Patent application title: GENE SILENCING OF SUGAR-DEPENDENT 1 IN JATROPHA CURCAS
Inventors:
IPC8 Class: AC12N1582FI
USPC Class:
800281
Class name: Multicellular living organisms and unmodified parts thereof and related processes method of introducing a polynucleotide molecule into or rearrangement of genetic material within a plant or plant part the polynucleotide alters fat, fatty oil, ester-type wax, or fatty acid production in the plant
Publication date: 2016-01-14
Patent application number: 20160010097
Abstract:
The present invention relates to the field of plant molecular biology and
gene silencing. More particularly, the present invention relates to gene
silencing of Sugar-dependent 1 (JcSDP1) in Jatropha curcas. JcSDP1
encodes a patatin-domain triacylglyerol lipase. Silencing of JcSDP1
enhances seed oil accumulation in J. curcas.Claims:
1. A method of increasing seed oil accumulation in Jatropha comprising
modulating the activity of a patatin-domain triacylglycerol lipase in a
Jatropha plant to cause an increased seed oil accumulation.
2. The method of claim 1, wherein the modulation is reducing the activity of the lipase.
3. The method of claim 1, wherein the lipase is encoded by a sugar-dependent-1 (SDP1) gene.
4. The method of claim 3, wherein the lipase has the amino acid sequence set forth in SEQ ID NO:2.
5. The method of claim 4, wherein the lipase is encoded by a nucleic acid having a nucleotide sequence set forth in SEQ ID NO:1, or nucleotides 874-3450 of SEQ ID NO:1, or nucleotides 874-3447 of SEQ ID NO:1.
6. The method of claim 1, wherein the reduction in activity of the lipase is mediated by RNAi or VIGS.
7. The method of claim 6, wherein the activity of the lipase is modulated in seeds of the Jatropha plant.
8. The method of claim 7, wherein the modulation in Jatropha seeds is accomplished using a Jatropha seed specific promoter.
9. The method of claim 8, wherein the Jatropha seed specific promoter is a Jatropha curcas SDP1 promoter.
10. The method of claim 9, wherein the Jatropha curcas SDP1 promoter has the nucleotide sequence set forth in nucleotides 1-722 of SEQ ID NO:1 or promoter functional fragment thereof.
11. The method of claim 1, wherein the seed oil accumulation is increased by about 13% to about 30%.
12. The method of claim 6, wherein the RNAi is mediated by a nucleic acid comprising the nucleotide sequence set forth in SEQ ID NO:3.
13. An isolated nucleic acid comprising nucleotides 3161-3533 of SEQ ID NO:1, wherein the nucleic acid, when used in RNAi, reduces expression of the sugar-dependent 1 (SDP1) gene.
14. The isolated nucleic acid of claim 13, wherein the nucleic acid comprises nucleotides 3161-3528 of SEQ ID NO:1.
15. The isolated nucleic acid of claim 13 which further comprises a plant operable promoter operably linked to the nucleic acid.
16. The isolated nucleic acid of claim 15, wherein the promoter is a seed specific promoter.
17. The isolated nucleic acid of claim 16, wherein the seed specific promoter is a Jatropha curcas SDP1 promoter.
18. The isolated nucleic acid of claim 17, wherein the Jatropha curcas SDP1 promoter comprises nucleotides 1-722 of SEQ ID NO:1.
19. An isolated nucleic acid that when used in RNAi reduces expression of the sugar-dependent 1 (SDP1) gene, wherein the nucleic acid comprises the nucleotide sequence set forth in SEQ ID NO:3.
20. A nucleic acid construct or a vector comprising the isolated nucleic acid of claim 13.
21. A transgenic plant cell or transgenic plant having the isolated nucleic acid of claim 13 stably integrated in its genome.
22. A method of making a transgenic plant cell comprising introducing the isolated nucleic acid of claim 13 into a plant cell and selecting a transgenic plant cell in which the isolated nucleic acid is stably integrated in its genome.
23. A method of making a transgenic plant comprising introducing the isolated nucleic acid of claim 13 into a plant cell, selecting a transgenic plant cell in which the isolated nucleic acid is stably integrated in its genome and regenerating a transgenic plant from the selected transgenic plant cell.
24. An isolated nucleic acid encoding a protein comprising the amino acid sequence set forth in SEQ ID NO:2.
25. The isolated nucleic acid of claim 24 which comprises the nucleotide sequence set forth in SEQ ID NO:1, or nucleotides 874-3450 of SEQ ID NO:1 or nucleotides 874-3447 of SEQ ID NO:1.
26. The isolated nucleic acid of claim 24 which further comprises a plant operable promoter operably linked to the nucleic acid.
27. The isolated nucleic acid of claim 26, wherein the promoter is a heterologous seed specific promoter.
Description:
CROSS-REFERENCE TO RELATED APPLICATION
[0001] The present application is related to and claims priority to U.S. Provisional Patent Application Ser. No. 61/773,386, filed 6 Mar. 2013. This application is incorporated herein by reference.
SEQUENCE SUBMISSION
[0002] The present application is being filed along with a Sequence Listing in electronic format. The Sequence Listing is entitled 2577229PCTSequencListing.txt, was created on 18 Feb. 2014 and is 43 kb in size. The information in the electronic format of the Sequence Listing is part of the present application and is incorporated herein by reference in its entirety.
BACKGROUND OF THE INVENTION
[0003] The present invention relates to the field of plant molecular biology and gene silencing. More particularly, the present invention relates to gene silencing of Sugar-dependent 1 (JcSDP1) in Jatropha curcas. JcSDP1 encodes a patatin-domain triacylglyerol lipase. Silencing of JcSDP1 enhances seed oil accumulation in J. curcas.
[0004] The publications and other materials used herein to illuminate the background of the invention, and in particular, cases to provide additional details respecting the practice, are incorporated by reference, and for convenience are referenced in the following text by author and date and are listed alphabetically by author in the appended bibliography.
[0005] The diminishing stock of fossil fuel has catalyzed a soaring demand for renewable energy sources world-wide. To meet this demand active research has been initiated relating to solar, wind, tidal and geothermal power generations during the last several decades. Likewise, there is also an increasing focus on biofuels, which are energy sources derived from renewable biomass. There are two main types of biofuels: bioethanol and biodiesel, which are generally used as gasoline and diesel additives, respectively. Bioethanol is mainly produced by fermentation using sugar or starch derived from crops such as sugar cane and corn. Biodiesel, on the other hand, is obtained by trans-esterification of plant oils and animal fats. In 2010, the global biofuel production reached 105 billion liters and provided 2.7% of the world's energy needs for transportation. It has been forecast that biofuel may account for more than a quarter of the world's demand for transportation fuels by 2050 (1).
[0006] Biodiesel is generally produced from oil seed crops such as rape in temperate countries and oil palm in the tropics. In the past several years, a small tree called Jatropha curcus grown in the tropical and subtropical regions has emerged as an attractive candidate crop for biodiesel production. Jatropha has several interesting attributes making it suitable for consideration as a biodiesel plant. Jatropha seeds contain up to 40% oil consisting of approximately 75% unsaturated fatty acids (2, 3), with a high level (around 47%) of linoleic acid (C18:2) (4). In addition to having a high oil content and favorable oil composition for biodiesel, Jatropha plants have a short gestation period and adapt well to a wide range of agro-climatic conditions (5, 6). Moreover, its ability to grow on marginal land reduces the possibility that Jatropha may compete with food crops for arable land.
[0007] Because of Jatropha has just been recently been domesticated much work remains to be done to improve its agronomic traits either by traditional breeding or by gene technology.
[0008] Given the commercial interest in Jatropha seed oil, it is not surprising that the immediate focus is on seed oil content and quality. With respect to the latter trait, Qu et al. (7) have recently reported that gene silencing of JcFAD2 can greatly enhance the proportion of oleic acids in seeds of transgenic Jatropha. Here, we addressed the issue of increasing levels of oil accumulation in Jatropha seeds by genetic modification.
[0009] Plant oil in seeds is stored as triacylglycerol (TAG) consisting of three fatty acid chains (usually C16 or C18) covalently linked to glycerol. Depending on the plant source, TAGs may contain fatty acids of different chain lengths and degree of saturation and the fatty acids may be decorated with diverse modifications. Plant TAGs are generally stored in small organelles called oil bodies which are assembled in developing seeds, flower petals, pollen grains and fruits of a huge number of plant species (8, 9). During seed germination TAGs are hydrolyzed into fatty acids and glycerol and this reaction is catalyzed by TAG lipases, which are widely distributed in plants but also found in animals and microorganisms (10). Among the known lipases, the unorthodox patatin-like TAG lipases (PTLs) are oil body-associated enzymes that play a major role in the initiation of TAG degradation in yeast, mammals and insects (11, 12). During seed germination, TAG lipases initiate TAG degradation into glycerol and free fatty acids and the released fatty acids are consumed through the beta-oxidation pathway which releases energy for early seedling growth (13, 14).
[0010] Recently, Eastmond (15, 16) has shown that the sugar-dependent 1 (SDP1) of Arabidopsis encodes a patatin-like acyl hydrolase domain. The encoded protein, SDP1, is specifically responsible for the first step of TAG degradation during Arabidopsis seed germination. This enzyme is also able to associate with oil body surface as well as the other reported PTLs. A T-DNA insertion SDP1 mutant allele, sdp1-5, displayed growth retardation on a sugar-deficient medium due to the deficiency of glycerol and free fatty acids, which are products of TAG degradation (15, 16). The study of Eastmond (15, 16) showed an accumulation of clustered oil bodies in seedling cotyledons and a higher TAG levels in sdp1-5 than WT (Col-0).
[0011] It is desired to increase seed oil content in Jatropha which would be an important agronomic trait for this biodiesel crop.
SUMMARY OF THE INVENTION
[0012] The present invention relates to the field of plant molecular biology and gene silencing. More particularly, the present invention relates to gene silencing of Sugar-dependent 1 (JcSDP1) in Jatropha curcas. JcSDP1 encodes a patatin-domain triacylglyerol lipase. Silencing of JcSDP1 enhances seed oil accumulation in J. curcas.
[0013] Thus, in one aspect, the present invention provides for In a first aspect, the present invention provides an isolated nucleic acid encoding a JcSDP1 protein comprising the amino acid sequence set forth in SEQ ID NO:2. In one embodiment, the nucleic acid comprises the nucleotide sequence set forth in SEQ ID NO:1. In another embodiment, the nucleic acid comprises the nucleotide sequence set forth as nucleotides 874-3450 of SEQ ID NO:1. In an additional embodiment, the nucleic acid comprises the nucleotide sequence set forth as nucleotides 874-3447 of SEQ ID NO:1. In a further embodiment, the nucleic acid further comprises a plant operable promoter operably linked to a nucleic acid comprising the coding sequence, or a nucleic acid comprising the 5' UTR (or portion thereof) and the coding sequence, or a nucleic acid comprising the coding sequence and the 3' UTR (or portion thereof) or a nucleic acid comprising the 5' UTR (or portion thereof), coding sequence and the 3' UTR (or portion thereof. In one embodiment, the promoter is a seed specific promoter. In another embodiment, the seed specific promoter is a Jatropha seed specific promoter. In a further embodiment, the Jatropha seed specific promoter is the JcSDP1 promoter. In one embodiment, the JcSDP1 promoter comprises nucleotides 1-722 of SEQ ID NO:1. In another embodiment, functional fragments of the JcSDP1 promoter can be used.
[0014] In a second aspect, the present invention provides a construct or vector comprising an isolated nucleic acid as described herein. In one embodiment, the construct or vector is an expression construct or vector. In another embodiment, the construct or vector further comprises a selectable marker. In a further embodiment, the construct or vector comprises a Cre-lox recombination marker free system.
[0015] In a third aspect, the present invention provides a transgenic plant comprising a nucleic acid or vector described herein. In one embodiment, the transgenic plant is a Jatropha plant.
[0016] In a fourth aspect, the present invention provides for the down regulation of a Jatropha SDP1 gene. In one embodiment, the down regulation of a Jatropha SDP1 gene involves using RNA interference (RNAi), including double stranded RNA, siRNA, microRNA, and hairpin RNA. In another embodiment, the down regulation of a Jatropha SDP1 gene involves using viral induced gene silencing (VIGS). In one embodiment, a nucleic acid is provided which down regulates the Jatropha SDP1 gene. In another embodiment, a nucleic acid is provided which down regulates the Jatropha curcas SDP1 gene. In some embodiments, the nucleic acid comprises nucleotides 3161 to 3533 of SEQ ID NO:1. In another embodiment, a suitable nucleic acid for use in down regulating the Jatropha SDP1 gene is selected using well known RNAi considerations and algorithms. In some embodiments, the nucleic acid further comprises a plant operable promoter operably linked to the nucleic acid. In one embodiment, the promoter is a seed specific promoter. In another embodiment, the seed specific promoter is a Jatropha seed promoter. In a further embodiment, the Jatropha seed specific promoter is the JcSDP1 promoter. In one embodiment, the JcSDP1 promoter comprises nucleotides 1-722 of SEQ ID NO:1. In another embodiment, functional fragments of the JcSDP1 promoter can be used. In an additional embodiment, the promoter comprises the JcSDP1 promoter and the 3' UTR (or portion thereof). In one embodiment, a nucleic acid suitable for down regulating Jatropha SDP1 gene is set forth in SEQ ID NO:3. According to this aspect, the present invention also provides a vector comprising an isolated nucleic acid as described herein. In one embodiment, the vector is an expression vector. In another embodiment, the vector further comprises a selectable marker. In a further embodiment, the vector comprises a Cre-lox recombination marker free system. According to this aspect, the present invention further provides a transgenic or infected plant or plant cell comprising a nucleic acid or vector described herein. In one embodiment, the transgenic or infected plant or plant cell is a Jatropha plant or plant cell. In some embodiments, the down regulation of a Jatropha SDP1 gene provides enhanced seed oil accumulation in a transgenic or infected Jatropha plant, such as Jatropha curcas.
[0017] In a fifth aspect, the present invention provides methods of increasing seed oil accumulation in Jatropha, such as Jatropha curcas. In one embodiment, a method involves modulating the level of activity of a Sugar-dependent 1 (SDP1) gene, such as JcSDP1 gene, which encodes a patatin-domain triacylglycerol lipase in the host Jatropha cell or Jatropha plant. The level of activity of the lipase can be reduced by reducing expression of the SDP1 gene. In one embodiment, the modulation of the level of activity of the lipase is a reduction in the activity of the lipase. The level of activity of the lipase can be reduced by using RNAi techniques described herein in which the lipase is the target for the RNAi. Alternatively, the level of activity of the lipase can be reduced using VIGS techniques as described herein in which at least a partial fragment of the target gene is used. In one embodiment, Jatropha RNAi plants accumulate about 13% to about 30% higher seed oil than control plants.
BRIEF DESCRIPTION OF THE DRAWINGS
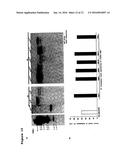
[0018] FIGS. 1A-1G show sdp1-5 mutant accumulates a higher TAG levels than WT (Col-0) in mature seeds. FIG. 1A: Scanning electron microscopy (SEM) showing the seed surface structure of WT (Col-0) and sdp1-5. FIG. 1B: Comparative dry seed weight of WT (Col-0) and sdp1-5. FIG. 1C: Total amount of fatty acids per seed of WT (Col-0) and sdp1-5. FIG. 1D: Relative fatty acids of dried seeds of WT (Col-0) and sdp1-5. FIG. 1E: Fatty acid profiles of WT (Col-0) and sdp1-5 seeds. FIG. 1F: Seed eicosenoic acid (20:1) content. FIG. 1G: TLC separation of neutral lipid fractions from WT (Col-0) and three lines of sdp1-5 null mutant. 300 μg of neutral lipids were fractionated by TLC on silica gel plates. TAG, Triacylglycerol (Triolein, Sigma T7140); FFA, Free Fatty Acid (Oleic acid, Sigma 75090), Mix, mix TAG and FFA, SE, sterol ester; FFA, free fatty acid; DAG, diacylglycerol. H) Profiling of relative amounts of FFA and TAG by GC/MS. The absolute amount was calculated using the C15:0 as an internal control by comparing their peak areas. *P<0.05 or **P<0.01 versus WT (Col-0). Each experiment was performed with 100 seeds per line with 5 biological replicates. Error bar shows SD, standard deviation (n=5). DW, Dry weight
[0019] FIGS. 2A-2C show sdp1-5 mutant seeds produce more oil bodies. FIG. 2A: TEM analysis of oil body distribution in WT (Col-0) and sdp1-5 mature seeds. The scale bar is 5 μm. PSV; protein storage vacuole, CW; cell wall, OB; oil body. FIG. 2B: Numbers of oil bodies per cell in mature seeds of WT (Col-0) and sdp1-5. A cell has an average of 196 μm2 cross-sectional area (Mean value with SD; n=5). FIG. 2C: Expression levels of AtOLE1 (At4g25140), AtOLE2 (At5g40420), and AtOLE3 (At5g51210) in early developing stages of seeds (3 to 5 DAP) from WT (Col-0) and sdp1-5. Actin1 (At2g37620) was used as an internal control. *P<0.05, **P<0.01, or ***P<0.001 versus WT (Col-0). There were 5 biological replicates. SD, standard deviation (n=5). DAP: Day after pollination.
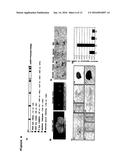
[0020] FIGS. 3A and 3B show cloning of JcSDP1 gene from Jatropha plants and complementation of the Arabidopsis sdp1-5 mutant. FIG. 3A: Deduced amino acid sequence of JcSDP1 (SEQ ID NO:2) and alignment to the AtSDP1 amino acid sequence (SEQ ID NO:4). Bold line; patatin domain, star (*****); site-1 protease (S1P) target sequence, RXXL (SEQ ID NO:5), GXGXXG (SEQ ID NO:6); oxyanion hole motif, GXSXG (SEQ ID NO:7); lipase consensus motif with catalytic serine. FIG. 3B: Complementation of sdp1-5 with JcSDP1. WT (Col-0), sdp1-5, and CaMV35S:JcSDP1/sdp1-5 transgenic plants were grown on MS medium with or without sucrose (1%).
[0021] FIGS. 4A-4E show JcSDP1 promoter and its expression analysis in ProJcSDP1:GUS transgenic plants. FIG. 4A: Composition of putative cis-elements in JcSDP1 promoter. FIG. 4B: Transient ProJcSDP1:GUS expression in Jatropha fruit and leaf. FIG. 4C: Heterologous expressions of ProJcSDP1:GUS in transgenic Arabidopsis plants. a) 14 day-old seedling, b-c) inflorescence, d-e) rosette leaf, f) siliques, g) seeds at different developing stages, h) young seeds (3 to 4 DAP, globular-stage embryo), i) mid-stage seeds (9 to 10 DAP, mature green embryo). DAP: Days after pollination. FIG. 4D: Sugar-dependent expression of ProJcSDP1:GUS transgenic plants (14 day-old seedling). 1% or 3% fructose, glucose, and sucrose were used as a sugar source and mannitol was used as a control for osmotic stress. FIG. 4E: JcSDP1 gene expression profiles using quantitative real-time PCR at different seed developing stages (S1; 1WAF, S2; 2 to 3WAF, S3; 4 to 6WAF, and S4; 7 to 8WAF, WAF: weeks after fertilization JcTubulin expression levels were used as an internal control. Mean values are given with SD (n=3).
[0022] FIGS. 5A-5E show construction of ProJcSDP1:JcSDP1-RNAi transgenic Jatropha plants and analysis of seed oils. FIG. 5A: Structure of the inducible ProJcSDP:SDP1-RNAi marker-free construction. FIG. 5B: ProJcSDP:SDP1-RNAi marker-free transgenic plants; a-c) T1 generation of dried seeds. d) Overall phenotype of T1 transgenic plants generated from T1 embryos. e) Size comparison of the 5th leaf between control (CK) plants and JcSDP1-RNAi transgenic line #158. FIG. 5C: Total fatty acid analysis in mature endosperm of T1 seed. FIG. 5D: Fatty acid profile in JcSDP1-RNAi transgenic lines. FIG. 5E: JcSDP1 transcript levels in transgenic lines. JcTubulin expression levels were used as an internal control. Numbers refer to transgenic line numbers. Asterisks indicated statistically significant difference compared with the control, *P<0.05, **P<0.01, or ***P<0.001 versus CK, in different biological replicates. CK, 35S:GFP control.
[0023] FIGS. 6A-6C show schematic diagram of SDP1 gene structure and insertion position of T-DNA in the sdp1-5 allele. FIG. 6A: The T-DNA is inserted in the first exon of SDP1 resulting in a null mutation. FIG. 6B: Isolation of the homozygous sdp1-5 mutant by RT-PCR. FIG. 6C: Expression levels of AtSDP1 in early developing seeds (3 to 5 DAP). Actin1 (At2g37620) expression levels were used as an internal control. *P<0.05, **P<0.01, or ***P<0.001 versus Col-0 for 5 biological replicates. SD, standard deviation (n=5). DAP: Days after pollination.
[0024] FIG. 7 shows expression profiles of fatty acid synthesis-related genes in early developing seeds of WT (Col-0) and sdp1-5. The cDNA library was synthesized from total mRNA derived from seeds (3 to 5 DAP). DAP: Days after pollination. Mean values are given with SD (n=3). ACP1(acyl carrier protein; At3g05020), FAD2(oleate desaturase; At3g12120), FAD3(linoleate desaturase; At2g29980), FAE1(fatty acid elongase; At4g34520), Cy-PK(cytosol pyruvate kinase; At5g52920), Ch-PK(chloroplast pyruvate kinase: At3g22960), KASI(ketoacyl-ACP Synthase I; At5g46290), KASIII(ketoacyl-ACP synthase III; At1g62640), BCCP2(biotin carboxyl carrier protein; At5g15530), and SUS2(sucrose synthase 2; At5g49190).
[0025] FIGS. 8A and 8B shows analysis of lipid composition of mature dried endosperm of JcSDP1-RNAi by TLC and GC/MS. FIG. 8A: Total lipids were extracted from mature endosperm of CK and JcSDP1-RNAi transgenic line #158 and 300 μg of total neutral lipids were separated by TLC on silica gel plate and stained with Iodine (I2). SE, sterol ester; TAG, triacylglycerol; FFA, free fatty acid; DAG, diacylglycerol. FIG. 8B: Profiling of relative amounts of FFA and TAG by GC/MS. The absolute amount was calculated using the C15:0 as an internal control by comparing their peak areas.
[0026] FIG. 9 shows a schematic diagram of a JcSDP1-RNAi sequence in accordance with one embodiment of the invention. A sequence for this construction is shown in SEQ ID NO:3
[0027] FIGS. 10A and 10B show Southern blot analysis of T0 and T1 transgenic plants expressing JcSDP1-RNAi. FIG. 10A: Total genomic DNA were digested with XhoI restriction enzyme and hybridized with HPT probe. FIG. 10B: Total lipid content (%, w/w) in individual transgenic line carrying the JcSDP1-RNAi construct.
DETAILED DESCRIPTION OF THE INVENTION
[0028] The present invention relates to the field of plant molecular biology and gene silencing. More particularly, the present invention relates to gene silencing of Sugar-dependent 1 (JcSDP1) in Jatropha curcas. JcSDP1 encodes a patatin-domain triacylglyerol lipase. Silencing of JcSDP1 enhances seed oil accumulation in J. curcas.
[0029] Unless defined otherwise, all technical and scientific terms used herein have the same meaning as is commonly understood by one of skill in the art to which the invention belongs.
[0030] As used herein, "allele" refers to any of one or more alternative forms of a gene locus, all of which alleles relate to a trait or characteristic. In a diploid cell or organism, the two alleles of a given gene occupy corresponding loci on a pair of homologous chromosomes.
[0031] As used herein, "gene" refers to a nucleic acid sequence that encompasses a 5' promoter region associated with the expression of the gene product, any intron and exon regions and 3' or 5' untranslated regions associated with the expression of the gene product.
[0032] As used herein, "genotype" refers to the genetic constitution of a cell or organism.
[0033] As used herein, "phenotype" refers to the detectable characteristics of a cell or organism, which characteristics are the manifestation of gene expression.
[0034] The terms "polynucleotide," nucleic acid" and "nucleic acid molecule are used interchangeably herein to refer to a polymer of nucleotides which may be a natural or synthetic linear and sequential array of nucleotides and/or nucleosides, including deoxyribonucleic acid, ribonucleic acid, and derivatives thereof. It includes chromosomal DNA, self-replicating plasmids, infectious polymers of DNA or RNA and DNA or RNA that performs a primarily structural role. Unless otherwise indicated, nucleic acids or polynucleotide are written left to right in 5' to 3' orientation, Nucleotides are referred to by their commonly accepted single-letter codes. Numeric ranges are inclusive of the numbers defining the range.
[0035] The terms "polypeptide," "peptide," and "protein" are used interchangeably herein to refer to a polymer of amino acid residues. The terms apply to amino acid polymers in which one or more amino acid residue is an artificial chemical analogue of a corresponding naturally occurring amino acid, as well as to naturally occurring amino acid polymers Amino acids may be referred to by their commonly known three-letter or one-letter symbols. Amino acid sequences are written left to right in amino to carboxy orientation, respectively. Numeric ranges are inclusive of the numbers defining the range.
[0036] Thus, in one aspect, the present invention provides for In a first aspect, the present invention provides an isolated nucleic acid encoding a JcSDP1 protein comprising the amino acid sequence set forth in SEQ ID NO:2. In one embodiment, the nucleic acid comprises the nucleotide sequence set forth in SEQ ID NO: 1. In another embodiment, the nucleic acid comprises the nucleotide sequence set forth as nucleotides 874-3450 of SEQ ID NO: 1. In an additional embodiment, the nucleic acid comprises the nucleotide sequence set forth as nucleotides 874-3447 of SEQ ID NO: 1. In a further embodiment, the nucleic acid further comprises a plant operable promoter operably linked to a nucleic acid comprising the coding sequence, or a nucleic acid comprising the 5' UTR (or portion thereof) and the coding sequence, or a nucleic acid comprising the coding sequence and the 3' UTR (or portion thereof) or a nucleic acid comprising the 5' UTR (or portion thereof), coding sequence and the 3' UTR (or portion thereof. In another embodiment, the seed specific promoter is a Jatropha seed specific promoter. In a further embodiment, the Jatropha seed specific promoter is the JcSDP1 promoter. In one embodiment, the JcSDP1 promoter comprises nucleotides 1-722 of SEQ ID NO:1. In another embodiment, functional fragments of the JcSDP1 promoter can be used. Useful functional fragments of the JcSDP1 promoter can be identified by the skilled artisan using conventional techniques well known in the art including, but not limited to promoter analysis software, promoter cis-element software and the like.
[0037] In a second aspect, the present invention provides a construct or vector comprising an isolated nucleic acid as described herein. In one embodiment, the construct or vector is an expression construct or vector. In another embodiment, the construct or vector further comprises a selectable marker. In a further embodiment, the construct or vector comprises a Cre-lox recombination marker free system.
[0038] In a third aspect, the present invention provides a transgenic plant comprising a nucleic acid or vector described herein. In one embodiment, the transgenic plant is a Jatropha plant.
[0039] In a fourth aspect, the present invention provides for the down regulation of a Jatropha SDP1 gene. In one embodiment, the down regulation of a Jatropha SDP1 gene involves using RNA interference (RNAi), including double stranded RNA, siRNA, microRNA, and hairpin RNA. In another embodiment, the down regulation of a Jatropha SDP1 gene involves using viral induced gene silencing (VIGS). In one embodiment, a nucleic acid is provided which down regulates the Jatropha SDP1 gene. In another embodiment, a nucleic acid is provided which down regulates the Jatropha curcas SDP1 gene. In some embodiments, the nucleic acid comprises nucleotides 3161 to 3533 of SEQ ID NO:1. In another embodiment, a suitable nucleic acid for use in down regulating the Jatropha SDP1 gene is selected using well known RNAi considerations and algorithms. In some embodiments, the nucleic acid further comprises a plant operable promoter operably linked to the nucleic acid. In one embodiment, the promoter is a seed specific promoter. In another embodiment, the seed specific promoter is a Jatropha seed promoter. In a further embodiment, the Jatropha seed specific promoter is the JcSDP1 promoter. In one embodiment, the JcSDP1 promoter comprises nucleotides 1-722 of SEQ ID NO: 1. In another embodiment, functional fragments of the JcSDP1 promoter can be used. In an additional embodiment, the promoter comprises the JcSDP1 promoter and the 3' UTR (or portion thereof). In one embodiment, a nucleic acid suitable for down regulating Jatropha SDP1 gene is set forth in SEQ ID NO:3. According to this aspect, the present invention also provides a vector comprising an isolated nucleic acid as described herein. In one embodiment, the vector is an expression vector. In another embodiment, the vector further comprises a selectable marker. In a further embodiment, the vector comprises a Cre-lox recombination marker free system. According to this aspect, the present invention further provides a transgenic or infected plant or plant cell comprising a nucleic acid or vector described herein. In one embodiment, the transgenic or infected plant or plant cell is a Jatropha plant or plant cell. In some embodiments, the down regulation of a Jatropha SDP1 gene provides enhanced seed oil accumulation in a transgenic or infected Jatropha plant, such as Jatropha curcas.
[0040] According to this aspect, the nucleic acid is selected to inhibit expression of the native gene or to silence the native gene within a plant's tissues to achieve a desired phenotype. In one embodiment, expression of the native gene is inhibited. Such inhibition might be accomplished, for example, with transformation of a plant cell to comprise a promoter linked to an antisense nucleotide sequence, hairpin, RNA interfering molecule, double stranded RNA, microRNA or other nucleic acid molecule, such that tissue-preferred expression of the molecule interferes with translation of the mRNA of the native DNA sequence or otherwise inhibits expression of the native DNA sequence in plant cells. For further description of RNAi techniques or microRNA techniques, see, e.g., U.S. Pat. Nos. 5,034,323; 6,326,527; 6,452,067; 6,573,099; 6,753,139; and 6,777,588. See also International Publication Nos. WO 97/01952, WO 98/36083, WO 98/53083, WO 99/32619 and WO 01/75164; and U.S. Patent Application Publication Nos. 2003/0175965, 2003/0175783, 2003/0180945, 2004/0214330, 2005/0244858, 2005/0277610, 2006/0130176, 2007/0265220, 2008/0313773, 2009/0094711, 2009/0215860, 2009/0308041, 2010/0058498 and 2011/0091975. RNAi molecules or microRNA molecules can be prepared by the skilled artisan using techniques well known in the art, including techniques for the selection and testing of RNAi molecules and microRNA molecules that are useful for down regulating a Jatropha SDP1 gene. In another embodiment, the native gene may be silenced by using VIGS. Such silencing may be accomplished by infecting a Jatropha plant with a VIGS system that contains at least a partial fragment of a candidate gene to be silenced. For further description of VIGS systems, see International Publication Nos. WO 2010/080071 and WO 2010/144058.
[0041] The construct typically includes regulatory regions operatively linked to the 5' side of the nucleic acid described herein (such as a nucleic acid encoding a Jatropha SDP1 protein or a nucleic acid encoding an RNAi molecule to down regulate a Jatropha SSDP1 gene) and/or to the 3' side of the nucleic acid. A cassette containing all of these elements is also referred to herein as an expression cassette. The expression cassettes may additionally contain 5' leader sequences in the expression cassette construct. The regulatory regions (i.e., promoters, transcriptional regulatory regions, and translational termination regions) and/or the polynucleotide encoding a signal anchor may be native/analogous to the host cell or to each other. The promoters and tissue-specific promoters, such as seed promoters and especially Jatropha seed promoters, are particularly useful for preparing constructs for the transformation of Jatropha. Alternatively, the regulatory regions and/or the polynucleotide encoding a signal anchor may be heterologous to the host cell or to each other. See, U.S. Pat. No. 7,205,453 and U.S. Patent Application Publication Nos. 2006/0218670, 2006/0248616 and 20090100536, and the references cited therein. The expression cassettes may additionally contain 5' leader sequences in the expression cassette construct. Such leader sequences can act to enhance translation. Translation leaders are known in the art and include those described in International Publication No. WO 2008/094127 and the references cited therein.
[0042] A number of promoters can be used in the practice of the invention. The promoters can be selected based on the desired outcome. That is, the nucleic acids can be combined with constitutive, tissue-preferred, or other promoters for expression in the host cell of interest. Such constitutive promoters include, for example, the core promoter of the Rsyn7 (WO 99/48338 and U.S. Pat. No. 6,072,050); the core CaMV 35S promoter (Odell et al., 1985); rice actin (McElroy et al., 1990); ubiquitin (Christensen and Quail, 1989; Christensen et al., 1992); pEMU (Last et al., 1991); MAS (Velten et al., 1984); ALS promoter (U.S. Pat. No. 5,659,026), and the like. Other constitutive promoters include, for example, those disclosed in U.S. Pat. Nos. 5,608,149; 5,608,144; 5,604,121; 5,569,597; 5,466,785; 5,399,680; 5,268,463; and 5,608,142.
[0043] Other promoters include inducible promoters, particularly from a pathogen-inducible promoter. Such promoters include those from pathogenesis-related proteins (PR proteins), which are induced following infection by a pathogen; e.g., PR proteins, SAR proteins, beta-1,3-glucanase, chitinase, etc. Other promoters include those that are expressed locally at or near the site of pathogen infection. In further embodiments, the promoter may be a wound-inducible promoter. In other embodiments, chemical-regulated promoters can be used to modulate the expression of a gene in a plant through the application of an exogenous chemical regulator. The promoter may be a chemical-inducible promoter, where application of the chemical induces gene expression, or a chemical-repressible promoter, where application of the chemical represses gene expression. In addition, tissue-preferred promoters can be utilized to target enhanced expression of a polynucleotide of interest within a particular plant tissue. Each of these promoters are described in U.S. Pat. Nos. 6,506,962, 6,575,814, 6,972,349 and 7,301,069 and in U.S. Patent Application Publication Nos. 2007/0061917 and 2007/0143880. Any other recourse seed specific promoter can be used to, for example, Jatropha oleosin promoter (Popluechai et al., 2011), 2S storage protein promoter, the Jatropha SDP1 promoter described herein, and the like.
[0044] Generally, the expression cassette may additionally comprise a selectable marker gene for the selection of transformed cells. Selectable marker genes are utilized for the selection of transformed cells or tissues. Usually, the plant selectable marker gene will encode antibiotic resistance, with suitable genes including at least one set of genes coding for resistance to the antibiotic spectinomycin, the streptomycin phosphotransferase (spt) gene coding for streptomycin resistance, the neomycin phosphotransferase (nptII) gene encoding kanamycin or geneticin resistance, the hygromycin phosphotransferase (hpt or aphiv) gene encoding resistance to hygromycin, acetolactate synthase (als) genes. Alternatively, the plant selectable marker gene will encode herbicide resistance such as resistance to the sulfonylurea-type herbicides, glufosinate, glyphosate, ammonium, bromoxynil, imidazolinones, and 2,4-dichlorophenoxyacetate (2,4-D), including genes coding for resistance to herbicides which act to inhibit the action of glutamine synthase such as phosphinothricin or basta (e.g., the bar gene). See generally, International Publication No. WO 02/36782, U.S. Pat. No. 7,205,453 and U.S. Patent Application Publication Nos. 2006/0218670, 2006/0248616, 2007/0143880 and 2009/0100536, and the references cited therein. See also, Jefferson et al. (1991); De Wet et al. (1987); Goff et al. (1990); Kain et al. (1995) and Chiu et al. (1996). This list of selectable marker genes is not meant to be limiting. Any selectable marker gene can be used. The selectable marker gene is also under control of a promoter operable in the plant species to be transformed. Such promoters include those described in International Publication No. WO 2008/094127 and the references cited therein.
[0045] Alternatively, the expression cassette may additionally comprise a Cre-lox recombination marker free system, such as described by Zuo et al. (2001). Such a system is useful for producing selection marker free transgenic Jatropha plants.
[0046] In preparing the expression cassette, the various DNA fragments may be manipulated, so as to provide for the DNA sequences in the proper orientation and, as appropriate, in the proper reading frame. Toward this end, adapters or linkers may be employed to join the DNA fragments or other manipulations may be involved to provide for convenient restriction sites, removal of superfluous DNA, removal of restriction sites, or the like. For this purpose, in vitro mutagenesis, primer repair, restriction, annealing, resubstitutions, e.g. transitions and transversions may be involved.
[0047] Once a nucleic acid has been cloned into an expression vector, it may be introduced into a plant cell using conventional transformation procedures. The term "plant cell" is intended to encompass any cell derived from a plant including undifferentiated tissues such as callus and suspension cultures, as well as plant seeds, pollen or plant embryos. Plant tissues suitable for transformation include leaf tissues, root tissues, meristems, protoplasts, hypocotyls, cotyledons, scutellum, shoot apex, root, immature embryo, pollen, and anther. "Transformation" means the directed modification of the genome of a cell by the external application of recombinant DNA from another cell of different genotype, leading to its uptake and integration into the subject cell's genome. In this manner, genetically modified plants, plant cells, plant tissue, seed, and the like can be obtained.
[0048] DNA constructs containing the promoters of the present invention can be used to transform any plant and particularly cotton plants. The constructs may be introduced into the genome of the desired plant host by a variety of conventional techniques. Techniques for transforming a wide variety of higher plant species are well known and described in the technical and scientific literature. Transformation protocols may vary depending on the type of plant or plant cell, i.e., monocot or dicot, targeted for transformation, as is well known to the skilled artisan. For example, the DNA construct may be introduced directly into the genomic DNA of the plant cell using techniques such as electroporation and microinjection of plant cell protoplasts, or the DNA constructs can be introduced directly to plant tissue using ballistic methods, such as DNA particle bombardment. Alternatively, the DNA constructs may be combined with suitable T-DNA flanking regions and introduced into a conventional Agrobacterium tumefaciens host vector. The virulence functions of the Agrobacterium tumefaciens host will direct the insertion of the construct and adjacent marker into the plant cell DNA when the cell is infected by the bacteria. Thus, any method, which provides for effective transformation/transfection may be employed. See, for example, U.S. Pat. Nos. 7,241,937, 7,273,966 and 7,291,765 and U.S. Patent Application Publication Nos. 2007/0231905 and 2008/0010704 and references cited therein. See also, International Publication Nos. WO 2005/103271 and WO 2008/094127 and references cited therein. Techniques which have been used to transform oil palm include biolistic-mediated transformation and Agrobacterium-mediated transformation. See, for example, Masli et al. (2009); Omidvar et al. (2008); Parveez et al. (2008); Abdullah et al. (2005); Parveez et al. (2000); Chowdhury, et al. (1997); U.S. Patent Application Publication No. 2009/0038032; and International Publication No. WO 2010/071608.
[0049] Transformed plant cells which are derived by any of the above transformation techniques can be cultured to regenerate a whole plant which possesses the transformed genotype and thus the desired phenotype, e.g., a transgenic plant. A "transgenic plant" is a plant into which foreign DNA has been introduced. A "transgenic plant" encompasses all descendants, hybrids, and crosses thereof, whether reproduced sexually or asexually, and which continue to harbor the foreign DNA. Regeneration techniques rely on manipulation of certain phytohormones in a tissue culture growth medium, typically relying on a biocide and/or herbicide marker which has been introduced together with the desired nucleotide sequences. See for example, International Publication Nos. WO 2008/094127 and WO 2010/071608 and references cited therein.
[0050] The foregoing methods for transformation are typically used for producing a transgenic variety in which the expression cassette is stably incorporated. After the expression cassette is stably incorporated in transgenic plants, it can be transferred to other plants by sexual crossing. In one embodiment, the transgenic variety could then be crossed, with another (non-transformed or transformed) variety, in order to produce a new transgenic variety. Alternatively, a genetic trait which has been engineered into a particular Jatropha line using the foregoing transformation techniques could be moved into another line using traditional backcrossing techniques that are well known in the plant breeding arts. For example, a backcrossing approach could be used to move an engineered trait from a public, non-elite variety into an elite variety, or from a variety containing a foreign gene in its genome into a variety or varieties which do not contain that gene. As used herein, "crossing" can refer to a simple X by Y cross, or the process of backcrossing, depending on the context. Any of a number of standard breeding techniques can be used, depending upon the species to be crossed.
[0051] Once transgenic plants of this type are produced, the plants themselves can be cultivated in accordance with conventional procedures. Transgenic seeds can, of course, be recovered from the transgenic plants. These seeds can then be planted in the soil and cultivated using conventional procedures to produce transgenic plants. The culitvated transgenic plants will express the DNA of interest in a tissue-preferred or tissue-specific manner as described herein.
[0052] In a fifth aspect, the present invention provides methods of increasing seed oil accumulation in Jatropha, such as Jatropha curcas. In one embodiment, a method involves modulating the level of activity of a Sugar-dependent 1 (SDP1) gene, such as JcSDP1 gene, which encodes a patatin-domain triacylglycerol lipase in the host Jatropha cell or Jatropha plant. The level of activity of the lipase can be reduced by reducing expression of the SDP1 gene. In one embodiment, the modulation of the level of activity of the lipase is a reduction in the activity of the lipase. The level of activity of the lipase can be reduced by using RNAi techniques described herein in which the lipase is the target for the RNAi. Alternatively, the level of activity of the lipase can be reduced using VIGS techniques as described herein in which at least a partial fragment of the target gene is used. In one embodiment, Jatropha RNAi plants accumulate about 13% to about 30% higher seed oil than control plants.
[0053] The present invention shows that SDP1-deficiency enhances seed oil accumulation in the model plant Arabidopsis. Based on this result, RNAi technology and the native JcSDP1 promoter was used to generate transgenic Jatropha plants with reduced endogenous SDP1 expression. The present invention shows that SDP1-deficient transgenic Jatropha plants, such as those produce by RNAi technology, accumulate about 13% to about 30% higher seed oil than control plants. Free fatty acid content in seed was reduced from about 27% in WT to about 8.5% in SDP1-deficient transgenic Jatropha plants, such as those produce by RNAi technology. This transgenic technology leads to an enhanced agronomic trait for this biodiesel crop.
[0054] The practice of the present invention employs, unless otherwise indicated, conventional techniques of chemistry, molecular biology, microbiology, recombinant DNA, genetics, immunology, cell biology, cell culture and transgenic biology, which are within the skill of the art. See, e.g., Maniatis et al., 1982, Molecular Cloning (Cold Spring Harbor Laboratory Press, Cold Spring Harbor, N.Y.); Sambrook et al., 1989, Molecular Cloning, 2nd Ed. (Cold Spring Harbor Laboratory Press, Cold Spring Harbor, N.Y.); Sambrook and Russell, 2001, Molecular Cloning, 3rd Ed. (Cold Spring Harbor Laboratory Press, Cold Spring Harbor, N.Y.); Ausubel et al., 1992), Current Protocols in Molecular Biology (John Wiley & Sons, including periodic updates); Glover, 1985, DNA Cloning (IRL Press, Oxford); Russell, 1984, Molecular biology of plants: a laboratory course manual (Cold Spring Harbor Laboratory Press, Cold Spring Harbor, N.Y.); Anand, Techniques for the Analysis of Complex Genomes, (Academic Press, New York, 1992); Guthrie and Fink, Guide to Yeast Genetics and Molecular Biology (Academic Press, New York, 1991); Harlow and Lane, 1988, Antibodies, (Cold Spring Harbor Laboratory Press, Cold Spring Harbor, N.Y.); Nucleic Acid Hybridization (B. D. Hames & S. J. Higgins eds. 1984); Transcription And Translation (B. D. Hames & S. J. Higgins eds. 1984); Culture Of Animal Cells (R. I. Freshney, Alan R. Liss, Inc., 1987); Immobilized Cells And Enzymes (IRL Press, 1986); B. Perbal, A Practical Guide To Molecular Cloning (1984); the treatise, Methods In Enzymology (Academic Press, Inc., N.Y.); Methods In Enzymology, Vols. 154 and 155 (Wu et al. eds.), Immunochemical Methods In Cell And Molecular Biology (Mayer and Walker, eds., Academic Press, London, 1987); Handbook Of Experimental Immunology, Volumes I-IV (D. M. Weir and C. C. Blackwell, eds., 1986); Riott, Essential Immunology, 6th Edition, Blackwell Scientific Publications, Oxford, 1988; Fire et al., RNA Interference Technology: From Basic Science to Drug Development, Cambridge University Press, Cambridge, 2005; Schepers, RNA Interference in Practice, Wiley-VCH, 2005; Engelke, RNA Interference (RNAi): The Nuts & Bolts of siRNA Technology, DNA Press, 2003; Gott, RNA Interference, Editing, and Modification: Methods and Protocols (Methods in Molecular Biology), Human Press, Totowa, N. J., 2004; Sohail, Gene Silencing by RNA Interference: Technology and Application, CRC, 2004.
EXAMPLES
[0055] The present invention is described by reference to the following Examples, which are offered by way of illustration and are not intended to limit the invention in any manner. Standard techniques well known in the art or the techniques specifically described below were utilized. The following abbreviations are used: DAP: Days After Pollination; FAME: Fatty Acid Methyl Ester; FFA: Free Fatty Acid; GC: Gas Chromatography; GC/MS: Gas Chromatography/Mass Spectrometry; GUS: beta-glucuronidase; PTLs: Patatin-like TAG Lipases; qRT-PCR: Quantitative real time PCR; RNAi: RNA interference; SEM: Scanning Electron Microscopy; SDP1: Sugar-dependent1; TAGs: Triacylglycerols; TEM: Transmission Electron Microscopy; TLC: Thin Layer Chromatography; WAF: Weeks After Fertilization.
Example 1
Materials and Methods
[0056] Plant Materials and Growth Condition:
[0057] Arabidopsis Col-0 (Arabidopsis thaliana ecotype Columbia-0) was grown on soil with sand. Homozygous sdp1-5 mutant (Salk--076697, Arabidopsis Biological Resource Center, Ohio State University, Columbus, USA) seeds were stratified at 4° C. for 3 days on soil and cultured in a growth chamber under 16 hr-light/8 hr-dark cycles, 23° C.±3° C. and white light (100-150 μEm-2s-1 photosynthetically active radiation). Rosette leaves were harvested from 3 or 4 week-old plants and other organs such as inflorescence stems, cauline leaves, flowers, siliques, and developing seeds were harvested from 6 week-old plants. Seeds from siliques of 3 to 5 DAP (DAP: day after pollination) were used as the early stage of seed maturation in Arabidopsis. Leaves and developing fruits (S1; 1WAF, S2; 2 to 3WAF, S3; 4 to 6WAF, and S4; 7 to 8WAF, WAF: weeks after fertilization) were collected from Jatropha plants grown in a greenhouse.
[0058] RNA Isolation and Quantitative Real-Time PCR:
[0059] Total mRNA was isolated from plant samples using TRIzol reagent (Invitrogen, St. Louis, Mo., USA) according to the manufacturer's instructions. cDNA was synthesized with 1 μg total mRNA using MMLV Superscript II (Promega, Madison, Wis., USA) after DNase I treatment (Roche Applied Science, Mannheim, Germany). Quantitative real-time PCR assay was performed with an ABI 7900 sequence Detection. System (Applied-Biosystems, Foster City, Calif., USA). Power SYBR Green PCR Master Mix (Applied-Biosystems) reagent protocol was used but reduced the volume to 10 μL per reaction. As controls, the species-specific tubulin and actin primer sets for Jatropha curcas and Arabidopsis thaliana, respectively, were used. Fold change values of the target gene transcripts were subsequently normalized by dividing the ΔCt values with the ΔCt values of each control gene transcript. All real time PCR experiments were performed in triplicates using different biological samples.
[0060] Isolation of JcSDP1 Full Length cDNA and its Promoter:
[0061] A partial SDP1 sequence in our Jatropha curcas EST database was found. Using this partial sequence, 5' or 3' cDNA RACE primers were designed and RACE experiments were performed using BD SMART® RACE cDNA Amplification Kit (BD Bioscience Clontech, Franklin Lakes, N.J., USA). One μg total RNA derived from developing seeds (S2 to S3) was used for the generation of 5' or 3' RACE pools. The 5' and 3' cDNA ends were obtained by touchdown PCR with advantage 2 PCR Enzyme System (BD Bioscience Clontech) following the manufacturer's instructions. To specifically amplify the products, the primary RACE products were diluted and mixed with a nested gene specific primer (GSP2) and the nested universal primer mixture (AP2). The PCR products were gel purified, cloned into pDrive Cloning Vector (Qiagen, Dusseldorf, Germany) to obtain full-length JcSDP1 cDNA (2577-bp). The Universal GenomeWalker kit (BD Bioscience Clontech) was used to isolate the promoter fragment of the JcSDP1 gene. Genomic DNA was restricted by DraI, EcoRV, PvuII, SspI, and StuI endonuclease and five libraries of adaptor-ligated genomic fragments were constructed. These genomic DNA libraries were used as templates for the PCR reactions for promoter isolation. For each round of genome walking, the primary PCR products were amplified by a gene-specific primer (GSP1) and the outer adaptor primer (AP1). To the second PCR reaction mix, the primary products were diluted and used as templates with a nested gene specific primer (GSP2) and the nested adaptor primer (AP2). The secondary PCR products were then analyzed on agarose gels and the relevant DNA fragment purified with QIAEXII Gel Extraction Kit (Qiagen), cloned into pDrive Cloning Vector (Qiagen) and sequenced. Potential cis-elements in the promoter region were analyzed using computational analytical methods available on two public web-sites, PlantCare (http://bioinformatics.psb.ugent.be/webtools/plantcare) and PLACE (http://www.dna.affrc.go.jp).
[0062] Construction of Pro JcSDP1:GUS Fusion Gene and Promoter Analysis:
[0063] pKGWFS7 destination vector including GFP or GUS reporter gene was used for promoter analysis. The 5'-flanking region of JcSDP1 was amplified using two specific primers, JcSDP1-PF1T and JcSDP1-PR1. This promoter was 873-bp in length, including 149-bp of 5' untranslated region (UTR). The amplified PCR product was inserted into TOPO donor vector using the pENTR®/D TOPO Cloning Kit (Invitrogen), and then inserted into the destination vector, pKGWFS7, using the Gateway LR Clonase® II enzyme mix (Invitrogen). For plant transformation, the constructs were introduced into Agrobacterium tumefaciens strain AGL1 using electroporation. Constructs were transformed into WT (Col-0) or sdp1-5 mutant (Salk--076697) background through the floral dip method described by Clough and Bent (38).
[0064] For transient assay of JcSDP1 promoter expression, 10 μg of ProJcSDP1:GUS in pKGWFS7 plasmid DNA was coated with 1 μm diameter gold particles (2.5 mg gold particles, 200 μL of 2.5 M CaCl2 and 100 μL of 0.1 M Spermidine). After incubating on ice for 30 min, the pellet of DNA/gold particle was washed twice with 70% ethanol and re-suspended in 100% ethanol. Jatropha fruits and leaves were centered in a petri-dish containing MS agar medium (39). Fruits and leaves were bombarded at 1,350 psi with a biolistic helium gun device (Bio-rad PDS-1000/He, USA). After incubation for 2 days at 25° C. the bombarded tissues were analyzed by histochemical assays as described by Jafferson et al. (40). The tissues were incubated in GUS staining buffer [0.1 M Sodium phosphate (pH 7.0), 1 mM 5-bromo-4-chloro-3-indolyl-D-glucuronide (Sigma, St Louis, Mo., USA), 0.5 mM potassium ferrocyanide, 0.5 mM potassium ferricyanide, 10 mM Na2EDTA, and 0.1% Triton X-100] for 20 hrs at 37° C. Stained tissues were rinsed with 70% to 80% ethanol until pigments such as chlorophylls had been cleared. Selected organs of ProJcSDP1:GUS transgenic Arabidopsis lines were analyzed for GUS expression. To investigate the sugar-responsive expression of JcSDP1 promoter, we used 14 days-old seedlings of T2 lines and incubated for 24 hrs in an MS media with 1% or 3% of a sugar source: sucrose, fructose, and glucose. Mannitol was used as a control for osmotic stress.
[0065] Complementation of the JcSDP1/Sdp1-5 Mutation in Arabidopsis:
[0066] Full-length JcSDP1 cDNA was cloned into a vector harboring a tandem CaMV35S promoter and a Nos polyA addition sequence from pCAMBIA 1300 and transformed into Arabidopsis sdp1-5 mutant (Salk--076697). T1 transformants were selected on 40 μg/mL hygromycin and T2 seedlings were assayed for sugar responses on MS agar plates without sucrose.
[0067] Construction of Inducible Marker-Free ProJcSDP1: JcSDP1-RNAi Vector and Transformation to Jatropha Plants:
[0068] The pCCreloxP (pCCLB3) inducible marker free vector system was used as described by Qiu et al. (41). The T-DNA of pCCreloxP vector harbors a loxP fragment that consists of CRE-int, HPT and XVE genes (FIG. 5A). To silence JcSDP1 expression using RNAi a 373-bp fragment was selected that includes 290-bp 3' coding region of JcSDP1 cDNA with a stop codon and 83-bp of the 3' UTR region. The 373-bp JcSDP1 fragment was amplified with two primer sets, JcSDP1-RNAiF-XhoI and JcSDP1-RNAiR2-HindIII or JcSDP1-RNAiF-BamHI and JcSDP1-RNAiR2-PstI, used for the sense orientation or antisense orientation, respectively, and the fragments were then cloned into pBluescript SK intron vector (pBS-Ski) which has an intron (156-bp) to generate pJcSDP1-RNAi as previously described (42). ProJcSDP1 was amplified with two primers, JcSDP1-PF1-ApaI and JcSDP1-PR1-XhoI and then restricted with ApaI/XhoI. To insert the ProJcSDP1 (JcSDP1 promoter) fragment into the pJcSDP1-RNAi construct, we used ApaI and XhoI restriction enzyme sites in the pJcSDP1-RNAi. A 226-bp fragment containing the CaMV35S polyA addition sequence (T35S) was amplified with T35S-F-XbaI and T35S-R-Pml-SacII primers. The amplified T35S fragment was cleaved with XbaI/SacII restriction enzymes and then cloned into ProJcSDP1-RNAi. Finally, the ProJcSDP1:JcSDP1-RNAi:T35S fragment from the ProJcSDP1-RNAi construct was inserted into the pCCreloxP marker free vector at the ApaI and PmlI sites. All constructs were introduced into Agrobacterium tumefaciens strain AGL1 using electroporation and then transformed into Jatropha using cotyledon explants (43).
[0069] Fatty Acid Analysis in Arabidopsis and Jatropha:
[0070] We used gas chromatography (GC) to analyze fatty acid profiles of the sdp1-5 mutant. (44). 100 dried seeds of each line were weighed and samples were transmethylated at 85° C. for 2 hrs in a reaction buffer (1 mL of 3 M HCl-methanol (Sigma), 25 μL of BHT solution (Sigma), and 300 μL of toluene (Sigma)). 50 μg of pentadecanoic acid (C15:0, Sigma) was added to each sample as an internal standard. After being cooled down to room temperature, 1.5 mL of 0.9% NaCl (w/v) was added to the mix and the fatty acid methyl esters (FAMEs) were extracted two times with 1 mL of hexane. Extracts were evaporated under nitrogen and then dissolved in 100 μL of the hexane. The final extracts were analyzed with GC using a flame ionization detector (FID) on Agilent 6890 (Agilent, Santa Clara, Calif., USA) employing helium as the carrier gas. Total fatty acids were estimated by comparing the total FAME peak area (pA*sec) to that of the C15:0.
[0071] To analyze fatty acid content in Jatropha transgenic lines, dried endosperm of seed was used as described previously (7). Jatropha samples were transmethylated at 70° C. for 20 min in a reaction buffer (1 mL of 3 N methanolic-HCl, 400 μL of 2,2, dimethoxypropane (Sigma) and 50 μg of pentadecanoic acid (C15:0)). After being cooled down to room temperature, the fatty acid methyl esters (FAMEs) were washed with 1 mL of water and 1 mL hexane twice. The extracts were evaporated under nitrogen and then dissolved in 500 μL of hexane. The final samples were analyzed by GCMS-QP2010 (Shimadzu Corporation, Japan). Standard deviation was calculated based on several different plants.
[0072] Analysis of Lipids by TLC and Quantification by GC/MS:
[0073] Seed lipids were extracted with hexane three times (7, 44). After determination of total lipid amount, 300 μg of neutral lipid were fractionated by TLC (Thin Layer Chromatography) on silica gel plates in a running solvent mixture, hexane: ethyl acetic acid: acetic acid (90:10:1 by vol). Triolein (Sigma T7140) and Oleic acid (Sigma 75090) were used as a standard of TAG and of FFA, respectively. TLC plates were exposed to iodine (I2) vapor for visualization. The separated neutral lipid species including TAG and FFA were recovered from the plates using hexane and quantified by GC/MS after conversion to their corresponding methyl esters by methanolic-HCl method according to Li et al. (44). The absolute amount was calculated against the C15:0 internal standard by comparing their relative peak areas.
[0074] TEM and SEM Analysis:
[0075] TEM was performed with mature dried seeds of WT (Col-0) and sdp1-5 mutant. Seeds were embedded in resin and sectioned by an ultramicrotome (Leica Ultracut UCT; Leica, Germany) set at 70-nm thickness. Sectioned samples were placed onto 300 mesh cooper grids. Sections were observed and pictures were taken with transmission electron microscope (JEM-1230; JEOL, Japan) at 120 kV. For SEM analysis dried seeds were mounted directly and observed using a JSM-6360LV (JEOL, Japan) with an acceleration voltage of 20 kV.
[0076] Seed Weight and Size Measurement:
[0077] Mature seeds were harvested from WT (Col-0) and sdp1-5 mutant grown under the same conditions. Sample size of 100 seeds per WT (Col-0) or sdp1-5 mutant was used to obtain an average seed weight with at least five times biological replications. Average values are given along with SD (n=5). The Leica DM5000B microscope (Germany) and Image) analysis software were used to measure seed sizes. Average values are given along with SD (n=10).
[0078] Primers:
[0079] Sequence information on primers used in the experiments is set forth in Table 1 The SEQ ID NO: is set forth in parenthesis.
TABLE-US-00001 TABLE 1 Primer Sequences Promoter analysis (SEQ ID NO:) JcSDP1-PF1T CACCtacatccagtagattgcacgtcacacacta (8) JcSDP1-PR1 ctctaaaagtttcgtgttttggatgatttgct (9) Gene construction JcSDP1-CF-Sal ttggtcgacatggatataagtaatgaggccaatgt (10) JcSDP1-CR-Nhe attgctagcaccatccacagaactttgatcctgtc (11) JcSDP1-RNAiF-Xho atactcgagtccataatgggattgtgttgaacgtcgt (12) JcSDP1-RNAiR2-H3 tcgaagcttgctagtggcacttgatattaaggtgat (13) JcSDP1-RNAiF-Bm ataggatcctccataatgggattgtgttgaacgtcgt (14) JcSDP1-RNAiR2-Pst tctttctgcagctagtggcacttgatattaaggt (15) JcSDP1-PF1-ApaI tatgggccctacatccagtagattgcacgtcacaca (16) JcSDP1-PR1-Xho atgctcgagctctaaaagtttcgtgttttggatgattt (17) T35S-F-Xba acttctagacggccatgctagagtccgcaa (18) T35S-R-Pml-SacII ttaccgcggcacgtgaggtcactggatttt (19) MF genotyping HygF aaaaagcctgaactcaccgcgacgtct (20) HygR tacttctacacagccatcggtcca (21) MF-P1 ctgaattgtcgaggtcgaagatc (22) JcSDP1-R16 agcagccaaatgggtctgtcc (23) T35S-R cttctcattatcggtggtgaacat (24) qRT-PCR At2g37620 AtActin F ctctgtctggattggagggtc (25) AtActin R gcttgagaaatggtcggaaa (26) At4g25140 AtOleo1-F tgccgctataaccgttttctcttgga (27) AtOleo1-R atgttccccaccagtatgttgctgtcc (28) At5g40420 AtOleo2-F atgcggttggctacgcaggacaa (29) AtOleo2-R tcatgcagccgtcgtcctcccet (30) At5g51210 AtOle3-F tggaatagccgccattaccgcctt (31) AtOle3-R agaaacttgttggtgttggactccaatg (32) At5g04040 AtSDP1-QF atgccatcgattgggaacca (33) AtSDP1-QR caccggttcttgtaaaccggaat (34) JcTubulin F gaggctggatctggcaaacacgtt (35) JcTubulin R tgtgtaatgacctctagcaaaatta (36) JcSDP1-QF cataatgggattgtgttgaacgtcgt (37) JcSDP1-QR gtcatcatcgtcatccccagcata (38) Fatty acid related genes in Arabidopsis At3g12120 AtFAD2-F cctcagcctctctcttacttggctt (39) AtFAD2-R ccaggagaagtaagggacgag (40) At2g29980 AtFAD3-F ctacttgttggtccataatgttcgtca (41) AtFAD3-R cgtagataactccattccttgcct (42) At4g34520 AtFAE1-F gtcttaaccaactttttcaacctctgtt (43) AtFAE1-R ccaaaccgaaaacagtgaaagcaaagagt (44) At3g05020 AtACP1-F aatctatccttcaacctccgccgt (45) AtACP1-R agagaatctgctccaaggtcagct (46) At5g15530 AtBCCP2-F ggatctcctttccctccgatgttt (47) AtBCCP2-R ataaattcagagagctcggcgggt (48) At3g22960 AtCh-PK-F tggcatcactggtctcccgatgt (49) AtCh-PK-R tcatacctcttgatttcagtaacgaga (50) At5g52920 AtCy-PK-F agtcactatcgtccttccggcacaa (51) AtCy-PK-R cgctctgtacgattgctatttcctct (52) At5g49190 AtSUS2-F tcaagtcagttcactgcagatctaat (53) AtSUS2-R caactcggtaaagaccaggcatagtgaa (54) At5g46290 AtKAS1-F tcgcaaaacacacatcacacac (55) AtKAS1-R gtgattgacgatttgatggtaag (56) At1g62640 AtKASIII-F tggtcttcctgatctgcc (57) AtKASIII-R ccgcttctcactgcctcat (58)
[0080] Protein and Carbohydrate Analysis:
[0081] Protein content in Jatropha transgenic lines was analyzed according to Focks and Benning (45) with 50 mg of dried endosperm. Protein amounts were measured by the Lowry DC protein assay (Bio-Rad) using gamma-globulin as a standard. To analyze carbohydrate content, 50 mg of dried endosperm were homogenized in 200 μL of assay buffer and centrifuged at full speed. The extracted supernatant was used for carbohydrate quantification using Total Carbohydrate Assay Kit (Sigma). D-glucose was used as a standard for calibration.
[0082] gDNA Isolation and Southern Blot:
[0083] Total genomic DNA was isolated from leaves from transgenic or control (CK; 35S:GFP) plants grown in a greenhouse. We used CTAB (cetyltrimethylammonium bromide, Sigma) method (46). Genomic DNA was digested with restriction enzymes and separated on 0.8% agarose gels. The gels were processed and blotted onto Hybond-N.sup.+ membranes (Roche, Germany) following standard procedures (47). Probes were prepared with PCR DIG Labelling Mix using specific primer sets for Hygromycin phosphotransferase (HPT) gene and JcSDP1 gene. Hybridization was performed using the PCR DIG detection kit following the supplier's instructions (Roche, Germany).
Example 2
Sdp1-5 Accumulates TAG in Mature Dried Seeds
[0084] We obtained from the ABRC stock center an Arabidopsis mutant (Salk--076697) with T-DNA insertion in the SDP1 (At5g04040) locus and this mutant was designated sdp1-5 [15]. Using RT-PCR as a screen we obtained 3 homozygous lines (#24, 30 and 33) of the sdp1-5 null allele and one line (#33) was chosen for further experiments (Figure S1B). SDP1 transcript levels in sdp1-5 (#33) were 20-fold lower than in WT (Col-0) during early stages of seed development (3 to 5 DAP, DAP: Day after pollination) (FIG. 6C).
[0085] We first examined the effect of SDP1 deficiency on seed development, total fatty acid content and fatty acid profile. Scanning Electron Microscopy (SEM) analysis showed that sdp1-5 seeds were slightly larger than those of WT (Col-0) in length and width (FIG. 1A; Table 2). In addition, sdp1-5 seeds displayed an increase in dry weight of around 11.5% as compared to WT. To investigate the role of SDP1 in seed oil accumulation, total oil content and fatty acid composition in dried seeds were compared between WT (Col-0) and the sdp1-5 mutant. FIG. 1 B and C show that the average dry seed weight of WT (Col-0) was about 19 μg containing approximately 5.54 μg of total fatty acids. The seed lipid content obtained by us is very similar to those reported by others, which is around 30-35% of dry seed weight. On the other hand, sdp1-5 seeds had an average dry weight of around 22 μg per seed containing 7.17 μg of total fatty acids. Therefore, sdp1-5 seeds have about 10% higher levels of total fatty acids compared to WT (Col-0) seeds (FIG. 1D). In addition, there was a clear increase in the relative proportion of unsaturated fatty acids, such as linolenic acid (C18:3) and eicosenoic acid (C20:1) in sdp1-5 seeds (FIGS. 1E and 1F). To characterize TAG accumulation in mature dried seeds, we analyzed total neutral lipid from WT (Col-0) and 3 homozygous lines of the sdp1-5 null allele by thin layer chromatography on silica gel plates. FIG. 1G shows reduced levels of free fatty acids (FFA) in sdp1-5 compared to WT (Col-0). Triolein and oleic acid were used as standards of TAG and FFA, respectively. To obtain quantitative data, we analyzed the samples using GC/MS and pentadecanoic acid (C15:0) was used as an internal control for quantitation. The sdp1-5 mutant had about 4.25% FFA and 95.75% TAG compared to 13.35% FFA and 86.65% TAG in WT (Col-0) (FIG. 1H).
TABLE-US-00002 TABLE 2 Seed Weight and Size of WT (Col-0) and sdp1-5 Weight of seed Length of seed Width of seed Name (μg/seed)a (μm/seed)b (μm/seed)b Col-0 19.00 ± 1.10 481.12 ± 3.36 276.70 ± 10.58 sdp1-5 21.00 ± 0.57**c 522.86 ± 7.56*** 294.29 ± 15.12* aSeed weight determination using 100 mature seeds. Average values are given along with SD (n = 5). bThe length and width of seed were measured using mature dried seeds. Average values are given along with SD (n = 10). c*P < 0.05, **P < 0.01 or ***P < 0.001 versus WT (Col-0) seed.
[0086] To examine the molecular basis of these changes in lipid content and fatty acid profile, we analyzed transcript levels of key genes involved in fatty acid biosynthesis. We found FAD3 was up-regulated in sdp1-5 (FIG. 7). Previously, Puttick et al. (17) reported that in Arabidopsis over-expression of FAD3 leads to an accumulation of high levels of linolenic acid and that is inversely correlated with linoleic acid (18:2) levels. We also observed an inverse correlation between linolenic acid and linoleic acids levels in sdp1-5 seeds. In addition, we found an increased accumulation of eicosenoic acid, a very-long-chain fatty acid that has been used as a metabolic marker for storage TAGs formation in Arabidopsis seeds (18). We found sdp1-5 seeds accumulated 1.3 μg of eicosenoic acid per seed which was 40% higher than that of WT (Col-0) seeds. The increased eicosenoic acid level in sdp1-5 seeds was likely due to the up-regulated Fatty Acids Elongase 1 (FAE1) (FIG. 7). Our results suggest that SDP1-deficiency is closely correlated with seed size augmentation, the relative proportion of unsaturated fatty acids and the accumulation of TAGs in mature seeds.
