Patent application title: POLYMER TUBE HEAT EXCHANGER
Inventors:
Georgios S. Zafiris (Glastonbury, CT, US)
Georgios S. Zafiris (Glastonbury, CT, US)
John P. Wesson (Vernon, CT, US)
Satyam Bendapudi (Syracuse, NY, US)
Brian L. Deperro (Buffalo, NY, US)
IPC8 Class: AF28F2106FI
USPC Class:
165173
Class name: Heat exchange side-by-side tubular structures or tube sections with manifold type header or header plate
Publication date: 2014-09-18
Patent application number: 20140262183
Abstract:
A heat exchanger includes a plurality of polymer tubes extending between
and fluidly connecting an inlet manifold and an outlet manifold, thereby
providing a flow path inside said tubes for a first fluid between the
inlet manifold and the outlet manifold. The tubes are configured to have
space between the exterior surfaces thereof, thereby providing a fluid
flow path for a second fluid through the space between the exterior
surfaces of the tubes.Claims:
1. A heat exchanger, comprising: a plurality of polymer tubes extending
between and fluidly connecting an inlet manifold and an outlet manifold,
thereby providing a flow path inside said tubes for a first fluid between
the inlet manifold and the outlet manifold; said plurality of tubes
configured to have space between the exterior surfaces thereof, thereby
providing a fluid flow path for a second fluid through the space between
the exterior surfaces of the tubes; said polymer tubes comprising
polyarylene sulfide, polyarylether ketone, aromatic polysulfone, aromatic
polyetherimide, or blends thereof.
2. The heat exchanger of claim 1, wherein said polymer tubes have a diameter of 1 mm to 10 mm and a wall thickness of 0.1 mm to 1 mm.
3. The heat exchanger of claim 1, wherein the polymer tubes further comprise a nanoparticle thermal conductivity enhancing additive.
4. The heat exchanger of claim 1, wherein the polymer tubes consist essentially of polyarylene sulfide, polyarylether ketone, aromatic polysulfone, aromatic polyetherimide, or blends thereof.
5. The heat exchanger of claim 1, wherein the polymer tubes comprise a plurality of co-extruded polymer layers wherein at least one of the polymer layers comprises polyarylene sulfide, polyarylether ketone, aromatic polysulfone, aromatic polyetherimide, or blends thereof.
6. The heat exchanger of claim 5, wherein at least one of the polymer layers comprises a fluoropolymer.
7. The heat exchanger of claim 5, wherein said at least one of the polymer layers consists essentially of polyarylene sulfide.
8. The heat exchanger of claim 1, wherein the polymer tubes have a diameter of 2 mm to 5 mm.
9. The heat exchanger of claim 1, wherein the polymer tubes have a thickness of 0.2 mm to 0.5 mm.
10. The heat exchanger of claim 1, wherein the tubes have a circular cross-section.
11. The heat exchanger of claim 1, wherein the tubes have an ovular or elliptical cross-section having an aspect ratio of greater than 1 and less than or equal to 5.
12. The heat exchanger of claim 1, wherein the tubes have a rectangular cross-section.
13. The heat exchanger of claim 1, wherein the tubes include an integrated support.
14. The heat exchanger of claim 13, wherein the integrated support is external.
15. The heat exchanger of claim 1, wherein the tubes comprise polyarylene sulfide or polyarylether ketone having a crystallinity of 10% to 85%.
16. The heat exchanger of claim 1, wherein the heat exchanger is a module wherein the tubes and the inlet and outlet manifolds are all injection molded in a single injection molding shot.
17. The heat exchanger of claim 16, comprising a plurality of said heat exchanger modules stacked together.
18. The heat exchanger of claim 17, wherein adjacent modules are connected with a gasket to produce a fluid-tight seal, and are fastened together mechanically or adhesively.
19. The heat exchanger of claim 17, wherein modules on each end of the stack include a solid end-wall as part of their injection molded structure.
20. A method of making a heat exchanger tube, comprising extruding a thermoplastic composition comprising polyarylene sulfide or polyarylether ketone, and heating the extruded tube with radiant or convective heating under tension to increase crystallinity of the polyarylene sulfide or polyarylether ketone to 10% to 85%.
Description:
BACKGROUND OF THE INVENTION
[0001] The subject matter disclosed herein generally relates to heat exchangers and, more particularly, to heat exchangers having polymer tubes.
[0002] Heat exchangers are widely used in various applications, including but not limited to heating and cooling systems including hydronic fan coil units, heating and cooling in various industrial and chemical processes, heat recovery systems, and the like, to name but a few. Many heat exchangers for transferring heat from one fluid to another fluid utilize one or more tubes through which one fluid flows while a second fluid flows around the tubes. Heat from one of the fluids is transferred to the other fluid by conduction through the tube wall.
[0003] Heat exchanger tubes may be made from a variety of materials, including metals such as aluminum or copper. Metal heat exchanger tubes offer a number of different advantages, including high thermal conductivity and strength. However, metal heat exchanger tubes also suffer from a number of problems. For example, metal heat exchanger tubes can be expensive and time-consuming to manufacture, are subject to corrosion from certain types of fluids, are and can impose limitations on the design of the heat exchanger due to the limitations of metal fabrication techniques. Nylon plastic tubes have also been used for heat exchangers. Plastic tube heat exchangers, however, have not achieved widespread acceptance due to a number of problems such as low thermal conductivity and/or the necessity of high wall thicknesses in order to provide physical robustness to tubes under a wide variety of operating conditions such as high temperatures and/or pressures (e.g., in hydronic building heating systems, water temperatures can reach temperatures of 90° C. and pressures of 3100 kPa). Composite tubes have also been proposed that use plastic as part of the composite; however, such tubes can be expensive and difficult to manufacture, and composite tubes also may require high wall thicknesses to accommodate the various materials of the composite.
[0004] Therefore, there continues to be a need for new materials and/or configurations for heat exchanger tubes that offer benefits including but not limited to improved performance, ease of manufacture, and/or reduced cost.
BRIEF DESCRIPTION OF THE INVENTION
[0005] According to one aspect of the invention, a heat exchanger includes a plurality of polymer tubes extending between and fluidly connecting an inlet manifold and an outlet manifold, thereby providing a flow path inside said tubes for a first fluid between the inlet manifold and the outlet manifold. The tubes are configured to have space between the exterior surfaces thereof, thereby providing a fluid flow path for a second fluid through the space between the exterior surfaces of the tubes. The tubes further comprise polyarylene sulfide, polyarylether ketone, aromatic polysulfone, aromatic polyetherimide, or blends thereof. In an exemplary embodiment, the tubes have a diameter of 1 mm to 10 mm and a wall thickness of 0.1 mm to 1 mm.
BRIEF DESCRIPTION OF THE DRAWINGS
[0006] The subject matter which is regarded as the invention is particularly pointed out and distinctly claimed in the claims at the conclusion of the specification. The foregoing and other features, and advantages of the invention are apparent from the following detailed description taken in conjunction with the accompanying drawings in which:
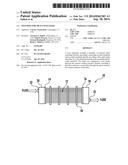
[0007] FIG. 1 is a schematic diagram depicting a front view of the tube portion of an exemplary heat exchanger;
[0008] FIG. 2 is a schematic diagram depicting a top view of the tube portion of the exemplary heat exchanger of FIG. 1;
[0009] FIG. 3 is a schematic diagram depicting an end view of the tube portion of the exemplary heat exchanger of FIG. 1;
[0010] FIGS. 4A-C depict a schematic diagram of an exemplary heat exchanger known as a spiral heat exchanger;
[0011] FIGS. 5A-B depict a schematic diagram of an exemplary heat exchanger known as a plate heat exchanger
[0012] FIG. 6 is a schematic diagram depicting a cross-sectional view of an exemplary tube structure having an integrated support; and
[0013] FIG. 7 is a schematic diagram depicting a cross-sectional view of an exemplary tube structure having an integrated support.
DETAILED DESCRIPTION OF THE INVENTION
[0014] The polymer tubes described herein are formed from a thermoplastic composition comprising polyarylene sulfide, polyarylether ketone, aromatic polysulfone, aromatic polyetherimide, or blends thereof. Each of these polymer families is known in the art and is commercially available in different variations. Polyarylene sulfide, as used herein, includes any polyarylene sulfide-series resin (polyarylene thioether-series resin), and may be a homopolymer or copolymer having a polyarylene sulfide backbone having --(Ar--S--)-- repeat units wherein Ar represents an arylene group. Exemplary arylene groups include p-phenylene, m-phenylene, o-phenylene group, substituted phenylene groups (e.g., an alkylphenylene group having substituent(s) such as a C1-5 alkyl group, or an arylphenylene group having substituent(s) such as phenyl group), p,p'-diphenylenesulfone, p,p'-biphenylene, p,p'-diphenylene ether, and p,p'-diphenylenecarbonyl. The polyarylene sulfide-series resin may be a homopolymer of a single repeat unit such as any of the above-mentioned repeat units, or may be a copolymer containing different repeat units such as any of the above-mentioned repeat units. In an exemplary embodiment, the polyarylene sulfide comprises at least 70 mol % p-phenylene sulfide repeat units. In another exemplary embodiment, the polyarylene sulfide comprises at least 90 mol % p-phenylene sulfide repeat units. In yet another exemplary embodiment, the polyarylene sulfide resin is a poly(p-phenylene sulfide) homopolymer. The polyarylene sulfide resin may be obtained by allowing a relative low molecular weight of a linear polymer to crosslink with oxidation, or a high molecular weight polymer having a substantially linear structure which may be obtained by condensation polymerization from a bifunctional monomer. In another exemplary embodiment, a branched or crosslinked polyarylene sulfide resin obtained by a polymerization of the monomer in combination with a monomer having functional groups. In yet another exemplary embodiment, the polyarylene sulfide may be a polybiphenylene sulfide (PBPS), a polyarylene sulfide ketone (PPSK), a polybiphenylene sulfide sulfone (PPSS), or other polyarylene sulfide variants as known in the art. In yet another exemplary embodiment, the polyarylene sulfide can be blended or alloyed with an elastomer. Exemplary number average molecular weights of the polyphenylene sulfide-series resin can range from about 300 to 30×104, more specifically about 400 to 10×104. The polyphenylene sulfide polymers are commercially available from Chevron Phillips Chemicals and Ticona Celanese under the brand names RYTON® and FORTRON®, respectively. The polyphenylene sulfide polymer alloys and blends are commercially available from Chevron Phillips Chemicals under the brand name XTEL.
[0015] As used herein, the term polyaryl ether ketones (PAEK) includes any of several polymer types containing aromatic rings, usually phenyl rings, linked primarily by ketone and ether groups in different sequences. Examples of PAEK resins include, but are not limited to, polyether ketones (PEK), polyether ether ketones (PEEK), polyether ketone ether ketone ketones (PEKEKK) and polyether ketone ketones (PEKK) and copolymers containing such groups as well as blends thereof. The PAEK polymers may comprise monomer units containing an aromatic ring, usually a phenyl ring, a ketone group and an ether group in any sequence. Low levels, for example less than 10 mole %, of addition linking groups may be present as long as they do not fundamentally alter the properties of the PAEK resin. PEEK is commercially available from Victrex Ltd. as VICTREX® PEEK. PEKEKK resin is commercially available from BASF as ULTRAPEK®. Polyether ether ketone resins are also available under the GATONE® and KETASPIRE® trade names from Solvay Co. and Solvay Advanced Polymers.
[0016] In some instances the first resin component is a polymer selected from the group consisting of at least one of polyaryl ether ketones, polyaryl ketones, polyether ketones and polyether ether ketones. Any of the PAEK polymers can be used which will have improved properties through blending with a second resin component according to the present invention. This encompasses the use of one or more polymers from any of the several classes of PAEK resins described herein or described in the references cited herein.
[0017] For example, several polyaryl ether ketones which are highly crystalline, with melting points above 300° C., can be used in blends according to the present invention. Examples of these crystalline polyaryl ether ketones are shown in the structures (I), (II), (III), (IV) and (V) below.
##STR00001##
[0018] The formation and properties of polyaryl ether ketones is well known in the art. For example, the electrophilic aromatic substitution (e.g., Friedel-Crafts catalyzed) reaction of aromatic diacyl halides with unsubstituted aromatic compounds such as diphenyl ether some of the early work is disclosed in, for example, U.S. Pat. No. 3,065,205. A broad range of resins can be formed, for example, by the nucleophilic aromatic substitution reaction of an activated aromatic dihalide and an aromatic diol or salt thereof, as shown, for example, in U.S. Pat. No. 4,175,175.
[0019] Other examples of crystalline polyaryl ether ketones which are suitable for use herein can be generically characterized as containing repeating units of the following formula (VI):
##STR00002##
wherein Ar is independently a divalent aromatic radical selected from phenylene, biphenylene or naphthylene, X is independently --O--, --C(O)--, --O--Ar--C(O)--, --S--, --SO2-- or a direct bond, and n is an integer of from 0 to about 10.
[0020] PAEK resins can be prepared by methods well known in the art. One such method comprises heating a substantially equimolar mixture of at least one bisphenol, often reacted as its bis-phenolate salt, and at least one of either a dihalobenzoid compound or, in other cases, at least one halophenol compound may be reacted to form polymer. In other instances mixtures of these compounds may be used. For example hydroquinone can be reacted with a dihalo aryl ketone, such a dichloro benzophenone of difluoro benzophenone to form a polyaryl ether ketone. In other cases dihydroxy aryl ketone, such as dihydroxy benzophenone can be polymerized with aryl dihalides such as dichloro benzene to form PAEK resins. In still other instances dihydroxy aryl ethers, such as dihydroxy diphenyl ether can be reacted with dihalo aryl ketones, such as difluoro benzophenone. In other variations dihydroxy compounds with no ether linkages, such as or dihydroxy biphenyl or hydroquinone may be reacted with dihalo compounds which may have both ether and ketone linkages, for instance bis-(dichloro phenyl)benzophenone. In other instances diaryl ether carboxylic acids or carboxylic acid halides can be polymerized to form polyaryl ether ketones. Examples of such compounds are diphenylether carboxylic acid, diphenyl ether carboxylic acid chloride, phenoxy-phenoxy benzoic acid, and mixtures thereof. In still other instances dicarboxylic acids or dicarboxylic acid halides can be condensed with diaryl ethers, for example iso- or tere-phthaloyl chlorides (or mixtures thereof) can be reacted with diphenyl ether, to form PAEK resins.
[0021] Polyaryl ether ketones may be produced by various processes, including the process as described in, for example, U.S. Pat. No. 4,176,222. This process comprises heating in the temperature range of 100 to 400° C., a substantially equimolar mixture of: (a) at least one bisphenol; and, (b.i) at least one dihalobenzenoid compound, and/or (b.ii) at least one halophenol, in which in the dihalobenzenoid compound or halophenol, the halogen atoms are activated by --C═O-- groups ortho or para thereto, with a mixture of sodium carbonate or bicarbonate and a second alkali metal carbonate or bicarbonate, the alkali metal of said second alkali metal carbonate or bicarbonate having a higher atomic number than that of sodium, the amount of the second alkali metal carbonate or bicarbonate being such that there are 0.001 to 0.2 gram atoms of the alkali metal of higher atomic number per gram atom of sodium, the total amount of alkali metal carbonate or bicarbonate being such that there is at least one alkali metal atom for each phenol group present, and thereafter separating the polymer from the alkali metal halide.
[0022] Polyaryl ether ketones may also be prepared by other processes. One example is a process in which reactants such as: (a) a dicarboxylic acid; (b) at least one divalent aromatic radical and at least one mono aromatic dicarboxylic acid and, (c) combinations of (a) and (b), are reacted in the presence of a fluoroalkane sulfonic acid, particularly trifluoromethane sulfonic acid. See, for example, U.S. Pat. No. 4,396,755. Additional polyaryl ether ketones may be prepared according to a process wherein aromatic diacyl compounds are polymerized with at least one aromatic compound and at least one mono acyl halide as described in, for example, U.S. Pat. No. 4,398,020. PAEK resin weight average molecular weight (Mw) may vary from 5,000 to 150,000 g/mole, specifically from about 10,000 to 80,000 g/mole
[0023] Aromatic polysulfone, as used herein includes any polymer having repeat units, a portion of which includes the structure (VI)
##STR00003##
In addition to this structure, the aromatic polysulfone backbone can include a number of other connecting units, including an isopropylidene linkage that is part of the repeat unit of the well-known bisphenol A-based polysulfone. In an exemplary embodiment, the aromatic polysulfone has the structure (VII)
##STR00004##
wherein X represents a divalent alkylene group of 1 to 6 carbon atoms (e.g., --C(CH3)n-- where n=1-6), a divalent sulfonyl group, or a direct bond.
[0024] There are several routes for the synthesis of polysulfones, but the one that has proved to be most practical and versatile over the years is by aromatic nucleophilic substitution. This polycondensation route is based on reaction of approximately equimolar quantities of 4,4'-dihalodiphenylsulfone (usually dichlorodiphenylsulfone (DCDPS)) with a bisphenol in the presence of a base to form aromatic ether bonds and eliminate alkali salt as a by-product. This route is widely used for the commercial manufacture of polysulfones on a commercial scale. Typical polysulfones include structures (VIII), (IX), and (x) below:
##STR00005##
nown as polysulfone, polyphenylenesulfone, and polyethersulfone, respectively. As with the above-described polyarylene sulfide and PAEK polymers, crosslinking or other functional groups and substituents can be included in the polymer structure to achieve target properties, as is known in the art. Exemplary molecular weights for aromatic polysulfones can range from 40,000 to 80,000, more specifically 50,000 to 70,000.
[0025] Aromatic polyetherimide, as used herein, includes any polymer that comprises repeat units of the formula (XI)
##STR00006##
wherein T is --O-- or a group of the formula --O--Z--O-- wherein the divalent bonds of the --O-- or the --O--Z--O-- group are in the 3,3', 3,4', 4,3', or the 4,4' positions, and wherein Z is defined above. In one embodiment, the polyimide, polyetherimide or polyetherimide sulfone may be a copolymer. Mixtures of the polyimide, polyetherimide or polyetherimide sulfone may also be employed.
[0026] The polyetherimide can be prepared by any of the methods well known to those skilled in the art, including the reaction of an aromatic bis(ether anhydride) of the formula (XII)
##STR00007##
with an organic diamine of the formula (XIII)
H2N--R1--NH2 (Formula XIII)
wherein T and R1 are defined as described above.
[0027] Examples of specific aromatic bis anhydrides and organic diamines are disclosed, for example, in U.S. Pat. Nos. 3,972,902 and 4,455,410. Illustrative examples of aromatic bis anhydrides include:
[0028] 3,3-bis[4-(3,4-dicarboxyphenoxy)phenyl]propane dianhydride;
[0029] 4,4'-bis(3,4-dicarboxyphenoxy)diphenyl ether dianhydride;
[0030] 4,4'-bis(3,4-dicarboxyphenoxy)diphenyl sulfide dianhydride;
[0031] 4,4'-bis(3,4-dicarboxyphenoxy)benzophenone dianhydride;
[0032] 4,4'-bis(3,4-dicarboxyphenoxy)diphenyl sulfone dianhydride;
[0033] 2,2-bis[4-(2,3-dicarboxyphenoxy)phenyl]propane dianhydride;
[0034] 4,4'-bis(2,3-dicarboxyphenoxy)diphenyl ether dianhydride;
[0035] 4,4'-bis(2,3-dicarboxyphenoxy)diphenyl sulfide dianhydride;
[0036] 4,4'-bis(2,3-dicarboxyphenoxy)benzophenone dianhydride;
[0037] 4,4'-bis(2,3-dicarboxyphenoxy)diphenyl sulfone dianhydride;
[0038] 4-(2,3-dicarboxyphenoxy)-4'-(3,4-dicarboxyphenoxy)diphenyl-2,2-propane dianhydride;
[0039] 4-(2,3-dicarboxyphenoxy)-4'-(3,4-dicarboxyphenoxy)diphenyl ether dianhydride;
[0040] 4-(2,3-dicarboxyphenoxy)-4'-(3,4-dicarboxyphenoxy)diphenyl sulfide dianhydride;
[0041] 4-(2,3-dicarboxyphenoxy)-4'-(3,4-dicarboxyphenoxy)benzophenone dianhydride; and,
[0042] 4-(2,3-dicarboxyphenoxy)-4'-(3,4-dicarboxyphenoxy)diphenyl sulfone dianhydride, as well as various mixtures thereof.
[0043] Another class of aromatic bis(ether anhydride)s included by formula (XII) above includes, but is not limited to, compounds wherein T is of the formula (XIV)
##STR00008##
and the ether linkages, for example, are preferably in the 3,3', 3,4', 4,3', or 4,4' positions, and mixtures thereof, and where Q is as defined above.
[0044] Any diamino compound may be employed. Examples of suitable compounds are ethylenediamine, propylenediamine, trimethylenediamine, diethylenetriamine, triethylenetertramine, hexamethylenediamine, heptamethylenediamine, octamethylenediamine, nonamethylenediamine, decamethylenediamine, 1,12-dodecanediamine, 1,18-octadecanediamine, 3-methylheptamethylenediamine, 4,4-dimethylheptamethylenediamine, 4-methylnonamethylenediamine, 5-methylnonamethylenediamine, 2,5-dimethylhexamethylenediamine, 2,5-dimethylheptamethylenediamine, 2,2-dimethylpropylenediamine, N-methyl-bis(3-aminopropyl)amine, 3-methoxyhexamethylenediamine, 1,2-bis(3-aminopropoxy)ethane, bis(3-aminopropyl) sulfide, 1,4-cyclohexanediamine, bis-(4-aminocyclohexyl)methane, m-phenylenediamine, p-phenylenediamine, 2,4-diaminotoluene, 2,6-diaminotoluene, m-xylylenediamine, p-xylylenediamine, 2-methyl-4,6-diethyl-1,3-phenylene-diamine, 5-methyl-4,6-diethyl-1,3-phenylene-diamine, benzidine, 3,3'-dimethylbenzidine, 3,3'-dimethoxybenzidine, 1,5-diaminonaphthalene, bis(4-aminophenyl)methane, bis(2-chloro-4-amino-3,5-diethylphenyl)methane, bis(4-aminophenyl) propane, 2,4-bis(p-amino-t-butyl) toluene, bis(p-amino-t-butylphenyl)ether, bis(p-methyl-o-aminophenyl)benzene, bis(p-methyl-o-aminopentyl)benzene, 1,3-diamino-4-isopropylbenzene, bis(4-aminophenyl) sulfide, bis(4-aminophenyl) sulfone, and bis(4-aminophenyl)ether. Mixtures of these compounds may also be used. Exemplary diamino compounds include aromatic diamines, specifically m- and p-phenylenediamine, sulfonyl dianiline and mixtures thereof.
[0045] In one embodiment, the polyetherimide resin comprises structural units according to formula (XI) wherein each R is independently p-phenylene or m-phenylene or a mixture thereof and T is a divalent radical of the formula (XV)
##STR00009##
[0046] Included among the many methods of making polyetherimides, are those disclosed in U.S. Pat. Nos. 3,847,867, 3,852,242, 3,803,085, 3,905,942, 3,983,093, and 4,443,591, the disclosures of which are incorporated herein by reference in their entirety. These patents mentioned for the purpose of teaching, by way of illustration, general and specific methods for preparing polyimides such as the polyetherimides described herein. Polyimides, polyetherimides and polyetherimide sulfones may have a melt index of about 0.1 to about 10 grams per minute (g/min), as measured by American Society for Testing Materials (ASTM) D1238 at 340 to about 370° C., using a 6.6 kilogram (kg) weight. In a one embodiment, the polyetherimide resin has a weight average molecular weight (Mw) of about 10,000 to about 150,000 grams per mole (g/mole), as measured by gel permeation chromatography, using a polystyrene standard. In another embodiment the polyetherimide has Mw of 20,000 to 60,000. Such polyetherimide resins typically have an intrinsic viscosity greater than about 0.2 deciliters per gram (dl/g), or, more specifically, about 0.35 to about 0.7 dl/g as measured in m-cresol at 25° C. Examples of some polyetherimides useful in blends described herein are listed in ASTM D5205 "Standard Classification System for Polyetherimide (PEI) Materials".
[0047] The composition can be further modified, if desired, by including one or more additives often used with thermoplastic polymers, some of which may serve to beneficially affect the chemical and physical properties. Examples are mineral fillers, including clay, reinforcing agents, for example, glass fibers, flakes or spheres, plasticizers, flame retardants, stabilizers, antioxidants, colorants, processing aids, and the like as known in the art. In an exemplary embodiment, the resin composition includes a nanoparticle additive for enhancing the thermal conductivity of the composition. Examples of such additives include aluminum nanoflakes or carbon nanoparticles, with particle sizes ranging from about 10 nm to about 250 nm, more specifically about 50 nm to about 200 nm, and even more specifically from about 50 nm to about 100 nm.
[0048] The resin compositions used in the present invention can be prepared and/or formed into tubes by a variety of methods involving intimate admixing of the materials with any additional additives desired in the formulation. Suitable procedures include solution blending and melt blending. Because of the availability of melt blending equipment in commercial polymer processing facilities, melt processing procedures may be readily used. Examples of equipment used in such melt compounding methods include: co-rotating and counter-rotating extruders, single screw extruders, disc-pack processors and various other types of extrusion equipment. In some instances, the compounded material exits the extruder through appropriately-shaped holes in a die and the resulting tubes are cooled by air or by passing through a water bath. The cooled tubes can then be cut to an appropriate length. In some exemplary embodiments, rapid cooling of the tubes can result in low levels of crystallinity that adversely impact physical properties (e.g., dimensional stability) of the polymer tube. In such cased, the above-described polymer compositions can be adjusted by post-production processing such as annealing at elevated temperatures to produce target crystallinities or other properties. Annealing temperatures can range from about 80° C. to about 180° C. Exemplary polymer compositions for which such annealing may be used include polyphenylene sulfides and/or polyarylether ketones, with which annealing can be used to produce target crystallinities of about 10% to about 85% (more specifically about 30% to about 80%), and the annealing temperature can vary with the choice of polymer (e.g., useful annealing temperatures for polyphenylene sulfides can range from about 80° C. to about 130° C. Annealing can be carried out with conventional heat sources such as radiant or convective heat sources either on-line with the tube extrusion process or off-line at some point in time after the tubes have been manufactured.
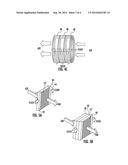
[0049] An exemplary heat exchanger is shown in schematic diagram form in FIG. 1. The particular configuration of heat exchanger is not critical, as any configuration that can utilize the tubes described herein can be used. Heat exchangers with which the invention can be practiced include, but are not limited to shell and tube heat exchangers, fan coil heat exchangers, spiral heat exchangers, and plate heat exchangers. FIG. 1 depicts a heat exchanger 10 such as for use in a typical fan coil heat exchanger unit. As shown in FIG. 1, a bank of tubes 12 extends between end plates 14 and 16. Tube supports 18 are disposed at positions between the end plates and have openings therein through which the tubes 12 pass. Inlet plenum 20 having inlet 22 receives a first fluid (e.g., water, ethylene glycol, or the like), which flows through the tubes 12 and out into an outlet plenum 24, from where it flows through outlet 25 and out of the heat exchanger. FIG. 2 depicts a top view of the heat exchanger 10, and shows the flow of a second fluid (e.g., air) between the tubes of the tube bank. FIG. 3 depicts an end view of the heat exchanger 10 taken at a cross-section through or at the end plate 14. End plate 14 has a number of holes 26 therein that receive the tubes 12. The tubes are sealingly attached to the end plate by conventional techniques, including but not limited to the use of an adhesive, laser welding, ultrasonic welding, or other thermoplastic welding techniques such as magnetic induction or microwave welding performed by magnetic induction or microwave heating of metal particle additives in the tube and/or end plate polymer compositions.
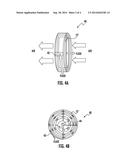
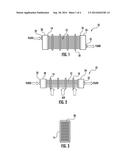
[0050] Alternative exemplary embodiments of heat exchangers are shown in FIGS. 4 and 5. FIGS. 4A and 4B depict side and end views, respectively, of a spiral heat exchanger 40 having a continuous spiral tube 12' with inlet 42 and outlet 44. Heat transfer fluid enters the spiral tube 12' at inlet 42 at the perimeter of the spiral and exits outlet 44 at the center of the spiral while air to be heated or cooled by the heat transfer fluid is directed in a perpendicular direction across the spiral tube 12'. Multiple spiral heat exchangers may be stacked in modular fashion as shown in FIG. 4C. FIG. 5A schematically depicts a plate heat exchanger 50 having heat transfer tubes 12'', inlet portion 52, and outlet portion 54. The plate heat exchanger is distinguished from the heat exchanger of FIG. 1 in that the tubes 12'' have a rectangular cross-section, and in that the entire assembly 50 can be injection molded in a single injection molding shot process. Multiple plate heat exchangers may be stacked in modular fashion as shown in FIG. 5B having two end modules 50' and one or more middle modules 50''. In an exemplary embodiment, stacked heat exchanger modules have gaskets between them and are fastened together with adhesive or with mechanical connections such as a threaded nut and bold connector or other known connectors. In another exemplary embodiment, modules on each end of the stack include a solid end wall as part of their injection molded structure.
[0051] The polymer tubes 12 may have different cross-sectional configurations, including but not limited to circular, ovular, elliptical, square, or rectangular. Tubes with ovular or elliptical cross-sections can have a cross-sectional aspect ratio of greater than 1 and less than or equal to 4. The wall thickness of the tubes can range from about 0.1 mm to about 1 mm, and more specifically from about 0.2 mm to about 0.5 mm. The diameter of circular tubes can range from about 1 mm to about 10 mm, and more specifically from about 2 mm to about 5 mm, and ovular or elliptical tubes can be sized to have cross-sectional areas equivalent to circular tubes with such diameters. Tubes can be formed using conventional extrusion techniques that are well-known in the art and do not require further detailed explanation herein. In another exemplary embodiment the liquid carrying channels can be of rectangular cross-section and of the above sizes and wall thicknesses, and are formed by injection molding along with the manifolds in one injection molding shot, such as for the plate heat exchanger of FIG. 5.
[0052] In some exemplary embodiments, a composition comprising the above-described polyarylene sulfide, polyarylether ketone, aromatic polysulfone, aromatic polyetherimide polymers is co-extruded as one or more layers in a multi-layer tube. In a further exemplary embodiment, a multi-layer tube also includes a barrier layer such as a fluoropolymer (e.g., polyvinylidene fluoride (PVDF), ethylene tetrafluoroethylene (ETFE), perfluoroalkoxy (PFA) resins, or polytetrafluoroethylene (PTFE). The adhesion of some fluoropolymers to adjacent layer(s) of a multi-layer tube structure can be enhanced through the inclusion of tie layers of adhesion-promoting polymers adjacent to the fluoropolymer layer(s) (e.g., PFA or ETFE, or polymer blends containing a fluoropolymer and one or more other polymers compatible with both the fluoropolymer and the adjacent polymer layers).
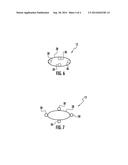
[0053] In another exemplary embodiment, the tubes can be extruded with integrated supports. Such supports can provide strength and integrity to the tube, and may be particularly useful for non-circular cross-section tubes. Integrated supports can be of any cross-sectional shape, including circular or rectangular, and can be internal or external to the tube. Internal supports may extend part way across the diameter of the tube or may be a web structure that extends completely across the tube diameter. The cross-sections of exemplary tubes with an integrated support structure are shown in FIG. 4 (internal support structure) and FIG. 5 (external support structure), where a tube 14 comprises a tube wall 28 and integrated support structures 30.
[0054] While the invention has been described in detail in connection with only a limited number of embodiments, it should be readily understood that the invention is not limited to such disclosed embodiments. Rather, the invention can be modified to incorporate any number of variations, alterations, substitutions or equivalent arrangements not heretofore described, but which are commensurate with the spirit and scope of the invention. Additionally, while various embodiments of the invention have been described, it is to be understood that aspects of the invention may include only some of the described embodiments. Accordingly, the invention is not to be seen as limited by the foregoing description, but is only limited by the scope of the appended claims.
User Contributions:
Comment about this patent or add new information about this topic: