Patent application title: Method for Making Radioactive Isotopic Gallium-67
Inventors:
Sun-Rong Huang (Taoyuan County, TW)
Ying-Ming Tsai (Taoyuan County, TW)
Jainn-Hsin Lu (Taoyuan County, TW)
Jenn-Tzong Chen (Taipei City, TW)
Wuu-Jyh Lin (Taoyuan County, TW)
Assignees:
ATOMIC ENERGY COUNCIL-INSTITUTE OF NUCLEAR ENERGY RESEARCH
IPC8 Class: AC25C122FI
USPC Class:
205340
Class name: Electrolysis: processes, compositions used therein, and methods of preparing the compositions electrolytic synthesis (process, composition, and method of preparing composition) utilizing electromagnetic wave energy during synthesis (e.g., visible light, etc.)
Publication date: 2011-09-08
Patent application number: 20110214995
Abstract:
Disclosed is a method for making carrier-free radioactive isotopic
gallium-67. Stable isotopic zinc-68 is turned into zinc-68 solid target
by disposition or electroplating. Then, the zinc-68 solid target is
subjected to a proton beam. A cyclotron is used to provide irradiation
from 15 to 40 MeV. After the irradiation, the zinc-68 solid target is
dissolved in concentrated acid and turned into solution that contains
zinc-65, zinc-68, gallium-67 and gallium-68. High concentrated
hydrochloric acid and resin are added into the solution for exchange of
ions. Zinc-68 liquid and gallium-67 liquid are filtered and separated
from the solution. Thus, pure gallium-67 liquid is produced. With a
vaporizer, the gallium-67 liquid is vaporized and turned into radioactive
isotopic gallium-67.Claims:
1. A method for making carrier-free radioactive isotopic gallium-67, the
method comprising the steps of: turning isotopic zinc-68 into a zinc-68
solid target by electroplating; subjecting the zinc-68 solid target to
proton irradiation; dissolving the zinc-68 solid target in strong acid,
thus providing solution that contains zinc-65, zinc-68, gallium-67 and
gallium-68; adding hydrochloric acid and resin in the solution for
exchange of ions; filtering the solution, thus providing zinc-68 solution
and gallium-67 solution; and evaporating the gallium-67 solution, thus
providing radioactive isotopic gallium-67.
2. The method according to claim 1, wherein the intensity of the proton irradiation is 15 to 40 MeV.
3. The method according to claim 1, wherein the strong acid used to dissolve the zinc-68 solid target is hydrochloric acid with concentration of 8N to 12N.
4. The method according to claim 1, wherein the concentration of the hydrochloric acid added in the solution is 8N to 12N.
Description:
TECHNICAL FIELD
[0001] The present disclosure relates to a method for making radioactive isotopic gallium-67 and, more particularly, to a method for rapidly filtering pure gallium-67 liquid.
DESCRIPTION OF THE RELATED ARTS
[0002] Gallium-67 (67Ga) is a radioactive isotope produced by a cyclotron. The nuclear properties of gallium-67 are short half life (t1/2=78 h) and medium energy (E.C.=100%, Er=93 KeV (38%), 185 KeV (23.6%) and 300 KeV (19%)). Therefore, gallium-67 is often used in single photon emission computerized tomography ("SPECT") for human organs. Substantial nuclear reaction in the production of gallium-67 includes 68Zn (p, 2n), 67Zn (d, 2n) and 65Cu (α, 2n). The yield of the production of gallium-67 is highest if high-concentration zinc-68 (higher than 97%) with proper thickness (0 to 100 μm) is used as a target subject to proper proton irradiation (0 to 22 Mev). By dissolving the irradiated target and chemical separation, carrier-free gallium-67 chloride and citrate are provided. The carrier-free gallium-67 chloride and citrate can be combined with other organic ligands and turned into nuclear medicine for medical treatment. However, the radioactive isotopes cannot be produced in Taiwan, and all of the gallium-68 necessary for the production of the nuclear medical citrate is imported difficultly, expensively.
[0003] The present disclosure is therefore intended to obviate or at least alleviate the problems encountered in prior art.
SUMMARY OF THE DISCLOSURE
[0004] It is the primary objective of the present disclosure to provide a method for making radioactive isotopic gallium-67.
[0005] To achieve the foregoing objective, isotopic zinc-68 is turned into a zinc-68 solid target by disposition or electroplating. Then, the zinc-68 solid target is subjected to proton irradiation. The irradiated zinc-68 solid target is dissolved in strong acid and turned into solution that contains zinc-65, zinc-68, gallium-67 and gallium-68. Hydrochloric acid and resin are added into the solution for exchange of ions. Zinc-68 solution and gallium-67 solution are separated from the solution by filtering. The gallium-67 solution is evaporated, thus providing radioactive isotopic gallium-67.
[0006] Other objectives, advantages and features of the present disclosure will be apparent from the following description referring to the attached drawings.
BRIEF DESCRIPTIONS OF THE DRAWINGS
[0007] The present disclosure will be described via detailed illustration of the preferred embodiment referring to the drawings wherein:
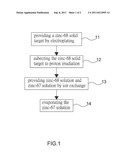
[0008] FIG. 1 is a flow chart of a method for making radio isotopic gallium-67 according to the preferred embodiment of the present disclosure; and
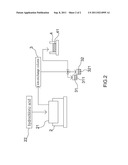
[0009] FIG. 2 is a scheme of equipment used in the method shown in FIG. 1 to make radio isotopic gallium-67.
DESCRIPTION OF THE PREFERRED EMBODIMENT
[0010] Referring to FIG. 1, there is shown a method for making radioactive isotopic gallium-67 according to the preferred embodiment of the present disclosure. Referring to FIG. 2, there is shown equipment used in the method to make radioactive isotopic gallium-67. The equipment includes a dissolving unit 2, an ion-exchange column 3, a zinc-68 solution-recycling unit 31, a waste liquid-recycling unit 32 and an evaporation unit 4.
[0011] At 11, zinc-68 is turned into a zinc-68 solid target 21 by electroplating.
[0012] At 12, the zinc-68 solid target 21 is subjected to proton irradiation in a cyclotron (not shown). The intensity of the proton irradiation in the cyclotron is 15 to 40 MeV.
[0013] At 13, the irradiated zinc-68 solid target 21 is dissolved in strong acid in the dissolving unit 2. Thus, there is provided solution containing zinc-68, gallium-66, gallium-67 and gallium-68. The strong acid can be hydrochloric acid with concentration of 8N to 12N and preferably 9N.
[0014] At 14, some of the solution is filled in the ion-exchange column 3. Hydrochloric acid, with concentration of 8N to 12N and preferably 9N, and resin are filled in the ion-exchange column 3 for ion exchange. Gallium-67 solution 41 is absorbed. Zinc-68 solution 311 is sent into the zinc-68 solution-recycling bottle 31. Low-concentration solution 32 is transferred into the waste liquid-recycling unit 32.
[0015] At 15, the gallium-67 solution is heated in an evaporation unit 4, so that the hydrochloric acid is evaporated and then radioactive isotopic gallium-67 is made.
[0016] The present disclosure has been described via the detailed illustration of the preferred embodiment. Those skilled in the art can derive variations from the preferred embodiment without departing from the scope of the present disclosure. Therefore, the preferred embodiment shall not limit the scope of the present disclosure defined in the claims.
User Contributions:
Comment about this patent or add new information about this topic:
| People who visited this patent also read: | |
| Patent application number | Title |
|---|---|
| 20140368995 | PORTABLE ELECTRONIC DEVICE |
| 20140368994 | WATERPROOF DOOR ASSEMBLY OF ELECTRONIC DEVICE |
| 20140368993 | USING SYNTHETIC SETAE ON MOBILE DEVICE HOUSING |
| 20140368992 | Methods For Establishing Thermal Joints Between Heat Spreaders and Heat Generating Components Using Thermoplastic and/or Self-Healing Thermal Interface Materials |
| 20140368991 | RENEWABLE ENERGY BASED DATACENTER COOLING |