Example 3
Sdp1-5 Mutant has Increased Numbers of Oil Bodies in Dried Seeds
[0087] Plant storage lipids, predominantly triacyglycerols (TAGs), are sequestered by monolayer phospholipids with embedded small proteins, such as oleosin, to form oil bodies (19). Previous work has highlighted the importance of oleosins for oil body structure and TAG accumulation in mature seeds. We used Transmission Electron Microscopy (TEM) to analyze the formation of oil bodies in sdp1-5 seeds. Mutant sdp1-5 seeds contained increased number of oil bodies but they were relatively smaller in size compared to those in WT (Col-0) seeds (FIG. 2A). The cross-sectional area of one mature seed cell was about 196 μm2. Within this area, there were around 175 oil bodies in WT (Col-0) but about 216 oil bodies in sdp1-5, representing an increase of approximately 23% (FIG. 2B). Because oleosins are important proteins for seed oil body formation we investigated expression levels of 3 different oleosin genes in sdp1-5 by qRT-PCR. FIG. 2C shows transcript levels of AtOLE1 (At4g25140) and AtOLE2 (At5g40420) were two fold higher in sdp1-5 compared to WT (Col-0). These results suggest the increased number of oil bodies in sdp1-5 is possibly due to the enhanced expression of oleosins.
Example 4
JcSDP1 an Ortholog of AtSDP1 Rescues the SDP1-Deficiency in Arabidopsis
[0088] To investigate the impact of SDP1 deficiency in an oil seed crop, we isolated full-length JcSDP1 cDNA from Jatropha seed RNA samples using 5' and 3' cRACE technology. JcSDP1 encodes a protein of 858 amino acids with a molecular mass of approximately 96 kDa. A BLAST search revealed that JcSDP1 has high sequence homology (76%) to the Arabidopsis SDP1 (At5g04040). JcSDP1 has at least three predicted trans-membrane domains and four Site-1 protease (SIP) target sequences at the N terminus (RXXL; SEQ ID NO:5). Moreover, JcSDP1 has a conserved patatin domain with lipase activity sequences such as the oxyanion hole motif (GXGXXG; SEQ ID NO:6) and a lipase consensus motif with a catalytic serine (GXSXG; SEQ ID NO:7) (FIG. 3A). The high similarity of sequences and domains between JcSDP1 and AtSDP1 implied a possible similar function of JcSDP1 in TAGs metabolism. To investigate the function of JcSDP1 in TAG degradation during early stages of seed germination, we transformed JcSDP1 under the control of a CaMV35S promoter into the Arabidopsis sdp1-5 mutant. The Arabidopsis sdp1-5 showed retarded growth on sugar depleted MS medium and but the retarded growth can be rescued by the supplementation of 1% sucrose (15). Seven day-old seedlings of sdp1-5 plants expressing the heterologous JcSDP1 displayed normal growth on MS medium without sucrose whereas sdp1-5 mutant displayed retarded growth (FIG. 3B). This result indicates that JcSDP1 is able to rescue the retarded growth phenotype of sdp1-5 providing evidence JcSDP1 is an ortholog of AtSDP1.
Example 5
Characterization of JcSDP1 Promoter
[0089] To specifically silence JcSDP1 gene expression in Jatropha, we isolated a native JcSDP1 promoter fragment from Jatropha genomic DNAs using a Genome Walker kit (Clontech). We cloned an approximately 0.7 kb fragment of JcSDP1 proximal to the 5'-UTR, which contained several putative cis-elements for gene expression and regulation. This region of the JcSDP1 promoter contains a TATA box and a CAAT box, located at -13 to -16 and -66 to -69, respectively. The promoter region also contains two putative sugar-responsive elements found in the alpha amylase gene, TATCCA and TAACAAA (20,21), located at -154 to -159 and -139 to -145, respectively. In addition the promoter fragment includes four E-box motifs, CANNTG (22, 23), which are likely involved in seed specific expression (FIG. 4A).
[0090] To see if the JcSDP1 promoter would show seed specific expressions, we generated a ProJcSDP1:GUS expression clone using the pKGWFS7 gateway vector system. The expression patterns of JcSDP1 promoter were examined in homologous and heterologous systems. To transiently analyze JcSDP1 expression in a homologous system, we introduced the ProJcSDP1:GUS fusion gene into developing fruits and leaf of Jatropha. We found the GUS gene was transiently expressed in developing Jatropha fruits, especially the endosperm part of the seed whereas leaf tissues did not show any GUS expression (FIG. 4B). This result suggests that the JcSDP1 promoter has seed-specific expression in Jatropha plant. We also analyzed JcSDP1-GUS expression in developing seeds of transgenic Arabidopsis plants expressing ProJcSDP1:GUS transgene. FIG. 4C shows that GUS expression was highly elevated at 3-4 days (globular embryos) after pollination (DAP) and also at 9-10 DAP (mature green embryos) (FIG. 4C-h and i) following the developmental stages described by Le et al. (24). These expression patterns were consistent with the expression pattern of JcSDP1 which was highly activated at stage S2 (3 WAF) (FIG. 4E). These results imply that the JcSDP1 promoter is controlled in a seed-specific and development dependent manner.
[0091] Using a cis-element prediction program we found that the JcSDP1 promoter carries two sugar-responsive alpha amylase elements, such as TATCCA and TAACAAA (20, 21). To investigate sugar responses of JcSDP1, we used 14 day-old transgenic Arabidopsis seedlings harboring the ProJcSDP1:GUS transgene. We examined the effects on GUS expression of sucrose, glucose, or fructose (1% or 3% concentration) supplementation in MS(-) media. As a control, mannose was used as a source of osmotic stress. FIG. 4D shows that the JcSDP1 promoter showed sugar-dependent expression, and the expression was especially responsive to sucrose and fructose.
Example 6
JcSDP1-RNAi Transgenic Jatropha Plants Accumulate Increased Storage Lipids in Their Endosperm
[0092] We were interested to see if the reduction of SDP1 expression in Jatropha plants would also lead to an increased seed oil accumulation. To this end, we generated JcSDP1-RNAi transgenic Jatropha plants. To specifically control lipid accumulation in the mature seeds without ectopic gene silencing, we placed the JcSDP1-RNAi transgene under the control of a native JcSDP1 promoter (FIG. 9; SEQ ID NO:3) which is specifically expressed in Jatropha seeds. Through a two step selection process using hygromycin and β-estradiol, marker free or non-marker free transgenic plants were screened. These plants were further confirmed by genotyping with specific primer sets for HygF and HygR for the hygromycin gene and P1 and R16 or P1 and T35S-R for the marker free transgene. Based on the genotyping results, we obtained chimeric (heterozygote) marker-free plants in which the antibiotic selection marker was partially removed by homologous recombination. The transgenic lines displayed normal growth patterns and growth rates, leaf number and leaf size (FIG. 5B, panels d and e). In contrast to the enlarged seed size found in Arabidopsis sdp1-5, T1 mature seeds from JcSDP1-RNAi plants were normal in size (FIG. 5B, panels a, b, and c). To investigate oil content in T1 mature seeds, we carefully separated endosperms from embryos. Whereas the endosperms were used for further molecular and biochemical analysis, the embryos were regenerated to maintain the transgenic lines. After transgenic plants were fully grown and mature, we harvested mature seeds from four individual transgenic plants for further studies. Compared to control (CK) plants (35S:GFP lines), all tested transgenic lines accumulated increased oil content per dry seed weight in their endosperm (FIG. 5C). Endosperm of the best transgenic line (#158) accumulated raw oil content to about 54% of the dry seed weight; this represented a 30% increase of total oil in transgenic plants compared to 35S:GFP control (CK) plants. We analyzed JcSDP1-RNAi transgenic line #158 to investigate possible changes in protein and carbohydrate content along with the increased in lipid accumulation in endosperm. The protein content per endosperm of JcSDP1-RNAi decreased about 7% compared to the CK (35S:GFP) plant as shown in Table 3. However, no difference in the carbohydrate content was detected between the two.
TABLE-US-00003 TABLE 3 Protein and Carbohydrate Contents in Endosperm of Control (CK; 35S:GFP) and JcSDP1-RNAi Transgenic Plants Protein Carbohydrate Total Lipid Content Content Content Genotype (%, w/w) (%, w/w) (%, w/w) CK (35S:GFP) 41.41 ± 2.19 14.93 ± 0.16 13.89 ± 0.12 JcSDP1-RNAi #158 54.17 ± 2.13 13.99 ± 0.19 13.92 ± 0.13 Values are the mean ± SE of measurements on endosperms from individual seed (n = 4) of JcSDP1-RNAi #158 T1 transgenic and control (CK; 35S:GFP) plants grown in a greenhouse.
[0093] To characterize TAG accumulation in mature endosperm, we analyzed total neutral lipid from CK and 3 individual lines of #158 transgenic plants by thin layer chromatography on silica gel plates. FIG. S3A shows reduced levels of free fatty acids (FFA) in the #158 transgenic line compared to CK. In addition, the #158 transgenic plants had about 8.49% FFA and 91.50% TAG compared to 26.83% FFA and 73.17% TAG in CK plants (FIG. 8B). We verified the number of T-DNA insertions in the best transgenic lines by Southern blot analysis (See Additional file 4). Among the T1 progeny plants of the parental line #158, we recovered a line (#158-8) that carried a single T-DNA insert and another line (#158-11) with multiple inserts. Regardless of the copy number of insertion, both #158-8 and #158-11 showed a very similar level of total seed lipid accumulation (FIGS. 10A and 10B).
[0094] To determine possible alterations in fatty acid profiles of JcSDP1-RNAi transgenic lines, we analyzed extracts by gas chromatography-mass spectrometry (GCMS-QP2010). FIG. 5D shows that JcSDP1-RNAi transgenic plants displayed a very similar fatty acid profile to that of CK plants. In contrast to the changes of fatty acids in the Arabidopsis mutant sdp1-5, we were unable to find any significant accumulation of C20:1 (less than 0.5% of total TAGs) in the transgenic Jatropha lines. The observed differences between the effects of SDP1 knock-down in Arabidopsis and Jatropha might be caused by physiological differences in oil accumulation. Indeed, reductions of JcSDP1 transcript levels were observed in JcSDP1-RNAi transgenic lines (FIG. 5E). Moreover, the increased oil accumulation in seeds was inversely correlated to the residual JcSDP1 transcript levels as assayed by quantitative real-time PCR (qRT-PCR). Taken together, we conclude that that suppression of JcSDP1 expression using RNAi technology can lead to enhanced oil accumulation in mature dried Jatropha seeds.
Example 7
SDP1-Deficiency Enhances Total Seed Oil Accumulation in Arabidopsis and Jatropha
[0095] The breakdown of TAGs into fatty acids is initiated by lipases many of which have been identified in eukaryotes including plants, yeast and animals (11, 12, 25). One of the characterized lipases is encoded by the Arabidopsis SUGAR DEPENDENT1 (SDP1). This lipase contains a patatin-like domain and is tightly associated with the oil body (13, 26, 27). Three SDP1 homologous genes have been identified in Arabidopsis and all 3 play important roles in storage oil mobilization (13, 14, 28). Besides its role in seed germination SDP1 is also active during the seed desiccation stage to hydrolyze TAGs (15).
[0096] Seed development in Arabidopsis consists of two major phases: early embryogenesis which is completed within 6 days after pollination (DAP) and seed maturation phase. The latter process can be further defined into three sub stages; early-maturation, mid-maturation and late-maturation. In the early-maturation stage embryos accumulate increasing amount of starch and begin to synthesize storage oils and proteins (7-10 DAP). During the mid-maturation stage (11-16 DAP) the amount of starch is dramatically reduced and this is inversely related to the accumulation of oils and proteins. Finally, in the late-maturation stage which is also known as desiccation stage, embryos gradually become metabolically quiescent with the exception that they synthesize sugars such as sucrose and trehalose to maintain integrity during the desiccation processes (29, 30). Chia et al. (14) reported that TAGs in oil bodies are important carbon sources for the proceeding of desiccation in late-maturation stage. During the desiccation process, around 10-14% of TAGs are consumed as carbon sources. Many enzymes, such as malate synthase (MS), isocitrate lyase (ICL), 3-ketoacyl CoA thiolase (KAT), hydroxyacyl CoA dehydrogenase (HD), enoyl hydratase (EH), and phosphoenolpyruvate carboxykinase (PEPCK), are involved to metabolize TAG (11, 12, 25). The Arabidopsis sdp1/sdp1L double knockout mutant display a similar phenotype as the glyoxylate cycle mutant id or 02, which lacks isocitrate lyase. These mutants are unable to synthesize carbohydrates from fatty acids (28, 31). In the sdp1-5, TAG degradation is almost completely blocked because SDP1 is the first enzyme for TAG metabolism in the desiccation process of the late-maturation stage (15, 28).
[0097] Based on these studies, we hypothesized that SDP1-deficiency might block TAG metabolism during the desiccation process thereby reducing the loss of TAG in the late-maturation stage. To test this hypothesis we isolated the Jatropha SDP1 homolog and showed that it is functional equivalent to the Arabidopsis SDP1 by genetic complementation of the Arabidopsis sdp1-5 mutant (FIG. 3B). Moreover, we showed SDP1-deficiency generated by RNAi technology produced a notable increase of seed oil accumulation in transgenic Jatropha plants. Our results suggest that the technology can be applied to enhance seed oil accumulation in other oil seed crops as well. It is possible that seeds of SDP1-deficient transgenic plants may be partially blocked in the desiccation process which may negatively impact their long-term shelf life. Moreover, the inhibition of TAG degradation may retard seedling growth and reduce seedling vigor. However, in the case of Jatropha, these issues can be mitigated by the industrial scale production of clonal transgenic plants by tissue culture. In addition, the issue of reduced seedling vigor can be obviated by germinating seeds in a sucrose-supplemented medium and selling germinated seedlings.
[0098] In this work, we have used the seed-specific promoter of JcSDP1 for regulated expression of the JcSDP1-related RNAi construct. The JcSDP1 promoter was chosen because we showed its activity peaks in the early stage of developing seed and gradually returns to basal levels in the late stage (FIG. 4E). Moreover, the JcSDP1 promoter is responsive to sugars, especially sucrose and fructose (FIG. 5). Based on these findings, we used the JcSDP1 promoter to express JcSDP1-related RNAi sequence so as to establish a feed-back inhibition system. For instance, if the gene silencing is not strong enough to knock down JcSDP1 expression, the residual JcSDP1 would degrade TAGs into free fatty acids which can be converted into sucrose, trehalose, and proteins. Once sucrose is elevated to a certain critical level in the endosperm, it can activate the JcSDP1 promoter which then in turn enhances the suppression of JcSDP1 gene expression. The gene silencing strategy we showed here has several advantages over the use of a CaMV35S promoter: 1) using the cognate promoter of a target gene can offer more specific regulation of the RNAi-dependent gene silencing against the target gene itself; 2) the end product of the target gene can be used for its own feed-back suppression; and 3) reduced ectopic expression of transgene.
Example 8
SDP1-Deficient Transgenic Jatropha Plants are Advantageous for the Process of Biodiesel Production
[0099] Biodiesel is commonly produced from crude oil by alkaline or acid-treatment processes which are known as trans-esterification (32, 33). Owing to the short reaction time and reduced energy consumption, the alkaline-treatment process is the preferred method for trans-esterification. For the alkaline-treatment process, crude oil should contain a very low levels of free fatty acids (FFAs) and moisture because a high level of FFA and water can transform trans-esterification into saponification leading to easy depletion of catalysts (34, 35). For crude oil containing a high amount of FFAs and water acid treatment is more suitable; however, the reaction time of acid treatment is very long and there is a greater requirement of alcohol. This dilemma has prompted studies to improve trans-esterification processes by chemical methods (36, 37). The deficiency of seed-specific SDP1 in Jatropha plant provides a solution to the dilemma. FFA levels of mature seeds are mainly determined by SDP1 during the desiccation step at the late-maturation stage (15, 28). Here, we showed that in mature dry seeds JcSDP1-RNAi transgenic lines contain higher TAG levels and lower FFA levels compared to control Jatropha plants (FIG. 1C-1D and FIG. 5C). Therefore, crude oils derived from SDP1-deficient transgenic Jatropha plants are a better substrate compared to WT crude oil for alkaline trans-esterification in biodiesel production.
[0100] The use of the terms "a" and "an" and "the" and similar referents in the context of describing the invention (especially in the context of the following claims) are to be construed to cover both the singular and the plural, unless otherwise indicated herein or clearly contradicted by context. The terms "comprising," "having," "including," and "containing" are to be construed as open-ended terms (i.e., meaning "including, but not limited to,") unless otherwise noted. Recitation of ranges of values herein are merely intended to serve as a shorthand method of referring individually to each separate value falling within the range, unless otherwise indicated herein, and each separate value is incorporated into the specification as if it were individually recited herein. All methods described herein can be performed in any suitable order unless otherwise indicated herein or otherwise clearly contradicted by context. The use of any and all examples, or exemplary language (e.g., "such as") provided herein, is intended merely to better illuminate the invention and does not pose a limitation on the scope of the invention unless otherwise claimed. No language in the specification should be construed as indicating any non-claimed element as essential to the practice of the invention.
[0101] Embodiments of this invention are described herein, including the best mode known to the inventors for carrying out the invention. Variations of those embodiments may become apparent to those of ordinary skill in the art upon reading the foregoing description. The inventors expect skilled artisans to employ such variations as appropriate, and the inventors intend for the invention to be practiced otherwise than as specifically described herein. Accordingly, this invention includes all modifications and equivalents of the subject matter recited in the claims appended hereto as permitted by applicable law. Moreover, any combination of the above-described elements in all possible variations thereof is encompassed by the invention unless otherwise indicated herein or otherwise clearly contradicted by context.
BIBLIOGRAPHY
[0102] 1. IEA says "Biofuels can displace 27% of transportation fuels by 2050 Washington". Platts. 20 Apr. 2011.
[0103] 2. Gubitz G M, Mittelbach M, Trabi M: Exploitation of the tropical oil seed plant Jatropha curcas L. Bioresource Technol 1999, 67:73-82.
[0104] 3. Carels N: Jatropha curcas: A review. Advances in Botanical Research 2009, 50:39-86.
[0105] 4. Adebowale K O, Adedire C O: Chemical composition and insecticidal properties of the underutilized Jatropha curcas seed oil. African Biotechnol 2006, 5:901-906.
[0106] 5. Openshaw K: A review of Jatropha curcas: an oil plant of unfulfilled promise. Biomass Bioenergy 2000, 19:1-15.
[0107] 6. Kumar S, Sharma S: An evaluation of multipurpose oil seed crop for industrial uses (Jatropha curcas L.). A Review Ind Crops Prod 2008, 28:1-10.
[0108] 7. Qu J, Mao H-Z, Chen W, Gao S-Q Bai Y-N, Sun Y-W, Geng Y-F, Ye J: Development of marker-free transgenic Jatropha plants with increased levels of seed oleic acid. Biotechnol Biofuels 2012, 5:10-20.
[0109] 8. Stymne S, Stobart A K: Triacylglycerol biosynthesis. In The Biochemistry of Plants, Lipids: Structure and Function. Volume 9, Stumpf P K editor (New York: Academic Press) 1987, 175-214.
[0110] 9. Murphy DJ: Plant Lipids: Biology, Utilization and Manipulation. Oxford, UK: Blackwell Publishing 2005.
[0111] 10. Dutta S, Ray L: Production and characterization of an alkaline thermostable crude lipase from an isolated strain of Bacillus cereus C7. Appl Biochem Biotechnol 2009, 159:142-154.
[0112] 11. Athenstaedt K, Daum G: YMR313c/TGL3 encodes a novel triacylglycerol lipase located in lipid particles of Saccharomyces cerevisiae. J Biol Chem 2003, 278:23317-23323.
[0113] 12. Gronke S, Mildner A, Fellert S, Tennagels N, Petry S, Muller G, Jackie H, Kuhnlein R P: Brummer lipase is an evolutionary conserved fat storage regulator in Drosophila. Cell Metab 2005, 1:323-330.
[0114] 13. Eastmond P J, Rawsthorne S: Coordinate changes in carbon partitioning and plastidial metabolism during the development of oilseed rape embryos. Plant Physiol 2000, 122:767-774.
[0115] 14. Chia T Y P, Pike M J, Rawsthorne S: Storage oil breakdown during embryo development of Brassica napus (L.). J Exp Botany 2005, 56:1285-1296.
[0116] 15. Eastmond P J: SUGAR-DEPENDENT1 encodes a patatin domain triacylglycerol lipase that initiates storage oil breakdown in germinating Arabidopsis seeds. Plant Cell 2006, 18:665-675.
[0117] 16. Eastmond P J: Monodehydroascorbate reductase4 is required for seed storage oil hydrolysis and postgerminative growth in Arabidopsis. Plant Cell 2007, 19:1376-1387.
[0118] 17. Puttick D, Dauk M, Lozinsky S, Smith M A: Overexpression of a FAD3 desaturase increases synthesis of a polymethylene-interrupted dienoic fatty acid in seeds of Arabidopsis thaliana L. Lipids 2009, 44:753-757.
[0119] 18. Lemieux B, Miguel M, Somerville C, Browse J: Mutants of Arabidopsis with alterations in seed lipid fatty acid composition. Threoretical and Applied Genetics 1990, 80:234-240.
[0120] 19. Huang A H C: Oleosins and oil bodies in seeds and other organs. Plant Physiol 1996, 110:1055-1061.
[0121] 20. Lu C-A, Lim E-K, Yu S-M: Sugar response sequence in the promoter of a rice alpha-amylase gene serves as a transcriptional enhancer. J Biol Chem 1998, 273:10120-10131.
[0122] 21. Lu C-A, Ho T D, Ho S-L, Yu S-M: Three novel MYB proteins with one DNA binding repeat mediate sugar and hormone regulation of alpha-amylase gene expression. Plant Cell 2002, 14:1963-1980.
[0123] 22. Stalberg K, Ellerstom M, Ezcurra I, Ablov S, Rask L: Disruption of an overlapping E-box/ABRE motif abolished high transcription of the napA storage-protein promoter in transgenic Brassica napus seeds. Planta 1996, 199:515-519.
[0124] 23. Hartmann U, Sagasser M, Mehrtens F, Stracke R, Weisshaar B: Differential combinatorial interactions of cis-acting elements recognized by R2R3-MYB, BZIP, and BHLH factors control light-responsive and tissue-specific activation of phenylpropanoid biosynthesis genes. Plant Mol Biol 2005, 57:155-171.
[0125] 24. Le B H, Cheng C, Bui A Q, Wagmaister J A, Henry K F, Pelletier J., Kwong L, Belmonte M, Kirkbride R, Horvath S, Drews G N, Fisher R L., Okamuro J K., Harada J J, Goldberg R B: Global analysis of gene activity during Arabidopsis seed development and identification of seed-specific transcription factors. Proc Natl Acad Sci USA 2010, 107:8063-8070.
[0126] 25. Kurat C F, Natter K, Petschnigg J, Wolinski H, Scheuringer K, Scholz H, Zimmermann R, Leber R, Zechner R, Kohlwein S D: Obese yeast: triglyceride lipolysis is functionally conserved from mammals to yeast. J Biol Chem 2006, 281:491-500.
[0127] 26. Eastmond P J: Cloning and characterization of the acid lipase from castor beans. J Biol Chem 2004, 279:45540-45545.
[0128] 27. van der Schoot C, Paul L K, Paul S B, Rinne P L H: Plant lipid bodies and cell-cell signaling: A new role for an old organelle? Plant signaling and Behavior 2011, 6:1732-1738.
[0129] 28. Kelly A L, Quettier A-L, Shaw E, Eastmond P J: Seed storage oil mobilization is important but not essential for germination or seedling establishment in Arabidopsis. Plant Physiol 2011, 157:866-875.
[0130] 29. Baud S, Boutin J-P, Miguel M, Lepiniec L, Rochat C: An integrated overview of seed development in Arabidopsis thaliana ecotype WS. Plant Physiol and Biochem 2002, 40:151-160.
[0131] 30. Baud S, Dubreucq B, Miguel M, Rochat C, Lepiniec L: Storage reserve accumulation in Arabidopsis: Metabolic and developmental control of seed filling. The Arabidopsis Book 2008, Vol. 6
[0132] 31. Eastmond P J, Germain V, Lange P R, Bryce J H, Smith S M, Graham I A: Postgerminative growth and lipid catabolism in oilseeds lacking the glyoxylate cycle. Proc Natl Acad Sci USA 2000, 97:5669-5674.
[0133] 32. Devanesan M G, Viruthagiri T, Sugumar N: Transesterification of Jatropha oil using immobilized Pseudomonas fluorescens. African J of Biotechnology 2007, 6:2497-2501.
[0134] 33. Thaiyasuit P, Pianthong K, Worapun I: Acid esterification-alkaline transesterification process for methyl ester production from crude rubber seed oil. J Oleo Sci 2012, 61:81-88.
[0135] 34. Freedman B, Pryde E H, Mounts T L: Variables affecting the yields of fatty esters from transesterified vegetable oils. J Am Oil Chem Soc 1984, 61:1638-1643.
[0136] 35. Canakci M, van Gerpen J: Biodiesel production via acid catalysis. Trans ASAE 1999, 42:1203-1210.
[0137] 36. Lopez D E, Goodwin J G, Bruce D A, Lotero E: Transesterification of triacetin with methanol on solid acid and base catalysts. Appl Catalysis 2005, 295:97-105.
[0138] 37. Lopez D E, Goodwin J G, Bruce D A, Furuta S: Esterification and transesterification using modified zirconia catalysts. Appl Catalysis 2008, 339:76-83.
[0139] 38. Clough S J, Bent A F: Floral dip: a simplified method for Agrobacterium-mediated transformation of Arabidopsis thaliana. Plant J 1995, 16:735-743.
[0140] 39. Murashige T, Skoog F: A revised medium for rapid growth and bioassays with tobacco culture. Physiol Plant 1962, 15:473-497.
[0141] 40. Jefferson R A, Kavanagh T A, Bevan M W: GUS fusion: β-glucuronidase as a sensitive and versatile gene fusion marker in higher plants. EMBO J 1987, 6:3901-3907.
[0142] 41. Qiu C, Sangha J S, Song F, Zhou Z, Yin A, Gu K, Tian D, Yang J, Yin Z: Production of marker-free transgenic rice expressing tissue-specific Bt gene. Plant Cell Reports 2010, 29:1097-1107.
[0143] 42. Guo H S, Fei J F, Xie Q, Chua N H: A chemical-regulated inducible RNAi system in plants. Plant J 2003, 34:383-392.
[0144] 43. Mao H Z, Ye J, Chua N H: Genetic transformation of Jatropha curcas. 2009, International Application No.:PCT/SG2009/000479 (WO 2010/071608).
[0145] 44. Li Y, Beisson F, Pollard M, Ohlrogge J: Oil content of Arabidopsis seeds: The influence of seed anatomy, light and plant-to-plant variation. Pytochemistry 2006, 67:904-915.
[0146] 45. Focks N, Benning C: wrinkled1: A novel, low-seed-oil mutant of Arabidopsis with a deficiency in the seed-specific regulation of carbohydrate metabolism. Plant Physiol 1988, 118:91-101.
[0147] 46. Allen G C, Flores-Vergara M A, Krasynanski S, Kumar S, Thompson W F: A modified protocol for rapid DNA isolation from plant tissues using cetyltrimethylammonium bromide. Nat Protoc 2006, 1:2320-2325.
[0148] 47. Sambrook J, Russell D W: Molecular cloning: a laboratory manual. 3rd edition. Cold Spring Harbor Laboratory Press, Cold Spring Harbor, N.Y.; 2001.
[0149] Abdullah, R. et al. (2005). Immature embryo: A useful tool for oil palm (Elaeis guineensis Jacq.) genetic transformation studies. Electronic Journal of Biotechnology [online] vol. 8, no. 1 [Apr. 15, 2005). Available from: http colon//www dot ejbiotechnology dot info/content/vol8/issue1/full/1/index.html.
[0150] Chiu, W. et al. (1996). Engineered GFP as a vital reporter in plants. Current Biology 6:325-330.
[0151] Chowdhury, M. K. U. et al. (1997). Evaluation of five promoters for use in transformation of oil palm (Elaeis guineensis Jacq.) Plant Cell Reports 16:277-281.
[0152] Christensen, A. H. and Quail, P. H. (1989). Sequence analysis and transcriptional regulation by heat shock of polyubiquitin transcripts from maize. Plant Mol Biol 12:619-632.
[0153] Christensen, A. H. et al. (1992). Maize polyubiquitin genes: structure, thermal perturbation of expression and transcript splicing, and promoter activity following transfer to protoplasts by electroporation. Plant Mol Biol 18:675-689.
[0154] De Wet, J. R. et al. (1987). Firefly luciferase gene: structure and expression in mammalian cells. Mol Cell Biol 7:725-737.
[0155] Goff, S. A. et al. (1990). Transactivation of anthocyanin biosynthetic genes following transfer of B regulatory genes into maize tissues. EMBO J 9:2517-2522.
[0156] Jefferson, R. A. et al. (1991). Plant Molecular Biology Manual, ed. Gelvin et al., Kluwer Academic Publishers, pp. 1-33.
[0157] Kain, S. R. et al. (1995). Green fluorescent protein as a reporter of gene expression and protein localization. Biotechniques 19:650-655.
[0158] Last, D. I. et al. (1991). pEmu: an improved promoter for gene expression in cereal cells. Theor Appl Genet 81:581-588.
[0159] Mash, D. I. A. et al. (2009). Transformation of oil palm using Agrobacterium tumefaciens. J Oil Palm Res 21:643-652.
[0160] McElroy, D. et al. (1990). Isolation of an efficient actin promoter for use in rice transformation. Plant Cell 2:163-171.
[0161] Odell, J. T. et al. (1985). Identification of DNA sequences required for activity of the cauliflower mosaic virus 35S promoter. Nature 313:810-812.
[0162] Omidvar, V. et al. (2008). A transient assay to evaluate the expression of polyhydroxybutyrate genes regulated by oil palm mesocarp-specific promoter. Plant Cell Rep 27:1451-1459.
[0163] Parveez, G. K. A. et al. (2000). Transgenic oil palm: production and projection. Biochemical Society Transactions 28:969-972.
[0164] Parveez, G. K. A. (2008). Biolistic mediated production of transgenic oil palm. Methods Mol Biol 477:301-320.
[0165] Popluechai, S., et al. (2011). Jatropha curcas oil body proteasome and oleosins: L-form JcOle3 as a potential phylogenetic marker. Plant Physiol Biochem 49: 352-356.
[0166] Velten, J. et al. (1984). Isolation of a dual plant promoter fragment from the Ti plasmid of Agrobacterium tumefaciens. EMBO J 3:2723-2730.
[0167] Zuo, J. et al. (2001). Chemical-regulated, site-specific DNA excision in transgenic plants. Nat Biotechnol 19:157-161.
Sequence CWU
1
1
5813835DNAJatropha
curcaspromoter(1)..(722)5'UTR(723)..(873)CDS(874)..(3450)3'UTR(3451)..(38-
35) 1tacatccagt agattgcacg tcacacacta atctacccgc cacgtatgtg cccattcaaa
60ctttgcgtca ctgtatcgct atctttatct ctttcacgtt tcccaatatt ttccctctct
120tctctcctta tagtaaagcc tatcaaccag tccccatctg tctctctcac tctataaagc
180cacgtttttt cttcttttcc cgggattatc ctatctgggt ttctcgggtt tctttttgaa
240ttccaattcc tgagttcgtt gccctttttc tgtttttctg tttgtttacg ttatgaatac
300ccatttggta tttgtattaa ttaaatagct ggtatatgtt atttgtgctt tttaggtttg
360agggttttgg ggtttggtta atcagcaaga tctgttaaga aagaaaggaa ggtgagagtt
420gagaggacag acccatttgg ctgcttttct tggaatttca atatttaagg ttggctttct
480gctgaattcg gttaagaggc tggattttgt gacttctata catccctttg gtgtataaat
540taagttttct tataggattg ctatctatcc acttttatct aacaaaagcc attatctttc
600ttttgcatag agagtctggt ggtgtaatat ttcaatcatt ttgtgattgg agaaacaatt
660tcagtcattt tattgtcttt atgcaattgt tagagctgtg cagaacatat aggtgtatat
720agtcaaaaca ttgccttgta attccttcaa ttgctgtctc ctagcaattg ttattgcttc
780ttaagtttta tattagcaaa tattcttgtt gagttacagg aattcccaat agcttcgatt
840tagcaaatca tccaaaacac gaaactttta gag atg gat ata agt aat gag gcc
894 Met Asp Ile Ser Asn Glu Ala
1 5
aat gtt gat ccc ttt cca att gga cct tca tca att att ggt aga act
942Asn Val Asp Pro Phe Pro Ile Gly Pro Ser Ser Ile Ile Gly Arg Thr
10 15 20
att gct ttt aga gtg tta ttc tgc aag tca atg gga caa ttg agg cgt
990Ile Ala Phe Arg Val Leu Phe Cys Lys Ser Met Gly Gln Leu Arg Arg
25 30 35
aga atc tat cat ttt tta ttg aat tac att tgt aga ctt agg gat ttt
1038Arg Ile Tyr His Phe Leu Leu Asn Tyr Ile Cys Arg Leu Arg Asp Phe
40 45 50 55
tgt gct tca atg gta tca tgg ttg cat ccg cgg aat cca caa gga ata
1086Cys Ala Ser Met Val Ser Trp Leu His Pro Arg Asn Pro Gln Gly Ile
60 65 70
ctg gca atg gtg aca ata att gct ttt gta ttg aaa agg tac aca aac
1134Leu Ala Met Val Thr Ile Ile Ala Phe Val Leu Lys Arg Tyr Thr Asn
75 80 85
gtg aaa tcg agg gct gag atg gca tat cgg agg aaa ttt tgg agg aat
1182Val Lys Ser Arg Ala Glu Met Ala Tyr Arg Arg Lys Phe Trp Arg Asn
90 95 100
atg atg aga act gca ttg aca tat gaa gag tgg gca cat gct gct aag
1230Met Met Arg Thr Ala Leu Thr Tyr Glu Glu Trp Ala His Ala Ala Lys
105 110 115
atg ctt gat aaa gag aca cca aag atg aac gaa tcg gac ctt tat gat
1278Met Leu Asp Lys Glu Thr Pro Lys Met Asn Glu Ser Asp Leu Tyr Asp
120 125 130 135
gaa gaa tta gtg aga aat aag ctt cag gag ctt cga cac cgt aga caa
1326Glu Glu Leu Val Arg Asn Lys Leu Gln Glu Leu Arg His Arg Arg Gln
140 145 150
gag gga tct ctt aga gat att atc ttt tgt atg aga gct gat ctt ata
1374Glu Gly Ser Leu Arg Asp Ile Ile Phe Cys Met Arg Ala Asp Leu Ile
155 160 165
aga aat ctt ggt aat atg tgc aac cca gag ctt cac aag ggc agg ctt
1422Arg Asn Leu Gly Asn Met Cys Asn Pro Glu Leu His Lys Gly Arg Leu
170 175 180
caa gtg cct aag ctt att aag gaa tac att gat gag gtc tca acc cag
1470Gln Val Pro Lys Leu Ile Lys Glu Tyr Ile Asp Glu Val Ser Thr Gln
185 190 195
ttg aga atg gtt tgt gac tca gat aca gag gag ctt tca ttg gag gaa
1518Leu Arg Met Val Cys Asp Ser Asp Thr Glu Glu Leu Ser Leu Glu Glu
200 205 210 215
aag ctt tct ttc atg cat gaa acg aga cat gca ttt ggg aga aca gct
1566Lys Leu Ser Phe Met His Glu Thr Arg His Ala Phe Gly Arg Thr Ala
220 225 230
ttg ctt ttg agt gga ggt gct tcc ctc gga gcc ttt cat gta ggt gtg
1614Leu Leu Leu Ser Gly Gly Ala Ser Leu Gly Ala Phe His Val Gly Val
235 240 245
gtt aag acg ctg gtg cag cat aag ctt ttg ccc aga ata att gct ggt
1662Val Lys Thr Leu Val Gln His Lys Leu Leu Pro Arg Ile Ile Ala Gly
250 255 260
tct agt gta gga tcc att atg tgt tct att gtc gcc acg aga gca tgg
1710Ser Ser Val Gly Ser Ile Met Cys Ser Ile Val Ala Thr Arg Ala Trp
265 270 275
cca gag ttg caa agt ttt ttt gaa gat tct ttg cat tca ttg cag ttt
1758Pro Glu Leu Gln Ser Phe Phe Glu Asp Ser Leu His Ser Leu Gln Phe
280 285 290 295
ttt gat caa atg ggc ggg ctt ttt act gta gtc aag agg gtc acg aca
1806Phe Asp Gln Met Gly Gly Leu Phe Thr Val Val Lys Arg Val Thr Thr
300 305 310
caa ggt gct gtc cat gaa ata cgg cag ttg caa tgg atg tta agg cat
1854Gln Gly Ala Val His Glu Ile Arg Gln Leu Gln Trp Met Leu Arg His
315 320 325
ctc aca agc aat ctt aca ttt cag gaa gct tat gat atg aca ggt cga
1902Leu Thr Ser Asn Leu Thr Phe Gln Glu Ala Tyr Asp Met Thr Gly Arg
330 335 340
att ctt gca atc aca gtt tgc tct cca agg aag cat gag cct cct aga
1950Ile Leu Ala Ile Thr Val Cys Ser Pro Arg Lys His Glu Pro Pro Arg
345 350 355
tgc ctt aac tac ctg act tca cct cat gtt gtc ata tgg agt gca gtc
1998Cys Leu Asn Tyr Leu Thr Ser Pro His Val Val Ile Trp Ser Ala Val
360 365 370 375
act gct tct tgt gcc ttt cct ggt ctt ttt gaa gcc cag gaa ctt atg
2046Thr Ala Ser Cys Ala Phe Pro Gly Leu Phe Glu Ala Gln Glu Leu Met
380 385 390
gct aag gat aga agt gga gaa att gtt cca tat cat cca ccc ttc aag
2094Ala Lys Asp Arg Ser Gly Glu Ile Val Pro Tyr His Pro Pro Phe Lys
395 400 405
ctg gaa cca gag gaa ggg tca agc acg tct gct cgt cgg tgg agg gat
2142Leu Glu Pro Glu Glu Gly Ser Ser Thr Ser Ala Arg Arg Trp Arg Asp
410 415 420
gga agc ttg gag att gat tta cct atg atg caa ttg aag gag cta ttc
2190Gly Ser Leu Glu Ile Asp Leu Pro Met Met Gln Leu Lys Glu Leu Phe
425 430 435
aat gtt aat cat ttt att gtg agt caa gca aat cct cac att gct cca
2238Asn Val Asn His Phe Ile Val Ser Gln Ala Asn Pro His Ile Ala Pro
440 445 450 455
tta ttg aga atg aag gag ttt ata aga gct tat ggc ggt aac ttt gct
2286Leu Leu Arg Met Lys Glu Phe Ile Arg Ala Tyr Gly Gly Asn Phe Ala
460 465 470
gca aag ctt gct cat ctc acc gag atg gaa gta aaa cat aga tgc agt
2334Ala Lys Leu Ala His Leu Thr Glu Met Glu Val Lys His Arg Cys Ser
475 480 485
cag gtg ttg gaa ctt ggt ttt cca tta ggt gga gtt gcc aag ctt ttt
2382Gln Val Leu Glu Leu Gly Phe Pro Leu Gly Gly Val Ala Lys Leu Phe
490 495 500
gct caa gat tgg gag gga gac gtc act gtt gtt atg cct gct aca ctc
2430Ala Gln Asp Trp Glu Gly Asp Val Thr Val Val Met Pro Ala Thr Leu
505 510 515
gct cag tac tca aaa att ata caa aat ccg act ctt gtg gag ctg caa
2478Ala Gln Tyr Ser Lys Ile Ile Gln Asn Pro Thr Leu Val Glu Leu Gln
520 525 530 535
aag gcg gcc aac caa ggg aga agg tgt aca tgg gag aaa ctt tca gcc
2526Lys Ala Ala Asn Gln Gly Arg Arg Cys Thr Trp Glu Lys Leu Ser Ala
540 545 550
ata aaa gcc aac tgc ggg att gag ctt tgt ctt gat gaa tgt gtc gca
2574Ile Lys Ala Asn Cys Gly Ile Glu Leu Cys Leu Asp Glu Cys Val Ala
555 560 565
att ctc aac cac atg cgt aga ctc aaa agg agt gct gag aga gca gct
2622Ile Leu Asn His Met Arg Arg Leu Lys Arg Ser Ala Glu Arg Ala Ala
570 575 580
gct gct tcg cat ggc ata cct aac cca agt act agt aat gtc aaa ttc
2670Ala Ala Ser His Gly Ile Pro Asn Pro Ser Thr Ser Asn Val Lys Phe
585 590 595
agt gct tct aga aga atc cct tct tgg aac tgc att gct aga gag aac
2718Ser Ala Ser Arg Arg Ile Pro Ser Trp Asn Cys Ile Ala Arg Glu Asn
600 605 610 615
tca aca ggg tca att gat gag ctt ctg act gat gtt gct tcc aca ttt
2766Ser Thr Gly Ser Ile Asp Glu Leu Leu Thr Asp Val Ala Ser Thr Phe
620 625 630
cat caa ggt gtt ggt gga tct gga gca act aca ggt aga aat ttg cgg
2814His Gln Gly Val Gly Gly Ser Gly Ala Thr Thr Gly Arg Asn Leu Arg
635 640 645
act cac cgc aac ata cat gat gga agc gac agc gaa tct gaa aat gta
2862Thr His Arg Asn Ile His Asp Gly Ser Asp Ser Glu Ser Glu Asn Val
650 655 660
gat ata act tct tgg aca agg tct ggt ggg cca ttg atg agg aca act
2910Asp Ile Thr Ser Trp Thr Arg Ser Gly Gly Pro Leu Met Arg Thr Thr
665 670 675
tca gca aat aaa ttt att gac ttc gtt caa aat ctt gat att gat gcc
2958Ser Ala Asn Lys Phe Ile Asp Phe Val Gln Asn Leu Asp Ile Asp Ala
680 685 690 695
gaa ttg acc aaa ggc ttg ttg act cat cct aac tct ccg ggg gct cct
3006Glu Leu Thr Lys Gly Leu Leu Thr His Pro Asn Ser Pro Gly Ala Pro
700 705 710
atg gga att agg gat cca ttt aat aca agt tct cgg gtg aca aca cct
3054Met Gly Ile Arg Asp Pro Phe Asn Thr Ser Ser Arg Val Thr Thr Pro
715 720 725
gag aga att tcg gaa agt gat ttt gaa ctg agg gat ttt agc aga tca
3102Glu Arg Ile Ser Glu Ser Asp Phe Glu Leu Arg Asp Phe Ser Arg Ser
730 735 740
tct cag act ggt tct agt att atg gtt act gaa ggc gat ctt ttg cag
3150Ser Gln Thr Gly Ser Ser Ile Met Val Thr Glu Gly Asp Leu Leu Gln
745 750 755
cca gaa aga atc cat aat ggg att gtg ttg aac gtc gta aag aag gaa
3198Pro Glu Arg Ile His Asn Gly Ile Val Leu Asn Val Val Lys Lys Glu
760 765 770 775
aac ttg gga ctc tca aat aga agc cag gat tca gaa aat tac aat gaa
3246Asn Leu Gly Leu Ser Asn Arg Ser Gln Asp Ser Glu Asn Tyr Asn Glu
780 785 790
att cct gaa tgt gtt cag ctt gat agg gat atg gat ggt agc tca gca
3294Ile Pro Glu Cys Val Gln Leu Asp Arg Asp Met Asp Gly Ser Ser Ala
795 800 805
tct gaa tat gct ggg gat gac gat gat gac gac aat gac aat gac aat
3342Ser Glu Tyr Ala Gly Asp Asp Asp Asp Asp Asp Asn Asp Asn Asp Asn
810 815 820
gac atc att act gta aca aac ttc tca aat gtg gta tct ccc att cct
3390Asp Ile Ile Thr Val Thr Asn Phe Ser Asn Val Val Ser Pro Ile Pro
825 830 835
gtt ccc aag gat gat tct ggg gta cat gag gga cag gat caa agt tct
3438Val Pro Lys Asp Asp Ser Gly Val His Glu Gly Gln Asp Gln Ser Ser
840 845 850 855
gtg gat ggt tag ttttgaggaa tatttctgtt gtgtgaacaa tttggtctat
3490Val Asp Gly
ctgattcccg gatcacctta atatcaagtg ccactagctg cagaaagagc agagaaaagc
3550aattattgct tgaacatccc catttcacgt cgttttcagg agataatggt cagggacagc
3610agagaataat caatcatttg ttagatagaa gatttgtata tgaaaccgga gagaaaagct
3670caacattttt agaagaatga tcgcatttga gatcaaattg tcttgaatat gccaaattat
3730ttattgtgta aaatgtattg tacataccac gcaatgtgca atgtgatttt catacgtaca
3790gttcaatgaa agaaacagat tttacatcaa aaaaaaaaaa aaaaa
38352858PRTJatropha curcas 2Met Asp Ile Ser Asn Glu Ala Asn Val Asp Pro
Phe Pro Ile Gly Pro 1 5 10
15 Ser Ser Ile Ile Gly Arg Thr Ile Ala Phe Arg Val Leu Phe Cys Lys
20 25 30 Ser Met
Gly Gln Leu Arg Arg Arg Ile Tyr His Phe Leu Leu Asn Tyr 35
40 45 Ile Cys Arg Leu Arg Asp Phe
Cys Ala Ser Met Val Ser Trp Leu His 50 55
60 Pro Arg Asn Pro Gln Gly Ile Leu Ala Met Val Thr
Ile Ile Ala Phe 65 70 75
80 Val Leu Lys Arg Tyr Thr Asn Val Lys Ser Arg Ala Glu Met Ala Tyr
85 90 95 Arg Arg Lys
Phe Trp Arg Asn Met Met Arg Thr Ala Leu Thr Tyr Glu 100
105 110 Glu Trp Ala His Ala Ala Lys Met
Leu Asp Lys Glu Thr Pro Lys Met 115 120
125 Asn Glu Ser Asp Leu Tyr Asp Glu Glu Leu Val Arg Asn
Lys Leu Gln 130 135 140
Glu Leu Arg His Arg Arg Gln Glu Gly Ser Leu Arg Asp Ile Ile Phe 145
150 155 160 Cys Met Arg Ala
Asp Leu Ile Arg Asn Leu Gly Asn Met Cys Asn Pro 165
170 175 Glu Leu His Lys Gly Arg Leu Gln Val
Pro Lys Leu Ile Lys Glu Tyr 180 185
190 Ile Asp Glu Val Ser Thr Gln Leu Arg Met Val Cys Asp Ser
Asp Thr 195 200 205
Glu Glu Leu Ser Leu Glu Glu Lys Leu Ser Phe Met His Glu Thr Arg 210
215 220 His Ala Phe Gly Arg
Thr Ala Leu Leu Leu Ser Gly Gly Ala Ser Leu 225 230
235 240 Gly Ala Phe His Val Gly Val Val Lys Thr
Leu Val Gln His Lys Leu 245 250
255 Leu Pro Arg Ile Ile Ala Gly Ser Ser Val Gly Ser Ile Met Cys
Ser 260 265 270 Ile
Val Ala Thr Arg Ala Trp Pro Glu Leu Gln Ser Phe Phe Glu Asp 275
280 285 Ser Leu His Ser Leu Gln
Phe Phe Asp Gln Met Gly Gly Leu Phe Thr 290 295
300 Val Val Lys Arg Val Thr Thr Gln Gly Ala Val
His Glu Ile Arg Gln 305 310 315
320 Leu Gln Trp Met Leu Arg His Leu Thr Ser Asn Leu Thr Phe Gln Glu
325 330 335 Ala Tyr
Asp Met Thr Gly Arg Ile Leu Ala Ile Thr Val Cys Ser Pro 340
345 350 Arg Lys His Glu Pro Pro Arg
Cys Leu Asn Tyr Leu Thr Ser Pro His 355 360
365 Val Val Ile Trp Ser Ala Val Thr Ala Ser Cys Ala
Phe Pro Gly Leu 370 375 380
Phe Glu Ala Gln Glu Leu Met Ala Lys Asp Arg Ser Gly Glu Ile Val 385
390 395 400 Pro Tyr His
Pro Pro Phe Lys Leu Glu Pro Glu Glu Gly Ser Ser Thr 405
410 415 Ser Ala Arg Arg Trp Arg Asp Gly
Ser Leu Glu Ile Asp Leu Pro Met 420 425
430 Met Gln Leu Lys Glu Leu Phe Asn Val Asn His Phe Ile
Val Ser Gln 435 440 445
Ala Asn Pro His Ile Ala Pro Leu Leu Arg Met Lys Glu Phe Ile Arg 450
455 460 Ala Tyr Gly Gly
Asn Phe Ala Ala Lys Leu Ala His Leu Thr Glu Met 465 470
475 480 Glu Val Lys His Arg Cys Ser Gln Val
Leu Glu Leu Gly Phe Pro Leu 485 490
495 Gly Gly Val Ala Lys Leu Phe Ala Gln Asp Trp Glu Gly Asp
Val Thr 500 505 510
Val Val Met Pro Ala Thr Leu Ala Gln Tyr Ser Lys Ile Ile Gln Asn
515 520 525 Pro Thr Leu Val
Glu Leu Gln Lys Ala Ala Asn Gln Gly Arg Arg Cys 530
535 540 Thr Trp Glu Lys Leu Ser Ala Ile
Lys Ala Asn Cys Gly Ile Glu Leu 545 550
555 560 Cys Leu Asp Glu Cys Val Ala Ile Leu Asn His Met
Arg Arg Leu Lys 565 570
575 Arg Ser Ala Glu Arg Ala Ala Ala Ala Ser His Gly Ile Pro Asn Pro
580 585 590 Ser Thr Ser
Asn Val Lys Phe Ser Ala Ser Arg Arg Ile Pro Ser Trp 595
600 605 Asn Cys Ile Ala Arg Glu Asn Ser
Thr Gly Ser Ile Asp Glu Leu Leu 610 615
620 Thr Asp Val Ala Ser Thr Phe His Gln Gly Val Gly Gly
Ser Gly Ala 625 630 635
640 Thr Thr Gly Arg Asn Leu Arg Thr His Arg Asn Ile His Asp Gly Ser
645 650 655 Asp Ser Glu Ser
Glu Asn Val Asp Ile Thr Ser Trp Thr Arg Ser Gly 660
665 670 Gly Pro Leu Met Arg Thr Thr Ser Ala
Asn Lys Phe Ile Asp Phe Val 675 680
685 Gln Asn Leu Asp Ile Asp Ala Glu Leu Thr Lys Gly Leu Leu
Thr His 690 695 700
Pro Asn Ser Pro Gly Ala Pro Met Gly Ile Arg Asp Pro Phe Asn Thr 705
710 715 720 Ser Ser Arg Val Thr
Thr Pro Glu Arg Ile Ser Glu Ser Asp Phe Glu 725
730 735 Leu Arg Asp Phe Ser Arg Ser Ser Gln Thr
Gly Ser Ser Ile Met Val 740 745
750 Thr Glu Gly Asp Leu Leu Gln Pro Glu Arg Ile His Asn Gly Ile
Val 755 760 765 Leu
Asn Val Val Lys Lys Glu Asn Leu Gly Leu Ser Asn Arg Ser Gln 770
775 780 Asp Ser Glu Asn Tyr Asn
Glu Ile Pro Glu Cys Val Gln Leu Asp Arg 785 790
795 800 Asp Met Asp Gly Ser Ser Ala Ser Glu Tyr Ala
Gly Asp Asp Asp Asp 805 810
815 Asp Asp Asn Asp Asn Asp Asn Asp Ile Ile Thr Val Thr Asn Phe Ser
820 825 830 Asn Val
Val Ser Pro Ile Pro Val Pro Lys Asp Asp Ser Gly Val His 835
840 845 Glu Gly Gln Asp Gln Ser Ser
Val Asp Gly 850 855 32053DNAArtificial
SequenceRNAi sequence 3gggccctaca tccagtagat tgcacgtcac acactaatct
acccgccacg tatgtgccca 60ttcaaacttt gcgtcactgt atcgctatct ttatctcttt
cacgtttccc aatattttcc 120ctctcttctc tccttatagt aaagcctatc aaccagtccc
catctgtctc tctcactcta 180taaagccacg ttttttcttc ttttcccggg attatcctat
ctgggtttct cgggtttctt 240tttgaattcc aattcctgag ttcgttgccc tttttctgtt
tttctgtttg tttacgttat 300gaatacccat ttggtatttg tattaattaa atagctggta
tatgttattt gtgcttttta 360ggtttgaggg ttttggggtt tggttaatca gcaagatctg
ttaagaaaga aaggaaggtg 420agagttgaga ggacagaccc atttggctgc ttttcttgga
atttcaatat ttaaggttgg 480ctttctgctg aattcggtta agaggctgga ttttgtgact
tctatacatc cctttggtgt 540ataaattaag ttttcttata ggattgctat ctatccactt
ttatctaaca aaagccatta 600tctttctttt gcatagagag tctggtggtg taatatttca
atcattttgt gattggagaa 660acaatttcag tcattttatt gtctttatgc aattgttaga
gctgtgcaga acatataggt 720gtatatagtc aaaacattgc cttgtaattc cttcaattgc
tgtctcctag caattgttat 780tgcttcttaa gttttatatt agcaaatatt cttgttgagt
tacaggaatt cccaatagct 840tcgatttagc aaatcatcca aaacacgaaa cttttagagc
tcgagtccat aatgggattg 900tgttgaacgt cgtaaagaag gaaaacttgg gactctcaaa
tagaagccag gattcagaaa 960attacaatga aattcctgaa tgtgttcagc ttgataggga
tatggatggt agctcagcat 1020ctgaatatgc tggggatgac gatgatgacg acaatgacaa
tgacaatgac atcattactg 1080taacaaactt ctcaaatgtg gtatctccca ttcctgttcc
caaggatgat tctggggtac 1140atgagggaca ggatcaaagt tctgtggatg gttagttttg
aggaatattt ctgttgtgtg 1200aacaatttgg tctatctgat tcccggatca ccttaatatc
aagtgccact agcaagcttg 1260attacgtaag tagatcttaa acacctacac cattttttta
atcactacta cccattgcat 1320tgaacaaact tcaagttctt cttagcttca gattaagaaa
gtacccttcc ttggctttgt 1380tgatgtggta ccattgtcca ttgtcttgtg tgtttgcaga
attcctgcag ctagtggcac 1440ttgatattaa ggtgatccgg gaatcagata gaccaaattg
ttcacacaac agaaatattc 1500ctcaaaacta accatccaca gaactttgat cctgtccctc
atgtacccca gaatcatcct 1560tgggaacagg aatgggagat accacatttg agaagtttgt
tacagtaatg atgtcattgt 1620cattgtcatt gtcgtcatca tcgtcatccc cagcatattc
agatgctgag ctaccatcca 1680tatccctatc aagctgaaca cattcaggaa tttcattgta
attttctgaa tcctggcttc 1740tatttgagag tcccaagttt tccttcttta cgacgttcaa
cacaatccca ttatggagga 1800tccactagtt ctagacggcc atgctagagt ccgcaaaaat
caccagtctc tctctacaaa 1860tctatctctc tctatttttc tccagaataa tgtgtgagta
gttcccagat aagggaatta 1920gggttcttat agggtttcgc tcatgtgttg agcatataag
aaacccttag tatgtatttg 1980tatttgtaaa atacttctat caataaaatt tctaattcct
aaaaccaaaa tccagtgacc 2040tcacgtgccg cgg
20534825PRTArabidopsis thaliana 4Met Asp Ile Ser
Asn Glu Ala Ser Val Asp Pro Phe Ser Ile Gly Pro 1 5
10 15 Ser Ser Ile Met Gly Arg Thr Ile Ala
Phe Arg Val Leu Phe Cys Arg 20 25
30 Ser Met Ser Gln Leu Arg Arg Asp Leu Phe Arg Phe Leu Leu
His Trp 35 40 45
Phe Leu Arg Phe Lys Leu Thr Val Ser Pro Phe Val Ser Trp Phe His 50
55 60 Pro Arg Asn Pro Gln
Gly Ile Leu Ala Val Val Thr Ile Ile Ala Phe 65 70
75 80 Val Leu Lys Arg Tyr Thr Asn Val Lys Ile
Lys Ala Glu Met Ala Tyr 85 90
95 Arg Arg Lys Phe Trp Arg Asn Met Met Arg Thr Ala Leu Thr Tyr
Glu 100 105 110 Glu
Trp Ala His Ala Ala Lys Met Leu Glu Lys Glu Thr Pro Lys Met 115
120 125 Asn Glu Ser Asp Leu Tyr
Asp Glu Glu Leu Val Lys Asn Lys Leu Gln 130 135
140 Glu Leu Arg His Arg Arg Gln Glu Gly Ser Leu
Arg Asp Ile Met Phe 145 150 155
160 Cys Met Arg Ala Asp Leu Val Arg Asn Leu Gly Asn Met Cys Asn Ser
165 170 175 Glu Leu
His Lys Gly Arg Leu Gln Val Pro Arg His Ile Lys Glu Tyr 180
185 190 Ile Asp Glu Val Ser Thr Gln
Leu Arg Met Val Cys Asn Ser Asp Ser 195 200
205 Glu Glu Leu Ser Leu Glu Glu Lys Leu Ser Phe Met
His Glu Thr Arg 210 215 220
His Ala Phe Gly Arg Thr Ala Leu Leu Leu Ser Gly Gly Ala Ser Leu 225
230 235 240 Gly Ala Phe
His Val Gly Val Val Arg Thr Leu Val Glu His Lys Leu 245
250 255 Leu Pro Arg Ile Ile Ala Gly Ser
Ser Val Gly Ser Ile Ile Cys Ala 260 265
270 Val Val Ala Ser Arg Ser Trp Pro Glu Leu Gln Ser Phe
Phe Glu Asn 275 280 285
Ser Leu His Ser Leu Gln Phe Phe Asp Gln Leu Gly Gly Val Phe Ser 290
295 300 Ile Val Lys Arg
Val Met Thr Gln Gly Ala Leu His Asp Ile Arg Gln 305 310
315 320 Leu Gln Cys Met Leu Arg Asn Leu Thr
Ser Asn Leu Thr Phe Gln Glu 325 330
335 Ala Tyr Asp Met Thr Gly Arg Ile Leu Gly Ile Thr Val Cys
Ser Pro 340 345 350
Arg Lys His Glu Pro Pro Arg Cys Leu Asn Tyr Leu Thr Ser Pro His
355 360 365 Val Val Ile Trp
Ser Ala Val Thr Ala Ser Cys Ala Phe Pro Gly Leu 370
375 380 Phe Glu Ala Gln Glu Leu Met Ala
Lys Asp Arg Ser Gly Glu Ile Val 385 390
395 400 Pro Tyr His Pro Pro Phe Asn Leu Asp Pro Glu Val
Gly Thr Lys Ser 405 410
415 Ser Ser Gly Arg Arg Trp Arg Asp Gly Ser Leu Glu Val Asp Leu Pro
420 425 430 Met Met Gln
Leu Lys Glu Leu Phe Asn Val Asn His Phe Ile Val Ser 435
440 445 Gln Ala Asn Pro His Ile Ala Pro
Leu Leu Arg Leu Lys Asp Leu Val 450 455
460 Arg Ala Tyr Gly Gly Arg Phe Ala Ala Lys Leu Ala His
Leu Val Glu 465 470 475
480 Met Glu Val Lys His Arg Cys Asn Gln Val Leu Glu Leu Gly Phe Pro
485 490 495 Leu Gly Gly Leu
Ala Lys Leu Phe Ala Gln Glu Trp Glu Gly Asp Val 500
505 510 Thr Val Val Met Pro Ala Thr Leu Ala
Gln Tyr Ser Lys Ile Ile Gln 515 520
525 Asn Pro Thr His Val Glu Leu Gln Lys Ala Ala Asn Gln Gly
Arg Arg 530 535 540
Cys Thr Trp Glu Lys Leu Ser Ala Ile Lys Ser Asn Cys Gly Ile Glu 545
550 555 560 Leu Ala Leu Asp Asp
Ser Val Ala Ile Leu Asn His Met Arg Arg Leu 565
570 575 Lys Lys Ser Ala Glu Arg Ala Ala Thr Ala
Thr Ser Ser Ser His His 580 585
590 Gly Leu Ala Ser Thr Thr Arg Phe Asn Ala Ser Arg Arg Ile Pro
Ser 595 600 605 Trp
Asn Val Leu Ala Arg Glu Asn Ser Thr Gly Ser Leu Asp Asp Leu 610
615 620 Val Thr Asp Asn Asn Leu
His Ala Ser Ser Gly Arg Asn Leu Ser Asp 625 630
635 640 Ser Glu Thr Glu Ser Val Glu Leu Ser Ser Trp
Thr Arg Thr Gly Gly 645 650
655 Pro Leu Met Arg Thr Ala Ser Ala Asn Lys Phe Ile Asp Phe Val Gln
660 665 670 Ser Leu
Asp Ile Asp Ile Ala Leu Val Arg Gly Phe Ser Ser Ser Pro 675
680 685 Asn Ser Pro Ala Val Pro Pro
Gly Gly Ser Phe Thr Pro Ser Pro Arg 690 695
700 Ser Ile Ala Ala His Ser Asp Ile Glu Ser Asn Ser
Asn Ser Asn Asn 705 710 715
720 Leu Gly Thr Ser Thr Ser Ser Ile Thr Val Thr Glu Gly Asp Leu Leu
725 730 735 Gln Pro Glu
Arg Thr Ser Asn Gly Phe Val Leu Asn Val Val Lys Arg 740
745 750 Glu Asn Leu Gly Met Pro Ser Ile
Gly Asn Gln Asn Thr Glu Leu Pro 755 760
765 Glu Ser Val Gln Leu Asp Ile Pro Glu Lys Glu Met Asp
Cys Ser Ser 770 775 780
Val Ser Glu His Glu Glu Asp Asp Asn Asp Asn Glu Glu Glu His Asn 785
790 795 800 Gly Ser Ser Leu
Val Thr Val Ser Ser Glu Asp Ser Gly Leu Gln Glu 805
810 815 Pro Val Ser Gly Ser Val Ile Asp Ala
820 825 54PRTArtificial Sequencepeptide motif
5Arg Xaa Xaa Leu 1 66PRTArtificial Sequencepeptide motif
6Gly Xaa Gly Xaa Xaa Gly 1 5 75PRTArtificial
Sequencepeptide motif 7Gly Xaa Ser Xaa Gly 1 5
834DNAArtificial Sequenceprimer sequence 8cacctacatc cagtagattg
cacgtcacac acta 34932DNAArtificial
Sequenceprimer sequence 9ctctaaaagt ttcgtgtttt ggatgatttg ct
321035DNAArtificial Sequenceprimer sequence
10ttggtcgaca tggatataag taatgaggcc aatgt
351135DNAArtificial Sequenceprimer sequence 11attgctagca ccatccacag
aactttgatc ctgtc 351237DNAArtificial
Sequenceprimer sequence 12atactcgagt ccataatggg attgtgttga acgtcgt
371336DNAArtificial Sequenceprimer sequence
13tcgaagcttg ctagtggcac ttgatattaa ggtgat
361437DNAArtificial Sequenceprimer sequence 14ataggatcct ccataatggg
attgtgttga acgtcgt 371534DNAArtificial
Sequenceprimer sequence 15tctttctgca gctagtggca cttgatatta aggt
341636DNAArtificial Sequenceprimer sequence
16tatgggccct acatccagta gattgcacgt cacaca
361738DNAArtificial Sequenceprimer sequence 17atgctcgagc tctaaaagtt
tcgtgttttg gatgattt 381830DNAArtificial
Sequenceprimer sequence 18acttctagac ggccatgcta gagtccgcaa
301930DNAArtificial Sequenceprimer sequence
19ttaccgcggc acgtgaggtc actggatttt
302027DNAArtificial Sequenceprimer sequence 20aaaaagcctg aactcaccgc
gacgtct 272124DNAArtificial
Sequenceprimer sequence 21tacttctaca cagccatcgg tcca
242223DNAArtificial Sequenceprimer sequence
22ctgaattgtc gaggtcgaag atc
232321DNAArtificial Sequenceprimer sequence 23agcagccaaa tgggtctgtc c
212424DNAArtificial
Sequenceprimer sequence 24cttctcatta tcggtggtga acat
242521DNAArtificial Sequenceprimer sequence
25ctctgtctgg attggagggt c
212620DNAArtificial Sequenceprimer sequence 26gcttgagaaa tggtcggaaa
202726DNAArtificial
Sequenceprimer sequence 27tgccgctata accgttttct cttgga
262827DNAArtificial Sequenceprimer sequence
28atgttcccca ccagtatgtt gctgtcc
272923DNAArtificial Sequenceprimer sequence 29atgcggttgg ctacgcagga caa
233023DNAArtificial
Sequenceprimer sequence 30tcatgcagcc gtcgtcctcc cct
233124DNAArtificial Sequenceprimer sequence
31tggaatagcc gccattaccg cctt
243228DNAArtificial Sequenceprimer sequence 32agaaacttgt tggtgttgga
ctccaatg 283320DNAArtificial
Sequenceprimer sequence 33atgccatcga ttgggaacca
203423DNAArtificial Sequenceprimer sequence
34caccggttct tgtaaaccgg aat
233524DNAArtificial Sequenceprimer sequence 35gaggctggat ctggcaaaca cgtt
243625DNAArtificial
Sequenceprimer sequence 36tgtgtaatga cctctagcaa aatta
253726DNAArtificial Sequenceprimer sequence
37cataatggga ttgtgttgaa cgtcgt
263824DNAArtificial Sequenceprimer sequence 38gtcatcatcg tcatccccag cata
243925DNAArtificial
Sequenceprimer sequence 39cctcagcctc tctcttactt ggctt
254021DNAArtificial Sequenceprimer sequence
40ccaggagaag taagggacga g
214127DNAArtificial Sequenceprimer sequence 41ctacttgttg gtccataatg
ttcgtca 274224DNAArtificial
Sequenceprimer sequence 42cgtagataac tccattcctt gcct
244328DNAArtificial Sequenceprimer sequence
43gtcttaacca actttttcaa cctctgtt
284429DNAArtificial Sequenceprimer sequence 44ccaaaccgaa aacagtgaaa
gcaaagagt 294524DNAArtificial
Sequenceprimer sequence 45aatctatcct tcaacctccg ccgt
244624DNAArtificial Sequenceprimer sequence
46agagaatctg ctccaaggtc agct
244724DNAArtificial Sequenceprimer sequence 47ggatctcctt tccctccgat gttt
244824DNAArtificial
Sequenceprimer sequence 48ataaattcag agagctcggc gggt
244923DNAArtificial Sequenceprimer sequence
49tggcatcact ggtctcccga tgt
235027DNAArtificial Sequenceprimer sequence 50tcatacctct tgatttcagt
aacgaga 275125DNAArtificial
Sequenceprimer sequence 51agtcactatc gtccttccgg cacaa
255226DNAArtificial Sequenceprimer sequence
52cgctctgtac gattgctatt tcctct
265326DNAArtificial Sequenceprimer sequence 53tcaagtcagt tcactgcaga
tctaat 265428DNAArtificial
Sequenceprimer sequence 54caactcggta aagaccaggc atagtgaa
285522DNAArtificial Sequenceprimer sequence
55tcgcaaaaca cacatcacac ac
225623DNAArtificial Sequenceprimer sequence 56gtgattgacg atttgatggt aag
235719DNAArtificial
Sequenceprimer sequence 57tggtcttcct gcttctgcc
195819DNAArtificial Sequenceprimer sequence
58ccgcttctca ctgcctcat
19
User Contributions:
Comment about this patent or add new information about this topic: