Patent application title: PROTEIN-BASED STREPTOCOCCUS PNEUMONIAE VACCINES
Inventors:
Yaffa Mizrachi-Nebenzahl (Beer Sheva, IL)
Yaffa Mizrachi-Nebenzahl (Beer Sheva, IL)
IPC8 Class: AA61K3909FI
USPC Class:
4241901
Class name: Antigen, epitope, or other immunospecific immunoeffector (e.g., immunospecific vaccine, immunospecific stimulator of cell-mediated immunity, immunospecific tolerogen, immunospecific immunosuppressor, etc.) amino acid sequence disclosed in whole or in part; or conjugate, complex, or fusion protein or fusion polypeptide including the same disclosed amino acid sequence derived from bacterium (e.g., mycoplasma, anaplasma, etc.)
Publication date: 2009-06-11
Patent application number: 20090148470
Inventors list |
Agents list |
Assignees list |
List by place |
Classification tree browser |
Top 100 Inventors |
Top 100 Agents |
Top 100 Assignees |
Usenet FAQ Index |
Documents |
Other FAQs |
Patent application title: PROTEIN-BASED STREPTOCOCCUS PNEUMONIAE VACCINES
Inventors:
Yaffa Mizrachi Nebenzahl
Agents:
WINSTON & STRAWN LLP;PATENT DEPARTMENT
Assignees:
Origin: WASHINGTON, DC US
IPC8 Class: AA61K3909FI
USPC Class:
4241901
Abstract:
The present invention is primarily directed to a method for preventing
infection of a mammalian subject with S. pneumoniae, wherein said method
comprises administering to said subject an effective amount of one or
more S. pneumoniae cell wall and/or cell membrane proteins and/or
immunogenically-active fragments, derivatives or modifications thereof,
wherein said proteins are selected from a defined group of immunogenic
proteins. The present invention further provides vaccine compositions
containing said cell wall and/or cell membrane proteins.Claims:
1. A method for protecting a mammalian subject against infection from S.
pneumoniae by administering to said subject a vaccine composition
comprising an isolated protein selected from one or more S. pneumoniae
cell wall or cell membrane proteins or immunogenically-active protein
fragments, derivatives or modifications thereof which are associated with
an age-dependent immune response and which are administered in an amount
effective to induce an immune response to S. pneumoniae to thus protect
said subject from infection from S. pneumoniae.
2. The method according to claim 1 wherein the one or more proteins are effective in all age groups, including those age groups which do not produce anti-S. pneumoniae antibodies following inoculation with polysaccharide-based vaccines.
3. The method according to claim 2, wherein the subject is an infant under four years of age.
4. The method according to claim 2 wherein the subject is an infant under two years of age.
5. The method according to claim 2, wherein the subject is an elderly subject.
6. The method of claim 1 wherein said S. pneumoniae cell wall and/or cell membrane protein is selected from the group consisting of: phosphoenolpyruvate protein phosphotransferase (Accession No. NP--345645, SEQ ID NO:4); phosphoglucomutase/phosphomannomutase family protein (Accession No. NP--346006, SEQ ID NO:5); trigger factor (Accession No. NP--344923, SEQ ID NO:6); elongation factor G/tetracycline resistance protein (tetO), (Accession No. NP--34481 1, SEQ ID NO:7); NADH oxidase (Accession No. NP--345923, SEQ ID NO:8); Aspartyl/glutamyl-tRNA amidotransferase subunit C (Accession No. NP--344960, SEQ ID NO:9); cell division protein FtsZ (Accession No. NP--346105, SEQ ID NO:10); L-lactate dehydrogenase (Accession No. NP--345686, SEQ ID NO:11); glyceraldehyde 3-phosphate dehydrogenase (GAPDH), (Accession No. NP--346439, SEQ ID NO:12); fructose-bisphosphate aldolase (Accession No. NP--345117, SEQ ID NO:13); UDP-glucose 4-epimerase (Accession No. NP--346261, SEQ ID NO:14); elongation factor Tu family protein (Accession No. NP--358192, SEQ ID NO:15); Bifunctional GMP synthase/glutamine amidotransferase protein (Accession No. NP--345899, SEQ ID NO:16); glutamyl-tRNA synthetase (Accession No. NP--346492, SEQ ID NO:17); glutamate dehydrogenase (Accession No. NP--345769, SEQ ID NO:18); Elongation factor TS (Accession No. NP--346622, SEQ ID NO:19); phosphoglycerate kinase (TIGR4) (Accession No. AAK74657, SEQ ID NO:20); 30S ribosomal protein S1 (Accession No. NP--345350, SEQ ID NO:21); 6-phosphogluconate dehydrogenase (Accession No. NP--357929, SEQ ID NO:22); aminopeptidase C (Accession No. NP--344819, SEQ ID NO:23); carbomoyl-phosphate synthase (large subunit) (Accession No. NP--345739, SEQ ID NO:24); PTS system, mannose-specific IIAB components (Accession No. NP--344822, SEQ ID NO:25); 30S ribosomal protein S2 (Accession No. NP--346623, SEQ ID NO:26); dihydroorotate dehydrogenase 1B (Accession No. NP--358460, SEQ ID NO:27); aspartate carbamoyltransferase catalytic subunit (Accession No. NP--345741, SEQ ID NO:28); elongation factor Tu (Accession No. NP--345941, SEQ ID NO:29); Pneumococcal surface immunogenic protein A (PsipA) (Accession No. NP--344634, SEQ ID NO:30); phosphoglycerate kinase (R6) (Accession No. NP--358035, SEQ ID NO:31); ABC transporter substrate-binding protein (Accession No. NP--344690, SEQ ID NO:32); endopeptidase O (Accession No. NP--346087, SEQ ID NO:33); Pneumococcal surface immunogenic protein B (PsipB) (Accession No. NP--358083, SEQ ID NO:34); Pneumococcal surface immunogenic protein C (PsipC) (Accession No. NP--345081, SEQ ID NO:35).
7. The method according to claim 1, wherein the vaccine composition includes one or more pharmaceutically acceptable adjuvants.
8. The method according to claim 1 wherein the immune response is effective against localized infection of a mucosal tissue by S. pneumoniae.
9. The method according to claim 8, wherein the one or more S. pneumoniae proteins to be administered are lectins.
10. The method according to claim 1, wherein the immune response is effective against systemic infection with S. pneumoniae.
11. The method according to claim 10, wherein the one or more S. pneumoniae proteins to be administered are non-lectins.
12. The method according to claim 1, wherein the S. pneumoniae proteins to be administered are a mixture of lectins and non-lectins.
13. The method according to claim 1, wherein the subject is a human subject.
14. The method of claim 1 wherein the subject is protected against S. pneumoniae infection by administration of the vaccine composition prior to occurrence of the infection.
15. A vaccine composition comprising as the active ingredient one or more isolated protein selected from S. pneumoniae cell wall or cell membrane proteins or immunogenically-active fragments, derivatives or modifications thereof which are associated with an age-dependent immune response, optionally together with one or more pharmaceutically acceptable adjuvants.
16. The vaccine composition according to claim 15, wherein said S. pneumoniae cell wall and/or cell membrane protein is selected from the group consisting of: phosphoenolpyruvate protein phosphotransferase (Accession No. NP--345645, SEQ ID NO:4); phosphoglucomutase/phosphomannomutase family protein (Accession No. NP--346006, SEQ ID NO:5); trigger factor (Accession No. NP--344923, SEQ ID NO:6); elongation factor G/tetracycline resistance protein (tetO), (Accession No. NP--344811, SEQ ID NO:7); NADH oxidase (Accession No. NP--345923, SEQ ID NO:8); Aspartyl/glutamyl-tRNA amidotransferase subunit C (Accession No. NP--344960, SEQ ID NO:9); cell division protein FtsZ (Accession No. NP--346105, SEQ ID NO:10); L-lactate dehydrogenase (Accession No. NP--345686, SEQ ID NO:11); glyceraldehyde 3-phosphate dehydrogenase (GAPDH), (Accession No. NP--346439, SEQ ID NO:12); fructose-bisphosphate aldolase (Accession No. NP--345117, SEQ ID NO:13); UDP-glucose 4-epimerase (Accession No. NP--346261, SEQ ID NO:14); elongation factor Tu family protein (Accession No. NP--358192, SEQ ID NO:15); Bifunctional GMP synthase/glutamine amidotransferase protein (Accession No. NP--345899, SEQ ID NO:16); glutamyl-tRNA synthetase (Accession No. NP--346492, SEQ ID NO:17); glutamate dehydrogenase (Accession No. NP--345769, SEQ ID NO:18); Elongation factor TS (Accession No. NP--346622, SEQ ID NO:19); phosphoglycerate kinase (TIGR4) (Accession No. AAK74657, SEQ ID NO:20); 30S ribosomal protein S1 (Accession No. NP--345350, SEQ ID NO:21); 6-phosphogluconate dehydrogenase (Accession No. NP--357929, SEQ ID NO:22); aminopeptidase C (Accession No. NP--344819, SEQ ID NO:23); carbomoyl-phosphate synthase (large subunit) (Accession No. NP--345739, SEQ ID NO:24); PTS system, mannose-specific IIAB components (Accession No. NP--344822, SEQ ID NO:25); 30S ribosomal protein S2 (Accession No. NP--346623, SEQ ID NO:26); dihydroorotate dehydrogenase 1B (Accession No. NP--358460, SEQ ID NO:27); aspartate carbamoyltransferase catalytic subunit (Accession No. NP--345741, SEQ ID NO:28); elongation factor Tu (Accession No. NP--345941, SEQ ID NO:29); Pneumococcal surface immunogenic protein A (PsipA) (Accession No. NP--344634, SEQ ID NO:30); phosphoglycerate kinase (R6) (Accession No. NP--358035, SEQ ID NO:31); ABC transporter substrate-binding protein (Accession No. NP--344690, SEQ ID NO:32); endopeptidase O (Accession No. NP--346087, SEQ ID NO:33); Pneumococcal surface immunogenic protein B (PsipB) (Accession No. NP--358083, SEQ ID NO:34); Pneumococcal surface immunogenic protein C (PsipC) (Accession No. NP--345081, SEQ ID NO:35).
17. The vaccine composition according to claim 15, wherein the one or more S. pneumoniae cell wall and/or cell membrane proteins are lectins.
18. The vaccine composition according to claim 15, wherein the one or more S. pneumoniae cell wall and/or cell membrane proteins are non-lectins.
19. The vaccine composition according to claim 15, wherein the one or more S. pneumoniae cell wall and/or cell membrane proteins are a mixture of lectins and non-lectins.
20. The vaccine composition according to claim 15, which is specifically formulated for an infant under four years of age, an infant under two years of age, or for an elderly subject.
Description:
CROSS REFERENCE TO RELATED APPLICATIONS
[0001]This application is a divisional of U.S. application Ser. No. 10/953,513 filed on Sep. 30, 2004, which is a continuation-in-part of PCT International application no. PCT/IL03/00271, filed on Apr. 1, 2003, which designated the U.S. and claims priority to U.S. provisional application No. 60/368,981, filed on Apr. 2, 2002. The entire contents of these applications are incorporated herein by reference.
FIELD OF THE INVENTION
[0002]The present invention relates to a method for vaccinating subjects against infection with Streptococcus pneumoniae. More specifically, the present invention provides a vaccination method based on the use of immunogenic S. pneumoniae cell wall and cell membrane proteins and/or immunogenically-active fragments thereof, as well as vaccine compositions comprising said proteins and/or fragments.
BACKGROUND OF THE INVENTION
[0003]The Gram-positive bacterium Streptococcus pneumoniae is a major cause of disease, suffering and death worldwide. Diseases caused by infection with this agent include otitis media, pneumonia, bacteremia, meningitis and sepsis. In some cases, infected individuals may become asymptomatic carriers of S. pneumoniae, thereby readily allowing the rapid spread of this infective agent throughout the population. In view of the serious consequences of infection with S. pneumoniae, as well as its rapid spread within and between populations, there is an urgent need for safe, effective vaccination regimes.
[0004]Current methods of vaccination are based on inoculation of the subject with polysaccharides obtained from the capsules of S. pneumoniae. While these polysaccharide-based vaccine preparations have been found to be reasonably efficacious when used to prevent infection in adult populations, they are significantly less useful in the treatment of young children (under two years of age) and the elderly. One commonly-used capsular polysaccharide 23-valent vaccine, for example, has been found to be only 60% effective in preventing S. pneumoniae invasive disease in elderly subjects and completely incapable of yielding clinically-useful antibody responses in the under-two age group [Shapiro E. D. et al. (1991) N. Engl. J. Med. 325: 1453-1460].
[0005]In an attempt to increase the immunogenicity of these vaccines, various compositions comprising capsular polysaccharides that have been conjugated with various carrier or adjuvant proteins have been used. Although vaccines of this type constitute an improvement in relation to the unconjugated polysaccharide vaccines, they have not overcome the problem of coverage, since they are effective against only about 10% of the 92 known capsular serotypes. Consequently, upon vaccination, repopulation with serotypes not present in the vaccine occurs.
[0006]In the cases of certain other bacteria of pathogenic importance for human and other mammalian species, vaccines comprising immunogenic virulence proteins are currently being developed. Such protein-based vaccines should be of particular value in the case of vulnerable subjects such as very young children, in view of the fact that such subjects are able to produce antibodies against foreign proteins. Unfortunately, very little is known of the molecular details of the life cycle of S. pneumoniae, or of the nature of role of the various virulence factors which are known or thought to be involved in targeting and infection of susceptible hosts.
[0007]It is a purpose of the present invention to provide a method for protecting individuals against infection with S. pneumoniae by the use of a protein-based vaccine.
[0008]It is another purpose of the invention to provide a protein-based vaccine that is prepared from the immunogenic cell wall and/or cell membrane proteins of S. pneumoniae.
[0009]It is yet another purpose of the invention to provide a vaccination method that overcomes the problems and drawbacks of prior art methods.
[0010]Further objects and advantages of the present invention will become apparent as the description proceeds.
SUMMARY OF THE INVENTION
[0011]It has now been found that it is possible to protect individuals against infection with S. pneumoniae by means of administering to said individuals a vaccine composition comprising one or more proteins isolated from the outer layers of the aforementioned bacteria and/or one or more immunogenically-active fragments, derivatives or modifications thereof. Unexpectedly, it was found that such vaccine compositions are effective against a wide range of different S. pneumoniae serotypes, and in all age groups, including those age groups which do not produce anti-S. pneumoniae antibodies following inoculation with polysaccharide-based vaccines.
[0012]It is to be noted that in the context of the present invention, the terms "fragments", "derivatives" and "modifications" are to be understood as follows:
[0013]"Fragment": a less than full length portion of the native sequence of the protein in question, wherein the sequence of said portion is essentially unchanged as compared to the relevant part of the sequence of the native protein.
[0014]"Derivative": a less than full length portion of the native sequence of the protein in question, wherein either the sequence further comprises (at its termini and/or within said sequence itself) non-native sequences, i.e. sequences which do not form part of the native protein in question. The term "derivative" also includes within its scope molecular species produced by conjugating chemical groups to the amino residue side chains of the native proteins or fragments thereof, wherein said chemical groups do not form part of the naturally-occurring amino acid residues present in said native proteins.
[0015]"Modification": a full length protein or less than full length portion thereof comprising non-native amino acid residues and sequences of such non-native residues, which have been introduced as a consequence of mutation of the native sequence (by either random or site-directed processes).
[0016]The term "immunogenically-active" is used herein in ordinary sense to refer to an entity (such as a protein or its fragment or derivative) that is capable of eliciting an immune response when introduced into a host subject.
[0017]The present invention is primarily directed to a method for preventing infection of mammalian subjects with S. pneumoniae, wherein said method comprises administering to a subject in need of such treatment an effective amount of one or more S. pneumoniae cell wall and/or cell membrane proteins and/or immunogenically-active fragments, derivatives or modifications thereof, wherein said proteins are selected from the group consisting of: phosphoenolpyruvate protein phosphotransferase, phosphomannomutase, trigger factor, elongation factor G, tetracycline resistance protein (tetO), DNA directed RNA polymerase alpha-chain, NADH oxidase, glutamyl-tRNA amidotransferase subunit A, N utilization substance protein A homolog, XAA-HIS dipeptidase, cell division protein ftsz, zinc metalloproteinase in SCAA 5' region (ORF 6), L-lactate dehydrogenase, glyceraldehyde 3-phosphate dehydrogenase (GAPDH), fructose-biphosphate aldolase, UDP-glucose 4-epimerase, GTP binding protein typA/BipA, GMP synthase, glutamyl-tRNA synthetase, NADP-specific glutamate dehydrogenase, Elongation factor TS, phosphoglycerate kinase (cell wall protein), pyridine nucleotide-disulfide oxidoreductase, 40S ribosomal protein S1, 6-phosphogluconate dehydrogenase, aminopeptidase C, carbomyl-phosphate synthase (large subunit), PTS system mannose-specific IIAB components, ribosomal protein S2, dihydroorotate dehydrogenase, aspartate carbamoyltransferase, elongation factor Tu, Pneumococcal surface immunogenic protein A (PsipA), phosphoglycerate kinase (cell membrane protein), ABC transporter substrate-binding protein, endopeptidase O, Pneumococcal surface immunogenic protein B (PsipB) and Pneumococcal surface immunogenic protein C (PsipC).
[0018]The means used to identify the aforementioned S. pneumoniae proteins, and their unique public access database accession codes will be disclosed and described hereinbelow.
[0019]In a preferred embodiment of the method of the present invention, one or more adjuvants may be optionally administered to the subject together with one or more of the aforementioned S. pneumoniae cell wall and/or cell membrane proteins or fragments thereof.
[0020]In one particularly preferred embodiment, the method of the present invention for preventing infection of mammalian subjects by S. pneumoniae comprises administering to a subject in need of such treatment an effective amount of S. pneumoniae glyceraldehyde 3-phosphate dehydrogenase (GAPDH).
[0021]In another particularly preferred embodiment, the method of the present invention for preventing infection of mammalian subjects by S. pneumoniae comprises administering to a subject in need of such treatment an effective amount of S. pneumoniae fructose-biphosphate aldolase.
[0022]In a further particularly preferred embodiment, the method of the present invention for preventing infection of mammalian subjects by S. pneumoniae comprises administering to a subject in need of such treatment an effective amount of one or more immunogenically-active fragments of the aforementioned S. pneumoniae fructose-biphosphate aldolase protein. Although many such active fragments may be generated from this protein and used in the presently-disclosed method, in an especially preferred embodiment, the active fragment used corresponds to the peptide encoded by the first 294 nucleotides of the fructose biphosphate aldolase gene (SP0605 Streptococcus pneumoniae TIGR4), referred to herein as SEQ ID no.1, and defined in the sequence listing that forms an integral part of the present disclosure.
[0023]In one preferred embodiment of the method of the invention, the cell wall and/or cell membrane proteins are S. pneumoniae proteins that are associated with an age-related immune response.
[0024]The term "age-related immune response" (as used throughout this application) indicates that the ability of subjects to produce antibodies to the protein or proteins causing said immune response increases with age. In the case of human subjects, said ability is measured over a time scale beginning with neonates and ending at approximately age four years and adults. In non-human mammalian subjects, the "age-related immune response" is measured over an age range extending from neonates to an age at which the immune system of the young mammal is at a stage of development comparable to that of a pre-puberty human child and adults.
[0025]In another preferred embodiment of the method of the invention, the cell wall and/or cell membrane proteins are lectins.
[0026]In another preferred embodiment of the method of the invention, the cell wall and/or cell membrane proteins are non-lectins.
[0027]In another preferred embodiment of the method of the invention, the cell wall and/or cell membrane proteins are a mixture of lectins and non-lectins.
[0028]The term "lectins" is used hereinabove and hereinbelow to indicate proteins having the ability to specifically bind to polysaccharides or oligosaccharides. Conversely, the term "non-lectins" is used to refer to proteins lacking the aforementioned saccharide-binding property.
[0029]In one preferred embodiment of the method of the invention, the mammalian subject is a human subject.
[0030]In another aspect, the present invention is directed to a vaccine composition comprising as the active ingredient one or more S. pneumoniae cell wall and/or cell membrane proteins and/or immunogenically-active fragments, derivatives or modifications thereof, wherein said proteins are selected from the group consisting of: phosphoenolpyruvate protein phosphotransferase, phosphomannomutase, trigger factor, elongation factor G, tetracycline resistance protein (tetO), DNA directed RNA polymerase alpha-chain, NADH oxidase, glutamyl-tRNA amidotransferase subunit A, N utilization substance protein A homolog, XAA-HIS dipeptidase, cell division protein ftsz, zinc metalloproteinase in SCAA 5' region (ORF 6), L-lactate dehydrogenase, glyceraldehyde 3-phosphate dehydrogenase (GAPDH), fructose-biphosphate aldolase, UDP-glucose 4-epimerase, GTP binding protein typA/BipA, GMP synthase, glutamyl-tRNA synthetase, NADP-specific glutamate dehydrogenase, Elongation factor TS, phosphoglycerate kinase (cell wall protein), pyridine nucleotide-disulfide oxidoreductase, 40S ribosomal protein S1, 6-phosphogluconate dehydrogenase, aminopeptidase C, carbomyl-phosphate synthase (large subunit), PTS system mannose-specific IIAB components, ribosomal protein S2, dihydroorotate dehydrogenase, aspartate carbamoyltransferase, elongation factor Tu, Pneumococcal surface immunogenic protein A (PsipA), phosphoglycerate kinase (cell membrane protein), ABC transporter substrate-binding protein and endopeptidase O, Pneumococcal surface immunogenic protein B (PsipB) and Pneumococcal surface immunogenic protein C (PsipC).
[0031]The vaccine compositions of the present invention may also contain other, non-immunologically-specific additives, diluents and excipients. For example, in many cases, the vaccine compositions of the present invention may contain--in addition to the S. pneumoniae cell-wall and/or cell-membrane protein(s)--one or more adjuvants.
[0032]In one particularly preferred embodiment, the vaccine composition of the present invention comprises an effective amount of S. pneumoniae glyceraldehyde 3-phosphate dehydrogenase (GAPDH) as the active ingredient.
[0033]In another particularly preferred embodiment of the present invention, the vaccine composition comprises an effective amount of S. pneumoniae fructose-biphosphate aldolase as the active ingredient.
[0034]In a further particularly preferred embodiment, the vaccine composition of the present invention for preventing infection of mammalian subjects by S. pneumoniae comprises an effective amount of one or more immunogenically-active fragments of the aforementioned S. pneumoniae fructose-biphosphate aldolase protein. Although many such active fragments may be generated from this protein and incorporated into the presently-disclosed composition, in an especially preferred embodiment, the active fragment used corresponds to the peptide encoded by the first 294 nucleotides of the fructose biphosphate aldolase gene (SP0605 Streptococcus pneumoniae TIGR4), herein referred to as SEQ ID no.1.
[0035]The aforementioned vaccine compositions may clearly be used for preventing infection of the mammalian subjects by S. pneumoniae. However, said vaccine composition is not restricted to this use alone. Rather it may be usefully employed to prevent infection by any infectious agent whose viability or proliferation may be inhibited by the antibodies generated by a host in response to the inoculation therein of the one or more S. pneumoniae proteins provided in said composition.
[0036]In one preferred embodiment of the vaccine composition of the present invention, the cell wall and/or cell membrane proteins are S. pneumoniae proteins that are associated with an age-related immune response.
[0037]In another preferred embodiment of the vaccine composition of the present invention, the cell wall and/or cell membrane proteins are lectins.
[0038]In another preferred embodiment of the vaccine composition of the present invention, the cell wall and/or cell membrane proteins are non-lectins.
[0039]In a further preferred embodiment of the vaccine composition of the present invention, the cell wall and/or cell membrane proteins are a mixture of lectins and non-lectins.
[0040]The present invention also encompasses the use of one or more S. pneumoniae cell wall and/or cell membrane proteins and/or immunogenically-active fragments, derivatives or modifications thereof in the preparation of a vaccine for use in the prevention of diseases and carrier states caused by said S pneumoniae, wherein said proteins are selected from the group consisting of: phosphoenolpyruvate protein phosphotransferase, phosphomannomutase, trigger factor, elongation factor G, tetracycline resistance protein (tetO), DNA directed RNA polymerase alpha-chain, NADH oxidase, glutamyl-tRNA amidotransferase subunit A, N utilization substance protein A homolog, XAA-HIS dipeptidase, cell division protein ftsz, zinc metalloproteinase in SCAA 5' region (ORF 6), L-lactate dehydrogenase, glyceraldehyde 3-phosphate dehydrogenase (GAPDH), fructose-biphosphate aldolase, UDP-glucose 4-epimerase, GTP binding protein typA/BipA, GMP synthase, glutamyl-tRNA synthetase, NADP-specific glutamate dehydrogenase, Elongation factor TS, phosphoglycerate kinase (cell wall protein), pyridine nucleotide-disulfide oxido-reductase, 40S ribosomal protein S1, 6-phosphogluconate dehydrogenase, aminopeptidase C, carbomyl-phosphate synthase (large subunit), PTS system mannose-specific IIAB components, ribosomal protein s2, dihydroorotate dehydrogenase, aspartate carbamoyltransferase, elongation factor Tu, Pneumococcal surface immunogenic protein A (PsipA), phosphoglycerate kinase (cell membrane protein), ABC transporter substrate-binding protein endopeptidase O, Pneumococcal surface immunogenic protein B (PsipB) and Pneumococcal surface immunogenic protein C (PsipC).
[0041]In one particularly preferred embodiment, the protein used in the preparation of the vaccine is S. pneumoniae glyceraldehyde 3-phosphate dehydrogenase (GAPDH).
[0042]In another particularly preferred embodiment of the present invention, the protein used in the preparation of the vaccine is S. pneumoniae fructose-biphosphate aldolase.
[0043]In a further particularly preferred embodiment of this aspect of the present invention, an immunogenically-active fragment of the aforementioned S. pneumoniae fructose-biphosphate aldolase protein is used in the preparation of the vaccine. Although many such active fragments may be generated from this protein and used in the preparation of the vaccine, in an especially preferred embodiment, the active fragment used corresponds to the peptide encoded by the first 294 nucleotides of the fructose biphosphate aldolase gene (SP0605 Streptococcus pneumoniae TIGR4), herein referred to as SEQ ID no.1.
[0044]Preferably, the cell wall and/or cell membrane proteins used in the preparation of said vaccine are S. pneumoniae proteins that are associated with an age-related immune response.
[0045]In another preferred embodiment, the cell wall and/or cell membrane proteins used in the preparation of the abovementioned vaccine are lectins.
[0046]In yet another preferred embodiment the cell wall and/or cell membrane proteins used in the preparation of the abovementioned vaccine are non-lectins.
[0047]In a further preferred embodiment, the cell wall and/or cell membrane proteins used in the preparation of the abovementioned vaccine are a mixture of lectins and non-lectins.
[0048]The present invention is also directed to one or more S. pneumoniae cell wall and/or cell membrane proteins and/or immunogenically-active fragments, derivatives or modifications thereof for use as a vaccine for the prevention of diseases and carrier states caused by said S pneumoniae, wherein said proteins are selected from the group consisting of: phosphoenolpyruvate protein phosphotransferase, phosphomannomutase, trigger factor, elongation factor G, tetracycline resistance protein (tetO), DNA directed RNA polymerase alpha-chain, NADH oxidase, glutamyl-tRNA amidotransferase subunit A, N utilization substance protein A homolog, XAA-HIS dipeptidase, cell division protein ftsz, zinc metalloproteinase in SCAA 5' region (ORF 6), L-lactate dehydrogenase, glyceraldehyde 3-phosphate dehydrogenase (GAPDH), fructose-biphosphate aldolase, UDP-glucose 4-epimerase, GTP binding protein typA/BipA, GMP synthase, glutamyl-tRNA synthetase, NADP-specific glutamate dehydrogenase, Elongation factor TS, phosphoglycerate kinase (cell wall protein), pyridine nucleotide-disulfide oxido-reductase, 40S ribosomal protein S1, 6-phosphogluconate dehydrogenase, aminopeptidase C, carbomyl-phosphate synthase (large subunit), PTS system mannose-specific IIAB components, ribosomal protein S2, dihydroorotate dehydrogenase, aspartate carbamoyltransferase, elongation factor Tu, Pneumococcal surface immunogenic protein A (PsipA), phosphoglycerate kinase (cell membrane protein), ABC transporter substrate-binding protein, endopeptidase O, Pneumococcal surface immunogenic protein B (PsipB) and Pneumococcal surface immunogenic protein C (PsipC).
[0049]In one particularly preferred embodiment, the protein for use as said vaccine is S. pneumoniae glyceraldehyde 3-phosphate dehydrogenase (GAPDH).
[0050]In another particularly preferred embodiment of the present invention, the protein for use as said vaccine is S. pneumoniae fructose-biphosphate aldolase.
[0051]In a further particularly preferred embodiment of this aspect of the present invention, an immunogenically-active fragment of the aforementioned S. pneumoniae fructose-biphosphate aldolase protein is provided for use as said vaccine. Although many such active fragments may be generated from this protein and used in the preparation of the vaccine, in an especially preferred embodiment, the active fragment used corresponds to the peptide encoded by the first 294 nucleotides of the fructose biphosphate aldolase gene (SP0605 Streptococcus pneumoniae TIGR4), herein referred to as SEQ ID no.1.
[0052]In one preferred embodiment, the cell wall and/or cell membrane proteins for use as described above are S. pneumoniae proteins associated with an age-related immune response.
[0053]In another preferred embodiment, the aforementioned cell wall and/or cell membrane proteins are lectins.
[0054]In a further preferred embodiment, the aforementioned cell wall and/or cell membrane proteins are non-lectins.
[0055]In a still further preferred embodiment, the aforementioned cell wall and/or cell membrane proteins are a mixture of lectins and non-lectins.
[0056]It is to be noted that when the S. pneumoniae proteins and/or fragments, derivatives or modifications thereof used in the aforementioned methods, compositions and vaccines are lectins, said methods, compositions and vaccines are particularly efficacious in the prevention of localized S. pneumoniae infections. In one preferred embodiment, the localized infections are infections of mucosal tissue, particularly of nasal and other respiratory mucosa.
[0057]Conversely, when the S. pneumoniae proteins and/or their fragments, derivatives or modifications used in the aforementioned methods, compositions and vaccines are non-lectins, said methods, compositions and vaccines are particularly efficacious in the prevention of intra-peritoneal and systemic S. pneumoniae infections.
[0058]All of the above and other characteristics and advantages of the present invention will be further understood from the following illustrative and non-limitative examples of preferred embodiments thereof.
BRIEF DESCRIPTION OF THE DRAWINGS
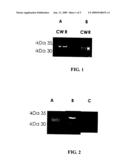
[0059]FIG. 1 is a photograph of a Western blot in which the sera of mice immunized with (A) recombinant GAPDH and (B) recombinant fructose-biphosphate aldolase are seen to recognize the corresponding native proteins (CW) (in an electrophoretically-separated total cell wall protein preparation), and the corresponding recombinant protein (R).
[0060]FIG. 2 is a photograph of a Western blot in which the sera of mice immunized with pVAC constructs containing the cDNA of S. pneumoniae fructose-biphosphate aldolase (A) and GAPDH (B) are seen to recognize the corresponding native proteins from electrophoretically-separated total cell wall protein preparation. Sera obtained following immunization with the pVAC parental plasmid did not recognize either of the two proteins (C).
[0061]FIG. 3 is a graph describing the ability of recombinant fructose-biphosphate aldolase (FIG. 3A) and GAPDH (FIG. 3B) to elicit a protective immune response to intraperitoneal and intranasal challenge with a lethal dose of S. pneumoniae in the mouse model system.
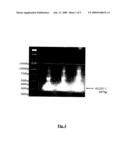
[0062]FIG. 4 is a photograph of a gel depicting the 297 base pair ALDO 1-containing fragment of S. pneumoniae fructose biphosphate aldolase.
[0063]FIG. 5 illustrates the use of the pHAT expression vector in cloning the ALDO 1 fragment of S. pneumoniae fructose biphosphate aldolase. FIG. 5A is a schematic illustration of the construction of the pHAT expression vector, while FIG. 5B depicts an agarose gel separation of ALDO 1 and the pHAT vector after restriction by Kpn1 and SacI enzymes.
[0064]FIG. 6 is a photograph of an agarose gel showing the 297 bp PCR amplification product (comprising ALDO 1) obtained from colonies transformed with the pHAT/ALDO 1 construct.
DETAILED DESCRIPTION OF PREFERRED EMBODIMENTS
[0065]Vaccination protects individuals (and by extension, populations) from the harmful effects of pathogenic agents, such as bacteria, by inducing a specific immunological response to said pathogenic agents in the vaccinated subject.
[0066]Vaccines are generally, but not exclusively, administered by means of injection, generally by way of the intramuscular, intradermal or subcutaneous routes. Some vaccines may also be administered by the intravenous, intraperitoneal, nasal or oral routes.
[0067]The S. pneumoniae-protein containing preparations of the invention can be administered as either single or multiple doses of an effective amount of said protein. The term "effective amount" is used herein to indicate that the vaccine is administered in an amount sufficient to induce or boost a specific immune response, such that measurable amounts (or an increase in the measurable amounts) of one or more antibodies directed against the S. pneumoniae proteins used may be detected in the serum or plasma of the vaccinated subject. The precise weight of protein or proteins that constitutes an "effective amount" will depend upon many factors including the age, weight and physical condition of the subject to be vaccinated. The precise quantity also depends upon the capacity of the subject's immune system to produce antibodies, and the degree of protection desired. Effective dosages can be readily established by one of ordinary skill in the art through routine trials establishing dose response curves. However, for the purposes of the present invention, effective amounts of the compositions of the invention can vary from 0.01-1,000 μg/ml per dose, more preferably 0.1-500 μg/ml per dose, wherein the usual dose size is 1 ml.
[0068]The vaccines of the present invention will generally comprise an effective amount of one or more S. pneumoniae proteins as the active component, suspended in an appropriate vehicle. In the case of intranasal formulations, for example, said formulations may include vehicles that neither cause irritation to the nasal mucosa nor significantly disturb ciliary function. Diluents such as water, aqueous saline may also be added. The nasal formulations may also contain preservatives including, but not limited to, chlorobutanol and benzalkonium chloride. A surfactant may be present to enhance absorption of the subject proteins by the nasal mucosa. An additional mode of antigen delivery may include an encapsulation technique, which involves complex coacervation of gelatin and chondroitin sulfate (Azhari R, Leong K W. 1991. Complex coacervation of chondroitin sulfate and gelatin and its use for encapsulation and slow release of a model protein. Proc. Symp. Control. Rel. 18:617; Brown K E, Leong K, Huang C H, Dalal R, Green G D, Haimes H B, Jimenez P A, Bathon J. 1998. Gelatin/chondroitin 6-sulfate microspheres for delivery of therapeutic proteins to the joint. Arthritis Rheum 41:2185-2195).
[0069]The present invention also encompasses within its scope the preparation and use of DNA vaccines. Vaccination methods and compositions of this type are well known in the art and are comprehensively described in many different articles, monographs and books (see, for example, chapter 11 of "Molecular Biotechnology: principles and applications of recombinant DNA" eds. B. R. Glick & J. J. Pasternak, ASM Press, Washington, D.C., 2nd edition, 1998). In principle, DNA vaccination is achieved by cloning the cDNAs for the desired immunogen into a suitable DNA vaccine vector, such as the pVAC vector (Invivogen). In the case of PVAC, the desired immunogenic proteins are targeted and anchored to the cell surface by cloning the gene of interest in frame between the IL2 signal sequence and the C-terminal transmembrane anchoring domain of human placental alkaline phosphatase. Such DNA vaccine vectors are specifically designed to stimulate humoral immune responses by intramuscular injection. The antigenic peptide produced on the surface of muscle cells is taken up by antigen presenting cells (APCs) and processed through the major histocompatibility complex (MHC) class II pathway.
[0070]Oral liquid preparations may be in the form of, for example, aqueous or oily suspension, solutions, emulsions, syrups or elixirs, or may be presented dry in tablet form or a product for reconstitution with water or other suitable vehicle before use. Such liquid preparations may contain conventional additives such as suspending agents, emulsifying agents, non-aqueous vehicles (which may include edible oils), or preservative.
[0071]However, in general, the vaccines of the present invention would normally be administered parenterally, by the intramuscular, intravenous, intradermal or subcutaneous routes, either by injection or by a rapid infusion method. Compositions for parenteral administration include sterile aqueous or non-aqueous solutions, suspensions, and emulsions. Examples of non-aqueous solvents are propylene glycol, polyethylene glycol, vegetable oils such as olive oil, and injectable organic esters such as ethyl oleate. Besides the abovementioned inert diluents and solvents, the vaccine compositions of the invention can also include adjuvants, wetting agents, emulsifying and suspending agents, or sweetening, flavoring, or perfuming agents.
[0072]The aforementioned adjuvants are substances that can be used to augment a specific immune response. Normally, the adjuvant and the composition are mixed prior to presentation to the immune system, or presented separately, but into the same site of the subject being vaccinated. Adjuvants that may be usefully employed in the preparation of vaccines include: oil adjuvants (for example, Freund's complete and incomplete adjuvants, that will be used in animal experiments only and is forbidden from use in humans), mineral salts, alum, silica, kaolin, and carbon, polynucleotides and certain natural substances of microbial origin. An additional mode of antigen delivery may include an encapsulation technique, which involves complex coacervation of gelatin and chondroitin sulfate (Azhari R, Leong K W. 1991. Complex coacervation of chondroitin sulfate and gelatin and its use for encapsulation and slow release of a model protein. Proc. Symp. Control. Rel. 18:617; Brown K E, Leong K, Huang C H, Dalal R, Green G D, Haimes H B, Jimenez P A, Bathon J. 1998. Gelatin/chondroitin 6-sulfate microspheres for delivery of therapeutic proteins to the joint. Arthritis Rheum 41:2185-2195).
[0073]Further examples of materials and methods useful in the preparation of vaccine compositions are well known to those skilled in the art. In addition, further details may be gleaned from Remington's Pharmaceutical Sciences, Mack Publishing Co, Easton, Pa., USA (1980).
[0074]The S. pneumoniae cell-wall and/or cell-membrane proteins for use in working the present invention may be obtained by directly purifying said proteins from cultures of S. pneumoniae by any of the standard techniques used to prepare and purify cell-surface proteins. Suitable methods are described in many biochemistry text-books, review articles and laboratory guides, including inter alia "Protein Structure: a practical approach" ed. T. E. Creighton, IRL Press, Oxford, UK (1989).
[0075]However, it is to be noted that such an approach suffers many practical limitations that present obstacles for producing commercially-viable quantities of the desired proteins. The S. pneumoniae proteins of the present invention may therefore be more conveniently prepared by means of recombinant biotechnological means, whereby the gene for the S. pneumoniae protein of interest is isolated and inserted into an appropriate expression vector system (such as a plasmid or phage), which is then introduced into a host cell that will permit large-scale production of said protein by means of, for example, overexpression.
[0076]As a first stage, the location of the genes of interest with the S. pneumoniae genome may be determined by reference to a complete-genome database such as the TIGR database that is maintained by the Institute for Genomic Research (web site: http colon double slash www dot tigr dot org slash). The selected sequence may, where appropriate, be isolated directly by the use of appropriate restriction endonucleases, or more effectively by means of PCR amplification. Suitable techniques are described in, for example, U.S. Pat. Nos. 4,683,195, 4,683,202, 4,800,159, 4,965,188, as well as in Innis et al. eds., PCR Protocols: A guide to method and applications.
[0077]Following amplification and/or restriction endonuclease digestion, the desired gene or gene fragment is ligated either initially into a cloning vector, or directly into an expression vector that is appropriate for the chosen host cell type. In the case of the S. pneumoniae proteins, Escherichia coli is the most useful expression host. However, many other cell types may be also be usefully employed including other bacteria, yeast cells, insect cells and mammalian cell systems.
[0078]High-level expression of the desired protein within the host cell may be achieved in several different ways (depending on the chosen expression vector) including expression as a fusion protein (e.g. with factor Xa or thrombin), expression as a His-tagged protein, dual vector systems, expression systems leading to incorporation of the recombinant protein inside inclusion bodies etc. The recombinant protein will then need to be isolated from the cell membrane, interior, inclusion body or (in the case of secreted proteins) the culture medium, by one of the many methods known in the art.
[0079]All of the above recombinant DNA and protein purification techniques are well known to all skilled artisans in the field, the details of said techniques being described in many standard works including "Molecular cloning: a laboratory manual" by Sambrook, J., Fritsch, E. F. & Maniatis, T., Cold Spring Harbor, N.Y., 2nd ed., 1989, which is incorporated herein by reference in its entirety.
[0080]As disclosed and explained hereinabove, each of the abovementioned embodiments of the invention may be based on the use of one or more intact, full length, cell wall and/or cell membrane proteins or, in the alternative, or in addition thereto, fragments, derivatives and modifications of said full length proteins. Fragments may be obtained by means of recombinant expression of selected regions of the cell wall protein gene(s). Alternatively, such fragments may be obtained by means of controlled, site-specific cleavage of the cell-wall proteins using one or more enzymes such as factor X, trypsin, chymotrypsin etc., as are well known in the art. Derivatives of the full length proteins or fragments thereof may be obtained by introducing non-native sequences within the DNA sequences encoding said proteins, followed by expression of said derivatized sequences. Derivatives may also be produced by conjugating non-native groups to the amino residue side chains of the cell wall proteins or protein fragments, using standard protein modification techniques. Modified cell wall proteins and protein fragments for use in the present invention may also be obtained by the use of site-directed mutagenesis techniques. Such techniques are well known in the art and are described, for example, in "Molecular cloning: a laboratory manual" by Sambrook, J., Fritsch, E. F. & Maniatis, T., Cold Spring Harbor, N.Y., 2nd ed., 1989. Of particular interest is the use of one or more of the preceding techniques to create fragments or derivatives possessing the desired epitopic sites, but lacking other domains which are responsible for adverse effects such as suppression of cellular immune responses. It is to be emphasized that all of the immediately preceding discussion of fragments, derivatives and mutants of the cell wall proteins disclosed herein are to be considered as an integral part of the present invention.
[0081]The following examples are provided for illustrative purposes and in order to more particularly explain and describe the present invention. The present invention, however, is not limited to the particular embodiments disclosed in the examples.
EXAMPLE 1
Prevention of S. Pneumoniae Infection In Mice By Inoculation With S. Pneumoniae Cell Wall Protein Fractions
Methods
[0082]Bacterial Cells
[0083]The bacterial strain used in this study was an S. pneumoniae serotype 3 strain. The bacteria were plated onto tryptic soy agar supplemented with 5% sheep erythrocytes and incubated for 17-18 hours at 37° C. under anaerobic conditions. The bacterial cells were then transferred to Todd-Hewitt broth supplemented with 0-5% yeast extract and grown to mid-late log phase. Bacteria were harvested and the pellets were stored at -70° C.
[0084]Purification of Cell Wall Proteins
[0085]Bacterial pellets were resuspended in phosphate buffered saline (PBS). The resulting pellets were then treated with mutanolysin to release cell wall components. Supernatants containing the CW proteins were then harvested. Subsequently, the bacteria were sonicated and centrifuged and the resulting pellet containing the bacteria membranes (m) were lysed with 0.5% TRITON X-100 detergent.
[0086]Fractionation of the Cell Wall Protein Mixture
[0087]Cell wall protein-containing supernatants were allowed to adhere to fetuin (a highly glycosylated pan-lectin binding protein) that was covalently bound to a sepharose column. Non-adherent molecules, obtained from the flow-through fraction were predominantly non-lectin molecules, while the column-adherent lectins were eluted with 50 mM ammonium acetate at pH 3.5.
[0088]Experimental: S. pneumoniae cell wall (CW) proteins were separated into lectin (CW-L) and non-lectin (NL) fractions by fetuin affinity chromatography, as described hereinabove. C57BL/6 and BALB/c mice were vaccinated with S. pneumoniae total CW (CW-T), CW-L and CW-NL protein preparations mixed with Freund's adjuvant, by means of the following procedure: each mouse was primed with 25 micrograms of CW-T, CW-NL and CW-L protein preparations intramuscularly, with complete Freund's adjuvant (CFA) and boosted with incomplete Freund's adjuvant (IFA), 4 and 7 weeks following priming. Western blots of the abovementioned protein preparations were probed with sera obtained a week after the last immunization. Animals were then challenged intranasally (IN) or intraperitoneally (IP) with 108 cfu of S. pneumoniae serotype 3, that caused 100% mortality in control mice immunized with CFA and boosted with IFA only within 96 hours post-inoculation. Vaccination with CW-L elicited partial protection against S. Pneumoniae IN and IP challenge (50% and 45% respectively). Vaccination with CW-T and CW-NL proteins elicited 70% and 65% protection against IP challenge, respectively. Vaccination with CW-T and CW-NL proteins elicited 85% and 50% protection against IN inoculation, respectively.
EXAMPLE 2
Determination of Age-Related Immunoreactivity To S. Pneumoniae Surface Proteins
[0089]The following study was carried out in order to investigate the age-related development of immunoreactivity to S. pneumoniae cell wall and cell membrane proteins.
[0090]Operating as described hereinabove in Example 1, a fraction containing cell wall proteins was obtained from a clinical isolate of S. pneumoniae. In addition, cell membrane proteins were recovered by solubilizing the membrane pellet (described hereinabove in Example 1) in 0.5% TRITON X-100 detergent. The cell wall and cell membrane proteins were separated by means of two-dimensional gel electrophoresis, wherein the proteins were separated using sodium dodecyl sulfate polyacrylamide gel electrophoresis (SDS-PAGE) in one dimension, and polyacrylamide gel isoelectric focusing in the other dimension.
[0091]The ability of serum prepared from blood samples of children aged 1.5, 2.5 and 3.5 years and adults to recognize the separated S. pneumoniae proteins was investigated by Western blot analysis according to the methods described by Rapola S. et al. [J. Infect. Dis. (2000) 182: 1146-52].
[0092]Putative identification of the separated protein spots obtained following the 2 D-electrophoresis was achieved by the use of the Matrix Assisted Laser Desorption/Ionization mass spectrometery (MALDI-MS) technique.
[0093]Results of the above analysis are summarized in the following table:
TABLE-US-00001 Age-dependent immunoreactivity to S. pneumoniae surface proteins (total, pH 4-6.5) =x spot protein age age age nr. per spot Homology 1.5 2.5 3.5 adult 1 2 DNA K/phosphoenolpyruvate protein phosphotransferase * * * * 3 1 Trigger factor * * * * 4 2 60 kDa chaperonin (proteinCPN60) (GroEL protein) ** * ** *** Elongation factor G/tetracycline resistance protein teto (TET(O)) 7 2 Glutamyl-tRNA amidotransferase subunit A/N utilization * ** ? *** substance protein A homolog. 11 2 Oligopeptide-binding protein amiA/aliA/aliB precursor ? ? ? ? Hypothetical zinc metalloproteinase in SCAA 5'region (ORF 6) 12 1 Pneumolysin (thio-activated cytolysin) * * ** 13 1 L-lactate dehydrogenase * ** * ? 14 1 Glyceraldehyde 3-phosphate dehydrogenase (GAPDH) * ** *** *** 15 1 Fructose-biphosphate aldolase ** *** *** *** 16 1 UDP-glucose 4-epimerase ** * ? ? 17 2 Elongation factor G/tetracycline resistance protein teto (TET(O)) * ** ? ? GTP binding protein typA/BipA 18 1 Pyruvate oxidase *** *** *** *** 22 1 Glutamyl-tRNA synthease * ** ? 23 1 NADP-specific glutamate dehydrogenase * * * 24 1 Glyceraldehyde 3-phosphate dehydrogenase (GAPDH) * ** *** *** 25 1 Enolase (2-phosphoglycerate dehydratase) * ** ** ** 27 1 Phosphoglycerate kinase * ** ** ** 29 1 Glucose-6-phosphate isomerase * * ** 30 2 40S ribosomal protein S1/6-phosphogluconate dehydrogenase ? ? 31 1 Aminopeptidase C ? ? new spots 33 * ** *** 55/62 ** *** *** *** 57/65 * * ** ** 58 ** * ? ?
[0094]The data presented in the preceding table indicate that there is an age-dependent development of immunoreactivity to several S. pneumoniae cell wall and cell surface proteins.
EXAMPLE 3
Prevention of S. Pneumoniae Infection In Mice By Inoculation With Recombinantly-Expressed S. pneumoniae cell surface Proteins
[0095]Cloning of Immunogenic S. Pneumoniae Surface Proteins
[0096]S. pneumoniae fructose-biphosphate aldolase (hereinafter referred to as "aldolase") and GAPDH proteins were cloned into the pHAT expression vector (Clontech) and expressed in E. coli BL21 cells (Promega Corp., USA) using standard laboratory procedures. Following lysis of the BL21 cells, recombinant proteins were purified by the use of immobilized metal affinity chromatography (IMAC) on Ni-NTa columns (Qiagen) and eluted with imidazole. In a separate set of experiments, S. pneumoniae aldolase cDNAs were cloned into the pVAC expression vector (Invivogen), a DNA vaccine vector specifically designed to stimulate a humoral immune response by intramuscular injection. Antigenic proteins are targeted and anchored to the cell surface by cloning the gene of interest in frame between the IL2 signal sequence and the C-terminal transmembrane anchoring domain of human placental alkaline phosphatase. The antigenic peptide produced on the surface of muscle cells is taken up by antigen presenting cells (APCs) and processed through the major histocompatibility complex (MHC) class II pathway.
[0097]Immunization
[0098]BALB/c and C57BL/6 mice (7 week old females) were intramuscularly immunized with 25 micrograms of either recombinant aldolase or recombinant GAPDH proteins together with either Freund's complete adjuvant (CFA) or an alum adjuvant. In a separate set of experiments, mice of the aforementioned strains were intramuscularly immunized with 50 micrograms of the pVAC-aldolase or pVAC-GAPDH constructs that were described hereinabove.
[0099]Assessment of Immunogenicity
[0100]The immunogenicity of recombinant S. pneumoniae aldolase and GAPDH proteins (1-2 micrograms of each one) was assessed by Western blot assay using serum of mice that had been immunized with either total cell wall proteins (CW-T) or with one of the recombinant proteins (as described hereinabove). The results obtained (FIG. 1) indicate that the sera of the immunized animals recognized both recombinant GAPDH and aldolase proteins, and the native GAPDH and aldolase proteins present in the CW-T mixture.
[0101]In a separate set of experiments the serum of mice that had been immunized with the pVAC-aldolase or pVAC-GAPDH constructs, as described above, was used to detect native aldolase and GAPDH, respectively in Western blots obtained from SDS-PAGE separations of CW-T proteins. The results obtained (FIG. 2) indicate that inoculation with the pVAC-based constructs is capable of eliciting an immune response. Sera of mice vaccinated with the parental pVAC plasmid (i.e. without insert) did not, however, react with the CW-T proteins.
[0102]Protective Vaccination
[0103]Following immunization with the recombinant proteins as described hereinabove, the mice were challenged intranasally with a lethal dose of 108 CFU of S. pneumoniae serotype 3. Only 10% of the control animals (immunization with either CFA or alum only) survived the bacterial challenge. However, 40% of the animals immunized with the recombinant aldolase protein in CFA and 43% of the animals immunized with the same protein in alum survived the challenge. Following immunization with the pVAC-aldolase construct, 33% of the animals survived as shown in FIG. 3A. With regard to recombinant GAPDH, 36% of the animals immunized with this recombinant protein (in either of the above-mentioned adjuvants) survived. Immunization with the pVAC-GAPDH construct, led to a survival rate of 40% as shown in FIG. 3B.
EXAMPLE 4
S. Pneumoniae Immunogenic Proteins
[0104]Operating essentially as in Example 2, the ability of serum prepared from blood samples of children aged 1.5, 2.5 and 3.5 years and adults to recognize the separated S. pneumoniae proteins was investigated by Western blot analysis according to the methods described by Rapola S. et al. [J. Infect. Dis. (2000) 182: 1146-52].
[0105]Identification of the separated protein spots obtained following the 2 D-electrophoresis was achieved by the use of the Matrix Assisted Laser Desorption/Ionization mass spectrometry (MALDI-MS) technique, and comparison of the partial amino acid sequences obtained thereby with the sequences contained in the TIGR4 and/or R6 databases (maintained by The Institute for Genomic Research).
[0106]The cell surface proteins found to be immunogenic (classified according to their cellular location--cell membrane or cell wall) are summarized in the following table:
TABLE-US-00002 List of immunogenic proteins accession code from spot TIGR 4 or R6 cell wall proteins: homology to 1 Phosphoenolpyruvate protein phosphotransferase NP345645 2 Probable phosphomannomutase NP346006 3 Trigger factor NP344923 4 Elongation factor G/tetracycline resistance protein teto (TET(O)) NP344811/P72533 5 DNA directed RNA polymerase alpha-chain G95027 6 NADH oxidase NP345923 7 Glutamyl-tRNA Amidotransferase subunit A NP344960 N utilization substance protein A homolog. P32727 8 XAA-HIS dipeptidase P45494 Cell division protein ftsz. NP346105 11 Hypothetical zinc metalloproteinase in SCAA 5'region (ORF 6) P42358 13 L-lactate dehydrogenase NP345686 14 Glyceraldehyde 3-phosphate dehydrogenase (GAPDH) NP346439 15 Fructose-biphosphate aldolase NP345117 16 UDP-glucose 4-epimerase NP346261 GTP binding protein typA/BipA NP358192 21 GMP synthase NP345899 22 Glutamyl-tRNA synthetase NP346492 23 NADP-specific glutamate dehydrogenase NP345679 26 Elongation factor TS NP346622 27 Phosphoglycerate kinase AAK74657 28 Probable pyridine nucleotide-disulfide oxido-reductase P77212 30 40S ribosomal protein S1 NP345350 6-phosphogluconate dehydrogenase NP357929 31 Aminopeptidase C NP344819 33 Carbamoyl-phosphate synthase, large subunit NP345739 57 PTS system, mannose-specific IIAB components NP344822 58 Ribosomal protein S2 NP346623 62 Dihydroorotate dehydrogenase NP358460 65 Aspartate carbamoyltransferase NP345741 membrane proteins: homology to 14 Elongation factor Tu NP345941 19 Pneumococcal surface immunogenic protein A (PsipA) NP344634 22 Phosphoglycerate kinase NP358035 40 ABC transporter, substrate-binding protein NP344690 lectins: homology to 10 Endopeptidase O NP346087 14 Pneumococcal surface immunogenic protein B (PsipB) NP358083 Pneumococcal surface immunogenic protein C (PsipC) NP345081
EXAMPLE 5
Preparation of An S. Pneumoniae Fructose Biphosphate Aldolase Fragment
[0107]A peptide referred to as ALDO 1, corresponding to the first 294 nucleotides of the coding sequence of the fructose biphosphate aldolase gene (SP0605 Streptococcus pneumoniae TIGR4) (see SEQ ID NO:1 of the sequence listing that forms an integral part of the present disclosure), was amplified from S. pneumoniae strain R6 genomic DNA by means of PCR with the following primers:
TABLE-US-00003 Forward (5'- GGT ACC ATG GCA ATC GTT TCA GCA-3', SEQ ID NO: 2) Reverse (5'- GAG CTC ACC AAC TTC GAT ACA CTC AAG-3', SEQ ID NO: 3).
[0108]The amplified product obtained thereby is shown in FIG. 4.
[0109]The Forward and Reverse primers contain Kpn1 and SacI recognition sequences, respectively. The primers flank the entire open reading frames. The amplified and Kpn1-SacI (Takara Bio Inc, Shiga, Japan) digested DNA-fragments were cloned into the pHAT expression vector (BD Biosciences Clontech, Palo Alto, Calif., USA), as illustrated in FIGS. 5A and 5B and transformed in DH5a ULTRAMAX ultracompetent E. coli cells.
[0110]Ampicillin-resistant transformants were cultured and plasmid DNA was analyzed by PCR. The pHAT-ALDO 1 vector was purified from DH5a ULTRAMAX cells using the Qiagen High Speed Plasmid Maxi Kit (Qiagen GMBH, Hilden, Germany) and transformed in E. coli host expression strain BL21(DE3)pLysS. PCR amplification of the ALDO 1 fragment from transformed positive colonies yielded the 297 bp fragment indicated in the gel shown in FIG. 6.
[0111]While specific embodiments of the invention have been described for the purpose of illustration, it will be understood that the invention may be carried out in practice by skilled persons with many modifications, variations and adaptations, without departing from its spirit or exceeding the bounds of the present invention.
Sequence CWU
1
351294DNAStreptococcus pneumoniae 1atggcaatcg tttcagcaga aaaatttgtc
caagcagccc gtgacaacgg ttatgcagtt 60ggtggattta acacaaacaa ccttgagtgg
actcaagcta tcttgcgcgc agcagaagct 120aaaaaagctc cagttttgat ccaaacttca
atgggtgctg ctaaatacat gggtggttac 180aaagttgctc gcaacttgat cgctaacctt
gttgaatcaa tgggtatcac tgtaccagta 240gctatccacc ttgaccacgg tcactacgaa
gatgcacttg agtgtatcga agtt 294224DNAArtificial SequencePRIMER
2ggtaccatgg caatcgtttc agca
24327DNAArtificial SequencePRIMER 3gagctcacca acttcgatac actcaag
274577PRTStreptococcus pneumoniae 4Met Thr
Glu Met Leu Lys Gly Ile Ala Ala Ser Asp Gly Val Ala Val1 5
10 15Ala Lys Ala Tyr Leu Leu Val Gln
Pro Asp Leu Ser Phe Glu Thr Ile20 25
30Thr Val Glu Asp Thr Asn Ala Glu Glu Ala Arg Leu Asp Ala Ala Leu35
40 45Gln Ala Ser Gln Asp Glu Leu Ser Val Ile
Arg Glu Lys Ala Val Gly50 55 60Thr Leu
Gly Glu Glu Ala Ala Gln Val Phe Asp Ala His Leu Met Val65
70 75 80Leu Ala Asp Pro Glu Met Ile
Ser Gln Ile Lys Glu Thr Ile Arg Ala85 90
95Lys Lys Val Asn Ala Glu Ala Gly Leu Lys Glu Val Thr Asp Met Phe100
105 110Ile Thr Ile Phe Glu Gly Met Glu Asp
Asn Pro Tyr Met Gln Glu Arg115 120 125Ala
Ala Asp Ile Arg Asp Val Thr Lys Arg Val Leu Ala Asn Leu Leu130
135 140Gly Lys Lys Leu Pro Asn Pro Ala Ser Ile Asn
Glu Glu Val Ile Val145 150 155
160Ile Ala His Asp Leu Thr Pro Ser Asp Thr Ala Gln Leu Asp Lys
Asn165 170 175Phe Val Lys Ala Phe Val Thr
Asn Ile Gly Gly Arg Thr Ser His Ser180 185
190Ala Ile Met Ala Arg Thr Leu Glu Ile Ala Ala Val Leu Gly Thr Asn195
200 205Asn Ile Thr Glu Ile Val Lys Asp Gly
Asp Ile Leu Ala Val Asn Gly210 215 220Ile
Thr Gly Glu Val Ile Ile Asn Pro Thr Asp Glu Gln Ala Ala Glu225
230 235 240Phe Lys Ala Ala Gly Glu
Ala Tyr Ala Lys Gln Lys Ala Glu Trp Ala245 250
255Leu Leu Lys Asp Ala Gln Thr Val Thr Ala Asp Gly Lys His Phe
Glu260 265 270Leu Ala Ala Asn Ile Gly Thr
Pro Lys Asp Val Glu Gly Val Asn Asn275 280
285Asn Gly Ala Glu Ala Val Gly Leu Tyr Arg Thr Glu Phe Leu Tyr Met290
295 300Asp Ser Gln Asp Phe Pro Thr Glu Asp
Glu Gln Tyr Glu Ala Tyr Lys305 310 315
320Ala Val Leu Glu Gly Met Asn Gly Lys Pro Val Val Val Arg
Thr Met325 330 335Asp Ile Gly Gly Asp Lys
Glu Leu Pro Tyr Phe Asp Met Pro His Glu340 345
350Met Asn Pro Phe Leu Gly Phe Arg Ala Leu Arg Ile Ser Ile Ser
Glu355 360 365Thr Gly Asp Ala Met Phe Arg
Thr Gln Ile Arg Ala Leu Leu Arg Ala370 375
380Ser Val His Gly Gln Leu Arg Ile Met Phe Pro Met Val Ala Leu Leu385
390 395 400Lys Glu Phe Arg
Ala Ala Lys Ala Val Phe Asp Glu Glu Lys Ala Asn405 410
415Leu Leu Ala Glu Gly Val Ala Val Ala Asp Asn Ile Gln Val
Gly Ile420 425 430Met Ile Glu Ile Pro Ala
Ala Ala Met Leu Ala Asp Gln Phe Ala Lys435 440
445Glu Val Asp Phe Phe Ser Ile Gly Thr Asn Asp Leu Ile Gln Tyr
Thr450 455 460Met Ala Ala Asp Arg Met Asn
Glu Gln Val Ser Tyr Leu Tyr Gln Pro465 470
475 480Tyr Asn Pro Ser Ile Leu Arg Leu Ile Asn Asn Val
Ile Lys Ala Ala485 490 495His Ala Glu Gly
Lys Trp Ala Gly Met Cys Gly Glu Met Ala Gly Asp500 505
510Gln Gln Ala Val Pro Leu Leu Val Gly Met Gly Leu Asp Glu
Phe Ser515 520 525Met Ser Ala Thr Ser Val
Leu Arg Thr Arg Ser Leu Met Lys Lys Leu530 535
540Asp Thr Ala Lys Met Glu Glu Tyr Ala Asn Arg Ala Leu Thr Glu
Cys545 550 555 560Ser Thr
Met Glu Glu Val Leu Glu Leu Gln Lys Glu Tyr Val Asn Phe565
570 575Asp5450PRTStreptococcus pneumoniae 5Met Gly Lys
Tyr Phe Gly Thr Asp Gly Val Arg Gly Glu Ala Asn Leu1 5
10 15Glu Leu Thr Pro Glu Leu Ala Phe Lys
Leu Gly Arg Phe Gly Gly Tyr20 25 30Val
Leu Ser Gln His Glu Thr Glu Ala Pro Lys Val Phe Val Gly Arg35
40 45Asp Thr Arg Ile Ser Gly Glu Met Leu Glu Ser
Ala Leu Val Ala Gly50 55 60Leu Leu Ser
Val Gly Ile His Val Tyr Lys Leu Gly Val Leu Ala Thr65 70
75 80Pro Ala Val Ala Tyr Leu Val Glu
Thr Glu Gly Ala Ser Ala Gly Val85 90
95Met Ile Ser Ala Ser His Asn Pro Ala Leu Asp Asn Gly Ile Lys Phe100
105 110Phe Gly Gly Asp Gly Phe Lys Leu Asp Asp
Glu Lys Glu Ala Glu Ile115 120 125Glu Ala
Leu Leu Asp Ala Glu Glu Asp Thr Leu Pro Arg Pro Ser Ala130
135 140Glu Gly Leu Gly Ile Leu Val Asp Tyr Pro Glu Gly
Leu Arg Lys Tyr145 150 155
160Glu Gly Tyr Leu Val Ser Thr Gly Thr Pro Leu Asp Gly Met Lys Val165
170 175Ala Leu Asp Thr Ala Asn Gly Ala Ala
Ser Thr Ser Ala Arg Gln Ile180 185 190Phe
Ala Asp Leu Gly Ala Gln Leu Thr Val Ile Gly Glu Thr Pro Asp195
200 205Gly Leu Asn Ile Asn Leu Asn Val Gly Ser Thr
His Pro Glu Ala Leu210 215 220Gln Glu Val
Val Lys Glu Ser Gly Ser Ala Ile Gly Leu Ala Phe Asp225
230 235 240Gly Asp Ser Asp Arg Leu Ile
Ala Val Asp Glu Asn Gly Asp Ile Val245 250
255Asp Gly Asp Lys Ile Met Tyr Ile Ile Gly Lys Tyr Leu Ser Glu Lys260
265 270Gly Gln Leu Ala Gln Asn Thr Ile Val
Thr Thr Val Met Ser Asn Leu275 280 285Gly
Phe His Lys Ala Leu Asn Arg Glu Gly Ile Asn Lys Ala Val Thr290
295 300Ala Val Gly Asp Arg Tyr Val Val Glu Glu Met
Arg Lys Ser Gly Tyr305 310 315
320Asn Leu Gly Gly Glu Gln Ser Gly His Val Ile Leu Met Asp Tyr
Asn325 330 335Thr Thr Gly Asp Gly Gln Leu
Ser Ala Val Gln Leu Thr Lys Ile Met340 345
350Lys Glu Thr Gly Lys Ser Leu Ser Glu Leu Ala Ala Glu Val Thr Ile355
360 365Tyr Pro Gln Lys Leu Val Asn Ile Arg
Val Glu Asn Val Met Lys Glu370 375 380Lys
Ala Met Glu Val Pro Ala Ile Lys Ala Ile Ile Glu Lys Met Glu385
390 395 400Glu Glu Met Ala Gly Asn
Gly Arg Ile Leu Val Arg Pro Ser Gly Thr405 410
415Glu Pro Leu Leu Arg Val Met Ala Glu Ala Pro Thr Thr Glu Glu
Val420 425 430Asn Tyr Tyr Val Asp Thr Ile
Thr Asp Val Val Arg Ala Glu Ile Gly435 440
445Ile Asp4506420PRTStreptococcus pneumoniae 6Met Ser Val Ser Phe Glu
Asn Lys Glu Thr Asn Arg Gly Val Leu Thr1 5
10 15Phe Thr Ile Ser Gln Asp Gln Ile Lys Pro Glu Leu
Asp Arg Val Phe20 25 30Lys Ser Val Lys
Lys Ser Leu Asn Val Pro Gly Phe Arg Lys Gly His35 40
45Leu Pro Arg Pro Ile Phe Asp Gln Lys Phe Gly Glu Glu Ala
Leu Tyr50 55 60Gln Asp Ala Met Asn Ala
Leu Leu Pro Asn Ala Tyr Glu Ala Ala Val65 70
75 80Lys Glu Ala Gly Leu Glu Val Val Ala Gln Pro
Lys Ile Asp Val Thr85 90 95Ser Met Glu
Lys Gly Gln Asp Trp Val Ile Thr Ala Glu Val Val Thr100
105 110Lys Pro Glu Val Lys Leu Gly Asp Tyr Lys Asn Leu
Glu Val Ser Val115 120 125Asp Val Glu Lys
Glu Val Thr Asp Ala Asp Val Glu Glu Arg Ile Glu130 135
140Arg Glu Arg Asn Asn Leu Ala Glu Leu Val Ile Lys Glu Ala
Ala Ala145 150 155 160Glu
Asn Gly Asp Thr Val Val Ile Asp Phe Val Gly Ser Ile Asp Gly165
170 175Val Glu Phe Asp Gly Gly Lys Gly Glu Asn Phe
Ser Leu Gly Leu Gly180 185 190Ser Gly Gln
Phe Ile Pro Gly Phe Glu Asp Gln Leu Val Gly His Ser195
200 205Ala Gly Glu Thr Val Asp Val Ile Val Thr Phe Pro
Glu Asp Tyr Gln210 215 220Ala Glu Asp Leu
Ala Gly Lys Glu Ala Lys Phe Val Thr Thr Ile His225 230
235 240Glu Val Lys Ala Lys Glu Val Pro Ala
Leu Asp Asp Glu Leu Ala Lys245 250 255Asp
Ile Asp Glu Glu Val Glu Thr Leu Ala Asp Leu Lys Glu Lys Tyr260
265 270Ser Lys Glu Leu Ala Ala Ala Lys Glu Glu Ala
Tyr Lys Asp Ala Val275 280 285Glu Gly Ala
Ala Ile Asp Thr Ala Val Glu Asn Ala Glu Ile Val Glu290
295 300Leu Pro Glu Glu Met Ile His Glu Glu Val His Arg
Ser Val Asn Glu305 310 315
320Phe Leu Gly Asn Leu Gln Arg Gln Gly Ile Asn Pro Asp Met Tyr Phe325
330 335Gln Ile Thr Gly Thr Thr Gln Glu Asp
Leu His Asn Gln Tyr Gln Ala340 345 350Glu
Ala Glu Ser Arg Thr Lys Thr Asn Leu Val Ile Glu Ala Val Ala355
360 365Lys Ala Glu Gly Phe Asp Ala Ser Glu Glu Glu
Ile Gln Lys Glu Val370 375 380Glu Gln Leu
Ala Ala Asp Tyr Asn Met Glu Val Ala Gln Val Gln Asn385
390 395 400Leu Leu Ser Ala Asp Met Leu
Lys His Asp Ile Thr Ile Lys Lys Ala405 410
415Val Glu Leu Ile4207693PRTStreptococcus pneumoniae 7Met Ala Arg Glu
Phe Ser Leu Glu Lys Thr Arg Asn Ile Gly Ile Met1 5
10 15Ala His Val Asp Ala Gly Lys Thr Thr Thr
Thr Glu Arg Ile Leu Tyr20 25 30Tyr Thr
Gly Lys Ile His Lys Ile Gly Glu Thr His Glu Gly Ala Ser35
40 45Gln Met Asp Trp Met Glu Gln Glu Gln Glu Arg Gly
Ile Thr Ile Thr50 55 60Ser Ala Ala Thr
Thr Ala Gln Trp Asn Asn His Arg Val Asn Ile Ile65 70
75 80Asp Thr Pro Gly His Val Asp Phe Thr
Ile Glu Val Gln Arg Ser Leu85 90 95Arg
Val Leu Asp Gly Ala Val Thr Val Leu Asp Ser Gln Ser Gly Val100
105 110Glu Pro Gln Thr Glu Thr Val Trp Arg Gln Ala
Thr Glu Tyr Gly Val115 120 125Pro Arg Ile
Val Phe Ala Asn Lys Met Asp Lys Ile Gly Ala Asp Phe130
135 140Leu Tyr Ser Val Ser Thr Leu His Asp Arg Leu Gln
Ala Asn Ala His145 150 155
160Pro Ile Gln Leu Pro Ile Gly Ser Glu Asp Asp Phe Arg Gly Ile Ile165
170 175Asp Leu Ile Lys Met Lys Ala Glu Ile
Tyr Thr Asn Asp Leu Gly Thr180 185 190Asp
Ile Leu Glu Glu Asp Ile Pro Ala Glu Tyr Leu Asp Gln Ala Gln195
200 205Glu Tyr Arg Glu Lys Leu Ile Glu Ala Val Ala
Glu Thr Asp Glu Glu210 215 220Leu Met Met
Lys Tyr Leu Glu Gly Glu Glu Ile Thr Asn Glu Glu Leu225
230 235 240Lys Ala Gly Ile Arg Lys Ala
Thr Ile Asn Val Glu Phe Phe Pro Val245 250
255Leu Cys Gly Ser Ala Phe Lys Asn Lys Gly Val Gln Leu Met Leu Asp260
265 270Ala Val Ile Asp Tyr Leu Pro Ser Pro
Leu Asp Ile Pro Ala Ile Lys275 280 285Gly
Ile Asn Pro Asp Thr Asp Ala Glu Glu Ile Arg Pro Ala Ser Asp290
295 300Glu Glu Pro Phe Ala Ala Leu Ala Phe Lys Ile
Met Thr Asp Pro Phe305 310 315
320Val Gly Arg Leu Thr Phe Phe Arg Val Tyr Ser Gly Val Leu Gln
Ser325 330 335Gly Ser Tyr Val Leu Asn Thr
Ser Lys Gly Lys Arg Glu Arg Ile Gly340 345
350Arg Ile Leu Gln Met His Ala Asn Ser Arg Gln Glu Ile Asp Thr Val355
360 365Tyr Ser Gly Asp Ile Ala Ala Ala Val
Gly Leu Lys Asp Thr Thr Thr370 375 380Gly
Asp Ser Leu Thr Asp Glu Lys Ala Lys Ile Ile Leu Glu Ser Ile385
390 395 400Asn Val Pro Glu Pro Val
Ile Gln Leu Met Val Glu Pro Lys Ser Lys405 410
415Ala Asp Gln Asp Lys Met Gly Ile Ala Leu Gln Lys Leu Ala Glu
Glu420 425 430Asp Pro Thr Phe Arg Val Glu
Thr Asn Val Glu Thr Gly Glu Thr Val435 440
445Ile Ser Gly Met Gly Glu Leu His Leu Asp Val Leu Val Asp Arg Met450
455 460Arg Arg Glu Phe Lys Val Glu Ala Asn
Val Gly Ala Pro Gln Val Ser465 470 475
480Tyr Arg Glu Thr Phe Arg Ala Ser Thr Gln Ala Arg Gly Phe
Phe Lys485 490 495Arg Gln Ser Gly Gly Lys
Gly Gln Phe Gly Asp Val Trp Ile Glu Phe500 505
510Thr Pro Asn Glu Glu Gly Lys Gly Phe Glu Phe Glu Asn Ala Ile
Val515 520 525Gly Gly Val Val Pro Arg Glu
Phe Ile Pro Ala Val Glu Lys Gly Leu530 535
540Val Glu Ser Met Ala Asn Gly Val Leu Ala Gly Tyr Pro Met Val Asp545
550 555 560Val Lys Ala Lys
Leu Tyr Asp Gly Ser Tyr His Asp Val Asp Ser Ser565 570
575Glu Thr Ala Phe Lys Ile Ala Ala Ser Leu Ser Leu Lys Glu
Ala Ala580 585 590Lys Ser Ala Gln Pro Ala
Ile Leu Glu Pro Met Met Leu Val Thr Ile595 600
605Thr Val Pro Glu Glu Asn Leu Gly Asp Val Met Gly His Val Thr
Ala610 615 620Arg Arg Gly Arg Val Asp Gly
Met Glu Ala His Gly Asn Ser Gln Ile625 630
635 640Val Arg Ala Tyr Val Pro Leu Ala Glu Met Phe Gly
Tyr Ala Thr Val645 650 655Leu Arg Ser Ala
Ser Gln Gly Arg Gly Thr Phe Met Met Val Phe Asp660 665
670His Tyr Glu Asp Val Pro Lys Ser Val Gln Glu Glu Ile Ile
Lys Lys675 680 685Asn Lys Gly Glu
Asp6908459PRTStreptococcus pneumoniae 8Met Ser Lys Ile Val Val Val Gly
Ala Asn His Ala Gly Thr Ala Cys1 5 10
15Ile Asn Thr Met Leu Asp Asn Phe Gly Asn Glu Asn Glu Ile
Val Val20 25 30Phe Asp Gln Asn Ser Asn
Ile Ser Phe Leu Gly Cys Gly Met Ala Leu35 40
45Trp Ile Gly Glu Gln Ile Asp Gly Ala Glu Gly Leu Phe Tyr Ser Asp50
55 60Lys Glu Lys Leu Glu Ala Lys Gly Ala
Lys Val Tyr Met Asn Ser Pro65 70 75
80Val Leu Ser Ile Asp Tyr Asp Asn Lys Val Val Thr Ala Glu
Val Glu85 90 95Gly Lys Glu His Lys Glu
Ser Tyr Glu Lys Leu Ile Phe Ala Thr Gly100 105
110Ser Thr Pro Ile Leu Pro Pro Ile Glu Gly Val Glu Ile Val Lys
Gly115 120 125Asn Arg Glu Phe Lys Ala Thr
Leu Glu Asn Val Gln Phe Val Lys Leu130 135
140Tyr Gln Asn Ala Glu Glu Val Ile Asn Lys Leu Ser Asp Lys Ser Gln145
150 155 160His Leu Asp Arg
Ile Ala Val Val Gly Gly Gly Tyr Ile Gly Val Glu165 170
175Leu Ala Glu Ala Phe Glu Arg Leu Gly Lys Glu Val Val Leu
Val Asp180 185 190Ile Val Asp Thr Val Leu
Asn Gly Tyr Tyr Asp Lys Asp Phe Thr Gln195 200
205Met Met Ala Lys Asn Leu Glu Asp His Asn Ile Arg Leu Ala Leu
Gly210 215 220Gln Thr Val Lys Ala Ile Glu
Gly Asp Gly Lys Val Glu Arg Leu Ile225 230
235 240Thr Asp Lys Glu Ser Phe Asp Val Asp Met Val Ile
Leu Ala Val Gly245 250 255Phe Arg Pro Asn
Thr Ala Leu Ala Gly Gly Lys Ile Glu Leu Phe Arg260 265
270Asn Gly Ala Phe Leu Val Asp Lys Lys Gln Glu Thr Ser Ile
Pro Asp275 280 285Val Tyr Ala Val Gly Asp
Cys Ala Thr Val Tyr Asp Asn Ala Arg Lys290 295
300Asp Thr Ser Tyr Ile Ala Leu Ala Ser Asn Ala Val Arg Thr Gly
Ile305 310 315 320Val Gly
Ala Tyr Asn Ala Cys Gly His Glu Leu Glu Gly Ile Gly Val325
330 335Gln Gly Ser Asn Gly Ile Ser Ile Tyr Gly Leu His
Met Val Ser Thr340 345 350Gly Leu Thr Leu
Glu Lys Ala Lys Ala Ala Gly Tyr Asn Ala Thr Glu355 360
365Thr Gly Phe Asn Asp Leu Gln Lys Pro Glu Phe Met Lys His
Asp Asn370 375 380His Glu Val Ala Ile Lys
Ile Val Phe Asp Lys Asp Ser Arg Glu Ile385 390
395 400Leu Gly Ala Gln Met Val Ser His Asp Ile Ala
Ile Ser Met Gly Ile405 410 415His Met Phe
Ser Leu Ala Ile Gln Glu His Val Thr Ile Asp Lys Leu420
425 430Ala Leu Thr Asp Leu Phe Phe Leu Pro His Phe Asn
Lys Pro Tyr Asn435 440 445Tyr Ile Thr Met
Ala Ala Leu Thr Ala Glu Lys450 4559100PRTStreptococcus
pneumoniae 9Met Lys Ile Thr Gln Glu Glu Val Thr His Val Ala Asn Leu Ser
Lys1 5 10 15Leu Arg Phe
Ser Glu Glu Glu Thr Ala Ala Phe Ala Thr Thr Leu Ser20 25
30Lys Ile Val Asp Met Val Glu Leu Leu Gly Glu Val Asp
Thr Thr Gly35 40 45Val Ala Pro Thr Thr
Thr Met Ala Asp Arg Lys Thr Val Leu Arg Pro50 55
60Asp Val Ala Glu Glu Gly Ile Asp Arg Asp Arg Leu Phe Lys Asn
Val65 70 75 80Pro Glu
Lys Asp Asn Tyr Tyr Ile Lys Val Pro Ala Ile Leu Asp Asn85
90 95Gly Gly Asp Ala10010419PRTStreptococcus pneumoniae
10Met Thr Phe Ser Phe Asp Thr Ala Ala Ala Gln Gly Ala Val Ile Lys1
5 10 15Val Ile Gly Val Gly Gly
Gly Gly Gly Asn Ala Ile Asn Arg Met Val20 25
30Asp Glu Gly Val Thr Gly Val Glu Phe Ile Ala Ala Asn Thr Asp Val35
40 45Gln Ala Leu Ser Ser Thr Lys Ala Glu
Thr Val Ile Gln Leu Gly Pro50 55 60Lys
Leu Thr Arg Gly Leu Gly Ala Gly Gly Gln Pro Glu Val Gly Arg65
70 75 80Lys Ala Ala Glu Glu Ser
Glu Glu Thr Leu Thr Glu Ala Ile Ser Gly85 90
95Ala Asp Met Val Phe Ile Thr Ala Gly Met Gly Gly Gly Ser Gly Thr100
105 110Gly Ala Ala Pro Val Ile Ala Arg
Ile Ala Lys Asp Leu Gly Ala Leu115 120
125Thr Val Gly Val Val Thr Arg Pro Phe Gly Phe Glu Gly Ser Lys Arg130
135 140Gly Gln Phe Ala Val Glu Gly Ile Asn
Gln Leu Arg Glu His Val Asp145 150 155
160Thr Leu Leu Ile Ile Ser Asn Asn Asn Leu Leu Glu Ile Val
Asp Lys165 170 175Lys Thr Pro Leu Leu Glu
Ala Leu Ser Glu Ala Asp Asn Val Leu Arg180 185
190Gln Gly Val Gln Gly Ile Thr Asp Leu Ile Thr Asn Pro Gly Leu
Ile195 200 205Asn Leu Asp Phe Ala Asp Val
Lys Thr Val Met Ala Asn Lys Gly Asn210 215
220Ala Leu Met Gly Ile Gly Ile Gly Ser Gly Glu Glu Arg Val Val Glu225
230 235 240Ala Ala Arg Lys
Ala Ile Tyr Ser Pro Leu Leu Glu Thr Thr Ile Asp245 250
255Gly Ala Glu Asp Val Ile Val Asn Val Thr Gly Gly Leu Asp
Leu Thr260 265 270Leu Ile Glu Ala Glu Glu
Ala Ser Gln Ile Val Asn Gln Ala Ala Gly275 280
285Gln Gly Val Asn Ile Trp Leu Gly Thr Ser Ile Asp Glu Ser Met
Arg290 295 300Asp Glu Ile Arg Val Thr Val
Val Ala Thr Gly Val Arg Gln Asp Arg305 310
315 320Val Glu Lys Val Val Ala Pro Gln Ala Arg Ser Ala
Thr Asn Tyr Arg325 330 335Glu Thr Val Lys
Pro Ala His Ser His Gly Phe Asp Arg His Phe Asp340 345
350Met Ala Glu Thr Val Glu Leu Pro Lys Gln Asn Pro Arg Arg
Leu Glu355 360 365Pro Thr Gln Ala Ser Ala
Phe Gly Asp Trp Asp Leu Arg Arg Glu Ser370 375
380Ile Val Arg Thr Thr Asp Ser Val Val Ser Pro Val Glu Arg Phe
Glu385 390 395 400Ala Pro
Ile Ser Gln Asp Glu Asp Glu Leu Asp Thr Pro Pro Phe Phe405
410 415Lys Asn Arg11328PRTStreptococcus pneumoniae 11Met
Thr Ser Thr Lys Gln His Lys Lys Val Ile Leu Val Gly Asp Gly1
5 10 15Ala Val Gly Ser Ser Tyr Ala
Phe Ala Leu Val Asn Gln Gly Ile Ala20 25
30Gln Glu Leu Gly Ile Ile Glu Ile Pro Gln Leu His Glu Lys Ala Val35
40 45Gly Asp Ala Leu Asp Leu Ser His Ala Leu
Ala Phe Thr Ser Pro Lys50 55 60Lys Ile
Tyr Ala Ala Gln Tyr Ser Asp Cys Ala Asp Ala Asp Leu Val65
70 75 80Val Ile Thr Ala Gly Ala Pro
Gln Lys Pro Gly Glu Thr Arg Leu Asp85 90
95Leu Val Gly Lys Asn Leu Ala Ile Asn Lys Ser Ile Val Thr Gln Val100
105 110Val Glu Ser Gly Phe Lys Gly Ile Phe
Leu Val Ala Ala Asn Pro Val115 120 125Asp
Val Leu Thr Tyr Ser Thr Trp Lys Phe Ser Gly Phe Pro Lys Glu130
135 140Arg Val Ile Gly Ser Gly Thr Ser Leu Asp Ser
Ala Arg Phe Arg Gln145 150 155
160Ala Leu Ala Glu Lys Leu Asp Val Asp Ala Arg Ser Val His Ala
Tyr165 170 175Ile Met Gly Glu His Gly Asp
Ser Glu Phe Ala Val Trp Ser His Ala180 185
190Asn Ile Ala Gly Val Asn Leu Glu Glu Phe Leu Lys Asp Thr Gln Asn195
200 205Val Gln Glu Ala Glu Leu Ile Glu Leu
Phe Glu Gly Val Arg Asp Ala210 215 220Ala
Tyr Thr Ile Ile Asn Lys Lys Gly Ala Thr Tyr Tyr Gly Ile Ala225
230 235 240Val Ala Leu Ala Arg Ile
Thr Lys Ala Ile Leu Asp Asp Glu Asn Ala245 250
255Val Leu Pro Leu Ser Val Phe Gln Glu Gly Gln Tyr Gly Val Glu
Asn260 265 270Val Phe Ile Gly Gln Pro Ala
Val Val Gly Ala His Gly Ile Val Arg275 280
285Pro Val Asn Ile Pro Leu Asn Asp Ala Glu Thr Gln Lys Met Gln Ala290
295 300Ser Ala Lys Glu Leu Gln Ala Ile Ile
Asp Glu Ala Trp Lys Asn Pro305 310 315
320Glu Phe Gln Glu Ala Ser Lys Asn32512335PRTStreptococcus
pneumoniae 12Met Val Val Lys Val Gly Ile Asn Gly Phe Gly Arg Ile Gly Arg
Leu1 5 10 15Ala Phe Arg
Arg Ile Gln Asn Val Glu Gly Val Glu Val Thr Arg Ile20 25
30Asn Asp Leu Thr Asp Pro Val Met Leu Ala His Leu Leu
Lys Tyr Asp35 40 45Thr Thr Gln Gly Arg
Phe Asp Gly Thr Val Glu Val Lys Glu Gly Gly50 55
60Phe Glu Val Asn Gly Lys Phe Ile Lys Val Ser Ala Glu Arg Asp
Pro65 70 75 80Glu Gln
Ile Asp Trp Ala Thr Asp Gly Val Glu Ile Val Leu Glu Ala85
90 95Thr Gly Phe Phe Ala Lys Lys Glu Ala Ala Glu Lys
His Leu Lys Gly100 105 110Gly Ala Lys Lys
Val Val Ile Thr Ala Pro Gly Gly Asn Asp Val Lys115 120
125Thr Val Val Phe Asn Thr Asn His Asp Val Leu Asp Gly Thr
Glu Thr130 135 140Val Ile Ser Gly Ala Ser
Cys Thr Thr Asn Cys Leu Ala Pro Met Ala145 150
155 160Lys Ala Leu Gln Asp Asn Phe Gly Val Val Glu
Gly Leu Met Thr Thr165 170 175Ile His Ala
Tyr Thr Gly Asp Gln Met Ile Leu Asp Gly Pro His Arg180
185 190Gly Gly Asp Leu Arg Arg Ala Arg Ala Gly Ala Ala
Asn Ile Val Pro195 200 205Asn Ser Thr Gly
Ala Ala Lys Ala Ile Gly Leu Val Ile Pro Glu Leu210 215
220Asn Gly Lys Leu Asp Gly Ser Ala Gln Arg Val Pro Thr Pro
Thr Gly225 230 235 240Ser
Val Thr Glu Leu Val Ala Val Leu Glu Lys Asn Val Thr Val Asp245
250 255Glu Val Asn Ala Ala Met Lys Ala Ala Ser Asn
Glu Ser Tyr Gly Tyr260 265 270Thr Glu Asp
Pro Ile Val Ser Ser Asp Ile Val Gly Met Ser Tyr Gly275
280 285Ser Leu Phe Asp Ala Thr Gln Thr Lys Val Leu Asp
Val Asp Gly Lys290 295 300Gln Leu Val Lys
Val Val Ser Trp Tyr Asp Asn Glu Met Ser Tyr Thr305 310
315 320Ala Gln Leu Val Arg Thr Leu Glu Tyr
Phe Ala Lys Ile Ala Lys325 330
33513293PRTStreptococcus pneumoniae 13Met Ala Ile Val Ser Ala Glu Lys Phe
Val Gln Ala Ala Arg Asp Asn1 5 10
15Gly Tyr Ala Val Gly Gly Phe Asn Thr Asn Asn Leu Glu Trp Thr
Gln20 25 30Ala Ile Leu Arg Ala Ala Glu
Ala Lys Lys Ala Pro Val Leu Ile Gln35 40
45Thr Ser Met Gly Ala Ala Lys Tyr Met Gly Gly Tyr Lys Val Ala Arg50
55 60Asn Leu Ile Ala Asn Leu Val Glu Ser Met
Gly Ile Thr Val Pro Val65 70 75
80Ala Ile His Leu Asp His Gly His Tyr Glu Asp Ala Leu Glu Cys
Ile85 90 95Glu Val Gly Tyr Thr Ser Ile
Met Phe Asp Gly Ser His Leu Pro Val100 105
110Glu Glu Asn Leu Lys Leu Ala Lys Glu Val Val Glu Lys Ala His Ala115
120 125Lys Gly Ile Ser Val Glu Ala Glu Val
Gly Thr Ile Gly Gly Glu Glu130 135 140Asp
Gly Ile Ile Gly Lys Gly Glu Leu Ala Pro Ile Glu Asp Ala Lys145
150 155 160Ala Met Val Glu Thr Gly
Ile Asp Phe Leu Ala Ala Gly Ile Gly Asn165 170
175Ile His Gly Pro Tyr Pro Val Asn Trp Glu Gly Leu Asp Leu Asp
His180 185 190Leu Gln Lys Leu Thr Glu Ala
Leu Pro Gly Phe Pro Ile Val Leu His195 200
205Gly Gly Ser Gly Ile Pro Asp Glu Gln Ile Gln Ala Ala Ile Lys Leu210
215 220Gly Val Ala Lys Val Asn Val Asn Thr
Glu Cys Gln Ile Ala Phe Ala225 230 235
240Asn Ala Thr Arg Lys Phe Ala Arg Asp Tyr Glu Ala Asn Glu
Ala Glu245 250 255Tyr Asp Lys Lys Lys Leu
Phe Asp Pro Arg Lys Phe Leu Ala Asp Gly260 265
270Val Lys Ala Ile Gln Ala Ser Val Glu Glu Arg Ile Asp Val Phe
Gly275 280 285Ser Glu Gly Lys
Ala29014336PRTStreptococcus pneumoniae 14Met Ala Ile Leu Val Thr Gly Gly
Ala Gly Tyr Ile Gly Ser His Thr1 5 10
15Val Val Glu Leu Leu Asn Leu Gly Lys Glu Val Ile Ile Val
Asp Asn20 25 30Leu Ser Asn Ser Ser Ile
Leu Val Leu Asp Arg Ile Glu Ala Ile Thr35 40
45Gly Ile Arg Pro Val Phe Tyr Glu Leu Asp Val Cys Asp Lys Gln Ala50
55 60Leu Arg Lys Val Phe Glu Gln Glu Ser
Ile Asp Ala Ala Ile His Phe65 70 75
80Ala Gly Tyr Lys Ala Val Gly Glu Ser Val Gln Lys Pro Val
Met Tyr85 90 95Tyr Lys Asn Asn Ile Met
Ser Thr Leu Ala Leu Val Glu Val Met Ser100 105
110Glu Phe Asn Val Lys Lys Ile Val Phe Ser Ser Ser Ala Thr Val
Tyr115 120 125Gly Ile Asn Asn Gln Ser Pro
Leu Ile Glu Thr Met Gln Thr Ser Ala130 135
140Thr Asn Pro Tyr Gly Tyr Thr Lys Val Met Leu Glu Gln Ile Leu Lys145
150 155 160Asp Val His Val
Ala Asp Ser Glu Trp Ser Ile Ala Leu Leu Arg Tyr165 170
175Phe Asn Pro Ile Gly Ala His Glu Ser Gly Leu Ile Gly Glu
Asp Pro180 185 190Ser Gly Ile Pro Asn Asn
Leu Met Pro Tyr Ile Ala Gln Val Ala Val195 200
205Gly Lys Leu Ser Glu Leu Ser Val Phe Gly Asn Asp Tyr Asp Thr
Leu210 215 220Asp Gly Thr Gly Val Arg Asp
Tyr Ile His Val Val Asp Leu Ala Ile225 230
235 240Gly His Ile Lys Ala Leu Glu Lys Val Ser Glu Lys
Thr Asp Val Tyr245 250 255Ile Tyr Asn Leu
Gly Ser Gly Glu Gly Thr Ser Val Leu Gln Leu Val260 265
270Asn Thr Phe Glu Ser Val Asn Lys Ile Pro Ile Pro Tyr Lys
Ile Val275 280 285Pro Arg Arg Ser Gly Asp
Val Ala Thr Cys Tyr Ala Asn Ala Asp Lys290 295
300Ala Tyr Lys Glu Leu Asn Trp Arg Thr Thr Lys Ser Ile Glu Asp
Met305 310 315 320Cys Arg
Asp Thr Trp Asn Trp Gln Ser Lys Asn Pro Asn Gly Tyr Asn325
330 33515620PRTStreptococcus pneumoniae 15Met Asn Ile
Ile Glu Glu Ile Met Thr Lys Leu Arg Glu Asp Ile Arg1 5
10 15Asn Ile Ala Ile Ile Ala His Val Asp
His Gly Lys Thr Thr Leu Val20 25 30Asp
Glu Leu Leu Lys Gln Ser Glu Thr Leu Asp Ala Arg Thr Glu Leu35
40 45Ala Glu Arg Ala Met Asp Ser Asn Asp Ile Glu
Lys Glu Arg Gly Ile50 55 60Thr Ile Leu
Ala Lys Asn Thr Ala Val Ala Tyr Asn Gly Thr Arg Ile65 70
75 80Asn Ile Met Asp Thr Pro Gly His
Ala Asp Phe Gly Gly Glu Val Glu85 90
95Arg Ile Met Lys Met Val Asp Gly Val Val Leu Val Val Asp Ala Tyr100
105 110Glu Gly Thr Met Pro Gln Thr Arg Phe Val
Leu Lys Lys Ala Leu Glu115 120 125Gln Asp
Leu Val Pro Ile Val Val Val Asn Lys Ile Asp Lys Pro Ser130
135 140Ala Arg Pro Ala Glu Val Val Asp Glu Val Leu Glu
Leu Phe Ile Glu145 150 155
160Leu Gly Ala Asp Asp Asp Gln Leu Asp Phe Pro Val Val Tyr Ala Ser165
170 175Ala Ile Asn Gly Thr Ser Ser Leu Ser
Asp Asp Pro Ala Asp Gln Glu180 185 190Ala
Thr Met Ala Pro Ile Phe Asp Thr Ile Ile Asp His Ile Pro Ala195
200 205Pro Val Asp Asn Ser Asp Glu Pro Leu Gln Phe
Gln Val Ser Leu Leu210 215 220Asp Tyr Asn
Asp Phe Val Gly Arg Ile Gly Ile Gly Arg Val Phe Arg225
230 235 240Gly Thr Val Lys Val Gly Asp
Gln Val Thr Leu Ser Lys Leu Asp Gly245 250
255Thr Thr Lys Asn Phe Arg Val Thr Lys Leu Phe Gly Phe Phe Gly Leu260
265 270Glu Arg Arg Glu Ile Gln Glu Ala Lys
Ala Gly Asp Leu Ile Ala Val275 280 285Ser
Gly Met Glu Asp Ile Phe Val Gly Glu Thr Ile Thr Pro Thr Asp290
295 300Ala Val Glu Ala Leu Pro Ile Leu His Ile Asp
Glu Pro Thr Leu Gln305 310 315
320Met Thr Phe Leu Val Asn Asn Ser Pro Phe Ala Gly Lys Glu Gly
Lys325 330 335Trp Val Thr Ser Arg Lys Val
Glu Glu Arg Leu Gln Ala Glu Leu Gln340 345
350Thr Asp Val Ser Leu Arg Val Asp Pro Thr Asp Ser Pro Asp Lys Trp355
360 365Thr Val Ser Gly Arg Gly Glu Leu His
Leu Ser Ile Leu Ile Glu Thr370 375 380Met
Arg Arg Glu Gly Tyr Glu Leu Gln Val Ser Arg Pro Glu Val Ile385
390 395 400Val Lys Glu Ile Asp Gly
Val Lys Cys Glu Pro Phe Glu Arg Val Gln405 410
415Ile Asp Thr Pro Glu Glu Tyr Gln Gly Ser Val Ile Gln Ser Leu
Ser420 425 430Glu Arg Lys Gly Glu Met Leu
Asp Met Ile Ser Thr Gly Asn Gly Gln435 440
445Thr Arg Leu Val Phe Leu Val Pro Ala Arg Gly Leu Ile Gly Tyr Ser450
455 460Thr Glu Phe Leu Ser Met Thr Arg Gly
Tyr Gly Ile Met Asn His Thr465 470 475
480Phe Asp Gln Tyr Leu Pro Leu Ile Pro Gly Glu Ile Gly Gly
Arg His485 490 495Arg Gly Ala Leu Val Ser
Ile Asp Ala Gly Lys Ala Thr Thr Tyr Ser500 505
510Ile Met Ser Ile Glu Glu Arg Gly Thr Ile Phe Val Asn Pro Gly
Thr515 520 525Glu Val Tyr Glu Gly Met Ile
Ile Gly Glu Asn Ser Arg Glu Asn Asp530 535
540Leu Thr Val Asn Ile Thr Lys Ala Lys Gln Met Thr Asn Val Arg Ser545
550 555 560Ala Thr Lys Asp
Gln Thr Ala Val Ile Lys Thr Pro Arg Ile Leu Thr565 570
575Leu Glu Glu Ser Leu Glu Phe Leu Asn Asp Asp Glu Tyr Met
Glu Val580 585 590Thr Pro Glu Ser Ile Arg
Leu Arg Lys Gln Ile Leu Asn Lys Ala Glu595 600
605Arg Glu Lys Ala Asn Lys Lys Lys Lys Ser Ala Glu610
615 62016520PRTStreptococcus pneumoniae 16Met Ser Asn
Ile Ser Thr Asp Leu Gln Asp Val Glu Lys Ile Ile Val1 5
10 15Leu Asp Tyr Gly Ser Gln Tyr Asn Gln
Leu Ile Ser Arg Arg Ile Arg20 25 30Glu
Ile Gly Val Phe Ser Glu Leu Lys Ser His Lys Ile Ser Ala Ala35
40 45Glu Val Arg Glu Val Asn Pro Val Gly Ile Ile
Leu Ser Gly Gly Pro50 55 60Asn Ser Val
Tyr Glu Asp Gly Ser Phe Asp Ile Asp Pro Glu Ile Phe65 70
75 80Glu Leu Gly Ile Pro Ile Leu Gly
Ile Cys Tyr Gly Met Gln Leu Leu85 90
95Thr His Lys Leu Gly Gly Lys Val Val Pro Ala Gly Asp Ala Gly Asn100
105 110Arg Glu Tyr Gly Gln Ser Thr Leu Thr His
Thr Pro Ser Ala Leu Phe115 120 125Glu Ser
Thr Pro Asp Glu Gln Thr Val Leu Met Ser His Gly Asp Ala130
135 140Val Thr Glu Ile Pro Ala Asp Phe Val Arg Thr Gly
Thr Ser Ala Asp145 150 155
160Cys Pro Tyr Ala Ala Ile Glu Asn Pro Asp Lys His Ile Tyr Gly Ile165
170 175Gln Phe His Pro Glu Val Arg His Ser
Val Tyr Gly Asn Asp Ile Leu180 185 190Arg
Asn Phe Ala Leu Asn Ile Cys Lys Ala Lys Gly Asp Trp Ser Met195
200 205Asp Asn Phe Ile Asp Met Gln Ile Lys Lys Ile
Arg Glu Thr Val Gly210 215 220Asp Lys Arg
Val Leu Leu Gly Leu Ser Gly Gly Val Asp Ser Ser Val225
230 235 240Val Gly Val Leu Leu Gln Lys
Ala Ile Gly Asp Gln Leu Ile Cys Ile245 250
255Phe Val Asp His Gly Leu Leu Arg Lys Gly Glu Ala Asp Gln Val Met260
265 270Asp Met Leu Gly Gly Lys Phe Gly Leu
Asn Ile Val Lys Ala Asp Ala275 280 285Ala
Lys Arg Phe Leu Asp Lys Leu Ala Gly Val Ser Asp Pro Glu Gln290
295 300Lys Arg Lys Ile Ile Gly Asn Glu Phe Val Tyr
Val Phe Asp Asp Glu305 310 315
320Ala Ser Lys Leu Lys Asp Val Lys Phe Leu Ala Gln Gly Thr Leu
Tyr325 330 335Thr Asp Val Ile Glu Ser Gly
Thr Asp Thr Ala Gln Thr Ile Lys Ser340 345
350His His Asn Val Gly Gly Leu Pro Glu Asp Met Gln Phe Glu Leu Ile355
360 365Glu Pro Leu Asn Thr Leu Tyr Lys Asp
Glu Val Arg Ala Leu Gly Thr370 375 380Glu
Leu Gly Met Pro Asp His Ile Val Trp Arg Gln Pro Phe Pro Gly385
390 395 400Pro Gly Leu Ala Ile Arg
Val Met Gly Glu Ile Thr Glu Glu Lys Leu405 410
415Glu Thr Val Arg Glu Ser Asp Ala Ile Leu Arg Glu Glu Ile Ala
Lys420 425 430Ala Gly Leu Asp Arg Asp Ile
Trp Gln Tyr Phe Thr Val Asn Thr Gly435 440
445Val Arg Ser Val Gly Val Met Gly Asp Gly Arg Thr Tyr Asp Tyr Thr450
455 460Ile Ala Ile Arg Ala Ile Thr Ser Ile
Asp Gly Met Thr Ala Asp Phe465 470 475
480Ala Lys Ile Pro Trp Glu Val Leu Gln Lys Ile Ser Val Arg
Ile Val485 490 495Asn Glu Val Asp His Val
Asn Arg Ile Val Tyr Asp Ile Thr Ser Lys500 505
510Pro Pro Ala Thr Val Glu Trp Glu515
52017486PRTStreptococcus pneumoniae 17Met Ser Lys Asp Ile Arg Val Arg Tyr
Ala Pro Ser Pro Thr Gly Leu1 5 10
15Leu His Ile Gly Asn Ala Arg Thr Ala Leu Phe Asn Tyr Leu Tyr
Ala20 25 30Arg His His Gly Gly Thr Phe
Leu Ile Arg Ile Glu Asp Thr Asp Arg35 40
45Lys Arg His Val Glu Asp Gly Glu Arg Ser Gln Leu Glu Asn Leu Arg50
55 60Trp Leu Gly Met Asp Trp Asp Glu Ser Pro
Glu Ser His Glu Asn Tyr65 70 75
80Arg Gln Ser Glu Arg Leu Asp Leu Tyr Gln Lys Tyr Ile Asp Gln
Leu85 90 95Leu Ala Glu Gly Lys Ala Tyr
Lys Ser Tyr Val Thr Glu Glu Glu Leu100 105
110Ala Ala Glu Arg Glu Arg Gln Glu Val Ala Gly Glu Thr Pro Arg Tyr115
120 125Ile Asn Glu Tyr Leu Gly Met Ser Glu
Glu Glu Lys Ala Ala Tyr Ile130 135 140Ala
Glu Arg Glu Ala Ala Gly Ile Ile Pro Thr Val Arg Leu Ala Val145
150 155 160Asn Glu Ser Gly Ile Tyr
Lys Trp His Asp Met Val Lys Gly Asp Ile165 170
175Glu Phe Glu Gly Gly Asn Ile Gly Gly Asp Trp Val Ile Gln Lys
Lys180 185 190Asp Gly Tyr Pro Thr Tyr Asn
Phe Ala Val Val Ile Asp Asp His Asp195 200
205Met Gln Ile Ser His Val Ile Arg Gly Asp Asp His Ile Ala Asn Thr210
215 220Pro Lys Gln Leu Met Val Tyr Glu Ala
Leu Gly Trp Glu Ala Pro Glu225 230 235
240Phe Gly His Met Thr Leu Ile Ile Asn Ser Glu Thr Gly Lys
Lys Leu245 250 255Ser Lys Arg Asp Thr Asn
Thr Leu Gln Phe Ile Glu Asp Tyr Arg Lys260 265
270Lys Gly Tyr Leu Pro Glu Ala Val Phe Asn Phe Ile Ala Leu Leu
Gly275 280 285Trp Asn Pro Gly Gly Glu Asp
Glu Ile Phe Ser Arg Glu Glu Phe Ile290 295
300Lys Leu Phe Asp Glu Asn Arg Leu Ser Lys Ser Pro Ala Ala Phe Asp305
310 315 320Gln Lys Lys Leu
Asp Trp Met Ser Asn Asp Tyr Ile Lys Asn Ala Asp325 330
335Leu Glu Thr Ile Phe Glu Met Ala Lys Pro Phe Leu Glu Glu
Ala Gly340 345 350Arg Leu Thr Asp Lys Ala
Glu Lys Leu Val Glu Leu Tyr Lys Pro Gln355 360
365Met Lys Ser Val Asp Glu Ile Ile Pro Leu Thr Asp Leu Phe Phe
Ser370 375 380Asp Phe Pro Glu Leu Thr Glu
Ala Glu Arg Glu Val Met Thr Gly Glu385 390
395 400Thr Val Pro Thr Val Leu Glu Ala Phe Lys Ala Lys
Leu Glu Ala Met405 410 415Thr Asp Asp Glu
Phe Val Thr Glu Asn Ile Phe Pro Gln Ile Lys Ala420 425
430Val Gln Lys Glu Thr Gly Ile Lys Gly Lys Asn Leu Phe Met
Pro Ile435 440 445Arg Ile Ala Val Ser Gly
Glu Met His Gly Pro Glu Leu Pro Asp Thr450 455
460Ile Phe Leu Leu Gly Arg Glu Lys Ser Ile Gln His Ile Glu Asn
Met465 470 475 480Leu Lys
Glu Ile Ser Lys48518448PRTStreptococcus pneumoniae 18Met Thr Ser Ala Lys
Glu Tyr Ile Gln Ser Val Phe Glu Thr Val Lys1 5
10 15Ala Arg Asn Gly His Glu Ala Glu Phe Leu Gln
Ala Val Glu Glu Phe20 25 30Phe Asn Thr
Leu Glu Pro Val Phe Glu Lys His Pro Glu Tyr Ile Glu35 40
45Glu Asn Ile Leu Ala Arg Ile Thr Glu Pro Glu Arg Val
Val Ser Phe50 55 60Arg Val Pro Trp Val
Asp Arg Asp Gly Lys Ile Gln Val Asn Arg Gly65 70
75 80Tyr Arg Val Gln Phe Asn Ser Ala Val Gly
Pro Tyr Lys Gly Gly Leu85 90 95Arg Phe
His Pro Thr Val Asn Gln Gly Ile Leu Lys Phe Leu Gly Phe100
105 110Glu Gln Ile Phe Lys Asn Val Leu Thr Gly Leu Pro
Ile Gly Gly Gly115 120 125Lys Gly Gly Ser
Asp Phe Asp Pro Lys Gly Lys Thr Asp Ala Glu Val130 135
140Met Arg Phe Cys Gln Ser Phe Met Thr Glu Leu Gln Lys His
Ile Gly145 150 155 160Pro
Ser Leu Asp Val Pro Ala Gly Asp Ile Gly Val Gly Gly Arg Glu165
170 175Ile Gly Tyr Leu Tyr Gly Gln Tyr Lys Arg Leu
Asn Gln Phe Asp Ala180 185 190Gly Val Leu
Thr Gly Lys Pro Leu Gly Phe Gly Gly Ser Leu Ile Arg195
200 205Pro Glu Ala Thr Gly Tyr Gly Leu Val Tyr Tyr Thr
Glu Glu Met Leu210 215 220Lys Ala Asn Gly
Asn Ser Phe Ala Gly Lys Lys Val Val Ile Ser Gly225 230
235 240Ser Gly Asn Val Ala Gln Tyr Ala Leu
Gln Lys Ala Thr Glu Leu Gly245 250 255Ala
Thr Val Ile Ser Val Ser Asp Ser Asn Gly Tyr Val Ile Asp Glu260
265 270Asn Gly Ile Asp Phe Asp Leu Leu Val Asp Val
Lys Glu Lys Arg Arg275 280 285Ala Arg Leu
Thr Glu Tyr Ala Ala Glu Lys Ala Thr Ala Thr Tyr His290
295 300Glu Gly Thr Val Trp Thr Tyr Ala Gly Asn Tyr Asp
Ile Ala Leu Pro305 310 315
320Cys Ala Thr Gln Asn Glu Ile Asn Gly Glu Ala Ala Lys Arg Leu Val325
330 335Ala Gln Gly Val Ile Cys Val Ser Glu
Gly Ala Asn Met Pro Ser Asp340 345 350Leu
Asp Ala Ile Lys Val Tyr Lys Glu Asn Gly Ile Phe Tyr Gly Pro355
360 365Ala Lys Ala Ala Asn Ala Gly Gly Val Ala Val
Ser Ala Leu Glu Met370 375 380Ser Gln Asn
Ser Leu Arg Leu Ser Trp Thr Arg Glu Glu Val Asp Gly385
390 395 400Arg Leu Lys Asp Ile Met Thr
Asn Ile Phe Asn Thr Ala Lys Thr Thr405 410
415Ser Glu Thr Tyr Gly Leu Asp Lys Asp Tyr Leu Ala Gly Ala Asn Ile420
425 430Ala Ala Phe Glu Asn Val Ala Asn Ala
Met Ile Ala Gln Gly Ile Val435 440
44519346PRTStreptococcus pneumoniae 19Met Ala Glu Ile Thr Ala Lys Leu Val
Lys Glu Leu Arg Glu Lys Ser1 5 10
15Gly Ala Gly Val Met Asp Ala Lys Lys Ala Leu Val Glu Thr Asp
Gly20 25 30Asp Ile Glu Lys Ala Ile Glu
Leu Leu Arg Glu Lys Gly Met Ala Lys35 40
45Ala Ala Lys Lys Ala Asp Arg Val Ala Ala Glu Gly Leu Thr Gly Val50
55 60Tyr Val Asn Gly Asn Val Ala Ala Val Ile
Glu Val Asn Ala Glu Thr65 70 75
80Asp Phe Val Ala Lys Asn Ala Gln Phe Val Glu Leu Val Asn Thr
Thr85 90 95Ala Lys Val Ile Ala Glu Gly
Lys Pro Ala Asn Asn Glu Glu Ala Leu100 105
110Ala Leu Ile Met Pro Ser Gly Glu Thr Leu Glu Ala Ala Tyr Val Ser115
120 125Ala Thr Ala Thr Ile Gly Glu Lys Ile
Ser Phe Arg Arg Phe Ala Leu130 135 140Ile
Glu Lys Thr Asp Ala Gln His Phe Gly Ala Tyr Gln His Asn Gly145
150 155 160Gly Arg Ile Gly Val Ile
Ser Val Val Glu Gly Gly Asp Glu Ala Leu165 170
175Ala Lys Gln Leu Ser Met His Ile Ala Ala Met Lys Pro Thr Val
Leu180 185 190Ser Tyr Lys Glu Leu Asp Glu
Gln Phe Val Lys Asp Glu Leu Ala Gln195 200
205Leu Asn His Val Ile Asp Gln Asp Asn Glu Ser Arg Ala Met Val Asn210
215 220Lys Pro Ala Leu Pro His Leu Lys Tyr
Gly Ser Lys Ala Gln Leu Thr225 230 235
240Asp Asp Val Ile Ala Gln Ala Glu Ala Asp Ile Lys Ala Glu
Leu Ala245 250 255Ala Glu Gly Lys Pro Glu
Lys Ile Trp Asp Lys Ile Ile Pro Gly Lys260 265
270Met Asp Arg Phe Met Leu Asp Asn Thr Lys Val Asp Gln Ala Tyr
Thr275 280 285Leu Leu Ala Gln Val Tyr Ile
Met Asp Asp Ser Lys Thr Val Glu Ala290 295
300Tyr Leu Glu Ser Val Asn Ala Ser Val Val Glu Phe Ala Arg Phe Glu305
310 315 320Val Gly Glu Gly
Ile Glu Lys Ala Ala Asn Asp Phe Glu Ala Glu Val325 330
335Ala Ala Thr Met Ala Ala Ala Leu Asn Asn340
34520398PRTStreptococcus pneumoniae 20Met Ala Lys Leu Thr Val Lys Asp
Val Asp Leu Lys Gly Lys Lys Val1 5 10
15Leu Val Arg Val Asp Phe Asn Val Pro Leu Lys Asp Gly Val
Ile Thr20 25 30Asn Asp Asn Arg Ile Thr
Ala Ala Leu Pro Thr Ile Lys Tyr Ile Ile35 40
45Glu Gln Gly Gly Arg Ala Ile Leu Phe Ser His Leu Gly Arg Val Lys50
55 60Glu Glu Ala Asp Lys Ala Gly Lys Ser
Leu Ala Pro Val Ala Ala Asp65 70 75
80Leu Ala Ala Lys Leu Gly Gln Asp Val Val Phe Pro Gly Val
Thr Arg85 90 95Gly Ala Glu Leu Glu Ala
Ala Ile Asn Ala Leu Glu Asp Gly Gln Val100 105
110Leu Leu Val Glu Asn Thr Arg Tyr Glu Asp Val Asp Gly Lys Lys
Glu115 120 125Ser Lys Asn Asp Pro Glu Leu
Gly Lys Tyr Trp Ala Ser Leu Gly Asp130 135
140Gly Ile Phe Val Asn Asp Ala Phe Gly Thr Ala His Arg Ala His Ala145
150 155 160Ser Asn Val Gly
Ile Ser Ala Asn Val Glu Lys Ala Val Ala Gly Phe165 170
175Leu Leu Glu Asn Glu Ile Ala Tyr Ile Gln Glu Ala Val Glu
Thr Pro180 185 190Glu Arg Pro Phe Val Ala
Ile Leu Gly Gly Ser Lys Val Ser Asp Lys195 200
205Ile Gly Val Ile Glu Asn Leu Leu Glu Lys Ala Asp Asn Val Leu
Ile210 215 220Gly Gly Gly Met Thr Tyr Thr
Phe Tyr Lys Ala Gln Gly Ile Glu Ile225 230
235 240Gly Asn Ser Leu Val Glu Glu Asp Lys Leu Asp Val
Ala Lys Ala Leu245 250 255Leu Glu Lys Ala
Asn Gly Lys Leu Ile Leu Pro Val Asp Ser Lys Glu260 265
270Ala Asn Ala Phe Ala Gly Tyr Thr Glu Val Arg Asp Thr Glu
Gly Glu275 280 285Ala Val Ser Glu Gly Phe
Leu Gly Leu Asp Ile Gly Pro Lys Ser Ile290 295
300Ala Lys Phe Asp Glu Ala Leu Thr Gly Ala Lys Thr Val Val Trp
Asn305 310 315 320Gly Pro
Met Gly Val Phe Glu Asn Pro Asp Phe Gln Ala Gly Thr Ile325
330 335Gly Val Met Asp Ala Ile Val Lys Gln Pro Gly Val
Lys Ser Ile Ile340 345 350Gly Gly Gly Asp
Ser Ala Ala Ala Ala Ile Asn Leu Gly Arg Ala Asp355 360
365Lys Phe Ser Trp Ile Ser Thr Gly Gly Gly Ala Ser Met Glu
Leu Leu370 375 380Glu Gly Lys Val Leu Pro
Gly Leu Ala Ala Leu Thr Glu Lys385 390
39521400PRTStreptococcus pneumoniae 21Met Asn Glu Phe Glu Asp Leu Leu Asn
Ser Val Ser Gln Val Glu Thr1 5 10
15Gly Asp Val Val Ser Ala Glu Val Leu Thr Val Asp Ala Thr Gln
Ala20 25 30Asn Val Ala Ile Ser Gly Thr
Gly Val Glu Gly Val Leu Thr Leu Arg35 40
45Glu Leu Thr Asn Asp Arg Asp Ala Asp Ile Asn Asp Phe Val Lys Val50
55 60Gly Glu Val Leu Asp Val Leu Val Leu Arg
Gln Val Val Gly Lys Asp65 70 75
80Thr Asp Thr Val Thr Tyr Leu Val Ser Lys Lys Arg Leu Glu Ala
Arg85 90 95Lys Ala Trp Asp Lys Leu Val
Gly Arg Glu Glu Glu Val Val Thr Val100 105
110Lys Gly Thr Arg Ala Val Lys Gly Gly Leu Ser Val Glu Phe Glu Gly115
120 125Val Arg Gly Phe Ile Pro Ala Ser Met
Leu Asp Thr Arg Phe Val Arg130 135 140Asn
Ala Glu Arg Phe Val Gly Gln Glu Phe Asp Thr Lys Ile Lys Glu145
150 155 160Val Asn Ala Lys Glu Asn
Arg Phe Ile Leu Ser Arg Arg Glu Val Val165 170
175Glu Ala Ala Thr Ala Ala Ala Arg Ala Glu Val Phe Gly Lys Leu
Ala180 185 190Val Gly Asp Val Val Thr Gly
Lys Val Ala Arg Ile Thr Ser Phe Gly195 200
205Ala Phe Val Asp Leu Gly Gly Val Asp Gly Leu Val His Leu Thr Glu210
215 220Leu Ser His Glu Arg Asn Val Ser Pro
Lys Ser Val Val Thr Val Gly225 230 235
240Glu Glu Ile Glu Val Lys Ile Leu Asp Leu Asn Glu Glu Glu
Gly Arg245 250 255Val Ser Leu Ser Leu Lys
Ala Thr Val Pro Gly Pro Trp Asp Gly Val260 265
270Glu Gln Lys Leu Ala Lys Gly Asp Val Val Glu Gly Thr Val Lys
Arg275 280 285Leu Thr Asp Phe Gly Ala Phe
Val Glu Val Leu Pro Gly Ile Asp Gly290 295
300Leu Val His Val Ser Gln Ile Ser His Lys Arg Ile Glu Asn Pro Lys305
310 315 320Glu Ala Leu Lys
Val Gly Gln Glu Val Gln Val Lys Val Leu Glu Val325 330
335Asn Ala Asp Ala Glu Arg Val Ser Leu Ser Ile Lys Ala Leu
Glu Glu340 345 350Arg Pro Ala Gln Glu Glu
Gly Gln Lys Glu Glu Lys Arg Ala Ala Arg355 360
365Pro Arg Arg Pro Arg Arg Gln Glu Lys Arg Asp Phe Glu Leu Pro
Glu370 375 380Thr Gln Thr Gly Phe Ser Met
Ala Asp Leu Phe Gly Asp Ile Glu Leu385 390
395 40022474PRTStreptococcus pneumoniae 22Met Thr Lys
Ala Asn Phe Gly Val Val Gly Met Ala Val Met Gly Arg1 5
10 15Asn Leu Ala Leu Asn Ile Glu Ser Arg
Gly Tyr Thr Val Ala Ile Tyr20 25 30Asn
Arg Ser Lys Glu Lys Thr Glu Asp Val Ile Ala Cys His Pro Glu35
40 45Lys Asn Phe Val Pro Ser Tyr Asp Val Glu Ser
Phe Val Asn Ser Ile50 55 60Glu Lys Pro
Arg Arg Ile Met Leu Met Val Gln Ala Gly Pro Gly Thr65 70
75 80Asp Ala Thr Ile Gln Ala Leu Leu
Pro His Leu Asp Lys Gly Asp Ile85 90
95Leu Ile Asp Gly Gly Asn Thr Phe Tyr Lys Asp Thr Ile Arg Arg Asn100
105 110Glu Glu Leu Ala Asn Ser Gly Ile Asn Phe
Ile Gly Thr Gly Val Ser115 120 125Gly Gly
Glu Lys Gly Ala Leu Glu Gly Pro Ser Ile Met Pro Gly Gly130
135 140Gln Lys Glu Ala Tyr Glu Leu Val Ala Asp Val Leu
Glu Glu Ile Ser145 150 155
160Ala Lys Ala Pro Glu Asp Gly Lys Pro Cys Val Thr Tyr Ile Gly Pro165
170 175Asp Gly Ala Gly His Tyr Val Lys Met
Val His Asn Gly Ile Glu Tyr180 185 190Gly
Asp Met Gln Leu Ile Ala Glu Ser Tyr Asp Leu Met Gln His Leu195
200 205Leu Gly Leu Ser Ala Glu Asp Met Ala Glu Ile
Phe Thr Glu Trp Asn210 215 220Lys Gly Glu
Leu Asp Ser Tyr Leu Ile Glu Ile Thr Ala Asp Ile Leu225
230 235 240Ser Arg Lys Asp Asp Glu Gly
Gln Asp Gly Pro Ile Val Asp Tyr Ile245 250
255Leu Asp Ala Ala Gly Asn Lys Gly Thr Gly Lys Trp Thr Ser Gln Ser260
265 270Ser Leu Asp Leu Gly Val Pro Leu Ser
Leu Ile Thr Glu Ser Val Phe275 280 285Ala
Arg Tyr Ile Ser Thr Tyr Lys Glu Glu Arg Val His Ala Ser Lys290
295 300Val Leu Pro Lys Pro Ala Ala Phe Asn Phe Glu
Gly Asp Lys Ala Glu305 310 315
320Leu Ile Glu Lys Ile Arg Gln Ala Leu Tyr Phe Ser Lys Ile Ile
Ser325 330 335Tyr Ala Gln Gly Phe Ala Gln
Leu Arg Val Ala Ser Lys Glu Asn Asn340 345
350Trp Asn Leu Pro Phe Ala Asp Ile Ala Ser Ile Trp Arg Asp Gly Cys355
360 365Ile Ile Arg Ser Arg Phe Leu Gln Lys
Ile Thr Asp Ala Tyr Asn Arg370 375 380Asp
Ala Asp Leu Ala Asn Leu Leu Leu Asp Glu Tyr Phe Leu Asp Val385
390 395 400Thr Ala Lys Tyr Gln Gln
Ala Val Arg Asp Ile Val Ala Leu Ala Val405 410
415Gln Ala Gly Val Pro Val Pro Thr Phe Ser Ala Ala Ile Thr Tyr
Phe420 425 430Asp Ser Tyr Arg Ser Ala Asp
Leu Pro Ala Asn Leu Ile Gln Ala Gln435 440
445Arg Asp Tyr Phe Gly Ala His Thr Tyr Gln Arg Lys Asp Lys Glu Gly450
455 460Thr Phe His Tyr Ser Trp Tyr Asp Glu
Lys465 47023444PRTStreptococcus pneumoniae 23Met Asn Ala
Ile Gln Glu Ser Phe Thr Asp Lys Leu Phe Ala Asn Tyr1 5
10 15Glu Ala Asn Val Lys Tyr Gln Ala Ile
Glu Asn Ala Ala Ser His Asn20 25 30Gly
Ile Phe Ala Ala Leu Glu Arg Arg Gln Ser His Val Asp Asn Thr35
40 45Pro Val Phe Ser Leu Asp Leu Thr Lys Asp Lys
Val Thr Asn Gln Lys50 55 60Ala Ser Gly
Arg Cys Trp Met Phe Ala Ala Leu Asn Thr Phe Arg His65 70
75 80Lys Leu Ile Ser Gln Tyr Lys Leu
Glu Asn Phe Glu Leu Ser Gln Ala85 90
95His Thr Phe Phe Trp Asp Lys Tyr Glu Lys Ser Asn Trp Phe Leu Glu100
105 110Gln Val Ile Ala Thr Ser Asp Gln Glu Leu
Thr Ser Arg Lys Val Ser115 120 125Phe Leu
Leu Gln Thr Pro Gln Gln Asp Gly Gly Gln Trp Asp Met Val130
135 140Val Ser Leu Phe Glu Lys Tyr Gly Val Val Pro Lys
Ser Val Tyr Pro145 150 155
160Glu Ser Val Ser Ser Ser Ser Ser Arg Glu Leu Asn Ala Ile Leu Asn165
170 175Lys Leu Leu Arg Gln Asp Ala Gln Ile
Leu Arg Asp Leu Leu Val Ser180 185 190Gly
Ala Asp Gln Ala Thr Val Gln Ala Lys Lys Glu Asp Leu Leu Gln195
200 205Glu Ile Phe Asn Phe Leu Ala Met Ser Leu Gly
Leu Pro Pro Arg Lys210 215 220Phe Asp Phe
Ala Tyr Arg Asp Lys Asp Asn Asn Tyr Lys Ser Glu Lys225
230 235 240Gly Ile Thr Pro Gln Glu Phe
Tyr Lys Lys Tyr Val Asn Leu Pro Leu245 250
255Glu Asp Tyr Val Ser Val Ile Asn Ala Pro Thr Ala Asp Lys Pro Tyr260
265 270Gly Lys Ser Tyr Thr Val Glu Met Leu
Gly Asn Val Val Gly Ser Arg275 280 285Ala
Val Arg Tyr Ile Asn Val Pro Met Glu Arg Leu Lys Glu Leu Ala290
295 300Ile Ala Gln Met Gln Ala Gly Glu Thr Val Trp
Phe Gly Ser Asp Val305 310 315
320Gly Gln Leu Ser Asn Arg Lys Ala Gly Ile Leu Ala Thr Asp Val
Tyr325 330 335Asp Phe Glu Ser Ser Met Asp
Ile Lys Leu Thr Gln Asp Lys Ala Gly340 345
350Arg Leu Asp Tyr Ser Glu Ser Leu Met Thr His Ala Met Val Leu Thr355
360 365Gly Val Asp Leu Asp Glu Asn Gly Lys
Ser Thr Lys Trp Lys Val Glu370 375 380Asn
Ser Trp Gly Asp Lys Val Gly Thr Asp Gly Tyr Phe Val Ala Ser385
390 395 400Asp Ala Trp Met Asp Glu
Tyr Thr Tyr Gln Ile Val Val Arg Lys Glu405 410
415Leu Leu Thr Ala Glu Glu Gln Ala Ala Tyr Gly Ala Glu Pro Ile
Val420 425 430Leu Ala Pro Trp Asp Pro Met
Gly Ala Leu Ala Glu435 440241058PRTStreptococcus
pneumoniae 24Met Pro Lys Arg Thr Asp Ile Gln Lys Ile Met Val Ile Gly Ser
Gly1 5 10 15Pro Ile Ile
Ile Gly Gln Ala Ala Glu Phe Asp Tyr Ala Gly Thr Gln20 25
30Ala Cys Leu Ser Leu Lys Glu Glu Gly Tyr Glu Val Val
Leu Val Asn35 40 45Ser Asn Pro Ala Thr
Ile Met Thr Asp Lys Glu Ile Ala Asp Lys Val50 55
60Tyr Ile Glu Pro Ile Thr Leu Glu Phe Val Thr Arg Ile Leu Arg
Lys65 70 75 80Glu Gly
Pro Asp Ala Leu Leu Pro Thr Leu Gly Gly Gln Thr Gly Leu85
90 95Asn Met Ala Met Glu Leu Ser Lys Asn Gly Ile Leu
Asp Glu Leu Gly100 105 110Val Glu Leu Leu
Gly Thr Lys Leu Ser Ala Ile Asp Gln Ala Glu Asp115 120
125Arg Asp Leu Phe Lys Gln Leu Met Glu Glu Leu Glu Gln Pro
Ile Pro130 135 140Glu Ser Glu Ile Val Asn
Thr Val Glu Glu Ala Val Ala Phe Ala Ala145 150
155 160Thr Ile Gly Tyr Pro Val Ile Val Arg Pro Ala
Phe Thr Leu Gly Gly165 170 175Thr Gly Gly
Gly Met Cys Ala Asn Glu Lys Glu Leu Arg Glu Ile Thr180
185 190Glu Asn Gly Leu Lys Leu Ser Pro Val Thr Gln Cys
Leu Ile Glu Arg195 200 205Ser Ile Ala Gly
Phe Lys Glu Ile Glu Tyr Glu Val Met Arg Asp Ser210 215
220Ala Asp Asn Ala Leu Val Val Cys Asn Met Glu Asn Phe Asp
Pro Val225 230 235 240Gly
Ile His Thr Gly Asp Ser Ile Val Phe Ala Pro Ala Gln Thr Met245
250 255Ser Asp Tyr Glu Asn Gln Met Leu Arg Asp Ala
Ser Leu Ser Ile Ile260 265 270Arg Ala Leu
Lys Ile Glu Gly Gly Cys Asn Val Gln Leu Ala Leu Asp275
280 285Pro Asn Ser Phe Lys Tyr Tyr Val Ile Glu Val Asn
Pro Arg Val Ser290 295 300Arg Ser Ser Ala
Leu Ala Ser Lys Ala Thr Gly Tyr Pro Ile Ala Lys305 310
315 320Leu Ala Ala Lys Ile Ala Val Gly Leu
Thr Leu Asp Glu Val Ile Asn325 330 335Pro
Val Thr Gly Ser Thr Tyr Ala Met Phe Glu Pro Ala Leu Asp Tyr340
345 350Val Val Ala Lys Ile Pro Arg Phe Pro Phe Asp
Lys Phe Glu Lys Gly355 360 365Glu Arg Arg
Leu Gly Thr Gln Met Lys Ala Thr Gly Glu Val Met Ala370
375 380Ile Gly Arg Asn Ile Glu Glu Ser Leu Leu Lys Ala
Cys Arg Ser Leu385 390 395
400Glu Ile Gly Val His His Asn Glu Ile Pro Glu Leu Ala Ala Val Ser405
410 415Asp Asp Ala Leu Ile Glu Lys Val Val
Lys Ala Gln Asp Asp Arg Leu420 425 430Phe
Tyr Val Ser Glu Ala Ile Arg Arg Gly Tyr Thr Pro Glu Glu Ile435
440 445Ala Glu Leu Thr Lys Ile Asp Ile Phe Tyr Leu
Asp Lys Leu Leu His450 455 460Ile Phe Glu
Ile Glu Gln Glu Leu Gly Ala His Pro Gln Asp Leu Glu465
470 475 480Val Leu Lys Thr Ala Lys Leu
Asn Gly Phe Ser Asp Arg Lys Ile Ala485 490
495Glu Leu Trp Gly Thr Thr Asp Asp Lys Val Arg Gln Leu Arg Leu Glu500
505 510Asn Lys Ile Val Pro Val Tyr Lys Met
Val Asp Thr Cys Ala Ala Glu515 520 525Phe
Asp Ser Glu Thr Pro Tyr Phe Tyr Ser Thr Tyr Gly Trp Glu Asn530
535 540Glu Ser Ile Arg Ser Asp Lys Glu Ser Val Leu
Val Leu Gly Ser Gly545 550 555
560Pro Ile Arg Ile Gly Gln Gly Val Glu Phe Asp Tyr Ala Thr Val
His565 570 575Ser Val Lys Ala Ile Gln Ala
Ala Gly Tyr Glu Ala Ile Ile Met Asn580 585
590Ser Asn Pro Glu Thr Val Ser Thr Asp Phe Ser Val Ser Asp Lys Leu595
600 605Tyr Phe Glu Pro Leu Thr Phe Glu Asp
Val Met Asn Val Ile Asp Leu610 615 620Glu
Gln Pro Lys Gly Val Ile Val Gln Phe Gly Gly Gln Thr Ala Ile625
630 635 640Asn Leu Ala Glu Pro Leu
Ala Lys Ala Gly Val Thr Ile Leu Gly Thr645 650
655Gln Val Ala Asp Leu Asp Arg Ala Glu Asp Arg Asp Leu Phe Glu
Gln660 665 670Ala Leu Lys Glu Leu Asp Ile
Pro Gln Pro Pro Gly Gln Thr Ala Thr675 680
685Asn Glu Glu Glu Ala Ala Leu Ala Ala Arg Lys Ile Gly Phe Pro Val690
695 700Leu Val Arg Pro Ser Tyr Val Leu Gly
Gly Arg Ala Met Glu Ile Val705 710 715
720Glu Asn Glu Glu Asp Leu Arg Ser Tyr Met Arg Thr Ala Val
Lys Ala725 730 735Ser Pro Asp His Pro Val
Leu Val Asp Ser Tyr Ile Val Gly Gln Glu740 745
750Cys Glu Val Asp Ala Ile Ser Asp Gly Lys Asn Val Leu Ile Pro
Gly755 760 765Ile Met Glu His Ile Glu Arg
Ala Gly Val His Ser Gly Asp Ser Met770 775
780Ala Val Tyr Pro Pro Gln Thr Leu Ser Gln Lys Val Gln Glu Thr Ile785
790 795 800Ala Asp Tyr Thr
Lys Arg Leu Ala Ile Gly Leu His Cys Leu Gly Met805 810
815Met Asn Ile Gln Phe Val Ile Lys Asp Glu Lys Val Tyr Val
Ile Glu820 825 830Val Asn Pro Arg Ala Ser
Arg Thr Val Pro Phe Leu Ser Lys Val Thr835 840
845Asn Ile Pro Met Ala Gln Val Ala Thr Lys Leu Ile Leu Gly Gln
Ser850 855 860Leu Ser Glu Leu Gly Tyr Gln
Asn Gly Leu Tyr Pro Glu Ser Thr Arg865 870
875 880Val His Ile Lys Ala Pro Val Phe Ser Phe Thr Lys
Leu Ala Lys Val885 890 895Asp Ser Leu Leu
Gly Pro Glu Met Lys Ser Thr Gly Glu Val Met Gly900 905
910Ser Asp Ala Thr Leu Glu Lys Ala Leu Tyr Lys Ala Phe Glu
Ala Ser915 920 925Tyr Leu His Leu Pro Thr
Phe Gly Asn Val Val Phe Thr Ile Ala Asp930 935
940Asp Ala Lys Glu Glu Ala Leu Asn Leu Ala Arg Arg Phe Gln Asn
Ile945 950 955 960Gly Tyr
Gly Ile Leu Ala Thr Glu Gly Thr Ala Ala Phe Phe Ala Ser965
970 975His Gly Leu Gln Ala Gln Pro Val Gly Lys Ile Gly
Asp Asp Asp Lys980 985 990Asp Ile Pro Ser
Phe Val Arg Lys Gly Arg Ile Gln Ala Ile Ile Asn995 1000
1005Thr Val Gly Thr Lys Arg Thr Ala Asp Glu Asp Gly
Glu Gln Ile1010 1015 1020Arg Arg Ser Ala
Ile Glu His Gly Val Pro Leu Phe Thr Ala Leu1025 1030
1035Asp Thr Ala Asn Ala Met Leu Lys Val Leu Glu Ser Arg
Ser Phe1040 1045 1050Val Thr Glu Ala
Ile105525332PRTStreptococcus pneumoniae 25Met Thr Ile Met Ser Ile Gly Ile
Ile Ile Ala Ser His Gly Glu Phe1 5 10
15Ala Ala Gly Ile His Gln Ser Gly Ser Met Ile Phe Gly Glu
Gln Glu20 25 30Lys Val Gln Val Val Thr
Phe Met Pro Asn Glu Gly Pro Asp Asp Leu35 40
45Tyr Ala Lys Phe Asn Asn Ala Val Ala Ala Phe Asp Ala Glu Asp Glu50
55 60Val Leu Val Leu Ala Asp Leu Trp Ser
Gly Ser Pro Phe Asn Gln Ala65 70 75
80Ser Arg Val Met Gly Glu Asn Pro Glu Arg Lys Phe Ala Ile
Ile Thr85 90 95Gly Leu Asn Leu Pro Met
Leu Ile Gln Ala Tyr Thr Glu Arg Leu Met100 105
110Asp Ala Ala Ala Gly Val Glu Lys Val Ala Ala Asn Ile Ile Lys
Glu115 120 125Ala Lys Asp Gly Ile Lys Ala
Leu Pro Glu Glu Leu Asn Pro Val Glu130 135
140Glu Val Ala Ser Ala Ala Ala Ala Pro Val Ala Gln Thr Ala Ile Pro145
150 155 160Glu Gly Thr Val
Ile Gly Asp Gly Lys Leu Lys Ile Asn Leu Ala Arg165 170
175Leu Asp Thr Arg Leu Leu His Gly Gln Val Ala Thr Ala Trp
Thr Pro180 185 190Asp Ser Lys Ala Asn Arg
Ile Ile Val Ala Ser Asp Asn Val Ala Lys195 200
205Asp Asp Leu Arg Lys Glu Leu Ile Lys Gln Ala Ala Pro Gly Asn
Val210 215 220Lys Ala Asn Val Val Pro Ile
Gln Lys Leu Ile Glu Ile Ser Lys Asp225 230
235 240Pro Arg Phe Gly Glu Thr His Ala Leu Ile Leu Phe
Glu Thr Pro Gln245 250 255Asp Ala Leu Arg
Ala Ile Glu Gly Gly Val Pro Ile Lys Thr Leu Asn260 265
270Val Gly Ser Met Ala His Ser Thr Gly Lys Thr Leu Val Asn
Thr Val275 280 285Leu Ser Met Asp Lys Glu
Asp Val Ala Thr Phe Glu Lys Met Arg Asp290 295
300Leu Gly Val Glu Phe Asp Val Arg Lys Val Pro Asn Asp Ser Lys
Lys305 310 315 320Asp Leu
Phe Asp Leu Ile Asn Lys Ala Asn Val Lys325
33026259PRTStreptococcus pneumoniae 26Met Ala Val Ile Ser Met Lys Gln Leu
Leu Glu Ala Gly Val His Phe1 5 10
15Gly His Gln Thr Arg Arg Trp Asn Pro Lys Met Ala Lys Tyr Ile
Phe20 25 30Thr Glu Arg Asn Gly Ile His
Val Ile Asp Leu Gln Gln Thr Val Lys35 40
45Tyr Ala Asp Gln Ala Tyr Asp Phe Met Arg Asp Ala Ala Ala Asn Asp50
55 60Ala Val Val Leu Phe Val Gly Thr Lys Lys
Gln Ala Ala Asp Ala Val65 70 75
80Ala Glu Glu Ala Val Arg Ser Gly Gln Tyr Phe Ile Asn His Arg
Trp85 90 95Leu Gly Gly Thr Leu Thr Asn
Trp Gly Thr Ile Gln Lys Arg Ile Ala100 105
110Arg Leu Lys Glu Ile Lys Arg Met Glu Glu Asp Gly Thr Phe Glu Val115
120 125Leu Pro Lys Lys Glu Val Ala Leu Leu
Asn Lys Gln Arg Ala Arg Leu130 135 140Glu
Lys Phe Leu Gly Gly Ile Glu Asp Met Pro Arg Ile Pro Asp Val145
150 155 160Met Tyr Val Val Asp Pro
His Lys Glu Gln Ile Ala Val Lys Glu Ala165 170
175Lys Lys Leu Gly Ile Pro Val Val Ala Met Val Asp Thr Asn Thr
Asp180 185 190Pro Asp Asp Ile Asp Val Ile
Ile Pro Ala Asn Asp Asp Ala Ile Arg195 200
205Ala Val Lys Leu Ile Thr Ala Lys Leu Ala Asp Ala Ile Ile Glu Gly210
215 220Arg Gln Gly Glu Asp Ala Val Ala Val
Glu Ala Glu Phe Ala Ala Leu225 230 235
240Glu Thr Gln Ala Asp Ser Ile Glu Glu Ile Val Glu Val Val
Glu Gly245 250 255Asp Asn
Ala27312PRTStreptococcus pneumoniae 27Met Thr Thr Asn Arg Leu Gln Val Ser
Leu Pro Gly Leu Asp Leu Lys1 5 10
15Asn Pro Ile Ile Pro Ala Ser Gly Cys Phe Gly Phe Gly Gln Glu
Tyr20 25 30Ala Lys Tyr Tyr Asp Leu Asn
Leu Leu Gly Ser Ile Met Ile Lys Ala35 40
45Thr Thr Leu Glu Pro Arg Phe Gly Asn Pro Thr Pro Arg Val Ala Glu50
55 60Thr Pro Ala Gly Met Leu Asn Ala Ile Gly
Leu Gln Asn Pro Gly Leu65 70 75
80Glu Val Val Leu Ala Glu Lys Leu Pro Trp Leu Glu Arg Glu Tyr
Pro85 90 95Asn Leu Pro Ile Ile Ala Asn
Val Ala Gly Phe Ser Lys Gln Glu Tyr100 105
110Ala Ala Val Ser His Gly Ile Ser Lys Ala Thr Asn Val Lys Ala Ile115
120 125Glu Leu Asn Ile Ser Cys Pro Asn Val
Asp His Cys Asn His Gly Leu130 135 140Leu
Ile Gly Gln Asp Pro Asp Leu Ala Tyr Asp Val Val Lys Ala Ala145
150 155 160Val Glu Ala Ser Glu Val
Pro Val Tyr Val Lys Leu Thr Pro Ser Val165 170
175Thr Asp Ile Val Thr Val Ala Lys Ala Ala Glu Asp Ala Gly Ala
Ser180 185 190Gly Leu Thr Met Ile Asn Thr
Leu Val Gly Met Arg Phe Asp Leu Lys195 200
205Thr Arg Lys Pro Ile Leu Ala Asn Gly Thr Gly Gly Met Ser Gly Pro210
215 220Ala Val Phe Pro Val Ala Leu Lys Leu
Ile Arg Gln Val Ala Gln Thr225 230 235
240Thr Asp Leu Pro Ile Ile Gly Met Gly Gly Val Asp Ser Thr
Glu Ala245 250 255Ala Leu Glu Met Tyr Leu
Ala Gly Ala Ser Ala Ile Gly Val Gly Thr260 265
270Ala Asn Phe Thr Asn Pro Tyr Ala Cys Pro Asp Ile Ile Glu Asn
Leu275 280 285Pro Lys Val Met Asp Lys Tyr
Gly Ile Ser Ser Leu Glu Glu Leu Arg290 295
300Gln Glu Val Lys Glu Ser Leu Arg305
31028307PRTStreptococcus pneumoniae 28Met Ser Glu Asn Gln Gln Ala Leu Asn
His Val Val Ser Met Glu Asp1 5 10
15Leu Thr Val Asp Gln Val Met Lys Leu Ile Lys Arg Gly Ile Glu
Phe20 25 30Lys Asn Gly Ala Gln Leu Pro
Tyr Glu Asp His Pro Ile Val Ser Asn35 40
45Leu Phe Phe Glu Asp Ser Thr Arg Thr His Lys Ser Phe Glu Val Ala50
55 60Glu Ile Lys Leu Gly Leu Glu Arg Leu Asp
Phe Asp Val Lys Thr Ser65 70 75
80Ser Val Asn Lys Gly Glu Thr Leu Tyr Asp Thr Ile Leu Thr Leu
Ser85 90 95Ala Leu Gly Val Asp Val Cys
Val Ile Arg His Pro Glu Val Asp Tyr100 105
110Tyr Arg Glu Leu Ile Ala Ser Pro Thr Ile Thr Thr Ser Ile Ile Asn115
120 125Gly Gly Asp Gly Ser Gly Gln His Pro
Ser Gln Ser Leu Leu Asp Leu130 135 140Met
Thr Ile Tyr Glu Glu Phe Gly His Phe Glu Gly Leu Lys Val Ala145
150 155 160Ile Ala Gly Asp Leu Asp
His Ser Arg Val Ala Lys Ser Asn Met Gln165 170
175Ile Leu Lys Arg Leu Gly Ala Glu Leu Phe Phe Ala Gly Pro Glu
Glu180 185 190Trp Arg Ser Gln Glu Phe Ala
Asp Tyr Gly Gln Phe Val Thr Ile Asp195 200
205Glu Ile Ile Asp Gln Val Asp Val Met Met Phe Leu Arg Val Gln His210
215 220Glu Arg His Asp Ser Gly Ala Val Phe
Ser Lys Glu Asp Tyr His Ala225 230 235
240Gln His Gly Leu Thr Gln Glu Arg Tyr Asp Arg Leu Lys Glu
Thr Ala245 250 255Ile Leu Met His Pro Ala
Pro Ile Asn Arg Asp Val Glu Ile Ala Asp260 265
270His Leu Val Glu Ala Pro Lys Ser Arg Ile Val Gln Gln Met Thr
Asn275 280 285Gly Val Phe Val Arg Met Ala
Ile Leu Glu Ser Val Leu Ala Ser Arg290 295
300Asn Ala Asn30529398PRTStreptococcus pneumoniae 29Met Ala Lys Glu Lys
Tyr Asp Arg Ser Lys Pro His Val Asn Ile Gly1 5
10 15Thr Ile Gly His Val Asp His Gly Lys Thr Thr
Leu Thr Ala Ala Ile20 25 30Thr Thr Val
Leu Ala Arg Arg Leu Pro Ser Ser Val Asn Gln Pro Lys35 40
45Asp Tyr Ala Ser Ile Asp Ala Ala Pro Glu Glu Arg Glu
Arg Gly Ile50 55 60Thr Ile Asn Thr Ala
His Val Glu Tyr Glu Thr Glu Lys Arg His Tyr65 70
75 80Ala His Ile Asp Ala Pro Gly His Ala Asp
Tyr Val Lys Asn Met Ile85 90 95Thr Gly
Ala Ala Gln Met Asp Gly Ala Ile Leu Val Val Ala Ser Thr100
105 110Asp Gly Pro Met Pro Gln Thr Arg Glu His Ile Leu
Leu Ser Arg Gln115 120 125Val Gly Val Lys
His Leu Ile Val Phe Met Asn Lys Val Asp Leu Val130 135
140Asp Asp Glu Glu Leu Leu Glu Leu Val Glu Met Glu Ile Arg
Asp Leu145 150 155 160Leu
Ser Glu Tyr Asp Phe Pro Gly Asp Asp Leu Pro Val Ile Gln Gly165
170 175Ser Ala Leu Lys Ala Leu Glu Gly Asp Ser Lys
Tyr Glu Asp Ile Val180 185 190Met Glu Leu
Met Asn Thr Val Asp Glu Tyr Ile Pro Glu Pro Glu Arg195
200 205Asp Thr Asp Lys Pro Leu Leu Leu Pro Val Glu Asp
Val Phe Ser Ile210 215 220Thr Gly Arg Gly
Thr Val Ala Ser Gly Arg Ile Asp Arg Gly Ile Val225 230
235 240Lys Val Asn Asp Glu Ile Glu Ile Val
Gly Ile Lys Glu Glu Thr Gln245 250 255Lys
Ala Val Val Thr Gly Val Glu Met Phe Arg Lys Gln Leu Asp Glu260
265 270Gly Leu Ala Gly Asp Asn Val Gly Val Leu Leu
Arg Gly Val Gln Arg275 280 285Asp Glu Ile
Glu Arg Gly Gln Val Ile Ala Lys Pro Gly Ser Ile Asn290
295 300Pro His Thr Lys Phe Lys Gly Glu Val Tyr Ile Leu
Thr Lys Glu Glu305 310 315
320Gly Gly Arg His Thr Pro Phe Phe Asn Asn Tyr Arg Pro Gln Phe Tyr325
330 335Phe Arg Thr Thr Asp Val Thr Gly Ser
Ile Glu Leu Pro Ala Gly Thr340 345 350Glu
Met Val Met Pro Gly Asp Asn Val Thr Ile Asp Val Glu Leu Ile355
360 365His Pro Ile Ala Val Glu Gln Gly Thr Thr Phe
Ser Ile Arg Glu Gly370 375 380Gly Arg Thr
Val Gly Ser Gly Met Val Thr Glu Ile Glu Ala385 390
3953046PRTStreptococcus pneumoniae 30Met Lys Ser Thr Lys Glu Glu
Ile Gln Thr Ile Lys Thr Leu Leu Lys1 5 10
15Asp Ser Arg Thr Ala Lys Tyr His Lys Arg Leu Gln Ile
Val Leu Phe20 25 30Cys Leu Met Gly Lys
Ser Tyr Lys Glu Ile Ile Glu Leu Leu35 40
4531398PRTStreptococcus pneumoniae 31Met Ala Lys Leu Thr Val Lys Asp Val
Asp Leu Lys Gly Lys Lys Val1 5 10
15Leu Val Arg Val Asp Phe Asn Val Pro Leu Lys Asp Gly Val Ile
Thr20 25 30Asn Asp Asn Arg Ile Thr Ala
Ala Leu Pro Thr Ile Lys Tyr Ile Ile35 40
45Glu Gln Gly Gly Arg Ala Ile Leu Phe Ser His Leu Gly Arg Val Lys50
55 60Glu Glu Ser Asp Lys Ala Gly Lys Ser Leu
Ala Pro Val Ala Ala Asp65 70 75
80Leu Ala Ala Lys Leu Gly Gln Asp Val Val Phe Pro Gly Val Thr
Arg85 90 95Gly Ala Glu Leu Glu Ala Ala
Ile Asn Ala Leu Glu Asp Gly Gln Val100 105
110Leu Leu Val Glu Asn Thr Arg Tyr Glu Asp Val Asp Gly Lys Lys Glu115
120 125Ser Lys Asn Asp Pro Glu Leu Gly Lys
Tyr Trp Ala Ser Leu Gly Asp130 135 140Gly
Ile Phe Val Asn Asp Ala Phe Gly Thr Ala His Arg Ala His Ala145
150 155 160Ser Asn Val Gly Ile Ser
Ala Asn Val Glu Lys Ala Val Ala Gly Phe165 170
175Leu Leu Glu Asn Glu Ile Ala Tyr Ile Gln Glu Ala Val Glu Thr
Pro180 185 190Glu Arg Pro Phe Val Ala Ile
Leu Gly Gly Ser Lys Val Ser Asp Lys195 200
205Ile Gly Val Ile Glu Asn Leu Leu Glu Lys Ala Asp Lys Val Leu Ile210
215 220Gly Gly Gly Met Thr Tyr Thr Phe Tyr
Lys Ala Gln Gly Ile Glu Ile225 230 235
240Gly Asn Ser Leu Val Glu Glu Asp Lys Leu Asp Val Ala Lys
Ala Leu245 250 255Leu Glu Lys Ala Asn Gly
Lys Leu Ile Leu Pro Val Asp Ser Lys Glu260 265
270Ala Asn Ala Phe Ala Gly Tyr Thr Glu Val Arg Asp Thr Glu Gly
Glu275 280 285Ala Val Ser Glu Gly Phe Leu
Gly Leu Asp Ile Gly Pro Lys Ser Ile290 295
300Ala Lys Phe Asp Glu Ala Leu Thr Gly Ala Lys Thr Val Val Trp Asn305
310 315 320Gly Pro Met Gly
Val Phe Glu Asn Pro Asp Phe Gln Ala Gly Thr Ile325 330
335Gly Val Met Asp Ala Ile Val Lys Gln Pro Gly Val Lys Ser
Ile Ile340 345 350Gly Gly Gly Asp Ser Ala
Ala Ala Ala Ile Asn Leu Gly Arg Ala Asp355 360
365Lys Phe Ser Trp Ile Ser Thr Gly Gly Gly Ala Ser Met Glu Leu
Leu370 375 380Glu Gly Lys Val Leu Pro Gly
Leu Ala Ala Leu Thr Glu Lys385 390
39532276PRTStreptococcus pneumoniae 32Met Lys Lys Ile Val Lys Tyr Ser Ser
Leu Ala Ala Leu Ala Leu Val1 5 10
15Ala Ala Gly Val Leu Ala Ala Cys Ser Gly Gly Ala Lys Lys Glu
Gly20 25 30Glu Ala Ala Ser Lys Lys Glu
Ile Ile Val Ala Thr Asn Gly Ser Pro35 40
45Lys Pro Phe Ile Tyr Glu Glu Asn Gly Glu Leu Thr Gly Tyr Glu Ile50
55 60Glu Val Val Arg Ala Ile Phe Lys Asp Ser
Asp Lys Tyr Asp Val Lys65 70 75
80Phe Glu Lys Thr Glu Trp Ser Gly Val Phe Ala Gly Leu Asp Ala
Asp85 90 95Arg Tyr Asn Met Ala Val Asn
Asn Leu Ser Tyr Thr Lys Glu Arg Ala100 105
110Glu Lys Tyr Leu Tyr Ala Ala Pro Ile Ala Gln Asn Pro Asn Val Leu115
120 125Val Val Lys Lys Asp Asp Ser Ser Ile
Lys Ser Leu Asp Asp Ile Gly130 135 140Gly
Lys Ser Thr Glu Val Val Gln Ala Thr Thr Ser Ala Lys Gln Leu145
150 155 160Glu Ala Tyr Asn Ala Glu
His Thr Asp Asn Pro Thr Ile Leu Asn Tyr165 170
175Thr Lys Ala Asp Leu Gln Gln Ile Met Val Arg Leu Ser Asp Gly
Gln180 185 190Phe Asp Tyr Lys Ile Phe Asp
Lys Ile Gly Val Glu Thr Val Ile Lys195 200
205Asn Gln Gly Leu Asp Asn Leu Lys Val Ile Glu Leu Pro Ser Asp Gln210
215 220Gln Pro Tyr Val Tyr Pro Leu Leu Ala
Gln Gly Gln Asp Glu Leu Lys225 230 235
240Ser Phe Val Asp Lys Arg Ile Lys Glu Leu Tyr Lys Asp Gly
Thr Leu245 250 255Glu Lys Leu Ser Lys Gln
Phe Phe Gly Asp Thr Tyr Leu Pro Ala Glu260 265
270Ala Asp Ile Lys27533630PRTStreptococcus pneumoniae 33Met Thr Arg
Tyr Gln Asp Asp Phe Tyr Asp Ala Ile Asn Gly Glu Trp1 5
10 15Gln Gln Thr Ala Glu Ile Pro Ala Asp
Lys Ser Gln Thr Gly Gly Phe20 25 30Val
Asp Leu Asp Gln Glu Ile Glu Asp Leu Met Leu Ala Thr Thr Asp35
40 45Lys Trp Leu Ala Gly Glu Glu Val Pro Glu Asp
Ala Ile Leu Glu Asn50 55 60Phe Val Lys
Tyr His Arg Leu Val Arg Asp Phe Asp Lys Arg Glu Ala65 70
75 80Asp Gly Ile Thr Pro Val Leu Pro
Leu Leu Lys Glu Phe Gln Glu Leu85 90
95Glu Thr Phe Ala Asp Phe Thr Ala Lys Leu Ala Glu Phe Glu Leu Ala100
105 110Gly Lys Pro Asn Phe Leu Pro Phe Gly Val
Ser Pro Asp Phe Met Asp115 120 125Ala Arg
Ile Asn Val Leu Trp Ala Ser Ala Pro Ser Thr Ile Leu Pro130
135 140Asp Thr Thr Tyr Tyr Ala Glu Glu His Pro Gln Arg
Glu Glu Leu Leu145 150 155
160Thr Leu Trp Lys Glu Ser Ser Ala Asn Leu Leu Lys Ala Tyr Asp Phe165
170 175Ser Asp Glu Glu Ile Glu Asp Leu Leu
Glu Lys Arg Leu Glu Leu Asp180 185 190Arg
Arg Val Ala Ala Val Val Leu Ser Asn Glu Glu Ser Ser Glu Tyr195
200 205Ala Lys Leu Tyr His Pro Tyr Ser Tyr Glu Asp
Phe Lys Lys Phe Ala210 215 220Pro Ala Leu
Pro Leu Asp Asp Phe Phe Lys Ala Val Ile Gly Gln Leu225
230 235 240Pro Asp Lys Val Ile Val Asp
Glu Glu Arg Phe Trp Gln Ala Ala Glu245 250
255Gln Phe Tyr Ser Glu Glu Ala Trp Ser Leu Leu Lys Ala Thr Leu Ile260
265 270Leu Ser Val Val Asn Leu Ser Thr Ser
Tyr Leu Thr Glu Asp Ile Arg275 280 285Val
Leu Ser Gly Ala Tyr Ser Arg Ala Leu Ser Gly Val Pro Glu Ala290
295 300Lys Asp Lys Val Lys Ala Ala Tyr His Leu Ala
Gln Glu Pro Phe Lys305 310 315
320Gln Ala Leu Gly Leu Trp Tyr Ala Arg Glu Lys Phe Ser Pro Glu
Ala325 330 335Lys Ala Asp Val Glu Lys Lys
Val Ala Thr Met Ile Asp Val Tyr Lys340 345
350Glu Arg Leu Leu Lys Asn Asp Trp Leu Thr Pro Glu Thr Cys Lys Gln355
360 365Ala Ile Val Lys Leu Asn Val Ile Lys
Pro Tyr Ile Gly Tyr Pro Glu370 375 380Glu
Leu Pro Ala Arg Tyr Lys Asp Lys Val Val Asn Glu Thr Ala Ser385
390 395 400Leu Phe Glu Asn Ala Leu
Ala Phe Ala Arg Val Glu Ile Lys His Ser405 410
415Trp Ser Lys Trp Asn Gln Pro Val Asp Tyr Lys Glu Trp Gly Met
Pro420 425 430Ala His Met Val Asn Ala Tyr
Tyr Asn Pro Gln Lys Asn Leu Ile Val435 440
445Phe Pro Ala Ala Ile Leu Gln Ala Pro Phe Tyr Asp Leu His Gln Ser450
455 460Ser Ser Ala Asn Tyr Gly Gly Ile Gly
Ala Val Ile Ala His Glu Ile465 470 475
480Ser His Ala Phe Asp Thr Asn Gly Ala Ser Phe Asp Glu Asn
Gly Ser485 490 495Leu Lys Asp Trp Trp Thr
Glu Ser Asp Tyr Ala Ala Phe Lys Glu Lys500 505
510Thr Gln Lys Val Ile Asp Gln Phe Asp Gly Gln Asp Ser Tyr Gly
Ala515 520 525Thr Ile Asn Gly Lys Leu Thr
Val Ser Glu Asn Val Ala Asp Leu Gly530 535
540Gly Ile Ala Ala Ala Leu Glu Ala Ala Lys Arg Glu Ala Asp Phe Ser545
550 555 560Ala Glu Glu Phe
Phe Tyr Asn Phe Gly Arg Ile Trp Arg Met Lys Gly565 570
575Arg Pro Glu Phe Met Lys Leu Leu Ala Ser Val Asp Val His
Ala Pro580 585 590Ala Lys Leu Arg Val Asn
Val Gln Val Pro Asn Phe Asp Asp Phe Phe595 600
605Thr Thr Tyr Asp Val Lys Glu Gly Asp Gly Met Trp Arg Ser Pro
Glu610 615 620Glu Arg Val Ile Ile Trp625
63034149PRTStreptococcus pneumoniae 34Met Ile Gly Val Val
Ala Arg Glu Asn Ala Ala Glu Gln Ile Lys Gln1 5
10 15Tyr Gln Lys Phe Thr Val Asn Ile Ser Asp Glu
Thr Ser Met Leu Ala20 25 30Met Glu Gln
Ala Gly Phe Ile Ser His Gln Glu Lys Leu Glu Arg Leu35 40
45Gly Val His Tyr Glu Ile Ser Glu Arg Thr Gln Ile Pro
Ile Leu Asp50 55 60Ala Cys Pro Leu Val
Leu Asp Cys Arg Val Asp Arg Ile Val Glu Glu65 70
75 80Asp Gly Ile Cys His Ile Phe Ala Lys Ile
Leu Glu Arg Leu Val Ala85 90 95Pro Glu
Leu Leu Asp Glu Lys Gly His Phe Lys Asn Gln Leu Phe Ala100
105 110Pro Thr Tyr Phe Met Gly Asp Gly Tyr Gln Arg Val
Tyr Arg Tyr Leu115 120 125Asp Lys Arg Val
Asp Met Lys Gly Ser Phe Ile Lys Lys Ala Arg Lys130 135
140Lys Asp Gly Lys Asn14535149PRTStreptococcus pneumoniae
35Met Ile Gly Val Val Ala Arg Glu Asn Ala Ala Glu Gln Ile Lys Gln1
5 10 15Tyr Gln Lys Phe Thr Val
Asn Ile Ser Asp Glu Thr Ser Met Leu Ala20 25
30Met Glu Gln Ala Gly Phe Ile Ser His Gln Glu Lys Leu Glu Arg Leu35
40 45Gly Val His Tyr Glu Ile Ser Glu Arg
Thr Gln Thr Pro Ile Leu Asp50 55 60Ala
Cys Pro Leu Val Leu Asp Cys Arg Val Asp Arg Ile Val Glu Glu65
70 75 80Asp Gly Ile Cys His Ile
Phe Ala Lys Ile Leu Glu Arg Leu Val Ala85 90
95Pro Glu Leu Leu Asp Glu Lys Gly His Phe Lys Asn Gln Leu Phe Ala100
105 110Pro Thr Tyr Phe Met Gly Asp Gly
Tyr Gln Arg Val Tyr Arg Tyr Leu115 120
125Asp Lys Arg Val Asp Met Lys Gly Ser Phe Ile Lys Lys Ala Arg Lys130
135 140Lys Asp Gly Lys Asn145
User Contributions:
comments("1"); ?> comment_form("1"); ?>Inventors list |
Agents list |
Assignees list |
List by place |
Classification tree browser |
Top 100 Inventors |
Top 100 Agents |
Top 100 Assignees |
Usenet FAQ Index |
Documents |
Other FAQs |
User Contributions:
Comment about this patent or add new information about this topic: