Patent application title: KLOTHO PROTEIN AND RELATED COMPOUNDS FOR THE TREATMENT AND DIAGNOSIS OF CANCER
Inventors:
H. Phillip Koeffler (Los Angeles, CA, US)
Ido Wolf (Or Yehuda, IL)
Ido Wolf (Or Yehuda, IL)
Tamar Rubinek (Or Yehuda, IL)
Bella Kaufman (Kiryat Ono, IL)
Lilach Abramovitch (Kfar Saba, IL)
IPC8 Class: AA61K3847FI
USPC Class:
424 9461
Class name: Enzyme or coenzyme containing hydrolases (3. ) (e.g., urease, lipase, asparaginase, muramidase, etc.) acting on glycosyl compound (3.2) (e.g., glycosidases lysozyme, nucleosidases, cellulase, etc.)
Publication date: 2010-12-30
Patent application number: 20100330062
Claims:
1-31. (canceled)
32. A method for treating cancer, comprising administering to a subject in need thereof a pharmaceutically effective amount of a composition comprising a klotho protein or a DNA encoding klotho protein.
33. The method of claim 32, wherein said cancer is an IGF-1 dependent cancer.
34. The method of claim 32, wherein said cancer is selected from the group consisting of colon cancer, prostate cancer, lung cancer, cervical cancer and ovarian cancer.
35. The method of claim 32, wherein said cancer is breast cancer.
36. The method of claim 32, wherein said cancer is pancreatic cancer.
37. The method of claim 32, wherein said klotho protein comprises native klotho protein.
38. The method of claim 32, wherein said klotho protein is a soluble protein.
39. The method of claim 32, wherein said klotho protein is selected from the group consisting of:a) an isolated polypeptide including the amino acid sequence selected from the group of amino acid sequences represented by SEQ ID NOS: 1, 2, 3, 4, 5, 11, 12, 13, 14 and 15;b) an isolated polypeptide having a homology of at least 80% with a polypeptide including the amino acid sequence selected from the group of amino acid sequences represented by SEQ ID NOS: 1, 2, 3, 4, 5, 11, 12, 13, 14 and 15;c) an isolated polypeptide comprising an amino acid sequence wherein at least one amino acid of the amino acid sequence of a polypeptide comprising an amino acid sequence selected from amino acid sequences represented by SEQ ID NOS: 1, 2, 3, 4, 5, 11, 12, 13, 14 and 15 is deleted, substituted or added; andd) an isolated polypeptide comprising an amino acid sequence selected from the group of amino acid sequences represented by residues 31 to 982 of SEQ ID NO: 1, residues 34 to 1012 of SEQ ID NO: 1, residues 34 to 549 of SEQ ID NO: 2 and residues 36 to 1014 of SEQ ID NO: 3.
40. The method of claim 39, wherein said homology is of at least 90% or more therewith.
41. The method of claim 32, wherein said DNA encoding klotho protein is selected from the group consisting of:a) DNA encoding a polypeptide comprising an amino acid sequence selected from the amino acid sequences represented by SEQ ID NOS: 1, 2, 3, 4, 5, 11, 12, 13, 14 and 15 or isolated polypeptides having a homology of at least 80% therewith or a DNA encoding a polypeptide comprising an amino acid sequence wherein at least one amino acid of the amino acid sequence of a polypeptide comprising an amino acid sequence selected from amino acid sequences represented by SEQ ID NOS: 1, 2, 3, 4, 5, 11, 12, 13, 14 and 15 is deleted, substituted or added;(b) DNA encoding a polypeptide comprising an amino acid sequence selected from the amino acid sequences represented by residues 31 to 982 of SEQ ID NO: 1, residues 34 to 1012 of SEQ ID NO: 1, residues 34 to 549 of SEQ ID NO: 2 and residues 36 to 1014 of SEQ ID NO: 3;c) DNA comprising a DNA sequence selected from DNA sequences represented by SEQ ID NOS: 6, 7, 8, 9 and 10; andd) DNA which hybridizes with a DNA described in (a), (b) or (c) under stringent conditions.
42. The method of claim 32, wherein said DNA encoding klotho protein is provided inserted in a recombinant vector suitable for gene therapy.
43. The method of claim 32, wherein said klotho protein or DNA encoding klotho protein is of a form selected from the group consisting of murine, human, bovine, canine or equine forms.
44. A pharmaceutical composition comprising a klotho protein or DNA encoding klotho protein and a pharmaceutically acceptable carrier for use as a medicament for treating cancer.
45. The composition of claim 44, wherein said cancer is an IGF-1 dependent cancer.
46. The composition of claim 45, wherein said cancer is selected from the group consisting of colon cancer, prostate cancer, lung cancer, cervical cancer and ovarian cancer.
47. The composition of claim 44, wherein said cancer is breast cancer.
48. The composition of claim 44, wherein said cancer is pancreatic cancer.
49. The composition of claim 44, wherein said klotho protein comprises native klotho protein.
50. The composition of claim 44, wherein said klotho protein is a soluble protein.
51. The composition of claim 44, wherein said klotho protein is selected from the group consisting of:a) an isolated polypeptide including the amino acid sequence selected from the group of amino acid sequences represented by SEQ ID NOS: 1, 2, 3, 4, 5, 11, 12, 13, 14 and 15;b) an isolated polypeptide having a homology of at least 80% with a polypeptide including the amino acid sequence selected from the group of amino acid sequences represented by SEQ ID NOS: 1, 2, 3, 4, 5, 11, 12, 13, 14 and 15;c) an isolated polypeptide comprising an amino acid sequence wherein at least one amino acid of the amino acid sequence of a polypeptide comprising an amino acid sequence selected from amino acid sequences represented by SEQ ID NOS: 1, 2, 3, 4, 5, 11, 12, 13, 14 and 15 is deleted, substituted or added; andd) an isolated polypeptide comprising an amino acid sequence selected from the group of amino acid sequences represented by residues 31 to 982 of SEQ ID NO: 1, residues 34 to 1012 of SEQ ID NO: 1, residues 34 to 549 of SEQ ID NO: 2 and residues 36 to 1014 of SEQ ID NO: 3.
52. The composition of claim 51, wherein said homology is of at least 90% or more therewith.
53. The composition of claim 44, wherein said DNA encoding klotho protein is selected from the group consisting of:a) DNA encoding a polypeptide comprising an amino acid sequence selected from the amino acid sequences represented by SEQ ID NOS: 1, 2, 3, 4, 5, 11, 12, 13, 14 and 15 or isolated polypeptides having a homology of at least 80% therewith or a DNA encoding a polypeptide comprising an amino acid sequence wherein at least one amino acid of the amino acid sequence of a polypeptide comprising an amino acid sequence selected from amino acid sequences represented by SEQ ID NOS: 1, 2, 3, 4, 5, 11, 12, 13, 14 and 15 is deleted, substituted or added;(b) DNA encoding a polypeptide comprising an amino acid sequence selected from the amino acid sequences represented by residues 31 to 982 of SEQ ID NO: 1, residues 34 to 1012 of SEQ ID NO: 1, residues 34 to 549 of SEQ ID NO: 2 and residues 36 to 1014 of SEQ ID NO: 3;c) DNA comprising a DNA sequence selected from DNA sequences represented by SEQ ID NOS: 6, 7, 8, 9 and 10; andd) DNA which hybridizes with a DNA described in (a), (b) or (c) under stringent conditions.
54. The composition of claim 44, wherein said DNA encoding klotho protein is provided inserted in a recombinant vector suitable for gene therapy.
55. The composition of claim 44, wherein said klotho protein or DNA encoding klotho protein is of a form selected from the group consisting of murine, human, bovine, canine or equine forms.
56. A method for the diagnosis of cancer in a subject, the method comprising:measuring a level of klotho protein present in a test sample obtained from the subject; andcomparing said level of klotho protein in said test sample to that of a reference samplewherein a reduced level of klotho protein in said test sample relative to that of said reference sample is indicative of the presence of cancer in the subject.
57. A method of predicting the development of cancer in a benign breast disease patient, the method comprising:measuring a level of klotho protein in a test sample obtained from a benign breast lesion; andcomparing said level of klotho protein in said test sample to that of a reference sample comprising breast tissue from an area adjacent to said lesionwherein a reduced level of klotho protein in said test sample relative to that of said reference sample is indicative of a relatively higher risk for developing breast cancer.
Description:
RELATED PATENT APPLICATIONS
[0001]The present application is the US National Phase of PCT/IL2008/000618 and claims priority from U.S. Provisional Patent Application No. 60/916,787 filed 8 May 2007 and from U.S. Provisional Patent Application No. 61/042,918 filed 7 Apr. 2008, both included by reference as if fully set forth herein.
FIELD AND BACKGROUND OF THE INVENTION
[0002]The present invention relates to the field of therapeutics, and more specifically to the use of the klotho protein for the treatment and diagnosis of cancer, such as breast cancer and pancreatic cancer as well as other IGF-1 dependent cancers.
[0003]Cancer is a group of diseases in which cells grow and divide without respect to normal limits, forming a tumor, and invading and destroying adjacent tissues. Cancer cells may spread to other locations in the body, resulting in a metastatic tumor composed of cells of the same type as those of the original tumor. Cancers are treated in a number of ways including surgery (excision of a tumor), radiation therapy (directed irradiation with X-rays to destroy cancer cells) and chemotherapy (administration of APIs that are more toxic to cancer cells than to non-cancer cells). All these therapies are associated with severe side-effects, and their activity in most types of metastatic cancers is limited.
[0004]Common forms of cancer include breast cancer and pancreatic cancer.
[0005]Breast cancer, in which malignant cells develop in the tissues of the breast, is the most common cancer among women, and the second most common cause of cancer death in women in the United States. Breast tissue is composed of lobes and ducts and also includes blood vessels and lymph vessels, which are connected to lymph nodes. Clusters of lymph nodes are found near the breast in the axilla, under the arm, above the collarbone, and in the chest. Several different types of breast cancer exist, which vary with respect to the types of cells in which they first appear, the symptoms which develop, and how frequently they occur.
[0006]Risk factors for breast cancer include older age, early age onset of menstruation, never having given birth, family history (mother or sister) of breast cancer, radiation therapy to the chest, increased density of breast tissue as identified by mammogram, use of hormones such as estrogen and progesterone, frequent alcohol consumption, race-associated factors, and the presence of particular genetic mutations.
[0007]In addition, the presence of benign breast disease is associated with an increased risk of breast cancer. Thus, proliferative lesions without atypia are associated with a 1.5- to 2-fold increased risk of breast cancer, whereas atypical hyperplasias are associated with a 4- to 5-fold increased risk. Currently, there are no markers which can predict increased risk of breast cancer among benign breast disease patients.
[0008]Breast cancer can metastasize to almost any other part of the body, including the lymph nodes, bones, liver, lungs, and brain. Metastatic breast cancer is incurable with a median survival period of about 2 years. Breast cancers are treated with surgery, radiation therapy, chemotherapy and, according to specific tumor characteristics, may also be treated with hormonal therapy or antibodies directed against human epidermal growth factor (HER2) protein. Treatment is also administered before (neo-adjuvant) or after (adjuvant) a surgery in which the primary tumor has been removed, in order to prevent disease recurrence. All forms of therapy, including chemotherapy, radiotherapy hormonotherapy and biological therapy may also be used.
[0009]Pancreatic cancer is a malignant tumor of the pancreas. Each year about 33,000 individuals in the United States are diagnosed with this condition, and more than 60,000 in Europe. Depending on the extent of the tumor at the time of diagnosis, the prognosis is generally regarded as poor. Median survival from diagnosis is around 3 to 6 months; 5-year survival is much less than 5%; and complete remission is extremely rare. With 37,170 cases diagnosed in the United States in 2007, and 33,700 deaths, pancreatic cancer has the highest fatality rate of all cancers and is the fourth highest cancer killer in the United States among both men and women. Although it accounts for only 2.5% of new cases, pancreatic cancer is responsible for 6% of cancer deaths each year.
[0010]Treatment of pancreatic cancer depends on the stage of the cancer, and includes surgical removal of tumors and chemotherapy. The Whipple procedure is the most common surgical treatment for pancreatic cancers. This procedure is a major surgical operation involving the pancreas, duodenum, and other organs, and carries a significant degree of risk.
[0011]Patients diagnosed with pancreatic cancer typically have a poor prognosis partly because the cancer usually causes no symptoms early on, leading to locally advanced or metastatic disease at time of diagnosis. As a result of the aggressive invasion and early metastasis characteristics of the disease, 90% of patients have surgically unresectable disease at the time of diagnosis.
[0012]Chemotherapeutic agents for treating cancers in general include alkylating agents, antimetabolites, anthracyclines, plant alkaloids, topoisomerase inhibitors, hormone receptor modulators, and hormone level modulators. Chemotherapeutic agents are generally associated with severe side effects, due mainly to the inability of the chemotherapeutic agent to distinguish between normal and healthy cells, such that certain fast-growing, normal cells are also attacked. These include blood cells forming in the bone marrow and cells in the digestive tract (mouth, stomach, intestines, esophagus), reproductive system (sexual organs), and hair follicles. Some chemotherapeutic agents may affect cells of vital organs, such as the heart, kidney, bladder, lungs, and nervous system.
[0013]Commonly used chemotherapeutic agents for treatment of pancreatic cancer and breast cancer are taxanes, anthracyclines and antimetabolites such as fluorouracil and gemcitabine. Combination therapy with doxorubicin or mitomycin, may also be used. All of these are associated with side effects. For example, fluorouracil commonly causes diarrhea, mouth ulcers, hair thinning or hair loss, and increased risk of infection; and gemcitabine is associated with nausea, swelling of the feet or legs or weight gain due to fluid retention, flu like symptoms such as headache, chills and fever, and increased risk of infection.
[0014]The only therapy currently available for metastatic pancreatic cancer is standard chemotherapy (normally using gencitabine, either alone or in combination with capecitabine, erlotinib or platinum agents), which adds about 1 to 2 months to the median survival time.
[0015]When the presence of cancer is suspected in a patient, an imaging study is first performed, such as x-ray, ultrasonography or computed tomography. Imaging studies show the presence, location and size of an abnormal mass, but not whether the mass is cancerous. Cancer can only be positively diagnosed by biopsy, which involves removal of a small tissue sample. The biopsy may be performed using fine needle aspiration, wherein a narrow needle is inserted into a lump or tumor, and cells are withdrawn.
[0016]Some types of tumors, including breast tumors, may require an excisional biopsy, wherein a whole lump is surgically removed.
[0017]Tissue obtained by biopsy is generally analyzed by microscopic analysis of histological sections, which is a time consuming process, requiring a certain amount of subjective evaluation by the pathologist conducting the analysis.
[0018]Given the importance of detecting cancer early in its development, and the potential harm that may occur as a result of false positives or false negatives from mammography and other techniques for screening of various types of cancer, a substantial need remains for a more accurate and less invasive means of determining the presence of cancer. Moreover, currently used techniques cannot predict cancer development among patients with benign breast cancer.
[0019]It has therefore been desirable to find a treatment for cancer, such as pancreatic cancer and breast cancer, which overcomes at least some of the drawbacks of the background art.
[0020]It is further desirable to find a method of diagnosis of cancer which overcomes at least some of the drawbacks of the background art.
SUMMARY OF THE INVENTION
[0021]Some embodiments of the present invention relate to the use of klotho protein and related compounds in the treatment and diagnosis of cancer, such as breast cancer and pancreatic cancer.
[0022]According to an aspect of some embodiments of the present invention, there is provided klotho protein or DNA encoding klotho protein for use in the treatment of cancer. According to some embodiments, the klotho protein or DNA is used together with a chemotherapeutic agent.
[0023]According to an aspect of some embodiments of the present invention, there is provided for the use of klotho protein or of DNA encoding klotho protein in the manufacture of a medicament for the treatment of cancer, substantially as described in the specification. According to some embodiments, the klotho protein or DNA is used together with a chemotherapeutic agent.
[0024]According to an aspect of some embodiments of the present invention, there is provided for the use of a composition comprising a pharmaceutically acceptable amount of a klotho protein or of DNA encoding klotho protein for the treatment of cancer. According to some embodiments, the klotho protein or DNA is used together with a chemotherapeutic agent. According to some embodiments, the composition further comprises a chemotherapeutic agent.
[0025]According to an aspect of some embodiments of the present invention, there is provided a method for treating cancer, comprising administering to a subject in need thereof a pharmaceutically effective amount of a composition comprising a klotho protein or a DNA encoding klotho protein. According to some embodiments, the method further comprises co-administration of a chemotherapeutic agent.
[0026]According to an aspect of some embodiments of the present invention, there is provided a pharmaceutical composition comprising a klotho protein or DNA encoding klotho protein and a pharmaceutically acceptable carrier for use as a medicament for treating cancer. According to some embodiments, the composition further comprises a chemotherapeutic agent.
[0027]According to some embodiments of the present invention, the cancer is an IGF-1 dependent cancer. In some embodiments, the IGF-1 dependent cancer is colon cancer, prostate cancer, lung cancer, cervical cancer or ovarian cancer.
[0028]According to some embodiments, the cancer is breast cancer.
[0029]According to some embodiments, the cancer is pancreatic cancer.
[0030]According to some embodiments, the klotho protein comprises native klotho protein.
[0031]According to some embodiments, the klotho protein is a soluble protein.
[0032]According to some embodiments, the klotho protein is selected from the group consisting of: a) an isolated polypeptide including the amino acid sequence selected from the group of amino acid sequences represented by SEQ ID NOS: 1, 2, 3, 4, 5, 11, 12, 13, 14 and 15; b) an isolated polypeptide having a homology of at least 80% or more with a polypeptide including the amino acid sequence selected from the group of amino acid sequences represented by SEQ ID NOS: 1, 2, 3, 4, 5, 11, 12, 13, 14 and 15; c) an isolated polypeptide comprising an amino acid sequence wherein at least one amino acid of the amino acid sequence of a polypeptide comprising an amino acid sequence selected from amino acid sequences represented by SEQ ID NOS: 1, 2, 3, 4, 5, 11, 12, 13, 14 and 15 is deleted, substituted or added; and d) an isolated polypeptide comprising an amino acid sequence selected from the group of amino acid sequences represented by residues 31 to 982 of SEQ ID NO: 1, residues 34 to 1012 of SEQ ID NO: 1, residues 34 to 549 of SEQ ID NO: 2 and residues 36 to 1014 of SEQ ID NO: 3. In some embodiments, the homology is of at least 90% or more, at least 95% or more and even of at least 98% or more.
[0033]According to some embodiments, the DNA encoding klotho protein is selected from the group consisting of: a) DNA encoding a polypeptide comprising an amino acid sequence selected from the amino acid sequences represented by SEQ ID NOS: 1, 2, 3, 4, 5, 11, 12, 13, 14 and 15 or isolated polypeptides having a homology of at least 80% (in some embodiments, the homology is of at least 90%, at least 95% and even of at least 98%) or more therewith or a DNA encoding a polypeptide comprising an amino acid sequence wherein at least one amino acid of the amino acid sequence of a polypeptide comprising an amino acid sequence selected from amino acid sequences represented by SEQ ID NOS: 1, 2, 3, 4, 5, 11, 12, 13, 14 and 15 is deleted, substituted or added; b) DNA encoding a polypeptide comprising an amino acid sequence selected from the amino acid sequences represented by residues 31 to 982 of SEQ ID NO: 1, residues 34 to 1012 of SEQ ID NO: 1, residues 34 to 549 of SEQ ID NO: 2 and residues 36 to 1014 of SEQ ID NO: 3, and c) DNA comprising a DNA sequence selected from DNA sequences represented by SEQ ID NOS: 6, 7, 8, 9 and 10; d) DNA which hybridizes with a DNA described in (a), (b) or (c) under stringent conditions.
[0034]According to some embodiments, the DNA encoding klotho protein is provided inserted in a recombinant vector suitable for gene therapy.
[0035]According to some embodiments, the klotho protein or DNA encoding klotho protein is of a form selected from the group consisting of murine, human, bovine, canine or equine forms.
[0036]According to some embodiments of methods or uses of the present invention, the klotho protein or DNA encoding klotho protein and the chemotherapeutic agent are administered in a single dosage form. According to some embodiments, the klotho protein or DNA encoding klotho protein and the chemotherapeutic agent are administered sequentially in separate dosage forms.
[0037]According to an aspect of some embodiments of the present invention, there is provided a composition comprising a combination of at least two active pharmaceutical ingredients, wherein a first active pharmaceutical ingredient is a klotho protein and a second active pharmaceutical ingredient is at least one chemotherapeutic agent, wherein the amount of the at least one klotho protein alone and the amount of the at least one chemotherapeutic agent alone is insufficient to achieve the therapeutic effect achieved by the administration of the combination of two or more of the agents.
[0038]According to an aspect of some embodiments of the present invention, there is provided a method for the diagnosis of cancer in a subject, the method comprising: measuring a level of klotho protein present in a test sample obtained from the subject; and comparing the level of klotho protein in the test sample to that of a reference sample wherein a reduced level of klotho protein in the test sample relative to that of the reference sample is indicative of the presence of cancer in the subject. In some embodiments, a reduced level of klotho protein in the test sample relative to that of the reference sample is indicative of an increased risk for developing cancer.
[0039]According to some embodiments, the cancer diagnosed is an IGF-1 dependent cancer, for example, colon cancer, prostate cancer, lung cancer, cervical cancer and ovarian cancer.
[0040]According to some embodiments, the diagnosed cancer is breast cancer.
[0041]According to some embodiments, the diagnosed cancer is pancreatic cancer.
[0042]According to some embodiments, the test sample comprises a biopsy of tissue from a breast tumor, and the reference sample comprises a biopsy of normal breast tissue adjacent to the breast tumor. According to some embodiments, the method is used for screening benign breast disease patients for the presence of breast cancer. According to some embodiments, the method is used for screening benign breast disease patients for evaluating the relative risk of developing of breast cancer.
[0043]According to an aspect of some embodiments of the present invention, there is provided a method of predicting the development of cancer in a benign breast disease patient, the method comprising: measuring a level of klotho protein in a test sample obtained from a benign breast lesion; and comparing the level of klotho protein in the test sample to that of a reference sample comprising breast tissue from an area adjacent to the lesion wherein a reduced level of klotho protein in the test sample relative to that of the reference sample is indicative of a relatively higher risk for developing breast cancer.
[0044]Unless otherwise defined, all technical and scientific terms used herein have the same meaning as commonly understood by one of ordinary skill in the art to which this invention pertains. In case of conflict, the patent specification, including definitions, will control.
[0045]As used herein, the term "treating" includes curing a condition, treating a condition, preventing a condition, treating symptoms of a condition, curing symptoms of a condition, ameliorating symptoms of a condition, treating effects of a condition, ameliorating effects of a condition, and preventing results of a condition.
[0046]As used herein, the terms "comprising", "including", "having" and grammatical variants thereof are to be taken as specifying the stated features, integers, steps or components but do not preclude the addition of one or more additional features, integers, steps, components or groups thereof. These terms encompass the terms "consisting of" and "consisting essentially of".
[0047]The phrase "consisting essentially of" or grammatical variants thereof when used herein are to be taken as specifying the stated features, integers, steps or components but do not preclude the addition of one or more additional features, integers, steps, components or groups thereof but only if the additional features, integers, steps, components or groups thereof do not materially alter the basic and novel characteristics of the claimed composition, device or method.
[0048]As used herein, the indefinite articles "a" and "an" mean "at least one" or "one or more" unless the context clearly dictates otherwise.
BRIEF DESCRIPTION OF THE FIGURES
[0049]Some embodiments of the invention are herein described, by way of example only, with reference to the accompanying figures. The description, together with the figures, makes apparent how embodiments of the invention may be practiced to those skilled in the art. It is stressed that the particulars shown in the figures are by way of example and for purposes of illustrative discussion of embodiments of the invention.
[0050]In the figures:
[0051]FIG. 1 shows immunohistochemical staining of klotho protein. Upper panel: Kidney section [magnifications: ×400, left and ×1000, right]. Middle panel: normal breast, shown at ×400, left and ×1000 magnifications. Lower panel, Ductal carcinoma in situ (DCIS, left) and invasive ductal carcinoma (IDC, right);
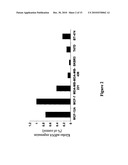
[0052]FIG. 2 is a bar chart showing klotho protein mRNA levels as determined by quantitative real-time RT-PCR in different breast cancer cell lines, relative to the expression in MCF-7;
[0053]FIG. 3a shows the results of a colony formation assay, in which colonies were stained with crystal violet and photographed;
[0054]FIG. 3b is a bar chart showing relative viability of stably transfected MCF-7 subclones (KL-HA) and control (pcDNA3);
[0055]FIG. 4a is a bar chart showing klotho protein mRNA expression in MCF-7 cells transfected with control siRNA (siRNAc) or three klotho protein-directed siRNA (siRNA1-3);
[0056]FIG. 4b is a bar chart showing relative viability of MCF-7 cells transfected with control siRNA (siRNAc) or three klotho protein-directed siRNA (siRNA1-3);
[0057]FIG. 5 shows an immunoblot of MCF-7 and HEK293 cells transfected with either klotho protein expressing vector (KL) or an empty vector (C);
[0058]FIG. 6a is a bar chart showing relative viability of MCF-7 cells transfected with either klotho protein expression plasmid (KL-HA) or a control vector (pcDNA3), and treated by IGF-1; (12.5 nM) for the indicated times;
[0059]FIG. 6b shows a representative plate of MCF-7 cells, after 96 hr of IGF-1 treatment;
[0060]FIG. 7a shows an immunoblot of MCF-7 cells transfected with either klotho protein-HA expression plasmid (KL-HA) or a control vector (pcDNA3) and treated with IGF-1 (15 min) for the indicated doses;
[0061]FIG. 7b shows an immunoblot of MCF-7 cells transfected with either klotho protein-HA expression plasmid (KL-HA) or a control vector (pcDNA3) and treated with IGF-1 (12.5 nM) for the indicated times;
[0062]FIG. 8 shows an immunoblot of MCF-7 and HEK293 cells transfected with either klotho protein-HA expression plasmid (KL-HA) or a control vector (pcDNA3) and stimulated with IGF-1 (12.5 nM, 15 min). Cell lysates were immunoblotted using anti-phosphorylated (p) and total (t) IGF-1R, p- and t-IRS-1, p- and t-AKT, p- and t-GSK3β, p- and t-ERK1/2 and C/EBPβ antibodies;
[0063]FIG. 9a shows an immunoblot of klotho protein silenced in MCF-7 cells using siRNA3 or control siRNA (si-C). Cell lysates were immunoblotted using anti p- and t-AKT antibody;
[0064]FIG. 9b shows an immunoblot of MCF-7 cells treated with soluble klotho protein and stimulated with IGF-1 (12.5 nM, 15 min). Cell lysates were immunoblotted using anti-phosphorylated (p) and total (t) IGF-1R;
[0065]FIG. 10 shows an immunoblot of MCF-7 cells stably expressing HA-klotho protein or a control vector, which were lysed and immunoprecipitated for klotho protein using anti-HA antibody and immunoblotted with anti-IGF-1R antibodies;
[0066]FIG. 11a shows an immunoblot of MCF-7 and HEK293 cells transfected and stimulated with insulin. Cell lysates were immunoblotted using anti-p- and t-insulin receptor (IR) and p- and t-AKT antibodies;
[0067]FIG. 11b shows an immunoblot of MCF-7, MDA-MB-231 and HEK293 cells transfected and stimulated with EGF. Cell lysates were immunoblotted using anti-phosphorylated (p) and total (t) ERK1/2 antibodies;
[0068]FIG. 12 shows immunohistochemical staining of normal pancreas cells and pancreatic carcinoma, at ×200 and ×400 magnification;
[0069]FIG. 13 shows a photograph of a colony formation assay, in which pancreatic carcinoma cell lines Colo357, MiaPaCa2, and Panc1 were transfected with either a HA-tagged klotho protein expression vector (pcDNA3-HA-KL) or an empty vector (pcDNA3) and colonies were stained with crystal violet;
[0070]FIG. 14 is a bar chart showing the results of an MTT proliferation assay;
[0071]FIG. 15 is a graph showing the effect of klotho protein on tumor size;
[0072]FIG. 16a is an immunoblot of Panel cells transfected with either klotho protein-HA expression plasmid (KL-HA) or a control vector (pcDNA3) using antibodies directed against phosphorylated (p) and total (t) IGF-1R, p- and t-AKT, p- and t-ERK1/2; and
[0073]FIG. 16b is an immunoblot of Panel cells treated with klotho protein and stimulated with IGF-1.
DESCRIPTION OF SOME EMBODIMENTS OF THE INVENTION
[0074]The present invention, in some embodiments, relates to the field of therapeutics, and more specifically to the use of klotho protein for the treatment and diagnosis of cancer. In some embodiments, the cancer is an IGF-1 dependent cancer, for example, colon cancer, prostate cancer, lung cancer, cervical cancer, ovarian cancer, but especially breast cancer and pancreatic cancer.
[0075]The klotho protein gene was identified as a potent suppressor of aging (Kuro-o et al., 1997). Mice homozygous for a mutated klotho protein allele exhibit a syndrome resembling human aging and suffer from osteoporosis, atherosclerosis and a short lifespan; while klotho protein over-expression extends lifespan and is associated with reduced fertility and insulin resistance (Kuro-o et al., 1997; Kurosu et al., 2005).
[0076]Anti-aging pharmaceutical compositions including klotho protein (human and murine) as well as DNA encoding this protein have been described in PCT/JP1997/004585 (published, inter alia, in EP 0 945 506 B1) which is included by reference as if fully set forth herein.
[0077]Compositions for reducing or increasing phosphorus and/or vitamin D level in blood, used for preventing and/or treating osteoporosis or rachitis, comprising klotho protein or anti-klotho protein antibody have been described in PCT/JP2004/006899 published as WO2004/100976.
[0078]Klotho protein (for example, SEQ ID NO: 1 from human kidney or SEQ ID NO: 3 from murine kidney) is a single pass transmembrane protein located at the cell membrane (Ito et al., 2000; Kuro-o et al., 1997; Matsumura et al., 1998; Shiraki-Iida et al., 1998) and also detected in the Golgi apparatus (Imura et al., 2007).
[0079]The extracellular domain of klotho protein is composed of two internal repeats, KL1 and KL2, which share amino acid sequence homology to β-glucosidase but lacks glucosidase catalytic activity (Kuro-o et al., 1997). The extracellular domain can be cleaved, shed into the serum and act as a circulating hormone (Imura et al., 2004). It has been shown recently that A Desintegrin and Metalloproteinase (ADAM) 10 and 17 participate in this process in response to insulin stimulation (Chen et al., 2007).
[0080]mRNA expression analysis identified klotho protein mainly in the distal renal tubules and the choroid plexus in the brain, but also in sex-hormone-responsive tissues including the placenta, testes and ovaries (Kuro-o et al., 1997; Ohyama et al., 1998; Shiraki-Iida et al., 1998). The expression of klotho protein in breast or pancreatic tissue has not hitherto been explored.
[0081]Klotho protein-modulated receptors include the insulin receptor (IR) (Kuro-o et al., 1997), the IGF-1 receptor (IGF-1R) and several fibroblast growth factor receptors (FGFR), but not the epidermal growth factor receptor (EGFR) (Kurosu et al., 2006; Urakawa et al., 2006). Treatment of cultured cells with soluble klotho protein inhibited insulin-induced glucose uptake, and reduced stimulation-induced phosphorylation of the IR, the IGF-1R and the insulin receptor substrates (IRS)-1 and -2 (Kurosu et al., 2005). Moreover, klotho protein-deficient mice are hypoglycemic and extremely sensitive to insulin (Kuro-o et al., 1997; Utsugi et al., 2000), while klotho protein over-expressing mice are associated with insulin resistance (Kurosu et al., 2005).
[0082]Klotho protein-induced inhibition of the IGF-1R may also affect the expression of the transcription factors CCAAT/enhancer-binding protein (C/EBP) α and β. These factors are down-regulated by the IGF-1 pathway and were recently identified as breast cancer growth suppressors (Gery et al., 2005; Gomis et al., 2006; Wolf et al., 2006a). The C/EBP family is involved in adipocyte differentiation and klotho protein-deficient mice have barely detected amounts of white adipose tissue (Kuro-o et al., 1997). Indeed, klotho protein has been identified recently as an inducer of adipocyte differentiation, and this activity is mediated through up-regulation of these transcription factors (Chihara et al., 2006).
[0083]Klotho protein may play a role in human diseases, and klotho protein polymorphism was associated with reduced life span, coronary heart disease and osteoporosis (Arking et al., 2003; Arking et al., 2002; Kim et al., 2006; Zarrabeitia et al., 2007).
[0084]Klotho protein has been shown to affect the activity of several signaling pathways, which may participate in breast cancer tumorigenesis, through modulation of ligand-dependent activation of their specific membranal receptors. Increased serum insulin levels are associated with adverse prognosis in breast cancer, high circulating IGF-1 levels are associated with increased risk of premenopausal breast cancer, and inhibition of the insulin and IGF-1 pathways inhibits growth of breast cancer cells (Bartucci et al., 2001; Wolf et al., 2006b; Yee, 2006).
[0085]Klotho protein expression and activities in breast cancer has never before been elucidated. As klotho protein is expressed in various hormone-responsive tissues, and as klotho protein activity is associated with inhibition of the IGF-1 and insulin pathways, it was considered by the present inventors that klotho protein may have tumor suppressor activities in breast cancer.
[0086]The present inventors have identified high klotho protein expression in normal breast tissue and low klotho protein expression in breast cancer; noted inhibition of breast cancer cell growth following over-expression of klotho protein, and growth enhancement of klotho protein-expressing cells following klotho protein knock-down; and revealed modulation of the IGF-1 and the insulin pathways by klotho protein. Taken together, the results suggest klotho protein as a novel breast cancer tumor suppressor.
[0087]Insulin like growth factor-1 (IGF-1) is also a powerful mediator of pancreatic cancer. Both IGF-1 and IGF-1 receptor (IGF-1R) are overexpressed in human pancreatic tumors as well as in pancreatic cancer cell lines (Bergmann, U., et al, 1995, Karna, E., et al, 2002). Blockage of the IGF-1R by a dominant negative inhibitor suppresses tumorigenicity both in vitro and in vivo and increase sensitivity of pancreatic tumors to radiation and chemotherapy-induced apoptosis (Edderkaoui, M., et al, 2007). Thus, the inventors consider that the IGF-1 pathway may serve as an attractive target for novel therapies against pancreatic cancer.
[0088]Klotho protein has previously been identified as an inhibitor of the IGF-1 system in hepatocytes and muscle cells. The effects of klotho protein on inhibition of the IGF-1 system in cancer cells, such as pancreatic and breast cancer cells, have not hitherto been studied.
[0089]The present inventors have found that klotho protein has an inhibitory effect on growth of cell lines, including breast cancer cell lines and pancreatic cancer cell lines. This is exceptionally surprising and entirely unexpected in light of the fact that Klotho protein has been demonstrated to have an anti-senescence and anti-apoptotic activity (see, inter alia,.
[0090]Immunohistochemistry analysis of klotho protein expression in both pancreatic cells and breast tissue arrays revealed high levels of klotho protein expression in normal breast samples and normal pancreatic cells, but very low expression in cancerous breast cells or cancerous pancreatic cells. In cancer samples, high klotho protein expression was associated with smaller tumor size and reduced K167 staining.
[0091]Forced expression of klotho protein reduced proliferation of MCF-7 and MDA-MB-231 breast cancer cells, while klotho protein silencing in MCF-7 cells, which normally express klotho protein, enhanced proliferation. Moreover, forced expression of klotho protein in these cells inhibited ligand-dependent IGF-1 and insulin pathways activation, and induced upregulation of the transcription factor CCAAT/enhancer-binding protein (C/EBP) β, a breast cancer growth inhibitor which is negatively regulated by the IGF-1-AKT axis. Co-immunoprecipitation revealed an interaction between klotho protein and the IGF-1 receptor. Klotho protein did not affect activation of the epidermal growth factor pathway.
[0092]Over-expression of klotho protein was similarly found to inhibit growth of pancreatic cell lines Panc1, Colo357 and Mia Paca, and to inhibit the IGF-1 pathway in Panc1 cells, by inhibition of phosphorylation of the IGF-1 receptor, and its downstream targets AKT1 and ERK1/2.
[0093]These data suggest klotho protein as a potential tumor suppressor and identify it as an inhibitor of the IGF-1 pathway, for example in human breast cancer or pancreatic cancer.
[0094]Insulin like growth factor-1 (IGF-1) is also a powerful mediator of other cancers, such as colon cancer, lung cancer, ovarian cancer, cervical cancer and prostate cancer. Thus, the data found by the Inventors suggest that klotho protein as a potential tumor suppressor in IGF-ldependent cancers in addition to human breast cancer or pancreatic cancer, for example colon cancer, lung cancer, ovarian cancer, cervical cancer and prostate cancer.
Treatment of Cancer
[0095]Thus, as discussed herein and as described in detail in the Examples section below, it has been surprisingly found by the present inventors that klotho protein is useful in affecting cancer cells and therefore in the treatment of cancer.
[0096]According to some embodiments, the present invention therefore provides use of klotho protein (including related compounds such as a fragment, an analog, a mimetic, a derivative or salt thereof) as a therapeutic agent for the treatment of cancer.
[0097]In some embodiments the present invention provides a method of treatment of cancer comprising administering to a subject in need thereof a pharmaceutically effective amount of a klotho protein.
[0098]In some embodiments, the present invention further provides the use of a composition comprising a pharmaceutically acceptable amount of a klotho protein for the treatment of cancer.
Types of Cancer
[0099]In some embodiments, the cancer is an IGF-1 dependent cancer. According to some aspects of embodiments of the present invention, the cancer is selected from the group consisting of breast cancer, pancreatic cancer, colon cancer, lung cancer, cervical cancer, ovarian cancer, and prostate cancer. According to some embodiments of the present invention, the cancer is breast cancer. According to some embodiments of the present invention, the cancer is pancreatic cancer.
[0100]According to some embodiments of the present invention, the cancer may be metastatic cancer.
Klotho Protein
[0101]The klotho protein may be any suitable klotho protein and may be made, isolated, and purified in any suitable fashion with which one skilled in the art, for example as described hereinbelow or in PCT/JP1997/004585 (published, inter alia, in EP 0 945 506 B1). Specific representative examples of klotho proteins suitable for use in implementing some embodiments of the teachings of the present invention include proteins described herein and in PCT/JP1997/004585
[0102]In some embodiments, a klotho protein used in implementing the teachings of the present invention is a complete, or substantially complete, klotho protein. Typical such complete klotho proteins are isolated polypeptides including the amino acid sequence selected from the group of amino acid sequences represented by SEQ ID NOS: 1, 3, 5, and 11. Although not wishing to be held to any one theory, it is believed that in some embodiments, a complete or substantially complete klotho protein that is administered to a subject is modified in the body of a subject (for example by cleavage of at least one peptide bond, shortening of the N-terminus and/or shortening of the C-terminus) and the thus-modified polypeptide provides the desired effect, e.g., treating cancer.
[0103]In some embodiments, a klotho protein used in implementing the teachings of the present invention is a cleaved extracellular portion of a complete klotho protein. Typical such extracellular portions of a klotho protein are isolated polypeptides including the amino acid sequence selected from the group of amino acid sequences represented by SEQ ID NOS: 2, 4, 12 and 13.
[0104]In some embodiments, a klotho protein used in implementing the teachings of the present invention is a portion of a complete klotho protein. Typical such portions of complete klotho proteins are isolated polypeptides including the amino acid sequence selected from the group of amino acid sequences represented by SEQ ID NOS: 14, 15 as well as solubilized klotho protein, see below.
[0105]In some embodiments, a klotho protein used in implementing the teachings of the present invention is a solubilized klotho protein, for example solubilized by removal of one or more amino acid residues from the C-terminus, the N-terminus or both the C-terminus and the N-terminus, as is known to one skilled in the art and as described, for example, in Kurosu et al., 2005. Typical such solubilized klotho proteins are isolated polypeptides including an amino acid sequence selected from the group of amino acid sequences represented by residues 31 to 982 of SEQ ID NO: 1 (see experimental section below), residues 34 to 1012 of SEQ ID NO: 1 (as disclosed in PCT/JP1997/004585), residues 34 to 549 of SEQ ID NO: 2 (as disclosed in PCT/JP1997/004585) and residues 36 to 1014 of SEQ ID NO: 3 (as disclosed in PCT/JP1997/004585).
[0106]In some embodiments, a klotho protein used in implementing the teachings of the present invention is an isolated polypeptides having a homology of at least 80% or more (e.g., at least 90%, at least 95% and even at least 98% homology) with a polypeptide including the amino acid sequence selected from the group of amino acid sequences represented by any of SEQ ID NOS: 1 to 5 or 11 to 15.
[0107]In some embodiments, a klotho protein used in implementing the teachings of the present invention is an isolated polypeptide comprising an amino acid sequence wherein at least one amino acid of the amino acid sequence of a polypeptide comprising an amino acid sequence selected from amino acid sequences represented by SEQ ID NOS: 1 to 5 or 11 to 15 is deleted, substituted or added.
[0108]It is important to note that in some embodiments it is preferred that a klotho protein administered to a subject be allogeneic (from the same species as the subject) as opposed to heterologous. For example, for treating cancer in humans, in some embodiments it is preferred to use a human klotho protein (e.g., SEQ ID NOS: 1, 2, 5 or 11 to 15). For example, for treating cancer in a mouse, in some embodiments it is preferred to use a murine klotho protein (e.g., SEQ ID NOS: 3 or 4).
[0109]According to some embodiments of the present invention, the klotho protein is a native protein, a derivative or analog thereof, or an active segment of the native protein.
[0110]According to some embodiments of the present invention, the klotho protein is a soluble or solublized form of klotho protein.
[0111]According to some embodiments, the Klotho protein is a pegylated klotho protein, for example, a protein substantially similar or identical to klotho proteins described herein that has been pegylated to improve pharmacokinetics or other parameters. Various advantages of pegylation and methods for pegylation of proteins such as klotho proteins are known in the art, see for example Ryan S M, Mantovani G, Wang X, Haddleton D M, Brayden D J "Advances in PEGylation of important biotech molecules: delivery aspects" in Expert Opin Drug Deliv. 2008, 5(4), 371-383.
[0112]Specific examples of klotho proteins suitable for implementing some embodiments of the teachings of the present invention are described herein with reference to the amino acid sequences in the appended sequence listing. The thus-described listed klotho proteins are summarized:
TABLE-US-00001 SEQ ID NO: species/source AA residues accession no. 1* human kidney 1012 AAQ41828 2* human kidney 549 AAQ41829 3* mouse kidney 1014 AAQ41830 4* mouse kidney 550 AAQ41831 5* human pancreas 1015 AAQ41832 11 human 1012 EAX08526 12 human 549 BAA24941 13 human 549 EAX08525 14 human 273 CAC94767 15 human 739 CAC94773 *fully described in PCT/JP1997/004585
Administration
[0113]The klotho protein may be administered by any suitable route, for example as described in PCT/JP1997/004585. For example, the klotho protein may be administered by parenteral (including intravenous, intradermal, intraperitoneal, intramuscular and subcutaneous) routes. Alternatively, the klotho protein may be administered by other delivery routes, including oral, nasal, buccal, sublingual, intra-tracheal, transdermal, transmucosal, and pulmonary. The klotho protein may be administered by continuous release or delivery, using, for example, an infusion pump, continuous infusion, controlled release formulations utilizing polymer, oil or water insoluble matrices.
[0114]Carriers or excipients known in the art can also be used to facilitate administration of the klotho protein. Examples of carriers and excipients include calcium carbonate, calcium phosphate, various sugars such as lactose, or types of starch, cellulose derivatives, gelatin, vegetable oils, polyethylene glycols and physiologically compatible solvents.
[0115]According to some embodiments, the klotho protein is administered in extended release form, which is capable of releasing the protein over a predetermined release period, such that a clinically effective plasma level of the klotho protein is maintained for at least 24 hours, such as at least 48 hours, at least 72 hours, at least one week, or at least one month.
[0116]According to some embodiments of the present invention, the klotho protein may be administered in combination with one or more chemotherapeutic agents, including, for example, alkylating agents, antimetabolites, anthracyclines, plant alkaloids, topoisomerase inhibitors, hormone receptor modulators, hormone level modulators, and other antitumour agents.
[0117]Examples of suitable alkylating agents include, without limitation, busulfan, carboplatin, carmustine, cisplatin, chloroambucil, cyclophosphamide, dacarbazine, hexamethylmelamine, ifosfamide, mechlorethamine, melphalan, oxoplatin, streptozocin, temozolomide, thiotepa, and uramustine.
[0118]Non-limiting examples of suitable antimetabolites include azathioprine, capecitabine, carmofur, cladribine, clofarabine, cytarabine, fludarabine, fluorouracil, gemcitabine, mercaptopurine, methotrexate, premetrexed, raltitrexed, tegafur, and tioguanine.
[0119]Suitable anthracyclines include, for example, daunorubicin, doxorubicin, epirubicin, idarubicin, mitoxantrone and valrubicin.
[0120]Examples of suitable plant alkaloids include docetaxel, paclitaxel, vinblastine, vincristine, vindesine, and vinorelbine. Examples of suitable topoisomerase inhibitors include include amsacrine, etoposide, etoposide phosphate, irinotecan, teniposide, and topotecan. Examples of suitable hormone receptor modulators include tamoxifen; and estrogen antagonists, such as faslodex. Examples of suitable hormone level modulators include aromatose inhibitors, such as letrozole, anastrazole and aromasin. Examples of other antitumor agents include dactinomycin, and other chemotherapeutic agents for treatment of obesity-related cancers, such as trastuzumab (herceptin), lapatinib, bevacizumab (avastin), cetuximab (erbitux), panitumumab, erlotinib, and sunitinib.
[0121]Preferably, the klotho protein and the chemotherapeutic agent are administered by subcutaneous or intravenous injection.
[0122]The chemotherapeutic agent may optionally be provided in a combined dosage form, together with the klotho protein. Alternatively, the chemotherapeutic agent may be provided in a separate dosage form, for co-administration or sequential administration, either before or after administration of the klotho protein.
[0123]The present invention further provides a composition comprising pharmaceutically acceptable amounts of a klotho protein and a chemotherapeutic agent as an additional active pharmaceutical ingredient.
[0124]The composition may optionally be provided in extended-release form, as described above with regard to the klotho protein alone.
[0125]According to some embodiments of the present invention, there is provided a composition comprising a combination of at least two active pharmaceutical ingredients, at least one of which is a klotho protein and at least one of which is a chemotherapeutic agent, wherein the amount of klotho protein and amount of the chemotherapeutic agent alone is insufficient to achieve the therapeutic effect achieved by the administration of the combination of two or more of the active pharmaceutical ingredients. The composition of the present invention comprises, in addition to the active pharmaceutical ingredients, a pharmaceutically acceptable carrier, and may optionally further comprise one or more pharmaceutically acceptable excipients.
[0126]Pharmaceutically acceptable carriers can be sterile liquids, such as water and oils, including those of petroleum, animal, vegetable or synthetic origin, such as peanut oil, soybean oil, mineral oil, sesame oil and the like. Water is a preferred carrier when the pharmaceutical composition is administered subcutaneously. Saline solutions and aqueous dextrose and glycerol solutions can also be employed as liquid carriers, particularly for injectable solutions.
[0127]If desired, solutions of the above dosage compositions may be thickened with a thickening agent such as methylcellulose. They may be prepared in emulsified form, such as either water in oil or oil in water. Any of a wide variety of pharmaceutically acceptable emulsifying agents may be employed including, for example, acacia powder, a non-ionic surfactant (such as a Tween), or an ionic surfactant (such as alkali polyether alcohol sulfates or sulfonates, e.g., a Triton).
[0128]In general, the composition of the present invention is prepared by mixing the ingredients following generally accepted procedures. For example, the selected components may be simply mixed in a blender or other standard device to produce a concentrated mixture which may then be adjusted to the final concentration and viscosity by the addition of water or thickening agent and possibly a buffer to control pH or an additional solute to control tonicity.
Treatment Using Gene Therapy
[0129]Discussed above were various embodiments of the present invention where a klotho protein, a polypeptide, was administered to a subject in order to treat cancer. In some embodiments, rather than administering a polypeptide, gene therapy is used, whereby a DNA encoding for a klotho protein is administered, in the usual way, to a subject. The thus-administered DNA causes the body of the subject to produce endogenous klotho protein in amounts sufficient to lead to the desired anti-cancer effect. In some embodiments, the DNA is delivered inserted in a recombinant vector (e.g., a bacteria) suitable for gene therapy.
[0130]Thus, according to some embodiments, the present invention therefore provides use of DNA-encoding for klotho protein (or related compounds) as a therapeutic agent for the treatment of cancer.
[0131]In some embodiments the present invention provides a method of treatment of cancer comprising administering to a subject in need thereof DNA encoding klotho protein or related compound, e.g., a fragment, an analog, a mimetic or a derivative thereof.
[0132]In some embodiments, the cancer is an IGF-1 dependent cancer. According to some aspects of embodiments of the present invention, the cancer is selected from the group consisting of breast cancer, pancreatic cancer, colon cancer, lung cancer, cervical cancer, ovarian cancer, and prostate cancer. According to some embodiments of the present invention, the cancer is breast cancer. According to some embodiments of the present invention, the cancer is pancreatic cancer.
[0133]According to some embodiments of the present invention, the cancer may be metastatic cancer.
[0134]The DNA encoding klotho protein may be a DNA for encoding any suitable klotho protein and may be made, isolated, purified, and provided in any suitable fashion with which one skilled in the art, for example as described in PCT/JP1997/004585 (published, inter alia, in EP 0 945 506 B1). For example, as is well known to one skilled in the art, a preferred way of providing a protein-encoding DNA, such as DNA encoding klotho protein, is by inserting the DNA in a recombinant vector, especially a recombinant vector suitable for gene therapy.
[0135]In some embodiments, the DNA encoding klotho protein is a DNA encoding a polypeptide comprising an amino acid sequence selected from the amino acid sequences represented by SEQ ID NOS: 1, 2, 3, 4 and 5 or isolated polypeptides having a homology of at least 80% or more therewith (e.g., at least 90%, at least 95% and even at least 98% homology therewith) or a DNA encoding a polypeptide comprising an amino acid sequence wherein at least one amino acid of the amino acid sequence of a polypeptide comprising an amino acid sequence selected from amino acid sequences represented by SEQ ID NOS: 1, 2, 3, 4 and 5 is deleted, substituted or added.
[0136]In some embodiments, the DNA encoding klotho protein is a DNA comprising a DNA sequence selected from DNA sequences represented by SEQ ID NOS: 6, 7, 8, 9 and 10.
[0137]In some embodiments, the DNA encoding klotho protein is a DNA which hybridizes with a DNA described above under stringent conditions. By "DNA which hybridizes under stringent conditions" is meant DNA obtained by colony hybridization, plaque hybridization or Southern blot hybridization using DNA encoding klotho protein, specifically including DNA identified after hybridization, using a filter on which colony- or plaque-derived DNA has been immobilized in the presence of 0.7 to 1.0 M NaCl at 65° C. and washing the resulting filter using 0.1 to 2×SSC solutions (the composition of 1×SSC solution comprises 150 mM sodium chloride and 15 mM sodium citrate) at 65° C. Hybridization can be carried out according to a method described, for example, in Molecular Cloning, A Laboratory Manual, the 2nd edition (Sambrook, Fritsch, & Maniatis eds., Cold Spring Harbor Laboratory Press, 1989). Specific examples of the DNA which hybridizes include DNA having a homology of 60% or more with a nucleotide sequence of the DNA encoding the polypeptide of an amino acid sequence selected from amino acid sequences represented by SEQ ID NOS: 1, 2, 3, 4 and 5, preferably DNA having a homology of 80% or more, and more preferably DNA having a homology of 95% or more.
[0138]It is important to note that in some embodiments it is preferred that the DNA administered to a subject encode allogeneic (from the same species as the subject) klotho protein, as opposed to heterologous klotho protein. For example, for treating cancer in humans, in some embodiments it is preferred to use a DNA encoding for human klotho protein (e.g., SEQ ID NOS: 6, 7 or 10). For example, for treating cancer in a mouse, in some embodiments it is preferred to use a DNA encoding for murine klotho protein (e.g., SEQ ID NOS: 8 or 9).
[0139]Specific examples of nucleic acids suitable for implementing some embodiments of the teachings of the present invention are described herein with reference to the nucleic acid sequences in the appended sequence listing. The thus-described listed nucleic acids are summarized:
TABLE-US-00002 encoding a polypeptide SEQ ID comprising an amino NO: acid sequence represented by: Accession nr. 6* SEQ ID NO: 1 AR343616 7* SEQ ID NO: 2 AR343617 8* SEQ ID NO: 3 AR343618 9* SEQ ID NO: 4 AR343619 10* SEQ ID NO: 5 AR343620 *fully described in PCT/JP1997/004585
[0140]The DNA encoding for klotho protein may be administered by any suitable route, for example as described in PCT/JP1997/004585. For example, in some embodiments the DNA is provided inserted in a recombinant vector (e.g., a bacteria), and the vector administered by parenteral routes.
[0141]According to some embodiments of the present invention, the DNA encoding for klotho protein may be administered in combination with one or more chemotherapeutic agents, as discussed above for administration of klotho protein as a polypeptide
[0142]As noted above, in some embodiments, the teachings of the present invention (e.g., administration of a klotho protein as a polypeptide or of DNA encoding therefore) are directed to treating cancer for example, to reduce or eliminate cancerous tumors and metastatic cells and tumors.
[0143]In some embodiments, the teachings of the present invention (e.g., administration of a klotho protein as a polypeptide or of DNA encoding therefore) are implemented to treat cancer as an adjuvant treatment, that is to say together with known modalities of cancer treatment.
[0144]In some embodiments, the teachings of the present invention (e.g., administration of a klotho protein as a polypeptide or of DNA encoding therefore) are implemented to treat cancer as a neo-adjuvant treatment, for example to reduce the size of a tumor prior to surgical excision thereof.
[0145]In some embodiments, the teachings of the present invention are implemented prophylactically (e.g., administration of a klotho protein as a polypeptide or of DNA encoding therefore). For example, in some embodiments, the present invention is implemented on a person who has not yet been diagnosed with cancer but is a member of a group at high risk of being diagnosed with cancer, for example has a genetic inclination to cancer (family history), a pathological indication of pre cancer (e.g., pre breast cancer), DCIS (ductal carcinoma in situ), clinically significant alcohol use, age or use of HRT (hormone replacement therapy). For example, in some embodiments, the present invention is implemented on a person whose cancer is in remission (complete or partial) but may be susceptible to a return of the disease.
Diagnosis
[0146]The present inventors have further surprisingly found that reduced expression of klotho protein correlates with increased tumor proliferative index, which provides an indication of the number of cells that are actively growing. A low proliferative index is associated with a slower growth rate.
[0147]In invasive ductal carcinomas, low klotho protein expression was associated with larger tumor size and higher expression of the cell proliferation marker, K167. Thus, it appears that klotho protein expression is down-regulated in the early stages of breast cancer development, such that low or decreased klotho protein expression may serve as a maker of breast cancer.
[0148]According to some embodiments of the present invention, there is provided a method for the diagnosis of cancer, comprising assaying the level of klotho protein present in a test sample obtained from a patient, and comparing the level of klotho protein to that of a reference sample, wherein a decreased level of klotho protein in the test sample, relative to that of the reference sample, is indicative of the presence of cancer in the patient.
[0149]According to some aspects of this embodiment of the present invention, the cancer is an IGF-1 dependent cancer, for example, breast cancer, pancreatic cancer, colon cancer, lung cancer, cervical cancer, ovarian cancer, or prostate cancer. According to some aspects of this embodiment of the present invention, the cancer is breast cancer or pancreatic cancer.
[0150]In embodiments, the test sample comprises a biopsy of tissue from a tumor, and the standard sample comprises a biopsy of normal tissue taken from an area adjacent to the tumor. Decreased klotho protein expression in the tumor tissue as opposed to the normal tissue indicates that the tumor is cancerous. For example, in embodiments wherein the cancer to be diagnosed is breast cancer, the test sample comprises a biopsy of tissue from a breast tumor, and the standard sample comprises a biopsy of normal breast tissue taken from an area adjacent to the breast tumor. Decreased klotho protein expression in the tumor tissue as opposed to the normal breast tissue indicates that the tumor is cancerous.
[0151]According to some embodiments of the present invention, there is provided a method of predicting the development of cancer in a benign breast disease patient, the method comprising measuring a level of klotho protein in a test sample obtained from a benign breast lesion; and comparing the level of klotho protein in the test sample to that of a reference sample comprising breast tissue from an area adjacent to the lesion; wherein a reduced level of klotho protein in the test sample relative to that of the reference sample is indicative of the presence of cancer in the patient or, in embodiments, is indicative of an increased risk for developing of cancer in the patient.
[0152]Exemplary embodiments of the invention are discussed hereinbelow with reference to specific materials, methods and examples. The material, methods and examples discussed herein are illustrative and not intended to be limiting. In some embodiments, methods and materials similar or equivalent to those described herein are used in the practice or testing of embodiments of the invention. It is to be understood that the invention is not necessarily limited in its application to the details of construction and the arrangement of the components and/or methods set forth in the following description and/or illustrated in the drawings. The invention is capable of other embodiments or of being practiced or carried out in various ways.
EXAMPLES
Materials and Methods
[0153]Chemicals, antibodies and constructs: Insulin and EGF were obtained from Sigma (St. Louis, Mo.). IGF-1 was obtained from PeproTech Inc (Rocky Hill, N.J.), and G418 from Invitrogen (Carlsbad, Calif.). Antibodies used in this study: anti-p53, -PARP, -IGF-1β receptor, and --C/EBPβ (Santa Cruz Biotechnology, Santa Cruz, Calif.), anti-phospho-AKT1 (S473), phospho-IGF-1R (Y1131), phospho-IR (Y1146) total pan-AKT, pGSK3β (S9) (Cell Signaling Technology, Danvers, Mass.), anti-diphosphorylated and -total ERK 1/2 (Sigma), phospho-IRS1 (Invitrogen), anti-HA (Covance, Princeton, N.J.). The klotho protein (for klotho having SEQ ID NO. 3) expression vector was a generous gift of Y. Nabeshima (Kyoto University, Japan).
[0154]siRNA: Klotho protein directed stealth siRNA duplex oligoribonucleotides and control siRNAs were both designed and purchased from Invitrogen. Lipofectamine 2000 (Invitrogen), and was used to transfect the cells with 100 pmoles of siRNA.
TABLE-US-00003 Si-1, directed against 1039-1063: ACCUUUCAUCUAUUCUGCCUGAUUU Si-2, directed against 1187-1211: CCUGAGGCAACUGCUUUCCUGGAUU Si-3, directed against 1274-1298: CACCAAGAGAGAUGAUGCCAAAUAU Si-C, scrambled: CACGAGAUAGAGUAGAACCAACUAU
[0155]Tissue arrays and immunohistochemistry analysis: Breast cancer tissue arrays were created, after IRB approval, from cancers diagnosed at Cedars-Sinai Medical Center from 1991 to 1998. Clinical parameters and follow-up information were obtained from hospital records. The tissue arrays were constructed using a manual arrayer (Beecher Instruments, MD) and contained three 1 mm samples for each specimen. For immunohistochemistry analysis, five-micron sections were cut from the tissue array. The slides were deparaffinized through xylenes and graded ethyl alcohols and then rinsed in water, followed by quenching of endogenous peroxidase activity by a 30% solution of hydrogen peroxidase in methanol for 10 minutes. Antigen retrieval was performed by boiling the slides in 0.0 μmol/L sodium citrate buffer pH 6.0 in a microwave oven at maximum power for 1 min and at 20% power for 9 minutes, followed by a 20 minutes cool down and rinses in wash buffer. The slides were then incubated for 1 hour with the anti-klotho protein antibody (Calbiochem, BD Biosciences, San Jose, Calif., 1:100 dilution) after blocking with normal serum for 30 minutes, reacted with the secondary antibody for 30 minutes and signal amplification and chromogen development were conducted for 30 minutes each. The stained slides were counterstained with hematoxylin and mounted. Antibody staining specificity was determined by staining kidney sections, and each run included appropriate positive and negative control slides. Staining was scored by percent of positive tumor cells and staining intensity.
[0156]Cells and transfections: Breast cancer cell lines were obtained from the American Type Culture Collection (Manassas, Va.). All transfections used LipofectAMINE 2000 (Invitrogen). Stable clones were generated by selection in complete culture medium containing 750 μg/ml G418.
[0157]Real Time Reverse Transcription-PCR(RT-PCR): Total RNA was extracted using the RNA isolation kit (Sigma) and processed to cDNA with RevertAid (Fermentas, Vilnius, Lithuania). Primers were designed using Primer Express (Applied Biosystems, Foster City, Calif.) and synthesized by IDT (Coralville, Iowa). Klotho protein specific primers were: Forward 5'-GCTCTCAAAGCCCACATACTG, reverse 5'-GCAGCATAACGATAGAGGCC.
[0158]Amplification reactions were performed with Platinum qPCR SuperMix (Invitrogen) in triplicates in an ABI Prism® 7000 (Applied Biosystems) as previously described (Wolf et al., 2007).
[0159]Western blot analysis: Cells were harvested and lysed for total protein extraction in RIPA buffer (50 mM Tris-Cl pH 7.4, 150 mM NaCl, 1% NP-40, 0.25% Na-deoxycholate, 1 mM EDTA, 1 mM NaF) together with a protease inhibitor cocktail (Sigma). 50 μg protein extracts were loaded on 10% polyacrylamide gels, separated electrophoretically and blotted from the gel onto nitrocellulose membrane (Schleicher & Schuell Bioscience GmbH, Dassel, DE). The membranes were then immunoblotted with the indicated antibodies.
[0160]Immunoprecipitation: Cells were harvested as for Western blot, and 400 μg protein lysate were incubated with IgG-Sepharose (Sigma) or protein A/G (Pierce, Rockford, Ill.) beads at 4° C. for 1 h to eliminate non-specifically bound proteins. These beads were subsequently used as a negative control. The supernatants were then incubated for 4 hours with either 2 μg anti-HA antibody (Sigma) or 2 μA of anti-IGF-1β-R (Santa Cruz Biotechnology) and IgG-Sepharose or protein A/G (Pierce, Rockford, Ill.), respectively, was added. Following an overnight incubation, the beads were washed 6 times in RIPA buffer and the immunoprecipitated materials were separated by SDS-PAGE and detected by Western blotting.
[0161]Colony assays: Two days following transfection with the indicated plasmids, G418 (750 μg/ml) was added to the culture media; and at day 14, the cells were stained using gentian violet. Untransfected cells were treated similarly, and all died within the 2 weeks of culture in the selection media. Quantification of the results was performed using AlphaImager 2000 (Alpha Innotech, CA).
[0162]3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT) viability assay: 7.5×103 cells/well were plated in 96-well plates, cultured in the appropriate culture media, and transfected with either control plasmid or klotho protein plasmid; and at indicated times, cells were cultured for two hours with 500 μg/ml MTT reagent (Sigma-Aldrich, St. Louis, Mo.). The medium was aspirated, and the cells were dissolved by dimethyl sulfoxide (DMSO). Absorbance of the formazan product was measured by an enzyme-linked immunosorbent assay reader.
[0163]Generation of soluble klotho protein: Soluble klotho protein was generated essentially as described (Kurosu et al., 2005). In brief, insect cells, which stably express V5-tagged soluble klotho protein, were generated using the Drosophila Expression System (Invitrogen). A cDNA fragment encoding the entire extracellular domain of mouse Klotho protein (amino acid residues 31 to 982 of SEQ ID NO: 3) was inserted into a pMT/BiP/V5-His expression vector, allowing inducible expression from metallothionein promoter and secretion of the recombinant protein with V5 nad 6×His tags at the C-terminus into conditioned medium. The expression vector was transfected into Drosophila Schneider cells by the calcium phosphate method together with pCoHYGRO selection vector. Stable transformants were selected with hygromycin B and cloned by limiting dilution to obtain clones with high expression levels. Expression of soluble Klotho protein was induced in serum-free medium containing 1 mM CuSO4 for 14 days. Klotho protein secreted in the conditioned medium was purified by affinity column chromatography using agarose affinity gel with anti-V5 antibodies (Sigma). Purified Klotho protein and known concentrations of albumin were subjected to SDS-PAGE followed by Sypro Ruby staining (Molecular Probe) to ascertain the concentration and purity of the protein. [0164]Statistical analysis: The study variables were compared between the study groups using Fisher's exact test for categorical variables. Pearson's correlation coefficient was used to determine the relationship between continuous variables.
I. Breast Cancer Studies
Klotho Protein Expression in Breast Tissue
[0165]Immunohistochemical analysis of klotho protein was conducted on breast tissue arrays consisting of 11 normal breast samples, 58 early stage invasive ductal carcinomas (IDC) and 47 pure ductal carcinomas in situ (DCIS) samples. Normal tissue adjacent to the IDC and DCIS was also analyzed. Human kidney sections, which are known to express high klotho protein levels only at the distal tubules (Kato et al., 2000), served as controls.
[0166]Clinical and pathological tumor characteristics of the 22 invasive breast cancer samples that showed klotho protein expression were compared to 36 invasive breast cancers that had no klotho protein expression. Results are shown in FIG. 1 and Table 1.
TABLE-US-00004 TABLE 1 Klotho protein expression in normal breast, DCIS* and invasive ductal carcinoma of the breast. Normal adjacent Normal Normal to adjacent to breast DCIS DCIS1 IDC2 IDC N = 11 N = 45 N = 47 N = 55 N = 58 N (%) N (%) N (%) N (%) N (%) Klotho absent 0 (0) 0 (0) 13 (28) 0 (0) 22 (38) protein low 0 (0) 4 (10) 26 (55) 5 (9) 23 (40) expression high 11 (100) 41 (90) 8 (17) 50 (91) 13 (22) DCIS, ductal carcinoma in situ; IDC, invasive ductal carcinoma. P < 0.0001 for comparisons of normal or adjacent normal samples to either DCIS or IDC samples
[0167]The relationship between klotho protein expression and tumor proliferative index was evaluated using K167 staining. The relationship between klotho protein expression and tumor size was also studied. Results are shown in Table 2.
TABLE-US-00005 TABLE 2 Distribution of study variables by klotho protein levels. No klotho protein klotho protein expression expression N = 22 N = 36 Overall P N (%) N (%) value Median Age (range) 52 (27-86) 51 (32-95) 0.48 Tumor size (T) 1 10 (45) 26 (72) 0.08 for 1 2 9 (41) 9 (25) vs 2-3 3 2 (9) 1 (3) Unknown 1 (5) 0 (0) Lymph nodes Negative 10 (45) 21 (58) 0.16 involvement (N) Positive 11 (50) 10 (28) Unknown 1 (5) 5 (14) Histology IDC 20 (91) 36 (100) 0.14 Other 2 (9) 0 (0) Grade 1-2 16 (73) 21 (58) 0.26 3 5 (22) 14 (39) Unknown 1 (5) 1 (3) ER Negative 2 (9) 8 (22) 0.3 Positive 16 (73) 24 (67) Unknown 4 (18) 4 (11) PR Negative 4 (18) 8 (22) 1 Positive 14 (64) 24 (67) Unknown 4 (18) 4 (11) KI67 1 6 (35) 20 (55) 0.04 2 4 (26) 0 (0) (1 vs 2, 3) 3 5 (13) 6 (18) Unknown 7 (26) 10 (22) IDC, invasive ductal carcinoma; ER, estrogen receptor; PR, progesterone receptor.
Klotho Protein Expression in Breast Cancer Cell Lines Using Real Time Polymerase Chain Reaction
[0168]The expression of klotho protein was analyzed in breast cancer cell lines (MCF-12A, MCF-7, MDA-MB-231, SK-BR3, T-47D, and BT-474), using real-time polymerase chain reaction (RT-PCR), which is used to amplify and simultaneously quantify a targeted DNA molecule. It enables both detection and quantification (as absolute number of copies or relative amount when normalized to DNA input or additional normalizing genes) of a specific sequence in a DNA sample.
[0169]The selected cell lines have different phenotypes and different expressions patterns of the estrogen receptor (ER), Her2 and p53, and thus represent various subclasses of breast cancer (Lacroix & Leclercq, 2004).
[0170]Results are shown in FIG. 2.
[0171]Effect of Over-Expression of Klotho Protein on Growth of Breast Cancer Cells
[0172]The effect of klotho protein on growth of breast cancer cells was first assessed using colony formation assays. MCF-7, MDA-MB-231 and the non-cancerous human embryonic kidney (HEK) 293 cells were transfected with either a HA-tagged klotho protein expression vector (pcDNA3-HA-KL) or an empty vector (pcDNA3). Transfected cells were cultured in media containing G418 for two weeks and stained to determine the number of surviving colonies.
[0173]In order to assess the long-term effects of klotho protein expression, MCF-7 and MDA-MB-231 clones, which stably expressed either pcDNA3 or pcDNA3-HA-KL, were generated. Proliferation was evaluated by 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT) assay after seeding for 24-72 h.
[0174]As some klotho protein expression was noted in MCF-7 cells (FIG. 2), the effects of down-regulation of klotho protein on these cells was studied. Three klotho protein-directed siRNAs were designed and tested for their ability to silence klotho protein expression in MCF-7 cells, compared to a control siRNA (siRNAc). Results are shown in FIGS. 4a and 4b.
Effect of Over-Expression of Klotho Protein on p53 Levels
[0175]In fibroblasts, klotho protein reduces senescence through down-regulation of p53 (de Oliveira, 2006). The effects of klotho protein on p53 levels in MCF-7 breast cancer cells and HEK 293 were therefore analyzed. Results are shown in FIG. 5.
Effect of Klotho Protein on IGF-1-Induced MCF-7Proliferation
[0176]Klotho protein is known to inhibit activation of the IGF-1 pathway in L6 myoblasts cells and rat hepatoma H4IIE cells (Kurosu et al., 2005). This pathway plays an important role in breast cancer tumorigenesis, and IGF-1 induces MCF-7 cell proliferation (Geier et al., 1995; Yee, 2006). In order to test the ability of klotho protein to modulate IGF-1-induced proliferation and survival, MCF-7 cells were transiently transfected with either pcDNA3 or pcDNA3-HA-KL, and grown in 0.5% serum with either IGF-1 or a control vehicle for 24-96 hr. Results are shown in FIG. 6.
Effect of Klotho Protein on Inhibition of Activation of the IGF-1/Insulin Pathway and Relationship with IGF-1R in Breast Cancer Cells
[0177]The effect of klotho protein was studied on IGF-1 pathway activation in MCF-7 breast cancer cells, which express high IGF-1R levels and showed an enhanced proliferation following IGF-1 treatment (Geier et al., 1995; Haimsohn et al., 2002). MDA-MB-231 cells, which express lower levels of these receptors, were also examined. HEK 293 cells, whose growth is not dependent on the IGF-1 pathway (El-Shewy et al., 2007) and is not inhibited by klotho protein, served as control.
[0178]In order to elucidate klotho protein activity as an inhibitor of IGF-1-induced activation, MCF-7 cells were transfected with either pcDNA3-HA-KL or an empty vector, starved for 48 hours, treated with IGF-1 for the indicated times and doses, and analyzed using Western blotting for the level of expression and phosphorylation of IGF-1R. Results are shown in FIGS. 7a and 7b.
[0179]In further experiments, MCF-7, MDA-MB-231 or HEK-293 cells were transfected with either pcDNA3-HA-KL or empty vector and treated with IGF-1 (12.5 nM, 15 min). Results are shown in FIG. 8.
[0180]The effect of silencing of klotho protein in MCF-7 cells, using siRNA3, was also studied. Results are shown in FIG. 9a.
[0181]As klotho protein may also be shed and act as a circulating hormone (Kuro-o et al., 1997; Matsumura et al., 1998), the ability of soluble klotho protein to inhibit activation of the IGF-1 pathway in MCF-7 cells was tested. V5-tagged soluble klotho protein was generated as described under "Materials and Methods", its expression and purity were examined using Coomassie staining and its concentration was determined (data not shown). MCF-7 cells were treated with klotho protein for 15 min and, stimulated with IGF-1 (12.5 nM, 15 min), and expression and phosphorylation of the IGF-1R was determined. Results are shown in FIG. 9b.
[0182]C/EBPβ is a down-stream target of the GSK3β pathway, which is suppressed following stimulation by IGF-1 (Li et al., 2007). It has tumor suppressor activities in breast cancer and is upregulated during klotho protein-induced adipocyte differentiation (Chihara et al., 2006). The effects of over-expression of klotho protein on C/EBPβ levels were therefore analyzed.
[0183]As klotho protein can bind FGFR and other membrane proteins, binding of klotho protein to the IGF-1R was examined. Co-immunoprecipitation assays were conducted using MCF-7 cells either stably (FIG. 10) or transiently (data not shown) expressing HA-klotho protein. HA-klotho protein was detected by Western blotting following immunoprecipitation of the IGF-1R, and the IGF-1R was detected following immunoprecipitation using HA-directed antibody.
Effect of Over-Expression of Klotho Protein on Insulin Stimulation and Activation of the EGF Pathway in Breast Cancer Cells
[0184]The effects of over-expression of klotho protein on the insulin-stimulated pathway were also examined. Results are shown in FIG. 11a and 11b. The EGF pathway, which plays an important role in the pathogenesis of breast cancer, is not modulated by klotho protein in either L6 or H4IIE cells (Kurosu et al., 2005). In order to evaluate the effects of klotho protein on this pathway in MCF-7, MDA-MB-231 and HEK293 breast cancer cells, these cells were transfected with either pcDNA3-HA-KL or an empty vector, starved for 48 hours, treated with EGF, and analyzed for the expression and activity of the ERK1/2, which are downstream targets of the EGF pathway.
II. Pancreatic Cancer Studies
Klotho Protein Expression in Pancreatic Cancer Cells
[0185]Tissue samples obtained from 3 normal pancreas and 12 pancreatic carcinoma were analyzed using immunohistochemical staining, at ×200 and ×400 magnification. Representative results are shown in FIG. 12a-12d.
[0186]Pancreatic carcinoma cell lines Colo357, MiaPaCa2, and Panc1 were also analyzed for klotho protein expression.
[0187]Effect of Over-Expression of Klotho Protein on Growth of Pancreatic Cancer Cells
[0188]The effect of over-expression of klotho protein on growth of pancreatic cancer cells was studied using both a colony formation assay and a 3-(4,5-Dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT) proliferation assay.
[0189]For the colony formation assay, pancreatic carcinoma cell lines Colo357, MiaPaCa2, and Panc1 were transfected with either a HA-tagged klotho protein expression vector (pcDNA3-HA-KL) or an empty vector (pcDNA3). Transfected cells were cultured in media containing G418 for two weeks. Colonies were stained with crystal violet and photographed. Results are shown in FIG. 13.
[0190]For the MTT proliferation assay, Panc1 cells (3×103/well) were plated in 96-well plates, cultured in the appropriate culture media containing 10% FCS, and treated with either soluble klotho protein (sol KL), the chemotherapeutic agent 5-Fluorouracil (5-FU), or a combination of sol KL and 5-FU. Medium alone served as control. 48 hr after incubation, cells were cultured for 2 hr with 500 μg/ml MTT reagent, the medium was aspirated, and the cells were dissolved in dimethyl sulfoxide (DMSO). Absorbance of the formazan product was measured by an enzyme-linked immunosorbent assay reader to provide a measure of viability. Results are shown in FIG. 14.
In Vivo Studies of the Effect of Klotho Protein on Pancreatic Cancer
[0191]Panc1 cells were harvested, washed twice with sterile PBS, counted and resuspended in Matrigel (BD Biosciences). Six-week-old female athymic nude mice were injected subcutaneously in both flanks with cells at a density of 0.5×106 cells/100 μl. The mice were treated with daily intra-peritoneal injections of either soluble klotho protein (prepared as described above): 200 ng/day, 500 ng/day, or vehicle control (saline). Five mice were used in each group. Tumor size was measured weekly with a linear caliper for up to 4 weeks, and tumor volume was estimated using the equation V=(a×b2)×0.5236, where "a" is the larger dimension and "b" the perpendicular diameter. Results are shown in FIG. 15.
Down-Regulation of the IGF-1 Pathway by Klotho Protein in Pancreatic Cancer Cell Lines.
[0192]Panc1 cells were transfected with either klotho protein-HA expression plasmid (KL-HA) or a control vector (pcDNA3). After 24 hrs, cells were serum starved for 48 hr and treated with IGF-1 (12.5 nM, 15 min). Following treatment, cells were harvested and proteins were resolved and immunoblotted using antibodies directed against phosphorylated (p) and total (t) IGF-1R, p- and t-AKT, p- and t-ERK1/2. Results are shown in FIG. 16a.
[0193]Since klotho protein is shed and may act as a circulating hormone, the ability of soluble klotho protein to inhibit activation of the IGF-1 pathway in Panc1 cells was tested. V5-tagged soluble klotho protein was generated as described [Kurosu H. et al., 2005]. Panc1 cells were treated with klotho protein for 10 min and stimulated with IGF-1 (12.5 nM, 15 min), and expression and phosphorylation of the IGF-1R, AKT and ERK1/2 was determined. Results are shown in FIG. 16b.
In Vivo Studies of the Effect of Klotho Protein on Various IGF-Dependent Cancers
[0194]Cells of various human IGF-1 dependent cancers are obtained in the usual way, counted and suspended in Matrigel (BD Biosciences). Six-week-old female athymic nude mice are injected subcutaneously in both flanks with cells at a usual density (e.g., 0.5×106 cells/100 μl). The mice are treated with daily intra-peritoneal injections of either a klotho protein (200 ng/day, 500 ng/day) or vehicle control (saline). Five mice are used in each group. Tumor size is measured weekly with a linear caliper for up to 4 weeks, and tumor volume is estimated using the equation V=(a×b2)×0.5236, where "a" is the larger dimension and "b" the perpendicular diameter. It is seen that the tumors in mice to which the saline control is administered have larger tumors than the mice to which klotho protein is administered.
[0195]As noted above, in these studies cancer cell representative of different IGF-1 dependent cancers are placed in mice to demonstrate the efficacy of the teachings of the present invention in treating a respective cancer in humans.
[0196]Amongst other cells, HCT116 cells are placed in mice and the mice treated as described above to demonstrate the efficacy of the teachings of the present invention in treating colon cancer in humans.
[0197]Amongst other cells, LanCap cells are placed in mice and the mice treated as described above to demonstrate the efficacy of the teachings of the present invention in treating prostate cancer in humans.
[0198]Amongst other cells, HeLa cells are placed in mice and the mice treated as described above to demonstrate the efficacy of the teachings of the present invention in treating cervical cancer in humans.
[0199]Amongst other cells, lung cancer cells (such as described in Noro R et al. BMC cancer 2006, 6, 277-289) are placed in mice and the mice treated as described above to demonstrate the efficacy of the teachings of the present invention in treating lung cancer in humans.
[0200]Amongst other cells, ovarian cancer cells (such as described in Masiakos P T et al. Clinical Cancer Research 1999, 5, 3488-3499) are placed in mice and the mice treated as described above to demonstrate the efficacy of the teachings of the present invention in treating ovarian cancer in humans.
[0201]The Klotho proteins used in the above studies are any of the Klotho proteins disclosed herein, including the isolated polypeptide sequences comprising an amino acid sequence represented by SEQ ID NOS: 1, 2, 3, 4, 5, 11, 12, 13, 14 and 15 and also an isolated polypeptide sequence comprising an amino acid sequence represented by residues 31 to 982 of SEQ ID NO: 1, residues 34 to 1012 of SEQ ID NO: 1, residues 34 to 549 of SEQ ID NO: 2 and residues 36 to 1014 of SEQ ID NO: 3 and also pegylated versions of the same.
Results
Klotho Protein Expression in Breast Cancer
[0202]Immunochemical staining showed that higher klotho protein expression was noted in all normal breast samples and in 90% of the normal breast samples adjacent to IDC or DCIS, compared to only 17% in DCIS and 22% in IDC (p, 0.0001, FIG. 1, Table 1)
[0203]A significant association between klotho protein expression and lower proliferative index, as evaluated by KI67 staining occurred (p=0.04, Table 2). A trend was also noted toward an association between no klotho protein expression and smaller tumor size (p=0.08, Table 2).
[0204]RT-PCR studies showed that high klotho protein expression was found in the non-malignant cell breast cell line MCF-12A, and in the well-differentiated MDF-7 cells, but that low klotho protein expression was found in the less differentiated cell lines MDA-MB-231, SK-BR3, and T-47D (FIG. 2).
[0205]Transfection assays of the MCF-7 and MDA-MB-231 breast cancer cell lines showed that transient klotho protein upregulation decreased clonal growth of these cells, as measured by colony assays, but did not affect the growth of human embryonic 293 cells. Stable overexpression of klotho protein significantly reduced breast cancer cell proliferation. Klotho protein expression reduced the number and size of surviving colonies of breast cancer cells by 84% and 72% (respectively), compared to empty vector-transfected controls (p<0.0005), but had no effect on HEK 293 growth (FIG. 3).
[0206]Proliferation as evaluated by MTT assay was up to 6-fold slower in klotho protein-expressing cells compared to the controls. Results for MCF-7 cells are shown in FIG. 3b.
[0207]Analyses of klotho protein mRNA levels following transfection with the various siRNAs revealed 88% reduction of klotho protein expression following transfection with siRNA3 (FIG. 4a). Proliferation of MCF-7 cells, as assessed by MTT assay, increased by 2.5-fold at 48 h following down-regulation of klotho protein using siRNA3, compared to transfection with siRNAc (p<0.05, FIG. 4b).
[0208]Over-expression of klotho protein was found to reduce p53 levels in HEK 293, but to increase p53 levels in MCF-7 cells (FIG. 5). No changes in expression or cleavage of the apoptosis associated protein polyADP-ribose polymerases (PARP) were noted following klotho protein over-expression of klotho protein in either cell line.
[0209]Klotho protein transfection completely inhibited IGF-1 induced cell proliferation in untreated MCF-7 cells, and this inhibition was only minimally restored following addition of IGF-1 to the cells. Thus, while IGF-1 increased by up to 40% cell-proliferation in control pcDNA3-transfected cells, cell proliferation in the HA-KL-transfected cells increased by only 25% (FIGS. 6a and 6b).
[0210]As shown in FIG. 7a, maximal activation of the IGF-1/insulin pathway occurred at 15 min of IGF-1 (12.5 nM) and this activation was inhibited in klotho protein over-expressing cells. Klotho protein over-expression in MCF-7 cells was associated with reduced phosphorylation not only of the IGF-1R, but also its downstream targets: IRS-1, AKT1, GSK313 and extracellular signal-regulated kinases (ERK)-1 and -2 (FIG. 7b). Smaller effects were noted in the MDA-MB-231 cells (data not shown), and only minor effects were noted in HEK 293 cells (FIG. 8).
[0211]An increased AKT phosphorylation following IGF-1 stimulation in siRNA3 transfected cells was noticed compared to siRNAc transfected cells (FIG. 9a).
[0212]The addition of soluble klotho protein inhibited phosphorylation of the IGF-1 receptor, as shown in FIG. 9b.
[0213]MCF-7 cells were also treated with a conditioned medium taken from klotho protein-over-expressing cells and found similar results, namely, inhibition of IGF-1 pathway activation (data not shown). These data suggest that the soluble form of klotho protein is a potent inhibitor of the IGF-1 pathway.
[0214]Analysis of the effects of over-expression of klotho protein on C/EBPβ levels showed significant up-regulation of C/EBPβ following over-expression of klotho protein in MCF-7 cells, and to a lesser extent in MDA-MB-231 cells, compared to cells transfected with an empty vector (FIG. 8).
[0215]Klotho protein inhibited the activation of the insulin pathway in MCF-7 cells but not HEK 293 cells (FIG. 11a). Over-expression of klotho protein affected neither the expression nor phosphorylation of ERK1/2, thus suggesting that the EGF pathway was not affected by over-expression of klotho protein (FIG. 11b).
Klotho Protein Expression in Pancreatic Cancer
[0216]As seen in FIGS. 12a and 12b, at ×200 and ×400 magnification, respectively, immunohistochemical staining revealed the presence of a high level of klotho protein expression in tissue obtained from a normal pancreas. In contrast, as seen in FIGS. 1c and 1d, very low levels of klotho protein expression were seen at ×200 and ×400 magnification, respectively, in tissue derived from a pancreatic carcinoma.
[0217]High klotho protein expression was noted in all normal pancreatic samples studied (n=3), but only in 2 out of 12 pancreatic cancer samples. 6 out of 12 carcinoma samples did not express detectable levels of klotho protein and 4 expressed only low levels of klotho protein (see Table 3 below).
[0218]Carcinoma cell lines Colo357 and MiaPaCa2 expressed high levels of klotho protein, while Panc1 expressed only low levels of klotho protein.
TABLE-US-00006 TABLE 3 Normal (n = 3) Carcinoma (n = 12) High klotho protein 3 2 (+) Weak klotho protein 0 4 (+/-) Undetectable (-) 0 6
[0219]FIG. 13 presents the results of a colony formation assay, which shows that klotho protein expression significantly reduced the number and size of surviving colonies, as compared to empty-vector transfected colonies.
[0220]As seen in FIG. 14, MTT proliferation assay showed that Panc1 cells showed a reduction in viability in response to soluble klotho protein. Treatment with 5 nM caused more than 40% reduction in viability. Moreover, the combination of soluble klotho protein and 5-FU (500 μM) showed additive effect: while 5-FU alone reduced viability by 50%, the combination reduced viability by 70%.
[0221]Animal studies showed that klotho protein treatment of mice resulted in a significant dose-response arrest of tumor growth, as seen in FIG. 15.
[0222]Klotho protein over-expression was found to be associated with reduced phosphorylation of the IGF-1R, and its downstream targets: AKT1 and ERK1/2, as shown in FIG. 16a. Similarly, the addition of soluble klotho protein inhibited phosphorylation of all three proteins, as shown in FIG. 16b.
[0223]Maximal activation occurred at 15 min of IGF-1 (12.5 nM) and this activation was inhibited in klotho protein over-expressing cells.
Discussion
[0224]The present inventors have identified, for the first time, tumor suppressor activities for klotho protein in breast cancer. High klotho protein expression was observed in normal breast, while low expression was noted in DCIS and invasive cancer, and re-introduction of klotho protein into breast cancer cells slowed their growth and was associated with inhibited activation of the insulin/IGF pathways and up-regulation of the tumor suppressor C/EBPβ.
[0225]Klotho protein is abundantly expressed in the distal convoluted renal tubules and in the choroids plexus in the brain. However, klotho protein is also expressed in various sex-hormone responsive organs including placenta, testis, prostate and ovary (Kuro-o et al., 1997). Thus, its expression in the normal mammary gland could be anticipated. The present analysis was conducted on a relatively large number of breast samples and corroborated staining specificity using well-established negative and positive controls. Moreover, a search of published breast cancer transcriptome profiles, using the Oncomine database (http://www.oncomine.org) further supported these findings and revealed reduced klotho protein expression in breast cancer, and an association between lower expression and adverse tumor characteristics.
[0226]The present observation of an association between low klotho protein expression and higher proliferation also indicate a tumor suppressor role for klotho protein in breast cancer. Very low expression of klotho protein was observed both in DCIS and invasive cancers, and preliminary observations indicate reduced klotho protein expression already in ductal hyperplasia (data not shown). These data suggest that loss of klotho protein expression is required in the early stages of breast cancer tumorigenesis. Thus, klotho protein expression pattern may serve as novel prognostic factor in breast cancer, and perhaps may be able to predict cancer development from pre-malignant lesions. However, a much larger number of samples is required in order to elucidate the prognostic significance of klotho protein expression.
[0227]Klotho protein is a type I transmembrane protein which localizes at the plasma membrane, as well as in the cytoplasm, where it dimerizes with Na+ K+-ATPase in the Golgi apparatus (Imura et al., 2007). In accordance with these data, immunostaining analysis revealed klotho protein expression both in the plasma membrane and the cytoplasm. However, klotho protein may also be shed and act as a circulating hormone (Chen et al., 2007).
[0228]The present inventors have identified inhibition of the IGF-1 pathway following either klotho protein over-expression in breast cancer cells, treatment of the cells with a soluble form of klotho protein or treatment of the cells with a condition medium taken from klotho protein over-expressing cells. Thus, klotho protein activities in the mammary gland may be mediated not only by expression within the cells but also through shedding of soluble klotho protein and a paracrine effect on surrounding cells. However, as klotho protein expression is lost only in DCIS or invasive cancers, and not in the adjacent normal mammary tissue, and as soluble klotho protein inhibits the growth of breast cancer cells in vitro, we hypothesize that even if a soluble form of klotho protein is excreted from the normal mammary cells, it either does not reach the tumor cells or is inactivated within the tumor, or its concentration is not sufficient to affect the nearby cancer cells.
[0229]Activity of the IGF-1R and the IR, and their down-stream signaling components, including IRS-1, Grb2, shc, PI-3K and AKT has been associated with breast cancer tumorigenesis. Moreover, specific IGF-1R inhibitors show anti-tumoral effects against breast cancer (Haluska et al., 2006; Mitsiades et al., 2004). Thus, loss of klotho protein-mediated inhibition of the insulin/IGF-1 pathways may serve as a novel mechanism involved in breast cancer tumorigenesis. Importantly, while klotho protein has been shown previously to inhibit activation of the IGF-1R without altering IGF-1R ability to bind IGF-1 (Kurosu et al., 2005), the present inventors have shown for the first time, a direct interaction between klotho protein and the IGF-1R. The mechanism by which klotho protein inhibits IGF-1 signaling however needs to be further explored.
[0230]Several lines of evidence suggest a growth inhibitory role for the transcription factor C/EBPβ in breast cancer tumorigenesis. C/EBPβ inhibits proliferation of breast cancer cells through up-regulation of 15-PGDH, the major prostaglandin catabolic enzyme, mediates TGF cytostatic response in metastatic breast cancer, and plays an important role in oncogene induced senescence (Gery et al., 2005; Gomis et al., 2006; Wolf et al., 2006a). Interestingly, C/EBPβ may serve as a link between klotho protein, the insulin/IGF-1 pathways and breast cancer growth inhibition. C/EBPβ is involved in klotho protein signaling and is up-regulated during klotho protein-induced adipocyte differentiation (Chihara et al., 2006). Moreover, the IGF-1 pathway can negatively regulate C/EBPβ by phosphorylation of GSK313 that leads to C/EBPβ down-regulation (Li et al., 2007). Our observation of C/EBPβ up-regulation following klotho protein over-expression suggests a role for the interaction between klotho protein, the IGF-1 pathway and C/EBPβ in breast cancer tumorigenesis.
[0231]A striking cell-type dependent klotho protein activity was observed. In MCF-7 cells, klotho protein reduces clonogenic proliferation, increases p53 expression, inhibits the insulin/IGF-1 pathways and increases the response to bFGF (data not shown), while the opposite effects are observed in the non-cancerous HEK 293 cells. Moreover, over-expression of klotho protein in vascular endothelial cells down-regulates p53 and reduces apoptosis and senescence (Ikushima et al., 2006), while its silencing in human primary fibroblasts up-regulates p53 and induces premature senescence (de Oliveira, 2006). Thus, the effects observed by the present inventors in breast cancer cells are opposite to the effects observed in various non-cancerous cells. Interestingly, differential growth inhibition has been shown recently following EGF pathway inhibition, which reduced breast cancer cell proliferation but enhanced myocyte growth (Spector et al., 2007). This phenomenon was attributed to the ability of normal myocytes to overcome stress by increasing their oxidative activity, compared to the cancer cells, which show only glycolytic activity. Perhaps similar mechanisms may also be involved in the differential response to over-expression of klotho protein and IGF-1 pathway inhibition. As klotho protein has been shown to play a role in cellular response to oxidative stress activities (Yamamoto et al., 2005), as well as in glucose metabolism (Utsugi et al., 2000), this hypothesis should be further explored.
[0232]In summary, the present inventors have identified klotho protein, for the first time, as a tumor suppressor gene in breast cancer and as a modulator of the IGF-1 pathway in breast cancer cells. As klotho protein is expressed in various sex hormone responsive organs, and as klotho protein may also serve as a circulating hormone, its role in the pathogenesis of other malignant diseases should also be explored.
[0233]It is appreciated that certain features of the invention, which are, for clarity, described in the context of separate embodiments, may also be provided in combination in a single embodiment. Conversely, various features of the invention, which are, for brevity, described in the context of a single embodiment, may also be provided separately or in any suitable subcombination or as suitable in any other described embodiment of the invention. Certain features described in the context of various embodiments are not to be considered essential features of those embodiments, unless the embodiment is inoperative without those elements.
[0234]Although the invention has been described in conjunction with specific embodiments thereof, it is evident that many alternatives, modifications and variations will be apparent to those skilled in the art. Accordingly, it is intended to embrace all such alternatives, modifications and variations that fall within the spirit and broad scope of the appended claims.
[0235]Citation or identification of any reference in this application shall not be construed as an admission that such reference is available as prior art to the present invention.
[0236]To the extent that section headings are used, they should not be construed as necessarily limiting.
REFERENCES
[0237]Arking D E, Becker D M, Yanek L R, Fallin D, Judge D P, Moy T F, et al. (2003). KLOTHO allele status and the risk of early-onset occult coronary artery disease. Am J Hum Genet. 72: 1154-1161. [0238]Arking D E, Krebsova A, Macek M Sr., Macek M Jr, Arking A, Mian I S, et al. (2002). Association of human aging with a functional variant of klotho. Proc Natl Acad Sci USA 99: 856-861. [0239]Bartucci M, Morelli C, Mauro L, Ando' S, Surmacz E. (2001). Differential Insulin-like Growth Factor I Receptor Signaling and Function in Estrogen Receptor (ER)-positive MCF-7 and ER-negative MDA-MB-231 Breast Cancer Cells. Cancer Res 61: 6747-6754. [0240]Bergmann U et al. Insulin-like growth factor I overexpression in human pancreatic cancer: evidence for autocrine and paracrine roles. Cancer Res. 1995 May 15; 55(10):2007-11. [0241]Chen C-D, Podvin S, Gillespie E. Leeman S E, Abraham C R. (2007). Insulin stimulates the cleavage and release of the extracellular domain of Klotho by ADAM10 and ADAM17. Proc Natl Acad Sci USA 104: 19796-19801. [0242]Chihara Y, Rakugi H, Ishikawa K, Ikushima M, Maekawa Y, Ohta J, et al. (2006). Klotho protein promotes adipocyte differentiation. Endocrinology 147: 3835-3842. [0243]de Oliveira R M (2006). Klotho RNAi induces premature senescence of human cells via a p53/p21 dependent pathway. FEBS Lett 580: 5753-5758. [0244]Edderkaoui M et al. Insulin-like growth factor-I receptor mediates the prosurvival effect of fibronectin. [0245]El-Shewy H M, Lee M-H, Obeid L M, Jaffa A A, Luttrell L M. (2007). The Insulin-like growth factor type 1 and insulin-like growth factor type 2/mannose-6-phosphate receptors independently regulate ERK1/2 activity in HEK293 cells. J Biol Chem 282: 26150-26157. [0246]Geier A, Beery R, Haimsohn M, Karasik A. (1995). Insulin-like growth factor-1 inhibits cell death induced by anticancer drugs in the MCF-7 cells: involvement of growth factors in drug resistance. Cancer Invest 13: 480-486. [0247]Gery S, Tanosaki S, Bose S, Bose N, Vadgama J, Koeffler H P. (2005). Down-regulation and growth inhibitory role of C/EBP {alpha} in breast cancer. Clin Cancer Res 11: 3184-3190. [0248]Gomis R R, Alarcon C, Nadal C, Van Poznak C, Massague J. (2006). C/EBP[beta] at the core of the TGF[beta] cytostatic response and its evasion in metastatic breast cancer cells. Cancer Cell 10: 203-214. [0249]Haimsohn M, Beery R, Karasik A, Kanety H, Geier A. (2002). Aurintricarboxylic acid induces a distinct activation of the IGF-I receptor signaling within MDA-231 Cells. Endocrinology 143: 837-845. [0250]Haluska P, Carboni J M, Loegering D A, Lee F Y, Wittman M, Saulnier M G, et al. (2006). In vitro and in vivo antitumor effects of the dual insulin-like growth factor-I/insulin receptor inhibitor, BMS-554417. Cancer Res 66: 362-371. [0251]Ikushima M, Rakugi H, Ishikawa K, Maekawa Y, Yamamoto K, Ohta J. (2006). Anti-apoptotic and anti-senescence effects of Klotho on vascular endothelial cells. Biochem Biophys Res Commun 339: 827-832. [0252]Imura A, Iwano A, Tohyama O, Tsuji Y, Nozaki K, Hashimoto N, et al. (2004). Secreted Klotho protein in sera and CSF: implication for post-translational cleavage in release of Klotho protein from cell membrane. FEBS Lett 565: 143-147. [0253]Imura A, Tsuji Y, Murata M, Maeda R, Kubota K, Iwano A, et al. (2007). {alpha}-Klotho as a Regulator of Calcium Homeostasis. Science 316: 1615-1618. [0254]Ito S, Kinoshita S, Shiraishi N, Nakagawa S, Sekine S, Fujimori T, et al. (2000). Molecular cloning and expression analyses of mouse [beta]klotho, which encodes a novel Klotho family protein. Mech Dev 98: 115-119. [0255]Karna E et al. Serum and tissue level of insulin-like growth factor-I (IGF-I) and IGF-I binding proteins as an index of pancreatitis and pancreatic cancer. Int J Exp Pathol. 2002 October; 83(5):239-45. [0256]Kato Y, Arakawa E, Kinoshita S, Shirai A, Furuya A, Yamano K, et al. (2000). Establishment of the anti-klotho monoclonal antibodies and detection of klotho protein in kidneys. Biochem Biophys Res Commun 267: 597-602. [0257]Kim Y, Kim J-H, Nam Y J, Kong M, Kim Y J, Yu K-H, et al. (2006). Klotho is a genetic risk factor for ischemic stroke caused by cardioembolism in Korean females. Neurosci Lett 407: 189-194. [0258]Kuro-o M, Matsumura Y, Aizawa H, Kawaguchi H, Suga T, Utsugi T, et al. (1997). Mutation of the mouse klotho gene leads to a syndrome resembling ageing. Nature 390: 45-51. [0259]Kurosu H, Ogawa Y, Miyoshi M, Yamamoto M, Nandi A, Rosenblatt K P, et al. (2006). Regulation of fibroblast growth factor-23 signaling by klotho. J Biol Chem 281: 6120-6123. [0260]Kurosu H et al. Suppression of aging in mice by the hormone Klotho. Science. 2005 Sep. 16; 309(5742):1829-33. J Biol. Chem. 2007 Sep. 14; 282(37):26646-55. [0261]Kurosu H, Yamamoto M, Clark J D, Pastor N, Nandi A, Gurnani P, et al. (2005). Suppression of aging in mice by the hormone klotho. Science 309: 1829-1833. [0262]Lacroix M, Leclercq G. (2004). Relevance of breast cancer cell lines as models for breast tumours: an update. Breast Cancer Res Treat 83: 249-289. [0263]Li X, Kim J W, Gronborg M, Urlaub H, Lane M D, Tang Q-Q. (2007). Role of cdk2 in the sequential phosphorylation/activation of C/EBPbeta during adipocyte differentiation. Proc Natl Acad Sci USA 104: 11597-11602. [0264]Matsumura Y, Aizawa H, Shiraki-Iida T, Nagai R, Kuro-o M, Nabeshima Y-i. (1998). Identification of the human klotho gene and its two transcripts encoding membrane and secreted klotho protein. Biochem Biophys Res Commun 242: 626-630. [0265]Mitsiades C S, Mitsiades N S, McMullan C J, Poulaki V, Shringarpure R, Akiyama M, et al. (2004). Inhibition of the insulin-like growth factor receptor-1 tyrosine kinase activity as a therapeutic strategy for multiple myeloma, other hematologic malignancies, and solid tumors. Cancer Cell 5: 221-230. [0266]Ohyama Y, Kurabayashi M, Masuda H, Nakamura T, Aihara Y, Kaname T, et al. (1998). Molecular cloning of rat klotho cDNA: markedly decreased expression of klotho by acute inflammatory stress. Biochem Biophys Res Commun 251: 920-925. [0267]Shiraki-Iida T, Aizawa H, Matsumura Y, Sekine S, Iida A, Anazawa H, et al. (1998). Structure of the mouse klotho gene and its two transcripts encoding membrane and secreted protein. FEBS Lett 424: 6-10. [0268]Spector N L, Yarden Y, Smith B, Lyass L, Trusk P, Pry K, et al. (2007). Activation of AMP-activated protein kinase by human EGF receptor 2/EGF receptor tyrosine kinase inhibitor protects cardiac cells. Proc Natl Acad Sci USA 104: 10607-10612. [0269]Urakawa I, Yamazaki Y, Shimada T, Iijima K, Hasegawa H, Okawa K, et al. (2006). Klotho converts canonical FGF receptor into a specific receptor for FGF23. Nature 444: 770-774. [0270]Utsugi T, Ohno T, Ohyama Y, Uchiyama T, Saito Y, Matsumura Y, et al. (2000). Decreased insulin production and increased insulin sensitivity in the klotho mutant mouse, a novel animal model for human aging. Metabolism 49: 1118-1123. [0271]Wolf I, Bose S, Williamson E A, Miller C W, Karlan B Y, Koeffler H P. (2007). FOXA1: Growth inhibitor and a favorable prognostic factor in human breast cancer. Int J Cancer 120: 1013-1022. [0272]Wolf I, O'Kelly J, Rubinek T, Tong M, Nguyen A, Lin B T, et al. (2006a). 15-Hydroxyprostaglandin dehydrogenase is a tumor suppressor of human breast cancer. Cancer Res 66: 7818-7823. [0273]Wolf I, Sadetzki S, Gluck I, Oberman B, Ben-David M, Papa M Z, et al. (2006b). Association between diabetes mellitus and adverse characteristics of breast cancer at presentation. Eur J Cancer 42: 1077-1082. [0274]Yamamoto M, Clark J D, Pastor J V, Gurnani P, Nandi A, Kurosu H, et al. (2005). Regulation of oxidative stress by the anti-aging hormone klotho. J Biol Chem 280: 38029-38034. [0275]Yee D. (2006). Targeting insulin-like growth factor pathways. Br J Cancer 94: 465-468. [0276]Zarrabeitia M, Hern indez J, Valero C, Zarrabeitia A, Ortiz F, Gonzalez-Macias J, et al. (2007). Klotho gene polymorphism and male bone mass. Calcif Tissue Int 80: 10-14.
Sequence CWU
1
SEQUENCE LISTING
<160> NUMBER OF SEQ ID NOS: 18
<210> SEQ ID NO 1
<211> LENGTH: 1012
<212> TYPE: PRT
<213> ORGANISM: homo sapiens
<300> PUBLICATION INFORMATION:
<302> TITLE: Polypeptide, Novel DNA and novel antibody
<308> DATABASE ACCESSION NUMBER: AAQ41828
<309> DATABASE ENTRY DATE: 2003-08-17
<310> PATENT DOCUMENT NUMBER: US 6,579,850
<311> PATENT FILING DATE: 1999-06-25
<312> PUBLICATION DATE: 2003-09-18
<313> RELEVANT RESIDUES IN SEQ ID NO: (1)..(1012)
<400> SEQUENCE: 1
Met Pro Ala Ser Ala Pro Pro Arg Arg Pro Arg Pro Pro Pro Pro Ser
1 5 10 15
Leu Ser Leu Leu Leu Val Leu Leu Gly Leu Gly Gly Arg Arg Leu Arg
20 25 30
Ala Glu Pro Gly Asp Gly Ala Gln Thr Trp Ala Arg Phe Ser Arg Pro
35 40 45
Pro Ala Pro Glu Ala Ala Gly Leu Phe Gln Gly Thr Phe Pro Asp Gly
50 55 60
Phe Leu Trp Ala Val Gly Ser Ala Ala Tyr Gln Thr Glu Gly Gly Trp
65 70 75 80
Gln Gln His Gly Lys Gly Ala Ser Ile Trp Asp Thr Phe Thr His His
85 90 95
Pro Leu Ala Pro Pro Gly Asp Ser Arg Asn Ala Ser Leu Pro Leu Gly
100 105 110
Ala Pro Ser Pro Leu Gln Pro Ala Thr Gly Asp Val Ala Ser Asp Ser
115 120 125
Tyr Asn Asn Val Phe Arg Asp Thr Glu Ala Leu Arg Glu Leu Gly Val
130 135 140
Thr His Tyr Arg Phe Ser Ile Ser Trp Ala Arg Val Leu Pro Asn Gly
145 150 155 160
Ser Ala Gly Val Pro Asn Arg Glu Gly Leu Arg Tyr Tyr Arg Arg Leu
165 170 175
Leu Glu Arg Leu Arg Glu Leu Gly Val Gln Pro Val Val Thr Leu Tyr
180 185 190
His Trp Asp Leu Pro Gln Arg Leu Gln Asp Ala Tyr Gly Gly Trp Ala
195 200 205
Asn Arg Ala Leu Ala Asp His Phe Arg Asp Tyr Ala Glu Leu Cys Phe
210 215 220
Arg His Phe Gly Gly Gln Val Lys Tyr Trp Ile Thr Ile Asp Asn Pro
225 230 235 240
Tyr Val Val Ala Trp His Gly Tyr Ala Thr Gly Arg Leu Ala Pro Gly
245 250 255
Ile Arg Gly Ser Pro Arg Leu Gly Tyr Leu Val Ala His Asn Leu Leu
260 265 270
Leu Ala His Ala Lys Val Trp His Leu Tyr Asn Thr Ser Phe Arg Pro
275 280 285
Thr Gln Gly Gly Gln Val Ser Ile Ala Leu Ser Ser His Trp Ile Asn
290 295 300
Pro Arg Arg Met Thr Asp His Ser Ile Lys Glu Cys Gln Lys Ser Leu
305 310 315 320
Asp Phe Val Leu Gly Trp Phe Ala Lys Pro Val Phe Ile Asp Gly Asp
325 330 335
Tyr Pro Glu Ser Met Lys Asn Asn Leu Ser Ser Ile Leu Pro Asp Phe
340 345 350
Thr Glu Ser Glu Lys Lys Phe Ile Lys Gly Thr Ala Asp Phe Phe Ala
355 360 365
Leu Cys Phe Gly Pro Thr Leu Ser Phe Gln Leu Leu Asp Pro His Met
370 375 380
Lys Phe Arg Gln Leu Glu Ser Pro Asn Leu Arg Gln Leu Leu Ser Trp
385 390 395 400
Ile Asp Leu Glu Phe Asn His Pro Gln Ile Phe Ile Val Glu Asn Gly
405 410 415
Trp Phe Val Ser Gly Thr Thr Lys Arg Asp Asp Ala Lys Tyr Met Tyr
420 425 430
Tyr Leu Lys Lys Phe Ile Met Glu Thr Leu Lys Ala Ile Lys Leu Asp
435 440 445
Gly Val Asp Val Ile Gly Tyr Thr Ala Trp Ser Leu Met Asp Gly Phe
450 455 460
Glu Trp His Arg Gly Tyr Ser Ile Arg Arg Gly Leu Phe Tyr Val Asp
465 470 475 480
Phe Leu Ser Gln Asp Lys Met Leu Leu Pro Lys Ser Ser Ala Leu Phe
485 490 495
Tyr Gln Lys Leu Ile Glu Lys Asn Gly Phe Pro Pro Leu Pro Glu Asn
500 505 510
Gln Pro Leu Glu Gly Thr Phe Pro Cys Asp Phe Ala Trp Gly Val Val
515 520 525
Asp Asn Tyr Ile Gln Val Asp Thr Thr Leu Ser Gln Phe Thr Asp Leu
530 535 540
Asn Val Tyr Leu Trp Asp Val His His Ser Lys Arg Leu Ile Lys Val
545 550 555 560
Asp Gly Val Val Thr Lys Lys Arg Lys Ser Tyr Cys Val Asp Phe Ala
565 570 575
Ala Ile Gln Pro Gln Ile Ala Leu Leu Gln Glu Met His Val Thr His
580 585 590
Phe Arg Phe Ser Leu Asp Trp Ala Leu Ile Leu Pro Leu Gly Asn Gln
595 600 605
Ser Gln Val Asn His Thr Ile Leu Gln Tyr Tyr Arg Cys Met Ala Ser
610 615 620
Glu Leu Val Arg Val Asn Ile Thr Pro Val Val Ala Leu Trp Gln Pro
625 630 635 640
Met Ala Pro Asn Gln Gly Leu Pro Arg Leu Leu Ala Arg Gln Gly Ala
645 650 655
Trp Glu Asn Pro Tyr Thr Ala Leu Ala Phe Ala Glu Tyr Ala Arg Leu
660 665 670
Cys Phe Gln Glu Leu Gly His His Val Lys Leu Trp Ile Thr Met Asn
675 680 685
Glu Pro Tyr Thr Arg Asn Met Thr Tyr Ser Ala Gly His Asn Leu Leu
690 695 700
Lys Ala His Ala Leu Ala Trp His Val Tyr Asn Glu Lys Phe Arg His
705 710 715 720
Ala Gln Asn Gly Lys Ile Ser Ile Ala Leu Gln Ala Asp Trp Ile Glu
725 730 735
Pro Ala Cys Pro Phe Ser Gln Lys Asp Lys Glu Val Ala Glu Arg Val
740 745 750
Leu Glu Phe Asp Ile Gly Trp Leu Ala Glu Pro Ile Phe Gly Ser Gly
755 760 765
Asp Tyr Pro Trp Val Met Arg Asp Trp Leu Asn Gln Arg Asn Asn Phe
770 775 780
Leu Leu Pro Tyr Phe Thr Glu Asp Glu Lys Lys Leu Ile Gln Gly Thr
785 790 795 800
Phe Asp Phe Leu Ala Leu Ser His Tyr Thr Thr Ile Leu Val Asp Ser
805 810 815
Glu Lys Glu Asp Pro Ile Lys Tyr Asn Asp Tyr Leu Glu Val Gln Glu
820 825 830
Met Thr Asp Ile Thr Trp Leu Asn Ser Pro Ser Gln Val Ala Val Val
835 840 845
Pro Trp Gly Leu Arg Lys Val Leu Asn Trp Leu Lys Phe Lys Tyr Gly
850 855 860
Asp Leu Pro Met Tyr Ile Ile Ser Asn Gly Ile Asp Asp Gly Leu His
865 870 875 880
Ala Glu Asp Asp Gln Leu Arg Val Tyr Tyr Met Gln Asn Tyr Ile Asn
885 890 895
Glu Ala Leu Lys Ala His Ile Leu Asp Gly Ile Asn Leu Cys Gly Tyr
900 905 910
Phe Ala Tyr Ser Phe Asn Asp Arg Thr Ala Pro Arg Phe Gly Leu Tyr
915 920 925
Arg Tyr Ala Ala Asp Gln Phe Glu Pro Lys Ala Ser Met Lys His Tyr
930 935 940
Arg Lys Ile Ile Asp Ser Asn Gly Phe Pro Gly Pro Glu Thr Leu Glu
945 950 955 960
Arg Phe Cys Pro Glu Glu Phe Thr Val Cys Thr Glu Cys Ser Phe Phe
965 970 975
His Thr Arg Lys Ser Leu Leu Ala Phe Ile Ala Phe Leu Phe Phe Ala
980 985 990
Ser Ile Ile Ser Leu Ser Leu Ile Phe Tyr Tyr Ser Lys Lys Gly Arg
995 1000 1005
Arg Ser Tyr Lys
1010
<210> SEQ ID NO 2
<211> LENGTH: 549
<212> TYPE: PRT
<213> ORGANISM: homo sapiens
<300> PUBLICATION INFORMATION:
<302> TITLE: Polypeptide, novel DNA and novel antibody
<308> DATABASE ACCESSION NUMBER: AAQ41829
<309> DATABASE ENTRY DATE: 2003-08-17
<310> PATENT DOCUMENT NUMBER: US 6,579,850
<311> PATENT FILING DATE: 1999-06-25
<312> PUBLICATION DATE: 2003-09-18
<313> RELEVANT RESIDUES IN SEQ ID NO: (1)..(549)
<400> SEQUENCE: 2
Met Pro Ala Ser Ala Pro Pro Arg Arg Pro Arg Pro Pro Pro Pro Ser
1 5 10 15
Leu Ser Leu Leu Leu Val Leu Leu Gly Leu Gly Gly Arg Arg Leu Arg
20 25 30
Ala Glu Pro Gly Asp Gly Ala Gln Thr Trp Ala Arg Phe Ser Arg Pro
35 40 45
Pro Ala Pro Glu Ala Ala Gly Leu Phe Gln Gly Thr Phe Pro Asp Gly
50 55 60
Phe Leu Trp Ala Val Gly Ser Ala Ala Tyr Gln Thr Glu Gly Gly Trp
65 70 75 80
Gln Gln His Gly Lys Gly Ala Ser Ile Trp Asp Thr Phe Thr His His
85 90 95
Pro Leu Ala Pro Pro Gly Asp Ser Arg Asn Ala Ser Leu Pro Leu Gly
100 105 110
Ala Pro Ser Pro Leu Gln Pro Ala Thr Gly Asp Val Ala Ser Asp Ser
115 120 125
Tyr Asn Asn Val Phe Arg Asp Thr Glu Ala Leu Arg Glu Leu Gly Val
130 135 140
Thr His Tyr Arg Phe Ser Ile Ser Trp Ala Arg Val Leu Pro Asn Gly
145 150 155 160
Ser Ala Gly Val Pro Asn Arg Glu Gly Leu Arg Tyr Tyr Arg Arg Leu
165 170 175
Leu Glu Arg Leu Arg Glu Leu Gly Val Gln Pro Val Val Thr Leu Tyr
180 185 190
His Trp Asp Leu Pro Gln Arg Leu Gln Asp Ala Tyr Gly Gly Trp Ala
195 200 205
Asn Arg Ala Leu Ala Asp His Phe Arg Asp Tyr Ala Glu Leu Cys Phe
210 215 220
Arg His Phe Gly Gly Gln Val Lys Tyr Trp Ile Thr Ile Asp Asn Pro
225 230 235 240
Tyr Val Val Ala Trp His Gly Tyr Ala Thr Gly Arg Leu Ala Pro Gly
245 250 255
Ile Arg Gly Ser Pro Arg Leu Gly Tyr Leu Val Ala His Asn Leu Leu
260 265 270
Leu Ala His Ala Lys Val Trp His Leu Tyr Asn Thr Ser Phe Arg Pro
275 280 285
Thr Gln Gly Gly Gln Val Ser Ile Ala Leu Ser Ser His Trp Ile Asn
290 295 300
Pro Arg Arg Met Thr Asp His Ser Ile Lys Glu Cys Gln Lys Ser Leu
305 310 315 320
Asp Phe Val Leu Gly Trp Phe Ala Lys Pro Val Phe Ile Asp Gly Asp
325 330 335
Tyr Pro Glu Ser Met Lys Asn Asn Leu Ser Ser Ile Leu Pro Asp Phe
340 345 350
Thr Glu Ser Glu Lys Lys Phe Ile Lys Gly Thr Ala Asp Phe Phe Ala
355 360 365
Leu Cys Phe Gly Pro Thr Leu Ser Phe Gln Leu Leu Asp Pro His Met
370 375 380
Lys Phe Arg Gln Leu Glu Ser Pro Asn Leu Arg Gln Leu Leu Ser Trp
385 390 395 400
Ile Asp Leu Glu Phe Asn His Pro Gln Ile Phe Ile Val Glu Asn Gly
405 410 415
Trp Phe Val Ser Gly Thr Thr Lys Arg Asp Asp Ala Lys Tyr Met Tyr
420 425 430
Tyr Leu Lys Lys Phe Ile Met Glu Thr Leu Lys Ala Ile Lys Leu Asp
435 440 445
Gly Val Asp Val Ile Gly Tyr Thr Ala Trp Ser Leu Met Asp Gly Phe
450 455 460
Glu Trp His Arg Gly Tyr Ser Ile Arg Arg Gly Leu Phe Tyr Val Asp
465 470 475 480
Phe Leu Ser Gln Asp Lys Met Leu Leu Pro Lys Ser Ser Ala Leu Phe
485 490 495
Tyr Gln Lys Leu Ile Glu Lys Asn Gly Phe Pro Pro Leu Pro Glu Asn
500 505 510
Gln Pro Leu Glu Gly Thr Phe Pro Cys Asp Phe Ala Trp Gly Val Val
515 520 525
Asp Asn Tyr Ile Gln Val Ser Gln Leu Thr Lys Pro Ile Ser Ser Leu
530 535 540
Thr Lys Pro Tyr His
545
<210> SEQ ID NO 3
<211> LENGTH: 1014
<212> TYPE: PRT
<213> ORGANISM: Mus musculus
<300> PUBLICATION INFORMATION:
<302> TITLE: Polypeptide, novel DNA and novel antibody
<308> DATABASE ACCESSION NUMBER: AAQ41830
<309> DATABASE ENTRY DATE: 2003-08-17
<310> PATENT DOCUMENT NUMBER: US 6,579,850
<311> PATENT FILING DATE: 1999-06-25
<312> PUBLICATION DATE: 2003-09-18
<313> RELEVANT RESIDUES IN SEQ ID NO: (1)..(1014)
<400> SEQUENCE: 3
Met Leu Ala Arg Ala Pro Pro Arg Arg Pro Pro Arg Leu Val Leu Leu
1 5 10 15
Arg Leu Leu Leu Leu His Leu Leu Leu Leu Ala Leu Arg Ala Arg Cys
20 25 30
Leu Ser Ala Glu Pro Gly Gln Gly Ala Gln Thr Trp Ala Arg Phe Ala
35 40 45
Arg Ala Pro Ala Pro Glu Ala Ala Gly Leu Leu His Asp Thr Phe Pro
50 55 60
Asp Gly Phe Leu Trp Ala Val Gly Ser Ala Ala Tyr Gln Thr Glu Gly
65 70 75 80
Gly Trp Arg Gln His Gly Lys Gly Ala Ser Ile Trp Asp Thr Phe Thr
85 90 95
His His Ser Gly Ala Ala Pro Ser Asp Ser Pro Ile Val Val Ala Pro
100 105 110
Ser Gly Ala Pro Ser Pro Pro Leu Ser Ser Thr Gly Asp Val Ala Ser
115 120 125
Asp Ser Tyr Asn Asn Val Tyr Arg Asp Thr Glu Gly Leu Arg Glu Leu
130 135 140
Gly Val Thr His Tyr Arg Phe Ser Ile Ser Trp Ala Arg Val Leu Pro
145 150 155 160
Asn Gly Thr Ala Gly Thr Pro Asn Arg Glu Gly Leu Arg Tyr Tyr Arg
165 170 175
Arg Leu Leu Glu Arg Leu Arg Glu Leu Gly Val Gln Pro Val Val Thr
180 185 190
Leu Tyr His Trp Asp Leu Pro Gln Arg Leu Gln Asp Thr Tyr Gly Gly
195 200 205
Trp Ala Asn Arg Ala Leu Ala Asp His Phe Arg Asp Tyr Ala Glu Leu
210 215 220
Cys Phe Arg His Phe Gly Gly Gln Val Lys Tyr Trp Ile Thr Ile Asp
225 230 235 240
Asn Pro Tyr Val Val Ala Trp His Gly Tyr Ala Thr Gly Arg Leu Ala
245 250 255
Pro Gly Val Arg Gly Ser Ser Arg Leu Gly Tyr Leu Val Ala His Asn
260 265 270
Leu Leu Leu Ala His Ala Lys Val Trp His Leu Tyr Asn Thr Ser Phe
275 280 285
Arg Pro Thr Gln Gly Gly Arg Val Ser Ile Ala Leu Ser Ser His Trp
290 295 300
Ile Asn Pro Arg Arg Met Thr Asp Tyr Asn Ile Arg Glu Cys Gln Lys
305 310 315 320
Ser Leu Asp Phe Val Leu Gly Trp Phe Ala Lys Pro Ile Phe Ile Asp
325 330 335
Gly Asp Tyr Pro Glu Ser Met Lys Asn Asn Leu Ser Ser Leu Leu Pro
340 345 350
Asp Phe Thr Glu Ser Glu Lys Arg Leu Ile Arg Gly Thr Ala Asp Phe
355 360 365
Phe Ala Leu Ser Phe Gly Pro Thr Leu Ser Phe Gln Leu Leu Asp Pro
370 375 380
Asn Met Lys Phe Arg Gln Leu Glu Ser Pro Asn Leu Arg Gln Leu Leu
385 390 395 400
Ser Trp Ile Asp Leu Glu Tyr Asn His Pro Pro Ile Phe Ile Val Glu
405 410 415
Asn Gly Trp Phe Val Ser Gly Thr Thr Lys Arg Asp Asp Ala Lys Tyr
420 425 430
Met Tyr Tyr Leu Lys Lys Phe Ile Met Glu Thr Leu Lys Ala Ile Arg
435 440 445
Leu Asp Gly Val Asp Val Ile Gly Tyr Thr Ala Trp Ser Leu Met Asp
450 455 460
Gly Phe Glu Trp His Arg Gly Tyr Ser Ile Arg Arg Gly Leu Phe Tyr
465 470 475 480
Val Asp Phe Leu Ser Gln Asp Lys Glu Leu Leu Pro Lys Ser Ser Ala
485 490 495
Leu Phe Tyr Gln Lys Leu Ile Glu Asp Asn Gly Phe Pro Pro Leu Pro
500 505 510
Glu Asn Gln Pro Leu Glu Gly Thr Phe Pro Cys Asp Phe Ala Trp Gly
515 520 525
Val Val Asp Asn Tyr Val Gln Val Asp Thr Thr Leu Ser Gln Phe Thr
530 535 540
Asp Pro Asn Val Tyr Leu Trp Asp Val His His Ser Lys Arg Leu Ile
545 550 555 560
Lys Val Asp Gly Val Val Ala Lys Lys Arg Lys Pro Tyr Cys Val Asp
565 570 575
Phe Ser Ala Ile Arg Pro Gln Ile Thr Leu Leu Arg Glu Met Arg Val
580 585 590
Thr His Phe Arg Phe Ser Leu Asp Trp Ala Leu Ile Leu Pro Leu Gly
595 600 605
Asn Gln Thr Gln Val Asn His Thr Val Leu His Phe Tyr Arg Cys Met
610 615 620
Ile Ser Glu Leu Val His Ala Asn Ile Thr Pro Val Val Ala Leu Trp
625 630 635 640
Gln Pro Ala Ala Pro His Gln Gly Leu Pro His Ala Leu Ala Lys His
645 650 655
Gly Ala Trp Glu Asn Pro His Thr Ala Leu Ala Phe Ala Asp Tyr Ala
660 665 670
Asn Leu Cys Phe Lys Glu Leu Gly His Trp Val Asn Leu Trp Ile Thr
675 680 685
Met Asn Glu Pro Asn Thr Arg Asn Met Thr Tyr Arg Ala Gly His His
690 695 700
Leu Leu Arg Ala His Ala Leu Ala Trp His Leu Tyr Asp Asp Lys Phe
705 710 715 720
Arg Ala Ala Gln Lys Gly Lys Ile Ser Ile Ala Leu Gln Ala Asp Trp
725 730 735
Ile Glu Pro Ala Cys Pro Phe Ser Gln Asn Asp Lys Glu Val Ala Glu
740 745 750
Arg Val Leu Glu Phe Asp Ile Gly Trp Leu Ala Glu Pro Ile Phe Gly
755 760 765
Ser Gly Asp Tyr Pro Arg Val Met Arg Asp Trp Leu Asn Gln Lys Asn
770 775 780
Asn Phe Leu Leu Pro Tyr Phe Thr Glu Asp Glu Lys Lys Leu Val Arg
785 790 795 800
Gly Ser Phe Asp Phe Leu Ala Val Ser His Tyr Thr Thr Ile Leu Val
805 810 815
Asp Trp Glu Lys Glu Asp Pro Met Lys Tyr Asn Asp Tyr Leu Glu Val
820 825 830
Gln Glu Met Thr Asp Ile Thr Trp Leu Asn Ser Pro Ser Gln Val Ala
835 840 845
Val Val Pro Trp Gly Leu Arg Lys Val Leu Asn Trp Leu Arg Phe Lys
850 855 860
Tyr Gly Asp Leu Pro Met Tyr Val Thr Ala Asn Gly Ile Asp Asp Asp
865 870 875 880
Pro His Ala Glu Gln Asp Ser Leu Arg Ile Tyr Tyr Ile Lys Asn Tyr
885 890 895
Val Asn Glu Ala Leu Lys Ala Tyr Val Leu Asp Asp Ile Asn Leu Cys
900 905 910
Gly Tyr Phe Ala Tyr Ser Leu Ser Asp Arg Ser Ala Pro Lys Ser Gly
915 920 925
Phe Tyr Arg Tyr Ala Ala Asn Gln Phe Glu Pro Lys Pro Ser Met Lys
930 935 940
His Tyr Arg Arg Ile Ile Asp Ser Asn Gly Phe Leu Gly Ser Gly Thr
945 950 955 960
Leu Gly Arg Phe Cys Pro Glu Glu Tyr Thr Val Cys Thr Glu Cys Gly
965 970 975
Phe Phe Gln Thr Arg Lys Ser Leu Leu Val Phe Ile Ser Phe Leu Val
980 985 990
Phe Thr Phe Ile Ile Ser Leu Ala Leu Ile Phe His Tyr Ser Lys Lys
995 1000 1005
Gly Gln Arg Ser Tyr Lys
1010
<210> SEQ ID NO 4
<211> LENGTH: 550
<212> TYPE: PRT
<213> ORGANISM: Mus musculus
<300> PUBLICATION INFORMATION:
<302> TITLE: Polypeptide, novel DNA and novel antibody
<308> DATABASE ACCESSION NUMBER: AAQ41831
<309> DATABASE ENTRY DATE: 2003-08-17
<310> PATENT DOCUMENT NUMBER: US 6,579,850
<311> PATENT FILING DATE: 1999-06-25
<312> PUBLICATION DATE: 2003-09-18
<313> RELEVANT RESIDUES IN SEQ ID NO: (1)..(550)
<400> SEQUENCE: 4
Met Leu Ala Arg Ala Pro Pro Arg Arg Pro Pro Arg Leu Val Leu Leu
1 5 10 15
Arg Leu Leu Leu Leu His Leu Leu Leu Leu Ala Leu Arg Ala Arg Cys
20 25 30
Leu Ser Ala Glu Pro Gly Gln Gly Ala Gln Thr Trp Ala Arg Phe Ala
35 40 45
Arg Ala Pro Ala Pro Glu Ala Ala Gly Leu Leu His Asp Thr Phe Pro
50 55 60
Asp Gly Phe Leu Trp Ala Val Gly Ser Ala Ala Tyr Gln Thr Glu Gly
65 70 75 80
Gly Trp Arg Gln His Gly Lys Gly Ala Ser Ile Trp Asp Thr Phe Thr
85 90 95
His His Ser Gly Ala Ala Pro Ser Asp Ser Pro Ile Val Val Ala Pro
100 105 110
Ser Gly Ala Pro Ser Pro Pro Leu Ser Ser Thr Gly Asp Val Ala Ser
115 120 125
Asp Ser Tyr Asn Asn Val Tyr Arg Asp Thr Glu Gly Leu Arg Glu Leu
130 135 140
Gly Val Thr His Tyr Arg Phe Ser Ile Ser Trp Ala Arg Val Leu Pro
145 150 155 160
Asn Gly Thr Ala Gly Thr Pro Asn Arg Glu Gly Leu Arg Tyr Tyr Arg
165 170 175
Arg Leu Leu Glu Arg Leu Arg Glu Leu Gly Val Gln Pro Val Val Thr
180 185 190
Leu Tyr His Trp Asp Leu Pro Gln Arg Leu Gln Asp Thr Tyr Gly Gly
195 200 205
Trp Ala Asn Arg Ala Leu Ala Asp His Phe Arg Asp Tyr Ala Glu Leu
210 215 220
Cys Phe Arg His Phe Gly Gly Gln Val Lys Tyr Trp Ile Thr Ile Asp
225 230 235 240
Asn Pro Tyr Val Val Ala Trp His Gly Tyr Ala Thr Gly Arg Leu Ala
245 250 255
Pro Gly Val Arg Gly Ser Ser Arg Leu Gly Tyr Leu Val Ala His Asn
260 265 270
Leu Leu Leu Ala His Ala Lys Val Trp His Leu Tyr Asn Thr Ser Phe
275 280 285
Arg Pro Thr Gln Gly Gly Arg Val Ser Ile Ala Leu Ser Ser His Trp
290 295 300
Ile Asn Pro Arg Arg Met Thr Asp Tyr Asn Ile Arg Glu Cys Gln Lys
305 310 315 320
Ser Leu Asp Phe Val Leu Gly Trp Phe Ala Lys Pro Ile Phe Ile Asp
325 330 335
Gly Asp Tyr Pro Glu Ser Met Lys Asn Asn Leu Ser Ser Leu Leu Pro
340 345 350
Asp Phe Thr Glu Ser Glu Lys Arg Leu Ile Arg Gly Thr Ala Asp Phe
355 360 365
Phe Ala Leu Cys Phe Gly Pro Thr Leu Ser Phe Gln Leu Leu Asp Pro
370 375 380
Asn Met Lys Phe Arg Gln Leu Glu Ser Pro Asn Leu Arg Gln Leu Leu
385 390 395 400
Ser Trp Ile Asp Leu Glu Tyr Asn His Pro Pro Ile Phe Ile Val Glu
405 410 415
Asn Gly Trp Phe Val Ser Gly Thr Thr Lys Arg Asp Asp Ala Lys Tyr
420 425 430
Met Tyr Tyr Leu Lys Lys Phe Ile Met Glu Thr Leu Lys Ala Ile Arg
435 440 445
Leu Asp Gly Val Asp Val Ile Gly Tyr Thr Ala Trp Ser Leu Met Asp
450 455 460
Gly Phe Glu Trp His Arg Gly Tyr Ser Ile Arg Arg Gly Leu Phe Tyr
465 470 475 480
Val Asp Phe Leu Ser Gln Asp Lys Glu Leu Leu Pro Lys Ser Ser Ala
485 490 495
Leu Phe Tyr Gln Lys Leu Ile Glu Asp Asn Gly Phe Pro Pro Leu Pro
500 505 510
Glu Asn Gln Pro Leu Glu Gly Thr Phe Pro Cys Asp Phe Ala Trp Gly
515 520 525
Val Val Asp Asn Tyr Val Gln Val Ser Pro Leu Thr Lys Pro Ser Val
530 535 540
Gly Leu Leu Leu Pro His
545 550
<210> SEQ ID NO 5
<211> LENGTH: 1015
<212> TYPE: PRT
<213> ORGANISM: homo sapiens
<300> PUBLICATION INFORMATION:
<302> TITLE: Polypeptide, novel DNA and novel antibody
<308> DATABASE ACCESSION NUMBER: AAQ41832
<309> DATABASE ENTRY DATE: 2003-08-17
<310> PATENT DOCUMENT NUMBER: US 6,579,850
<311> PATENT FILING DATE: 1999-06-25
<312> PUBLICATION DATE: 2003-09-18
<313> RELEVANT RESIDUES IN SEQ ID NO: (1)..(1015)
<400> SEQUENCE: 5
Met Ser Asn Gly Gly Leu Gln Arg Ser Val Ile Leu Ser Ala Leu Ile
1 5 10 15
Leu Leu Arg Ala Val Thr Gly Phe Ser Gly Asp Gly Arg Ala Ile Trp
20 25 30
Ser Lys Asn Pro Asn Phe Thr Pro Val Asn Glu Ser Gln Leu Phe Leu
35 40 45
Tyr Gly Thr Phe Pro Lys Asn Phe Phe Trp Gly Ile Gly Thr Gly Ala
50 55 60
Leu Gln Val Glu Gly Ser Trp Lys Lys Asp Gly Lys Gly Pro Ser Ile
65 70 75 80
Trp Asp His Phe Ile His Thr His Leu Lys Asn Val Ser Ser Thr Asn
85 90 95
Gly Ser Ser Asp Ser Tyr Ile Phe Leu Glu Lys Asp Leu Ser Ala Leu
100 105 110
Asp Phe Ile Gly Val Ser Phe Tyr Gln Phe Ser Ile Ser Trp Pro Arg
115 120 125
Leu Phe Pro Asp Gly Ile Val Thr Val Ala Asn Ala Lys Gly Leu Gln
130 135 140
Tyr Tyr Ser Thr Leu Leu Asp Ala Leu Val Leu Arg Asn Ile Glu Pro
145 150 155 160
Ile Val Thr Leu Tyr His Trp Asp Leu Pro Leu Ala Leu Gln Glu Lys
165 170 175
Tyr Gly Gly Trp Lys Asn Asp Thr Ile Ile Asp Ile Phe Asn Asp Tyr
180 185 190
Ala Thr Tyr Cys Phe Gln Met Phe Gly Asp Arg Val Lys Tyr Trp Ile
195 200 205
Thr Ile His Asn Pro Tyr Leu Val Ala Trp His Gly Tyr Gly Thr Gly
210 215 220
Met His Ala Pro Gly Glu Lys Gly Asn Leu Ala Ala Val Tyr Thr Val
225 230 235 240
Gly His Asn Leu Ile Lys Ala His Ser Lys Val Trp His Asn Tyr Asn
245 250 255
Thr His Phe Arg Pro His Gln Lys Gly Trp Leu Ser Ile Thr Leu Gly
260 265 270
Ser His Trp Ile Glu Pro Asn Arg Ser Glu Asn Thr Met Asp Ile Phe
275 280 285
Lys Cys Gln Gln Ser Met Val Ser Val Leu Gly Trp Phe Ala Asn Pro
290 295 300
Ile His Gly Asp Gly Asp Tyr Pro Glu Gly Met Arg Lys Lys Leu Phe
305 310 315 320
Ser Val Leu Pro Ile Phe Ser Glu Ala Glu Lys His Glu Met Arg Gly
325 330 335
Thr Ala Asp Phe Phe Ala Phe Ser Phe Gly Pro Asn Asn Phe Lys Pro
340 345 350
Leu Asn Thr Met Ala Lys Met Gly Gln Asn Val Ser Leu Asn Leu Arg
355 360 365
Glu Ala Leu Asn Trp Ile Lys Leu Glu Tyr Asn Asn Pro Arg Ile Leu
370 375 380
Ile Ala Glu Asn Gly Trp Phe Thr Asp Ser Arg Val Lys Thr Glu Asp
385 390 395 400
Thr Thr Ala Ile Tyr Met Met Lys Asn Phe Leu Ser Gln Val Leu Gln
405 410 415
Ala Ile Arg Leu Asp Glu Ile Arg Val Phe Gly Tyr Thr Ala Trp Ser
420 425 430
Leu Leu Asp Gly Phe Glu Trp Gln Asp Ala Tyr Thr Ile Arg Arg Gly
435 440 445
Leu Phe Tyr Val Asp Phe Asn Ser Lys Gln Lys Glu Arg Lys Pro Lys
450 455 460
Ser Ser Ala His Tyr Tyr Lys Gln Ile Ile Arg Glu Asn Gly Phe Ser
465 470 475 480
Leu Lys Glu Ser Thr Pro Asp Val Gln Gly Gln Phe Pro Cys Asp Phe
485 490 495
Ser Trp Gly Val Thr Glu Ser Val Leu Lys Pro Glu Ser Val Ala Ser
500 505 510
Ser Pro Gln Phe Ser Asp Pro His Leu Tyr Val Trp Asn Ala Thr Gly
515 520 525
Asn Arg Leu Leu His Arg Val Glu Gly Val Arg Leu Lys Thr Arg Pro
530 535 540
Ala Gln Cys Thr Asp Phe Val Asn Ile Lys Lys Gln Leu Glu Met Leu
545 550 555 560
Ala Arg Met Lys Val Thr His Tyr Arg Phe Ala Leu Asp Trp Ala Ser
565 570 575
Val Leu Pro Thr Gly Asn Leu Ser Ala Val Asn Arg Gln Ala Leu Arg
580 585 590
Tyr Tyr Arg Cys Val Val Ser Glu Gly Leu Lys Leu Gly Ile Ser Ala
595 600 605
Met Val Thr Leu Tyr Tyr Pro Thr His Ala His Leu Gly Leu Pro Glu
610 615 620
Pro Leu Leu His Ala Asp Gly Trp Leu Asn Pro Ser Thr Ala Glu Ala
625 630 635 640
Phe Gln Ala Tyr Ala Gly Leu Cys Phe Gln Glu Leu Gly Asp Leu Val
645 650 655
Lys Leu Trp Ile Thr Ile Asn Glu Pro Asn Arg Leu Ser Asp Ile Tyr
660 665 670
Asn Arg Ser Gly Asn Asp Thr Tyr Gly Ala Ala His Asn Leu Leu Val
675 680 685
Ala His Ala Leu Ala Trp Arg Leu Tyr Asp Gln Gln Phe Arg Pro Ser
690 695 700
Gln Arg Gly Ala Val Ser Leu Ser Leu His Ala Asp Trp Ala Glu Pro
705 710 715 720
Ala Asn Pro Tyr Ala Asp Ser His Trp Arg Ala Ala Glu Arg Phe Leu
725 730 735
Gln Phe Glu Ile Ala Trp Phe Ala Glu Pro Leu Phe Lys Thr Gly Asp
740 745 750
Tyr Pro Ala Ala Met Arg Glu Tyr Ile Ala Ser Lys His Arg Arg Gly
755 760 765
Leu Ser Ser Ser Ala Leu Pro Arg Leu Thr Glu Ala Glu Arg Arg Leu
770 775 780
Leu Lys Gly Thr Val Asp Phe Cys Ala Leu Asn His Phe Thr Thr Arg
785 790 795 800
Phe Val Met His Glu Gln Leu Ala Gly Ser Arg Tyr Asp Ser Asp Arg
805 810 815
Asp Ile Gln Phe Leu Gln Asp Ile Thr Arg Leu Ser Ser Pro Thr Arg
820 825 830
Leu Ala Val Ile Pro Trp Gly Val Arg Lys Leu Leu Arg Trp Val Arg
835 840 845
Arg Asn Tyr Gly Asp Met Asp Ile Tyr Ile Thr Ala Ser Gly Ile Asp
850 855 860
Asp Gln Ala Leu Glu Asp Asp Arg Leu Arg Lys Tyr Tyr Leu Gly Lys
865 870 875 880
Tyr Leu Gln Glu Val Leu Lys Ala Tyr Leu Ile Asp Lys Val Arg Ile
885 890 895
Lys Gly Tyr Tyr Ala Phe Lys Leu Ala Glu Glu Lys Ser Lys Pro Arg
900 905 910
Phe Gly Phe Phe Thr Ser Asp Phe Lys Ala Lys Ser Ser Ile Gln Phe
915 920 925
Tyr Asn Lys Val Ile Ser Ser Arg Gly Phe Pro Phe Glu Asn Ser Ser
930 935 940
Ser Arg Cys Ser Gln Thr Gln Glu Asn Thr Glu Cys Thr Val Cys Leu
945 950 955 960
Phe Leu Val Gln Lys Lys Pro Leu Ile Phe Leu Gly Cys Cys Phe Phe
965 970 975
Ser Thr Leu Val Leu Leu Leu Ser Ile Ala Ile Phe Gln Arg Gln Lys
980 985 990
Arg Arg Lys Phe Trp Lys Ala Lys Asn Leu Gln His Ile Pro Leu Lys
995 1000 1005
Lys Gly Lys Arg Val Val Ser
1010 1015
<210> SEQ ID NO 6
<211> LENGTH: 3163
<212> TYPE: DNA
<213> ORGANISM: homo sapiens
<300> PUBLICATION INFORMATION:
<302> TITLE: Polypeptide, novel DNA and novel antibody
<308> DATABASE ACCESSION NUMBER: AR343616
<309> DATABASE ENTRY DATE: 2003-08-17
<310> PATENT DOCUMENT NUMBER: US 6,579,850
<311> PATENT FILING DATE: 1999-06-25
<312> PUBLICATION DATE: 2003-09-18
<313> RELEVANT RESIDUES IN SEQ ID NO: (1)..(3163)
<400> SEQUENCE: 6
cgcgcagcat gcccgccagc gccccgccgc gccgcccgcg gccgccgccg ccgtcgctgt 60
cgctgctgct ggtgctgctg ggcctgggcg gccgccgcct gcgtgcggag ccgggcgacg 120
gcgcgcagac ctgggcccgt gtctcgcggc ctcctgcccc cgaggccgcg ggcctcttcc 180
agggcacctt ccccgacggc ttcctctggg ccgtgggcag cgccgcctac cagaccgagg 240
gcggctggca gcagcacggc aagggtgcgt ccatctggga cacgttcacc caccaccccc 300
tggcaccccc gggagactcc cggaacgcca gtctgccgtt gggcgccccg tcgccgctgc 360
agcccgccac cggggacgta gccagcgaca gctacaacaa cgtcttccgc gacacggagg 420
cgctgcgcga gctcggggtc actcactacc gcttctccat ctcgtgggcg cgagtgctcc 480
ccaatggcag cgcgggcgtc cccaaccgcg aggggctgcg ctactaccgg cgcctgctgg 540
agcggctgcg ggagctgggc gtgcagcccg tggtcaccct gtaccactgg gacctgcccc 600
agcgcctgca ggacgcctac ggcggctggg ccaaccgcgc cctggccgac cacttcaggg 660
attacgcgga gctctgcttc cgccacttcg gcggtcaggt caagtactgg atcaccatcg 720
acaaccccta cgtggtggcc tggcacggct acgccaccgg gcgcctggcc cccggcatcc 780
ggggcagccc gcggctcggg tacctggtgg cgcacaacct cctcctggct catgccaaag 840
tctggcatct ctacaatact tctttccgtc ccactcaggg aggtcaggtg tccattgccc 900
taagctctca ctggatcaat cctcgaagaa tgaccgacca cagcatcaaa gaatgtcaaa 960
aatctctgga ctttgtacta ggttggtttg ccaaacccgt atttattgat ggtgactatc 1020
ccgagagcat gaagaataac ctttcatcta ttctgcctga ttttactgaa tctgagaaaa 1080
agttcatcaa aggaactgct gacttttttg ctctttgctt tggacccacc ttgagttttc 1140
aacttttgga ccctcacatg aagttccgcc aattggaatc tcccaacctg aggcaactgc 1200
tttcctggat tgaccttgaa tttaaccatc ctcaaatatt tattgtggaa aatggctggt 1260
ttgtctcagg gaccaccaag agagatgatg ccaaatatat gtattacctc aaaaagttca 1320
tcatggaaac cttaaaagcc atcaagctgg atggggtgga tgtcatcggg tataccgcat 1380
ggtccctcat ggatggtttc gagtggcaca gaggttacag catcaggcgt ggactcttct 1440
atgttgactt tctaagccag gacaagatgt tgttgccaaa gtcttcagcc ttgttctacc 1500
aaaagctgat agagaaaaat ggcttccctc ctttacctga aaatcagccc ctagaaggga 1560
catttccctg tgactttgct tggggagttg ttgacaacta cattcaagta gataccactc 1620
tgtctcagtt taccgacctg aatgtttacc tgtgggatgt ccaccacagt aaaaggctta 1680
ttaaagtgga tggggttgtg accaagaaga ggaaatccta ctgtgttgac tttgctgcca 1740
tccagcccca gatcgcttta ctccaggaaa tgcacgttac acattttcgc ttctccctgg 1800
actgggccct gattctccct ctgggtaacc agtcccaggt gaaccacacc atcctgcagt 1860
actatcgctg catggccagc gagcttgtcc gtgtcaacat caccccagtg gtggccctgt 1920
ggcagcctat ggccccgaac caaggactgc cgcgcctcct ggccaggcag ggcgcctggg 1980
agaaccccta cactgccctg gcctttgcag agtatgcccg actgtgcttt caagagctcg 2040
gccatcacgt caagctttgg ataacgatga atgagccgta tacaaggaat atgacataca 2100
gtgctggcca caaccttctg aaggcccatg ccctggcttg gcatgtgtac aatgaaaagt 2160
ttaggcatgc tcagaatggg aaaatatcca tagccttgca ggctgattgg atagaacctg 2220
cctgcccttt ctcccaaaag gacaaagagg tggccgagag agttttggaa tttgacattg 2280
gctggctggc tgagcccatt ttcggctctg gagattatcc atgggtgatg agggactggc 2340
tgaaccaaag aaacaatttt cttcttcctt atttcactga agatgaaaaa aagctaatcc 2400
agggtacctt tgactttttg gctttaagcc attataccac catccttgta gactcagaaa 2460
aagaagatcc aataaaatac aatgattacc tagaagtgca agaaatgacc gacatcacgt 2520
ggctcaactc ccccagtcag gtggcggtag tgccctgggg gttgcgcaaa gtgctgaact 2580
ggctgaagtt caagtacgga gacctcccca tgtacataat atccaacgga atcgatgacg 2640
ggctgcatgc tgaggacgac cagctgaggg tgtattatat gcagaattac ataaacgaag 2700
ctctcaaagc ccacatactg gatggtatca atctttgcgg atactttgct tattcgttta 2760
acgaccgcac agctccgagg tttggcctct atcgttatgc tgcagatcag tttgagccca 2820
aggcatccat gaaacattac aggaaaatta ttgacagcaa tggtttcccg ggcccagaaa 2880
ctctggaaag attttgtcca gaagaattca ccgtgtgtac tgagtgcagt ttttttcaca 2940
cccgaaagtc tttactggct ttcatagctt ttctattttt tgcttctatt atttctctct 3000
cccttatatt ttactactcg aagaaaggca gaagaagtta caaatagttc tgaacatttt 3060
tctattcatt cattttgaaa taattatgca gacacatcag ctgttaacca tttgcacctc 3120
taagtgttgt gaaactgtaa atttcataca tttgacttct aga 3163
<210> SEQ ID NO 7
<211> LENGTH: 3435
<212> TYPE: DNA
<213> ORGANISM: homo sapiens
<300> PUBLICATION INFORMATION:
<302> TITLE: Polypeptide, novel DNA and novel antibody
<308> DATABASE ACCESSION NUMBER: AR343617
<309> DATABASE ENTRY DATE: 2003-08-17
<310> PATENT DOCUMENT NUMBER: US 6,579,850
<311> PATENT FILING DATE: 1999-06-25
<312> PUBLICATION DATE: 2003-09-18
<313> RELEVANT RESIDUES IN SEQ ID NO: (1)..(3435)
<400> SEQUENCE: 7
cgcgcagcat gcccgccagc gccccgccgc gccgcccgcg gccgccgccg ccgtcgctgt 60
cgctgctgct ggtgctgctg ggcctgggcg gccgccgcct gcgtgcggag ccgggcgacg 120
gcgcgcagac ctgggcccgt gtctcgcggc ctcctgcccc cgaggccgcg ggcctcttcc 180
agggcacctt ccccgacggc ttcctctggg ccgtgggcag cgccgcctac cagaccgagg 240
gcggctggca gcagcacggc aagggtgcgt ccatctggga cacgttcacc caccaccccc 300
tggcaccccc gggagactcc cggaacgcca gtctgccgtt gggcgccccg tcgccgctgc 360
agcccgccac cggggacgta gccagcgaca gctacaacaa cgtcttccgc gacacggagg 420
cgctgcgcga gctcggggtc actcactacc gcttctccat ctcgtgggcg cgagtgctcc 480
ccaatggcag cgcgggcgtc cccaaccgcg aggggctgcg ctactaccgg cgcctgctgg 540
agcggctgcg ggagctgggc gtgcagcccg tggtcaccct gtaccactgg gacctgcccc 600
agcgcctgca ggacgcctac ggcggctggg ccaaccgcgc cctggccgac cacttcaggg 660
attacgcgga gctctgcttc cgccacttcg gcggtcaggt caagtactgg atcaccatcg 720
acaaccccta cgtggtggcc tggcacggct acgccaccgg gcgcctggcc cccggcatcc 780
ggggcagccc gcggctcggg tacctggtgg cgcacaacct cctcctggct catgccaaag 840
tctggcatct ctacaatact tctttccgtc ccactcaggg aggtcaggtg tccattgccc 900
taagctctca ctggatcaat cctcgaagaa tgaccgacca cagcatcaaa gaatgtcaaa 960
aatctctgga ctttgtacta ggttggtttg ccaaacccgt atttattgat ggtgactatc 1020
ccgagagcat gaagaataac ctttcatcta ttctgcctga ttttactgaa tctgagaaaa 1080
agttcatcaa aggaactgct gacttttttg ctctttgctt tggacccacc ttgagttttc 1140
aacttttgga ccctcacatg aagttccgcc aattggaatc tcccaacctg aggcaactgc 1200
tttcctggat tgaccttgaa tttaaccatc ctcaaatatt tattgtggaa aatggctggt 1260
ttgtctcagg gaccaccaag agagatgatg ccaaatatat gtattacctc aaaaagttca 1320
tcatggaaac cttaaaagcc atcaagctgg atggggtgga tgtcatcggg tataccgcat 1380
ggtccctcat ggatggtttc gagtggcaca gaggttacag catcaggcgt ggactcttct 1440
atgttgactt tctaagccag gacaagatgt tgttgccaaa gtcttcagcc ttgttctacc 1500
aaaagctgat agagaaaaat ggcttccctc ctttacctga aaatcagccc ctagaaggga 1560
catttccctg tgactttgct tggggagttg ttgacaacta cattcaagta agtcagctga 1620
caaaaccaat cagcagtctc accaagccct atcactagta gataccactc tgtctcagtt 1680
taccgacctg aatgtttacc tgtgggatgt ccaccacagt aaaaggctta ttaaagtgga 1740
tggggttgtg accaagaaga ggaaatccta ctgtgttgac tttgctgcca tccagcccca 1800
gatcgcttta ctccaggaaa tgcacgttac acattttcgc ttctccctgg actgggccct 1860
gattctccct ctgggtaacc agtcccaggt gaaccacacc atcctgcagt actatcgctg 1920
catggccagc gagcttgtcc gtgtcaacat caccccagtg gtggccctgt ggcagcctat 1980
ggccccgaac caaggactgc cgcgcctcct ggccaggcag ggcgcctggg agaaccccta 2040
cactgccctg gcctttgcag agtatgcccg actgtgcttt caagagctcg gccatcacgt 2100
caagctttgg ataacgatga atgagccgta tacaaggaat atgacataca gtgctggcca 2160
caaccttctg aaggcccatg ccctggcttg gcatgtgtac aatgaaaagt ttaggcatgc 2220
tcagaatggg aaaatatcca tagccttgca ggctgattgg atagaacctg cctgcccttt 2280
ctcccaaaag gacaaagagg tggccgagag agttttggaa tttgacattg gctggctggc 2340
tgagcccatt ttcggctctg gagattatcc atgggtgatg agggactggc tgaaccaaag 2400
aaacaatttt cttcttcctt atttcactga agatgaaaaa aagctaatcc agggtacctt 2460
tgactttttg gctttaagcc attataccac catccttgta gactcagaaa aagaagatcc 2520
aataaaatac aatgattacc tagaagtgca agaaatgacc gacatcacgt ggctcaactc 2580
ccccagtcag gtggcggtag tgccctgggg gttgcgcaaa gtgctgaact ggctgaagtt 2640
caagtacgga gacctcccca tgtacataat atccaacgga atcgatgacg ggctgcatgc 2700
tgaggacgac cagctgaggg tgtattatat gcagaattac ataaacgaag ctctcaaagc 2760
ccacatactg gatggtatca atctttgcgg atactttgct tattcgttta acgaccgcac 2820
agctccgagg tttggcctct atcgttatgc tgcagatcag tttgagccca aggcatccat 2880
gaaacattac aggaaaatta ttgacagcaa tggtttcccg ggcccagaaa ctctggaaag 2940
attttgtcca gaagaattca ccgtgtgtac tgagtgcagt ttttttcaca cccgaaagtc 3000
tttactggct ttcatagctt ttctattttt tgcttctatt atttctctct cccttatatt 3060
ttactactcg aagaaaggca gaagaagtta caaatagttc tgaacatttt tctattcatt 3120
cattttgaaa taattatgca gacacatcag ctgttaacca tttgcacctc taagtgttgt 3180
gaaactgtaa atttcataca tttgacttct agaaaacatt tttgtggctt atgacagagg 3240
ttttgaaatg ggcataggtg atcgtaaaat attgaataat gcgaatagtg cctgaatttg 3300
ttctcttttt gggtgattaa aaaactgaca ggcactataa tttctgtaac acactaacaa 3360
aagcatgaaa aataggaacc acaccaatgc aacatttgtg cagaaatttg aatgacaaga 3420
ttaggaatat tttct 3435
<210> SEQ ID NO 8
<211> LENGTH: 5032
<212> TYPE: DNA
<213> ORGANISM: homo sapiens
<300> PUBLICATION INFORMATION:
<302> TITLE: Polypeptide, novel DNA and novel antibody
<308> DATABASE ACCESSION NUMBER: AR343618
<309> DATABASE ENTRY DATE: 2003-08-17
<310> PATENT DOCUMENT NUMBER: US 6,579,850
<311> PATENT FILING DATE: 1999-06-25
<312> PUBLICATION DATE: 2003-09-18
<313> RELEVANT RESIDUES IN SEQ ID NO: (1)..(5032)
<400> SEQUENCE: 8
cctcccggct cccgcagcat gctagcccgc gcccctcctc gccgcccgcc gcggctggtg 60
ctgctccgtt tgctgttgct gcatctgctg ctgctcgccc tgcgcgcccg ctgcctgagc 120
gctgagccgg gtcagggcgc gcagacctgg gctcgcttcg cgcgcgctcc tgccccagag 180
gccgctggcc tcctccacga caccttcccc gacggtttcc tctgggcggt aggcagcgcc 240
gcctatcaga ccgagggcgg ctggcgacag cacggcaaag gcgcgtccat ctgggacact 300
ttcacccatc actctggggc ggccccgtcc gactccccga tcgtcgtggc gccgtcgggt 360
gccccgtcgc ctcccctgtc ctccactgga gatgtggcca gcgatagtta caacaacgtc 420
taccgcgaca cagaggggct gcgcgaactg ggggtcaccc actaccgctt ctccatatcg 480
tgggcgcggg tgctccccaa tggcaccgcg ggcactccca accgcgaggg gctgcgctac 540
taccggcggc tgctggagcg gctgcgggag ctgggcgtgc agccggtggt taccctgtac 600
cattgggacc tgccacagcg cctgcaggac acctatggcg gatgggccaa tcgcgccctg 660
gccgaccatt tcagggatta tgccgagctc tgcttccgcc acttcggtgg tcaggtcaag 720
tactggatca ccattgacaa cccctacgtg gtggcctggc acgggtatgc caccgggcgc 780
ctggccccgg gcgtgagggg cagctccagg ctcgggtacc tggttgccca caacctactt 840
ttggctcatg ccaaagtctg gcatctctac aacacctctt tccgccccac acagggaggc 900
cgggtgtcta tcgccttaag ctcccattgg atcaatcctc gaagaatgac tgactataat 960
atcagagaat gccagaagtc tcttgacttt gtgctaggct ggtttgccaa acccatattt 1020
attgatggcg actacccaga gagtatgaag aacaacctct cgtctcttct gcctgatttt 1080
actgaatctg agaagaggct catcagagga actgctgact tttttgctct ctccttcgga 1140
ccaaccttga gctttcagct attggaccct aacatgaagt tccgccaatt ggagtctccc 1200
aacctgaggc agcttctgtc ttggatagat ctggaatata accaccctcc aatatttatt 1260
gtggaaaatg gctggtttgt ctcgggaacc accaaaaggg atgatgccaa atatatgtat 1320
tatctcaaga agttcataat ggaaacctta aaagcaatca gactggatgg ggtcgacgtc 1380
attgggtaca ccgcgtggtc gctcatggac ggtttcgagt ggcatagggg ctacagcatc 1440
cggcgaggac tcttctacgt tgactttctg agtcaggaca aggagctgtt gccaaagtct 1500
tcggccttgt tctaccaaaa gctgatagag gacaatggct ttcctccttt acctgaaaac 1560
cagccccttg aagggacatt tccctgtgac tttgcttggg gagttgttga caactacgtt 1620
caagtggaca ctactctctc tcagtttact gacccgaatg tctatctgtg ggatgtgcat 1680
cacagtaaga ggcttattaa agtagacggg gttgtagcca agaagagaaa accttactgt 1740
gttgatttct ctgccatccg gcctcagata accttacttc gagaaatgcg ggtcacccac 1800
tttcgcttct ccctggactg ggccctgatc ttgcctctgg gtaaccagac ccaagtgaac 1860
cacacggttc tgcacttcta ccgctgcatg atcagcgagc tggtgcacgc caacatcact 1920
ccagtggtgg ccctgtggca gccagcagcc ccgcaccaag gcctgccaca tgcccttgca 1980
aaacatgggg cctgggagaa cccgcacact gctctggcgt ttgcagacta cgcaaacctg 2040
tgttttaaag agttgggtca ctgggtcaat ctctggatca ccatgaacga gccaaacaca 2100
cggaacatga cctatcgtgc cgggcaccac ctcctgagag cccatgcctt ggcttggcat 2160
ctgtacgatg acaagtttag ggcggctcag aaaggcaaaa tatccatcgc cttgcaggct 2220
gactggatag aaccggcctg ccctttctct caaaatgaca aagaagtggc cgagagagtt 2280
ttggaatttg atataggctg gctggcagag cctatttttg gttccggaga ttatccacgt 2340
gtgatgaggg actggctgaa ccaaaaaaac aattttcttt tgccctattt caccgaagat 2400
gaaaaaaagc tagtccgggg ttcctttgac ttcctggcgg tgagtcatta caccaccatt 2460
ctggtagact gggaaaagga ggatccgatg aaatacaacg attacttgga ggtacaggag 2520
atgactgaca tcacatggct caactctccc agtcaggtgg cagtggtgcc ttgggggctg 2580
cgcaaagtgc tcaactggct aaggttcaag tacggagacc tcccgatgta tgtgacagcc 2640
aatggaatcg atgatgaccc ccacgccgag caagactcac tgaggatcta ttatattaag 2700
aattatgtga atgaggctct gaaagcctac gtgttggacg acatcaacct ttgtggctac 2760
tttgcgtatt cacttagtga tcgctcagct cccaagtctg gcttttatcg atatgctgcg 2820
aatcagtttg agcccaaacc atctatgaaa cattacagga gaattattga cagcaatggc 2880
ttcctgggtt ctggaacact gggaaggttt tgtccagaag aatacactgt gtgcaccgaa 2940
tgtggatttt ttcaaacccg gaagtctttg ctggtcttca tctcgtttct tgtttttact 3000
tttattattt ctcttgctct catttttcac tactccaaga aaggccagag aagttataag 3060
taatgtgaac gtctgcctgg ccattcgctt tgggatcaag atgtacacgc cgtcagccgt 3120
ttgcacctct ctgtgttgtg agccgcattc cacacatttc gattctagaa aacccttttt 3180
gtcatgggtg gtagaggttt taaacaggaa ttggtgagaa taaaatattg cagggtgaat 3240
ggtatctgaa tctgctctct ttggtggcaa ttacggaatt atactcacca cagtttctac 3300
agtgccccgg aatggaaggc atagaatacg gtagggataa cagtgccaag cagacagaag 3360
tttaaagaac aactttaggg acttgtttat ccatggccat ttttaaattc actcctgttg 3420
gggagtaaca ctctctcaat taccatctta acacctggac tttacctgat ccagttttac 3480
aaggtgaagt agaaaaatat ccagtaaagg tggccaagag ccctgagtcc agagcagccc 3540
attaagaagc actattccta ccaaatgctg ctaatgtcaa tttacaaata tacttagaaa 3600
gcacattatg gacatttgta ttcttgtgaa tgtttttgag gtgtgcccta aaccccagat 3660
ccttgagggc tttctcttac caactttcct ttcagagcct gcttgttgga gattcttccc 3720
cagccccctt cccctttccc tcttgctctg ccccacctcg ctccacccag cttgctccag 3780
cccaaagatt ctttatttgt ttctcattac cgaaggttgt gagccaccat gtggtttctg 3840
ggatttgaac tcatgacctc cggaggagct gtcatgctct taaccagccc atgttgaaga 3900
ttcttttgat aaatattcac aaaaaataaa gatgagccat gagctgttgg cctcttcgga 3960
agcggaaact gagtgatttg attgaacatc cttttatctt tgaccagacc ttggaatgaa 4020
tgcaatgacc tttcccacag gaagaaggag gagctctcag tcaaactgta aagaatgcct 4080
cttcagaata tgctgtcagt gcttggatgc catgatgttc aactttctta gtcgatccgg 4140
cagcaatcac agtgtgagca cactgggaac ctgtccttgc ggccgccgag atctaccgtg 4200
tgcttctgtg aagaggcttt gacgtagccc ctctttgagc tcttacacca tgctactgac 4260
ttctagaaag gctaattagg tcttcttcta cacctaatac cctaagtctt actgactctc 4320
acgggagaag tctctgtgct acacctgagt ggtcttattg ataaccctga taccagatca 4380
ggcaagataa atccgtcata gcaggcatgg ctacccttgc tgccacaggg tcacagcaca 4440
tagctcatca ccctgttatt cttcatcttg caatgtggta tggttttcct ggtgaatgat 4500
cagcttttgc tgtggtattc tttatacatc tggacttatt attgaaatca aatgctatag 4560
aatcaatagt ttattttatg tctatttttc ttgatcgcag agtaatatat attaattgta 4620
aaaaatttaa gaaacaaaaa ctatatgtaa agaaaaaatt ataatataat acagagatgc 4680
tgctgacagt tcctatgtgt tgtgttttgt atactgagat catgtgatac gtaggcatac 4740
atcttcttgg gtttttttgt ttttgttttt tgttttgttt tgttttgttt tggttttttg 4800
agatagggtt tctctgtata gccctggctg tcctggaact cactttgcag accaggctag 4860
cctcaaactc ttattcattt ttactgaagt aatttttctg tcattagtct tcaagagcaa 4920
aactttaata gttatggaga atattgccag aacagctcaa aactgtttta tttgttggtc 4980
caatttccca ttaattagtt caataataaa tatcatttag aaataaaaaa aa 5032
<210> SEQ ID NO 9
<211> LENGTH: 1650
<212> TYPE: DNA
<213> ORGANISM: homo sapiens
<300> PUBLICATION INFORMATION:
<302> TITLE: Polypeptide, novel DNA and novel antibody
<308> DATABASE ACCESSION NUMBER: AR343619
<309> DATABASE ENTRY DATE: 2003-08-17
<310> PATENT DOCUMENT NUMBER: US 6,579,850
<311> PATENT FILING DATE: 1999-06-25
<312> PUBLICATION DATE: 2003-09-18
<313> RELEVANT RESIDUES IN SEQ ID NO: (1)..(1650)
<400> SEQUENCE: 9
atgctagccc gcgcccctcc tcgccgcccg ccgcggctgg tgctgctccg tttgctgttg 60
ctgcatctgc tgctgctcgc cctgcgcgcc cgctgcctga gcgctgagcc gggtcagggc 120
gcgcagacct gggctcgctt cgcgcgcgct cctgccccag aggccgctgg cctcctccac 180
gacaccttcc ccgacggttt cctctgggcg gtaggcagcg ccgcctatca gaccgagggc 240
ggctggcgac agcacggcaa aggcgcgtcc atctgggaca ctttcaccca tcactctggg 300
gcggccccgt ccgactcccc gatcgtcgtg gcgccgtcgg gtgccccgtc gcctcccctg 360
tcctccactg gagatgtggc cagcgatagt tacaacaacg tctaccgcga cacagagggg 420
ctgcgcgaac tgggggtcac ccactaccgc ttctccatat cgtgggcgcg ggtgctcccc 480
aatggcaccg cgggcactcc caaccgcgag gggctgcgct actaccggcg gctgctggag 540
cggctgcggg agctgggcgt gcagccggtg gttaccctgt accattggga cctgccacag 600
cgcctgcagg acacctatgg cggatgggcc aatcgcgccc tggccgacca tttcagggat 660
tatgccgagc tctgcttccg ccacttcggt ggtcaggtca agtactggat caccattgac 720
aacccctacg tggtggcctg gcacgggtat gccaccgggc gcctggcccc gggcgtgagg 780
ggcagctcca ggctcgggta cctggttgcc cacaacctac ttttggctca tgccaaagtc 840
tggcatctct acaacacctc tttccgcccc acacagggag gccgggtgtc tatcgcctta 900
agctcccatt ggatcaatcc tcgaagaatg actgactata atatcagaga atgccagaag 960
tctcttgact ttgtgctagg ctggtttgcc aaacccatat ttattgatgg cgactaccca 1020
gagagtatga agaacaacct ctcgtctctt ctgcctgatt ttactgaatc tgagaagagg 1080
ctcatcagag gaactgctga cttttttgct ctctgcttcg gaccaacctt gagctttcag 1140
ctattggacc ctaacatgaa gttccgccaa ttggagtctc ccaacctgag gcagcttcts 1200
tcttggatag atctggaata taaccaccct ccaatattta ttgtggaaaa tggctggttt 1260
gtctcgggaa ccaccaaaag ggatgatgcc aaatatatgt attatctcaa gaagttcata 1320
atggaaacct taaaagcaat cagactggat ggggtcgacg tcattgggta caccgcgtgg 1380
tcgctcatgg acggtttcga gtggcatagg ggctacagca tccggcgagg actcttctac 1440
gttgactttc tgagtcagga caaggagctg ttgccaaagt cttcggcctt gttctaccaa 1500
aagctgatag aggacaatgg ctttcctcct ttacctgaaa accagcccct tgaagggaca 1560
tttccctgtg actttgcttg gggagttgtt gacaactacg tacaagtaag tcctttgaca 1620
aaacccagtg tcggcctctt gcttcctcac 1650
<210> SEQ ID NO 10
<211> LENGTH: 3460
<212> TYPE: DNA
<213> ORGANISM: homo sapiens
<300> PUBLICATION INFORMATION:
<302> TITLE: Polypeptide, novel DNA and novel antibody
<308> DATABASE ACCESSION NUMBER: AR343620
<309> DATABASE ENTRY DATE: 2003-08-17
<310> PATENT DOCUMENT NUMBER: US 6,579,850
<311> PATENT FILING DATE: 1999-06-25
<312> PUBLICATION DATE: 2003-09-18
<313> RELEVANT RESIDUES IN SEQ ID NO: (1)..(3460)
<400> SEQUENCE: 10
cagggaatga atggattttc ttcagcactg atgaaataac cacacgctat aggaatacaa 60
tgtccaacgg gggattgcaa agatctgtca tcctgtcagc acttattctg ctacgagctg 120
ttactggatt ctctggagat ggaagagcta tatggtctaa aaatcctaat tttactccgg 180
taaatgaaag tcagctgttt ctctatggca ctttccctaa aaactttttc tggggtattg 240
ggactggagc attgcaagtg gaagggagtt ggaagaagga tggaaaagga ccttctatat 300
gggatcattt catccacaca caccttaaaa atgtcagcag cacgaatggt tccagtgaca 360
gttatatttt tctggaaaaa gacttatcag ccctggattt tataggagtt tctttttatc 420
aattttcaat ttcctggcca aggcttttcc ccgatggaat agtaacagtt gccaacgcaa 480
aaggtctgca gtactacagt actcttctgg acgctctagt gcttagaaac attgaaccta 540
tagttacttt ataccactgg gatttgcctt tggcactaca agaaaaatat ggggggtgga 600
aaaatgatac cataatagat atcttcaatg actatgccac atactgtttc cagatgtttg 660
gggaccgtgt caaatattgg attacaattc acaacccata tctagtggct tggcatgggt 720
atgggacagg tatgcatgcc cctggagaga agggaaattt agcagctgtc tacactgtgg 780
gacacaactt gatcaaggct cactcgaaag tttggcataa ctacaacaca catttccgcc 840
cacatcagaa gggttggtta tcgatcacgt tgggatctca ttggatcgag ccaaaccggt 900
cggaaaacac gatggatata ttcaaatgtc aacaatccat ggtttctgtg cttggatggt 960
ttgccaaccc tatccatggg gatggcgact atccagaggg gatgagaaag aagttgttct 1020
ccgttctacc cattttctct gaagcagaga agcatgagat gagaggcaca gctgatttct 1080
ttgccttttc ttttggaccc aacaacttca agcccctaaa caccatggct aaaatgggac 1140
aaaatgtttc acttaattta agagaagcgc tgaactggat taaactggaa tacaacaacc 1200
ctcgaatctt gattgctgag aatggctggt tcacagacag tcgtgtgaaa acagaagaca 1260
ccacggccat ctacatgatg aagaatttcc tcagccaggt gcttcaagca ataaggttag 1320
atgaaatacg agtgtttggt tatactgcct ggtctctcct ggatggcttt gaatggcagg 1380
atgcttacac catccgccga ggattatttt atgtggattt taacagtaaa cagaaagagc 1440
ggaaacctaa gtcttcagca cactactaca aacagatcat acgagaaaat ggtttttctt 1500
taaaagagtc cacgccagat gtgcagggcc agtttccctg tgacttctcc tggggtgtca 1560
ctgaatctgt tcttaagccc gagtctgtgg cttcgtcccc acagttcagc gatcctcatc 1620
tgtacgtgtg gaacgccact ggcaacagac tgttgcaccg agtggaaggg gtgaggctga 1680
aaacacgacc cgctcaatgc acagattttg taaacatcaa aaaacaactt gagatgttgg 1740
caagaatgaa agtcacccac taccggtttg ctctggattg ggcctcggtc cttcccactg 1800
gcaacctgtc cgcggtgaac cgacaggccc tgaggtacta caggtgcgtg gtcagtgagg 1860
ggctgaagct tggcatctcc gcgatggtca ccctgtatta tccgacccac gcccacctag 1920
gcctccccga gcctctgttg catgccgacg ggtggctgaa cccatcgacg gccgaggcct 1980
tccaggccta cgctgggctg tgcttccagg agctggggga cctggtgaag ctctggatca 2040
ccatcaacga gcctaaccgg ctaagtgaca tctacaaccg ctctggcaac gacacctacg 2100
gggcggcgca caacctgctg gtggcccacg ccctggcctg gcgcctctac gaccagcagt 2160
tcaggccgtc acagcgcggg gccgtgtcgc tgtcgctgca cgcggactgg gcggaacccg 2220
ccaaccccta tgctgactcg cactggaggg cggccgagcg cttcctgcag ttcgagatcg 2280
cctggttcgc cgagccgctc ttcaagaccg gggactaccc cgcggccatg agggaataca 2340
ttgcctccaa gcaccgacgg gggctttcca gctcggccct gccgcgcctc accgaggccg 2400
aaaggaggct gctcaagggc acggtcgact tctgcgcgct caaccacttc accactaggt 2460
tcgtgatgca cgagcagctg gccggcagcc gctacgactc ggacagggac atccagtttc 2520
tgcaggacat cacccgcctg agctccccca cgcgcctggc tgtgattccc tggggggtgc 2580
gcaagctgct gcggtgggtc cggaggaact acggcgacat ggacatttac atcaccgcca 2640
gtggcatcga cgaccaggct ctggaggatg accggctccg gaagtactac ctagggaagt 2700
accttcagga ggtgctgaaa gcatacctga ttgataaagt cagaatcaaa ggctattatg 2760
cattcaaact ggctgaagag aaatctaaac ccagatttgg attcttcaca tctgatttta 2820
aagctaaatc ctcaatacaa ttttacaaca aagtgatcag cagcaggggc ttcccttttg 2880
agaacagtag ttctagatgc agtcagaccc aagaaaatac agagtgcact gtctgcttat 2940
tccttgtgca gaagaaacca ctgatattcc tgggttgttg cttcttctcc accctggttc 3000
tactcttatc aattgccatt tttcaaaggc agaagagaag aaagttttgg aaagcaaaaa 3060
acttacaaca cataccatta aagaaaggca agagagttgt tagctaaact gatctgtctg 3120
catgatagac agtttaaaaa ttcatcccag ttccatatgc tggtaactta caggagatat 3180
acctgtatta tagaaagaca atctgagata cagctgtaac caaggtgatg acaattgtct 3240
ctgctgtgtg gttcaaagaa cattccctta ggtgttgaca tcagtgaact cagttcttgg 3300
atgtaaacat aaaggcttca tcctgacagt aagctatgag gattacatgc tacattgctt 3360
cttaaagttt catcaactgt attccatcat tctgctttag ctttcatctc taccaatagc 3420
tacttgtggt acaataaatt atttttaaga agaaaaaaaa 3460
<210> SEQ ID NO 11
<211> LENGTH: 1012
<212> TYPE: PRT
<213> ORGANISM: homo sapiens
<300> PUBLICATION INFORMATION:
<308> DATABASE ACCESSION NUMBER: NP_004786
<309> DATABASE ENTRY DATE: 2008-04-20
<313> RELEVANT RESIDUES IN SEQ ID NO: (1)..(1012)
<400> SEQUENCE: 11
Met Pro Ala Ser Ala Pro Pro Arg Arg Pro Arg Pro Pro Pro Pro Ser
1 5 10 15
Leu Ser Leu Leu Leu Val Leu Leu Gly Leu Gly Gly Arg Arg Leu Arg
20 25 30
Ala Glu Pro Gly Asp Gly Ala Gln Thr Trp Ala Arg Phe Ser Arg Pro
35 40 45
Pro Ala Pro Glu Ala Ala Gly Leu Phe Gln Gly Thr Phe Pro Asp Gly
50 55 60
Phe Leu Trp Ala Val Gly Ser Ala Ala Tyr Gln Thr Glu Gly Gly Trp
65 70 75 80
Gln Gln His Gly Lys Gly Ala Ser Ile Trp Asp Thr Phe Thr His His
85 90 95
Pro Leu Ala Pro Pro Gly Asp Ser Arg Asn Ala Ser Leu Pro Leu Gly
100 105 110
Ala Pro Ser Pro Leu Gln Pro Ala Thr Gly Asp Val Ala Ser Asp Ser
115 120 125
Tyr Asn Asn Val Phe Arg Asp Thr Glu Ala Leu Arg Glu Leu Gly Val
130 135 140
Thr His Tyr Arg Phe Ser Ile Ser Trp Ala Arg Val Leu Pro Asn Gly
145 150 155 160
Ser Ala Gly Val Pro Asn Arg Glu Gly Leu Arg Tyr Tyr Arg Arg Leu
165 170 175
Leu Glu Arg Leu Arg Glu Leu Gly Val Gln Pro Val Val Thr Leu Tyr
180 185 190
His Trp Asp Leu Pro Gln Arg Leu Gln Asp Ala Tyr Gly Gly Trp Ala
195 200 205
Asn Arg Ala Leu Ala Asp His Phe Arg Asp Tyr Ala Glu Leu Cys Phe
210 215 220
Arg His Phe Gly Gly Gln Val Lys Tyr Trp Ile Thr Ile Asp Asn Pro
225 230 235 240
Tyr Val Val Ala Trp His Gly Tyr Ala Thr Gly Arg Leu Ala Pro Gly
245 250 255
Ile Arg Gly Ser Pro Arg Leu Gly Tyr Leu Val Ala His Asn Leu Leu
260 265 270
Leu Ala His Ala Lys Val Trp His Leu Tyr Asn Thr Ser Phe Arg Pro
275 280 285
Thr Gln Gly Gly Gln Val Ser Ile Ala Leu Ser Ser His Trp Ile Asn
290 295 300
Pro Arg Arg Met Thr Asp His Ser Ile Lys Glu Cys Gln Lys Ser Leu
305 310 315 320
Asp Phe Val Leu Gly Trp Phe Ala Lys Pro Val Phe Ile Asp Gly Asp
325 330 335
Tyr Pro Glu Ser Met Lys Asn Asn Leu Ser Ser Ile Leu Pro Asp Phe
340 345 350
Thr Glu Ser Glu Lys Lys Phe Ile Lys Gly Thr Ala Asp Phe Phe Ala
355 360 365
Leu Cys Phe Gly Pro Thr Leu Ser Phe Gln Leu Leu Asp Pro His Met
370 375 380
Lys Phe Arg Gln Leu Glu Ser Pro Asn Leu Arg Gln Leu Leu Ser Trp
385 390 395 400
Ile Asp Leu Glu Phe Asn His Pro Gln Ile Phe Ile Val Glu Asn Gly
405 410 415
Trp Phe Val Ser Gly Thr Thr Lys Arg Asp Asp Ala Lys Tyr Met Tyr
420 425 430
Tyr Leu Lys Lys Phe Ile Met Glu Thr Leu Lys Ala Ile Lys Leu Asp
435 440 445
Gly Val Asp Val Ile Gly Tyr Thr Ala Trp Ser Leu Met Asp Gly Phe
450 455 460
Glu Trp His Arg Gly Tyr Ser Ile Arg Arg Gly Leu Phe Tyr Val Asp
465 470 475 480
Phe Leu Ser Gln Asp Lys Met Leu Leu Pro Lys Ser Ser Ala Leu Phe
485 490 495
Tyr Gln Lys Leu Ile Glu Lys Asn Gly Phe Pro Pro Leu Pro Glu Asn
500 505 510
Gln Pro Leu Glu Gly Thr Phe Pro Cys Asp Phe Ala Trp Gly Val Val
515 520 525
Asp Asn Tyr Ile Gln Val Asp Thr Thr Leu Ser Gln Phe Thr Asp Leu
530 535 540
Asn Val Tyr Leu Trp Asp Val His His Ser Lys Arg Leu Ile Lys Val
545 550 555 560
Asp Gly Val Val Thr Lys Lys Arg Lys Ser Tyr Cys Val Asp Phe Ala
565 570 575
Ala Ile Gln Pro Gln Ile Ala Leu Leu Gln Glu Met His Val Thr His
580 585 590
Phe Arg Phe Ser Leu Asp Trp Ala Leu Ile Leu Pro Leu Gly Asn Gln
595 600 605
Ser Gln Val Asn His Thr Ile Leu Gln Tyr Tyr Arg Cys Met Ala Ser
610 615 620
Glu Leu Val Arg Val Asn Ile Thr Pro Val Val Ala Leu Trp Gln Pro
625 630 635 640
Met Ala Pro Asn Gln Gly Leu Pro Arg Leu Leu Ala Arg Gln Gly Ala
645 650 655
Trp Glu Asn Pro Tyr Thr Ala Leu Ala Phe Ala Glu Tyr Ala Arg Leu
660 665 670
Cys Phe Gln Glu Leu Gly His His Val Lys Leu Trp Ile Thr Met Asn
675 680 685
Glu Pro Tyr Thr Arg Asn Met Thr Tyr Ser Ala Gly His Asn Leu Leu
690 695 700
Lys Ala His Ala Leu Ala Trp His Val Tyr Asn Glu Lys Phe Arg His
705 710 715 720
Ala Gln Asn Gly Lys Ile Ser Ile Ala Leu Gln Ala Asp Trp Ile Glu
725 730 735
Pro Ala Cys Pro Phe Ser Gln Lys Asp Lys Glu Val Ala Glu Arg Val
740 745 750
Leu Glu Phe Asp Ile Gly Trp Leu Ala Glu Pro Ile Phe Gly Ser Gly
755 760 765
Asp Tyr Pro Trp Val Met Arg Asp Trp Leu Asn Gln Arg Asn Asn Phe
770 775 780
Leu Leu Pro Tyr Phe Thr Glu Asp Glu Lys Lys Leu Ile Gln Gly Thr
785 790 795 800
Phe Asp Phe Leu Ala Leu Ser His Tyr Thr Thr Ile Leu Val Asp Ser
805 810 815
Glu Lys Glu Asp Pro Ile Lys Tyr Asn Asp Tyr Leu Glu Val Gln Glu
820 825 830
Met Thr Asp Ile Thr Trp Leu Asn Ser Pro Ser Gln Val Ala Val Val
835 840 845
Pro Trp Gly Leu Arg Lys Val Leu Asn Trp Leu Lys Phe Lys Tyr Gly
850 855 860
Asp Leu Pro Met Tyr Ile Ile Ser Asn Gly Ile Asp Asp Gly Leu His
865 870 875 880
Ala Glu Asp Asp Gln Leu Arg Val Tyr Tyr Met Gln Asn Tyr Ile Asn
885 890 895
Glu Ala Leu Lys Ala His Ile Leu Asp Gly Ile Asn Leu Cys Gly Tyr
900 905 910
Phe Ala Tyr Ser Phe Asn Asp Arg Thr Ala Pro Arg Phe Gly Leu Tyr
915 920 925
Arg Tyr Ala Ala Asp Gln Phe Glu Pro Lys Ala Ser Met Lys His Tyr
930 935 940
Arg Lys Ile Ile Asp Ser Asn Gly Phe Pro Gly Pro Glu Thr Leu Glu
945 950 955 960
Arg Phe Cys Pro Glu Glu Phe Thr Val Cys Thr Glu Cys Ser Phe Phe
965 970 975
His Thr Arg Lys Ser Leu Leu Ala Phe Ile Ala Phe Leu Phe Phe Ala
980 985 990
Ser Ile Ile Ser Leu Ser Leu Ile Phe Tyr Tyr Ser Lys Lys Gly Arg
995 1000 1005
Arg Ser Tyr Lys
1010
<210> SEQ ID NO 12
<211> LENGTH: 1012
<212> TYPE: PRT
<213> ORGANISM: homo sapiens
<300> PUBLICATION INFORMATION:
<308> DATABASE ACCESSION NUMBER: CAH71888
<309> DATABASE ENTRY DATE: 2008-02-24
<313> RELEVANT RESIDUES IN SEQ ID NO: (1)..(1012)
<400> SEQUENCE: 12
Met Pro Ala Ser Ala Pro Pro Arg Arg Pro Arg Pro Pro Pro Pro Ser
1 5 10 15
Leu Ser Leu Leu Leu Val Leu Leu Gly Leu Gly Gly Arg Arg Leu Arg
20 25 30
Ala Glu Pro Gly Asp Gly Ala Gln Thr Trp Ala Arg Phe Ser Arg Pro
35 40 45
Pro Ala Pro Glu Ala Ala Gly Leu Phe Gln Gly Thr Phe Pro Asp Gly
50 55 60
Phe Leu Trp Ala Val Gly Ser Ala Ala Tyr Gln Thr Glu Gly Gly Trp
65 70 75 80
Gln Gln His Gly Lys Gly Ala Ser Ile Trp Asp Thr Phe Thr His His
85 90 95
Pro Leu Ala Pro Pro Gly Asp Ser Arg Asn Ala Ser Leu Pro Leu Gly
100 105 110
Ala Pro Ser Pro Leu Gln Pro Ala Thr Gly Asp Val Ala Ser Asp Ser
115 120 125
Tyr Asn Asn Val Phe Arg Asp Thr Glu Ala Leu Arg Glu Leu Gly Val
130 135 140
Thr His Tyr Arg Phe Ser Ile Ser Trp Ala Arg Val Leu Pro Asn Gly
145 150 155 160
Ser Ala Gly Val Pro Asn Arg Glu Gly Leu Arg Tyr Tyr Arg Arg Leu
165 170 175
Leu Glu Arg Leu Arg Glu Leu Gly Val Gln Pro Val Val Thr Leu Tyr
180 185 190
His Trp Asp Leu Pro Gln Arg Leu Gln Asp Ala Tyr Gly Gly Trp Ala
195 200 205
Asn Arg Ala Leu Ala Asp His Phe Arg Asp Tyr Ala Glu Leu Cys Phe
210 215 220
Arg His Phe Gly Gly Gln Val Lys Tyr Trp Ile Thr Ile Asp Asn Pro
225 230 235 240
Tyr Val Val Ala Trp His Gly Tyr Ala Thr Gly Arg Leu Ala Pro Gly
245 250 255
Ile Arg Gly Ser Pro Arg Leu Gly Tyr Leu Val Ala His Asn Leu Leu
260 265 270
Leu Ala His Ala Lys Val Trp His Leu Tyr Asn Thr Ser Phe Arg Pro
275 280 285
Thr Gln Gly Gly Gln Val Ser Ile Ala Leu Ser Ser His Trp Ile Asn
290 295 300
Pro Arg Arg Met Thr Asp His Ser Ile Lys Glu Cys Gln Lys Ser Leu
305 310 315 320
Asp Phe Val Leu Gly Trp Phe Ala Lys Pro Val Phe Ile Asp Gly Asp
325 330 335
Tyr Pro Glu Ser Met Lys Asn Asn Leu Ser Ser Ile Leu Pro Asp Phe
340 345 350
Thr Glu Ser Glu Lys Lys Phe Ile Lys Gly Thr Ala Asp Phe Phe Ala
355 360 365
Leu Cys Phe Gly Pro Thr Leu Ser Phe Gln Leu Leu Asp Pro His Met
370 375 380
Lys Phe Arg Gln Leu Glu Ser Pro Asn Leu Arg Gln Leu Leu Ser Trp
385 390 395 400
Ile Asp Leu Glu Phe Asn His Pro Gln Ile Phe Ile Val Glu Asn Gly
405 410 415
Trp Phe Val Ser Gly Thr Thr Lys Arg Asp Asp Ala Lys Tyr Met Tyr
420 425 430
Tyr Leu Lys Lys Phe Ile Met Glu Thr Leu Lys Ala Ile Lys Leu Asp
435 440 445
Gly Val Asp Val Ile Gly Tyr Thr Ala Trp Ser Leu Met Asp Gly Phe
450 455 460
Glu Trp His Arg Gly Tyr Ser Ile Arg Arg Gly Leu Phe Tyr Val Asp
465 470 475 480
Phe Leu Ser Gln Asp Lys Met Leu Leu Pro Lys Ser Ser Ala Leu Phe
485 490 495
Tyr Gln Lys Leu Ile Glu Lys Asn Gly Phe Pro Pro Leu Pro Glu Asn
500 505 510
Gln Pro Leu Glu Gly Thr Phe Pro Cys Asp Phe Ala Trp Gly Val Val
515 520 525
Asp Asn Tyr Ile Gln Val Asp Thr Thr Leu Ser Gln Phe Thr Asp Leu
530 535 540
Asn Val Tyr Leu Trp Asp Val His His Ser Lys Arg Leu Ile Lys Val
545 550 555 560
Asp Gly Val Val Thr Lys Lys Arg Lys Ser Tyr Cys Val Asp Phe Ala
565 570 575
Ala Ile Gln Pro Gln Ile Ala Leu Leu Gln Glu Met His Val Thr His
580 585 590
Phe Arg Phe Ser Leu Asp Trp Ala Leu Ile Leu Pro Leu Gly Asn Gln
595 600 605
Ser Gln Val Asn His Thr Ile Leu Gln Tyr Tyr Arg Cys Met Ala Ser
610 615 620
Glu Leu Val Arg Val Asn Ile Thr Pro Val Val Ala Leu Trp Gln Pro
625 630 635 640
Met Ala Pro Asn Gln Gly Leu Pro Arg Leu Leu Ala Arg Gln Gly Ala
645 650 655
Trp Glu Asn Pro Tyr Thr Ala Leu Ala Phe Ala Glu Tyr Ala Arg Leu
660 665 670
Cys Phe Gln Glu Leu Gly His His Val Lys Leu Trp Ile Thr Met Asn
675 680 685
Glu Pro Tyr Thr Arg Asn Met Thr Tyr Ser Ala Gly His Asn Leu Leu
690 695 700
Lys Ala His Ala Leu Ala Trp His Val Tyr Asn Glu Lys Phe Arg His
705 710 715 720
Ala Gln Asn Gly Lys Ile Ser Ile Ala Leu Gln Ala Asp Trp Ile Glu
725 730 735
Pro Ala Cys Pro Phe Ser Gln Lys Asp Lys Glu Val Ala Glu Arg Val
740 745 750
Leu Glu Phe Asp Ile Gly Trp Leu Ala Glu Pro Ile Phe Gly Ser Gly
755 760 765
Asp Tyr Pro Trp Val Met Arg Asp Trp Leu Asn Gln Arg Asn Asn Phe
770 775 780
Leu Leu Pro Tyr Phe Thr Glu Asp Glu Lys Lys Leu Ile Gln Gly Thr
785 790 795 800
Phe Asp Phe Leu Ala Leu Ser His Tyr Thr Thr Ile Leu Val Asp Ser
805 810 815
Glu Lys Glu Asp Pro Ile Lys Tyr Asn Asp Tyr Leu Glu Val Gln Glu
820 825 830
Met Thr Asp Ile Thr Trp Leu Asn Ser Pro Ser Gln Val Ala Val Val
835 840 845
Pro Trp Gly Leu Arg Lys Val Leu Asn Trp Leu Lys Phe Lys Tyr Gly
850 855 860
Asp Leu Pro Met Tyr Ile Ile Ser Asn Gly Ile Asp Asp Gly Leu His
865 870 875 880
Ala Glu Asp Asp Gln Leu Arg Val Tyr Tyr Met Gln Asn Tyr Ile Asn
885 890 895
Glu Ala Leu Lys Ala His Ile Leu Asp Gly Ile Asn Leu Cys Gly Tyr
900 905 910
Phe Ala Tyr Ser Phe Asn Asp Arg Thr Ala Pro Arg Phe Gly Leu Tyr
915 920 925
Arg Tyr Ala Ala Asp Gln Phe Glu Pro Lys Ala Ser Met Lys His Tyr
930 935 940
Arg Lys Ile Ile Asp Ser Asn Gly Phe Pro Gly Pro Glu Thr Leu Glu
945 950 955 960
Arg Phe Cys Pro Glu Glu Phe Thr Val Cys Thr Glu Cys Ser Phe Phe
965 970 975
His Thr Arg Lys Ser Leu Leu Ala Phe Ile Ala Phe Leu Phe Phe Ala
980 985 990
Ser Ile Ile Ser Leu Ser Leu Ile Phe Tyr Tyr Ser Lys Lys Gly Arg
995 1000 1005
Arg Ser Tyr Lys
1010
<210> SEQ ID NO 13
<211> LENGTH: 1012
<212> TYPE: PRT
<213> ORGANISM: homo sapiens
<300> PUBLICATION INFORMATION:
<308> DATABASE ACCESSION NUMBER: BAA24940
<309> DATABASE ENTRY DATE: 2004-11-03
<313> RELEVANT RESIDUES IN SEQ ID NO: (1)..(1012)
<400> SEQUENCE: 13
Met Pro Ala Ser Ala Pro Pro Arg Arg Pro Arg Pro Pro Pro Gln Ser
1 5 10 15
Leu Ser Leu Leu Leu Val Leu Leu Gly Leu Gly Gly Arg Arg Leu Arg
20 25 30
Ala Glu Pro Gly Asp Gly Ala Gln Thr Trp Ala Arg Phe Ser Arg Pro
35 40 45
Pro Ala Pro Glu Ala Ala Gly Leu Phe Gln Gly Thr Phe Pro Asp Gly
50 55 60
Phe Leu Trp Ala Val Gly Ser Ala Ala Tyr Gln Thr Glu Gly Gly Trp
65 70 75 80
Gln Gln His Gly Lys Gly Ala Ser Ile Trp Asp Thr Phe Thr His His
85 90 95
Pro Leu Ala Pro Pro Gly Asp Ser Arg Asn Ala Ser Leu Pro Leu Gly
100 105 110
Ala Pro Ser Pro Leu Gln Pro Ala Thr Gly Asp Val Ala Ser Asp Ser
115 120 125
Tyr Asn Asn Val Phe Arg Asp Thr Glu Ala Leu Arg Glu Leu Gly Val
130 135 140
Thr His Tyr Arg Phe Ser Ile Ser Trp Ala Arg Val Leu Pro Asn Gly
145 150 155 160
Ser Ala Gly Val Pro Asn Arg Glu Gly Leu Arg Tyr Tyr Arg Arg Leu
165 170 175
Leu Glu Arg Leu Arg Glu Leu Gly Val Gln Pro Val Val Thr Leu Tyr
180 185 190
His Trp Asp Leu Pro Gln Arg Leu Gln Asp Ala Tyr Gly Gly Trp Ala
195 200 205
Asn Arg Ala Leu Ala Asp His Phe Arg Asp Tyr Ala Glu Leu Cys Phe
210 215 220
Arg His Phe Gly Gly Gln Val Lys Tyr Trp Ile Thr Ile Asp Asn Pro
225 230 235 240
Tyr Val Val Ala Trp His Gly Tyr Ala Thr Gly Arg Leu Ala Pro Gly
245 250 255
Ile Arg Gly Ser Pro Arg Leu Gly Tyr Leu Val Ala His Asn Leu Leu
260 265 270
Leu Ala His Ala Lys Val Trp His Leu Tyr Asn Thr Ser Phe Arg Pro
275 280 285
Thr Gln Gly Gly Gln Val Ser Ile Ala Leu Ser Ser His Trp Ile Asn
290 295 300
Pro Arg Arg Met Thr Asp His Ser Ile Lys Glu Cys Gln Lys Ser Leu
305 310 315 320
Asp Phe Val Leu Gly Trp Phe Ala Lys Pro Val Phe Ile Asp Gly Asp
325 330 335
Tyr Pro Glu Ser Met Lys Asn Asn Leu Ser Ser Ile Leu Pro Asp Phe
340 345 350
Thr Glu Ser Glu Lys Lys Phe Ile Lys Gly Thr Ala Asp Phe Phe Ala
355 360 365
Leu Cys Phe Gly Pro Thr Leu Ser Phe Gln Leu Leu Asp Pro His Met
370 375 380
Lys Phe Arg Gln Leu Glu Ser Pro Asn Leu Arg Gln Leu Leu Ser Trp
385 390 395 400
Ile Asp Leu Glu Phe Asn His Pro Gln Ile Phe Ile Val Glu Asn Gly
405 410 415
Trp Phe Val Ser Gly Thr Thr Lys Arg Asp Asp Ala Lys Tyr Met Tyr
420 425 430
Tyr Leu Lys Lys Phe Ile Met Glu Thr Leu Lys Ala Ile Lys Leu Asp
435 440 445
Gly Val Asp Val Ile Gly Tyr Thr Ala Trp Ser Leu Met Asp Gly Phe
450 455 460
Glu Trp His Arg Gly Tyr Ser Ile Arg Arg Gly Leu Phe Tyr Val Asp
465 470 475 480
Phe Leu Ser Gln Asp Lys Met Leu Leu Pro Lys Ser Ser Ala Leu Phe
485 490 495
Tyr Gln Lys Leu Ile Glu Lys Asn Gly Phe Pro Pro Leu Pro Glu Asn
500 505 510
Gln Pro Leu Glu Gly Thr Phe Pro Cys Asp Phe Ala Trp Gly Val Val
515 520 525
Asp Asn Tyr Ile Gln Val Asp Thr Thr Leu Ser Gln Phe Thr Asp Leu
530 535 540
Asn Val Tyr Leu Trp Asp Val His His Ser Lys Arg Leu Ile Lys Val
545 550 555 560
Asp Gly Val Val Thr Lys Lys Arg Lys Ser Tyr Cys Val Asp Phe Ala
565 570 575
Ala Ile Gln Pro Gln Ile Ala Leu Leu Gln Glu Met His Val Thr His
580 585 590
Phe Arg Phe Ser Leu Asp Trp Ala Leu Ile Leu Pro Leu Gly Asn Gln
595 600 605
Ser Gln Val Asn His Thr Ile Leu Gln Tyr Tyr Arg Cys Met Ala Ser
610 615 620
Glu Leu Val Arg Val Asn Ile Thr Pro Val Val Ala Leu Trp Gln Pro
625 630 635 640
Met Ala Pro Asn Gln Gly Leu Pro Arg Leu Leu Ala Arg Gln Gly Ala
645 650 655
Trp Glu Asn Pro Tyr Thr Ala Leu Ala Phe Ala Glu Tyr Ala Arg Leu
660 665 670
Cys Phe Gln Glu Leu Gly His His Val Lys Leu Trp Ile Thr Met Asn
675 680 685
Glu Pro Tyr Thr Arg Asn Met Thr Tyr Ser Ala Gly His Asn Leu Leu
690 695 700
Lys Ala His Ala Leu Ala Trp His Val Tyr Asn Glu Lys Phe Arg His
705 710 715 720
Ala Gln Asn Gly Lys Ile Ser Ile Ala Leu Gln Ala Asp Trp Ile Glu
725 730 735
Pro Ala Cys Pro Phe Ser Gln Lys Asp Lys Glu Val Ala Glu Arg Val
740 745 750
Leu Glu Phe Asp Ile Gly Trp Leu Ala Glu Pro Ile Phe Gly Ser Gly
755 760 765
Asp Tyr Pro Trp Val Met Arg Asp Trp Leu Asn Gln Arg Asn Asn Phe
770 775 780
Leu Leu Pro Tyr Phe Thr Glu Asp Glu Lys Lys Leu Ile Gln Gly Thr
785 790 795 800
Phe Asp Phe Leu Ala Leu Ser His Tyr Thr Thr Ile Leu Val Asp Ser
805 810 815
Glu Lys Glu Asp Pro Ile Lys Tyr Asn Asp Tyr Leu Glu Val Gln Glu
820 825 830
Met Thr Asp Ile Thr Trp Leu Asn Ser Pro Ser Gln Val Ala Val Val
835 840 845
Pro Trp Gly Leu Arg Lys Val Leu Asn Trp Leu Lys Phe Lys Tyr Gly
850 855 860
Asp Leu Pro Met Tyr Ile Ile Ser Asn Gly Ile Asp Asp Gly Leu His
865 870 875 880
Ala Glu Asp Asp Gln Leu Arg Val Tyr Tyr Met Gln Asn Tyr Ile Asn
885 890 895
Glu Ala Leu Lys Ala His Ile Leu Asp Gly Ile Asn Leu Cys Gly Tyr
900 905 910
Phe Ala Tyr Ser Phe Asn Asp Arg Thr Ala Pro Arg Phe Gly Leu Tyr
915 920 925
Arg Tyr Ala Ala Asp Gln Phe Glu Pro Lys Ala Ser Met Lys His Tyr
930 935 940
Arg Lys Ile Ile Asp Ser Asn Gly Phe Pro Gly Pro Glu Thr Leu Glu
945 950 955 960
Arg Phe Cys Pro Glu Glu Phe Thr Val Cys Thr Glu Cys Ser Phe Phe
965 970 975
His Thr Arg Lys Ser Leu Leu Ala Phe Ile Ala Phe Leu Phe Phe Ala
980 985 990
Ser Ile Ile Ser Leu Ser Leu Ile Phe Tyr Tyr Ser Lys Lys Gly Arg
995 1000 1005
Arg Ser Tyr Lys
1010
<210> SEQ ID NO 14
<211> LENGTH: 1012
<212> TYPE: PRT
<213> ORGANISM: homo sapiens
<300> PUBLICATION INFORMATION:
<308> DATABASE ACCESSION NUMBER: EAX08526
<309> DATABASE ENTRY DATE: 2006-12-18
<313> RELEVANT RESIDUES IN SEQ ID NO: (1)..(1012)
<400> SEQUENCE: 14
Met Pro Ala Ser Ala Pro Pro Arg Arg Pro Arg Pro Pro Pro Pro Ser
1 5 10 15
Leu Ser Leu Leu Leu Val Leu Leu Gly Leu Gly Gly Arg Arg Leu Arg
20 25 30
Ala Glu Pro Gly Asp Gly Ala Gln Thr Trp Ala Arg Phe Ser Arg Pro
35 40 45
Pro Ala Pro Glu Ala Ala Gly Leu Phe Gln Gly Thr Phe Pro Asp Gly
50 55 60
Phe Leu Trp Ala Val Gly Ser Ala Ala Tyr Gln Thr Glu Gly Gly Trp
65 70 75 80
Gln Gln His Gly Lys Gly Ala Ser Ile Trp Asp Thr Phe Thr His His
85 90 95
Pro Leu Ala Pro Pro Gly Asp Ser Arg Asn Ala Ser Leu Pro Leu Gly
100 105 110
Ala Pro Ser Pro Leu Gln Pro Ala Thr Gly Asp Val Ala Ser Asp Ser
115 120 125
Tyr Asn Asn Val Phe Arg Asp Thr Glu Ala Leu Arg Glu Leu Gly Val
130 135 140
Thr His Tyr Arg Phe Ser Ile Ser Trp Ala Arg Val Leu Pro Asn Gly
145 150 155 160
Ser Ala Gly Val Pro Asn Arg Glu Gly Leu Arg Tyr Tyr Arg Arg Leu
165 170 175
Leu Glu Arg Leu Arg Glu Leu Gly Val Gln Pro Val Val Thr Leu Tyr
180 185 190
His Trp Asp Leu Pro Gln Arg Leu Gln Asp Ala Tyr Gly Gly Trp Ala
195 200 205
Asn Arg Ala Leu Ala Asp His Phe Arg Asp Tyr Ala Glu Leu Cys Phe
210 215 220
Arg His Phe Gly Gly Gln Val Lys Tyr Trp Ile Thr Ile Asp Asn Pro
225 230 235 240
Tyr Val Val Ala Trp His Gly Tyr Ala Thr Gly Arg Leu Ala Pro Gly
245 250 255
Ile Arg Gly Ser Pro Arg Leu Gly Tyr Leu Val Ala His Asn Leu Leu
260 265 270
Leu Ala His Ala Lys Val Trp His Leu Tyr Asn Thr Ser Phe Arg Pro
275 280 285
Thr Gln Gly Gly Gln Val Ser Ile Ala Leu Ser Ser His Trp Ile Asn
290 295 300
Pro Arg Arg Met Thr Asp His Ser Ile Lys Glu Cys Gln Lys Ser Leu
305 310 315 320
Asp Phe Val Leu Gly Trp Phe Ala Lys Pro Val Phe Ile Asp Gly Asp
325 330 335
Tyr Pro Glu Ser Met Lys Asn Asn Leu Ser Ser Ile Leu Pro Asp Phe
340 345 350
Thr Glu Ser Glu Lys Lys Phe Ile Lys Gly Thr Ala Asp Phe Phe Ala
355 360 365
Leu Cys Phe Gly Pro Thr Leu Ser Phe Gln Leu Leu Asp Pro His Met
370 375 380
Lys Phe Arg Gln Leu Glu Ser Pro Asn Leu Arg Gln Leu Leu Ser Trp
385 390 395 400
Ile Asp Leu Glu Phe Asn His Pro Gln Ile Phe Ile Val Glu Asn Gly
405 410 415
Trp Phe Val Ser Gly Thr Thr Lys Arg Asp Asp Ala Lys Tyr Met Tyr
420 425 430
Tyr Leu Lys Lys Phe Ile Met Glu Thr Leu Lys Ala Ile Lys Leu Asp
435 440 445
Gly Val Asp Val Ile Gly Tyr Thr Ala Trp Ser Leu Met Asp Gly Phe
450 455 460
Glu Trp His Arg Gly Tyr Ser Ile Arg Arg Gly Leu Phe Tyr Val Asp
465 470 475 480
Phe Leu Ser Gln Asp Lys Met Leu Leu Pro Lys Ser Ser Ala Leu Phe
485 490 495
Tyr Gln Lys Leu Ile Glu Lys Asn Gly Phe Pro Pro Leu Pro Glu Asn
500 505 510
Gln Pro Leu Glu Gly Thr Phe Pro Cys Asp Phe Ala Trp Gly Val Val
515 520 525
Asp Asn Tyr Ile Gln Val Asp Thr Thr Leu Ser Gln Phe Thr Asp Leu
530 535 540
Asn Val Tyr Leu Trp Asp Val His His Ser Lys Arg Leu Ile Lys Val
545 550 555 560
Asp Gly Val Val Thr Lys Lys Arg Lys Ser Tyr Cys Val Asp Phe Ala
565 570 575
Ala Ile Gln Pro Gln Ile Ala Leu Leu Gln Glu Met His Val Thr His
580 585 590
Phe Arg Phe Ser Leu Asp Trp Ala Leu Ile Leu Pro Leu Gly Asn Gln
595 600 605
Ser Gln Val Asn His Thr Ile Leu Gln Tyr Tyr Arg Cys Met Ala Ser
610 615 620
Glu Leu Val Arg Val Asn Ile Thr Pro Val Val Ala Leu Trp Gln Pro
625 630 635 640
Met Ala Pro Asn Gln Gly Leu Pro Arg Leu Leu Ala Arg Gln Gly Ala
645 650 655
Trp Glu Asn Pro Tyr Thr Ala Leu Ala Phe Ala Glu Tyr Ala Arg Leu
660 665 670
Cys Phe Gln Glu Leu Gly His His Val Lys Leu Trp Ile Thr Met Asn
675 680 685
Glu Pro Tyr Thr Arg Asn Met Thr Tyr Ser Ala Gly His Asn Leu Leu
690 695 700
Lys Ala His Ala Leu Ala Trp His Val Tyr Asn Glu Lys Phe Arg His
705 710 715 720
Ala Gln Asn Gly Lys Ile Ser Ile Ala Leu Gln Ala Asp Trp Ile Glu
725 730 735
Pro Ala Cys Pro Phe Ser Gln Lys Asp Lys Glu Val Ala Glu Arg Val
740 745 750
Leu Glu Phe Asp Ile Gly Trp Leu Ala Glu Pro Ile Phe Gly Ser Gly
755 760 765
Asp Tyr Pro Trp Val Met Arg Asp Trp Leu Asn Gln Arg Asn Asn Phe
770 775 780
Leu Leu Pro Tyr Phe Thr Glu Asp Glu Lys Lys Leu Ile Gln Gly Thr
785 790 795 800
Phe Asp Phe Leu Ala Leu Ser His Tyr Thr Thr Ile Leu Val Asp Ser
805 810 815
Glu Lys Glu Asp Pro Ile Lys Tyr Asn Asp Tyr Leu Glu Val Gln Glu
820 825 830
Met Thr Asp Ile Thr Trp Leu Asn Ser Pro Ser Gln Val Ala Val Val
835 840 845
Pro Trp Gly Leu Arg Lys Val Leu Asn Trp Leu Lys Phe Lys Tyr Gly
850 855 860
Asp Leu Pro Met Tyr Ile Ile Ser Asn Gly Ile Asp Asp Gly Leu His
865 870 875 880
Ala Glu Asp Asp Gln Leu Arg Val Tyr Tyr Met Gln Asn Tyr Ile Asn
885 890 895
Glu Ala Leu Lys Ala His Ile Leu Asp Gly Ile Asn Leu Cys Gly Tyr
900 905 910
Phe Ala Tyr Ser Phe Asn Asp Arg Thr Ala Pro Arg Phe Gly Leu Tyr
915 920 925
Arg Tyr Ala Ala Asp Gln Phe Glu Pro Lys Ala Ser Met Lys His Tyr
930 935 940
Arg Lys Ile Ile Asp Ser Asn Gly Phe Pro Gly Pro Glu Thr Leu Glu
945 950 955 960
Arg Phe Cys Pro Glu Glu Phe Thr Val Cys Thr Glu Cys Ser Phe Phe
965 970 975
His Thr Arg Lys Ser Leu Leu Ala Phe Ile Ala Phe Leu Phe Phe Ala
980 985 990
Ser Ile Ile Ser Leu Ser Leu Ile Phe Tyr Tyr Ser Lys Lys Gly Arg
995 1000 1005
Arg Ser Tyr Lys
1010
<210> SEQ ID NO 15
<211> LENGTH: 549
<212> TYPE: PRT
<213> ORGANISM: homo sapiens
<300> PUBLICATION INFORMATION:
<308> DATABASE ACCESSION NUMBER: BAA24941
<309> DATABASE ENTRY DATE: 2004-11-02
<313> RELEVANT RESIDUES IN SEQ ID NO: (1)..(549)
<400> SEQUENCE: 15
Met Pro Ala Ser Ala Pro Pro Arg Arg Pro Arg Pro Pro Pro Gln Ser
1 5 10 15
Leu Ser Leu Leu Leu Val Leu Leu Gly Leu Gly Gly Arg Arg Leu Arg
20 25 30
Ala Glu Pro Gly Asp Gly Ala Gln Thr Trp Ala Arg Phe Ser Arg Pro
35 40 45
Pro Ala Pro Glu Ala Ala Gly Leu Phe Gln Gly Thr Phe Pro Asp Gly
50 55 60
Phe Leu Trp Ala Val Gly Ser Ala Ala Tyr Gln Thr Glu Gly Gly Trp
65 70 75 80
Gln Gln His Gly Lys Gly Ala Ser Ile Trp Asp Thr Phe Thr His His
85 90 95
Pro Leu Ala Pro Pro Gly Asp Ser Arg Asn Ala Ser Leu Pro Leu Gly
100 105 110
Ala Pro Ser Pro Leu Gln Pro Ala Thr Gly Asp Val Ala Ser Asp Ser
115 120 125
Tyr Asn Asn Val Phe Arg Asp Thr Glu Ala Leu Arg Glu Leu Gly Val
130 135 140
Thr His Tyr Arg Phe Ser Ile Ser Trp Ala Arg Val Leu Pro Asn Gly
145 150 155 160
Ser Ala Gly Val Pro Asn Arg Glu Gly Leu Arg Tyr Tyr Arg Arg Leu
165 170 175
Leu Glu Arg Leu Arg Glu Leu Gly Val Gln Pro Val Val Thr Leu Tyr
180 185 190
His Trp Asp Leu Pro Gln Arg Leu Gln Asp Ala Tyr Gly Gly Trp Ala
195 200 205
Asn Arg Ala Leu Ala Asp His Phe Arg Asp Tyr Ala Glu Leu Cys Phe
210 215 220
Arg His Phe Gly Gly Gln Val Lys Tyr Trp Ile Thr Ile Asp Asn Pro
225 230 235 240
Tyr Val Val Ala Trp His Gly Tyr Ala Thr Gly Arg Leu Ala Pro Gly
245 250 255
Ile Arg Gly Ser Pro Arg Leu Gly Tyr Leu Val Ala His Asn Leu Leu
260 265 270
Leu Ala His Ala Lys Val Trp His Leu Tyr Asn Thr Ser Phe Arg Pro
275 280 285
Thr Gln Gly Gly Gln Val Ser Ile Ala Leu Ser Ser His Trp Ile Asn
290 295 300
Pro Arg Arg Met Thr Asp His Ser Ile Lys Glu Cys Gln Lys Ser Leu
305 310 315 320
Asp Phe Val Leu Gly Trp Phe Ala Lys Pro Val Phe Ile Asp Gly Asp
325 330 335
Tyr Pro Glu Ser Met Lys Asn Asn Leu Ser Ser Ile Leu Pro Asp Phe
340 345 350
Thr Glu Ser Glu Lys Lys Phe Ile Lys Gly Thr Ala Asp Phe Phe Ala
355 360 365
Leu Cys Phe Gly Pro Thr Leu Ser Phe Gln Leu Leu Asp Pro His Met
370 375 380
Lys Phe Arg Gln Leu Glu Ser Pro Asn Leu Arg Gln Leu Leu Ser Trp
385 390 395 400
Ile Asp Leu Glu Phe Asn His Pro Gln Ile Phe Ile Val Glu Asn Gly
405 410 415
Trp Phe Val Ser Gly Thr Thr Lys Arg Asp Asp Ala Lys Tyr Met Tyr
420 425 430
Tyr Leu Lys Lys Phe Ile Met Glu Thr Leu Lys Ala Ile Lys Leu Asp
435 440 445
Gly Val Asp Val Ile Gly Tyr Thr Ala Trp Ser Leu Met Asp Gly Phe
450 455 460
Glu Trp His Arg Gly Tyr Ser Ile Arg Arg Gly Leu Phe Tyr Val Asp
465 470 475 480
Phe Leu Ser Gln Asp Lys Met Leu Leu Pro Lys Ser Ser Ala Leu Phe
485 490 495
Tyr Gln Lys Leu Ile Glu Lys Asn Gly Phe Pro Pro Leu Pro Glu Asn
500 505 510
Gln Pro Leu Glu Gly Thr Phe Pro Cys Asp Phe Ala Trp Gly Val Val
515 520 525
Asp Asn Tyr Ile Gln Val Ser Gln Leu Thr Lys Pro Ile Ser Ser Leu
530 535 540
Thr Lys Pro Tyr His
545
<210> SEQ ID NO 16
<211> LENGTH: 549
<212> TYPE: PRT
<213> ORGANISM: homo sapiens
<300> PUBLICATION INFORMATION:
<308> DATABASE ACCESSION NUMBER: EAX08525
<309> DATABASE ENTRY DATE: 2006-12-18
<313> RELEVANT RESIDUES IN SEQ ID NO: (1)..(549)
<400> SEQUENCE: 16
Met Pro Ala Ser Ala Pro Pro Arg Arg Pro Arg Pro Pro Pro Pro Ser
1 5 10 15
Leu Ser Leu Leu Leu Val Leu Leu Gly Leu Gly Gly Arg Arg Leu Arg
20 25 30
Ala Glu Pro Gly Asp Gly Ala Gln Thr Trp Ala Arg Phe Ser Arg Pro
35 40 45
Pro Ala Pro Glu Ala Ala Gly Leu Phe Gln Gly Thr Phe Pro Asp Gly
50 55 60
Phe Leu Trp Ala Val Gly Ser Ala Ala Tyr Gln Thr Glu Gly Gly Trp
65 70 75 80
Gln Gln His Gly Lys Gly Ala Ser Ile Trp Asp Thr Phe Thr His His
85 90 95
Pro Leu Ala Pro Pro Gly Asp Ser Arg Asn Ala Ser Leu Pro Leu Gly
100 105 110
Ala Pro Ser Pro Leu Gln Pro Ala Thr Gly Asp Val Ala Ser Asp Ser
115 120 125
Tyr Asn Asn Val Phe Arg Asp Thr Glu Ala Leu Arg Glu Leu Gly Val
130 135 140
Thr His Tyr Arg Phe Ser Ile Ser Trp Ala Arg Val Leu Pro Asn Gly
145 150 155 160
Ser Ala Gly Val Pro Asn Arg Glu Gly Leu Arg Tyr Tyr Arg Arg Leu
165 170 175
Leu Glu Arg Leu Arg Glu Leu Gly Val Gln Pro Val Val Thr Leu Tyr
180 185 190
His Trp Asp Leu Pro Gln Arg Leu Gln Asp Ala Tyr Gly Gly Trp Ala
195 200 205
Asn Arg Ala Leu Ala Asp His Phe Arg Asp Tyr Ala Glu Leu Cys Phe
210 215 220
Arg His Phe Gly Gly Gln Val Lys Tyr Trp Ile Thr Ile Asp Asn Pro
225 230 235 240
Tyr Val Val Ala Trp His Gly Tyr Ala Thr Gly Arg Leu Ala Pro Gly
245 250 255
Ile Arg Gly Ser Pro Arg Leu Gly Tyr Leu Val Ala His Asn Leu Leu
260 265 270
Leu Ala His Ala Lys Val Trp His Leu Tyr Asn Thr Ser Phe Arg Pro
275 280 285
Thr Gln Gly Gly Gln Val Ser Ile Ala Leu Ser Ser His Trp Ile Asn
290 295 300
Pro Arg Arg Met Thr Asp His Ser Ile Lys Glu Cys Gln Lys Ser Leu
305 310 315 320
Asp Phe Val Leu Gly Trp Phe Ala Lys Pro Val Phe Ile Asp Gly Asp
325 330 335
Tyr Pro Glu Ser Met Lys Asn Asn Leu Ser Ser Ile Leu Pro Asp Phe
340 345 350
Thr Glu Ser Glu Lys Lys Phe Ile Lys Gly Thr Ala Asp Phe Phe Ala
355 360 365
Leu Cys Phe Gly Pro Thr Leu Ser Phe Gln Leu Leu Asp Pro His Met
370 375 380
Lys Phe Arg Gln Leu Glu Ser Pro Asn Leu Arg Gln Leu Leu Ser Trp
385 390 395 400
Ile Asp Leu Glu Phe Asn His Pro Gln Ile Phe Ile Val Glu Asn Gly
405 410 415
Trp Phe Val Ser Gly Thr Thr Lys Arg Asp Asp Ala Lys Tyr Met Tyr
420 425 430
Tyr Leu Lys Lys Phe Ile Met Glu Thr Leu Lys Ala Ile Lys Leu Asp
435 440 445
Gly Val Asp Val Ile Gly Tyr Thr Ala Trp Ser Leu Met Asp Gly Phe
450 455 460
Glu Trp His Arg Gly Tyr Ser Ile Arg Arg Gly Leu Phe Tyr Val Asp
465 470 475 480
Phe Leu Ser Gln Asp Lys Met Leu Leu Pro Lys Ser Ser Ala Leu Phe
485 490 495
Tyr Gln Lys Leu Ile Glu Lys Asn Gly Phe Pro Pro Leu Pro Glu Asn
500 505 510
Gln Pro Leu Glu Gly Thr Phe Pro Cys Asp Phe Ala Trp Gly Val Val
515 520 525
Asp Asn Tyr Ile Gln Val Ser Gln Leu Thr Lys Pro Ile Ser Ser Leu
530 535 540
Thr Lys Pro Tyr His
545
<210> SEQ ID NO 17
<211> LENGTH: 273
<212> TYPE: PRT
<213> ORGANISM: homo sapiens
<300> PUBLICATION INFORMATION:
<308> DATABASE ACCESSION NUMBER: CAC94767
<309> DATABASE ENTRY DATE: 2005-04-18
<313> RELEVANT RESIDUES IN SEQ ID NO: (1)..(273)
<400> SEQUENCE: 17
Met Pro Ala Ser Ala Pro Pro Arg Arg Pro Arg Pro Pro Pro Pro Ser
1 5 10 15
Leu Ser Leu Leu Leu Val Leu Leu Gly Leu Gly Gly Arg Arg Leu Arg
20 25 30
Ala Glu Pro Gly Asp Gly Ala Gln Thr Trp Ala Arg Phe Ser Arg Pro
35 40 45
Pro Ala Pro Glu Ala Ala Gly Leu Phe Gln Gly Thr Phe Pro Asp Gly
50 55 60
Phe Leu Trp Ala Val Gly Ser Ala Ala Tyr Gln Thr Glu Gly Gly Trp
65 70 75 80
Gln Gln His Gly Lys Gly Ala Ser Ile Trp Asp Thr Phe Thr His His
85 90 95
Pro Leu Ala Pro Pro Gly Asp Ser Arg Asn Ala Ser Leu Pro Leu Gly
100 105 110
Ala Pro Ser Pro Leu Gln Pro Ala Thr Gly Asp Val Ala Ser Asp Ser
115 120 125
Tyr Asn Asn Val Phe Arg Asp Thr Glu Ala Leu Arg Glu Leu Gly Val
130 135 140
Thr His Tyr Arg Phe Ser Ile Ser Trp Ala Arg Val Leu Pro Asn Gly
145 150 155 160
Ser Ala Gly Val Pro Asn Arg Glu Gly Leu Arg Tyr Tyr Arg Arg Leu
165 170 175
Leu Glu Arg Leu Arg Glu Leu Gly Val Gln Pro Val Val Thr Leu Tyr
180 185 190
His Trp Asp Leu Pro Gln Arg Leu Gln Asp Ala Tyr Gly Gly Trp Ala
195 200 205
Asn Arg Ala Leu Ala Asp His Phe Arg Asp Tyr Ala Glu Leu Cys Phe
210 215 220
Arg His Phe Gly Gly Gln Val Lys Tyr Trp Ile Thr Ile Asp Asn Pro
225 230 235 240
Tyr Val Val Ala Trp His Gly Tyr Ala Thr Gly Arg Leu Ala Pro Gly
245 250 255
Ile Arg Gly Ser Pro Arg Leu Gly Tyr Leu Val Ala His Asn Leu Leu
260 265 270
Leu
<210> SEQ ID NO 18
<211> LENGTH: 739
<212> TYPE: PRT
<213> ORGANISM: homo sapiens
<300> PUBLICATION INFORMATION:
<308> DATABASE ACCESSION NUMBER: CAC94773
<309> DATABASE ENTRY DATE: 2005-04-18
<313> RELEVANT RESIDUES IN SEQ ID NO: (1)..(739)
<400> SEQUENCE: 18
Ala His Ala Lys Val Trp His Leu Tyr Asn Thr Ser Phe Arg Pro Thr
1 5 10 15
Gln Gly Gly Gln Val Ser Ile Ala Leu Ser Ser His Trp Ile Asn Pro
20 25 30
Arg Arg Met Thr Asp His Ser Ile Lys Glu Cys Gln Lys Ser Leu Asp
35 40 45
Phe Val Leu Gly Trp Phe Ala Lys Pro Val Phe Ile Asp Gly Asp Tyr
50 55 60
Pro Glu Ser Met Lys Asn Asn Leu Ser Ser Ile Leu Pro Asp Phe Thr
65 70 75 80
Glu Ser Glu Lys Lys Phe Ile Lys Gly Thr Ala Asp Phe Phe Ala Leu
85 90 95
Cys Phe Gly Pro Thr Leu Ser Phe Gln Leu Leu Asp Pro His Met Lys
100 105 110
Phe Arg Gln Leu Glu Ser Pro Asn Leu Arg Gln Leu Leu Ser Trp Ile
115 120 125
Asp Leu Glu Phe Asn His Pro Gln Ile Phe Ile Val Glu Asn Gly Trp
130 135 140
Phe Val Ser Gly Thr Thr Lys Arg Asp Asp Ala Lys Tyr Met Tyr Tyr
145 150 155 160
Leu Lys Lys Phe Ile Met Glu Thr Leu Lys Ala Ile Lys Leu Asp Gly
165 170 175
Val Asp Val Ile Gly Tyr Thr Ala Trp Ser Leu Met Asp Gly Phe Glu
180 185 190
Trp His Arg Gly Tyr Ser Ile Arg Arg Gly Leu Phe Tyr Val Asp Phe
195 200 205
Leu Ser Gln Asp Lys Met Leu Leu Pro Lys Ser Ser Ala Leu Phe Tyr
210 215 220
Gln Lys Leu Ile Glu Lys Asn Gly Phe Pro Pro Leu Pro Glu Asn Gln
225 230 235 240
Pro Leu Glu Gly Thr Phe Pro Cys Asp Phe Ala Trp Gly Val Val Asp
245 250 255
Asn Tyr Ile Gln Val Asp Thr Thr Leu Ser Gln Phe Thr Asp Leu Asn
260 265 270
Val Tyr Leu Trp Asp Val His His Ser Lys Arg Leu Ile Lys Val Asp
275 280 285
Gly Val Val Thr Lys Lys Arg Lys Ser Tyr Cys Val Asp Phe Ala Ala
290 295 300
Ile Gln Pro Gln Ile Ala Leu Leu Gln Glu Met His Val Thr His Phe
305 310 315 320
Arg Phe Ser Leu Asp Trp Ala Leu Ile Leu Pro Leu Gly Asn Gln Ser
325 330 335
Gln Val Asn His Thr Ile Leu Gln Tyr Tyr Arg Cys Met Ala Ser Glu
340 345 350
Leu Val Arg Val Asn Ile Thr Pro Val Val Ala Leu Trp Gln Pro Met
355 360 365
Ala Pro Asn Gln Gly Leu Pro Arg Leu Leu Ala Arg Gln Gly Ala Trp
370 375 380
Glu Asn Pro Tyr Thr Ala Leu Ala Phe Ala Glu Tyr Ala Arg Leu Cys
385 390 395 400
Phe Gln Glu Leu Gly His His Val Lys Leu Trp Ile Thr Met Asn Glu
405 410 415
Pro Tyr Thr Arg Asn Met Thr Tyr Ser Ala Gly His Asn Leu Leu Lys
420 425 430
Ala His Ala Leu Ala Trp His Val Tyr Asn Glu Lys Phe Arg His Ala
435 440 445
Gln Asn Gly Lys Ile Ser Ile Ala Leu Gln Ala Asp Trp Ile Glu Pro
450 455 460
Ala Cys Pro Phe Ser Gln Lys Asp Lys Glu Val Ala Glu Arg Val Leu
465 470 475 480
Glu Phe Asp Ile Gly Trp Leu Ala Glu Pro Ile Phe Gly Ser Gly Asp
485 490 495
Tyr Pro Trp Val Met Arg Asp Trp Leu Asn Gln Arg Asn Asn Phe Leu
500 505 510
Leu Pro Tyr Phe Thr Glu Asp Glu Lys Lys Leu Ile Gln Gly Thr Phe
515 520 525
Asp Phe Leu Ala Leu Ser His Tyr Thr Thr Ile Leu Val Asp Ser Glu
530 535 540
Lys Glu Asp Pro Ile Lys Tyr Asn Asp Tyr Leu Glu Val Gln Glu Met
545 550 555 560
Thr Asp Ile Thr Trp Leu Asn Ser Pro Ser Gln Val Ala Val Val Pro
565 570 575
Trp Gly Leu Arg Lys Val Leu Asn Trp Leu Lys Phe Lys Tyr Gly Asp
580 585 590
Leu Pro Met Tyr Ile Ile Ser Asn Gly Ile Asp Asp Gly Leu His Ala
595 600 605
Glu Asp Asp Gln Leu Arg Val Tyr Tyr Met Gln Asn Tyr Ile Asn Glu
610 615 620
Ala Leu Lys Ala His Ile Leu Asp Gly Ile Asn Leu Cys Gly Tyr Phe
625 630 635 640
Ala Tyr Ser Phe Asn Asp Arg Thr Ala Pro Arg Phe Gly Leu Tyr Arg
645 650 655
Tyr Ala Ala Asp Gln Phe Glu Pro Lys Ala Ser Met Lys His Tyr Arg
660 665 670
Lys Ile Ile Asp Ser Asn Gly Phe Pro Gly Pro Glu Thr Leu Glu Arg
675 680 685
Phe Cys Pro Glu Glu Phe Thr Val Cys Thr Glu Cys Ser Phe Phe His
690 695 700
Thr Arg Lys Ser Leu Leu Ala Phe Ile Ala Phe Leu Phe Phe Ala Ser
705 710 715 720
Ile Ile Ser Leu Ser Leu Ile Phe Tyr Tyr Ser Lys Lys Gly Arg Arg
725 730 735
Ser Tyr Lys
User Contributions:
Comment about this patent or add new information about this topic: