our hypothetical nucleic acid molecule of interest into pENTR7, between the XmnI and XhoI sites. Once this is accomplished, several optional protocols using the Entry Clone pENTR7 may be followed:
[0532] Option 3:
[0533] Since the nucleic acid molecule of interest has been amplified with PCR, it may be desirable to sequence it. To do this, transfer the nucleic acid molecule of interest from the Entry Vector into a vector that has priming sites for the standard sequencing primers. Such a vector is pDEST6 (FIGS. 26A, 26B). This Destination Vector places the nucleic acid molecule of interest in the opposite orientation to the lac promoter (which is leaky--see Example 3 above). If the gene product is toxic to E. coli, this Destination Vector will minimize its toxicity.
[0534] Option 4:
[0535] While the sequencing is going on, you might wish to check the expression of the nucleic acid molecule of interest in, for example, CHO cells, by recombining the nucleic acid molecule of interest into a CMV promoter vector (pDEST7, FIG. 27; or pDEST12, FIG. 32), or into a baculovirus vector (pDEST8, FIG. 28; or pDEST10, FIG. 30) for expression in insect cells. Both of these vectors will transcribe the coding sequence of your nucleic acid molecule of interest, and translate it from the ATG of the PCR product using the Kozak bases upstream of the XmnI site.
Option 5:
[0536] If you wish to purify protein, for example to make antibodies, you can clone the nucleic acid molecule of interest into a His6 fusion vector, pDEST2 (FIG. 22). Since the nucleic acid molecule of interest is cloned downstream of the TEV protease cleavage domain of pENTR7 (FIG. 16), the amino acid sequence of the protein produced will be:
TABLE-US-00011 (SEQ ID NO: 34) [------attB1-----] TEV protease NH2-MSYYHHHHHHGITSLYKKAGF ↓ TM----COOH
[0537] The attB site and the restriction sites used to make the Destination and Entry Vectors are translated into the underlined 11 amino acids (GITSLYKKAGF) (SEQ ID NO:35). Cleavage with TEV protease (arrow) leaves two amino acids, GT, on the amino end of the gene product.
[0538] See FIG. 55 for an example of a nucleic acid molecule of interest, the chloramphenicol acetyl transferase (CAT) gene, cloned into pENTR7 (FIG. 16) as a blunt (amino)-XhoI (carboxy) fragment, then cloned by recombination into the His6 fusion vector pDEST2 (FIG. 22).
Option 6:
[0539] If the His6 fusion protein is insoluble, you may go on and try a GST fusion. The appropriate Destination vector is pDEST3 (FIG. 23).
Option 7:
[0540] If you need to make RNA probes and prefer SP6 RNA polymerase, you can make the top strand RNA with your nucleic acid molecule of interest cloned into pSPORT+ (pDEST5 (FIGS. 25A, 25B)), and the bottom strand RNA with the nucleic acid molecule of interest cloned into pSPORT(-) (pDEST6 (FIGS. 26A, 26B)). Opposing promoters for Ti RNA polymerase and SP6 RNA polymerase are also present in these clones.
Option 8:
[0541] It is often worthwhile to clone your nucleic acid molecule of interest into a variety of Destination Vectors in the same experiment. For example, if the number of colonies varies widely when the various recombination reactions are transformed into E. coli, this may be an indication that the nucleic acid molecule of interest is toxic in some contexts. (This problem is more clearly evident when a positive control gene is used for each Destination Vector.) Specifically, if many more colonies are obtained when the nucleic acid molecule of interest is recombined into pDEST6 than in pDEST5, there is a good chance that leakiness of the lac promoter is causing some expression of the nucleic acid molecule of interest in pSPORT "+" (which is not harmful in pDEST6 because the nucleic acid molecule of interest is in the opposite orientation).
Example 16
Demonstration of a One-Tube Transfer of a PCR Product (or Expression Clone) to Expression Clone Via a Recombinational Cloning Reaction
[0542] In the BxP recombination (Entry or Gateward) reaction described herein, a DNA segment flanked by attB1 and attB2 sites in a plasmid conferring ampicillin resistance was transferred by recombination into an attP plasmid conferring kanamycin resistance, which resulted in a product molecule wherein the DNA segment was flanked by attL sites (attL1 and attL2). This product plasmid comprises an "attL Entry Clone" molecule, because it can react with a "attR Destination Vector" molecule via the LxR (Destination) reaction, resulting in the transfer of the DNA segment to a new (ampicillin resistant) vector. In the previously described examples, it was necessary to transform the BxP reaction products into E. coli, select kanamycin resistant colonies, grow those colonies in liquid culture, and prepare miniprep DNA, before reacting this DNA with a Destination Vector in an LxR reaction.
[0543] The goal of the following experiment was to eliminate the transformation and miniprep DNA steps, by adding the BxP Reaction products directly to an LxR Reaction. This is especially appropriate when the DNA segment flanked by attB sites is a PCR product instead of a plasmid, because the PCR product cannot give ampicillin-resistant colonies upon transformation, whereas attB plasmids (in general) carry an ampicillin resistance gene. Thus use of a PCR product flanked by attB sites in a BxP Reaction allows one to select for the ampicillin resistance encoded by the desired attB product of a subsequent LxR Reaction.
[0544] Two reactions were prepared: Reaction A, negative control, no attB PCR product, (8 μl) contained 50 ng pEZC7102 (attP Donor plasmid, confers kanamycin resistance) and 2 μl BxP Clonase (22 ng/μl Int protein and 8 ng/μl IHF protein) in BxP buffer (25 mM Tris HCl, pH 7.8, 70 mM KCl, 5 mM spermidine, 0.5 mM EDTA, 250 μg/ml BSA). Reaction B (24 μl) contained 150 ng pEZC7102, 6 μl BxP Clonase, and 120 ng of the attB-tet-PCR product in the same buffer as reaction A. The attB-tet-PCR product comprised the tetracycline resistance gene of plasmid pBR322, amplified with two primers containing either attB1 or attB2 sites, and having 4 Gs at their 5' ends, as described earlier.
[0545] The two reactions were incubated at 25° C. for 30 minutes. Then aliquots of these reactions were added to new components that comprised LxR Reactions or appropriate controls for the LxR Reaction. Five new reactions were thus produced:
Reaction 1:
[0546] 5 μl of reaction A was added to a 5 μl LxR Reaction containing 25 ng NcoI-cut pEZC8402 (the attR Destination Vector plasmid) in LxR buffer (37.5 mM Tris HCl, pH 7.7, 16.5 mM NaCl, 35 mM KCl, 5 mM spermidine, 375 μg/ml BSA), and 1 μl of GATEWAY® LR Clonase® Enzyme Mix (total volume of 10 μl).
Reaction 2:
[0547] Same as reaction 1, except 5 μl of reaction B (positive) were added instead of reaction A (negative).
Reaction 3:
[0548] Same as reaction 2, except that the amounts of Nco-cut pEZC8402 and GATEWAY® LR Clonase® Enzyme Mix were doubled, to 50 ng and 2 μl, respectively.
Reaction 4:
[0549] Same as reaction 2, except that 25 ng of pEZ11104 (a positive control attL Entry Clone plasmid) were added in addition to the aliquot of reaction B.
Reaction 5:
[0550] Positive control LxR Reaction, containing 25 ng NcoI-cut pEZC8402, 25 ng pEZ11104, 37.5 mM Tris HCl pH 7.7, 16.5 mM NaCl, 35 mM KCl, 5 mM spermidine, 375 μg/ml BSA and 1 μl GATEWAY® LR Clonase® Enzyme Mix in a total volume of 5 μl.
[0551] All five reactions were incubated at 25° C. for 30 minutes. Then, 1 μl aliquots of each of the above five reactions, plus 1 μl from the remaining volume of Reaction B, the standard BxP Reaction, were used to transform 50 μl competent DH5α E. coli. DNA and cells were incubated on ice for 15 min., heat shocked at 42° C. for 45 sec., and 450 μl SOC were added. Each tube was incubated with shaking at 37° C. for 60 min. Aliquots of 100 μl and 400 μl of each transformation were plated on LB plates containing either 50 μg/ml kanamycin or 100 μg/ml ampicillin (see Table 2). A transformation with 10 pg of pUC19 DNA (plated on LB-amp100) served as a control on the transformation efficiency of the DH5α cells. Following incubation overnight at 37° C., the number of colonies on each plate was determined.
[0552] Results of these reactions are shown in Table 2.
TABLE-US-00012 TABLE 2 Reaction No: 1 2 3 4 5 6 Number of Colonies Vol. Neg. 1X 2X LxR LxR BxP plated: Control pEZC8402 pEZC8402 Reaction Reaction Reaction BxP and LR and LR with Pos. alone alone Reaction Clonase.sup. ® Clonase.sup. ® Control DNA 100 μl 2 1 8 9 ~1000 ~1000 400 μl 5 10 35 62 >2000 >2000 Selection: Kan Amp Amp Amp Amp Kan *(Transformation with pUC 19 DNA yielded 1.4 × 109 CFU/μg DNA.)
[0553] 34 of the 43 colonies obtained from Reaction 3 were picked into 2 ml Terrific Broth with 100 μg/ml ampicillin and these cultures were grown overnight, with shaking, at 37° C. 27 of the 34 cultures gave at least moderate growth, and of these 24 were used to prepare miniprep DNA, using the standard protocol. These 24 DNAs were initially analyzed as supercoiled (SC) DNA on a 1% agarose gel to identify those with inserts and to estimate the sizes of the inserts. Fifteen of the 24 samples displayed SC DNA of the size predicted (5553 bp) if tetx7102 had correctly recombined with pEZC8402 to yield tetx8402. One of these samples contained two plasmids, one of ˜5500 bp and a one of ˜3500 bp. The majority of the remaining clones were approximately 4100 bp in size.
[0554] All 15 of the clones displaying SC DNA of predicted size (˜5500 bp) were analyzed by two different double digests with restriction endonucleases to confirm the structure of the expected product: tetx8402. (See plasmid maps, FIGS. 57-59) In one set of digests, the DNAs were treated with Not I and Eco RI, which should cut the predicted product just outside both attB sites, releasing the tee insert on a fragment of 1475 bp. In the second set of digests, the DNAs were digested with NotI and with NruI. NruI cleaves asymmetrically within the subcloned tee insert, and together with NotI will release a fragment of 1019 bp.
[0555] Of the 15 clones analyzed by double restriction digestion, 14 revealed the predicted sizes of fragments for the expected product.
Interpretation:
[0556] The DNA components of Reaction B, pEZC7102 and attB-tet-PCR, are shown in FIG. 56. The desired product of BxP Reaction B is tetx7102, depicted in FIG. 57. The LxR Reaction recombines the product of the BxP Reaction, tetx7102 (FIG. 57), with the Destination Vector, pEZC8402, shown in FIG. 58. The LxR Reaction with tetx7102 plus pEZC8402 is predicted to yield the desired product tetx8402, shown in FIG. 59.
[0557] Reaction 2, which combined the BxP Reaction and LxR Reaction, gave few colonies beyond those of the negative control Reaction. In contrast, Reaction 3, with twice the amount of pEZC8402 (FIG. 58) and LxR Clonase, yielded a larger number of colonies. These colonies were analyzed further, by restriction digestion, to confirm the presence of expected product. Reaction 4 included a known amount of attL Entry Clone plasmid in the combined BxP-plus-LxR reaction. But reaction 4 yielded only about 1% of the colonies obtained when the same DNA was used in a LxR reaction alone, Reaction 6. This result suggests that the LxR reaction may be inhibited by components of the BxP reaction.
[0558] Restriction endonuclease analysis of the products of Reaction 3 revealed that a sizeable proportion of the colonies (14 of the 34 analyzed) contained the desired tee subclone, tetx8402 (FIG. 59).
[0559] The above results establish the feasibility of performing first a BxP recombination reaction followed by a LxR recombination reaction--in the same tube--simply by adding the appropriate buffer mix, recombination proteins, and DNAs to a completed BxP reaction. This method should prove useful as a faster method to convert attB-containing PCR products into different Expression Clones, eliminating the need to isolate first the intermediate attL-PCR insert subclones, before recombining these with Destination Vectors. This may prove especially valuable for automated applications of these reactions.
[0560] This same one-tube approach allows for the rapid transfer of nucleic acid molecules contained in attB plasmid clones into new functional vectors as well. As in the above examples, attL subclones generated in a BxP Reaction can be recombined directly with various Destination Vectors in a LxR reaction. The only additional requirement for using attB plasmids, instead of attB-containing PCR products, is that the Destination Vector(s) employed must contain a different selection marker from the one present on the attB plasmid itself and the attP vector.
[0561] Two alternative protocols for a one-tube reaction have also proven useful and somewhat more optimal than the conditions described above.
Alternative 1:
[0562] Reaction buffer contained 50 mM Tris-HCl (pH 7.5), 50 mM NaCl, 0.25 mM EDTA, 2.5 mM spermidine, and 200 μg/ml BSA. After a 16 (or 3) hour incubation of the PCR product (100 ng)+attP Donor plasmid (100 ng)+GATEWAY® BP Clonase® Enzyme Mix+Destination Vector (100 ng), 2 μl of GATEWAY® LR Clonase® Enzyme Mix (per 10 μl reaction mix) was added and the mixture was incubated an additional 6 (or 2) hours at 25° C. Stop solution was then added as above and the mixture was incubated at 37° C. as above and transformed by electroporation with 1 μl directly into electrocompetent host cells. Results of this series of experiments demonstrated that longer incubation times (16 hours vs. 3 hours for the BP Reaction, 6 hours vs. 2 hours for the LR Reaction) resulted in about twice as many colonies being obtained as for the shorter incubation times. With two independent genes, 10/10 colonies having the correct cloning patterns were obtained.
Alternative 2:
[0563] A standard BP Reaction under the reaction conditions described above for Alternative 1 was performed for 2 hours at 25° C. Following the BP Reaction, the following components were added to the reaction mixture in a total volume of 7 μl:
[0564] 20 mM Tris-HCl, pH 7.5
[0565] 100 mM NaCl
[0566] 5 μg/ml Xis-His6
[0567] 15% glycerol
[0568] ˜1000 ng of Destination Vector The reaction mixture was then incubated for 2 hours at 25° C., and 2.5 μl of stop solution (containing 2 μg/ml proteinase K) was added and the mixture was incubated at 37° C. for an additional 10 minutes. Chemically competent host cells were then transformed with 2 μl of the reaction mixture, or electrocompetent host cells (e.g., EMax DH10B cells; Life Technologies, Inc.) were electroporated with 2 μl of the reaction mixture per 25-40 μl of cells. Following transformation, mixtures were diluted with SOC, incubated at 37° C., and plated as described above on media selecting for the selection markers on the Destination Vector and the Entry clone (B×P reaction product). Analogous results to those described for Alternative 1 were obtained with these reaction conditions--a higher level of colonies containing correctly recombined reaction products were observed.
Example 17
Demonstration of a One-Tube Transfer of a PCR Product (or Expression Clone) to Expression Clone Via a Recombinational Cloning Reaction
[0569] Single-tube transfer of PCR product DNA or Expression Clones into Expression Clones by recombinational cloning has also been accomplished using a procedure modified from that described in Example 16. This procedure is as follows:
[0570] Perform a standard BP (Gateward) Reaction (see Examples 9 and 10) in 20 μl volume at 25° C. for 1 hour.
[0571] After the incubation is over, take a 10 μl aliquot from the 20 μl total volume and add 1 μl of Proteinase K (2 mg/ml) and incubate at 37° C. for 10 minutes. This first aliquot can be used for transformation and gel assay of BP reaction analysis. Plate BP reaction transformation on LB plates with Kanamycin (50 ug/ml).
[0572] Add the following reagents to the remaining 10 μl aliquot of the BP reaction:
[0573] 1 μl of 0.75 M NaCl
[0574] 2 μl of destination vector (150 ng/μl)
[0575] 4 μl of LR Clonase® (after thawing and brief mixing)
[0576] Mix all reagents well and incubate at 25° C. for 3 hours. Stop the reaction at the end of incubation with 1.7 μl of Proteinase K (2 mg/ml) and incubate at 37° C. for 10 minutes.
[0577] Transform 2 μl of the completed reaction into 100 μl of competent cells. Plate 100 μl and 400 μl on LB plates with Ampicilin (100 μg/ml).
Notes:
[0578] If your competent cells are less than 108 CFU/μg, and you are concerned about getting enough colonies, you can improve the yield several fold by incubating the BP reaction for 6-20 hours. Electroporation also can yield better colony output than chemical transformation.
[0579] PCR products greater than about 5-6 kb show significantly lower cloning efficiency in the BP reaction. In this case, we recommend using longer incubation times for both BP and LR steps.
[0580] If you want to move your insert gene into several destination vectors simultaneously, then scale up the initial BP reaction volume so that you have a 10 μl aliquot for adding each destination vector.
Example 18
Optimization of GATEWAY® Clonase® Enzyme Compositions
[0581] The enzyme compositions containing Int and IHF (for BP Reactions) were optimized using a standard functional recombinational cloning reaction (a BP reaction) between attB-containing plasmids and attP-containing plasmids, according to the following protocol:
Materials and Methods:
[0582] Substrates:
[0583] AttP--supercoiled pDONR201
[0584] AttB--linear ˜1 Kb [3H]PCR product amplified from pEZC7501
[0585] Proteins:
[0586] IntH6--His6-carboxy-tagged λ Integrase
[0587] IHF--Integration Host Factor
[0588] Clonase:
[0589] 50 ng/μl IntH6 and 20 ng/μl IHF, admixed in 25 mM Tris-HCl (pH 7.5),
[0590] 22 mM NaCl, 5 mM EDTA, 1 mg/ml BSA, 5 mM Spermidine, and 50% glycerol.
[0591] Reaction Mixture (Total Volume of 40 μl):
[0592] 1000 ng AttP plasmid
[0593] 600 ng AttB [3H] PCR product
[0594] 8 μl Clonase (400 ng IntH6, 160 ng IHF) in 25 mM Tris-HCl (pH 7.5),
[0595] 22 mM NaCl, 5 mM EDTA, 1 mg/ml BSA, 5 mM Spermidine, 5 mM DTT.
[0596] Reaction mixture was incubated for 1 hour at 25° C., 4 μl of 2 μg/μl proteinase K was added and mixture was incubated for an additional 20 minutes at 37° C. Mixture was then extracted with an equal volume of Phenol/Chloroform/Isoamyl alcohol. The aqueous layer was then collected, and 0.1 volumes of 3 M sodium acetate and 2 volumes of cold 100% ethanol were added. Tubes were then spun in a microcentrifuge at maximum RPM for 10 minutes at room temperature. Ethanol was decanted, and pellets were rinsed with 70% ethanol and re-centrifuged as above. Ethanol was decanted, and pellets were allowed to air dry for 5-10 minutes and then dissolved in 20 μl of 33 mM Tris-Acetate (pH 7.8), 66 mM potassium acetate, 10 mM magnesium acetate, 1 mM DTT, and 1 mM ATP. 2 units of exonuclease V (e.g., Plasmid Safe; EpiCentre, Inc., Madison, Wis.) was then added, and the mixture was incubated at 37° C. for 30 minutes.
[0597] Samples were then TCA-washed by spotting 30 μl of reaction mixture onto a Whatman GF/C filter, washing filters once with 10% TCA+1% NaPPi for 10 minutes, three times with 5% TCA for 5 minutes each, and twice with ethanol for 5 minutes each. Filters were then dried under a heat lamp, placed into a scintillation vial, and counted on a β liquid scintillation counter (LSC).
[0598] The principle behind this assay is that, after exonuclease V digestion, only double-stranded circular DNA survives in an acid-insoluble form. All DNA substrates and products that have free ends are digested to an acid-soluble form and are not retained on the filters. Therefore, only the 3H-labeled attB linear DNA which ends up in circular form after both inter- and intramolecular integration is complete is resistant to digestion and is recovered as acid-insoluble product. Optimal enzyme and buffer formulations in the Clonase compositions therefore are those that give the highest levels of circularized 3H-labeled attB-containing sequences, as determined by highest cpm in the LSC. Although this assay was designed for optimization of GATEWAY® BP Clonase® Enzyme Mix compositions (Int+IHF), the same type of assay may be performed to optimize GATEWAY® LR Clonase® Enzyme Mix compositions (Int+IHF+Xis), except that the reaction mixtures would comprise 1000 ng of AttR (instead of AttP) and 600 ng of AttL (instead of AttB), and 40 ng of His6-carboxy-tagged Xis (XisH6) in addition to the IntH6 and IHF.
Example 19
Testing Functionality of Entry and Destination Vectors
[0599] As part of assessment of the functionality of particular vectors of the invention, it is important to functionally test the ability of the vectors to recombine. This assessment can be carried out by performing a recombinational cloning reaction (as schematized in FIGS. 2, 4, and 5A and 5B, and as described herein and in commonly owned U.S. application Ser. No. 08/486,139, filed Jun. 7, 1995, Ser. No. 08/663,002, filed Jun. 7, 1996 (now U.S. Pat. No. 5,888,732), Ser. No. 09/005,476, filed Jan. 12, 1998, and Ser. No. 09/177,387, filed Oct. 23, 1998, the disclosures of all of which are incorporated by reference herein in their entireties), by transforming E. coli and scoring colony forming units. However, an alternative assay may also be performed to allow faster, more simple assessment of the functionality of a given Entry or Destination Vector by agarose gel electrophoresis. The following is a description of such an in vitro assay.
Materials and Methods:
[0600] Plasmid templates pEZC1301 (FIG. 84) and pEZC1313 (FIG. 85), each containing a single wild type att site, were used for the generation of PCR products containing attL or attR sites, respectively. Plasmid templates were linearized with AlwNI, phenol extracted, ethanol precipitated and dissolved in TE to a concentration of 1 ng/μl.
TABLE-US-00013 PCR primers (capital letters represent base changes from wildtype): attL1 (SEQ ID NO: 36) gggg agcct gcttttttGtacAaa gttggcatta taaaaaagca ttgc; attL2 (SEQ ID NO: 37) gggg agcct gctttCttGtacAaa gttggcatta taaaaaagca ttgc; attL right (SEQ ID NO: 38) tgttgccggg aagctagagt aa; attR1 (SEQ ID NO: 39) gggg Acaag ttTgtaCaaaaaagc tgaacgaga aacgtaaaat; attR2 (SEQ ID NO: 40) gggg Acaag ttTgtaCaaGaaagc tgaacgaga aacgtaaaat; attR right (SEQ ID NO: 41) ca gacggcatga tgaacctgaa
PCR primers were dissolved in TE to a concentration of 500 pmol/μl. Primer mixes were prepared, consisting of attL1+attLright primers, attL2+attLright primers, attR1+attRright primers, and attR2+attRright primers, each mix containing 20 pmol/μl of each primer.
[0601] PCR Reactions:
1 μl plasmid template (1 ng) 1 μl primer pairs (20 pmoles of each)
3 μl of H20
45 μl of Platinum PCR SuperMix® (Life Technologies, Inc.)
[0602] Cycling Conditions (Performed in MJ Thermocycler):
[0603] 95° C./2 minutes
[0604] 94° C./30 seconds
[0605] 25 cycles of 58° C./30 seconds and 72° C./1.5 minutes
[0606] 72° C./5 minutes
[0607] 5° C./hold
[0608] The resulting attL PCR product was 1.5 kb, and the resulting attR PCR product was 1.0 kb.
[0609] PCR reactions were PEG/MgCl2 precipitated by adding 150 μl H2O and 100 μl of 3×PEG/MgCl2 solution followed by centrifugation. The PCR products were dissolved in 50 μl of TE. Quantification of the PCR product was performed by gel electrophoresis of 1 μl and was estimated to be 50-100 ng/μl.
[0610] Recombination reactions of PCR products containing attL or attR sites with GATEWAY® plasmids was performed as follows:
[0611] 8 μl of H2O
[0612] 2 μl of attL or attR PCR product (100-200 ng)
[0613] 2 μl of GATEWAY® plasmid (100 ng)
[0614] 4 μl of 5× Destination buffer
[0615] 4 μl of GATEWAY® LR Clonase® Enzyme Mix
[0616] 20 μl total volume (the reactions can be scaled down to a 5 μl total volume by adjusting the volumes of the components to about 1/4 of those shown above, while keeping the stoichiometries the same).
[0617] Clonase reactions were incubated at 25° C. for 2 hours. 2 μl of proteinase K (2 mg/ml) was added to stop the reaction. 10 μl was then run on a 1% agarose gel. Positive control reactions were performed by reacting attL1 PCR product (1.0 kb) with attR1 PCR product (1.5 kb) and by similarly reacting attL2 PCR product with attR2 PCR product to observe the formation of a larger (2.5 kb) recombination product. Negative controls were similarly performed by reacting attL1 PCR product with attR2 PCR product and vice versa or reactions of attL PCR product with an attL plasmid, etc.
[0618] In alternative assays, to test attB Entry vectors, plasmids containing single attP sites were used. Plasmids containing single att sites could also be used as recombination substrates in general to test all Entry and Destination vectors (i.e., those containing attL, attR, attB and attP sites). This would eliminate the need to do PCR reactions.
Results:
[0619] Destination and Entry plasmids when reacted with appropriate att-containing PCR products formed linear recombinant molecules that could be easily visualized on an agarose gel when compared to control reactions containing no attL or attR PCR product. Thus, the functionality of Destination and Entry vectors constructed according to the invention may be determined either by carrying out the Destination or Entry recombination reactions as depicted in FIGS. 2, 4, and 5A and 5B, or more rapidly by carrying out the linearization assay described in this Example.
Example 20
PCR Cloning Using Universal Adapter-Primers
[0620] As described herein, the cloning of PCR products using the GATEWAY® PCR Cloning System (Life Technologies, Inc.; Rockville, Md.) requires the addition of attB sites (attB1 and attB2) to the ends of gene-specific primers used in the PCR reaction. The protocols described in the preceding Examples suggest that the user add 29 bp (25 bp containing the attB site plus four G residues) to the gene-specific primer. It would be advantageous to high volume users of the GATEWAY® PCR Cloning System to generate attB-containing PCR product using universal attB adapter-primers in combination with shorter gene-specific primers containing a specified overlap to the adapters. The following experiments demonstrate the utility of this strategy using universal attB adapter-primers and gene-specific primers containing overlaps of various lengths from 6 bp to 18 bp. The results demonstrate that gene-specific primers with overlaps of 10 bp to 18 bp can be used successfully in PCR amplifications with universal attB adapter-primers to generate full-length PCR products. These PCR products can then be successfully cloned with high fidelity in a specified orientation using the GATEWAY® PCR Cloning System.
Methods and Results:
[0621] To demonstrate that universal attB adapter-primers can be used with gene-specific primers containing partial attB sites in PCR reactions to generate full-length PCR product, a small 256 bp region of the human hemoglobin cDNA was chosen as a target so that intermediate sized products could be distinguished from full-length products by agarose gel electrophoresis.
The following oligonucleotides were used:
TABLE-US-00014 (SEQ ID NO: 42) B1-Hgb: GGGG ACA AGT TTG TAC AAA AAA GCA GGC T-5'-Hgb*; B2-Hgb: (SEQ ID NO: 43) GGGG ACC ACT TTG TAC AAG AAA GCT GGG T-3'-Hgb**; 18B1-Hgb: (SEQ ID NO: 44) TG TAC AAA AAA GCA GGC T-5'-Hgb; 18B2-Hgb: (SEQ ID NO: 45) TG TAC AAG AAA GCT GGG T-3'-Hgb; 15B1-Hgb: (SEQ ID NO: 46) AC AAA AAA GCA GGC T-5'-Hgb; 15B2-Hgb: (SEQ ID NO: 47) AC AAG AAA GCT GGG T-3'-Hgb; 12B1-Hgb: (SEQ ID NO: 48) AA AAA GCA GGC T-5'-Hgb; 12B2-Hgb: (SEQ ID NO: 49) AG AAA GCT GGG T-3'-Hgb; 11B1-Hgb: (SEQ ID NO: 50) A AAA GCA GGC T-5'-Hgb; 11B2-Hgb: (SEQ ID NO: 51) G AAA GCT GGG T-3'-Hgb; 10B1-Hgb: (SEQ ID NO: 52) AAA GCA GGC T-5'-Hgb; 10B2-Hgb: (SEQ ID NO: 53) AAA GCT GGG T-3'-Hgb; 9B1-Hgb: AA GCA GGC T-5'-Hgb 9B2-Hgb: AA GCT GGG T-3'-Hgb 8B1-Hgb: A GCA GGC T-5'-Hgb 8B2-Hgb: A GCT GGG T-3'-Hgb 7B1-Hgb: GCA GGC T-5'-Hgb 7B2-Hgb: GCT GGG T-3'-Hgb 6B1-Hgb: CA GGC T-5'-Hgb 6B2-Hgb: CT GGG T-3'-Hgb attB1 adapter: (SEQ ID NO: 54) GGGG ACA AGT TTG TAC AAA AAA GCA GGC T; attB2 adapter: (SEQ ID NO: 55) GGGG ACC ACT TTG TAC AAG AAA GCT GGG T; (SEQ ID NO: 56) *-5'-Hgb = GTC ACT AGC CTG TGG AGC AAG A; (SEQ ID NO: 57) **-3'-Hgb = AGG ATG GCA GAG GGA GAC GAC A
[0622] The aim of these experiments was to develop a simple and efficient universal adapter PCR method to generate attB containing PCR products suitable for use in the GATEWAY® PCR Cloning System. The reaction mixtures and thermocycling conditions should be simple and efficient so that the universal adapter PCR method could be routinely applicable to any PCR product cloning application.
[0623] PCR reaction conditions were initially found that could successfully amplify predominately full-length PCR product using gene-specific primers containing 18 bp and 15 bp overlap with universal attB primers. These conditions are outlined below:
[0624] 10 pmoles of gene-specific primers
[0625] 10 pmoles of universal attB adapter-primers
[0626] 1 ng of plasmid containing the human hemoglobin cDNA.
[0627] 100 ng of human leukocyte cDNA library DNA.
[0628] 5 μl of 10× PLATINUM Taq HiFi® reaction buffer (Life Technologies, Inc.)
[0629] 2 μl of 50 mM MgSO4
[0630] 1 μl of 10 mM dNTPs
[0631] 0.2 μl of PLATINUM Taq HiFi® (1.0 unit)
[0632] H2O to 50 μl total reaction volume
Cycling Conditions:
[0633] 25 × | 95 ° C . / 5 min 94 ° C . / 15 sec 50 ° C . / 30 sec 68 ° C . / 1 min 68 ° C . / 5 min 5 ° C . / hold ##EQU00002##
[0634] To assess the efficiency of the method, 2 μl ( 1/25) of the 50 μl PCR reaction was electrophoresed in a 3% Agarose-1000 gel. With overlaps of 12 bp or less, smaller intermediate products containing one or no universal attB adapter predominated the reactions. Further optimization of PCR reaction conditions was obtained by titrating the amounts of gene-specific primers and universal attB adapter-primers. The PCR reactions were set up as outlined above except that the amounts of primers added were:
0, 1, 3 or 10 pmoles of gene-specific primers 0, 10, 30 or 100 pmoles of adapter-primers
Cycling Conditions:
[0635] 25 × | 95 ° C . / 3 min 94 ° C . / 15 sec 50 ° C . / 45 sec 68 ° C . / 1 min 68 ° C . / 5 min 5 ° C . / hold ##EQU00003##
[0636] The use of limiting amounts of gene-specific primers (3 pmoles) and excess adapter-primers (30 pmoles) reduced the amounts of smaller intermediate products. Using these reaction conditions the overlap necessary to obtain predominately full-length PCR product was reduced to 12 bp. The amounts of gene-specific and adapter-primers was further optimized in the following PCR reactions:
0, 1, 2 or 3 pmoles of gene-specific primers 0, 30, 40 or 50 pmoles of adapter-primers
Cycling Conditions:
[0637] 25 × | 95 ° C . / 3 min 94 ° C . / 15 sec 48 ° C . / 1 min 68 ° C . / 1 min 68 ° C . / 5 min 5 ° C . / hold ##EQU00004##
[0638] The use of 2 pmoles of gene-specific primers and 40 pmoles of adapter-primers further reduced the amounts of intermediate products and generated predominately full-length PCR products with gene-specific primers containing an 11 bp overlap. The success of the PCR reactions can be assessed in any PCR application by performing a no adapter control. The use of limiting amounts of gene-specific primers should give faint or barely visible bands when 1/25 to 1/10 of the PCR reaction is electrophoresed on a standard agarose gel. Addition of the universal attB adapter-primers should generate a robust PCR reaction with a much higher overall yield of product.
[0639] PCR products from reactions using the 18 bp, 15 bp, 12 bp, 11 bp and 10 bp overlap gene-specific primers were purified using the CONCERT® Rapid PCR Purification System (PCR products greater than 500 bp can be PEG precipitated). The purified PCR products were subsequently cloned into an attP containing plasmid vector using the GATEWAY® PCR Cloning System (Life Technologies, Inc.; Rockville, Md.) and transformed into E. coli. Colonies were selected and counted on the appropriate antibiotic media and screened by PCR for correct inserts and orientation.
[0640] Raw PCR products (unpurified) from the attB adapter PCR of a plasmid clone of part of the human beta-globin (Hgb) gene were also used in GATEWAY® PCR Cloning System reactions. PCR products generated with the full attB B1/B2-Hgb, the 12B1/B2, 11B1/B2 and 10B1/B2 attB overlap Hgb primers were successfully cloned into the GATEWAY® pENTR21 attP vector (FIG. 49). 24 colonies from each (24×4=96 total) were tested and each was verified by PCR to contain correct inserts. The cloning efficiency expressed as cfu/ml is shown below:
TABLE-US-00015 Primer Used cfu/ml Hgb full attB 8,700 Hgb 12 bp overlap 21,000 Hgb 11 bp overlap 20,500 Hgb 10 bp overlap 13,500 GFP control 1,300
[0641] Interestingly, the overlap PCR products cloned with higher efficiency than did the full attB PCR product. Presumably, and as verified by visualization on agarose gel, the adapter PCR products were slightly cleaner than was the full attB PCR product. The differences in colony output may also reflect the proportion of PCR product molecules with intact attB sites.
[0642] Using the attB adapter PCR method, PCR primers with 12 bp attB overlaps were used to amplify cDNAs of different sizes (ranging from 1 to 4 kb) from a leukocyte cDNA library and from first strand cDNA prepared from HeLa total RNA. While three of the four cDNAs were able to be amplified by this method, a non-specific amplification product was also observed that under some conditions would interfere with the gene-specific amplification. This non-specific product was amplified in reactions containing the attB adapter-primers alone without any gene-specific overlap primers present. The non-specific amplification product was reduced by increasing the stringency of the PCR reaction and lowering the attB adapter PCR primer concentration.
[0643] These results indicate that the adapter-primer PCR approach described in this Example will work well for cloned genes. These results also demonstrate the development of a simple and efficient method to amplify PCR products that are compatible with the GATEWAY® PCR Cloning System that allows the use of shorter gene-specific primers that partially overlap universal attB adapter-primers. In routine PCR cloning applications, the use of 12 bp overlaps is recommended. The methods described in this Example can thus reduce the length of gene-specific primers by up to 17 residues or more, resulting in a significant savings in oligonucleotide costs for high volume users of the GATEWAY® PCR Cloning System. In addition, using the methods and assays described in this Example, one of ordinary skill can, using only routine experimentation, design and use analogous primer-adapters based on or containing other recombination sites or fragments thereof, such as attL, attR, attP, lox, FRT, etc.
Example 21
Mutational Analysis of the Bacteriophage Lambda attL and attR Sites: Determinants of Att Site Specificity In Site-Specific Recombination
[0644] To investigate the determinants of att site specificity, the bacteriophage lambda attL and attR sites were systematically mutagenized. As noted herein, the determinants of specificity have previously been localized to the 7 bp overlap region (TTTATAC, which is defined by the cut sites for the integrase protein and is the region where strand exchange takes place) within the 15 bp core region (GCTTTTTTATACTAA) (SEQ ID NO:58) which is identical in all four lambda att sites, attB, attP, attL and attR. This core region, however, has not heretofore been systematically mutagenized and examined to define precisely which mutations produce unique changes in att site specificity.
[0645] Therefore, to examine the effect of att sequence on site specificity, mutant attL and attR sites were generated by PCR and tested in an in vitro site-specific recombination assay. In this way all possible single base pair changes within the 7 bp overlap region of the core att site were generated as well as five additional changes outside the 7 bp overlap but within the 15 bp core att site. Each attL PCR substrate was tested in the in vitro recombination assay with each of the attR PCR substrates.
Methods
[0646] To examine both the efficiency and specificity of recombination of mutant attL and attR sites, a simple in vitro site-specific recombination assay was developed. Since the core regions of attL and attR lie near the ends of these sites, it was possible to incorporate the desired nucleotide base changes within PCR primers and generate a series of PCR products containing mutant attL and attR sites. PCR products containing attL and attR sites were used as substrates in an in vitro reaction with GATEWAY® LR Clonase® Enzyme Mix (Life Technologies, Inc.; Rockville, Md.). Recombination between a 1.5 kb attL PCR product and a 1.0 kb attR PCR product resulted in a 2.5 kb recombinant molecule that was monitored using agarose gel electrophoresis and ethidium bromide staining.
[0647] Plasmid templates pEZC1301 (FIG. 84) and pEZC1313 (FIG. 85), each containing a single wild type attL or attR site, respectively, were used for the generation of recombination substrates. The following list shows primers that were used in PCR reactions to generate the attL PCR products that were used as substrates in L×R Clonase reactions (capital letters represent changes from the wild-type sequence, and the underline represents the 7 bp overlap region within the 15 bp core att site; a similar set of PCR primers was used to prepare the attR PCR products containing matching mutations):
GATEWAY® sites (note: attL2 sequence in GATEWAY® plasmids begins "accca" while the attL2 site in this example begins "agcct" to reflect wild-type attL outside the core region.):
TABLE-US-00016 attL1: (SEQ ID NO: 36) gggg agcct gcttttttGtacAaa gttggcatta taaaaa- agca ttgc attL2: (SEQ ID NO: 37) gggg agcct gctttCttGtacAaa gttggcatta taaaaa- agca ttgc Wild-type: attL0: (SEQ ID NO: 59) gggg agcct gcttttttatactaa gttggcatta taaaaa- agca ttgc Single base changes from wild-type: attLT1A: (SEQ ID NO: 60) gggg agcct gctttAttatactaa gttggcatta taaaaa- agca ttgc attLT1C: (SEQ ID NO: 61) gggg agcct gctttCttatactaa gttggcatta taaaaa- agca ttgc attLT1G: (SEQ ID NO: 62) gggg agcct gctttGttatactaa gttggcatta taaaaa- agca ttgc attLT2A: (SEQ ID NO: 63) gggg agcct gcttttAtatactaa gttggcatta taaaaa- agca ttgc attLT2C: (SEQ ID NO: 64) gggg agcct gcttttCtatactaa gttggcatta taaaaa- agca ttgc attLT2G: (SEQ ID NO: 65) gggg agcct gcttttGtatactaa gttggcatta taaaa- aagca ttgc attLT3A: (SEQ ID NO: 66) gggg agcct gctttttAatactaa gttggcatta taaaa- aagca ttgc attLT3C: (SEQ ID NO: 67) gggg agcct gctttttCatactaa gttggcatta taaaa- aagca ttgc attLT3G: (SEQ ID NO: 68) gggg agcct gctttttGatactaa gttggcatta taaaa- aagca ttgc attLA4C: (SEQ ID NO: 69) gggg agcct gcttttttCtactaa gttggcatta taaaa- aagca ttgc attLA4G: (SEQ ID NO: 70) gggg agcct gcttttttGtactaa gttggcatta taaaa- aagca ttgc attLA4T: (SEQ ID NO: 71) gggg agcct gcttttttTtactaa gttggcatta taaaa- aagca ttgc attLT5A: (SEQ ID NO: 72) gggg agcct gcttttttaAactaa gttggcatta taaaa- aagca ttgc attLT5C: (SEQ ID NO: 73) gggg agcct gcttttttaCactaa gttggcatta taaaa- aagca ttgc attLT5G: (SEQ ID NO: 74) gggg agcct gcttttttaGactaa gttggcatta taaaa- aagca ttgc attLA6C: (SEQ ID NO: 75) gggg agcct gcttttttatCctaa gttggcatta taaaa- aagca ttgc attLA6G: (SEQ ID NO: 76) gggg agcct gcttttttatGctaa gttggcatta taaaa- aagca ttgc attLA6T: (SEQ ID NO: 77) gggg agcct gcttttttatTctaa gttggcatta taaaa- aagca ttgc attLC7A: (SEQ ID NO: 78) gggg agcct gcttttttataAtaa gttggcatta taaaa- aagca ttgc attLC7G: (SEQ ID NO: 79) gggg agcct gcttttttataGtaa gttggcatta taaaa- aagca ttgc attLC7T: (SEQ ID NO: 80) gggg agcct gcttttttataTtaa gttggcatta taaaa- aagca ttgc Single base changes outside of the 7 bp overlap: attL8: (SEQ ID NO: 81) gggg agcct Acttttttatactaa gttggcatta taaaa- aagca ttgc attL9: (SEQ ID NO: 82) gggg agcct gcCtttttatactaa gttggcatta taaaaa- agca ttgc attL10: (SEQ ID NO: 83) gggg agcct gcttCtttatactaa gttggcatta taaaaa- agca ttgc attL14: (SEQ ID NO: 84) gggg agcct gcttttttatacCaa gttggcatta taaaaa- agca ttgc attL15: (SEQ ID NO: 85) gggg agcct gcttttttatactaG gttggcatta taaaaa- agca ttgc
Note: additional vectors wherein the first nine bases are gggg agcca (i.e., substituting an adenine for the thymine in the position immediately preceding the 15-bp core region), which may or may not contain the single base pair substitutions (or deletions) outlined above, can also be used in these experiments.
[0648] Recombination reactions of attL- and attR-containing PCR products was performed as follows:
[0649] 8 μl of H20
[0650] 2 μl of attL PCR product (100 ng)
[0651] 2 μl of attR PCR product (100 ng)
[0652] 4 μl of 5× buffer
[0653] 4 μl of GATEWAY® LR Clonase® Enzyme Mix
[0654] 20 μl total volume
Clonase reactions were incubated at 25° C. for 2 hours. 2 μl of 10× Clonase stop solution (proteinase K, 2 mg/ml) were added to stop the reaction. 10 μl were run on a 1% agarose gel.
Results
[0655] Each attL PCR substrate was tested in the in vitro recombination assay with each of the attR PCR substrates. Changes within the first three positions of the 7 bp overlap (TTTATAC) strongly altered the specificity of recombination. These mutant att sites each recombined as well as the wild-type, but only with their cognate partner mutant; they did not recombine detectably with any other att site mutant. In contrast, changes in the last four positions (TTTATAC) only partially altered specificity; these mutants recombined with their cognate mutant as well as wild-type att sites and recombined partially with all other mutant att sites except for those having mutations in the first three positions of the 7 bp overlap. Changes outside of the 7 bp overlap were found not to affect specificity of recombination, but some did influence the efficiency of recombination.
[0656] Based on these results, the following rules for att site specificity were determined:
[0657] Only changes within the 7 bp overlap affect specificity.
[0658] Changes within the first 3 positions strongly affect specificity.
[0659] Changes within the last 4 positions weakly affect specificity.
[0660] Mutations that affected the overall efficiency of the recombination reaction were also assessed by this method. In these experiments, a slightly increased (less than 2-fold) recombination efficiency with attLT1A and attLC7T substrates was observed when these substrates were reacted with their cognate attR partners. Also observed were mutations that decreased recombination efficiency (approximately 2-3 fold), including attLA6G, attL14 and attL15. These mutations presumably reflect changes that affect Int protein binding at the core att site.
[0661] The results of these experiments demonstrate that changes within the first three positions of the 7 bp overlap (TTTATAC) strongly altered the specificity of recombination (i.e., att sequences with one or more mutations in the first three thymidines would only recombine with their cognate partners and would not cross-react with any other att site mutation). In contrast, mutations in the last four positions (TTTATAC) only partially altered specificity (i.e., att sequences with one or more mutations in the last four base positions would cross-react partially with the wild-type att site and all other mutant att sites, except for those having mutations in one or more of the first three positions of the 7 bp overlap). Mutations outside of the 7 bp overlap were not found to affect specificity of recombination, but some were found to influence (i.e., to cause a decrease in) the efficiency of recombination.
Example 22
Discovery of Att Site Mutations that Increase the Cloning Efficiency of GATEWAY® Cloning Reactions
[0662] In experiments designed to understand the determinants of att site specificity, point mutations in the core region of attL were made. Nucleic acid molecules containing these mutated attL sequences were then reacted in an LR reaction with nucleic acid molecules containing the cognate attR site (i.e., an attR site containing a mutation corresponding to that in the attL site), and recombinational efficiency was determined as described above. Several mutations located in the core region of the att site were noted that either slightly increased (less than 2-fold) or decreased (between 2-4-fold) the efficiency of the recombination reaction (Table 3).
TABLE-US-00017 TABLE 3 Effects of attL mutations on Recombination Reactions. SEQ Effect on Site ID NO: Sequence Recombination attL0 86 agcctgcttttttata ctaagttggcatta attL5 87 agcctgctttAttata slightly ctaagttggcatta increased attL6 88 agcctgcttttttata slightly Ttaagttggcatta increased attL13 89 agcctgcttttttatG decreased ctaagttggcatta attL14 90 agcctgcttttttata decreased cCaagttggcatta attL15 91 agcctgcttttttata decreased ctaGgttggcatta con- 92 CAACTTnnTnnnAnnA sensus AGTTG
[0663] It was also noted that these mutations presumably reflected changes that either increased or decreased, respectively, the relative affinity of the integrase protein for binding the core att site. A consensus sequence for an integrase core-binding site (CAACTTNNT) has been inferred in the literature but not directly tested (see, e.g., Ross and Landy, Cell 33:261-272 (1983)). This consensus core integrase-binding sequence was established by comparing the sequences of each of the four core att sites found in attP and attB as well as the sequences of five non-att sites that resemble the core sequence and to which integrase has been shown to bind in vitro. These experiments suggest that many more att site mutations might be identified which increase the binding of integrase to the core att site and thus increase the efficiency of GATEWAY® cloning reactions.
Example 23
Effects of Core Region Mutations on Recombination Efficiency
[0664] To directly compare the cloning efficiency of mutations in the att site core region, single base changes were made in the attB2 site of an attB1-TET-attB2 PCR product. Nucleic acid molecules containing these mutated attB2 sequences were then reacted in a BP reaction with nucleic acid molecules containing non-cognate attP sites (i.e., wildtype attP2), and recombinational efficiency was determined as described above The cloning efficiency of these mutant attB2 containing PCR products compared to standard attB1-TET-attB2 PCR product are shown in Table 4.
TABLE-US-00018 TABLE 4 Efficiency of Recombination With Mutated attB2 Sites. SEQ Cloning Site ID NO: Sequence Mutation Efficiency attB0 93 tcaagttagtataaaaaagcaggct attB1 94 ggggacaagtttgtacaaaaaagcaggct attB2 95 ggggaccactttgtacaagaaagctgggt 100% attB2.1 96 ggggaAcactttgtacaagaaagctgggt C→A 40% attB2.2 97 ggggacAactttgtacaagaaagctgggt C→A 131% attB2.3 98 ggggaccCctttgtacaagaaagctgggt A→C 4% attB2.4 99 ggggaccaAtttgtacaagaaagctgggt C→A 11% attB2.5 100 ggggaccacGttgtacaagaaagctgggt T→G 4% attB2.6 101 ggggaccactGtgtacaagaaagctgggt T→G 6% attB2.7 102 ggggaccacttGgtacaagaaagctgggt T→G 1% attB2.8 103 ggggaccactttTtacaagaaagctgggt G→T 0.5%
[0665] As noted above, a single base change in the attB2.2 site increased the cloning efficiency of the attB1-TET-attB2.2 PCR product to 131% compared to the attB1-TET-attB2 PCR product. Interestingly, this mutation changes the integrase core binding site of attB2 to a sequence that matches more closely the proposed consensus sequence.
[0666] Additional experiments were performed to directly compare the cloning efficiency of an attB1-TET-attB2 PCR product with a PCR product that contained attB sites containing the proposed consensus sequence (see Example 22) of an integrase core binding site. The following attB sites were used to amplify attB-TET PCR products:
TABLE-US-00019 attB1 (SEQ ID NO: 104) ggggacaagtttgtacaaaaaagcaggct attB1.6 SEQ ID NO: 105) ggggacaaCtttgtacaaaaaagTTggct attB2 (SEQ ID NO: 106) ggggaccactttgtacaagaaagctgggt attB2.10 (SEQ ID NO: 107) ggggacAactttgtacaagaaagTtgggt
[0667] BP reactions were carried out between 300 ng (100 fmoles) of pDONR201 (FIG. 49A) with 80 ng (80 fmoles) of attB-TET PCR product in a 20 μl volume with incubation for 1.5 hrs at 25° C., creating pENTR201-TET Entry clones. A comparison of the cloning efficiencies of the above-noted attB sites in BP reactions is shown in Table 5.
TABLE-US-00020 TABLE 5 Cloning efficiency of BP Reactions. Fold PCR product CFU/ml Increase B1-tet-B2 7,500 B1.6-tet-B2 12,000 1.6 x B1-tet-B2.10 20,900 2.8 x B1.6-tet-B2.10 30,100 4.0 x
[0668] These results demonstrate that attB PCR products containing sequences that perfectly match the proposed consensus sequence for integrase core binding sites can produce Entry clones with four-fold higher efficiency than standard Gateway attB1 and attB2 PCR products.
[0669] The entry clones produced above were then transferred to pDEST20 (FIG. 40A) via LR reactions (300 ng (64 fmoles) pDEST20 mixed with 50 ng (77 fmoles) of the respective pENTR201-TET Entry clone in 20 μl volume; incubated for 1 hr incubation at 25° C.). The efficiencies of cloning for these reactions are compared in Table 6.
TABLE-US-00021 TABLE 6 Cloning Efficiency of LR Reactions. Fold pENTR201-TET x pDRST20 CFU/ml Increase L1-tet-L2 5,800 L1.6-tet-L2 8,000 1.4 L1-tet-L2.10 10,000 1.7 L1.6-tet-L2.10 9,300 1.6
[0670] These results demonstrate that the mutations introduced into attB1.6 and attB2.10 that transfer with the gene into entry clones slightly increase the efficiency of LR reactions. Thus, the present invention encompasses not only mutations in attB sites that increase recombination efficiency, but also to the corresponding mutations that result in the attL sites created by the BP reaction.
[0671] To examine the increased cloning efficiency of the attB1.6-TET-attB2.10 PCR product over a range of PCR product amounts, experiments analogous to those described above were performed in which the amount of attB PCR product was titrated into the reaction mixture. The results are shown in Table 7.
TABLE-US-00022 TABLE 7 Titration of attB PCR products. Amount of attB PCR Fold product (ng) PCR product CFU/ml Increase 20 attB1-TET-attB2 3,500 6.1 attB1.6-TET-attB2.10 21,500 50 attB1-TET-attB2 9,800 5.0 attB1.6-TET-attB2.10 49,000 100 attB1-TET-attB2 18,800 2.8 attB1.6-TET-attB2.10 53,000 200 attB1-TET-attB2 19,000 2.5 attB1.6-TET-attB2.10 48,000
[0672] These results demonstrate that as much as a six-fold increase in cloning efficiency is achieved with the attB1.6-TET-attB2.10 PCR product as compared to the standard attB1-TET-attB2 PCR product at the 20 ng amount.
Example 24
Determination of attB Sequence Requirements for Optimum Recombination Efficiency
[0673] To examine the sequence requirements for attB and to determine which attB sites would clone with the highest efficiency from populations of degenerate attB sites, a series of experiments was performed. Degenerate PCR primers were designed which contained five bases of degeneracy in the B-arm of the attB site. These degenerate sequences would thus transfer with the gene into Entry clone in BP reactions and subsequently be transferred with the gene into expression clones in LR reactions. The populations of degenerate attB and attL sites could thus be cycled from attB to attL back and forth for any number of cycles. By altering the reaction conditions at each transfer step (for example by decreasing the reaction time and/or decreasing the concentration of DNA) the reaction can be made increasingly more stringent at each cycle and thus enrich for populations of attB and attL sites that react more efficiently.
[0674] The following degenerate PCR primers were used to amplify a 500 bp fragment from pUC18 which contained the lacZ alpha fragment (only the attB portion of each primer is shown):
TABLE-US-00023 attB1 (SEQ ID NO: 108) GGGG ACAAGTTTGTACAAA AAAGC AGGCT attB1n16-20 (SEQ ID NO: 109) GGGG ACAAGTTTGTACAAA nnnnn-AGGCT attB1n21-25 (SEQ ID NO: 110) GGGG ACAAGTTTGTACAAA AAAGC-nnnnn attB 2 (SEQ ID NO: 111) GGGG ACCACTTTGTACAAG AAAGC TGGGT attB2n16-20 (SEQ ID NO: 112) GGGG ACCACTTTGTACAAG nnnnn-TGGGT attB2n21-25 (SEQ ID NO: 113) GGGG ACCACTTTGTACAAG AAAGC-nnnnn
The starting population size of degenerate att sites is 45 or 1024 molecules. Four different populations were transferred through two BP reactions and two LR reactions. Following transformation of each reaction, the population of transformants was amplified by growth in liquid media containing the appropriate selection antibiotic. DNA was prepared from the population of clones by alkaline lysis miniprep and used in the next reaction. The results of the BP and LR cloning reactions are shown below.
TABLE-US-00024 BP-1, overnight reactions cfu/ml percent of control attB1-LacZa-attB2 78,500 100% attB1n16-20-LacZa-attB2 1,140 1.5% attB1n21-25-LacZa-attB2 11,100 14% attB1-LacZa-attB2n16-20 710 0.9% attB1-LacZa-attB2n21-25 16,600 21%
TABLE-US-00025 LR-1, pENTR201-LacZa x pDEST20/EcoRI, 1 hr reactions cfu/ml percent of control attL1-LacZa-attL2 20,000 100% attL1n16-20-LacZa-attL2 2,125 11% attL1n21-25-LacZa-attL2 2,920 15% attL1-LacZa-attL2n16-20 3,190 16% attL1-LacZa-attL2n21-25 1,405 7%
TABLE-US-00026 BP-2, pEXP20-LacZa/ScaI x pDONR 201, 1 hr reactions cfu/ml percent of control attB1-LacZa-attB2 48,600 100% attB1n16-20-LacZa-attB2 22,800 47% attB1n21-25-LacZa-attB2 31,500 65% attB1-LacZa-attB2n16-20 42,400 87% attB1-LacZa-attB2n21-25 34,500 71%
TABLE-US-00027 LR-2, pENTR201-LacZa x pDEST6/NcoI, 1 hr reactions cfu/ml percent of control attL1-LacZa-attL2 23,000 100% attL1n16-20-LacZa-attL2 49,000 213% attL1n21-25-LacZa-attL2 18,000 80% attL1-LacZa-attL2n16-20 37,000 160% attL1-LacZa-attL2n21-25 57,000 250%
[0675] These results demonstrate that at each successive transfer, the cloning efficiency of the entire population of att sites increases, and that there is a great deal of flexibility in the definition of an attB site. Specific clones may be isolated from the above reactions, tested individually for recombination efficiency, and sequenced. Such new specificities may then be compared to known examples to guide the design of new sequences with new recombination specificities. In addition, based on the enrichment and screening protocols described herein, one of ordinary skill can easily identify and use sequences in other recombination sites, e.g., other att sites, lox, FRT, etc., that result in increased specificity in the recombination reactions using nucleic acid molecules containing such sequences.
Example 25
Design of att Site PCR Adapter-Primers
[0676] Additional studies were performed to design gene-specific primers with 12 bp of attB1 and attB2 at their 5'-ends. The optimal primer design for att-containing primers is the same as for any PCR primers: the gene-specific portion of the primers should ideally have a Tm of >50° C. at 50 mM salt (calculation of Tm is based on the formula 59.9+41(% GC)-675/n).
TABLE-US-00028 Primers: 12 bp attB1 (SEQ ID NO: 114): AA AAA GCA GGC TNN-forward gene-specific primer 12 bp attB2 (SEQ ID NO: 115): A GAA AGC TGG GTN-reverse gene-specific primer attB1 adapter primer (SEQ ID NO: 116): GGGGACAAGTTTGTACAAAAAA-GCAGGCT attB2 adapter primer (SEQ ID NO: 117): GGGGACCACTTTGTACAAGAAA-GCTGGGT
Protocol:
[0677] (1) Mix 200 ng of cDNA library or 1 ng of plasmid clone DNA (alternatively, genomic DNA or RNA could be used) with 10 pmoles of gene specific primers in a 50 μl PCR reaction, using one or more polypeptides having DNA polymerase activity such as those described herein. (The addition of greater than 10 pmoles of gene-specific primers can decrease the yield of attB PCR product. In addition, if RNA is used, a standard reverse transcriptase-PCR (RT-PCR) protocol should be followed; see, e.g., Gerard, G. F., et al., FOCUS 11:60 (1989); Myers, T. W., and Gelfand, D. H., Biochem. 30:7661 (1991); Freeman, W. N., et al., BioTechniques 20:782 (1996); and U.S. application Ser. No. 09/064,057, filed Apr. 22, 1998, the disclosures of all of which are incorporated herein by reference.)
1st PCR Profile:
[0678] (a) 95° C. for 3 minutes (b) 10 cycles of:
[0679] (i) 94° C. for 15 seconds
[0680] (ii) 50° C.* for 30 seconds *The optimal annealing temperature is determined by the calculated Tm of the gene-specific part of the primer.
[0681] (iii) 68° C. for 1 minute/kb of target amplicon
(c) 68° C. for 5 minutes (d) 10° C. hold
[0682] (2) Transfer 10 μl to a 40 μl PCR reaction mix containing 35 pmoles each of the attB1 and attB2 adapter primers.
PCR Profile:
[0683] (a) 95° C. for 1 minute (b) 5 cycles of:
[0684] (i) 94° C. for 15 seconds
[0685] (ii) 45° C.* for 30 seconds *The optimal annealing temperature is determined by the calculated Tm of the gene-specific part of the primer.
[0686] (iii) 68° C. for 1 minute/kb of target amplicon
(c) 15-20 cycles** of: ** 15 cycles is sufficient for low complexity targets.
[0687] (i) 94° C. for 15 seconds
[0688] (ii) 55° C.* for 30 seconds *The optimal annealing temperature is determined by the calculated Tm of the gene-specific part of the primer.
[0689] (iii) 68° C. for 1 minute/kb of target amplicon
(d) 68° C. for 5 minutes (e) 10° C. hold
Notes:
[0690] 1. It is useful to perform a no-adapter primer control to assess the yield of attB PCR product produced.
[0691] 2. Linearized template usually results in slightly greater yield of PCR product.
Example 26
One-Tube Recombinational Cloning Using the GATEWAY® Cloning System
[0692] To provide for easier and more rapid cloning using the GATEWAY® cloning system, we have designed a protocol whereby the BP and LR reactions may be performed in a single tube (a "one-tube" protocol). The following is an example of such a one-tube protocol; in this example, an aliquot of the BP reaction is taken before adding the LR components, but the BP and LR reactions may be performed in a one-tube protocol without first taking the BP aliquot:
TABLE-US-00029 Reaction Component Volume attB DNA (100-200 ng/25 μl reaction) 1-12.5 μl .sup. attP DNA (pDONR201) 150 ng/μl 2.5 μl 5X BP Reaction Buffer 5.0 μl Tris-EDTA (to 20 μl).sup. BP Clonase 5.0 μl Total vol. 25 μl
[0693] After the above components were mixed in a single tube, the reaction mixtures were incubated for 4 hours at 25° C. A 5 μl aliquot of reaction mixture was removed, and 0.5 μl of 10× stop solution was added to this reaction mixture and incubated for 10 minutes at 37° C. Competent cells were then transformed with 1-2 μl of the BP reaction per 100 μl of cells; this transformation yielded colonies of Entry Clones for isolation of individual Entry Clones and for quantitation of the BP Reaction efficiency.
[0694] To the remaining 20 μl of BP reaction mixture, the following components of the LR reaction were added:
TABLE-US-00030 Reaction Component Final Concentration Volume Added NaCl 0.75M 1 μl Destination Vector 150 ng/ul 3 μl LR Clonase 6 μl Total vol. 30 μl
[0695] After the above components were mixed in a single tube, the reaction mixtures were incubated for 2 hours at 25° C. 3 μl of 10× stop solution was added, and the mixture was incubated for 10 minutes at 37° C. Competent cells were then transformed with 1-2 μl of the reaction mixture per 100 μl of cells
Notes:
[0696] 1. If desired, the Destination Vector can be added to the initial BP reaction.
[0697] 2. The reactions can be scaled down by 2×, if desired.
[0698] 3. Shorter incubation times for the BP and/or LR reactions can be used (scaled to the desired cloning efficiencies of the reaction), but a lower number of colonies will typically result.
[0699] 4. To increase the number of colonies obtained by several fold, incubate the BP reaction for 6-20 hours and increase the LR reaction to 3 hours. Electroporation also works well with 1-2 ul of the PK-treated reaction mixture.
[0700] 5. PCR products greater than about 5 kb may show significantly lower cloning efficiency in the BP reaction. In this case, we recommend using a one-tube reaction with longer incubation times (e.g., 6-18 hours) for both the BP and LR steps.
Example 27
Relaxation of Destination Vectors During the LR Reaction
[0701] To further optimize the LR Reaction, the composition of the LR Reaction buffer was modified from that described above and this modified buffer was used in a protocol to examine the impact of enzymatic relaxation of Destination Vectors during the LR Reaction.
[0702] LR Reactions were set up as usual (see, e.g., Example 6), except that 5×BP Reaction Buffer (see Example 5) was used for the LR Reaction. To accomplish Destination Vector relaxation during the LR Reaction, Topoisomerase I (Life Technologies, Inc., Rockville, Md.; Catalogue No. 38042-016) was added to the reaction mixture at a final concentration of ˜15U per μg of total DNA in the reaction (for example, for reaction mixtures with a total of 400 ng DNA in the 20 μl LR Reaction, ˜6 units of Topoisomerase I was added). Reaction mixtures were set up as follows:
TABLE-US-00031 Reaction Component Volume ddH2O 6.5 μl.sup. 4X BP Reaction Buffer 5 μl 100 ng single chain/linear pENTR CAT, 50 ng/μl 2 μl 300 ng single chain/linear pDEST6, 150 ng/μl 2 μl Topoisomerase I, 15 U/ml 0.5 μl.sup. LR Clonase 4 μl
[0703] Reaction mixtures were incubated at 25° C. for 1 hour, and 2 μl of 2 μg/μl Proteinase K was then added and mixtures incubated for 10 minutes at 37° C. to stop the LR Reaction. Competent cells were then transformed as described in the preceding examples. The results of these studies demonstrated that relaxation of substrates in the LR reaction using Topoisomerase I resulted in a 2- to 10-fold increase in colony output compared to those LR reactions performed without including Topoisomerase I.
[0704] Having now fully described the present invention in some detail by way of illustration and example for purposes of clarity of understanding, it will be obvious to one of ordinary skill in the art that the same can be performed by modifying or changing the invention within a wide and equivalent range of conditions, formulations and other parameters without affecting the scope of the invention or any specific embodiment thereof, and that such modifications or changes are intended to be encompassed within the scope of the appended claims.
[0705] All publications, patents and patent applications mentioned in this specification are indicative of the level of skill of those skilled in the art to which this invention pertains, and are herein incorporated by reference to the same extent as if each individual publication, patent or patent application was specifically and individually indicated to be incorporated by reference.
Sequence CWU
1
1
303125DNAArtificial SequenceDescription of Artificial Sequence Synthetic
attB1 site oligonucleotide 1acaagtttgt acaaaaaagc aggct
25225DNAArtificial SequenceDescription of
Artificial Sequence Synthetic attB2 site oligonucleotide 2acccagcttt
cttgtacaaa gtggt
253233DNAArtificial SequenceDescription of Artificial Sequence Synthetic
attP1 site polynucleotide 3tacaggtcac taataccatc taagtagttg
attcatagtg actggatatg ttgtgtttta 60cagtattatg tagtctgttt tttatgcaaa
atctaattta atatattgat atttatatca 120ttttacgttt ctcgttcagc ttttttgtac
aaagttggca ttataaaaaa gcattgctca 180tcaatttgtt gcaacgaaca ggtcactatc
agtcaaaata aaatcattat ttg 2334233DNAArtificial
SequenceDescription of Artificial Sequence Synthetic attP2
polynucleotide 4caaataatga ttttattttg actgatagtg acctgttcgt tgcaacaaat
tgataagcaa 60tgctttctta taatgccaac tttgtacaag aaagctgaac gagaaacgta
aaatgatata 120aatatcaata tattaaatta gattttgcat aaaaaacaga ctacataata
ctgtaaaaca 180caacatatcc agtcactatg aatcaactac ttagatggta ttagtgacct
gta 2335125DNAArtificial SequenceDescription of Artificial
Sequence Synthetic attL1 polynucleotide 5acaagtttgt acaaaaaagc
tgaacgagaa acgtaaaatg atataaatat caatatatta 60aattagattt tgcataaaaa
acagactaca taatactgta aaacacaaca tatccagtca 120ctatg
1256135DNAArtificial
SequenceDescription of Artificial Sequence Synthetic attL2
polynucleotide 6gcaggtcgac catagtgact ggatatgttg tgttttacag tattatgtag
tctgtttttt 60atgcaaaatc taatttaata tattgatatt tatatcattt tacgtttctc
gttcagcttt 120cttgtacaaa gtggt
1357100DNAArtificial SequenceDescription of Artificial
Sequence Synthetic attR1 polynucleotide 7caaataatga ttttattttg
actgatagtg acctgttcgt tgcaacaaat tgataagcaa 60tgctttttta taatgccaac
tttgtacaaa aaagcaggct 1008100DNAArtificial
SequenceDescription of Artificial Sequence Synthetic attR2
polynucleotide 8caaataatga ttttattttg actgatagtg acctgttcgt tgcaacaaat
tgataagcaa 60tgctttctta taatgccaac tttgtacaag aaagctgggt
100915DNAArtificial SequenceDescription of Artificial
Sequence Synthetic 15 bp core region of attB, attP, attL and attR
oligonucleotide 9gcttttttat actaa
151030DNAArtificial SequenceDescription of Artificial
Sequence Synthetic attL5 oligonucleotide 10agcctgcttt attatactaa
gttggcatta 301130DNAArtificial
SequenceDescription of Artificial Sequence Synthetic attL6
oligonucleotide 11agcctgcttt tttatattaa gttggcatta
301229DNAArtificial SequenceDescription of Artificial
Sequence Synthetic attB1.6 oligonucleotide 12ggggacaact ttgtacaaaa
aagttggct 291329DNAArtificial
SequenceDescription of Artificial Sequence Synthetic attB2.2
oligonucleotide 13ggggacaact ttgtacaaga aagctgggt
291429DNAArtificial SequenceDescription of Artificial
Sequence Synthetic attB2.10 oligonucleotide 14ggggacaact ttgtacaaga
aagttgggt 291538DNAArtificial
SequenceDescription of Artificial Sequence Synthetic attB2(-1)
primer 15cccagctttc ttgtacaaag tggtnnnnnn nnnnnnnn
381638DNAArtificial SequenceDescription of Artificial Sequence
Synthetic attB2(-2) primer 16ccagctttct tgtacaaagt ggtnnnnnnn
nnnnnnnn 381738DNAArtificial
SequenceDescription of Artificial Sequence Synthetic attB2(-3)
primer 17cagctttctt gtacaaagtg gtnnnnnnnn nnnnnnnn
381838DNAArtificial SequenceDescription of Artificial Sequence
Synthetic attB2(-4) primer 18agctttcttg tacaaagtgg tnnnnnnnnn
nnnnnnnn 381939DNAArtificial
SequenceDescription of Artificial Sequence Synthetic attB1- and
attB2-derived primer 19acaagtttgt acaaaaaagc aggctnnnnn nnnnnnnnn
392039DNAArtificial SequenceDescription of Artificial
Sequence Synthetic attB1- and attB2-derived primer 20accactttgt
acaagaaagc tgggtnnnnn nnnnnnnnn
392132DNAArtificial SequenceDescription of Artificial Sequence Synthetic
attB1- and attB2-derived primer 21tgtacaaaaa agcaggctnn nnnnnnnnnn nn
322232DNAArtificial SequenceDescription
of Artificial Sequence Synthetic attB1- and attB2-derived primer
22tgtacaagaa agctgggtnn nnnnnnnnnn nn
322329DNAArtificial SequenceDescription of Artificial Sequence Synthetic
attB1- and attB2-derived primer 23acaaaaaagc aggctnnnnn nnnnnnnnn
292429DNAArtificial SequenceDescription
of Artificial Sequence Synthetic attB1- and attB2-derived primer
24acaagaaagc tgggtnnnnn nnnnnnnnn
292526DNAArtificial SequenceDescription of Artificial Sequence Synthetic
attB1- and attB2-derived primer 25aaaaagcagg ctnnnnnnnn nnnnnn
262626DNAArtificial SequenceDescription
of Artificial Sequence Synthetic attB1- and attB2-derived primer
26agaaagctgg gtnnnnnnnn nnnnnn
262725DNAArtificial SequenceDescription of Artificial Sequence Synthetic
attB1- and attB2-derived primer 27aaaagcaggc tnnnnnnnnn nnnnn
252825DNAArtificial SequenceDescription
of Artificial Sequence Synthetic attB1- and attB2-derived primer
28gaaagctggg tnnnnnnnnn nnnnn
252924DNAArtificial SequenceDescription of Artificial Sequence Synthetic
attB1- and attB2-derived primer 29aaagcaggct nnnnnnnnnn nnnn
243024DNAArtificial SequenceDescription
of Artificial Sequence Synthetic attB1- and attB2-derived primer
30aaagctgggt nnnnnnnnnn nnnn
243129DNAArtificial SequenceDescription of Artificial Sequence Synthetic
attB1 primer 31ggggacaagt ttgtacaaaa aagcaggct
293229DNAArtificial SequenceDescription of Artificial
Sequence Synthetic attB2 primer 32ggggaccact ttgtacaaga aagctgggt
293327DNAArtificial
SequenceDescription of Artificial Sequence Synthetic XhoI Insertion
primer 33atgnnnnnnn nntaactcga gnnnnnn
273430PRTArtificial SequenceDescription of Artificial Sequence
Synthetic attB1 fused into a His6 fusion vector polypeptide 34Met
Ser Tyr Tyr His His His His His His Gly Ile Thr Ser Leu Tyr 1
5 10 15 Lys Lys Ala Gly Phe Glu
Asn Leu Tyr Phe Gln Gly Thr Met 20 25
30 3511PRTArtificial SequenceDescription of Artificial
Sequence Synthetic attB Amino Acid Sequence peptide 35Gly Ile Thr
Ser Leu Tyr Lys Lys Ala Gly Phe 1 5 10
3648DNAArtificial SequenceDescription of Artificial Sequence Synthetic
attL1 PCR primer 36ggggagcctg cttttttgta caaagttggc attataaaaa
agcattgc 483748DNAArtificial SequenceDescription of
Artificial Sequence Synthetic attL2 PCR primer 37ggggagcctg
ctttcttgta caaagttggc attataaaaa agcattgc
483822DNAArtificial SequenceDescription of Artificial Sequence Synthetic
attL Right PCR primer 38tgttgccggg aagctagagt aa
223943DNAArtificial SequenceDescription of
Artificial Sequence Synthetic attR1 PCR primer 39ggggacaagt
ttgtacaaaa aagctgaacg agaaacgtaa aat
434043DNAArtificial SequenceDescription of Artificial Sequence Synthetic
attR2 oligonucleotide 40ggggacaagt ttgtacaaga aagctgaacg agaaacgtaa
aat 434122DNAArtificial SequenceDescription of
Artificial Sequence Synthetic attR Right oligonucleotide
41cagacggcat gatgaacctg aa
224229DNAArtificial SequenceDescription of Artificial Sequence Synthetic
B1-Hgb oligonucleotide 42ggggacaagt ttgtacaaaa aagcaggct
294329DNAArtificial SequenceDescription of
Artificial Sequence Synthetic B2-Hgb oligonucleotide 43ggggaccact
ttgtacaaga aagctgggt
294418DNAArtificial SequenceDescription of Artificial Sequence Synthetic
18B1-Hgb oligonucleotide 44tgtacaaaaa agcaggct
184518DNAArtificial SequenceDescription of
Artificial Sequence Synthetic 18B2-Hgb oligonucleotide 45tgtacaagaa
agctgggt
184615DNAArtificial SequenceDescription of Artificial Sequence Synthetic
15B1-Hgb oligonucleotide 46acaaaaaagc aggct
154715DNAArtificial SequenceDescription of
Artificial Sequence Synthetic 15B2-Hgb oligonucleotide 47acaagaaagc
tgggt
154812DNAArtificial SequenceDescription of Artificial Sequence Synthetic
12B1-Hgb oligonucleotide 48aaaaagcagg ct
124912DNAArtificial SequenceDescription of
Artificial Sequence Synthetic 12B2-Hgb oligonucleotide 49agaaagctgg
gt
125011DNAArtificial SequenceDescription of Artificial Sequence Synthetic
11B1-Hgb oligonucleotide 50aaaagcaggc t
115111DNAArtificial SequenceDescription of
Artificial Sequence Synthetic 11B2-Hgb oligonucleotide 51gaaagctggg
t
115210DNAArtificial SequenceDescription of Artificial Sequence Synthetic
10B1-Hgb oligonucleotide 52aaagcaggct
105310DNAArtificial SequenceDescription of
Artificial Sequence Synthetic 10B2-Hgb oligonucleotide 53aaagctgggt
105429DNAArtificial SequenceDescription of Artificial Sequence Synthetic
attB1 adapter oligonucleotide 54ggggacaagt ttgtacaaaa aagcaggct
295529DNAArtificial SequenceDescription
of Artificial Sequence Synthetic attB2 adapter oligonucleotide
55ggggaccact ttgtacaaga aagctgggt
295622DNAArtificial SequenceDescription of Artificial Sequence Synthetic
-5' -Hgb oligonucleotide 56gtcactagcc tgtggagcaa ga
225722DNAArtificial SequenceDescription of
Artificial Sequence Synthetic -3' -Hgb oligonucleotide 57aggatggcag
agggagacga ca
225815DNAArtificial SequenceDescription of Artificial Sequence Synthetic
15 bp Core Region of attB, attP, attL and attR oligonucleotide
58gcttttttat actaa
155948DNAArtificial SequenceDescription of Artificial Sequence Synthetic
attL0 PCR primer 59ggggagcctg cttttttata ctaagttggc attataaaaa
agcattgc 486048DNAArtificial SequenceDescription of
Artificial Sequence Synthetic attLT1A PCR primer 60ggggagcctg
ctttattata ctaagttggc attataaaaa agcattgc
486148DNAArtificial SequenceDescription of Artificial Sequence Synthetic
attLT1C PCR primer 61ggggagcctg ctttcttata ctaagttggc attataaaaa
agcattgc 486248DNAArtificial SequenceDescription of
Artificial Sequence Synthetic attLT1G PCR primer 62ggggagcctg
ctttgttata ctaagttggc attataaaaa agcattgc
486348DNAArtificial SequenceDescription of Artificial Sequence Synthetic
attLT2A PCR primer 63ggggagcctg cttttatata ctaagttggc attataaaaa
agcattgc 486448DNAArtificial SequenceDescription of
Artificial Sequence Synthetic attLT2C PCR primer 64ggggagcctg
cttttctata ctaagttggc attataaaaa agcattgc
486548DNAArtificial SequenceDescription of Artificial Sequence Synthetic
attLT2G PCR primer 65ggggagcctg cttttgtata ctaagttggc attataaaaa
agcattgc 486648DNAArtificial SequenceDescription of
Artificial Sequence Synthetic attLT3A PCR primer 66ggggagcctg
ctttttaata ctaagttggc attataaaaa agcattgc
486748DNAArtificial SequenceDescription of Artificial Sequence Synthetic
attLT3C PCR primer 67ggggagcctg ctttttcata ctaagttggc attataaaaa
agcattgc 486848DNAArtificial SequenceDescription of
Artificial Sequence Synthetic attLT3G PCR primer 68ggggagcctg
ctttttgata ctaagttggc attataaaaa agcattgc
486948DNAArtificial SequenceDescription of Artificial Sequence Synthetic
attLA4C PCR primer 69ggggagcctg cttttttcta ctaagttggc attataaaaa
agcattgc 487048DNAArtificial SequenceDescription of
Artificial Sequence Synthetic attLA4G PCR primer 70ggggagcctg
cttttttgta ctaagttggc attataaaaa agcattgc
487148DNAArtificial SequenceDescription of Artificial Sequence Synthetic
attLA4T PCR primer 71ggggagcctg ctttttttta ctaagttggc attataaaaa
agcattgc 487248DNAArtificial SequenceDescription of
Artificial Sequence Synthetic attLT5A PCR primer 72ggggagcctg
cttttttaaa ctaagttggc attataaaaa agcattgc
487348DNAArtificial SequenceDescription of Artificial Sequence Synthetic
attLT5C PCR primer 73ggggagcctg cttttttaca ctaagttggc attataaaaa
agcattgc 487448DNAArtificial SequenceDescription of
Artificial Sequence Synthetic attLT5G PCR primer 74ggggagcctg
cttttttaga ctaagttggc attataaaaa agcattgc
487548DNAArtificial SequenceDescription of Artificial Sequence Synthetic
attLA6C PCR primer 75ggggagcctg cttttttatc ctaagttggc attataaaaa
agcattgc 487648DNAArtificial SequenceDescription of
Artificial Sequence Synthetic attLA6G PCR primer 76ggggagcctg
cttttttatg ctaagttggc attataaaaa agcattgc
487748DNAArtificial SequenceDescription of Artificial Sequence Synthetic
attLA6T PCR primer 77ggggagcctg cttttttatt ctaagttggc attataaaaa
agcattgc 487848DNAArtificial SequenceDescription of
Artificial Sequence Synthetic attLC7A PCR primer 78ggggagcctg
cttttttata ataagttggc attataaaaa agcattgc
487948DNAArtificial SequenceDescription of Artificial Sequence Synthetic
attLC7G PCR primer 79ggggagcctg cttttttata gtaagttggc attataaaaa
agcattgc 488048DNAArtificial SequenceDescription of
Artificial Sequence Synthetic attLC7T PCR primer 80ggggagcctg
cttttttata ttaagttggc attataaaaa agcattgc
488148DNAArtificial SequenceDescription of Artificial Sequence Synthetic
attL8 oligonucleotide 81ggggagccta cttttttata ctaagttggc attataaaaa
agcattgc 488248DNAArtificial SequenceDescription of
Artificial Sequence Synthetic attL9 oligonucleotide 82ggggagcctg
cctttttata ctaagttggc attataaaaa agcattgc
488348DNAArtificial SequenceDescription of Artificial Sequence Synthetic
attL10 oligonucleotide 83ggggagcctg cttctttata ctaagttggc attataaaaa
agcattgc 488448DNAArtificial SequenceDescription of
Artificial Sequence Synthetic attL14 oligonucleotide 84ggggagcctg
cttttttata ccaagttggc attataaaaa agcattgc
488548DNAArtificial SequenceDescription of Artificial Sequence Synthetic
attL15 oligonucleotide 85ggggagcctg cttttttata ctaggttggc attataaaaa
agcattgc 488630DNAArtificial SequenceDescription of
Artificial Sequence Synthetic attL0 oligonucleotide 86agcctgcttt
tttatactaa gttggcatta
308730DNAArtificial SequenceDescription of Artificial Sequence Synthetic
attL5 oligonucleotide 87agcctgcttt attatactaa gttggcatta
308830DNAArtificial SequenceDescription of
Artificial Sequence Synthetic attL6 oligonucleotide 88agcctgcttt
tttatattaa gttggcatta
308930DNAArtificial SequenceDescription of Artificial Sequence Synthetic
attL13 oligonucleotide 89agcctgcttt tttatgctaa gttggcatta
309030DNAArtificial SequenceDescription of
Artificial Sequence Synthetic attL14 oligonucleotide 90agcctgcttt
tttataccaa gttggcatta
309130DNAArtificial SequenceDescription of Artificial Sequence Synthetic
attL15 oligonucleotide 91agcctgcttt tttatactag gttggcatta
309221DNAArtificial SequenceDescription of
Artificial Sequence Synthetic consensus sequence for integrase
core-binding 92caacttnntn nnannaagtt g
219325DNAArtificial SequenceDescription of Artificial Sequence
Synthetic attB0 oligonucleotide 93tcaagttagt ataaaaaagc aggct
259429DNAArtificial
SequenceDescription of Artificial Sequence Synthetic attB1
oligonucleotide 94ggggacaagt ttgtacaaaa aagcaggct
299529DNAArtificial SequenceDescription of Artificial
Sequence Synthetic attB2 oligonucleotide 95ggggaccact ttgtacaaga
aagctgggt 299629DNAArtificial
SequenceDescription of Artificial Sequence Synthetic attB2.1
oligonucleotide 96ggggaacact ttgtacaaga aagctgggt
299729DNAArtificial SequenceDescription of Artificial
Sequence Synthetic attB2.2 oligonucleotide 97ggggacaact ttgtacaaga
aagctgggt 299829DNAArtificial
SequenceDescription of Artificial Sequence Synthetic attB2.3
oligonucleotide 98ggggacccct ttgtacaaga aagctgggt
299929DNAArtificial SequenceDescription of Artificial
Sequence Synthetic attB2.4 oligonucleotide 99ggggaccaat ttgtacaaga
aagctgggt 2910029DNAArtificial
SequenceDescription of Artificial Sequence Synthetic attB2.5
oligonucleotide 100ggggaccacg ttgtacaaga aagctgggt
2910129DNAArtificial SequenceDescription of Artificial
Sequence Synthetic attB2.6 oligonucleotide 101ggggaccact gtgtacaaga
aagctgggt 2910229DNAArtificial
SequenceDescription of Artificial Sequence Synthetic attB2.7
oligonucleotide 102ggggaccact tggtacaaga aagctgggt
2910329DNAArtificial SequenceDescription of Artificial
Sequence Synthetic attB2.8 oligonucleotide 103ggggaccact ttttacaaga
aagctgggt 2910429DNAArtificial
SequenceDescription of Artificial Sequence Synthetic attB1
Amplification Site oligonucleotide 104ggggacaagt ttgtacaaaa aagcaggct
2910529DNAArtificial
SequenceDescription of Artificial Sequence Synthetic attB1.6
Amplification Site oligonucleotide 105ggggacaact ttgtacaaaa aagttggct
2910629DNAArtificial
SequenceDescription of Artificial Sequence Synthetic attB2
Amplification Site oligonucleotide 106ggggaccact ttgtacaaga aagctgggt
2910729DNAArtificial
SequenceDescription of Artificial Sequence Synthetic attB2.10
Amplification Site oligonucleotide 107ggggacaact ttgtacaaga aagttgggt
2910829DNAArtificial
SequenceDescription of Artificial Sequence Synthetic attB1 PCR
primer 108ggggacaagt ttgtacaaaa aagcaggct
2910929DNAArtificial SequenceDescription of Artificial Sequence
Synthetic attB1n16-20 PCR primer 109ggggacaagt ttgtacaaan nnnnaggct
2911029DNAArtificial
SequenceDescription of Artificial Sequence Synthetic attB1n21-25 PCR
primer 110ggggacaagt ttgtacaaaa aagcnnnnn
2911129DNAArtificial SequenceDescription of Artificial Sequence
Synthetic attB2 PCR primer 111ggggaccact ttgtacaaga aagctgggt
2911229DNAArtificial SequenceDescription
of Artificial Sequence Synthetic attB2n16-20 PCR primer
112ggggaccact ttgtacaagn nnnntgggt
2911329DNAArtificial SequenceDescription of Artificial Sequence Synthetic
attB2n21-25 PCR primer 113ggggaccact ttgtacaaga aagcnnnnn
2911414DNAArtificial SequenceDescription of
Artificial Sequence Synthetic 12bp attB1 forward gene-specific
primer 114aaaaagcagg ctnn
1411513DNAArtificial SequenceDescription of Artificial Sequence
Synthetic 12bp attB2 reverse gene-specific primer 115agaaagctgg gtn
1311629DNAArtificial SequenceDescription of Artificial Sequence Synthetic
attB1 adapter primer 116ggggacaagt ttgtacaaaa aagcaggct
2911729DNAArtificial SequenceDescription of
Artificial Sequence Synthetic attB2 adapter primer 117ggggaccact
ttgtacaaga aagctgggt
291182717DNAArtificial SequenceDescription of Artificial Sequence
Synthetic Entry Vector pENTR1A polynucleotide 118ctgacggatg
gcctttttgc gtttctacaa actcttcctg ttagttagtt acttaagctc 60gggccccaaa
taatgatttt attttgactg atagtgacct gttcgttgca acaaattgat 120aagcaatgct
tttttataat gccaactttg tacaaaaaag caggctttaa aggaaccaat 180tcagtcgact
ggatccggta ccgaattcgc ttactaaaag ccagataaca gtatgcgtat 240ttgcgcgctg
atttttgcgg tataagaata tatactgata tgtatacccg aagtatgtca 300aaaagaggtg
tgcttctaga atgcagttta aggtttacac ctataaaaga gagagccgtt 360atcgtctgtt
tgtggatgta cagagtgata ttattgacac gcccgggcga cggatagtga 420tccccctggc
cagtgcacgt ctgctgtcag ataaagtctc ccgtgaactt tacccggtgg 480tgcatatcgg
ggatgaaagc tggcgcatga tgaccaccga tatggccagt gtgccggtct 540ccgttatcgg
ggaagaagtg gctgatctca gccaccgcga aaatgacatc aaaaacgcca 600ttaacctgat
gttctgggga atatagaatt cgcggccgca ctcgagatat ctagacccag 660ctttcttgta
caaagttggc attataagaa agcattgctt atcaatttgt tgcaacgaac 720aggtcactat
cagtcaaaat aaaatcatta tttgccatcc agctgcagct ctggcccgtg 780tctcaaaatc
tctgatgtta cattgcacaa gataaaaata tatcatcatg aacaataaaa 840ctgtctgctt
acataaacag taatacaagg ggtgttatga gccatattca acgggaaacg 900tcgaggccgc
gattaaattc caacatggat gctgatttat atgggtataa atgggctcgc 960gataatgtcg
ggcaatcagg tgcgacaatc tatcgcttgt atgggaagcc cgatgcgcca 1020gagttgtttc
tgaaacatgg caaaggtagc gttgccaatg atgttacaga tgagatggtc 1080agactaaact
ggctgacgga atttatgcct cttccgacca tcaagcattt tatccgtact 1140cctgatgatg
catggttact caccactgcg atccccggaa aaacagcatt ccaggtatta 1200gaagaatatc
ctgattcagg tgaaaatatt gttgatgcgc tggcagtgtc cctgcgccgg 1260ttgcattcga
ttcctgtttg taattgtcct tttaacagcg atcgcgtatt tcgtctcgct 1320caggcgcaat
cacgaatgaa taacggtttg gttgatgcga gtgattttga tgacgagcgt 1380aatggctggc
ctgttgaaca agtctggaaa gaaatgcata aacttttgcc attctcaccg 1440gattcagtcg
tcactcatgg tgatttctca cttgataacc ttatttttga cgaggggaaa 1500ttaataggtt
gtattgatgt tggacgagtc ggaatcgcag accgatacca ggatcttgcc 1560atcctatgga
actgcctcgg tgagttttct ccttcattac agaaacggct ttttcaaaaa 1620tatggtattg
ataatcctga tatgaataaa ttgcagtttc atttgatgct cgatgagttt 1680ttctaatcag
aattggttaa ttggttgtaa cattattcag attgggcccc gttccactga 1740gcgtcagacc
ccgtagaaaa gatcaaagga tcttcttgag atcctttttt tctgcgcgta 1800atctgctgct
tgcaaacaaa aaaaccaccg ctaccagcgg tggtttgttt gccggatcaa 1860gagctaccaa
ctctttttcc gaaggtaact ggcttcagca gagcgcagat accaaatact 1920gttcttctag
tgtagccgta gttaggccac cacttcaaga actctgtagc accgcctaca 1980tacctcgctc
tgctaatcct gttaccagtg gctgctgcca gtggcgataa gtcgtgtctt 2040accgggttgg
actcaagacg atagttaccg gataaggcgc agcggtcggg ctgaacgggg 2100ggttcgtgca
cacagcccag cttggagcga acgacctaca ccgaactgag atacctacag 2160cgtgagctat
gagaaagcgc cacgcttccc gaagggagaa aggcggacag gtatccggta 2220agcggcaggg
tcggaacagg agagcgcacg agggagcttc cagggggaaa cgcctggtat 2280ctttatagtc
ctgtcgggtt tcgccacctc tgacttgagc gtcgattttt gtgatgctcg 2340tcaggggggc
ggagcctatg gaaaaacgcc agcaacgcgg cctttttacg gttcctggcc 2400ttttgctggc
cttttgctca catgttcttt cctgcgttat cccctgattc tgtggataac 2460cgtattaccg
ctagcatgga tctcggggac gtctaactac taagcgagag tagggaactg 2520ccaggcatca
aataaaacga aaggctcagt cggaagactg ggcctttcgt tttatctgtt 2580gtttgtcggt
gaacgctctc ctgagtagga caaatccgcc gggagcggat ttgaacgttg 2640tgaagcaacg
gcccggaggg tggcgggcag gacgcccgcc ataaactgcc aggcatcaaa 2700ctaagcagaa
ggccatc
27171192718DNAArtificial SequenceDescription of Artificial Sequence
Synthetic Entry Vector pENTR2B polynucleotide 119ctgacggatg
gcctttttgc gtttctacaa actcttcctg ttagttagtt acttaagctc 60gggccccaaa
taatgatttt attttgactg atagtgacct gttcgttgca acaaattgat 120aagcaatgct
tttttataat gccaactttg tacaaaaaag caggctggcg ccggaaccaa 180ttcagtcgac
tggatccggt accgaattcg cttactaaaa gccagataac agtatgcgta 240tttgcgcgct
gatttttgcg gtataagaat atatactgat atgtataccc gaagtatgtc 300aaaaagaggt
gtgcttctag aatgcagttt aaggtttaca cctataaaag agagagccgt 360tatcgtctgt
ttgtggatgt acagagtgat attattgaca cgcccgggcg acggatggtg 420atccccctgg
ccagtgcacg tctgctgtca gataaagtct cccgtgaact ttacccggtg 480gtgcatatcg
gggatgaaag ctggcgcatg atgaccaccg atatggccag tgtgccggtc 540tccgttatcg
gggaagaagt ggctgatctc agccaccgcg aaaatgacat caaaaacgcc 600attaacctga
tgttctgggg aatatagaat tcgcggccgc actcgagata tctagaccca 660gctttcttgt
acaaagttgg cattataaga aagcattgct tatcaatttg ttgcaacgaa 720caggtcacta
tcagtcaaaa taaaatcatt atttgccatc cagctgcagc tctggcccgt 780gtctcaaaat
ctctgatgtt acattgcaca agataaaaat atatcatcat gaacaataaa 840actgtctgct
tacataaaca gtaatacaag gggtgttatg agccatattc aacgggaaac 900gtcgaggccg
cgattaaatt ccaacatgga tgctgattta tatgggtata aatgggctcg 960cgataatgtc
gggcaatcag gtgcgacaat ctatcgcttg tatgggaagc ccgatgcgcc 1020agagttgttt
ctgaaacatg gcaaaggtag cgttgccaat gatgttacag atgagatggt 1080cagactaaac
tggctgacgg aatttatgcc tcttccgacc atcaagcatt ttatccgtac 1140tcctgatgat
gcatggttac tcaccactgc gatccccgga aaaacagcat tccaggtatt 1200agaagaatat
cctgattcag gtgaaaatat tgttgatgcg ctggcagtgt tcctgcgccg 1260gttgcattcg
attcctgttt gtaattgtcc ttttaacagc gatcgcgtat ttcgtctcgc 1320tcaggcgcaa
tcacgaatga ataacggttt ggttgatgcg agtgattttg atgacgagcg 1380taatggctgg
cctgttgaac aagtctggaa agaaatgcat aaacttttgc cattctcacc 1440ggattcagtc
gtcactcatg gtgatttctc acttgataac cttatttttg acgaggggaa 1500attaataggt
tgtattgatg ttggacgagt cggaatcgca gaccgatacc aggatcttgc 1560catcctatgg
aactgcctcg gtgagttttc tccttcatta cagaaacggc tttttcaaaa 1620atatggtatt
gataatcctg atatgaataa attgcagttt catttgatgc tcgatgagtt 1680tttctaatca
gaattggtta attggttgta acattattca gattgggccc cgttccactg 1740agcgtcagac
cccgtagaaa agatcaaagg atcttcttga gatccttttt ttctgcgcgt 1800aatctgctgc
ttgcaaacaa aaaaaccacc gctaccagcg gtggtttgtt tgccggatca 1860agagctacca
actctttttc cgaaggtaac tggcttcagc agagcgcaga taccaaatac 1920tgttcttcta
gtgtagccgt agttaggcca ccacttcaag aactctgtag caccgcctac 1980atacctcgct
ctgctaatcc tgttaccagt ggctgctgcc agtggcgata agtcgtgtct 2040taccgggttg
gactcaagac gatagttacc ggataaggcg cagcggtcgg gctgaacggg 2100gggttcgtgc
acacagccca gcttggagcg aacgacctac accgaactga gatacctaca 2160gcgtgagcta
tgagaaagcg ccacgcttcc cgaagggaga aaggcggaca ggtatccggt 2220aagcggcagg
gtcggaacag gagagcgcac gagggagctt ccagggggaa acgcctggta 2280tctttatagt
cctgtcgggt ttcgccacct ctgacttgag cgtcgatttt tgtgatgctc 2340gtcagggggg
cggagcctat ggaaaaacgc cagcaacgcg gcctttttac ggttcctggc 2400cttttgctgg
ccttttgctc acatgttctt tcctgcgtta tcccctgatt ctgtggataa 2460ccgtattacc
gctagcatgg atctcgggga cgtctaacta ctaagcgaga gtagggaact 2520gccaggcatc
aaataaaacg aaaggctcag tcggaagact gggcctttcg ttttatctgt 2580tgtttgtcgg
tgaacgctct cctgagtagg acaaatccgc cgggagcgga tttgaacgtt 2640gtgaagcaac
ggcccggagg gtggcgggca ggacgcccgc cataaactgc caggcatcaa 2700actaagcaga
aggccatc
27181202723DNAArtificial SequenceDescription of Artificial Sequence
Synthetic Entry Vector pENTR3C polynucleotide 120ctgacggatg
gcctttttgc gtttctacaa actcttcctg ttagttagtt acttaagctc 60gggccccaaa
taatgatttt attttgactg atagtgacct gttcgttgca acaaattgat 120aagcaatgct
tttttataat gccaactttg tacaaaaaag caggctcttt aaaggaacca 180attcagtcga
ctggatccgg taccgaattc gatcgcttac taaaagccag ataacagtat 240gcgtatttgc
gcgctgattt ttgcggtata agaatatata ctgatatgta tacccgaagt 300atgtcaaaaa
gaggtgtgct tctagaatgc agtttaaggt ttacacctat aaaagagaga 360gccgttatcg
tctgtttgtg gatgtacaga gtgatattat tgacacgccc gggcgacgga 420tggtgatccc
cctggccagt gcacgtctgc tgtcagataa agtctcccgt gaactttacc 480cggtggtgca
tatcggggat gaaagctggc gcatgatgac caccgatatg gccagtgtgc 540cggtctccgt
tatcggggaa gaagtggctg atctcagcca ccgcgaaaat gacatcaaaa 600acgccattaa
cctgatgttc tggggaatat agaattcgcg gccgcactcg agatatctag 660acccagcttt
cttgtacaaa gttggcatta taagaaagca ttgcttatca atttgttgca 720acgaacaggt
cactatcagt caaaataaaa tcattatttg ccatccagct gcagctctgg 780cccgtgtctc
aaaatctctg atgttacatt gcacaagata aaaatatatc atcatgaaca 840ataaaactgt
ctgcttacat aaacagtaat acaaggggtg ttatgagcca tattcaacgg 900gaaacgtcga
ggccgcgatt aaattccaac atggatgctg atttatatgg gtataaatgg 960gctcgcgata
atgtcgggca atcaggtgcg acaatctatc gcttgtatgg gaagcccgat 1020gcgccagagt
tgtttctgaa acatggcaaa ggtagcgttg ccaatgatgt tacagatgag 1080atggtcagac
taaactggct gacggaattt atgcctcttc cgaccatcaa gcattttatc 1140cgtactcctg
atgatgcatg gttactcacc actgcgatcc ccggaaaaac agcattccag 1200gtattagaag
aatatcctga ttcaggtgaa aatattgttg atgcgctggc agtgttcctg 1260cgccggttgc
attcgattcc tgtttgtaat tgtcctttta acagcgatcg cgtatttcgt 1320ctcgctcagg
cgcaatcacg aatgaataac ggtttggttg atgcgagtga ttttgatgac 1380gagcgtaatg
gctggcctgt tgaacaagtc tggaaagaaa tgcataaact tttgccattc 1440tcaccggatt
cagtcgtcac tcatggtgat ttctcacttg ataaccttat ttttgacgag 1500gggaaattaa
taggttgtat tgatgttgga cgagtcggaa tcgcagaccg ataccaggat 1560cttgccatcc
tatggaactg cctcggtgag ttttctcctt cattacagaa acggcttttt 1620caaaaatatg
gtattgataa tcctgatatg aataaattgc agtttcattt gatgctcgat 1680gagtttttct
aatcagaatt ggttaattgg ttgtaacatt attcagattg ggccccgttc 1740cactgagcgt
cagaccccgt agaaaagatc aaaggatctt cttgagatcc tttttttctg 1800cgcgtaatct
gctgcttgca aacaaaaaaa ccaccgctac cagcggtggt ttgtttgccg 1860gatcaagagc
taccaactct ttttccgaag gtaactggct tcagcagagc gcagatacca 1920aatactgttc
ttctagtgta gccgtagtta ggccaccact tcaagaactc tgtagcaccg 1980cctacatacc
tcgctctgct aatcctgtta ccagtggctg ctgccagtgg cgataagtcg 2040tgtcttaccg
ggttggactc aagacgatag ttaccggata aggcgcagcg gtcgggctga 2100acggggggtt
cgtgcacaca gcccagcttg gagcgaacga cctacaccga actgagatac 2160ctacagcgtg
agctatgaga aagcgccacg cttcccgaag ggagaaaggc ggacaggtat 2220ccggtaagcg
gcagggtcgg aacaggagag cgcacgaggg agcttccagg gggaaacgcc 2280tggtatcttt
atagtcctgt cgggtttcgc cacctctgac ttgagcgtcg atttttgtga 2340tgctcgtcag
gggggcggag cctatggaaa aacgccagca acgcggcctt tttacggttc 2400ctggcctttt
gctggccttt tgctcacatg ttctttcctg cgttatcccc tgattctgtg 2460gataaccgta
ttaccgctag catggatctc ggggacgtct aactactaag cgagagtagg 2520gaactgccag
gcatcaaata aaacgaaagg ctcagtcgga agactgggcc tttcgtttta 2580tctgttgttt
gtcggtgaac gctctcctga gtaggacaaa tccgccggga gcggatttga 2640acgttgtgaa
gcaacggccc ggagggtggc gggcaggacg cccgccataa actgccaggc 2700atcaaactaa
gcagaaggcc atc
27231212720DNAArtificial SequenceDescription of Artificial Sequence
Synthetic Entry Vector pENTR4 polynucleotide 121ctgacggatg
gcctttttgc gtttctacaa actcttcctg ttagttagtt acttaagctc 60gggccccaaa
taatgatttt attttgactg atagtgacct gttcgttgca acaaattgat 120aagcaatgct
tttttataat gccaactttg tacaaaaaag caggctccac catgggaacc 180aattcagtcg
actggatccg gtaccgaatt cgcttactaa aagccagata acagtatgcg 240tatttgcgcg
ctgatttttg cggtataaga atatatactg atatgtatac ccgaagtatg 300tcaaaaagag
gtgtgcttct agaatgcagt ttaaggttta cacctataaa agagagagcc 360gttatcgtct
gtttgtggat gtacagagtg atattattga cacgcccggg cgacggatgg 420tgatccccct
ggccagtgca cgtctgctgt cagataaagt ctcccgtgaa ctttacccgg 480tggtgcatat
cggggatgaa agctggcgca tgatgaccac cgatatggcc agtgtgccgg 540tctccgttat
cggggaagaa gtggctgatc tcagccaccg cgaaaatgac atcaaaaacg 600ccattaacct
gatgttctgg ggaatataga attcgcggcc gcactcgaga tatctagacc 660cagctttctt
gtacaaagtt ggcattataa gaaagcattg cttatcaatt tgttgcaacg 720aacaggtcac
tatcagtcaa aataaaatca ttatttgcca tccagctgca gctctggccc 780gtgtctcaaa
atctctgatg ttacattgca caagataaaa atatatcatc atgaacaata 840aaactgtctg
cttacataaa cagtaataca aggggtgtta tgagccatat tcaacgggaa 900acgtcgaggc
cgcgattaaa ttccaacatg gatgctgatt tatatgggta taaatgggct 960cgcgataatg
tcgggcaatc aggtgcgaca atctatcgct tgtatgggaa gcccgatgcg 1020ccagagttgt
ttctgaaaca tggcaaaggt agcgttgcca atgatgttac agatgagatg 1080gtcagactaa
actggctgac ggaatttatg cctcttccga ccatcaagca ttttatccgt 1140actcctggtg
atgcatggtt actcaccact gcgatccccg gaaaaacagc attccaggta 1200ttagaagaat
atcctgattc aggtgaaaat attgttgatg cgctggcagt gttcctgcgc 1260cggttgcatt
cgattcctgt ttgtaattgt ccttttaaca gcgatcgcgt atttcgtctc 1320gctcaggcgc
aatcacgaat gaataacggt ttggttgatg cgagtgattt tgatgacgag 1380cgtaatggct
ggcctgttga acaagtctgg aaagaaatgc ataaactttt gccattctca 1440ccggattcag
tcgtcactca tggtgatttc tcacttgata accttatttt tgacgagggg 1500aaattaatag
gttgtattga tgttggacga gtcggaatcg cagaccgata ccaggatctt 1560gccatcctat
ggaactgcct cggtgagttt tctccttcat tacagaaacg gctttttcaa 1620aaatatggta
ttgataatcc tgatatgaat aaattgcagt ttcatttgat gctcgatgag 1680tttttctaat
cagaattggt taattggttg taacattatt cagattgggc cccgttccac 1740tgagcgtcag
accccgtaga aaagatcaaa ggatcttctt gagatccttt ttttctgcgc 1800gtaatctgct
gcttgcaaac aaaaaaacca ccgctaccag cggtggtttg tttgccggat 1860caagagctac
caactctttt tccgaaggta actggcttca gcagagcgca gataccaaat 1920actgttcttc
tagtgtagcc gtagttaggc caccacttca agaactctgt agcaccgcct 1980acatacctcg
ctctgctaat cctgttacca gtggctgctg ccagtggcga taagtcgtgt 2040cttaccgggt
tggactcaag acgatagtta ccggataagg cgcagcggtc gggctgaacg 2100gggggttcgt
gcacacagcc cagcttggag cgaacgacct acaccgaact gagataccta 2160cagcgtgagc
tatgagaaag cgccacgctt cccgaaggga gaaaggcgga caggtatccg 2220gtaagcggca
gggtcggaac aggagagcgc acgagggagc ttccaggggg aaacgcctgg 2280tatctttata
gtcctgtcgg gtttcgccac ctctgacttg agcgtcgatt tttgtgatgc 2340tcgtcagggg
ggcggagcct atggaaaaac gccagcaacg cggccttttt acggttcctg 2400gccttttgct
ggccttttgc tcacatgttc tttcctgcgt tatcccctga ttctgtggat 2460aaccgtatta
ccgctagcat ggatctcggg gacgtctaac tactaagcga gagtagggaa 2520ctgccaggca
tcaaataaaa cgaaaggctc agtcggaaga ctgggccttt cgttttatct 2580gttgtttgtc
ggtgaacgct ctcctgagta ggacaaatcc gccgggagcg gatttgaacg 2640ttgtgaagca
acggcccgga gggtggcggg caggacgccc gccataaact gccaggcatc 2700aaactaagca
gaaggccatc
27201222720DNAArtificial SequenceDescription of Artificial Sequence
Synthetic Entry Vector pENTR5 polynucleotide 122ctgacggatg
gcctttttgc gtttctacaa actcttcctg ttagttagtt acttaagctc 60gggccccaaa
taatgatttt attttgactg atagtgacct gttcgttgca acaaattgat 120aagcaatgct
tttttataat gccaactttg tacaaaaaag caggctttca tatgggaacc 180aattcagtcg
actggatccg gtaccgaatt cgcttactaa aagccagata acagtatgcg 240tatttgcgcg
ctgatttttg cggtataaga atatatactg atatgtatac ccgaagtatg 300tcaaaaagag
gtgtgcttct agaatgcagt ttaaggttta cacctataaa agagagagcc 360gttatcgtct
gtttgtggat gtacagagtg atattattga cacgcccggg cgacggatgg 420tgatccccct
ggccagtgca cgtctgctgt cagataaagt ctcccgtgaa ctttacccgg 480tggtgcatat
cggggatgaa agctggcgca tgatgaccac cgatatggcc agtgtgccgg 540tctccgttat
cggggaagaa gtggctgatc tcagccaccg cgaaaatgac atcaaaaacg 600ccattaacct
gatgttctgg ggaatataga attcgcggcc gcactcgaga tatctagacc 660cagctttctt
gtacaaagtt ggcattataa gaaagcattg cttatcaatt tgttgcaacg 720aacaggtcac
tatcagtcaa aataaaatca ttatttgcca tccagctgca gctctggccc 780gtgtctcaaa
atctctgatg ttacattgca caagataaaa atatatcatc atgaacaata 840aaactgtctg
cttacataaa cagtaataca aggggtgtta tgagccatat tcaacgggaa 900acgtcgaggc
cgcgattaaa ttccaacatg gatgctgatt tatatgggta taaatgggct 960cgcgataatg
tcgggcaatc aggtgcgaca atctatcgct tgtatgggaa gcccgatgcg 1020ccagagttgt
ttctgaaaca tggcaaaggt agcgttgcca atgatgttac agatgagatg 1080gtcagactaa
actggctgac ggaatttatg cctcttccga ccatcaagca ttttatccgt 1140actcctgatg
atgcatggtt actcaccact gcgatccccg gaaaaacagc attccaggta 1200ttagaagaat
atcctgattc aggtgaaaat attgttgatg cgctggcagt gttcctgcgc 1260cggttgcatt
cgattcctgt ttgtaattgt ccttttaaca gcgatcgcgt atttcgtctc 1320gctcaggcgc
aatcacgaat gaataacggt ttggttgatg cgagtgattt tgatgacgag 1380cgtaatggct
ggcctgttga acaagtctgg aaagaaatgc ataaactttt gccattctca 1440ccggattcag
tcgtcactca tggtgatttc tcacttgata accttatttt tgacgagggg 1500aaattaatag
gttgtattga tgttggacga gtcggaatcg cagaccgata ccaggatctt 1560gccatcctat
ggaactgcct cggtgagttt tctccttcat tacagaaacg gctttttcaa 1620aaatatggta
ttgataatcc tgatatgaat aaattgcagt ttcatttgat gctcgatgag 1680tttttctaat
cagaattggt taattggttg taacattatt cagattgggc cccgttccac 1740tgagcgtcag
accccgtaga aaagatcaaa ggatcttctt gagatccttt ttttctgcgc 1800gtaatctgct
gcttgcaaac aaaaaaacca ccgctaccag cggtggtttg tttgccggat 1860caagagctac
caactctttt tccgaaggta actggcttca gcagagcgca gataccaaat 1920actgttcttc
tagtgtagcc gtagttaggc caccacttca agaactctgt agcaccgcct 1980acatacctcg
ctctgctaat cctgttacca gtggctgctg ccagtggcga taagtcgtgt 2040cttaccgggt
tggactcaag acgatagtta ccggataagg cgcagcggtc gggctgaacg 2100gggggttcgt
gcacacagcc cagcttggag cgaacgacct acaccgaact gagataccta 2160cagcgtgagc
tatgagaaag cgccacgctt cccgaaggga gaaaggcgga caggtatccg 2220gtaagcggca
gggtcggaac aggagagcgc acgagggagc ttccaggggg aaacgcctgg 2280tatctttata
gtcctgtcgg gtttcgccac ctctgacttg agcgtcgatt tttgtgatgc 2340tcgtcagggg
ggcggagcct atggaaaaac gccagcaacg cggccttttt acggttcctg 2400gccttttgct
ggccttttgc tcacatgttc tttcctgcgt tatcccctga ttctgtggat 2460aaccgtatta
ccgctagcat ggatctcggg gacgtctaac tactaagcga gagtagggaa 2520ctgccaggca
tcgaataaaa cgaaaggctc agtcggaaga ctgggccttt cgttttatct 2580gttgtttgtc
ggtgaacgct ctcctgagta ggacaaatcc gccgggagcg gatttgaacg 2640ttgtgaagca
acggcccgga gggtggcggg caggacgccc gccataaact gccaggcatc 2700aaactaagca
gaaggccatc
27201232717DNAArtificial SequenceDescription of Artificial Sequence
Synthetic Entry Vector pENTR6 polynucleotide 123ctgacggatg
gcctttttgc gtttctacaa actcttcctg ttagttagtt acttaagctc 60gggccccaaa
taatgatttt attttgactg atagtgacct gttcgttgca acaaattgat 120aagcaatgct
tttttataat gccaactttg tacaaaaaag caggctgcat gcgaaccaat 180tcagtcgact
ggatccggta ccgaattcgc ttactaaaag ccagataaca gtatgcgtat 240ttgcgcgctg
atttttgcgg tataagaata tatactgata tgtatacccg aagtatgtca 300aaaagaggtg
tgcttctaga atgcagttta aggtttacac ctataaaaga gagagccgtt 360atcgtctgtt
tgtggatgta cagagtgata ttattgacac gcccgggcga cggatggtga 420tccccctggc
cagtgcacgt ctgctgtcag ataaagtctc ccgtgaactt tacccggtgg 480tgcatatcgg
ggatgaaagc tggcgcatga tgaccaccga tatggccagt gtgccggtct 540ccgttatcgg
ggaagaagtg gctgatctca gccaccgcga aaatgacatc aaaaacgcca 600ttaacctgat
gttctgggga atatagaatt cgcggccgca ctcgagatat ctagacccag 660ctttcttgta
caaagttggc attataagaa agcattgctt atcaatttgt tgcaacgaac 720aggtcactat
cagtcaaaat aaaatcatta tttgccatcc agctgcagct ctggcccgtg 780tctcaaaatc
tctgatgtta cattgcacaa gataaaaata tatcatcatg aacaataaaa 840ctgtctgctt
acataaacag taatacaagg ggtgttatga gccatattca acgggaaacg 900tcgaggccgc
gattaaattc caacatggat gctgatttat atgggtataa atgggctcgc 960gataatgtcg
ggcaatcagg tgcgacaatc tatcgcttgt atgggaagcc cgatgcgcca 1020gagttgtttc
tgaaacatgg caaaggtagc gttgccaatg atgttacaga tgagatggtc 1080agactaaact
ggctgacgga atttatgcct cttccgacca tcaagcattt tatccgtact 1140cctgatgatg
catggttact caccactgcg atccccggaa aaacagcatt ccaggtatta 1200gaagaatatc
ctgattcagg tgaaaatatt gttgatgcgc tggcagtgtt cctgcgccgg 1260ttgcattcga
ttcctgtttg taattgtcct tttaacagcg atcgcgtatt tcgtctcgct 1320caggcgcaat
cacgaatgaa taacggtttg gttgatgcga gtgattttga tgacgagcgt 1380aatggctggc
ctgttgaaca agtctggaaa gaaatgcata aacttttgcc attctcaccg 1440gattcagtcg
tcactcatgg tgatttctca cttgataacc ttatttttga cgaggggaaa 1500ttaataggtt
gtattgatgt tggacgagtc ggaatcgcag accgatacca ggatcttgcc 1560atcctatgga
actgcctcgg tgagttttct ccttcattac agaaacggct ttttcaaaaa 1620tatggtattg
ataatcctga tatgaataaa ttgcagtttc atttgatgct cgatgagttt 1680ttctaatcag
aattggttaa ttggttgtaa cattattcag attgggcccc gttccactga 1740gcgtcagacc
ccgtagaaaa gatcaaagga tcttcttgag atcctttttt tctgcgcgta 1800atctgctgct
tgcaaacaaa aaaaccaccg ctaccagcgg tggtttgttt gccggatcaa 1860gagctaccaa
ctctttttcc gaaggtaact ggcttcagca gagcgcagat accaaatact 1920gttcttctag
tgtagccgta gttaggccac cacttcaaga actctgtagc accgcctaca 1980tacctcgctc
tgctaatcct gttaccagtg gctgctgcca gtggcgataa gtcgtgtctt 2040accgggttgg
actcaagacg atagttaccg gataaggcgc agcggtcggg ctgaacgggg 2100ggttcgtgca
cacagcccag cttggagcga acgacctaca ccgaactgag atacctacag 2160cgtgagctat
gagaaagcgc cacgcttccc gaagggagaa aggcggacag gtatccggta 2220agcggcaggg
tcggaacagg agagcgcacg agggagcttc cagggggaaa cgcctggtat 2280ctttatagtc
ctgtcgggtt tcgccacctc tgacttgagc gtcgattttt gtgatgctcg 2340tcaggggggc
ggagcctatg gaaaaacgcc agcaacgcgg cctttttacg gttcctggcc 2400ttttgctggc
cttttgctca catgttcttt cctgcgttat cccctgattc tgtggataac 2460cgtattaccg
ctagcatgga tctcggggac gtctaactac taagcgagag tagggaactg 2520ccaggcatca
aataaaacga aaggctcagt cggaagactg ggcctttcgt tttatctgtt 2580gtttgtcggt
gaacgctctc ctgagtagga caaatccgcc gggagcggat ttgaacgttg 2640tgaagcaacg
gcccggaggg tggcgggcag gacgcccgcc ataaactgcc aggcatcaaa 2700ctaagcagaa
ggccatc
27171242738DNAArtificial SequenceDescription of Artificial Sequence
Synthetic Entry Vector pENTR7 polynucleotide 124ctgacggatg
gcctttttgc gtttctacaa actcttcctg ttagttagtt acttaagctc 60gggccccaaa
taatgatttt attttgactg atagtgacct gttcgttgca acaaattgat 120aagcaatgct
tttttataat gccaactttg tacaaaaaag caggctttga aaacctgtat 180tttcaaggaa
ccgtttcatg catcgtcgac tggatccggt accgaattcg cttactaaaa 240gccagataac
agtatgcgta tttgcgcgct gatttttgcg gtataagaat atatactgat 300atgtataccc
gaagtatgtc aaaaagaggt gtgcttctag aatgcagttt aaggtttaca 360cctataaaag
agagagccgt tatcgtctgt ttgtggatgt acagagtgat attattgaca 420cgcccgggcg
acggatagtg atccccctgg ccagtgcacg tctgctgtca gataaagtct 480cccgtgaact
ttacccggtg gtgcatatcg gggatgaaag ctggcgcatg atgaccaccg 540atatggccag
tgtgccggtc tccgttatcg gggaagaagt ggctgatctc agccaccgcg 600aaaatgacat
caaaaacgcc attaacctga tgttctgggg aatatagaat tcgcggccgc 660actcgagata
tctagaccca gctttcttgt acaaagttgg cattataaga aagcattgct 720tatcaatttg
ttgcaacgaa caggtcacta tcagtcaaaa taaaatcatt atttgccatc 780cagctgcagc
tctggcccgt gtctcaaaat ctctgatgtt acattgcaca agataaaaat 840atatcatcat
gaacaataaa actgtctgct tacataaaca gtaatacaag gggtgttatg 900agccatattc
aacgggaaac gtcgaggccg cgattaaatt ccaacatgga tgctgattta 960tatgggtata
aatgggctcg cgataatgtc gggcaatcag gtgcgacaat ctatcgcttg 1020tatgggaagc
ccgatgcgcc agagttgttt ctgaaacatg gcaaaggtag cgttgccaat 1080gatgttacag
atgagatggt cagactaaac tggctgacgg aatttatgcc tcttccgacc 1140atcaagcatt
ttatccgtac tcctgatgat gcatggttac tcaccactgc gatccccgga 1200aaaacagcat
tccaggtatt agaagaatat cctgattcag gtgaaaatat tgttgatgcg 1260ctggcagtgt
tcctgcgccg gttgcattcg attcctgttt gtaattgtcc ttttaacagc 1320gatcgcgtat
ttcgtctcgc tcaggcgcaa tcacgaatga ataacggttt ggttgatgcg 1380agtgattttg
atgacgagcg taatggctgg cctgttgaac aagtctggaa agaaatgcat 1440aaacttttgc
cattctcacc ggattcagtc gtcactcatg gtgatttctc acttgataac 1500cttatttttg
acgaggggaa attaataggt tgtattgatg ttggacgagt cggaatcgca 1560gaccgatacc
aggatcttgc catcctatgg aactgcctcg gtgagttttc tccttcatta 1620cagaaacggc
tttttcaaaa atatggtatt gataatcctg atatgaataa attgcagttt 1680catttgatgc
tcgatgagtt tttctaatca gaattggtta attggttgta acattattca 1740gattgggccc
cgttccactg agcgtcagac cccgtagaaa agatcaaagg atcttcttga 1800gatccttttt
ttctgcgcgt aatctgctgc ttgcaaacaa aaaaaccacc gctaccagcg 1860gtggtttgtt
tgccggatca agagctacca actctttttc cgaaggtaac tggcttcagc 1920agagcgcaga
taccaaatac tgttcttcta gtgtagccgt agttaggcca ccacttcaag 1980aactctgtag
caccgcctac atacctcgct ctgctaatcc tgttaccagt ggctgctgcc 2040agtggcgata
agtcgtgtct taccgggttg gactcaagac gatagttacc ggataaggcg 2100cagcggtcgg
gctgaacggg gggttcgtgc acacagccca gcttggagcg aacgacctac 2160accgaactga
gatacctaca gcgtgagcta tgagaaagcg ccacgcttcc cgaagggaga 2220aaggcggaca
ggtatccggt aagcggcagg gtcggaacag gagagcgcac gagggagctt 2280ccagggggaa
acgcctggta tctttatagt cctgtcgggt ttcgccacct ctgacttgag 2340cgtcgatttt
tgtgatgctc gtcagggggg cggagcctat ggaaaaacgc cagcaacgcg 2400gcctttttac
ggttcctggc cttttgctgg ccttttgctc acatgttctt tcctgcgtta 2460tcccctgatt
ctgtggataa ccgtattacc gctagcatgg atctcgggga cgtctaacta 2520ctaagcgaga
gtagggaact gccaggcatc aaataaaacg aaaggctcag tcggaagact 2580gggcctttcg
ttttatctgt tgtttgtcgg tgaacgctct cctgagtagg acaaatccgc 2640cgggagcgga
tttgaacgtt gtgaagcaac ggcccggagg gtggcgggca ggacgcccgc 2700cataaactgc
caggcatcaa actaagcaga aggccatc
27381252735DNAArtificial SequenceDescription of Artificial Sequence
Synthetic Entry Vector pENTR8 polynucleotide 125ctgacggatg
gcctttttgc gtttctacaa actcttcctg ttagttagtt acttaagctc 60gggccccaaa
taatgatttt attttgactg atagtgacct gttcgttgca acaaattgat 120aagcaatgct
tttttataat gccaactttg tacaaaaaag caggctttga aaacctgtat 180tttcaaggaa
ccatggacct agtcgactgg atccggtacc gaattcgctt actaaaagcc 240agataacagt
atgcgtattt gcgcgctgat ttttgcggta taagaatata tactgatatg 300tatacccgaa
gtatgtcaaa aagaggtgtg cttctagaat gcagtttaag gtttacacct 360ataaaagaga
gagccgttat cgtctgtttg tggatgtaca gagtgatatt attgacacgc 420ccgggcgacg
gatagtgatc cccctggcca gtgcacgtct gctgtcagat aaagtctccc 480gtgaacttta
cccggtggtg catatcgggg atgaaagctg gcgcatgatg accaccgata 540tggccagtgt
gccggtctcc gttatcgggg aagaagtggc tgatctcagc caccgcgaaa 600atgacatcaa
aaacgccatt aacctgatgt tctggggaat atagaattcg cggccgcact 660cgagatatct
agacccagct ttcttgtaca aagttggcat tataagaaag cattgcttat 720caatttgttg
caacgaacag gtcactatca gtcaaaataa aatcattatt tgccatccag 780ctgcagctct
ggcccgtgtc tcaaaatctc tgatgttaca ttgcacaaga taaaaatata 840tcatcatgaa
caataaaact gtctgcttac ataaacagta atacaagggg tgttatgagc 900catattcaac
gggaaacgtc gaggccgcga ttaaattcca acatggatgc tgatttatat 960gggtataaat
gggctcgcga taatgtcggg caatcaggtg cgacaatcta tcgcttgtat 1020gggaagcccg
atgcgccaga gttgtttctg aaacatggca aaggtagcgt tgccaatgat 1080gttacagatg
agatggtcag actaaactgg ctgacggaat ttatgcctct tccgaccatc 1140aagcatttta
tccgtactcc tgatgatgca tggttactca ccactgcgat ccccggaaaa 1200acagcattcc
aggtattaga agaatatcct gattcaggtg aaaatattgt tgatgcgctg 1260gcagtgtccc
tgcgccggtt gcattcgatt cctgtttgta attgtccttt taacagcgat 1320cgcgtatttc
gtctcgctca ggcgcaatca cgaatgaata acggtttggt tgatgcgagt 1380gattttgatg
acgagcgtaa tggctggcct gttgaacaag tctggaaaga aatgcataaa 1440cttttgccat
tctcaccgga ttcagtcgtc actcatggtg atttctcact tgataacctt 1500atttttgacg
aggggaaatt aataggttgt attgatgttg gacgagtcgg aatcgcagac 1560cgataccagg
atcttgccat cctatggaac tgcctcggtg agttttctcc ttcattacag 1620aaacggcttt
ttcaaaaata tggtattgat aatcctgata tgaataaatt gcagtttcat 1680ttgatgctcg
atgagttttt ctaatcagaa ttggttaatt ggttgtaaca ttattcagat 1740tgggccccgt
tccactgagc gtcagacccc gtagaaaaga tcaaaggatc ttcttgagat 1800cctttttttc
tgcgcgtaat ctgctgcttg caaacaaaaa aaccaccgct accagcggtg 1860gtttgtttgc
cggatcaaga gctaccaact ctttttccga aggtaactgg cttcagcaga 1920gcgcagatac
caaatactgt tcttctagtg tagccgtagt taggccacca cttcaagaac 1980tctgtagcac
cgcctacata cctcgctctg ctaatcctgt taccagtggc tgctgccagt 2040ggcgataagt
cgtgtcttac cgggttggac tcaagacgat agttaccgga taaggcgcag 2100cggtcgggct
gaacgggggg ttcgtgcaca cagcccagct tggagcgaac gacctacacc 2160gaactgagat
acctacagcg tgagctatga gaaagcgcca cgcttcccga agggagaaag 2220gcggacaggt
atccggtaag cggcagggtc ggaacaggag agcgcacgag ggagcttcca 2280gggggaaacg
cctggtatct ttatagtcct gtcgggtttc gccacctctg acttgagcgt 2340cgatttttgt
gatgctcgtc aggggggcgg agcctatgga aaaacgccag caacgcggcc 2400tttttacggt
tcctggcctt ttgctggcct tttgctcaca tgttctttcc tgcgttatcc 2460cctgattctg
tggataaccg tattaccgct agcatggatc tcggggacgt ctaactacta 2520agcgagagta
gggaactgcc aggcatcaaa taaaacgaaa ggctcagtcg gaagactggg 2580cctttcgttt
tatctgttgt ttgtcggtga acgctctcct gagtaggaca aatccgccgg 2640gagcggattt
gaacgttgtg aagcaacggc ccggagggtg gcgggcagga cgcccgccat 2700aaactgccag
gcatcaaact aagcagaagg ccatc
27351262735DNAArtificial SequenceDescription of Artificial Sequence
Synthetic Entry Vector pENTR9 polynucleotide 126ctgacggatg
gcctttttgc gtttctacaa actcttcctg ttagttagtt acttaagctc 60gggccccaaa
taatgatttt attttgactg atagtgacct gttcgttgca acaaattgat 120aagcaatgct
tttttataat gccaactttg tacaaaaaag caggctttga aaacctgtat 180tttcaaggac
atatgagatc tgtcgactgg atccggtacc gaattcgctt actaaaagcc 240agataacagt
atgcgtattt gcgcgctgat ttttgcggta taagaatata tactgatatg 300tatacccgaa
gtatgtcaaa aagaggtgtg cttctagaat gcagtttaag gtttacacct 360ataaaagaga
gagccgttat cgtctgtttg tggatgtaca gagtgatatt attgacacgc 420ccgggcgacg
gatagtgatc cccctggcca gtgcacgtct gctgtcagat aaagtctccc 480gtgaacttta
cccggtggtg catatcgggg atgaaagctg gcgcatgatg accaccgata 540tggccagtgt
gccggtctcc gttatcgggg aagaagtggc tgatctcagc caccgcgaaa 600atgacatcaa
aaacgccatt aacctgatgt tctggggaat atagaattcg cggccgcact 660cgagatatct
agacccagct ttcttgtaca aagttggcat tataagaaag cattgcttat 720caatttgttg
caacgaacag gtcactatca gtcaaaataa aatcattatt tgccatccag 780ctgcagctct
ggcccgtgtc tcaaaatctc tgatgttaca ttgcacaaga taaaaatata 840tcatcatgaa
caataaaact gtctgcttac ataaacagta atacaagggg tgttatgagc 900catattcaac
gggaaacgtc gaggccgcga ttaaattcca acatggatgc tgatttatat 960gggtataaat
gggctcgcga taatgtcggg caatcaggtg cgacaatcta tcgcttgtat 1020gggaagcccg
atgcgccaga gttgtttctg aaacatggca aaggtagcgt tgccaatgat 1080gttacagatg
agatggtcag actaaactgg ctgacggaat ttatgcctct tccgaccatc 1140aagcatttta
tccgtactcc tgatgatgca tggttactca ccactgcgat ccccggaaaa 1200acagcattcc
aggtattaga agaatatcct gattcaggtg aaaatattgt tgatgcgctg 1260gcagtgtccc
tgcgccggtt gcattcgatt cctgtttgta attgtccttt taacagcgat 1320cgcgtatttc
gtctcgctca ggcgcaatca cgaatgaata acggtttggt tgatgcgagt 1380gattttgatg
acgagcgtaa tggctggcct gttgaacaag tctggaaaga aatgcataaa 1440cttttgccat
tctcaccgga ttcagtcgtc actcatggtg atttctcact tgataacctt 1500atttttgacg
aggggaaatt aataggttgt attgatgttg gacgagtcgg aatcgcagac 1560cgataccagg
atcttgccat cctatggaac tgcctcggtg agttttctcc ttcattacag 1620aaacggcttt
ttcaaaaata tggtattgat aatcctgata tgaataaatt gcagtttcat 1680ttgatgctcg
atgagttttt ctaatcagaa ttggttaatt ggttgtaaca ttattcagat 1740tgggccccgt
tccactgagc gtcagacccc gtagaaaaga tcaaaggatc ttcttgagat 1800cctttttttc
tgcgcgtaat ctgctgcttg caaacaaaaa aaccaccgct accagcggtg 1860gtttgtttgc
cggatcaaga gctaccaact ctttttccga aggtaactgg cttcagcaga 1920gcgcagatac
caaatactgt tcttctagtg tagccgtagt taggccacca cttcaagaac 1980tctgtagcac
cgcctacata cctcgctctg ctaatcctgt taccagtggc tgctgccagt 2040ggcgataagt
cgtgtcttac cgggttggac tcaagacgat agttaccgga taaggcgcag 2100cggtcgggct
gaacgggggg ttcgtgcaca cagcccagct tggagcgaac gacctacacc 2160gaactgagat
acctacagcg tgagctatga gaaagcgcca cgcttcccga agggagaaag 2220gcggacaggt
atccggtaag cggcagggtc ggaacaggag agcgcacgag ggagcttcca 2280gggggaaacg
cctggtatct ttatagtcct gtcgggtttc gccacctctg acttgagcgt 2340cgatttttgt
gatgctcgtc aggggggcgg agcctatgga aaaacgccag caacgcggcc 2400tttttacggt
tcctggcctt ttgctggcct tttgctcaca tgttctttcc tgcgttatcc 2460cctgattctg
tggataaccg tattaccgct agcatggatc tcggggacgt ctaactacta 2520agcgagagta
gggaactgcc aggcatcaaa taaaacgaaa ggctcagtcg gaagactggg 2580cctttcgttt
tatctgttgt ttgtcggtga acgctctcct gagtaggaca aatccgccgg 2640gagcggattt
gaacgttgtg aagcaacggc ccggagggtg gcgggcagga cgcccgccat 2700aaactgccag
gcatcaaact aagcagaagg ccatc
27351272738DNAArtificial SequenceDescription of Artificial Sequence
Synthetic Entry Vector pENTR10 polynucleotide 127ctgacggatg
gcctttttgc gtttctacaa actcttcctg ttagttagtt acttaagctc 60gggccccaaa
taatgatttt attttgactg atagtgacct gttcgttgca acaaattgat 120aagcaatgct
tttttataat gccaactttg tacaaaaaag caggcttcga actaaggaaa 180tacttacata
tgggaaccaa ttcagtcgac tggatccggt accgaattcg cttactaaaa 240gccagataac
agtatgcgta tttgcgcgct gatttttgcg gtataagaat atatactgat 300atgtataccc
gaagtatgtc aaaaagaggt gtgcttctag aatgcagttt aaggtttaca 360cctataaaag
agagagccgt tatcgtctgt ttgtggatgt acagagtgat attattgaca 420cgcccgggcg
acggatggtg atccccctgg ccagtgcacg tctgctgtca gataaagtct 480cccgtgaact
ttacccggtg gtgcatatcg gggatgaaag ctggcgcatg atgaccaccg 540atatggccag
tgtgccggtc tccgttatcg gggaagaagt ggctgatctc agccaccgcg 600aaaatgacat
caaaaacgcc attaacctga tgttctgggg aatatagaat tcgcggccgc 660actcgagata
tctagaccca gctttcttgt acaaagttgg cattataaga aagcattgct 720tatcaatttg
ttgcaacgaa caggtcacta tcagtcaaaa taaaatcatt atttgccatc 780cagctgcagc
tctggcccgt gtctcaaaat ctctgatgtt acattgcaca agataaaaat 840atatcatcat
gaacaataaa actgtctgct tacataaaca gtaatacaag gggtgttatg 900agccatattc
aacgggaaac gtcgaggccg cgattaaatt ccaacatgga tgctgattta 960tatgggtata
aatgggctcg cgataatgtc gggcaatcag gtgcgacaat ctatcgcttg 1020tatgggaagc
ccgatgcgcc agagttgttt ctgaaacatg gcaaaggtag cgttgccaat 1080gatgttacag
atgagatggt cagactaaac tggctgacgg aatttatgcc tcttccgacc 1140atcaagcatt
ttatccgtac tcctgatgat gcatggttac tcaccactgc gatccccgga 1200aaaacagcat
tccaggtatt agaagaatat cctgattcag gtgaaaatat tgttgatgcg 1260ctggcagtgt
tcctgcgccg gttgcattcg attcctgttt gtaattgtcc ttttaacagc 1320gatcgcgtat
ttcgtctcgc tcaggcgcaa tcacgaatga ataacggttt ggttgatgcg 1380agtgattttg
atgacgagcg taatggctgg cctgttgaac aagtctggaa agaaatgcat 1440aaacttttgc
cattctcacc ggattcagtc gtcactcatg gtgatttctc acttgataac 1500cttatttttg
acgaggggaa attaataggt tgtattgatg ttggacgagt cggaatcgca 1560gaccgatacc
aggatcttgc catcctatgg aactgcctcg gtgagttttc tccttcatta 1620cagaaacggc
tttttcaaaa atatggtatt gataatcctg atatgaataa attgcagttt 1680catttgatgc
tcgatgagtt tttctaatca gaattggtta attggttgta acattattca 1740gattgggccc
cgttccactg agcgtcagac cccgtagaaa agatcaaagg atcttcttga 1800gatccttttt
ttctgcgcgt aatctgctgc ttgcaaacaa aaaaaccacc gctaccagcg 1860gtggtttgtt
tgccggatca agagctacca actctttttc cgaaggtaac tggcttcagc 1920agagcgcaga
taccaaatac tgttcttcta gtgtagccgt agttaggcca ccacttcaag 1980aactctgtag
caccgcctac atacctcgct ctgctaatcc tgttaccagt ggctgctgcc 2040agtggcgata
agtcgtgtct taccgggttg gactcaagac gatagttacc ggataaggcg 2100cagcggtcgg
gctgaacggg gggttcgtgc acacagccca gcttggagcg aacgacctac 2160accgaactga
gatacctaca gcgtgagcta tgagaaagcg ccacgcttcc cgaagggaga 2220aaggcggaca
ggtatccggt aagcggcagg gtcggaacag gagagcgcac gagggagctt 2280ccagggggaa
acgcctggta tctttatagt cctgtcgggt ttcgccacct ctgacttgag 2340cgtcgatttt
tgtgatgctc gtcagggggg cggagcctat ggaaaaacgc cagcaacgcg 2400gcctttttac
ggttcctggc cttttgctgg ccttttgctc acatgttctt tcctgcgtta 2460tcccctgatt
ctgtggataa ccgtattacc gctagcatgg atctcgggga cgtctaacta 2520ctaagcgaga
gtagggaact gccaggcatc gaataaaacg aaaggctcag tcggaagact 2580gggcctttcg
ttttatctgt tgtttgtcgg tgaacgctct cctgagtagg acaaatccgc 2640cgggagcgga
tttgaacgtt gtgaagcaac ggcccggagg gtggcgggca ggacgcccgc 2700cataaactgc
caggcatcaa actaagcaga aggccatc
27381282744DNAArtificial SequenceDescription of Artificial Sequence
Synthetic Entry Vector pENTR11 polynucleotide 128ctgacggatg
gcctttttgc gtttctacaa actcttcctg ttagttagtt acttaagctc 60gggccccaaa
taatgatttt attttgactg atagtgacct gttcgttgca acaaattgat 120aagcaatgct
tttttataat gccaactttg tacaaaaaag caggcttcga aggagataga 180accaattctc
taaggaaata cttaaccatg gtcgactgga tccggtaccg aattcgctta 240ctaaaagcca
gataacagta tgcgtatttg cgcgctgatt tttgcggtat aagaatatat 300actgatatgt
atacccgaag tatgtcaaaa agaggtgtgc ttctagaatg cagtttaagg 360tttacaccta
taaaagagag agccgttatc gtctgtttgt ggatgtacag agtgatatta 420ttgacacgcc
cgggcgacgg atagtgatcc ccctggccag tgcacgtctg ctgtcagata 480aagtctcccg
tgaactttac ccggtggtgc atatcgggga tgaaagctgg cgcatgatga 540ccaccgatat
ggccagtgtg ccggtctccg ttatcgggga agaagtggct gatctcagcc 600accgcgaaaa
tgacatcaaa aacgccatta acctgatgtt ctggggaata tagaattcgc 660ggccgcactc
gagatatcta gacccagctt tcttgtacaa agttggcatt ataagaaagc 720attgcttatc
aatttgttgc aacgaacagg tcactatcag tcaaaataaa atcattattt 780gccatccagc
tgcagctctg gcccgtgtct caaaatctct gatgttacat tgcacaagat 840aaaaatatat
catcatgaac aataaaactg tctgcttaca taaacagtaa tacaaggggt 900gttatgagcc
atattcaacg ggaaacgtcg aggccgcgat taaattccaa catggatgct 960gatttatatg
ggtataaatg ggctcgcgat aatgtcgggc aatcaggtgc gacaatctat 1020cgcttgtatg
ggaagcccga tgcgccagag ttgtttctga aacatggcaa aggtagcgtt 1080gccaatgatg
ttacagatga gatggtcaga ctaaactggc tgacggaatt tatgcctctt 1140ccgaccatca
agcattttat ccgtactcct gatgatgcat ggttactcac cactgcgatc 1200cccggaaaaa
cagcattcca ggtattagaa gaatatcctg attcaggtga aaatattgtt 1260gatgcgctgg
cagtgttcct gcgccggttg cattcgattc ctgtttgtaa ttgtcctttt 1320aacagcgatc
gcgtatttcg tctcgctcag gcgcaatcac gaatgaataa cggtttggtt 1380gatgcgagtg
attttgatga cgagcgtaat ggctggcctg ttgaacaagt ctggaaagaa 1440atgcataaac
ttttgccatt ctcaccggat tcagtcgtca ctcatggtga tttctcactt 1500gataacctta
tttttgacga ggggaaatta ataggttgta ttgatgttgg acgagtcgga 1560atcgcagacc
gataccagga tcttgccatc ctatggaact gcctcggtga gttttctcct 1620tcattacaga
aacggctttt tcaaaaatat ggtattgata atcctgatat gaataaattg 1680cagtttcatt
tgatgctcga tgagtttttc taatcagaat tggttaattg gttgtaacat 1740tattcagatt
gggccccgtt ccactgagcg tcagaccccg tagaaaagat caaaggatct 1800tcttgagatc
ctttttttct gcgcgtaatc tgctgcttgc aaacaaaaaa accaccgcta 1860ccagcggtgg
tttgtttgcc ggatcaagag ctaccaactc tttttccgaa ggtaactggc 1920ttcagcagag
cgcagatacc aaatactgtt cttctagtgt agccgtagtt aggccaccac 1980ttcaagaact
ctgtagcacc gcctacatac ctcgctctgc taatcctgtt accagtggct 2040gctgccagtg
gcgataagtc gtgtcttacc gggttggact caagacgata gttaccggat 2100aaggcgcagc
ggtcgggctg aacggggggt tcgtgcacac agcccagctt ggagcgaacg 2160acctacaccg
aactgagata cctacagcgt gagctatgag aaagcgccac gcttcccgaa 2220gggagaaagg
cggacaggta tccggtaagc ggcagggtcg gaacaggaga gcgcacgagg 2280gagcttccag
ggggaaacgc ctggtatctt tatagtcctg tcgggtttcg ccacctctga 2340cttgagcgtc
gatttttgtg atgctcgtca ggggggcgga gcctatggaa aaacgccagc 2400aacgcggcct
ttttacggtt cctggccttt tgctggcctt ttgctcacat gttctttcct 2460gcgttatccc
ctgattctgt ggataaccgt attaccgcta gcatggatct cggggacgtc 2520taactactaa
gcgagagtag ggaactgcca ggcatcaaat aaaacgaaag gctcagtcgg 2580aagactgggc
ctttcgtttt atctgttgtt tgtcggtgaa cgctctcctg agtaggacaa 2640atccgccggg
agcggatttg aacgttgtga agcaacggcc cggagggtgg cgggcaggac 2700gcccgccata
aactgccagg catcaaacta agcagaaggc catc
27441296464DNAArtificial SequenceDescription of Artificial Sequence
Synthetic pDEST1 polynucleotide 129gtttgacagc ttatcatcga ctgcacggtg
caccaatgct tctggcgtca ggcagccatc 60ggaagctgtg gtatggctgt gcaggtcgta
aatcactgca taattcgtgt cgctcaaggc 120gcactcccgt tctggataat gttttttgcg
ccgacatcat aacggttctg gcaaatattc 180tgaaatgagc tgttgacaat taatcatccg
gtccgtataa tctgtggaat tgtgagcggg 240ataacaattt catcgcgagg taccaagcta
tcacaagttt gtacaaaaaa gctgaacgag 300aaacgtaaaa tgatataaat atcaatatat
taaattagat tttgcataaa aaacagacta 360cataatactg taaaacacaa catatccagt
cactatggcg gccgctaagt tggcagcatc 420acccgacgca ctttgcgccg aataaatacc
tgtgacggaa gatcacttcg cagaataaat 480aaatcctggt gtccctgttg ataccgggaa
gccctgggcc aacttttggc gaaaatgaga 540cgttgatcgg cacgtaagag gttccaactt
tcaccataat gaaataagat cactaccggg 600cgtatttttt gagttatcga gattttcagg
agctaaggaa gctaaaatgg agaaaaaaat 660cactggatat accaccgttg atatatccca
atggcatcgt aaagaacatt ttgaggcatt 720tcagtcagtt gctcaatgta cctataacca
gaccgttcag ctggatatta cggccttttt 780aaagaccgta aagaaaaata agcacaagtt
ttatccggcc tttattcaca ttcttgcccg 840cctgatgaat gctcatccgg aattccgtat
ggcaatgaaa gacggtgagc tggtgatatg 900ggatagtgtt cacccttgtt acaccgtttt
ccatgagcaa actgaaacgt tttcatcgct 960ctggagtgaa taccacgacg atttccggca
gtttctacac atatattcgc aagatgtggc 1020gtgttacggt gaaaacctgg cctatttccc
taaagggttt attgagaata tgtttttcgt 1080ctcagccaat ccctgggtga gtttcaccag
ttttgattta aacgtggcca atatggacaa 1140cttcttcgcc cccgttttca ccatgggcaa
atattatacg caaggcgaca aggtgctgat 1200gccgctggcg attcaggttc atcatgccgt
ctgtgatggc ttccatgtcg gcagaatgct 1260taatgaatta caacagtact gcgatgagtg
gcagggcggg gcgtaaacgc gtggatccgg 1320cttactaaaa gccagataac agtatgcgta
tttgcgcgct gatttttgcg gtataagaat 1380atatactgat atgtataccc gaagtatgtc
aaaaagaggt gtgctatgaa gcagcgtatt 1440acagtgacag ttgacagcga cagctatcag
ttgctcaagg catatatgat gtcaatatct 1500ccggtctggt aagcacaacc atgcagaatg
aagcccgtcg tctgcgtgcc gaacgctgga 1560aagcggaaaa tcaggaaggg atggctgagg
tcgcccggtt tattgaaatg aacggctctt 1620ttgctgacga gaacagggac tggtgaaatg
cagtttaagg tttacaccta taaaagagag 1680agccgttatc gtctgtttgt ggatgtacag
agtgatatta ttgacacgcc cgggcgacgg 1740atggtgatcc ccctggccag tgcacgtctg
ctgtcagata aagtctcccg tgaactttac 1800ccggtggtgc atatcgggga tgaaagctgg
cgcatgatga ccaccgatat ggccagtgtg 1860ccggtctccg ttatcgggga agaagtggct
gatctcagcc accgcgaaaa tgacatcaaa 1920aacgccatta acctgatgtt ctggggaata
taaatgtcag gctcccttat acacagccag 1980tctgcaggtc gaccatagtg actggatatg
ttgtgtttta cagtattatg tagtctgttt 2040tttatgcaaa atctaattta atatattgat
atttatatca ttttacgttt ctcgttcagc 2100tttcttgtac aaagtggtga tagcttggct
gttttggcgg atgagagaag attttcagcc 2160tgatacagat taaatcagaa cgcagaagcg
gtctgataaa acagaatttg cctggcggca 2220gtagcgcggt ggtcccacct gaccccatgc
cgaactcaga agtgaaacgc cgtagcgccg 2280atggtagtgt ggggtctccc catgcgagag
tagggaactg ccaggcatca aataaaacga 2340aaggctcagt cgaaagactg ggcctttcgt
tttatctgtt gtttgtcggt gaacgctctc 2400ctgagtagga caaatccgcc gggagcggat
ttgaacgttg cgaagcaacg gcccggaggg 2460tggcgggcag gacgcccgcc ataaactgcc
aggcatcaaa ttaagcagaa ggccatcctg 2520acggatggcc tttttgcgtt tctacaaact
ctttttgttt atttttctaa atacattcaa 2580atatgtatcc gctcatgaga caataaccct
gataaatgct tcaataatat tgaaaaagga 2640agagtatgag tattcaacat ttccgtgtcg
cccttattcc cttttttgcg gcattttgcc 2700ttcctgtttt tgctcaccca gaaacgctgg
tgaaagtaaa agatgctgaa gatcagttgg 2760gtgcacgagt gggttacatc gaactggatc
tcaacagcgg taagatcctt gagagttttc 2820gccccgaaga acgttttcca atgatgagca
cttttaaagt tctgctatgt ggcgcggtat 2880tatcccgtgt tgacgccggg caagagcaac
tcggtcgccg catacactat tctcagaatg 2940acttggttga gtactcacca gtcacagaaa
agcatcttac ggatggcatg acagtaagag 3000aattatgcag tgctgccata accatgagtg
ataacactgc ggccaactta cttctgacaa 3060cgatcggagg accgaaggag ctaaccgctt
ttttgcacaa catgggggat catgtaactc 3120gccttgatcg ttgggaaccg gagctgaatg
aagccatacc aaacgacgag cgtgacacca 3180cgatgcctac agcaatggca acaacgttgc
gcaaactatt aactggcgaa ctacttactc 3240tagcttcccg gcaacaatta atagactgga
tggaggcgga taaagttgca ggaccacttc 3300tgcgctcggc ccttccggct ggctggttta
ttgctgataa atctggagcc ggtgagcgtg 3360ggtctcgcgg tatcattgca gcactggggc
cagatggtaa gccctcccgt atcgtagtta 3420tctacacgac ggggagtcag gcaactatgg
atgaacgaaa tagacagatc gctgagatag 3480gtgcctcact gattaagcat tggtaactgt
cagaccaagt ttactcatat atactttaga 3540ttgatttaaa acttcatttt taatttaaaa
ggatctaggt gaagatcctt tttgataatc 3600tcatgaccaa aatcccttaa cgtgagtttt
cgttccactg agcgtcagac cccgtagaaa 3660agatcaaagg atcttcttga gatccttttt
ttctgcgcgt aatctgctgc ttgcaaacaa 3720aaaaaccacc gctaccagcg gtggtttgtt
tgccggatca agagctacca actctttttc 3780cgaaggtaac tggcttcagc agagcgcaga
taccaaatac tgtccttcta gtgtagccgt 3840agttaggcca ccacttcaag aactctgtag
caccgcctac atacctcgct ctgctaatcc 3900tgttaccagt ggctgctgcc agtggcgata
agtcgtgtct taccgggttg gactcaagac 3960gatagttacc ggataaggcg cagcggtcgg
gctgaacggg gggttcgtgc acacagccca 4020gcttggagcg aacgacctac accgaactga
gatacctaca gcgtgagcta tgagaaagcg 4080ccacgcttcc cgaagggaga aaggcggaca
ggtatccggt aagcggcagg gtcggaacag 4140gagagcgcac gagggagctt ccagggggaa
acgcctggta tctttatagt cctgtcgggt 4200ttcgccacct ctgacttgag cgtcgatttt
tgtgatgctc gtcagggggg cggagcctat 4260ggaaaaacgc cagcaacgcg gcctttttac
ggttcctggc cttttgctgg ccttttgctc 4320acatgttctt tcctgcgtta tcccctgatt
ctgtggataa ccgtattacc gcctttgagt 4380gagctgatac cgctcgccgc agccgaacga
ccgagcgcag cgagtcagtg agcgaggaag 4440cggaagagcg cctgatgcgg tattttctcc
ttacgcatct gtgcggtatt tcacaccgca 4500taattttgtt aaaattcgcg ttaaattttt
gttaaatcag ctcatttttt aaccaatagg 4560ccgaaatcgg caaaatccct tataaatcaa
aagaatagac cgagataggg ttgagtgttg 4620ttccagtttg gaacaagagt ccactattaa
agaacgtgga ctccaacgtc aaagggcgaa 4680aaaccgtcta tcagggcgat ggcccactac
gtgaaccatc accctaatca agttttttgg 4740ggtcgaggtg ccgtaaagca ctaaatcgga
accctaaagg gagcccccga tttagagctt 4800gacggggaaa gccggcgaac gtggcgagaa
aggaagggaa gaaagcgaaa ggagcgggcg 4860ctagggcgct ggcaagtgta gcggtcacgc
tgcgcgtaac caccacaccc gccgcgctta 4920atgcgccgct acagggcgcg tccattcgcc
attcaggctg ctatggtgca ctctcagtac 4980aatctgctct gatgccgcat agttaagcca
gtaccagtca cgtagcgata tcggagtgta 5040tacactccgc tatcgctacg tgactgggtc
atggctgcgc cccgacaccc gccaacaccc 5100gctgacgcgc cctgacgggc ttgtctgctc
ccggcatccg cttacagaca agctgtgacc 5160gtctccggga gctgcatgtg tcagaggttt
tcaccgtcat caccgaaacg cgcgaggcag 5220cagatcaatt cgcgcgcgaa ggcgaagcgg
catgcattta cgttgacacc atcgaatggt 5280gcaaaacctt tcgcggtatg gcatgatagc
gcccggaaga gagtcaattc agggtggtga 5340atgtgaaacc agtaacgtta tacgatgtcg
cagagtatgc cggtgtctct tatcagaccg 5400tttcccgcgt ggtgaaccag gccagccacg
tttctgcgaa aacgcgggaa aaagtggaag 5460cggcgatggc ggagctgaat tacattccca
accgcgtggc acaacaactg gcgggcaaac 5520agtcgttgct gattggcgtt gccacctcca
gtctggccct gcacgcgccg tcgcaaattg 5580tcgcggcgat taaatctcgc gccgatcaac
tgggtgccag cgtggtggtg tcgatggtag 5640aacgaagcgg cgtcgaagcc tgtaaagcgg
cggtgcacaa tcttctcgcg caacgcgtca 5700gtgggctgat cattaactat ccgctggatg
accaggatgc cattgctgtg gaagctgcct 5760gcactaatgt tccggcgtta tttcttgatg
tctctgacca gacacccatc aacagtatta 5820ttttctccca tgaagacggt acgcgactgg
gcgtggagca tctggtcgca ttgggtcacc 5880agcaaatcgc gctgttagcg ggcccattaa
gttctgtctc ggcgcgtctg cgtctggctg 5940gctggcataa atatctcact cgcaatcaaa
ttcagccgat agcggaacgg gaaggcgact 6000ggagtgccat gtccggtttt caacaaacca
tgcaaatgct gaatgagggc atcgttccca 6060ctgcgatgct ggttgccaac gatcagatgg
cgctgggcgc aatgcgcgcc attaccgagt 6120ccgggctgcg cgttggtgcg gatatctcgg
tagtgggata cgacgatacc gaagacagct 6180catgttatat cccgccgtta accaccatca
aacaggattt tcgcctgctg gggcaaacca 6240gcgtggaccg cttgctgcaa ctctctcagg
gccaggcggt gaagggcaat cagctgttgc 6300ccgtctcact ggtgaaaaga aaaaccaccc
tggcacccaa tacgcaaacc gcctctcccc 6360gcgcgttggc cgattcatta atgcagctgg
cacgacaggt ttcccgactg gaaagcgggc 6420agtgagcgca acgcaattaa tgtgagttag
cgcgaattga tctg 64641306553DNAArtificial
SequenceDescription of Artificial Sequence Synthetic pDEST2
polynucleotide 130ggcggtgcac aatcttctcg cgcaacgcgt cagtgggctg atcattaact
atccgctgga 60tgaccaggat gccattgctg tggaagctgc ctgcactaat gttccggcgt
tatttcttga 120tgtctctgac cagacaccca tcaacagtat tattttctcc catgaagacg
gtacgcgact 180gggcgtggag catctggtcg cattgggtca ccagcaaatc gcgctgttag
cgggcccatt 240aagttctgtc tcggcgcgtc tgcgtctggc tggctggcat aaatatctca
ctcgcaatca 300aattcagccg atagcggaac gggaaggcga ctggagtgcc atgtccggtt
ttcaacaaac 360catgcaaatg ctgaatgagg gcatcgttcc cactgcgatg ctggttgcca
acgatcagat 420ggcgctgggc gcaatgcgcg ccattaccga gtccgggctg cgcgttggtg
cggatatctc 480ggtagtggga tacgacgata ccgaagacag ctcatgttat atcccgccgt
caaccaccat 540caaacaggat tttcgcctgc tggggcaaac cagcgtggac cgcttgctgc
aactctctca 600gggccaggcg gtgaagggca atcagctgtt gcccgtctca ctggtgaaaa
gaaaaaccac 660cctggcaccc aatacgcaaa ccgcctctcc ccgcgcgttg gccgattcat
taatgcagct 720ggcacgacag gtttcccgac tggaaagcgg gcagtgagcg caacgcaatt
aatgtgagtt 780agcgcgaatt gatctggttt gacagcttat catcgactgc acggtgcacc
aatgcttctg 840gcgtcaggca gccatcggaa gctgtggtat ggctgtgcag gtcgtaaatc
actgcataat 900tcgtgtcgct caaggcgcac tcccgttctg gataatgttt tttgcgccga
catcataacg 960gttctggcaa atattctgaa atgagctgtt gacaattaat catccggtcc
gtataatctg 1020tggaattgtg agcggataac aatttcacac aggaaacaga ccatgtcgta
ctaccatcac 1080catcaccatc acggcatcac aagtttgtac aaaaaagctg aacgagaaac
gtaaaatgat 1140ataaatatca atatattaaa ttagattttg cataaaaaac agactacata
atactgtaaa 1200acacaacata tccagtcact atggcggccg ctaagttggc agcatcaccc
gacgcacttt 1260gcgccgaata aatacctgtg acggaagatc acttcgcaga ataaataaat
cctggtgtcc 1320ctgttgatac cgggaagccc tgggccaact tttggcgaaa atgagacgtt
gatcggcacg 1380taagaggttc caactttcac cataatgaaa taagatcact accgggcgta
ttttttgagt 1440tatcgagatt ttcaggagct aaggaagcta aaatggagaa aaaaatcact
ggatatacca 1500ccgttgatat atcccaatgg catcgtaaag aacattttga ggcatttcag
tcagttgctc 1560aatgtaccta taaccagacc gttcagctgg atattacggc ctttttaaag
accgtaaaga 1620aaaataagca caagttttat ccggccttta ttcacattct tgcccgcctg
atgaatgctc 1680atccggaatt ccgtatggca atgaaagacg gtgagctggt gatatgggat
agtgttcacc 1740cttgttacac cgttttccat gagcaaactg aaacgttttc atcgctctgg
agtgaatacc 1800acgacgattt ccggcagttt ctacacatat attcgcaaga tgtggcgtgt
tacggtgaaa 1860acctggccta tttccctaaa gggtttattg agaatatgtt tttcgtctca
gccaatccct 1920gggtgagttt caccagtttt gatttaaacg tggccaatat ggacaacttc
ttcgcccccg 1980ttttcaccat gggcaaatat tatacgcaag gcgacaaggt gctgatgccg
ctggcgattc 2040aggttcatca tgccgtctgt gatggcttcc atgtcggcag aatgcttaat
gaattacaac 2100agtactgcga tgagtggcag ggcggggcgt aaacgcgtgg atccggctta
ctaaaagcca 2160gataacagta tgcgtatttg cgcgctgatt tttgcggtat aagaatatat
actgatatgt 2220atacccgaag tatgtcaaaa agaggtgtgc tatgaagcag cgtattacag
tgacagttga 2280cagcgacagc tatcagttgc tcaaggcata tatgatgtca atatctccgg
tctggtaagc 2340acaaccatgc agaatgaagc ccgtcgtctg cgtgccgaac gctggaaagc
ggaaaatcag 2400gaagggatgg ctgaggtcgc ccggtttatt gaaatgaacg gctcttttgc
tgacgagaac 2460agggactggt gaaatgcagt ttaaggttta cacctataaa agagagagcc
gttatcgtct 2520gtttgtggat gtacagagtg atattattga cacgcccggg cgacggatgg
tgatccccct 2580ggccagtgca cgtctgctgt cagataaagt ctcccgtgaa ctttacccgg
tggtgcatat 2640cggggatgaa agctggcgca tgatgaccac cgatatggcc agtgtgccgg
tctccgttat 2700cggggaagaa gtggctgatc tcagccaccg cgaaaatgac atcaaaaacg
ccattaacct 2760gatgttctgg ggaatataaa tgtcaggctc ccttatacac agccagtctg
caggtcgacc 2820atagtgactg gatatgttgt gttttacagt attatgtagt ctgtttttta
tgcaaaatct 2880aatttaatat attgatattt atatcatttt acgtttctcg ttcagctttc
ttgtacaaag 2940tggtgatgcc catatgggaa ttcaaaggcc tacgtcgacg agctcactag
tcgcggccgc 3000ttctagagga tccctcgagg catgcggtac caagcttggc tgttttggcg
gatgagagaa 3060gattttcagc ctgatacaga ttaaatcaga acgcagaagc ggtctgataa
aacagaattt 3120gcctggcggc agtagcgcgg tggtcccacc tgaccccatg ccgaactcag
aagtgaaacg 3180ccgtagcgcc gatggtagtg tggggtctcc ccatgcgaga gtagggaact
gccaggcatc 3240aaataaaacg aaaggctcag tcgaaagact gggcctttcg ttttatctgt
tgtttgtcgg 3300tgaacgctct cctgagtagg acaaatccgc cgggagcgga tttgaacgtt
gcgaagcaac 3360ggcccggagg gtggcgggca ggacgcccgc cataaactgc caggcatcaa
attaagcaga 3420aggccatcct gacggatggc ctttttgcgt ttctacaaac tctttttgtt
tatttttcta 3480aatacattca aatatgtatc cgctcatgag acaataaccc tgataaatgc
ttcaataata 3540ttgaaaaagg aagagtatga gtattcaaca tttccgtgtc gcccttattc
ccttttttgc 3600ggcattttgc cttcctgttt ttgctcaccc agaaacgctg gtgaaagtaa
aagatgctga 3660agatcagttg ggtgcacgag tgggttacat cgaactggat ctcaacagcg
gtaagatcct 3720tgagagtttt cgccccgaag aacgttttcc aatgatgagc acttttaaag
ttctgctatg 3780tggcgcggta ttatcccgtg ttgacgccgg gcaagagcaa ctcggtcgcc
gcatacacta 3840ttctcagaat gacttggttg agtactcacc agtcacagaa aagcatctta
cggatggcat 3900gacagtaaga gaattatgca gtgctgccat aaccatgagt gataacactg
cggccaactt 3960acttctgaca acgatcggag gaccgaagga gctaaccgct tttttgcaca
acatggggga 4020tcatgtaact cgccttgatc gttgggaacc ggagctgaat gaagccatac
caaacgacga 4080gcgtgacacc acgatgccta cagcaatggc aacaacgttg cgcaaactat
taactggcga 4140actacttact ctagcttccc ggcaacaatt aatagactgg atggaggcgg
ataaagttgc 4200aggaccactt ctgcgctcgg cccttccggc tggctggttt attgctgata
aatctggagc 4260cggtgagcgt gggtctcgcg gtatcattgc agcactgggg ccagatggta
agccctcccg 4320tatcgtagtt atctacacga cggggagtca ggcaactatg gatgaacgaa
atagacagat 4380cgctgagata ggtgcctcac tgattaagca ttggtaactg tcagaccaag
tttactcata 4440tatactttag attgatttaa aacttcattt ttaatttaaa aggatctagg
tgaagatcct 4500ttttgataat ctcatgacca aaatccctta acgtgagttt tcgttccact
gagcgtcaga 4560ccccgtagaa aagatcaaag gatcttcttg agatcctttt tttctgcgcg
taatctgctg 4620cttgcaaaca aaaaaaccac cgctaccagc ggtggtttgt ttgccggatc
aagagctacc 4680aactcttttt ccgaaggtaa ctggcttcag cagagcgcag ataccaaata
ctgtccttct 4740agtgtagccg tagttaggcc accacttcaa gaactctgta gcaccgccta
catacctcgc 4800tctgctaatc ctgttaccag tggctgctgc cagtggcgat aagtcgtgtc
ttaccgggtt 4860ggactcaaga cgatagttac cggataaggc gcagcggtcg ggctgaacgg
ggggttcgtg 4920cacacagccc agcttggagc gaacgaccta caccgaactg agatacctac
agcgtgagct 4980atgagaaagc gccacgcttc ccgaagggag aaaggcggac aggtatccgg
taagcggcag 5040ggtcggaaca ggagagcgca cgagggagct tccaggggga aacgcctggt
atctttatag 5100tcctgtcggg tttcgccacc tctgacttga gcgtcgattt ttgtgatgct
cgtcaggggg 5160gcggagccta tggaaaaacg ccagcaacgc ggccttttta cggttcctgg
ccttttgctg 5220gccttttgct cacatgttct ttcctgcgtt atcccctgat tctgtggata
accgtattac 5280cgcctttgag tgagctgata ccgctcgccg cagccgaacg accgagcgca
gcgagtcagt 5340gagcgaggaa gcggaagagc gcctgatgcg gtattttctc cttacgcatc
tgtgcggtat 5400ttcacaccgc ataattttgt taaaattcgc gttaaatttt tgttaaatca
gctcattttt 5460taaccaatag gccgaaatcg gcaaaatccc ttataaatca aaagaataga
ccgagatagg 5520gttgagtgtt gttccagttt ggaacaagag tccactatta aagaacgtgg
actccaacgt 5580caaagggcga aaaaccgtct atcagggcga tggcccacta cgtgaaccat
caccctaatc 5640aagttttttg gggtcgaggt gccgtaaagc actaaatcgg aaccctaaag
ggagcccccg 5700atttagagct tgacggggaa agccggcgaa cgtggcgaga aaggaaggga
agaaagcgaa 5760aggagcgggc gctagggcgc tggcaagtgt agcggtcacg ctgcgcgtaa
ccaccacacc 5820cgccgcgctt aatgcgccgc tacagggcgc gtcccattcg ccattcaggc
tgctatggtg 5880cactctcagt acaatctgct ctgatgccgc atagttaagc cagtatacac
tccgctatcg 5940ctacgtgact gggtcatggc tgcgccccga cacccgccaa cacccgctga
cgcgccctga 6000cgggcttgtc tgctcccggc atccgcttac agacaagctg tgaccgtctc
cgggagctgc 6060atgtgtcaga ggttttcacc gtcatcaccg aaacgcgcga ggcagcagat
caattcgcgc 6120gcgaaggcga agcggcatgc atttacgttg acaccatcga atggtgcaaa
acctttcgcg 6180gtatggcatg atagcgcccg gaagagagtc aattcagggt ggtgaatgtg
aaaccagtaa 6240cgttatacga tgtcgcagag tatgccggtg tctcttatca gaccgtttcc
cgcgtggtga 6300accaggccag ccacgtttct gcgaaaacgc gggaaaaagt ggaagcggcg
atggcggagc 6360tgaattacat tcccaaccgc gtggcacaac aactggcggg caaacagtcg
ttgctgattg 6420gcgttgccac ctccagtctg gccctgcacg cgccgtcgca aattgtcgcg
gcgattaaat 6480ctcgcgccga tcaactgggt gccagcgtgg tggtgtcgat ggtagaacga
agcggcgtcg 6540aagcctgtaa agc
65531316823DNAArtificial SequenceDescription of Artificial
Sequence Synthetic pDEST3 polynucleotide 131acgttatcga ctgcacggtg
caccaatgct tctggcgtca ggcagccatc ggaagctgtg 60gtatggctgt gcaggtcgta
aatcactgca taattcgtgt cgctcaaggc gcactcccgt 120tctggataat gttttttgcg
ccgacatcat aacggttctg gcaaatattc tgaaatgagc 180tgttgacaat taatcatcgg
ctcgtataat gtgtggaatt gtgagcggat aacaatttca 240cacaggaaac agtattcatg
tcccctatac taggttattg gaaaattaag ggccttgtgc 300aacccactcg acttcttttg
gaatatcttg aagaaaaata tgaagagcat ttgtatgagc 360gcgatgaagg tgataaatgg
cgaaacaaaa agtttgaatt gggtttggag tttcccaatc 420ttccttatta tattgatggt
gatgttaaat taacacagtc tatggccatc atacgttata 480tagctgacaa gcacaacatg
ttgggtggtt gtccaaaaga gcgtgcagag atttcaatgc 540ttgaaggagc ggttttggat
attagatacg gtgtttcgag aattgcatat agtaaagact 600ttgaaactct caaagttgat
tttcttagca agctacctga aatgctgaaa atgttcgaag 660atcgtttatg tcataaaaca
tatttaaatg gtgatcatgt aacccatcct gacttcatgt 720tgtatgacgc tcttgatgtt
gttttataca tggacccaat gtgcctggat gcgttcccaa 780aattagtttg ttttaaaaaa
cgtattgaag ctatcccaca aattgataag tacttgaaat 840ccagcaagta tatagcatgg
cctttgcagg gctggcaagc cacgtttggt ggtggcgacc 900atcctccaaa atcggatctg
gttccgcgtg gatctcgtcg tgcatctgtt ggatccccat 960caacaagttt gtacaaaaaa
gctgaacgag aaacgtaaaa tgatataaat atcaatatat 1020taaattagat tttgcataaa
aaacagacta cataatactg taaaacacaa catatccagt 1080cactatggcg gccgctaagt
tggcagcatc acccgacgca ctttgcgccg aataaatacc 1140tgtgacggaa gatcacttcg
cagaataaat aaatcctggt gtccctgttg ataccgggaa 1200gccctgggcc aacttttggc
gaaaatgaga cgttgatcgg cacgtaagag gttccaactt 1260tcaccataat gaaataagat
cactaccggg cgtatttttt gagttatcga gattttcagg 1320agctaaggaa gctaaaatgg
agaaaaaaat cactggatat accaccgttg atatatccca 1380atggcatcgt aaagaacatt
ttgaggcatt tcagtcagtt gctcaatgta cctataacca 1440gaccgttcag ctggatatta
cggccttttt aaagaccgta aagaaaaata agcacaagtt 1500ttatccggcc tttattcaca
ttcttgcccg cctgatgaat gctcatccgg aattccgtat 1560ggcaatgaaa gacggtgagc
tggtgatatg ggatagtgtt cacccttgtt acaccgtttt 1620ccatgagcaa actgaaacgt
tttcatcgct ctggagtgaa taccacgacg atttccggca 1680gtttctacac atatattcgc
aagatgtggc gtgttacggt gaaaacctgg cctatttccc 1740taaagggttt attgagaata
tgtttttcgt ctcagccaat ccctgggtga gtttcaccag 1800ttttgattta aacgtggcca
atatggacaa cttcttcgcc cccgttttca ccatgggcaa 1860atattatacg caaggcgaca
aggtgctgat gccgctggcg attcaggttc atcatgccgt 1920ctgtgatggc ttccatgtcg
gcagaatgct taatgaatta caacagtact gcgatgagtg 1980gcagggcggg gcgtaaagat
ctggatccgg cttactaaaa gccagataac agtatgcgta 2040tttgcgcgct gatttttgcg
gtataagaat atatactgat atgtataccc gaagtatgtc 2100aaaaagaggt gtgctatgaa
gcagcgtatt acagtgacag ttgacagcga cagctatcag 2160ttgctcaagg catatatgat
gtcaatatct ccggtctggt aagcacaacc atgcagaatg 2220aagcccgtcg tctgcgtgcc
gaacgctgga aagcggaaaa tcaggaaggg atggctgagg 2280tcgcccggtt tattgaaatg
aacggctctt ttgctgacga gaacagggac tggtgaaatg 2340cagtttaagg tttacaccta
taaaagagag agccgttatc gtctgtttgt ggatgtacag 2400agtgatatta ttgacacgcc
cgggcgacgg atggtgatcc ccctggccag tgcacgtctg 2460ctgtcagata aagtctcccg
tgaactttac ccggtggtgc atatcgggga tgaaagctgg 2520cgcatgatga ccaccgatat
ggccagtgtg ccggtctccg ttatcgggga agaagtggct 2580gatctcagcc accgcgaaaa
tgacatcaaa aacgccatta acctgatgtt ctggggaata 2640taaatgtcag gctcccttat
acacagccag tctgcaggtc gaccatagtg actggatatg 2700ttgtgtttta cagtattatg
tagtctgttt tttatgcaaa atctaattta atatattgat 2760atttatatca ttttacgttt
ctcgttcagc tttcttgtac aaagtggttg atgggaattc 2820atcgtgactg actgacgatc
tgcctcgcgc gtttcggtga tgacggtgaa aacctctgac 2880acatgcagct cccggagacg
gtcacagctt gtctgtaagc ggatgccggg agcagacaag 2940cccgtcaggg cgcgtcagcg
ggtgttggcg ggtgtcgggg cgcagccatg acccagtcac 3000gtagcgatag cggagtgtat
aattcttgaa gacgaaaggg cctcgtgata cgcctatttt 3060tataggttaa tgtcatgata
ataatggttt cttagacgtc aggtggcact tttcggggaa 3120atgtgcgcgg aacccctatt
tgtttatttt tctaaataca ttcaaatatg tatccgctca 3180tgagacaata accctgataa
atgcttcaat aatattgaaa aaggaagagt atgagtattc 3240aacatttccg tgtcgccctt
attccctttt ttgcggcatt ttgccttcct gtttttgctc 3300acccagaaac gctggtgaaa
gtaaaagatg ctgaagatca gttgggtgca cgagtgggtt 3360acatcgaact ggatctcaac
agcggtaaga tccttgagag ttttcgcccc gaagaacgtt 3420ttccaatgat gagcactttt
aaagttctgc tatgtggcgc ggtattatcc cgtgttgacg 3480ccgggcaaga gcaactcggt
cgccgcatac actattctca gaatgacttg gttgagtact 3540caccagtcac agaaaagcat
cttacggatg gcatgacagt aagagaatta tgcagtgctg 3600ccataaccat gagtgataac
actgcggcca acttacttct gacaacgatc ggaggaccga 3660aggagctaac cgcttttttg
cacaacatgg gggatcatgt aactcgcctt gatcgttggg 3720aaccggagct gaatgaagcc
ataccaaacg acgagcgtga caccacgatg cctgcagcaa 3780tggcaacaac gttgcgcaaa
ctattaactg gcgaactact tactctagct tcccggcaac 3840aattaataga ctggatggag
gcggataaag ttgcaggacc acttctgcgc tcggcccttc 3900cggctggctg gtttattgct
gataaatctg gagccggtga gcgtgggtct cgcggtatca 3960ttgcagcact ggggccagat
ggtaagccct cccgtatcgt agttatctac acgacgggga 4020gtcaggcaac tatggatgaa
cgaaatagac agatcgctga gataggtgcc tcactgatta 4080agcattggta actgtcagac
caagtttact catatatact ttagattgat ttaaaacttc 4140atttttaatt taaaaggatc
taggtgaaga tcctttttga taatctcatg accaaaatcc 4200cttaacgtga gttttcgttc
cactgagcgt cagaccccgt agaaaagatc aaaggatctt 4260cttgagatcc tttttttctg
cgcgtaatct gctgcttgca aacaaaaaaa ccaccgctac 4320cagcggtggt ttgtttgccg
gatcaagagc taccaactct ttttccgaag gtaactggct 4380tcagcagagc gcagatacca
aatactgtcc ttctagtgta gccgtagtta ggccaccact 4440tcaagaactc tgtagcaccg
cctacatacc tcgctctgct aatcctgtta ccagtggctg 4500ctgccagtgg cgataagtcg
tgtcttaccg ggttggactc aagacgatag ttaccggata 4560aggcgcagcg gtcgggctga
acggggggtt cgtgcacaca gcccagcttg gagcgaacga 4620cctacaccga actgagatac
ctacagcgtg agctatgaga aagcgccacg cttcccgaag 4680ggagaaaggc ggacaggtat
ccggtaagcg gcagggtcgg aacaggagag cgcacgaggg 4740agcttccagg gggaaacgcc
tggtatcttt atagtcctgt cgggtttcgc cacctctgac 4800ttgagcgtcg atttttgtga
tgctcgtcag gggggcggag cctatggaaa aacgccagca 4860acgcggcctt tttacggttc
ctggcctttt gctggccttt tgctcacatg ttctttcctg 4920cgttatcccc tgattctgtg
gataaccgta ttaccgcctt tgagtgagct gataccgctc 4980gccgcagccg aacgaccgag
cgcagcgagt cagtgagcga ggaagcggaa gagcgcctga 5040tgcggtattt tctccttacg
catctgtgcg gtatttcaca ccgcataaat tccgacacca 5100tcgaatggtg caaaaccttt
cgcggtatgg catgatagcg cccggaagag agtcaattca 5160gggtggtgaa tgtgaaacca
gtaacgttat acgatgtcgc agagtatgcc ggtgtctctt 5220atcagaccgt ttcccgcgtg
gtgaaccagg ccagccacgt ttctgcgaaa acgcgggaaa 5280aagtggaagc ggcgatggcg
gagctgaatt acattcccaa ccgcgtggca caacaactgg 5340cgggcaaaca gtcgttgctg
attggcgttg ccacctccag tctggccctg cacgcgccgt 5400cgcaaattgt cgcggcgatt
aaatctcgcg ccgatcaact gggtgccagc gtggtggtgt 5460cgatggtaga acgaagcggc
gtcgaagcct gtaaagcggc ggtgcacaat cttctcgcgc 5520aacgcgtcag tgggctgatc
attaactatc cgctggatga ccaggatgcc attgctgtgg 5580aagctgcctg cactaatgtt
ccggcgttat ttcttgatgt ctctgaccag acacccatca 5640acagtattat tttctcccat
gaagacggta cgcgactggg cgtggagcat ctggtcgcat 5700tgggtcacca gcaaatcgcg
ctgttagcgg gcccattaag ttctgtctcg gcgcgtctgc 5760gtctggctgg ctggcataaa
tatctcactc gcaatcaaat tcagccgata gcggaacggg 5820aaggcgactg gagtgccatg
tccggttttc aacaaaccat gcaaatgctg aatgagggca 5880tcgttcccac tgcgatgctg
gttgccaacg atcagatggc gctgggcgca atgcgcgcca 5940ttaccgagtc cgggctgcgc
gttggtgcgg atatctcggt agtgggatac gacgataccg 6000aagacagctc atgttatatc
ccgccgttaa ccaccatcaa acaggatttt cgcctgctgg 6060ggcaaaccag cgtggaccgc
ttgctgcaac tctctcaggg ccaggcggtg aagggcaatc 6120agctgttgcc cgtctcactg
gtgaaaagaa aaaccaccct ggcgcccaat acgcaaaccg 6180cctctccccg cgcgttggcc
gattcattaa tgcagctggc acgacaggtt tcccgactgg 6240aaagcgggca gtgagcgcaa
cgcaattaat gtgagttagc tcactcatta ggcaccccag 6300gctttacact ttatgcttcc
ggctcgtatg ttgtgtggaa ttgtgagcgg ataacaattt 6360cacacaggaa acagctatga
ccatgattac ggattcactg gccgtcgttt tacaacgtcg 6420tgactgggaa aaccctggcg
ttacccaact taatcgcctt gcagcacatc cccctttcgc 6480cagctggcgt aatagcgaag
aggcccgcac cgatcgccct tcccaacagt tgcgcagcct 6540gaatggcgaa tggcgctttg
cctggtttcc ggcaccagaa gcggtgccgg aaagctggct 6600ggagtgcgat cttcctgagg
ccgatactgt cgtcgtcccc tcaaactggc agatgcacgg 6660ttacgatgcg cccatctaca
ccaacgtaac ctatcccatt acggtcaatc cgccgtttgt 6720tcccacggag aatccgacgg
gttgttactc gctcacattt aatgttgatg aaagctggct 6780acaggaaggc cagacgcgaa
ttatttttga tggcgttgga att 68231326964DNAArtificial
SequenceDescription of Artificial Sequence Synthetic pDEST4
polynucleotide 132ctatccgctg gatgaccagg atgccattgc tgtggaagct gcctgcacta
atgttccggc 60gttatttctt gatgtctctg accagacacc catcaacagt attattttct
cccatgaaga 120cggtacgcga ctgggcgtgg agcatctggt cgcattgggt caccagcaaa
tcgcgctgtt 180agcgggccca ttaagttctg tctcggcgcg tctgcgtctg gctggctggc
ataaatatct 240cactcgcaat caaattcagc cgatagcgga acgggaaggc gactggagtg
ccatgtccgg 300ttttcaacaa accatgcaaa tgctgaatga gggcatcgtt cccactgcga
tgctggttgc 360caacgatcag atggcgctgg gcgcaatgcg cgccattacc gagtccgggc
tgcgcgttgg 420tgcggatatc tcggtagtgg gatacgacga taccgaagac agctcatgtt
atatcccgcc 480gtcaaccacc atcaaacagg attttcgcct gctggggcaa accagcgtgg
accgcttgct 540gcaactctct cagggccagg cggtgaaggg caatcagctg ttgcccgtct
cactggtgaa 600aagaaaaacc accctggcac ccaatacgca aaccgcctct ccccgcgcgt
tggccgattc 660attaatgcag ctggcacgac aggtttcccg actggaaagc gggcagtgag
cgcaacgcaa 720ttaatgtgag ttagcgcgaa ttgatctggt ttgacagctt atcatcgact
gcacggtgca 780ccaatgcttc tggcgtcagg cagccatcgg aagctgtggt atggctgtgc
aggtcgtaaa 840tcactgcata attcgtgtcg ctcaaggcgc actcccgttc tggataatgt
tttttgcgcc 900gacatcataa cggttctggc aaatattctg aaatgagctg ttgacaatta
atcatccggt 960ccgtataatc tgtggaattg tgagcggata acaatttcac acaggaaaca
gaccatgggt 1020catcatcatc atcatcacga ttacgatatc ccaacgaccg aaaacctgta
ttttcagggc 1080gcccatatga gcgataaaat tattcacctg actgacgaca gttttgacac
ggatgtactc 1140aaagcggacg gggcgatcct cgtcgatttc tgggcagagt ggtgcggtcc
gtgcaaaatg 1200atcgccccga ttctggatga aatcgctgac gaatatcagg gcaaactgac
cgttgcaaaa 1260ctgaacatcg atcaaaaccc tggcactgcg ccgaaatatg gcatccgtgg
tatcccgact 1320ctgctgctgt tcaaaaacgg tgaagtggcg gcaaccaaag tgggtgcact
gtctaaaggt 1380cagttgaaag agttcctcga cgctaacctg gccggttctg gttctggtga
tgacgatgac 1440aaggtaccca tcacaagttt gtacaaaaaa gctgaacgag aaacgtaaaa
tgatataaat 1500atcaatatat taaattagat tttgcataaa aaacagacta cataatactg
taaaacacaa 1560catatccagt cactatggcg gccgctaagt tggcagcatc acccgacgca
ctttgcgccg 1620aataaatacc tgtgacggaa gatcacttcg cagaataaat aaatcctggt
gtccctgttg 1680ataccgggaa gccctgggcc aacttttggc gaaaatgaga cgttgatcgg
cacgtaagag 1740gttccaactt tcaccataat gaaataagat cactaccggg cgtatttttt
gagttatcga 1800gattttcagg agctaaggaa gctaaaatgg agaaaaaaat cactggatat
accaccgttg 1860atatatccca atggcatcgt aaagaacatt ttgaggcatt tcagtcagtt
gctcaatgta 1920cctataacca gaccgttcag ctggatatta cggccttttt aaagaccgta
aagaaaaata 1980agcacaagtt ttatccggcc tttattcaca ttcttgcccg cctgatgaat
gctcatccgg 2040aattccgtat ggcaatgaaa gacggtgagc tggtgatatg ggatagtgtt
cacccttgtt 2100acaccgtttt ccatgagcaa actgaaacgt tttcatcgct ctggagtgaa
taccacgacg 2160atttccggca gtttctacac atatattcgc aagatgtggc gtgttacggt
gaaaacctgg 2220cctatttccc taaagggttt attgagaata tgtttttcgt ctcagccaat
ccctgggtga 2280gtttcaccag ttttgattta aacgtggcca atatggacaa cttcttcgcc
cccgttttca 2340ccatgggcaa atattatacg caaggcgaca aggtgctgat gccgctggcg
attcaggttc 2400atcatgccgt ctgtgatggc ttccatgtcg gcagaatgct taatgaatta
caacagtact 2460gcgatgagtg gcagggcggg gcgtaaacgc gtggatccgg cttactaaaa
gccagataac 2520agtatgcgta tttgcgcgct gatttttgcg gtataagaat atatactgat
atgtataccc 2580gaagtatgtc aaaaagaggt gtgctatgaa gcagcgtatt acagtgacag
ttgacagcga 2640cagctatcag ttgctcaagg catatatgat gtcaatatct ccggtctggt
aagcacaacc 2700atgcagaatg aagcccgtcg tctgcgtgcc gaacgctgga aagcggaaaa
tcaggaaggg 2760atggctgagg tcgcccggtt tattgaaatg aacggctctt ttgctgacga
gaacagggac 2820tggtgaaatg cagtttaagg tttacaccta taaaagagag agccgttatc
gtctgtttgt 2880ggatgtacag agtgatatta ttgacacgcc cgggcgacgg atggtgatcc
ccctggccag 2940tgcacgtctg ctgtcagata aagtctcccg tgaactttac ccggtggtgc
atatcgggga 3000tgaaagctgg cgcatgatga ccaccgatat ggccagtgtg ccggtctccg
ttatcgggga 3060agaagtggct gatctcagcc accgcgaaaa tgacatcaaa aacgccatta
acctgatgtt 3120ctggggaata taaatgtcag gctcccttat acacagccag tctgcaggtc
gaccatagtg 3180actggatatg ttgtgtttta cagtattatg tagtctgttt tttatgcaaa
atctaattta 3240atatattgat atttatatca ttttacgttt ctcgttcagc tttcttgtac
aaagtggtga 3300tggggatcct ctagagtcga cctgcagtaa tcgtacaggg tagtacaaat
aaaaaaggca 3360cgtcagatga cgtgcctttt ttcttgtgag cagtaagctt ggctgttttg
gcggatgaga 3420gaagattttc agcctgatac agattaaatc agaacgcaga agcggtctga
taaaacagaa 3480tttgcctggc ggcagtagcg cggtggtccc acctgacccc atgccgaact
cagaagtgaa 3540acgccgtagc gccgatggta gtgtggggtc tccccatgcg agagtaggga
actgccaggc 3600atcaaataaa acgaaaggct cagtcgaaag actgggcctt tcgttttatc
tgttgtttgt 3660cggtgaacgc tctcctgagt aggacaaatc cgccgggagc ggatttgaac
gttgcgaagc 3720aacggcccgg agggtggcgg gcaggacgcc cgccataaac tgccaggcat
caaattaagc 3780agaaggccat cctgacggat ggcctttttg cgtttctaca aactcttttt
gtttattttt 3840ctaaatacat tcaaatatgt atccgctcat gagacaataa ccctgataaa
tgcttcaata 3900atattgaaaa aggaagagta tgagtattca acatttccgt gtcgccctta
ttcccttttt 3960tgcggcattt tgccttcctg tttttgctca cccagaaacg ctggtgaaag
taaaagatgc 4020tgaagatcag ttgggtgcac gagtgggtta catcgaactg gatctcaaca
gcggtaagat 4080ccttgagagt tttcgccccg aagaacgttt tccaatgatg agcactttta
aagttctgct 4140atgtggcgcg gtattatccc gtgttgacgc cgggcaagag caactcggtc
gccgcataca 4200ctattctcag aatgacttgg ttgagtactc accagtcaca gaaaagcatc
ttacggatgg 4260catgacagta agagaattat gcagtgctgc cataaccatg agtgataaca
ctgcggccaa 4320cttacttctg acaacgatcg gaggaccgaa ggagctaacc gcttttttgc
acaacatggg 4380ggatcatgta actcgccttg atcgttggga accggagctg aatgaagcca
taccaaacga 4440cgagcgtgac accacgatgc ctacagcaat ggcaacaacg ttgcgcaaac
tattaactgg 4500cgaactactt actctagctt cccggcaaca attaatagac tggatggagg
cggataaagt 4560tgcaggacca cttctgcgct cggcccttcc ggctggctgg tttattgctg
ataaatctgg 4620agccggtgag cgtgggtctc gcggtatcat tgcagcactg gggccagatg
gtaagccctc 4680ccgtatcgta gttatctaca cgacggggag tcaggcaact atggatgaac
gaaatagaca 4740gatcgctgag ataggtgcct cactgattaa gcattggtaa ctgtcagacc
aagtttactc 4800atatatactt tagattgatt taaaacttca tttttaattt aaaaggatct
aggtgaagat 4860cctttttgat aatctcatga ccaaaatccc ttaacgtgag ttttcgttcc
actgagcgtc 4920agaccccgta gaaaagatca aaggatcttc ttgagatcct ttttttctgc
gcgtaatctg 4980ctgcttgcaa acaaaaaaac caccgctacc agcggtggtt tgtttgccgg
atcaagagct 5040accaactctt tttccgaagg taactggctt cagcagagcg cagataccaa
atactgtcct 5100tctagtgtag ccgtagttag gccaccactt caagaactct gtagcaccgc
ctacatacct 5160cgctctgcta atcctgttac cagtggctgc tgccagtggc gataagtcgt
gtcttaccgg 5220gttggactca agacgatagt taccggataa ggcgcagcgg tcgggctgaa
cggggggttc 5280gtgcacacag cccagcttgg agcgaacgac ctacaccgaa ctgagatacc
tacagcgtga 5340gctatgagaa agcgccacgc ttcccgaagg gagaaaggcg gacaggtatc
cggtaagcgg 5400cagggtcgga acaggagagc gcacgaggga gcttccaggg ggaaacgcct
ggtatcttta 5460tagtcctgtc gggtttcgcc acctctgact tgagcgtcga tttttgtgat
gctcgtcagg 5520ggggcggagc ctatggaaaa acgccagcaa cgcggccttt ttacggttcc
tggccttttg 5580ctggcctttt gctcacatgt tctttcctgc gttatcccct gattctgtgg
ataaccgtat 5640taccgccttt gagtgagctg ataccgctcg ccgcagccga acgaccgagc
gcagcgagtc 5700agtgagcgag gaagcggaag agcgcctgat gcggtatttt ctccttacgc
atctgtgcgg 5760tatttcacac cgcataattt tgttaaaatt cgcgttaaat ttttgttaaa
tcagctcatt 5820ttttaaccaa taggccgaaa tcggcaaaat cccttataaa tcaaaagaat
agaccgagat 5880agggttgagt gttgttccag tttggaacaa gagtccacta ttaaagaacg
tggactccaa 5940cgtcaaaggg cgaaaaaccg tctatcaggg cgatggccca ctacgtgaac
catcacccta 6000atcaagtttt ttggggtcga ggtgccgtaa agcactaaat cggaacccta
aagggagccc 6060ccgatttaga gcttgacggg gaaagccggc gaacgtggcg agaaaggaag
ggaagaaagc 6120gaaaggagcg ggcgctaggg cgctggcaag tgtagcggtc acgctgcgcg
taaccaccac 6180acccgccgcg cttaatgcgc cgctacaggg cgcgtccatt cgccattcag
gctgctatgg 6240tgcactctca gtacaatctg ctctgatgcc gcatagttaa gccagtatac
actccgctat 6300cgctacgtga ctgggtcatg gctgcgcccc gacacccgcc aacacccgct
gacgcgccct 6360gacgggcttg tctgctcccg gcatccgctt acagacaagc tgtgaccgtc
tccgggagct 6420gcatgtgtca gaggttttca ccgtcatcac cgaaacgcgc gaggcagcag
atcaattcgc 6480gcgcgaaggc gaagcggcat gcatttacgt tgacaccatc gaatggtgca
aaacctttcg 6540cggtatggca tgatagcgcc cggaagagag tcaattcagg gtggtgaatg
tgaaaccagt 6600aacgttatac gatgtcgcag agtatgccgg tgtctcttat cagaccgttt
cccgcgtggt 6660gaaccaggcc agccacgttt ctgcgaaaac gcgggaaaaa gtggaagcgg
cgatggcgga 6720gctgaattac attcccaacc gcgtggcaca acaactggcg ggcaaacagt
cgttgctgat 6780tggcgttgcc acctccagtc tggccctgca cgcgccgtcg caaattgtcg
cggcgattaa 6840atctcgcgcc gatcaactgg gtgccagcgt ggtggtgtcg atggtagaac
gaagcggcgt 6900cgaagcctgt aaagcggcgg tgcacaatct tctcgcgcaa cgcgtcagtn
gggctgatca 6960ttaa
69641335957DNAArtificial SequenceDescription of Artificial
Sequence Synthetic pDEST5 polynucleotide 133aggcacccca ggctttacac
tttatgcttc cggctcgtat gttgtgtgga attgtgagcg 60gataacaatt tcacacagga
aacagctatg accatgatta cgccaagctc taatacgact 120cactataggg aaagctggta
cgcctgcagg taccggtccg gaattcccgg gtcgacgatc 180acaagtttgt acaaaaaagc
tgaacgagaa acgtaaaatg atataaatat caatatatta 240aattagattt tgcataaaaa
acagactaca taatactgta aaacacaaca tatccagtca 300ctatggcggc cgctaagttg
gcagcatcac ccgacgcact ttgcgccgaa taaatacctg 360tgacggaaga tcacttcgca
gaataaataa atcctggtgt ccctgttgat accgggaagc 420cctgggccaa cttttggcga
aaatgagacg ttgatcggca cgtaagaggt tccaactttc 480accataatga aataagatca
ctaccgggcg tattttttga gttatcgaga ttttcaggag 540ctaaggaagc taaaatggag
aaaaaaatca ctggatatac caccgttgat atatcccaat 600ggcatcgtaa agaacatttt
gaggcatttc agtcagttgc tcaatgtacc tataaccaga 660ccgttcagct ggatattacg
gcctttttaa agaccgtaaa gaaaaataag cacaagtttt 720atccggcctt tattcacatt
cttgcccgcc tgatgaatgc tcatccggaa ttccgtatgg 780caatgaaaga cggtgagctg
gtgatatggg atagtgttca cccttgttac accgttttcc 840atgagcaaac tgaaacgttt
tcatcgctct ggagtgaata ccacgacgat ttccggcagt 900ttctacacat atattcgcaa
gatgtggcgt gttacggtga aaacctggcc tatttcccta 960aagggtttat tgagaatatg
tttttcgtct cagccaatcc ctgggtgagt ttcaccagtt 1020ttgatttaaa cgtggccaat
atggacaact tcttcgcccc cgttttcacc atgggcaaat 1080attatacgca aggcgacaag
gtgctgatgc cgctggcgat tcaggttcat catgccgtct 1140gtgatggctt ccatgtcggc
agaatgctta atgaattaca acagtactgc gatgagtggc 1200agggcggggc gtaaacgcgt
ggatccggct tactaaaagc cagataacag tatgcgtatt 1260tgcgcgctga tttttgcggt
ataagaatat atactgatat gtatacccga agtatgtcaa 1320aaagaggtgt gctatgaagc
agcgtattac agtgacagtt gacagcgaca gctatcagtt 1380gctcaaggca tatatgatgt
caatatctcc ggtctggtaa gcacaaccat gcagaatgaa 1440gcccgtcgtc tgcgtgccga
acgctggaaa gcggaaaatc aggaagggat ggctgaggtc 1500gcccggttta ttgaaatgaa
cggctctttt gctgacgaga acagggactg gtgaaatgca 1560gtttaaggtt tacacctata
aaagagagag ccgttatcgt ctgtttgtgg atgtacagag 1620tgatattatt gacacgcccg
ggcgacggat ggtgatcccc ctggccagtg cacgtctgct 1680gtcagataaa gtctcccgtg
aactttaccc ggtggtgcat atcggggatg aaagctggcg 1740catgatgacc accgatatgg
ccagtgtgcc ggtctccgtt atcggggaag aagtggctga 1800tctcagccac cgcgaaaatg
acatcaaaaa cgccattaac ctgatgttct ggggaatata 1860aatgtcaggc tcccttatac
acagccagtc tgcaggtcga ccatagtgac tggatatgtt 1920gtgttttaca gtattatgta
gtctgttttt tatgcaaaat ctaatttaat atattgatat 1980ttatatcatt ttacgtttct
cgttcagctt tcttgtacaa agtggtgatc actagtcggc 2040ggccgctcta gaggatccaa
gcttacgtac gcgtgcatgc gacgtcatag ctcttctata 2100gtgtcaccta aattcaattc
actggccgtc gttttacaac gtcgtgactg ggaaaaccct 2160ggcgttaccc aacttaatcg
ccttgcagca catccccctt tcgccagctg gcgtaatagc 2220gaagaggccc gcaccgatcg
cccttcccaa cagttgcgca gcctgaatgg cgaatggacg 2280cgccctgtag cggcgcatta
agcgcggcgg gtgtggtggt tacgcgcagc gtgaccgcta 2340cacttgccag cgccctagcg
cccgctcctt tcgctttctt cccttccttt ctcgccacgt 2400tcgccggctt tccccgtcaa
gctctaaatc gggggctccc tttagggttc cgatttagtg 2460ctttacggca cctcgacccc
aaaaaacttg attagggtga tggttcacgt agtgggccat 2520cgccctgata gacggttttt
cgccctttga cgttggagtc cacgttcttt aatagtggac 2580tcttgttcca aactggaaca
acactcaacc ctatctcggt ctattctttt gatttataag 2640ggattttgcc gatttcggcc
tattggttaa aaaatgagct gatttaacaa aaatttaacg 2700cgaattttaa caaaatatta
acgtttacaa tttcaggtgg cacttttcgg ggaaatgtgc 2760gcggaacccc tatttgttta
tttttctaaa tacattcaaa tatgtatccg ctcatgagac 2820aataaccctg ataaatgctt
caataatatt gaaaaaggaa gagtatgagt attcaacatt 2880tccgtgtcgc ccttattccc
ttttttgcgg cattttgcct tcctgttttt gctcacccag 2940aaacgctggt gaaagtaaaa
gatgctgaag atcagttggg tgcacgagtg ggttacatcg 3000aactggatct caacagcggt
aagatccttg agagttttcg ccccgaagaa cgttttccaa 3060tgatgagcac ttttaaagtt
ctgctatgtg gcgcggtatt atcccgtatt gacgccgggc 3120aagagcaact cggtcgccgc
atacactatt ctcagaatga cttggttgag tactcaccag 3180tcacagaaaa gcatcttacg
gatggcatga cagtaagaga attatgcagt gctgccataa 3240ccatgagtga taacactgcg
gccaacttac ttctgacaac gatcggagga ccgaaggagc 3300taaccgcttt tttgcacaac
atgggggatc atgtaactcg ccttgatcgt tgggaaccgg 3360agctgaatga agccatacca
aacgacgagc gtgacaccac gatgcctgta gcaatggcaa 3420caacgttgcg caaactatta
actggcgaac tacttactct agcttcccgg caacaattaa 3480tagactggat ggaggcggat
aaagttgcag gaccacttct gcgctcggcc cttccggctg 3540gctggtttat tgctgataaa
tctggagccg gtgagcgtgg gtctcgcggt atcattgcag 3600cactggggcc agatggtaag
ccctcccgta tcgtagttat ctacacgacg gggagtcagg 3660caactatgga tgaacgaaat
agacagatcg ctgagatagg tgcctcactg attaagcatt 3720ggtaactgtc agaccaagtt
tactcatata tactttagat tgatttaaaa cttcattttt 3780aatttaaaag gatctaggtg
aagatccttt ttgataatct catgaccaaa atcccttaac 3840gtgagttttc gttccactga
gcgtcagacc ccgtagaaaa gatcaaagga tcttcttgag 3900atcctttttt tctgcgcgta
atctgctgct tgcaaacaaa aaaaccaccg ctaccagcgg 3960tggtttgttt gccggatcaa
gagctaccaa ctctttttcc gaaggtaact ggcttcagca 4020gagcgcagat accaaatact
gtccttctag tgtagccgta gttaggccac cacttcaaga 4080actctgtagc accgcctaca
tacctcgctc tgctaatcct gttaccagtg gctgctgcca 4140gtggcgataa gtcgtgtctt
accgggttgg actcaagacg atagttaccg gataaggcgc 4200agcggtcggg ctgaacgggg
ggttcgtgca cacagcccag cttggagcga acgacctaca 4260ccgaactgag atacctacag
cgtgagcatt gagaaagcgc cacgcttccc gaagggagaa 4320aggcggacag gtatccggta
agcggcaggg tcggaacagg agagcgcacg agggagcttc 4380cagggggaaa cgcctggtat
ctttatagtc ctgtcgggtt tcgccacctc tgacttgagc 4440gtcgattttt gtgatgctcg
tcaggggggc ggagcctatg gaaaaacgcc agcaacgcgg 4500cctttttacg gttcctggcc
ttttgctggc cttttgctca catgttcttt cctgcgttat 4560cccctgattc tgtggataac
cgtattaccg cctttgagtg agctgatacc gctcgccgca 4620gccgaacgac cgagcgcagc
gagtcagtga gcgaggaagc ggaagagcgc ccaatacgca 4680aaccgcctct ccccgcgcgt
tggccgattc attaatgcag agcttgcaat tcgcgcgcga 4740aggcgaagcg gcatttacgt
tgacaccatc gaatggcgca aaacctttcg cggtatggca 4800tgatagcgcc cggaagagag
tcaattcagg gtggtgaatg tgaaaccagt aacgttatac 4860gatgtcgcag agtatgccgg
tgtctcttat cagaccgttt cccgcgtggt gaaccaggcc 4920agccacgttt ctgcgaaaac
gcgggaaaaa gtggaagcgg cgatggcgga gctgaattac 4980attcccaacc gcgtggcaca
acaactggcg ggcaaacagt cgttgctgat tggcgttgcc 5040acctccagtc tggccctgca
cgcgccgtcg caaattgtcg cggcgattaa atctcgcgcc 5100gatcaactgg gtgccagcgt
ggtggtgtcg atggtagaac gaagcggcgt cgaagcctgt 5160aaagcggcgg tgcacaatct
tctcgcgcaa cgggtcagtg ggctgatcat taactatccg 5220ctggatgacc aggatgccat
tgctgtggaa gctgcctgca ctaatgttcc ggcgttattt 5280cttgatgtct ctgaccagac
acccatcaac agtattattt tctcccatga agacggtacg 5340cgactgggcg tggagcatct
ggtcgcattg ggtcaccagc aaatcgcgct gttagcgggc 5400ccattaagtt ctgtctcggc
gcgtctgcgt ctggctggct ggcataaata tctcactcgc 5460aatcaaattc agccgatagc
ggaacgggaa ggcgactgga gtgccatgtc cggttttcaa 5520caaaccatgc aaatgctgaa
tgagggcatc gttcccactg cgatgctggt tgccaacgat 5580cagatggcgc tgggcgcaat
gcgcgccatt accgagtccg ggctgcgcgt tggtgcggat 5640atctcggtag tgggatacga
cgataccgaa gacagctcat gttatatccc gccgtcaacc 5700accatcaaac aggattttcg
cctgctgggg caaaccagcg tggaccgctt gctgcaactc 5760tctcagggcc aggcggtgaa
gggcaatcag ctgttgcccg tctcactggt gaaaagaaaa 5820accaccctgg cgcccaatac
gcaaaccgcc tctccccgcg cgttggccga ttcattaatg 5880cagctggcac gacaggtttc
ccgactggaa agcgggcagt gagcgcaacg caattaatgt 5940gagttagctc actcatt
59571345957DNAArtificial
SequenceDescription of Artificial Sequence Synthetic pDEST6
polynucleotide 134taacgccagg gttttcccag tcacgacgtt gtaaaacgac ggccagtgaa
ttgaatttag 60gtgacactat agaagagcta tgacgtcgca tgcacgcgta cgtaagcttg
gatcctctag 120agcggccgcc gactagtgat cacaagtttg tacaaaaaag ctgaacgaga
aacgtaaaat 180gatataaata tcaatatatt aaattagatt ttgcataaaa aacagactac
ataatactgt 240aaaacacaac atatccagtc actatggcgg ccgctaagtt ggcagcatca
cccgacgcac 300tttgcgccga ataaatacct gtgacggaag atcacttcgc agaataaata
aatcctggtg 360tccctgttga taccgggaag ccctgggcca acttttggcg aaaatgagac
gttgatcggc 420acgtaagagg ttccaacttt caccataatg aaataagatc actaccgggc
gtattttttg 480agttatcgag attttcagga gctaaggaag ctaaaatgga gaaaaaaatc
actggatata 540ccaccgttga tatatcccaa tggcatcgta aagaacattt tgaggcattt
cagtcagttg 600ctcaatgtac ctataaccag accgttcagc tggatattac ggccttttta
aagaccgtaa 660agaaaaataa gcacaagttt tatccggcct ttattcacat tcttgcccgc
ctgatgaatg 720ctcatccgga attccgtatg gcaatgaaag acggtgagct ggtgatatgg
gatagtgttc 780acccttgtta caccgttttc catgagcaaa ctgaaacgtt ttcatcgctc
tggagtgaat 840accacgacga tttccggcag tttctacaca tatattcgca agatgtggcg
tgttacggtg 900aaaacctggc ctatttccct aaagggttta ttgagaatat gtttttcgtc
tcagccaatc 960cctgggtgag tttcaccagt tttgatttaa acgtggccaa tatggacaac
ttcttcgccc 1020ccgttttcac catgggcaaa tattatacgc aaggcgacaa ggtgctgatg
ccgctggcga 1080ttcaggttca tcatgccgtc tgtgatggct tccatgtcgg cagaatgctt
aatgaattac 1140aacagtactg cgatgagtgg cagggcgggg cgtaaacgcg tggatccggc
ttactaaaag 1200ccagataaca gtatgcgtat ttgcgcgctg atttttgcgg tataagaata
tatactgata 1260tgtatacccg aagtatgtca aaaagaggtg tgctatgaag cagcgtatta
cagtgacagt 1320tgacagcgac agctatcagt tgctcaaggc atatatgatg tcaatatctc
cggtctggta 1380agcacaacca tgcagaatga agcccgtcgt ctgcgtgccg aacgctggaa
agcggaaaat 1440caggaaggga tggctgaggt cgcccggttt attgaaatga acggctcttt
tgctgacgag 1500aacagggact ggtgaaatgc agtttaaggt ttacacctat aaaagagaga
gccgttatcg 1560tctgtttgtg gatgtacaga gtgatattat tgacacgccc gggcgacgga
tggtgatccc 1620cctggccagt gcacgtctgc tgtcagataa agtctcccgt gaactttacc
cggtggtgca 1680tatcggggat gaaagctggc gcatgatgac caccgatatg gccagtgtgc
cggtctccgt 1740tatcggggaa gaagtggctg atctcagcca ccgcgaaaat gacatcaaaa
acgccattaa 1800cctgatgttc tggggaatat aaatgtcagg ctcccttata cacagccagt
ctgcaggtcg 1860accatagtga ctggatatgt tgtgttttac agtattatgt agtctgtttt
ttatgcaaaa 1920tctaatttaa tatattgata tttatatcat tttacgtttc tcgttcagct
ttcttgtaca 1980aagtggtgat cgtcgacccg ggaattccgg accggtacct gcaggcgtac
cagctttccc 2040tatagtgagt cgtattagag cttggcgtaa tcatggtcat agctgtttcc
tgtgtgaaat 2100tgttatccgc tcacaattcc acacaacata cgagccggaa gcataaagtg
taaagcctgg 2160ggtgcctaat gagtgagcta actcacatta attgcgttgc gctcactgcc
cgctttccag 2220tcgggaaacc tgtcgtgcca gctgcattaa tgaatcggcc aacgcgcggg
gagaggcggt 2280ttgcgtattg ggcgccaggg tggtttttct tttcaccagt gagacgggca
acagctgatt 2340gcccttcacc gcctggccct gagagagttg cagcaagcgg tccacgctgg
tttgccccag 2400caggcgaaaa tcctgtttga tggtggttga cggcgggata taacatgagc
tgtcttcggt 2460atcgtcgtat cccactaccg agatatccgc accaacgcgc agcccggact
cggtaatggc 2520gcgcattgcg cccagcgcca tctgatcgtt ggcaaccagc atcgcagtgg
gaacgatgcc 2580ctcattcagc atttgcatgg tttgttgaaa accggacatg gcactccagt
cgccttcccg 2640ttccgctatc ggctgaattt gattgcgagt gagatattta tgccagccag
ccagacgcag 2700acgcgccgag acagaactta atgggcccgc taacagcgcg atttgctggt
gacccaatgc 2760gaccagatgc tccacgccca gtcgcgtacc gtcttcatgg gagaaaataa
tactgttgat 2820gggtgtctgg tcagagacat caagaaataa cgccggaaca ttagtgcagg
cagcttccac 2880agcaatggca tcctggtcat ccagcggata gttaatgatc agcccactga
cccgttgcgc 2940gagaagattg tgcaccgccg ctttacaggc ttcgacgccg cttcgttcta
ccatcgacac 3000caccacgctg gcacccagtt gatcggcgcg agatttaatc gccgcgacaa
tttgcgacgg 3060cgcgtgcagg gccagactgg aggtggcaac gccaatcagc aacgactgtt
tgcccgccag 3120ttgttgtgcc acgcggttgg gaatgtaatt cagctccgcc atcgccgctt
ccactttttc 3180ccgcgttttc gcagaaacgt ggctggcctg gttcaccacg cgggaaacgg
tctgataaga 3240gacaccggca tactctgcga catcgtataa cgttactggt ttcacattca
ccaccctgaa 3300ttgactctct tccgggcgct atcatgccat accgcgaaag gttttgcgcc
attcgatggt 3360gtcaacgtaa atgccgcttc gccttcgcgc gcgaattgca agctctgcat
taatgaatcg 3420gccaacgcgc ggggagaggc ggtttgcgta ttgggcgctc ttccgcttcc
tcgctcactg 3480actcgctgcg ctcggtcgtt cggctgcggc gagcggtatc agctcactca
aaggcggtaa 3540tacggttatc cacagaatca ggggataacg caggaaagaa catgtgagca
aaaggccagc 3600aaaaggccag gaaccgtaaa aaggccgcgt tgctggcgtt tttccatagg
ctccgccccc 3660ctgacgagca tcacaaaaat cgacgctcaa gtcagaggtg gcgaaacccg
acaggactat 3720aaagatacca ggcgtttccc cctggaagct ccctcgtgcg ctctcctgtt
ccgaccctgc 3780cgcttaccgg atacctgtcc gcctttctcc cttcgggaag cgtggcgctt
tctcaatgct 3840cacgctgtag gtatctcagt tcggtgtagg tcgttcgctc caagctgggc
tgtgtgcacg 3900aaccccccgt tcagcccgac cgctgcgcct tatccggtaa ctatcgtctt
gagtccaacc 3960cggtaagaca cgacttatcg ccactggcag cagccactgg taacaggatt
agcagagcga 4020ggtatgtagg cggtgctaca gagttcttga agtggtggcc taactacggc
tacactagaa 4080ggacagtatt tggtatctgc gctctgctga agccagttac cttcggaaaa
agagttggta 4140gctcttgatc cggcaaacaa accaccgctg gtagcggtgg tttttttgtt
tgcaagcagc 4200agattacgcg cagaaaaaaa ggatctcaag aagatccttt gatcttttct
acggggtctg 4260acgctcagtg gaacgaaaac tcacgttaag ggattttggt catgagatta
tcaaaaagga 4320tcttcaccta gatcctttta aattaaaaat gaagttttaa atcaatctaa
agtatatatg 4380agtaaacttg gtctgacagt taccaatgct taatcagtga ggcacctatc
tcagcgatct 4440gtctatttcg ttcatccata gttgcctgac tccccgtcgt gtagataact
acgatacggg 4500agggcttacc atctggcccc agtgctgcaa tgataccgcg agacccacgc
tcaccggctc 4560cagatttatc agcaataaac cagccagccg gaagggccga gcgcagaagt
ggtcctgcaa 4620ctttatccgc ctccatccag tctattaatt gttgccggga agctagagta
agtagttcgc 4680cagttaatag tttgcgcaac gttgttgcca ttgctacagg catcgtggtg
tcacgctcgt 4740cgtttggtat ggcttcattc agctccggtt cccaacgatc aaggcgagtt
acatgatccc 4800ccatgttgtg caaaaaagcg gttagctcct tcggtcctcc gatcgttgtc
agaagtaagt 4860tggccgcagt gttatcactc atggttatgg cagcactgca taattctctt
actgtcatgc 4920catccgtaag atgcttttct gtgactggtg agtactcaac caagtcattc
tgagaatagt 4980gtatgcggcg accgagttgc tcttgcccgg cgtcaatacg ggataatacc
gcgccacata 5040gcagaacttt aaaagtgctc atcattggaa aacgttcttc ggggcgaaaa
ctctcaagga 5100tcttaccgct gttgagatcc agttcgatgt aacccactcg tgcacccaac
tgatcttcag 5160catcttttac tttcaccagc gtttctgggt gagcaaaaac aggaaggcaa
aatgccgcaa 5220aaaagggaat aagggcgaca cggaaatgtt gaatactcat actcttcctt
tttcaatatt 5280attgaagcat ttatcagggt tattgtctca tgagcggata catatttgaa
tgtatttaga 5340aaaataaaca aataggggtt ccgcgcacat ttccccgaaa agtgccacct
gaaattgtaa 5400acgttaatat tttgttaaaa ttcgcgttaa atttttgtta aatcagctca
ttttttaacc 5460aataggccga aatcggcaaa atcccttata aatcaaaaga atagaccgag
atagggttga 5520gtgttgttcc agtttggaac aagagtccac tattaaagaa cgtggactcc
aacgtcaaag 5580ggcgaaaaac cgtctatcag ggcgatggcc cactacgtga accatcaccc
taatcaagtt 5640ttttggggtc gaggtgccgt aaagcactaa atcggaaccc taaagggagc
ccccgattta 5700gagcttgacg gggaaagccg gcgaacgtgg cgagaaagga agggaagaaa
gcgaaaggag 5760cgggcgctag ggcgctggca agtgtagcgg tcacgctgcg cgtaaccacc
acacccgccg 5820cgcttaatgc gccgctacag ggcgcgtcca ttcgccattc aggctgcgca
actgttggga 5880agggcgatcg gtgcgggcct cttcgctatt acgccagctg gcgaaagggg
gatgtgctgc 5940aaggcgatta agttggg
59571356025DNAArtificial SequenceDescription of Artificial
Sequence Synthetic pDEST7 polynucleotide 135attatcatga cattaaccta
taaaaatagg cgtagtacga ggccctttca ctcattagat 60gcatgtcgtt acataactta
cggtaaatgg cccgcctggc tgaccgccca acgacccccg 120cccattgacg tcaataatga
cgtatgttcc catagtaacg ccaataggga ctttccattg 180acgtcaatgg gtggagtatt
tacggtaaac tgcccacttg gcagtacatc aagtgtatca 240tatgccaagt acgcccccta
ttgacgtcaa tgacggtaaa tggcccgcct ggcattatgc 300ccagtacatg accttatggg
actttcctac ttggcagtac atctacgtat tagtcatcgc 360tattaccatg gtgatgcggt
tttggcagta catcaatggg cgtggatagc ggtttgactc 420acggggattt ccaagtctcc
accccattga cgtcaatggg agtttgtttt ggcaccaaaa 480tcaacgggac tttccaaaat
gtcgtaacaa ctccgcccca ttgacgcaaa tgggcggtag 540gcgtgtacgg tgggaggtct
atataagcag agctcgttta gtgaaccgtc agatcgcctg 600gagacgccat ccacgctgtt
ttgacctcca tagaagacac cgggaccgat ccagcctccg 660gactctagcc taggccgcgg
agcggataac aatttcacac aggaaacagc tatgaccatt 720aggcctttgc aaaaagctat
ttaggtgaca ctatagaagg tacgcctgca ggtaccggat 780cacaagtttg tacaaaaaag
ctgaacgaga aacgtaaaat gatataaata tcaatatatt 840aaattagatt ttgcataaaa
aacagactac ataatactgt aaaacacaac atatccagtc 900actatggcgg ccgcattagg
caccccaggc tttacacttt atgcttccgg ctcgtataat 960gtgtggattt tgagttagga
tccgtcgaga ttttcaggag ctaaggaagc taaaatggag 1020aaaaaaatca ctggatatac
caccgttgat atatcccaat ggcatcgtaa agaacatttt 1080gaggcatttc agtcagttgc
tcaatgtacc tataaccaga ccgttcagct ggatattacg 1140gcctttttaa agaccgtaaa
gaaaaataag cacaagtttt atccggcctt tattcacatt 1200cttgcccgcc tgatgaatgc
tcatccggaa ttccgtatgg caatgaaaga cggtgagctg 1260gtgatatggg atagtgttca
cccttgttac accgttttcc atgagcaaac tgaaacgttt 1320tcatcgctct ggagtgaata
ccacgacgat ttccggcagt ttctacacat atattcgcaa 1380gatgtggcgt gttacggtga
aaacctggcc tatttcccta aagggtttat tgagaatatg 1440tttttcgtct cagccaatcc
ctgggtgagt ttcaccagtt ttgatttaaa cgtggccaat 1500atggacaact tcttcgcccc
cgttttcacc atgggcaaat attatacgca aggcgacaag 1560gtgctgatgc cgctggcgat
tcaggttcat catgccgtct gtgatggctt ccatgtcggc 1620agaatgctta atgaattaca
acagtactgc gatgagtggc agggcggggc gtaaacgcgt 1680ggatccggct tactaaaagc
cagataacag tatgcgtatt tgcgcgctga tttttgcggt 1740ataagaatat atactgatat
gtatacccga agtatgtcaa aaagaggtgt gctatgaagc 1800agcgtattac agtgacagtt
gacagcgaca gctatcagtt gctcaaggca tatatgatgt 1860caatatctcc ggtctggtaa
gcacaaccat gcagaatgaa gcccgtcgtc tgcgtgccga 1920acgctggaaa gcggaaaatc
aggaagggat ggctgaggtc gcccggttta ttgaaatgaa 1980cggctctttt gctgacgaga
acagggactg gtgaaatgca gtttaaggtt tacacctata 2040aaagagagag ccgttatcgt
ctgtttgtgg atgtacagag tgatattatt gacacgcccg 2100ggcgacggat ggtgatcccc
ctggccagtg cacgtctgct gtcagataaa gtctcccgtg 2160aactttaccc ggtggtgcat
atcggggatg aaagctggcg catgatgacc accgatatgg 2220ccagtgtgcc ggtctccgtt
atcggggaag aagtggctga tctcagccac cgcgaaaatg 2280acatcaaaaa cgccattaac
ctgatgttct ggggaatata aatgtcaggc tcccttatac 2340acagccagtc tgcaggtcga
ccatagtgac tggatatgtt gtgttttaca gtattatgta 2400gtctgttttt tatgcaaaat
ctaatttaat atattgatat ttatatcatt ttacgtttct 2460cgttcagctt tcttgtacaa
agtggtgatc gcgtgcatgc gacgtcatag ctctctccct 2520atagtgagtc gtattataag
ctaggcactg gccgtcgttt tacaacgtcg tgactgggaa 2580aactgctagc ttgggatctt
tgtgaaggaa ccttacttct gtggtgtgac ataattggac 2640aaactaccta cagagattta
aagctctaag gtaaatataa aatttttaag tgtataatgt 2700gttaaactag ctgcatatgc
ttgctgcttg agagttttgc ttactgagta tgatttatga 2760aaatattata cacaggagct
agtgattcta attgtttgtg tattttagat tcacagtccc 2820aaggctcatt tcaggcccct
cagtcctcac agtctgttca tgatcataat cagccatacc 2880acatttgtag aggttttact
tgctttaaaa aacctcccac acctccccct gaacctgaaa 2940cataaaatga atgcaattgt
tgttgttaac ttgtttattg cagcttataa tggttacaaa 3000taaagcaata gcatcacaaa
tttcacaaat aaagcatttt tttcactgca ttctagttgt 3060ggtttgtcca aactcatcaa
tgtatcttat catgtctgga tcgatcctgc attaatgaat 3120cggccaacgc gcggggagag
gcggtttgcg tattggctgg cgtaatagcg aagaggcccg 3180caccgatcgc ccttcccaac
agttgcgcag cctgaatggc gaatgggacg cgccctgtag 3240cggcgcatta agcgcggcgg
gtgtggtggt tacgcgcagc gtgaccgcta cacttgccag 3300cgccctagcg cccgctcctt
tcgctttctt cccttccttt ctcgccacgt tcgccggctt 3360tccccgtcaa gctctaaatc
gggggctccc tttagggttc cgatttagtg ctttacggca 3420cctcgacccc aaaaaacttg
attagggtga tggttcacgt agtgggccat cgccctgata 3480gacggttttt cgccctttga
cgttggagtc cacgttcttt aatagtggac tcttgttcca 3540aactggaaca acactcaacc
ctatctcggt ctattctttt gatttataag ggattttgcc 3600gatttcggcc tattggttaa
aaaatgagct gatttaacaa aaatttaacg cgaattttaa 3660caaaatatta acgtttacaa
tttcaggtgg cacttttcgg ggaaatgtgc gcggaacccc 3720tatttgttta tttttctaaa
tacattcaaa tatgtatccg ctcatgccag gtcttggact 3780ggtgagaacg gcttgctcgg
cagcttcgat gtgtgctgga gggagaataa aggtctaaga 3840tgtgcgatag agggaagtcg
cattgaatta tgtgctgtgt agggatcgct ggtatcaaat 3900atgtgtgccc acccctggca
tgagacaata accctgataa atgcttcaat aatattgaaa 3960aaggaagagt atgagtattc
aacatttccg tgtcgccctt attccctttt ttgcggcatt 4020ttgccttcct gtttttgctc
acccagaaac gctggtgaaa gtaaaagatg ctgaagatca 4080gttgggtgca cgagtgggtt
acatcgaact ggatctcaac agcggtaaga tccttgagag 4140ttttcgcccc gaagaacgtt
ttccaatgat gagcactttt aaagttctgc tatgtggcgc 4200ggtattatcc cgtattgacg
ccgggcaaga gcaactcggt cgccgcatac actattctca 4260gaatgacttg gttgagtact
caccagtcac agaaaagcat cttacggatg gcatgacagt 4320aagagaatta tgcagtgctg
ccataaccat gagtgataac actgcggcca acttacttct 4380gacaacgatc ggaggaccga
aggagctaac cgcttttttg cacaacatgg gggatcatgt 4440aactcgcctt gatcgttggg
aaccggagct gaatgaagcc ataccaaacg acgagcgtga 4500caccacgatg cctgtagcaa
tggcaacaac gttgcgcaaa ctattaactg gcgaactact 4560tactctagct tcccggcaac
aattaataga ctggatggag gcggataaag ttgcaggacc 4620acttctgcgc tcggcccttc
cggctggctg gtttattgct gataaatctg gagccggtga 4680gcgtgggtct cgcggtatca
ttgcagcact ggggccagat ggtaagccct cccgtatcgt 4740agttatctac acgacgggga
gtcaggcaac tatggatgaa cgaaatagac agatcgctga 4800gataggtgcc tcactgatta
agcattggta actgtcagac caagtttact catatatact 4860ttagattgat ttaaaacttc
atttttaatt taaaaggatc taggtgaaga tcctttttga 4920taatctcatg ccataacttc
gtataatgta tgctatacga agttatggca tgaccaaaat 4980cccttaacgt gagttttcgt
tccactgagc gtcagacccc gtagaaaaga tcaaaggatc 5040ttcttgagat cctttttttc
tgcgcgtaat ctgctgcttg caaacaaaaa aaccaccgct 5100accagcggtg gtttgtttgc
cggatcaaga gctaccaact ctttttccga aggtaactgg 5160cttcagcaga gcgcagatac
caaatactgt ccttctagtg tagccgtagt taggccacca 5220cttcaagaac tctgtagcac
cgcctacata cctcgctctg ctaatcctgt taccagtggc 5280tgctgccagt ggcgataagt
cgtgtcttac cgggttggac tcaagacgat agttaccgga 5340taaggcgcag cggtcgggct
gaacgggggg ttcgtgcaca cagcccagct tggagcgaac 5400gacctacacc gaactgagat
acctacagcg tgagcattga gaaagcgcca cgcttcccga 5460agggagaaag gcggacaggt
atccggtaag cggcagggtc ggaacaggag agcgcacgag 5520ggagcttcca gggggaaacg
cctggtatct ttatagtcct gtcgggtttc gccacctctg 5580acttgagcgt cgatttttgt
gatgctcgtc aggggggcgg agcctatgga aaaacgccag 5640caacgcggcc tttttacggt
tcctggcctt ttgctggcct tttgctcaca tgttctttcc 5700tgcgttatcc cctgattctg
tggataaccg tattaccgcc tttgagtgag ctgataccgc 5760tcgccgcagc cgaacgaccg
agcgcagcga gtcagtgagc gaggaagcgg aagagcgccc 5820aatacgcaaa ccgcctctcc
ccgcgcgttg gccgattcat taatgcagag cttgcaattc 5880gcgcgttttt caatattatt
gaagcattta tcagggttat tgtctcatga gcggatacat 5940atttgaatgt atttagaaaa
ataaacaaat aggggttccg cgcacatttc cccgaaaagt 6000gccacctgac gtctaagaaa
ccatt 60251366526DNAArtificial
SequenceDescription of Artificial Sequence Synthetic pDEST8
polynucleotide 136cgtatactcc ggaatattaa tagatcatgg agataattaa aatgataacc
atctcgcaaa 60taaataagta ttttactgtt ttcgtaacag ttttgtaata aaaaaaccta
taaatattcc 120ggattattca taccgtccca ccatcgggcg cggatcatca caagtttgta
caaaaaagct 180gaacgagaaa cgtaaaatga tataaatatc aatatattaa attagatttt
gcataaaaaa 240cagactacat aatactgtaa aacacaacat atccagtcac tatggcggcc
gctaagttgg 300cagcatcacc cgacgcactt tgcgccgaat aaatacctgt gacggaagat
cacttcgcag 360aataaataaa tcctggtgtc cctgttgata ccgggaagcc ctgggccaac
ttttggcgaa 420aatgagacgt tgatcggcac gtaagaggtt ccaactttca ccataatgaa
ataagatcac 480taccgggcgt attttttgag ttatcgagat tttcaggagc taaggaagct
aaaatggaga 540aaaaaatcac tggatatacc accgttgata tatcccaatg gcatcgtaaa
gaacattttg 600aggcatttca gtcagttgct caatgtacct ataaccagac cgttcagctg
gatattacgg 660cctttttaaa gaccgtaaag aaaaataagc acaagtttta tccggccttt
attcacattc 720ttgcccgcct gatgaatgct catccggaat tccgtatggc aatgaaagac
ggtgagctgg 780tgatatggga tagtgttcac ccttgttaca ccgttttcca tgagcaaact
gaaacgtttt 840catcgctctg gagtgaatac cacgacgatt tccggcagtt tctacacata
tattcgcaag 900atgtggcgtg ttacggtgaa aacctggcct atttccctaa agggtttatt
gagaatatgt 960ttttcgtctc agccaatccc tgggtgagtt tcaccagttt tgatttaaac
gtggccaata 1020tggacaactt cttcgccccc gttttcacca tgggcaaata ttatacgcaa
ggcgacaagg 1080tgctgatgcc gctggcgatt caggttcatc atgccgtctg tgatggcttc
catgtcggca 1140gaatgcttaa tgaattacaa cagtactgcg atgagtggca gggcggggcg
taaacgcgtg 1200gatccggctt actaaaagcc agataacagt atgcgtattt gcgcgctgat
ttttgcggta 1260taagaatata tactgatatg tatacccgaa gtatgtcaaa aagaggtgtg
ctatgaagca 1320gcgtattaca gtgacagttg acagcgacag ctatcagttg ctcaaggcat
atatgatgtc 1380aatatctccg gtctggtaag cacaaccatg cagaatgaag cccgtcgtct
gcgtgccgaa 1440cgctggaaag cggaaaatca ggaagggatg gctgaggtcg cccggtttat
tgaaatgaac 1500ggctcttttg ctgacgagaa cagggactgg tgaaatgcag tttaaggttt
acacctataa 1560aagagagagc cgttatcgtc tgtttgtgga tgtacagagt gatattattg
acacgcccgg 1620gcgacggatg gtgatccccc tggccagtgc acgtctgctg tcagataaag
tctcccgtga 1680actttacccg gtggtgcata tcggggatga aagctggcgc atgatgacca
ccgatatggc 1740cagtgtgccg gtctccgtta tcggggaaga agtggctgat ctcagccacc
gcgaaaatga 1800catcaaaaac gccattaacc tgatgttctg gggaatataa atgtcaggct
cccttataca 1860cagccagtct gcaggtcgac catagtgact ggatatgttg tgttttacag
tattatgtag 1920tctgtttttt atgcaaaatc taatttaata tattgatatt tatatcattt
tacgtttctc 1980gttcagcttt cttgtacaaa gtggtgatag cttgtcgaga agtactagag
gatcataatc 2040agccatacca catttgtaga ggttttactt gctttaaaaa acctcccaca
cctccccctg 2100aacctgaaac ataaaatgaa tgcaattgtt gttgttaact tgtttattgc
agcttataat 2160ggttacaaat aaagcaatag catcacaaat ttcacaaata aagcattttt
ttcactgcat 2220tctagttgtg gtttgtccaa actcatcaat gtatcttatc atgtctggat
ctgatcactg 2280cttgagccta ggagatccga accagataag tgaaatctag ttccaaacta
ttttgtcatt 2340tttaattttc gtattagctt acgacgctac acccagttcc catctatttt
gtcactcttc 2400cctaaataat ccttaaaaac tccatttcca cccctcccag ttcccaacta
ttttgtccgc 2460ccacagcggg gcatttttct tcctgttatg tttttaatca aacatcctgc
caactccatg 2520tgacaaaccg tcatcttcgg ctactttttc tctgtcacag aatgaaaatt
tttctgtcat 2580ctcttcgtta ttaatgtttg taattgactg aatatcaacg cttatttgca
gcctgaatgg 2640cgaatggacg cgccctgtag cggcgcatta agcgcggcgg gtgtggtggt
tacgcgcagc 2700gtgaccgcta cacttgccag cgccctagcg cccgctcctt tcgctttctt
cccttccttt 2760ctcgccacgt tcgccggctt tccccgtcaa gctctaaatc gggggctccc
tttagggttc 2820cgatttagtg ctttacggca cctcgacccc aaaaaacttg attagggtga
tggttcacgt 2880agtgggccat cgccctgata gacggttttt cgccctttga cgttggagtc
cacgttcttt 2940aatagtggac tcttgttcca aactggaaca acactcaacc ctatctcggt
ctattctttt 3000gatttataag ggattttgcc gatttcggcc tattggttaa aaaatgagct
gatttaacaa 3060aaatttaacg cgaattttaa caaaatatta acgtttacaa tttcaggtgg
cacttttcgg 3120ggaaatgtgc gcggaacccc tatttgttta tttttctaaa tacattcaaa
tatgtatccg 3180ctcatgagac aataaccctg ataaatgctt caataatatt gaaaaaggaa
gagtatgagt 3240attcaacatt tccgtgtcgc ccttattccc ttttttgcgg cattttgcct
tcctgttttt 3300gctcacccag aaacgctggt gaaagtaaaa gatgctgaag atcagttggg
tgcacgagtg 3360ggttacatcg aactggatct caacagcggt aagatccttg agagttttcg
ccccgaagaa 3420cgttttccaa tgatgagcac ttttaaagtt ctgctatgtg gcgcggtatt
atcccgtatt 3480gacgccgggc aagagcaact cggtcgccgc atacactatt ctcagaatga
cttggttgag 3540tactcaccag tcacagaaaa gcatcttacg gatggcatga cagtaagaga
attatgcagt 3600gctgccataa ccatgagtga taacactgcg gccaacttac ttctgacaac
gatcggagga 3660ccgaaggagc taaccgcttt tttgcacaac atgggggatc atgtaactcg
ccttgatcgt 3720tgggaaccgg agctgaatga agccatacca aacgacgagc gtgacaccac
gatgcctgta 3780gcaatggcaa caacgttgcg caaactatta actggcgaac tacttactct
agcttcccgg 3840caacaattaa tagactggat ggaggcggat aaagttgcag gaccacttct
gcgctcggcc 3900cttccggctg gctggtttat tgctgataaa tctggagccg gtgagcgtgg
gtctcgcggt 3960atcattgcag cactggggcc agatggtaag ccctcccgta tcgtagttat
ctacacgacg 4020gggagtcagg caactatgga tgaacgaaat agacagatcg ctgagatagg
tgcctcactg 4080attaagcatt ggtaactgtc agaccaagtt tactcatata tactttagat
tgatttaaaa 4140cttcattttt aatttaaaag gatctaggtg aagatccttt ttgataatct
catgaccaaa 4200atcccttaac gtgagttttc gttccactga gcgtcagacc ccgtagaaaa
gatcaaagga 4260tcttcttgag atcctttttt tctgcgcgta atctgctgct tgcaaacaaa
aaaaccaccg 4320ctaccagcgg tggtttgttt gccggatcaa gagctaccaa ctctttttcc
gaaggtaact 4380ggcttcagca gagcgcagat accaaatact gtccttctag tgtagccgta
gttaggccac 4440cacttcaaga actctgtagc accgcctaca tacctcgctc tgctaatcct
gttaccagtg 4500gctgctgcca gtggcgataa gtcgtgtctt accgggttgg actcaagacg
atagttaccg 4560gataaggcgc agcggtcggg ctgaacgggg ggttcgtgca cacagcccag
cttggagcga 4620acgacctaca ccgaactgag atacctacag cgtgagcatt gagaaagcgc
cacgcttccc 4680gaagggagaa aggcggacag gtatccggta agcggcaggg tcggaacagg
agagcgcacg 4740agggagcttc cagggggaaa cgcctggtat ctttatagtc ctgtcgggtt
tcgccacctc 4800tgacttgagc gtcgattttt gtgatgctcg tcaggggggc ggagcctatg
gaaaaacgcc 4860agcaacgcgg cctttttacg gttcctggcc ttttgctggc cttttgctca
catgttcttt 4920cctgcgttat cccctgattc tgtggataac cgtattaccg cctttgagtg
agctgatacc 4980gctcgccgca gccgaacgac cgagcgcagc gagtcagtga gcgaggaagc
ggaagagcgc 5040ctgatgcggt attttctcct tacgcatctg tgcggtattt cacaccgcag
accagccgcg 5100taacctggca aaatcggtta cggttgagta ataaatggat gccctgcgta
agcgggtgtg 5160ggcggacaat aaagtcttaa actgaacaaa atagatctaa actatgacaa
taaagtctta 5220aactagacag aatagttgta aactgaaatc agtccagtta tgctgtgaaa
aagcatactg 5280gacttttgtt atggctaaag caaactcttc attttctgaa gtgcaaattg
cccgtcgtat 5340taaagagggg cgtggccaag ggcatggtaa agactatatt cgcggcgttg
tgacaattta 5400ccgaacaact ccgcggccgg gaagccgatc tcggcttgaa cgaattgtta
ggtggcggta 5460cttgggtcga tatcaaagtg catcacttct tcccgtatgc ccaactttgt
atagagagcc 5520actgcgggat cgtcaccgta atctgcttgc acgtagatca cataagcacc
aagcgcgttg 5580gcctcatgct tgaggagatt gatgagcgcg gtggcaatgc cctgcctccg
gtgctcgccg 5640gagactgcga gatcatagat atagatctca ctacgcggct gctcaaacct
gggcagaacg 5700taagccgcga gagcgccaac aaccgcttct tggtcgaagg cagcaagcgc
gatgaatgtc 5760ttactacgga gcaagttccc gaggtaatcg gagtccggct gatgttggga
gtaggtggct 5820acgtctccga actcacgacc gaaaagatca agagcagccc gcatggattt
gacttggtca 5880gggccgagcc tacatgtgcg aatgatgccc atacttgagc cacctaactt
tgttttaggg 5940cgactgccct gctgcgtaac atcgttgctg ctgcgtaaca tcgttgctgc
tccataacat 6000caaacatcga cccacggcgt aacgcgcttg ctgcttggat gcccgaggca
tagactgtac 6060aaaaaaacag tcataacaag ccatgaaaac cgccactgcg ccgttaccac
cgctgcgttc 6120ggtcaaggtt ctggaccagt tgcgtgagcg catacgctac ttgcattaca
gtttacgaac 6180cgaacaggct tatgtcaact gggttcgtgc cttcatccgt ttccacggtg
tgcgtcaccc 6240ggcaaccttg ggcagcagcg aagtcgaggc atttctgtcc tggctggcga
acgagcgcaa 6300ggtttcggtc tccacgcatc gtcaggcatt ggcggccttg ctgttcttct
acggcaaggt 6360gctgtgcacg gatctgccct ggcttcagga gatcggaaga cctcggccgt
cgcggcgctt 6420gccggtggtg ctgaccccgg atgaagtggt tcgcatcctc ggttttctgg
aaggcgagca 6480tcgtttgttc gcccaggact ctagctatag ttctagtggt tggcta
652613712464DNAArtificial SequenceDescription of Artificial
Sequence Synthetic pDEST9 polynucleotide 137agcaagtggt tccggacagg
cttgggggcc gaactggagg tggcactaac atctaggtat 60gaggtagagg gctgcaaaag
tatcctcata gccatggcca ccttggcgag ggacattaag 120gcgtttaaga aattgagagg
acctgttata cacctctacg gcggtcctag attggtgcgt 180taatacacag aattctgatt
ggatcccggt ccgaagcgcg ctttcccatc acaagtttgt 240acaaaaaagc tgaacgagaa
acgtaaaatg atataaatat caatatatta aattagattt 300tgcataaaaa acagactaca
taatactgta aaacacaaca tatccagtca ctatggcggc 360cgctaagttg gcagcatcac
ccgacgcact ttgcgccgaa taaatacctg tgacggaaga 420tcacttcgca gaataaataa
atcctggtgt ccctgttgat accgggaagc cctgggccaa 480cttttggcga aaatgagacg
ttgatcggca cgtaagaggt tccaactttc accataatga 540aataagatca ctaccgggcg
tattttttga gttatcgaga ttttcaggag ctaaggaagc 600taaaatggag aaaaaaatca
ctggatatac caccgttgat atatcccaat ggcatcgtaa 660agaacatttt gaggcatttc
agtcagttgc tcaatgtacc tataaccaga ccgttcagct 720ggatattacg gcctttttaa
agaccgtaaa gaaaaataag cacaagtttt atccggcctt 780tattcacatt cttgcccgcc
tgatgaatgc tcatccggaa ttccgtatgg caatgaaaga 840cggtgagctg gtgatatggg
atagtgttca cccttgttac accgttttcc atgagcaaac 900tgaaacgttt tcatcgctct
ggagtgaata ccacgacgat ttccggcagt ttctacacat 960atattcgcaa gatgtggcgt
gttacggtga aaacctggcc tatttcccta aagggtttat 1020tgagaatatg tttttcgtct
cagccaatcc ctgggtgagt ttcaccagtt ttgatttaaa 1080cgtggccaat atggacaact
tcttcgcccc cgttttcacc atgggcaaat attatacgca 1140aggcgacaag gtgctgatgc
cgctggcgat tcaggttcat catgccgtct gtgatggctt 1200ccatgtcggc agaatgctta
atgaattaca acagtactgc gatgagtggc agggcggggc 1260gtaaagatct ggatccggct
tactaaaagc cagataacag tatgcgtatt tgcgcgctga 1320tttttgcggt ataagaatat
atactgatat gtatacccga agtatgtcaa aaagaggtgt 1380gctatgaagc agcgtattac
agtgacagtt gacagcgaca gctatcagtt gctcaaggca 1440tatatgatgt caatatctcc
ggtctggtaa gcacaaccat gcagaatgaa gcccgtcgtc 1500tgcgtgccga acgctggaaa
gcggaaaatc aggaagggat ggctgaggtc gcccggttta 1560ttgaaatgaa cggctctttt
gctgacgaga acagggactg gtgaaatgca gtttaaggtt 1620tacacctata aaagagagag
ccgttatcgt ctgtttgtgg atgtacagag tgatattatt 1680gacacgcccg ggcgacggat
ggtgatcccc ctggccagtg cacgtctgct gtcagataaa 1740gtctcccgtg aactttaccc
ggtggtgcat atcggggatg aaagctggcg catgatgacc 1800accgatatgg ccagtgtgcc
ggtctccgtt atcggggaag aagtggctga tctcagccac 1860cgcgaaaatg acatcaaaaa
cgccattaac ctgatgttct ggggaatata aatgtcaggc 1920tcccttatac acagccagtc
tgcaggtcga ccatagtgac tggatatgtt gtgttttaca 1980gtattatgta gtctgttttt
tatgcaaaag tgctaattta atatattgat atttatatca 2040ttttacgttt ctcgttcagc
tttcttgtac aaagtggtga tgggaactcg agttcactag 2100tcgatcccgc ggccgctttc
gaacctaggc aagcatgcgg gcccagtggg taattaattg 2160aattacatcc ctacgcaaac
gttttacggc cgccggtggc gcccgcgccc ggcggcccgt 2220ccttggccgt tgcaggccac
tccggtggct cccgtcgtcc ccgacttcca ggcccagcag 2280atgcagcaac tcatcagcgc
cgtaaatgcg ctgacaatga gacagaacgc aattgctcct 2340gctaggagct taattcgacg
aataattgga tttttatttt attttgcaat tggtttttaa 2400tatttccaaa aaaaaaaaaa
aaaaaaaaaa aaaaaaaaaa aaaaaaaaaa aaaaaaaaaa 2460aaaaaaaaaa aaaaaaacta
gaaatcgcga tttctagtct gcattaatga atcggccaac 2520gcgcggggag aggcggtttg
cgtattgggc gctcttccgc ttcctcgctc actgactcgc 2580tgcgctcggt cgttcggctg
cggcgagcgg tatcagctca ctcaaaggcg gtaatacggt 2640tatccacaga atcaggggat
aacgcaggaa agaacatgtg agcaaaaggc cagcaaaagg 2700ccaggaaccg taaaaaggcc
gcgttgctgg cgtttttcca taggctccgc ccccctgacg 2760agcatcacaa aaatcgacgc
tcaagtcaga ggtggcgaaa cccgacagga ctataaagat 2820accaggcgtt tccccctgga
agctccctcg tgcgctctcc tgttccgacc ctgccgctta 2880ccggatacct gtccgccttt
ctcccttcgg gaagcgtggc gctttctcaa tgctcgcgct 2940gtaggtatct cagttcggtg
taggtcgttc gctccaagct gggctgtgtg cacgaacccc 3000ccgttcagcc cgaccgctgc
gccttatccg gtaactatcg tcttgagtcc aacccggtaa 3060gacacgactt atcgccactg
gcagcagcca ctggtaacag gattagcaga gcgaggtatg 3120taggcggtgc tacagagttc
ttgaagtggt ggcctaacta cggctacact agaaggacag 3180tatttggtat ctgcgctctg
ctgaagccag ttaccttcgg aaaaagagtt ggtagctctt 3240gatccggcaa acaaaccacc
gctggtagcg gtggtttttt tgtttgcaag cagcagatta 3300cgcgcagaaa aaaaggatct
caagaagatc ctttgatctt ttctacgggg tctgacgctc 3360agtggaacga aaactcacgt
taagggattt tggtcatgag attatcaaaa aggatcttca 3420cctagatcct tttaaattaa
aaatgaagtt ttaaatcaat ctaaagtata tatgagtaaa 3480cttggtctga cagttaccaa
tgcttaatca gtgaggcacc tatctcagcg atctgtctat 3540ttcgttcatc catagttgcc
tgactccccg tcgtgtagat aactacgata cgggagggct 3600taccatctgg ccccagtgct
gcaatgatac cgcgagaccc acgctcaccg gctccagatt 3660tatcagcaat aaaccagcca
gccggaaggg ccgagcgcag aagtggtcct gcaactttat 3720ccgcctccat ccagtctatt
aattgttgcc gggaagctag agtaagtagt tcgccagtta 3780atagtttgcg caacgttgtt
gccattgcta caggcatcgt ggtgtcacgc tcgtcgtttg 3840gtatggcttc attcagctcc
ggttcccaac gatcaaggcg agttacatga tcccccatgt 3900tgtgcaaaaa agcggttagc
tccttcggtc ctccgatcgt tgtcagaagt aagttggccg 3960cagtgttatc actcatggtt
atggcagcac tgcataattc tcttactgtc atgccatccg 4020taagatgctt ttctgtgact
ggtgagtact caaccaagtc attctgagaa tagtgtatgc 4080ggcgaccgag ttgctcttgc
ccggcgtcaa tacgggataa taccgcgcca catagcagaa 4140ctttaaaagt gctcatcatt
ggaaaacgtt cttcggggcg aaaactctca aggatcttac 4200cgctgttgag atccagttcg
atgtaaccca ctcgtgcacc caactgatct tcagcatctt 4260ttactttcac cagcgtttct
gggtgagcaa aaacaggaag gcaaaatgcc gcaaaaaagg 4320gaataagggc gacacggaaa
tgttgaatac tcatactctt cctttttcaa tattattgaa 4380gcatttatca gggttattgt
ctcatgagcg gatacatatt tgaatgtatt tagaaaaata 4440aacaaatagg ggttccgcgc
acatttcccc gaaaagtgcc acctgacgtc taagaaacca 4500ttattatcat gacattaacc
tataaaaata ggcgtatcac gaggcccttt cgtctcgcgc 4560gtttcggtga tgacggtgaa
aacctctgac acatgcagct cccggagacg gtcacagctt 4620ctgtctaagc ggatgccggg
agcagacaag cccgtcaggg cgcgtcagcg ggtgttggcg 4680ggtgtcgggg ctggcttaac
tatgcggcat cagagcagat tgtactgaga gtgcaccata 4740tcgacgctct cccttatgcg
actcctgcat taggaagcag cccagtacta ggttgaggcc 4800gttgagcacc gccgccgcaa
ggaatggtgc atgcaaggag atggcgccca acagtccccc 4860ggccacgggg cctgccacca
tacccacgcc gaaacaagcg ctcatgagcc cgaagtggcg 4920agcccgatct tccccatcgg
tgatgtcggc gatataggcg ccagcaaccg cacctgtggc 4980gccggtgatg ccggccacga
tgcgtccggc gtagaggatc tggctagcga tgaccctgct 5040gattggttcg ctgaccattt
ccggggtgcg gaacggcgtt accagaaact cagaaggttc 5100gtccaaccaa accgactctg
acggcagttt acgagagaga tgatagggtc tgcttcagta 5160agccagatgc tacacaatta
ggcttgtaca tattgtcgtt agaacgcggc tacaattaat 5220acataacctt atgtatcata
cacatacgat ttaggtgaca ctatagatgg cggatgtgtg 5280acatacacga cgccaaaaga
ttttgttcca gctcctgcca cctccgctac gcgagagatt 5340aaccacccac gatggccgcc
aaagtgcatg ttgatattga ggctgacagc ccattcatca 5400agtctttgca gaaggcattt
ccgtcgttcg aggtggagtc attgcaggtc acaccaaatg 5460accatgcaaa tgccagagca
ttttcgcacc tggctaccaa attgatcgag caggagactg 5520acaaagacac actcatcttg
gatatcggca gtgcgccttc caggagaatg atgtctacgc 5580acaaatacca ctgcgtatgc
cctatgcgca gcgcagaaga ccccgaaagg ctcgatagct 5640acgcaaagaa actggcagcg
gcctccggga aggtgctgga tagagagatc gcaggaaaaa 5700tcaccgacct gcagaccgtc
atggctacgc cagacgctga atctcctacc ttttgcctgc 5760atacagacgt cacgtgtcgt
acggcagccg aagtggccgt ataccaggac gtgtatgctg 5820tacatgcacc aacatcgctg
taccatcagg cgatgaaagg tgtcagaacg gcgtattgga 5880ttgggtttga caccaccccg
tttatgtttg acgcgctagc aggcgcgtat ccaacctacg 5940ccacaaactg ggccgacgag
caggtgttac aggccaggaa cataggactg tgtgcagcat 6000ccttgactga gggaagactc
ggcaaactgt ccattctccg caagaagcaa ttgaaacctt 6060gcgacacagt catgttctcg
gtaggatcta cattgtacac tgagagcaga aagctactga 6120ggagctggca cttaccctcc
gtattccacc tgaaaggtaa acaatccttt acctgtaggt 6180gcgataccat cgtatcatgt
gaagggtacg tagttaagaa aatcactatg tgccccggcc 6240tgtacggtaa aacggtaggg
tacgccgtga cgtatcacgc ggagggattc ctagtgtgca 6300agaccacaga cactgtcaaa
ggagaaagag tctcattccc tgtatgcacc tacgtcccct 6360caaccatctg tgatcaaatg
actggcatac tagcgaccga cgtcacaccg gaggacgcac 6420agaagttgtt agtgggattg
aatcagagga tagttgtgaa cggaagaaca cagcgaaaca 6480ctaacacgat gaagaactat
ctgcttccga ttgtggccgt cgcatttagc aagtgggcga 6540gggaatacaa ggcagacctt
gatgatgaaa aacctctggg tgtccgagag aggtcactta 6600cttgctgctg cttgtgggca
tttaaaacga ggaagatgca caccatgtac aagaaaccag 6660acacccagac aatagtgaag
gtgccttcag agtttaactc gttcgtcatc ccgagcctat 6720ggtctacagg cctcgcaatc
ccagtcagat cacgcattaa gatgcttttg gccaagaaga 6780ccaagcgaga gttaatacct
gttctcgacg cgtcgtcagc cagggatgct gaacaagagg 6840agaaggagag gttggaggcc
gagctgacta gagaagcctt accacccctc gtccccatcg 6900cgccggcgga gacgggagtc
gtcgacgtcg acgttgaaga actagagtat cacgcaggtg 6960caggggtcgt ggaaacacct
cgcagcgcgt tgaaagtcac cgcacagccg aacgacgtac 7020tactaggaaa ttacgtagtt
ctgtccccgc agaccgtgct caagagctcc aagttggccc 7080ccgtgcaccc tctagcagag
caggtgaaaa taataacaca taacgggagg gccggcggtt 7140accaggtcga cggatatgac
ggcagggtcc tactaccatg tggatcggcc attccggtcc 7200ctgagtttca ggctttgagc
gagagcgcca ctatggtgta caacgaaagg gagttcgtca 7260acaggaaact ataccatatt
gccgttcacg gaccctcgct gaacaccgac gaggagaact 7320acgagaaagt cagagctgaa
agaactgacg ccgagtacgt gttcgacgta gataaaaaat 7380gctgcgtcaa gagagaggaa
gcgtcgggtt tggtgttggt gggagagcta accaaccccc 7440cgttccatga attcgcctac
gaagggctga agatcaggcc gtcggcacca tataagacta 7500cagtagtagg agtctttggg
gttccgggat caggcaagtc tgctattatt aagagcctcg 7560tgaccaaaca cgatctggtc
accagcggca agaaggagaa ctgccaggaa atagttaacg 7620acgtgaagaa gcaccgcggg
aaggggacaa gtagggaaaa cagtgactcc atcctgctaa 7680acgggtgtcg tcgtgccgtg
gacatcctat atgtggacga ggctttcgct tgccattccg 7740gtactctgct ggccctaatt
gctcttgtta aacctcggag caaagtggtg ttatgcggag 7800accccaagca atgcggattc
ttcaatatga tgcagcttaa ggtgaacttc aaccacaaca 7860tctgcactga agtatgtcat
aaaagtatat ccagacgttg cacgcgtcca gtcacggcca 7920tcgtgtctac gttgcactac
ggaggcaaga tgcgcacgac caacccgtgc aacaaaccca 7980taatcataga caccacagga
cagaccaagc ccaagccagg agacatcgtg ttaacatgct 8040tccgaggctg ggcaaagcag
ctgcagttgg actaccgtgg acacgaagtc atgacagcag 8100cagcatctca gggcctcacc
cgcaaagggg tatacgccgt aaggcagaag gtgaatgaaa 8160atcccttgta tgcccctgcg
tcggagcacg tgaatgtact gctgacgcgc actgaggata 8220ggctggtgtg gaaaacgctg
gccggcgatc cctggattaa ggtcctatca aacattccac 8280agggtaactt tacggccaca
ttggaagaat ggcaagaaga acacgacaaa ataatgaagg 8340tgattgaagg accggctgcg
cctgtggacg cgttccagaa caaagcgaac gtgtgttggg 8400cgaaaagcct ggtgcctgtc
ctggacactg ccggaatcag attgacagca gaggagtgga 8460gcaccataat tacagcattt
aaggaggaca gagcttactc tccagtggtg gccttgaatg 8520aaatttgcac caagtactat
ggagttgacc tggacagtgg cctgttttct gccccgaagg 8580tgtccctgta ttacgagaac
aaccactggg ataacagacc tggtggaagg atgtatggat 8640tcaatgccgc aacagctgcc
aggctggaag ctagacatac cttcctgaag gggcagtggc 8700atacgggcaa gcaggcagtt
atcgcagaaa gaaaaatcca accgctttct gtgctggaca 8760atgtaattcc tatcaaccgc
aggctgccgc acgccctggt ggctgagtac aagacggtta 8820aaggcagtag ggttgagtgg
ctggtcaata aagtaagagg gtaccacgtc ctgctggtga 8880gtgagtacaa cctggctttg
cctcgacgca gggtcacttg gttgtcaccg ctgaatgtca 8940caggcgccga taggtgctac
gacctaagtt taggactgcc ggctgacgcc ggcaggttcg 9000acttggtctt tgtgaacatt
cacacggaat tcagaatcca ccactaccag cagtgtgtcg 9060accacgccat gaagctgcag
atgcttgggg gagatgcgct acgactgcta aaacccggcg 9120gcatcttgat gagagcttac
ggatacgccg ataaaatcag cgaagccgtt gtttcctcct 9180taagcagaaa gttctcgtct
gcaagagtgt tgcgcccgga ttgtgtcacc agcaatacag 9240aagtgttctt gctgttctcc
aactttgaca acggaaagag accctctacg ctacaccaga 9300tgaataccaa gctgagtgcc
gtgtatgccg gagaagccat gcacacggcc gggtgtgcac 9360catcctacag agttaagaga
gcagacatag ccacgtgcac agaagcggct gtggttaacg 9420cagctaacgc ccgtggaact
gtaggggatg gcgtatgcag ggccgtggcg aagaaatggc 9480cgtcagcctt taagggagca
gcaacaccag tgggcacaat taaaacagtc atgtgcggct 9540cgtaccccgt catccacgct
gtagcgccta atttctctgc cacgactgaa gcggaagggg 9600accgcgaatt ggccgctgtc
taccgggcag tggccgccga agtaaacaga ctgtcactga 9660gcagcgtagc catcccgctg
ctgtccacag gagtgttcag cggcggaaga gataggctgc 9720agcaatccct caaccatcta
ttcacagcaa tggacgccac ggacgctgac gtgaccatct 9780actgcagaga caaaagttgg
gagaagaaaa tccaggaagc cattgacatg aggacggctg 9840tggagttgct caatgatgac
gtggagctga ccacagactt ggtgagagtg cacccggaca 9900gcagcctggt gggtcgtaag
ggctacagta ccactgacgg gtcgctgtac tcgtactttg 9960aaggtacgaa attcaaccag
gctgctattg atatggcaga gatactgacg ttgtggccca 10020gactgcaaga ggcaaacgaa
cagatatgcc tatacgcgct gggcgaaaca atggacaaca 10080tcagatccaa atgtccggtg
aacgattccg attcatcaac acctcccagg acagtgccct 10140gcctgtgccg ctacgcaatg
acagcagaac ggatcgcccg ccttaggtca caccaagtta 10200aaagcatggt ggtttgctca
tcttttcccc tcccgaaata ccatgtagat ggggtgcaga 10260aggtaaagtg cgagaaggtt
ctcctgttcg acccgacggt accttcagtg gttagtccgc 10320ggaagtatgc cgcatctacg
acggaccact cagatcggtc gttacgaggg tttgacttgg 10380actggaccac cgactcgtct
tccactgcca gcgataccat gtcgctaccc agtttgcagt 10440cgtgtgacat cgactcgatc
tacgagccaa tggctcccat agtagtgacg gctgacgtac 10500accctgaacc cgcaggcatc
gcggacctgg cggcagatgt gcaccctgaa cccgcagacc 10560atgtggacct ggagaacccg
attcctccac cgcgcccgaa gagagctgca taccttgcct 10620cccgcgcggc ggagcgaccg
gtgccggcgc cgagaaagcc gacgcctgcc ccaaggactg 10680cgtttaggaa caagctgcct
ttgacgttcg gcgactttga cgagcacgag gtcgatgcgt 10740tggcctccgg gattactttc
ggagacttcg acgacgtcct gcgactaggc cgcgcgggtg 10800catatatttt ctcctcggac
actggcagcg gacatttaca acaaaaatcc gttaggcagc 10860acaatctcca gtgcgcacaa
ctggatgcgg tccaggagga gaaaatgtac ccgccaaaat 10920tggatactga gagggagaag
ctgttgctgc tgaaaatgca gatgcaccca tcggaggcta 10980ataagagtcg ataccagtct
cgcaaagtgg agaacatgaa agccacggtg gtggacaggc 11040tcacatcggg ggccagattg
tacacgggag cggacgtagg ccgcatacca acatacgcgg 11100ttcggtaccc ccgccccgtg
tactccccta ccgtgatcga aagattctca agccccgatg 11160tagcaatcgc agcgtgcaac
gaatacctat ccagaaatta cccaacagtg gcgtcgtacc 11220agataacaga tgaatacgac
gcatacttgg acatggttga cgggtcggat agttgcttgg 11280acagagcgac attctgcccg
gcgaagctcc ggtgctaccc gaaacatcat gcgtaccacc 11340agccgactgt acgcagtgcc
gtcccgtcac cctttcagaa cacactacag aacgtgctag 11400cggctgccac caagagaaac
tgcaacgtca cgcaaatgcg agaactaccc accatggact 11460cggcagtgtt caacgtggag
tgcttcaagc gctatgcctg ctccggagaa tattgggaag 11520aatatgctaa acaacctatc
cggataacca ctgagaacat cactacctat gtgaccaaat 11580tgaaaggccc gaaagctgct
gccttgttcg ctaagaccca caacttggtt ccgctgcagg 11640aggttcccat ggacagattc
acggtcgaca tgaaacgaga tgtcaaagtc actccaggga 11700cgaaacacac agaggaaaga
cccaaagtcc aggtaattca agcagcggag ccattggcga 11760ccgcttacct gtgcggcatc
cacagggaat tagtaaggag actaaatgct gtgttacgcc 11820ctaacgtgca cacattgttt
gatatgtcgg ccgaagactt tgacgcgatc atcgcctctc 11880acttccaccc aggagacccg
gttctagaga cggacattgc atcattcgac aaaagccagg 11940acgactcctt ggctcttaca
ggtttaatga tcctcgaaga tctaggggtg gatcagtacc 12000tgctggactt gatcgaggca
gcctttgggg aaatatccag ctgtcaccta ccaactggca 12060cgcgcttcaa gttcggagct
atgatgaaat cgggcatgtt tctgactttg tttattaaca 12120ctgttttgaa catcaccata
gcaagcaggg tactggagca gagactcact gactccgcct 12180gtgcggcctt catcggcgac
gacaacatcg ttcacggagt gatctccgac aagctgatgg 12240cggagaggtg cgcgtcgtgg
gtcaacatgg aggtgaagat cattgacgct gtcatgggcg 12300aaaaaccccc atatttttgt
gggggattca tagtttttga cagcgtcaca cagaccgcct 12360gccgtgtttc agacccactt
aagcgcctgt tcaagttggg taagccgcta acagctgaag 12420acaagcagga cgaagacagg
cgacgagcac tgagtgacga ggtt 124641386708DNAArtificial
SequenceDescription of Artificial Sequence Synthetic pDEST10
polynucleotide 138ccccggatga agtggttcgc atcctcggtt ttctggaagg cgagcatcgt
ttgttcgccc 60aggactctag ctatagttct agtggttggc tacgtatact ccggaatatt
aatagatcat 120ggagataatt aaaatgataa ccatctcgca aataaataag tattttactg
ttttcgtaac 180agttttgtaa taaaaaaacc tataaatatt ccggattatt cataccgtcc
caccatcggg 240cgcggatctc ggtccgaaac catgtcgtac taccatcacc atcaccatca
cgattacgat 300atcccaacga ccgaaaacct gtattttcag ggcatcacaa gtttgtacaa
aaaagctgaa 360cgagaaacgt aaaatgatat aaatatcaat atattaaatt agattttgca
taaaaaacag 420actacataat actgtaaaac acaacatatc cagtcactat ggcggccgct
aagttggcag 480catcacccga cgcactttgc gccgaataaa tacctgtgac ggaagatcac
ttcgcagaat 540aaataaatcc tggtgtccct gttgataccg ggaagccctg ggccaacttt
tggcgaaaat 600gagacgttga tcggcacgta agaggttcca actttcacca taatgaaata
agatcactac 660cgggcgtatt ttttgagtta tcgagatttt caggagctaa ggaagctaaa
atggagaaaa 720aaatcactgg atataccacc gttgatatat cccaatggca tcgtaaagaa
cattttgagg 780catttcagtc agttgctcaa tgtacctata accagaccgt tcagctggat
attacggcct 840ttttaaagac cgtaaagaaa aataagcaca agttttatcc ggcctttatt
cacattcttg 900cccgcctgat gaatgctcat ccggaattcc gtatggcaat gaaagacggt
gagctggtga 960tatgggatag tgttcaccct tgttacaccg ttttccatga gcaaactgaa
acgttttcat 1020cgctctggag tgaataccac gacgatttcc ggcagtttct acacatatat
tcgcaagatg 1080tggcgtgtta cggtgaaaac ctggcctatt tccctaaagg gtttattgag
aatatgtttt 1140tcgtctcagc caatccctgg gtgagtttca ccagttttga tttaaacgtg
gccaatatgg 1200acaacttctt cgcccccgtt ttcaccatgg gcaaatatta tacgcaaggc
gacaaggtgc 1260tgatgccgct ggcgattcag gttcatcatg ccgtctgtga tggcttccat
gtcggcagaa 1320tgcttaatga attacaacag tactgcgatg agtggcaggg cggggcgtaa
acgcgtggat 1380ccggcttact aaaagccaga taacagtatg cgtatttgcg cgctgatttt
tgcggtataa 1440gaatatatac tgatatgtat acccgaagta tgtcaaaaag aggtgtgcta
tgaagcagcg 1500tattacagtg acagttgaca gcgacagcta tcagttgctc aaggcatata
tgatgtcaat 1560atctccggtc tggtaagcac aaccatgcag aatgaagccc gtcgtctgcg
tgccgaacgc 1620tggaaagcgg aaaatcagga agggatggct gaggtcgccc ggtttattga
aatgaacggc 1680tcttttgctg acgagaacag ggactggtga aatgcagttt aaggtttaca
cctataaaag 1740agagagccgt tatcgtctgt ttgtggatgt acagagtgat attattgaca
cgcccgggcg 1800acggatggtg atccccctgg ccagtgcacg tctgctgtca gataaagtct
cccgtgaact 1860ttacccggtg gtgcatatcg gggatgaaag ctggcgcatg atgaccaccg
atatggccag 1920tgtgccggtc tccgttatcg gggaagaagt ggctgatctc agccaccgcg
aaaatgacat 1980caaaaacgcc attaacctga tgttctgggg aatataaatg tcaggctccc
ttatacacag 2040ccagtctgca ggtcgaccat agtgactgga tatgttgtgt tttacagtat
tatgtagtct 2100gttttttatg caaaatctaa tttaatatat tgatatttat atcattttac
gtttctcgtt 2160cagctttctt gtacaaagtg gtgatgccat ggatccggaa ttcaaaggcc
tacgtcgacg 2220agctcaacta gtgcggccgc tttcgaatct agagcctgca gtctcgaggc
atgcggtacc 2280aagcttgtcg agaagtacta gaggatcata atcagccata ccacatttgt
agaggtttta 2340cttgctttaa aaaacctccc acacctcccc ctgaacctga aacataaaat
gaatgcaatt 2400gttgttgtta acttgtttat tgcagcttat aatggttaca aataaagcaa
tagcatcaca 2460aatttcacaa ataaagcatt tttttcactg cattctagtt gtggtttgtc
caaactcatc 2520aatgtatctt atcatgtctg gatctgatca ctgcttgagc ctaggagatc
cgaaccagat 2580aagtgaaatc tagttccaaa ctattttgtc atttttaatt ttcgtattag
cttacgacgc 2640tacacccagt tcccatctat tttgtcactc ttccctaaat aatccttaaa
aactccattt 2700ccacccctcc cagttcccaa ctattttgtc cgcccacagc ggggcatttt
tcttcctgtt 2760atgtttttaa tcaaacatcc tgccaactcc atgtgacaaa ccgtcatctt
cggctacttt 2820ttctctgtca cagaatgaaa atttttctgt catctcttcg ttattaatgt
ttgtaattga 2880ctgaatatca acgcttattt gcagcctgaa tggcgaatgg gacgcgccct
gtagcggcgc 2940attaagcgcg gcgggtgtgg tggttacgcg cagcgtgacc gctacacttg
ccagcgccct 3000agcgcccgct cctttcgctt tcttcccttc ctttctcgcc acgttcgccg
gctttccccg 3060tcaagctcta aatcgggggc tccctttagg gttccgattt agtgctttac
ggcacctcga 3120ccccaaaaaa cttgattagg gtgatggttc acgtagtggg ccatcgccct
gatagacggt 3180ttttcgccct ttgacgttgg agtccacgtt ctttaatagt ggactcttgt
tccaaactgg 3240aacaacactc aaccctatct cggtctattc ttttgattta taagggattt
tgccgatttc 3300ggcctattgg ttaaaaaatg agctgattta acaaaaattt aacgcgaatt
ttaacaaaat 3360attaacgttt acaatttcag gtggcacttt tcggggaaat gtgcgcggaa
cccctatttg 3420tttatttttc taaatacatt caaatatgta tccgctcatg agacaataac
cctgataaat 3480gcttcaataa tattgaaaaa ggaagagtat gagtattcaa catttccgtg
tcgcccttat 3540tccctttttt gcggcatttt gccttcctgt ttttgctcac ccagaaacgc
tggtgaaagt 3600aaaagatgct gaagatcagt tgggtgcacg agtgggttac atcgaactgg
atctcaacag 3660cggtaagatc cttgagagtt ttcgccccga agaacgtttt ccaatgatga
gcacttttaa 3720agttctgcta tgtggcgcgg tattatcccg tattgacgcc gggcaagagc
aactcggtcg 3780ccgcatacac tattctcaga atgacttggt tgagtactca ccagtcacag
aaaagcatct 3840tacggatggc atgacagtaa gagaattatg cagtgctgcc ataaccatga
gtgataacac 3900tgcggccaac ttacttctga caacgatcgg aggaccgaag gagctaaccg
cttttttgca 3960caacatgggg gatcatgtaa ctcgccttga tcgttgggaa ccggagctga
atgaagccat 4020accaaacgac gagcgtgaca ccacgatgcc tgtagcaatg gcaacaacgt
tgcgcaaact 4080attaactggc gaactactta ctctagcttc ccggcaacaa ttaatagact
ggatggaggc 4140ggataaagtt gcaggaccac ttctgcgctc ggcccttccg gctggctggt
ttattgctga 4200taaatctgga gccggtgagc gtgggtctcg cggtatcatt gcagcactgg
ggccagatgg 4260taagccctcc cgtatcgtag ttatctacac gacggggagt caggcaacta
tggatgaacg 4320aaatagacag atcgctgaga taggtgcctc actgattaag cattggtaac
tgtcagacca 4380agtttactca tatatacttt agattgattt aaaacttcat ttttaattta
aaaggatcta 4440ggtgaagatc ctttttgata atctcatgac caaaatccct taacgtgagt
tttcgttcca 4500ctgagcgtca gaccccgtag aaaagatcaa aggatcttct tgagatcctt
tttttctgcg 4560cgtaatctgc tgcttgcaaa caaaaaaacc accgctacca gcggtggttt
gtttgccgga 4620tcaagagcta ccaactcttt ttccgaaggt aactggcttc agcagagcgc
agataccaaa 4680tactgtcctt ctagtgtagc cgtagttagg ccaccacttc aagaactctg
tagcaccgcc 4740tacatacctc gctctgctaa tcctgttacc agtggctgct gccagtggcg
ataagtcgtg 4800tcttaccggg ttggactcaa gacgatagtt accggataag gcgcagcggt
cgggctgaac 4860ggggggttcg tgcacacagc ccagcttgga gcgaacgacc tacaccgaac
tgagatacct 4920acagcgtgag cattgagaaa gcgccacgct tcccgaaggg agaaaggcgg
acaggtatcc 4980ggtaagcggc agggtcggaa caggagagcg cacgagggag cttccagggg
gaaacgcctg 5040gtatctttat agtcctgtcg ggtttcgcca cctctgactt gagcgtcgat
ttttgtgatg 5100ctcgtcaggg gggcggagcc tatggaaaaa cgccagcaac gcggcctttt
tacggttcct 5160ggccttttgc tggccttttg ctcacatgtt ctttcctgcg ttatcccctg
attctgtgga 5220taaccgtatt accgcctttg agtgagctga taccgctcgc cgcagccgaa
cgaccgagcg 5280cagcgagtca gtgagcgagg aagcggaaga gcgcctgatg cggtattttc
tccttacgca 5340tctgtgcggt atttcacacc gcagaccagc cgcgtaacct ggcaaaatcg
gttacggttg 5400agtaataaat ggatgccctg cgtaagcggg tgtgggcgga caataaagtc
ttaaactgaa 5460caaaatagat ctaaactatg acaataaagt cttaaactag acagaatagt
tgtaaactga 5520aatcagtcca gttatgctgt gaaaaagcat actggacttt tgttatggct
aaagcaaact 5580cttcattttc tgaagtgcaa attgcccgtc gtattaaaga ggggcgtggc
caagggcatg 5640gtaaagacta tattcgcggc gttgtgacaa tttaccgaac aactccgcgg
ccgggaagcc 5700gatctcggct tgaacgaatt gttaggtggc ggtacttggg tcgatatcaa
agtgcatcac 5760ttcttcccgt atgcccaact ttgtatagag agccactgcg ggatcgtcac
cgtaatctgc 5820ttgcacgtag atcacataag caccaagcgc gttggcctca tgcttgagga
gattgatgag 5880cgcggtggca atgccctgcc tccggtgctc gccggagact gcgagatcat
agatatagat 5940ctcactacgc ggctgctcaa acctgggcag aacgtaagcc gcgagagcgc
caacaaccgc 6000ttcttggtcg aaggcagcaa gcgcgatgaa tgtcttacta cggagcaagt
tcccgaggta 6060atcggagtcc ggctgatgtt gggagtaggt ggctacgtct ccgaactcac
gaccgaaaag 6120atcaagagca gcccgcatgg atttgacttg gtcagggccg agcctacatg
tgcgaatgat 6180gcccatactt gagccaccta actttgtttt agggcgactg ccctgctgcg
taacatcgtt 6240gctgctgcgt aacatcgttg ctgctccata acatcaaaca tcgacccacg
gcgtaacgcg 6300cttgctgctt ggatgcccga ggcatagact gtacaaaaaa acagtcataa
caagccatga 6360aaaccgccac tgcgccgtta ccaccgctgc gttcggtcaa ggttctggac
cagttgcgtg 6420agcgcatacg ctacttgcat tacagtttac gaaccgaaca ggcttatgtc
aactgggttc 6480gtgccttcat ccgtttccac ggtgtgcgtc acccggcaac cttgggcagc
agcgaagtcg 6540aggcatttct gtcctggctg gcgaacgagc gcaaggtttc ggtctccacg
catcgtcagg 6600cattggcggc cttgctgttc ttctacggca aggtgctgtg cacggatctg
ccctggcttc 6660aggagatcgg aagacctcgg ccgtcgcggc gcttgccggt ggtgctga
67081397026DNAArtificial SequenceDescription of Artificial
Sequence Synthetic pDEST11 polynucleotide 139cgagtttacc actccctatc
agtgatagag aaaagtgaaa gtcgagttta ccactcccta 60tcagtgatag agaaaagtga
aagtcgagtt taccactccc tatcagtgat agagaaaagt 120gaaagtcgag tttaccactc
cctatcagtg atagagaaaa gtgaaagtcg agtttaccac 180tccctatcag tgatagagaa
aagtgaaagt cgagtttacc actccctatc agtgatagag 240aaaagtgaaa gtcgagttta
ccactcccta tcagtgatag agaaaagtga aagtcgagct 300cggtacccgg gtcgagtagg
cgtgtacggt gggaggccta tataagcaga gctcgtttag 360tgaaccgtca gatcgcctgg
agacgccatc cacgctgttt tgacctccat agaagacacc 420gggaccgatc cagcctccgc
ggccccgaat tcgagctcgg tacccgggga tcctctagag 480tcgaggtcga cggtatcgat
aagcttgata tcaacaagtt tgtacaaaaa agctgaacga 540gaaacgtaaa atgatataaa
tatcaatata ttaaattaga ttttgcataa aaaacagact 600acataatact gtaaaacaca
acatatccag tcactatggc ggccgctaag ttggcagcat 660cacccgacgc actttgcgcc
gaataaatac ctgtgacgga agatcacttc gcagaataaa 720taaatcctgg tgtccctgtt
gataccggga agccctgggc caacttttgg cgaaaatgag 780acgttgatcg gcacgtaaga
ggttccaact ttcaccataa tgaaataaga tcactaccgg 840gcgtattttt tgagttatcg
agattttcag gagctaagga agctaaaatg gagaaaaaaa 900tcactggata taccaccgtt
gatatatccc aatggcatcg taaagaacat tttgaggcat 960ttcagtcagt tgctcaatgt
acctataacc agaccgttca gctggatatt acggcctttt 1020taaagaccgt aaagaaaaat
aagcacaagt tttatccggc ctttattcac attcttgccc 1080gcctgatgaa tgctcatccg
gaattccgta tggcaatgaa agacggtgag ctggtgatat 1140gggatagtgt tcacccttgt
tacaccgttt tccatgagca aactgaaacg ttttcatcgc 1200tctggagtga ataccacgac
gatttccggc agtttctaca catatattcg caagatgtgg 1260cgtgttacgg tgaaaacctg
gcctatttcc ctaaagggtt tattgagaat atgtttttcg 1320tctcagccaa tccctgggtg
agtttcacca gttttgattt aaacgtggcc aatatggaca 1380acttcttcgc ccccgttttc
accatgggca aatattatac gcaaggcgac aaggtgctga 1440tgccgctggc gattcaggtt
catcatgccg tctgtgatgg cttccatgtc ggcagaatgc 1500ttaatgaatt acaacagtac
tgcgatgagt ggcagggcgg ggcgtaaaga tctggatccg 1560gcttactaaa agccagataa
cagtatgcgt atttgcgcgc tgatttttgc ggtataagaa 1620tatatactga tatgtatacc
cgaagtatgt caaaaagagg tgtgctatga agcagcgtat 1680tacagtgaca gttgacagcg
acagctatca gttgctcaag gcatatatga tgtcaatatc 1740tccggtctgg taagcacaac
catgcagaat gaagcccgtc gtctgcgtgc cgaacgctgg 1800aaagcggaaa atcaggaagg
gatggctgag gtcgcccggt ttattgaaat gaacggctct 1860tttgctgacg agaacaggga
ctggtgaaat gcagtttaag gtttacacct ataaaagaga 1920gagccgttat cgtctgtttg
tggatgtaca gagtgatatt attgacacgc ccgggcgacg 1980gatggtgatc cccctggcca
gtgcacgtct gctgtcagat aaagtctccc gtgaacttta 2040cccggtggtg catatcgggg
atgaaagctg gcgcatgatg accaccgata tggccagtgt 2100gccggtctcc gttatcgggg
aagaagtggc tgatctcagc caccgcgaaa atgacatcaa 2160aaacgccatt aacctgatgt
tctggggaat ataaatgtca ggctccctta tacacagcca 2220gtctgcaggt cgaccatagt
gactggatat gttgtgtttt acagtattat gtagtctgtt 2280ttttatgcaa aatctaattt
aatatattga tatttatatc attttacgtt tctcgttcag 2340ctttcttgta caaagtggtt
gatatcgaat tcctgcagcc cgggggatcc actagttcta 2400gagcactgcg atgagtggca
gggcggggcg taattttttt aaggcagtta ttggtgccct 2460taaacgcctg gtgctacgcc
tgaataagtg ataataagcg gatgaatggc agaaattcgc 2520cggatctttg tgaaggaacc
ttacttctgt ggtgtgacat aattggacaa actacctaca 2580gagatttaaa gctctaaggt
aaatataaaa tttttaagtg tataatgtgt taaactactg 2640attctaattg tttgtgtatt
ttagattcca acctatggaa ctgatgaatg ggagcagtgg 2700tggaatgcct ttaatgagga
aaacctgttt tgctcagaag aaatgccatc tagtgatgat 2760gaggctactg ctgactctca
acattctact cctccaaaaa agaagagaaa ggtagaagac 2820cccaaggact ttccttcaga
attgctaagt tttttgagtc atgctgtgtt tagtaataga 2880actcttgctt gctttgctat
ttacaccaca aaggaaaaag ctgcactgct atacaagaaa 2940attatggaaa aatattctgt
aacctttata agtaggcata acagttataa tcataacata 3000ctgttttttc ttactccaca
caggcataga gtgtctgcta ttaataacta tgctcaaaaa 3060ttgtgtacct ttagcttttt
aatttgtaaa ggggttaata aggaatattt gatgtatagt 3120gccttgacta gagatcataa
tcagccatac cacatttgta gaggttttac ttgctttaaa 3180aaacctccca cacctccccc
tgaacctgaa acataaaatg aatgcaattg ttgttgttaa 3240cttgtttatt gcagcttata
atggttacaa ataaagcaat agcatcacaa atttcacaaa 3300taaagcattt ttttcactgc
attctagttg tggtttgtcc aaactcatca atgtatctta 3360tcatgtctgg atccccagga
agctcctctg tgtcctcata aaccctaacc tcctctactt 3420gagaggacat tccaatcata
ggctgcccat ccaccctctg tgtcctcctg ttaattaggt 3480cacttaacaa aaaggaaatt
gggtaggggt ttttcacaga ccgctttcta agggtaattt 3540taaaatatct gggaagtccc
ttccactgct gtgttccaga agtgttggta aacagcccac 3600aaatgtcaac agcagaaaca
tacaagctgt cagctttgca caagggccca acaccctgct 3660catcaagaag cactgtggtt
gctgtgttag taatgtgcaa aacaggaggc acattttccc 3720cacctgtgta ggttccaaaa
tatctagtgt tttcattttt acttggatca ggaacccagc 3780actccactgg ataagcatta
tccttatcca aaacagcctt gtggtcagtg ttcatctgct 3840gactgtcaac tgtagcattt
tttggggtta cagtttgagc aggatatttg gtcctgtagt 3900ttgctaacac accctgcagc
tccaaaggtt ccccaccaac agcaaaaaaa tgaaaatttg 3960acccttgaat gggttttcca
gcaccatttt catgagtttt ttgtgtccct gaatgcaagt 4020ttaacatagc agttacccca
ataacctcag ttttaacagt aacagcttcc cacatcaaaa 4080tatttccaca ggttaagtcc
tcatttaaat taggcaaagg aattgctcta gagcggccgc 4140caccgcggtg gagctccaat
tcgccctata gtgagtcgta ttacgcgcgc tcactggccg 4200tcgttttaca acgtcgtgac
tgggaaaacc ctggcgttac ccaacttaat cgccttgcag 4260cacatccccc tttcgccagc
tggcgtaata gcgaagaggc ccgcaccgat cgcccttccc 4320aacagttgcg cagcctgaat
ggcgaatggg acgcgccctg tagcggcgca ttaagcgcgg 4380cgggtgtggt ggttacgcgc
agcgtgaccg ctacacttgc cagcgcccta gcgcccgctc 4440ctttcgcttt cttcccttcc
tttctcgcca cgttcgccgg ctttccccgt caagctctaa 4500atcgggggct ccctttaggg
ttccgattta gtgctttacg gcacctcgac cccaaaaaac 4560ttgattaggg tgatggttca
cgtagtgggc catcgccctg atagacggtt tttcgccctt 4620tgacgttgga gtccacgttc
tttaatagtg gactcttgtt ccaaactgga acaacactca 4680accctatctc ggtctattct
tttgatttat aagggatttt gccgatttcg gcctattggt 4740taaaaaatga gctgatttaa
caaaaattta acgcgaattt taacaaaata ttaacgctta 4800caatttaggt ggcacttttc
ggggaaatgt gcgcggaacc cctatttgtt tatttttcta 4860aatacattca aatatgtatc
cgctcatgag acaataaccc tgataaatgc ttcaataata 4920ttgaaaaagg aagagtatga
gtattcaaca tttccgtgtc gcccttattc ccttttttgc 4980ggcattttgc cttcctgttt
ttgctcaccc agaaacgctg gtgaaagtaa aagatgctga 5040agatcagttg ggtgcacgag
tgggttacat cgaactggat ctcaacagcg gtaagatcct 5100tgagagtttt cgccccgaag
aacgttttcc aatgatgagc acttttaaag ttctgctatg 5160tggcgcggta ttatcccgta
ttgacgccgg gcaagagcaa ctcggtcgcc gcatacacta 5220ttctcagaat gacttggttg
agtactcacc agtcacagaa aagcatctta cggatggcat 5280gacagtaaga gaattatgca
gtgctgccat aaccatgagt gataacactg cggccaactt 5340acttctgaca acgatcggag
gaccgaagga gctaaccgct tttttgcaca acatggggga 5400tcatgtaact cgccttgatc
gttgggaacc ggagctgaat gaagccatac caaacgacga 5460gcgtgacacc acgatgcctg
tagcaatggc aacaacgttg cgcaaactat taactggcga 5520actacttact ctagcttccc
ggcaacaatt aatagactgg atggaggcgg ataaagttgc 5580aggaccactt ctgcgctcgg
cccttccggc tggctggttt attgctgata aatctggagc 5640cggtgagcgt gggtctcgcg
gtatcattgc agcactgggg ccagatggta agccctcccg 5700tatcgtagtt atctacacga
cggggagtca ggcaactatg gatgaacgaa atagacagat 5760cgctgagata ggtgcctcac
tgattaagca ttggtaactg tcagaccaag tttactcata 5820tatactttag attgatttaa
aacttcattt ttaatttaaa aggatctagg tgaagatcct 5880ttttgataat ctcatgacca
aaatccctta acgtgagttt tcgttccact gagcgtcaga 5940ccccgtagaa aagatcaaag
gatcttcttg agatcctttt tttctgcgcg taatctgctg 6000cttgcaaaca aaaaaaccac
cgctaccagc ggtggtttgt ttgccggatc aagagctacc 6060aactcttttt ccgaaggtaa
ctggcttcag cagagcgcag ataccaaata ctgtccttct 6120agtgtagccg tagttaggcc
accacttcaa gaactctgta gcaccgccta catacctcgc 6180tctgctaatc ctgttaccag
tggctgctgc cagtggcgat aagtcgtgtc ttaccgggtt 6240ggactcaaga cgatagttac
cggataaggc gcagcggtcg ggctgaacgg ggggttcgtg 6300cacacagccc agcttggagc
gaacgaccta caccgaactg agatacctac agcgtgagct 6360atgagaaagc gccacgcttc
ccgaagggag aaaggcggac aggtatccgg taagcggcag 6420ggtcggaaca ggagagcgca
cgagggagct tccaggggga aacgcctggt atctttatag 6480tcctgtcggg tttcgccacc
tctgacttga gcgtcgattt ttgtgatgct cgtcaggggg 6540gcggagccta tggaaaaacg
ccagcaacgc ggccttttta cggttcctgg ccttttgctg 6600gccttttgct cacatgttct
ttcctgcgtt atcccctgat tctgtggata accgtattac 6660cgcctttgag tgagctgata
ccgctcgccg cagccgaacg accgagcgca gcgagtcagt 6720gagcgaggaa gcggaagagc
gcccaatacg caaaccgcct ctccccgcgc gttggccgat 6780tcattaatgc agctggcacg
acaggtttcc cgactggaaa gcgggcagtg agcgcaacgc 6840aattaatgtg agttagctca
ctcattaggc accccaggct ttacacttta tgcttccggc 6900tcgtatgttg tgtggaattg
tgagcggata acaatttcac acaggaaaca gctatgacca 6960tgattacgcc aagcgcgcaa
ttaaccctca ctaaagggaa caaaagctgg gtaccgggcc 7020ccccct
70261407278DNAArtificial
SequenceDescription of Artificial Sequence Synthetic pDEST12.2
polynucleotide 140ggggggcgga gcctatggaa aaacgccagc aacgcggcct ttttacggtt
cctggccttt 60tgctggcctt ttgctcacat gttctttcct gcgttatccc ctgattctgt
ggataaccgt 120attaccgcct ttgagtgagc tgataccgct cgccgcagcc gaacgaccga
gcgcagcgag 180tcagtgagcg aggaagcgga agagctcgcg aatgcatgtc gttacataac
ttacggtaaa 240tggcccgcct ggctgaccgc ccaacgaccc ccgcccattg acgtcaataa
tgacgtatgt 300tcccatagta acgccaatag ggactttcca ttgacgtcaa tgggtggagt
atttacggta 360aactgcccac ttggcagtac atcaagtgta tcatatgcca agtacgcccc
ctattgacgt 420caatgacggt aaatggcccg cctggcatta tgcccagtac atgaccttat
gggactttcc 480tacttggcag tacatctacg tattagtcat cgctattacc atggtgatgc
ggttttggca 540gtacatcaat gggcgtggat agcggtttga ctcacgggga tttccaagtc
tccaccccat 600tgacgtcaat gggagtttgt tttggcacca aaatcaacgg gactttccaa
aatgtcgtaa 660caactccgcc ccattgacgc aaatgggcgg taggcgtgta cggtgggagg
tctatataag 720cagagctcgt ttagtgaacc gtcagatcgc ctggagacgc catccacgct
gttttgacct 780ccatagaaga caccgggacc gatccagcct ccggactcta gcctaggccg
cgggacggat 840aacaatttca cacaggaaac agctatgacc attaggcctt tgcaaaaagc
tatttaggtg 900acactataga aggtacgcct gcaggtaccg gatcacaagt ttgtacaaaa
aagctgaacg 960agaaacgtaa aatgatataa atatcaatat attaaattag attttgcata
aaaaacagac 1020tacataatac tgtaaaacac aacatatcca gtcactatgg cggccgcatt
aggcacccca 1080ggctttacac tttatgcttc cggctcgtat aatgtgtgga ttttgagtta
ggatccgtcg 1140agattttcag gagctaagga agctaaaatg gagaaaaaaa tcactggata
taccaccgtt 1200gatatatccc aatggcatcg taaagaacat tttgaggcat ttcagtcagt
tgctcaatgt 1260acctataacc agaccgttca gctggatatt acggcctttt taaagaccgt
aaagaaaaat 1320aagcacaagt tttatccggc ctttattcac attcttgccc gcctgatgaa
tgctcatccg 1380gaattccgta tggcaatgaa agacggtgag ctggtgatat gggatagtgt
tcacccttgt 1440tacaccgttt tccatgagca aactgaaacg ttttcatcgc tctggagtga
ataccacgac 1500gatttccggc agtttctaca catatattcg caagatgtgg cgtgttacgg
tgaaaacctg 1560gcctatttcc ctaaagggtt tattgagaat atgtttttcg tctcagccaa
tccctgggtg 1620agtttcacca gttttgattt aaacgtggcc aatatggaca acttcttcgc
ccccgttttc 1680accatgggca aatattatac gcaaggcgac aaggtgctga tgccgctggc
gattcaggtt 1740catcatgccg tctgtgatgg cttccatgtc ggcagaatgc ttaatgaatt
acaacagtac 1800tgcgatgagt ggcagggcgg ggcgtaaacg cgtggatccg gcttactaaa
agccagataa 1860cagtatgcgt atttgcgcgc tgatttttgc ggtataagaa tatatactga
tatgtatacc 1920cgaagtatgt caaaaagagg tgtgctatga agcagcgtat tacagtgaca
gttgacagcg 1980acagctatca gttgctcaag gcatatatga tgtcaatatc tccggtctgg
taagcacaac 2040catgcagaat gaagcccgtc gtctgcgtgc cgaacgctgg aaagcggaaa
atcaggaagg 2100gatggctgag gtcgcccggt ttattgaaat gaacggctct tttgctgacg
agaacaggga 2160ctggtgaaat gcagtttaag gtttacacct ataaaagaga gagccgttat
cgtctgtttg 2220tggatgtaca gagtgatatt attgacacgc ccgggcgacg gatggtgatc
cccctggcca 2280gtgcacgtct gctgtcagat aaagtctccc gtgaacttta cccggtggtg
catatcgggg 2340atgaaagctg gcgcatgatg accaccgata tggccagtgt gccggtctcc
gttatcgggg 2400aagaagtggc tgatctcagc caccgcgaaa atgacatcaa aaacgccatt
aacctgatgt 2460tctggggaat ataaatgtca ggctccctta tacacagcca gtctgcaggt
cgaccatagt 2520gactggatat gttgtgtttt acagtattat gtagtctgtt ttttatgcaa
aatctaattt 2580aatatattga tatttatatc attttacgtt tctcgttcag ctttcttgta
caaagtggtg 2640atcgcgtgca tgcgacgtca tagctctctc cctatagtga gtcgtattat
aagctaggca 2700ctggccgtcg ttttacaacg tcgtgactgg gaaaactgct agcttgggat
ctttgtgaag 2760gaaccttact tctgtggtgt gacataattg gacaaactac ctacagagat
ttaaagctct 2820aaggtaaata taaaattttt aagtgtataa tgtgttaaac tagctgcata
tgcttgctgc 2880ttgagagttt tgcttactga gtatgattta tgaaaatatt atacacagga
gctagtgatt 2940ctaattgttt gtgtatttta gattcacagt cccaaggctc atttcaggcc
cctcagtcct 3000cacagtctgt tcatgatcat aatcagccat accacatttg tagaggtttt
acttgcttta 3060aaaaacctcc cacacctccc cctgaacctg aaacataaaa tgaatgcaat
tgttgttgtt 3120aacttgttta ttgcagctta taatggttac aaataaagca atagcatcac
aaatttcaca 3180aataaagcat ttttttcact gcattctagt tgtggtttgt ccaaactcat
caatgtatct 3240tatcatgtct ggatcgatcc tgcattaatg aatcggccaa cgcgcgggga
gaggcggttt 3300gcgtattggc tggcgtaata gcgaagaggc ccgcaccgat cgcccttccc
aacagttgcg 3360cagcctgaat ggcgaatggg acgcgccctg tagcggcgca ttaagcgcgg
cgggtgtggt 3420ggttacgcgc agcgtgaccg ctacacttgc cagcgcccta gcgcccgctc
ctttcgcttt 3480cttcccttcc tttctcgcca cgttcgccgg ctttccccgt caagctctaa
atcgggggct 3540ccctttaggg ttccgattta gtgctttacg gcacctcgac cccaaaaaac
ttgattaggg 3600tgatggttca cgtagtgggc catcgccctg atagacggtt tttcgccctt
tgacgttgga 3660gtccacgttc tttaatagtg gactcttgtt ccaaactgga acaacactca
accctatctc 3720ggtctattct tttgatttat aagggatttt gccgatttcg gcctattggt
taaaaaatga 3780gctgatttaa caaatattta acgcgaattt taacaaaata ttaacgttta
caatttcgcc 3840tgatgcggta ttttctcctt acgcatctgt gcggtatttc acaccgcata
cgcggatctg 3900cgcagcacca tggcctgaaa taacctctga aagaggaact tggttaggta
ccttctgagg 3960cggaaagaac cagctgtgga atgtgtgtca gttagggtgt ggaaagtccc
caggctcccc 4020agcaggcaga agtatgcaaa gcatgcatct caattagtca gcaaccaggt
gtggaaagtc 4080cccaggctcc ccagcaggca gaagtatgca aagcatgcat ctcaattagt
cagcaaccat 4140agtcccgccc ctaactccgc ccatcccgcc cctaactccg cccagttccg
cccattctcc 4200gccccatggc tgactaattt tttttattta tgcagaggcc gaggccgcct
cggcctctga 4260gctattccag aagtagtgag gaggcttttt tggaggccta ggcttttgca
aaaagcttga 4320ttcttctgac acaacagtct cgaacttaag gctagagcca ccatgattga
acaagatgga 4380ttgcacgcag gttctccggc cgcttgggtg gagaggctat tcggctatga
ctgggcacaa 4440cagacaatcg gctgctctga tgccgccgtg ttccggctgt cagcgcaggg
gcgcccggtt 4500ctttttgtca agaccgacct gtccggtgcc ctgaatgaac tgcaggacga
ggcagcgcgg 4560ctatcgtggc tggccacgac gggcgttcct tgcgcagctg tgctcgacgt
tgtcactgaa 4620gcgggaaggg actggctgct attgggcgaa gtgccggggc aggatctcct
gtcatctcac 4680cttgctcctg ccgagaaagt atccatcatg gctgatgcaa tgcggcggct
gcatacgctt 4740gatccggcta cctgcccatt cgaccaccaa gcgaaacatc gcatcgagcg
agcacgtact 4800cggatggaag ccggtcttgt cgatcaggat gatctggacg aagagcatca
ggggctcgcg 4860ccagccgaac tgttcgccag gctcaaggcg cgcatgcccg acggcgagga
tctcgtcgtg 4920acccatggcg atgcctgctt gccgaatatc atggtggaaa atggccgctt
ttctggattc 4980atcgactgtg gccggctggg tgtggcggac cgctatcagg acatagcgtt
ggctacccgt 5040gatattgctg aagagcttgg cggcgaatgg gctgaccgct tcctcgtgct
ttacggtatc 5100gccgctcccg attcgcagcg catcgccttc tatcgccttc ttgacgagtt
cttctgagcg 5160ggactctggg gttcgaaatg accgaccaag cgacgcccaa cctgccatca
cgatggccgc 5220aataaaatat ctttattttc attacatctg tgtgttggtt ttttgtgtga
atcgatagcg 5280ataaggatcc gcgtatggtg cactctcagt acaatctgct ctgatgccgc
atagttaagc 5340cagccccgac acccgccaac acccgctgac gcgccctgac gggcttgtct
gctcccggca 5400tccgcttaca gacaagctgt gaccgtctcc gggagctgca tgtgtcagag
gttttcaccg 5460tcatcaccga aacgcgcgag acgaaagggc ctcgtgatac gcctattttt
ataggttaat 5520gtcatgataa taatggtttc ttagacgtca ggtggcactt ttcggggaaa
tgtgcgcgga 5580acccctattt gtttattttt ctaaatacat tcaaatatgt atccgctcat
gagacaataa 5640ccctgataaa tgcttcaata atattgaaaa aggaagagta tgagtattca
acatttccgt 5700gtcgccctta ttcccttttt tgcggcattt tgccttcctg tttttgctca
cccagaaacg 5760ctggtgaaag taaaagatgc tgaagatcag ttgggtgcac gagtgggtta
catcgaactg 5820gatctcaaca gcggtaagat ccttgagagt tttcgccccg aagaacgttt
tccaatgatg 5880agcactttta aagttctgct atgtggcgcg gtattatccc gtattgacgc
cgggcaagag 5940caactcggtc gccgcataca ctattctcag aatgacttgg ttgagtactc
accagtcaca 6000gaaaagcatc ttacggatgg catgacagta agagaattat gcagtgctgc
cataaccatg 6060agtgataaca ctgcggccaa cttacttctg acaacgatcg gaggaccgaa
ggagctaacc 6120gcttttttgc acaacatggg ggatcatgta actcgccttg atcgttggga
accggagctg 6180aatgaagcca taccaaacga cgagcgtgac accacgatgc ctgtagcaat
ggcaacaacg 6240ttgcgcaaac tattaactgg cgaactactt actctagctt cccggcaaca
attaatagac 6300tggatggagg cggataaagt tgcaggacca cttctgcgct cggcccttcc
ggctggctgg 6360tttattgctg ataaatctgg agccggtgag cgtgggtctc gcggtatcat
tgcagcactg 6420gggccagatg gtaagccctc ccgtatcgta gttatctaca cgacggggag
tcaggcaact 6480atggatgaac gaaatagaca gatcgctgag ataggtgcct cactgattaa
gcattggtaa 6540ctgtcagacc aagtttactc atatatactt tagattgatt taaaacttca
tttttaattt 6600aaaaggatct aggtgaagat cctttttgat aatctcatga ccaaaatccc
ttaacgtgag 6660ttttcgttcc actgagcgtc agaccccgta gaaaagatca aaggatcttc
ttgagatcct 6720ttttttctgc gcgtaatctg ctgcttgcaa acaaaaaaac caccgctacc
agcggtggtt 6780tgtttgccgg atcaagagct accaactctt tttccgaagg taactggctt
cagcagagcg 6840cagataccaa atactgtcct tctagtgtag ccgtagttag gccaccactt
caagaactct 6900gtagcaccgc ctacatacct cgctctgcta atcctgttac cagtggctgc
tgccagtggc 6960gataagtcgt gtcttaccgg gttggactca agacgatagt taccggataa
ggcgcagcgg 7020tcgggctgaa cggggggttc gtgcacacag cccagcttgg agcgaacgac
ctacaccgaa 7080ctgagatacc tacagcgtga gcattgagaa agcgccacgc ttcccgaagg
gagaaaggcg 7140gacaggtatc cggtaagcgg cagggtcgga acaggagagc gcacgaggga
gcttccaggg 7200ggaaacgcct ggtatcttta tagtcctgtc gggtttcgcc acctctgact
tgagcgtcga 7260tttttgtgat gctcgtca
72781415848DNAArtificial SequenceDescription of Artificial
Sequence Synthetic pDEST13 polynucleotide 141ttcactggcc gtcgttttac
aacgtcgtga ctgggaaaac cctggcgtta cccaacttaa 60tcgccttgca gcacatcccc
ctttcgccag ctggcgtaat agcgaagagg cccgcaccga 120tcgcccttcc caacagttgc
gcagcctgaa tggcgaatgg cgcctgatgc ggtattttct 180ccttacgcat ctgtgcggta
tttcacaccg catatggtgc actctcagta caatctgctc 240tgatgccgca tagttaagcc
agccccgaca cccgccaaca cccgctgacg cgccctgacg 300ggcttgtctg ctcccggcat
ccgcttacag acaagctgtg accgtctccg ggagctgcat 360gtgtcagagg ttttcaccgt
catcaccgaa acgcgcgaga cgaaagggcc tcgtgatacg 420cctattttta taggttaatg
tcatgataat aatggtttct tagacgtcag gtggcacttt 480tcggggaaat gtgcgcggaa
cccctatttg tttatttttc taaatacatt caaatatgta 540tccgctcatg agacaataac
cctgataaat gcttcaataa tattgaaaaa ggaagagtat 600gagtattcaa catttccgtg
tcgcccttat tccctttttt gcggcatttt gccttcctgt 660ttttgctcac ccagaaacgc
tggtgaaagt aaaagatgct gaagatcagt tgggtgcacg 720agtgggttac atcgaactgg
atctcaacag cggtaagatc cttgagagtt ttcgccccga 780agaacgtttt ccaatgatga
gcacttttaa agttctgcta tgtggcgcgg tattatcccg 840tattgacgcc gggcaagagc
aactcggtcg ccgcatacac tattctcaga atgacttggt 900tgagtactca ccagtcacag
aaaagcatct tacggatggc atgacagtaa gagaattatg 960cagtgctgcc ataaccatga
gtgataacac tgcggccaac ttacttctga caacgatcgg 1020aggaccgaag gagctaaccg
cttttttgca caacatgggg gatcatgtaa ctcgccttga 1080tcgttgggaa ccggagctga
atgaagccat accaaacgac gagcgtgaca ccacgatgcc 1140tgtagcaatg gcaacaacgt
tgcgcaaact attaactggc gaactactta ctctagcttc 1200ccggcaacaa ttaatagact
ggatggaggc ggataaagtt gcaggaccac ttctgcgctc 1260ggcccttccg gctggctggt
ttattgctga taaatctgga gccggtgagc gtgggtctcg 1320cggtatcatt gcagcactgg
ggccagatgg taagccctcc cgtatcgtag ttatctacac 1380gacggggagt caggcaacta
tggatgaacg aaatagacag atcgctgaga taggtgcctc 1440actgattaag cattggtaac
tgtcagacca agtttactca tatatacttt agattgattt 1500aaaacttcat ttttaattta
aaaggatcta ggtgaagatc ctttttgata atctcatgac 1560caaaatccct taacgtgagt
tttcgttcca ctgagcgtca gaccccgtag aaaagatcaa 1620aggatcttct tgagatcctt
tttttctgcg cgtaatctgc tgcttgcaaa caaaaaaacc 1680accgctacca gcggtggttt
gtttgccgga tcaagagcta ccaactcttt ttccgaaggt 1740aactggcttc agcagagcgc
agataccaaa tactgttctt ctagtgtagc cgtagttagg 1800ccaccacttc aagaactctg
tagcaccgcc tacatacctc gctctgctaa tcctgttacc 1860agtggctgct gccagtggcg
ataagtcgtg tcttaccggg ttggactcaa gacgatagtt 1920accggataag gcgcagcggt
cgggctgaac ggggggttcg tgcacacagc ccagcttgga 1980gcgaacgacc tacaccgaac
tgagatacct acagcgtgag cattgagaaa gcgccacgct 2040tcccgaaggg agaaaggcgg
acaggtatcc ggtaagcggc agggtcggaa caggagagcg 2100cacgagggag cttccagggg
gaaacgcctg gtatctttat agtcctgtcg ggtttcgcca 2160cctctgactt gagcgtcgat
ttttgtgatg ctcgtcaggg gggcggagcc tatggaaaaa 2220cgccagcaac gcggcctttt
tacggttcct ggccttttgc tggccttttg ctcacatgtt 2280ctttcctgcg ttatcccctg
attctgtgga taaccgtatt accgcctttg agtgagctga 2340taccgctcgc cgcagccgaa
cgaccgagcg cagcgagtca gtgagcgagg aagcggaaga 2400gcgcccaata cgcaaaccgc
ctctccccgc gcgttggccg attcattaat gcagctggca 2460cgacaggttt cccgactgga
aagcgggcag tgagcgcaac gcaattaatg tgagttagct 2520cactcattag gcaccccagg
ctttacactt tatgcttccg gctcgtatgt tgtgtggaat 2580tgtgagcgga taacaatttc
acacaggaaa cagctatgac catgattacg ccaagcttgg 2640ctgcaggtga tgattatcag
ccagcagaga ttaaggaaaa cagacaggtt tattgagcgc 2700ttatctttcc ctttattttt
gctgcggtaa gtcgcataaa aaccattctt cataattcaa 2760tccatttact atgttatgtt
ctgaggggag tgaaaattcc cctaattcga tgaagattct 2820tgctcaattg ttatcagcta
tgcgccgacc agaacacctt gccgatcagc caaacgtctc 2880ttcaggccac tgactagcga
taactttccc cacaacggaa caactctcat tgcatgggat 2940cattgggtac tgtgggttta
gtggttgtaa aaacacctga ccgctatccc tgatcagttt 3000cttgaaggta aactcatcac
ccccaagtct ggctatgcag aaatcacctg gctcaacagc 3060ctgctcaggg tcaacgagaa
ttaacattcc gtcaggaaag cttggcttgg agcctgttgg 3120tgcggtcatg gaattacctt
caacctcaag ccagaatgca gaatcactgg cttttttggt 3180tgtgcttacc catctctccg
catcaccttt ggtaaaggtt ctaagcttag gtgagaacat 3240ccctgcctga acatgagaaa
aaacagggta ctcatactca cttctaagtg acggctgcat 3300actaaccgct tcatacatct
cgtagatttc tctggcgatt gaagggctaa attcttcaac 3360gctaactttg agaatttttg
caagcaatgc ggcgttataa gcatttaatg cattgatgcc 3420attaaataaa gcaccaacgc
ctgactgccc catccccatc ttgtctgcga cagattcctg 3480ggataagcca agttcatttt
tctttttttc ataaattgct ttaaggcgac gtgcgtcctc 3540aagctgctct tgtgttaatg
gtttcttttt tgtgctcata cgttaaatct atcaccgcaa 3600gggataaata tctaacaccg
tgcgtgttga ctattttacc tctggcggtg ataatggttg 3660catgtactaa ggaggttgta
tggaacaacg cataaccctg aaagattatg caatgcgctt 3720tgggcaaacc aagacagcta
aagatctctc acctaccaaa caatgccccc ctgcaaaaaa 3780taaattcata taaaaaacat
acagataacc atctgcggtg ataaattatc tctggcggtg 3840ttgacataaa taccactggc
ggtgatactg agcacatcag caggacgcac tgaccaccat 3900gaaggtgacg ctcttaaaaa
ttaagccctg aagaagggca gcattcaaag cagaaggctt 3960tggggtgtgt gatacgaaac
gaagcattgg gatcatcaca agtttgtaca aaaaagctga 4020acgagaaacg taaaatgata
taaatatcaa tatattaaat tagattttgc ataaaaaaca 4080gactacataa tactgtaaaa
cacaacatat ccagtcacta tggcggccgc taagttggca 4140gcatcacccg acgcactttg
cgccgaataa atacctgtga cggaagatca cttcgcagaa 4200taaataaatc ctggtgtccc
tgttgatacc gggaagccct gggccaactt ttggcgaaaa 4260tgagacgttg atcggcacgt
aagaggttcc aactttcacc ataatgaaat aagatcacta 4320ccgggcgtat tttttgagtt
atcgagattt tcaggagcta aggaagctaa aatggagaaa 4380aaaatcactg gatataccac
cgttgatata tcccaatggc atcgtaaaga acattttgag 4440gcatttcagt cagttgctca
atgtacctat aaccagaccg ttcagctgga tattacggcc 4500tttttaaaga ccgtaaagaa
aaataagcac aagttttatc cggcctttat tcacattctt 4560gcccgcctga tgaatgctca
tccggaattc cgtatggcaa tgaaagacgg tgagctggtg 4620atatgggata gtgttcaccc
ttgttacacc gttttccatg agcaaactga aacgttttca 4680tcgctctgga gtgaatacca
cgacgatttc cggcagtttc tacacatata ttcgcaagat 4740gtggcgtgtt acggtgaaaa
cctggcctat ttccctaaag ggtttattga gaatatgttt 4800ttcgtctcag ccaatccctg
ggtgagtttc accagttttg atttaaacgt ggccaatatg 4860gacaacttct tcgcccccgt
tttcaccatg ggcaaatatt atacgcaagg cgacaaggtg 4920ctgatgccgc tggcgattca
ggttcatcat gccgtctgtg atggcttcca tgtcggcaga 4980atgcttaatg aattacaaca
gtactgcgat gagtggcagg gcggggcgta aacgcgtgga 5040tccggcttac taaaagccag
ataacagtat gcgtatttgc gcgctgattt ttgcggtata 5100agaatatata ctgatatgta
tacccgaagt atgtcaaaaa gaggtgtgct atgaagcagc 5160gtattacagt gacagttgac
agcgacagct atcagttgct caaggcatat atgatgtcaa 5220tatctccggt ctggtaagca
caaccatgca gaatgaagcc cgtcgtctgc gtgccgaacg 5280ctggaaagcg gaaaatcagg
aagggatggc tgaggtcgcc cggtttattg aaatgaacgg 5340ctcttttgct gacgagaaca
gggactggtg aaatgcagtt taaggtttac acctataaaa 5400gagagagccg ttatcgtctg
tttgtggatg tacagagtga tattattgac acgcccgggc 5460gacggatggt gatccccctg
gccagtgcac gtctgctgtc agataaagtc tcccgtgaac 5520tttacccggt ggtgcatatc
ggggatgaaa gctggcgcat gatgaccacc gatatggcca 5580gtgtgccggt ctccgttatc
ggggaagaag tggctgatct cagccaccgc gaaaatgaca 5640tcaaaaacgc cattaacctg
atgttctggg gaatataaat gtcaggctcc gttatacaca 5700gccagtctgc aggtcgacca
tagtgactgg atatgttgtg ttttacagta ttatgtagtc 5760tgttttttat gcaaaatcta
atttaatata ttgatattta tatcatttta cgtttctcgt 5820tcagctttct tgtacaaagt
ggtgataa 58481426422DNAArtificial
SequenceDescription of Artificial Sequence Synthetic pDEST14
polynucleotide 142cgatcccgcg aaattaatac gactcactat agggagacca caacggtttc
cctctagatc 60acaagtttgt acaaaaaagc tgaacgagaa acgtaaaatg atataaatat
caatatatta 120aattagattt tgcataaaaa acagactaca taatactgta aaacacaaca
tatccagtca 180ctatggcggc cgctaagttg gcagcatcac ccgacgcact ttgcgccgaa
taaatacctg 240tgacggaaga tcacttcgca gaataaataa atcctggtgt ccctgttgat
accgggaagc 300cctgggccaa cttttggcga aaatgagacg ttgatcggca cgtaagaggt
tccaactttc 360accataatga aataagatca ctaccgggcg tattttttga gttatcgaga
ttttcaggag 420ctaaggaagc taaaatggag aaaaaaatca ctggatatac caccgttgat
atatcccaat 480ggcatcgtaa agaacatttt gaggcatttc agtcagttgc tcaatgtacc
tataaccaga 540ccgttcagct ggatattacg gcctttttaa agaccgtaaa gaaaaataag
cacaagtttt 600atccggcctt tattcacatt cttgcccgcc tgatgaatgc tcatccggaa
ttccgtatgg 660caatgaaaga cggtgagctg gtgatatggg atagtgttca cccttgttac
accgttttcc 720atgagcaaac tgaaacgttt tcatcgctct ggagtgaata ccacgacgat
ttccggcagt 780ttctacacat atattcgcaa gatgtggcgt gttacggtga aaacctggcc
tatttcccta 840aagggtttat tgagaatatg tttttcgtct cagccaatcc ctgggtgagt
ttcaccagtt 900ttgatttaaa cgtggccaat atggacaact tcttcgcccc cgttttcacc
atgggcaaat 960attatacgca aggcgacaag gtgctgatgc cgctggcgat tcaggttcat
catgccgtct 1020gtgatggctt ccatgtcggc agaatgctta atgaattaca acagtactgc
gatgagtggc 1080agggcggggc gtaaacgcgt ggatccggct tactaaaagc cagataacag
tatgcgtatt 1140tgcgcgctga tttttgcggt ataagaatat atactgatat gtatacccga
agtatgtcaa 1200aaagaggtgt gctatgaagc agcgtattac agtgacagtt gacagcgaca
gctatcagtt 1260gctcaaggca tatatgatgt caatatctcc ggtctggtaa gcacaaccat
gcagaatgaa 1320gcccgtcgtc tgcgtgccga acgctggaaa gcggaaaatc aggaagggat
ggctgaggtc 1380gcccggttta ttgaaatgaa cggctctttt gctgacgaga acagggactg
gtgaaatgca 1440gtttaaggtt tacacctata aaagagagag ccgttatcgt ctgtttgtgg
atgtacagag 1500tgatattatt gacacgcccg ggcgacggat ggtgatcccc ctggccagtg
cacgtctgct 1560gtcagataaa gtctcccgtg aactttaccc ggtggtgcat atcggggatg
aaagctggcg 1620catgatgacc accgatatgg ccagtgtgcc ggtctccgtt atcggggaag
aagtggctga 1680tctcagccac cgcgaaaatg acatcaaaaa cgccattaac ctgatgttct
ggggaatata 1740aatgtcaggc tcccttatac acagccagtc tgcaggtcga ccatagtgac
tggatatgtt 1800gtgttttaca gtattatgta gtctgttttt tatgcaaaat ctaatttaat
atattgatat 1860ttatatcatt ttacgtttct cgttcagctt tcttgtacaa agtggtgatg
atccggctgc 1920taacaaagcc cgaaaggaag ctgagttggc tgctgccacc gctgagcaat
aactagcata 1980accccttggg gcctctaaac gggtcttgag gggttttttg ctgaaaggag
gaactatatc 2040cggatatcca caggacgggt gtggtcgcca tgatcgcgta gtcgatagtg
gctccaagta 2100gcgaagcgag caggactggg cggcggccaa agcggtcgga cagtgctccg
agaacgggtg 2160cgcatagaaa ttgcatcaac gcatatagcg ctagcagcac gccatagtga
ctggcgatgc 2220tgtcggaatg gacgatatcc cgcaagaggc ccggcagtac cggcataacc
aagcctatgc 2280ctacagcatc cagggtgacg gtgccgagga tgacgatgag cgcattgtta
gatttcatac 2340acggtgcctg actgcgttag caatttaact gtgataaact accgcattaa
agcttatcga 2400tgataagctg tcaaacatga gaattcttga agacgaaagg gcctcgtgat
acgcctattt 2460ttataggtta atgtcatgat aataatggtt tcttagacgt caggtggcac
ttttcgggga 2520aatgtgcgcg gaacccctat ttgtttattt ttctaaatac attcaaatat
gtatccgctc 2580atgagacaat aaccctgata aatgcttcaa taatattgaa aaaggaagag
tatgagtatt 2640caacatttcc gtgtcgccct tattcccttt tttgcggcat tttgccttcc
tgtttttgct 2700cacccagaaa cgctggtgaa agtaaaagat gctgaagatc agttgggtgc
acgagtgggt 2760tacatcgaac tggatctcaa cagcggtaag atccttgaga gttttcgccc
cgaagaacgt 2820tttccaatga tgagcacttt taaagttctg ctatgtggcg cggtattatc
ccgtgttgac 2880gccgggcaag agcaactcgg tcgccgcata cactattctc agaatgactt
ggttgagtac 2940tcaccagtca cagaaaagca tcttacggat ggcatgacag taagagaatt
atgcagtgct 3000gccataacca tgagtgataa cactgcggcc aacttacttc tgacaacgat
cggaggaccg 3060aaggagctaa ccgctttttt gcacaacatg ggggatcatg taactcgcct
tgatcgttgg 3120gaaccggagc tgaatgaagc cataccaaac gacgagcgtg acaccacgat
gcctgcagca 3180atggcaacaa cgttgcgcaa actattaact ggcgaactac ttactctagc
ttcccggcaa 3240caattaatag actggatgga ggcggataaa gttgcaggac cacttctgcg
ctcggccctt 3300ccggctggct ggtttattgc tgataaatct ggagccggtg agcgtgggtc
tcgcggtatc 3360attgcagcac tggggccaga tggtaagccc tcccgtatcg tagttatcta
cacgacgggg 3420agtcaggcaa ctatggatga acgaaataga cagatcgctg agataggtgc
ctcactgatt 3480aagcattggt aactgtcaga ccaagtttac tcatatatac tttagattga
tttaaaactt 3540catttttaat ttaaaaggat ctaggtgaag atcctttttg ataatctcat
gaccaaaatc 3600ccttaacgtg agttttcgtt ccactgagcg tcagaccccg tagaaaagat
caaaggatct 3660tcttgagatc ctttttttct gcgcgtaatc tgctgcttgc aaacaaaaaa
accaccgcta 3720ccagcggtgg tttgtttgcc ggatcaagag ctaccaactc tttttccgaa
ggtaactggc 3780ttcagcagag cgcagatacc aaatactgtc cttctagtgt agccgtagtt
aggccaccac 3840ttcaagaact ctgtagcacc gcctacatac ctcgctctgc taatcctgtt
accagtggct 3900gctgccagtg gcgataagtc gtgtcttacc gggttggact caagacgata
gttaccggat 3960aaggcgcagc ggtcgggctg aacggggggt tcgtgcacac agcccagctt
ggagcgaacg 4020acctacaccg aactgagata cctacagcgt gagctatgag aaagcgccac
gcttcccgaa 4080gggagaaagg cggacaggta tccggtaagc ggcagggtcg gaacaggaga
gcgcacgagg 4140gagcttccag ggggaaacgc ctggtatctt tatagtcctg tcgggtttcg
ccacctctga 4200cttgagcgtc gatttttgtg atgctcgtca ggggggcgga gcctatggaa
aaacgccagc 4260aacgcggcct ttttacggtt cctggccttt tgctggcctt ttgctcacat
gttctttcct 4320gcgttatccc ctgattctgt ggataaccgt attaccgcct ttgagtgagc
tgataccgct 4380cgccgcagcc gaacgaccga gcgcagcgag tcagtgagcg aggaagcgga
agagcgcctg 4440atgcggtatt ttctccttac gcatctgtgc ggtatttcac accgcatata
tggtgcactc 4500tcagtacaat ctgctctgat gccgcatagt taagccagta tacactccgc
tatcgctacg 4560tgactgggtc atggctgcgc cccgacaccc gccaacaccc gctgacgcgc
cctgacgggc 4620ttgtctgctc ccggcatccg cttacagaca agctgtgacc gtctccggga
gctgcatgtg 4680tcagaggttt tcaccgtcat caccgaaacg cgcgaggcag ctgcggtaaa
gctcatcagc 4740gtggtcgtga agcgattcac agatgtctgc ctgttcatcc gcgtccagct
cgttgagttt 4800ctccagaagc gttaatgtct ggcttctgat aaagcgggcc atgttaaggg
cggttttttc 4860ctgtttggtc actgatgcct ccgtgtaagg gggatttctg ttcatggggg
taatgatacc 4920gatgaaacga gagaggatgc tcacgatacg ggttactgat gatgaacatg
cccggttact 4980ggaacgttgt gagggtaaac aactggcggt atggatgcgg cgggaccaga
gaaaaatcac 5040tcagggtcaa tgccagcgct tcgttaatac agatgtaggt gttccacagg
gtagccagca 5100gcatcctgcg atgcagatcc ggaacataat ggtgcagggc gctgacttcc
gcgtttccag 5160actttacgaa acacggaaac cgaagaccat tcatgttgtt gctcaggtcg
cagacgtttt 5220gcagcagcag tcgcttcacg ttcgctcgcg tatcggtgat tcattctgct
aaccagtaag 5280gcaaccccgc cagcctagcc gggtcctcaa cgacaggagc acgatcatgc
gcacccgtgg 5340ccaggaccca acgctgcccg agatgcgccg cgtgcggctg ctggagatgg
cggacgcgat 5400ggatatgttc tgccaagggt tggtttgcgc attcacagtt ctccgcaaga
attgattggc 5460tccaattctt ggagtggtga atccgttagc gaggtgccgc cggcttccat
tcaggtcgag 5520gtggcccggc tccatgcacc gcgacgcaac gcggggaggc agacaaggta
tagggcggcg 5580cctacaatcc atgccaaccc gttccatgtg ctcgccgagg cggcataaat
cgccgtgacg 5640atcagcggtc cagtgatcga agttaggctg gtaagagccg cgagcgatcc
ttgaagctgt 5700ccctgatggt cgtcatctac ctgcctggac agcatggcct gcaacgcggg
catcccgatg 5760ccgccggaag cgagaagaat cataatgggg aaggccatcc agcctcgcgt
cgcgaacgcc 5820agcaagacgt agcccagcgc gtcggccgcc atgccggcga taatggcctg
cttctcgccg 5880aaacgtttgg tggcgggacc agtgacgaag gcttgagcga gggcgtgcaa
gattccgaat 5940accgcaagcg acaggccgat catcgtcgcg ctccagcgaa agcggtcctc
gccgaaaatg 6000acccagagcg ctgccggcac ctgtcctacg agttgcatga taaagaagac
agtcataagt 6060gcggcgacga tagtcatgcc ccgcgcccac cggaaggagc tgactgggtt
gaaggctctc 6120aagggcatcg gtcgatcgac gctctccctt atgcgactcc tgcattagga
agcagcccag 6180tagtaggttg aggccgttga gcaccgccgc cgcaaggaat ggtgcatgca
aggagatggc 6240gcccaacagt cccccggcca cggggcctgc caccataccc acgccgaaac
aagcgctcat 6300gagcccgaag tggcgagccc gatcttcccc atcggtgatg tcggcgatat
aggcgccagc 6360aaccgcacct gtggcgccgg tgatgccggc cacgatgcgt ccggcgtaga
ggatcgagat 6420ct
64221437013DNAArtificial SequenceDescription of Artificial
Sequence Synthetic pDEST15 polynucleotide 143atcgagatct cgatcccgcg
aaattaatac gactcactat agggagacca caacggtttc 60cctctagaaa taattttgtt
taactttaag aaggagatat acatatgtcc cctatactag 120gttattggaa aattaagggc
cttgtgcaac ccactcgact tcttttggaa tatcttgaag 180aaaaatatga agagcatttg
tatgagcgcg atgaaggtga taaatggcga aacaaaaagt 240ttgaattggg tttggagttt
cccaatcttc cttattatat tgatggtgat gttaaattaa 300cacagtctat ggccatcata
cgttatatag ctgacaagca caacatgttg ggtggttgtc 360caaaagagcg tgcagagatt
tcaatgcttg aaggagcggt tttggatatt agatacggtg 420tttcgagaat tgcatatagt
aaagactttg aaactctcaa agttgatttt cttagcaagc 480tacctgaaat gctgaaaatg
ttcgaagatc gtttatgtca taaaacatat ttaaatggtg 540atcatgtaac ccatcctgac
ttcatgttgt atgacgctct tgatgttgtt ttatacatgg 600acccaatgtg cctggatgcg
ttcccaaaat tagtttgttt taaaaaacgt attgaagcta 660tcccacaaat tgataagtac
ttgaaatcca gcaagtatat agcatggcct ttgcagggct 720ggcaagccac gtttggtggt
ggcgaccatc ctccaaaatc ggatctggtt ccgcgtccat 780ggtcgaatca aacaagtttg
tacaaaaaag ctgaacgaga aacgtaaaat gatataaata 840tcaatatatt aaattagatt
ttgcataaaa aacagactac ataatactgt aaaacacaac 900atatccagtc actatggcgg
ccgcattagg caccccaggc tttacacttt atgcttccgg 960ctcgtataat gtgtggattt
tgagttagga tccgtcgaga ttttcaggag ctaaggaagc 1020taaaatggag aaaaaaatca
ctggatatac caccgttgat atatcccaat ggcatcgtaa 1080agaacatttt gaggcatttc
agtcagttgc tcaatgtacc tataaccaga ccgttcagct 1140ggatattacg gcctttttaa
agaccgtaaa gaaaaataag cacaagtttt atccggcctt 1200tattcacatt cttgcccgcc
tgatgaatgc tcatccggaa ttccgtatgg caatgaaaga 1260cggtgagctg gtgatatggg
atagtgttca cccttgttac accgttttcc atgagcaaac 1320tgaaacgttt tcatcgctct
ggagtgaata ccacgacgat ttccggcagt ttctacacat 1380atattcgcaa gatgtggcgt
gttacggtga aaacctggcc tatttcccta aagggtttat 1440tgagaatatg tttttcgtct
cagccaatcc ctgggtgagt ttcaccagtt ttgatttaaa 1500cgtggccaat atggacaact
tcttcgcccc cgttttcacc atgggcaaat attatacgca 1560aggcgacaag gtgctgatgc
cgctggcgat tcaggttcat catgccgtct gtgatggctt 1620ccatgtcggc agaatgctta
atgaattaca acagtactgc gatgagtggc agggcggggc 1680gtaatctaga ggatccggct
tactaaaagc cagataacag tatgcgtatt tgcgcgctga 1740tttttgcggt ataagaatat
atactgatat gtatacccga agtatgtcaa aaagaggtgt 1800gctatgaagc agcgtattac
agtgacagtt gacagcgaca gctatcagtt gctcaaggca 1860tatatgatgt caatatctcc
ggtctggtaa gcacaaccat gcagaatgaa gcccgtcgtc 1920tgcgtgccga acgctggaaa
gcggaaaatc aggaagggat ggctgaggtc gcccggttta 1980ttgaaatgaa cggctctttt
gctgacgaga acagggactg gtgaaatgca gtttaaggtt 2040tacacctata aaagagagag
ccgttatcgt ctgtttgtgg atgtacagag tgatattatt 2100gacacgcccg ggcgacggat
ggtgatcccc ctggccagtg cacgtctgct gtcagataaa 2160gtctcccgtg aactttaccc
ggtggtgcat atcggggatg aaagctggcg catgatgacc 2220accgatatgg ccagtgtgcc
ggtctccgtt atcggggaag aagtggctga tctcagccac 2280cgcgaaaatg acatcaaaaa
cgccattaac ctgatgttct ggggaatata aatgtcaggc 2340tcccttatac acagccagtc
tgcaggtcga ccatagtgac tggatatgtt gtgttttaca 2400gtattatgta gtctgttttt
tatgcaaaat ctaatttaat atattgatat ttatatcatt 2460ttacgtttct cgttcagctt
tcttgtacaa agtggtttga ttcgacccgg gatccggctg 2520ctaacaaagc ccgaaaggaa
gctgagttgg ctgctgccac cgctgagcaa taactagcat 2580aaccccttgg ggcctctaaa
cgggtcttga ggggtttttt gctgaaagga ggaactatat 2640ccggatatcc acaggacggg
tgtggtcgcc atgatcgcgt agtcgatagt ggctccaagt 2700agcgaagcga gcaggactgg
gcggcggcca aagcggtcgg acagtgctcc gagaacgggt 2760gcgcatagaa attgcatcaa
cgcatatagc gctagcagca cgccatagtg actggcgatg 2820ctgtcggaat ggacgatatc
ccgcaagagg cccggcagta ccggcataac caagcctatg 2880cctacagcat ccagggtgac
ggtgccgagg atgacgatga gcgcattgtt agatttcata 2940cacggtgcct gactgcgtta
gcaatttaac tgtgataaac taccgcatta aagcttatcg 3000atgataagct gtcaaacatg
agaattcttg aagacgaaag ggcctcgtga tacgcctatt 3060tttataggtt aatgtcatga
taataatggt ttcttagacg tcaggtggca cttttcgggg 3120aaatgtgcgc ggaaccccta
tttgtttatt tttctaaata cattcaaata tgtatccgct 3180catgagacaa taaccctgat
aaatgcttca ataatattga aaaaggaaga gtatgagtat 3240tcaacatttc cgtgtcgccc
ttattccctt ttttgcggca ttttgccttc ctgtttttgc 3300tcacccagaa acgctggtga
aagtaaaaga tgctgaagat cagttgggtg cacgagtggg 3360ttacatcgaa ctggatctca
acagcggtaa gatccttgag agttttcgcc ccgaagaacg 3420ttttccaatg atgagcactt
ttaaagttct gctatgtggc gcggtattat cccgtgttga 3480cgccgggcaa gagcaactcg
gtcgccgcat acactattct cagaatgact tggttgagta 3540ctcaccagtc acagaaaagc
atcttacgga tggcatgaca gtaagagaat tatgcagtgc 3600tgccataacc atgagtgata
acactgcggc caacttactt ctgacaacga tcggaggacc 3660gaaggagcta accgcttttt
tgcacaacat gggggatcat gtaactcgcc ttgatcgttg 3720ggaaccggag ctgaatgaag
ccataccaaa cgacgagcgt gacaccacga tgcctgcagc 3780aatggcaaca acgttgcgca
aactattaac tggcgaacta cttactctag cttcccggca 3840acaattaata gactggatgg
aggcggataa agttgcagga ccacttctgc gctcggccct 3900tccggctggc tggtttattg
ctgataaatc tggagccggt gagcgtgggt ctcgcggtat 3960cattgcagca ctggggccag
atggtaagcc ctcccgtatc gtagttatct acacgacggg 4020gagtcaggca actatggatg
aacgaaatag acagatcgct gagataggtg cctcactgat 4080taagcattgg taactgtcag
accaagttta ctcatatata ctttagattg atttaaaact 4140tcatttttaa tttaaaagga
tctaggtgaa gatccttttt gataatctca tgaccaaaat 4200cccttaacgt gagttttcgt
tccactgagc gtcagacccc gtagaaaaga tcaaaggatc 4260ttcttgagat cctttttttc
tgcgcgtaat ctgctgcttg caaacaaaaa aaccaccgct 4320accagcggtg gtttgtttgc
cggatcaaga gctaccaact ctttttccga aggtaactgg 4380cttcagcaga gcgcagatac
caaatactgt ccttctagtg tagccgtagt taggccacca 4440cttcaagaac tctgtagcac
cgcctacata cctcgctctg ctaatcctgt taccagtggc 4500tgctgccagt ggcgataagt
cgtgtcttac cgggttggac tcaagacgat agttaccgga 4560taaggcgcag cggtcgggct
gaacgggggg ttcgtgcaca cagcccagct tggagcgaac 4620gacctacacc gaactgagat
acctacagcg tgagctatga gaaagcgcca cgcttcccga 4680agggagaaag gcggacaggt
atccggtaag cggcagggtc ggaacaggag agcgcacgag 4740ggagcttcca gggggaaacg
cctggtatct ttatagtcct gtcgggtttc gccacctctg 4800acttgagcgt cgatttttgt
gatgctcgtc aggggggcgg agcctatgga aaaacgccag 4860caacgcggcc tttttacggt
tcctggcctt ttgctggcct tttgctcaca tgttctttcc 4920tgcgttatcc cctgattctg
tggataaccg tattaccgcc tttgagtgag ctgataccgc 4980tcgccgcagc cgaacgaccg
agcgcagcga gtcagtgagc gaggaagcgg aagagcgcct 5040gatgcggtat tttctcctta
cgcatctgtg cggtatttca caccgcatat atggtgcact 5100ctcagtacaa tctgctctga
tgccgcatag ttaagccagt atacactccg ctatcgctac 5160gtgactgggt catggctgcg
ccccgacacc cgccaacacc cgctgacgcg ccctgacggg 5220cttgtctgct cccggcatcc
gcttacagac aagctgtgac cgtctccggg agctgcatgt 5280gtcagaggtt ttcaccgtca
tcaccgaaac gcgcgaggca gctgcggtaa agctcatcag 5340cgtggtcgtg aagcgattca
cagatgtctg cctgttcatc cgcgtccagc tcgttgagtt 5400tctccagaag cgttaatgtc
tggcttctga taaagcgggc catgttaagg gcggtttttt 5460cctgtttggt cactgatgcc
tccgtgtaag ggggatttct gttcatgggg gtaatgatac 5520cgatgaaacg agagaggatg
ctcacgatac gggttactga tgatgaacat gcccggttac 5580tggaacgttg tgagggtaaa
caactggcgg tatggatgcg gcgggaccag agaaaaatca 5640ctcagggtca atgccagcgc
ttcgttaata cagatgtagg tgttccacag ggtagccagc 5700agcatcctgc gatgcagatc
cggaacataa tggtgcaggg cgctgacttc cgcgtttcca 5760gactttacga aacacggaaa
ccgaagacca ttcatgttgt tgctcaggtc gcagacgttt 5820tgcagcagca gtcgcttcac
gttcgctcgc gtatcggtga ttcattctgc taaccagtaa 5880ggcaaccccg ccagcctagc
cgggtcctca acgacaggag cacgatcatg cgcacccgtg 5940gccaggaccc aacgctgccc
gagatgcgcc gcgtgcggct gctggagatg gcggacgcga 6000tggatatgtt ctgccaaggg
ttggtttgcg cattcacagt tctccgcaag aattgattgg 6060ctccaattct tggagtggtg
aatccgttag cgaggtgccg ccggcttcca ttcaggtcga 6120ggtggcccgg ctccatgcac
cgcgacgcaa cgcggggagg cagacaaggt atagggcggc 6180gcctacaatc catgccaacc
cgttccatgt gctcgccgag gcggcataaa tcgccgtgac 6240gatcagcggt ccagtgatcg
aagttaggct ggtaagagcc gcgagcgatc cttgaagctg 6300tccctgatgg tcgtcatcta
cctgcctgga cagcatggcc tgcaacgcgg gcatcccgat 6360gccgccggaa gcgagaagaa
tcataatggg gaaggccatc cagcctcgcg tcgcgaacgc 6420cagcaagacg tagcccagcg
cgtcggccgc catgccggcg ataatggcct gcttctcgcc 6480gaaacgtttg gtggcgggac
cagtgacgaa ggcttgagcg agggcgtgca agattccgaa 6540taccgcaagc gacaggccga
tcatcgtcgc gctccagcga aagcggtcct cgccgaaaat 6600gacccagagc gctgccggca
cctgtcctac gagttgcatg ataaagaaga cagtcataag 6660tgcggcgacg atagtcatgc
cccgcgccca ccggaaggag ctgactgggt tgaaggctct 6720caagggcatc ggtcgatcga
cgctctccct tatgcgactc ctgcattagg aagcagccca 6780gtagtaggtt gaggccgttg
agcaccgccg ccgcaaggaa tggtgcatgc aaggagatgg 6840cgcccaacag tcccccggcc
acggggcctg ccaccatacc cacgccgaaa caagcgctca 6900tgagcccgaa gtggcgagcc
cgatcttccc catcggtgat gtcggcgata taggcgccag 6960caaccgcacc tgtggcgccg
gtgatgccgg ccacgatgcg tccggcgtag agg 70131446675DNAArtificial
SequenceDescription of Artificial Sequence Synthetic pDEST16
polynucleotide 144agatctcgat cccgcgaaat taatacgact cactataggg agaccacaac
ggtttccctc 60tagaaataat tttgtttaac tttaagaagg agatatacat atgagcgata
aaattattca 120cctgactgac gacagttttg acacggatgt actcaaagcg gacggggcga
tcctcgtcga 180tttctgggca gagtggtgcg gtccgtgcaa aatgatcgcc ccgattctgg
atgaaatcgc 240tgacgaatat cagggcaaac tgaccgttgc aaaactgaac atcgatcaaa
accctggcac 300tgcgccgaaa tatggcatcc gtggtatccc gactctgctg ctgttcaaaa
acggtgaagt 360ggcggcaacc aaagtgggtg cactgtctaa aggtcagttg aaagagttcc
tcgacgctaa 420cctggccggt tctggttctg gtgatgacga tgacaagatc acaagtttgt
acaaaaaagc 480tgaacgagaa acgtaaaatg atataaatat caatatatta aattagattt
tgcataaaaa 540acagactaca taatactgta aaacacaaca tatccagtca ctatggcggc
cgcattaggc 600accccaggct ttacacttta tgcttccggc tcgtataatg tgtggatttt
gagttaggat 660ccggcgagat tttcaggagc taaggaagct aaaatggaga aaaaaatcac
tggatatacc 720accgttgata tatcccaatg gcatcgtaaa gaacattttg aggcatttca
gtcagttgct 780caatgtacct ataaccagac cgttcagctg gatattacgg cctttttaaa
gaccgtaaag 840aaaaataagc acaagtttta tccggccttt attcacattc ttgcccgcct
gatgaatgct 900catccggaat tccgtatggc aatgaaagac ggtgagctgg tgatatggga
tagtgttcac 960ccttgttaca ccgttttcca tgagcaaact gaaacgtttt catcgctctg
gagtgaatac 1020cacgacgatt tccggcagtt tctacacata tattcgcaag atgtggcgtg
ttacggtgaa 1080aacctggcct atttccctaa agggtttatt gagaatatgt ttttcgtctc
agccaatccc 1140tgggtgagtt tcaccagttt tgatttaaac gtggccaata tggacaactt
cttcgccccc 1200gttttcacca tgggcaaata ttatacgcaa ggcgacaagg tgctgatgcc
gctggcgatt 1260caggttcatc atgccgtctg tgatggcttc catgtcggca gaatgcttaa
tgaattacaa 1320cagtactgcg atgagtggca gggcggggcg taaacgcgtg gatccggctt
actaaaagcc 1380agataacagt atgcgtattt gcgcgctgat ttttgcggta taagaatata
tactgatatg 1440tatacccgaa gtatgtcaaa aagaggtgtg ctatgaagca gcgtattaca
gtgacagttg 1500acagcgacag ctatcagttg ctcaaggcat atatgatgtc aatatctccg
gtctggtaag 1560cacaaccatg cagaatgaag cccgtcgtct gcgtgccgaa cgctggaaag
cggaaaatca 1620ggaagggatg gctgaggtcg cccggtttat tgaaatgaac ggctcttttg
ctgacgagaa 1680cagggactgg tgaaatgcag tttaaggttt acacctataa aagagagagc
cgttatcgtc 1740tgtttgtgga tgtacagagt gatattattg acacgcccgg gcgacggatg
gtgatccccc 1800tggccagtgc acgtctgctg tcagataaag tctcccgtga actttacccg
gtggtgcata 1860tcggggatga aagctggcgc atgatgacca ccgatatggc cagtgtgccg
gtctccgtta 1920tcggggaaga agtggctgat ctcagccacc gcgaaaatga catcaaaaac
gccattaacc 1980tgatgttctg gggaatataa atgtcaggct cccttataca cagccagtct
gcaggtcgac 2040catagtgact ggatatgttg tgttttacag tattatgtag tctgtttttt
atgcaaaatc 2100taatttaata tattgatatt tatatcattt tacgtttctc gttcagcttt
cttgtacaaa 2160gtggtgatga tccggctgct aacaaagccc gaaaggaagc tgagttggct
gctgccaccg 2220ctgagcaata actagcataa ccccttgggg cctctaaacg ggtcttgagg
ggttttttgc 2280tgaaaggagg aactatatcc ggatatccac aggacgggtg tggtcgccat
gatcgcgtag 2340tcgatagtgg ctccaagtag cgaagcgagc aggactgggc ggcggccaaa
gcggtcggac 2400agtgctccga gaacgggtgc gcatagaaat tgcatcaacg catatagcgc
tagcagcacg 2460ccatagtgac tggcgatgct gtcggaatgg acgatatccc gcaagaggcc
cggcagtacc 2520ggcataacca agcctatgcc tacagcatcc agggtgacgg tgccgaggat
gacgatgagc 2580gcattgttag atttcataca cggtgcctga ctgcgttagc aatttaactg
tgataaacta 2640ccgcattaaa gcttatcgat gataagctgt caaacatgag aattcttgaa
gacgaaaggg 2700cctcgtgata cgcctatttt tataggttaa tgtcatgata ataatggttt
cttagacgtc 2760aggtggcact tttcggggaa atgtgcgcgg aacccctatt tgtttatttt
tctaaataca 2820ttcaaatatg tatccgctca tgagacaata accctgataa atgcttcaat
aatattgaaa 2880aaggaagagt atgagtattc aacatttccg tgtcgccctt attccctttt
ttgcggcatt 2940ttgccttcct gtttttgctc acccagaaac gctggtgaaa gtaaaagatg
ctgaagatca 3000gttgggtgca cgagtgggtt acatcgaact ggatctcaac agcggtaaga
tccttgagag 3060ttttcgcccc gaagaacgtt ttccaatgat gagcactttt aaagttctgc
tatgtggcgc 3120ggtattatcc cgtgttgacg ccgggcaaga gcaactcggt cgccgcatac
actattctca 3180gaatgacttg gttgagtact caccagtcac agaaaagcat cttacggatg
gcatgacagt 3240aagagaatta tgcagtgctg ccataaccat gagtgataac actgcggcca
acttacttct 3300gacaacgatc ggaggaccga aggagctaac cgcttttttg cacaacatgg
gggatcatgt 3360aactcgcctt gatcgttggg aaccggagct gaatgaagcc ataccaaacg
acgagcgtga 3420caccacgatg cctgcagcaa tggcaacaac gttgcgcaaa ctattaactg
gcgaactact 3480tactctagct tcccggcaac aattaataga ctggatggag gcggataaag
ttgcaggacc 3540acttctgcgc tcggcccttc cggctggctg gtttattgct gataaatctg
gagccggtga 3600gcgtgggtct cgcggtatca ttgcagcact ggggccagat ggtaagccct
cccgtatcgt 3660agttatctac acgacgggga gtcaggcaac tatggatgaa cgaaatagac
agatcgctga 3720gataggtgcc tcactgatta agcattggta actgtcagac caagtttact
catatatact 3780ttagattgat ttaaaacttc atttttaatt taaaaggatc taggtgaaga
tcctttttga 3840taatctcatg accaaaatcc cttaacgtga gttttcgttc cactgagcgt
cagaccccgt 3900agaaaagatc aaaggatctt cttgagatcc tttttttctg cgcgtaatct
gctgcttgca 3960aacaaaaaaa ccaccgctac cagcggtggt ttgtttgccg gatcaagagc
taccaactct 4020ttttccgaag gtaactggct tcagcagagc gcagatacca aatactgtcc
ttctagtgta 4080gccgtagtta ggccaccact tcaagaactc tgtagcaccg cctacatacc
tcgctctgct 4140aatcctgtta ccagtggctg ctgccagtgg cgataagtcg tgtcttaccg
ggttggactc 4200aagacgatag ttaccggata aggcgcagcg gtcgggctga acggggggtt
cgtgcacaca 4260gcccagcttg gagcgaacga cctacaccga actgagatac ctacagcgtg
agctatgaga 4320aagcgccacg cttcccgaag ggagaaaggc ggacaggtat ccggtaagcg
gcagggtcgg 4380aacaggagag cgcacgaggg agcttccagg gggaaacgcc tggtatcttt
atagtcctgt 4440cgggtttcgc cacctctgac ttgagcgtcg atttttgtga tgctcgtcag
gggggcggag 4500cctatggaaa aacgccagca acgcggcctt tttacggttc ctggcctttt
gctggccttt 4560tgctcacatg ttctttcctg cgttatcccc tgattctgtg gataaccgta
ttaccgcctt 4620tgagtgagct gataccgctc gccgcagccg aacgaccgag cgcagcgagt
cagtgagcga 4680ggaagcggaa gagcgcctga tgcggtattt tctccttacg catctgtgcg
gtatttcaca 4740ccgcatatat ggtgcactct cagtacaatc tgctctgatg ccgcatagtt
aagccagtat 4800acactccgct atcgctacgt gactgggtca tggctgcgcc ccgacacccg
ccaacacccg 4860ctgacgcgcc ctgacgggct tgtctgctcc cggcatccgc ttacagacaa
gctgtgaccg 4920tctccgggag ctgcatgtgt cagaggtttt caccgtcatc accgaaacgc
gcgaggcagc 4980tgcggtaaag ctcatcagcg tggtcgtgaa gcgattcaca gatgtctgcc
tgttcatccg 5040cgtccagctc gttgagtttc tccagaagcg ttaatgtctg gcttctgata
aagcgggcca 5100tgttaagggc ggttttttcc tgtttggtca ctgatgcctc cgtgtaaggg
ggatttctgt 5160tcatgggggt aatgataccg atgaaacgag agaggatgct cacgatacgg
gttactgatg 5220atgaacatgc ccggttactg gaacgttgtg agggtaaaca actggcggta
tggatgcggc 5280gggaccagag aaaaatcact cagggtcaat gccagcgctt cgttaataca
gatgtaggtg 5340ttccacaggg tagccagcag catcctgcga tgcagatccg gaacataatg
gtgcagggcg 5400ctgacttccg cgtttccaga ctttacgaaa cacggaaacc gaagaccatt
catgttgttg 5460ctcaggtcgc agacgttttg cagcagcagt cgcttcacgt tcgctcgcgt
atcggtgatt 5520cattctgcta accagtaagg caaccccgcc agcctagccg ggtcctcaac
gacaggagca 5580cgatcatgcg cacccgtggc caggacccaa cgctgcccga gatgcgccgc
gtgcggctgc 5640tggagatggc ggacgcgatg gatatgttct gccaagggtt ggtttgcgca
ttcacagttc 5700tccgcaagaa ttgattggct ccaattcttg gagtggtgaa tccgttagcg
aggtgccgcc 5760ggcttccatt caggtcgagg tggcccggct ccatgcaccg cgacgcaacg
cggggaggca 5820gacaaggtat agggcggcgc ctacaatcca tgccaacccg ttccatgtgc
tcgccgaggc 5880ggcataaatc gccgtgacga tcagcggtcc agtgatcgaa gttaggctgg
taagagccgc 5940gagcgatcct tgaagctgtc cctgatggtc gtcatctacc tgcctggaca
gcatggcctg 6000caacgcgggc atcccgatgc cgccggaagc gagaagaatc ataatgggga
aggccatcca 6060gcctcgcgtc gcgaacgcca gcaagacgta gcccagcgcg tcggccgcca
tgccggcgat 6120aatggcctgc ttctcgccga aacgtttggt ggcgggacca gtgacgaagg
cttgagcgag 6180ggcgtgcaag attccgaata ccgcaagcga caggccgatc atcgtcgcgc
tccagcgaaa 6240gcggtcctcg ccgaaaatga cccagagcgc tgccggcacc tgtcctacga
gttgcatgat 6300aaagaagaca gtcataagtg cggcgacgat agtcatgccc cgcgcccacc
ggaaggagct 6360gactgggttg aaggctctca agggcatcgg tcgatcgacg ctctccctta
tgcgactcct 6420gcattaggaa gcagcccagt agtaggttga ggccgttgag caccgccgcc
gcaaggaatg 6480gtgcatgcaa ggagatggcg cccaacagtc ccccggccac ggggcctgcc
accataccca 6540cgccgaaaca agcgctcatg agcccgaagt ggcgagcccg atcttcccca
tcggtgatgt 6600cggcgatata ggcgccagca accgcacctg tggcgccggt gatgccggcc
acgatgcgtc 6660cggcgtagag gatcg
66751456354DNAArtificial SequenceDescription of Artificial
Sequence Synthetic pDEST17 polynucleotide 145cgatcccgcg aaattaatac
gactcactat agggagacca caacggtttc cctctagaaa 60taattttgtt taactttaag
aaggagatat acatatgtcg tactaccatc accatcacca 120tcacctcgaa tcaacaagtt
tgtacaaaaa agctgaacga gaaacgtaaa atgatataaa 180tatcaatata ttaaattaga
ttttgcataa aaaacagact acataatact gtaaaacaca 240acatatccag tcactatggc
ggccgcatta ggcaccccag gctttacact ttatgcttcc 300ggctcgtata atgtgtggat
tttgagttag gatccgtcga gattttcagg agctaaggaa 360gctaaaatgg agaaaaaaat
cactggatat accaccgttg atatatccca atggcatcgt 420aaagaacatt ttgaggcatt
tcagtcagtt gctcaatgta cctataacca gaccgttcag 480ctggatatta cggccttttt
aaagaccgta aagaaaaata agcacaagtt ttatccggcc 540tttattcaca ttcttgcccg
cctgatgaat gctcatccgg aattccgtat ggcaatgaaa 600gacggtgagc tggtgatatg
ggatagtgtt cacccttgtt acaccgtttt ccatgagcaa 660actgaaacgt tttcatcgct
ctggagtgaa taccacgacg atttccggca gtttctacac 720atatattcgc aagatgtggc
gtgttacggt gaaaacctgg cctatttccc taaagggttt 780attgagaata tgtttttcgt
ctcagccaat ccctgggtga gtttcaccag ttttgattta 840aacgtggcca atatggacaa
cttcttcgcc cccgttttca ccatgggcaa atattatacg 900caaggcgaca aggtgctgat
gccgctggcg attcaggttc atcatgccgt ctgtgatggc 960ttccatgtcg gcagaatgct
taatgaatta caacagtact gcgatgagtg gcagggcggg 1020gcgtaaagat ctggatccgg
cttactaaaa gccagataac agtatgcgta tttgcgcgct 1080gatttttgcg gtataagaat
atatactgat atgtataccc gaagtatgtc aaaaagaggt 1140gtgctatgaa gcagcgtatt
acagtgacag ttgacagcga cagctatcag ttgctcaagg 1200catatatgat gtcaatatct
ccggtctggt aagcacaacc atgcagaatg aagcccgtcg 1260tctgcgtgcc gaacgctgga
aagcggaaaa tcaggaaggg atggctgagg tcgcccggtt 1320tattgaaatg aacggctctt
ttgctgacga gaacagggac tggtgaaatg cagtttaagg 1380tttacaccta taaaagagag
agccgttatc gtctgtttgt ggatgtacag agtgatatta 1440ttgacacgcc cgggcgacgg
atggtgatcc ccctggccag tgcacgtctg ctgtcagata 1500aagtctcccg tgaactttac
ccggtggtgc atatcgggga tgaaagctgg cgcatgatga 1560ccaccgatat ggccagtgtg
ccggtctccg ttatcgggga agaagtggct gatctcagcc 1620accgcgaaaa tgacatcaaa
aacgccatta acctgatgtt ctggggaata taaatgtcag 1680gctcccttat acacagccag
tctgcaggtc gaccatagtg actggatatg ttgtgtttta 1740cagtattatg tagtctgttt
tttatgcaaa atctaattta atatattgat atttatatca 1800ttttacgttt ctcgttcagc
tttcttgtac aaagtggttg attcgaggct gctaacaaag 1860cccgaaagga agctgagttg
gctgctgcca ccgctgagca ataactagca taaccccttg 1920gggcctctaa acgggtcttg
aggggttttt tgctgaaagg aggaactata tccggatatc 1980cacaggacgg gtgtggtcgc
catgatcgcg tagtcgatag tggctccaag tagcgaagcg 2040agcaggactg ggcggcggcc
aaagcggtcg gacagtgctc cgagaacggg tgcgcataga 2100aattgcatca acgcatatag
cgctagcagc acgccatagt gactggcgat gctgtcggaa 2160tggacgatat cccgcaagag
gcccggcagt accggcataa ccaagcctat gcctacagca 2220tccagggtga cggtgccgag
gatgacgatg agcgcattgt tagatttcat acacggtgcc 2280tgactgcgtt agcaatttaa
ctgtgataaa ctaccgcatt aaagcttatc gatgataagc 2340tgtcaaacat gagaattctt
gaagacgaaa gggcctcgtg atacgcctat ttttataggt 2400taatgtcatg ataataatgg
tttcttagac gtcaggtggc acttttcggg gaaatgtgcg 2460cggaacccct atttgtttat
ttttctaaat acattcaaat atgtatccgc tcatgagaca 2520ataaccctga taaatgcttc
aataatattg aaaaaggaag agtatgagta ttcaacattt 2580ccgtgtcgcc cttattccct
tttttgcggc attttgcctt cctgtttttg ctcacccaga 2640aacgctggtg aaagtaaaag
atgctgaaga tcagttgggt gcacgagtgg gttacatcga 2700actggatctc aacagcggta
agatccttga gagttttcgc cccgaagaac gttttccaat 2760gatgagcact tttaaagttc
tgctatgtgg cgcggtatta tcccgtgttg acgccgggca 2820agagcaactc ggtcgccgca
tacactattc tcagaatgac ttggttgagt actcaccagt 2880cacagaaaag catcttacgg
atggcatgac agtaagagaa ttatgcagtg ctgccataac 2940catgagtgat aacactgcgg
ccaacttact tctgacaacg atcggaggac cgaaggagct 3000aaccgctttt ttgcacaaca
tgggggatca tgtaactcgc cttgatcgtt gggaaccgga 3060gctgaatgaa gccataccaa
acgacgagcg tgacaccacg atgcctgcag caatggcaac 3120aacgttgcgc aaactattaa
ctggcgaact acttactcta gcttcccggc aacaattaat 3180agactggatg gaggcggata
aagttgcagg accacttctg cgctcggccc ttccggctgg 3240ctggtttatt gctgataaat
ctggagccgg tgagcgtggg tctcgcggta tcattgcagc 3300actggggcca gatggtaagc
cctcccgtat cgtagttatc tacacgacgg ggagtcaggc 3360aactatggat gaacgaaata
gacagatcgc tgagataggt gcctcactga ttaagcattg 3420gtaactgtca gaccaagttt
actcatatat actttagatt gatttaaaac ttcattttta 3480atttaaaagg atctaggtga
agatcctttt tgataatctc atgaccaaaa tcccttaacg 3540tgagttttcg ttccactgag
cgtcagaccc cgtagaaaag atcaaaggat cttcttgaga 3600tccttttttt ctgcgcgtaa
tctgctgctt gcaaacaaaa aaaccaccgc taccagcggt 3660ggtttgtttg ccggatcaag
agctaccaac tctttttccg aaggtaactg gcttcagcag 3720agcgcagata ccaaatactg
tccttctagt gtagccgtag ttaggccacc acttcaagaa 3780ctctgtagca ccgcctacat
acctcgctct gctaatcctg ttaccagtgg ctgctgccag 3840tggcgataag tcgtgtctta
ccgggttgga ctcaagacga tagttaccgg ataaggcgca 3900gcggtcgggc tgaacggggg
gttcgtgcac acagcccagc ttggagcgaa cgacctacac 3960cgaactgaga tacctacagc
gtgagctatg agaaagcgcc acgcttcccg aagggagaaa 4020ggcggacagg tatccggtaa
gcggcagggt cggaacagga gagcgcacga gggagcttcc 4080agggggaaac gcctggtatc
tttatagtcc tgtcgggttt cgccacctct gacttgagcg 4140tcgatttttg tgatgctcgt
caggggggcg gagcctatgg aaaaacgcca gcaacgcggc 4200ctttttacgg ttcctggcct
tttgctggcc ttttgctcac atgttctttc ctgcgttatc 4260ccctgattct gtggataacc
gtattaccgc ctttgagtga gctgataccg ctcgccgcag 4320ccgaacgacc gagcgcagcg
agtcagtgag cgaggaagcg gaagagcgcc tgatgcggta 4380ttttctcctt acgcatctgt
gcggtatttc acaccgcata tatggtgcac tctcagtaca 4440atctgctctg atgccgcata
gttaagccag tatacactcc gctatcgcta cgtgactggg 4500tcatggctgc gccccgacac
ccgccaacac ccgctgacgc gccctgacgg gcttgtctgc 4560tcccggcatc cgcttacaga
caagctgtga ccgtctccgg gagctgcatg tgtcagaggt 4620tttcaccgtc atcaccgaaa
cgcgcgaggc agctgcggta aagctcatca gcgtggtcgt 4680gaagcgattc acagatgtct
gcctgttcat ccgcgtccag ctcgttgagt ttctccagaa 4740gcgttaatgt ctggcttctg
ataaagcggg ccatgttaag ggcggttttt tcctgtttgg 4800tcactgatgc ctccgtgtaa
gggggatttc tgttcatggg ggtaatgata ccgatgaaac 4860gagagaggat gctcacgata
cgggttactg atgatgaaca tgcccggtta ctggaacgtt 4920gtgagggtaa acaactggcg
gtatggatgc ggcgggacca gagaaaaatc actcagggtc 4980aatgccagcg cttcgttaat
acagatgtag gtgttccaca gggtagccag cagcatcctg 5040cgatgcagat ccggaacata
atggtgcagg gcgctgactt ccgcgtttcc agactttacg 5100aaacacggaa accgaagacc
attcatgttg ttgctcaggt cgcagacgtt ttgcagcagc 5160agtcgcttca cgttcgctcg
cgtatcggtg attcattctg ctaaccagta aggcaacccc 5220gccagcctag ccgggtcctc
aacgacagga gcacgatcat gcgcacccgt ggccaggacc 5280caacgctgcc cgagatgcgc
cgcgtgcggc tgctggagat ggcggacgcg atggatatgt 5340tctgccaagg gttggtttgc
gcattcacag ttctccgcaa gaattgattg gctccaattc 5400ttggagtggt gaatccgtta
gcgaggtgcc gccggcttcc attcaggtcg aggtggcccg 5460gctccatgca ccgcgacgca
acgcggggag gcagacaagg tatagggcgg cgcctacaat 5520ccatgccaac ccgttccatg
tgctcgccga ggcggcataa atcgccgtga cgatcagcgg 5580tccagtgatc gaagttaggc
tggtaagagc cgcgagcgat ccttgaagct gtccctgatg 5640gtcgtcatct acctgcctgg
acagcatggc ctgcaacgcg ggcatcccga tgccgccgga 5700agcgagaaga atcataatgg
ggaaggccat ccagcctcgc gtcgcgaacg ccagcaagac 5760gtagcccagc gcgtcggccg
ccatgccggc gataatggcc tgcttctcgc cgaaacgttt 5820ggtggcggga ccagtgacga
aggcttgagc gagggcgtgc aagattccga ataccgcaag 5880cgacaggccg atcatcgtcg
cgctccagcg aaagcggtcc tcgccgaaaa tgacccagag 5940cgctgccggc acctgtccta
cgagttgcat gataaagaag acagtcataa gtgcggcgac 6000gatagtcatg ccccgcgccc
accggaagga gctgactggg ttgaaggctc tcaagggcat 6060cggtcgatcg acgctctccc
ttatgcgact cctgcattag gaagcagccc agtagtaggt 6120tgaggccgtt gagcaccgcc
gccgcaagga atggtgcatg caaggagatg gcgcccaaca 6180gtcccccggc cacggggcct
gccaccatac ccacgccgaa acaagcgctc atgagcccga 6240agtggcgagc ccgatcttcc
ccatcggtga tgtcggcgat ataggcgcca gcaaccgcac 6300ctgtggcgcc ggtgatgccg
gccacgatgc gtccggcgta gaggatcgag atct 63541466613DNAArtificial
SequenceDescription of Artificial Sequence Synthetic pDEST18
polynucleotide 146gacgcgccct gtagcggcgc attaagcgcg gcgggtgtgg tggttacgcg
cagcgtgacc 60gctacacttg ccagcgccct agcgcccgct cctttcgctt tcttcccttc
ctttctcgcc 120acgttcgccg gctttccccg tcaagctcta aatcgggggc tccctttagg
gttccgattt 180agtgctttac ggcacctcga ccccaaaaaa cttgattagg gtgatggttc
acgtagtggg 240ccatcgccct gatagacggt ttttcgccct ttgacgttgg agtccacgtt
ctttaatagt 300ggactcttgt tccaaactgg aacaacactc aaccctatct cggtctattc
ttttgattta 360taagggattt tgccgatttc ggcctattgg ttaaaaaatg agctgattta
acaaaaattt 420aacgcgaatt ttaacaaaat attaacgttt acaatttcag gtggcacttt
tcggggaaat 480gtgcgcggaa cccctatttg tttatttttc taaatacatt caaatatgta
tccgctcatg 540agacaataac cctgataaat gcttcaataa tattgaaaaa ggaagagtat
gagtattcaa 600catttccgtg tcgcccttat tccctttttt gcggcatttt gccttcctgt
ttttgctcac 660ccagaaacgc tggtgaaagt aaaagatgct gaagatcagt tgggtgcacg
agtgggttac 720atcgaactgg atctcaacag cggtaagatc cttgagagtt ttcgccccga
agaacgtttt 780ccaatgatga gcacttttaa agttctgcta tgtggcgcgg tattatcccg
tattgacgcc 840gggcaagagc aactcggtcg ccgcatacac tattctcaga atgacttggt
tgagtactca 900ccagtcacag aaaagcatct tacggatggc atgacagtaa gagaattatg
cagtgctgcc 960ataaccatga gtgataacac tgcggccaac ttacttctga caacgatcgg
aggaccgaag 1020gagctaaccg cttttttgca caacatgggg gatcatgtaa ctcgccttga
tcgttgggaa 1080ccggagctga atgaagccat accaaacgac gagcgtgaca ccacgatgcc
tgtagcaatg 1140gcaacaacgt tgcgcaaact attaactggc gaactactta ctctagcttc
ccggcaacaa 1200ttaatagact ggatggaggc ggataaagtt gcaggaccac ttctgcgctc
ggcccttccg 1260gctggctggt ttattgctga taaatctgga gccggtgagc gtgggtctcg
cggtatcatt 1320gcagcactgg ggccagatgg taagccctcc cgtatcgtag ttatctacac
gacggggagt 1380caggcaacta tggatgaacg aaatagacag atcgctgaga taggtgcctc
actgattaag 1440cattggtaac tgtcagacca agtttactca tatatacttt agattgattt
aaaacttcat 1500ttttaattta aaaggatcta ggtgaagatc ctttttgata atctcatgac
caaaatccct 1560taacgtgagt tttcgttcca ctgagcgtca gaccccgtag aaaagatcaa
aggatcttct 1620tgagatcctt tttttctgcg cgtaatctgc tgcttgcaaa caaaaaaacc
accgctacca 1680gcggtggttt gtttgccgga tcaagagcta ccaactcttt ttccgaaggt
aactggcttc 1740agcagagcgc agataccaaa tactgtcctt ctagtgtagc cgtagttagg
ccaccacttc 1800aagaactctg tagcaccgcc tacatacctc gctctgctaa tcctgttacc
agtggctgct 1860gccagtggcg ataagtcgtg tcttaccggg ttggactcaa gacgatagtt
accggataag 1920gcgcagcggt cgggctgaac ggggggttcg tgcacacagc ccagcttgga
gcgaacgacc 1980tacaccgaac tgagatacct acagcgtgag cattgagaaa gcgccacgct
tcccgaaggg 2040agaaaggcgg acaggtatcc ggtaagcggc agggtcggaa caggagagcg
cacgagggag 2100cttccagggg gaaacgcctg gtatctttat agtcctgtcg ggtttcgcca
cctctgactt 2160gagcgtcgat ttttgtgatg ctcgtcaggg gggcggagcc tatggaaaaa
cgccagcaac 2220gcggcctttt tacggttcct ggccttttgc tggccttttg ctcacatgtt
ctttcctgcg 2280ttatcccctg attctgtgga taaccgtatt accgcctttg agtgagctga
taccgctcgc 2340cgcagccgaa cgaccgagcg cagcgagtca gtgagcgagg aagcggaaga
gcgcctgatg 2400cggtattttc tccttacgca tctgtgcggt atttcacacc gcagaccagc
cgcgtaacct 2460ggcaaaatcg gttacggttg agtaataaat ggatgccctg cgtaagcggg
tgtgggcgga 2520caataaagtc ttaaactgaa caaaatagat ctaaactatg acaataaagt
cttaaactag 2580acagaatagt tgtaaactga aatcagtcca gttatgctgt gaaaaagcat
actggacttt 2640tgttatggct aaagcaaact cttcattttc tgaagtgcaa attgcccgtc
gtattaaaga 2700ggggcgtggc caagggcatg gtaaagacta tattcgcggc gttgtgacaa
tttaccgaac 2760aactccgcgg ccgggaagcc gatctcggct tgaacgaatt gttaggtggc
ggtacttggg 2820tcgatatcaa agtgcatcac ttcttcccgt atgcccaact ttgtatagag
agccactgcg 2880ggatcgtcac cgtaatctgc ttgcacgtag atcacataag caccaagcgc
gttggcctca 2940tgcttgagga gattgatgag cgcggtggca atgccctgcc tccggtgctc
gccggagact 3000gcgagatcat agatatagat ctcactacgc ggctgctcaa acctgggcag
aacgtaagcc 3060gcgagagcgc caacaaccgc ttcttggtcg aaggcagcaa gcgcgatgaa
tgtcttacta 3120cggagcaagt tcccgaggta atcggagtcc ggctgatgtt gggagtaggt
ggctacgtct 3180ccgaactcac gaccgaaaag atcaagagca gcccgcatgg atttgacttg
gtcagggccg 3240agcctacatg tgcgaatgat gcccatactt gagccaccta actttgtttt
agggcgactg 3300ccctgctgcg taacatcgtt gctgctgcgt aacatcgttg ctgctccata
acatcaaaca 3360tcgacccacg gcgtaacgcg cttgctgctt ggatgcccga ggcatagact
gtacaaaaaa 3420acagtcataa caagccatga aaaccgccac tgcgccgtta ccaccgctgc
gttcggtcaa 3480ggttctggac cagttgcgtg agcgcatacg ctacttgcat tacagtttac
gaaccgaaca 3540ggcttatgtc aactgggttc gtgccttcat ccgtttccac ggtgtgcgtc
acccggcaac 3600cttgggcagc agcgaagtcg aggcatttct gtcctggctg gcgaacgagc
gcaaggtttc 3660ggtctccacg catcgtcagg cattggcggc cttgctgttc ttctacggca
aggtgctgtg 3720cacggatctg ccctggcttc aggagatcgg aagacctcgg ccgtcgcggc
gcttgccggt 3780ggtgctgacc ccggatgaag tggttcgcat cctcggtttt ctggaaggcg
agcatcgttt 3840gttcgcccag gactctagct atagttctag tggttggcta cgtatcgagc
aagaaaataa 3900aacgccaaac gcgttggagt cttgtgtgct atttttacaa agattcagaa
atacgcatca 3960cttacaacaa gggggactat gaaattatgc attttgagga tgccgggacc
tttaattcaa 4020cccaacacaa tatattatag ttaaataaga attatttatc aaatcatttg
tatattaatt 4080aaaatactat actgtaaatt acattttatt tacaatgagg atcatcacaa
gtttgtacaa 4140aaaagctgaa cgagaaacgt aaaatgatat aaatatcaat atattaaatt
agattttgca 4200taaaaaacag actacataat actgtaaaac acaacatatc cagtcactat
ggcggccgct 4260aagttggcag catcacccga cgcactttgc gccgaataaa tacctgtgac
ggaagatcac 4320ttcgcagaat aaataaatcc tggtgtccct gttgataccg ggaagccctg
ggccaacttt 4380tggcgaaaat gagacgttga tcggcacgta agaggttcca actttcacca
taatgaaata 4440agatcactac cgggcgtatt ttttgagtta tcgagatttt caggagctaa
ggaagctaaa 4500atggagaaaa aaatcactgg atataccacc gttgatatat cccaatggca
tcgtaaagaa 4560cattttgagg catttcagtc agttgctcaa tgtacctata accagaccgt
tcagctggat 4620attacggcct ttttaaagac cgtaaagaaa aataagcaca agttttatcc
ggcctttatt 4680cacattcttg cccgcctgat gaatgctcat ccggaattcc gtatggcaat
gaaagacggt 4740gagctggtga tatgggatag tgttcaccct tgttacaccg ttttccatga
gcaaactgaa 4800acgttttcat cgctctggag tgaataccac gacgatttcc ggcagtttct
acacatatat 4860tcgcaagatg tggcgtgtta cggtgaaaac ctggcctatt tccctaaagg
gtttattgag 4920aatatgtttt tcgtctcagc caatccctgg gtgagtttca ccagttttga
tttaaacgtg 4980gccaatatgg acaacttctt cgcccccgtt ttcaccatgg gcaaatatta
tacgcaaggc 5040gacaaggtgc tgatgccgct ggcgattcag gttcatcatg ccgtctgtga
tggcttccat 5100gtcggcagaa tgcttaatga attacaacag tactgcgatg agtggcaggg
cggggcgtaa 5160acgcgtggat ccggcttact aaaagccaga taacagtatg cgtatttgcg
cgctgatttt 5220tgcggtataa gaatatatac tgatatgtat acccgaagta tgtcaaaaag
aggtgtgcta 5280tgaagcagcg tattacagtg acagttgaca gcgacagcta tcagttgctc
aaggcatata 5340tgatgtcaat atctccggtc tggtaagcac aaccatgcag aatgaagccc
gtcgtctgcg 5400tgccgaacgc tggaaagcgg aaaatcagga agggatggct gaggtcgccc
ggtttattga 5460aatgaacggc tcttttgctg acgagaacag ggactggtga aatgcagttt
aaggtttaca 5520cctataaaag agagagccgt tatcgtctgt ttgtggatgt acagagtgat
attattgaca 5580cgcccgggcg acggatggtg atccccctgg ccagtgcacg tctgctgtca
gataaagtct 5640cccgtgaact ttacccggtg gtgcatatcg gggatgaaag ctggcgcatg
atgaccaccg 5700atatggccag tgtgccggtc tccgttatcg gggaagaagt ggctgatctc
agccaccgcg 5760aaaatgacat caaaaacgcc attaacctga tgttctgggg aatataaatg
tcaggctccc 5820ttatacacag ccagtctgca ggtcgaccat agtgactgga tatgttgtgt
tttacagtat 5880tatgtagtct gttttttatg caaaatctaa tttaatatat tgatatttat
atcattttac 5940gtttctcgtt cagctttctt gtacaaagtg gtgatagctt gtcgagaagt
actagaggat 6000cataatcagc cataccacat ttgtagaggt tttacttgct ttaaaaaacc
tcccacacct 6060ccccctgaac ctgaaacata aaatgaatgc aattgttgtt gttaacttgt
ttattgcagc 6120ttataatggt tacaaataaa gcaatagcat cacaaatttc acaaataaag
catttttttc 6180actgcattct agttgtggtt tgtccaaact catcaatgta tcttatcatg
tctggatctg 6240atcactgctt gagcctagga gatccgaacc agataagtga aatctagttc
caaactattt 6300tgtcattttt aattttcgta ttagcttacg acgctacacc cagttcccat
ctattttgtc 6360actcttccct aaataatcct taaaaactcc atttccaccc ctcccagttc
ccaactattt 6420tgtccgccca cagcggggca tttttcttcc tgttatgttt ttaatcaaac
atcctgccaa 6480ctccatgtga caaaccgtca tcttcggcta ctttttctct gtcacagaat
gaaaattttt 6540ctgtcatctc ttcgttatta atgtttgtaa ttgactgaat atcaacgctt
atttgcagcc 6600tgaatggcga atg
66131476668DNAArtificial SequenceDescription of Artificial
Sequence Synthetic pDEST19 polynucleotide 147agtggttcgc atcctcggtt
ttctggaagg cgagcatcgt ttgttcgccc aggactctag 60ctatagttct agtggttggc
tacgtatatc aaatacttgt aggtgacgcc gtcatctttc 120cattgtaacg taaatggcaa
cttgtagatg aacgcgctgt caaaaaaccg gccagtttct 180tccacaaact cgcgcacggc
tgtctcgtaa acttttgcgt cgcaacaatc gcgatgacct 240cgtggtatgg aaattttttc
taaaaaagtg tcgttcatgt cggcggcggg cgcgttcgcg 300ctccggtacg cgcgacgggc
acacagcagg acagccttgt ccggctcgat tatcataaac 360aatcctgcag gcatgcaagc
tcggatcatc acaagtttgt acaaaaaagc tgaacgagaa 420acgtaaaatg atataaatat
caatatatta aattagattt tgcataaaaa acagactaca 480taatactgta aaacacaaca
tatccagtca ctatggcggc cgctaagttg gcagcatcac 540ccgacgcact ttgcgccgaa
taaatacctg tgacggaaga tcacttcgca gaataaataa 600atcctggtgt ccctgttgat
accgggaagc cctgggccaa cttttggcga aaatgagacg 660ttgatcggca cgtaagaggt
tccaactttc accataatga aataagatca ctaccgggcg 720tattttttga gttatcgaga
ttttcaggag ctaaggaagc taaaatggag aaaaaaatca 780ctggatatac caccgttgat
atatcccaat ggcatcgtaa agaacatttt gaggcatttc 840agtcagttgc tcaatgtacc
tataaccaga ccgttcagct ggatattacg gcctttttaa 900agaccgtaaa gaaaaataag
cacaagtttt atccggcctt tattcacatt cttgcccgcc 960tgatgaatgc tcatccggaa
ttccgtatgg caatgaaaga cggtgagctg gtgatatggg 1020atagtgttca cccttgttac
accgttttcc atgagcaaac tgaaacgttt tcatcgctct 1080ggagtgaata ccacgacgat
ttccggcagt ttctacacat atattcgcaa gatgtggcgt 1140gttacggtga aaacctggcc
tatttcccta aagggtttat tgagaatatg tttttcgtct 1200cagccaatcc ctgggtgagt
ttcaccagtt ttgatttaaa cgtggccaat atggacaact 1260tcttcgcccc cgttttcacc
atgggcaaat attatacgca aggcgacaag gtgctgatgc 1320cgctggcgat tcaggttcat
catgccgtct gtgatggctt ccatgtcggc agaatgctta 1380atgaattaca acagtactgc
gatgagtggc agggcggggc gtaaacgcgt ggatccggct 1440tactaaaagc cagataacag
tatgcgtatt tgcgcgctga tttttgcggt ataagaatat 1500atactgatat gtatacccga
agtatgtcaa aaagaggtgt gctatgaagc agcgtattac 1560agtgacagtt gacagcgaca
gctatcagtt gctcaaggca tatatgatgt caatatctcc 1620ggtctggtaa gcacaaccat
gcagaatgaa gcccgtcgtc tgcgtgccga acgctggaaa 1680gcggaaaatc aggaagggat
ggctgaggtc gcccggttta ttgaaatgaa cggctctttt 1740gctgacgaga acagggactg
gtgaaatgca gtttaaggtt tacacctata aaagagagag 1800ccgttatcgt ctgtttgtgg
atgtacagag tgatattatt gacacgcccg ggcgacggat 1860ggtgatcccc ctggccagtg
cacgtctgct gtcagataaa gtctcccgtg aactttaccc 1920ggtggtgcat atcggggatg
aaagctggcg catgatgacc accgatatgg ccagtgtgcc 1980ggtctccgtt atcggggaag
aagtggctga tctcagccac cgcgaaaatg acatcaaaaa 2040cgccattaac ctgatgttct
ggggaatata aatgtcaggc tcccttatac acagccagtc 2100tgcaggtcga ccatagtgac
tggatatgtt gtgttttaca gtattatgta gtctgttttt 2160tatgcaaaat ctaatttaat
atattgatat ttatatcatt ttacgtttct cgttcagctt 2220tcttgtacaa agtggtgatc
gagaagtact agaggatcat aatcagccat accacatttg 2280tagaggtttt acttgcttta
aaaaacctcc cacacctccc cctgaacctg aaacataaaa 2340tgaatgcaat tgttgttgtt
aacttgttta ttgcagctta taatggttac aaataaagca 2400atagcatcac aaatttcaca
aataaagcat ttttttcact gcattctagt tgtggtttgt 2460ccaaactcat caatgtatct
tatcatgtct ggatctgatc actgcttgag cctaggagat 2520ccgaaccaga taagtgaaat
ctagttccaa actattttgt catttttaat tttcgtatta 2580gcttacgacg ctacacccag
ttcccatcta ttttgtcact cttccctaaa taatccttaa 2640aaactccatt tccacccctc
ccagttccca actattttgt ccgcccacag cggggcattt 2700ttcttcctgt tatgttttta
atcaaacatc ctgccaactc catgtgacaa accgtcatct 2760tcggctactt tttctctgtc
acagaatgaa aatttttctg tcatctcttc gttattaatg 2820tttgtaattg actgaatatc
aacgcttatt tgcagcctga atggcgaatg gacgcgccct 2880gtagcggcgc attaagcgcg
gcgggtgtgg tggttacgcg cagcgtgacc gctacacttg 2940ccagcgccct agcgcccgct
cctttcgctt tcttcccttc ctttctcgcc acgttcgccg 3000gctttccccg tcaagctcta
aatcgggggc tccctttagg gttccgattt agtgctttac 3060ggcacctcga ccccaaaaaa
cttgattagg gtgatggttc acgtagtggg ccatcgccct 3120gatagacggt ttttcgccct
ttgacgttgg agtccacgtt ctttaatagt ggactcttgt 3180tccaaactgg aacaacactc
aaccctatct cggtctattc ttttgattta taagggattt 3240tgccgatttc ggcctattgg
ttaaaaaatg agctgattta acaaaaattt aacgcgaatt 3300ttaacaaaat attaacgttt
acaatttcag gtggcacttt tcggggaaat gtgcgcggaa 3360cccctatttg tttatttttc
taaatacatt caaatatgta tccgctcatg agacaataac 3420cctgataaat gcttcaataa
tattgaaaaa ggaagagtat gagtattcaa catttccgtg 3480tcgcccttat tccctttttt
gcggcatttt gccttcctgt ttttgctcac ccagaaacgc 3540tggtgaaagt aaaagatgct
gaagatcagt tgggtgcacg agtgggttac atcgaactgg 3600atctcaacag cggtaagatc
cttgagagtt ttcgccccga agaacgtttt ccaatgatga 3660gcacttttaa agttctgcta
tgtggcgcgg tattatcccg tattgacgcc gggcaagagc 3720aactcggtcg ccgcatacac
tattctcaga atgacttggt tgagtactca ccagtcacag 3780aaaagcatct tacggatggc
atgacagtaa gagaattatg cagtgctgcc ataaccatga 3840gtgataacac tgcggccaac
ttacttctga caacgatcgg aggaccgaag gagctaaccg 3900cttttttgca caacatgggg
gatcatgtaa ctcgccttga tcgttgggaa ccggagctga 3960atgaagccat accaaacgac
gagcgtgaca ccacgatgcc tgtagcaatg gcaacaacgt 4020tgcgcaaact attaactggc
gaactactta ctctagcttc ccggcaacaa ttaatagact 4080ggatggaggc ggataaagtt
gcaggaccac ttctgcgctc ggcccttccg gctggctggt 4140ttattgctga taaatctgga
gccggtgagc gtgggtctcg cggtatcatt gcagcactgg 4200ggccagatgg taagccctcc
cgtatcgtag ttatctacac gacggggagt caggcaacta 4260tggatgaacg aaatagacag
atcgctgaga taggtgcctc actgattaag cattggtaac 4320tgtcagacca agtttactca
tatatacttt agattgattt aaaacttcat ttttaattta 4380aaaggatcta ggtgaagatc
ctttttgata atctcatgac caaaatccct taacgtgagt 4440tttcgttcca ctgagcgtca
gaccccgtag aaaagatcaa aggatcttct tgagatcctt 4500tttttctgcg cgtaatctgc
tgcttgcaaa caaaaaaacc accgctacca gcggtggttt 4560gtttgccgga tcaagagcta
ccaactcttt ttccgaaggt aactggcttc agcagagcgc 4620agataccaaa tactgtcctt
ctagtgtagc cgtagttagg ccaccacttc aagaactctg 4680tagcaccgcc tacatacctc
gctctgctaa tcctgttacc agtggctgct gccagtggcg 4740ataagtcgtg tcttaccggg
ttggactcaa gacgatagtt accggataag gcgcagcggt 4800cgggctgaac ggggggttcg
tgcacacagc ccagcttgga gcgaacgacc tacaccgaac 4860tgagatacct acagcgtgag
cattgagaaa gcgccacgct tcccgaaggg agaaaggcgg 4920acaggtatcc ggtaagcggc
agggtcggaa caggagagcg cacgagggag cttccagggg 4980gaaacgcctg gtatctttat
agtcctgtcg ggtttcgcca cctctgactt gagcgtcgat 5040ttttgtgatg ctcgtcaggg
gggcggagcc tatggaaaaa cgccagcaac gcggcctttt 5100tacggttcct ggccttttgc
tggccttttg ctcacatgtt ctttcctgcg ttatcccctg 5160attctgtgga taaccgtatt
accgcctttg agtgagctga taccgctcgc cgcagccgaa 5220cgaccgagcg cagcgagtca
gtgagcgagg aagcggaaga gcgcctgatg cggtattttc 5280tccttacgca tctgtgcggt
atttcacacc gcagaccagc cgcgtaacct ggcaaaatcg 5340gttacggttg agtaataaat
ggatgccctg cgtaagcggg tgtgggcgga caataaagtc 5400ttaaactgaa caaaatagat
ctaaactatg acaataaagt cttaaactag acagaatagt 5460tgtaaactga aatcagtcca
gttatgctgt gaaaaagcat actggacttt tgttatggct 5520aaagcaaact cttcattttc
tgaagtgcaa attgcccgtc gtattaaaga ggggcgtggc 5580caagggcatg gtaaagacta
tattcgcggc gttgtgacaa tttaccgaac aactccgcgg 5640ccgggaagcc gatctcggct
tgaacgaatt gttaggtggc ggtacttggg tcgatatcaa 5700agtgcatcac ttcttcccgt
atgcccaact ttgtatagag agccactgcg ggatcgtcac 5760cgtaatctgc ttgcacgtag
atcacataag caccaagcgc gttggcctca tgcttgagga 5820gattgatgag cgcggtggca
atgccctgcc tccggtgctc gccggagact gcgagatcat 5880agatatagat ctcactacgc
ggctgctcaa acctgggcag aacgtaagcc gcgagagcgc 5940caacaaccgc ttcttggtcg
aaggcagcaa gcgcgatgaa tgtcttacta cggagcaagt 6000tcccgaggta atcggagtcc
ggctgatgtt gggagtaggt ggctacgtct ccgaactcac 6060gaccgaaaag atcaagagca
gcccgcatgg atttgacttg gtcagggccg agcctacatg 6120tgcgaatgat gcccatactt
gagccaccta actttgtttt agggcgactg ccctgctgcg 6180taacatcgtt gctgctgcgt
aacatcgttg ctgctccata acatcaaaca tcgacccacg 6240gcgtaacgcg cttgctgctt
ggatgcccga ggcatagact gtacaaaaaa acagtcataa 6300caagccatga aaaccgccac
tgcgccgtta ccaccgctgc gttcggtcaa ggttctggac 6360cagttgcgtg agcgcatacg
ctacttgcat tacagtttac gaaccgaaca ggcttatgtc 6420aactgggttc gtgccttcat
ccgtttccac ggtgtgcgtc acccggcaac cttgggcagc 6480agcgaagtcg aggcatttct
gtcctggctg gcgaacgagc gcaaggtttc ggtctccacg 6540catcgtcagg cattggcggc
cttgctgttc ttctacggca aggtgctgtg cacggatctg 6600ccctggcttc aggagatcgg
aagacctcgg ccgtcgcggc gcttgccggt ggtgctgacc 6660ccggatga
66681487066DNAArtificial
SequenceDescription of Artificial Sequence Synthetic pDEST20
polynucleotide 148ccactgcgcc gttaccaccg ctgcgttcgg tcaaggttct ggaccagttg
cgtgagcgca 60tacgctactt gcattacagt ttacgaaccg aacaggctta tgtcaactgg
gttcgtgcct 120tcatccgttt ccacggtgtg cgtcacccgg caaccttggg cagcagcgaa
gtcgaggcat 180ttctgtcctg gctggcgaac gagcgcaagg tttcggtctc cacgcatcgt
caggcattgg 240cggccttgct gttcttctac ggcaaggtgc tgtgcacgga tctgccctgg
cttcaggaga 300tcggaagacc tcggccgtcg cggcgcttgc cggtggtgct gaccccggat
gaagtggttc 360gcatcctcgg ttttctggaa ggcgagcatc gtttgttcgc ccaggactct
agctatagtt 420ctagtggttg gctacgtata ctccggaata ttaatagatc atggagataa
ttaaaatgat 480aaccatctcg caaataaata agtattttac tgttttcgta acagttttgt
aataaaaaaa 540cctataaata ttccggatta ttcataccgt cccaccatcg ggcgcggatc
catggcccct 600atactaggtt attggaaaat taagggcctt gtgcaaccca ctcgacttct
tttggaatat 660cttgaagaaa aatatgaaga gcatttgtat gagcgcgatg aaggtgataa
atggcgaaac 720aaaaagtttg aattgggttt ggagtttccc aatcttcctt attatattga
tggtgatgtt 780aaattaacac agtctatggc catcatacgt tatatagctg acaagcacaa
catgttgggt 840ggttgtccaa aagagcgtgc agagatttca atgcttgaag gagcggtttt
ggatattaga 900tacggtgttt cgagaattgc atatagtaaa gactttgaaa ctctcaaagt
tgattttctt 960agcaagctac ctgaaatgct gaaaatgttc gaagatcgtt tatgtcataa
aacatattta 1020aatggtgatc atgtaaccca tcctgacttc atgttgtatg acgctcttga
tgttgtttta 1080tacatggacc caatgtgcct ggatgcgttc ccaaaattag tttgttttaa
aaaacgtatt 1140gaagctatcc cacaaattga taagtacttg aaatccagca agtatatagc
atggcctttg 1200cagggctggc aagccacgtt tggtggtggc gaccatcctc caaaatcgga
tctggttccg 1260cgtcataatc aaacaagttt gtacaaaaaa gctgaacgag aaacgtaaaa
tgatataaat 1320atcaatatat taaattagat tttgcataaa aaacagacta cataatactg
taaaacacaa 1380catatccagt cactatggcg gccgcattag gcaccccagg ctttacactt
tatgcttccg 1440gctcgtatgt tgtgtggatt ttgagttagg atccggcgag attttcagga
gctaaggaag 1500ctaaaatgga gaaaaaaatc actggatata ccaccgttga tatatcccaa
tggcatcgta 1560aagaacattt tgaggcattt cagtcagttg ctcaatgtac ctataaccag
accgttcagc 1620tggatattac ggccttttta aagaccgtaa agaaaaataa gcacaagttt
tatccggcct 1680ttattcacat tcttgcccgc ctgatgaatg ctcatccgga attccgtatg
gcaatgaaag 1740acggtgagct ggtgatatgg gatagtgttc acccttgtta caccgttttc
catgagcaaa 1800ctgaaacgtt ttcatcgctc tggagtgaat accacgacga tttccggcag
tttctacaca 1860tatattcgca agatgtggcg tgttacggtg aaaacctggc ctatttccct
aaagggttta 1920ttgagaatat gtttttcgtc tcagccaatc cctgggtgag tttcaccagt
tttgatttaa 1980acgtggccaa tatggacaac ttcttcgccc ccgttttcac catgggcaaa
tattatacgc 2040aaggcgacaa ggtgctgatg ccgctggcga ttcaggttca tcatgccgtc
tgtgatggct 2100tccatgtcgg cagaatgctt aatgaattac aacagtactg cgatgagtgg
cagggcgggg 2160cgtaatctag aggatccggc ttactaaaag ccagataaca gtatgcgtat
ttgcgcgctg 2220atttttgcgg tataagaata tatactgata tgtatacccg aagtatgtca
aaaagaggtg 2280tgctatgaag cagcgtatta cagtgacagt tgacagcgac agctatcagt
tgctcaaggc 2340atatatgatg tcaatatctc cggtctggta agcacaacca tgcagaatga
agcccgtcgt 2400ctgcgtgccg aacgctggaa agcggaaaat caggaaggga tggctgaggt
cgcccggttt 2460attgaaatga acggctcttt tgctgacgag aacagggact ggtgaaatgc
agtttaaggt 2520ttacacctat aaaagagaga gccgttatcg tctgtttgtg gatgtacaga
gtgatattat 2580tgacacgccc gggcgacgga tggtgatccc cctggccagt gcacgtctgc
tgtcagataa 2640agtctcccgt gaactttacc cggtggtgca tatcggggat gaaagctggc
gcatgatgac 2700caccgatatg gccagtgtgc cggtctccgt tatcggggaa gaagtggctg
atctcagcca 2760ccgcgaaaat gacatcaaaa acgccattaa cctgatgttc tggggaatat
aaatgtcagg 2820ctcccttata cacagccagt ctgcaggtcg accatagtga ctggatatgt
tgtgttttac 2880agtattatgt agtctgtttt ttatgcaaaa tctaatttaa tatattgata
tttatatcat 2940tttacgtttc tcgttcagct ttcttgtaca aagtggtttg atagcttgtc
gagaagtact 3000agaggatcat aatcagccat accacatttg tagaggtttt acttgcttta
aaaaacctcc 3060cacacctccc cctgaacctg aaacataaaa tgaatgcaat tgttgttgtt
aacttgttta 3120ttgcagctta taatggttac aaataaagca atagcatcac aaatttcaca
aataaagcat 3180ttttttcact gcattctagt tgtggtttgt ccaaactcat caatgtatct
tatcatgtct 3240ggatctgatc actgcttgag cctaggagat ccgaaccaga taagtgaaat
ctagttccaa 3300actattttgt catttttaat tttcgtatta gcttacgacg ctacacccag
ttcccatcta 3360ttttgtcact cttccctaaa taatccttaa aaactccatt tccacccctc
ccagttccca 3420actattttgt ccgcccacag cggggcattt ttcttcctgt tatgttttta
atcaaacatc 3480ctgccaactc catgtgacaa accgtcatct tcggctactt tttctctgtc
acagaatgaa 3540aatttttctg tcatctcttc gttattaatg tttgtaattg actgaatatc
aacgcttatt 3600tgcagcctga atggcgaatg gacgcgccct gtagcggcgc attaagcgcg
gcgggtgtgg 3660tggttacgcg cagcgtgacc gctacacttg ccagcgccct agcgcccgct
cctttcgctt 3720tcttcccttc ctttctcgcc acgttcgccg gctttccccg tcaagctcta
aatcgggggc 3780tccctttagg gttccgattt agtgctttac ggcacctcga ccccaaaaaa
cttgattagg 3840gtgatggttc acgtagtggg ccatcgccct gatagacggt ttttcgccct
ttgacgttgg 3900agtccacgtt ctttaatagt ggactcttgt tccaaactgg aacaacactc
aaccctatct 3960cggtctattc ttttgattta taagggattt tgccgatttc ggcctattgg
ttaaaaaatg 4020agctgattta acaaaaattt aacgcgaatt ttaacaaaat attaacgttt
acaatttcag 4080gtggcacttt tcggggaaat gtgcgcggaa cccctatttg tttatttttc
taaatacatt 4140caaatatgta tccgctcatg agacaataac cctgataaat gcttcaataa
tattgaaaaa 4200ggaagagtat gagtattcaa catttccgtg tcgcccttat tccctttttt
gcggcatttt 4260gccttcctgt ttttgctcac ccagaaacgc tggtgaaagt aaaagatgct
gaagatcagt 4320tgggtgcacg agtgggttac atcgaactgg atctcaacag cggtaagatc
cttgagagtt 4380ttcgccccga agaacgtttt ccaatgatga gcacttttaa agttctgcta
tgtggcgcgg 4440tattatcccg tattgacgcc gggcaagagc aactcggtcg ccgcatacac
tattctcaga 4500atgacttggt tgagtactca ccagtcacag aaaagcatct tacggatggc
atgacagtaa 4560gagaattatg cagtgctgcc ataaccatga gtgataacac tgcggccaac
ttacttctga 4620caacgatcgg aggaccgaag gagctaaccg cttttttgca caacatgggg
gatcatgtaa 4680ctcgccttga tcgttgggaa ccggagctga atgaagccat accaaacgac
gagcgtgaca 4740ccacgatgcc tgtagcaatg gcaacaacgt tgcgcaaact attaactggc
gaactactta 4800ctctagcttc ccggcaacaa ttaatagact ggatggaggc ggataaagtt
gcaggaccac 4860ttctgcgctc ggcccttccg gctggctggt ttattgctga taaatctgga
gccggtgagc 4920gtgggtctcg cggtatcatt gcagcactgg ggccagatgg taagccctcc
cgtatcgtag 4980ttatctacac gacggggagt caggcaacta tggatgaacg aaatagacag
atcgctgaga 5040taggtgcctc actgattaag cattggtaac tgtcagacca agtttactca
tatatacttt 5100agattgattt aaaacttcat ttttaattta aaaggatcta ggtgaagatc
ctttttgata 5160atctcatgac caaaatccct taacgtgagt tttcgttcca ctgagcgtca
gaccccgtag 5220aaaagatcaa aggatcttct tgagatcctt tttttctgcg cgtaatctgc
tgcttgcaaa 5280caaaaaaacc accgctacca gcggtggttt gtttgccgga tcaagagcta
ccaactcttt 5340ttccgaaggt aactggcttc agcagagcgc agataccaaa tactgtcctt
ctagtgtagc 5400cgtagttagg ccaccacttc aagaactctg tagcaccgcc tacatacctc
gctctgctaa 5460tcctgttacc agtggctgct gccagtggcg ataagtcgtg tcttaccggg
ttggactcaa 5520gacgatagtt accggataag gcgcagcggt cgggctgaac ggggggttcg
tgcacacagc 5580ccagcttgga gcgaacgacc tacaccgaac tgagatacct acagcgtgag
cattgagaaa 5640gcgccacgct tcccgaaggg agaaaggcgg acaggtatcc ggtaagcggc
agggtcggaa 5700caggagagcg cacgagggag cttccagggg gaaacgcctg gtatctttat
agtcctgtcg 5760ggtttcgcca cctctgactt gagcgtcgat ttttgtgatg ctcgtcaggg
gggcggagcc 5820tatggaaaaa cgccagcaac gcggcctttt tacggttcct ggccttttgc
tggccttttg 5880ctcacatgtt ctttcctgcg ttatcccctg attctgtgga taaccgtatt
accgcctttg 5940agtgagctga taccgctcgc cgcagccgaa cgaccgagcg cagcgagtca
gtgagcgagg 6000aagcggaaga gcgcctgatg cggtattttc tccttacgca tctgtgcggt
atttcacacc 6060gcagaccagc cgcgtaacct ggcaaaatcg gttacggttg agtaataaat
ggatgccctg 6120cgtaagcggg tgtgggcgga caataaagtc ttaaactgaa caaaatagat
ctaaactatg 6180acaataaagt cttaaactag acagaatagt tgtaaactga aatcagtcca
gttatgctgt 6240gaaaaagcat actggacttt tgttatggct aaagcaaact cttcattttc
tgaagtgcaa 6300attgcccgtc gtattaaaga ggggcgtggc caagggcatg gtaaagacta
tattcgcggc 6360gttgtgacaa tttaccgaac aactccgcgg ccgggaagcc gatctcggct
tgaacgaatt 6420gttaggtggc ggtacttggg tcgatatcaa agtgcatcac ttcttcccgt
atgcccaact 6480ttgtatagag agccactgcg ggatcgtcac cgtaatctgc ttgcacgtag
atcacataag 6540caccaagcgc gttggcctca tgcttgagga gattgatgag cgcggtggca
atgccctgcc 6600tccggtgctc gccggagact gcgagatcat agatatagat ctcactacgc
ggctgctcaa 6660acctgggcag aacgtaagcc gcgagagcgc caacaaccgc ttcttggtcg
aaggcagcaa 6720gcgcgatgaa tgtcttacta cggagcaagt tcccgaggta atcggagtcc
ggctgatgtt 6780gggagtaggt ggctacgtct ccgaactcac gaccgaaaag atcaagagca
gcccgcatgg 6840atttgacttg gtcagggccg agcctacatg tgcgaatgat gcccatactt
gagccaccta 6900actttgtttt agggcgactg ccctgctgcg taacatcgtt gctgctgcgt
aacatcgttg 6960ctgctccata acatcaaaca tcgacccacg gcgtaacgcg cttgctgctt
ggatgcccga 7020ggcatagact gtacaaaaaa acagtcataa caagccatga aaaccg
706614911713DNAArtificial SequenceDescription of Artificial
Sequence Synthetic pDEST21 polynucleotide 149tttattatgt tacaatatgg
aagggaactt tacacttctc ctatgcacat atattaatta 60aagtccaatg ctagtagaga
aggggggtaa cacccctccg cgctcttttc cgattttttt 120ctaaaccgtg gaatatttcg
gatatccttt tgttgtttcc gggtgtacaa tatggacttc 180ctcttttctg gcaaccaaac
ccatacatcg ggattcctat aataccttcg ttggtctccc 240taacatgtag gtggcggagg
ggagatatac aatagaacag ataccagaca agacataatg 300ggctaaacaa gactacacca
attacactgc ctcattgatg gtggtacata acgaactaat 360actgtagccc tagacttgat
agccatcatc atatcgaagt ttcactaccc tttttccatt 420tgccatctat tgaagtaata
ataggcgcat gcaacttctt ttcttttttt ttcttttctc 480tctcccccgt tgttgtctca
ccatatccgc aatgacaaaa aaaatgatgg aagacactaa 540aggaaaaaat taacgacaaa
gacagcacca acagatgtcg ttgttccaga gctgatgagg 600ggtatcttcg aacacacgaa
actttttcct tccttcattc acgcacacta ctctctaatg 660agcaacggta tacggccttc
cttccagtta cttgaatttg aaataaaaaa agtttgccgc 720tttgctatca agtataaata
gacctgcaat tattaatctt ttgtttcctc gtcattgttc 780tcgttccctt tcttccttgt
ttctttttct gcacaatatt tcaagctata ccaagcatac 840aatcaactcc aagcttgaag
caagcctcct gaaagatgaa gctactgtct tctatcgaac 900aagcatgcga tatttgccga
cttaaaaagc tcaagtgctc caaagaaaaa ccgaagtgcg 960ccaagtgtct gaagaacaac
tgggagtgtc gctactctcc caaaaccaaa aggtctccgc 1020tgactagggc acatctgaca
gaagtggaat caaggctaga aagactggaa cagctatttc 1080tactgatttt tcctcgagaa
gaccttgaca tgattttgaa aatggattct ttacaggata 1140taaaagcatt gttaacagga
ttatttgtac aagataatgt gaataaagat gccgtcacag 1200atagattggc ttcagtggag
actgatatgc ctctaacatt gagacagcat agaataagtg 1260cgacatcatc atcggaagag
agtagtaaca aaggtcaaag acagttgact gtatcgtcga 1320ggtcgaatca aacaagtttg
tacaaaaaag ctgaacgaga aacgtaaaat gatataaata 1380tcaatatatt aaattagatt
ttgcataaaa aacagactac ataatactgt aaaacacaac 1440atatccagtc actatggcgg
ccgctaagtt ggcagcatca cccgacgcac tttgcgccga 1500ataaatacct gtgacggaag
atcacttcgc agaataaata aatcctggtg tccctgttga 1560taccgggaag ccctgggcca
acttttggcg aaaatgagac gttgatcggc acgtaagagg 1620ttccaacttt caccataatg
aaataagatc actaccgggc gtattttttg agttatcgag 1680attttcagga gctaaggaag
ctaaaatgga gaaaaaaatc actggatata ccaccgttga 1740tatatcccaa tggcatcgta
aagaacattt tgaggcattt cagtcagttg ctcaatgtac 1800ctataaccag accgttcagc
tggatattac ggccttttta aagaccgtaa agaaaaataa 1860gcacaagttt tatccggcct
ttattcacat tcttgcccgc ctgatgaatg ctcatccgga 1920attccgtatg gcaatgaaag
acggtgagct ggtgatatgg gatagtgttc acccttgtta 1980caccgttttc catgagcaaa
ctgaaacgtt ttcatcgctc tggagtgaat accacgacga 2040tttccggcag tttctacaca
tatattcgca agatgtggcg tgttacggtg aaaacctggc 2100ctatttccct aaagggttta
ttgagaatat gtttttcgtc tcagccaatc cctgggtgag 2160tttcaccagt tttgatttaa
acgtggccaa tatggacaac ttcttcgccc ccgttttcac 2220catgggcaaa tattatacgc
aaggcgacaa ggtgctgatg ccgctggcga ttcaggttca 2280tcatgccgtc tgtgatggct
tccatgtcgg cagaatgctt aatgaattac aacagtactg 2340cgatgagtgg cagggcgggg
cgtaatctag aggatccggc ttactaaaag ccagataaca 2400gtatgcgtat ttgcgcgctg
atttttgcgg tataagaata tatactgata tgtatacccg 2460aagtatgtca aaaagaggtg
tgctatgaag cagcgtatta cagtgacagt tgacagcgac 2520agctatcagt tgctcaaggc
atatatgatg tcaatatctc cggtctggta agcacaacca 2580tgcagaatga agcccgtcgt
ctgcgtgccg aacgctggaa agcggaaaat caggaaggga 2640tggctgaggt cgcccggttt
attgaaatga acggctcttt tgctgacgag aacagggact 2700ggtgaaatgc agtttaaggt
ttacacctat aaaagagaga gccgttatcg tctgtttgtg 2760gatgtacaga gtgatattat
tgacacgccc gggcgacgga tggtgatccc cctggccagt 2820gcacgtctgc tgtcagataa
agtctcccgt gaactttacc cggtggtgca tatcggggat 2880gaaagctggc gcatgatgac
caccgatatg gccagtgtgc cggtctccgt tatcggggaa 2940gaagtggctg atctcagcca
ccgcgaaaat gacatcaaaa acgccattaa cctgatgttc 3000tggggaatat aaatgtcagg
ctcccttata cacagccagt ctgcaggtcg accatagtga 3060ctggatatgt tgtgttttac
agtattatgt agtctgtttt ttatgcaaaa tctaatttaa 3120tatattgata tttatatcat
tttacgtttc tcgttcagct ttcttgtaca aagtggtttg 3180atggccgcta agtaagtaag
acgtcgagct ctaagtaagt aacggccgcc accgcggtgg 3240agctttggac ttcttcgcca
gaggtttggt caagtctcca atcaaggttg tcggcttgtc 3300taccttgcca gaaatttacg
aaaagatgga aaagggtcaa atcgttggta gatacgttgt 3360tgacacttct aaataagcga
atttcttatg atttatgatt tttattatta aataagttat 3420aaaaaaaata agtgtataca
aattttaaag tgactcttag gttttaaaac gaaaattctt 3480attcttgagt aactctttcc
tgtaggtcag gttgctttct caggtatagc atgaggtcgc 3540tcttattgac cacacctcta
ccggcatgcc gagcaaatgc ctgcaaatcg ctccccattt 3600cacccaattg tagatatgct
aactccagca atgagttgat gaatctcggt gtgtatttta 3660tgtcctcaga ggacaatacc
tgttgtaatc gttcttccac acggatccca attcgcccta 3720tagtgagtcg tattacaatt
cactggccgt cgttttacaa cgtcgtgact gggaaaaccc 3780tggcgttacc caacttaatc
gccttgcagc acatccccct ttcgccagct ggcgtaatag 3840cgaagaggcc cgcaccgatc
gcccttccca acagttgcgc agcctgaatg gcgaatggac 3900gcgccctgta gcggcgcatt
aagcgcggcg ggtgtggtgg ttacgcgcag cgtgaccgct 3960acacttgcca gcgccctagc
gcccgctcct ttcgctttct tcccttcctt tctcgccacg 4020ttcgccggct ttccccgtca
agctctaaat cgggggctcc ctttagggtt ccgatttagt 4080gctttacggc acctcgaccc
caaaaaactt gattagggtg atggttcacg tagtgggcca 4140tcgccctgat agacggtttt
tcgccctttg acgttggagt ccacgttctt taatagtgga 4200ctcttgttcc aaactggaac
aacactcaac cctatctcgg tctattcttt tgatttataa 4260gggattttgc cgatttcggc
ctattggtta aaaaatgagc tgatttaaca aaaatttaac 4320gcgaatttta acaaaatatt
aacgtttaca atttcctgat gcggtatttt ctccttacgc 4380atctgtgcgg tatttcacac
cgcatatcga ccggtcgagg agaacttcta gtatatccac 4440atacctaata ttattgcctt
attaaaaatg gaatcggaac aattacatca aaatccacat 4500tctcttcaaa atcaattgtc
ctgtacttcc ttgttcatgt gtgttcaaaa acgttatatt 4560tataggataa ttatactcta
tttctcaaca agtaattggt tgtttggccg agcggtctaa 4620ggcgcctgat tcaagaaata
tcttgaccgc agttaactgt gggaatactc aggtatcgta 4680agatgcaaga gttcgaatct
cttagcaacc attatttttt tcctcaacat aacgagaaca 4740cacaggggcg ctatcgcaca
gaatcaaatt cgatgactgg aaattttttg ttaatttcag 4800aggtcgcctg acgcatatac
ctttttcaac tgaaaaattg ggagaaaaag gaaaggtgag 4860aggccggaac cggcttttca
tatagaatag agaagcgttc atgactaaat gcttgcatca 4920caatacttga agttgacaat
attatttaag gacctattgt tttttccaat aggtggttag 4980caatcgtctt actttctaac
ttttcttacc ttttacattt cagcaatata tatatatatt 5040tcaaggatat accattctaa
tgtctgcccc tatgtctgcc cctaagaaga tcgtcgtttt 5100gccaggtgac cacgttggtc
aagaaatcac agccgaagcc attaaggttc ttaaagctat 5160ttctgatgtt cgttccaatg
tcaagttcga tttcgaaaat catttaattg gtggtgctgc 5220tatcgatgct acaggtgtcc
cacttccaga tgaggcgctg gaagcctcca agaaggttga 5280tgccgttttg ttaggtgctg
tgggtggtcc taaatggggt accggtagtg ttagacctga 5340acaaggttta ctaaaaatcc
gtaaagaact tcaattgtac gccaacttaa gaccatgtaa 5400ctttgcatcc gactctcttt
tagacttatc tccaatcaag ccacaatttg ctaaaggtac 5460tgacttcgtt gttgtcagag
aattagtggg aggtatttac tttggtaaga gaaaggaaga 5520cgatggtgat ggtgtcgctt
gggatagtga acaatacacc gttccagaag tgcaaagaat 5580cacaagaatg gccgctttca
tggccctaca acatgagcca ccattgccta tttggtcctt 5640ggataaagct aatgttttgg
cctcttcaag attatggaga aaaactgtgg aggaaaccat 5700caagaacgaa ttccctacat
tgaaggttca acatcaattg attgattctg ccgccatgat 5760cctagttaag aacccaaccc
acctaaatgg tattataatc accagcaaca tgtttggtga 5820tatcatctcc gatgaagcct
ccgttatccc aggttccttg ggtttgttgc catctgcgtc 5880cttggcctct ttgccagaca
agaacaccgc atttggtttg tacgaaccat gccacggttc 5940tgctccagat ttgccaaaga
ataaggttga ccctatcgcc actatcttgt ctgctgcaat 6000gatgttgaaa ttgtcattga
acttgcctga agaaggtaag gccattgaag atgcagttaa 6060aaaggttttg gatgcaggta
tcagaactgg tgatttaggt ggttccaaca gtaccaccga 6120agtcggtgat gctgtcgccg
aagaagttaa gaaaatcctt gcttaaaaag attctctttt 6180tttatgatat ttgtacataa
actttataaa tgaaattcat aatagaaacg acacgaaatt 6240acaaaatgga atatgttcat
agggtagacg aaactatata cgcaatctac atacatttat 6300caagaaggag aaaaaggagg
atagtaaagg aatacaggta agcaaattga tactaatggc 6360tcaacgtgat aaggaaaaag
aattgcactt taacattaat attgacaagg aggagggcac 6420cacacaaaaa gttaggtgta
acagaaaatc atgaaactac gattcctaat ttgatattgg 6480aggattttct ctaaaaaaaa
aaaaatacaa caaataaaaa acactcaatg acctgaccat 6540ttgatggagt ttaagtcaat
accttcttga accatttccc ataatggtga aagttccctc 6600aagaatttta ctctgtcaga
aacggcctta cgacgtagtc gatatggtgc actctcagta 6660caatctgctc tgatgccgca
tagttaagcc agccccgaca cccgccaaca cccgctgacg 6720cgccctgacg ggcttgtctg
ctcccggcat ccgcttacag acaagctgtg accgtctccg 6780ggagctgcat gtgtcagagg
ttttcaccgt catcaccgaa acgcgcgaga cgaaagggcc 6840tcgtgatacg cctattttta
taggttaatg tcatgataat aatggtttct taggacggat 6900cgcttgcctg taacttacac
gcgcctcgta tcttttaatg atggaataat ttgggaattt 6960actctgtgtt tatttatttt
tatgttttgt atttggattt tagaaagtaa ataaagaagg 7020tagaagagtt acggaatgaa
gaaaaaaaaa taaacaaagg tttaaaaaat ttcaacaaaa 7080agcgtacttt acatatatat
ttattagaca agaaaagcag attaaataga tatacattcg 7140attaacgata agtaaaatgt
aaaatcacag gattttcgtg tgtggtcttc tacacagaca 7200agatgaaaca attcggcatt
aatacctgag agcaggaaga gcaagataaa aggtagtatt 7260tgttggcgat ccccctagag
tcttttacat cttcggaaaa caaaaactat tttttcttta 7320atttcttttt ttactttcta
tttttaattt atatatttat attaaaaaat ttaaattata 7380attattttta tagcacgtga
tgaaaaggac ccaggtggca cttttcgggg aaatgtgcgc 7440ggaaccccta tttgtttatt
tttctaaata cattcaaata tgtatccgct catgagacaa 7500taaccctgat aaatgcttca
ataatctgca gctctggccc gtgtctcaaa atctctgatg 7560ttacattgca caagataaaa
atatatcatc atgaacaata aaactgtctg cttacataaa 7620cagtaataca aggggtgtta
tgagccatat tcaacgggaa acgtcttgct ggaggccgcg 7680attaaattcc aacatggatg
ctgatttata tgggtataaa tgggctcgcg ataatgtcgg 7740gcaatcaggt gcgacaatct
ttcgattgta tgggaagccc gatgcgccag agttgtttct 7800gaaacatggc aaaggtagcg
ttgccaatga tgttacagat gagatggtca gactaaactg 7860gctgacggaa tttatgcctc
ttccgaccat caagcatttt atccgtactc ctgatgatgc 7920atggttactc accactgcga
tccgcgggaa aacagcattc caggtattag aagaatatcc 7980tgattcaggt gaaaatattg
ttgatgcgct ggcagtgttc ctgcgccggt tgcattcgat 8040tcctgtttgt aattgtcctt
ttaacagcga tcgcgtattt cgtctcgctc aggcgcaatc 8100acgaatgaat aacggtttgg
ttgatgcgag tgattttgat gacgagcgta atggctggcc 8160tgttgaacaa gtctggaaag
aaatgcatac gcttttgcca ttctcaccgg attcagtcgt 8220cactcatggt gatttctcac
ttgataacct tatttttgac gaggggaaat taataggttg 8280tattgatgtt ggacgagtcg
gaatcgcaga ccgataccag gatcttgcca tcctatggaa 8340ctgcctcggt gagttttctc
cttcattaca gaaacggctt tttcaaaaat atggtattga 8400taatcctgat atgaataaat
tgcagtttca tttgatgctc gatgagtttt tctaatcaga 8460attggttaat tggttgtaac
actggcagag cattacgctg acttgacggg acggcgcatg 8520accaaaatcc cttaacgtga
gttttcgttc cactgagcgt cagaccccgt agaaaagatc 8580aaaggatctt cttgagatcc
tttttttctg cgcgtaatct gctgcttgca aacaaaaaaa 8640ccaccgctac cagcggtggt
ttgtttgccg gatcaagagc taccaactct ttttccgaag 8700gtaactggct tcagcagagc
gcagatacca aatactgtcc ttctagtgta gccgtagtta 8760ggccaccact tcaagaactc
tgtagcaccg cctacatacc tcgctctgct aatcctgtta 8820ccagtggctg ctgccagtgg
cgataagtcg tgtcttaccg ggttggactc aagacgatag 8880ttaccggata aggcgcagcg
gtcgggctga acggggggtt cgtgcacaca gcccagcttg 8940gagcgaacga cctacaccga
actgagatac ctacagcgtg agcattgaga aagcgccacg 9000cttcccgaag ggagaaaggc
ggacaggtat ccggtaagcg gcagggtcgg aacaggagag 9060cgcacgaggg agcttccagg
ggggaacgcc tggtatcttt atagtcctgt cgggtttcgc 9120cacctctgac ttgagcgtcg
atttttgtga tgctcgtcag gggggccgag cctatggaaa 9180aacgccagca acgcggcctt
tttacggttc ctggcctttt gctggccttt tgctcacatg 9240ttctttcctg cgttatcccc
tgattctgtg gataaccgta ttaccgcctt tgagtgagct 9300gataccgctc gccgcagccg
aacgaccgag cgcagcgagt cagtgagcga ggaagcggaa 9360gagcgcccaa tacgcaaacc
gcctctcccc gcgcgttggc cgattcatta atgcagctgg 9420cacgacaggt ttcccgactg
gaaagcgggc agtgagcgca acgcaattaa tgtgagttac 9480ctcactcatt aggcacccca
ggctttacac tttatgcttc cggctcctat gttgtgtgga 9540attgtgagcg gataacaatt
tcacacagga aacagctatg accatgatta cgccaagctc 9600ggaattaacc ctcactaaag
ggaacaaaag ctggtaccga tcccgagctt tgcaaattaa 9660agccttcgag cgtcccaaaa
ccttctcaag caaggttttc agtataatgt tacatgcgta 9720cacgcgtctg tacagaaaaa
aaagaaaaat ttgaaatata aataacgttc ttaatactaa 9780cataactata aaaaaataaa
tagggaccta gacttcaggt tgtctaactc cttccttttc 9840ggttagagcg gatgtggggg
gagggcgtga atgtaagcgt gacataacta attacatgat 9900atcgacaaag gaaaaggggc
ctgtttactc acaggctttt ttcaagtagg taattaagtc 9960gtttctgtct ttttccttct
tcaacccacc aaaggccatc ttggtacttt tttttttttt 10020tttttttttt tttttttttt
tttttttttt tttttttttt tttttttttt tttttttttt 10080tttttttttt tttttttttt
tcatagaaat aatacagaag tagatgttga attagattaa 10140actgaagata tataatttat
tggaaaatac atagagcttt ttgttgatgc gcttaagcga 10200tcaattcaac aacaccacca
gcagctctga ttttttcttc agccaacttg gagacgaatc 10260tagctttgac gataactgga
acatttggaa ttctaccctt acccaagatc ttaccgtaac 10320cggctgccaa agtgtcaata
actggagcag tttccttaga agcagatttc aagtattggt 10380ctctcttgtc ttctgggatc
aatgtccaca atttgtccaa gttcaagact ggcttccaga 10440aatgagcttg ttgcttgtgg
aagtatctca taccaacctt accgaaataa cctggatggt 10500atttatccat gttaattctg
tggtgatgtt gaccaccggc catacctcta ccaccggggt 10560gctttctgtg cttaccgata
cgacctttac cggctgagac gtgacctctg tgctttctag 10620tcttagtgaa tctggaaggc
attcttgatt agttggatga ttgttctggg atttaatgca 10680aaaatcactt aagaaggaaa
atcaacggag aaagcaaacg ccatcttaaa tatacgggat 10740acagatgaaa gggtttgaac
ctatctggaa aatagcatta aacaagcgaa aaactgcgag 10800gaaaattgtt tgcgtctctg
cgggctattc acgcgccaga ggaaaatagg aaaaataaca 10860gggcattaga aaaataattt
tgattttggt aatgtgtggg tcctggtgta cagatgttac 10920attggttaca gtactcttgt
ttttgctgtg tttttcgatg aatctccaaa atggttgtta 10980gcacatggaa gagtcaccga
tgctaagtta tctctatgta agctacgtgg cgtgactttt 11040gatgaagccg cacaagagat
acaggattgg caactgcaaa tagaatctgg ggatcccccc 11100tcgagatccg ggatcgaaga
aatgatggta aatgaaatag gaaatcaagg agcatgaagg 11160caaaagacaa atataagggt
cgaacgaaaa ataaagtgaa aagtgttgat atgatgtatt 11220tggctttgcg gcgccgaaaa
aacgagttta cgcaattgca caatcatgct gactctgtgg 11280cggacccgcg ctcttgccgg
cccggcgata acgctgggcg tgaggctgtg cccggcggag 11340ttttttgcgc ctgcattttc
caaggtttac cctgcgctaa ggggcgagat tggagaagca 11400ataagaatgc cggttggggt
tgcgatgatg acgaccacga caactggtgt cattatttaa 11460gttgccgaaa gaacctgagt
gcatttgcaa catgagtata ctagaagaat gagccaagac 11520ttgcgagacg cgagtttgcc
ggtggtgcga acaatagagc gaccatgacc ttgaaggtga 11580gacgcgcata accgctagag
tactttgaag aggaaacagc aatagggttg ctaccagtat 11640aaatagacag gtacatacaa
cactggaaat ggttgtctgt ttgagtacgc tttcaattca 11700tttgggtgtg cac
117131508923DNAArtificial
SequenceDescription of Artificial Sequence Synthetic pDEST22
polynucleotide 150ttcatttggg tgtgcacttt attatgttac aatatggaag ggaactttac
acttctccta 60tgcacatata ttaattaaag tccaatgcta gtagagaagg ggggtaacac
ccctccgcgc 120tcttttccga tttttttcta aaccgtggaa tatttcggat atccttttgt
tgtttccggg 180tgtacaatat ggacttcctc ttttctggca accaaaccca tacatcggga
ttcctataat 240accttcgttg gtctccctaa catgtaggtg gcggagggga gatatacaat
agaacagata 300ccagacaaga cataatgggc taaacaagac tacaccaatt acactgcctc
attgatggtg 360gtacataacg aactaatact gtagccctag acttgatagc catcatcata
tcgaagtttc 420actacccttt ttccatttgc catctattga agtaataata ggcgcatgca
acttcttttc 480tttttttttc ttttctctct cccccgttgt tgtctcacca tatccgcaat
gacaaaaaaa 540atgatggaag acactaaagg aaaaaattaa cgacaaagac agcaccaaca
gatgtcgttg 600ttccagagct gatgaggggt atcttcgaac acacgaaact ttttccttcc
ttcattcacg 660cacactactc tctaatgagc aacggtatac ggccttcctt ccagttactt
gaatttgaaa 720taaaaaaagt ttgccgcttt gctatcaagt ataaatagac ctgcaattat
taatcttttg 780tttcctcgtc attgttctcg ttccctttct tccttgtttc tttttctgca
caatatttca 840agctatacca agcatacaat caactccaag cttatgccca agaagaagcg
gaaggtctcg 900agcggcgcca attttaatca aagtgggaat attgctgata gctcattgtc
cttcactttc 960actaacagta gcaacggtcc gaacctcata acaactcaaa caaattctca
agcgctttca 1020caaccaattg cctcctctaa cgttcatgat aacttcatga ataatgaaat
cacggctagt 1080aaaattgatg atggtaataa ttcaaaacca ctgtcacctg gttggacgga
ccaaactgcg 1140tataacgcgt ttggaatcac tacagggatg tttaatacca ctacaatgga
tgatgtatat 1200aactatctat tcgatgatga agatacccca ccaaacccaa aaaaagaggg
tgggtcgaat 1260caaacaagtt tgtacaaaaa agctgaacga gaaacgtaaa atgatataaa
tatcaatata 1320ttaaattaga ttttgcataa aaaacagact acataatact gtaaaacaca
acatatccag 1380tcactatggc ggccgctaag ttggcagcat cacccgacgc actttgcgcc
gaataaatac 1440ctgtgacgga agatcacttc gcagaataaa taaatcctgg tgtccctgtt
gataccggga 1500agccctgggc caacttttgg cgaaaatgag acgttgatcg gcacgtaaga
ggttccaact 1560ttcaccataa tgaaataaga tcactaccgg gcgtattttt tgagttatcg
agattttcag 1620gagctaagga agctaaaatg gagaaaaaaa tcactggata taccaccgtt
gatatatccc 1680aatggcatcg taaagaacat tttgaggcat ttcagtcagt tgctcaatgt
acctataacc 1740agaccgttca gctggatatt acggcctttt taaagaccgt aaagaaaaat
aagcacaagt 1800tttatccggc ctttattcac attcttgccc gcctgatgaa tgctcatccg
gaattccgta 1860tggcaatgaa agacggtgag ctggtgatat gggatagtgt tcacccttgt
tacaccgttt 1920tccatgagca aactgaaacg ttttcatcgc tctggagtga ataccacgac
gatttccggc 1980agtttctaca catatattcg caagatgtgg cgtgttacgg tgaaaacctg
gcctatttcc 2040ctaaagggtt tattgagaat atgtttttcg tctcagccaa tccctgggtg
agtttcacca 2100gttttgattt aaacgtggcc aatatggaca acttcttcgc ccccgttttc
accatgggca 2160aatattatac gcaaggcgac aaggtgctga tgccgctggc gattcaggtt
catcatgccg 2220tctgtgatgg cttccatgtc ggcagaatgc ttaatgaatt acaacagtac
tgcgatgagt 2280ggcagggcgg ggcgtaatct agaggatccg gcttactaaa agccagataa
cagtatgcgt 2340atttgcgcgc tgatttttgc ggtataagaa tatatactga tatgtatacc
cgaagtatgt 2400caaaaagagg tgtgctatga agcagcgtat tacagtgaca gttgacagcg
acagctatca 2460gttgctcaag gcatatatga tgtcaatatc tccggtctgg taagcacaac
catgcagaat 2520gaagcccgtc gtctgcgtgc cgaacgctgg aaagcggaaa atcaggaagg
gatggctgag 2580gtcgcccggt ttattgaaat gaacggctct tttgctgacg agaacaggga
ctggtgaaat 2640gcagtttaag gtttacacct ataaaagaga gagccgttat cgtctgtttg
tggatgtaca 2700gagtgatatt attgacacgc ccgggcgacg gatggtgatc cccctggcca
gtgcacgtct 2760gctgtcagat aaagtctccc gtgaacttta cccggtggtg catatcgggg
atgaaagctg 2820gcgcatgatg accaccgata tggccagtgt gccggtctcc gttatcgggg
aagaagtggc 2880tgatctcagc caccgcgaaa atgacatcaa aaacgccatt aacctgatgt
tctggggaat 2940ataaatgtca ggctccctta tacacagcca gtctgcaggt cgaccatagt
gactggatat 3000gttgtgtttt acagtattat gtagtctgtt ttttatgcaa aatctaattt
aatatattga 3060tatttatatc attttacgtt tctcgttcag ctttcttgta caaagtggtt
tgatggccgc 3120taagtaagta agacgtcgag ctctaagtaa gtaacggccg ccaccgcggt
ggagctttgg 3180acttcttcgc cagaggtttg gtcaagtctc caatcaaggt tgtcggcttg
tctaccttgc 3240cagaaattta cgaaaagatg gaaaagggtc aaatcgttgg tagatacgtt
gttgacactt 3300ctaaataagc gaatttctta tgatttatga tttttattat taaataagtt
ataaaaaaaa 3360taagtgtata caaattttaa agtgactctt aggttttaaa acgaaaattc
ttattcttga 3420gtaactcttt cctgtaggtc aggttgcttt ctcaggtata gcatgaggtc
gctcttattg 3480accacacctc taccggcatg ccgagcaaat gcctgcaaat cgctccccat
ttcacccaat 3540tgtagatatg ctaactccag caatgagttg atgaatctcg gtgtgtattt
tatgtcctca 3600gaggacaata cctgttgtaa tcgttcttcc acacggatcc caattcgccc
tatagtgagt 3660cgtattacaa ttcactggcc gtcgttttac aacgtcgtga ctgggaaaac
cctggcgtta 3720cccaacttaa tcgccttgca gcacatcccc ctttcgccag ctggcgtaat
agcgaagagg 3780cccgcaccga tcgcccttcc caacagttgc gcagcctgaa tggcgaatgg
acgcgccctg 3840tagcggcgca ttaagcgcgg cgggtgtggt ggttacgcgc agcgtgaccg
ctacacttgc 3900cagcgcccta gcgcccgctc ctttcgcttt cttcccttcc tttctcgcca
cgttcgccgg 3960ctttccccgt caagctctaa atcgggggct ccctttaggg ttccgattta
gtgctttacg 4020gcacctcgac cccaaaaaac ttgattaggg tgatggttca cgtagtgggc
catcgccctg 4080atagacggtt tttcgccctt tgacgttgga gtccacgttc tttaatagtg
gactcttgtt 4140ccaaactgga acaacactca accctatctc ggtctattct tttgatttat
aagggatttt 4200gccgatttcg gcctattggt taaaaaatga gctgatttaa caaaaattta
acgcgaattt 4260taacaaaata ttaacgttta caatttcctg atgcggtatt ttctccttac
gcatctgtgc 4320ggtatttcac accgcaggca agtgcacaaa caatacttaa ataaatacta
ctcagtaata 4380acctatttct tagcattttt gacgaaattt gctattttgt tagagtcttt
tacaccattt 4440gtctccacac ctccgcttac atcaacacca ataacgccat ttaatctaag
cgcatcacca 4500acattttctg gcgtcagtcc accagctaac ataaaatgta agctttcggg
gctctcttgc 4560cttccaaccc agtcagaaat cgagttccaa tccaaaagtt cacctgtccc
acctgcttct 4620gaatcaaaca agggaataaa cgaatgaggt ttctgtgaag ctgcactgag
tagtatgttg 4680cagtcttttg gaaatacgag tcttttaata actggcaaac cgaggaactc
ttggtattct 4740tgccacgact catctccatg cagttggacg atatcaatgc cgtaatcatt
gaccagagcc 4800aaaacatcct ccttaggttg attacgaaac acgccaacca agtatttcgg
agtgcctgaa 4860ctatttttat atgcttttac aagacttgaa attttccttg caataaccgg
gtcaattgtt 4920ctctttctat tgggcacaca tataataccc agcaagtcag catcggaatc
tagagcacat 4980tctgcggcct ctgtgctctg caagccgcaa actttcacca atggaccaga
actacctgtg 5040aaattaataa cagacatact ccaagctgcc tttgtgtgct taatcacgta
tactcacgtg 5100ctcaatagtc accaatgccc tccctcttgg ccctctcctt ttcttttttc
gaccgaatta 5160attcttaatc ggcaaaaaaa gaaaagctcc ggatcaagat tgtacgtaag
gtgacaagct 5220atttttcaat aaagaatatc ttccactact gccatctggc gtcataactg
caaagtacac 5280atatattacg atgctgtcta ttaaatgctt cctatattat atatatagta
atgtcgttta 5340tggtgcactc tcagtacaat ctgctctgat gccgcatagt taagccagcc
ccgacacccg 5400ccaacacccg ctgacgcgcc ctgacgggct tgtctgctcc cggcatccgc
ttacagacaa 5460gctgtgaccg tctccgggag ctgcatgtgt cagaggtttt caccgtcatc
accgaaacgc 5520gcgagacgaa agggcctcgt gatacgccta tttttatagg ttaatgtcat
gataataatg 5580gtttcttagg acggatcgct tgcctgtaac ttacacgcgc ctcgtatctt
ttaatgatgg 5640aataatttgg gaatttactc tgtgtttatt tatttttatg ttttgtattt
ggattttaga 5700aagtaaataa agaaggtaga agagttacgg aatgaagaaa aaaaaataaa
caaaggttta 5760aaaaatttca acaaaaagcg tactttacat atatatttat tagacaagaa
aagcagatta 5820aatagatata cattcgatta acgataagta aaatgtaaaa tcacaggatt
ttcgtgtgtg 5880gtcttctaca cagacaagat gaaacaattc ggcattaata cctgagagca
ggaagagcaa 5940gataaaaggt agtatttgtt ggcgatcccc ctagagtctt ttacatcttc
ggaaaacaaa 6000aactattttt tctttaattt ctttttttac tttctatttt taatttatat
atttatatta 6060aaaaatttaa attataatta tttttatagc acgtgatgaa aaggacccag
gtggcacttt 6120tcggggaaat gtgcgcggaa cccctatttg tttatttttc taaatacatt
caaatatgta 6180tccgctcatg agacaataac cctgataaat gcttcaataa tattgaaaaa
ggaagagtat 6240gagtattcaa catttccgtg tcgcccttat tccctttttt gcggcatttt
gccttcctgt 6300ttttgctcac ccagaaacgc tggtgaaagt aaaagatgct gaagatcagt
tgggtgcacg 6360agtgggttac atcgaactgg atctcaacag cggtaagatc cttgagagtt
ttcgccccga 6420agaacgtttt ccaatgatga gcacttttaa agttctgcta tgtggcgcgg
tattatcccg 6480tattgacgcc gggcaagagc aactcggtcg ccgcatacac tattctcaga
atgacttggt 6540tgagtactca ccagtcacag aaaagcatct tacggatggc atgacagtaa
gagaattatg 6600cagtgctgcc ataaccatga gtgataacac tgcggccaac ttacttctga
caacgatcgg 6660aggaccgaag gagctaaccg ctttttttca caacatgggg gatcatgtaa
ctcgccttga 6720tcgttgggaa ccggagctga atgaagccat accaaacgac gagcgtgaca
ccacgatgcc 6780tgtagcaatg gcaacaacgt tgcgcaaact attaactggc gaactactta
ctctagcttc 6840ccggcaacaa ttaatagact ggatggaggc ggataaagtt gcaggaccac
ttctgcgctc 6900ggcccttccg gctggctggt ttattgctga taaatctgga gccggtgagc
gtgggtctcg 6960cggtatcatt gcagcactgg ggccagatgg taagccctcc cgtatcgtag
ttatctacac 7020gacgggcagt caggcaacta tggatgaacg aaatagacag atcgctgaga
taggtgcctc 7080actgattaag cattggtaac tgtcagacca agtttactca tatatacttt
agattgattt 7140aaaacttcat ttttaattta aaaggatcta ggtgaagatc ctttttgata
atctcatgac 7200caaaatccct taacgtgagt tttcgttcca ctgagcgtca gaccccgtag
aaaagatcaa 7260aggatcttct tgagatcctt tttttctgcg cgtaatctgc tgcttgcaaa
caaaaaaacc 7320accgctacca gcggtggttt gtttgccgga tcaagagcta ccaactcttt
ttccgaaggt 7380aactggcttc agcagagcgc agataccaaa tactgtcctt ctagtgtagc
cgtagttagg 7440ccaccacttc aagaactctg tagcaccgcc tacatacctc gctctgctaa
tcctgttacc 7500agtggctgct gccagtggcg ataagtcgtg tcttaccggg ttggactcaa
gacgatagtt 7560accggataag gcgcagcggt cgggctgaac ggggggttcg tgcacacagc
ccagcttgga 7620gcgaacgacc tacaccgaac tgagatacct acagcgtgag cattgagaaa
gcgccacgct 7680tcccgaaggg agaaaggcgg acaggtatcc ggtaagcggc agggtcggaa
caggagagcg 7740cacgagggag cttccagggg ggaacgcctg gtatctttat agtcctgtcg
ggtttcgcca 7800cctctgactt gagcgtcgat ttttgtgatg ctcgtcaggg gggccgagcc
tatggaaaaa 7860cgccagcaac gcggcctttt tacggttcct ggccttttgc tggccttttg
ctcacatgtt 7920ctttcctgcg ttatcccctg attctgtgga taaccgtatt accgcctttg
agtgagctga 7980taccgctcgc cgcagccgaa cgaccgagcg cagcgagtca gtgagcgagg
aagcggaaga 8040gcgcccaata cgcaaaccgc ctctccccgc gcgttggccg attcattaat
gcagctggca 8100cgacaggttt cccgactgga aagcgggcag tgagcgcaac gcaattaatg
tgagttacct 8160cactcattag gcaccccagg ctttacactt tatgcttccg gctcctatgt
tgtgtggaat 8220tgtgagcgga taacaatttc acacaggaaa cagctatgac catgattacg
ccaagctcgg 8280aattaaccct cactaaaggg aacaaaagct gggtaccggg ccccccctcg
agatccggga 8340tcgaagaaat gatggtaaat gaaataggaa atcaaggagc atgaaggcaa
aagacaaata 8400taagggtcga acgaaaaata aagtgaaaag tgttgatatg atgtatttgg
ctttgcggcg 8460ccgaaaaaac gagtttacgc aattgcacaa tcatgctgac tctgtggcgg
acccgcgctc 8520ttgccggccc ggcgataacg ctgggcgtga ggctgtgccc ggcggagttt
tttgcgcctg 8580cattttccaa ggtttaccct gcgctaaggg gcgagattgg agaagcaata
agaatgccgg 8640ttggggttgc gatgatgacg accacgacaa ctggtgtcat tatttaagtt
gccgaaagaa 8700cctgagtgca tttgcaacat gagtatacta gaagaatgag ccaagacttg
cgagacgcga 8760gtttgccggt ggtgcgaaca atagagcgac catgaccttg aaggtgagac
gcgcataacc 8820gctagagtac tttgaagagg aaacagcaat agggttgcta ccagtataaa
tagacaggta 8880catacaacac tggaaatggt tgtctgtttg agtacgcttt caa
89231516264DNAArtificial SequenceDescription of Artificial
Sequence Synthetic pDEST23 polynucleotide 151tcttccccat cggtgatgtc
ggcgatatag gcgccagcaa ccgcacctgt ggcgccggtg 60atgccggcca cgatgcgtcc
ggcgtagagg atcgagatct cgatcccgcg aaattaatac 120gactcactat agggagacca
caacggtttc cctctagatc acaagtttgt acaaaaaagc 180tgaacgagaa acgtaaaatg
atataaatat caatatatta aattagattt tgcataaaaa 240acagactaca taatactgta
aaacacaaca tatccagtca ctatggcggc cgcattaggc 300accccaggct ttacacttta
tgcttccggc tcgtataatg tgtggatttt gagttaggat 360ccggcgagat tttcaggagc
taaggaagct aaaatggaga aaaaaatcac tggatatacc 420accgttgata tatcccaatg
gcatcgtaaa gaacattttg aggcatttca gtcagttgct 480caatgtacct ataaccagac
cgttcagctg gatattacgg cctttttaaa gaccgtaaag 540aaaaataagc acaagtttta
tccggccttt attcacattc ttgcccgcct gatgaatgct 600catccggaat tccgtatggc
aatgaaagac ggtgagctgg tgatatggga tagtgttcac 660ccttgttaca ccgttttcca
tgagcaaact gaaacgtttt catcgctctg gagtgaatac 720cacgacgatt tccggcagtt
tctacacata tattcgcaag atgtggcgtg ttacggtgaa 780aacctggcct atttccctaa
agggtttatt gagaatatgt ttttcgtctc agccaatccc 840tgggtgagtt tcaccagttt
tgatttaaac gtggccaata tggacaactt cttcgccccc 900gttttcacca tgggcaaata
ttatacgcaa ggcgacaagg tgctgatgcc gctggcgatt 960caggttcatc atgccgtctg
tgatggcttc catgtcggca gaatgcttaa tgaattacaa 1020cagtactgcg atgagtggca
gggcggggcg taaacgcgtg gatccggctt actaaaagcc 1080agataacagt atgcgtattt
gcgcgctgat ttttgcggta taagaatata tactgatatg 1140tatacccgaa gtatgtcaaa
aagaggtgtg ctatgaagca gcgtattaca gtgacagttg 1200acagcgacag ctatcagttg
ctcaaggcat atatgatgtc aatatctccg gtctggtaag 1260cacaaccatg cagaatgaag
cccgtcgtct gcgtgccgaa cgctggaaag cggaaaatca 1320ggaagggatg gctgaggtcg
cccggtttat tgaaatgaac ggctcttttg ctgacgagaa 1380cagggactgg tgaaatgcag
tttaaggttt acacctataa aagagagagc cgttatcgtc 1440tgtttgtgga tgtacagagt
gatattattg acacgcccgg gcgacggatg gtgatccccc 1500tggccagtgc acgtctgctg
tcagataaag tctcccgtga actttacccg gtggtgcata 1560tcggggatga aagctggcgc
atgatgacca ccgatatggc cagtgtgccg gtctccgtta 1620tcggggaaga agtggctgat
ctcagccacc gcgaaaatga catcaaaaac gccattaacc 1680tgatgttctg gggaatataa
atgtcaggct cccttataca cagccagtct gcaggtcgac 1740catagtgact ggatatgttg
tgttttacag tattatgtag tctgtttttt atgcaaaatc 1800taatttaata tattgatatt
tatatcattt tacgtttctc gttcagcttt cttgtacaaa 1860gtggtgatta tgtcgtacta
ccatcaccat caccatcacc tcgatgagca ataactagca 1920taaccccttg gggcctctaa
acgggtcttg aggggttttt tgctgaaagg aggaactata 1980tccggatatc cacaggacgg
gtgtggtcgc catgatcgcg tagtcgatag tggctccaag 2040tagcgaagcg agcaggactg
ggcggcggcc aaagcggtcg gacagtgctc cgagaacggg 2100tgcgcataga aattgcatca
acgcatatag cgctagcagc acgccatagt gactggcgat 2160gctgtcggaa tggacgatat
cccgcaagag gcccggcagt accggcataa ccaagcctat 2220gcctacagca tccagggtga
cggtgccgag gatgacgatg agcgcattgt tagatttcat 2280acacggtgcc tgactgcgtt
agcaatttaa ctgtgataaa ctaccgcatt aaagcttatc 2340gatgataagc tgtcaaacat
gagaattctt gaagacgaaa gggcctcgtg atacgcctat 2400ttttataggt taatgtcatg
ataataatgg tttcttagac gtcaggtggc acttttcggg 2460gaaatgtgcg cggaacccct
atttgtttat ttttctaaat acattcaaat atgtatccgc 2520tcatgagaca ataaccctga
taaatgcttc aataatattg aaaaaggaag agtatgagta 2580ttcaacattt ccgtgtcgcc
cttattccct tttttgcggc attttgcctt cctgtttttg 2640ctcacccaga aacgctggtg
aaagtaaaag atgctgaaga tcagttgggt gcacgagtgg 2700gttacatcga actggatctc
aacagcggta agatccttga gagttttcgc cccgaagaac 2760gttttccaat gatgagcact
tttaaagttc tgctatgtgg cgcggtatta tcccgtgttg 2820acgccgggca agagcaactc
ggtcgccgca tacactattc tcagaatgac ttggttgagt 2880actcaccagt cacagaaaag
catcttacgg atggcatgac agtaagagaa ttatgcagtg 2940ctgccataac catgagtgat
aacactgcgg ccaacttact tctgacaacg atcggaggac 3000cgaaggagct aaccgctttt
ttgcacaaca tgggggatca tgtaactcgc cttgatcgtt 3060gggaaccgga gctgaatgaa
gccataccaa acgacgagcg tgacaccacg atgcctgcag 3120caatggcaac aacgttgcgc
aaactattaa ctggcgaact acttactcta gcttcccggc 3180aacaattaat agactggatg
gaggcggata aagttgcagg accacttctg cgctcggccc 3240ttccggctgg ctggtttatt
gctgataaat ctggagccgg tgagcgtggg tctcgcggta 3300tcattgcagc actggggcca
gatggtaagc cctcccgtat cgtagttatc tacacgacgg 3360ggagtcaggc aactatggat
gaacgaaata gacagatcgc tgagataggt gcctcactga 3420ttaagcattg gtaactgtca
gaccaagttt actcatatat actttagatt gatttaaaac 3480ttcattttta atttaaaagg
atctaggtga agatcctttt tgataatctc atgaccaaaa 3540tcccttaacg tgagttttcg
ttccactgag cgtcagaccc cgtagaaaag atcaaaggat 3600cttcttgaga tccttttttt
ctgcgcgtaa tctgctgctt gcaaacaaaa aaaccaccgc 3660taccagcggt ggtttgtttg
ccggatcaag agctaccaac tctttttccg aaggtaactg 3720gcttcagcag agcgcagata
ccaaatactg tccttctagt gtagccgtag ttaggccacc 3780acttcaagaa ctctgtagca
ccgcctacat acctcgctct gctaatcctg ttaccagtgg 3840ctgctgccag tggcgataag
tcgtgtctta ccgggttgga ctcaagacga tagttaccgg 3900ataaggcgca gcggtcgggc
tgaacggggg gttcgtgcac acagcccagc ttggagcgaa 3960cgacctacac cgaactgaga
tacctacagc gtgagctatg agaaagcgcc acgcttcccg 4020aagggagaaa ggcggacagg
tatccggtaa gcggcagggt cggaacagga gagcgcacga 4080gggagcttcc agggggaaac
gcctggtatc tttatagtcc tgtcgggttt cgccacctct 4140gacttgagcg tcgatttttg
tgatgctcgt caggggggcg gagcctatgg aaaaacgcca 4200gcaacgcggc ctttttacgg
ttcctggcct tttgctggcc ttttgctcac atgttctttc 4260ctgcgttatc ccctgattct
gtggataacc gtattaccgc ctttgagtga gctgataccg 4320ctcgccgcag ccgaacgacc
gagcgcagcg agtcagtgag cgaggaagcg gaagagcgcc 4380tgatgcggta ttttctcctt
acgcatctgt gcggtatttc acaccgcata tatggtgcac 4440tctcagtaca atctgctctg
atgccgcata gttaagccag tatacactcc gctatcgcta 4500cgtgactggg tcatggctgc
gccccgacac ccgccaacac ccgctgacgc gccctgacgg 4560gcttgtctgc tcccggcatc
cgcttacaga caagctgtga ccgtctccgg gagctgcatg 4620tgtcagaggt tttcaccgtc
atcaccgaaa cgcgcgaggc agctgcggta aagctcatca 4680gcgtggtcgt gaagcgattc
acagatgtct gcctgttcat ccgcgtccag ctcgttgagt 4740ttctccagaa gcgttaatgt
ctggcttctg ataaagcggg ccatgttaag ggcggttttt 4800tcctgtttgg tcactgatgc
ctccgtgtaa gggggatttc tgttcatggg ggtaatgata 4860ccgatgaaac gagagaggat
gctcacgata cgggttactg atgatgaaca tgcccggtta 4920ctggaacgtt gtgagggtaa
acaactggcg gtatggatgc ggcgggacca gagaaaaatc 4980actcagggtc aatgccagcg
cttcgttaat acagatgtag gtgttccaca gggtagccag 5040cagcatcctg cgatgcagat
ccggaacata atggtgcagg gcgctgactt ccgcgtttcc 5100agactttacg aaacacggaa
accgaagacc attcatgttg ttgctcaggt cgcagacgtt 5160ttgcagcagc agtcgcttca
cgttcgctcg cgtatcggtg attcattctg ctaaccagta 5220aggcaacccc gccagcctag
ccgggtcctc aacgacagga gcacgatcat gcgcacccgt 5280ggccaggacc caacgctgcc
cgagatgcgc cgcgtgcggc tgctggagat ggcggacgcg 5340atggatatgt tctgccaagg
gttggtttgc gcattcacag ttctccgcaa gaattgattg 5400gctccaattc ttggagtggt
gaatccgtta gcgaggtgcc gccggcttcc attcaggtcg 5460aggtggcccg gctccatgca
ccgcgacgca acgcggggag gcagacaagg tatagggcgg 5520cgcctacaat ccatgccaac
ccgttccatg tgctcgccga ggcggcataa atcgccgtga 5580cgatcagcgg tccagtgatc
gaagttaggc tggtaagagc cgcgagcgat ccttgaagct 5640gtccctgatg gtcgtcatct
acctgcctgg acagcatggc ctgcaacgcg ggcatcccga 5700tgccgccgga agcgagaaga
atcataatgg ggaaggccat ccagcctcgc gtcgcgaacg 5760ccagcaagac gtagcccagc
gcgtcggccg ccatgccggc gataatggcc tgcttctcgc 5820cgaaacgttt ggtggcggga
ccagtgacga aggcttgagc gagggcgtgc aagattccga 5880ataccgcaag cgacaggccg
atcatcgtcg cgctccagcg aaagcggtcc tcgccgaaaa 5940tgacccagag cgctgccggc
acctgtccta cgagttgcat gataaagaag acagtcataa 6000gtgcggcgac gatagtcatg
ccccgcgccc accggaagga gctgactggg ttgaaggctc 6060tcaagggcat cggtcgatcg
acgctctccc ttatgcgact cctgcattag gaagcagccc 6120agtagtaggt tgaggccgtt
gagcaccgcc gccgcaagga atggtgcatg caaggagatg 6180gcgcccaaca gtcccccggc
cacggggcct gccaccatac ccacgccgaa acaagcgctc 6240atgagcccga agtggcgagc
ccga 62641526961DNAArtificial
SequenceDescription of Artificial Sequence Synthetic pDEST24
polynucleotide 152atcgagatct cgatcccgcg aaattaatac gactcactat agggagacca
caacggtttc 60cctctagatc acaagtttgt acaaaaaagc tgaacgagaa acgtaaaatg
atataaatat 120caatatatta aattagattt tgcataaaaa acagactaca taatactgta
aaacacaaca 180tatccagtca ctatggcggc cgcattaggc accccaggct ttacacttta
tgcttccggc 240tcgtataatg tgtggatttt gagttaggat ccggcgagat tttcaggagc
taaggaagct 300aaaatggaga aaaaaatcac tggatatacc accgttgata tatcccaatg
gcatcgtaaa 360gaacattttg aggcatttca gtcagttgct caatgtacct ataaccagac
cgttcagctg 420gatattacgg cctttttaaa gaccgtaaag aaaaataagc acaagtttta
tccggccttt 480attcacattc ttgcccgcct gatgaatgct catccggaat tccgtatggc
aatgaaagac 540ggtgagctgg tgatatggga tagtgttcac ccttgttaca ccgttttcca
tgagcaaact 600gaaacgtttt catcgctctg gagtgaatac cacgacgatt tccggcagtt
tctacacata 660tattcgcaag atgtggcgtg ttacggtgaa aacctggcct atttccctaa
agggtttatt 720gagaatatgt ttttcgtctc agccaatccc tgggtgagtt tcaccagttt
tgatttaaac 780gtggccaata tggacaactt cttcgccccc gttttcacca tgggcaaata
ttatacgcaa 840ggcgacaagg tgctgatgcc gctggcgatt caggttcatc atgccgtctg
tgatggcttc 900catgtcggca gaatgcttaa tgaattacaa cagtactgcg atgagtggca
gggcggggcg 960taaacgcgtg gatccggctt actaaaagcc agataacagt atgcgtattt
gcgcgctgat 1020ttttgcggta taagaatata tactgatatg tatacccgaa gtatgtcaaa
aagaggtgtg 1080ctatgaagca gcgtattaca gtgacagttg acagcgacag ctatcagttg
ctcaaggcat 1140atatgatgtc aatatctccg gtctggtaag cacaaccatg cagaatgaag
cccgtcgtct 1200gcgtgccgaa cgctggaaag cggaaaatca ggaagggatg gctgaggtcg
cccggtttat 1260tgaaatgaac ggctcttttg ctgacgagaa cagggactgg tgaaatgcag
tttaaggttt 1320acacctataa aagagagagc cgttatcgtc tgtttgtgga tgtacagagt
gatattattg 1380acacgcccgg gcgacggatg gtgatccccc tggccagtgc acgtctgctg
tcagataaag 1440tctcccgtga actttacccg gtggtgcata tcggggatga aagctggcgc
atgatgacca 1500ccgatatggc cagtgtgccg gtctccgtta tcggggaaga agtggctgat
ctcagccacc 1560gcgaaaatga catcaaaaac gccattaacc tgatgttctg gggaatataa
atgtcaggct 1620cccttataca cagccagtct gcaggtcgac catagtgact ggatatgttg
tgttttacag 1680tattatgtag tctgtttttt atgcaaaatc taatttaata tattgatatt
tatatcattt 1740tacgtttctc gttcagcttt cttgtacaaa gtggtgatta tgtcccctat
actaggttat 1800tggaaaatta agggccttgt gcaacccact cgacttcttt tggaatatct
tgaagaaaaa 1860tatgaagagc atttgtatga gcgcgatgaa ggtgataaat ggcgaaacaa
aaagtttgaa 1920ttgggtttgg agtttcccaa tcttccttat tatattgatg gtgatgttaa
attaacacag 1980tctatggcca tcatacgtta tatagctgac aagcacaaca tgttgggtgg
ttgtccaaaa 2040gagcgtgcag agatttcaat gcttgaagga gcggttttgg atattagata
cggtgtttcg 2100agaattgcat atagtaaaga ctttgaaact ctcaaagttg attttcttag
caagctacct 2160gaaatgctga aaatgttcga agatcgttta tgtcataaaa catatttaaa
tggtgatcat 2220gtaacccatc ctgacttcat gttgtatgac gctcttgatg ttgttttata
catggaccca 2280atgtgcctgg atgcgttccc aaaattagtt tgttttaaaa aacgtattga
agctatccca 2340caaattgata agtacttgaa atccagcaag tatatagcat ggcctttgca
gggctggcaa 2400gccacgtttg gtggtggcga ccatcctcca aaatcggatc tggttccgcg
tccatgggga 2460tccggctgct aacaaagccc gaaaggaagc tgagttggct gctgccaccg
ctgagcaata 2520actagcataa ccccttgggg cctctaaacg ggtcttgagg ggttttttgc
tgaaaggagg 2580aactatatcc ggatatccac aggacgggtg tggtcgccat gatcgcgtag
tcgatagtgg 2640ctccaagtag cgaagcgagc aggactgggc ggcggccaaa gcggtcggac
agtgctccga 2700gaacgggtgc gcatagaaat tgcatcaacg catatagcgc tagcagcacg
ccatagtgac 2760tggcgatgct gtcggaatgg acgatatccc gcaagaggcc cggcagtacc
ggcataacca 2820agcctatgcc tacagcatcc agggtgacgg tgccgaggat gacgatgagc
gcattgttag 2880atttcataca cggtgcctga ctgcgttagc aatttaactg tgataaacta
ccgcattaaa 2940gcttatcgat gataagctgt caaacatgag aattcttgaa gacgaaaggg
cctcgtgata 3000cgcctatttt tataggttaa tgtcatgata ataatggttt cttagacgtc
aggtggcact 3060tttcggggaa atgtgcgcgg aacccctatt tgtttatttt tctaaataca
ttcaaatatg 3120tatccgctca tgagacaata accctgataa atgcttcaat aatattgaaa
aaggaagagt 3180atgagtattc aacatttccg tgtcgccctt attccctttt ttgcggcatt
ttgccttcct 3240gtttttgctc acccagaaac gctggtgaaa gtaaaagatg ctgaagatca
gttgggtgca 3300cgagtgggtt acatcgaact ggatctcaac agcggtaaga tccttgagag
ttttcgcccc 3360gaagaacgtt ttccaatgat gagcactttt aaagttctgc tatgtggcgc
ggtattatcc 3420cgtgttgacg ccgggcaaga gcaactcggt cgccgcatac actattctca
gaatgacttg 3480gttgagtact caccagtcac agaaaagcat cttacggatg gcatgacagt
aagagaatta 3540tgcagtgctg ccataaccat gagtgataac actgcggcca acttacttct
gacaacgatc 3600ggaggaccga aggagctaac cgcttttttg cacaacatgg gggatcatgt
aactcgcctt 3660gatcgttggg aaccggagct gaatgaagcc ataccaaacg acgagcgtga
caccacgatg 3720cctgcagcaa tggcaacaac gttgcgcaaa ctattaactg gcgaactact
tactctagct 3780tcccggcaac aattaataga ctggatggag gcggataaag ttgcaggacc
acttctgcgc 3840tcggcccttc cggctggctg gtttattgct gataaatctg gagccggtga
gcgtgggtct 3900cgcggtatca ttgcagcact ggggccagat ggtaagccct cccgtatcgt
agttatctac 3960acgacgggga gtcaggcaac tatggatgaa cgaaatagac agatcgctga
gataggtgcc 4020tcactgatta agcattggta actgtcagac caagtttact catatatact
ttagattgat 4080ttaaaacttc atttttaatt taaaaggatc taggtgaaga tcctttttga
taatctcatg 4140accaaaatcc cttaacgtga gttttcgttc cactgagcgt cagaccccgt
agaaaagatc 4200aaaggatctt cttgagatcc tttttttctg cgcgtaatct gctgcttgca
aacaaaaaaa 4260ccaccgctac cagcggtggt ttgtttgccg gatcaagagc taccaactct
ttttccgaag 4320gtaactggct tcagcagagc gcagatacca aatactgtcc ttctagtgta
gccgtagtta 4380ggccaccact tcaagaactc tgtagcaccg cctacatacc tcgctctgct
aatcctgtta 4440ccagtggctg ctgccagtgg cgataagtcg tgtcttaccg ggttggactc
aagacgatag 4500ttaccggata aggcgcagcg gtcgggctga acggggggtt cgtgcacaca
gcccagcttg 4560gagcgaacga cctacaccga actgagatac ctacagcgtg agctatgaga
aagcgccacg 4620cttcccgaag ggagaaaggc ggacaggtat ccggtaagcg gcagggtcgg
aacaggagag 4680cgcacgaggg agcttccagg gggaaacgcc tggtatcttt atagtcctgt
cgggtttcgc 4740cacctctgac ttgagcgtcg atttttgtga tgctcgtcag gggggcggag
cctatggaaa 4800aacgccagca acgcggcctt tttacggttc ctggcctttt gctggccttt
tgctcacatg 4860ttctttcctg cgttatcccc tgattctgtg gataaccgta ttaccgcctt
tgagtgagct 4920gataccgctc gccgcagccg aacgaccgag cgcagcgagt cagtgagcga
ggaagcggaa 4980gagcgcctga tgcggtattt tctccttacg catctgtgcg gtatttcaca
ccgcatatat 5040ggtgcactct cagtacaatc tgctctgatg ccgcatagtt aagccagtat
acactccgct 5100atcgctacgt gactgggtca tggctgcgcc ccgacacccg ccaacacccg
ctgacgcgcc 5160ctgacgggct tgtctgctcc cggcatccgc ttacagacaa gctgtgaccg
tctccgggag 5220ctgcatgtgt cagaggtttt caccgtcatc accgaaacgc gcgaggcagc
tgcggtaaag 5280ctcatcagcg tggtcgtgaa gcgattcaca gatgtctgcc tgttcatccg
cgtccagctc 5340gttgagtttc tccagaagcg ttaatgtctg gcttctgata aagcgggcca
tgttaagggc 5400ggttttttcc tgtttggtca ctgatgcctc cgtgtaaggg ggatttctgt
tcatgggggt 5460aatgataccg atgaaacgag agaggatgct cacgatacgg gttactgatg
atgaacatgc 5520ccggttactg gaacgttgtg agggtaaaca actggcggta tggatgcggc
gggaccagag 5580aaaaatcact cagggtcaat gccagcgctt cgttaataca gatgtaggtg
ttccacaggg 5640tagccagcag catcctgcga tgcagatccg gaacataatg gtgcagggcg
ctgacttccg 5700cgtttccaga ctttacgaaa cacggaaacc gaagaccatt catgttgttg
ctcaggtcgc 5760agacgttttg cagcagcagt cgcttcacgt tcgctcgcgt atcggtgatt
cattctgcta 5820accagtaagg caaccccgcc agcctagccg ggtcctcaac gacaggagca
cgatcatgcg 5880cacccgtggc caggacccaa cgctgcccga gatgcgccgc gtgcggctgc
tggagatggc 5940ggacgcgatg gatatgttct gccaagggtt ggtttgcgca ttcacagttc
tccgcaagaa 6000ttgattggct ccaattcttg gagtggtgaa tccgttagcg aggtgccgcc
ggcttccatt 6060caggtcgagg tggcccggct ccatgcaccg cgacgcaacg cggggaggca
gacaaggtat 6120agggcggcgc ctacaatcca tgccaacccg ttccatgtgc tcgccgaggc
ggcataaatc 6180gccgtgacga tcagcggtcc agtgatcgaa gttaggctgg taagagccgc
gagcgatcct 6240tgaagctgtc cctgatggtc gtcatctacc tgcctggaca gcatggcctg
caacgcgggc 6300atcccgatgc cgccggaagc gagaagaatc ataatgggga aggccatcca
gcctcgcgtc 6360gcgaacgcca gcaagacgta gcccagcgcg tcggccgcca tgccggcgat
aatggcctgc 6420ttctcgccga aacgtttggt ggcgggacca gtgacgaagg cttgagcgag
ggcgtgcaag 6480attccgaata ccgcaagcga caggccgatc atcgtcgcgc tccagcgaaa
gcggtcctcg 6540ccgaaaatga cccagagcgc tgccggcacc tgtcctacga gttgcatgat
aaagaagaca 6600gtcataagtg cggcgacgat agtcatgccc cgcgcccacc ggaaggagct
gactgggttg 6660aaggctctca agggcatcgg tcgatcgacg ctctccctta tgcgactcct
gcattaggaa 6720gcagcccagt agtaggttga ggccgttgag caccgccgcc gcaaggaatg
gtgcatgcaa 6780ggagatggcg cccaacagtc ccccggccac ggggcctgcc accataccca
cgccgaaaca 6840agcgctcatg agcccgaagt ggcgagcccg atcttcccca tcggtgatgt
cggcgatata 6900ggcgccagca accgcacctg tggcgccggt gatgccggcc acgatgcgtc
cggcgtagag 6960g
69611536652DNAArtificial SequenceDescription of Artificial
Sequence Synthetic pDEST25 polynucleotide 153ccggaagcga gaagaatcat
aatggggaag gccatccagc ctcgcgtcgc gaacgccagc 60aagacgtagc ccagcgcgtc
ggccgccatg ccggcgataa tggcctgctt ctcgccgaaa 120cgtttggtgg cgggaccagt
gacgaaggct tgagcgaggg cgtgcaagat tccgaatacc 180gcaagcgaca ggccgatcat
cgtcgcgctc cagcgaaagc ggtcctcgcc gaaaatgacc 240cagagcgctg ccggcacctg
tcctacgagt tgcatgataa agaagacagt cataagtgcg 300gcgacgatag tcatgccccg
cgcccaccgg aaggagctga ctgggttgaa ggctctcaag 360ggcatcggtc gatcgacgct
ctcccttatg cgactcctgc attaggaagc agcccagtag 420taggttgagg ccgttgagca
ccgccgccgc aaggaatggt gcatgcaagg agatggcgcc 480caacagtccc ccggccacgg
ggcctgccac catacccacg ccgaaacaag cgctcatgag 540cccgaagtgg cgagcccgat
cttccccatc ggtgatgtcg gcgatatagg cgccagcaac 600cgcacctgtg gcgccggtga
tgccggccac gatgcgtccg gcgtagagga tcgagatctc 660gatcccgcga aattaatacg
actcactata gggagaccac aacggtttcc ctctagatca 720caagtttgta caaaaaagct
gaacgagaaa cgtaaaatga tataaatatc aatatattaa 780attagatttt gcataaaaaa
cagactacat aatactgtaa aacacaacat atccagtcac 840tatggcggcc gcattaggca
ccccaggctt tacactttat gcttccggct cgtataatgt 900gtggattttg agttaggatc
cggcgagatt ttcaggagct aaggaagcta aaatggagaa 960aaaaatcact ggatatacca
ccgttgatat atcccaatgg catcgtaaag aacattttga 1020ggcatttcag tcagttgctc
aatgtaccta taaccagacc gttcagctgg atattacggc 1080ctttttaaag accgtaaaga
aaaataagca caagttttat ccggccttta ttcacattct 1140tgcccgcctg atgaatgctc
atccggaatt ccgtatggca atgaaagacg gtgagctggt 1200gatatgggat agtgttcacc
cttgttacac cgttttccat gagcaaactg aaacgttttc 1260atcgctctgg agtgaatacc
acgacgattt ccggcagttt ctacacatat attcgcaaga 1320tgtggcgtgt tacggtgaaa
acctggccta tttccctaaa gggtttattg agaatatgtt 1380tttcgtctca gccaatccct
gggtgagttt caccagtttt gatttaaacg tggccaatat 1440ggacaacttc ttcgcccccg
ttttcaccat gggcaaatat tatacgcaag gcgacaaggt 1500gctgatgccg ctggcgattc
aggttcatca tgccgtctgt gatggcttcc atgtcggcag 1560aatgcttaat gaattacaac
agtactgcga tgagtggcag ggcggggcgt aaacgcgtgg 1620atccggctta ctaaaagcca
gataacagta tgcgtatttg cgcgctgatt tttgcggtat 1680aagaatatat actgatatgt
atacccgaag tatgtcaaaa agaggtgtgc tatgaagcag 1740cgtattacag tgacagttga
cagcgacagc tatcagttgc tcaaggcata tatgatgtca 1800atatctccgg tctggtaagc
acaaccatgc agaatgaagc ccgtcgtctg cgtgccgaac 1860gctggaaagc ggaaaatcag
gaagggatgg ctgaggtcgc ccggtttatt gaaatgaacg 1920gctcttttgc tgacgagaac
agggactggt gaaatgcagt ttaaggttta cacctataaa 1980agagagagcc gttatcgtct
gtttgtggat gtacagagtg atattattga cacgcccggg 2040cgacggatgg tgatccccct
ggccagtgca cgtctgctgt cagataaagt ctcccgtgaa 2100ctttacccgg tggtgcatat
cggggatgaa agctggcgca tgatgaccac cgatatggcc 2160agtgtgccgg tctccgttat
cggggaagaa gtggctgatc tcagccaccg cgaaaatgac 2220atcaaaaacg ccattaacct
gatgttctgg ggaatataaa tgtcaggctc ccttatacac 2280agccagtctg caggtcgacc
atagtgactg gatatgttgt gttttacagt attatgtagt 2340ctgtttttta tgcaaaatct
aatttaatat attgatattt atatcatttt acgtttctcg 2400ttcagctttc ttgtacaaag
tggtgattat gagcgataaa attattcacc tgactgacga 2460cagttttgac acggatgtac
tcaaagcgga cggggcgatc ctcgtcgatt tctgggcaga 2520gtggtgcggt ccgtgcaaaa
tgatcgcccc gattctggat gaaatcgctg acgaatatca 2580gggcaaactg accgttgcaa
aactgaacat cgatcaaaac cctggcactg cgccgaaata 2640tggcatccgt ggtatcccga
ctctgctgct gttcaaaaac ggtgaagtgg cggcaaccaa 2700agtgggtgca ctgtctaaag
gtcagttgaa agagttcctc gacgctaacc tggccggttc 2760tggttctggt gatgacgatg
acaaggtacc cggggatcga tccggctgct aacaaagccc 2820gaaaggaagc tgagttggct
gctgccaccg ctgagcaata actagcataa ccccttgggg 2880cctctaaacg ggtcttgagg
ggttttttgc tgaaaggagg aactatatcc ggatatccac 2940aggacgggtg tggtcgccat
gatcgcgtag tcgatagtgg ctccaagtag cgaagcgagc 3000aggactgggc ggcggccaaa
gcggtcggac agtgctccga gaacgggtgc gcatagaaat 3060tgcatcaacg catatagcgc
tagcagcacg ccatagtgac tggcgatgct gtcggaatgg 3120acgatatccc gcaagaggcc
cggcagtacc ggcataacca agcctatgcc tacagcatcc 3180agggtgacgg tgccgaggat
gacgatgagc gcattgttag atttcataca cggtgcctga 3240ctgcgttagc aatttaactg
tgataaacta ccgcattaaa gcttatcgat gataagctgt 3300caaacatgag aattcttgaa
gacgaaaggg cctcgtgata cgcctatttt tataggttaa 3360tgtcatgata ataatggttt
cttagacgtc aggtggcact tttcggggaa atgtgcgcgg 3420aacccctatt tgtttatttt
tctaaataca ttcaaatatg tatccgctca tgagacaata 3480accctgataa atgcttcaat
aatattgaaa aaggaagagt atgagtattc aacatttccg 3540tgtcgccctt attccctttt
ttgcggcatt ttgccttcct gtttttgctc acccagaaac 3600gctggtgaaa gtaaaagatg
ctgaagatca gttgggtgca cgagtgggtt acatcgaact 3660ggatctcaac agcggtaaga
tccttgagag ttttcgcccc gaagaacgtt ttccaatgat 3720gagcactttt aaagttctgc
tatgtggcgc ggtattatcc cgtgttgacg ccgggcaaga 3780gcaactcggt cgccgcatac
actattctca gaatgacttg gttgagtact caccagtcac 3840agaaaagcat cttacggatg
gcatgacagt aagagaatta tgcagtgctg ccataaccat 3900gagtgataac actgcggcca
acttacttct gacaacgatc ggaggaccga aggagctaac 3960cgcttttttg cacaacatgg
gggatcatgt aactcgcctt gatcgttggg aaccggagct 4020gaatgaagcc ataccaaacg
acgagcgtga caccacgatg cctgcagcaa tggcaacaac 4080gttgcgcaaa ctattaactg
gcgaactact tactctagct tcccggcaac aattaataga 4140ctggatggag gcggataaag
ttgcaggacc acttctgcgc tcggcccttc cggctggctg 4200gtttattgct gataaatctg
gagccggtga gcgtgggtct cgcggtatca ttgcagcact 4260ggggccagat ggtaagccct
cccgtatcgt agttatctac acgacgggga gtcaggcaac 4320tatggatgaa cgaaatagac
agatcgctga gataggtgcc tcactgatta agcattggta 4380actgtcagac caagtttact
catatatact ttagattgat ttaaaacttc atttttaatt 4440taaaaggatc taggtgaaga
tcctttttga taatctcatg accaaaatcc cttaacgtga 4500gttttcgttc cactgagcgt
cagaccccgt agaaaagatc aaaggatctt cttgagatcc 4560tttttttctg cgcgtaatct
gctgcttgca aacaaaaaaa ccaccgctac cagcggtggt 4620ttgtttgccg gatcaagagc
taccaactct ttttccgaag gtaactggct tcagcagagc 4680gcagatacca aatactgtcc
ttctagtgta gccgtagtta ggccaccact tcaagaactc 4740tgtagcaccg cctacatacc
tcgctctgct aatcctgtta ccagtggctg ctgccagtgg 4800cgataagtcg tgtcttaccg
ggttggactc aagacgatag ttaccggata aggcgcagcg 4860gtcgggctga acggggggtt
cgtgcacaca gcccagcttg gagcgaacga cctacaccga 4920actgagatac ctacagcgtg
agctatgaga aagcgccacg cttcccgaag ggagaaaggc 4980ggacaggtat ccggtaagcg
gcagggtcgg aacaggagag cgcacgaggg agcttccagg 5040gggaaacgcc tggtatcttt
atagtcctgt cgggtttcgc cacctctgac ttgagcgtcg 5100atttttgtga tgctcgtcag
gggggcggag cctatggaaa aacgccagca acgcggcctt 5160tttacggttc ctggcctttt
gctggccttt tgctcacatg ttctttcctg cgttatcccc 5220tgattctgtg gataaccgta
ttaccgcctt tgagtgagct gataccgctc gccgcagccg 5280aacgaccgag cgcagcgagt
cagtgagcga ggaagcggaa gagcgcctga tgcggtattt 5340tctccttacg catctgtgcg
gtatttcaca ccgcatatat ggtgcactct cagtacaatc 5400tgctctgatg ccgcatagtt
aagccagtat acactccgct atcgctacgt gactgggtca 5460tggctgcgcc ccgacacccg
ccaacacccg ctgacgcgcc ctgacgggct tgtctgctcc 5520cggcatccgc ttacagacaa
gctgtgaccg tctccgggag ctgcatgtgt cagaggtttt 5580caccgtcatc accgaaacgc
gcgaggcagc tgcggtaaag ctcatcagcg tggtcgtgaa 5640gcgattcaca gatgtctgcc
tgttcatccg cgtccagctc gttgagtttc tccagaagcg 5700ttaatgtctg gcttctgata
aagcgggcca tgttaagggc ggttttttcc tgtttggtca 5760ctgatgcctc cgtgtaaggg
ggatttctgt tcatgggggt aatgataccg atgaaacgag 5820agaggatgct cacgatacgg
gttactgatg atgaacatgc ccggttactg gaacgttgtg 5880agggtaaaca actggcggta
tggatgcggc gggaccagag aaaaatcact cagggtcaat 5940gccagcgctt cgttaataca
gatgtaggtg ttccacaggg tagccagcag catcctgcga 6000tgcagatccg gaacataatg
gtgcagggcg ctgacttccg cgtttccaga ctttacgaaa 6060cacggaaacc gaagaccatt
catgttgttg ctcaggtcgc agacgttttg cagcagcagt 6120cgcttcacgt tcgctcgcgt
atcggtgatt cattctgcta accagtaagg caaccccgcc 6180agcctagccg ggtcctcaac
gacaggagca cgatcatgcg cacccgtggc caggacccaa 6240cgctgcccga gatgcgccgc
gtgcggctgc tggagatggc ggacgcgatg gatatgttct 6300gccaagggtt ggtttgcgca
ttcacagttc tccgcaagaa ttgattggct ccaattcttg 6360gagtggtgaa tccgttagcg
aggtgccgcc ggcttccatt caggtcgagg tggcccggct 6420ccatgcaccg cgacgcaacg
cggggaggca gacaaggtat agggcggcgc ctacaatcca 6480tgccaacccg ttccatgtgc
tcgccgaggc ggcataaatc gccgtgacga tcagcggtcc 6540agtgatcgaa gttaggctgg
taagagccgc gagcgatcct tgaagctgtc cctgatggtc 6600gtcatctacc tgcctggaca
gcatggcctg caacgcgggc atcccgatgc cg 66521547481DNAArtificial
SequenceDescription of Artificial Sequence Synthetic pDEST26
polynucleotide 154gtaaactgcc cacttggcag tacatcaagt gtatcatatg ccaagtacgc
cccctattga 60cgtcaatgac ggtaaatggc ccgcctggca ttatgcccag tacatgacct
tatgggactt 120tcctacttgg cagtacatct acgtattagt catcgctatt accatggtga
tgcggttttg 180gcagtacatc aatgggcgtg gatagcggtt tgactcacgg ggatttccaa
gtctccaccc 240cattgacgtc aatgggagtt tgttttggca ccaaaatcaa cgggactttc
caaaatgtcg 300taacaactcc gccccattga cgcaaatggg cggtaggcgt gtacggtggg
aggtctatat 360aagcagagct cgtttagtga accgtcagat cgcctggaga cgccatccac
gctgttttga 420cctccataga agacaccggg accgatccag cctccggact ctagcctagg
ccgcggacca 480tggcgtacta ccatcaccat caccatcact ctagatcaac aagtttgtac
aaaaaagctg 540aacgagaaac gtaaaatgat ataaatatca atatattaaa ttagattttg
cataaaaaac 600agactacata atactgtaaa acacaacata tccagtcact atggcggccg
cattaggcac 660cccaggcttt acactttatg cttccggctc gtataatgtg tggattttga
gttaggatcc 720ggcgagattt tcaggagcta aggaagctaa aatggagaaa aaaatcactg
gatataccac 780cgttgatata tcccaatggc atcgtaaaga acattttgag gcatttcagt
cagttgctca 840atgtacctat aaccagaccg ttcagctgga tattacggcc tttttaaaga
ccgtaaagaa 900aaataagcac aagttttatc cggcctttat tcacattctt gcccgcctga
tgaatgctca 960tccggaattc cgtatggcaa tgaaagacgg tgagctggtg atatgggata
gtgttcaccc 1020ttgttacacc gttttccatg agcaaactga aacgttttca tcgctctgga
gtgaatacca 1080cgacgatttc cggcagtttc tacacatata ttcgcaagat gtggcgtgtt
acggtgaaaa 1140cctggcctat ttccctaaag ggtttattga gaatatgttt ttcgtctcag
ccaatccctg 1200ggtgagtttc accagttttg atttaaacgt ggccaatatg gacaacttct
tcgcccccgt 1260tttcaccatg ggcaaatatt atacgcaagg cgacaaggtg ctgatgccgc
tggcgattca 1320ggttcatcat gccgtctgtg atggcttcca tgtcggcaga atgcttaatg
aattacaaca 1380gtactgcgat gagtggcagg gcggggcgta aagatctgga tccggcttac
taaaagccag 1440ataacagtat gcgtatttgc gcgctgattt ttgcggtata agaatatata
ctgatatgta 1500tacccgaagt atgtcaaaaa gaggtgtgct atgaagcagc gtattacagt
gacagttgac 1560agcgacagct atcagttgct caaggcatat atgatgtcaa tatctccggt
ctggtaagca 1620caaccatgca gaatgaagcc cgtcgtctgc gtgccgaacg ctggaaagcg
gaaaatcagg 1680aagggatggc tgaggtcgcc cggtttattg aaatgaacgg ctcttttgct
gacgagaaca 1740gggactggtg aaatgcagtt taaggtttac acctataaaa gagagagccg
ttatcgtctg 1800tttgtggatg tacagagtga tattattgac acgcccgggc gacggatggt
gatccccctg 1860gccagtgcac gtctgctgtc agataaagtc tcccgtgaac tttacccggt
ggtgcatatc 1920ggggatgaaa gctggcgcat gatgaccacc gatatggcca gtgtgccggt
ctccgttatc 1980ggggaagaag tggctgatct cagccaccgc gaaaatgaca tcaaaaacgc
cattaacctg 2040atgttctggg gaatataaat gtcaggctcc cttatacaca gccagtctgc
aggtcgacca 2100tagtgactgg atatgttgtg ttttacagta ttatgtagtc tgttttttat
gcaaaatcta 2160atttaatata ttgatattta tatcatttta cgtttctcgt tcagctttct
tgtacaaagt 2220ggttgatcgc gtgcatgcga cgtcatagct ctctccctat agtgagtcgt
attataagct 2280aggcactggc cgtcgtttta caacgtcgtg actgggaaaa ctgctagctt
gggatctttg 2340tgaaggaacc ttacttctgt ggtgtgacat aattggacaa actacctaca
gagatttaaa 2400gctctaaggt aaatataaaa tttttaagtg tataatgtgt taaactagct
gcatatgctt 2460gctgcttgag agttttgctt actgagtatg atttatgaaa atattataca
caggagctag 2520tgattctaat tgtttgtgta ttttagattc acagtcccaa ggctcatttc
aggcccctca 2580gtcctcacag tctgttcatg atcataatca gccataccac atttgtagag
gttttacttg 2640ctttaaaaaa cctcccacac ctccccctga acctgaaaca taaaatgaat
gcaattgttg 2700ttgttaactt gtttattgca gcttataatg gttacaaata aagcaatagc
atcacaaatt 2760tcacaaataa agcatttttt tcactgcatt ctagttgtgg tttgtccaaa
ctcatcaatg 2820tatcttatca tgtctggatc gatcctgcat taatgaatcg gccaacgcgc
ggggagaggc 2880ggtttgcgta ttggctggcg taatagcgaa gaggcccgca ccgatcgccc
ttcccaacag 2940ttgcgcagcc tgaatggcga atgggacgcg ccctgtagcg gcgcattaag
cgcggcgggt 3000gtggtggtta cgcgcagcgt gaccgctaca cttgccagcg ccctagcgcc
cgctcctttc 3060gctttcttcc cttcctttct cgccacgttc gccggctttc cccgtcaagc
tctaaatcgg 3120gggctccctt tagggttccg atttagtgct ttacggcacc tcgaccccaa
aaaacttgat 3180tagggtgatg gttcacgtag tgggccatcg ccctgataga cggtttttcg
ccctttgacg 3240ttggagtcca cgttctttaa tagtggactc ttgttccaaa ctggaacaac
actcaaccct 3300atctcggtct attcttttga tttataaggg attttgccga tttcggccta
ttggttaaaa 3360aatgagctga tttaacaaat atttaacgcg aattttaaca aaatattaac
gtttacaatt 3420tcgcctgatg cggtattttc tccttacgca tctgtgcggt atttcacacc
gcatacgcgg 3480atctgcgcag caccatggcc tgaaataacc tctgaaagag gaacttggtt
aggtaccttc 3540tgaggcggaa agaaccagct gtggaatgtg tgtcagttag ggtgtggaaa
gtccccaggc 3600tccccagcag gcagaagtat gcaaagcatg catctcaatt agtcagcaac
caggtgtgga 3660aagtccccag gctccccagc aggcagaagt atgcaaagca tgcatctcaa
ttagtcagca 3720accatagtcc cgcccctaac tccgcccatc ccgcccctaa ctccgcccag
ttccgcccat 3780tctccgcccc atggctgact aatttttttt atttatgcag aggccgaggc
cgcctcggcc 3840tctgagctat tccagaagta gtgaggaggc ttttttggag gcctaggctt
ttgcaaaaag 3900cttgattctt ctgacacaac agtctcgaac ttaaggctag agccaccatg
attgaacaag 3960atggattgca cgcaggttct ccggccgctt gggtggagag gctattcggc
tatgactggg 4020cacaacagac aatcggctgc tctgatgccg ccgtgttccg gctgtcagcg
caggggcgcc 4080cggttctttt tgtcaagacc gacctgtccg gtgccctgaa tgaactgcag
gacgaggcag 4140cgcggctatc gtggctggcc acgacgggcg ttccttgcgc agctgtgctc
gacgttgtca 4200ctgaagcggg aagggactgg ctgctattgg gcgaagtgcc ggggcaggat
ctcctgtcat 4260ctcaccttgc tcctgccgag aaagtatcca tcatggctga tgcaatgcgg
cggctgcata 4320cgcttgatcc ggctacctgc ccattcgacc accaagcgaa acatcgcatc
gagcgagcac 4380gtactcggat ggaagccggt cttgtcgatc aggatgatct ggacgaagag
catcaggggc 4440tcgcgccagc cgaactgttc gccaggctca aggcgcgcat gcccgacggc
gaggatctcg 4500tcgtgaccca tggcgatgcc tgcttgccga atatcatggt ggaaaatggc
cgcttttctg 4560gattcatcga ctgtggccgg ctgggtgtgg cggaccgcta tcaggacata
gcgttggcta 4620cccgtgatat tgctgaagag cttggcggcg aatgggctga ccgcttcctc
gtgctttacg 4680gtatcgccgc tcccgattcg cagcgcatcg ccttctatcg ccttcttgac
gagttcttct 4740gagcgggact ctggggttcg aaatgaccga ccaagcgacg cccaacctgc
catcacgatg 4800gccgcaataa aatatcttta ttttcattac atctgtgtgt tggttttttg
tgtgaatcga 4860tagcgataag gatccgcgta tggtgcactc tcagtacaat ctgctctgat
gccgcatagt 4920taagccagcc ccgacacccg ccaacacccg ctgacgcgcc ctgacgggct
tgtctgctcc 4980cggcatccgc ttacagacaa gctgtgaccg tctccgggag ctgcatgtgt
cagaggtttt 5040caccgtcatc accgaaacgc gcgagacgaa agggcctcgt gatacgccta
tttttatagg 5100ttaatgtcat gataataatg gtttcttaga cgtcaggtgg cacttttcgg
ggaaatgtgc 5160gcggaacccc tatttgttta tttttctaaa tacattcaaa tatgtatccg
ctcatgagac 5220aataaccctg ataaatgctt caataatatt gaaaaaggaa gagtatgagt
attcaacatt 5280tccgtgtcgc ccttattccc ttttttgcgg cattttgcct tcctgttttt
gctcacccag 5340aaacgctggt gaaagtaaaa gatgctgaag atcagttggg tgcacgagtg
ggttacatcg 5400aactggatct caacagcggt aagatccttg agagttttcg ccccgaagaa
cgttttccaa 5460tgatgagcac ttttaaagtt ctgctatgtg gcgcggtatt atcccgtatt
gacgccgggc 5520aagagcaact cggtcgccgc atacactatt ctcagaatga cttggttgag
tactcaccag 5580tcacagaaaa gcatcttacg gatggcatga cagtaagaga attatgcagt
gctgccataa 5640ccatgagtga taacactgcg gccaacttac ttctgacaac gatcggagga
ccgaaggagc 5700taaccgcttt tttgcacaac atgggggatc atgtaactcg ccttgatcgt
tgggaaccgg 5760agctgaatga agccatacca aacgacgagc gtgacaccac gatgcctgta
gcaatggcaa 5820caacgttgcg caaactatta actggcgaac tacttactct agcttcccgg
caacaattaa 5880tagactggat ggaggcggat aaagttgcag gaccacttct gcgctcggcc
cttccggctg 5940gctggtttat tgctgataaa tctggagccg gtgagcgtgg gtctcgcggt
atcattgcag 6000cactggggcc agatggtaag ccctcccgta tcgtagttat ctacacgacg
gggagtcagg 6060caactatgga tgaacgaaat agacagatcg ctgagatagg tgcctcactg
attaagcatt 6120ggtaactgtc agaccaagtt tactcatata tactttagat tgatttaaaa
cttcattttt 6180aatttaaaag gatctaggtg aagatccttt ttgataatct catgaccaaa
atcccttaac 6240gtgagttttc gttccactga gcgtcagacc ccgtagaaaa gatcaaagga
tcttcttgag 6300atcctttttt tctgcgcgta atctgctgct tgcaaacaaa aaaaccaccg
ctaccagcgg 6360tggtttgttt gccggatcaa gagctaccaa ctctttttcc gaaggtaact
ggcttcagca 6420gagcgcagat accaaatact gtccttctag tgtagccgta gttaggccac
cacttcaaga 6480actctgtagc accgcctaca tacctcgctc tgctaatcct gttaccagtg
gctgctgcca 6540gtggcgataa gtcgtgtctt accgggttgg actcaagacg atagttaccg
gataaggcgc 6600agcggtcggg ctgaacgggg ggttcgtgca cacagcccag cttggagcga
acgacctaca 6660ccgaactgag atacctacag cgtgagcatt gagaaagcgc cacgcttccc
gaagggagaa 6720aggcggacag gtatccggta agcggcaggg tcggaacagg agagcgcacg
agggagcttc 6780cagggggaaa cgcctggtat ctttatagtc ctgtcgggtt tcgccacctc
tgacttgagc 6840gtcgattttt gtgatgctcg tcaggggggc ggagcctatg gaaaaacgcc
agcaacgcgg 6900cctttttacg gttcctggcc ttttgctggc cttttgctca catgttcttt
cctgcgttat 6960cccctgattc tgtggataac cgtattaccg cctttgagtg agctgatacc
gctcgccgca 7020gccgaacgac cgagcgcagc gagtcagtga gcgaggaagc ggaagagcgc
ccaatacgca 7080aaccgcctct ccccgcgcgt tggccgattc attaatgcag agcttgcaat
tcgcgcgttt 7140ttcaatatta ttgaagcatt tatcagggtt attgtctcat gagcggatac
atatttgaat 7200gtatttagaa aaataaacaa ataggggttc cgcgcacatt tccccgaaaa
gtgccacctg 7260acgtctaaga aaccattatt atcatgacat taacctataa aaataggcgt
agtacgaggc 7320cctttcactc attagatgca tgtcgttaca taacttacgg taaatggccc
gcctggctga 7380ccgcccaacg acccccgccc attgacgtca ataatgacgt atgttcccat
agtaacgcca 7440atagggactt tccattgacg tcaatgggtg gagtatttac g
74811558123DNAArtificial SequenceDescription of Artificial
Sequence Synthetic pDEST27 polynucleotide 155ataagcagag ctcgtttagt
gaaccgtcag atcgcctgga gacgccatcc acgctgtttt 60gacctccata gaagacaccg
ggaccgatcc agcctccgga ctctagccta ggccgcggac 120catggcccct atactaggtt
attggaaaat taagggcctt gtgcaaccca ctcgacttct 180tttggaatat cttgaagaaa
aatatgaaga gcatttgtat gagcgcgatg aaggtgataa 240atggcgaaac aaaaagtttg
aattgggttt ggagtttccc aatcttcctt attatattga 300tggtgatgtt aaattaacac
agtctatggc catcatacgt tatatagctg acaagcacaa 360catgttgggt ggttgtccaa
aagagcgtgc agagatttca atgcttgaag gagcggtttt 420ggatattaga tacggtgttt
cgagaattgc atatagtaaa gactttgaaa ctctcaaagt 480tgattttctt agcaagctac
ctgaaatgct gaaaatgttc gaagatcgtt tatgtcataa 540aacatattta aatggtgatc
atgtaaccca tcctgacttc atgttgtatg acgctcttga 600tgttgtttta tacatggacc
caatgtgcct ggatgcgttc ccaaaattag tttgttttaa 660aaaacgtatt gaagctatcc
cacaaattga taagtacttg aaatccagca agtatatagc 720atggcctttg cagggctggc
aagccacgtt tggtggtggc gaccatcctc caaaatcgga 780tctggttccg cgttctagat
caacaagttt gtacaaaaaa gctgaacgag aaacgtaaaa 840tgatataaat atcaatatat
taaattagat tttgcataaa aaacagacta cataatactg 900taaaacacaa catatccagt
cactatggcg gccgcattag gcaccccagg ctttacactt 960tatgcttccg gctcgtataa
tgtgtggatt ttgagttagg atccggcgag attttcagga 1020gctaaggaag ctaaaatgga
gaaaaaaatc actggatata ccaccgttga tatatcccaa 1080tggcatcgta aagaacattt
tgaggcattt cagtcagttg ctcaatgtac ctataaccag 1140accgttcagc tggatattac
ggccttttta aagaccgtaa agaaaaataa gcacaagttt 1200tatccggcct ttattcacat
tcttgcccgc ctgatgaatg ctcatccgga attccgtatg 1260gcaatgaaag acggtgagct
ggtgatatgg gatagtgttc acccttgtta caccgttttc 1320catgagcaaa ctgaaacgtt
ttcatcgctc tggagtgaat accacgacga tttccggcag 1380tttctacaca tatattcgca
agatgtggcg tgttacggtg aaaacctggc ctatttccct 1440aaagggttta ttgagaatat
gtttttcgtc tcagccaatc cctgggtgag tttcaccagt 1500tttgatttaa acgtggccaa
tatggacaac ttcttcgccc ccgttttcac catgggcaaa 1560tattatacgc aaggcgacaa
ggtgctgatg ccgctggcga ttcaggttca tcatgccgtc 1620tgtgatggct tccatgtcgg
cagaatgctt aatgaattac aacagtactg cgatgagtgg 1680cagggcgggg cgtaaagatc
tggatccggc ttactaaaag ccagataaca gtatgcgtat 1740ttgcgcgctg atttttgcgg
tataagaata tatactgata tgtatacccg aagtatgtca 1800aaaagaggtg tgctatgaag
cagcgtatta cagtgacagt tgacagcgac agctatcagt 1860tgctcaaggc atatatgatg
tcaatatctc cggtctggta agcacaacca tgcagaatga 1920agcccgtcgt ctgcgtgccg
aacgctggaa agcggaaaat caggaaggga tggctgaggt 1980cgcccggttt attgaaatga
acggctcttt tgctgacgag aacagggact ggtgaaatgc 2040agtttaaggt ttacacctat
aaaagagaga gccgttatcg tctgtttgtg gatgtacaga 2100gtgatattat tgacacgccc
gggcgacgga tggtgatccc cctggccagt gcacgtctgc 2160tgtcagataa agtctcccgt
gaactttacc cggtggtgca tatcggggat gaaagctggc 2220gcatgatgac caccgatatg
gccagtgtgc cggtctccgt tatcggggaa gaagtggctg 2280atctcagcca ccgcgaaaat
gacatcaaaa acgccattaa cctgatgttc tggggaatat 2340aaatgtcagg ctcccttata
cacagccagt ctgcaggtcg accatagtga ctggatatgt 2400tgtgttttac agtattatgt
agtctgtttt ttatgcaaaa tctaatttaa tatattgata 2460tttatatcat tttacgtttc
tcgttcagct ttcttgtaca aagtggttga tcgcgtgcat 2520gcgacgtcat agctctctcc
ctatagtgag tcgtattata agctaggcac tggccgtcgt 2580tttacaacgt cgtgactggg
aaaactgcta gcttgggatc tttgtgaagg aaccttactt 2640ctgtggtgtg acataattgg
acaaactacc tacagagatt taaagctcta aggtaaatat 2700aaaattttta agtgtataat
gtgttaaact agctgcatat gcttgctgct tgagagtttt 2760gcttactgag tatgatttat
gaaaatatta tacacaggag ctagtgattc taattgtttg 2820tgtattttag attcacagtc
ccaaggctca tttcaggccc ctcagtcctc acagtctgtt 2880catgatcata atcagccata
ccacatttgt agaggtttta cttgctttaa aaaacctccc 2940acacctcccc ctgaacctga
aacataaaat gaatgcaatt gttgttgtta acttgtttat 3000tgcagcttat aatggttaca
aataaagcaa tagcatcaca aatttcacaa ataaagcatt 3060tttttcactg cattctagtt
gtggtttgtc caaactcatc aatgtatctt atcatgtctg 3120gatcgatcct gcattaatga
atcggccaac gcgcggggag aggcggtttg cgtattggct 3180ggcgtaatag cgaagaggcc
cgcaccgatc gcccttccca acagttgcgc agcctgaatg 3240gcgaatggga cgcgccctgt
agcggcgcat taagcgcggc gggtgtggtg gttacgcgca 3300gcgtgaccgc tacacttgcc
agcgccctag cgcccgctcc tttcgctttc ttcccttcct 3360ttctcgccac gttcgccggc
tttccccgtc aagctctaaa tcgggggctc cctttagggt 3420tccgatttag tgctttacgg
cacctcgacc ccaaaaaact tgattagggt gatggttcac 3480gtagtgggcc atcgccctga
tagacggttt ttcgcccttt gacgttggag tccacgttct 3540ttaatagtgg actcttgttc
caaactggaa caacactcaa ccctatctcg gtctattctt 3600ttgatttata agggattttg
ccgatttcgg cctattggtt aaaaaatgag ctgatttaac 3660aaatatttaa cgcgaatttt
aacaaaatat taacgtttac aatttcgcct gatgcggtat 3720tttctcctta cgcatctgtg
cggtatttca caccgcatac gcggatctgc gcagcaccat 3780ggcctgaaat aacctctgaa
agaggaactt ggttaggtac cttctgaggc ggaaagaacc 3840agctgtggaa tgtgtgtcag
ttagggtgtg gaaagtcccc aggctcccca gcaggcagaa 3900gtatgcaaag catgcatctc
aattagtcag caaccaggtg tggaaagtcc ccaggctccc 3960cagcaggcag aagtatgcaa
agcatgcatc tcaattagtc agcaaccata gtcccgcccc 4020taactccgcc catcccgccc
ctaactccgc ccagttccgc ccattctccg ccccatggct 4080gactaatttt ttttatttat
gcagaggccg aggccgcctc ggcctctgag ctattccaga 4140agtagtgagg aggctttttt
ggaggcctag gcttttgcaa aaagcttgat tcttctgaca 4200caacagtctc gaacttaagg
ctagagccac catgattgaa caagatggat tgcacgcagg 4260ttctccggcc gcttgggtgg
agaggctatt cggctatgac tgggcacaac agacaatcgg 4320ctgctctgat gccgccgtgt
tccggctgtc agcgcagggg cgcccggttc tttttgtcaa 4380gaccgacctg tccggtgccc
tgaatgaact gcaggacgag gcagcgcggc tatcgtggct 4440ggccacgacg ggcgttcctt
gcgcagctgt gctcgacgtt gtcactgaag cgggaaggga 4500ctggctgcta ttgggcgaag
tgccggggca ggatctcctg tcatctcacc ttgctcctgc 4560cgagaaagta tccatcatgg
ctgatgcaat gcggcggctg catacgcttg atccggctac 4620ctgcccattc gaccaccaag
cgaaacatcg catcgagcga gcacgtactc ggatggaagc 4680cggtcttgtc gatcaggatg
atctggacga agagcatcag gggctcgcgc cagccgaact 4740gttcgccagg ctcaaggcgc
gcatgcccga cggcgaggat ctcgtcgtga cccatggcga 4800tgcctgcttg ccgaatatca
tggtggaaaa tggccgcttt tctggattca tcgactgtgg 4860ccggctgggt gtggcggacc
gctatcagga catagcgttg gctacccgtg atattgctga 4920agagcttggc ggcgaatggg
ctgaccgctt cctcgtgctt tacggtatcg ccgctcccga 4980ttcgcagcgc atcgccttct
atcgccttct tgacgagttc ttctgagcgg gactctgggg 5040ttcgaaatga ccgaccaagc
gacgcccaac ctgccatcac gatggccgca ataaaatatc 5100tttattttca ttacatctgt
gtgttggttt tttgtgtgaa tcgatagcga taaggatccg 5160cgtatggtgc actctcagta
caatctgctc tgatgccgca tagttaagcc agccccgaca 5220cccgccaaca cccgctgacg
cgccctgacg ggcttgtctg ctcccggcat ccgcttacag 5280acaagctgtg accgtctccg
ggagctgcat gtgtcagagg ttttcaccgt catcaccgaa 5340acgcgcgaga cgaaagggcc
tcgtgatacg cctattttta taggttaatg tcatgataat 5400aatggtttct tagacgtcag
gtggcacttt tcggggaaat gtgcgcggaa cccctatttg 5460tttatttttc taaatacatt
caaatatgta tccgctcatg agacaataac cctgataaat 5520gcttcaataa tattgaaaaa
ggaagagtat gagtattcaa catttccgtg tcgcccttat 5580tccctttttt gcggcatttt
gccttcctgt ttttgctcac ccagaaacgc tggtgaaagt 5640aaaagatgct gaagatcagt
tgggtgcacg agtgggttac atcgaactgg atctcaacag 5700cggtaagatc cttgagagtt
ttcgccccga agaacgtttt ccaatgatga gcacttttaa 5760agttctgcta tgtggcgcgg
tattatcccg tattgacgcc gggcaagagc aactcggtcg 5820ccgcatacac tattctcaga
atgacttggt tgagtactca ccagtcacag aaaagcatct 5880tacggatggc atgacagtaa
gagaattatg cagtgctgcc ataaccatga gtgataacac 5940tgcggccaac ttacttctga
caacgatcgg aggaccgaag gagctaaccg cttttttgca 6000caacatgggg gatcatgtaa
ctcgccttga tcgttgggaa ccggagctga atgaagccat 6060accaaacgac gagcgtgaca
ccacgatgcc tgtagcaatg gcaacaacgt tgcgcaaact 6120attaactggc gaactactta
ctctagcttc ccggcaacaa ttaatagact ggatggaggc 6180ggataaagtt gcaggaccac
ttctgcgctc ggcccttccg gctggctggt ttattgctga 6240taaatctgga gccggtgagc
gtgggtctcg cggtatcatt gcagcactgg ggccagatgg 6300taagccctcc cgtatcgtag
ttatctacac gacggggagt caggcaacta tggatgaacg 6360aaatagacag atcgctgaga
taggtgcctc actgattaag cattggtaac tgtcagacca 6420agtttactca tatatacttt
agattgattt aaaacttcat ttttaattta aaaggatcta 6480ggtgaagatc ctttttgata
atctcatgac caaaatccct taacgtgagt tttcgttcca 6540ctgagcgtca gaccccgtag
aaaagatcaa aggatcttct tgagatcctt tttttctgcg 6600cgtaatctgc tgcttgcaaa
caaaaaaacc accgctacca gcggtggttt gtttgccgga 6660tcaagagcta ccaactcttt
ttccgaaggt aactggcttc agcagagcgc agataccaaa 6720tactgtcctt ctagtgtagc
cgtagttagg ccaccacttc aagaactctg tagcaccgcc 6780tacatacctc gctctgctaa
tcctgttacc agtggctgct gccagtggcg ataagtcgtg 6840tcttaccggg ttggactcaa
gacgatagtt accggataag gcgcagcggt cgggctgaac 6900ggggggttcg tgcacacagc
ccagcttgga gcgaacgacc tacaccgaac tgagatacct 6960acagcgtgag cattgagaaa
gcgccacgct tcccgaaggg agaaaggcgg acaggtatcc 7020ggtaagcggc agggtcggaa
caggagagcg cacgagggag cttccagggg gaaacgcctg 7080gtatctttat agtcctgtcg
ggtttcgcca cctctgactt gagcgtcgat ttttgtgatg 7140ctcgtcaggg gggcggagcc
tatggaaaaa cgccagcaac gcggcctttt tacggttcct 7200ggccttttgc tggccttttg
ctcacatgtt ctttcctgcg ttatcccctg attctgtgga 7260taaccgtatt accgcctttg
agtgagctga taccgctcgc cgcagccgaa cgaccgagcg 7320cagcgagtca gtgagcgagg
aagcggaaga gcgcccaata cgcaaaccgc ctctccccgc 7380gcgttggccg attcattaat
gcagagcttg caattcgcgc gtttttcaat attattgaag 7440catttatcag ggttattgtc
tcatgagcgg atacatattt gaatgtattt agaaaaataa 7500acaaataggg gttccgcgca
catttccccg aaaagtgcca cctgacgtct aagaaaccat 7560tattatcatg acattaacct
ataaaaatag gcgtagtacg aggccctttc actcattaga 7620tgcatgtcgt tacataactt
acggtaaatg gcccgcctgg ctgaccgccc aacgaccccc 7680gcccattgac gtcaataatg
acgtatgttc ccatagtaac gccaataggg actttccatt 7740gacgtcaatg ggtggagtat
ttacggtaaa ctgcccactt ggcagtacat caagtgtatc 7800atatgccaag tacgccccct
attgacgtca atgacggtaa atggcccgcc tggcattatg 7860cccagtacat gaccttatgg
gactttccta cttggcagta catctacgta ttagtcatcg 7920ctattaccat ggtgatgcgg
ttttggcagt acatcaatgg gcgtggatag cggtttgact 7980cacggggatt tccaagtctc
caccccattg acgtcaatgg gagtttgttt tggcaccaaa 8040atcaacggga ctttccaaaa
tgtcgtaaca actccgcccc attgacgcaa atgggcggta 8100ggcgtgtacg gtgggaggtc
tat 81231564396DNAArtificial
SequenceDescription of Artificial Sequence Synthetic pEXP501
polynucleotide 156ccattcgcca ttcaggctgc gcaactgttg ggaagggcga tcggtgcggg
cctcttcgct 60attacgccag ccaatacgca aaccgcctct ccccgcgcgt tggccgattc
attaatgcag 120gatcgatcca gacatgataa gatacattga tgagtttgga caaaccacaa
ctagaatgca 180gtgaaaaaaa tgctttattt gtgaaatttg tgatgctatt gctttatttg
taaccattat 240aagctgcaat aaacaagtta acaacaacaa ttgcattcat tttatgtttc
aggttcaggg 300ggaggtgtgg gaggtttttt aaagcaagta aaacctctac aaatgtggta
tggctgatta 360tgatcatgaa cagactgtga ggactgaggg gcctgaaatg agccttggga
ctgtgaatct 420aaaatacaca aacaattaga atcactagct cctgtgtata atattttcat
aaatcatact 480cagtaagcaa aactctcaag cagcaagcat atgcagctag tttaacacat
tatacactta 540aaaattttat atttacctta gagctttaaa tctctgtagg tagtttgtcc
aattatgtca 600caccacagaa gtaaggttcc ttcacaaaga tcccaagcta gcagttttcc
cagtcacgac 660gttgtaaaac gacggccagt gcctagctta taatacgact cactataggg
accactttgt 720acaagaaagc tgggtacgcg taagcttggg cccctcgagg gatcctctag
agcggccgcc 780gactagtgag ctcgtcgacg atatcccggg aattccggac cggtaccagc
ctgctttttt 840gtacaaactt gttctatagt gtcacctaaa taggcctaat ggtcatagct
gtttcctgtg 900tgaaattgtt atccgctccg cggcctaggc tagagtccgg aggctggatc
ggtcccggtg 960tcttctatgg aggtcaaaac agcgtggatg gcgtctccag gcgatctgac
ggttcactaa 1020acgagctctg cttatataga cctcccaccg tacacgccta ccgcccattt
gcgtcaatgg 1080ggcggagttg ttacgacatt ttggaaagtc ccgttgattt tggtgccaaa
acaaactccc 1140attgacgtca atggggtgga gacttggaaa tccccgtgag tcaaaccgct
atccacgccc 1200attgatgtac tgccaaaacc gcatcaccat ggtaatagcg atgactaata
cgtagatgta 1260ctgccaagta ggaaagtccc ataaggtcat gtactgggca taatgccagg
cgggccattt 1320accgtcattg acgtcaatag ggggcgtact tggcatatga tacacttgat
gtactgccaa 1380gtgggcagtt taccgtaaat actccaccca ttgacgtcaa tggaaagtcc
ctattggcgt 1440tactatggga acatacgtca ttattgacgt caatgggcgg gggtcgttgg
gcggtcagcc 1500aggcgggcca tttaccgtaa gttatgtaac gacatgcatc taatgagtga
aagggcctcg 1560tactacgcct atttttatag gttaatgtca tgataataat ggtttcttag
acgtcaggtg 1620gcacttttcg gggaaatgtg cgcggaaccc ctatttgttt atttttctaa
atacattcaa 1680atatgtatcc gctcatgaga caataaccct gataaatgct tcaataatat
tgaaaaacgc 1740gcgaattgca agctctgcat taatgaatcg gccaacgcgc ggggagaggc
ggtttgcgta 1800ttgggcgctc ttccgcttcc tcgctcactg actcgctgcg ctcggtcgtt
cggctgcggc 1860gagcggtatc agctcactca aaggcggtaa tacggttatc cacagaatca
ggggataacg 1920caggaaagaa catgtgagca aaaggccagc aaaaggccag gaaccgtaaa
aaggccgcgt 1980tgctggcgtt tttccatagg ctccgccccc ctgacgagca tcacaaaaat
cgacgctcaa 2040gtcagaggtg gcgaaacccg acaggactat aaagatacca ggcgtttccc
cctggaagct 2100ccctcgtgcg ctctcctgtt ccgaccctgc cgcttaccgg atacctgtcc
gcctttctcc 2160cttcgggaag cgtggcgctt tctcaatgct cacgctgtag gtatctcagt
tcggtgtagg 2220tcgttcgctc caagctgggc tgtgtgcacg aaccccccgt tcagcccgac
cgctgcgcct 2280tatccggtaa ctatcgtctt gagtccaacc cggtaagaca cgacttatcg
ccactggcag 2340cagccactgg taacaggatt agcagagcga ggtatgtagg cggtgctaca
gagttcttga 2400agtggtggcc taactacggc tacactagaa ggacagtatt tggtatctgc
gctctgctga 2460agccagttac cttcggaaaa agagttggta gctcttgatc cggcaaacaa
accaccgctg 2520gtagcggtgg tttttttgtt tgcaagcagc agattacgcg cagaaaaaaa
ggatctcaag 2580aagatccttt gatcttttct acggggtctg acgctcagtg gaacgaaaac
tcacgttaag 2640ggattttggt catgccataa cttcgtatag catacattat acgaagttat
ggcatgagat 2700tatcaaaaag gatcttcacc tagatccttt taaattaaaa atgaagtttt
aaatcaatct 2760aaagtatata tgagtaaact tggtctgaca gttaccaatg cttaatcagt
gaggcaccta 2820tctcagcgat ctgtctattt cgttcatcca tagttgcctg actccccgtc
gtgtagataa 2880ctacgatacg ggagggctta ccatctggcc ccagtgctgc aatgataccg
cgagacccac 2940gctcaccggc tccagattta tcagcaataa accagccagc cggaagggcc
gagcgcagaa 3000gtggtcctgc aactttatcc gcctccatcc agtctattaa ttgttgccgg
gaagctagag 3060taagtagttc gccagttaat agtttgcgca acgttgttgc cattgctaca
ggcatcgtgg 3120tgtcacgctc gtcgtttggt atggcttcat tcagctccgg ttcccaacga
tcaaggcgag 3180ttacatgatc ccccatgttg tgcaaaaaag cggttagctc cttcggtcct
ccgatcgttg 3240tcagaagtaa gttggccgca gtgttatcac tcatggttat ggcagcactg
cataattctc 3300ttactgtcat gccatccgta agatgctttt ctgtgactgg tgagtactca
accaagtcat 3360tctgagaata gtgtatgcgg cgaccgagtt gctcttgccc ggcgtcaata
cgggataata 3420ccgcgccaca tagcagaact ttaaaagtgc tcatcattgg aaaacgttct
tcggggcgaa 3480aactctcaag gatcttaccg ctgttgagat ccagttcgat gtaacccact
cgtgcaccca 3540actgatcttc agcatctttt actttcacca gcgtttctgg gtgagcaaaa
acaggaaggc 3600aaaatgccgc aaaaaaggga ataagggcga cacggaaatg ttgaatactc
atactcttcc 3660tttttcaata ttattgaagc atttatcagg gttattgtct catgccaggg
gtgggcacac 3720atatttgata ccagcgatcc ctacacagca cataattcaa tgcgacttcc
ctctatcgca 3780catcttagac ctttattctc cctccagcac acatcgaagc tgccgagcaa
gccgttctca 3840ccagtccaag acctggcatg agcggataca tatttgaatg tatttagaaa
aataaacaaa 3900taggggttcc gcgcacattt ccccgaaaag tgccacctga aattgtaaac
gttaatattt 3960tgttaaaatt cgcgttaaat ttttgttaaa tcagctcatt ttttaaccaa
taggccgaaa 4020tcggcaaaat cccttataaa tcaaaagaat agaccgagat agggttgagt
gttgttccag 4080tttggaacaa gagtccacta ttaaagaacg tggactccaa cgtcaaaggg
cgaaaaaccg 4140tctatcaggg cgatggccca ctacgtgaac catcacccta atcaagtttt
ttggggtcga 4200ggtgccgtaa agcactaaat cggaacccta aagggagccc ccgatttaga
gcttgacggg 4260gaaagccggc gaacgtggcg agaaaggaag ggaagaaagc gaaaggagcg
ggcgctaggg 4320cgctggcaag tgtagcggtc acgctgcgcg taaccaccac acccgccgcg
cttaatgcgc 4380cgctacaggg cgcgtc
43961574470DNAArtificial SequenceDescription of Artificial
Sequence Synthetic pDONR201 polynucleotide 157gttaacgcta gcatggatct
cgggccccaa ataatgattt tattttgact gatagtgacc 60tgttcgttgc aacaaattga
tgagcaatgc ttttttataa tgccaacttt gtacaaaaaa 120gctgaacgag aaacgtaaaa
tgatataaat atcaatatat taaattagat tttgcataaa 180aaacagacta cataatactg
taaaacacaa catatccagt cactatgaat caactactta 240gatggtatta gtgacctgta
gtcgaccgac agccttccaa atgttcttcg ggtgatgctg 300ccaacttagt cgaccgacag
ccttccaaat gttcttctca aacggaatcg tcgtatccag 360cctactcgct attgtcctca
atgccgtatt aaatcataaa aagaaataag aaaaagaggt 420gcgagcctct tttttgtgtg
acaaaataaa aacatctacc tattcatata cgctagtgtc 480atagtcctga aaatcatctg
catcaagaac aatttcacaa ctcttatact tttctcttac 540aagtcgttcg gcttcatctg
gattttcagc ctctatactt actaaacgtg ataaagtttc 600tgtaatttct actgtatcga
cctgcagact ggctgtgtat aagggagcct gacatttata 660ttccccagaa catcaggtta
atggcgtttt tgatgtcatt ttcgcggtgg ctgagatcag 720ccacttcttc cccgataacg
gagaccggca cactggccat atcggtggtc atcatgcgcc 780agctttcatc cccgatatgc
accaccgggt aaagttcacg ggagacttta tctgacagca 840gacgtgcact ggccaggggg
atcaccatcc gtcgcccggg cgtgtcaata atatcactct 900gtacatccac aaacagacga
taacggctct ctcttttata ggtgtaaacc ttaaactgca 960tttcaccagt ccctgttctc
gtcagcaaaa gagccgttca tttcaataaa ccgggcgacc 1020tcagccatcc cttcctgatt
ttccgctttc cagcgttcgg cacgcagacg acgggcttca 1080ttctgcatgg ttgtgcttac
cagaccggag atattgacat catatatgcc ttgagcaact 1140gatagctgtc gctgtcaact
gtcactgtaa tacgctgctt catagcacac ctctttttga 1200catacttcgg gtatacatat
cagtatatat tcttataccg caaaaatcag cgcgcaaata 1260cgcatactgt tatctggctt
ttagtaagcc ggatccacgc gattacgccc cgccctgcca 1320ctcatcgcag tactgttgta
attcattaag cattctgccg acatggaagc catcacagac 1380ggcatgatga acctgaatcg
ccagcggcat cagcaccttg tcgccttgcg tataatattt 1440gcccatggtg aaaacggggg
cgaagaagtt gtccatattg gccacgttta aatcaaaact 1500ggtgaaactc acccagggat
tggctgagac gaaaaacata ttctcaataa accctttagg 1560gaaataggcc aggttttcac
cgtaacacgc cacatcttgc gaatatatgt gtagaaactg 1620ccggaaatcg tcgtggtatt
cactccagag cgatgaaaac gtttcagttt gctcatggaa 1680aacggtgtaa caagggtgaa
cactatccca tatcaccagc tcaccgtctt tcattgccat 1740acggaattcc ggatgagcat
tcatcaggcg ggcaagaatg tgaataaagg ccggataaaa 1800cttgtgctta tttttcttta
cggtctttaa aaaggccgta atatccagct gaacggtctg 1860gttataggta cattgagcaa
ctgactgaaa tgcctcaaaa tgttctttac gatgccattg 1920ggatatatca acggtggtat
atccagtgat ttttttctcc attttagctt ccttagctcc 1980tgaaaatctc gataactcaa
aaaatacgcc cggtagtgat cttatttcat tatggtgaaa 2040gttggaacct cttacgtgcc
gatcaacgtc tcattttcgc caaaagttgg cccagggctt 2100cccggtatca acagggacac
caggatttat ttattctgcg aagtgatctt ccgtcacagg 2160tatttattcg gcgcaaagtg
cgtcgggtga tgctgccaac ttagtcgact acaggtcact 2220aataccatct aagtagttga
ttcatagtga ctggatatgt tgtgttttac agtattatgt 2280agtctgtttt ttatgcaaaa
tctaatttaa tatattgata tttatatcat tttacgtttc 2340tcgttcagct ttcttgtaca
aagttggcat tataagaaag cattgcttat caatttgttg 2400caacgaacag gtcactatca
gtcaaaataa aatcattatt tgccatccag ctgcagctct 2460ggcccgtgtc tcaaaatctc
tgatgttaca ttgcacaaga taaaaatata tcatcatgaa 2520caataaaact gtctgcttac
ataaacagta atacaagggg tgttatgagc catattcaac 2580gggaaacgtc gaggccgcga
ttaaattcca acatggatgc tgatttatat gggtataaat 2640gggctcgcga taatgtcggg
caatcaggtg cgacaatcta tcgcttgtat gggaagcccg 2700atgcgccaga gttgtttctg
aaacatggca aaggtagcgt tgccaatgat gttacagatg 2760agatggtcag actaaactgg
ctgacggaat ttatgcctct tccgaccatc aagcatttta 2820tccgtactcc tgatgatgca
tggttactca ccactgcgat ccccggaaaa acagcattcc 2880aggtattaga agaatatcct
gattcaggtg aaaatattgt tgatgcgctg gcagtgttcc 2940tgcgccggtt gcattcgatt
cctgtttgta attgtccttt taacagcgat cgcgtatttc 3000gtctcgctca ggcgcaatca
cgaatgaata acggtttggt tgatgcgagt gattttgatg 3060acgagcgtaa tggctggcct
gttgaacaag tctggaaaga aatgcataaa cttttgccat 3120tctcaccgga ttcagtcgtc
actcatggtg atttctcact tgataacctt atttttgacg 3180aggggaaatt aataggttgt
attgatgttg gacgagtcgg aatcgcagac cgataccagg 3240atcttgccat cctatggaac
tgcctcggtg agttttctcc ttcattacag aaacggcttt 3300ttcaaaaata tggtattgat
aatcctgata tgaataaatt gcagtttcat ttgatgctcg 3360atgagttttt ctaatcagaa
ttggttaatt ggttgtaaca ctggcagagc attacgctga 3420cttgacggga cggcgcaagc
tcatgaccaa aatcccttaa cgtgagtttt cgttccactg 3480agcgtcagac cccgtagaaa
agatcaaagg atcttcttga gatccttttt ttctgcgcgt 3540aatctgctgc ttgcaaacaa
aaaaaccacc gctaccagcg gtggtttgtt tgccggatca 3600agagctacca actctttttc
cgaaggtaac tggcttcagc agagcgcaga taccaaatac 3660tgtccttcta gtgtagccgt
agttaggcca ccacttcaag aactctgtag caccgcctac 3720atacctcgct ctgctaatcc
tgttaccagt ggctgctgcc agtggcgata agtcgtgtct 3780taccgggttg gactcaagac
gatagttacc ggataaggcg cagcggtcgg gctgaacggg 3840gggttcgtgc acacagccca
gcttggagcg aacgacctac accgaactga gatacctaca 3900gcgtgagcta tgagaaagcg
ccacgcttcc cgaagggaga aaggcggaca ggtatccggt 3960aagcggcagg gtcggaacag
gagagcgcac gagggagctt ccagggggaa acgcctggta 4020tctttatagt cctgtcgggt
ttcgccacct ctgacttgag cgtcgatttt tgtgatgctc 4080gtcagggggg cggagcctat
ggaaaaacgc cagcaacgcg gcctttttac ggttcctggc 4140cttttgctgg ccttttgctc
acatgttctt tcctgcgtta tcccctgatt ctgtggataa 4200ccgtattacc gctagccagg
aagagtttgt agaaacgcaa aaaggccatc cgtcaggatg 4260gccttctgct tagtttgatg
cctggcagtt tatggcgggc gtcctgcccg ccaccctccg 4320ggccgttgct tcacaacgtt
caaatccgct cccggcggat ttgtcctact caggagagcg 4380ttcaccgaca aacaacagat
aaaacgaaag gcccagtctt ccgactgagc ctttcgtttt 4440atttgatgcc tggcagttcc
ctactctcgc 44701584204DNAArtificial
SequenceDescription of Artificial Sequence Synthetic pDONR202
polynucleotide 158cggcattgag gacaatagcg agtaggctgg atacgacgat tccgtttgag
aagaacattt 60ggaaggctgt cggtcgacta agttggcagc atcacccgaa gaacatttgg
aaggctgtcg 120gtcgactaca ggtcactaat accatctaag tagttgattc atagtgactg
gatatgttgt 180gttttacagt attatgtagt ctgtttttta tgcaaaatct aatttaatat
attgatattt 240atatcatttt acgtttctcg ttcagctttt ttgtacaaag ttggcattat
aaaaaagcat 300tgctcatcaa tttgttgcaa cgaacaggtc actatcagtc aaaataaaat
cattatttgg 360ggcccgagat ccatgctagc ggtaatacgg ttatccacag aatcagggga
taacgcagga 420aagaacatgt gagcaaaagg ccagcaaaag gccaggaacc gtaaaaaggc
cgcgttgctg 480gcgtttttcc ataggctccg cccccctgac gagcatcaca aaaatcgacg
ctcaagtcag 540aggtggcgaa acccgacagg actataaaga taccaggcgt ttccccctgg
aagctccctc 600gtgcgctctc ctgttccgac cctgccgctt accggatacc tgtccgcctt
tctcccttcg 660ggaagcgtgg cgctttctca tagctcacgc tgtaggtatc tcagttcggt
gtaggtcgtt 720cgctccaagc tgggctgtgt gcacgaaccc cccgttcagc ccgaccgctg
cgccttatcc 780ggtaactatc gtcttgagtc caacccggta agacacgact tatcgccact
ggcagcagcc 840actggtaaca ggattagcag agcgaggtat gtaggcggtg ctacagagtt
cttgaagtgg 900tggcctaact acggctacac tagaaggaca gtatttggta tctgcgctct
gctgaagcca 960gttaccttcg gaaaaagagt tggtagctct tgatccggca aacaaaccac
cgctggtagc 1020ggtggttttt ttgtttgcaa gcagcagatt acgcgcagaa aaaaaggatc
tcaagaagat 1080cctttgatct tttctacggg gtctgacgct cagtggaacg aaaactcacg
ttaagggatt 1140ttggtcatga gcttgcgccg tcccgtcaag tcagcgtaat gctctgccag
tgttacaacc 1200aattaaccaa ttctgattag aaaaactcat cgagcatcaa atgaaactgc
aatttattca 1260tatcaggatt atcaatacca tatttttgaa aaagccgttt ctgtaatgaa
ggagaaaact 1320caccgaggca gttccatagg atggcaagat cctggtatcg gtctgcgatt
ccgactcgtc 1380caacatcaat acaacctatt aatttcccct cgtcaaaaat aaggttatca
agtgagaaat 1440caccatgagt gacgactgaa tccggtgaga atggcaaaag tttatgcatt
tctttccaga 1500cttgttcaac aggccagcca ttacgctcgt catcaaaatc actcgcatca
accaaaccgt 1560tattcattcg tgattgcgcc tgagcgagac gaaatacgcg atcgctgtta
aaaggacaat 1620tacaaacagg aatcgaatgc aaccggcgca ggaacactgc cagcgcatca
acaatatttt 1680cacctgaatc aggatattct tctaatacct ggaatgctgt ttttccgggg
atcgcagtgg 1740tgagtaacca tgcatcatca ggagtacgga taaaatgctt gatggtcgga
agaggcataa 1800attccgtcag ccagtttagt ctgaccatct catctgtaac atcattggca
acgctacctt 1860tgccatgttt cagaaacaac tctggcgcat cgggcttccc atacaagcga
tagattgtcg 1920cacctgattg cccgacatta tcgcgagccc atttataccc atataaatca
gcatccatgt 1980tggaatttaa tcgcggcctc gacgtttccc gttgaatatg gctcataaca
ccccttgtat 2040tactgtttat gtaagcagac agttttattg ttcatgatga tatattttta
tcttgtgcaa 2100tgtaacatca gagattttga gacacgggcc agagctgcag ctggatggca
aataatgatt 2160ttattttgac tgatagtgac ctgttcgttg caacaaattg ataagcaatg
ctttcttata 2220atgccaactt tgtacaagaa agctgaacga gaaacgtaaa atgatataaa
tatcaatata 2280ttaaattaga ttttgcataa aaaacagact acataatact gtaaaacaca
acatatccag 2340tcactatgaa tcaactactt agatggtatt agtgacctgt agtcgactaa
gttggcagca 2400tcacccgacg cactttgcgc cgaataaata cctgtgacgg aagatcactt
cgcagaataa 2460ataaatcctg gtgtccctgt tgataccggg aagccctggg ccaacttttg
gcgaaaatga 2520gacgttgatc ggcacgtaag aggttccaac tttcaccata atgaaataag
atcactaccg 2580ggcgtatttt ttgagttatc gagattttca ggagctaagg aagctaaaat
ggagaaaaaa 2640atcactggat ataccaccgt tgatatatcc caatggcatc gtaaagaaca
ttttgaggca 2700tttcagtcag ttgctcaatg tacctataac cagaccgttc agctggatat
tacggccttt 2760ttaaagaccg taaagaaaaa taagcacaag ttttatccgg cctttattca
cattcttgcc 2820cgcctgatga atgctcatcc ggaattccgt atggcaatga aagacggtga
gctggtgata 2880tgggatagtg ttcacccttg ttacaccgtt ttccatgagc aaactgaaac
gttttcatcg 2940ctctggagtg aataccacga cgatttccgg cagtttctac acatatattc
gcaagatgtg 3000gcgtgttacg gtgaaaacct ggcctatttc cctaaagggt ttattgagaa
tatgtttttc 3060gtctcagcca atccctgggt gagtttcacc agttttgatt taaacgtggc
caatatggac 3120aacttcttcg cccccgtttt caccatgggc aaatattata cgcaaggcga
caaggtgctg 3180atgccgctgg cgattcaggt tcatcatgcc gtctgtgatg gcttccatgt
cggcagaatg 3240cttaatgaat tacaacagta ctgcgatgag tggcagggcg gggcgtaatc
gcgtggatcc 3300ggcttactaa aagccagata acagtatgcg tatttgcgcg ctgatttttg
cggtataaga 3360atatatactg atatgtatac ccgaagtatg tcaaaaagag gtgtgctatg
aagcagcgta 3420ttacagtgac agttgacagc gacagctatc agttgctcaa ggcatatatg
atgtcaatat 3480ctccggtctg gtaagcacaa ccatgcagaa tgaagcccgt cgtctgcgtg
ccgaacgctg 3540gaaagcggaa aatcaggaag ggatggctga ggtcgcccgg tttattgaaa
tgaacggctc 3600ttttgctgac gagaacaggg actggtgaaa tgcagtttaa ggtttacacc
tataaaagag 3660agagccgtta tcgtctgttt gtggatgtac agagtgatat tattgacacg
cccgggcgac 3720ggatggtgat ccccctggcc agtgcacgtc tgctgtcaga taaagtctcc
cgtgaacttt 3780acccggtggt gcatatcggg gatgaaagct ggcgcatgat gaccaccgat
atggccagtg 3840tgccggtctc cgttatcggg gaagaagtgg ctgatctcag ccaccgcgaa
aatgacatca 3900aaaacgccat taacctgatg ttctggggaa tataaatgtc aggctccctt
atacacagcc 3960agtctgcagg tcgatacagt agaaattaca gaaactttat cacgtttagt
aagtatagag 4020gctgaaaatc cagatgaagc cgaacgactt gtaagagaaa agtataagag
ttgtgaaatt 4080gttcttgatg cagatgattt tcaggactat gacactagcg tatatgaata
ggtagatgtt 4140tttattttgt cacacaaaaa agaggctcgc acctcttttt cttatttctt
tttatgattt 4200aata
42041594208DNAArtificial SequenceDescription of Artificial
Sequence Synthetic pDONR203 polynucleotide 159gcgttcggca cgcagacgac
gggcttcatt ctgcatggtt gtgcttacca gaccggagat 60attgacatca tatatgcctt
gagcaactga tagctgtcgc tgtcaactgt cactgtaata 120cgctgcttca tagcacacct
ctttttgaca tacttcgggt atacatatca gtatatattc 180ttataccgca aaaatcagcg
cgcaaatacg catactgtta tctggctttt agtaagccgg 240atccacgcgt ttacgccccg
ccctgccact catcgcagta ctgttgtaat tcattaagca 300ttctgccgac atggaagcca
tcacagacgg catgatgaac ctgaatcgcc agcggcatca 360gcaccttgtc gccttgcgta
taatatttgc ccatggtgaa aacgggggcg aagaagttgt 420ccatattggc cacgtttaaa
tcaaaactgg tgaaactcac ccagggattg gctgagacga 480aaaacatatt ctcaataaac
cctttaggga aataggccag gttttcaccg taacacgcca 540catcttgcga atatatgtgt
agaaactgcc ggaaatcgtc gtggtattca ctccagagcg 600atgaaaacgt ttcagtttgc
tcatggaaaa cggtgtaaca agggtgaaca ctatcccata 660tcaccagctc accgtctttc
attgccatac ggaattccgg atgagcattc atcaggcggg 720caagaatgtg aataaaggcc
ggataaaact tgtgcttatt tttctttacg gtctttaaaa 780aggccgtaat atccagctga
acggtctggt tataggtaca ttgagcaact gactgaaatg 840cctcaaaatg ttctttacga
tgccattggg atatatcaac ggtggtatat ccagtgattt 900ttttctccat tttagcttcc
ttagctcctg aaaatctcga taactcaaaa aatacgcccg 960gtagtgatct tatttcatta
tggtgaaagt tggaacctct tacgtgccga tcaacgtctc 1020attttcgcca aaagttggcc
cagggcttcc cggtatcaac agggacacca ggatttattt 1080attctgcgaa gtgatcttcc
gtcacaggta tttattcggc gcaaagtgcg tcgggtgatg 1140ctgccaactt agtcgactac
aggtcactaa taccatctaa gtagttgatt catagtgact 1200ggatatgttg tgttttacag
tattatgtag tctgtttttt atgcaaaatc taatttaata 1260tattgatatt tatatcattt
tacgtttctc gttcagcttt cttgtacaaa gttggcatta 1320taagaaagca ttgcttatca
atttgttgca acgaacaggt cactatcagt caaaataaaa 1380tcattatttg ccatccagct
agcggtaata cggttatcca cagaatcagg ggataacgca 1440ggaaagaaca tgtgagcaaa
aggccagcaa aaggccagga accgtaaaaa ggccgcgttg 1500ctggcgtttt tccataggct
ccgcccccct gacgagcatc acaaaaatcg acgctcaagt 1560cagaggtggc gaaacccgac
aggactataa agataccagg cgtttccccc tggaagctcc 1620ctcgtgcgct ctcctgttcc
gaccctgccg cttaccggat acctgtccgc ctttctccct 1680tcgggaagcg tggcgctttc
tcatagctca cgctgtaggt atctcagttc ggtgtaggtc 1740gttcgctcca agctgggctg
tgtgcacgaa ccccccgttc agcccgaccg ctgcgcctta 1800tccggtaact atcgtcttga
gtccaacccg gtaagacacg acttatcgcc actggcagca 1860gccactggta acaggattag
cagagcgagg tatgtaggcg gtgctacaga gttcttgaag 1920tggtggccta actacggcta
cactagaaga acagtatttg gtatctgcgc tctgctgaag 1980ccagttacct tcggaaaaag
agttggtagc tcttgatccg gcaaacaaac caccgctggt 2040agcggtggtt tttttgtttg
caagcagcag attacgcgca gaaaaaaagg atctcaagaa 2100gatcctttga tcttttctac
ggggtctgac gctcagtgga acgaaaactc acgttaaggg 2160attttggtca tgagcttgcg
ccgtcccgtc aagtcagcgt aatgctctgc cagtgttaca 2220accaattaac caattctgat
tagaaaaact catcgagcat caaatgaaac tgcaatttat 2280tcatatcagg attatcaata
ccatattttt gaaaaagccg tttctgtaat gaaggagaaa 2340actcaccgag gcagttccat
aggatggcaa gatcctggta tcggtctgcg attccgactc 2400gtccaacatc aatacaacct
attaatttcc cctcgtcaaa aataaggtta tcaagtgaga 2460aatcaccatg agtgacgact
gaatccggtg agaatggcaa aagtttatgc atttctttcc 2520agacttgttc aacaggccag
ccattacgct cgtcatcaaa atcactcgca tcaaccaaac 2580cgttattcat tcgtgattgc
gcctgagcga gacgaaatac gcgatcgctg ttaaaaggac 2640aattacaaac aggaatcgaa
tgcaaccggc gcaggaacac tgccagcgca tcaacaatat 2700tttcacctga atcaggatat
tcttctaata cctggaatgc tgtttttccg gggatcgcag 2760tggtgagtaa ccatgcatca
tcaggagtac ggataaaatg cttgatggtc ggaagaggca 2820taaattccgt cagccagttt
agtctgacca tctcatctgt aacatcattg gcaacgctac 2880ctttgccatg tttcagaaac
aactctggcg catcgggctt cccatacaag cgatagattg 2940tcgcacctga ttgcccgaca
ttatcgcgag cccatttata cccatataaa tcagcatcca 3000tgttggaatt taatcgcggc
ctcgacgttt cccgttgaat atggctcata acaccccttg 3060tattactgtt tatgtaagca
gacagtttta ttgttcatga tgatatattt ttatcttgtg 3120caatgtaaca tcagagattt
tgagacacgg gccagagctg cagctagcat ggatctcggg 3180ccccaaataa tgattttatt
ttgactgata gtgacctgtt cgttgcaaca aattgatgag 3240caatgctttt ttataatgcc
aactttgtac aaaaaagctg aacgagaaac gtaaaatgat 3300ataaatatca atatattaaa
ttagattttg cataaaaaac agactacata atactgtaaa 3360acacaacata tccagtcact
atgaatcaac tacttagatg gtattagtga cctgtagtcg 3420accgacagcc ttccaaatgt
tcttcgggtg atgctgccaa cttagtcgac cgacagcctt 3480ccaaatgttc ttctcaaacg
gaatcgtcgt atccagccta ctcgctattg tcctcaatgc 3540cgtattaaat cataaaaaga
aataagaaaa agaggtgcga gcctcttttt tgtgtgacaa 3600aataaaaaca tctacctatt
catatacgct agtgtcatag tcctgaaaat catctgcatc 3660aagaacaatt tcacaactct
tatacttttc tcttacaagt cgttcggctt catctggatt 3720ttcagcctct atacttacta
aacgtgataa agtttctgta atttctactg tatcgacctg 3780cagactggct gtgtataagg
gagcctgaca tttatattcc ccagaacatc aggttaatgg 3840cgtttttgat gtcattttcg
cggtggctga gatcagccac ttcttccccg ataacggaga 3900ccggcacact ggccatatcg
gtggtcatca tgcgccagct ttcatccccg atatgcacca 3960ccgggtaaag ttcacgggag
actttatctg acagcagacg tgcactggcc agggggatca 4020ccatccgtcg cccgggcgtg
tcaataatat cactctgtac atccacaaac agacgataac 4080ggctctctct tttataggtg
taaaccttaa actgcatttc accagtccct gttctcgtca 4140gcaaaagagc cgttcatttc
aataaaccgg gcgacctcag ccatcccttc ctgattttcc 4200gctttcca
42081604165DNAArtificial
SequenceDescription of Artificial Sequence Synthetic pDONR204
polynucleotide 160cggcattgag gacaatagcg agtaggctgg atacgacgat tccgtttgag
aagaacattt 60ggaaggctgt cggtcgacta caggtcacta ataccatcta agtagttgaa
tcatagtgac 120tggatatgtt gtgttttaca gtattatgta gtctgttttt tatgcaaaat
ctaatttaat 180atattgatat ttatatcatt ttacgtttct cgttcagctt ttttgtacaa
agttggcatt 240ataaaaaagc attgcttatc aatttgttgc aacgaacagg tcactatcag
tcaaaataaa 300atcattattt ggggcccgag atccatgcta gctgcagtgc gcagggcccg
tgtctcaaaa 360tctctgatgt tacattgcac aagataaaaa tatatcatca tgaacaataa
aactgtctgc 420ttacataaac agtaatacaa ggggtgttat gagccatatt caacgggaaa
cgtcttgctg 480gaggccgcga ttaaattcca acatggatgc tgatttatat gggtataaat
gggctcgcga 540taatgtcggg caatcaggtg cgacaatctt tcgattgtat gggaagcccg
atgcgccaga 600gttgtttctg aaacatggca aaggtagcgt tgccaatgat gttacagatg
agatggtcag 660actaaactgg ctgacggaat ttatgcctct tccgaccatc aagcatttta
tccgtactcc 720tgatgatgca tggttactca ccactgcgat ccgcgggaaa acagcattcc
aggtattaga 780agaatatcct gattcaggtg aaaatattgt tgatgcgctg gcagtgttcc
tgcgccggtt 840gcattcgatt cctgtttgta attgtccttt taacagcgat cgcgtatttc
gtctcgctca 900ggcgcaatca cgaatgaata acggtttggt tgatgcgagt gattttgatg
acgagcgtaa 960tggctggcct gttgaacaag tctggaaaga aatgcatacg cttttgccat
tctcaccgga 1020ttcagtcgtc actcatggtg atttctcact tgataacctt atttttgacg
aggggaaatt 1080aataggttgt attgatgttg gacgagtcgg aatcgcagac cgataccagg
atcttgccat 1140cctatggaac tgcctcggtg agttttctcc ttcattacag aaacggcttt
ttcaaaaata 1200tggtattgat aatcctgata tgaataaatt gcagtttcat ttgatgctcg
atgagttttt 1260ctaatcagaa ttggttaatt ggttgtaaca ctggcagagc attacgctga
cttgacggga 1320cggcgncatg accaaaatcc cttaacgtga gttttcgttc cactgagcgt
cagaccccgt 1380agaaaagatc aaaggatctt cttgagatcc tttttttctg cgcgtaatct
gctgcttgca 1440aacaaaaaaa ccaccgctac cagcggtggt ttgtttgccg gatcaagagc
taccaactct 1500ttttccgaag gtaactggct tcagcagagc gcagatacca aatactgtcc
ttctagtgta 1560gccgtagtta ggccaccact tcaagaactc tgtagcaccg cctacatacc
tcgctctgct 1620aatcctgtta ccagtggctg ctgccagtgg cgataagtcg tgtcttaccg
ggttggactc 1680aagacgatag ttaccggata aggcgcagcg gtcgggctga acggggggtt
cgtgcacaca 1740gcccagcttg gagcgaacga cctacaccga actgagatac ctacagcgtg
agctatgaga 1800aagcgccacg cttcccgaag ggagaaaggc ggacaggtat ccggtaagcg
gcagggtcgg 1860aacaggagag cgcacgaggg agcttccagg gggaaacgcc tggtatcttt
atagtcctgt 1920cgggtttcgc cacctctgac ttgagcgtcg atttttgtga tgctcgtcag
gggggcggag 1980cctatggaaa aacgccagca acgcggcctt tttacggttc ctggcctttt
gctggccttt 2040tgctcacatg ttctttcctg cgttatcccc tgattctgtg gataaccgta
ttaccgctag 2100ctggatcggc aaataatgat tttattttga ctgatagtga cctgttcgtt
gcaacaaatt 2160gataagcaat gcttttttat aatgccaact ttgtacaaga aagctgaacg
agaaacgtaa 2220aatgatataa atatcaatat attaaattag attttgcata aaaaacagac
tacataatac 2280tgtaaaacac aacatatcca gtcactatga ttcaactact tagatggtat
tagtgacctg 2340tagtcgacta agttggcagc atcacccgac gcactttgcg ccgaataaat
acctgtgacg 2400gaagatcact tcgcagaata aataaatcct ggtgtccctg ttgataccgg
gaagccctgg 2460gccaactttt ggcgaaaatg agacgttgat cggcacattt cacaactctt
atacttttct 2520cttacaagtc gttcggcttc atctggattt tcagcctcta tacttactaa
acgtgataaa 2580gtttctgtaa tttctactgt atcgacctgc agactggctg tgtataacgg
agcctgacat 2640ttatattccc cagaacatca ggttaatggc gtttttgatg tcattttcgc
ggtggctgag 2700atcagccact tcttccccga taacggagac cggcacactg gccatatcgg
tggtcatcat 2760gcgccagctt tcatccccga tatgcaccac cgggtaaagt tcacgggaga
ctttatctga 2820cagcagacgt gcactggcca gggggatcac catccgtcgc ccgggcgtgt
caataatatc 2880actctgtaca tccacaaaca gacgataacg gctctctctt ttataggtgt
aaaccttaaa 2940ctgcatttca ccagtccctg ttctcgtcag caaaagagcc gttcatttca
ataaaccggg 3000cgacctcagc catcccttcc tgattttccg ctttccagcg ttcggcacgc
agacgacggg 3060cttcattctg catggttgtg cttaccagac cggagatatt gacatcatat
atgccttgag 3120caactgatag ctgtcgctgt caactgtcac tgtaatacgc tgcttcatag
cacacctctt 3180tttgacatac ttcgggtata catatcagta tatattctta taccgcaaaa
atcagcgcgc 3240aaatacgcat actgttatct ggcttttagt aagccggatc cacgcgttta
cgccccgccc 3300tgccactcat cgcagtactg ttgtaattca ttaagcattc tgccgacatg
gaagccatca 3360cagacggcat gatgaacctg aatcgccagc ggcatcagca ccttgtcgcc
ttgcgtataa 3420tatttgccca tggtgaaaac gggggcgaag aagttgtcca tattggccac
gtttaaatca 3480aaactggtga aactcaccca gggattggct gagacgaaaa acatattctc
aataaaccct 3540ttagggaaat aggccaggtt ttcaccgtaa cacgccacat cttgcgaata
tatgtgtaga 3600aactgccgga aatcgtcgtg gtattcactc cagagcgatg aaaacgtttc
agtttgctca 3660tggaaaacgg tgtaacaagg gtgaacacta tcccatatca ccagctcacc
gtctttcatt 3720gccatacgga attccggatg agcattcatc aggcgggcaa gaatgtgaat
aaaggccgga 3780taaaacttgt gcttattttt ctttacggtc tttaaaaagg ccgtaatatc
cagctgaacg 3840gtctggttat aggtacattg agcaactgac tgaaatgcct caaaatgttc
tttacgatgc 3900cattgggata tatcaacggt ggtatatcca gtgatttttt tctccatttt
agcttcctta 3960gctcctgaaa atctcgataa ctcaaaaaat acgcccggta gtgatcttat
ttcattatgg 4020tgaaagttgg aacctcttac tgttcttgat gcagatgatt ttcaggacta
tgacactagc 4080atatatgaat aggtagatgt ttttattttg tcacacaaaa aagaggctcg
cacctctttt 4140tcttatttct ttttatgatt taata
41651614939DNAArtificial SequenceDescription of Artificial
Sequence Synthetic pDONR205 polynucleotide 161ggcatcagca ccttgtcgcc
ttgcgtataa tatttgccca tggtgaaaac gggggcgaag 60aagttgtcca tattggccac
gtttaaatca aaactggtga aactcaccca gggattggct 120gagacgaaaa acatattctc
aataaaccct ttagggaaat aggccaggtt ttcaccgtaa 180cacgccacat cttgcgaata
tatgtgtaga aactgccgga aatcgtcgtg gtattcactc 240cagagcgatg aaaacgtttc
agtttgctca tggaaaacgg tgtaacaagg gtgaacacta 300tcccatatca ccagctcacc
gtctttcatt gccatacgga attccggatg agcattcatc 360aggcgggcaa gaatgtgaat
aaaggccgga taaaacttgt gcttattttt ctttacggtc 420tttaaaaagg ccgtaatatc
cagctgaacg gtctggttat aggtacattg agcaactgac 480tgaaatgcct caaaatgttc
tttacgatgc cattgggata tatcaacggt ggtatatcca 540gtgatttttt tctccatttt
agcttcctta gctcctgaaa atctcgataa ctcaaaaaat 600acgcccggta gtgatcttat
ttcattatgg tgaaagttgg aacctcttac gtgccgatca 660acgtctcatt ttcgccaaaa
gttggcccag ggcttcccgg tatcaacagg gacaccagga 720tttatttatt ctgcgaagtg
atcttccgtc acaggtattt attcggcgca aagtgcgtcg 780ggtgatgctg ccaacttagt
cgactacagg tcactaatac catctaagta gttgattcat 840agtgactgga tatgttgtgt
tttacagtat tatgtagtct gttttttatg caaaatctaa 900tttaatatat tgatatttat
atcattttac gtttctcgtt cagctttctt gtacaaagtt 960ggcattataa gaaagcattg
cttatcaatt tgttgcaacg aacaggtcac tatcagtcaa 1020aataaaatca ttatttgcca
tccagctgca gctctggccc gtgtctcaaa atctctgatg 1080ttacattgca caagataaaa
atatatcatc atgaattctc atgtttgaca gcttatcatc 1140gataagcttt aatgcggtag
tttatcacag ttaaattgct aacgcagtca ggcaccgtgt 1200atgaaatcta acaatgcgct
catcgtcatc ctcggcaccg tcaccctgga tgctgtaggc 1260ataggcttgg ttatgccggt
actgccgggc ctcttgcggg atatcgtcca ttccgacagc 1320atcgccagtc actatggcgt
gctgctagcg ctatatgcgt tgatgcaatt tctatgcgca 1380cccgttctcg gagcactgtc
cgaccgcttt ggccgccgcc cagtcctgct cgcttcgcta 1440cttggagcca ctatcgacta
cgcgatcatg gcgaccacac ccgtcctgtg gatcctctac 1500gccggacgca tcgtggccgg
catcaccggc gccacaggtg cggttgctgg cgcctatatc 1560gccgacatca ccgatgggga
agatcgggct cgccacttcg ggctcatgag cgcttgtttc 1620ggcgtgggta tggtggcagg
ccccgtggcc gggggactgt tgggcgccat ctccttgcat 1680gcaccattcc ttgcggcggc
ggtgctcaac ggcctcaacc tactactggg ctgcttccta 1740atgcaggagt cgcataaggg
agagcgtcga ccgatgccct tgagagcctt caacccagtc 1800agctccttcc ggtgggcgcg
gggcatgact atcgtcgccg cacttatgac tgtcttcttt 1860atcatgcaac tcgtaggaca
ggtgccggca gcgctctggg tcattttcgg cgaggaccgc 1920tttcgctgga gcgcgacgat
gatcggcctg tcgcttgcgg tattcggaat cttgcacgcc 1980ctcgctcaag ccttcgtcac
tggtcccgcc accaaacgtt tcggcgagaa gcaggccatt 2040atcgccggca tggcggccga
cgcgctgggc tacgtcttgc tggcgttcgc gacgcgaggc 2100tggatggcct tccccattat
gattcttctc gcttccggcg gcatcgggat gcccgcgttg 2160caggccatgc tgtccaggca
ggtagatgac gaccatcagg gacagcttca aggatcgctc 2220gcggctctta ccagcctaac
ttcgatcatt ggaccgctga tcgtcacggc gatttatgcc 2280gcctcggcga gcacatggaa
cgggttggca tggattgtag gcgccgccct ataccttgtc 2340tgcctccccg cgttgcgtcg
cggtgcatgg agccgggcca cctcgacctg aatggaagcc 2400ggcggcacct cgctaacgga
ttcaccactc caagaattgg agccaatcaa ttcttgcgga 2460gaactgtgaa tgcgcaaacc
aacccttggc agaacatatc catcgcatga ccaaaatccc 2520ttaacgtgag ttttcgttcc
actgagcgtc agaccccgta gaaaagatca aaggatcttc 2580ttgagatcct ttttttctgc
gcgtaatctg ctgcttgcaa acaaaaaaac caccgctacc 2640agcggtggtt tgtttgccgg
atcaagagct accaactctt tttccgaagg taactggctt 2700cagcagagcg cagataccaa
atactgtcct tctagtgtag ccgtagttag gccaccactt 2760caagaactct gtagcaccgc
ctacatacct cgctctgcta atcctgttac cagtggctgc 2820tgccagtggc gataagtcgt
gtcttaccgg gttggactca agacgatagt taccggataa 2880ggcgcagcgg tcgggctgaa
cggggggttc gtgcacacag cccagcttgg agcgaacgac 2940ctacaccgaa ctgagatacc
tacagcgtga gctatgagaa agcgccacgc ttcccgaagg 3000gagaaaggcg gacaggtatc
cggtaagcgg cagggtcgga acaggagagc gcacgaggga 3060gcttccaggg ggaaacgcct
ggtatcttta tagtcctgtc gggtttcgcc acctctgact 3120tgagcgtcga tttttgtgat
gctcgtcagg ggggcggagc ctatggaaaa acgccagcaa 3180cgcggccttt ttacggttcc
tggccttttg ctggcctttt gctcacatgt tctttcctgc 3240gttatcccct gattctgtgg
ataaccgtat taccgctagc caggaagagt ttgtagaaac 3300gcaaaaaggc catccgtcag
gatggccttc tgcttagttt gatgcctggc agtttatggc 3360gggcgtcctg cccgccaccc
tccgggccgt tgcttcacaa cgttcaaatc cgctcccggc 3420ggatttgtcc tactcaggag
agcgttcacc gacaaacaac agataaaacg aaaggcccag 3480tcttccgact gagcctttcg
ttttatttga tgcctggcag ttccctactc tcgcgttaac 3540gctagcatgg atctcgggcc
ccaaataatg attttatttt gactgatagt gacctgttcg 3600ttgcaacaaa ttgatgagca
atgctttttt ataatgccaa ctttgtacaa aaaagctgaa 3660cgagaaacgt aaaatgatat
aaatatcaat atattaaatt agattttgca taaaaaacag 3720actacataat actgtaaaac
acaacatatc cagtcactat gaatcaacta cttagatggt 3780attagtgacc tgtagtcgac
cgacagcctt ccaaatgttc ttcgggtgat gctgccaact 3840tagtcgaccg acagccttcc
aaatgttctt ctcaaacgga atcgtcgtat ccagcctact 3900cgctattgtc ctcaatgccg
tattaaatca taaaaagaaa taagaaaaag aggtgcgagc 3960ctcttttttg tgtgacaaaa
taaaaacatc tacctattca tatacgctag tgtcatagtc 4020ctgaaaatca tctgcatcaa
gaacaatttc acaactctta tacttttctc ttacaagtcg 4080ttcggcttca tctggatttt
cagcctctat acttactaaa cgtgataaag tttctgtaat 4140ttctactgta tcgacctgca
gactggctgt gtataaggga gcctgacatt tatattcccc 4200agaacatcag gttaatggcg
tttttgatgt cattttcgcg gtggctgaga tcagccactt 4260cttccccgat aacggagacc
ggcacactgg ccatatcggt ggtcatcatg cgccagcttt 4320catccccgat atgcaccacc
gggtaaagtt cacgggagac tttatctgac agcagacgtg 4380cactggccag ggggatcacc
atccgtcgcc cgggcgtgtc aataatatca ctctgtacat 4440ccacaaacag acgataacgg
ctctctcttt tataggtgta aaccttaaac tgcatttcac 4500cagtccctgt tctcgtcagc
aaaagagccg ttcatttcaa taaaccgggc gacctcagcc 4560atcccttcct gattttccgc
tttccagcgt tcggcacgca gacgacgggc ttcattctgc 4620atggttgtgc ttaccagacc
ggagatattg acatcatata tgccttgagc aactgatagc 4680tgtcgctgtc aactgtcact
gtaatacgct gcttcatagc acacctcttt ttgacatact 4740tcgggtatac atatcagtat
atattcttat accgcaaaaa tcagcgcgca aatacgcata 4800ctgttatctg gcttttagta
agccggatcc acgcgattac gccccgccct gccactcatc 4860gcagtactgt tgtaattcat
taagcattct gccgacatgg aagccatcac agacggcatg 4920atgaacctga atcgccagc
49391625156DNAArtificial
SequenceDescription of Artificial Sequence Synthetic pDONR206
polynucleotide 162cggcattgag gacaatagcg agtaggctgg atacgacgat tccgtttgag
aagaacattt 60ggaaggctgt cggtcgacta caggtcacta ataccatcta agtagttgaa
tcatagtgac 120tggatatgtt gtgttttaca gtattatgta gtctgttttt tatgcaaaat
ctaatttaat 180atattgatat ttatatcatt ttacgtttct cgttcagctt ttttgtacaa
agttggcatt 240ataaaaaagc attgcttatc aatttgttgc aacgaacagg tcactatcag
tcaaaataaa 300atcattattt ggggcccgag atccatgcta gcggtaatac ggttatccac
agaatcaggg 360gataacgcag gaaagaacat gtgagcaaaa ggccagcaaa aggccaggaa
ccgtaaaaag 420gccgcgttgc tggcgttttt ccataggctc cgcccccctg acgagcatca
caaaaatcga 480cgctcaagtc agaggtggcg aaacccgaca ggactataaa gataccaggc
gtttccccct 540ggaagctccc tcgtgcgctc tcctgttccg accctgccgc ttaccggata
cctgtccgcc 600tttctccctt cgggaagcgt ggcgctttct catagctcac gctgtaggta
tctcagttcg 660gtgtaggtcg ttcgctccaa gctgggctgt gtgcacgaac cccccgttca
gcccgaccgc 720tgcgccttat ccggtaacta tcgtcttgag tccaacccgg taagacacga
cttatcgcca 780ctggcagcag ccactggtaa caggattagc agagcgaggt atgtaggcgg
tgctacagag 840ttcttgaagt ggtggcctaa ctacggctac actagaagga cagtatttgg
tatctgcgct 900ctgctgaagc cagttacctt cggaaaaaga gttggtagct cttgatccgg
caaacaaacc 960accgctggta gcggtggttt ttttgtttgc aagcagcaga ttacgcgcag
aaaaaaagga 1020tctcaagaag atcctttgat cttttctacg gggtctgacg ctcagtggaa
cgaaaactca 1080cgttaaggga ttttggtcat gncgccgtcc cgtcaagtca gcgtaatgct
ctgccagtgt 1140tacaaccaat taaccaattc tgattagaaa aactcatcga gcatcaaatg
aaactgcaat 1200ttattcatat caggattatc aataccatat ttttgaaaaa gccgtttctg
taatgaagga 1260gaaaactcac cgaggcagtt ccataggatg gcaagatcct ggtatcggtc
tgcgattccg 1320actcgtccaa catcaataca acctattagc cgaggtcttc cgatctcctg
aagccagggc 1380agatccgtgc acagcacctt gccgtagaag aacagcaagg ccgccaatgc
ctgacgatgc 1440gtggagaccg aaaccttgcg ctcgttcgcc agccaggaca gaaatgcctc
gacttcgctg 1500ctgcccaagg ttgccgggtg acgcacaccg tggaaacgga tgaaggcacg
aacccagttg 1560acataagcct gttcggttcg taaactgtaa tgcaagtagc gtatgcgctc
acgcaactgg 1620tccagaacct tgaccgaacg cagcggtggt aacggcgcag tggcggtttt
catggcttgt 1680tatgactgtt tttttgtaca gtctatgcct cgggcatcca agcagcaagc
gcgttacgcc 1740gtgggtcgat gtttgatgtt atggagcagc aacgatgtta cgcagcagca
acgatgttac 1800gcagcagggc agtcgcccta aaacaaagtt aggtggctca agtatgggca
tcattcgcac 1860atgtaggctc ggccctgacc aagtcaaatc catgcgggct gctcttgatc
ttttcggtcg 1920tgagttcgga gacgtagcca cctactccca acatcagccg gactccgatt
acctcgggaa 1980cttgctccgt agtaagacat tcatcgcgct tgctgccttc gaccaagaag
cggttgttgg 2040cgctctcgcg gcttacgttc tgcccaggtt tgagcagccg cgtagtgaga
tctatatcta 2100tgatctcgca gtctccggcg agcaccggag gcagggcatt gccaccgcgc
tcatcaatct 2160cctcaagcat gaggccaacg cgcttggtgc ttatgtgatc tacgtgcaag
cagattacgg 2220tgacgatccc gcagtggctc tctatacaaa gttgggcata cgggaagaag
tgatgcactt 2280tgatatcgac ccaagtaccg ccacctaaca attcgttcaa gccgagatcg
gcttcccggc 2340ctaatttccc ctcgtcaaaa ataaggttat caagtgagaa atcaccatga
gtgacgactg 2400aatccggtga gaatggcaaa agcgtatgca tttctttcca gacttgttca
acaggccagc 2460cattacgctc gtcatcaaaa tcactcgcat caaccaaacc gttattcatt
cgtgattgcg 2520cctgagcgag acgaaatacg cgatcgctgt taaaaggaca attacaaaca
ggaatcgaat 2580gcaaccggcg caggaacact gccagcgcat caacaatatt ttcacctgaa
tcaggatatt 2640cttctaatac ctggaatgct gttttcccgc ggatcgcagt ggtgagtaac
catgcatcat 2700caggagtacg gataaaatgc ttgatggtcg gaagaggcat aaattccgtc
agccagttta 2760gtctgaccat ctcatctgta acatcattgg caacgctacc tttgccatgt
ttcagaaaca 2820actctggcgc atcgggcttc ccatacaatc gaaagattgt cgcacctgat
tgcccgacat 2880tatcgcgagc ccatttatac ccatataaat cagcatccat gttggaattt
aatcgcggcc 2940tccagcaaga cgtttcccgt tgaatatggc tcataacacc ccttgtatta
ctgtttatgt 3000aagcagacag ttttattgtt catgatgata tatttttatc ttgtgcaatg
taacatcaga 3060gattttgaga cacgggcccn gcgcactgca gctggatcgg caaataatga
ttttattttg 3120actgatagtg acctgttcgt tgcaacaaat tgataagcaa tgctttttta
taatgccaac 3180tttgtacaag aaagctgaac gagaaacgta aaatgatata aatatcaata
tattaaatta 3240gattttgcat aaaaaacaga ctacataata ctgtaaaaca caacatatcc
agtcactatg 3300attcaactac ttagatggta ttagtgacct gtagtcgact aagttggcag
catcacccga 3360cgcactttgc gccgaataaa tacctgtgac ggaagatcac ttcgcagaat
aaataaatcc 3420tggtgtccct gttgataccg ggaagccctg ggccaacttt tggcgaaaat
gagacgttga 3480tcggcacgta agaggttcca actttcacca taatgaaata agatcactac
cgggcgtatt 3540ttttgagtta tcgagatttt caggagctaa ggaagctaaa atggagaaaa
aaatcactgg 3600atataccacc gttgatatat cccaatggca tcgtaaagaa cattttgagg
catttcagtc 3660agttgctcaa tgtacctata accagaccgt tcagctggat attacggcct
ttttaaagac 3720cgtaaagaaa aataagcaca agttttatcc ggcctttatt cacattcttg
cccgcctgat 3780gaatgctcat ccggaattcc gtatggcaat gaaagacggt gagctggtga
tatgggatag 3840tgttcaccct tgttacaccg ttttccatga gcaaactgaa acgttttcat
cgctctggag 3900tgaataccac gacgatttcc ggcagtttct acacatatat tcgcaagatg
tggcgtgtta 3960cggtgaaaac ctggcctatt tccctaaagg gtttattgag aatatgtttt
tcgtctcagc 4020caatccctgg gtgagtttca ccagttttga tttaaacgtg gccaatatgg
acaacttctt 4080cgcccccgtt ttcaccatgg gcaaatatta tacgcaaggc gacaaggtgc
tgatgccgct 4140ggcgattcag gttcatcatg ccgtctgtga tggcttccat gtcggcagaa
tgcttaatga 4200attacaacag tactgcgatg agtggcaggg cggggcgtaa acgcgtggat
ccggcttact 4260aaaagccaga taacagtatg cgtatttgcg cgctgatttt tgcggtataa
gaatatatac 4320tgatatgtat acccgaagta tgtcaaaaag aggtgtgcta tgaagcagcg
tattacagtg 4380acagttgaca gcgacagcta tcagttgctc aaggcatata tgatgtcaat
atctccggtc 4440tggtaagcac aaccatgcag aatgaagccc gtcgtctgcg tgccgaacgc
tggaaagcgg 4500aaaatcagga agggatggct gaggtcgccc ggtttattga aatgaacggc
tcttttgctg 4560acgagaacag ggactggtga aatgcagttt aaggtttaca cctataaaag
agagagccgt 4620tatcgtctgt ttgtggatgt acagagtgat attattgaca cgcccgggcg
acggatggtg 4680atccccctgg ccagtgcacg tctgctgtca gataaagtct cccgtgaact
ttacccggtg 4740gtgcatatcg gggatgaaag ctggcgcatg atgaccaccg atatggccag
tgtgccggtc 4800tccgttatcg gggaagaagt ggctgatctc agccaccgcg aaaatgacat
caaaaacgcc 4860attaacctga tgttctgggg aatataaatg tcaggctccg ttatacacag
ccagtctgca 4920ggtcgataca gtagaaatta cagaaacttt atcacgttta gtaagtatag
aggctgaaaa 4980tccagatgaa gccgaacgac ttgtaagaga aaagtataag agttgtgaaa
ttgttcttga 5040tgcagatgat tttcaggact atgacactag catatatgaa taggtagatg
tttttatttt 5100gtcacacaaa aaagaggctc gcacctcttt ttcttatttc tttttatgat
ttaata 515616321DNAArtificial SequenceDescription of Artificial
Sequence Synthetic attR1 Reading Frame A oligonucleotide
163atcacaagtt tgtacaaaaa a
2116422DNAArtificial SequenceDescription of Artificial Sequence Synthetic
attR1 Reading Frame B oligonucleotide 164atcaacaagt ttgtacaaaa aa
2216523DNAArtificial
SequenceDescription of Artificial Sequence Synthetic attR1 Reading
Frame C oligonucleotide 165atcaaacaag tttgtacaaa aaa
2316621DNAArtificial SequenceDescription of
Artificial Sequence Synthetic attR2 Reading Frame A oligonucleotide
166tttcttgtac aaagtggtga t
2116722DNAArtificial SequenceDescription of Artificial Sequence Synthetic
attR2 Reading Frame B oligonucleotide 167tttcttgtac aaagtggttg at
2216823DNAArtificial
SequenceDescription of Artificial Sequence Synthetic attR2 Reading
Frame C oligonucleotide 168tttcttgtac aaagtggttc gat
2316923DNAArtificial SequenceDescription of
Artificial Sequence Synthetic attR1 Reading Frame C (Alternative B)
oligonucleotide 169atcaaacaag tttgtacaaa aaa
2317023DNAArtificial SequenceDescription of Artificial
Sequence Synthetic attR2 Reading Frame C (Alternative B)
oligonucleotide 170tttcttgtac aaagtggttt gat
2317130DNAArtificial SequenceDescription of Artificial
Sequence Synthetic attR1 Reading Frame A Cassette oligonucleotide
171nnnnnnatca caagtttgta caaaaaagct
3017233DNAArtificial SequenceDescription of Artificial Sequence Synthetic
attR1 Reading Frame B Cassette oligonucleotide 172nnnnnnnnat
caacaagttt gtacaaaaaa gct
3317333DNAArtificial SequenceDescription of Artificial Sequence Synthetic
attR1 Reading Frame C Cassette oligonucleotide 173nnnnnnnatc
aaacaagttt gtacaaaaaa gct
331744554DNAArtificial SequenceDescription of Artificial Sequence
Synthetic prfC Parent III polynucleotide 174gcgcccaata cgcaaaccgc
ctctccccgc gcgttggccg attcattaat gcagctggca 60cgacaggttt cccgactgga
aagcgggcag tgagcgcaac gcaattaatg tgagttagct 120cactcattag gcaccccagg
ctttacactt tatgcttccg gctcgtatgt tgtgtggaat 180tgtgagcgga taacaatttc
acacaggaaa cagctatgac catgattacg ccaagcttgc 240atgcctgcag gtcgactcta
gaggatcccc gggtaccgat atcaaacaag tttgtacaaa 300aaagctgaac gagaaacgta
aaatgatata aatatcaata tattaaatta gattttgcat 360aaaaaacaga ctacataata
ctgtaaaaca caacatatcc agtcactatg gcggccgcta 420agttggcagc atcacccgac
gcactttgcg ccgaataaat acctgtgacg gaagatcact 480tcgcagaata aataaatcct
ggtgtccctg ttgataccgg gaagccctgg gccaactttt 540ggcgaaaatg agacgttgat
cggcacgtaa gaggttccaa ctttcaccat aatgaaataa 600gatcactacc gggcgtattt
tttgagttat cgagattttc aggagctaag gaagctaaaa 660tggagaaaaa aatcactgga
tataccaccg ttgatatatc ccaatggcat cgtaaagaac 720attttgaggc atttcagtca
gttgctcaat gtacctataa ccagaccgtt cagctggata 780ttacggcctt tttaaagacc
gtaaagaaaa ataagcacaa gttttatccg gcctttattc 840acattcttgc ccgcctgatg
aatgctcatc cggaattccg tatggcaatg aaagacggtg 900agctggtgat atgggatagt
gttcaccctt gttacaccgt tttccatgag caaactgaaa 960cgttttcatc gctctggagt
gaataccacg acgatttccg gcagtttcta cacatatatt 1020cgcaagatgt ggcgtgttac
ggtgaaaacc tggcctattt ccctaaaggg tttattgaga 1080atatgttttt cgtctcagcc
aatccctggg tgagtttcac cagttttgat ttaaacgtgg 1140ccaatatgga caacttcttc
gcccccgttt tcaccatggg caaatattat acgcaaggcg 1200acaaggtgct gatgccgctg
gcgattcagg ttcatcatgc cgtctgtgat ggcttccatg 1260tcggcagaat gcttaatgaa
ttacaacagt actgcgatga gtggcagggc ggggcgtaat 1320ctagaggatc cggcttacta
aaagccagat aacagtatgc gtatttgcgc gctgattttt 1380gcggtataag aatatatact
gatatgtata cccgaagtat gtcaaaaaga ggtgtgctat 1440gaagcagcgt attacagtga
cagttgacag cgacagctat cagttgctca aggcatatat 1500gatgtcaata tctccggtct
ggtaagcaca accatgcaga atgaagcccg tcgtctgcgt 1560gccgaacgct ggaaagcgga
aaatcaggaa gggatggctg aggtcgcccg gtttattgaa 1620atgaacggct cttttgctga
cgagaacagg gactggtgaa atgcagttta aggtttacac 1680ctataaaaga gagagccgtt
atcgtctgtt tgtggatgta cagagtgata ttattgacac 1740gcccgggcga cggatggtga
tccccctggc cagtgcacgt ctgctgtcag ataaagtctc 1800ccgtgaactt tacccggtgg
tgcatatcgg ggatgaaagc tggcgcatga tgaccaccga 1860tatggccagt gtgccggtct
ccgttatcgg ggaagaagtg gctgatctca gccaccgcga 1920aaatgacatc aaaaacgcca
ttaacctgat gttctgggga atataaatgt caggctccgt 1980tatacacagc cagtctgcag
gtcgaccata gtgactggat atgttgtgtt ttacagtatt 2040atgtagtctg ttttttatgc
aaaatctaat ttaatatatt gatatttata tcattttacg 2100tttctcgttc agctttcttg
tacaaagtgg ttcgatatcg gtaccgagct cgaattcact 2160ggccgtcgtt ttacaacgtc
gtgactggga aaaccctggc gttacccaac ttaatcgcct 2220tgcagcacat ccccctttcg
ccagctggcg taatagcgaa gaggcccgca ccgatcgccc 2280ttcccaacag ttgcgcagcc
tgaatggcga atggcgcctg atgcggtatt ttctccttac 2340gcatctgtgc ggtatttcac
accgcatatg gtgcactctc agtacaatct gctctgatgc 2400cgcatagtta agccagcccc
gacacccgcc aacacccgct gacgcgccct gacgggcttg 2460tctgctcccg gcatccgctt
acagacaagc tgtgaccgtc tccgggagct gcatgtgtca 2520gaggttttca ccgtcatcac
cgaaacgcgc gagacgaaag ggcctcgtga tacgcctatt 2580tttataggtt aatgtcatga
taataatggt ttcttagacg tcaggtggca cttttcgggg 2640aaatgtgcgc ggaaccccta
tttgtttatt tttctaaata cattcaaata tgtatccgct 2700catgagacaa taaccctgat
aaatgcttca ataatattga aaaaggaaga gtatgagtat 2760tcaacatttc cgtgtcgccc
ttattccctt ttttgcggca ttttgccttc ctgtttttgc 2820tcacccagaa acgctggtga
aagtaaaaga tgctgaagat cagttgggtg cacgagtggg 2880ttacatcgaa ctggatctca
acagcggtaa gatccttgag agttttcgcc ccgaagaacg 2940ttttccaatg atgagcactt
ttaaagttct gctatgtggc gcggtattat cccgtattga 3000cgccgggcaa gagcaactcg
gtcgccgcat acactattct cagaatgact tggttgagta 3060ctcaccagtc acagaaaagc
atcttacgga tggcatgaca gtaagagaat tatgcagtgc 3120tgccataacc atgagtgata
acactgcggc caacttactt ctgacaacga tcggaggacc 3180gaaggagcta accgcttttt
tgcacaacat gggggatcat gtaactcgcc ttgatcgttg 3240ggaaccggag ctgaatgaag
ccataccaaa cgacgagcgt gacaccacga tgcctgtagc 3300aatggcaaca acgttgcgca
aactattaac tggcgaacta cttactctag cttcccggca 3360acaattaata gactggatgg
aggcggataa agttgcagga ccacttctgc gctcggccct 3420tccggctggc tggtttattg
ctgataaatc tggagccggt gagcgtgggt ctcgcggtat 3480cattgcagca ctggggccag
atggtaagcc ctcccgtatc gtagttatct acacgacggg 3540gagtcaggca actatggatg
aacgaaatag acagatcgct gagataggtg cctcactgat 3600taagcattgg taactgtcag
accaagttta ctcatatata ctttagattg atttaaaact 3660tcatttttaa tttaaaagga
tctaggtgaa gatccttttt gataatctca tgaccaaaat 3720cccttaacgt gagttttcgt
tccactgagc gtcagacccc gtagaaaaga tcaaaggatc 3780ttcttgagat cctttttttc
tgcgcgtaat ctgctgcttg caaacaaaaa aaccaccgct 3840accagcggtg gtttgtttgc
cggatcaaga gctaccaact ctttttccga aggtaactgg 3900cttcagcaga gcgcagatac
caaatactgt ccttctagtg tagccgtagt taggccacca 3960cttcaagaac tctgtagcac
cgcctacata cctcgctctg ctaatcctgt taccagtggc 4020tgctgccagt ggcgataagt
cgtgtcttac cgggttggac tcaagacgat agttaccgga 4080taaggcgcag cggtcgggct
gaacgggggg ttcgtgcaca cagcccagct tggagcgaac 4140gacctacacc gaactgagat
acctacagcg tgagctatga gaaagcgcca cgcttcccga 4200agggagaaag gcggacaggt
atccggtaag cggcagggtc ggaacaggag agcgcacgag 4260ggagcttcca gggggaaacg
cctggtatct ttatagtcct gtcgggtttc gccacctctg 4320acttgagcgt cgatttttgt
gatgctcgtc aggggggcgg agcctatgga aaaacgccag 4380caacgcggcc tttttacggt
tcctggcctt ttgctggcct tttgctcaca tgttctttcc 4440tgcgttatcc cctgattctg
tggataaccg tattaccgcc tttgagtgag ctgataccgc 4500tcgccgcagc cgaacgaccg
agcgcagcga gtcagtgagc gaggaagcgg aaga 45541757141DNAArtificial
SequenceDescription of Artificial Sequence Synthetic pDEST28
polynucleotide 175atgcatgtcg ttacataact tacggtaaat ggcccgcctg gctgaccgcc
caacgacccc 60cgcccattga cgtcaataat gacgtatgtt cccatagtaa cgccaatagg
gactttccat 120tgacgtcaat gggtggagta tttacggtaa actgcccact tggcagtaca
tcaagtgtat 180catatgccaa gtacgccccc tattgacgtc aatgacggta aatggcccgc
ctggcattat 240gcccagtaca tgaccttatg ggactttcct acttggcagt acatctacgt
attagtcatc 300gctattacca tggtgatgcg gttttggcag tacatcaatg ggcgtggata
gcggtttgac 360tcacggggat ttccaagtct ccaccccatt gacgtcaatg ggagtttgtt
ttggcaccaa 420aatcaacggg actttccaaa atgtcgtaac aactccgccc cattgacgca
aatgggcggt 480aggcgtgtac ggtgggaggt ctatataagc agagctctcc ctatcagtga
tagagatctc 540cctatcagtg atagagatcg tcgacgagct cgtttagtga accgtcagat
cgcctggaga 600cgccatccac gctgttttga cctccataga agacaccggg accgatccag
cctccggact 660ctagaggatc cctaccggtg atatcctcga gcccatcaac aagtttgtac
aaaaaagctg 720aacgagaaac gtaaaatgat ataaatatca atatattaaa ttagattttg
cataaaaaac 780agactacata atactgtaaa acacaacata tccagtcact atggcggccg
cattaggcac 840cccaggcttt acactttatg cttccggctc gtataatgtg tggattttga
gttaggatcc 900ggcgagattt tcaggagcta aggaagctaa aatggagaaa aaaatcactg
gatataccac 960cgttgatata tcccaatggc atcgtaaaga acattttgag gcatttcagt
cagttgctca 1020atgtacctat aaccagaccg ttcagctgga tattacggcc tttttaaaga
ccgtaaagaa 1080aaataagcac aagttttatc cggcctttat tcacattctt gcccgcctga
tgaatgctca 1140tccggaattc cgtatggcaa tgaaagacgg tgagctggtg atatgggata
gtgttcaccc 1200ttgttacacc gttttccatg agcaaactga aacgttttca tcgctctgga
gtgaatacca 1260cgacgatttc cggcagtttc tacacatata ttcgcaagat gtggcgtgtt
acggtgaaaa 1320cctggcctat ttccctaaag ggtttattga gaatatgttt ttcgtctcag
ccaatccctg 1380ggtgagtttc accagttttg atttaaacgt ggccaatatg gacaacttct
tcgcccccgt 1440tttcaccatg ggcaaatatt atacgcaagg cgacaaggtg ctgatgccgc
tggcgattca 1500ggttcatcat gccgtctgtg atggcttcca tgtcggcaga atgcttaatg
aattacaaca 1560gtactgcgat gagtggcagg gcggggcgta aagatctgga tccggcttac
taaaagccag 1620ataacagtat gcgtatttgc gcgctgattt ttgcggtata agaatatata
ctgatatgta 1680tacccgaagt atgtcaaaaa gaggtgtgct atgaagcagc gtattacagt
gacagttgac 1740agcgacagct atcagttgct caaggcatat atgatgtcaa tatctccggt
ctggtaagca 1800caaccatgca gaatgaagcc cgtcgtctgc gtgccgaacg ctggaaagcg
gaaaatcagg 1860aagggatggc tgaggtcgcc cggtttattg aaatgaacgg ctcttttgct
gacgagaaca 1920gggactggtg aaatgcagtt taaggtttac acctataaaa gagagagccg
ttatcgtctg 1980tttgtggatg tacagagtga tattattgac acgcccgggc gacggatggt
gatccccctg 2040gccagtgcac gtctgctgtc agataaagtc tcccgtgaac tttacccggt
ggtgcatatc 2100ggggatgaaa gctggcgcat gatgaccacc gatatggcca gtgtgccggt
ctccgttatc 2160ggggaagaag tggctgatct cagccaccgc gaaaatgaca tcaaaaacgc
cattaacctg 2220atgttctggg gaatataaat gtcaggctcc cttatacaca gccagtctgc
aggtcgacca 2280tagtgactgg atatgttgtg ttttacagta ttatgtagtc tgttttttat
gcaaaatcta 2340atttaatata ttgatattta tatcatttta cgtttctcgt tcagctttct
tgtacaaagt 2400ggttgatggg cggccgctct agagggccca agcttacgcg tgcatgcgac
gtcatagctc 2460tctccctata gtgagtcgta ttataagcta ggcactggcc gtcgttttac
aacgtcgtga 2520ctgggaaaac tgctagcttg ggatctttgt gaaggaacct tacttctgtg
gtgtgacata 2580attggacaaa ctacctacag agatttaaag ctctaaggta aatataaaat
ttttaagtgt 2640ataatgtgtt aaactagctg catatgcttg ctgcttgaga gttttgctta
ctgagtatga 2700tttatgaaaa tattatacac aggagctagt gattctaatt gtttgtgtat
tttagattca 2760cagtcccaag gctcatttca ggcccctcag tcctcacagt ctgttcatga
tcataatcag 2820ccataccaca tttgtagagg ttttacttgc tttaaaaaac ctcccacacc
tccccctgaa 2880cctgaaacat aaaatgaatg caattgttgt tgttaacttg tttattgcag
cttataatgg 2940ttacaaataa agcaatagca tcacaaattt cacaaataaa gcattttttt
cactgcattc 3000tagttgtggt ttgtccaaac tcatcaatgt atcttatcat gtctggatcg
atcctgcatt 3060aatgaatcgg ccaacgcgcg gggagaggcg gtttgcgtat tggctggcgt
aatagcgaag 3120aggcccgcac cgatcgccct tcccaacagt tgcgcagcct gaatggcgaa
tgggacgcgc 3180cctgtagcgg cgcattaagc gcggcgggtg tggtggttac gcgcagcgtg
accgctacac 3240ttgccagcgc cctagcgccc gctcctttcg ctttcttccc ttcctttctc
gccacgttcg 3300ccggctttcc ccgtcaagct ctaaatcggg ggctcccttt agggttccga
tttagtgctt 3360tacggcacct cgaccccaaa aaacttgatt agggtgatgg ttcacgtagt
gggccatcgc 3420cctgatagac ggtttttcgc cctttgacgt tggagtccac gttctttaat
agtggactct 3480tgttccaaac tggaacaaca ctcaacccta tctcggtcta ttcttttgat
ttataaggga 3540ttttgccgat ttcggcctat tggttaaaaa atgagctgat ttaacaaata
tttaacgcga 3600attttaacaa aatattaacg tttacaattt cgcctgatgc ggtattttct
ccttacgcat 3660ctgtgcggta tttcacaccg catacgcgga tctgcgcagc accatggcct
gaaataacct 3720ctgaaagagg aacttggtta ggtaccttct gaggcggaaa gaaccagctg
tggaatgtgt 3780gtcagttagg gtgtggaaag tccccaggct ccccagcagg cagaagtatg
caaagcatgc 3840atctcaatta gtcagcaacc aggtgtggaa agtccccagg ctccccagca
ggcagaagta 3900tgcaaagcat gcatctcaat tagtcagcaa ccatagtccc gcccctaact
ccgcccatcc 3960cgcccctaac tccgcccagt tccgcccatt ctccgcccca tggctgacta
atttttttta 4020tttatgcaga ggccgaggcc gcctcggcct ctgagctatt ccagaagtag
tgaggaggct 4080tttttggagg cctaggcttt tgcaaaaagc ttgattcttc tgacacaaca
gtctcgaact 4140taagaccatg gccaagcctt tgtctcaaga agaatccacc ctcattgaaa
gagcaacggc 4200tacaatcaac agcatcccca tctctgaaga ctacagcgtc gccagcgcag
ctctctctag 4260cgacggccgc atcttcactg gtgtcaatgt atatcatttt actgggggac
cttgtgcaga 4320actcgtggtg ctgggcactg ctgctgctgc ggcagctggc aacctgactt
gtatcgtcgc 4380gatcggaaat gagaacaggg gcatcttgag cccctgcgga cggtgccgac
aggtgcttct 4440cgatctgcat cctgggatca aagccatagt gaaggacagt gatggacagc
cgacggcagt 4500tgggattcgt gaattgctgc cctctggtta tgtgtgggag ggctaagcac
ttcgtggccg 4560agttcgaaat gaccgaccaa gcgacgccca acctgccatc acgatggccg
caataaaata 4620tctttatttt cattacatct gtgtgttggt tttttgtgtg aatcgatagc
gataaggatc 4680cgcgtatggt gcactctcag tacaatctgc tctgatgccg catagttaag
ccagccccga 4740cacccgccaa cacccgctga cgcgccctga cgggcttgtc tgctcccggc
atccgcttac 4800agacaagctg tgaccgtctc cgggagctgc atgtgtcaga ggttttcacc
gtcatcaccg 4860aaacgcgcga gacgaaaggg cctcgtgata cgcctatttt tataggttaa
tgtcatgata 4920ataatggttt cttagacgtc aggtggcact tttcggggaa atgtgcgcgg
aacccctatt 4980tgtttatttt tctaaataca ttcaaatatg tatccgctca tgagacaata
accctgataa 5040atgcttcaat aatattgaaa aaggaagagt atgagtattc aacatttccg
tgtcgccctt 5100attccctttt ttgcggcatt ttgccttcct gtttttgctc acccagaaac
gctggtgaaa 5160gtaaaagatg ctgaagatca gttgggtgca cgagtgggtt acatcgaact
ggatctcaac 5220agcggtaaga tccttgagag ttttcgcccc gaagaacgtt ttccaatgat
gagcactttt 5280aaagttctgc tatgtggcgc ggtattatcc cgtattgacg ccgggcaaga
gcaactcggt 5340cgccgcatac actattctca gaatgacttg gttgagtact caccagtcac
agaaaagcat 5400cttacggatg gcatgacagt aagagaatta tgcagtgctg ccataaccat
gagtgataac 5460actgcggcca acttacttct gacaacgatc ggaggaccga aggagctaac
cgcttttttg 5520cacaacatgg gggatcatgt aactcgcctt gatcgttggg aaccggagct
gaatgaagcc 5580ataccaaacg acgagcgtga caccacgatg cctgtagcaa tggcaacaac
gttgcgcaaa 5640ctattaactg gcgaactact tactctagct tcccggcaac aattaataga
ctggatggag 5700gcggataaag ttgcaggacc acttctgcgc tcggcccttc cggctggctg
gtttattgct 5760gataaatctg gagccggtga gcgtgggtct cgcggtatca ttgcagcact
ggggccagat 5820ggtaagccct cccgtatcgt agttatctac acgacgggga gtcaggcaac
tatggatgaa 5880cgaaatagac agatcgctga gataggtgcc tcactgatta agcattggta
actgtcagac 5940caagtttact catatatact ttagattgat ttaaaacttc atttttaatt
taaaaggatc 6000taggtgaaga tcctttttga taatctcatg accaaaatcc cttaacgtga
gttttcgttc 6060cactgagcgt cagaccccgt agaaaagatc aaaggatctt cttgagatcc
tttttttctg 6120cgcgtaatct gctgcttgca aacaaaaaaa ccaccgctac cagcggtggt
ttgtttgccg 6180gatcaagagc taccaactct ttttccgaag gtaactggct tcagcagagc
gcagatacca 6240aatactgtcc ttctagtgta gccgtagtta ggccaccact tcaagaactc
tgtagcaccg 6300cctacatacc tcgctctgct aatcctgtta ccagtggctg ctgccagtgg
cgataagtcg 6360tgtcttaccg ggttggactc aagacgatag ttaccggata aggcgcagcg
gtcgggctga 6420acggggggtt cgtgcacaca gcccagcttg gagcgaacga cctacaccga
actgagatac 6480ctacagcgtg agcattgaga aagcgccacg cttcccgaag ggagaaaggc
ggacaggtat 6540ccggtaagcg gcagggtcgg aacaggagag cgcacgaggg agcttccagg
gggaaacgcc 6600tggtatcttt atagtcctgt cgggtttcgc cacctctgac ttgagcgtcg
atttttgtga 6660tgctcgtcag gggggcggag cctatggaaa aacgccagca acgcggcctt
tttacggttc 6720ctggcctttt gctggccttt tgctcacatg ttctttcctg cgttatcccc
tgattctgtg 6780gataaccgta ttaccgcctt tgagtgagct gataccgctc gccgcagccg
aacgaccgag 6840cgcagcgagt cagtgagcga ggaagcggaa gagcgcccaa tacgcaaacc
gcctctcccc 6900gcgcgttggc cgattcatta atgcagagct tgcaattcgc gcgtttttca
atattattga 6960agcatttatc agggttattg tctcatgagc ggatacatat ttgaatgtat
ttagaaaaat 7020aaacaaatag gggttccgcg cacatttccc cgaaaagtgc cacctgacgt
ctaagaaacc 7080attattatca tgacattaac ctataaaaat aggcgtagta cgaggccctt
tcactcatta 7140g
71411767156DNAArtificial SequenceDescription of Artificial
Sequence Synthetic pDEST29 polynucleotide 176atgcatgtcg ttacataact
tacggtaaat ggcccgcctg gctgaccgcc caacgacccc 60cgcccattga cgtcaataat
gacgtatgtt cccatagtaa cgccaatagg gactttccat 120tgacgtcaat gggtggagta
tttacggtaa actgcccact tggcagtaca tcaagtgtat 180catatgccaa gtacgccccc
tattgacgtc aatgacggta aatggcccgc ctggcattat 240gcccagtaca tgaccttatg
ggactttcct acttggcagt acatctacgt attagtcatc 300gctattacca tggtgatgcg
gttttggcag tacatcaatg ggcgtggata gcggtttgac 360tcacggggat ttccaagtct
ccaccccatt gacgtcaatg ggagtttgtt ttggcaccaa 420aatcaacggg actttccaaa
atgtcgtaac aactccgccc cattgacgca aatgggcggt 480aggcgtgtac ggtgggaggt
ctatataagc agagctctcc ctatcagtga tagagatctc 540cctatcagtg atagagatcg
tcgacgagct cgtttagtga accgtcagat cgcctggaga 600cgccatccac gctgttttga
cctccataga agacaccggg accgatccag cctccggacc 660atggcgtact accatcacca
tcaccatcac accggtgata tcctcgagcc catcacaagt 720ttgtacaaaa aagctgaacg
agaaacgtaa aatgatataa atatcaatat attaaattag 780attttgcata aaaaacagac
tacataatac tgtaaaacac aacatatcca gtcactatgg 840cggccgcatt aggcacccca
ggctttacac tttatgcttc cggctcgtat aatgtgtgga 900ttttgagtta ggatccggcg
agattttcag gagctaagga agctaaaatg gagaaaaaaa 960tcactggata taccaccgtt
gatatatccc aatggcatcg taaagaacat tttgaggcat 1020ttcagtcagt tgctcaatgt
acctataacc agaccgttca gctggatatt acggcctttt 1080taaagaccgt aaagaaaaat
aagcacaagt tttatccggc ctttattcac attcttgccc 1140gcctgatgaa tgctcatccg
gaattccgta tggcaatgaa agacggtgag ctggtgatat 1200gggatagtgt tcacccttgt
tacaccgttt tccatgagca aactgaaacg ttttcatcgc 1260tctggagtga ataccacgac
gatttccggc agtttctaca catatattcg caagatgtgg 1320cgtgttacgg tgaaaacctg
gcctatttcc ctaaagggtt tattgagaat atgtttttcg 1380tctcagccaa tccctgggtg
agtttcacca gttttgattt aaacgtggcc aatatggaca 1440acttcttcgc ccccgttttc
accatgggca aatattatac gcaaggcgac aaggtgctga 1500tgccgctggc gattcaggtt
catcatgccg tctgtgatgg cttccatgtc ggcagaatgc 1560ttaatgaatt acaacagtac
tgcgatgagt ggcagggcgg ggcgtaaacg cgtggatccg 1620gcttactaaa agccagataa
cagtatgcgt atttgcgcgc tgatttttgc ggtataagaa 1680tatatactga tatgtatacc
cgaagtatgt caaaaagagg tgtgctatga agcagcgtat 1740tacagtgaca gttgacagcg
acagctatca gttgctcaag gcatatatga tgtcaatatc 1800tccggtctgg taagcacaac
catgcagaat gaagcccgtc gtctgcgtgc cgaacgctgg 1860aaagcggaaa atcaggaagg
gatggctgag gtcgcccggt ttattgaaat gaacggctct 1920tttgctgacg agaacaggga
ctggtgaaat gcagtttaag gtttacacct ataaaagaga 1980gagccgttat cgtctgtttg
tggatgtaca gagtgatatt attgacacgc ccgggcgacg 2040gatggtgatc cccctggcca
gtgcacgtct gctgtcagat aaagtctccc gtgaacttta 2100cccggtggtg catatcgggg
atgaaagctg gcgcatgatg accaccgata tggccagtgt 2160gccggtctcc gttatcgggg
aagaagtggc tgatctcagc caccgcgaaa atgacatcaa 2220aaacgccatt aacctgatgt
tctggggaat ataaatgtca ggctccgtta tacacagcca 2280gtctgcaggt cgaccatagt
gactggatat gttgtgtttt acagtattat gtagtctgtt 2340ttttatgcaa aatctaattt
aatatattga tatttatatc attttacgtt tctcgttcag 2400ctttcttgta caaagtggtg
atgggcggcc gctctagagg gcccaagctt acgcgtgcat 2460gcgacgtcat agctctctcc
ctatagtgag tcgtattata agctaggcac tggccgtcgt 2520tttacaacgt cgtgactggg
aaaactgcta gcttgggatc tttgtgaagg aaccttactt 2580ctgtggtgtg acataattgg
acaaactacc tacagagatt taaagctcta aggtaaatat 2640aaaattttta agtgtataat
gtgttaaact agctgcatat gcttgctgct tgagagtttt 2700gcttactgag tatgatttat
gaaaatatta tacacaggag ctagtgattc taattgtttg 2760tgtattttag attcacagtc
ccaaggctca tttcaggccc ctcagtcctc acagtctgtt 2820catgatcata atcagccata
ccacatttgt agaggtttta cttgctttaa aaaacctccc 2880acacctcccc ctgaacctga
aacataaaat gaatgcaatt gttgttgtta acttgtttat 2940tgcagcttat aatggttaca
aataaagcaa tagcatcaca aatttcacaa ataaagcatt 3000tttttcactg cattctagtt
gtggtttgtc caaactcatc aatgtatctt atcatgtctg 3060gatcgatcct gcattaatga
atcggccaac gcgcggggag aggcggtttg cgtattggct 3120ggcgtaatag cgaagaggcc
cgcaccgatc gcccttccca acagttgcgc agcctgaatg 3180gcgaatggga cgcgccctgt
agcggcgcat taagcgcggc gggtgtggtg gttacgcgca 3240gcgtgaccgc tacacttgcc
agcgccctag cgcccgctcc tttcgctttc ttcccttcct 3300ttctcgccac gttcgccggc
tttccccgtc aagctctaaa tcgggggctc cctttagggt 3360tccgatttag tgctttacgg
cacctcgacc ccaaaaaact tgattagggt gatggttcac 3420gtagtgggcc atcgccctga
tagacggttt ttcgcccttt gacgttggag tccacgttct 3480ttaatagtgg actcttgttc
caaactggaa caacactcaa ccctatctcg gtctattctt 3540ttgatttata agggattttg
ccgatttcgg cctattggtt aaaaaatgag ctgatttaac 3600aaatatttaa cgcgaatttt
aacaaaatat taacgtttac aatttcgcct gatgcggtat 3660tttctcctta cgcatctgtg
cggtatttca caccgcatac gcggatctgc gcagcaccat 3720ggcctgaaat aacctctgaa
agaggaactt ggttaggtac cttctgaggc ggaaagaacc 3780agctgtggaa tgtgtgtcag
ttagggtgtg gaaagtcccc aggctcccca gcaggcagaa 3840gtatgcaaag catgcatctc
aattagtcag caaccaggtg tggaaagtcc ccaggctccc 3900cagcaggcag aagtatgcaa
agcatgcatc tcaattagtc agcaaccata gtcccgcccc 3960taactccgcc catcccgccc
ctaactccgc ccagttccgc ccattctccg ccccatggct 4020gactaatttt ttttatttat
gcagaggccg aggccgcctc ggcctctgag ctattccaga 4080agtagtgagg aggctttttt
ggaggcctag gcttttgcaa aaagcttgat tcttctgaca 4140caacagtctc gaacttaaga
ccatggccaa gcctttgtct caagaagaat ccaccctcat 4200tgaaagagca acggctacaa
tcaacagcat ccccatctct gaagactaca gcgtcgccag 4260cgcagctctc tctagcgacg
gccgcatctt cactggtgtc aatgtatatc attttactgg 4320gggaccttgt gcagaactcg
tggtgctggg cactgctgct gctgcggcag ctggcaacct 4380gacttgtatc gtcgcgatcg
gaaatgagaa caggggcatc ttgagcccct gcggacggtg 4440ccgacaggtg cttctcgatc
tgcatcctgg gatcaaagcc atagtgaagg acagtgatgg 4500acagccgacg gcagttggga
ttcgtgaatt gctgccctct ggttatgtgt gggagggcta 4560agcacttcgt ggccgagttc
gaaatgaccg accaagcgac gcccaacctg ccatcacgat 4620ggccgcaata aaatatcttt
attttcatta catctgtgtg ttggtttttt gtgtgaatcg 4680atagcgataa ggatccgcgt
atggtgcact ctcagtacaa tctgctctga tgccgcatag 4740ttaagccagc cccgacaccc
gccaacaccc gctgacgcgc cctgacgggc ttgtctgctc 4800ccggcatccg cttacagaca
agctgtgacc gtctccggga gctgcatgtg tcagaggttt 4860tcaccgtcat caccgaaacg
cgcgagacga aagggcctcg tgatacgcct atttttatag 4920gttaatgtca tgataataat
ggtttcttag acgtcaggtg gcacttttcg gggaaatgtg 4980cgcggaaccc ctatttgttt
atttttctaa atacattcaa atatgtatcc gctcatgaga 5040caataaccct gataaatgct
tcaataatat tgaaaaagga agagtatgag tattcaacat 5100ttccgtgtcg cccttattcc
cttttttgcg gcattttgcc ttcctgtttt tgctcaccca 5160gaaacgctgg tgaaagtaaa
agatgctgaa gatcagttgg gtgcacgagt gggttacatc 5220gaactggatc tcaacagcgg
taagatcctt gagagttttc gccccgaaga acgttttcca 5280atgatgagca cttttaaagt
tctgctatgt ggcgcggtat tatcccgtat tgacgccggg 5340caagagcaac tcggtcgccg
catacactat tctcagaatg acttggttga gtactcacca 5400gtcacagaaa agcatcttac
ggatggcatg acagtaagag aattatgcag tgctgccata 5460accatgagtg ataacactgc
ggccaactta cttctgacaa cgatcggagg accgaaggag 5520ctaaccgctt ttttgcacaa
catgggggat catgtaactc gccttgatcg ttgggaaccg 5580gagctgaatg aagccatacc
aaacgacgag cgtgacacca cgatgcctgt agcaatggca 5640acaacgttgc gcaaactatt
aactggcgaa ctacttactc tagcttcccg gcaacaatta 5700atagactgga tggaggcgga
taaagttgca ggaccacttc tgcgctcggc ccttccggct 5760ggctggttta ttgctgataa
atctggagcc ggtgagcgtg ggtctcgcgg tatcattgca 5820gcactggggc cagatggtaa
gccctcccgt atcgtagtta tctacacgac ggggagtcag 5880gcaactatgg atgaacgaaa
tagacagatc gctgagatag gtgcctcact gattaagcat 5940tggtaactgt cagaccaagt
ttactcatat atactttaga ttgatttaaa acttcatttt 6000taatttaaaa ggatctaggt
gaagatcctt tttgataatc tcatgaccaa aatcccttaa 6060cgtgagtttt cgttccactg
agcgtcagac cccgtagaaa agatcaaagg atcttcttga 6120gatccttttt ttctgcgcgt
aatctgctgc ttgcaaacaa aaaaaccacc gctaccagcg 6180gtggtttgtt tgccggatca
agagctacca actctttttc cgaaggtaac tggcttcagc 6240agagcgcaga taccaaatac
tgtccttcta gtgtagccgt agttaggcca ccacttcaag 6300aactctgtag caccgcctac
atacctcgct ctgctaatcc tgttaccagt ggctgctgcc 6360agtggcgata agtcgtgtct
taccgggttg gactcaagac gatagttacc ggataaggcg 6420cagcggtcgg gctgaacggg
gggttcgtgc acacagccca gcttggagcg aacgacctac 6480accgaactga gatacctaca
gcgtgagcat tgagaaagcg ccacgcttcc cgaagggaga 6540aaggcggaca ggtatccggt
aagcggcagg gtcggaacag gagagcgcac gagggagctt 6600ccagggggaa acgcctggta
tctttatagt cctgtcgggt ttcgccacct ctgacttgag 6660cgtcgatttt tgtgatgctc
gtcagggggg cggagcctat ggaaaaacgc cagcaacgcg 6720gcctttttac ggttcctggc
cttttgctgg ccttttgctc acatgttctt tcctgcgtta 6780tcccctgatt ctgtggataa
ccgtattacc gcctttgagt gagctgatac cgctcgccgc 6840agccgaacga ccgagcgcag
cgagtcagtg agcgaggaag cggaagagcg cccaatacgc 6900aaaccgcctc tccccgcgcg
ttggccgatt cattaatgca gagcttgcaa ttcgcgcgtt 6960tttcaatatt attgaagcat
ttatcagggt tattgtctca tgagcggata catatttgaa 7020tgtatttaga aaaataaaca
aataggggtt ccgcgcacat ttccccgaaa agtgccacct 7080gacgtctaag aaaccattat
tatcatgaca ttaacctata aaaataggcg tagtacgagg 7140ccctttcact cattag
71561777544DNAArtificial
SequenceDescription of Artificial Sequence Synthetic pDEST30
polynucleotide 177atgcatgtcg ttacataact tacggtaaat ggcccgcctg gctgaccgcc
caacgacccc 60cgcccattga cgtcaataat gacgtatgtt cccatagtaa cgccaatagg
gactttccat 120tgacgtcaat gggtggagta tttacggtaa actgcccact tggcagtaca
tcaagtgtat 180catatgccaa gtacgccccc tattgacgtc aatgacggta aatggcccgc
ctggcattat 240gcccagtaca tgaccttatg ggactttcct acttggcagt acatctacgt
attagtcatc 300gctattacca tggtgatgcg gttttggcag tacatcaatg ggcgtggata
gcggtttgac 360tcacggggat ttccaagtct ccaccccatt gacgtcaatg ggagtttgtt
ttggcaccaa 420aatcaacggg actttccaaa atgtcgtaac aactccgccc cattgacgca
aatgggcggt 480aggcgtgtac ggtgggaggt ctatataagc agagctctcc ctatcagtga
tagagatctc 540cctatcagtg atagagatcg tcgacgagct cgtttagtga accgtcagat
cgcctggaga 600cgccatccac gctgttttga cctccataga agacaccggg accgatccag
cctccggact 660ctagaggatc cctaccggtg atatcctcga gcccatcaac aagtttgtac
aaaaaagctg 720aacgagaaac gtaaaatgat ataaatatca atatattaaa ttagattttg
cataaaaaac 780agactacata atactgtaaa acacaacata tccagtcact atggcggccg
cattaggcac 840cccaggcttt acactttatg cttccggctc gtataatgtg tggattttga
gttaggatcc 900ggcgagattt tcaggagcta aggaagctaa aatggagaaa aaaatcactg
gatataccac 960cgttgatata tcccaatggc atcgtaaaga acattttgag gcatttcagt
cagttgctca 1020atgtacctat aaccagaccg ttcagctgga tattacggcc tttttaaaga
ccgtaaagaa 1080aaataagcac aagttttatc cggcctttat tcacattctt gcccgcctga
tgaatgctca 1140tccggaattc cgtatggcaa tgaaagacgg tgagctggtg atatgggata
gtgttcaccc 1200ttgttacacc gttttccatg agcaaactga aacgttttca tcgctctgga
gtgaatacca 1260cgacgatttc cggcagtttc tacacatata ttcgcaagat gtggcgtgtt
acggtgaaaa 1320cctggcctat ttccctaaag ggtttattga gaatatgttt ttcgtctcag
ccaatccctg 1380ggtgagtttc accagttttg atttaaacgt ggccaatatg gacaacttct
tcgcccccgt 1440tttcaccatg ggcaaatatt atacgcaagg cgacaaggtg ctgatgccgc
tggcgattca 1500ggttcatcat gccgtctgtg atggcttcca tgtcggcaga atgcttaatg
aattacaaca 1560gtactgcgat gagtggcagg gcggggcgta aagatctgga tccggcttac
taaaagccag 1620ataacagtat gcgtatttgc gcgctgattt ttgcggtata agaatatata
ctgatatgta 1680tacccgaagt atgtcaaaaa gaggtgtgct atgaagcagc gtattacagt
gacagttgac 1740agcgacagct atcagttgct caaggcatat atgatgtcaa tatctccggt
ctggtaagca 1800caaccatgca gaatgaagcc cgtcgtctgc gtgccgaacg ctggaaagcg
gaaaatcagg 1860aagggatggc tgaggtcgcc cggtttattg aaatgaacgg ctcttttgct
gacgagaaca 1920gggactggtg aaatgcagtt taaggtttac acctataaaa gagagagccg
ttatcgtctg 1980tttgtggatg tacagagtga tattattgac acgcccgggc gacggatggt
gatccccctg 2040gccagtgcac gtctgctgtc agataaagtc tcccgtgaac tttacccggt
ggtgcatatc 2100ggggatgaaa gctggcgcat gatgaccacc gatatggcca gtgtgccggt
ctccgttatc 2160ggggaagaag tggctgatct cagccaccgc gaaaatgaca tcaaaaacgc
cattaacctg 2220atgttctggg gaatataaat gtcaggctcc cttatacaca gccagtctgc
aggtcgacca 2280tagtgactgg atatgttgtg ttttacagta ttatgtagtc tgttttttat
gcaaaatcta 2340atttaatata ttgatattta tatcatttta cgtttctcgt tcagctttct
tgtacaaagt 2400ggttgatggg cggccgctct agagggccca agcttacgcg tgcatgcgac
gtcatagctc 2460tctccctata gtgagtcgta ttataagcta ggcactggcc gtcgttttac
aacgtcgtga 2520ctgggaaaac tgctagcttg ggatctttgt gaaggaacct tacttctgtg
gtgtgacata 2580attggacaaa ctacctacag agatttaaag ctctaaggta aatataaaat
ttttaagtgt 2640ataatgtgtt aaactagctg catatgcttg ctgcttgaga gttttgctta
ctgagtatga 2700tttatgaaaa tattatacac aggagctagt gattctaatt gtttgtgtat
tttagattca 2760cagtcccaag gctcatttca ggcccctcag tcctcacagt ctgttcatga
tcataatcag 2820ccataccaca tttgtagagg ttttacttgc tttaaaaaac ctcccacacc
tccccctgaa 2880cctgaaacat aaaatgaatg caattgttgt tgttaacttg tttattgcag
cttataatgg 2940ttacaaataa agcaatagca tcacaaattt cacaaataaa gcattttttt
cactgcattc 3000tagttgtggt ttgtccaaac tcatcaatgt atcttatcat gtctggatcg
atcctgcatt 3060aatgaatcgg ccaacgcgcg gggagaggcg gtttgcgtat tggctggcgt
aatagcgaag 3120aggcccgcac cgatcgccct tcccaacagt tgcgcagcct gaatggcgaa
tgggacgcgc 3180cctgtagcgg cgcattaagc gcggcgggtg tggtggttac gcgcagcgtg
accgctacac 3240ttgccagcgc cctagcgccc gctcctttcg ctttcttccc ttcctttctc
gccacgttcg 3300ccggctttcc ccgtcaagct ctaaatcggg ggctcccttt agggttccga
tttagtgctt 3360tacggcacct cgaccccaaa aaacttgatt agggtgatgg ttcacgtagt
gggccatcgc 3420cctgatagac ggtttttcgc cctttgacgt tggagtccac gttctttaat
agtggactct 3480tgttccaaac tggaacaaca ctcaacccta tctcggtcta ttcttttgat
ttataaggga 3540ttttgccgat ttcggcctat tggttaaaaa atgagctgat ttaacaaata
tttaacgcga 3600attttaacaa aatattaacg tttacaattt cgcctgatgc ggtattttct
ccttacgcat 3660ctgtgcggta tttcacaccg catacgcgga tctgcgcagc accatggcct
gaaataacct 3720ctgaaagagg aacttggtta ggtaccttct gaggcggaaa gaaccagctg
tggaatgtgt 3780gtcagttagg gtgtggaaag tccccaggct ccccagcagg cagaagtatg
caaagcatgc 3840atctcaatta gtcagcaacc aggtgtggaa agtccccagg ctccccagca
ggcagaagta 3900tgcaaagcat gcatctcaat tagtcagcaa ccatagtccc gcccctaact
ccgcccatcc 3960cgcccctaac tccgcccagt tccgcccatt ctccgcccca tggctgacta
atttttttta 4020tttatgcaga ggccgaggcc gcctcggcct ctgagctatt ccagaagtag
tgaggaggct 4080tttttggagg cctaggcttt tgcaaaaagc ttgattcttc tgacacaaca
gtctcgaact 4140taaggctaga gccaccatga ttgaacaaga tggattgcac gcaggttctc
cggccgcttg 4200ggtggagagg ctattcggct atgactgggc acaacagaca atcggctgct
ctgatgccgc 4260cgtgttccgg ctgtcagcgc aggggcgccc ggttcttttt gtcaagaccg
acctgtccgg 4320tgccctgaat gaactgcagg acgaggcagc gcggctatcg tggctggcca
cgacgggcgt 4380tccttgcgca gctgtgctcg acgttgtcac tgaagcggga agggactggc
tgctattggg 4440cgaagtgccg gggcaggatc tcctgtcatc tcaccttgct cctgccgaga
aagtatccat 4500catggctgat gcaatgcggc ggctgcatac gcttgatccg gctacctgcc
cattcgacca 4560ccaagcgaaa catcgcatcg agcgagcacg tactcggatg gaagccggtc
ttgtcgatca 4620ggatgatctg gacgaagagc atcaggggct cgcgccagcc gaactgttcg
ccaggctcaa 4680ggcgcgcatg cccgacggcg aggatctcgt cgtgacccat ggcgatgcct
gcttgccgaa 4740tatcatggtg gaaaatggcc gcttttctgg attcatcgac tgtggccggc
tgggtgtggc 4800ggaccgctat caggacatag cgttggctac ccgtgatatt gctgaagagc
ttggcggcga 4860atgggctgac cgcttcctcg tgctttacgg tatcgccgct cccgattcgc
agcgcatcgc 4920cttctatcgc cttcttgacg agttcttctg agcgggactc tggggttcga
aatgaccgac 4980caagcgacgc ccaacctgcc atcacgatgg ccgcaataaa atatctttat
tttcattaca 5040tctgtgtgtt ggttttttgt gtgaatcgat agcgataagg atccgcgtat
ggtgcactct 5100cagtacaatc tgctctgatg ccgcatagtt aagccagccc cgacacccgc
caacacccgc 5160tgacgcgccc tgacgggctt gtctgctccc ggcatccgct tacagacaag
ctgtgaccgt 5220ctccgggagc tgcatgtgtc agaggttttc accgtcatca ccgaaacgcg
cgagacgaaa 5280gggcctcgtg atacgcctat ttttataggt taatgtcatg ataataatgg
tttcttagac 5340gtcaggtggc acttttcggg gaaatgtgcg cggaacccct atttgtttat
ttttctaaat 5400acattcaaat atgtatccgc tcatgagaca ataaccctga taaatgcttc
aataatattg 5460aaaaaggaag agtatgagta ttcaacattt ccgtgtcgcc cttattccct
tttttgcggc 5520attttgcctt cctgtttttg ctcacccaga aacgctggtg aaagtaaaag
atgctgaaga 5580tcagttgggt gcacgagtgg gttacatcga actggatctc aacagcggta
agatccttga 5640gagttttcgc cccgaagaac gttttccaat gatgagcact tttaaagttc
tgctatgtgg 5700cgcggtatta tcccgtattg acgccgggca agagcaactc ggtcgccgca
tacactattc 5760tcagaatgac ttggttgagt actcaccagt cacagaaaag catcttacgg
atggcatgac 5820agtaagagaa ttatgcagtg ctgccataac catgagtgat aacactgcgg
ccaacttact 5880tctgacaacg atcggaggac cgaaggagct aaccgctttt ttgcacaaca
tgggggatca 5940tgtaactcgc cttgatcgtt gggaaccgga gctgaatgaa gccataccaa
acgacgagcg 6000tgacaccacg atgcctgtag caatggcaac aacgttgcgc aaactattaa
ctggcgaact 6060acttactcta gcttcccggc aacaattaat agactggatg gaggcggata
aagttgcagg 6120accacttctg cgctcggccc ttccggctgg ctggtttatt gctgataaat
ctggagccgg 6180tgagcgtggg tctcgcggta tcattgcagc actggggcca gatggtaagc
cctcccgtat 6240cgtagttatc tacacgacgg ggagtcaggc aactatggat gaacgaaata
gacagatcgc 6300tgagataggt gcctcactga ttaagcattg gtaactgtca gaccaagttt
actcatatat 6360actttagatt gatttaaaac ttcattttta atttaaaagg atctaggtga
agatcctttt 6420tgataatctc atgaccaaaa tcccttaacg tgagttttcg ttccactgag
cgtcagaccc 6480cgtagaaaag atcaaaggat cttcttgaga tccttttttt ctgcgcgtaa
tctgctgctt 6540gcaaacaaaa aaaccaccgc taccagcggt ggtttgtttg ccggatcaag
agctaccaac 6600tctttttccg aaggtaactg gcttcagcag agcgcagata ccaaatactg
tccttctagt 6660gtagccgtag ttaggccacc acttcaagaa ctctgtagca ccgcctacat
acctcgctct 6720gctaatcctg ttaccagtgg ctgctgccag tggcgataag tcgtgtctta
ccgggttgga 6780ctcaagacga tagttaccgg ataaggcgca gcggtcgggc tgaacggggg
gttcgtgcac 6840acagcccagc ttggagcgaa cgacctacac cgaactgaga tacctacagc
gtgagcattg 6900agaaagcgcc acgcttcccg aagggagaaa ggcggacagg tatccggtaa
gcggcagggt 6960cggaacagga gagcgcacga gggagcttcc agggggaaac gcctggtatc
tttatagtcc 7020tgtcgggttt cgccacctct gacttgagcg tcgatttttg tgatgctcgt
caggggggcg 7080gagcctatgg aaaaacgcca gcaacgcggc ctttttacgg ttcctggcct
tttgctggcc 7140ttttgctcac atgttctttc ctgcgttatc ccctgattct gtggataacc
gtattaccgc 7200ctttgagtga gctgataccg ctcgccgcag ccgaacgacc gagcgcagcg
agtcagtgag 7260cgaggaagcg gaagagcgcc caatacgcaa accgcctctc cccgcgcgtt
ggccgattca 7320ttaatgcaga gcttgcaatt cgcgcgtttt tcaatattat tgaagcattt
atcagggtta 7380ttgtctcatg agcggataca tatttgaatg tatttagaaa aataaacaaa
taggggttcc 7440gcgcacattt ccccgaaaag tgccacctga cgtctaagaa accattatta
tcatgacatt 7500aacctataaa aataggcgta gtacgaggcc ctttcactca ttag
75441787559DNAArtificial SequenceDescription of Artificial
Sequence Synthetic pDEST31 polynucleotide 178atgcatgtcg ttacataact
tacggtaaat ggcccgcctg gctgaccgcc caacgacccc 60cgcccattga cgtcaataat
gacgtatgtt cccatagtaa cgccaatagg gactttccat 120tgacgtcaat gggtggagta
tttacggtaa actgcccact tggcagtaca tcaagtgtat 180catatgccaa gtacgccccc
tattgacgtc aatgacggta aatggcccgc ctggcattat 240gcccagtaca tgaccttatg
ggactttcct acttggcagt acatctacgt attagtcatc 300gctattacca tggtgatgcg
gttttggcag tacatcaatg ggcgtggata gcggtttgac 360tcacggggat ttccaagtct
ccaccccatt gacgtcaatg ggagtttgtt ttggcaccaa 420aatcaacggg actttccaaa
atgtcgtaac aactccgccc cattgacgca aatgggcggt 480aggcgtgtac ggtgggaggt
ctatataagc agagctctcc ctatcagtga tagagatctc 540cctatcagtg atagagatcg
tcgacgagct cgtttagtga accgtcagat cgcctggaga 600cgccatccac gctgttttga
cctccataga agacaccggg accgatccag cctccggacc 660atggcgtact accatcacca
tcaccatcac accggtgata tcctcgagcc catcacaagt 720ttgtacaaaa aagctgaacg
agaaacgtaa aatgatataa atatcaatat attaaattag 780attttgcata aaaaacagac
tacataatac tgtaaaacac aacatatcca gtcactatgg 840cggccgcatt aggcacccca
ggctttacac tttatgcttc cggctcgtat aatgtgtgga 900ttttgagtta ggatccggcg
agattttcag gagctaagga agctaaaatg gagaaaaaaa 960tcactggata taccaccgtt
gatatatccc aatggcatcg taaagaacat tttgaggcat 1020ttcagtcagt tgctcaatgt
acctataacc agaccgttca gctggatatt acggcctttt 1080taaagaccgt aaagaaaaat
aagcacaagt tttatccggc ctttattcac attcttgccc 1140gcctgatgaa tgctcatccg
gaattccgta tggcaatgaa agacggtgag ctggtgatat 1200gggatagtgt tcacccttgt
tacaccgttt tccatgagca aactgaaacg ttttcatcgc 1260tctggagtga ataccacgac
gatttccggc agtttctaca catatattcg caagatgtgg 1320cgtgttacgg tgaaaacctg
gcctatttcc ctaaagggtt tattgagaat atgtttttcg 1380tctcagccaa tccctgggtg
agtttcacca gttttgattt aaacgtggcc aatatggaca 1440acttcttcgc ccccgttttc
accatgggca aatattatac gcaaggcgac aaggtgctga 1500tgccgctggc gattcaggtt
catcatgccg tctgtgatgg cttccatgtc ggcagaatgc 1560ttaatgaatt acaacagtac
tgcgatgagt ggcagggcgg ggcgtaaacg cgtggatccg 1620gcttactaaa agccagataa
cagtatgcgt atttgcgcgc tgatttttgc ggtataagaa 1680tatatactga tatgtatacc
cgaagtatgt caaaaagagg tgtgctatga agcagcgtat 1740tacagtgaca gttgacagcg
acagctatca gttgctcaag gcatatatga tgtcaatatc 1800tccggtctgg taagcacaac
catgcagaat gaagcccgtc gtctgcgtgc cgaacgctgg 1860aaagcggaaa atcaggaagg
gatggctgag gtcgcccggt ttattgaaat gaacggctct 1920tttgctgacg agaacaggga
ctggtgaaat gcagtttaag gtttacacct ataaaagaga 1980gagccgttat cgtctgtttg
tggatgtaca gagtgatatt attgacacgc ccgggcgacg 2040gatggtgatc cccctggcca
gtgcacgtct gctgtcagat aaagtctccc gtgaacttta 2100cccggtggtg catatcgggg
atgaaagctg gcgcatgatg accaccgata tggccagtgt 2160gccggtctcc gttatcgggg
aagaagtggc tgatctcagc caccgcgaaa atgacatcaa 2220aaacgccatt aacctgatgt
tctggggaat ataaatgtca ggctccgtta tacacagcca 2280gtctgcaggt cgaccatagt
gactggatat gttgtgtttt acagtattat gtagtctgtt 2340ttttatgcaa aatctaattt
aatatattga tatttatatc attttacgtt tctcgttcag 2400ctttcttgta caaagtggtg
atgggcggcc gctctagagg gcccaagctt acgcgtgcat 2460gcgacgtcat agctctctcc
ctatagtgag tcgtattata agctaggcac tggccgtcgt 2520tttacaacgt cgtgactggg
aaaactgcta gcttgggatc tttgtgaagg aaccttactt 2580ctgtggtgtg acataattgg
acaaactacc tacagagatt taaagctcta aggtaaatat 2640aaaattttta agtgtataat
gtgttaaact agctgcatat gcttgctgct tgagagtttt 2700gcttactgag tatgatttat
gaaaatatta tacacaggag ctagtgattc taattgtttg 2760tgtattttag attcacagtc
ccaaggctca tttcaggccc ctcagtcctc acagtctgtt 2820catgatcata atcagccata
ccacatttgt agaggtttta cttgctttaa aaaacctccc 2880acacctcccc ctgaacctga
aacataaaat gaatgcaatt gttgttgtta acttgtttat 2940tgcagcttat aatggttaca
aataaagcaa tagcatcaca aatttcacaa ataaagcatt 3000tttttcactg cattctagtt
gtggtttgtc caaactcatc aatgtatctt atcatgtctg 3060gatcgatcct gcattaatga
atcggccaac gcgcggggag aggcggtttg cgtattggct 3120ggcgtaatag cgaagaggcc
cgcaccgatc gcccttccca acagttgcgc agcctgaatg 3180gcgaatggga cgcgccctgt
agcggcgcat taagcgcggc gggtgtggtg gttacgcgca 3240gcgtgaccgc tacacttgcc
agcgccctag cgcccgctcc tttcgctttc ttcccttcct 3300ttctcgccac gttcgccggc
tttccccgtc aagctctaaa tcgggggctc cctttagggt 3360tccgatttag tgctttacgg
cacctcgacc ccaaaaaact tgattagggt gatggttcac 3420gtagtgggcc atcgccctga
tagacggttt ttcgcccttt gacgttggag tccacgttct 3480ttaatagtgg actcttgttc
caaactggaa caacactcaa ccctatctcg gtctattctt 3540ttgatttata agggattttg
ccgatttcgg cctattggtt aaaaaatgag ctgatttaac 3600aaatatttaa cgcgaatttt
aacaaaatat taacgtttac aatttcgcct gatgcggtat 3660tttctcctta cgcatctgtg
cggtatttca caccgcatac gcggatctgc gcagcaccat 3720ggcctgaaat aacctctgaa
agaggaactt ggttaggtac cttctgaggc ggaaagaacc 3780agctgtggaa tgtgtgtcag
ttagggtgtg gaaagtcccc aggctcccca gcaggcagaa 3840gtatgcaaag catgcatctc
aattagtcag caaccaggtg tggaaagtcc ccaggctccc 3900cagcaggcag aagtatgcaa
agcatgcatc tcaattagtc agcaaccata gtcccgcccc 3960taactccgcc catcccgccc
ctaactccgc ccagttccgc ccattctccg ccccatggct 4020gactaatttt ttttatttat
gcagaggccg aggccgcctc ggcctctgag ctattccaga 4080agtagtgagg aggctttttt
ggaggcctag gcttttgcaa aaagcttgat tcttctgaca 4140caacagtctc gaacttaagg
ctagagccac catgattgaa caagatggat tgcacgcagg 4200ttctccggcc gcttgggtgg
agaggctatt cggctatgac tgggcacaac agacaatcgg 4260ctgctctgat gccgccgtgt
tccggctgtc agcgcagggg cgcccggttc tttttgtcaa 4320gaccgacctg tccggtgccc
tgaatgaact gcaggacgag gcagcgcggc tatcgtggct 4380ggccacgacg ggcgttcctt
gcgcagctgt gctcgacgtt gtcactgaag cgggaaggga 4440ctggctgcta ttgggcgaag
tgccggggca ggatctcctg tcatctcacc ttgctcctgc 4500cgagaaagta tccatcatgg
ctgatgcaat gcggcggctg catacgcttg atccggctac 4560ctgcccattc gaccaccaag
cgaaacatcg catcgagcga gcacgtactc ggatggaagc 4620cggtcttgtc gatcaggatg
atctggacga agagcatcag gggctcgcgc cagccgaact 4680gttcgccagg ctcaaggcgc
gcatgcccga cggcgaggat ctcgtcgtga cccatggcga 4740tgcctgcttg ccgaatatca
tggtggaaaa tggccgcttt tctggattca tcgactgtgg 4800ccggctgggt gtggcggacc
gctatcagga catagcgttg gctacccgtg atattgctga 4860agagcttggc ggcgaatggg
ctgaccgctt cctcgtgctt tacggtatcg ccgctcccga 4920ttcgcagcgc atcgccttct
atcgccttct tgacgagttc ttctgagcgg gactctgggg 4980ttcgaaatga ccgaccaagc
gacgcccaac ctgccatcac gatggccgca ataaaatatc 5040tttattttca ttacatctgt
gtgttggttt tttgtgtgaa tcgatagcga taaggatccg 5100cgtatggtgc actctcagta
caatctgctc tgatgccgca tagttaagcc agccccgaca 5160cccgccaaca cccgctgacg
cgccctgacg ggcttgtctg ctcccggcat ccgcttacag 5220acaagctgtg accgtctccg
ggagctgcat gtgtcagagg ttttcaccgt catcaccgaa 5280acgcgcgaga cgaaagggcc
tcgtgatacg cctattttta taggttaatg tcatgataat 5340aatggtttct tagacgtcag
gtggcacttt tcggggaaat gtgcgcggaa cccctatttg 5400tttatttttc taaatacatt
caaatatgta tccgctcatg agacaataac cctgataaat 5460gcttcaataa tattgaaaaa
ggaagagtat gagtattcaa catttccgtg tcgcccttat 5520tccctttttt gcggcatttt
gccttcctgt ttttgctcac ccagaaacgc tggtgaaagt 5580aaaagatgct gaagatcagt
tgggtgcacg agtgggttac atcgaactgg atctcaacag 5640cggtaagatc cttgagagtt
ttcgccccga agaacgtttt ccaatgatga gcacttttaa 5700agttctgcta tgtggcgcgg
tattatcccg tattgacgcc gggcaagagc aactcggtcg 5760ccgcatacac tattctcaga
atgacttggt tgagtactca ccagtcacag aaaagcatct 5820tacggatggc atgacagtaa
gagaattatg cagtgctgcc ataaccatga gtgataacac 5880tgcggccaac ttacttctga
caacgatcgg aggaccgaag gagctaaccg cttttttgca 5940caacatgggg gatcatgtaa
ctcgccttga tcgttgggaa ccggagctga atgaagccat 6000accaaacgac gagcgtgaca
ccacgatgcc tgtagcaatg gcaacaacgt tgcgcaaact 6060attaactggc gaactactta
ctctagcttc ccggcaacaa ttaatagact ggatggaggc 6120ggataaagtt gcaggaccac
ttctgcgctc ggcccttccg gctggctggt ttattgctga 6180taaatctgga gccggtgagc
gtgggtctcg cggtatcatt gcagcactgg ggccagatgg 6240taagccctcc cgtatcgtag
ttatctacac gacggggagt caggcaacta tggatgaacg 6300aaatagacag atcgctgaga
taggtgcctc actgattaag cattggtaac tgtcagacca 6360agtttactca tatatacttt
agattgattt aaaacttcat ttttaattta aaaggatcta 6420ggtgaagatc ctttttgata
atctcatgac caaaatccct taacgtgagt tttcgttcca 6480ctgagcgtca gaccccgtag
aaaagatcaa aggatcttct tgagatcctt tttttctgcg 6540cgtaatctgc tgcttgcaaa
caaaaaaacc accgctacca gcggtggttt gtttgccgga 6600tcaagagcta ccaactcttt
ttccgaaggt aactggcttc agcagagcgc agataccaaa 6660tactgtcctt ctagtgtagc
cgtagttagg ccaccacttc aagaactctg tagcaccgcc 6720tacatacctc gctctgctaa
tcctgttacc agtggctgct gccagtggcg ataagtcgtg 6780tcttaccggg ttggactcaa
gacgatagtt accggataag gcgcagcggt cgggctgaac 6840ggggggttcg tgcacacagc
ccagcttgga gcgaacgacc tacaccgaac tgagatacct 6900acagcgtgag cattgagaaa
gcgccacgct tcccgaaggg agaaaggcgg acaggtatcc 6960ggtaagcggc agggtcggaa
caggagagcg cacgagggag cttccagggg gaaacgcctg 7020gtatctttat agtcctgtcg
ggtttcgcca cctctgactt gagcgtcgat ttttgtgatg 7080ctcgtcaggg gggcggagcc
tatggaaaaa cgccagcaac gcggcctttt tacggttcct 7140ggccttttgc tggccttttg
ctcacatgtt ctttcctgcg ttatcccctg attctgtgga 7200taaccgtatt accgcctttg
agtgagctga taccgctcgc cgcagccgaa cgaccgagcg 7260cagcgagtca gtgagcgagg
aagcggaaga gcgcccaata cgcaaaccgc ctctccccgc 7320gcgttggccg attcattaat
gcagagcttg caattcgcgc gtttttcaat attattgaag 7380catttatcag ggttattgtc
tcatgagcgg atacatattt gaatgtattt agaaaaataa 7440acaaataggg gttccgcgca
catttccccg aaaagtgcca cctgacgtct aagaaaccat 7500tattatcatg acattaacct
ataaaaatag gcgtagtacg aggccctttc actcattag 755917912288DNAArtificial
SequenceDescription of Artificial Sequence Synthetic pDEST32
polynucleotide 179gacgaaaggg cctcgtgata cgcctatttt tataggttaa tgtcatgata
ataatggttt 60cttaggacgg atcgcttgcc tgtaacttac acgcgcctcg tatcttttaa
tgatggaata 120atttgggaat ttactctgtg tttatttatt tttatgtttt gtatttggat
tttagaaagt 180aaataaagaa ggtagaagag ttacggaatg aagaaaaaaa aataaacaaa
ggtttaaaaa 240atttcaacaa aaagcgtact ttacatatat atttattaga caagaaaagc
agattaaata 300gatatacatt cgattaacga taagtaaaat gtaaaatcac aggattttcg
tgtgtggtct 360tctacacaga caagatgaaa caattcggca ttaatacctg agagcaggaa
gagcaagata 420aaaggtagta tttgttggcg atccccctag agtcttttac atcttcggaa
aacaaaaact 480attttttctt taatttcttt ttttactttc tatttttaat ttatatattt
atattaaaaa 540atttaaatta taattatttt tatagcacgt gatgaaaagg acccaggtgg
cacttttcgg 600ggaaatgtgc gcggaacccc tatttgttta tttttctaaa tacattcaaa
tatgtatccg 660ctcatgagac aataaccctg ataaatgctt caataatctg cagtgcgcag
ggcccgtgtc 720tcaaaatctc tgatgttaca ttgcacaaga taaaaatata tcatcatgaa
caataaaact 780gtctgcttac ataaacagta atacaagggg tgttatgagc catattcaac
gggaaacgtc 840ttgctggagg ccgcgattaa attccaacat ggatgctgat ttatatgggt
ataaatgggc 900tcggtagcca accactagaa ctatagctag agtcctgggc gaacaaacga
tgctcgcctt 960ccagaaaacc gaggatgcga accacttcat ccggggtcag caccaccggc
aagcgccgcg 1020acggccgagg tcttccgatc tcctgaagcc agggcagatc cgtgcacagc
accttgccgt 1080agaagaacag caaggccgcc aatgcctgac gatgcgtgga gaccgaaacc
ttgcgctcgt 1140tcgccagcca ggacagaaat gcctcgactt cgctgctgcc caaggttgcc
gggtgacgca 1200caccgtggaa acggatgaag gcacgaaccc agttgacata agcctgttcg
gttcgtaaac 1260tgtaatgcaa gtagcgtatg cgctcacgca actggtccag aaccttgacc
gaacgcagcg 1320gtggtaacgg cgcagtggcg gttttcatgg cttgttatga ctgttttttt
gtacagtcta 1380tgcctcgggc atccaagcag caagcgcgtt acgccgtggg tcgatgtttg
atgttatgga 1440gcagcaacga tgttacgcag cagcaacgat gttacgcagc agggcagtcg
ccctaaaaca 1500aagttaggtg gctcaagtat gggcatcatt cgcacatgta ggctcggccc
tgaccaagtc 1560aaatccatgc gggctgctct tgatcttttc ggtcgtgagt tcggagacgt
agccacctac 1620tcccaacatc agccggactc cgattacctc gggaacttgc tccgtagtaa
gacattcatc 1680gcgcttgctg ccttcgacca agaagcggtt gttggcgctc tcgcggctta
cgttctgccc 1740aggtttgagc agccgcgtag tgagatctat atctatgatc tcgcagtctc
cggcgagcac 1800cggaggcagg gcattgccac cgcgctcatc aatctcctca agcatgaggc
caacgcgctt 1860ggtgcttatg tgatctacgt gcaagcagat tacggtgacg atcccgcagt
ggctctctat 1920acaaagttgg gcatacggga agaagtgatg cactttgata tcgacccaag
taccgccacc 1980taacaattcg ttcaagccga gatcggcttc ccggcctaat aggttgtatt
gatgttggac 2040gagtcggaat cgcagaccga taccaggatc ttgccatcct atggaactgc
ctcggtgagt 2100tttctccttc attacagaaa cggctttttc aaaaatatgg tattgataat
cctgatatga 2160ataaattgca gtttcatttg atgctcgatg agtttttcta atcagaattg
gttaattggt 2220tgtaacactg gcagagcatt acgctgactt gacgggacgg cgncatgacc
aaaatccctt 2280aacgtgagtt ttcgttccac tgagcgtcag accccgtaga aaagatcaaa
ggatcttctt 2340gagatccttt ttttctgcgc gtaatctgct gcttgcaaac aaaaaaacca
ccgctaccag 2400cggtggtttg tttgccggat caagagctac caactctttt tccgaaggta
actggcttca 2460gcagagcgca gataccaaat actgtccttc tagtgtagcc gtagttaggc
caccacttca 2520agaactctgt agcaccgcct acatacctcg ctctgctaat cctgttacca
gtggctgctg 2580ccagtggcga taagtcgtgt cttaccgggt tggactcaag acgatagtta
ccggataagg 2640cgcagcggtc gggctgaacg gggggttcgt gcacacagcc cagcttggag
cgaacgacct 2700acaccgaact gagataccta cagcgtgagc attgagaaag cgccacgctt
cccgaaggga 2760gaaaggcgga caggtatccg gtaagcggca gggtcggaac aggagagcgc
acgagggagc 2820ttccaggggg gaacgcctgg tatctttata gtcctgtcgg gtttcgccac
ctctgacttg 2880agcgtcgatt tttgtgatgc tcgtcagggg ggccgagcct atggaaaaac
gccagcaacg 2940cggccttttt acggttcctg gccttttgct ggccttttgc tcacatgttc
tttcctgcgt 3000tatcccctga ttctgtggat aaccgtatta ccgcctttga gtgagctgat
accgctcgcc 3060gcagccgaac gaccgagcgc agcgagtcag tgagcgagga agcggaagag
cgcccaatac 3120gcaaaccgcc tctccccgcg cgttggccga ttcattaatg cagctggcac
gacaggtttc 3180ccgactggaa agcgggcagt gagcgcaacg caattaatgt gagttacctc
actcattagg 3240caccccaggc tttacacttt atgcttccgg ctcctatgtt gtgtggaatt
gtgagcggat 3300aacaatttca cacaggaaac agctatgacc atgattacgc caagctcgga
attaaccctc 3360actaaaggga acaaaagctg gtaccgatcc cgagctttgc aaattaaagc
cttcgagcgt 3420cccaaaacct tctcaagcaa ggttttcagt ataatgttac atgcgtacac
gcgtctgtac 3480agaaaaaaaa gaaaaatttg aaatataaat aacgttctta atactaacat
aactataaaa 3540aaataaatag ggacctagac ttcaggttgt ctaactcctt ccttttcggt
tagagcggat 3600gtggggggag ggcgtgaatg taagcgtgac ataactaatt acatgatatc
gacaaaggaa 3660aaggggcctg tttactcaca ggcttttttc aagtaggtaa ttaagtcgtt
tctgtctttt 3720tccttcttca acccaccaaa ggccatcttg gtactttttt tttttttttt
tttttttttt 3780tttttttttt tttttttttt tttttttttt tttttttttt tttttttttt
tttttttttt 3840ttttttttca tagaaataat acagaagtag atgttgaatt agattaaact
gaagatatat 3900aatttattgg aaaatacata gagctttttg ttgatgcgct taagcgatca
attcaacaac 3960accaccagca gctctgattt tttcttcagc caacttggag acgaatctag
ctttgacgat 4020aactggaaca tttggaattc tacccttacc caagatctta ccgtaaccgg
ctgccaaagt 4080gtcaataact ggagcagttt ccttagaagc agatttcaag tattggtctc
tcttgtcttc 4140tgggatcaat gtccacaatt tgtccaagtt caagactggc ttccagaaat
gagcttgttg 4200cttgtggaag tatctcatac caaccttacc gaaataacct ggatggtatt
tatccatgtt 4260aattctgtgg tgatgttgac caccggccat acctctacca ccggggtgct
ttctgtgctt 4320accgatacga cctttaccgg ctgagacgtg acctctgtgc tttctagtct
tagtgaatct 4380ggaaggcatt cttgattagt tggatgattg ttctgggatt taatgcaaaa
atcacttaag 4440aaggaaaatc aacggagaaa gcaaacgcca tcttaaatat acgggataca
gatgaaaggg 4500tttgaaccta tctggaaaat agcattaaac aagcgaaaaa ctgcgaggaa
aattgtttgc 4560gtctctgcgg gctattcacg cgccagagga aaataggaaa aataacaggg
cattagaaaa 4620ataattttga ttttggtaat gtgtgggtcc tggtgtacag atgttacatt
ggttacagta 4680ctcttgtttt tgctgtgttt ttcgatgaat ctccaaaatg gttgttagca
catggaagag 4740tcaccgatgc taagttatct ctatgtaagc tacgtggcgt gacttttgat
gaagccgcac 4800aagagataca ggattggcaa ctgcaaatag aatctgggga tcccccctcg
agatccggga 4860tcgaagaaat gatggtaaat gaaataggaa atcaaggagc atgaaggcaa
aagacaaata 4920taagggtcga acgaaaaata aagtgaaaag tgttgatatg atgtatttgg
ctttgcggcg 4980ccgaaaaaac gagtttacgc aattgcacaa tcatgctgac tctgtggcgg
acccgcgctc 5040ttgccggccc ggcgataacg ctgggcgtga ggctgtgccc ggcggagttt
tttgcgcctg 5100cattttccaa ggtttaccct gcgctaaggg gcgagattgg agaagcaata
agaatgccgg 5160ttggggttgc gatgatgacg accacgacaa ctggtgtcat tatttaagtt
gccgaaagaa 5220cctgagtgca tttgcaacat gagtatacta gaagaatgag ccaagacttg
cgagacgcga 5280gtttgccggt ggtgcgaaca atagagcgac catgaccttg aaggtgagac
gcgcataacc 5340gctagagtac tttgaagagg aaacagcaat agggttgcta ccagtataaa
tagacaggta 5400catacaacac tggaaatggt tgtctgtttg agtacgcttt caattcattt
gggtgtgcac 5460tttattatgt tacaatatgg aagggaactt tacacttctc ctatgcacat
atattaatta 5520aagtccaatg ctagtagaga aggggggtaa cacccctccg cgctcttttc
cgattttttt 5580ctaaaccgtg gaatatttcg gatatccttt tgttgtttcc gggtgtacaa
tatggacttc 5640ctcttttctg gcaaccaaac ccatacatcg ggattcctat aataccttcg
ttggtctccc 5700taacatgtag gtggcggagg ggagatatac aatagaacag ataccagaca
agacataatg 5760ggctaaacaa gactacacca attacactgc ctcattgatg gtggtacata
acgaactaat 5820actgtagccc tagacttgat agccatcatc atatcgaagt ttcactaccc
tttttccatt 5880tgccatctat tgaagtaata ataggcgcat gcaacttctt ttcttttttt
ttcttttctc 5940tctcccccgt tgttgtctca ccatatccgc aatgacaaaa aaaatgatgg
aagacactaa 6000aggaaaaaat taacgacaaa gacagcacca acagatgtcg ttgttccaga
gctgatgagg 6060ggtatcttcg aacacacgaa actttttcct tccttcattc acgcacacta
ctctctaatg 6120agcaacggta tacggccttc cttccagtta cttgaatttg aaataaaaaa
agtttgccgc 6180tttgctatca agtataaata gacctgcaat tattaatctt ttgtttcctc
gtcattgttc 6240tcgttccctt tcttccttgt ttctttttct gcacaatatt tcaagctata
ccaagcatac 6300aatcaactcc aagcttgaag caagcctcct gaaagatgaa gctactgtct
tctatcgaac 6360aagcatgcga tatttgccga cttaaaaagc tcaagtgctc caaagaaaaa
ccgaagtgcg 6420ccaagtgtct gaagaacaac tgggagtgtc gctactctcc caaaaccaaa
aggtctccgc 6480tgactagggc acatctgaca gaagtggaat caaggctaga aagactggaa
cagctatttc 6540tactgatttt tcctcgagaa gaccttgaca tgattttgaa aatggattct
ttacaggata 6600taaaagcatt gttaacagga ttatttgtac aagataatgt gaataaagat
gccgtcacag 6660atagattggc ttcagtggag actgatatgc ctctaacatt gagacagcat
agaataagtg 6720cgacatcatc atcggaagag agtagtaaca aaggtcaaag acagttgact
gtatcgtcga 6780ggtcgaatca aacaagtttg tacaaaaaag ctgaacgaga aacgtaaaat
gatataaata 6840tcaatatatt aaattagatt ttgcataaaa aacagactac ataatactgt
aaaacacaac 6900atatccagtc actatggcgg ccgctaagtt ggcagcatca cccgacgcac
tttgcgccga 6960ataaatacct gtgacggaag atcacttcgc agaataaata aatcctggtg
tccctgttga 7020taccgggaag ccctgggcca acttttggcg aaaatgagac gttgatcggc
acgtaagagg 7080ttccaacttt caccataatg aaataagatc actaccgggc gtattttttg
agttatcgag 7140attttcagga gctaaggaag ctaaaatgga gaaaaaaatc actggatata
ccaccgttga 7200tatatcccaa tggcatcgta aagaacattt tgaggcattt cagtcagttg
ctcaatgtac 7260ctataaccag accgttcagc tggatattac ggccttttta aagaccgtaa
agaaaaataa 7320gcacaagttt tatccggcct ttattcacat tcttgcccgc ctgatgaatg
ctcatccgga 7380attccgtatg gcaatgaaag acggtgagct ggtgatatgg gatagtgttc
acccttgtta 7440caccgttttc catgagcaaa ctgaaacgtt ttcatcgctc tggagtgaat
accacgacga 7500tttccggcag tttctacaca tatattcgca agatgtggcg tgttacggtg
aaaacctggc 7560ctatttccct aaagggttta ttgagaatat gtttttcgtc tcagccaatc
cctgggtgag 7620tttcaccagt tttgatttaa acgtggccaa tatggacaac ttcttcgccc
ccgttttcac 7680catgggcaaa tattatacgc aaggcgacaa ggtgctgatg ccgctggcga
ttcaggttca 7740tcatgccgtc tgtgatggct tccatgtcgg cagaatgctt aatgaattac
aacagtactg 7800cgatgagtgg cagggcgggg cgtaatctag aggatccggc ttactaaaag
ccagataaca 7860gtatgcgtat ttgcgcgctg atttttgcgg tataagaata tatactgata
tgtatacccg 7920aagtatgtca aaaagaggtg tgctatgaag cagcgtatta cagtgacagt
tgacagcgac 7980agctatcagt tgctcaaggc atatatgatg tcaatatctc cggtctggta
agcacaacca 8040tgcagaatga agcccgtcgt ctgcgtgccg aacgctggaa agcggaaaat
caggaaggga 8100tggctgaggt cgcccggttt attgaaatga acggctcttt tgctgacgag
aacagggact 8160ggtgaaatgc agtttaaggt ttacacctat aaaagagaga gccgttatcg
tctgtttgtg 8220gatgtacaga gtgatattat tgacacgccc gggcgacgga tggtgatccc
cctggccagt 8280gcacgtctgc tgtcagataa agtctcccgt gaactttacc cggtggtgca
tatcggggat 8340gaaagctggc gcatgatgac caccgatatg gccagtgtgc cggtctccgt
tatcggggaa 8400gaagtggctg atctcagcca ccgcgaaaat gacatcaaaa acgccattaa
cctgatgttc 8460tggggaatat aaatgtcagg ctcccttata cacagccagt ctgcaggtcg
accatagtga 8520ctggatatgt tgtgttttac agtattatgt agtctgtttt ttatgcaaaa
tctaatttaa 8580tatattgata tttatatcat tttacgtttc tcgttcagct ttcttgtaca
aagtggtttg 8640atggccgcta agtaagtaag acgtcgagct ctaagtaagt aacggccgcc
accgcggtgg 8700agctttggac ttcttcgcca gaggtttggt caagtctcca atcaaggttg
tcggcttgtc 8760taccttgcca gaaatttacg aaaagatgga aaagggtcaa atcgttggta
gatacgttgt 8820tgacacttct aaataagcga atttcttatg atttatgatt tttattatta
aataagttat 8880aaaaaaaata agtgtataca aattttaaag tgactcttag gttttaaaac
gaaaattctt 8940gttcttgagt aactctttcc tgtaggtcag gttgctttct caggtatagc
atgaggtcgc 9000tcttattgac cacacctcta ccggcatgcc gagcaaatgc ctgcaaatcg
ctccccattt 9060cacccaattg tagatatgct aactccagca atgagttgat gaatctcggt
gtgtatttta 9120tgtcctcaga ggacaatacc tgttgtaatc gttcttccac acggatccca
attcgcccta 9180tagtgagtcg tattacaatt cactggccgt cgttttacaa cgtcgtgact
gggaaaaccc 9240tggcgttacc caacttaatc gccttgcagc acatccccct ttcgccagct
ggcgtaatag 9300cgaagaggcc cgcaccgatc gcccttccca acagttgcgc agcctgaatg
gcgaatggac 9360gcgccctgta gcggcgcatt aagcgcggcg ggtgtggtgg ttacgcgcag
cgtgaccgct 9420acacttgcca gcgccctagc gcccgctcct ttcgctttct tcccttcctt
tctcgccacg 9480ttcgccggct ttccccgtca agctctaaat cgggggctcc ctttagggtt
ccgatttagt 9540gctttacggc acctcgaccc caaaaaactt gattagggtg atggttcacg
tagtgggcca 9600tcgccctgat agacggtttt tcgccctttg acgttggagt ccacgttctt
taatagtgga 9660ctcttgttcc aaactggaac aacactcaac cctatctcgg tctattcttt
tgatttataa 9720gggattttgc cgatttcggc ctattggtta aaaaatgagc tgatttaaca
aaaatttaac 9780gcgaatttta acaaaatatt aacgtttaca atttcctgat gcggtatttt
ctccttacgc 9840atctgtgcgg tatttcacac cgcatatcga ccggtcgagg agaacttcta
gtatatccac 9900atacctaata ttattgcctt attaaaaatg gaatcggaac aattacatca
aaatccacat 9960tctcttcaaa atcaattgtc ctgtacttcc ttgttcatgt gtgttcaaaa
acgttatatt 10020tataggataa ttatactcta tttctcaaca agtaattggt tgtttggccg
agcggtctaa 10080ggcgcctgat tcaagaaata tcttgaccgc agttaactgt gggaatactc
aggtatcgta 10140agatgcaaga gttcgaatct cttagcaacc attatttttt tcctcaacat
aacgagaaca 10200cacaggggcg ctatcgcaca gaatcaaatt cgatgactgg aaattttttg
ttaatttcag 10260aggtcgcctg acgcatatac ctttttcaac tgaaaaattg ggagaaaaag
gaaaggtgag 10320aggccggaac cggcttttca tatagaatag agaagcgttc atgactaaat
gcttgcatca 10380caatacttga agttgacaat attatttaag gacctattgt tttttccaat
aggtggttag 10440caatcgtctt actttctaac ttttcttacc ttttacattt cagcaatata
tatatatatt 10500tcaaggatat accattctaa tgtctgcccc tatgtctgcc cctaagaaga
tcgtcgtttt 10560gccaggtgac cacgttggtc aagaaatcac agccgaagcc attaaggttc
ttaaagctat 10620ttctgatgtt cgttccaatg tcaagttcga tttcgaaaat catttaattg
gtggtgctgc 10680tatcgatgct acaggtgtcc cacttccaga tgaggcgctg gaagcctcca
agaaggttga 10740tgccgttttg ttaggtgctg tgggtggtcc taaatggggt accggtagtg
ttagacctga 10800acaaggttta ctaaaaatcc gtaaagaact tcaattgtac gccaacttaa
gaccatgtaa 10860ctttgcatcc gactctcttt tagacttatc tccaatcaag ccacaatttg
ctaaaggtac 10920tgacttcgtt gttgtcagag aattagtggg aggtatttac tttggtaaga
gaaaggaaga 10980cgatggtgat ggtgtcgctt gggatagtga acaatacacc gttccagaag
tgcaaagaat 11040cacaagaatg gccgctttca tggccctaca acatgagcca ccattgccta
tttggtcctt 11100ggataaagct aatgttttgg cctcttcaag attatggaga aaaactgtgg
aggaaaccat 11160caagaacgaa ttccctacat tgaaggttca acatcaattg attgattctg
ccgccatgat 11220cctagttaag aacccaaccc acctaaatgg tattataatc accagcaaca
tgtttggtga 11280tatcatctcc gatgaagcct ccgttatccc aggttccttg ggtttgttgc
catctgcgtc 11340cttggcctct ttgccagaca agaacaccgc atttggtttg tacgaaccat
gccacggttc 11400tgctccagat ttgccaaaga ataaggttga ccctatcgcc actatcttgt
ctgctgcaat 11460gatgttgaaa ttgtcattga acttgcctga agaaggtaag gccattgaag
atgcagttaa 11520aaaggttttg gatgcaggta tcagaactgg tgatttaggt ggttccaaca
gtaccaccga 11580agtcggtgat gctgtcgccg aagaagttaa gaaaatcctt gcttaaaaag
attctctttt 11640tttatgatat ttgtacataa actttataaa tgaaattcat aatagaaacg
acacgaaatt 11700acaaaatgga atatgttcat agggtagacg aaactatata cgcaatctac
atacatttat 11760caagaaggag aaaaaggagg atagtaaagg aatacaggta agcaaattga
tactaatggc 11820tcaacgtgat aaggaaaaag aattgcactt taacattaat attgacaagg
aggagggcac 11880cacacaaaaa gttaggtgta acagaaaatc atgaaactac gattcctaat
ttgatattgg 11940aggattttct ctaaaaaaaa aaaaatacaa caaataaaaa acactcaatg
acctgaccat 12000ttgatggagt ttaagtcaat accttcttga accatttccc ataatggtga
aagttccctc 12060aagaatttta ctctgtcaga aacggcctta cgacgtagtc gatatggtgc
actctcagta 12120caatctgctc tgatgccgca tagttaagcc agccccgaca cccgccaaca
cccgctgacg 12180cgccctgacg ggcttgtctg ctcccggcat ccgcttacag acaagctgtg
accgtctccg 12240ggagctgcat gtgtcagagg ttttcaccgt catcaccgaa acgcgcga
122881808815DNAArtificial SequenceDescription of Artificial
Sequence Synthetic pDEST33 polynucleotide 180gccttacgca tctgtgcggt
atttcacacc gcaggcaagt gcacaaacaa tacttaaata 60aatactactc agtaataacc
tatttcttag catttttgac gaaatttgct attttgttag 120agtcttttac accatttgtc
tccacacctc cgcttacatc aacaccaata acgccattta 180atctaagcgc atcaccaaca
ttttctggcg tcagtccacc agctaacata aaatgtaagc 240tttcggggct ctcttgcctt
ccaacccagt cagaaatcga gttccaatcc aaaagttcac 300ctgtcccacc tgcttctgaa
tcaaacaagg gaataaacga atgaggtttc tgtgaagctg 360cactgagtag tatgttgcag
tcttttggaa atacgagtct tttaataact ggcaaaccga 420ggaactcttg gtattcttgc
cacgactcat ctccatgcag ttggacgata tcaatgccgt 480aatcattgac cagagccaaa
acatcctcct taggttgatt acgaaacacg ccaaccaagt 540atttcggagt gcctgaacta
tttttatatg cttttacaag acttgaaatt ttccttgcaa 600taaccgggtc aattgttctc
tttctattgg gcacacatat aatacccagc aagtcagcat 660cggaatctag agcacattct
gcggcctctg tgctctgcaa gccgcaaact ttcaccaatg 720gaccagaact acctgtgaaa
ttaataacag acatactcca agctgccttt gtgtgcttaa 780tcacgtatac tcacgtgctc
aatagtcacc aatgccctcc ctcttggccc tctccttttc 840ttttttcgac cgaattaatt
cttaatcggc aaaaaaagaa aagctccgga tcaagattgt 900acgtaaggtg acaagctatt
tttcaataaa gaatatcttc cactactgcc atctggcgtc 960ataactgcaa agtacacata
tattacgatg ctgtctatta aatgcttcct atattatata 1020tatagtaatg tcgtttatgg
tgcactctca gtacaatctg ctctgatgcc gcatagttaa 1080gccagccccg acacccgcca
acacccgctg acgcgccctg acgggcttgt ctgctcccgg 1140catccgctta cagacaagct
gtgaccgtct ccgggagctg catgtgtcag aggttttcac 1200cgtcatcacc gaaacgcgcg
agacgaaagg gcctcgtgat acgcctattt ttataggtta 1260atgtcatgat aataatggtt
tcttaggacg gatcgcttgc ctgtaactta cacgcgcctc 1320gtatctttta atgatggaat
aatttgggaa tttactctgt gtttatttat ttttatgttt 1380tgtatttgga ttttagaaag
taaataaaga aggtagaaga gttacggaat gaagaaaaaa 1440aaataaacaa aggtttaaaa
aatttcaaca aaaagcgtac tttacatata tatttattag 1500acaagaaaag cagattaaat
agatatacat tcgattaacg ataagtaaaa tgtaaaatca 1560caggattttc gtgtgtggtc
ttctacacag acaagatgaa acaattcggc attaatacct 1620gagagcagga agagcaagat
aaaaggtagt atttgttggc gatcccccta gagtctttta 1680catcttcgga aaacaaaaac
tattttttct ttaatttctt tttttacttt ctatttttaa 1740tttatatatt tatattaaaa
aatttaaatt ataattattt ttatagcacg tgatgaaaag 1800gacccaggtg gcacttttcg
gggaaatgtg cgcggaaccc ctatttgttt atttttctaa 1860atacattcaa atatgtatcc
gctcatgaga caataaccct gataaatgct tcaataatat 1920tgaaaaagga agagtatgag
tattcaacat ttccgtgtcg cccttattcc cttttttgcg 1980gcattttgcc ttcctgtttt
tgctcaccca gaaacgctgg tgaaagtaaa agatgctgaa 2040gatcagttgg gtgcacgagt
gggttacatc gaactggatc tcaacagcgg taagatcctt 2100gagagttttc gccccgaaga
acgttttcca atgatgagca cttttaaagt tctgctatgt 2160ggcgcggtat tatcccgtat
tgacgccggg caagagcaac tcggtcgccg catacactat 2220tctcagaatg acttggttga
gtactcacca gtcacagaaa agcatcttac ggatggcatg 2280acagtaagag aattatgcag
tgctgccata accatgagtg ataacactgc ggccaactta 2340cttctgacaa cgatcggagg
accgaaggag ctaaccgctt tttttcacaa catgggggat 2400catgtaactc gccttgatcg
ttgggaaccg gagctgaatg aagccatacc aaacgacgag 2460cgtgacacca cgatgcctgt
agcaatggca acaacgttgc gcaaactatt aactggcgaa 2520ctacttactc tagcttcccg
gcaacaatta atagactgga tggaggcgga taaagttgca 2580ggaccacttc tgcgctcggc
ccttccggct ggctggttta ttgctgataa atctggagcc 2640ggtgagcgtg ggtctcgcgg
tatcattgca gcactggggc cagatggtaa gccctcccgt 2700atcgtagtta tctacacgac
gggcagtcag gcaactatgg atgaacgaaa tagacagatc 2760gctgagatag gtgcctcact
gattaagcat tggtaactgt cagaccaagt ttactcatat 2820atactttaga ttgatttaaa
acttcatttt taatttaaaa ggatctaggt gaagatcctt 2880tttgataatc tcatgaccaa
aatcccttaa cgtgagtttt cgttccactg agcgtcagac 2940cccgtagaaa agatcaaagg
atcttcttga gatccttttt ttctgcgcgt aatctgctgc 3000ttgcaaacaa aaaaaccacc
gctaccagcg gtggtttgtt tgccggatca agagctacca 3060actctttttc cgaaggtaac
tggcttcagc agagcgcaga taccaaatac tgtccttcta 3120gtgtagccgt agttaggcca
ccacttcaag aactctgtag caccgcctac atacctcgct 3180ctgctaatcc tgttaccagt
ggctgctgcc agtggcgata agtcgtgtct taccgggttg 3240gactcaagac gatagttacc
ggataaggcg cagcggtcgg gctgaacggg gggttcgtgc 3300acacagccca gcttggagcg
aacgacctac accgaactga gatacctaca gcgtgagcat 3360tgagaaagcg ccacgcttcc
cgaagggaga aaggcggaca ggtatccggt aagcggcagg 3420gtcggaacag gagagcgcac
gagggagctt ccagggggga acgcctggta tctttatagt 3480cctgtcgggt ttcgccacct
ctgacttgag cgtcgatttt tgtgatgctc gtcagggggg 3540ccgagcctat ggaaaaacgc
cagcaacgcg gcctttttac ggttcctggc cttttgctgg 3600ccttttgctc acatgttctt
tcctgcgtta tcccctgatt ctgtggataa ccgtattacc 3660gcctttgagt gagctgatac
cgctcgccgc agccgaacga ccgagcgcag cgagtcagtg 3720agcgaggaag cggaagagcg
cccaatacgc aaaccgcctc tccccgcgcg ttggccgatt 3780cattaatgca gctggcacga
caggtttccc gactggaaag cgggcagtga gcgcaacgca 3840attaatgtga gttacctcac
tcattaggca ccccaggctt tacactttat gcttccggct 3900cctatgttgt gtggaattgt
gagcggataa caatttcaca caggaaacag ctatgaccat 3960gattacgcca agctcggaat
taaccctcac taaagggaac aaaagctggg taccgggccc 4020cccctcgaga tccgggatcg
aagaaatgat ggtaaatgaa ataggaaatc aaggagcatg 4080aaggcaaaag acaaatataa
gggtcgaacg aaaaataaag tgaaaagtgt tgatatgatg 4140tatttggctt tgcggcgccg
aaaaaacgag tttacgcaat tgcacaatca tgctgactct 4200gtggcggacc cgcgctcttg
ccggcccggc gataacgctg ggcgtgaggc tgtgcccggc 4260ggagtttttt gcgcctgcat
tttccaaggt ttaccctgcg ctaaggggcg agattggaga 4320agcaataaga atgccggttg
gggttgcgat gatgacgacc acgacaactg gtgtcattat 4380ttaagttgcc gaaagaacct
gagtgcattt gcaacatgag tatactagaa gaatgagcca 4440agacttgcga gacgcgagtt
tgccggtggt gcgaacaata gagcgaccat gaccttgaag 4500gtgagacgcg cataaccgct
agagtacttt gaagaggaaa cagcaatagg gttgctacca 4560gtataaatag acaggtacat
acaacactgg aaatggttgt ctgtttgagt acgctttcaa 4620ttcatttggg tgtgcacttt
attatgttac aatatggaag ggaactttac acttctccta 4680tgcacatata ttaattaaag
tccaatgcta gtagagaagg ggggtaacac ccctccgcgc 4740tcttttccga tttttttcta
aaccgtggaa tatttcggat atccttttgt tgtttccggg 4800tgtacaatat ggacttcctc
ttttctggca accaaaccca tacatcggga ttcctataat 4860accttcgttg gtctccctaa
catgtaggtg gcggagggga gatatacaat agaacagata 4920ccagacaaga cataatgggc
taaacaagac tacaccaatt acactgcctc attgatggtg 4980gtacataacg aactaatact
gtagccctag acttgatagc catcatcata tcgaagtttc 5040actacccttt ttccatttgc
catctattga agtaataata ggcgcatgca acttcttttc 5100tttttttttc ttttctctct
cccccgttgt tgtctcacca tatccgcaat gacaaaaaaa 5160atgatggaag acactaaagg
aaaaaattaa cgacaaagac agcaccaaca gatgtcgttg 5220ttccagagct gatgaggggt
atcttcgaac acacgaaact ttttccttcc ttcattcacg 5280cacactactc tctaatgagc
aacggtatac ggccttcctt ccagttactt gaatttgaaa 5340taaaaaaagt ttgccgcttt
gctatcaagt ataaatagac ctgcaattat taatcttttg 5400tttcctcgtc attgttctcg
ttccctttct tccttgtttc tttttctgca caatatttca 5460agctatacca agcatacaat
caactccaag cttatgccca agaagaagcg gaaggtctcg 5520agcggcgcca attttaatca
aagtgggaat attgctgata gctcattgtc cttcactttc 5580actaacagta gcaacggtcc
gaacctcata acaactcaaa caaattctca agcgctttca 5640caaccaattg cctcctctaa
cgttcatgat aacttcatga ataatgaaat cacggctagt 5700aaaattgatg atggtaataa
ttcaaaacca ctgtcacctg gttggacgga ccaaactgcg 5760tataacgcgt ttggaatcac
tacagggatg tttaatacca ctacaatgga tgatgtatat 5820aactatctat tcgatgatga
agatacccca ccaaacccaa aaaaagaggg tgggtcgaat 5880caaacaagtt tgtacaaaaa
agctgaacga gaaacgtaaa atgatataaa tatcaatata 5940ttaaattaga ttttgcataa
aaaacagact acataatact gtaaaacaca acatatccag 6000tcactatggc ggccgctaag
ttggcagcat cacccgacgc actttgcgcc gaataaatac 6060ctgtgacgga agatcacttc
gcagaataaa taaatcctgg tgtccctgtt gataccggga 6120agccctgggc caacttttgg
cgaaaatgag acgttgatcg gcacgtaaga ggttccaact 6180ttcaccataa tgaaataaga
tcactaccgg gcgtattttt tgagttatcg agattttcag 6240gagctaagga agctaaaatg
gagaaaaaaa tcactggata taccaccgtt gatatatccc 6300aatggcatcg taaagaacat
tttgaggcat ttcagtcagt tgctcaatgt acctataacc 6360agaccgttca gctggatatt
acggcctttt taaagaccgt aaagaaaaat aagcacaagt 6420tttatccggc ctttattcac
attcttgccc gcctgatgaa tgctcatccg gaattccgta 6480tggcaatgaa agacggtgag
ctggtgatat gggatagtgt tcacccttgt tacaccgttt 6540tccatgagca aactgaaacg
ttttcatcgc tctggagtga ataccacgac gatttccggc 6600agtttctaca catatattcg
caagatgtgg cgtgttacgg tgaaaacctg gcctatttcc 6660ctaaagggtt tattgagaat
atgtttttcg tctcagccaa tccctgggtg agtttcacca 6720gttttgattt aaacgtggcc
aatatggaca acttcttcgc ccccgttttc accatgggca 6780aatattatac gcaaggcgac
aaggtgctga tgccgctggc gattcaggtt catcatgccg 6840tctgtgatgg cttccatgtc
ggcagaatgc ttaatgaatt acaacagtac tgcgatgagt 6900ggcagggcgg ggcgtaatct
agaggatccg gcttactaaa agccagataa cagtatgcgt 6960atttgcgcgc tgatttttgc
ggtataagaa tatatactga tatgtatacc cgaagtatgt 7020caaaaagagg tgtgctatga
agcagcgtat tacagtgaca gttgacagcg acagctatca 7080gttgctcaag gcatatatga
tgtcaatatc tccggtctgg taagcacaac catgcagaat 7140gaagcccgtc gtctgcgtgc
cgaacgctgg aaagcggaaa atcaggaagg gatggctgag 7200gtcgcccggt ttattgaaat
gaacggctct tttgctgacg agaacaggga ctggtgaaat 7260gcagtttaag gtttacacct
ataaaagaga gagccgttat cgtctgtttg tggatgtaca 7320gagtgatatt attgacacgc
ccgggcgacg gatggtgatc cccctggcca gtgcacgtct 7380gctgtcagat aaagtctccc
gtgaacttta cccggtggtg catatcgggg atgaaagctg 7440gcgcatgatg accaccgata
tggccagtgt gccggtctcc gttatcgggg aagaagtggc 7500tgatctcagc caccgcgaaa
atgacatcaa aaacgccatt aacctgatgt tctggggaat 7560ataaatgtca ggctccgtta
tacacagcca gtctgcaggt cgaccatagt gactggatat 7620gttgtgtttt acagtattat
gtagtctgtt ttttatgcaa aatctaattt aatatattga 7680tatttatatc attttacgtt
tctcgttcag ctttcttgta caaagtggtt tgatggccgc 7740taagtaagta agacgtcgag
ctccctatag tgagtcgtat tacactggcc gtcgttttac 7800aacgtcgtga ctgggaaaac
accggtgagc tctaagtaag taacggccgc caccgcggtg 7860gagctttgga cttcttcgcc
agaggtttgg tcaagtctcc aatcaaggtt gtcggcttgt 7920ctaccttgcc agaaatttac
gaaaagatgg aaaagggtca aatcgttggt agatacgttg 7980ttgacacttc taaataagcg
aatttcttat gatttatgat ttttattatt aaataagtta 8040taaaaaaaat aagtgtatac
aaattttaaa gtgactctta ggttttaaaa cgaaaattct 8100tgttcttgag taactctttc
ctgtaggtca ggttgctttc tcaggtatag catgaggtcg 8160ctcttattga ccacacctct
accggcatgc cgagcaaatg cctgcaaatc gctccccatt 8220tcacccaatt gtagatatgc
taactccagc aatgagttga tgaatctcgg tgtgtatttt 8280atgtcctcag aggacaatac
ctgttgtaat cgttcttcca cacggatccg catcaggcga 8340aattgtaaac gttaatattt
tgttaaaatt cgcgttaaat atttgttaaa tcagctcatt 8400ttttaaccaa taggccgaaa
tcggcaaaat cccttataaa tcaaaagaat agaccgagat 8460agggttgagt gttgttccag
tttggaacaa gagtccacta ttaaagaacg tggactccaa 8520cgtcaaaggg cgaaaaaccg
tctatcaggg cgatggccca ctacgtgaac catcacccta 8580atcaagtttt ttggggtcga
ggtgccgtaa agcactaaat cggaacccta aagggagccc 8640ccgatttaga gcttgacggg
gaaagccggc gaacgtggcg agaaaggaag ggaagaaagc 8700gaaaggagcg ggcgctaggg
cgctggcaag tgtagcggtc acgctgcgcg taaccaccac 8760acccgccgcg cttaatgcgc
cgctacaggg cgcgtcccat tcgccattca ctgca 88151817114DNAArtificial
SequenceDescription of Artificial Sequence Synthetic pDEST34
polynucleotide 181atcgagatct cgatcccgcg aaattaatac gactcactat agggagacca
caacggtttc 60cctctagatc acaagtttgt acaaaaaagc tgaacgagaa acgtaaaatg
atataaatat 120caatatatta aattagattt tgcataaaaa acagactaca taatactgta
aaacacaaca 180tatccagtca ctatggcggc cgcattaggc accccaggct ttacacttta
tgcttccggc 240tcgtataatg tgtggatttt gagttaggat ccggcgagat tttcaggagc
taaggaagct 300aaaatggaga aaaaaatcac tggatatacc accgttgata tatcccaatg
gcatcgtaaa 360gaacattttg aggcatttca gtcagttgct caatgtacct ataaccagac
cgttcagctg 420gatattacgg cctttttaaa gaccgtaaag aaaaataagc acaagtttta
tccggccttt 480attcacattc ttgcccgcct gatgaatgct catccggaat tccgtatggc
aatgaaagac 540ggtgagctgg tgatatggga tagtgttcac ccttgttaca ccgttttcca
tgagcaaact 600gaaacgtttt catcgctctg gagtgaatac cacgacgatt tccggcagtt
tctacacata 660tattcgcaag atgtggcgtg ttacggtgaa aacctggcct atttccctaa
agggtttatt 720gagaatatgt ttttcgtctc agccaatccc tgggtgagtt tcaccagttt
tgatttaaac 780gtggccaata tggacaactt cttcgccccc gttttcacca tgggcaaata
ttatacgcaa 840ggcgacaagg tgctgatgcc gctggcgatt caggttcatc atgccgtctg
tgatggcttc 900catgtcggca gaatgcttaa tgaattacaa cagtactgcg atgagtggca
gggcggggcg 960taaacgcgtg gatccggctt actaaaagcc agataacagt atgcgtattt
gcgcgctgat 1020ttttgcggta taagaatata tactgatatg tatacccgaa gtatgtcaaa
aagaggtgtg 1080ctatgaagca gcgtattaca gtgacagttg acagcgacag ctatcagttg
ctcaaggcat 1140atatgatgtc aatatctccg gtctggtaag cacaaccatg cagaatgaag
cccgtcgtct 1200gcgtgccgaa cgctggaaag cggaaaatca ggaagggatg gctgaggtcg
cccggtttat 1260tgaaatgaac ggctcttttg ctgacgagaa cagggactgg tgaaatgcag
tttaaggttt 1320acacctataa aagagagagc cgttatcgtc tgtttgtgga tgtacagagt
gatattattg 1380acacgcccgg gcgacggatg gtgatccccc tggccagtgc acgtctgctg
tcagataaag 1440tctcccgtga actttacccg gtggtgcata tcggggatga aagctggcgc
atgatgacca 1500ccgatatggc cagtgtgccg gtctccgtta tcggggaaga agtggctgat
ctcagccacc 1560gcgaaaatga catcaaaaac gccattaacc tgatgttctg gggaatataa
atgtcaggct 1620cccttataca cagccagtct gcaggtcgac catagtgact ggatatgttg
tgttttacag 1680tattatgtag tctgtttttt atgcaaaatc taatttaata tattgatatt
tatatcattt 1740tacgtttctc gttcagcttt cttgtacaaa gtggtgatta tgtcccctat
actaggttat 1800tggaaaatta agggccttgt gcaacccact cgacttcttt tggaatatct
tgaagaaaaa 1860tatgaagagc atttgtatga gcgcgatgaa ggtgataaat ggcgaaacaa
aaagtttgaa 1920ttgggtttgg agtttcccaa tcttccttat tatattgatg gtgatgttaa
attaacacag 1980tctatggcca tcatacgtta tatagctgac aagcacaaca tgttgggtgg
ttgtccaaaa 2040gagcgtgcag agatttcaat gcttgaagga gcggttttgg atattagata
cggtgtttcg 2100agaattgcat atagtaaaga ctttgaaact ctcaaagttg attttcttag
caagctacct 2160gaaatgctga aaatgttcga agatcgttta tgtcataaaa catatttaaa
tggtgatcat 2220gtaacccatc ctgacttcat gttgtatgac gctcttgatg ttgttttata
catggaccca 2280atgtgcctgg atgcgttccc aaaattagtt tgttttaaaa aacgtattga
agctatccca 2340caaattgata agtacttgaa atccagcaag tatatagcat ggcctttgca
gggctggcaa 2400gccacgtttg gtggtggcga ccatcctcca aaatcggatc tggttccgcg
tccatgggga 2460tccggctgct aacaaagccc gaaaggaagc tgagttggct gctgccaccg
ctgagcgctt 2520cccgataagg gagcaggcca gtaaaagcat tacccgtggt ggggttcccg
agcggccaaa 2580gggagcagac tctaaatctg ccgtcatcga cttcgaaggt tcgaatcctt
cccccaccac 2640catcactttc aaaagtgaat tcgctgagca ataactagca taaccccttg
gggcctctaa 2700acgggtcttg aggggttttt tgctgaaagg aggaactata tccggatatc
cacaggacgg 2760gtgtggtcgc catgatcgcg tagtcgatag tggctccaag tagcgaagcg
agcaggactg 2820ggcggcggcc aaagcggtcg gacagtgctc cgagaacggg tgcgcataga
aattgcatca 2880acgcatatag cgctagcagc acgccatagt gactggcgat gctgtcggaa
tggacgatat 2940cccgcaagag gcccggcagt accggcataa ccaagcctat gcctacagca
tccagggtga 3000cggtgccgag gatgacgatg agcgcattgt tagatttcat acacggtgcc
tgactgcgtt 3060agcaatttaa ctgtgataaa ctaccgcatt aaagcttatc gatgataagc
tgtcaaacat 3120gagaattctt gaagacgaaa gggcctcgtg atacgcctat ttttataggt
taatgtcatg 3180ataataatgg tttcttagac gtcaggtggc acttttcggg gaaatgtgcg
cggaacccct 3240atttgtttat ttttctaaat acattcaaat atgtatccgc tcatgagaca
ataaccctga 3300taaatgcttc aataatattg aaaaaggaag agtatgagta ttcaacattt
ccgtgtcgcc 3360cttattccct tttttgcggc attttgcctt cctgtttttg ctcacccaga
aacgctggtg 3420aaagtaaaag atgctgaaga tcagttgggt gcacgagtgg gttacatcga
actggatctc 3480aacagcggta agatccttga gagttttcgc cccgaagaac gttttccaat
gatgagcact 3540tttaaagttc tgctatgtgg cgcggtatta tcccgtgttg acgccgggca
agagcaactc 3600ggtcgccgca tacactattc tcagaatgac ttggttgagt actcaccagt
cacagaaaag 3660catcttacgg atggcatgac agtaagagaa ttatgcagtg ctgccataac
catgagtgat 3720aacactgcgg ccaacttact tctgacaacg atcggaggac cgaaggagct
aaccgctttt 3780ttgcacaaca tgggggatca tgtaactcgc cttgatcgtt gggaaccgga
gctgaatgaa 3840gccataccaa acgacgagcg tgacaccacg atgcctgcag caatggcaac
aacgttgcgc 3900aaactattaa ctggcgaact acttactcta gcttcccggc aacaattaat
agactggatg 3960gaggcggata aagttgcagg accacttctg cgctcggccc ttccggctgg
ctggtttatt 4020gctgataaat ctggagccgg tgagcgtggg tctcgcggta tcattgcagc
actggggcca 4080gatggtaagc cctcccgtat cgtagttatc tacacgacgg ggagtcaggc
aactatggat 4140gaacgaaata gacagatcgc tgagataggt gcctcactga ttaagcattg
gtaactgtca 4200gaccaagttt actcatatat actttagatt gatttaaaac ttcattttta
atttaaaagg 4260atctaggtga agatcctttt tgataatctc atgaccaaaa tcccttaacg
tgagttttcg 4320ttccactgag cgtcagaccc cgtagaaaag atcaaaggat cttcttgaga
tccttttttt 4380ctgcgcgtaa tctgctgctt gcaaacaaaa aaaccaccgc taccagcggt
ggtttgtttg 4440ccggatcaag agctaccaac tctttttccg aaggtaactg gcttcagcag
agcgcagata 4500ccaaatactg tccttctagt gtagccgtag ttaggccacc acttcaagaa
ctctgtagca 4560ccgcctacat acctcgctct gctaatcctg ttaccagtgg ctgctgccag
tggcgataag 4620tcgtgtctta ccgggttgga ctcaagacga tagttaccgg ataaggcgca
gcggtcgggc 4680tgaacggggg gttcgtgcac acagcccagc ttggagcgaa cgacctacac
cgaactgaga 4740tacctacagc gtgagctatg agaaagcgcc acgcttcccg aagggagaaa
ggcggacagg 4800tatccggtaa gcggcagggt cggaacagga gagcgcacga gggagcttcc
agggggaaac 4860gcctggtatc tttatagtcc tgtcgggttt cgccacctct gacttgagcg
tcgatttttg 4920tgatgctcgt caggggggcg gagcctatgg aaaaacgcca gcaacgcggc
ctttttacgg 4980ttcctggcct tttgctggcc ttttgctcac atgttctttc ctgcgttatc
ccctgattct 5040gtggataacc gtattaccgc ctttgagtga gctgataccg ctcgccgcag
ccgaacgacc 5100gagcgcagcg agtcagtgag cgaggaagcg gaagagcgcc tgatgcggta
ttttctcctt 5160acgcatctgt gcggtatttc acaccgcata tatggtgcac tctcagtaca
atctgctctg 5220atgccgcata gttaagccag tatacactcc gctatcgcta cgtgactggg
tcatggctgc 5280gccccgacac ccgccaacac ccgctgacgc gccctgacgg gcttgtctgc
tcccggcatc 5340cgcttacaga caagctgtga ccgtctccgg gagctgcatg tgtcagaggt
tttcaccgtc 5400atcaccgaaa cgcgcgaggc agctgcggta aagctcatca gcgtggtcgt
gaagcgattc 5460acagatgtct gcctgttcat ccgcgtccag ctcgttgagt ttctccagaa
gcgttaatgt 5520ctggcttctg ataaagcggg ccatgttaag ggcggttttt tcctgtttgg
tcactgatgc 5580ctccgtgtaa gggggatttc tgttcatggg ggtaatgata ccgatgaaac
gagagaggat 5640gctcacgata cgggttactg atgatgaaca tgcccggtta ctggaacgtt
gtgagggtaa 5700acaactggcg gtatggatgc ggcgggacca gagaaaaatc actcagggtc
aatgccagcg 5760cttcgttaat acagatgtag gtgttccaca gggtagccag cagcatcctg
cgatgcagat 5820ccggaacata atggtgcagg gcgctgactt ccgcgtttcc agactttacg
aaacacggaa 5880accgaagacc attcatgttg ttgctcaggt cgcagacgtt ttgcagcagc
agtcgcttca 5940cgttcgctcg cgtatcggtg attcattctg ctaaccagta aggcaacccc
gccagcctag 6000ccgggtcctc aacgacagga gcacgatcat gcgcacccgt ggccaggacc
caacgctgcc 6060cgagatgcgc cgcgtgcggc tgctggagat ggcggacgcg atggatatgt
tctgccaagg 6120gttggtttgc gcattcacag ttctccgcaa gaattgattg gctccaattc
ttggagtggt 6180gaatccgtta gcgaggtgcc gccggcttcc attcaggtcg aggtggcccg
gctccatgca 6240ccgcgacgca acgcggggag gcagacaagg tatagggcgg cgcctacaat
ccatgccaac 6300ccgttccatg tgctcgccga ggcggcataa atcgccgtga cgatcagcgg
tccagtgatc 6360gaagttaggc tggtaagagc cgcgagcgat ccttgaagct gtccctgatg
gtcgtcatct 6420acctgcctgg acagcatggc ctgcaacgcg ggcatcccga tgccgccgga
agcgagaaga 6480atcataatgg ggaaggccat ccagcctcgc gtcgcgaacg ccagcaagac
gtagcccagc 6540gcgtcggccg ccatgccggc gataatggcc tgcttctcgc cgaaacgttt
ggtggcggga 6600ccagtgacga aggcttgagc gagggcgtgc aagattccga ataccgcaag
cgacaggccg 6660atcatcgtcg cgctccagcg aaagcggtcc tcgccgaaaa tgacccagag
cgctgccggc 6720acctgtccta cgagttgcat gataaagaag acagtcataa gtgcggcgac
gatagtcatg 6780ccccgcgccc accggaagga gctgactggg ttgaaggctc tcaagggcat
cggtcgatcg 6840acgctctccc ttatgcgact cctgcattag gaagcagccc agtagtaggt
tgaggccgtt 6900gagcaccgcc gccgcaagga atggtgcatg caaggagatg gcgcccaaca
gtcccccggc 6960cacggggcct gccaccatac ccacgccgaa acaagcgctc atgagcccga
agtggcgagc 7020ccgatcttcc ccatcggtga tgtcggcgat ataggcgcca gcaaccgcac
ctgtggcgcc 7080ggtgatgccg gccacgatgc gtccggcgta gagg
71141825584DNAArtificial SequenceDescription of Artificial
Sequence Synthetic pDONR207 polynucleotide 182gcgagagtag ggaactgcca
ggcatcaaat aaaacgaaag gctcagtcgg aagactgggc 60ctttcgtttt atctgttgtt
tgtcggtgaa cgctctcctg agtaggacaa atccgccggg 120agcggatttg aacgttgtga
agcaacggcc cggagggtgg cgggcaggac gcccgccata 180aactgccagg catcaaacta
agcagaaggc catcctgacg gatggccttt ttgcgtttct 240acaaactctt cctggctagc
ggtaatacgg ttatccacag aatcagggga taacgcagga 300aagaacatgt gagcaaaagg
ccagcaaaag gccaggaacc gtaaaaaggc cgcgttgctg 360gcgtttttcc ataggctccg
cccccctgac gagcatcaca aaaatcgacg ctcaagtcag 420aggtggcgaa acccgacagg
actataaaga taccaggcgt ttccccctgg aagctccctc 480gtgcgctctc ctgttccgac
cctgccgctt accggatacc tgtccgcctt tctcccttcg 540ggaagcgtgg cgctttctca
tagctcacgc tgtaggtatc tcagttcggt gtaggtcgtt 600cgctccaagc tgggctgtgt
gcacgaaccc cccgttcagc ccgaccgctg cgccttatcc 660ggtaactatc gtcttgagtc
caacccggta agacacgact tatcgccact ggcagcagcc 720actggtaaca ggattagcag
agcgaggtat gtaggcggtg ctacagagtt cttgaagtgg 780tggcctaact acggctacac
tagaaggaca gtatttggta tctgcgctct gctgaagcca 840gttaccttcg gaaaaagagt
tggtagctct tgatccggca aacaaaccac cgctggtagc 900ggtggttttt ttgtttgcaa
gcagcagatt acgcgcagaa aaaaaggatc tcaagaagat 960cctttgatct tttctacggg
gtctgacgct cagtggaacg aaaactcacg ttaagggatt 1020ttggtcatga gcttgcgccg
tcccgtcaag tcagcgtaat gctctgccag tgttacaacc 1080aattaaccaa ttctgattag
aaaaactcat cgagcatcaa atgaaactgc aatttattca 1140tatcaggatt atcaatacca
tatttttgaa aaagccgttt ctgtaatgaa ggagaaaact 1200caccgaggca gttccatagg
atggcaagat cctggtatcg gtctgcgatt ccgactcgtc 1260caacatcaat acaacctatt
agtagccaac cactagaact atagctagag tcctgggcga 1320acaaacgatg ctcgccttcc
agaaaaccga ggatgcgaac cacttcatcc ggggtcagca 1380ccaccggcaa gcgccgcgac
ggccgaggtc ttccgatctc ctgaagccag ggcagatccg 1440tgcacagcac cttgccgtag
aagaacagca aggccgccaa tgcctgacga tgcgtggaga 1500ccgaaacctt gcgctcgttc
gccagccagg acagaaatgc ctcgacttcg ctgctgccca 1560aggttgccgg gtgacgcaca
ccgtggaaac ggatgaaggc acgaacccag ttgacataag 1620cctgttcggt tcgtaaactg
taatgcaagt agcgtatgcg ctcacgcaac tggtccagaa 1680ccttgaccga acgcagcggt
ggtaacggcg cagtggcggt tttcatggct tgttatgact 1740gtttttttgt acagtctatg
cctcgggcat ccaagcagca agcgcgttac gccgtgggtc 1800gatgtttgat gttatggagc
agcaacgatg ttacgcagca gcaacgatgt tacgcagcag 1860ggcagtcgcc ctaaaacaaa
gttaggtggc tcaagtatgg gcatcattcg cacatgtagg 1920ctcggccctg accaagtcaa
atccatgcgg gctgctcttg atcttttcgg tcgtgagttc 1980ggagacgtag ccacctactc
ccaacatcag ccggactccg attacctcgg gaacttgctc 2040cgtagtaaga cattcatcgc
gcttgctgcc ttcgaccaag aagcggttgt tggcgctctc 2100gcggcttacg ttctgcccag
gtttgagcag ccgcgtagtg agatctatat ctatgatctc 2160gcagtctccg gcgagcaccg
gaggcagggc attgccaccg cgctcatcaa tctcctcaag 2220catgaggcca acgcgcttgg
tgcttatgtg atctacgtgc aagcagatta cggtgacgat 2280cccgcagtgg ctctctatac
aaagttgggc atacgggaag aagtgatgca ctttgatatc 2340gacccaagta ccgccaccta
acaattcgtt caagccgaga tcggcttccc ggcctaattt 2400cccctcgtca aaaataaggt
tatcaagtga gaaatcacca tgagtgacga ctgaatccgg 2460tgagaatggc aaaagtttat
gcatttcttt ccagacttgt tcaacaggcc agccattacg 2520ctcgtcatca aaatcactcg
catcaaccaa accgttattc attcgtgatt gcgcctgagc 2580gagacgaaat acgcgatcgc
tgttaaaagg acaattacaa acaggaatcg aatgcaaccg 2640gcgcaggaac actgccagcg
catcaacaat attttcacct gaatcaggat attcttctaa 2700tacctggaat gctgtttttc
cggggatcgc agtggtgagt aaccatgcat catcaggagt 2760acggataaaa tgcttgatgg
tcggaagagg cataaattcc gtcagccagt ttagtctgac 2820catctcatct gtaacatcat
tggcaacgct acctttgcca tgtttcagaa acaactctgg 2880cgcatcgggc ttcccataca
agcgatagat tgtcgcacct gattgcccga cattatcgcg 2940agcccattta tacccatata
aatcagcatc catgttggaa tttaatcgcg gcctcgacgt 3000ttcccgttga atatggctca
taacaccccc tgtattactg tttatgtaag cagacagttt 3060tattgttcat gatgatatat
ttttatcttg tgcaatgtaa catcagagat tttgagacac 3120gggccagagc tgcagctgga
tggcaaataa tgattttatt ttgactgata gtgacctgtt 3180cgttgcaaca aattgataag
caatgctttc ttataatgcc aactttgtac aagaaagctg 3240aacgagaaac gtaaaatgat
ataaatatca atatattaaa ttagattttg cataaaaaac 3300agactacata atactgtaaa
acacaacata tccagtcact atgaatcaac tacttagatg 3360gtattagtga cctgtagtcg
actaagttgg cagcatcacc cgacgcactt tgcgccgaat 3420aaatacctgt gacggaagat
cacttcgcag aataaataaa tcctggtgtc cctgttgata 3480ccgggaagcc ctgggccaac
tttggcgaaa atgagacgtt gatcggcacg taagaggttc 3540caactttcac cataatgaaa
taagatcact accgggcgta ttttttgagt tatcgagatt 3600ttcaggagct aaggaagcta
aaatggagaa aaaaatcact ggatatacca ccgttgatat 3660atcccaatgg catcgtaaag
aacattttga ggcatttcag tcagttgctc aatgtaccta 3720taaccagacc gttcagctgg
atattacggc ctttttaaag accgtaaaga aaaataagca 3780caagttttat ccggccttta
ttcacattct tgcccgcctg atgaatgctc atccggaatt 3840ccgtatggca atgaaagacg
gtgagctggt gatatgggat agtgttcacc cttgttacac 3900cgttttccat gagcaaactg
aaacgttttc atcgctctgg agtgaatacc acgacgattt 3960ccggcagttt ctacacatat
attcgcaaga tgtggcgtgt tacggtgaaa acctggccta 4020tttccctaaa gggtttattg
agaatatgtt tttcgtctca gccaatccct gggtgagttt 4080caccagtttt gatttaaacg
tggccaatat ggacaacttc ttcgcccccg ttttcaccat 4140gggcaaatat tatacgcaag
gcgacaaggt gctgatgccg ctggcgattc aggttcatca 4200tgccgtctgt gatggcttcc
atgtcggcag aatgcttaat gaattacaac agtactgcga 4260tgagtggcag ggcggggcgt
aatcgcgtgg atccggctta ctaaaagcca gataacagta 4320tgcgtatttg cgcgctgatt
tttgcggtat aagaatatat actgatatgt atacccgaag 4380tatgtcaaaa agaggtgtgc
tatgaagcag cgtattacag tgacagttga cagcgacagc 4440tatcagttgc tcaaggcata
tatgatgtca atatctccgg tctggtaagc acaaccatgc 4500agaatgaagc ccgtcgtctg
cgtgccgaac gctggaaagc ggaaaatcag gaagggatgg 4560ctgaggtcgc ccggtttatt
gaaatgaacg gctcttttgc tgacgagaac agggactggt 4620gaaatgcagt ttaaggttta
cacctataaa agagagagcc gttatcgtct gtttgtggat 4680gtacagagtg atattattga
cacgcccggg cgacggatgg tgatccccct ggccagtgca 4740cgtctgctgt cagataaagt
ctcccgtgaa ctttacccgg tggtgcatat cggggatgaa 4800agctggcgca tgatgaccac
cgatatggcc agtgtgccgg tctccgttat cggggaagaa 4860gtggctgatc tcagccaccg
cgaaaatgac atcaaaaacg ccattaacct gatgttctgg 4920ggaatataaa tgtcaggctc
ccttatacac agccagtctg caggtcgata cagtagaaat 4980tacagaaact ttatcacgtt
tagtaagtat agaggctgaa aatccagatg aagccgaacg 5040acttgtaaga gaaaagtata
agagttgtga aattgttctt gatgcagatg attttcagga 5100ctatgacact agcgtatatg
aataggtaga tgtttttatt ttgtcacaca aaaaagaggc 5160tcgcacctct ttttcttatt
tctttttatg atttaatacg gcattgagga caatagcgag 5220taggctggat acgacgattc
cgtttgagaa gaacatttgg aaggctgtcg gtcgactaag 5280ttggcagcat cacccgaaga
acatttggaa ggctgtcggt cgactacagg tcactaatac 5340catctaagta gttgattcat
agtgactgga tatgttgtgt tttacagtat tatgtagtct 5400gttttttatg caaaatctaa
tttaatatat tgatatttat atcattttac gtttctcgtt 5460cagctttttt gtacaaagtt
ggcattataa aaaagcattg ctcatcaatt tgttgcaacg 5520aacaggtcac tatcagtcaa
aataaaatca ttatttgggg cccgagatcc atgctagcgt 5580taac
55841837038DNAArtificial
SequenceDescription of Artificial Sequence Synthetic pMAB85
polynucleotide 183gccttacgca tctgtgcggt atttcacacc gcaggcaagt gcacaaacaa
tacttaaata 60aatactactc agtaataacc tatttcttag catttttgac gaaatttgct
attttgttag 120agtcttttac accatttgtc tccacacctc cgcttacatc aacaccaata
acgccattta 180atctaagcgc atcaccaaca ttttctggcg tcagtccacc agctaacata
aaatgtaagc 240tttcggggct ctcttgcctt ccaacccagt cagaaatcga gttccaatcc
aaaagttcac 300ctgtcccacc tgcttctgaa tcaaacaagg gaataaacga atgaggtttc
tgtgaagctg 360cactgagtag tatgttgcag tcttttggaa atacgagtct tttaataact
ggcaaaccga 420ggaactcttg gtattcttgc cacgactcat ctccatgcag ttggacgata
tcaatgccgt 480aatcattgac cagagccaaa acatcctcct taggttgatt acgaaacacg
ccaaccaagt 540atttcggagt gcctgaacta tttttatatg cttttacaag acttgaaatt
ttccttgcaa 600taaccgggtc aattgttctc tttctattgg gcacacatat aatacccagc
aagtcagcat 660cggaatctag agcacattct gcggcctctg tgctctgcaa gccgcaaact
ttcaccaatg 720gaccagaact acctgtgaaa ttaataacag acatactcca agctgccttt
gtgtgcttaa 780tcacgtatac tcacgtgctc aatagtcacc aatgccctcc ctcttggccc
tctccttttc 840ttttttcgac cgaattaatt cttaatcggc aaaaaaagaa aagctccgga
tcaagattgt 900acgtaaggtg acaagctatt tttcaataaa gaatatcttc cactactgcc
atctggcgtc 960ataactgcaa agtacacata tattacgatg ctgtctatta aatgcttcct
atattatata 1020tatagtaatg tcgtttatgg tgcactctca gtacaatctg ctctgatgcc
gcatagttaa 1080gccagccccg acacccgcca acacccgctg acgcgccctg acgggcttgt
ctgctcccgg 1140catccgctta cagacaagct gtgaccgtct ccgggagctg catgtgtcag
aggttttcac 1200cgtcatcacc gaaacgcgcg agacgaaagg gcctcgtgat acgcctattt
ttataggtta 1260atgtcatgat aataatggtt tcttaggacg gatcgcttgc ctgtaactta
cacgcgcctc 1320gtatctttta atgatggaat aatttgggaa tttactctgt gtttatttat
ttttatgttt 1380tgtatttgga ttttagaaag taaataaaga aggtagaaga gttacggaat
gaagaaaaaa 1440aaataaacaa aggtttaaaa aatttcaaca aaaagcgtac tttacatata
tatttattag 1500acaagaaaag cagattaaat agatatacat tcgattaacg ataagtaaaa
tgtaaaatca 1560caggattttc gtgtgtggtc ttctacacag acaagatgaa acaattcggc
attaatacct 1620gagagcagga agagcaagat aaaaggtagt atttgttggc gatcccccta
gagtctttta 1680catcttcgga aaacaaaaac tattttttct ttaatttctt tttttacttt
ctatttttaa 1740tttatatatt tatattaaaa aatttaaatt ataattattt ttatagcacg
tgatgaaaag 1800gacccaggtg gcacttttcg gggaaatgtg cgcggaaccc ctatttgttt
atttttctaa 1860atacattcaa atatgtatcc gctcatgaga caataaccct gataaatgct
tcaataatat 1920tgaaaaagga agagtatgag tattcaacat ttccgtgtcg cccttattcc
cttttttgcg 1980gcattttgcc ttcctgtttt tgctcaccca gaaacgctgg tgaaagtaaa
agatgctgaa 2040gatcagttgg gtgcacgagt gggttacatc gaactggatc tcaacagcgg
taagatcctt 2100gagagttttc gccccgaaga acgttttcca atgatgagca cttttaaagt
tctgctatgt 2160ggcgcggtat tatcccgtat tgacgccggg caagagcaac tcggtcgccg
catacactat 2220tctcagaatg acttggttga gtactcacca gtcacagaaa agcatcttac
ggatggcatg 2280acagtaagag aattatgcag tgctgccata accatgagtg ataacactgc
ggccaactta 2340cttctgacaa cgatcggagg accgaaggag ctaaccgctt tttttcacaa
catgggggat 2400catgtaactc gccttgatcg ttgggaaccg gagctgaatg aagccatacc
aaacgacgag 2460cgtgacacca cgatgcctgt agcaatggca acaacgttgc gcaaactatt
aactggcgaa 2520ctacttactc tagcttcccg gcaacaatta atagactgga tggaggcgga
taaagttgca 2580ggaccacttc tgcgctcggc ccttccggct ggctggttta ttgctgataa
atctggagcc 2640ggtgagcgtg ggtctcgcgg tatcattgca gcactggggc cagatggtaa
gccctcccgt 2700atcgtagtta tctacacgac gggcagtcag gcaactatgg atgaacgaaa
tagacagatc 2760gctgagatag gtgcctcact gattaagcat tggtaactgt cagaccaagt
ttactcatat 2820atactttaga ttgatttaaa acttcatttt taatttaaaa ggatctaggt
gaagatcctt 2880tttgataatc tcatgaccaa aatcccttaa cgtgagtttt cgttccactg
agcgtcagac 2940cccgtagaaa agatcaaagg atcttcttga gatccttttt ttctgcgcgt
aatctgctgc 3000ttgcaaacaa aaaaaccacc gctaccagcg gtggtttgtt tgccggatca
agagctacca 3060actctttttc cgaaggtaac tggcttcagc agagcgcaga taccaaatac
tgtccttcta 3120gtgtagccgt agttaggcca ccacttcaag aactctgtag caccgcctac
atacctcgct 3180ctgctaatcc tgttaccagt ggctgctgcc agtggcgata agtcgtgtct
taccgggttg 3240gactcaagac gatagttacc ggataaggcg cagcggtcgg gctgaacggg
gggttcgtgc 3300acacagccca gcttggagcg aacgacctac accgaactga gatacctaca
gcgtgagcat 3360tgagaaagcg ccacgcttcc cgaagggaga aaggcggaca ggtatccggt
aagcggcagg 3420gtcggaacag gagagcgcac gagggagctt ccagggggga acgcctggta
tctttatagt 3480cctgtcgggt ttcgccacct ctgacttgag cgtcgatttt tgtgatgctc
gtcagggggg 3540ccgagcctat ggaaaaacgc cagcaacgcg gcctttttac ggttcctggc
cttttgctgg 3600ccttttgctc acatgttctt tcctgcgtta tcccctgatt ctgtggataa
ccgtattacc 3660gcctttgagt gagctgatac cgctcgccgc agccgaacga ccgagcgcag
cgagtcagtg 3720agcgaggaag cggaagagcg cccaatacgc aaaccgcctc tccccgcgcg
ttggccgatt 3780cattaatgca gctggcacga caggtttccc gactggaaag cgggcagtga
gcgcaacgca 3840attaatgtga gttacctcac tcattaggca ccccaggctt tacactttat
gcttccggct 3900cctatgttgt gtggaattgt gagcggataa caatttcaca caggaaacag
ctatgaccat 3960gattacgcca agctcggaat taaccctcac taaagggaac aaaagctggg
taccgggccc 4020cccctcgaga tccgggatcg aagaaatgat ggtaaatgaa ataggaaatc
aaggagcatg 4080aaggcaaaag acaaatataa gggtcgaacg aaaaataaag tgaaaagtgt
tgatatgatg 4140tatttggctt tgcggcgccg aaaaaacgag tttacgcaat tgcacaatca
tgctgactct 4200gtggcggacc cgcgctcttg ccggcccggc gataacgctg ggcgtgaggc
tgtgcccggc 4260ggagtttttt gcgcctgcat tttccaaggt ttaccctgcg ctaaggggcg
agattggaga 4320agcaataaga atgccggttg gggttgcgat gatgacgacc acgacaactg
gtgtcattat 4380ttaagttgcc gaaagaacct gagtgcattt gcaacatgag tatactagaa
gaatgagcca 4440agacttgcga gacgcgagtt tgccggtggt gcgaacaata gagcgaccat
gaccttgaag 4500gtgagacgcg cataaccgct agagtacttt gaagaggaaa cagcaatagg
gttgctacca 4560gtataaatag acaggtacat acaacactgg aaatggttgt ctgtttgagt
acgctttcaa 4620ttcatttggg tgtgcacttt attatgttac aatatggaag ggaactttac
acttctccta 4680tgcacatata ttaattaaag tccaatgcta gtagagaagg ggggtaacac
ccctccgcgc 4740tcttttccga tttttttcta aaccgtggaa tatttcggat atccttttgt
tgtttccggg 4800tgtacaatat ggacttcctc ttttctggca accaaaccca tacatcggga
ttcctataat 4860accttcgttg gtctccctaa catgtaggtg gcggagggga gatatacaat
agaacagata 4920ccagacaaga cataatgggc taaacaagac tacaccaatt acactgcctc
attgatggtg 4980gtacataacg aactaatact gtagccctag acttgatagc catcatcata
tcgaagtttc 5040actacccttt ttccatttgc catctattga agtaataata ggcgcatgca
acttcttttc 5100tttttttttc ttttctctct cccccgttgt tgtctcacca tatccgcaat
gacaaaaaaa 5160atgatggaag acactaaagg aaaaaattaa cgacaaagac agcaccaaca
gatgtcgttg 5220ttccagagct gatgaggggt atcttcgaac acacgaaact ttttccttcc
ttcattcacg 5280cacactactc tctaatgagc aacggtatac ggccttcctt ccagttactt
gaatttgaaa 5340taaaaaaagt ttgccgcttt gctatcaagt ataaatagac ctgcaattat
taatcttttg 5400tttcctcgtc attgttctcg ttccctttct tccttgtttc tttttctgca
caatatttca 5460agctatacca agcatacaat caactccaag cttatgccca agaagaagcg
gaaggtctcg 5520agcggcgcca attttaatca aagtgggaat attgctgata gctcattgtc
cttcactttc 5580actaacagta gcaacggtcc gaacctcata acaactcaaa caaattctca
agcgctttca 5640caaccaattg cctcctctaa cgttcatgat aacttcatga ataatgaaat
cacggctagt 5700aaaattgatg atggtaataa ttcaaaacca ctgtcacctg gttggacgga
ccaaactgcg 5760tataacgcgt ttggaatcac tacagggatg tttaatacca ctacaatgga
tgatgtatat 5820aactatctat tcgatgatga agatacccca ccaaacccaa aaaaagaggg
tgggtcgatc 5880acaagtttgt acaaaaaagc aggcttgtcg accccgggaa ttcagatcta
ctagtgcggc 5940cgcacgcgta cccagctttc ttgtacaaag tggtgacgtc gagctcccta
tagtgagtcg 6000tattacactg gccgtcgttt tacaacgtcg tgactgggaa aacaccggtg
agctctaagt 6060aagtaacggc cgccaccgcg gtggagcttt ggacttcttc gccagaggtt
tggtcaagtc 6120tccaatcaag gttgtcggct tgtctacctt gccagaaatt tacgaaaaga
tggaaaaggg 6180tcaaatcgtt ggtagatacg ttgttgacac ttctaaataa gcgaatttct
tatgatttat 6240gatttttatt attaaataag ttataaaaaa aataagtgta tacaaatttt
aaagtgactc 6300ttaggtttta aaacgaaaat tcttgttctt gagtaactct ttcctgtagg
tcaggttgct 6360ttctcaggta tagcatgagg tcgctcttat tgaccacacc tctaccggca
tgccgagcaa 6420atgcctgcaa atcgctcccc atttcaccca attgtagata tgctaactcc
agcaatgagt 6480tgatgaatct cggtgtgtat tttatgtcct cagaggacaa tacctgttgt
aatcgttctt 6540ccacacggat ccgcatcagg cgaaattgta aacgttaata ttttgttaaa
attcgcgtta 6600aatatttgtt aaatcagctc attttttaac caataggccg aaatcggcaa
aatcccttat 6660aaatcaaaag aatagaccga gatagggttg agtgttgttc cagtttggaa
caagagtcca 6720ctattaaaga acgtggactc caacgtcaaa gggcgaaaaa ccgtctatca
gggcgatggc 6780ccactacgtg aaccatcacc ctaatcaagt tttttggggt cgaggtgccg
taaagcacta 6840aatcggaacc ctaaagggag cccccgattt agagcttgac ggggaaagcc
ggcgaacgtg 6900gcgagaaagg aagggaagaa agcgaaagga gcgggcgcta gggcgctggc
aagtgtagcg 6960gtcacgctgc gcgtaaccac cacacccgcc gcgcttaatg cgccgctaca
gggcgcgtcc 7020cattcgccat tcactgca
70381847146DNAArtificial SequenceDescription of Artificial
Sequence Synthetic pMAB86 polynucleotide 184gacgaaaggg cctcgtgata
cgcctatttt tataggttaa tgtcatgata ataatggttt 60cttaggacgg atcgcttgcc
tgtaacttac acgcgcctcg tatcttttaa tgatggaata 120atttgggaat ttactctgtg
tttatttatt tttatgtttt gtatttggat tttagaaagt 180aaataaagaa ggtagaagag
ttacggaatg aagaaaaaaa aataaacaaa ggtttaaaaa 240atttcaacaa aaagcgtact
ttacatatat atttattaga caagaaaagc agattaaata 300gatatacatt cgattaacga
taagtaaaat gtaaaatcac aggattttcg tgtgtggtct 360tctacacaga caagatgaaa
caattcggca ttaatacctg agagcaggaa gagcaagata 420aaaggtagta tttgttggcg
atccccctag agtcttttac atcttcggaa aacaaaaact 480attttttctt taatttcttt
ttttactttc tatttttaat ttatatattt atattaaaaa 540atttaaatta taattatttt
tatagcacgt gatgaaaagg acccaggtgg cacttttcgg 600ggaaatgtgc gcggaacccc
tatttgttta tttttctaaa tacattcaaa tatgtatccg 660ctcatgagac aataaccctg
ataaatgctt caataatatt gaaaaaggaa gagtatgagt 720attcaacatt tccgtgtcgc
ccttattccc ttttttgcgg cattttgcct tcctgttttt 780gctcacccag aaacgctggt
gaaagtaaaa gatgctgaag atcagttggg tgcacgagtg 840ggttacatcg aactggatct
caacagcggt aagatccttg agagttttcg ccccgaagaa 900cgttttccaa tgatgagcac
ttttaaagtt ctgctatgtg gcgcggtatt atcccgtatt 960gacgccgggc aagagcaact
cggtcgccgc atacactatt ctcagaatga cttggttgag 1020tactcaccag tcacagaaaa
gcatcttacg gatggcatga cagtaagaga attatgcagt 1080gctgccataa ccatgagtga
taacactgcg gccaacttac ttctgacaac gatcggagga 1140ccgaaggagc taaccgcttt
ttttcacaac atgggggatc atgtaactcg ccttgatcgt 1200tgggaaccgg agctgaatga
agccatacca aacgacgagc gtgacaccac gatgcctgta 1260gcaatggcaa caacgttgcg
caaactatta actggcgaac tacttactct agcttcccgg 1320caacaattaa tagactggat
ggaggcggat aaagttgcag gaccacttct gcgctcggcc 1380cttccggctg gctggtttat
tgctgataaa tctggagccg gtgagcgtgg gtctcgcggt 1440atcattgcag cactggggcc
agatggtaag ccctcccgta tcgtagttat ctacacgacg 1500ggcagtcagg caactatgga
tgaacgaaat agacagatcg ctgagatagg tgcctcactg 1560attaagcatt ggtaactgtc
agaccaagtt tactcatata tactttagat tgatttaaaa 1620cttcattttt aatttaaaag
gatctaggtg aagatccttt ttgataatct catgaccaaa 1680atcccttaac gtgagttttc
gttccactga gcgtcagacc ccgtagaaaa gatcaaagga 1740tcttcttgag atcctttttt
tctgcgcgta atctgctgct tgcaaacaaa aaaaccaccg 1800ctaccagcgg tggtttgttt
gccggatcaa gagctaccaa ctctttttcc gaaggtaact 1860ggcttcagca gagcgcagat
accaaatact gtccttctag tgtagccgta gttaggccac 1920cacttcaaga actctgtagc
accgcctaca tacctcgctc tgctaatcct gttaccagtg 1980gctgctgcca gtggcgataa
gtcgtgtctt accgggttgg actcaagacg atagttaccg 2040gataaggcgc agcggtcggg
ctgaacgggg ggttcgtgca cacagcccag cttggagcga 2100acgacctaca ccgaactgag
atacctacag cgtgagcatt gagaaagcgc cacgcttccc 2160gaagggagaa aggcggacag
gtatccggta agcggcaggg tcggaacagg agagcgcacg 2220agggagcttc caggggggaa
cgcctggtat ctttatagtc ctgtcgggtt tcgccacctc 2280tgacttgagc gtcgattttt
gtgatgctcg tcaggggggc cgagcctatg gaaaaacgcc 2340agcaacgcgg cctttttacg
gttcctggcc ttttgctggc cttttgctca catgttcttt 2400cctgcgttat cccctgattc
tgtggataac cgtattaccg cctttgagtg agctgatacc 2460gctcgccgca gccgaacgac
cgagcgcagc gagtcagtga gcgaggaagc ggaagagcgc 2520ccaatacgca aaccgcctct
ccccgcgcgt tggccgattc attaatgcag ctggcacgac 2580aggtttcccg actggaaagc
gggcagtgag cgcaacgcaa ttaatgtgag ttacctcact 2640cattaggcac cccaggcttt
acactttatg cttccggctc ctatgttgtg tggaattgtg 2700agcggataac aatttcacac
aggaaacagc tatgaccatg attacgccaa gctcggaatt 2760aaccctcact aaagggaaca
aaagctgggt accgggcccc ccctcgagat ccgggatcga 2820agaaatgatg gtaaatgaaa
taggaaatca aggagcatga aggcaaaaga caaatataag 2880ggtcgaacga aaaataaagt
gaaaagtgtt gatatgatgt atttggcttt gcggcgccga 2940aaaaacgagt ttacgcaatt
gcacaatcat gctgactctg tggcggaccc gcgctcttgc 3000cggcccggcg ataacgctgg
gcgtgaggct gtgcccggcg gagttttttg cgcctgcatt 3060ttccaaggtt taccctgcgc
taaggggcga gattggagaa gcaataagaa tgccggttgg 3120ggttgcgatg atgacgacca
cgacaactgg tgtcattatt taagttgccg aaagaacctg 3180agtgcatttg caacatgagt
atactagaag aatgagccaa gacttgcgag acgcgagttt 3240gccggtggtg cgaacaatag
agcgaccatg accttgaagg tgagacgcgc ataaccgcta 3300gagtactttg aagaggaaac
agcaataggg ttgctaccag tataaataga caggtacata 3360caacactgga aatggttgtc
tgtttgagta cgctttcaat tcatttgggt gtgcacttta 3420ttatgttaca atatggaagg
gaactttaca cttctcctat gcacatatat taattaaagt 3480ccaatgctag tagagaaggg
gggtaacacc cctccgcgct cttttccgat ttttttctaa 3540accgtggaat atttcggata
tccttttgtt gtttccgggt gtacaatatg gacttcctct 3600tttctggcaa ccaaacccat
acatcgggat tcctataata ccttcgttgg tctccctaac 3660atgtaggtgg cggaggggag
atatacaata gaacagatac cagacaagac ataatgggct 3720aaacaagact acaccaatta
cactgcctca ttgatggtgg tacataacga actaatactg 3780tagccctaga cttgatagcc
atcatcatat cgaagtttca ctaccctttt tccatttgcc 3840atctattgaa gtaataatag
gcgcatgcaa cttcttttct ttttttttct tttctctctc 3900ccccgttgtt gtctcaccat
atccgcaatg acaaaaaaaa tgatggaaga cactaaagga 3960aaaaattaac gacaaagaca
gcaccaacag atgtcgttgt tccagagctg atgaggggta 4020tcttcgaaca cacgaaactt
tttccttcct tcattcacgc acactactct ctaatgagca 4080acggtatacg gccttccttc
cagttacttg aatttgaaat aaaaaaagtt tgccgctttg 4140ctatcaagta taaatagacc
tgcaattatt aatcttttgt ttcctcgtca ttgttctcgt 4200tccctttctt ccttgtttct
ttttctgcac aatatttcaa gctataccaa gcatacaatc 4260aactccaagc ttatgcccaa
gaagaagcgg aaggtctcga gcggcgccaa ttttaatcaa 4320agtgggaata ttgctgatag
ctcattgtcc ttcactttca ctaacagtag caacggtccg 4380aacctcataa caactcaaac
aaattctcaa gcgctttcac aaccaattgc ctcctctaac 4440gttcatgata acttcatgaa
taatgaaatc acggctagta aaattgatga tggtaataat 4500tcaaaaccac tgtcacctgg
ttggacggac caaactgcgt ataacgcgtt tggaatcact 4560acagggatgt ttaataccac
tacaatggat gatgtatata actatctatt cgatgatgaa 4620gataccccac caaacccaaa
aaaagagggt gggtcgatca caagtttgta caaaaaagca 4680ggcttgtcga ccccgggaat
tcagatctac tagtgcggcc gcacgcgtac ccagctttct 4740tgtacaaagt ggtgacgtcg
agctctaagt aagtaacggc cgccaccgcg gtggagcttt 4800ggacttcttc gccagaggtt
tggtcaagtc tccaatcaag gttgtcggct tgtctacctt 4860gccagaaatt tacgaaaaga
tggaaaaggg tcaaatcgtt ggtagatacg ttgttgacac 4920ttctaaataa gcgaatttct
tatgatttat gatttttatt attaaataag ttataaaaaa 4980aataagtgta tacaaatttt
aaagtgactc ttaggtttta aaacgaaaat tcttgttctt 5040gagtaactct ttcctgtagg
tcaggttgct ttctcaggta tagcatgagg tcgctcttat 5100tgaccacacc tctaccggca
tgccgagcaa atgcctgcaa atcgctcccc atttcaccca 5160attgtagata tgctaactcc
agcaatgagt tgatgaatct cggtgtgtat tttatgtcct 5220cagaggacaa tacctgttgt
aatcgttctt ccacacggat cccaattcgc cctatagtga 5280gtcgtattac aattcactgg
ccgtcgtttt acaacgtcgt gactgggaaa accctggcgt 5340tacccaactt aatcgccttg
cagcacatcc ccctttcgcc agctggcgta atagcgaaga 5400ggcccgcacc gatcgccctt
cccaacagtt gcgcagcctg aatggcgaat ggacgcgccc 5460tgtagcggcg cattaagcgc
ggcgggtgtg gtggttacgc gcagcgtgac cgctacactt 5520gccagcgccc tagcgcccgc
tcctttcgct ttcttccctt cctttctcgc cacgttcgcc 5580ggctttcccc gtcaagctct
aaatcggggg ctccctttag ggttccgatt tagtgcttta 5640cggcacctcg accccaaaaa
acttgattag ggtgatggtt cacgtagtgg gccatcgccc 5700tgatagacgg tttttcgccc
tttgacgttg gagtccacgt tctttaatag tggactcttg 5760ttccaaactg gaacaacact
caaccctatc tcggtctatt cttttgattt ataagggatt 5820ttgccgattt cggcctattg
gttaaaaaat gagctgattt aacaaaaatt taacgcgaat 5880tttaacaaaa tattaacgtt
tacaatttcc tgatgcggta ttttctcctt acgcatctgt 5940gcggtatttc acaccgcagg
caagtgcaca aacaatactt aaataaatac tactcagtaa 6000taacctattt cttagcattt
ttgacgaaat ttgctatttt gttagagtct tttacaccat 6060ttgtctccac acctccgctt
acatcaacac caataacgcc atttaatcta agcgcatcac 6120caacattttc tggcgtcagt
ccaccagcta acataaaatg taagctttcg gggctctctt 6180gccttccaac ccagtcagaa
atcgagttcc aatccaaaag ttcacctgtc ccacctgctt 6240ctgaatcaaa caagggaata
aacgaatgag gtttctgtga agctgcactg agtagtatgt 6300tgcagtcttt tggaaatacg
agtcttttaa taactggcaa accgaggaac tcttggtatt 6360cttgccacga ctcatctcca
tgcagttgga cgatatcaat gccgtaatca ttgaccagag 6420ccaaaacatc ctccttaggt
tgattacgaa acacgccaac caagtatttc ggagtgcctg 6480aactattttt atatgctttt
acaagacttg aaattttcct tgcaataacc gggtcaattg 6540ttctctttct attgggcaca
catataatac ccagcaagtc agcatcggaa tctagagcac 6600attctgcggc ctctgtgctc
tgcaagccgc aaactttcac caatggacca gaactacctg 6660tgaaattaat aacagacata
ctccaagctg cctttgtgtg cttaatcacg tatactcacg 6720tgctcaatag tcaccaatgc
cctccctctt ggccctctcc ttttcttttt tcgaccgaat 6780taattcttaa tcggcaaaaa
aagaaaagct ccggatcaag attgtacgta aggtgacaag 6840ctatttttca ataaagaata
tcttccacta ctgccatctg gcgtcataac tgcaaagtac 6900acatatatta cgatgctgtc
tattaaatgc ttcctatatt atatatatag taatgtcgtt 6960tatggtgcac tctcagtaca
atctgctctg atgccgcata gttaagccag ccccgacacc 7020cgccaacacc cgctgacgcg
ccctgacggg cttgtctgct cccggcatcc gcttacagac 7080aagctgtgac cgtctccggg
agctgcatgt gtcagaggtt ttcaccgtca tcaccgaaac 7140gcgcga
714618564DNAArtificial
SequenceDescription of Artificial Sequence Synthetic pENTR1A
multiple cloning site oligonucleotide 185act ttg tac aaa aaa gca ggc ttt
aaa gga acc aat tca gtc gac tgg 48Thr Leu Tyr Lys Lys Ala Gly Phe
Lys Gly Thr Asn Ser Val Asp Trp 1 5
10 15 atc cgg tac cga att c
64Ile Arg Tyr Arg Ile
20
18621PRTArtificial
SequenceDescription of Artificial Sequence Synthetic pENTR1A
multiple cloning site peptide 186Thr Leu Tyr Lys Lys Ala Gly Phe Lys Gly
Thr Asn Ser Val Asp Trp 1 5 10
15 Ile Arg Tyr Arg Ile 20 18749DNAArtificial
SequenceDescription of Artificial Sequence Synthetic pENTR1A
multiple cloning site oligonucleotide 187gaattcgcgg ccgcactcga gatatctaga
cccagctttc ttgtacaaa 4918862DNAArtificial
SequenceDescription of Artificial Sequence Synthetic pENTR2B
multiple cloning site oligonucleotide 188ttg tac aaa aaa gca ggc tgg cgc
cgg aac caa ttc agt cga ctg gat 48Leu Tyr Lys Lys Ala Gly Trp Arg
Arg Asn Gln Phe Ser Arg Leu Asp 1 5
10 15 ccg gta ccg aat tc
62Pro Val Pro Asn
20
18920PRTArtificial
SequenceDescription of Artificial Sequence Synthetic pENTR2B
multiple cloning site peptide 189Leu Tyr Lys Lys Ala Gly Trp Arg Arg Asn
Gln Phe Ser Arg Leu Asp 1 5 10
15 Pro Val Pro Asn 20 19050DNAArtificial
SequenceDescription of Artificial Sequence Synthetic pENTR2B
multiple cloning site oligonucleotide 190g aat tcg cgg ccg cac tcg aga
tat cta gac cca gct ttc ttg tac aaa 49Asn Ser Arg Pro His Ser Arg Tyr
Leu Asp Pro Ala Phe Leu Tyr Lys 1 5
10 15 g
5019116PRTArtificial
SequenceDescription of Artificial Sequence Synthetic pENTR2B
multiple cloning site peptide 191Asn Ser Arg Pro His Ser Arg Tyr Leu Asp
Pro Ala Phe Leu Tyr Lys 1 5 10
15 19269DNAArtificial SequenceDescription of Artificial
Sequence Synthetic pENTR3C multiple cloning site oligonucleotide
192ttg tac aaa aaa gca ggc tct tta aag gaa cca att cag tcg act gga
48Leu Tyr Lys Lys Ala Gly Ser Leu Lys Glu Pro Ile Gln Ser Thr Gly
1 5 10 15
tcc ggt acc gaa ttc gatcgc
69Ser Gly Thr Glu Phe
20
19321PRTArtificial SequenceDescription of Artificial Sequence Synthetic
pENTR3C multiple cloning site peptide 193Leu Tyr Lys Lys Ala Gly Ser
Leu Lys Glu Pro Ile Gln Ser Thr Gly 1 5
10 15 Ser Gly Thr Glu Phe 20
19450DNAArtificial SequenceDescription of Artificial Sequence Synthetic
pENTR3C multiple cloning site oligonucleotide 194g aat tcg cgg ccg cac
tcg aga tat cta gac cca gct ttc ttg tac aaa 49Asn Ser Arg Pro His Ser
Arg Tyr Leu Asp Pro Ala Phe Leu Tyr Lys 1 5
10 15 g
5019516PRTArtificial
SequenceDescription of Artificial Sequence Synthetic pENTR3C
multiple cloning site peptide 195Asn Ser Arg Pro His Ser Arg Tyr Leu Asp
Pro Ala Phe Leu Tyr Lys 1 5 10
15 19664DNAArtificial SequenceDescription of Artificial
Sequence Synthetic pENTR4 multiple cloning site oligonucleotide
196ttg tac aaa aaa gca ggc tcc acc atg gga acc aat tca gtc gac tgg
48Leu Tyr Lys Lys Ala Gly Ser Thr Met Gly Thr Asn Ser Val Asp Trp
1 5 10 15
atc cgg tac cga att c
64Ile Arg Tyr Arg Ile
20
19721PRTArtificial SequenceDescription of Artificial Sequence Synthetic
pENTR4 multiple cloning site peptide 197Leu Tyr Lys Lys Ala Gly Ser
Thr Met Gly Thr Asn Ser Val Asp Trp 1 5
10 15 Ile Arg Tyr Arg Ile 20
19850DNAArtificial SequenceDescription of Artificial Sequence Synthetic
pENTR4 multiple cloning site oligonucleotide 198g aat tcg cgg ccg cac
tcg aga tat cta gac cca gct ttc ttg tac aaa 49Asn Ser Arg Pro His Ser
Arg Tyr Leu Asp Pro Ala Phe Leu Tyr Lys 1 5
10 15 g
5019916PRTArtificial
SequenceDescription of Artificial Sequence Synthetic pENTR4 multiple
cloning site peptide 199Asn Ser Arg Pro His Ser Arg Tyr Leu Asp Pro Ala
Phe Leu Tyr Lys 1 5 10
15 20066DNAArtificial SequenceDescription of Artificial Sequence
Synthetic pENTR5 multiple cloning site oligonucleotide 200ttg tac
aaa aaa gca ggc ttt cat atg gga acc aat tca gtc gac tgg 48Leu Tyr
Lys Lys Ala Gly Phe His Met Gly Thr Asn Ser Val Asp Trp 1
5 10 15 atc cgg tac cga att
cgc 66Ile Arg Tyr Arg Ile
20
20121PRTArtificial
SequenceDescription of Artificial Sequence Synthetic pENTR5 multiple
cloning site peptide 201Leu Tyr Lys Lys Ala Gly Phe His Met Gly Thr Asn
Ser Val Asp Trp 1 5 10
15 Ile Arg Tyr Arg Ile 20 20251DNAArtificial
SequenceDescription of Artificial Sequence Synthetic pENTR5 multiple
cloning site oligonucleotide 202agaattcgcg gccgcactcg agatatctag
acccagcttt cttgtacaaa g 5120363DNAArtificial
SequenceDescription of Artificial Sequence Synthetic pENTR6 multiple
cloning site oligonucleotide 203ttg tac aaa aaa gca ggc tgc atg cga acc
aat tca gtc gac tgg atc 48Leu Tyr Lys Lys Ala Gly Cys Met Arg Thr
Asn Ser Val Asp Trp Ile 1 5 10
15 cgg tac cga att cgc
63Arg Tyr Arg Ile
20
20420PRTArtificial SequenceDescription of Artificial Sequence
Synthetic pENTR6 multiple cloning site peptide 204Leu Tyr Lys Lys
Ala Gly Cys Met Arg Thr Asn Ser Val Asp Trp Ile 1 5
10 15 Arg Tyr Arg Ile 20
20551DNAArtificial SequenceDescription of Artificial Sequence Synthetic
pENTR6 multiple cloning site oligonucleotide 205agaattcgcg gccgcactcg
agatatctag acccagcttt cttgtacaaa g 5120684DNAArtificial
SequenceDescription of Artificial Sequence Synthetic pENTR7 multiple
cloning site oligonucleotide 206ttg tac aaa aaa gca ggc ttt gaa aac ctg
tat ttt caa gga acc gtt 48Leu Tyr Lys Lys Ala Gly Phe Glu Asn Leu
Tyr Phe Gln Gly Thr Val 1 5 10
15 tca tgc atc gtc gac tgg atc cgg tac cga att cgc
84Ser Cys Ile Val Asp Trp Ile Arg Tyr Arg Ile
20 25
20727PRTArtificial SequenceDescription of Artificial
Sequence Synthetic pENTR7 multiple cloning site peptide 207Leu Tyr
Lys Lys Ala Gly Phe Glu Asn Leu Tyr Phe Gln Gly Thr Val 1 5
10 15 Ser Cys Ile Val Asp Trp Ile
Arg Tyr Arg Ile 20 25
20851DNAArtificial SequenceDescription of Artificial Sequence Synthetic
pENTR7 multiple cloning site oligonucleotide 208agaattcgcg gccgcactcg
agatatctag acccagcttt cttgtacaaa g 5120981DNAArtificial
SequenceDescription of Artificial Sequence Synthetic pENTR8 multiple
cloning site oligonucleotide 209ttg tac aaa aaa gca ggc ttt gaa aac ctg
tat ttt caa gga acc atg 48Leu Tyr Lys Lys Ala Gly Phe Glu Asn Leu
Tyr Phe Gln Gly Thr Met 1 5 10
15 gac cta gtc gac tgg atc cgg tac cga att cgc
81Asp Leu Val Asp Trp Ile Arg Tyr Arg Ile
20 25
21026PRTArtificial SequenceDescription of
Artificial Sequence Synthetic pENTR8 multiple cloning site peptide
210Leu Tyr Lys Lys Ala Gly Phe Glu Asn Leu Tyr Phe Gln Gly Thr Met 1
5 10 15 Asp Leu Val Asp
Trp Ile Arg Tyr Arg Ile 20 25
21151DNAArtificial SequenceDescription of Artificial Sequence Synthetic
pENTR8 multiple cloning site oligonucleotide 211agaattcgcg gccgcactcg
agatatctag acccagcttt cttgtacaaa g 5121281DNAArtificial
SequenceDescription of Artificial Sequence Synthetic pENTR9 multiple
cloning site oligonucleotide 212ttg tac aaa aaa gca ggc ttt gaa aac ctg
tat ttt caa gga cat atg 48Leu Tyr Lys Lys Ala Gly Phe Glu Asn Leu
Tyr Phe Gln Gly His Met 1 5 10
15 aga tct gtc gac tgg atc cgg tac cga att cgc
81Arg Ser Val Asp Trp Ile Arg Tyr Arg Ile
20 25
21326PRTArtificial SequenceDescription of Artificial
Sequence Synthetic pENTR9 multiple cloning site peptide 213Leu Tyr
Lys Lys Ala Gly Phe Glu Asn Leu Tyr Phe Gln Gly His Met 1 5
10 15 Arg Ser Val Asp Trp Ile Arg
Tyr Arg Ile 20 25 21451DNAArtificial
SequenceDescription of Artificial Sequence Synthetic pENTR9 multiple
cloning site oligonucleotide 214agaattcgcg gccgcactcg agatatctag
acccagcttt cttgtacaaa g 5121584DNAArtificial
SequenceDescription of Artificial Sequence Synthetic pENTR10
multiple cloning site oligonucleotide 215ttg tac aaa aaa gca ggc ttc gaa
cta agg aaa tac tta cat atg gga 48Leu Tyr Lys Lys Ala Gly Phe Glu
Leu Arg Lys Tyr Leu His Met Gly 1 5
10 15 acc aat tca gtc gac tgg atc cgg tac cga
att cgc 84Thr Asn Ser Val Asp Trp Ile Arg Tyr Arg
Ile 20 25
21627PRTArtificial SequenceDescription
of Artificial Sequence Synthetic pENTR10 multiple cloning site
peptide 216Leu Tyr Lys Lys Ala Gly Phe Glu Leu Arg Lys Tyr Leu His Met
Gly 1 5 10 15 Thr
Asn Ser Val Asp Trp Ile Arg Tyr Arg Ile 20
25 21751DNAArtificial SequenceDescription of Artificial Sequence
Synthetic pENTR10 multiple cloning site oligonucleotide
217agaattcgcg gccgcactcg agatatctag acccagcttt cttgtacaaa g
5121888DNAArtificial SequenceDescription of Artificial Sequence Synthetic
pENTR11 multiple cloning site oligonucleotide 218ttg tac aaa aaa gca
ggc ttc gaa gga gat aga acc aat tct cta agg 48Leu Tyr Lys Lys Ala
Gly Phe Glu Gly Asp Arg Thr Asn Ser Leu Arg 1 5
10 15 aaa tac tta acc atg gtc gac
tgg atc cgg tac cga att c 88Lys Tyr Leu Thr Met Val Asp
Trp Ile Arg Tyr Arg Ile 20
25 21929PRTArtificial
SequenceDescription of Artificial Sequence Synthetic pENTR11
multiple cloning site peptide 219Leu Tyr Lys Lys Ala Gly Phe Glu Gly Asp
Arg Thr Asn Ser Leu Arg 1 5 10
15 Lys Tyr Leu Thr Met Val Asp Trp Ile Arg Tyr Arg Ile
20 25 22050DNAArtificial
SequenceDescription of Artificial Sequence Synthetic pENTR11
multiple cloning site oligonucleotide 220g aat tcg cgg ccg cac tcg aga
tat cta gac cca gct ttc ttg tac aaa 49 Asn Ser Arg Pro His Ser Arg
Tyr Leu Asp Pro Ala Phe Leu Tyr Lys 1 5
10 15 g
5022116PRTArtificial
SequenceDescription of Artificial Sequence Synthetic pENTR11
multiple cloning site peptide 221Asn Ser Arg Pro His Ser Arg Tyr Leu Asp
Pro Ala Phe Leu Tyr Lys 1 5 10
15 222120DNAArtificial SequenceDescription of Artificial
Sequence Synthetic pDEST1 polynucleotide 222atgagctgtt gacaattaat
catccggctc gtataatgtg tggaattgtg agcggataac 60aatttcacac aggaaacaga
caggtatagg atcacaagtt tgtacaaaaa agctgaacga 120223153DNAArtificial
SequenceDescription of Artificial Sequence Synthetic pDEST2
polynucleotide 223aatattctga aatgagctgt tgacaattaa tcatccggtc cgtataatct
gtggaattgt 60gagcggataa caatttcaca caggaaacag acc atg tcg tac tac cat
cac cat 114 Met Ser Tyr Tyr His His His
1 5 cac cat cac ggc atc
aca agt ttgtacaaaa aagctgaa 153His His His Gly Ile
Thr Ser 10
22414PRTArtificial
SequenceDescription of Artificial Sequence Synthetic pDEST2 peptide
224Met Ser Tyr Tyr His His His His His His Gly Ile Thr Ser 1
5 10 225153DNAArtificial
SequenceDescription of Artificial Sequence Synthetic pDEST3
polynucleotide 225cggttctggc aaatattctg aaatgagctg ttgacaatta atcatcggct
cgtataatgt 60gtggaattgt gagcggataa caatttcaca caggaaacag tattc atg
tcc cct ata 117 Met Ser Pro Ile
1 cta ggttattgga aaattaaggg
ccttgtgcaa ccc 153Leu
5
2265PRTArtificial
SequenceDescription of Artificial Sequence Synthetic pDEST3 peptide
226Met Ser Pro Ile Leu 1 5 227102DNAArtificial
SequenceDescription of Artificial Sequence Synthetic pDEST3
polynucleotide 227ctggttccg cgt gga tct cgt cgt gca tct gtt gga tcc cca
tca aca agt 51 Arg Gly Ser Arg Arg Ala Ser Val Gly Ser Pro Ser Thr
Ser 1 5 10
ttg tac aaa aaa gctgaacgag aaacgtaaaa tgatataaat atcaatata
102Leu Tyr Lys Lys
15
22818PRTArtificial SequenceDescription of Artificial Sequence
Synthetic pDEST3 peptide 228Arg Gly Ser Arg Arg Ala Ser Val Gly Ser
Pro Ser Thr Ser Leu Tyr 1 5 10
15 Lys Lys 229204DNAArtificial SequenceDescription of
Artificial Sequence Synthetic pDEST4 polynucleotide 229gcaaatattc
tgaaatgagc tgttgacaat taatcatccg gtccgtataa tctgtggaat 60tgtgagcgga
taacaatttc acacaggaaa cagacc atg ggt cat cat cat cat 114 Met Gly
His His His His 1
5 cat cac gat tac gat atc cca acg acc gaa aac ctg tat ttt
cag ggc 162His His Asp Tyr Asp Ile Pro Thr Thr Glu Asn Leu Tyr Phe
Gln Gly 10 15 20
gcc cat atg agc gat aaa att att cac ctg act gac gac agt
204Ala His Met Ser Asp Lys Ile Ile His Leu Thr Asp Asp Ser
25 30 35
23036PRTArtificial SequenceDescription of Artificial Sequence
Synthetic pDEST4 polypeptide 230Met Gly His His His His His His Asp
Tyr Asp Ile Pro Thr Thr Glu 1 5 10
15 Asn Leu Tyr Phe Gln Gly Ala His Met Ser Asp Lys Ile Ile
His Leu 20 25 30
Thr Asp Asp Ser 35 231204DNAArtificial
SequenceDescription of Artificial Sequence Synthetic pDEST5
polynucleotide 231aggcacccca ggctttacac tttatgcttc cggctcgtat gttgtgtgga
attgtgagcg 60gataacaatt tcacacagga aacagctatg accatgatta cgccaagctc
taatacgact 120cactataggg aaagctggta cgcctgcagg taccggtccg gaattcccgg
gtcgacgatc 180acaagtttgt acaaaaaagc tgaa
204232204DNAArtificial SequenceDescription of Artificial
Sequence Synthetic pDEST5 polynucleotide 232tttacgtttc tcgttcagct
ttcttgtaca aagtggtgat cactagtcgg cggccgctct 60agaggatcca agcttacgta
cgcgtgcatg cgacgtcata gctcttctat agtgtcacct 120aaattcaatt cactggccgt
cgttttacaa cgtcgtgact gggaaaaccc tggcgttacc 180caacttaatc gccttgcagc
acat 204233204DNAArtificial
SequenceDescription of Artificial Sequence Synthetic pDEST6
polynucleotide 233taacgccagg gttttcccag tcacgacgtt gtaaaacgac ggccagtgaa
ttgaatttag 60gtgacactat agaagagcta tgacgtcgca tgcacgcgta cgtaagcttg
gatcctctag 120agcggccgcc gactagtgat cacaagtttg tacaaaaaag ctgaacgaga
aacgtaaaat 180gatataaata tcaatatatt aaat
204234255DNAArtificial SequenceDescription of Artificial
Sequence Synthetic pDEST6 polynucleotide 234tatttatatc attttacgtt
tctcgttcag ctttcttgta caaagtggtg atcgtcgacc 60cgggaattcc ggaccggtac
ctgcaggcgt accagctttc cctatagtga gtcgtattag 120agcttggcgt aatcatggtc
atagctgttt cctgtgtgaa attgttatcc gctcacaatt 180ccacacaaca tacgagccgg
aagcataaag tgtaaagcct ggggtgccta atgagtgagc 240taactcacat taatt
255235306DNAArtificial
SequenceDescription of Artificial Sequence Synthetic pDEST7
polynucleotide 235ccattgacgc aaatgggcgg taggcgtgta cggtgggagg tctatataag
cagagctcgt 60ttagtgaacc gtcagatcgc ctggagacgc catccacgct gttttgacct
ccatagaaga 120caccgggacc gatccagcct ccggactcta gcctaggccg cggagcggat
aacaatttca 180cacaggaaac agctatgacc actaggcttt tgcaaaaagc tatttaggtg
acactataga 240aggtacgcct gcaggtaccg gtccggaatt cccatcacaa gtttgtacaa
aaaagctgaa 300cgagaa
306236204DNAArtificial SequenceDescription of Artificial
Sequence Synthetic pDEST8 polynucleotide 236cgtatactcc ggaatattaa
tagatcatgg agataattaa aatgataacc atctcgcaaa 60taaataagta ttttactgtt
ttcgtaacag ttttgtaata aaaaaaccta taaatattcc 120ggattattca taccgtccca
ccatcgggcg cggatcatca caagtttgta caaaaaagct 180gaacgagaaa cgtaaaatga
tata 204237153DNAArtificial
SequenceDescription of Artificial Sequence Synthetic pDEST9
polynucleotide 237ttggcgaggg acattaaggc gtttaagaaa ttgagaggac ctgttataca
cctctacggc 60ggtcctagat tggtgcgtta atacacagaa ttctgattgg atcccggtcc
gaagcgcgct 120ttcccatcaa caagtttgta caaaaaagct gaa
153238204DNAArtificial SequenceDescription of Artificial
Sequence Synthetic pDEST10 polynucleotide 238aaataagtat tttactgttt
tcgtaacagt tttgtaataa aaaaacctat aaatattccg 60gattattcat accgtcccac
catcgggcgc ggatctcggt ccgaaacc atg tcg tac 117 Met Ser Tyr
1
tac cat cac cat cac cat cac gat tac gat atc cca acg acc gaa aac
165Tyr His His His His His His Asp Tyr Asp Ile Pro Thr Thr Glu Asn
5 10 15
ctg tat ttt cag ggc atc aca agt ttg tac aaa aaa gct
204Leu Tyr Phe Gln Gly Ile Thr Ser Leu Tyr Lys Lys
20 25 30
23931PRTArtificial SequenceDescription of Artificial Sequence
Synthetic pDEST10 polypeptide 239Met Ser Tyr Tyr His His His His His
His Asp Tyr Asp Ile Pro Thr 1 5 10
15 Thr Glu Asn Leu Tyr Phe Gln Gly Ile Thr Ser Leu Tyr Lys
Lys 20 25 30
240204DNAArtificial SequenceDescription of Artificial Sequence Synthetic
pDEST11 polynucleotide 240tagtgaaccg tcagatcgcc tggagacgcc atccacgctg
ttttgacctc catagaagac 60accgggaccg atccagcctc cgcggccccg aattcgagct
cggtacccgg ggatcctcta 120gagtcgaggt cgacggtatc gataagcttg atatcaacaa
gtttgtacaa aaaagctgaa 180cgagaaacgt aaaatgatat aaat
204241255DNAArtificial SequenceDescription of
Artificial Sequence Synthetic pDEST12.2 polynucleotide 241accgtcagat
cgcctggaga cgccatccac gctgttttga cctccataga agacaccggg 60accgatccag
cctccggact ctagcctagg ccgcggagcg gataacaatt tcacacagga 120aacagctatg
accattaggc ctttgcaaaa agctatttag gtgacactat agaaggtacg 180cctgcaggta
ccggtccgga attcccatca acaagtttgt acaaaaaagc tgaacgagaa 240acgtaaaatg
atata
255242300DNAArtificial SequenceDescription of Artificial Sequence
Synthetic pDEST13 polynucleotide 242tgggcaaacc aagacagcta aagatctctc
acctaccaaa caatgccccc ctgcaaaaaa 60taaattcata taaaaaacat acagataacc
atctgcggtg ataaattatc tctggcggtg 120ttgacataaa taccactggc ggtgatactg
agcacatcag caggacgcac tgaccaccat 180gaaggtgacg ctcttaaaaa ttaagccctg
aagaagggca gcattcaaag cagaaggctt 240tggggtgtgt gatacgaaac gaagcattgg
gatcatcaca agtttgtaca aaaaagctga 300243120DNAArtificial
SequenceDescription of Artificial Sequence Synthetic pDEST14
polynucleotide 243tgccggccac gatgcgtccg gcgtagagga tcgagatctc gatcccgcga
aattaatacg 60actcactata gggagaccac aacggtttcc ctctagatca caagtttgta
caaaaaagct 120244204DNAArtificial SequenceDescription of Artificial
Sequence Synthetic pDEST15 polynucleotide 244natcgagatc tcgatcccgc
gaaattaata cgactcacta tagggagacc acaacggttt 60ccctctagaa ataattttgt
ttaactttaa gaaggagata tacat atg tcc cct ata 117 Met Ser Pro Ile
1
cta ggttattgga aaattaaggg ccttgtgcaa cccactcgac ttcttttgga
170Leu
5
atatcttgaa gaaaaatatg aagagcattt gtat
2042455PRTArtificial SequenceDescription of Artificial Sequence Synthetic
pDEST15 peptide 245Met Ser Pro Ile Leu 1 5
246153DNAArtificial SequenceDescription of Artificial Sequence Synthetic
pDEST15 polynucleotide 246cagggctggc aagccacgtt tggtggtggc gaccatcctc
caaaatcgga tctggttccg 60cgtccatgg tcg aat caa aca agt ttg tac aaa aaa
gct gaacgagaaa 109 Ser Asn Gln Thr Ser Leu Tyr Lys Lys Ala
1 5 10
cgtaaaatga tataaatatc aatatattaa attagatttt gcat
15324710PRTArtificial SequenceDescription of Artificial
Sequence Synthetic pDEST15 peptide 247Ser Asn Gln Thr Ser Leu Tyr
Lys Lys Ala 1 5 10 248153DNAArtificial
SequenceDescription of Artificial Sequence Synthetic pDEST16
multiple cloning site polynucleotide 248gatctcgatc ccgcgaaatt aatacgactc
actataggga gaccacaacg gtttccctct 60agaaataatt ttgtttaact ttaagaagga
gatatacat atg agc gat aaa 111 Met Ser Asp Lys
1 attattcacc
tgactgacga cagttttgac acggatgtac tc
1532494PRTArtificial SequenceDescription of Artificial Sequence Synthetic
pDEST16 multiple cloning site peptide 249Met Ser Asp Lys 1
250153DNAArtificial SequenceDescription of Artificial Sequence
Synthetic pDEST16 multiple cloning site polynucleotide 250gtggcggcaa
ccaaagtggg tgcactgtct aaaggtcagt tgaaagagtt cctcgacgct 60aacctggccg
gttctggttc t ggt gat gac gat gac aag atc aca agt ttg 111 Gly Asp
Asp Asp Asp Lys Ile Thr Ser Leu 1
5 10 tac aaa aaa gct gaacgagaaa cgtaaaatga
tataaatatc 153Tyr Lys Lys Ala
25114PRTArtificial SequenceDescription
of Artificial Sequence Synthetic pDEST16 multiple cloning site
peptide 251Gly Asp Asp Asp Asp Lys Ile Thr Ser Leu Tyr Lys Lys Ala 1
5 10 252153DNAArtificial
SequenceDescription of Artificial Sequence Synthetic pDEST17
multiple cloning site polynucleotide 252gatcccgcga aattaatacg actcactata
gggagaccac aacggtttcc ctctagaaat 60aattttgttt aactttaaga aggagatata
cat atg tcg tac tac cat cac cat 114 Met Ser Tyr Tyr His His His
1 5 cac
cat cac ctc gaa tca aca agt ttg tac aaa aaa gct 153His
His His Leu Glu Ser Thr Ser Leu Tyr Lys Lys Ala
10 15 20
25320PRTArtificial SequenceDescription of Artificial Sequence Synthetic
pDEST17 multiple cloning site peptide 253Met Ser Tyr Tyr His His His
His His His Leu Glu Ser Thr Ser Leu 1 5
10 15 Tyr Lys Lys Ala 20
254420DNAArtificial SequenceDescription of Artificial Sequence Synthetic
pDEST18 p10 Promoter polynucleotide 254gaagacctcg gccgtcgcgg
cgcttgccgg tggtgctgac cccggatgaa gtggttcgca 60tcctcggttt tctggaaggc
gagcatcgtt tgttcgccca ggactctagc tatagttcta 120gtggttggct acgtatcgag
caagaaaata aaacgccaaa cgcgttggag tcttgtgtgc 180tatttttaca aagattcaga
aatacgcatc acttacaaca agggggacta tgaaattatg 240cattttgagg atgccgggac
ctttaattca acccaacaca atatattata gttaaataag 300aattatttat caaatcattt
gtatattaat taaaatacta tactgtaaat tacattttat 360ttacaatgag gatcatcaca
agtttgtaca aaaaagctga acgagaaacg taaaatgata 420255300DNAArtificial
SequenceDescription of Artificial Sequence Synthetic pDEST19 39K
Promoter polynucleotide 255ggtgacgccg tcatctttcc attgtaacgt aaatggcaac
ttgtagatga acgcgctgtc 60aaaaaaccgg ccagtttctt ccacaaactc gcgcacggct
gtctcgtaaa cttttgcgtc 120gcaacaatcg cgatgacctc gtggtatgga aattttttct
aaaaaagtgt cgttcatgtc 180ggcggcggcg ttcgcgctcc ggtacgcgcg acgggcacac
agcaggacag ccttgtccgg 240ctcgattatc ataaacaatc ctgcaggcat gcaagctgga
tcatcacaag tttgtacaaa 300256204DNAArtificial SequenceDescription of
Artificial Sequence Synthetic pDEST20 Polyhedron Promoter
polynucleotide 256ggctacgtat actccggaat attaatagat catggagata attaaaatga
taaccatctc 60gcaaataaat aagtatttta ctgttttcgt aacagttttg taataaaaaa
acctataaat 120attccggatt attcataccg tcccaccatc gggcgcggat cc atg gcc
cct ata 174 Met Ala Pro Ile
1 ctaggttatt ggaaaattaa gggccttgtg
2042574PRTArtificial SequenceDescription of
Artificial Sequence Synthetic pDEST20 Polyhedron Promoter peptide
257Met Ala Pro Ile 1 25895DNAArtificial SequenceDescription
of Artificial Sequence Synthetic pDEST20 Polyhedron Promoter
oligonucleotide 258tcg gat ctg gtt ccg cgt cat aat caa aca agt ttg tac
aaa aaa gct 48Ser Asp Leu Val Pro Arg His Asn Gln Thr Ser Leu Tyr
Lys Lys Ala 1 5 10
15 gaacgagaaa cgtaaaatga tataaatatc aatatattaa attagat
9525916PRTArtificial SequenceDescription of Artificial
Sequence Synthetic pDEST20 Polyhedron Promoter peptide 259Ser Asp
Leu Val Pro Arg His Asn Gln Thr Ser Leu Tyr Lys Lys Ala 1 5
10 15 260204DNAArtificial
SequenceDescription of Artificial Sequence Synthetic pDEST21
Promoter region polynucleotide 260ttgccgcttt gctatcaagt ataaatagac
ctgcaattat taatcttttg tttcctcgtc 60attgttctcg ttccctttct tccttgtttc
tttttctgca caatatttca agctatacca 120agcatacaat caactccaag cttgaagcaa
gcctcctgaa ag atg aag cta ctg 174 Met Lys Leu Leu
1 tct
tct atcgaacaag catgcgatat ttgc 204Ser
Ser 5
2616PRTArtificial SequenceDescription of Artificial Sequence Synthetic
pDEST21 Promoter region peptide 261Met Lys Leu Leu Ser Ser 1
5 262102DNAArtificial SequenceDescription of Artificial Sequence
Synthetic pDEST21 Promoter region polynucleotide 262gaagagagta
gtaacaaagg tcaaagacag ttgact gta tcg tcg agg tcg aat 54 Val Ser
Ser Arg Ser Asn 1
5 caa aca agt ttg tac aaa aaa gct gaacgagaaa
cgtaaaatga tata 102Gln Thr Ser Leu Tyr Lys Lys Ala
10
26314PRTArtificial SequenceDescription of
Artificial Sequence Synthetic pDEST21 Promoter region peptide 263Val
Ser Ser Arg Ser Asn Gln Thr Ser Leu Tyr Lys Lys Ala 1 5
10 264255DNAArtificial SequenceDescription
of Artificial Sequence Synthetic pDEST22 Promoter region
polynucleotide 264acgcacacta ctctctaatg agcaacggta tacggccttc cttccagtta
cttgaatttg 60aaataaaaaa agtttgccgc tttgctatca agtataaata gacctgcaat
tattaatctt 120ttgtttcctc gtcattgttc tcgttccctt tcttccttgt ttctttttct
gcacaatatt 180tcaagctata ccaagcatac aatcaactcc aagctt atg ccc aag aag
228 Met Pro Lys Lys
1 aagcggaagg tctcgagcgg cgccaat
2552654PRTArtificial SequenceDescription of
Artificial Sequence Synthetic pDEST22 Promoter region peptide 265Met
Pro Lys Lys 1 26682DNAArtificial SequenceDescription of
Artificial Sequence Synthetic pDEST22 oligonucleotide 266gaagataccc
caccaaaccc aaaaaaa gag ggt ggg tcg aat caa aca agt ttg 54 Glu
Gly Gly Ser Asn Gln Thr Ser Leu 1
5 tac aaa aaa gct gaacgagaaa cgtaaa
82Tyr Lys Lys Ala
10
26713PRTArtificial
SequenceDescription of Artificial Sequence Synthetic pDEST22 peptide
267Glu Gly Gly Ser Asn Gln Thr Ser Leu Tyr Lys Lys Ala 1 5
10 268102DNAArtificial SequenceDescription
of Artificial Sequence Synthetic pDEST23 T7 promoter polynucleotide
268atcccgcgaa attaatacga ctcactatag ggagaccaca acggtttccc tctagatcac
60aagtttgtac aaaaaagctg aacgagaaac gtaaaatgat at
102269153DNAArtificial SequenceDescription of Artificial Sequence
Synthetic pDEST23 T7 promoter polynucleotide 269tttttatgca
aaatctaatt taatatattg atatttatat cattttacgt ttctcgttca 60gct ttc ttg
tac aaa gtg gtg att atg tcg tac tac cat cac cat cac 108Ala Phe Leu
Tyr Lys Val Val Ile Met Ser Tyr Tyr His His His His 1
5 10 15 cat cac ctc
gat gag caa taactagcat aaccccttgg ggcctct 153His His Leu
Asp Glu Gln
20
27022PRTArtificial SequenceDescription of Artificial Sequence Synthetic
pDEST23 T7 promoter peptide 270Ala Phe Leu Tyr Lys Val Val Ile Met Ser
Tyr Tyr His His His His 1 5 10
15 His His Leu Asp Glu Gln 20
271102DNAArtificial SequenceDescription of Artificial Sequence Synthetic
pDEST24 T7 promoter polynucleotide 271atcgagatct cgatcccgcg
aaattaatac gactcactat agggagacca caacggtttc 60cctctagatc acaagtttgt
acaaaaaagc tgaacgagaa ac 102272102DNAArtificial
SequenceDescription of Artificial Sequence Synthetic pDEST24 T7
promoter polynucleotide 272tcattttacg tttctcgttc a gct ttc ttg tac aaa
gtg gtg att atg tcc 51 Ala Phe Leu Tyr Lys Val Val Ile Met Ser
1 5 10
cct ata cta ggttattgga aaattaaggg ccttgtgcaa cccactcgac tt
102Pro Ile Leu
27313PRTArtificial SequenceDescription of Artificial Sequence
Synthetic pDEST24 T7 promoter peptide 273Ala Phe Leu Tyr Lys Val Val
Ile Met Ser Pro Ile Leu 1 5 10
274102DNAArtificial SequenceDescription of Artificial Sequence
Synthetic pDEST25 T7 promoter polynucleotide 274nagatctcga
tcccgcgaaa ttaatacgac tcactatagg gagaccacaa cggtttccct 60ctagatcaca
agtttgtaca aaaaagctga acgagaaacg ta
102275102DNAArtificial SequenceDescription of Artificial Sequence
Synthetic pDEST25 T7 promoter polynucleotide 275ttttacgttt ctcgttca
gct ttc ttg tac aaa gtg gtg att atg agc gat 51 Ala Phe Leu Tyr
Lys Val Val Ile Met Ser Asp 1
5 10 aaa att att cacctgactg acgacagttt
tgacacggat gtactcaaag cg 102Lys Ile Ile
27614PRTArtificial
SequenceDescription of Artificial Sequence Synthetic pDEST25 T7
promoter peptide 276Ala Phe Leu Tyr Lys Val Val Ile Met Ser Asp Lys Ile
Ile 1 5 10
277306DNAArtificial SequenceDescription of Artificial Sequence Synthetic
pDEST26 CMV promoter polynucleotide 277ttgacgtcaa tgggagtttg
ttttggcacc aaaatcaacg ggactttcca aaatgtcgta 60acaactccgc cccattgacg
caaatgggcg gtaggcgtgt acggtgggag gtctatataa 120gcagagctcg tttagtgaac
cgtcagatcg cctggagacg ccatccacgc tgttttgacc 180tccatagaag acaccgggac
cgatccagcc tccggactct agcctaggcc gcggacc 237atg gcg tac tac cat cac
cat cac cat cac tct aga tca aca agt ttg 285Met Ala Tyr Tyr His His
His His His His Ser Arg Ser Thr Ser Leu 1 5
10 15 tac aaa aaa gct
gaacgagaa 306Tyr Lys Lys Ala
20
27820PRTArtificial SequenceDescription of Artificial Sequence Synthetic
pDEST26 CMV promoter peptide 278Met Ala Tyr Tyr His His His His His
His Ser Arg Ser Thr Ser Leu 1 5 10
15 Tyr Lys Lys Ala 20 279255DNAArtificial
SequenceDescription of Artificial Sequence Synthetic pDEST27
promoter polynucleotide 279nacggtggga ggtctatata agcagagctc gtttagtgaa
ccgtcagatc gcctggagac 60gccatccacg ctgttttgac ctccatagaa gacaccggga
ccgatccagc ctccggactc 120tagcctaggc cgcggacc atg gcc cct ata cta
ggttattgga aaattaaggg 173 Met Ala Pro Ile Leu
1 5
ccttgtgcaa cccactcgac ttcttttgga atatcttgaa gaaaaatatg
aagagcattt 233gtatgagcgc gatgaaggtg at
2552805PRTArtificial SequenceDescription of Artificial
Sequence Synthetic pDEST27 promoter peptide 280Met Ala Pro Ile Leu 1
5 28187DNAArtificial SequenceDescription of Artificial
Sequence Synthetic pDEST27 promoter oligonucleotide 281tttggtggtg
gcgaccatcc tccaaaatcg gatctg gtt ccg cgt tct aga tca 54 Val Pro
Arg Ser Arg Ser 1
5 aca agt ttg tac aaa aaa gct gaacgagaaa cg
87Thr Ser Leu Tyr Lys Lys Ala
10
28213PRTArtificial SequenceDescription of
Artificial Sequence Synthetic pDEST27 promoter peptide 282Val Pro
Arg Ser Arg Ser Thr Ser Leu Tyr Lys Lys Ala 1 5
10 283405DNAArtificial SequenceDescription of
Artificial Sequence Synthetic pEXP501 polynucleotide 283agagctcgtt
tagtgaaccg tcagatcgcc tggagacgcc atccacgctg ttttgacctc 60catagaagac
accgggaccg atccagcctc cggactctag cctaggccgc ggagcggata 120acaatttcac
acaggaaaca gctatgacca ttaggcctat ttaggtgaca ctatagaaca 180agtttgtaca
aaaaagcagg ctggtaccgg tccggaattc ccgggatatc gtcgacgagc 240tcactagtcg
gcggccgctc tagagtatcc ctcgaggggc ccaagcttac gcgtacccag 300ctttcttgta
caaagtggtc cctatagtga gtcgtattat aagctaggca ctggccgtcg 360ttttacaacg
tcgtgactgg gaaaactgct agcttgggat ctttg
405284153DNAArtificial SequenceDescription of Artificial Sequence
Synthetic His6-CAT polynucleotide 284cggataacaa tttcacacag
gaaacagacc atg tcg tac tac cat cac cat cac 54 Met Ser Tyr Tyr His
His His His 1 5
cat cac ggc atc aca agt ttg tac aaa aaa gca ggc ttt
gaa aac ctg 102His His Gly Ile Thr Ser Leu Tyr Lys Lys Ala Gly Phe
Glu Asn Leu 10 15 20
tat ttt caa gga acc atg gag aaa aaa atc act gga tat
acc acc gtt 150Tyr Phe Gln Gly Thr Met Glu Lys Lys Ile Thr Gly Tyr
Thr Thr Val 25 30 35
40 gat
153Asp
28541PRTArtificial SequenceDescription of Artificial
Sequence Synthetic His6-CAT polypeptide 285Met Ser Tyr Tyr His His
His His His His Gly Ile Thr Ser Leu Tyr 1 5
10 15 Lys Lys Ala Gly Phe Glu Asn Leu Tyr Phe Gln
Gly Thr Met Glu Lys 20 25
30 Lys Ile Thr Gly Tyr Thr Thr Val Asp 35
40 28610DNAArtificial SequenceDescription of Artificial Sequence
Synthetic consensus oligonucleotide 286magtcactat
1028723DNAArtificial
SequenceDescription of Artificial Sequence Synthetic attB1- and
attB2-derived primer 287aagcaggctn nnnnnnnnnn nnn
2328823DNAArtificial SequenceDescription of
Artificial Sequence Synthetic attB1- and attB2-derived primer
288aagctgggtn nnnnnnnnnn nnn
2328922DNAArtificial SequenceDescription of Artificial Sequence Synthetic
attB1- and attB2-derived primer 289agcaggctnn nnnnnnnnnn nn
2229022DNAArtificial
SequenceDescription of Artificial Sequence Synthetic attB1- and
attB2-derived primer 290agctgggtnn nnnnnnnnnn nn
2229121DNAArtificial SequenceDescription of
Artificial Sequence Synthetic attB1- and attB2-derived primer
291gcaggctnnn nnnnnnnnnn n
2129221DNAArtificial SequenceDescription of Artificial Sequence Synthetic
attB1- and attB2-derived primer 292gctgggtnnn nnnnnnnnnn n
2129320DNAArtificial
SequenceDescription of Artificial Sequence Synthetic attB1- and
attB2-derived primer 293caggctnnnn nnnnnnnnnn
2029420DNAArtificial SequenceDescription of
Artificial Sequence Synthetic attB1- and attB2-derived primer
294ctgggtnnnn nnnnnnnnnn
2029510DNAUnknownDescription of Unknown Eukaryotic initiating
oligonucleotide 295gccaccatgg
1029616DNAArtificial SequenceDescription of Artificial
Sequence Synthetic oligonucleotide 296atgnnnnnnn nntaac
1629720DNAArtificial
SequenceDescription of Artificial Sequence Synthetic oligonucleotide
297tcgagttann nnnnnnncat
2029830DNAArtificial SequenceDescription of Artificial Sequence Synthetic
oligonucleotide 298nnn aca agt ttg tac aaa aaa gca ggc tnn
30Thr Ser Leu Tyr Lys Lys Ala Gly
1 5
2998PRTArtificial SequenceDescription of Artificial
Sequence Synthetic peptide 299Thr Ser Leu Tyr Lys Lys Ala Gly 1
5 30033DNAArtificial SequenceDescription of
Artificial Sequence Synthetic oligonucleotide 300nnnnac cca gct ttc
ttg tac aaa gtg gtn nnn 33Pro Ala Phe Leu Tyr
Lys Val Val 1 5
3018PRTArtificial
SequenceDescription of Artificial Sequence Synthetic peptide 301Pro
Ala Phe Leu Tyr Lys Val Val 1 5
30251DNAArtificial SequenceDescription of Artificial Sequence Synthetic
oligonucleotide 302gat gac gat gac aag gta ccc atc aca agt ttg tac aaa
aaa gctgaacga 51Asp Asp Asp Asp Lys Val Pro Ile Thr Ser Leu Tyr Lys
Lys 1 5 10
30314PRTArtificial SequenceDescription of
Artificial Sequence Synthetic peptide 303Asp Asp Asp Asp Lys Val Pro
Ile Thr Ser Leu Tyr Lys Lys 1 5 10
User Contributions:
Comment about this patent or add new information about this topic:
| People who visited this patent also read: | |
| Patent application number | Title |
|---|---|
| 20160337534 | IMAGE FORMING APPARATUS AND RECORDING MEDIUM |
| 20160337533 | PORTABLE TERMINAL AND METHOD |
| 20160337531 | INFORMATION PROCESSING SYSTEM, INFORMATION PROCESSING APPARATUS, AND NON-TRANSITORY COMPUTER READABLE MEDIUM |
| 20160337530 | INFORMATION PROCESSING APPARATUS THAT CONTROLS DISPLAY AT TIME OF OCCURRENCE OF ABNORMALITY, METHOD OF CONTROLLING THE SAME, AND STORAGE MEDIUM |
| 20160337528 | IMAGE FORMING APPARATUS AND IMAGE FORMING METHOD THAT ARE NORMALLY RESETTABLE IN MODE WHERE OPERATING VOLTAGE IS NOT SUPPLIED TO MAIN CPU |