Patent application title: MODIFIED METAL SURFACE AND METHOD FOR PREPARING THE SAME USING AN ELECTROCHEMICAL PROCESS
Inventors:
Sasha Omanovic (Brossard, CA)
Jeffrey Harvey (Orleans, CA)
Hesam Dadafarin (Montreal, CA)
IPC8 Class: AC25D902FI
USPC Class:
205 50
Class name: Electrolysis: processes, compositions used therein, and methods of preparing the compositions product produced by electrolysis involving electrolytic marking, battery electrode active material forming, electroforming, or electrolytic coating
Publication date: 2012-04-12
Patent application number: 20120085652
Abstract:
The present invention relates to a metal or alloy substrate having a
modified surface, and more particularly a stainless steel substrate,
produced by the use of an electrochemical process. The invention also
relates generally to a method for producing a stable and covalent
association between self-assembled layers and a surface of a metal
substrate, and more particularly a stainless steel substrate. The metal
or alloy substrate, and more particularly the stainless steel substrate,
is modified by the covalent attachment of self-assembled monolayers on
the surface by using an electrochemical process. These functionalized
self-assembled monolayers provide a link to irreversibly attach or
immobilize an active or nonactive agent.Claims:
1. A method for attaching self-assembled monolayers onto a surface of a
stainless steel substrate comprising contacting said surface with a
solution comprising an alkanethiol compound, and electrochemically
depositing said alkanethiol compound on the surface using a nickel-based
alloy auxiliary electrode to covalently attach the self-assembly
monolayers onto said surface.
2. The method of claim 1, wherein the surface is pretreated by polishing said surface and/or etching said surface with oxalic acid.
3. The method of claim 1, wherein the solution is an ionically-conductive aqueous or an ethanolic electrolyte solution.
4. The method of claim 1, wherein the nickel-based alloy auxiliary electrode is a nichrome or a nickel auxiliary electrode.
5. The method of claim 1, wherein the self-assembled monolayers are formed by a cyclic application of a polarization potential.
6. The method of claim 5, wherein the polarization potential is between -1.8 volts and 0 volt vs. SCE (Saturated Calomel Electrode) and is applied for at least 25 cycles, preferably for 1 to 150 cycles.
7. The method of claim 1, wherein the solution is preconditioned by a cyclic potentiodynamic polarization of a nickel-based alloy auxiliary and working electrode.
8. The method of claim 7, wherein the polarization is performed at a scan rate of 100 mV/s between -1.8 volts and 0 volt vs. SCE for 50 to 400 cycles and for, preferably, 250 cycles.
9. The method of claim 1, further comprising covalently attaching to an end group of the self-assembled monolayers a nonactive agent or an active agent selected from the group consisting of proteins, pharmaceuticals, drugs, polymers, antibodies, bioorganic molecules, biologically active agents, preferably fibronectin, and metal nanoparticles.
10. The method of claim 9, wherein the end group of the self-assembled monolayers is chemically modified before covalently attaching the nonactive agent or the active agent.
11. A method for attaching self-assembled monolayers onto a surface of a metal substrate comprising contacting said surface with a solution comprising an alkanethiol compound, and electrochemically depositing said alkanethiol compound on the surface using a nickel-based alloy auxiliary electrode to covalently attach the self-assembly monolayers onto said surface.
12. The method of claim 11, wherein the substrate is nickel titanium (NiTi).
13. The method of claim 11, wherein the substrate is cobalt-chrome alloy (CoCr).
14. A stainless steel substrate having a modified surface comprising self-assembled monolayers covalently attached to the surface, wherein said self-assembled monolayers are prepared by contacting said surface with a solution comprising an alkanethiol compound, and electrochemically depositing said alkanethiol compound on the surface using a nickel-based alloy auxiliary electrode.
15. The stainless steel substrate of claim 14, further comprising a nonactive agent or an active agent covalently attached to an end group of the self-assembled monolayers, the active agent being selected from the group consisting of proteins, pharmaceuticals, drugs, polymers, antibodies, bioorganic molecules, biologically active agents, preferably fibronectin, and metal nanoparticles.
16. A metal substrate having a modified surface comprising self-assembled monolayers covalently attached to the metal surface, wherein said self-assembled monolayers are prepared by contacting said metal surface with a solution comprising an alkanethiol compound, and electrochemically depositing said alkanethiol compound on the metal surface using a nickel-based alloy auxiliary electrode.
17. The metal substrate of claim 16, wherein the substrate is nickel titanium (NiTi).
18. The metal substrate of claim 16, wherein the substrate is cobalt-chrome alloy (CoCr).
19. The metal substrate of claim 16, further comprising a nonactive agent or an active agent covalently attached to an end group of the self-assembled monolayers, the active agent being selected from the group consisting of proteins, pharmaceuticals, drugs, polymers, antibodies, bioorganic molecules, biologically active agents, preferably fibronectin, and metal nanoparticles.
20. A medical device having a metal substrate with a modified surface comprising self-assembled monolayers covalently attached to the metal surface, wherein said self-assembled monolayers are prepared by contacting said surface with a solution comprising an alkanethiol compound, and electrochemically depositing said alkanethiol compound on the surface using a nickel-based alloy auxiliary electrode.
Description:
CROSS-REFERENCED TO RELATED APPLICATION
[0001] This application claims the benefit of U.S. Provisional Patent Application No. 61/391,335, filed Oct. 8, 2010, which is hereby incorporated by reference.
FIELD
[0002] The invention relates generally to a metal or metal alloy substrate having a surface modified by an electrochemical process, and more particularly to a metal or metal alloy substrate of a medical device, such as stainless steel. The invention also relates generally to a method for producing a stable and covalent association between self-assembled monolayers and a surface of a metal or metal alloy substrate, such as stainless steel, using an electrochemical process.
BACKGROUND
[0003] Stainless steel, cobalt chrome (cobalt chromium) alloys, titanium, titanium alloys, nitinol, platinum, iridium and gold are metallic materials used in medical devices such as joint replacements, dental implants, vascular stents and other devices used to replace, support or enhance a biological structure. Although these biomaterials are relatively biocompatible and have satisfactory mechanical properties, corrosion resistance of some of them (e.g., stainless steel, nitinol) can be affected by the stability of a surface oxide layer on the metallic material. Therefore, when the oxide layer is removed or disrupted in any way, corrosion issues may arise which can affect the surrounding tissue and/or cause mechanical failure of the implant. It may also be desirable to attach or coat the metallic biomaterial with biologically active agents such as proteins, pharmaceutical compounds, drugs, polymers, antibodies, (bio)organic molecules, metal or polymer nanoparticles in order to provide a therapeutic effect or enhance the biocompatibility of the implanted device. However, there are problems associated with the stable attachment of such agents to metal surfaces under certain conditions, such as in vivo.
[0004] For example, medical grade stainless steel alloy 316 L is used extensively in the medical device, pharmaceutical, and semiconductor industries due to its general corrosion resistance, smoothness, biocompatibility and cleanability after electropolishing treatment. However, if the protective oxide layer of a stainless steel device is disrupted, it will result in unsatisfactory corrosion resistance, which can result in dissolution of toxic Cr and Ni species, pitting corrosion, which could cause mechanical failure of the device, and high degree of thrombogenicity. Another disadvantage of stainless steel is the poor polymer surface attachment such as that seen in polymer-coated stainless steel medical devices.
[0005] As a specific example, medical grade stainless steel is used in the manufacture of vascular stents. Indeed, angioplasty followed by metallic stenting is the minimally invasive procedure of choice for treating atherosclerotic lesions in coronary arteries due to its relatively high success rate, reasonable cost, and minimal length of hospitalization. However, despite the biocompatibility of the medical grade stainless steel, stent implantation can cause trauma to the blood exposed vascular wall, thereby, denuding a monolayer of cells from the vascular wall called the endothelium. This event initiates an exaggerated response to the vascular injury whereby vascular smooth muscle cells proliferate inwardly causing the re-closure of the artery in a process known as in-stent restenosis.
[0006] Stents are used not only as a mechanical intervention but also as a vehicle for providing biological therapy. Biologically active agents such as proteins, pharmaceuticals, drugs, polymers, antibodies, (bio)organic molecules, polymers and a wide range of metal nanoparticles may be chosen for coating of or incorporation into, for example, the stent walls in order to minimize in-stent restenosis. Other agents may also be desirable for other effects. Indeed, in the vascular art, fibronectin has been used in various implant applications in order to promote cell/surface interactions and to produce a biocompatible surface. Indeed, it is well known in the art that fibronectins play a large number of roles in the human body including: wound healing, angiogenesis, and inflammatory responses. Fibronectins also have a profound effect on a cell's ability to adhere, proliferate, and spread on surfaces. However, in these applications, fibronectin has been only physisorbed (physically adsorbed) on implant surfaces. In the case of blood-contacting implants, this approach is not desirable, due to the rapid removal of the adsorbed fibronectin under the blood sheer force, leaving the implant surface naked and open to adsorption of undesirable proteins. Implantable biomaterials made of other metals and metal alloys, such as nitinol (Nickel Titanium alloy), titanium alloy, Cobalt-Chrome alloy or cobalt chromium alloy, present similar drawbacks to stainless steel.
[0007] Therefore, there is a need to develop an improved metal surface that substantially overcomes or reduces the aforesaid drawbacks. For example, a metal surface that has improved corrosion resistance, or improved polymer or other material attachment; or a method for producing such an improved metal surface.
SUMMARY
[0008] Aspects and embodiments of the present inventions reduce the aforesaid difficulties and disadvantages.
[0009] A first aspect of the present invention provides a method of modifying a surface of a metal or metal alloy substrate, for example of an implantable medical device, using an electrochemical process to covalently or irreversibly attached self-assembled monolayers (SAMs) onto the surface. A second aspect of the present invention provides a surface of a metal or a metal alloy substrate to which can be irreversibly and/or covalently attached self-assembled monolayers. The metal or metal alloys can be stainless steel, nickel titanium (NiTi), cobalt-chrome alloys (CoCr) (also known as cobalt chromium alloy) or the like. Active or nonactive agents can be irreversibly and/or covalently attached to the self-assembled monolayers on the surface.
[0010] By substrate is meant any substance or material having a surface which can be modified as defined according to the embodiments of the present invention.
[0011] One embodiment of the first aspect describes a new method of modifying a surface of a stainless steel substrate using an electrochemical process to covalently attach self-assembled monolayers on the surface. The stainless steel can be medical or industrial grade, preferably stainless steel 316L. However, metals other than stainless steel can be used. This attachment is stable, irreversible and robust. These functionalized self-assembled monolayers provide a link to irreversibly attach or immobilize, for example, an active agent such as pharmaceuticals, antibodies, bioactive peptides, drugs, polymers or metal nanoparticles on a stainless steel surface or nonactive agents. Furthermore, this technique gives steel surfaces improved properties as, for example, a better corrosion protection or enhancement of polymer adhesion. Indeed, experimental results have demonstrated the significant stability of the films of self-assembled monolayers rendering them very robust for practical biomedical and industrial applications. The monolayer-modified stainless steel surfaces have demonstrated improved species attachment over simply physically adsorbed species of the prior art which were easily removed.
[0012] An embodiment of this aspect of the present invention provides a method for attaching or linking or immobilizing self-assembled monolayers (SAM) onto a stainless steel surface comprising contacting said stainless steel surface with a solution comprising an alkanethiol compound, and electrochemically depositing said alkanethiol compound on the stainless steel surface using a nickel-based alloy auxiliary electrode to covalently attach the self-assembly monolayers onto said stainless steel surface. In an embodiment, the stainless steel surface is pretreated by polishing said surface and/or etching said surface with oxalic acid. The solution containing the alkanethiol compounds is an ionically-conductive aqueous or an ethanolic electrolyte solution. The nickel-based alloy electrode used in the present invention is a nichrome or a nickel auxiliary electrode. In a preferred embodiment, the nickel-based alloy electrode is a nichrome auxiliary electrode. In the method of the present invention, the self-assembled monolayers are formed by a cyclic application of polarization potential. In a preferred embodiment, the potential is between -1.8 volts and 0 volt vs. SCE (Saturated Calomel Electrode) and is applied for 1 to 150 cycles at a scan rate of 50 mV/s. In a most preferred embodiment, the potential is applied for at least 25 cycles. Preferably, the solution is preconditioned by a cyclic potentiodynamic polarization of a nickel-based alloy auxiliary and working electrode. The polarization is performed at a scan rate of 100 mV/s between -1.8 volts and 0 volt vs. SCE for 50 to 400 cycles. In a preferred embodiment, the polarization is performed for 250 cycles. In a preferred embodiment, the preconditioning of the solution containing the electrolytes is performed with a nichrome auxiliary and working electrode.
[0013] The SAMs prepared using the alkanethiol compound comprise COOH, NH2, OH and CH3 end groups. The end group can be of a single type or a mixture of different types of end groups.
[0014] In another embodiment, after the covalent, stable and/or irreversible attachment of the SAMs on the surface, the latter is then ready to covalently receive an active or nonactive agent which can result in irreversibly immobilizing the agent on the surface using the SAMs as a chemical link. In one embodiment, the active agent is selected from the group consisting of proteins, pharmaceuticals, drugs, polymers, antibodies, bioorganic molecules, biologically active agents and metal nanoparticles. In a preferred embodiment, the biologically active agent is fibronectin. The person skilled in the art will understand that, in order to properly attach the active or nonactive agent on the SAMs, the end groups of the latter can be chemically modified before the covalent attachment of the active agent. For example, the end groups of the self-assembled monolayers are chemically modified by a solution of n-hydroxysuccinimide or 1-ethyl-3-[3-dimethyl aminopropyl]carbodiimide or by using a glutaraldehyde linking. Other modifications are possible in manners that would be known to the persons skilled in the art.
[0015] Alternative embodiments of this first aspect differ from the embodiments described above in that, instead of stainless steel as a substrate, any other metal or metal alloy, alone or in combination, can be used. For example, NiTi, CoCr, or the like can be used.
[0016] From the second aspect, an embodiment provides for a modified surface of a stainless steel substrate comprising self-assembled monolayers (SAM) covalently attached to the surface, wherein said self-assembled monolayers are prepared by contacting said surface with a solution comprising an alkanethiol compound, and electrochemically depositing said alkanethiol compound on the surface using a nickel-based alloy auxiliary electrode to covalently attach the self-assembly monolayers onto said surface. Industrial or medical grade stainless steel may be used in this embodiment, preferably stainless steel 316L. In other embodiments, metals other than stainless steel can be used. In an embodiment, the stainless steel surface is pretreated by polishing said surface and/or etching said surface with oxalic acid. The solution containing the alkanethiol compounds is an ionically-conductive aqueous or an ethanolic electrolyte solution. The nickel-based alloy electrode used in the present invention is a nichrome or a nickel auxiliary electrode. In a preferred embodiment, the nickel-based alloy electrode is a nichrome auxiliary electrode. In the method of the present invention, the self-assembled monolayers are formed by a cyclic application of a polarization potential. In a preferred embodiment, the potential is between -1.8 volts and 0 volt vs. SCE and is applied for 1 to 150 cycles at a scan rate of 50 mV/s. In a most preferred embodiment, the potential is applied for at least 25 cycles. Preferably, the solution is preconditioned by a cyclic potentiodynamic polarization of a nickel-based alloy auxiliary and working electrode. The polarization is performed at a scan rate of 100 mV/s between -1.8 volts and 0 volt vs. SCE for 50 to 400 cycles. In a preferred embodiment, the polarization is performed for 250 cycles. In a preferred embodiment, the preconditioning of the solution containing the electrolytes is performed with a nichrome auxiliary and working electrode.
[0017] The SAMs on the surface prepared using alkanethiol compound comprise COOH, NH2, OH and CH3 end groups. The end groups can be of a single type or a mixture of different types.
[0018] The modified surface may further comprise an active an active or nonactive agent attached thereto. The nonactive or active agent can be covalently attached to the end group of the SAMs. This may result in irreversibly immobilizing the active or nonactive agent on the surface using the monolayer film as a chemical link. The active agent is selected from the group consisting of proteins, pharmaceuticals, drugs, polymers, antibodies, bioorganic molecules, biologically active agents, and metal nanoparticles. In a preferred embodiment, the biologically active agent is fibronectin. The person skilled in the art will understand that, in order to properly attach the active agent on the SAMs, the end groups of the latter can be chemically modified before the covalent attachment of the active or nonactive agent. For example, the end groups of the self-assembled monolayers are chemically modified by a solution of n-hydroxysuccinimide or 1-ethyl-3-[3-dimethyl aminopropyl]carbodiimide or by using a glutaraldehyde linking. Other modification methods are included as would be apparent to person skilled in the art.
[0019] Alternative embodiments of this second aspect differ from the embodiments described above in that, instead of stainless steel as a substrate, any other metal or metal alloys, alone or in any combination can be used. For example, NiTi, CoCr, or the like can be used.
[0020] The stainless steel or other metal or metal alloy substrate can form the whole or part of a medical device. In a preferred embodiment, the medical device can be a stent or other vascular implant, a bone or other orthopaedic implants, dental implants and materials, fixation screws and other fixation devices, and surgical tools. In one embodiment, the medical device is a stent. By medical device is meant any product used in diagnosis, therapy or surgery of patients. Embodiments of the present invention can also be used to modify metal surfaces to obtain better corrosion protection or enhancement of polymer adhesion, for sensors development or for regulation of surface friction in industrial application also.
BRIEF DESCRIPTION OF THE DRAWINGS
[0021] Further aspects and advantages of the present invention will become better understood with reference to the description in association with the following drawings in which:
[0022] FIG. 1 is a schematic diagram of an electrochemical cell used for formation of self-assembled monolayers (SAMs) on a surface of a metal substrate according to an embodiment of the present invention where the metal substrate is stainless steel 316L;
[0023] FIG. 2 is a schematic diagram of the binding of fibronectin (FN) to a COOH-terminated alkanethiol SAM irreversibly formed on a 316L stainless steel surface according to an embodiment of the present invention, when 1-Ethyl-3-[3-dimethyl aminopropyl]carbodiimide (EDC) and N-hydroxysuccinimide (NHS) were used to bind FN to the SAM;
[0024] FIG. 3 is schematic diagram of the binding of fibronectin (FN) to a NH2-terminated alkanethiol SAM irreversibly formed on a 316L stainless steel surface according to an embodiment of the present invention, when glutaraldehyde was used to bind FN to the SAM;
[0025] FIG. 4 is a graphic showing first to twenty-fifth cyclic voltammagram sweep of a 316L stainless steel in a solution of (top) 1 mM 11-amino-undecanethiol and (bottom) 10 mM mercapto-undecanoic acid in 0.1 M NaClO4 in denatured ethanol; scan rate, 100 mVs-1, according to an embodiment of the present invention;
[0026] FIG. 5 is graphic showing a cyclic voltammagram of a 316L stainless steel electrode in 0.1 M NaClO4 in denatured ethanol. (v=100 mVs-1), where no NH2-terminated or COOH-terminated SAM precursor molecules are present in the electrolyte, according to an embodiment of the present invention;
[0027] FIG. 6 is PM-IRRAS spectra of an NH2-terminated SAM formed by polarizing a 316L stainless steel coupon in a solution of 1 mM 11-amino-undecanethiol between 0 and -1.8 VSCE in 0.1 M NaClO4 in denatured ethanol at a scan rate=100 mVs-1 where inset (A) represents the NH2-terminated SAM, according to an embodiment of the present invention;
[0028] FIG. 7 is a graphic showing the normalized area of vsymCH2 peak of a SAM formed on a 316L stainless steel surface after sonication in 0.16 M NaCl, denatured ethanol or 0.1 M NaOH, according to an embodiment of the present invention;
[0029] FIG. 8 is a graphic showing the normalized area of vsymCH2 peak of a COOH-terminated SAM formed on a 316L stainless steel surface using the procedure described in FIG. 7, according to an embodiment of the present invention;
[0030] FIG. 9 is a graphic showing the contact angle of COOH-terminated SAM-modified 316L stainless steel samples as a function of immersion time in aqueous 0.16 M NaCl wherein the SAM was formed on the 316L stainless steel surface using the procedure described in FIG. 7, according to an embodiment of the present invention;
[0031] FIG. 10 is a graphic showing the general corrosion protection efficiency of a COOH-terminated SAM formed on 316L stainless steel samples as a function of immersion time in aqueous 0.16 M NaCl wherein the SAM was formed on the 316L stainless steel surface using the procedure described in FIG. 7, according to an embodiment of the present invention;
[0032] FIG. 11 illustrates the 1600 cm-1 region PM-IRRAS spectra of a COO.sup.-/Metal-terminated SAM, where COO.sup.-/Metal denotes a metal complex and the inset represents the COO.sup.-/Metal SAM, in which M represents the metal, according to an embodiment of the present invention;
[0033] FIG. 12 illustrates the evidence of the destruction of the COO--/Metal complex by the immersion of the freshly-formed COOH-terminated SAM (dashed line) in phosphoric acid pH 1.5 for 30 minutes wherein the `free` COOH peak at 1712 cm-1 increases upon immersion (solid line), according to an embodiment of the present invention;
[0034] FIG. 13 illustrates the evidence of the destruction of the COO--/Metal complex by the immersion of the freshly-formed COOH-terminated SAM in various acids of pH 1.5 for 30 minutes, according to an embodiment of the present invention;
[0035] FIG. 14 illustrates the 1600 cm-1 PM-IRRAS spectra of changes to a NH2 SAM on 316L SS before (dark line) and after (pale line) reacting with a 20% v/v solution of glutaraldehyde for 1 hr, according to an embodiment of the present invention;
[0036] FIG. 15 illustrates the 1600 cm-1 region PM-IRRAS spectra of BSA (top) and FN (bottom) immobilized onto glutaraldehyde modified NH2 SAMs, wherein the pale line represents the situation before binding the protein, while the solid line represents the situation after binding the protein, according to an embodiment of the present invention;
[0037] FIG. 16 illustrates the 1600 cm-1 region PM-IRRAS spectra of (BSA) stability after sonication in aqueous 0.1 M NaOH. (top) BSA covalently bound to the NH2+GLU SAM, (bottom) BSA physically adsorbed (physisorbed) on the NH2 SAM, wherein the peaks are Amide 11600-1700 cm-1 (shaded) and Amide II 1485-1580 cm-1, according to an embodiment of the present invention;
[0038] FIG. 17 illustrates the PM-IRRAS spectra of NHS SAM ester on a 316L stainless steel surface after 2 hr immersion of a COOH-terminated SAM formed on a 316L stainless steel surface, in 75 mM EDC and 15 mM NHS, wherein the insert is the molecular schematic representing NHS ester SAM, according to an embodiment of the present invention;
[0039] FIG. 18 illustrates the 1600 cm-1 region PM-IRRAS spectra of the NHS and EDC reaction with a COOH-terminated SAM formed on a 316L stainless steel surface, according to an embodiment of the present invention;
[0040] FIG. 19 illustrates the normalized area of the NHS asymmetric carbonyl peak C═O at approximately 1740 cm-1 and the free unreacted COOH peak at approximately 1710 cm-1 as a function of time, wherein the error bars represent ±1 standard deviation from the mean, n=3, according to an embodiment of the present invention;
[0041] FIG. 20 illustrates the 1600 cm-1 region PM-IRRAS spectra of (top) FN covalently bound to a COOH-terminated SAM formed on a 316L stainless steel surface, and (bottom) FN physically adsorbed on a bare 316L stainless steel surface in which (a) represents a NHS ester peaks 1824 cm-1, 1789 cm-1, 1744 cm-1, (b) represents an Amide II 1600-1700 cm-1, (c) represents vasymCOO.sup.- 1542-1591 cm-1 with Amide II 1485-1580 cm-1, (d) represents symCOO.sup.- with δCH2 1456 cm-1 and (e) represents only Amide II 1485-1580 cm-1, Amide I peak is shaded to represent quantity of FN for visual effect, according to an embodiment of the present invention;
[0042] FIG. 21 is a graphic showing the normalized amide I peak area representing fibronectin (FN), wherein FN is covalently bound to a COOH-terminated SAM formed on a 316L stainless steel surface (left) and FN is physically adsorbed on bare 316L stainless steel surface (right), according to an embodiment of the present invention;
[0043] FIG. 22 is a graphic showing the HUVE cell attachment onto an FN-modified, COOH-modified and bare 316L stainless steel substrates for 1, 2 and 4 hrs of incubation, wherein the initial incubation was 50,000 cells/cm2 and the error bars are ±95% confidence interval, n=4, according to an embodiment of the present invention;
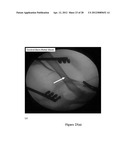
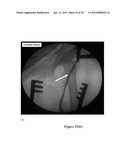
[0044] FIGS. 23 (a) and (b) are two angiograms showing the effect of a 316L stainless steel stent surface modification by covalently attached FN, wherein FIG. 23 (a) shows a commercial bare-metal stent, and FIG. 23 (b) shows a FN-modified commercial stent, according to an embodiment of the present invention;
[0045] FIG. 24 is a PM-IRRAS spectrum of an COOH-terminated SAM formed by polarizing a NiTi coupon in a solution of 1 mM 11-amino-undecanethiol between 0 and -1.8 VSCE in 0.1 M NaClO4 in denatured ethanol at a scan rate=50 mVs-1 (22 cycles), according to an embodiment of the present invention;
[0046] FIG. 25 is a PM-IRRAS spectrum of an COOH-terminated SAM formed by polarizing a NiTi coupon in a solution of 1 mM 11-amino-undecanethiol between 0 and -1.8 VSCE in 0.1 M NaClO4 in denatured ethanol at a scan rate=50 mVs-1, according to an embodiment of the present invention;
[0047] FIG. 26 is a PM-IRRAS spectrum of NHS SAM ester on a NiTi surface after 2 hours of immersion of a COOH-terminated SAM formed on a NiTi surface, in 75 mM EDC and 15 mM NHS, according to an embodiment of the present invention; and
[0048] FIG. 27 is showing the 1600 cm-1 region PM-IRRAS spectra of FN covalently bound to a COOH-terminated SAM formed on a NiTi surface, where the FN -amide I (1660 cm-1) and amide II (1540 cm-1) peaks are visible.
DETAILED DESCRIPTION
[0049] This invention is not limited in its application to the details of construction and the arrangement of components set forth in the following description or illustrated in the drawings. The invention is capable of other embodiments and of being practiced or of being carried out in various ways. Also, the phraseology and terminology used herein is for the purpose of description and should not be regarded as limiting. The use of "including", "comprising", or "having", "containing essentially", "involving" and variations thereof herein, is meant to encompass the items listed thereafter as well as, optionally, additional items. In the following description, the same numerical references refer to similar elements. In the drawings, like reference characters designate like or similar parts.
[0050] In one aspect of the present invention, an agent or active agent is covalently attached to a surface of a metal or alloy substrate through the use of self-assembled monolayers (SAMs). This covalent bonding will secure the active or nonactive agent to the surface. In a preferred embodiment of this aspect, the surface is a metallic surface of a medical device. Therefore, this covalent bonding will secure the active or nonactive agent to the medical device so that it is not removed, for example, by blood sheer stress or by the device storage in air or liquid media (corrosive or not). In a preferred embodiment of this aspect, the substrate is stainless steel, one of the most popular biomaterials in medical devices and especially in the manufacture of stents. More preferably, the stainless steel is medical grade stainless steel 316 L. In further embodiments, the substrate is Nitinol (NiTi) or Cobalt Chrome alloys (CoCr) or the like.
[0051] For ease of discussion, the nonactive or active agents detailed herein will be described with reference to a biocompatible protein such as fibronectin. However, the nonactive or active agents of the present invention may be any suitable molecule(s) that can be chemically attached to the SAMs as, for example, proteins, pharmaceuticals, drugs, polymers, antibodies, (bio)organic molecules, and a wide range of metal nanoparticles, and the like. In a preferred embodiment, the active agent is a biologically active agent. A person skilled in the art will have no difficulty in selecting an appropriate active agent in relation with the desired effect, function and/or treatment.
[0052] Examples of medical devices that may be coated with a nonactive or active agents by the method of the present invention include, but are not limited to stents (as coronary artery stents), conduits, cardiovascular valves, bone and dental implants, fixation screws, surgical tools, sensors, neural electrodes.
[0053] As known in the art, a SAM is an organized layer of molecules in which one end of the molecule, the "head group", shows a special affinity for a substrate. SAM usually also presents a "tail" with a functional group at the terminal end. In the present invention, a SAM is used as a platform for immobilizing a biologically active agent on a stainless steel surface. SAMs could also be used for a platform for drug delivery from, for example, a coronary stent. Alkanethiols are the most commonly used molecules for SAMs. Alkanethiols are molecules with an alkyl chain, (CH2)n chain, as the back bone, a tail end-group, and a SH head group. They are usually used on noble metal substrates, such as gold, because of the strong affinity of sulfur for these metals.
[0054] It is well recognized that the formation of SAMs on stainless steel represents a very challenging task. Indeed, the surface of steel is complex and heterogeneous containing predominantly Cr, Fe, Ni, and Mn oxides in multiple oxidation states. The strong affinity for 316 L stainless steel to form oxides (passive film) makes it relatively resistant to corrosion under certain conditions. Unfortunately strongly bound oxides make standard thiol assembly through sulfur metallic bonding difficult. Only the head group siloxanes Si(OEth)3 and phosphonates (PO3) can be used on oxide film surfaces without significant prior modification or driving force. In contrast, carboxylic acid (COOH) and thiols (SH) require pretreatments or electrochemical force to actively drive SAMs onto the steel surface. Other metals or metal alloys present similar challenges and modifying their surfaces.
[0055] Surprisingly, the Applicant has found a novel method for producing a stable association between SAMs and a metal surface, such as stainless steel, using an electrochemical process to chemically functionalize the surface. Indeed, the Applicant has demonstrated that this electrochemical method, combining the use of alkanethiol compounds and a nickel-base alloy electrode, enables the formation of covalent, stable and robust SAMs on the surface. This method is simple and highly reproducible, which is a major improvement as the formation of SAMs on stainless steel and other metals represents a very challenging task.
[0056] Furthermore, the Applicant has surprisingly discovered that this electrochemical process to chemically functionalize the stainless steel surface is also applicable to other metal surfaces such as NiTi and CoCr alloys. However, even though they both offer a better corrosion resistance and a higher biocompatibility than stainless steel, they are, as stainless steel, thrombogenic and therefore not a good choice for long-term implantation. Consequently, because NiTi and CoCr alloys present similar drawbacks as stainless steel (biocompatibility, thrombogenicity, negative cell/surface interactions), the rationale for modifying their surface is the same as for the stainless steel.
[0057] An embodiment of the first aspect of the present invention is directed to the formation or the attachment or the linking of SAMs on a stainless steel surface and, more particularly, on a stainless surface of an implantable medical device. To do so, functionalized surfaces prepared by SAMs of alkanethiols with --NH2 or --COOH, as tail group are used to covalently link, for example, a biologically active agent to the stainless steel surface. As will be understood by the person skilled in the art, SAMs of alkanethiols presenting different tail groups, such as --OH-- and --CH3 tail groups, can be used in the present invention.
[0058] Further, nonactive or active agents can be covalently attached to the tail group of the SAMs. In the present invention, the SAMs are extremely stable on the stainless steel surface because the bond strength is very high. In a preferred embodiment, fibronectin or any other active agent, is then covalently attached to the SAMs. The person skilled in the art will know which tail groups of the SAMs can be used in relation with particular agents.
[0059] The present invention provides a modified stainless steel surface with SAMs of alkanethiols and a method for modifying a surface of a stainless steel substrate using an electrochemical process comprising providing a surface, and contacting the surface with an alkanethiol SAM comprising, for example, of COOH and/or NH2 and/or OH and/or CH3 end group(s). It will be understood by the person skilled in the art that SAMs of alkanethiols presenting other tail groups can be used in the purpose of the present invention. The SAMs on the surface can be prepared using alkanethiol presenting a single type of end or tail group or a mixture of different types of end groups. The person skilled in the art will know how to select the combination in order to produce the desired functionalized SAMs on stainless steel. This method is also applicable to other metal substrates such as NiTi or CoCr or the like.
[0060] In further detail and in order to modify or coat a metal or metal alloy surface encompassed by the present invention, the sample surface of the metal or alloy, preferably stainless steel, is pretreated, for example, by polishing and/or etching in oxalic acid and then sonicated in an ultrasonic bath containing ethanol to remove polishing residues and degrease the surface. However, it will be understood that any techniques known in the art for the pretreatment of the stainless steel surface or any metal surface encompassed by the present invention can be used in the present invention. The sample is then immersed in an electrochemical cell containing the SAM precursor(s) at a specific concentration dissolved in an ionically-conductive aqueous solution or an ethanolic electrolyte solution. The person skilled in the art will know which type of solution has to be used in order to prepare the formation of SAMs. The SAMs are formed by cyclically and potentiodynamically polarizing the sample between two desired polarization potentials for a desired number of cycles by using a nickel-based alloy auxiliary electrode. In a preferred embodiment, the nickel-based alloy auxiliary electrode is a nickel or a nichrome auxiliary electrode. In a most preferred embodiment, the auxiliary electrode is a nichrome auxiliary electrode. The desired potential range is between -1.8 volts and 0 volt vs. SCE (Saturated Calomel Electrode). The number of cycles is between one and 200, preferably between one and 150 and, in a preferred embodiment, the number of cycles is 25. The sample is then sonicated in an ultrasonic bath to remove loosely-attached SAM precursor molecules. The sample is then ready to covalently receive an active agent or a biologically active agent or a combination of agents. This will result in irreversibly immobilizing the agents on the steel or metal surface using the monolayer film as a chemical link. The person skilled in the art will understand that, before the step of attaching or linking the SAM on the metal or stainless steel surface, the solution containing the electrolytes is preconditioned by a cyclic potentiodynamic polarization of a nickel-based alloy auxiliary electrode and a working electrode at a scan rate of 100 mV/s between -1.8 volts and 0 volt vs. SCE. The polarization is performed for 50 to 400 cycles but preferably for 250 cycles. In a preferred embodiment, the nickel-based alloy auxiliary and working electrode is a nickel or a nichrome electrode. In a most preferred embodiment, the auxiliary or the working electrode is a nichrome electrode.
[0061] The following examples demonstrate the present invention in greater details. These examples are not intended to limit the scope of the invention in any way.
[0062] The Applicant has developed a novel electrochemical method for producing a stable, strong and covalent association between SAMs and a stainless steel surface. Furthermore, the Applicant has surprisingly demonstrated that this novel electrochemical method is also applicable to other metal surfaces such as NiTi or CoCr for producing a stable and covalent association between SAMs and the metal surface. SAMs of alkanethiols with --NH2 or --COOH or --OH or --CH3 terminal tail group were formed on the surface of stainless steel 316 L by using a nichrome (NiCr) wire as the counter or auxiliary electrode. The person skilled in the art will understand that any nickel-based alloy electrode containing a higher concentration of nickel, i.e., at least 10%, can be used in the formation of the functionalized SAMs on stainless steel. Furthermore, it will be understood that a nickel auxiliary electrode can be used in the present invention in the formation of the functionalized SAMs on stainless steel. The strong, stable, covalent attachment and uniformity of the SAMs on the stainless steel substrate was confirmed using the following techniques: polarization modulation infrared absorption reflection spectroscopy (PM-IRRAS), contact angle measurements (CA) and electrochemical impedance spectroscopy (EIS). Furthermore, biological active agent immobilization on stainless steel was performed using the biocompatible protein fibronectin, and bovine serum albumin as a control sample. In order to covalently attach the fibronectin to the SAMs on the stainless steel surface, the COOH group on the SAM was reacted with aqueous 1-ethyl-3-3dimethyl aminopropyl carbodiimide (EDC) and hydroxysuccinimide (NHS), or NH2 group was reacted with aqueous glutaraldehyde. The covalent attachment of the fibronectin to the SAMs on the stainless steel was confirmed using PM-IRRAS, EIS and CA in combination with sonication in various organic and inorganic solutions and compared to physically adsorb fibronectin on bare stainless steel which was rapidly removed under this stress. Cell adhesion experiments were conducted using endothelial cells to investigate if the modified stainless steel surface was a better substrate for endothelial cell attachment rather than a bare stainless steel surface. The Applicant has demonstrated that the fibronectin-modified 316 L stainless steel was indeed significantly more biocompatible then the naked stainless steel surface, as evidenced by the increased number of endothelial cells on the modified surface.
Materials and Methods:
Materials:
[0063] Table 1 summarizes the source of the different chemicals and products used.
TABLE-US-00001 TABLE 1 Catalogue Chemical Number Use Self-Assembled Monolayers 11-mercapto- 450561 (Aldrich) COOH SAM undecanoic acid 1-undecanethiol 510467 (Aldrich) CH3 SAM 11-Amino-1- 111027-004 NH2 SAM undecanethiol (Assemblon) 11-mercapto-1- 447528 (Aldrich) OH SAM undecanol Solvents, Salts and Acids sodium chloride 71381 (Sigma) corrosion and sonication sodium hydroxide 5M SS256B-500 protein removal (Fisher) potassium phosphate 7778770 (Sigma) phosphate buffer solution dibasic sodium perchlorate FL020797 electrolyte (Fisher) denatured alcohol A407P-4 (Fisher) cleaning and solvent acetic acid A35-500 (Fisher) de-carboxylate COOH SAMs phosphoric acid A242-500 '' (Fisher) malonic acid AC12526-2500 '' (Acros) citric acid A940-500 '' (Fisher) hydrochloric acid A144-500 '' (Fisher) sulfuric acid A510-500 '' (Fisher) perchloric acid A2287-LB '' (Fisher) Protein Binding to Self Assembled Monolayers/Coupons bovine serum albumin A9418 (Sigma) proof of SAM protein (BSA) covalent binding Fibronectin 354008 (BD increase cell adhesion to Bioscience) 316L SS 1-ethyl-3-[3dimethyl 3450 (Fluka) catalyst BSA and FN aminopropyl] covalent binding (EDC) carbodiimide glutaraldehyde FL080202 BSA and FN covalent (Fisher) binding (GLU) n-hydroxysuccinimide 130672 (Sigma) BSA and FN covalent binding (NHS) 316L stainless steel 9298K131 machined into 0.5'' coupons (McMaster Caar) diameter × 2 mm thick coupons 316L SS NiCr (nichrome) wire 8880K72 as electrodes (McMaster Caar) Cell Attachment and Proliferation quick cell proliferation Bioscience quantifies number of essay kit (K301-500) cells in culture fetal bovine serum 12483 (Gibco) cell growth agent penicillin/streptomycin/ 450-202-EL cell growth anti- glutamine (Wisent) bacteria/virus
Substrate Preparation:
[0064] Identical 316 L stainless steel coupons (stent material) having a diameter of 127 mm and a thickness of 2 mm were used for cell testing, PM-IRRAS investigation and electrochemical measurements. They were machined from a 316 L stainless steel rod (9298K131 McMaster Caar).
[0065] Table 2 presents the chemical composition (wt %) of the stainless steel samples used.
TABLE-US-00002 TABLE 2 Fe Cr Ni K Mo Mn bal 16.22 10.31 0.031 3.2 1.48 Cu Si C Nb S Vn 0.57 0.38 0.065 0.034 0.043 --
[0066] Before further modification, the samples were polished with 1000, 2400, and 4000 grit SiC paper and rinsed and sonicated with water and denatured alcohol, acetone and chloroform for 10 minutes each to remove any residues from the polishing steps. The samples were dried in ambient air before use. If the SiC residue is not removed its presence is visible by large peaks between 1000-1100 cm-1 in the PM-IRRAS spectra representing Si species which are detrimental to film integrity.
Electrochemical Setup for Binding SAMs on Stainless Steel:
[0067] SAM deposition was performed in an electrochemical cell, containing SAM-forming precursor(s) dissolved in 0.1M NaClO4 in denatured ethanol. The 316L stainless steel sample was the working electrode. NiCr (McMaster Caar 880K72) was used as a counter-electrode (or as an auxiliary electrode), and a commercial saturated calomel electrode as a reference electrode (FIG. 1--SS316L+NiCr hook servers as a cradle for a flat 316L stainless steel sample (working electrode, WE) and to provide electrical contact with the external electrical circuit. SCE represents a saturated calomel electrode (reference electrode, RE), while NiCr is the counter (or auxiliary) electrode (CE). Ar is used to purge the solution (optional).
[0068] The SAMs were formed by potendiodynamically polarizing the 316L stainless steel sample between -1.8 volts and 0 volt vs. SCE, at a scan rate of 50 mVs-1, and for minimum of 25 cycles. The potential can be applied for 1 to 150 cycles. Before the SAM formation, the electrolyte was first pre-conditioned in the same electrochemical cell containing 0.1M NaClO4 in denatured ethanol (but no SAM precursors) by cyclic potentiodynamic polarization of a NiCr auxiliary and working electrode, which is in this step used instead of the 316L stainless steel sample. This preconditioning step of the electrolyte is necessary in order to successfully realize the covalent association of the SAMs on the stainless steel surface and in order to get stable SAMs. The polarization was performed at a scan rate of 100 mVs-1 between -1.8 volts and 0 volt vs. SCE for 50 to 400 cycles, but preferably 250 cycles. The person skilled in the art will understand that this technique of producing SAMs on a stainless steel surface can also work in aqueous solutions rather than in organic solvents.
[0069] After SAMs binding, the coupons were rinsed with deionized water and denatured ethanol, before and after a 5 minute sonication in ethanol, to remove any physically adsorbed molecules from the surface unless otherwise stated. The potentiostat used for these experiments was an Ecochemie Autolab PGSTAT30 potentiostat/galvanostat equipped with GPES v.4.9.5 software.
[0070] Homogeneous and mixed SAMs of any surface composition can be prepared using the above procedure. For example, a binary SAM (--S--(CH)n--COOHn%/X100%-n where X=OH, NH2 and CH3) can be advantageous in order to control the surface physicochemical properties (i.e., charge and wettability). This SAM can be prepared by dissolving the COOH precursor (--S--(CH)n--COOH) and the X precursor (--S--(CH)n--X) in the electrolyte in the corresponding proper molar ratios.
Fibronectin (FN) or Bovine Serum Albumin (BSA) Binding to COOH-Terminated SAMs Using 1-ethyl-3-[3-dimethyl aminopropyl]carbodiimide (EDC) and n-hydroxysuccinimide (NHS):
[0071] To covalently bind FN (or any other protein) to the 316 L stainless steel surface, EDC and NHS chemistry was used. COOH SAMs were first formed using the electrochemical techniques as described above. It has been found, as discussed later, that the SAM formation resulted in the formation of a SAM-COO.sup.-/Ni complex, thus making the COO.sup.- end group chemically inactive. In order to activate the SAM end group, the substrates were immersed in a weak acid for 30 minutes (any of the weak acids listed in Table 1 as "de-carboxylate COOH SAMs" can be used, but phosphoric acid, at a pH 1.5, gave the best results). The samples were then rinsed with deionized water and denatured ethanol several times and dried in ambient air. Then, the modified surfaces were immersed in 15 mM NHS and 75 mM EDC dissolved in deionized water for 2 hrs (FIG. 2) [1-4].
[0072] After the NHS/EDC immersion, the substrates were rinsed again in the same way and allowed to dry in ambient air. The now NHS ester SAM substrates were placed in a 2 μg/mL FN solution to immobilize the protein. The protein solution was prepared by first making a 100 mL phosphate buffer stock solution containing 0.05 M KH2PO4. The pH was adjusted to 6.0 using 5.0 M NaOH stock. The protein was weighed, then allowed to gently dissolve for 1 hr at room temperature in the phosphate buffer solution. Sonication and vigorous stifling can be detrimental to FN integrity and therefore was not used. The substrates were then incubated overnight in the protein solution to immobilize FN to the surface at 4° C. Although the adsorption kinetics may be slower at 4° C., it was determined that this temperature was suitable to lower protein denaturation. It will be understood by the person skilled in the art that the same procedure can be used to bind any protein or active agents.
[0073] A new phosphate buffer solution, pH 6.0, prepared as mentioned above was made and 2.0 M NaCl was added to it. This solution was used to remove physically adsorbed protein from the surface. The FN immobilized coupons were then placed in the 2.0 M NaCl and phosphate buffer solution for 2 hrs to remove any physically adsorbed FN. Then the substrates were rinsed with and stored in a solution of non-saline phosphate buffer 6.0 pH solution until cell testing. Before adding to cell culture media, the substrates were rinsed with deionized water and air dried in air to remove any phosphate buffer residue.
Fibronectin or Bovine Serum Albumin Binding to NH2-Terminated SAMs Using Glutaraldehyde Linking:
[0074] Binding FN (or any other protein or active agents) to the 316 L stainless steel surface was also achieved through the NH2-terminated SAM. The NH2-terminated SAMs were formed on the surface in the same way as the COOH-terminated SAM described previously.
[0075] The NH2-terminated SAM surface was first immersed in a solution of glutaraldehyde (GLU) 20% v/v for 1 hr, to form a stable imine bond (N═C) between the NH2 SAM and GLU (FIG. 3, step 3). After this, the samples were sonicated in denatured ethanol for 10 min to remove any non-chemically bound GLU from the surface. The surface was rinsed with dionized water and denatured ethanol and dried in ambient air. FN or BSA were then bound to the free aldehyde group of the NH2+GLU SAM forming an imide bond [1-4].
[0076] The protein immobilization was achieved in a similar manner than the protein immobilization on COOH SAMs. First, a 100 mL phosphate buffer stock solution containing 0.05 M KH2PO4 was made. The pH was adjusted to 6.0 using 5.0 M NaOH stock. The protein was weighed to make a final concentration of 2 μg/mL FN or BSA and allowed to gently dissolve for 1 hr at room temperature in the phosphate buffer solution. However, in this case, the NH2+GLU SAM substrates were placed in a 2 μg/mL FN or BSA solution to immobilize the proteins through reactive aldehyde groups (FIG. 3, step 4). The incubation was conducted overnight in the protein solution to immobilize FN or BSA to the surface at 4° C. and rinsed with deionized water and ethanol and allowed to dry before further use. It will be understood by the person skilled in the art that any protein or any active agent can be attached to the NH2-terminated SAM using the above procedure. Furthermore, the person skilled in the art will know how to adapt the methodology having regard to the type of active agent.
Electrochemical Setup for Binding SAMs on NiTi:
[0077] The methodology for the electrochemical binding of SAMs on NiTi is the same as the one described with stainless steel. SAM deposition was performed in an electrochemical cell, containing SAM-forming precursor(s) dissolved in 0.1M NaClO4 in denatured ethanol. The NiTi sample was the working electrode. NiCr (McMaster Caar 880K72) was used as a counter-electrode (or as an auxiliary electrode), and a commercial saturated calomel electrode as a reference electrode.
[0078] The SAMs were formed by potendiodynamically polarizing the NiTi sample between -1.8 volts and 0 volt vs. SCE, at a scan rate of 50 mVs-1, and for 22 cycles. Before the SAM formation, the electrolyte was first pre-conditioned in the same electrochemical cell containing 0.1M NaClO4 in denatured ethanol (but no SAM precursors) by cyclic potentiodynamic polarization of a NiCr auxiliary and working electrode, which is in this step used instead of the NiTi sample. This preconditioning step of the electrolyte is necessary in order to successfully realize the covalent association of the SAMs on the NiTi surface and in order to get stable SAMs. The polarization was performed at a scan rate of 100 mVs-1 between -1.8 volts and 0 volt vs. SCE for 50 to 400 cycles, but preferably 250 cycles. The person skilled in the art will understand that this technique of producing SAMs on a NiTi surface can also work in aqueous solutions rather than in organic solvents.
[0079] After SAMs binding, the coupons were rinsed with deionized water and denatured ethanol, before and after a 5 minute sonication in ethanol, to remove any physically adsorbed molecules from the surface unless otherwise stated. The potentiostat used for these experiments was an Ecochemie Autolab PGSTAT30 potentiostat/galvanostat equipped with GPES v.4.9.5 software.
[0080] Homogeneous and mixed SAMs of any surface composition can be prepared using the above procedure. For example, a binary SAM (--S--(CH)n--COOHn%/X100%-n where X=OH, NH2 and CH3) can be advantageous in order to control the surface physicochemical properties (i.e., charge and wettability). This SAM can be prepared by dissolving the COOH precursor (--S--(CH)n--COOH) and the X precursor (--S--(CH)n--X) in the electrolyte in the corresponding proper molar ratios.
[0081] Finally, FN was covalently linked to the electrochemically modified NiTi surface using the same technique as previously described with stainless steel.
Instrumentation and Chemical Analysis:
Polarization Modulation Infrared Reflection Absorption Spectroscopy (PM-IRRAS) (Bruker Optics) Settings:
[0082] To increase spectral resolution at a specific region, such as CH2 stretching or amide region, the photoelastic modulator (Hinds PEM-90) wavenumber was set to either 2900 cm-1 or 1600 cm-1 respectively. The halfwave retardation was set to λ/2 or 0.5 for 278 scans at a resolution of 3 cm-1. The angle of the incident beam was 80° with respect to the surface normal. Aperture setting was 6 mm. Bruker OPUS Spectroscopy software v. 6.7 was used for recording, smoothing, peak picking, baseline corrections and arithmetic manipulations. To obtain clear spectral peaks, the spectrum of the SAM-covered sample was subtracted from a bare and freshly polished 316 L stainless steel sample. A liquid nitrogen cooled MCT detector was used in all measurements.
Contact Angle Goniometry (CA):
[0083] Static contact angles were registered using an OCA 20 Goniometer from Dataphysics. The data manipulation was performed by SCA 200 Software at room temperature with 5 μl drop size using deionized water as the solvent. Typically 5 repeat measurements were taken for each surface and the mean and standard deviation were calculated to attain a final contact angle.
Electrochemical Impedance Spectroscopy (EIS):
[0084] Electrochemie Autolab PGSTAT30/FRA2 was used for electrochemical impedance spectroscopy measurements. SAM-modified and bare (control) 316L stainless steel samples were incubated in 0.16M NaCl aqueous solution and at selected times an EIS measurement was performed at open circuit potential. The recorded spectra were then modeled using an electrical equivalent circuit and the corresponding corrosion resistance was determined.
Endothelial Cell Attachment:
[0085] To evaluate the response of modified substrates on human umbilical vascular endothelial, HUVE, cells, attachment tests were conducted. The three substrates investigated included: Bare 316 L stainless steel as a control, COOH-terminated SAMs on 316 L stainless steel and FN covalently attached to COOH-terminated SAMs on 316 L stainless steel, COOH and COOH+FN respectively. HUVE cells were incubated in MCDB 131 medium with 10% fetal bovine serum (12483 Gibco) containing 1% penicillin, streptomycin and L-glutamine (Wisent 450-202-EL) for 1, 2 and 4 hrs.
[0086] After incubation each well was rinsed by pipetting 300 μL of media of 3 times to remove any non attached cells. Then, to quantify the cells, 30 μL (9.1% total volume) of the Quick Cell Proliferation Assay Kit (K301-2500) BD Biosciences was pipetted into the wells containing the substrate and media and incubated for 2 hrs at 37° C. Afterwards, 10 μL was pipetted from each well and added to a new 96 well-plate and shaken for 1 minute. Absorbances were read using the Beckamn Coulter AD340 Well Plate Reader set to 450 nm [5].
Results and Discussion:
Monolayer Assembly to 316 L Stainless Steel:
[0087] As mentioned previously, individual alkane molecules with long chains (usually >C6) under certain conditions can form high density structures that can be used to give metallic surfaces with entirely new defined surface chemistry (ex. CH3, COOH, NH2 or OH functional groups). For these films to be of practical use, the individual molecules must be firmly attached to the substrate through a head group. This is in contrast to Langmuir-Blodgett films which are not necessarily immobilized to a surface, but rather physisorbed. In the present invention, both NH2 and COOH functionalized 316 L stainless steel surfaces were made in order to ultimately covalently bind an active agent, such as fibronectin, to the surfaces. CH3- and OH-terminated SAMs were also developed.
[0088] In addition, the person skilled in the art will understand that forming mixed CH3-, OH-, COOH- and/or NH2-terminated binary SAMs could be beneficial since the surface wettability and charge could be regulated this way.
Alkanethiol Self-Assembly on Stainless Steel Using Potentiodynamic Polarization:
[0089] In order to obviate the drawbacks of known techniques in the art, a new procedure using potentiodynamic polarization was discovered to provide the electrochemical assembly of alkenethiols SAMs on stainless steel. Contrary to the techniques known in the art, the method of the present invention is reproducible, provides stable and reactive SAMs, and allows a high surface coverage of the substrate.
[0090] From the literature, it is clear that thiol SAMs had been formed on other well-defined metal surfaces such as copper [6, 7], silver, [8] and nickel [9] by applying controlled potential in an electrochemical cell containing the metal substrate as the working electrode and the alkanethiol molecule in solution.
[0091] The Applicant has surprisingly discovered that when a Nichrome (NiCr) wire, or any nickel-base alloy wire, was used as a counter electrode in electrochemical experiments, extremely stable SAMs could be formed. It was determined that the NiCr wire, or any nickel-base alloy wire, was instrumental in SAM binding and if the NiCr electrode, or any nickel-base alloy electrode, was replaced by titanium, stainless steel or graphite, the stable (irreversibly and covalently attached) SAMs would not form.
[0092] FIG. 4 represents the graphical output of the current response vs the applied polarization potential during the SAM assembly, which is also known as a cyclic voltammagram, CV. While monitoring the current response during the potential sweeps, a clear trend in the cathodic region was observed: the current amplitude was decreased with the number of sweeps. To determine if this effect was a result of alkanethiol assembly on the surface, a control experiment was run, in which the exact same conditions were investigated without any SAM molecules. The result is shown in FIG. 5. As the CV shows, even after 25 sweeps, the CV response remained fairly unchanged, demonstrating that the decreasing current trend in FIG. 4 is indeed due to the SAM formation. Namely, in this potential region (ca. negative of -1.2V), the hydrogen evolution reaction occurs. The rate of this reaction, and thus the measured current, depends on the surface area available for the reaction. Due to the SAM formation on the surface, the surface area available for the hydrogen reaction decreases with each sweep, thus the current density decreased. This is not the case with the control sample (FIG. 5). Therefore the key difference in the CVs in the presence of SAMs compared to the control is that there appears to be a blocking of electron transfer through the 316L stainless steel surface. This is due to the buildup of an electrically-insulating SAM on the steel surface. The current blocking effects of SAMs are well known [10]. The current blocking effect is summarized in Table 3.
TABLE-US-00003 TABLE 3 Alkanethiol current blocking effect during film assembly Current I/mA At -1.8 VSCE No SAM NH2 SAM COOH SAM 1st sweep -3.6 -4.36 -5.89 25th sweep -3.05 -0.73 -0.60 % difference 15.2 83.3 89.8
Effect of Number of Cyclic Sweeps on Sam Formation:
[0093] To validate that SAMs were being deposited on the 316 L stainless steel electrode during these cyclic sweeps, PM-IRRAS spectroscopy was used to identify chemical groups of the SAM. It was determined that when the number of sweeps was increased from 1, 5, 10, 15, and 25, the PM-IRRAS peaks between 2917-2930 cm-1 and 2845-2856 cm-1 continued to rise. This is consistent with the lowering of current in CV measurements with number of scans observed in FIG. 6.
[0094] These absorbance peaks represent the methylene CH2 asymmetric (vasymCH2) and symmetric (vsymCH2) stretching vibrations respectively of the alkane chains. Both the shape and wavenumber of the methylene peaks can be used to identify film structure. The lower the peak wavenumber, moving towards 2917 cm-1 vasymCH2 and 2850 cm-1 vsymCH2, the more ordered the film. These films typically had peaks between 2924-2928 cm-1 and vsymCH2 between 2852-2855 cm-1 therefore the SAM was considered liquid like with random alkane chain packing and gauche defects [11]. A perfectly ordered alkanethiol SAM would give the vasymCH2 peak <2918 cm-1 [12] In general the broader the peak, the less ordered the SAM. Sharp well defined peaks at lower wavenumbers indicate a trans conformation in a semi-ordered state [13].
NH2 and COOH SAM Stability:
[0095] In order to use self-assembled monolayers effectively as biomedical implant coatings, or for any other application, they must show a high level of stability in vivo. The stability of SAMs formed by the Applicant was tested, under sonification, in the following three solutions: (1) denatured alcohol (85% ethanol, 15% methanol), (2) 0.16 M NaCl and (3) 0.1 M NaOH (pH 13). These three solutions were chosen because (1) ethanol is a general solvent used to clean samples between stages while building the device and for sterilization of implants, (2) corrosive 0.16 NaCl is used to simulate chloride concentrations found in the human body, and 0.1 M NaOH is used as an extreme alkaline solvent to go beyond conditions that may be experienced in the human body and is well known to remove physically adsorbed proteins. After the sonication, each sample was withdrawn and rinsed with ethanol and reverse osmosis treated water to remove NaCl and NaOH crystals from the surfaces, for PM-IRRAS measurement. The results for COOH and NH2 terminated SAMs are shown in FIG. 7 (The SAM was made by 25 sweeps of a 316L stainless steel coupon in a (top) 1 mM 11-amino-undecanethiol and (bottom) 10 mM mercapto-undecanoic acid in 0.1 M NaClO4 solution in denatured ethanol. The potential range used was between 0 and -1.8 VSCE; scan rate, 100 mVs-1. Error bars are 95% confidence intervals, n=3.).
[0096] For the NH2-terminated SAMs (FIG. 7, top) the trend and SAM quantity remaining after sonication was similar for all of the different solvents. This suggests that the solution chemistry was not a determining factor for the stability of NH2 SAMs. The most destructive parameter was the sonication itself and spontaneous dissolution of non-tightly bound molecules into the solvent. Since alkane stretching remained in all cases, it is concluded that most of SAM molecules are attached to the 316 L stainless steel surface covalently. Indeed, it is known in the art that any physically adsorbed molecules would be easily removed by such aggressive treatments.
[0097] In the case of the COOH-terminated SAM (FIG. 7, bottom), it seems that after an initial large drop (40-60%) in SAM surface concentration (after 1 hour), the SAM surface amount remains relatively constant, with a much lower rate of SAM detachment over the remaining hours of sonication. This trend was shown for all three sonication solutions.
[0098] However, the 0.1 M NaOH seemed to remove a larger amount of SAM from the surface. The explanation for this is that some of the COOH groups could have been bonded to the steel through a COO.sup.- metal carboxylate system and the high alkalinity of the NaOH solution was more successful at "delaminating" the molecules from the surface. In terms of other stability tests, COOH SAMs were also exposed to strong acids for 30 minutes HCl, H2SO4, and HNO3 and still remained secured to the surface, again demonstrating chemical rather than physical attachment to the surface.
[0099] It should also be mentioned that the zero-time samples in FIG. 7 might have some SAM molecules present in the second layer and/or physisorbed on the surface. These molecules were not removed by `simple` rinsing, and were thus detected by PM-IRRAS. However, these molecules were easily removed by sonication. Therefore, the relative amount of SAM remaining chemically-bond to the surface after sonication, FIG. 7, is actually higher than that shown in the figure.
[0100] The stability of COOH-terminated SAM on 316L stainless steel samples was also evaluated over a period of 8 days of contestant immersion in aqueous 0.16 M NaCl. FIG. 8 (the graphic shows the normalized CH2 area as a function of immersion time in aqueous 0.16 M NaCl. Error bars are 95% confidence intervals, n=3) demonstrates that more than 70% of the chemically-bound SAM remained on the surface after the 8th day. It is believed that this percentage is even higher due to the fact explained under the above paragraph. Similarly, contact angle measurement, FIG. 9 (error bars are 95% confidence intervals, n=3), proves the existence of the COOH-terminated SAM on the 316L stainless steel surface after 7 days of constant immersion in aqueous 0.16 M NaCl, as the value of contact angle remained high (in comparison to the control bare surface) and stable.
[0101] The influence of the COOH-terminated SAM on the general corrosion stability of the 316L stainless steel surface was also examined over a longer immersion-time period using EIS measurements. In these measurements, both the control (bare) 316L stainless steel samples and the COOH-modified 316L stainless steel samples were kept in corrosive 0.16M aqueous NaCl over a period of 6 days. Each day an EIS measurement was performed at open-circuit-potential, and the corresponding corrosion resistance was calculated by modeling the EIS spectra. By comparing the corrosion resistance of the SAM-modified sample to that of the control sample, the corresponding corrosion inhibition efficiency of the formed SAM was calculated and presented in FIG. 10 (the data was obtained from EIS measurements. Error bars are 95% confidence intervals, n=3). It is evident that the corrosion inhibition efficiency of the SAM is very high (ca. 90%), which demonstrates that the COOH-terminated SAM quite efficiently protects the underlying 316L stainless steel surface from corroding. This is important as the general corrosion of 316L stainless steel in a human body results in dissolution of toxic nickel and chromium species into the surrounding tissue, and the formed SAM significantly inhibit this process and thus increases both the biocompatibility and mechanical stability of the implanted 316L stainless steel structure (stent, screws, bone implants, etc.).
[0102] The Applicant concludes from these results that the alkanethiol SAMs adhered to a stainless steel surface and remained on the surface of the stainless steel even after aggressive removal attempts. The alkanethiol SAMs are stable on the stainless steel surface. These results validate the use of alkanethiols SAMs on stainless steel in biomedical or industrial applications.
COO--/Metal Complex Formation in COOH-Terminated SAMs:
[0103] In the case of the COOH SAM, the primary purpose was to use it as a linking molecule to further attach n-hydroxysuccinimide, NHS. However, when experimentation was performed to immobilize NHS, the NHS ester was not forming at all on freshly formed COOH SAMs. The PM-IRRAS spectroscopy shows the existence of metal carboxylate (FIG. 11). Carboxylates in solution have been well studied using infrared spectroscopy and can be formed in a variety of proposed physical structures, by electron sharing of a metal species and the oxygens from the carboxylic acid moiety.
[0104] Again, since the formation of the COOH SAM was strictly for application of binding proteins/antibodies or drugs, and this was not attainable with freshly-formed COOH SAMs, due to the formation of carboxylates (FIG. 11), a suitable method had to be developed to destroy the carboxylate/metal complex and produce chemically active COOH groups, while at the same time make sure not to remove the COOH SAM molecules from the surface.
[0105] In order to address this problem, the Applicant has surprisingly found that the immersion of the complexated carboxylate COO.sup.-/Metal SAM into a solution in which the species of the solution would have a thermodynamic tendency to scavenge the metal from the COO.sup.-/Metal SAM surface. It was theorized that weak acids could do this since weak acids, like acetic acid, CH3COOH, tend to form carboxylates in the presence of metal ions. An example is acetic acid turning into sodium acetate CH3COO.sup.-(Na.sup.+) in the presence of Na.sup.+ ions. In this case Ni, Fe or Cr ions which were present during the cyclic procedure will act just as the Na.sup.+ ion.
[0106] If this theory was valid the following would hold: (1) PM-IRRAS spectroscopy would show a substantial increase in the free v(C═O) group at approximately 1712 cm-1 and decrease in the vasymCOO.sup.- and vsymCOO.sup.- bands, as indicated by the arrows in FIG. 12. With the carboxylic acid group metal-free, it would be possible to undergo chemical reactions such as the n-hydroxylsuccuimide reaction. FIG. 12 demonstrates that phosphoric acid was able to destroy most of the metal (M) from the COOH carboxylate. The rise in 1712 cm-1 free v(C═O) bond and v(C--OH) at 1250 cm-1 with the lowering of the vasym(COO.sup.-) and vsym(COO.sup.-) peaks between 1542-1591 cm-1 and 1410-1448 cm-1 showed the quantity of COO.sup.-/Metal bonding had lowered significantly and that the first prediction was true.
[0107] The use of phosphoric acid at pH 1.5 was very efficient in removing the COO.sup.-/Metal carboxylate complexes. In fact, phosphoric acid gave the best results among all the investigated acids, and almost completely removed the carboxylate complex (almost no v(COO.sup.-) peaks were observed after the sample immersion, FIG. 12). This result not only shows that phosphoric acid is a good acid to use to remove carboxylates from the surface, but since the COOH SAM is still intact on the surface with intact methylene vibration in the 2900 cm-1 and 1456 cm-1 regions, the predominant attachment of the COOH-terminated SAM to the 316L SS surface must be through the sulfur group.
[0108] Other acids were also studied for the purpose of removing the COO.sup.-/Metal complexes, and the PM-IRRAS spectra are shown in FIG. 13 ((the increase of `free` COOH peak at ca. 1712 cm-1 and the decrease of the COO-- peak at ca. 1550-1600 cm-1 is the evidence of the degree of complex removal). The strong acids as hydrochloric, nitric and sulfuric acids did not significantly alter the chemical structure of the COOH SAMs that were in the carboxylate state. This was because strong acids dissociate completely, and therefore would have no inclination to cleave the metal, from the COO.sup.-/Metal SAM surface, and associate with SAM. In contrast, the weak acids; acetic, citric, malonic and phosphoric acid do have a tendency to form non-dissociated metal systems in solution, and were therefore able to remove the metal from the SAM surface. The effectiveness of removing the metal from the carboxylated SAMs went in the order: phosphoric (pKa 2.12, 7.21, and 12.32)>citric (pKa 3.14, 4.77 and 6.39)=malonic (pKa 2.83)> and acetic (pKa 4.76) acid.
Protein Immobilization on 316L stainless steel:
Glutaraldehyde Modified NH2 SAMs:
[0109] One possible way to use NH2 SAMs as linking molecules is to expose them to a solution of glutaraldehyde, which is a linear alkane five carbon molecule with aldehyde groups (HC═O) at both ends (FIG. 3). The aldehydes readily react with free amine groups (NH2) to form an imine (N═C) bond (FIG. 3, steps 2 and 3). This bond is particularly strong. Once this has occurred, one free aldehyde of the new complex remains exposed to solution and functionalizes the surface. In this state, proteins are particularly susceptible to chemical binding to this surface via the same imine bond reaction through amine groups on the protein (FIG. 3, step 4).
[0110] The use of glutaraldehyde to link proteins to amine terminated SAMs has been demonstrated many times in the literature [14-18]. The progress of this reaction in the present invention was monitored using PM-IRRAS. FIG. 14 shows that the glutaraldehyde reaction modified most of the free amines of the SAM surface, which were represented by EN-H vibration centered at 1589 cm-1. They were converted to imines, represented by a broad peak of vN═C at 1657 cm-1 [19, 20]. This reaction was quite fast requiring only 1 hr of NH2 SAM immersion in a 20% v/v solution of glutaraldehyde.
Proof of Covalently Immobilized BSA and FN to NH2 SAMs:
[0111] NH2 SAMs modified with glutaraldehyde were then incubated in BSA and FN solutions, to form the structure presented in FIG. 3 step 4. The PM-IRRAS spectra demonstrating the presence of the proteins is shown in FIG. 15.
[0112] All proteins can be represented by two peaks known as Amide I≈1600-1700 cm-1 and Amide II≈1485-1580 cm-1 bands. These bands are attributed to C═O stretching, C--N stretching, in plane N--H bending and some C--N stretching; vibrations which are common in most proteins [21]. These peaks are clearly visible in FIG. 15 and indicate the presence of BSA and FN proteins.
[0113] A control experiment was carried out in which two groups of samples were run simultaneously with BSA as the model protein to ascertain if the BSA was really immobilized on the NH2+GLU SAM surface. The control was an NH2 SAM which should only have physically adsorbed BSA on the surface (no GLU was used to further modify this SAM). These two samples were sonicated in 0.1 M NaOH for 10 minutes and measured using PM-IRRAS before and after. The results are shown in FIG. 16.
[0114] In both cases the two amide peaks are visible before the sonication, confirming that BSA is indeed present on the surfaces. However, after the sonication the amide peaks disappeared from the spectra for the physisorbed BSA, indicating that BSA was removed through the sonication. On the other hand, the spectra of the covalently immobilized BSA shows the presence of the amide peaks, indicating that the protein remained on this surface. The peaks are smaller than those before the sonication, which is to be expected since not all the BSA will react with the aldehyde groups on the surface and multiple BSA layers may have formed that did not contact the surface directly, but were identified by PM-IRRAS. This demonstrated that using NH2 SAMs with glutaraldehyde linking is a valid method for irreversibly and covalently immobilized proteins onto 316L stainless steel.
N-Hydroxysuccinimide Ester Modified COOH SAMs:
[0115] Since carboxylic acid SAMs on 316L SS displayed the desired surface chemistry (free C═O) after acid immersion and the SAMs were determined stable in a range of aggressive environments, they were then further modified to link proteins to the surface just as NH2 SAMs were. However the procedure is slightly different for COOH SAMs. As shown in the FIG. 2, it was the 1-Ethyl-3-3dimethyl aminopropyl carbodiimide (EDC) and hydroxysuccinimide (NHS) chemistry that was used for protein immobilization.
[0116] In this reaction, EDC binds to the COOH SAM forms an intermediate complex with the COOH SAM. However, the formed complex is then easily replaced by NHS to form an ester on the COOH SAM surface (FIG. 2, steps 2 to 4) denoted NHS ester SAM.
[0117] The formation of an NHS ester SAM was necessary, because it was a reaction intermediate, that could easily be replaced by the amine groups from the protein to form a stable peptide bond on the 316L SS surface FIG. 2 step 5. As mentioned previously this chemistry can also be used to attach other amine containing molecules, including certain antibodies or medications or peptides. To validate the presence of NHS molecule covalently bonded to the COOH SAM surface, PM-IRRAS spectra were analyzed. The presences of the peaks representing NHS attachment is shown in FIG. 17.
[0118] The peak frequently observed between 1814-1824 cm-1 is due to carbonyl stretch of the NHS as an ester formation on the surface, demonstrating that this is chemisorption rather than physisorption. The other peaks at 1744 cm-1 and 1789 cm-1 is asymmetric and symmetric carbonyl C═O stretching of the cyclic carbon ring from the NHS molecule.
[0119] The optimal soaking time for the COOH SAM reacting with NHS and EDC was determined to be around 2 hrs. The peak at approximately 1744 cm-1 was used to represent the quantity of NHS conjugation to the surface, and the peak at 1710 cm-1 (FIG. 18 (the increase of the 1744 cm-1 peak intensity evidences an increase in the amount of NHS bound to the COOH-terminated SAM)) was used to represent un-reacted and free carboxylic acid of the COOH SAM. This reaction progression is clearly evident in FIG. 18. The initial reaction was rapid as shown by a large increase the NHS ester formation peak. In conjunction, as time progressed over 2 hours, the peak around 1710 cm-1, representing free carboxylic acid, was significantly diminished.
[0120] To easily visualize the information shown in FIG. 18, the normalized integrated intensity (area) of those peaks as function of time is shown in FIG. 19. It can be seen from the graph that a plateau of bound NHS is reached around 60 minutes and there is no statistical difference in the amount of NHS bound to the surface for increasing the reaction time. However given these operating conditions when building up these SAMs, this reaction was run for 120 minutes to increase confidence that the reaction had in fact fully gone to completion.
Proof of Covalently Immobilized Fibronectin to COOH-SAMs:
[0121] When a protein contacts the NHS ester, the NHS ester bond is broken and substituted with a more stable NH2 amine from the protein as shown in FIG. 2. The advantage of this design as discussed previously is that the protein is now more firmly attached to the surface and robust to removal under shear flow compared to simply physisorbed protein. The protein removal tests were conducted exactly the same way as the BSA removal experiments for the NH2+glutaraldehyde SAMs (FIG. 16); sonication in 0.1 M NaOH for 10 minutes. The results are presented in FIG. 20.
[0122] The results of this experiment indicated that after 0.1 M NaOH sonication, the NHS ester peak at 1744 cm-1 was completely removed (top plot). This was to be expected since NHS esters are particularly unstable in water over the long term and prone to hydrolysis. Therefore, the use of 0.1 M NaOH speeds up the process. The major observation in FIG. 20 is that after sonication, physisorbed FN was completely removed from the 316L stainless steel surface, while a significant amount of the FN covalently attached to the NHS SAM still remained on the 136L stainless steel surface. The quantification of the amounts was made by the integrated intensity of the amide peaks in the PM-IRRAS spectra and presented in FIG. 21 (the samples were sonicated in aqueous 0.1 M NaOH for 10 minutes, followed by dionized water and denatured ethanol rinsing, error bars represent 95% confidence intervals, n=3).
[0123] The results demonstrate that the physisorbed FN (control surface) was completely removed from the surface, while 48% (0.05 confidence interval) of FN remained on the NHS-modified surface. Actually, although the chemically modified 316L stainless steel surface was immersed in 2.0 M NaCl and then thoroughly rinsed with water before the stability experiment (`before sonication` in FIG. 21), the person skilled in the art will understand that this was not sufficient to completely remove all the physisorbed FN, which was in turn efficiently removed during the sonication in 0.1M NaOH. Thus, it has been demonstrated that the amount of FN remained irreversibly attached to the SAM-modified surface is actually larger than 48%.
[0124] These tests have demonstrated that NHS/EDC chemistry can efficiently be used to immobilize fibronectin to a stainless steel surface, and that the protein displayed superior surface stability compared to physically adsorbed fibronectin on bare stainless steel.
Endothelial Cell Attachment on Covalently Immobilized Fibronectin on COOH SAMs:
[0125] The Applicant has demonstrated that fibronectin can be efficiently attached to the 316L stainless steel surface using the novel electrochemical SAM-formation method developed by the present invention. The next step was to demonstrate that such a modified 316L stainless steel surface was a better substrate for endothelial cell attachment than the bare 316L stainless steel surface, which is commonly used for coronary stents. As known in the vascular art, fibronectin plays a strong role in cell attachment and subsequent proliferation on substrates [22, 23]. To investigate this, the fibronectin-coated COOH-SAM 316L stainless steel surface was tested for EC/surface interactions (cell attachment) and the results are presented in FIG. 22.
[0126] The cell density on the fibronectin-coated COOH-SAM 316L stainless steel surface was measured and compared to that on a control (bare 316L stainless steel) and a COOH-terminated SAM. The results of the experiment indicate the trend of increasing cell attachment with time on all substrates, demonstrating that none of the surfaces are cytotoxic. However, the fibronectin-coated COOH-SAM 316L stainless steel surface showed approximately 22%, 21%, and 25% improvement in cell density compared to the bare 316L stainless steel surface at 1, 2 and 4 hrs, respectively, at a significance of p<0.05 in all cases. The increase was even larger when compared to the COOH-terminated surface.
Angiogram--In-Vivo Experiments with Stent Implantation in an Animal
[0127] Using an embodiment of the method of the present invention, the surface of a 316L stainless steel stent was modified to covalently attach fibronectin. The modified stent was implanted in a femoral artery of a Beagle dog. As a control, a commercial 316L stainless steel stent was implanted. The angiogram was recorded 28 days post implantation. FIG. 23(a) shows that in-stent restenosis occurred at the stent implantation site (see arrow). On the contrary, the artery in which the FN-modified 316L stainless steel stent was implanted (FIG. 23(b)), is completely patent and does not show any signs of in-stent restenosis. This clearly demonstrates that the active agent covalently/irreversibly linked to the SAMs modified surfaces rendered the stent surface more biocompatible and functional.
[0128] These examples show that stable and robust SAMs of functionalized --NH2 and --COOH were formed on the surface of 316L stainless steel employing the electrochemical potentiodynamic polarization method involving the use of NiCr as a counter electrode. Furthermore, fibronectin was covalently/irreversibly linked to the --NH2 and --COOH SAMs modified surfaces. Indeed, the covalently immobilized proteins showed a stable and strong attachment compared to physically adsorbed and non-covalently bonded proteins on bare stainless steel. Finally, endothelial cell attachment to the fibronectin-modified 316 L stainless steel surface was significantly enhanced compared to a bare 316 stainless steel surface in vitro, indicating the higher biocompatibility of the surface for blood-contacting implant.
Monolayer Assembly to NiTi:
[0129] The electrochemical SAM-formation procedure demonstrated on stainless steel was also used to form an COOH-terminated SAM on a NiTi surface. The evidence of the presence of SAM on the NiTi surface is presented in FIG. 24, which shows the presence of the two characteristic CH2 vibrations. Indeed, the spectrum shows two CH2 peaks at 2926 and 2853 cm-1 evidencing the presence of the SAM on the surface. This demonstrated that an embodiment of the method of the present invention used to form the SAM on stainless steel also effectively produced the same SAM on NiTi.
[0130] The presence of the COOH-terminated SAM on a NiTi surface is also evidenced in FIG. 25 (the spectrum was recorded after 30 minute immersion of the sample in phosphoric acid), which shows the bending vibrations of the CH2 SAM group between 1400 and 1500 cm-1. The modified sample was immersed for 30 minutes in phosphoric acid pH 1.5, prior recording the spectra, in order to remove the COO-metal complex. The removal of the complex is evidenced by the presence of the `free` COOH vibration at 1710 cm-1. The wide compositional vibration between 1400 and 1500 cm-1 is related to symCOO-- with CH2. This demonstrated that an embodiment of the method of the present invention used to form the SAM on stainless steel also effectively produced the same SAM on NiTi.
[0131] As shown, in FIG. 26, the COOH-terminated SAM-modified NiTi surface was further modified with NHS to render the end COOH group chemically reactive for further protein binding. The PM-IRRAS spectrum in FIG. 26 evidences the presence of NHS on the surface and its chemical bond with the COOH-terminated SAM. Note the absence of the `free` COOH group vibration at 1710 cm-1, which demonstrates that COOH completely reacted with NHS. This demonstrated that an embodiment of the method of the present invention used to form the SAM is effective in producing a chemically reactive SAM on NiTi, which can be further modified by any desired chemical moiety.
[0132] Furthermore, the PM-IRRAS spectrum shown in FIG. 27 demonstrates the presence of FN on the NiTi surface. The spectrum was taken after 20 minutes of sonication of the FN-modified NiTi sample in 0.1M NaOH, which would be sufficient to remove any protein layer that is physisorbed on the surface. Hence, the spectra in FIG. 27 demonstrates that FN is indeed chemically attached to the NiTi surface and is very stable.
REFERENCES
[0133] Each of the following are incorporated by reference into the present application. To the extent that a definition or discussion of a term in any of the references disagrees with or undermines the definitions and disclosure set forth in the specification, the specification definitions and disclosure apply. [0134] 1. Briand E., et al., Building of an immunosensor: How can the composition and structure of the thiol attachment layer affect the immunosensor efficiency?, Biosensors and Bioelectronics 2006. 22: p. 440-448. [0135] 2. Shavel A., Gaponik N., and Eychmuller A., Covalent Linking of CdTe Nanocrystals to Amino-Functionalized Surfaces. ChemPhysChem, 2005(6): p. 449-451. [0136] 3. Delvaux M. and Demoustier-Champagne S., Immobilisation of glucose oxidase within metallic nanotubes arrays for application to enzyme biosensors. Biosensors and Bioelectronics, 2003. 18: p. 943-951. [0137] 4. Patel N., et al., Immobilization of Protein Molecules onto Homogeneous and Mixed Carboxylate-Terminated Self-Assembled Monolayers. Langmuir, 1997(13): p. 6485-6490. [0138] 5. BioVision, Quick Cell Proliferation Assay Kit II, B.R. Products, Editor: 980 Linda Vista Avenue, Mountain View, Calif. [0139] 6. Petrovic Z., Modificiranje povrsina samoorganizirajucim monoslojevima organskih molekula, in Fakultet kumijkog inzenjerstva tehnologije. 2008, Zagrebu. [0140] 7. Azzaroni O., Schilardi P L., and Salvarezza R C., Metal electrodeposition on self-assembled monolayers: a versatile tool for pattern transfer on metal thin films. Electrochemica Acta, 2003. 48: p. 3107-3114. [0141] 8. Esplandiu M. and Hagenstrom H., Functionalized self-assembled monolayers and their influence on silver electrodeposition. Solid State Ionics 2002. 150: p. 39-52. [0142] 9. Benji S., et al., Electrochemical Self-Assembly of Alkanethiolate Molecules on Ni(111) and Polycrystalline Ni Surfaces. Journal of Physical Chemistry B, 2005. 109(49): p. 23450-23460. [0143] 10. Volmer M., Stratmann M., and Viefhaus H., Electrochemical and Electron Spectroscopic Investigations of Iron Surfaces Modified with Thiols. Surface and Interface Analysis, 1990. 16(27): p. 282. [0144] 11. Liakos I L., et al., Comparative study of self-assembly of a range of monofunctional aliphatic molecules on magnetron-sputter aluminum. Surface and Interface Analysis, 2004. 36: p. 347-354. [0145] 12. Raman A., et al., Formation of Self-Assembled Monolayers of Alkylphosphonic Acid on the Native Oxide Surface of SS316L. Langmuir, 2006. 22: p. 6469-6472. [0146] 13. Yan L., et al., Formation and reaction of interchain carboxylic anhydride groups on self-assembled monolayers on gold Langmuir, 1997. 13: p. 6704-6712. [0147] 14. Zamborini F P. and Crooks R M., Corrosion Passivation of Gold by n-Alkanethiol Self-Assembled Monolayers Effect of Chain Length and End Group. Langmuir, 1998. 14(12): p. 3279-3286. [0148] 15. Sun J-J., Fang H-Q., and Chen H-Y., Immobilization of horseradish peroxidase on a self-assembled monolayer modified gold electrode for the detection of hydrogen peroxide. Analyst, 1998(123): p. 1365-1366. [0149] 16. Zawisza I., et al., Electrochemical, neutron reflectivity and in situ PM-FT-IRRAS studies of a monolayer of n-ocadecanol at a Au(111) electrode surface. Electrochemica Acta, 2004. 49: p. 3651-3664. [0150] 17. Toda M., et al., Complement activation on surfaces carrying amino groups. Biomaterials, 2008. 29: p. 407-417. [0151] 18. Frey B. and Corn R., Covalent Attachment and Derivatization of Poly(L-lysine) Monolayers on Gold Surfaces As Characterized by Polarization-Modulation FT-IR Spectroscopy. Anal. Chem., 1996. 68: p. 3187-3193. [0152] 19. Jirousova J. and Davidek J., Reactions of n-Hexanal with Glycine in Model Systems. Z. Lebensm. Unters.-Forsch, 1975(157): p. 269-276. [0153] 20. Pradier C-M., et al., Specific binding of avidin to biotin immobilised on modified gold surfaces Fourier transform infrared reflection absorption spectroscopy analysis. Surface Science, 2002(502-503): p. 193-202. [0154] 21. Stuart B., ed. Infrared Spectrsocpy: Fundmentals and Applications. 2004, Jon Wiley & Sons Inc.: Chichester, West Sussex, England. [0155] 22. Johansson S., et al., Fibronectin-Integrin Interactions. Frontiers in Bioscience, 1997(2): p. 126-146. [0156] 23. McKeown-Longo P. and Mosher D F., Binding of Plasma Fibronectin to Cell Layers of Human Skin Fibroblasts. The Journal of Cell Biology, 1983. 97: p. 466-472.
[0157] It should be appreciated that the invention is not limited to the particular embodiments described and illustrated herein but includes all modifications and variations falling within the scope of the invention as defined in the appended claims.
[0158] For example, although the present invention has been described in relation with a medical application, industrial application requiring the modification of a metal surface for covalently linking different types of agents can also be used. Furthermore, although the present invention has been described in relation to stainless steel, NiTi or CoCr, any types or grades of stainless steel or metal presenting similar drawbacks as stainless steel can be used in the performance of the invention.
User Contributions:
Comment about this patent or add new information about this topic:
| People who visited this patent also read: | |
| Patent application number | Title |
|---|---|
| 20150163471 | CAMERA ARRAY SYSTEM |
| 20150163470 | DEVICE AND METHOD FOR DISPLAYING INFORMATION IN LIGHT ILLUMINATED AREA AHEAD OF VEHICLE |
| 20150163469 | PROJECTION VIDEO DISPLAY APPARATUS |
| 20150163468 | PROJECTOR |
| 20150163467 | ILLUMINATION DEVICE, PROJECTION TYPE IMAGE DISPLAY DEVICE, AND OPTICAL DEVICE |